Abstract
Marmosets are becoming more utilized in biomedical research due to multiple advantages including (1) a nonhuman primate of a smaller size with less cost for housing, (2) physiologic similarities to humans, (3) translatable hepatic metabolism, (4) higher numbers of litters per year, (5) genome is sequenced, molecular reagents are available, (6) immunologically similar to humans, (7) transgenic marmosets with germline transmission have been produced, and (8) are naturally occurring hematopoietic chimeras. With more use of marmosets, disease surveillance over a wide range of ages of marmosets has been performed. This has led to a better understanding of the disease management of spontaneous diseases that can occur in colonies. Knowledge of clinical signs and histologic lesions can assist in maximizing the colony’s health, allowing for improved outcomes in translational studies within biomedical research. Here, we describe some basic husbandry, biology, common spontaneous diseases, and animal model applications for the common marmoset in biomedical research.
Introduction
The new world primate, the common marmoset (Callithrix jacchus), is in the primate family Callitrichidae and originates from forests in Brazil, where family groups have a diet of fruit, insects, eggs, and exudates from the gum tree known to be rich in calcium and carbohydrates. 1 The primate family is characterized by a small body size (less than 300-450 g) and dense short fur. All phalanges have claws with the exception of the hallux, which has a flattened nail. 2 While claws likely account for poor foot and hand manipulation, the nail is advantageous in grasping, which is important for tree gouging.2,3 The genus Callithrix (true marmosets) have enlarged incisors and reduced canines. 3 The common marmoset is the most frequently used species of this genus in biomedical research. Over the past two decades, marmosets have been more frequently used in preclinical, toxicology, and safety assessment of drugs.4-30
Of all the nonhuman primate (NHP) species, the small size of the marmoset translates to smaller enclosure requirements (compared with other NHP), and the physiologic similarity with humans makes the common marmoset very attractive for biomedical research. The European Union (EU) and US guidelines have defined minimum requirements for cage enclosure floor area and heights for housing marmosets in laboratories. 31 Due to their arboreal nature, the vertical cage height is more important than the floor area. The density of housing is also an important consideration as high marmoset density can influence disease incidence. Common marmosets can only be used for biomedical studies in the EU and US when animals are the offspring of marmosets bred in colonies, not wild-captured. It is estimated that research facilities currently house more than 6000 marmosets worldwide, with about 2500 of marmosets located in Japan, 1900 in North America, 1000 in Europe, 800 in South America, and an unknown number of marmosets located in China. 32
In breeding colonies, marmosets are typically bred in small monogamic groups in which only the dominant female reproduces. Marmosets can produce more offspring per year compared with any other NHP used in research. For example, a marmoset breeding pair can produce ~3 litters in 2 years with 2 to 4 offspring per litter compared cynomolgus macaques that at our institution produce 1 offspring in 1 to 2 years. For large litters, at some institutions, weaker marmoset newborns are humanely euthanized or fostered to another pair to support 2 or more offspring per litter. Females become reproductively mature at 1.5 years and breeding pairs produce litters usually for 5 to 7 years. Institutional colonies are convenient, practical, and reduce costs, with adequate housing space and a substantial institutional need. In a recent workshop, participants explored ways to ensure research conducted in marmosets is performed ethically with considerations of assessing potential harms and benefits. Participants discussed a variety of practical steps that the research community could take to proactively guide in improving research practices while enforcing ethical research. 32
Marmoset Models in Biomedical Research
Further characterization of marmosets including the sequencing of the genome, 33 the creation of transgenic marmosets with germline transmission, 34 and the availability of a marmoset specific microarray 35 has increased the applicability of marmoset biomedical research. Important in translational research, many surface receptors on marmoset lymphocytes and other cells have been found to cross-react with monoclonal antibodies against corresponding human epitopes demonstrating conserved nature and applicability of marmosets when modeling human diseases.36-38 Marmosets are born as naturally occurring hematopoietic chimeras, yet genetically distinct and tolerant to each other cells. 39 This natural chimerism allows for a cell transfer without inducing an alloresponse. Induced pluripotent stem cells have been made from common marmoset fetal liver cells. 40
For the past 15 to 20 years within the pharmacological industry, there has been increased product development despite the limited availability of NHP such as baboon and macaques. This has resulted in an increased use of marmosets for pharmacology and preclinical toxicity testing. 17 Due to the small size, a 400-g marmoset versus a 5 to 7 kg cynomolgus macaque, a marmoset requires much less material (drug, vaccine) for the early optimization and safety assessment studies, reducing costs, and the time for product development. The smaller blood volumes obtained from a marmoset historically were considered a limitation, but with the development of analytical methods for rodents and assays such as 96-cell-plate ELISAs, microliter amounts are acceptable for diagnostics and pharmacology purposes.
Marmosets breed well in colonies and provide a more controlled population for testing cohorts and toxicological datasets. Infectious disease surveillance is necessary for controlled, healthy test populations. 41 Trained technicians find that marmosets are relatively easy to restrain for examination, ultrasound, administration of test articles and medications, and drawing blood samples. Urine can be easily collected in rat metabolic cages since marmosets are of similar size. For pharmacokinetics studies, marmosets and humans share the same expression and inducers of drug-metabolizing enzymes like CYP1A2, which is responsible for Phase 1 metabolic activation of many hepatic compounds. In contrast, CYP1A2 is not expressed in the cynomolgus macaque liver, making the marmoset a better translational model. 42
Marmosets Have Been Used as a Model for Aging
Due to the relatively shorter life span of 15 to 16 years and the smaller body size, studies conducted in marmosets versus other NHP (rhesus or baboon) may be more advantageous. 43 One point that must be considered is the affect the environment has on aging. Barrier versus no barrier colonies age at different rates and have variable incidence of diseases which impact lifespan. One closed Japanese marmoset colony reported extending the lifespan of marmosets over 20 years but details on diseases in this colony have not been published. 44 Since neurodegeneration is prevalent in aging humans, there has been much interest in the neurodegeneration of aging marmosets. Aging marmosets have brain amyloid deposition, but this does also vary between colonies. 45 Amyloid has not yet been associated with dementia in marmosets. Neurogenesis in the dentate gyrus does decrease linearly with age in marmosets and mechanisms of this decline could be studied further. A study was done looking at inhibition of mTOR signaling by giving rapamycin at 1 mg/kg/day to marmosets for up to 14 months without adverse effects. 46 This work is now underway to evaluate the effect of rapamycin on aging.
Marmosets Have Been Used as a Model for Neurologic Disease Research: Parkinson’s Disease and Alzheimer’s Disease
Marmosets have been used as a model for neurologic disease research. 47 Parkinson’s disease is one of the most common neurodegenerative disorder in humans second to Alzheimer’s disease, of which 95% of cases do not have known genetic etiology.48,49 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) is the most frequently used neurotoxin to induce Parkinson’s disease by mitochondrial dysfunction in animal models. 50 The MPTP-induced Parkinson’s disease model using common marmosets provides several benefits including reproducibility of parkinsonian behaviors more comparable to those in human versus those observed in rodent models.51-54
Common marmosets are also a useful model for Alzheimer’s disease. This disease is primarily characterized by extracellular aggregation of β-amyloid (Aβ) and intracellular neurofibrillary tau tangles. 55 Marmosets can develop amyloidosis in their brain by intracerebral injection of Aβ fibrils homogenates56,57 as well as spontaneous senile plaques at old age.57,58 Moreover, the marmoset model can offer an opportunity to investigate underlying mechanism. It has been shown that the progression and development of the disease is affected by immune response in marmoset models.47,59
Polyglutamine Repeat Neurologic Diseases Have Been Modeled in Marmosets
A neurodegenerative disease, induced by polyglutamine (polyQ) repeats has been produced in transgenic marmosets, showing progressive neurological symptoms including motor impairment. These transgenic marmosets were produced by lentiviral introduction of the human ataxin 3 gene with 120 CAG repeats encoding an expanded polyQ DNA stretch. No offspring showed neurological symptoms at birth, but marmosets with higher transgene expression developed neurological symptoms of varying degrees at 3 to 4 months after birth, followed by gradual decreases in body weight gain, spontaneous activity, and grip strength. Pathological examinations revealed intranuclear polyQ protein inclusions, neuronal loss, gliosis, fourth ventricle enlargement suggesting cerebellar atrophy which recapitulates the neuropathological features of polyQ disease patients such as Spinocerebellar ataxia type 3 (SCA3). Successful germline transgene transmission was confirmed in the second-generation offspring derived from the symptomatic transgenic marmoset gamete. Whether this model can exhibit the generation to generation further expansion of CAG repeats, still remains to be determined. 60 The marmoset model of experimental autoimmune encephalomyelitis (EAE) is an important translational model for multiple sclerosis (MS).
Multiple sclerosis, a chronic autoimmune neurological disease, is characterized by white matter lesions including focal demyelination with varying degree of inflammation, axonal injury, edema, and blood-brain barrier dysfunction.61,62 Experimental autoimmune encephalomyelitis model, established by injecting myelin or myelin components in various species, has been widely used to mimic the disease. Unlike the rodent models, the marmoset EAE model offers the most relevant pathologic features such as presence of MS-like white matter and gray matter lesions in both brain and spinal cord.63-65 The marmoset has also been used for mechanistic investigation for a role of Epstein-Barr virus (EBV) in MS owing to the natural infection of EBV homologue. 66
Common Marmosets Have Been Used as a Model for Visual and Auditory Neuroscience Research
Using marmosets offers opportunities to study comparative visual neuroscience with application to the human brain.67,68 Marmosets are born with less developed visual system, which allows the study of basic development of the visual system at the postnatal stage.69,70 The lissencephalic brain of marmosets, unlike that of macaques, allows for functional mapping of visual cortex. 67 Similarly, marmosets have provided valuable insights in auditory research. 68 Because of their vocal communication, marmosets have been used to study sound coding and the auditory system response, vocal production, and its perception.71-75 The creation of transgenic lines of marmosets could be useful for studying various visual or auditory diseases of human.34,76
Common Marmosets are Used in Biocontainment Research
Marmosets have been used for biocontainment infectious disease research, including a variety of viral hemorrhagic fever caused by filoviruses and arenaviruses, Eastern Equine Encephalitis (EEEV), Severe Acute Respiratory Syndrome (SARS-CoV), Middle East Respiratory Syndrome (MERS-CoV), Influenza, Q fever (Coxiella burnetii), and dengue virus infection.77-81 Albeit marmosets are a proven animal model for the foregoing coronaviral diseases, however, a recent study of COVID-19 has shown marmosets exhibited only mild responses after SARS-CoV-2 infections. 82 Their susceptibility to wild types of these infectious agents, mimicking disease progression and pathophysiology of human diseases, has proved that marmosets are an appropriate small animal model for these diseases, while some of the agents, filovirus specifically, need to be adapted for rodent study. 83 Compared with other NHPs, that is, macaques, more manageable husbandry owing to their small size can offer advantages in manipulating experiments in high containment conditions.77,84
The Common Marmoset has Been Used in Preclinical Testing
Although marmosets are not the primary species for consideration for nonclinical studies, they have served as a useful NHP model for pharmacokinetics, safety pharmacology, and safety assessment for various small and large molecules.15,17,25,26 Justification for the use of marmosets stems from shared pharmacologic mechanisms, xenobiotic metabolisms, or previous study experience for the same class of drug. 25 The common marmoset shows a high degree of homology with human in immunoglobulins and T-cell receptors, which make this species a relevant model for immune toxicity testing.27,28 Marmosets also may have higher relevancy to humans regarding carcinogenicity than that of rats,29,30 yet few carcinogenicity studies have recently been done and this could be a missed opportunity in an NHP model.
Common Spontaneous Diseases
This common marmoset review on diseases is a brief overview and not considered complete, yet other reviews on marmoset diseases are available.85-87 At our institution, since we have a breeding colony of more than 200 marmosets, we review pathology from a wide age range of marmosets. This enables our clinical and pathology team to observe a range of spontaneous diseases that occur in captive marmosets. Some of these diseases are subclinical and are discovered at necropsy. Others warrant veterinary treatment and can impact research applications. As will be discussed in the following sections, especially as animals age, some of these conditions could occur in the same animal and at the same time and may be mechanistically related.
Common Inflammatory Diseases of Unknown Origin
Inflammatory conditions affecting the kidneys, livers, and the intestines have been the most problematic in common marmoset colonies and have caused animals to chronically lose weight. Reviewing the historic literature, there are references to Marmoset Wasting Syndrome that lack diagnostic specificity and this term should not be perpetuated in the literature. 32 There are multiple diseases that could also cause wasting and weight loss in marmosets, such as amyloidosis, thus relying on the pathological assessment for a specific diagnosis is best when characterizing or describing marmosets with chronic weight loss. When used together, serum albumin of <3.5 g/dL and body weight of <325 g, identified 100% of the marmosets affected with concurrent bone and gastrointestinal disease. 88 In marmosets with untreatable weight loss, we use a necropsy protocol that systematically samples multiple bones, multiple sections of the entire gastrointestinal tract, and major visceral organs to completely survey the case. Frequently, these animals will have a diagnosis of lymphocytic enteritis, colitis, and sometimes gastritis. This disease is one of the major causes of morbidity and mortality in captive marmosets. 89 It is clinically characterized by chronic weight loss, commonly with concurrent alopecia and ±diarrhea. 89 Monthly weigh-ins are important to detect this problem in the colony. Blood tests have revealed various abnormalities including anemia, hypoproteinemia, hypoalbuminemia, and increases in serum aspartate aminotransferase and alkaline phosphatase.88,90-92 The most consistently associated histological feature is chronic lymphoplasmacytic inflammation in the intestines, where inflammatory infiltrates (predominantly T cells) are found in the lamina propria and epithelium with villous atrophy, fusing and blunting and crypt hyperplasia (Figure 1).89,91,93,94
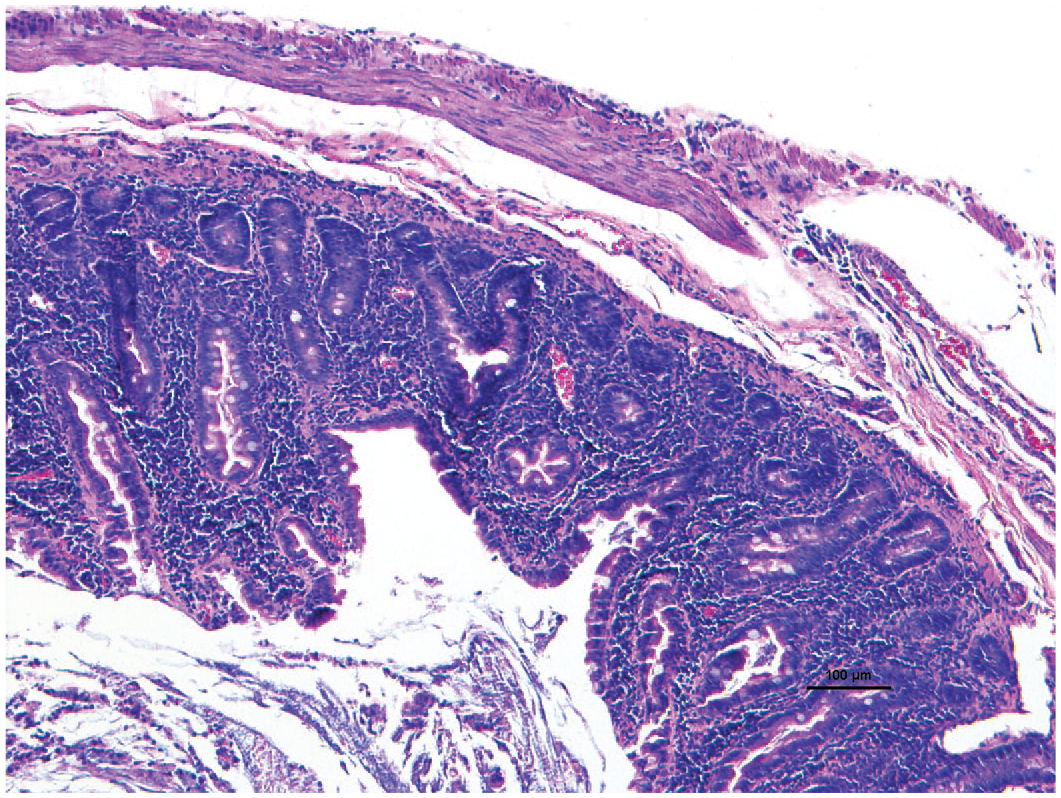
Chronic enteritis. Lymphoplasmacytic inflammation expanding the jejunum lamina propria with blunted small intestinal villi in a common marmoset. 10X H&E.
While several hypotheses have been proposed including food sensitivity95,96 and parasitic infestation, 90 its etiology and underlying mechanisms have not been explored and are likely multifactorial.97,98 However, recent reports suggest that two clinical conditions are closely related, chronic weight loss and a complex metabolic bone disease that also occurs in some (not all) common marmosets. It is probable that the intestinal disease impairs nutrient absorption and efficient vitamin D absorption, contributing to bone disease.88,99 Furthermore, there has been work to investigate a mechanism of hypoalbuminemia that demonstrated elevated α1-proteinase inhibitor concentrations in feces, reflecting protein intestinal loss in chronic lymphocytic enteritis affected animals with chronic weight loss. 98
Decreased body weight and serum albumin level serve as predictive markers for chronic lymphocytic enteritis within colonies. 88 Recent reports proposed elevated serum matrix metalloproteinase 9 and urinary lipid metabolites, including prostaglandins, as potential biomarkers.100,101 For treatment, glucocorticoids such as budesonide are an effective therapeutic option to improve clinical signs in marmosets with chronic lymphocytic enteritis. 89 A recent report demonstrated that tranexamic acid, a plasmin inhibitor, has a therapeutic potential by suppressing certain inflammatory cytokines. 102 The incidence of marmoset wasting syndrome has increased in the last 40 years. 86
Another differential diagnosis for diarrhea and chronic weight loss in a marmoset is systemic Amyloid A amyloidosis. In a retrospective analysis of 86 marmosets more than 1 year of age, 17% were found to have amyloid deposits in one or more organs in animals who failed to thrive. 103 The material was examined by amino acid sequence analysis and was composed on serum amyloid A (SAA-related protein). This type of amyloidosis is “secondary” as associated with an inflammatory process. 103
Bone Diseases Linked to Inflammatory Intestinal Disease are Present in Some Colonies, but Not All
The previously described gastrointestinal disease malabsorption likely impacts the next disease to be discussed, metabolic bone disease. Bone disease only occurs in some institutional colonies with GI disease for unknown reasons. 104 It manifests as various bone lesions that can be seen radiographically and depends on the age (growing vs adults) and nutrition status. The bone disease is caused by the deficiency of vitamin D and imbalance of calcium-phosphorus levels and often presents as a fractured bone. 105 Historically at our institution, the three most prevalent bone lesions found in common marmosets are rickets, fibrous osteodystrophy (FOD), and osteopenia. 106 These lesions are frequently not present alone and are observed concurrently. For example, all rickets-affected young animals showed additional histologic features of FOD and osteopenia. In adults, FOD and osteopenia are features that are shared. 106
Rickets, which resembles vitamin D–dependent rickets type II, is the most common bone disease in young marmosets who have active growth plates. 107 The rickets cases were characterized by widened physis (compared with nonaffected control animals) with a variable degree of metaphyseal flaring in radiographs. Histologically, rickets is characterized by a widened zone of hypertrophic chondrocytes within the growth plates in long bone and irregular cartilage extending into thickened trabeculae in the primary spongiosa of metaphysis. 106 Unfortunately, affected animals showing histological lesions did not always have radiographic lesions of rickets. In contrast, osteomalacia, the equivalent of rickets in adult animals, cannot be diagnosed routinely histologically because it is more challenging to recognize in bone sections which have been decalcified. 106 Increased unmineralized osteoid is the pathognomonic feature of osteomalacia, therefore, tissue sections without decalcification are needed to assess the degree of osteoid mineralization.
Historically, FOD is one of the common bone diseases affecting marmosets at our institution. Radiographically, bones will have punctate areas of bone lysis (moth-eaten pattern) (Figure 2). Multiple bones can be affected including long bones, maxilla, mandible, and vertebrae. Histologically, the cortical bone is reduced and forms anastomosing trabeculae with abundant fibrosis between trabecula (Figure 3). The areas of osteoclastic osteolysis of cortex and trabecula correspond to the radiographic moth-eaten pattern. FOD was found exclusively in marmosets who had the inflammatory gastrointestinal disease described earlier.88,106 Vitamin D deficiency negatively affects healthy bone density in marmosets. 99 Given that marmosets are highly sensitive to calcium and vitamin D deficiencies, the marmoset diet should include a sufficient amount of vitamin D (~7800 IU vitamin D3/kg dry matter basis). 106 In previous studies, PTH levels were higher in animals with FOD or osteopenia compared with control animals. 88 PTH is induced by low circulating levels of calcium or vitamin D3 due to concurrent gastrointestinal inflammation followed by nutrient malabsorption.88,99
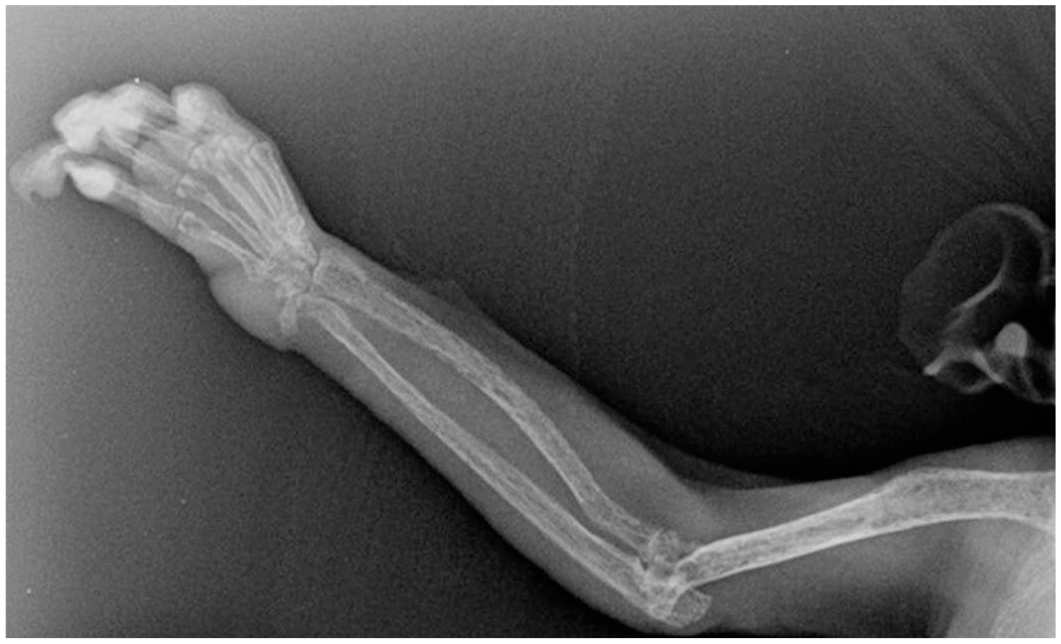
Fibrous osteodystrophy. Humeral and radial osteopenia with moth-eaten appearance in radiograph in a common marmoset.
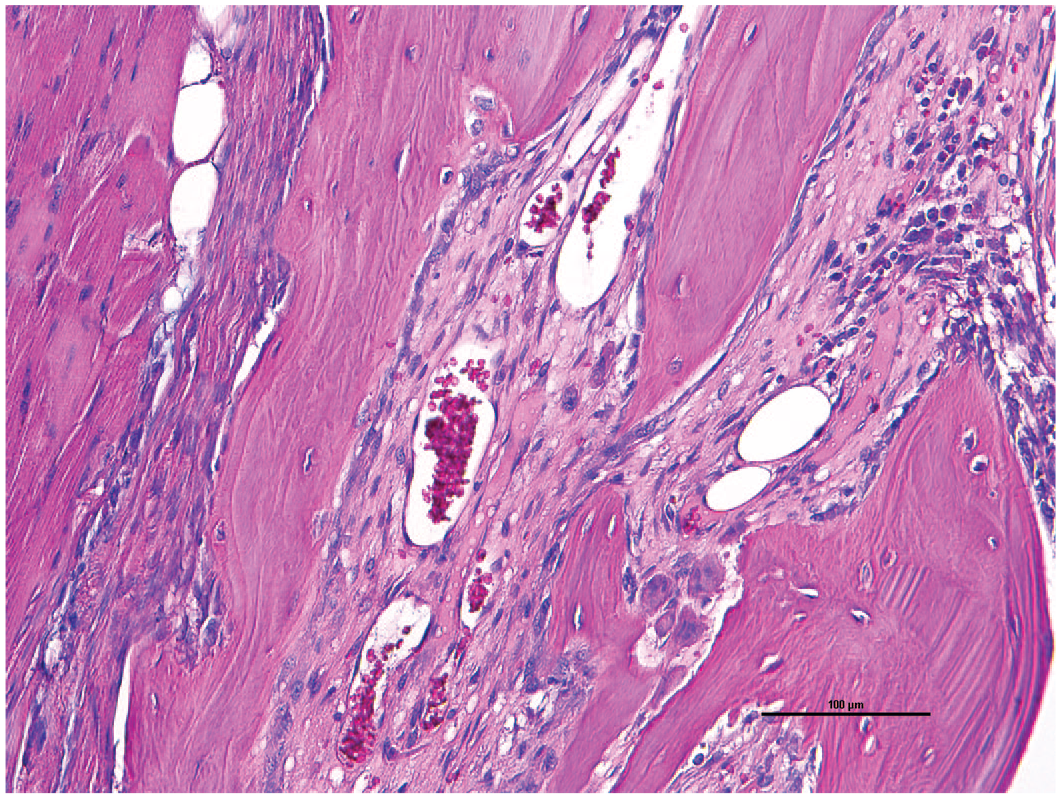
Fibrous osteodystrophy. Cortex is replaced with fibrosis between the trabeculae in a common marmoset. 20X H&E.
Finally, osteopenia is also prevalent in some marmoset colonies. Radiographical features showed generalized diffuse decreases in radiodensity of the cortex and medulla. Histologically, there is morphological evidence of overall decreased cortical thickness with variable endosteal resorption and cortical trabeculation. For a clinical diagnosis, a work-up generally requires bone densitometry also called dual-energy X-ray absorptiometry (DEXA). 99 There have been efforts to develop quantitative analysis techniques for radiographic digital images. 88 However, only histological evaluation of decreased cortical or trabecular bone has defined the diagnosis. 106 At our institution, osteopenia bone lesions in marmosets are also highly correlated to lesions of the inflammatory gastrointestinal disease and chronic weight loss in marmosets.88,106
Another comorbidity within marmoset colonies is chronic renal disease which can be subclinical but also does impact concurrent diseases. Tardif et al. 108 report 75% of marmosets above 8 years of age had renal lesions including interstitial nephritis, amyloidosis, and glomerular lesions at necropsy but only 17% had renal disease as the primary cause of death. In aging marmosets, spontaneous, progressive glomerulonephropathy is commonly seen. This disease may be similar to IgM and IgA nephropathy in humans. 109 Since these animals do not display clinical signs of renal disease prior to death, diagnosis is based on histopathology. 109 Histopathologic lesions are characterized into four grades based on severity, starting with mild interstitial nephritis, regenerative tubules, and interstitial inflammation that progresses to severe glomerular involvement (Figure 4).
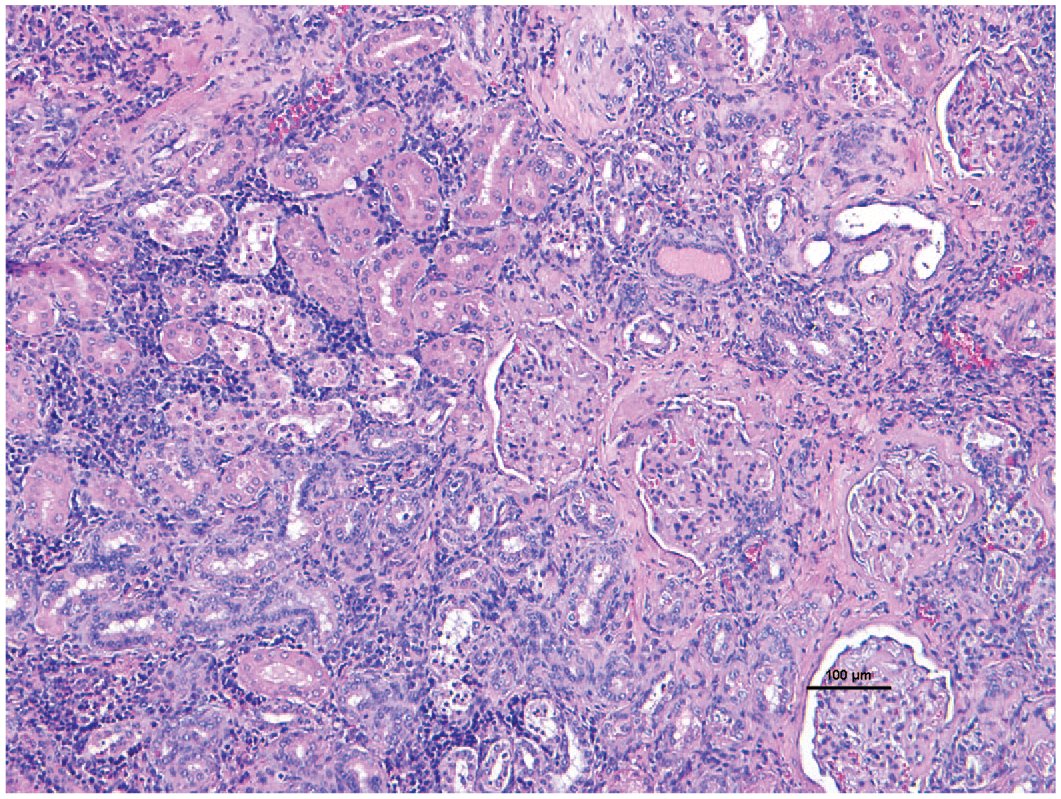
Chronic renal disease. Renal interstitial fibrosis with lymphocytic infiltrates and focal glomerular fibrosis/sclerosis. Tubules are attenuated and occasionally filled with hyaline casts in a common marmoset. 10X H&E.
Other Important Diseases of Marmoset Colonies
There are other less common diseases that are observed in marmoset colonies. A clinical differential diagnosis in a marmoset with sudden onset of lethargy, anorexia, or sudden death without premonitory signs is a Klebsiella pneumonia infection. 110 Klebsiella pneumonia is a zoonotic, opportunistic bacterium found in the nasopharyngeal and intestinal microbiomes in animals and humans. In marmosets, infection can present with diarrhea, but K pneumonia can also cause septicemia and pneumonia (hence its name). In some colonies, there are individual marmosets who are Klebsiella sp. rectal culture positive but otherwise healthy and asymptomatic. Klebsiella pneumonia polysaccharide capsule is a virulence factor. When K pneumonia outbreaks do occur, multiple organs are involved as was seen recently in a marmoset colony outbreak in Brazil. 111
Infectious disease is prevented in marmoset colonies by quarantining new arrivals to a colony for 1 to 3 months, known to prevent the spread of measles or parasites such as Giardia. Yet, with these measures, marmosets are still susceptible to a number of bacteria and viruses. Some colonies are monitored for tuberculosis with intradermal TB tests. Marmosets are susceptible to viruses from other species. For example, historically, viral infections can arise from herpes simplex 1 (from humans) and herpes tamarinus (from tamarins) resulting in high morbidity and mortality in colonies. Callitrichid herpesvirus 3 is observed in more than 60% of marmosets above 3 years of age in North American colonies, but is usually subclinical, and has been linked to GI lymphomas. Infectious gastrointestinal diseases can result from enteropathogenic Escherichia coli, Salmonella enterica, Clostridium difficile, and K pneumoniae infections in marmoset colonies. Necropsies should be performed on all deceased marmosets to monitor disease prevalence and prevent the spread of infectious diseases. 32
In contrast to weight loss, obesity can be a problem in some colonies. 112 A 500-g marmoset is considered obese. There may be a relationship between obesity and prenatal factors such as litter sizes and birth weights, but mechanisms have not been explored. Other less prevalent diseases occur in marmosets and a complete inventory of spontaneous diseases in marmoset was recently reviewed. 113
Marmosets Rarely Develop Spontaneous Cancers
Common marmosets have several naturally occurring cancers although neoplasia is uncommon in this species compared with other NHPs. The most common site is the gastrointestinal tract in which oral and nasopharyngeal carcinomas, and small intestinal adenocarcinoma can arise.114-116 While small intestinal adenocarcinomas are rare in other NHPs and man, it is the most common neoplasm affecting morbidity and mortality in common marmosets, even with extensive metastasis.116,117 In this example, an adenocarcinoma from the small intestines has metastasized to the lung (Figures 5 and 6). Less common or rarely reported malignant neoplasia includes malignant nephroblastoma, rhabdomyosarcoma, lymphoma, myeloid sarcoma, and apocrine gland adenocarcinoma.85,118-122 In addition, there are few neoplasia-causing viruses in common marmosets including Saimiriine herpesvirus 1 which can induce leukemia and lymphoma.123,124
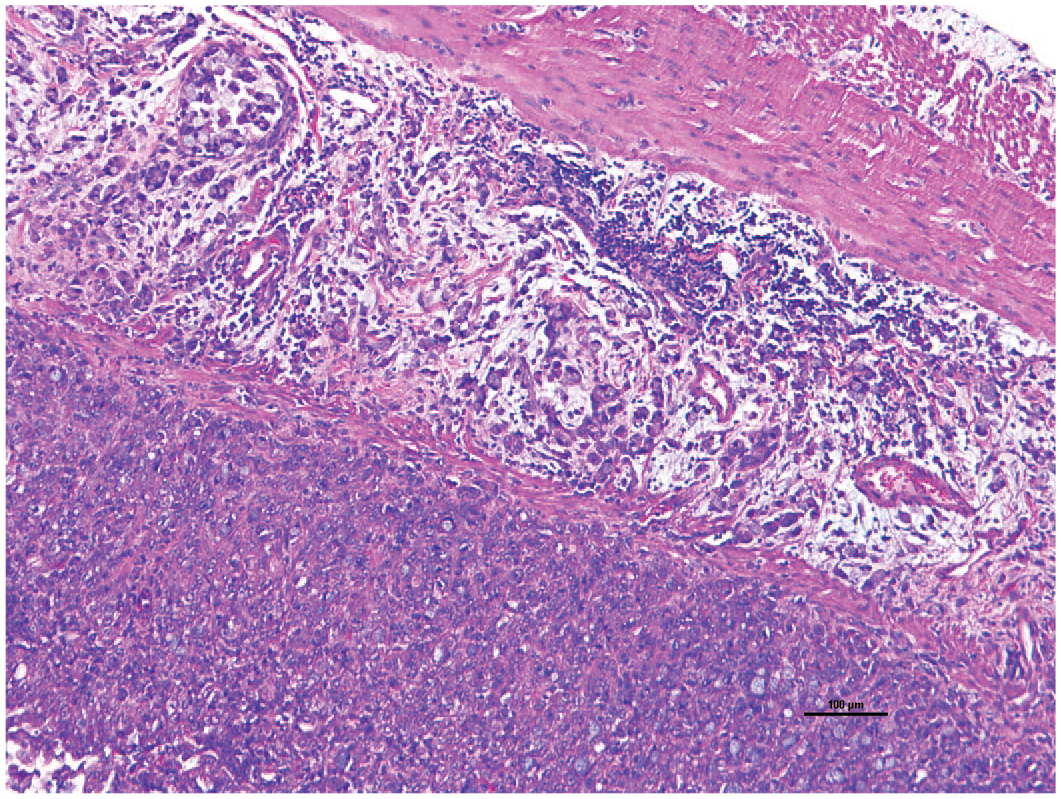
Duodenal adenocarcinoma. Histological examination revealed infiltrative mucinous adenocarcinoma of the duodenum in a common marmoset. 10X H&E.
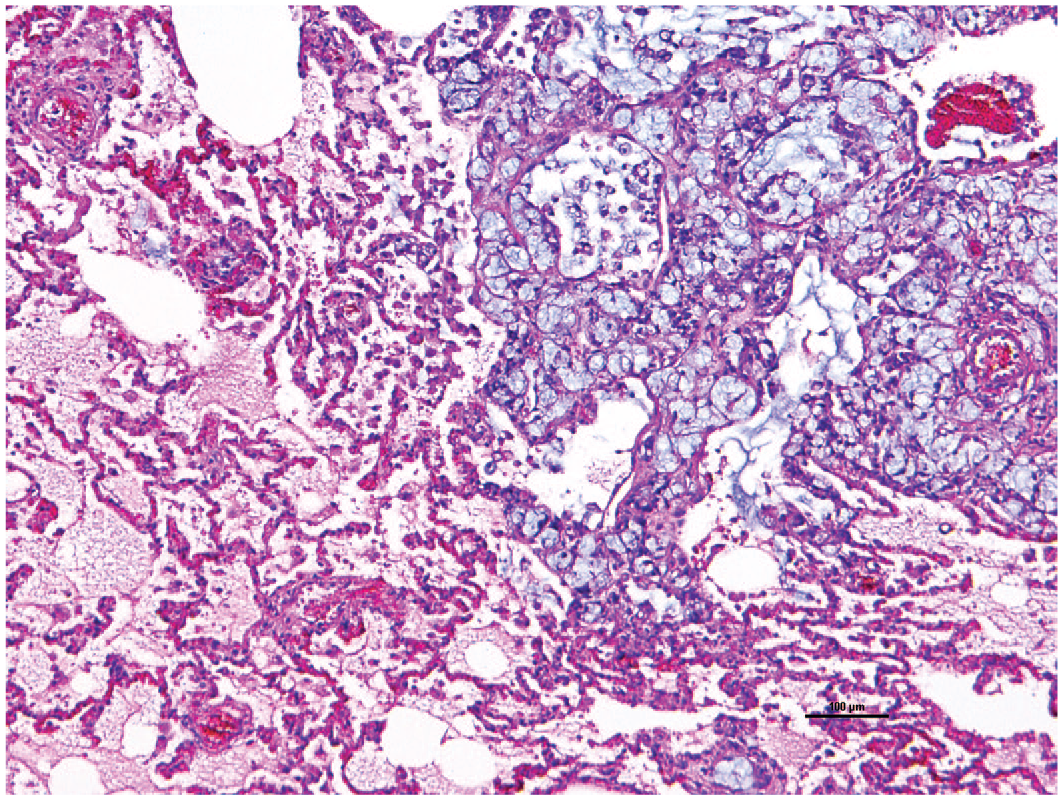
Lung metastases from duodenal carcinoma. Histological examination revealed infiltrative carcinoma with signet ring features or mucin deposition. 10X H&E.
In summary, marmosets are becoming more utilized in biomedical research due to multiple advantages including (1) NHP of smaller size, less cost for housing, (2) physiologic similarities to humans, (3) translatable hepatic metabolism, (4) higher numbers of litters per year, (5) genome is sequenced, molecular reagents are available, (6) immunologically similar to humans, (7) transgenic animals with germline transmission have been produced, and (8) are naturally occurring hematopoietic chimeras. With more use of marmosets, disease surveillance over a wide range of ages of marmosets has been performed. This has led to a better understanding of disease management of these spontaneous diseases that can occur in colonies. Knowledge of clinical signs and histologic lesions can assist in maximizing the colony’s health, allowing for improved outcomes in translational studies within biomedical research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
