Abstract
Spontaneous progressive glomerulonephropathy often occurs in common marmosets. However, there are few detailed reports concerning the age-related progressive process of glomerular changes. We discuss the glomerular changes in the early stage and the progressive changes in the advanced stage of nephropathy. We investigated the kidneys of common marmosets (2–11 years old; 9 males and 12 females) using hematoxylin and eosin, periodic acid–Schiff, periodic acid–methenamine-silver, and Masson’s trichrome (MT) stains and a transmission electron microscope. There was no remarkable change in urine cytology, hematology, or blood chemistry. In the early stage of nephropathy, effacement of podocyte foot processes was observed ultrastructurally even though there were no marked glomerular lesions in the light microscopy. Subsequently, mesangial proliferation occurred from the hilar to peripheral side along the tuft. In the middle stage, red deposits were visible at the glomerular basement membrane (GBM) and the mesangial region directly under the GBM (paramesangial area) with the MT stain. Electron dense deposits were seen at the same area. In the advanced stage, the irregularity became prominent with or without dense deposits. It is necessary to investigate in detail whether the change of podocyte in the early stage was immuno-mediated or due to podocyte failure.
Introduction
Spontaneous progressive glomerulonephropathy often occurs in common marmosets (Callithrix jacchus). These renal lesions are characterized by an increase in mesangial cells and matrix in the glomerular hilum, interstitial inflammatory cell infiltration, regeneration and hyperplasia of tubular epithelia, and proteinaceous cast in the tubuli. Glomerular sclerosis and tubulointerstitial nephritis were found in the advanced lesions. Previously, these renal lesions were described as “IgM nephropathy” because the frequency of the IgM deposition to the mesangium was high (Brack 1988, 1995; Brack and Weber 1995). Also, it was reported that IgA was involved in this nephropathy (Brack et al. 1999).
Recently, a correlation between expansion of mesangial matrix and age and between glomerular and tubulointerstitial lesions was reported by Isobe et al. (2012). They revealed that the increase in mesangial matrix progressed with age, and the tubulointerstitial lesions were correlated with the glomerular lesions. However, detailed morphological features of the primary and progressive glomerular lesions are not yet well known. The investigations of these morphological features are expected to contribute to the identification of specific pathogenesis of glomerulonephropathy in common marmosets.
In this study, we investigated the kidneys of common marmosets (2–11 years old; 9 males and 12 females), and the light microscopic findings were graded in 5 stages according to the progress. Moreover, we clarified the correlation of the renal lesions with age, and the ultrastructural features corresponding to each grade of the light microscopic lesions. This study revealed the primary glomerular change in the early stage and progressive glomerular change in the advanced stage of nephropathy as well as the relationship between the glomerular lesions and age of common marmosets.
Materials and Methods
Animals
Twenty-one common marmosets (C. jacchus) were evaluated. Nine were purchased from CLEA Japan Inc. (Tokyo, Japan), and the rest were bred at the Kashima laboratory of Mitsubishi Chemical Medience Corporation. Their ages ranged from 2 to 11 at the time of necropsy. There were 9 males and 12 females. The animals were housed in cages in an animal room maintained at a temperature of 26 ± 3°C and a humidity of 55 ± 20%. They were given CMS-1M (CLEA Japan Inc., Tokyo, Japan) and tap water. Prior to starting the study, the protocol was reviewed by the Institutional Animal Care and Use Committee and approved by the General Manager of the test facility in accordance with the “Guidelines for Animal Studies” (Nonclinical Research Center, Drug Development Service Segment, Mitsubishi Chemical Medience Corporation).
Urinalysis, Urine Cytology, Hematology, and Blood Chemistry
The animals were euthanized under anesthesia with pentobarbital sodium. Blood samples were collected from the abdominal vein. Urine samples were collected directly from the urinary bladder during the necropsy. The blood samples and urine samples were immediately used for the examination.
The number of red blood cell (RBC) and white blood cell (WBC) was measured with XT-2000iV (Sysmex Corporation, Hyogo, Japan). Serum urea nitrogen, creatinine, total protein, serum albumin, total serum cholesterol, and electrolyte were measured with TBA-200FR (Toshiba Medical System Corporation, Tochigi, Japan). Urinalysis was performed with the dipstick method for urinary protein, occult blood, and pH (Clinitech500, Siemense Healthcare Diagnostics K.K., Tokyo, Japan). Urine cytology was performed with urine sediment for crystal, RBC, WBC, epithelial cell, and cast. The measurement ranges of qualitative urinalysis and the criteria for urinary cytology are shown in Table 1.
Animal information, grade of renal lesion, urinalysis, urine cytology, hematology, and blood chemistry.
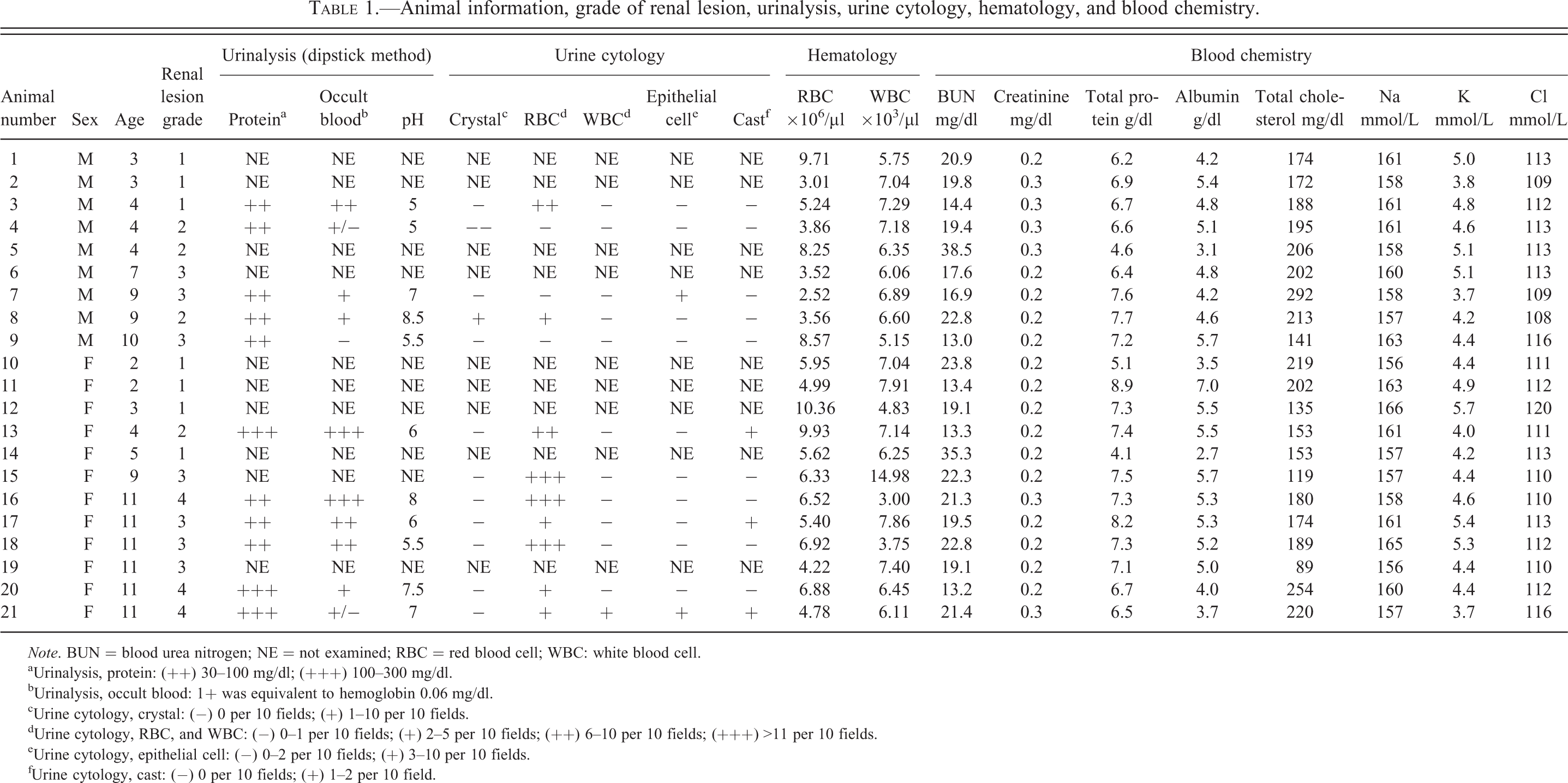
Note. BUN = blood urea nitrogen; NE = not examined; RBC = red blood cell; WBC: white blood cell.
aUrinalysis, protein: (++) 30–100 mg/dl; (+++) 100–300 mg/dl.
bUrinalysis, occult blood: 1+ was equivalent to hemoglobin 0.06 mg/dl.
cUrine cytology, crystal: (−) 0 per 10 fields; (+) 1–10 per 10 fields.
dUrine cytology, RBC, and WBC: (−) 0–1 per 10 fields; (+) 2–5 per 10 fields; (++) 6–10 per 10 fields; (+++) >11 per 10 fields.
eUrine cytology, epithelial cell: (−) 0–2 per 10 fields; (+) 3–10 per 10 fields.
fUrine cytology, cast: (−) 0 per 10 fields; (+) 1–2 per 10 field.
Histopathology
The kidneys were fixed in 10% neutral-buffered formalin, routinely embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (H&E), periodic acid–Schiff (PAS), periodic acid–methenamine-silver (PAM), and Masson’s trichrome (MT). The lesions were classified into five grades (0–4) based on the score of glomerular, tubular, and interstitial lesions with the H&E stain. The score of glomerular lesion corresponds with the previous report (Isobe et al. 2012).
Transmission Electron Microscopy (TEM)
Formalin-fixed kidneys from 4 animals (nos. 2, 5, 7, and 21) of each grade were used for the TEM. The cortex of each kidney was cut into small blocks and washed in distilled water for 30 min. They were postfixed in 1% osmium tetroxide and embedded in epoxy resin. Ultrathin sections were mounted on copper grids, stained with uranyl acetate and lead citrate, and examined with H-7600 TEM (Hitachi High-Tech Fielding Corporation, Tokyo, Japan).
Statistical Analysis
For each grade of renal lesion, data were presented as mean ± SD. Statistical analyses were performed using Microsoft Office Excel 2003 (Microsoft Corporation, Redmond, WA). The data were analyzed by one-way analysis of variance. Differences between groups were considered significant when p < .05.
Results
Urinalysis, Urine Cytology, Hematology, and Blood Chemistry
The results of urinalysis, urine cytology, hematology, and blood chemistry are shown in Table 1. Urinary protein was positive in all cases and occult blood was also positive in most cases. Urine pH ranged from 5 to 8. The urine of 4 males (nos. 1, 2, 5, and 6) and 6 females (nos. 10, 11, 12, 14, 15, and 19) could not be examined because it was directly collected from the urinary bladder during the necropsy. Conspicuous abnormalities were not found in the hematology or blood chemistry except for the WBC of animal number 15 (14.98 × 103 µl).
Light Microscopic Findings
The histopathological changes of the kidney were classified into 5 grades based on the histopathological score with the H&E stain. The distribution of the lesions in the whole kidney was classified into “focal” or “multifocal” (<80%) and “diffuse” (>80%). The distribution of the lesions in the whole glomerulus was classified into “segmental” (a lesion involving a part of the glomerulus) and “global” (a lesion involving the whole glomerulus). The glomerular lesion scoring was performed as follows; no change as score 1, increase in mesangial matrix predominantly in the hilar area in the glomerulus or segmental mesangial proliferation (focal or multifocal) as score 2, increase in mesangial matrix predominantly in the hilar area in the glomerulus or global mesangial proliferation (diffuse) as score 3. The regeneration tubuli and hyaline cast scoring was performed as follows; no change as score 0, few (within 4 areas) and focal or multifocal as score 1, many (up to 5 areas) or diffuse as score 2. The interstitial cell infiltration scoring was performed as follows; no change as score 0, sporadically in the hilar area of the kidney or the perivascular area as score 1, few (within 4 areas) and focal or multifocal as score 2, many (up to 5 areas) or diffuse as score 3. The interstitial fibrosis scoring was performed as follows; no change as score 0, focal as score 1, diffuse (more severe than score 1) as score 2 (Table 2). Finally, the renal lesion grading was performed as follows; the total score 0 to 3 as the renal lesion grade 0, 4 to 5 as grade 1, 6 to 7 as grade 2, 8 to 10 as grade 3, and 11 to 12 as grade 4.
Criteria for histopathological score.

Note. aSegmental, a lesion involving a part of the glomerulus; global, a lesion involving the whole glomerulus.
bFocal/multifocal, a lesion involving a part of the kidney (<80%); diffuse, a lesion involving all or nearly all area (>80%).
The grade of each animal is shown in Tables 1 and 3. The kidneys of 3 males (nos. 1, 2, and 3) and 4 females (nos. 10, 11, 12, and 14) were classified as grade 1. Ages ranged from 2 to 5 years old. Three males (nos. 4, 5, and 8) and 1 female (no. 13) were classified as grade 2. Ages ranged from 2 to 9 years old. Three males (nos. 6, 7, and 9) and 4 females (nos. 15, 17, 18, and 19) were classified as grade 3. Ages ranged from 7 to 11 years old. Three females (nos. 16, 20, and 21) were classified as grade 4. They were all 11 years old. The grade of renal lesion tended to become higher with age. However, there was no clear difference between the sex and grade of renal lesion.
Histopathological score and grade of renal lesion.
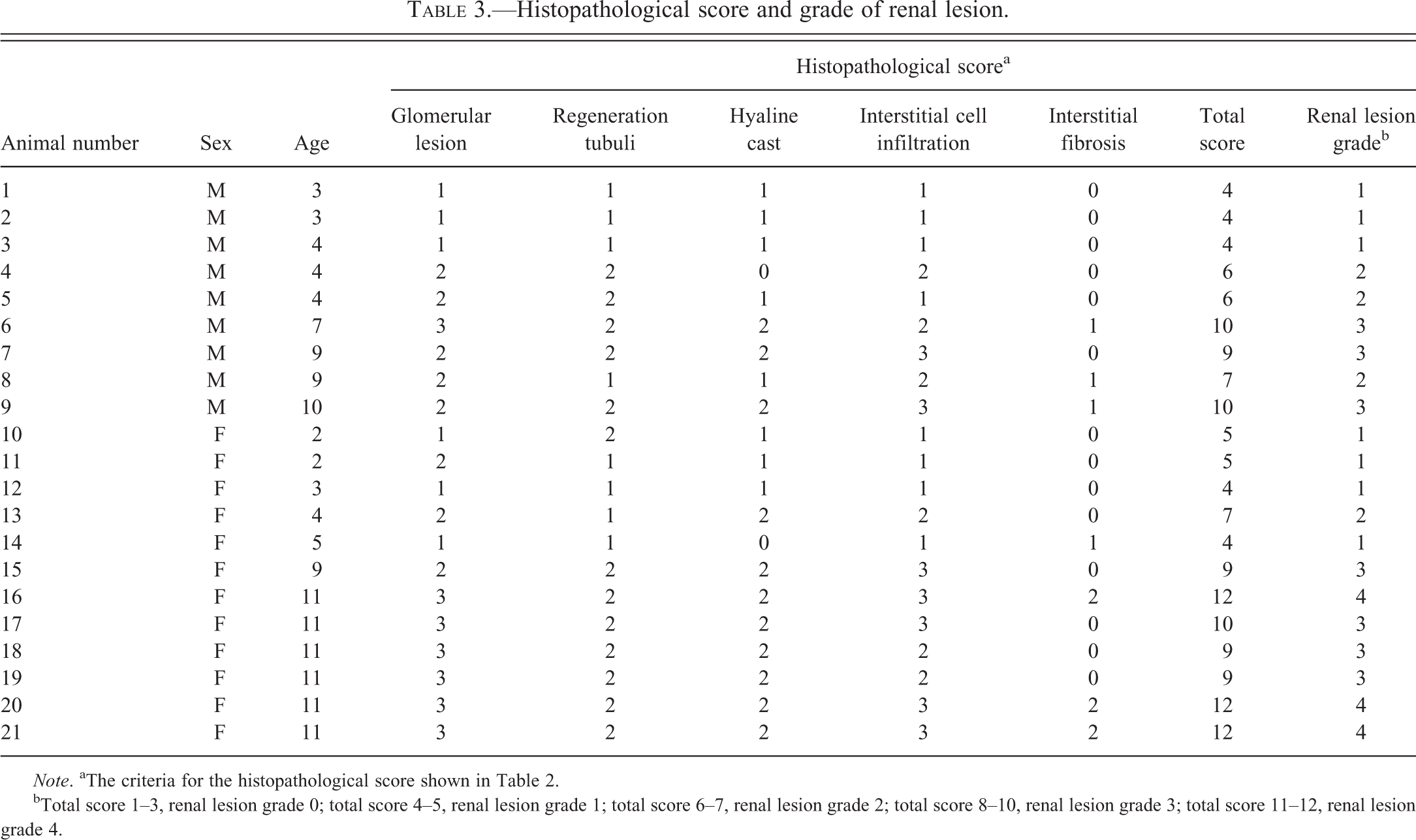
Note. aThe criteria for the histopathological score shown in Table 2.
bTotal score 1–3, renal lesion grade 0; total score 4–5, renal lesion grade 1; total score 6–7, renal lesion grade 2; total score 8–10, renal lesion grade 3; total score 11–12, renal lesion grade 4.
The details of the histopathological findings observed with the H&E, PAS, PAM, and MT stains are described below: Grade 0: No animal was classified as grade 0 Grade 1: Glomerular lesions could not be detected by light microscopy in these cases (Figure 1A and B). Instead, tubulointerstitial lesions, such as hyaline cast in tubuli, focal regenerative tubuli, and focal interstitial lymphocytic infiltration were observed sporadically (Figure 1C). Grade 2: Enlarged glomeruli were present focally or multifocally. Focal or multifocal proliferation of mesangial cells at the glomerular hilum and extraglomerular mesangial cells (known as Lacis cells or Goormaghtigh cells) were obvious in this stage (Figure 2A). However, neither the peripheral mesangial proliferation nor the change of capillary loop was seen in the PAM- (Figure 2B), PAS-, or MT-stained sections. Tubulointerstitial lesions greater than those of grade 1 were observed (Figure 2C). Grade 3: Abnormal glomeruli were present multifocally or diffusely. These glomeruli showed slight proliferation of mesangial cells and a conspicuous increase in mesangial matrix along the glomerular tuft (Figure 3A and B). The capillary loops were partially dilated, but the structure of the capillary lumen remained intact. In the PAM- and MT-stained sections, the glomerular capillary loops showed partial ruggedness and thickening. In the MT-stained sections, a red deposit (about 10 µm diameter) was seen in the mesangial region directly under the glomerular basement membrane (GBM; paramesangial area; Figure 3C). Tubulointerstitial lesions were more frequent and greater than those of grade 2. Grade 4: Abnormal glomeruli were present diffusely. Mesangial matrix proliferation expanded to the peripheral glomerular tuft. However, the mesangial cell proliferation in the peripheral glomerular tuft was not remarkable (Figure 4A). Eosinophilic mesangial deposit was seen in the paramesangial area (Figure 4A; arrow). A number of capillaries collapsed. The Bowman’s capsule tended to dilate, and its basement membrane thickened. Some of the glomeruli partially adhered to the Bowman’s capsule. In the PAM- and MT-stained sections, the capillary walls had some loop-shaped double outlines with or without red deposits (Figure 4B; arrow and arrow heads). There were many red depositions not only in the capillary walls but also in the paramesangium (Figure 4B; arrows). Interstitial lesions including fibrosis were spread widely (Figure 4C). Diffuse tubular lesions were also present partially with hyperplastic tubuli (atypical tubular regeneration)
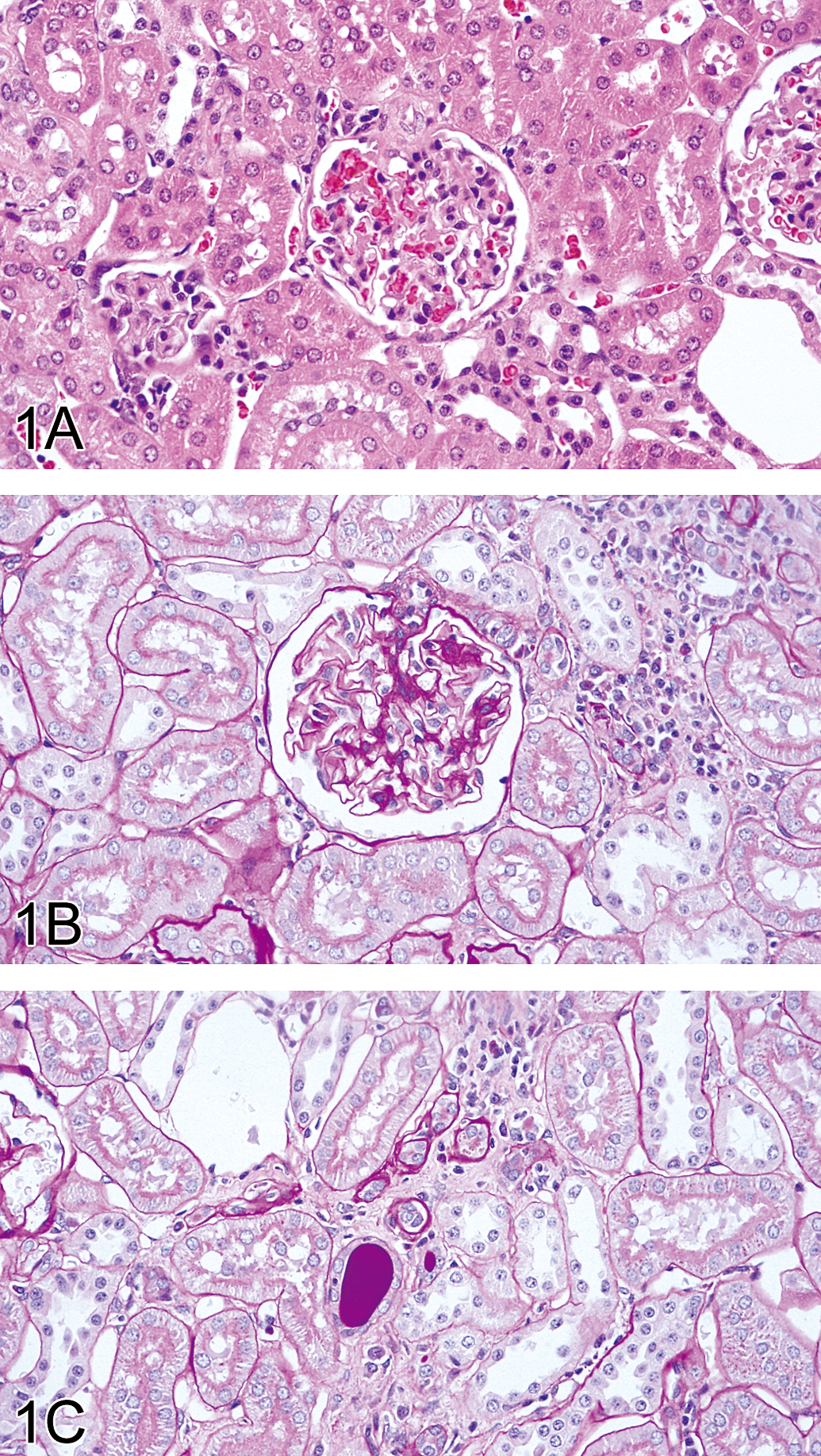
Kidney of animal no. 2 classified as grade 1. (A) No remarkable change in glomerulus H&E. Original magnification objective = 40×. (B) No proliferation of mesangium in glomerulus and the cell infiltration in the interstitium. PAS stain. Original magnification objective = 40×. (C) Interstitial cell infiltration and hyaline cast in the tubuli. PAS stain. Original magnification objective = 40×.
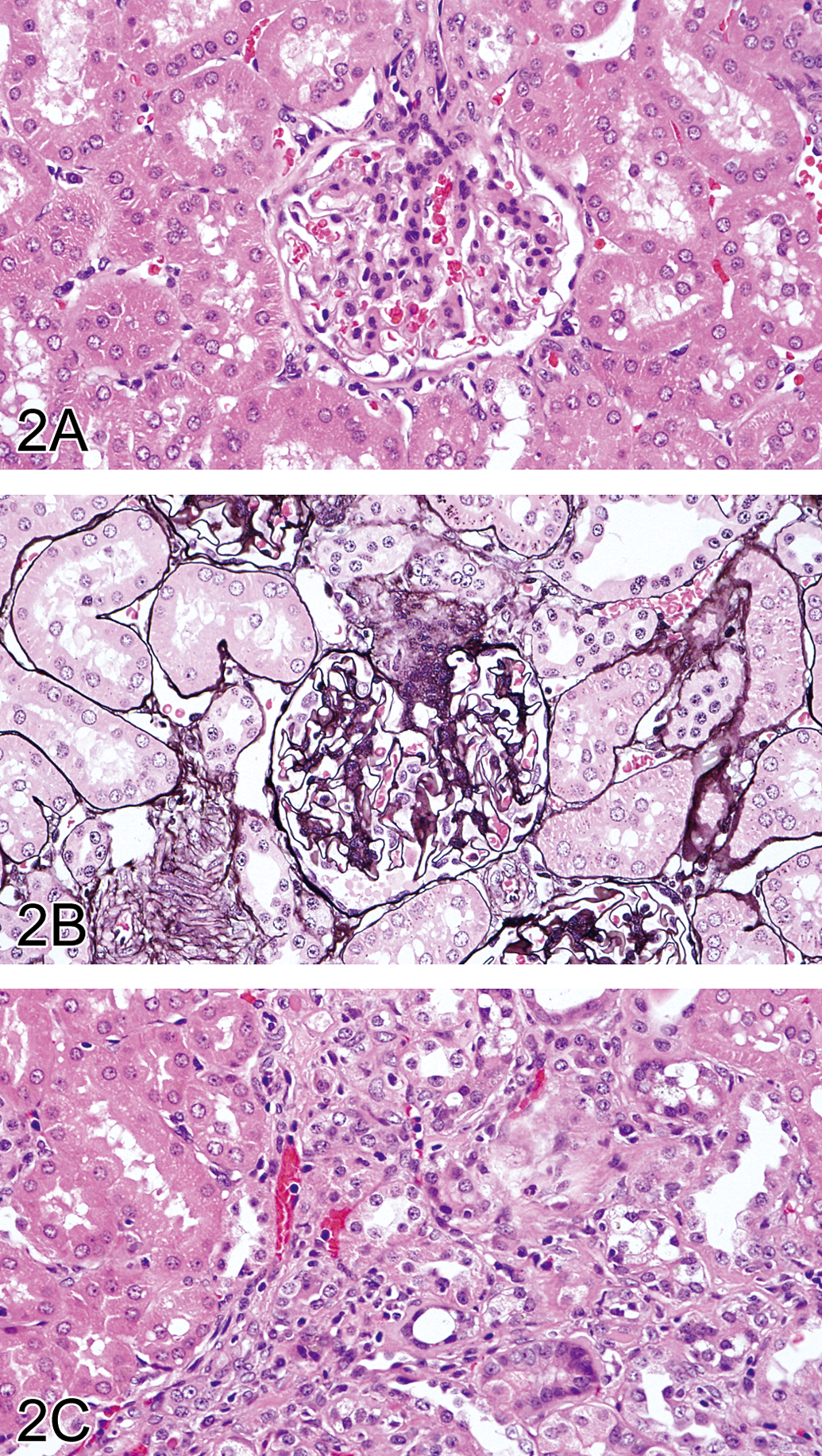
Kidney of animal no. 5 classified as grade 2. (A, B) Proliferation of mesangial cell at the glomerular hilum. A, H&E; B, PAM stain. Original magnification objective = 40×. (C) Focal regenerative tubuli and an inflammatory cell infiltration in the interstitium. H&E. Original magnification objective = 40×.
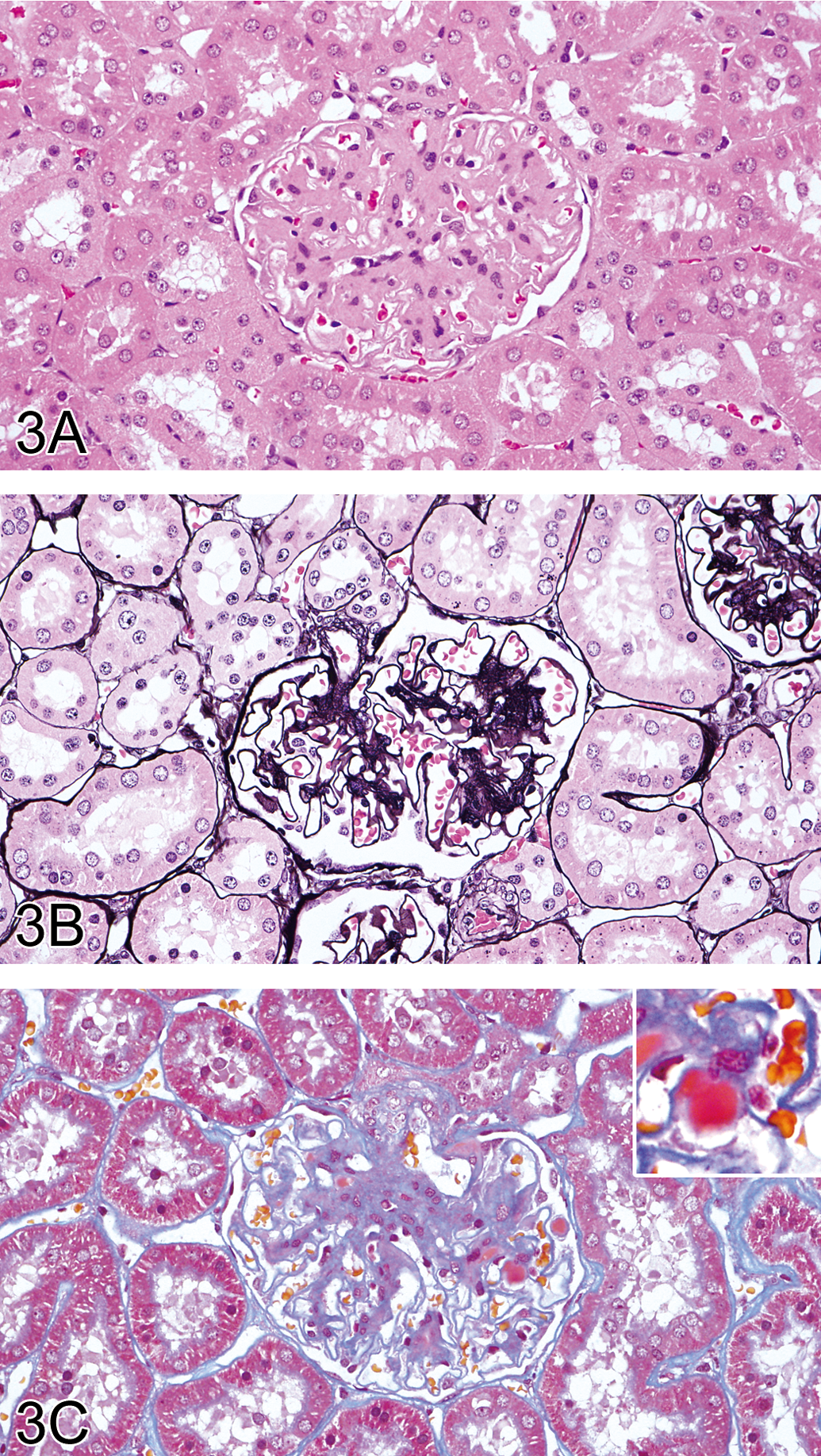
Kidney of animal no. 7 classified as grade 3. (A, B) Enlargement of glomerulus and an increase in mesangial matrix along the tuft. A, H&E; B, PAM stain. Original magnification objective = 40×. (C) An increase in mesangial matrix and red deposits in the paramesangial area (inset of Figure 3C). MT stain. Original magnification objective = 40×.
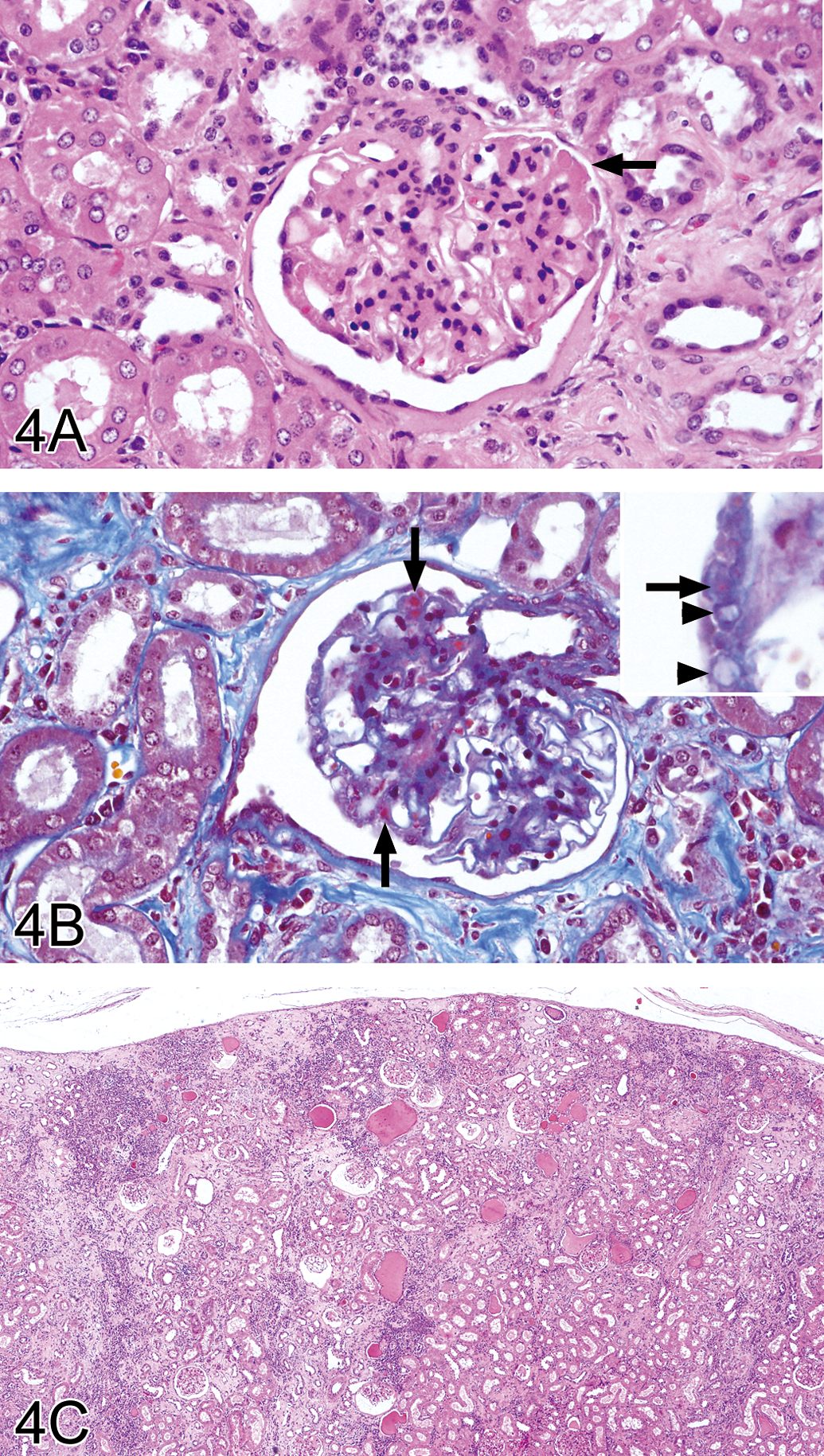
Kidney of animal no. 21 classified as grade 4. (A) Diffuse mesangial matrix proliferation, eosinophilic mesangial deposit (arrow), and thickening of basement membrane of Bowman’s capsule. H&E. Original magnification objective = 40×. (B) Some loop-shaped double outlines of capillary walls (arrow heads) and many red depositions not only in the capillary walls but also in the paramesangium (arrows). MT stain. Original magnification objective = 40×. (C) Interstitial fibrosis, inflammatory cell infiltration, and hyaline cast in the tubule. H&E. Original magnification objective = 4×.
Transmission Electron Microscopic Findings
In the glomeruli of a representative case (no. 2) classified as grade 1, no significant change was seen in the mesangial area. The structure of the lamina densa, lamina rara interna, and lamina rara externa of the GBM appeared normal, but the GBM partially and slightly thickened (about 400 nm thickness). Effacement of foot processes of the epithelial cells (podocytes) was observed near the thickened GBM (Figure 5).
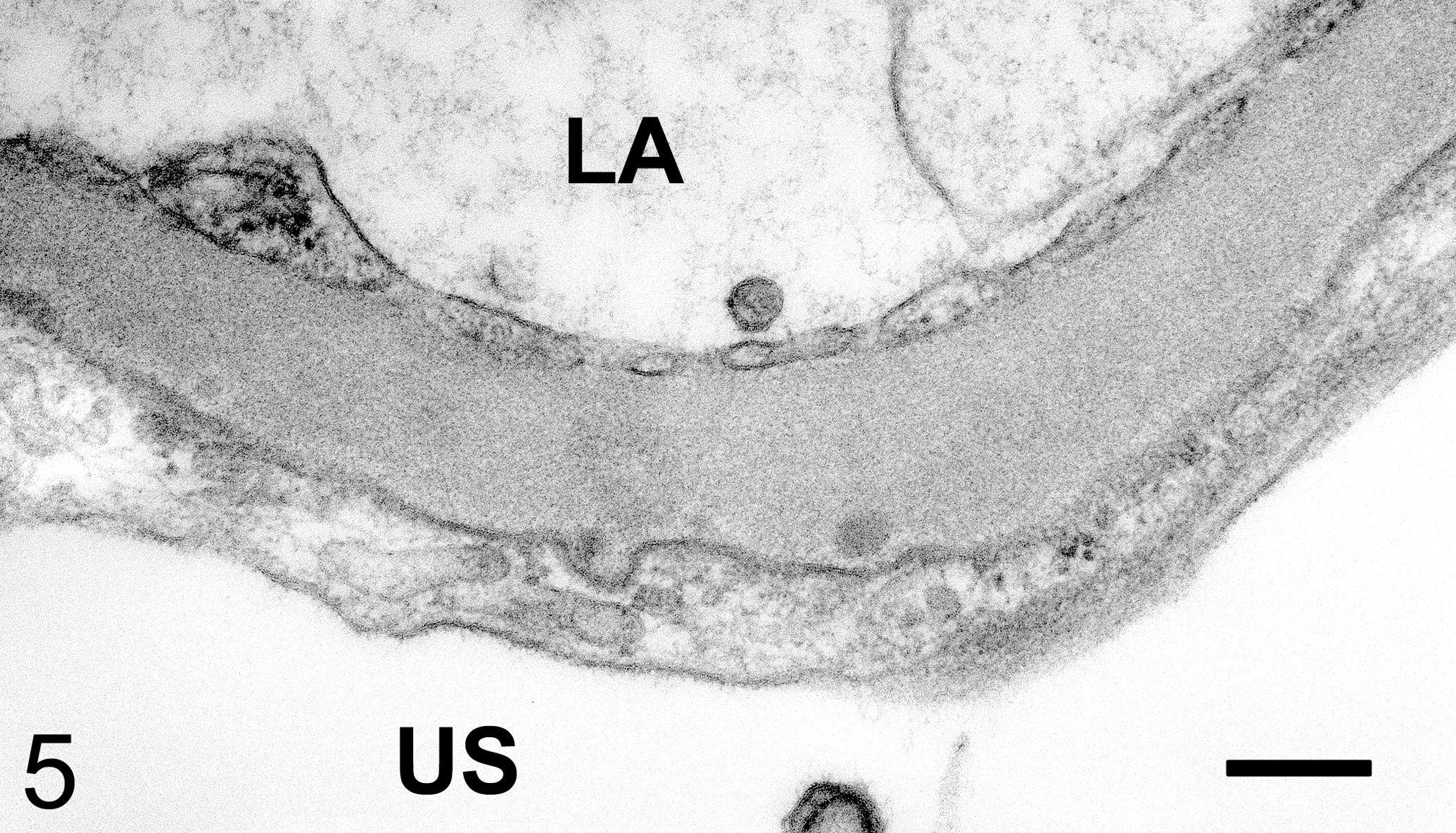
Glomerulus from animal no.2 classified as grade 1. Slight thickening of capillary basement membrane and effacement of podocyte foot processes. Transmission electron micrograph. LA, luminal area; US, urinary space. Bar: 250 nm.
In the glomeruli of a representative case (no. 5) classified as grade 2, an increase in the mesangial matrix and proliferation of mesangial cells in the hilum were obvious (Figure 6A). The GBM was much thicker in grade 2 than in grade 1, and the trilaminar structure was partially unclear because the lamina rara interna and lamina densa frayed and separated. Effacement of podocyte foot processes expanded much wider in grade 2 than in grade 1 (Figure 6B).
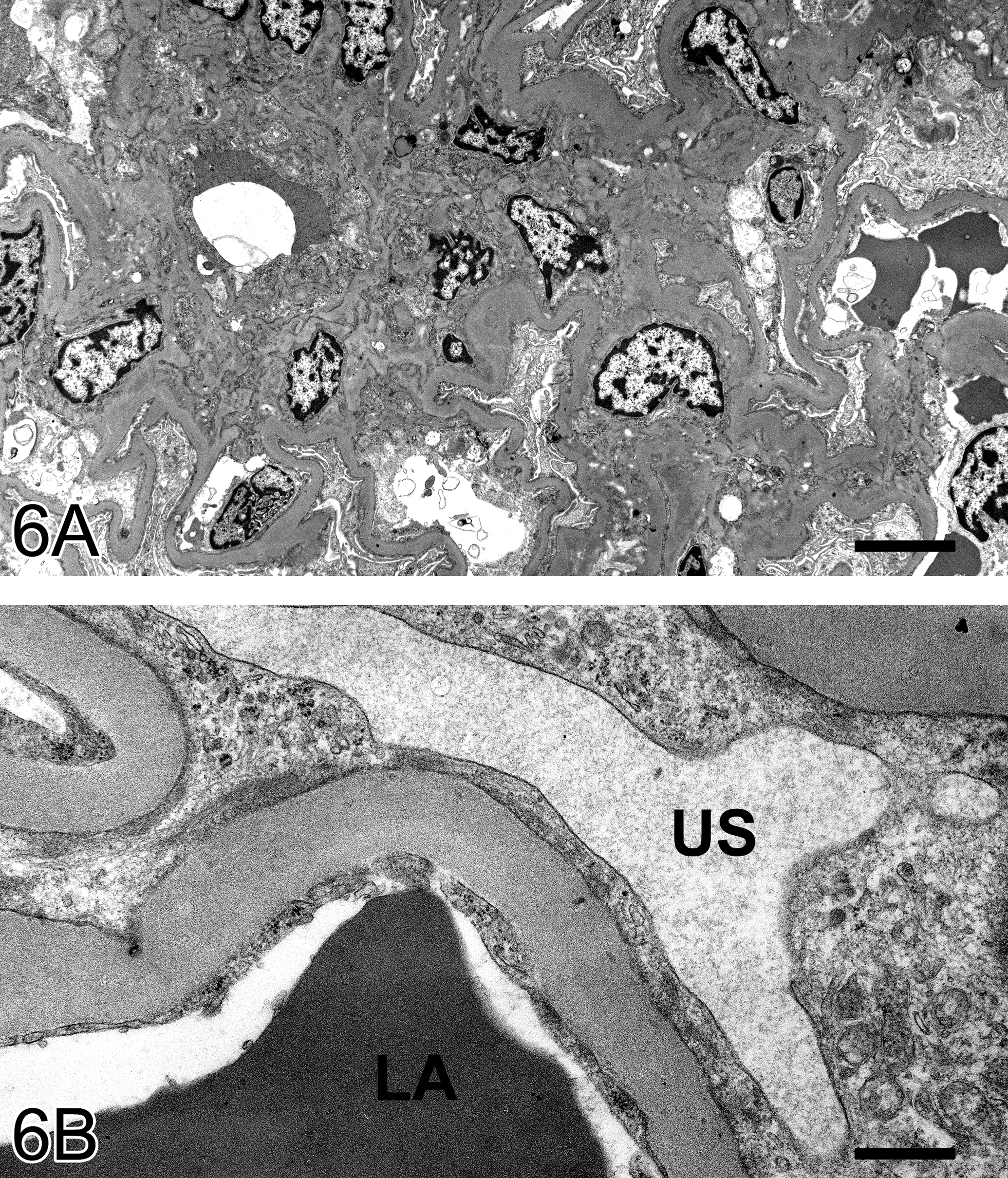
Glomerulus from animal no. 5 classified as grade 2. (A) An increase in the mesangial matrix and proliferation of mesangial cells in the hilum. (B) More thickening of capillary basement membrane than in grade 1, and a partially irregular of trilaminar structure. Transmission electron micrograph. LA, luminal area; US, urinary space. Bar: A, 5 µm; B, 667 nm.
In the glomeruli of a representative case (no. 7) classified as grade 3, the mesangial matrix increased in the mesangial area. The electron dense deposit suggested to be corresponding to the red deposits in the MT-stained sections was observed in the paramesangial and mesangial area (Figure 7A; arrows). The uneven outline of the GBM, especially the epithelial side, was widely observed, and the lamina densa partially laminated and reticulated (Figure 7B). As the lamina densa partially showed low electron density, the distinction between the lamina densa and lamina rara was not obvious. Effacement of podocyte foot processes was observed near the irregular GBM more widely in grade 3 than in grade 2.

Glomerulus from animal no. 7 classified as grade 3. (A) The electron dense deposit in the paramesangial and mesangial area (arrows). (B) The irregular outlines of the GBM, especially epithelial side, and the lamina densa partially separated. Transmission electron micrograph. LA, luminal area; US, urinary space. Bar: A, 3.3 µm; B, 1.3 µm.
A low electron density and an empty area were seen focally and sporadically at the peripheral GBM in the glomeruli of a representative case (no. 21) classified as grade 4 (Figure 8A). The invasion of mesangial cells was recognized in the subendothelial layer accompanied with their matrix near the paramesangial area (mesangial interposition; Figure 8B; arrows). Some organelles such as mitochondria and rough endoplasmic reticulum developed in mesangial cells. Effacement of podocyte foot processes and appearance of microvilli on the urinary space side were evident in podocytes. The GBM showed marked irregularity and protruded to the epithelial side, partially to the endothelial side. A large number of electron dense deposits developed within the protruded GBM and shaped like humps (Figure 8C; arrows). The electron dense deposits partly washed out (dissolved). The granular structures and cytoplasm including cellular organella were observed in the washed-out areas (Figure 8C; arrow heads). These lesions of the GBM may correspond to the loop-shaped double outlines and red deposits in the light microscopy.
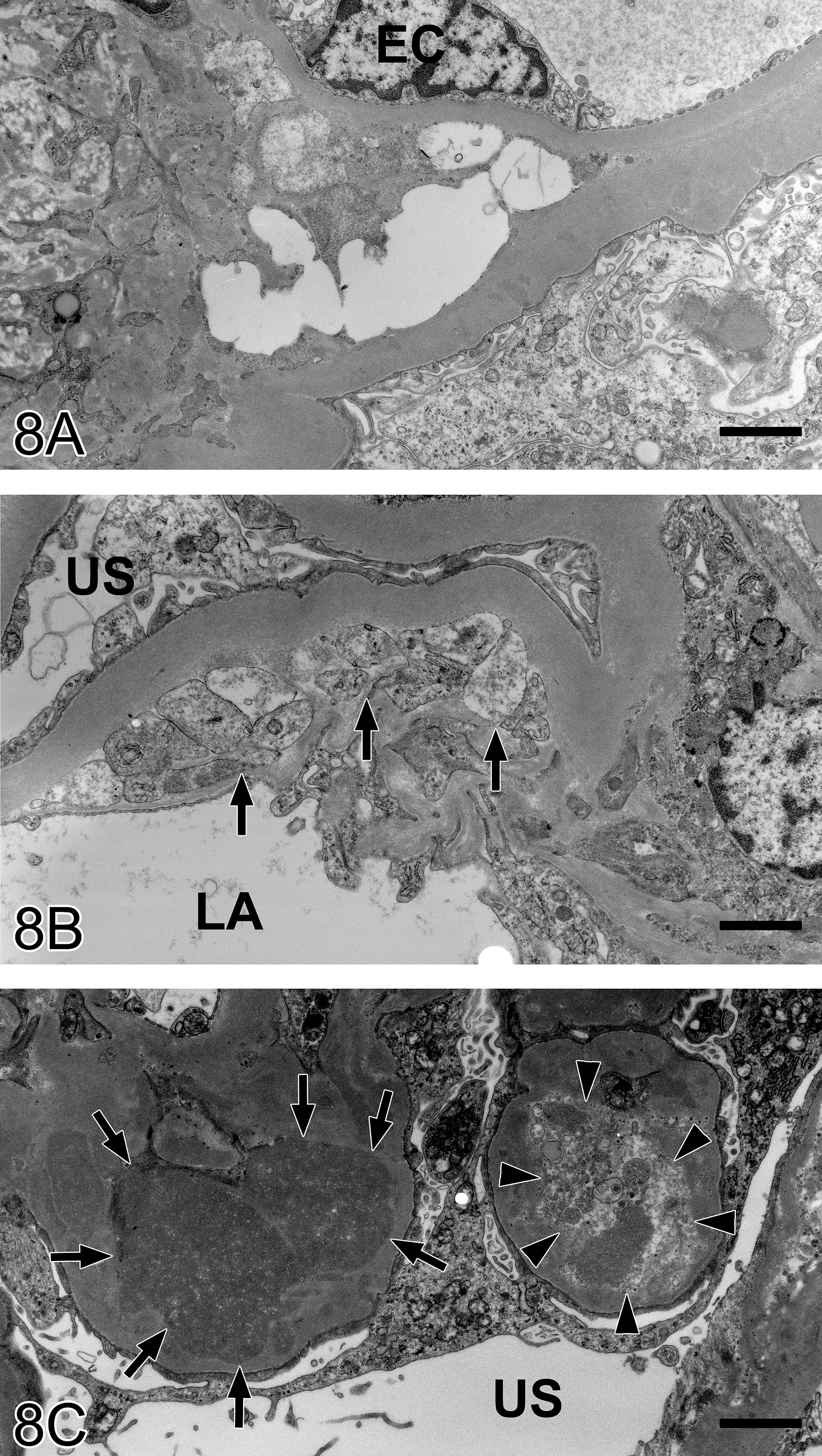
Glomerulus from animal no. 21 classified as grade 4. (A) Low electron density and an empty area in the mesangial area. (B) Mesangial interposition near the paramesangial area (arrows). (C) A number of electron dense deposits within the protruded GBM (arrows), dissolving the electron dense deposits and a granular structure in the washed-out area (arrow heads). Transmission electron micrograph. EC, endothelial cell; LA, luminal area; US, urinary space. Bar: A, 2.0 µm; B, 1.4 µm; C, 2.0 µm.
Statistical Analysis
There were no significant parameters for any grade of renal lesions (Table 4). The correlation between the grade of renal lesion and these parameters remained unclear.
Hematology and blood chemistry for each grade of renal lesion.
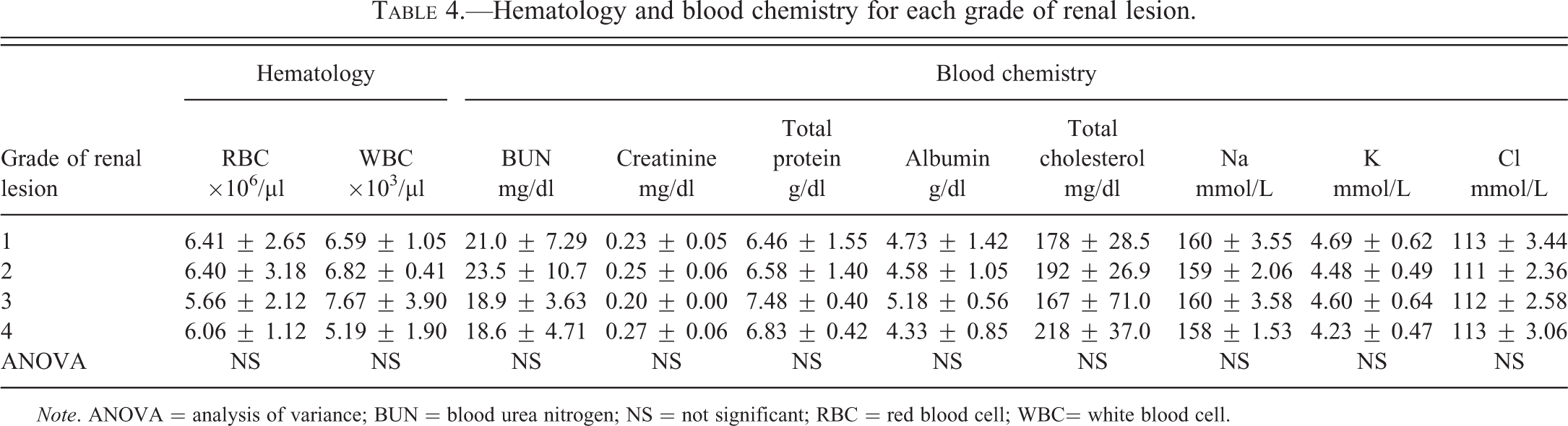
Note. ANOVA = analysis of variance; BUN = blood urea nitrogen; NS = not significant; RBC = red blood cell; WBC= white blood cell.
Discussion
We revealed morphological details of the age-related glomerulonephropathy in this study. The lesions in the early stage included effacement of podocyte foot processes and partial thickening of the GBM. They were followed by proliferation of mesangial matrix and cells, which became visible in the glomerular hilum, and gradually expanded along the tuft. The deposits were recognized at the paramesangial area and GBM in the middle stage by the MT stain and electron microscopy. The GBM showed irregular outlines of the epithelial side and a decrease in density of the lamina densa, and the irregularity became prominent with or without dense deposits in the advanced stage.
Effacement of podocyte foot processes and thickening of the GBM were likely to be the first event of this glomerulonephropathy because these lesions were observed before being visible in the light microscopy. Similar ultrastructural findings in humans are known as minor glomerular abnormalities. The term minor glomerular abnormalities is the morphological classification proposed by World Health Organization and not a clinical diagnosis. The category includes minimal change disease (Churg and Sobin 1982). The morphological features are that the glomeruli appear as normal or minor changes by light microscopy, and effacement of podocyte foot processes is seen by TEM. This change is not particular to certain glomerulonephritis because the effacement of podocyte foot processes is seen in various glomerulonephritis. Therefore, these morphological features indicate the early stage of glomerulonephropathy, but no pathognomonic signs to a specific glomerulonephropathy. However, IgM deposition was found in the glomeruli in a previous report on the common marmoset nephropathy (Brack 1995). IgM nephropathy in humans was classified as one of the diseases, which is a minimal change disease or focal segmental glomerulosclerosis (Al-Eisa et al. 1996; Maruyama et al. 2006; Pardo et al. 1984). Therefore, it is necessary to investigate in detail whether the change of podocyte in the early stage of the glomerulonephropathy was also immuno-mediated. It is because there is a possibility that the cause of the effacement was a podocyte failure (Chugh, Clement, and Macé 2011; Wei and Reiser 2011). The mechanism of the early change still remains unclear.
The red deposit in the MT-stained sections and the electron dense deposit suggested that the common marmoset nephropathy was also an immuno-mediated nephropathy. A histopathological feature of human IgA nephropathy was the eosinophilic mesangial deposit showing red in the MT-stained sections (D’Amico 1989). In a previous study, IgA immuno complex and anti-gliadin IgA were found in the common marmoset nephropathy (Brack et al. 1999). Moreover, a wasting syndrome often occurs in common marmosets. The syndrome was a systemic wasting disease including tubulointerstitial nephritis (Brack and Rothe 1981). The syndrome involves IgA-gliadin antibodies in the glomeruli and circulating immune complexes that contain IgA (Schroeder et al. 1999). Thus, the previous reports showed that immunoglobulin is deeply correlated with the renal lesions in the common marmosets. A hump-like formation and the electron dense deposit were similar to the lesions in membranous nephropathy in humans. The membranous nephropathy in humans was characterized by electron dense deposits at the subepithelial aspect of the GBM in the early stage and the poor reaction in the mesangium (Short and Mallick 1989). Thus, the mesangial reactions of glomerulonephropathy in common marmosets were different from the membranous nephropathy in humans. Our histopathological results suggested that immunoglobulin was involved in the expression of glomerular disorder, but the nephropathy was not the same as membranous nephropathy in humans.
Proliferation of the mesangial matrix being more remarkable than mesangial cells is considered one of the features of the glomerulonephropathy in common marmosets. The same mesangial proliferation pattern was reported in a previous study in common marmosets (Brack 1988; Brack and Weber 1995). Nonhuman primates often have the spontaneous progressive nephropathy. For example, spontaneous mesangioproliferative glomerulonephritis was often found in pig-tailed macaques (Giddens et al. 1981; Boyce, Giddens, and Seifert 1981). The features of the disease were age-related aggravations with tubulointerstitial lesions, similar to the common marmosets. However, global mesangial cell proliferation and no GBM change are different from common marmosets when it was examined by a light microscope. Thus, the glomerulonephropathy in common marmosets is the particular nephropathy morphologically.
Immunohistochemical stain could not be performed in this study since we did not prepare frozen sections of the kidney. We suggest that immunohistochemical examinations are essential to reveal the relationship between the primary and progress lesions and immunoglobulin deposition in future studies. Moreover, the relationship between the histopathological grade of glomerulonephropathy and our routine examination parameters remains unclear. We suggest that measuring sensitive biomarkers of glomerular injury such as plasma cystatin C, urinary b2-microglobulin, and kidney injury molecule-1 may facilitate the ability to judge the degree of progressive glomerulonephropathy in common marmosets (Pinches et al. 2012a, 2012b, 2012c).
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
We would like to thank Dr. Masami Suzuki and Dr. Kaori Isobe of Chugai Pharmaceutical Co., Ltd. for valuable comments on this study.
