Abstract
Cancer survivors may experience long-term adverse effects of cancer treatments such as premature ovarian failure and infertility. We aimed to investigate the potential effects and toxicity of bortezomib (BTZ) as an effective anticancer drug on ovaries, raise awareness to the negative consequences of the treatment, and help increase the quality of life after treatment. Mice were distributed into bortezomib (BTZ1, BTZ2) and saline-injected control groups (C1, C2) at a dose of 1 mg/kg twice per week for 6 weeks. We sacrificed C1, BTZ1 groups at day 1 and C2, BTZ2 groups at 4 weeks after the last injection. Ovary samples were examined using histopathological and immunohistochemical methods. Ovarian follicle impairment was detected on BTZ-treated mice and was associated with a statistically significant decreased population of primordial and antral follicles compared with control groups. In experimental groups, Caspase-3 and Ki67 expressions were increased, whereas estrogen receptor alpha (ERα) and progesterone receptor (PR) expressions were decreased in various developmental stages of follicles. BTZ specifically targets granulosa cells by inducing granulosa cell apoptosis and may have long-term effects on follicles. Bortezomib treatment may adversely affect ovarian function by accelerating ovarian reserve depletion and changing ERα and PR hormone levels that can cause fertility problems in the long term.
Introduction
Novel approaches in cancer therapy have led to increased rates of cancer survivors who then may experience long-term adverse effects of cancer treatments including the impact on fertility. Women surviving from cancer may face potential risks of chemotherapy on the female genital system such as reduced ovulation, reduction in ovarian size, amenorrhea, and difference in hormone levels.1,2 Thus, it is essential to know the potential long-term effects of new neoplastic drugs to prevent some of the side effects and help increase the quality of life after treatment.
The major component of the ubiquitin-proteasome system (UPS) is the proteasome complex, which exists in both cytoplasm and nucleus of eukaryotic cells. 3 UPS has crucial functions, including the fact that many intracellular proteins are degraded through this pathway. 4 UPS substrates include proteins involved in cellular processes such as cell cycle, growth transcription, DNA repair, cell survival, and apoptosis 4 that perform important roles on cellular homeostasis. 5
Bortezomib, an inhibitor of 26S, is a water-soluble dipeptidyl boronic acid. 6 It is the first approved proteasome inhibitor used in clinic for the treatment of the second most common hematological cancer, multiple myeloma (MM). 7 Bortezomib has additionally demonstrated antitumor activity in the treatment of certain solid tumors such as lymphoma, ovarian, prostate, lung, pancreatic, colorectal and gastroesophageal cancer, melanoma, and neuroblastoma.8-11 Bortezomib induces apoptosis in tumor cells and functions by distribution of intrinsic and extrinsic cell signaling pathways, such as cell cycle regulation, cell adhesion, and gene transcription. 12 This activity leads cancer cells to induce apoptosis and inhibits migration of cancer cells and tumor angiogenesis. 13 The most common serious side effects experienced by patients using this medicine are asthenic conditions, neutropenia, thrombocytopenia, peripheral neuropathy, anemia, and heart failure.14,15
There are existing studies on the effects of bortezomib on reproductive system. Studies about this chemotherapeutic agent mostly focused on the male genital system; however, there is very limited information on the effects of BTZ on the female reproductive organs. Researchers reported that BTZ causes long-term testicular dysfunction and testicular toxicity induced via upregulation of oxidative stress AMP-activated protein kinase activation and prevents normal development of germ cells.16,17 Unfortunately, we can only reach animal toxicology data about the female genital system from Food and Drug Administration (FDA) reports on rat and hamster ovary cells and pregnant rabbits. FDA reported that BTZ is not genotoxic based on Ames test applied in vitro hamster ovary, whereas teratological effects were found in rat and rabbit. However, there was no formal assessment on fertility or during peri- and postnatal development. 15
Estrogen is a primary steroid hormone and has essential functions in the female reproduction and other systems such as neuroendocrine, vascular, skeletal, and immune. 18 Estrogen acts by binding to two specific different nuclear receptors: estrogen receptor alpha (ERα) and estrogen receptor beta (ERβ). Studies indicated that ERα knockout mouse is infertile because the depletion of this gene disrupts the ovarian and uterine function. 19 ERα is predominantly expressed in the tissues including uterus, ovaries, cervix, vagina, and breasts in females. 20 Another important hormone in the female reproductive system is progesterone (P4). P4 mainly performs the establishment of pregnancy and maintenance and also plays crucial roles in the ovary such as regulating ovulation and luteinization. 21 The biological effects of P4 are mediated via progesterone receptor (PR) (which is expressed as two isoforms PRA and PRB) that belongs to ligand-dependent nuclear receptor superfamily. 22 PRA specifically functions in ovary and is essential for ovulation and uterus. 23
Ovary is one of the most toxicant drug-sensitive organ that includes various cell types and follicles and can be the potential target for damage during anticancer therapies. In light of all this information, for the first time we aimed to understand the pathogenesis of chemotherapy-induced ovarian toxicity of bortezomib by assessing the expressions of apoptosis marker Caspase-3 (Cas-3), proliferation marker Ki67, as well as ERα and PR because of the essential roles on female reproduction.
Materials and Methods
Animals
All experiments were conducted in adult C57BL/6 female mice (n = 24) aged 9 weeks. Mice were housed in a temperature, humidity-controlled vivarium (12-hour light: dark cycle) and were fed ad libitum. Animals were fed with a soy-free scientific 5V01 diet (Lab Diet PMI Nutrition International LLC, Brentwood, MO) to eliminate the estrogen-like effects of phytoestrogens. The general condition of the mice was closely monitored daily and body weight was recorded twice weekly before the injections. Animal Welfare Act was in accordance with institutional guidelines of Burdur Mehmet Akif Ersoy University and the study was approved by the Veterinary Faculty Experimentation Ethics Committee of Mehmet Akif Ersoy University (number 348-2017).
Drug Preparation and Treatment
Bortezomib (3.5 mg powder) was obtained from the pharmacy (Millennium Pharmaceuticals, Cambridge, MA) and suspended in 3.5 ml, 0.9% sterile saline solution. Injections were administered by intraperitoneally in a volume of 0.01 ml/g body weight via 28-gauge needles. Mice were distributed into four different groups (n = 6/group): BTZ treatment groups 1 and 2 (BTZ1, BTZ2) and control groups 1 and 2 (C1, C2). BTZ dose was chosen to be 1 mg/kg, two times a week (42.8 mg/m2), similar to the standard dose used in cancer patients with a schedule of 1.3 mg/m2 four times each, 21 days during eight cycles (41.6 mg/m2).12,24,25 In this study, we administered 3 cycles of treatment consecutively. The first cycle of therapy was constituted by manufacturer on days 1-4-8 and 11. 26 Mice were injected either 1 mg/kg BTZ in saline (BTZ1, BTZ2) or with saline alone as a vehicle (C1, C2) twice per week for 6 weeks, on days 1-4-8-11-15-18-22-25-29-32-36-39. 12 At day 40, the day after the last injection, mice from BTZ1 and C1 group were sacrificed for analyses. The animals from BTZ2 and C2 groups were left untreated for an additional 4-week period and they were sacrificed at the end of this period 12 (Figure 1) for further analyses to evaluate the potential recovery in ovaries.
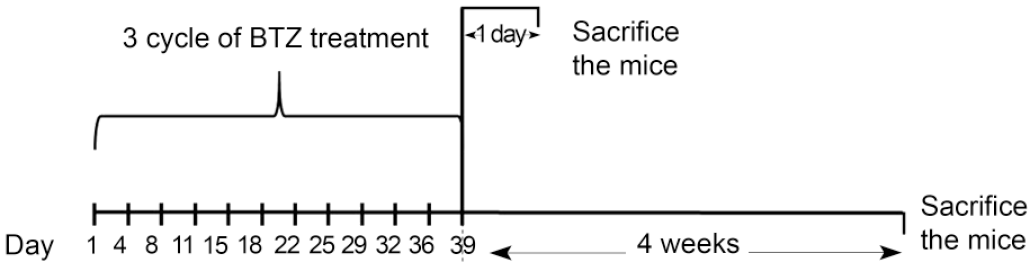
Timeline of the experimental procedure for bortezomib administration: The chart shows mice that were treated with bortezomib 2 times per week for 6 weeks period (injection was done on 1-4-11-15-18-22-25-29-32-36-39th days) and sacrificed after 1 day or 4 weeks after the last injection. For this study, animals were treated with bortezomib or saline as a vehicle (1 mg/kg of body weight, n = 6) as indicated above.
Histopathological Examination and Classification of Follicles
Mice were euthanized by cervical dislocation under general anesthesia with intraperitoneal injection of ketamine/xylazine (ketamine: 80 mg/kg; xylazine: 7.5 mg/kg) at the end of each treatment protocol. Ovary tissues from different groups were isolated and fixed in Bouin’s solution, embedded in paraffin, and five serial sections were scored from each paraffin block (5 μm). Sections were stained with hematoxylin and eosin (H&E) (Sigma-Aldrich, St.Louis, MO, USA). Afterward, the slides were dehydrated in 95% and 100% ethanol in succession for 1 minute followed by a 5-minute incubation in xylene. Finally, the slides were mounted on glass coverslips and analyzed under the microscope (Olympus BX51) to detect BTZ-induced differences in structure and number of follicles. Follicles were classified as resting follicles; primordial and growing follicles; primary, secondary, antral, and mature follicle; preovulatory. Primordial follicles have single layer of flattened pre-granulosa cells. Primary follicles consist of the primary oocyte surrounded by one expanded layer of cuboidal granulosa cells. Secondary follicles included primary oocyte surrounded by zona pellucida and 2 or more layers of granulosa cells. Antral follicle has the primary oocyte surrounded by zona pellucida and stratified granulosa cells that show the large, fluid-filled antral cavities, whereas preovulatory follicle shows a very large single antrum with fluid-filled cavity, has a primary oocyte surrounded by zona pellucida and granulosa cells that forms cumulus cells 27 (Supplemental Figure 1). All ovarian follicles of every fifth consecutive histological section were counted and categorized according to their developmental stage. We multiplied the number of follicles by 5 to get a value representative for each ovary follicle number. 2 Stained ovary sections were analyzed by two experienced pathologists who were blind to the experimental data.
Immunohistochemistry
Immunohistochemistry (IHC) was performed on Bouin-fixed, paraffin-embedded, 5-µm-thick sections from ovaries using ERα, PR, Cas-3, and Ki67 with streptavidin-biotin complex peroxidase (StrepABC-P) method (UltraVision Large Detection System-Thermo Sci, Waltham, MA). After deparaffinization, antigen retrieval was performed using citrate buffer solution (pH 6.0) for ERα, PR, and Cas-3, slides in the microwave for 2 × 5 minutes at 800 W and for 5 minutes at 600 W, whereas proteinase K was used for Ki67 slides as an antigen retrieval solution, for 20 minutes. Then all slides were incubated in H2O2 for 15 minutes to block endogenous peroxidase activity. Primary antibodies used were anti-ERα (polyclonal, Abcam, 1:100), anti-caspase-3 antibody (polyclonal, Thermo Scientific, 1:100), anti-Ki67 (polyclonal, Thermo Scientific, 1:300), and anti-PR (AB52) (monoclonal, Santa Cruz, 1:50). All primary antibodies were applied for 1 hour at 37°C as described by the manufacturer. Visualization was performed with 3 amino-9-ethylcarbazole (AEC) substrate. Sections were counterstained with Mayer’s hematoxylin. For negative control, phosphate-buffered saline (PBS) was used instead of primary antibodies. Immunoreactivity for each antibody was assessed by counting the 10 different microscopic fields and scored as negative (−) (no positive cells), mild positivity (+) (<10% positive cells), moderate positivity (++) (10%-50% positive cells), and strong positivity (+++) (>50% positive cells) by two blinded pathologists.
Statistical Analysis
Descriptive statistics were presented as mean ± standard error (SE). The normality assumption was checked with Shapiro-Wilk test and the homogeneity of variances assumption was checked with Levene’s test. The cell counts among the treatment groups (C1, BTZ1, C2, and BTZ2) were compared in each developmental stage using Kruskal-Wallis test, since the normality and homogeneity of variances assumptions were not satisfied in any of the developmental stages. In case of any statistically significant difference among the treatment groups, Bonferroni multiple comparison test was conducted to perform all possible pairwise comparisons. SPSS 25 (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.) was used to evaluate the data. A P value of <.05 was assumed to be the statistical significance level.
Result
Histopathological Effects of Bortezomib Treatment on Ovaries
The H&E staining was performed on ovarian sections for histological examination. Serial sections of ovaries were examined under light microscope and follicles of each section were classified and counted based on their developmental stage. We detected a depletion in the population of ovarian follicles at various developmental stages in BTZ1 compared with both control groups (C1 and C2) and BTZ2 compared with C2 groups, respectively (Figure 3A and B) and the decrease in the population of primordial follicles was statistically significant (P < .05) Moreover, the decrease in the population of antral follicles only in BTZ2 compared with C1 was statistically significant (P < .05) (Table 1). Statistical differences between developmental stages of follicles in each treatment groups are given in Table 1 and scatter plots showing the distributions of cell counts at the different developmental stages in each treatment group are given in Figure 2.
Mouse ovarian follicle numbers, mean ± standard error of the mean (mean ± SE) at different developmental stages of mice (6 animals per group) sacrificed, 1 day (C1, BTZ1) and 4 weeks (C2, BTZ2) after the last injection of bortezomib or saline (1 mg/kg) as a vehicle.
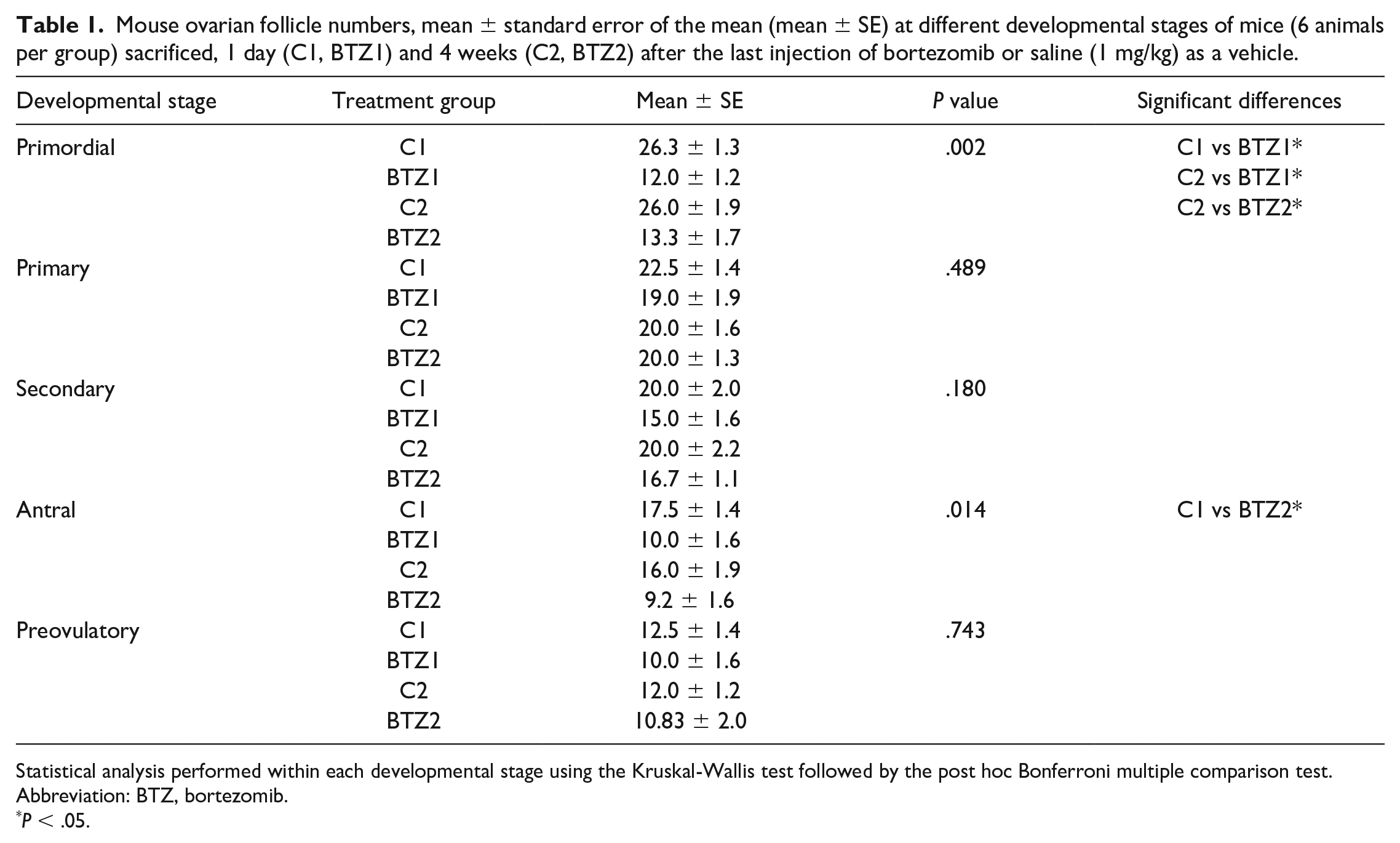
Statistical analysis performed within each developmental stage using the Kruskal-Wallis test followed by the post hoc Bonferroni multiple comparison test.
Abbreviation: BTZ, bortezomib.
P < .05.
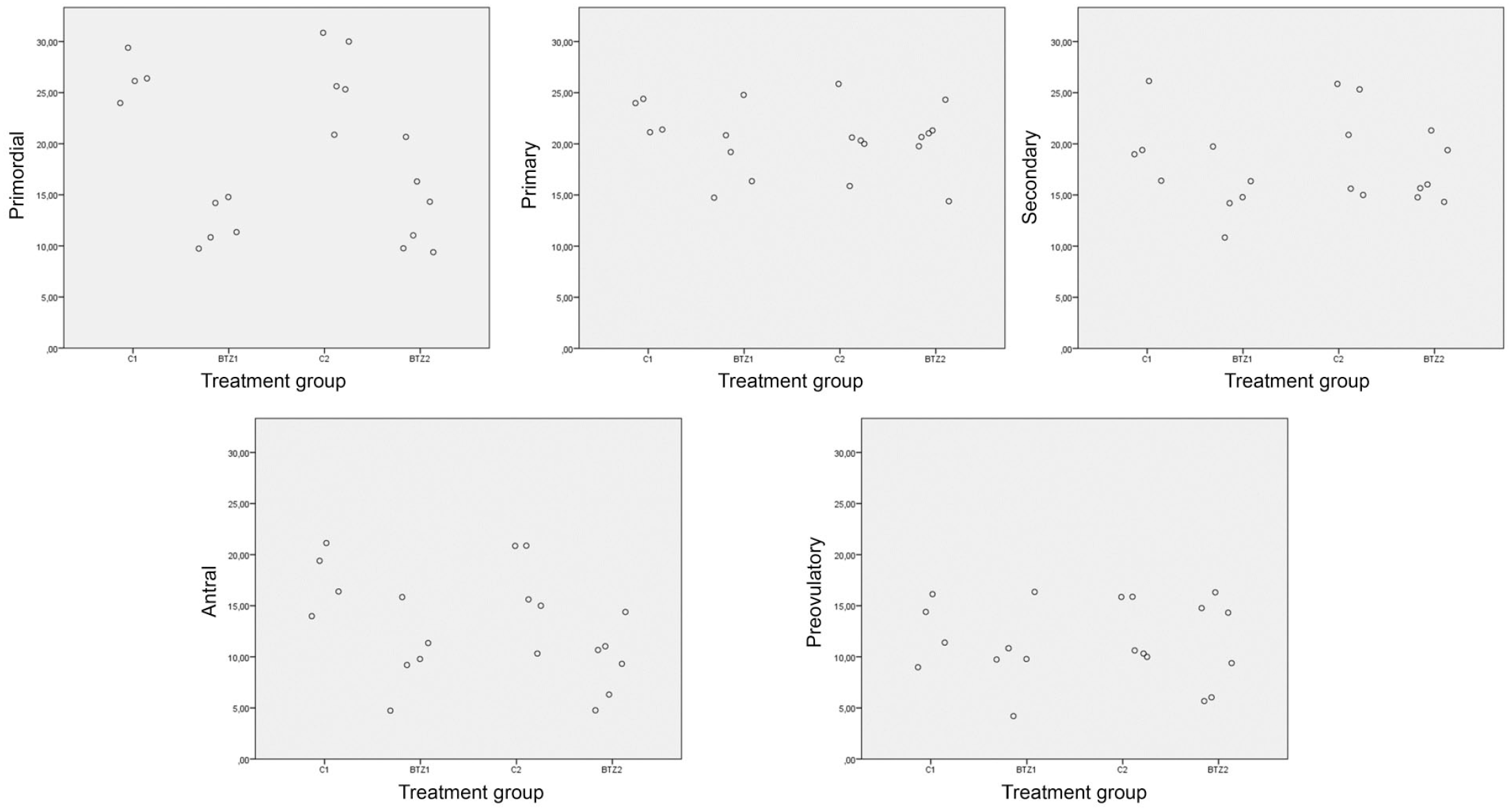
Scatter plots showing the distributions of cell counts at the different developmental stages in each treatment group, 1 day (C1, BTZ1) and 4 weeks (C2, BTZ2) after the 1 mg/kg injection of bortezomib and saline. BTZ indicates bortezomib.
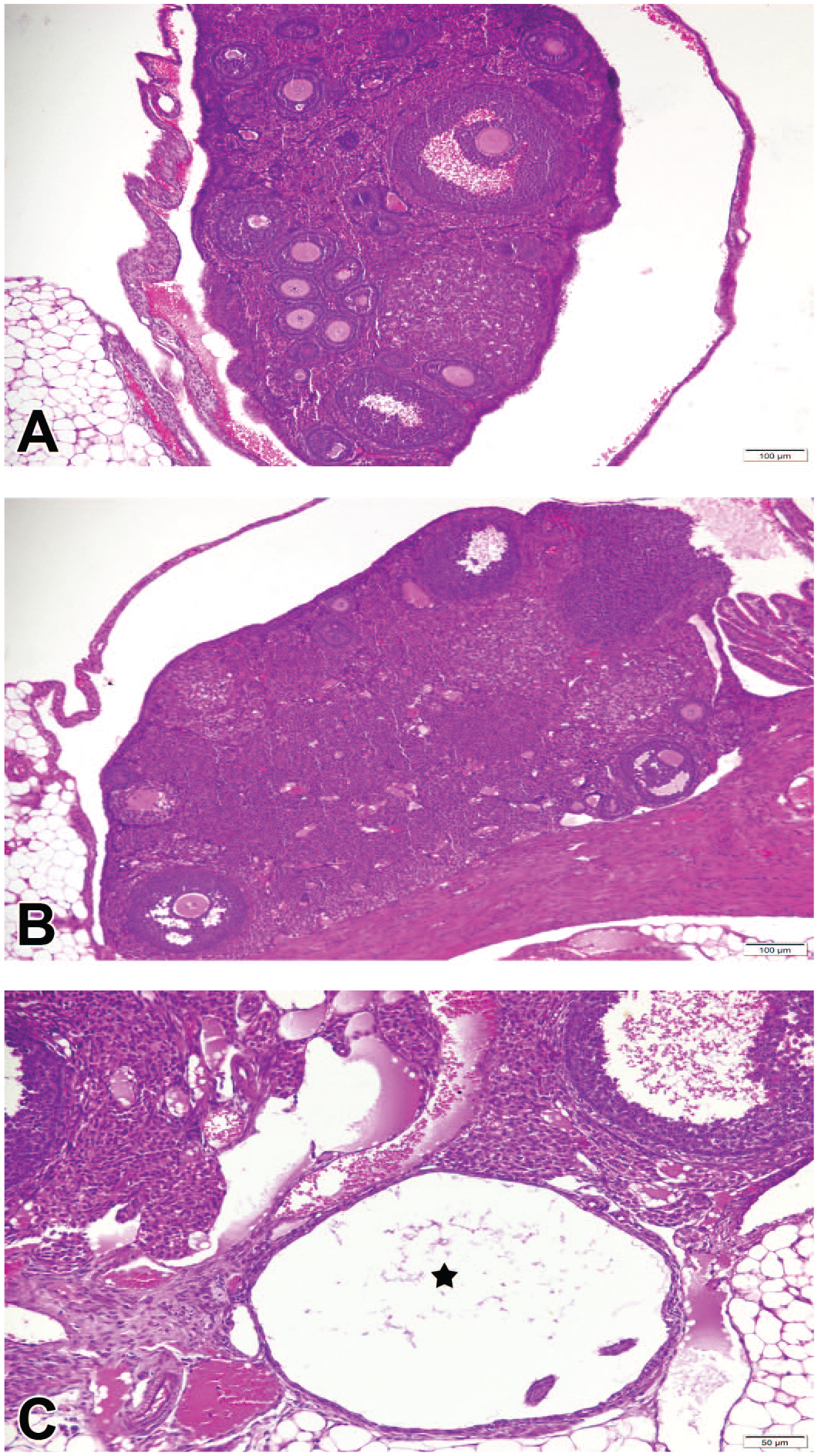
Photomicrographs of ovarian sections, H&E. (A) Control group, sections showing multiple growing follicles in various stages. (B) BTZ2 group, depletion in the population of primordial, primary, secondary, and antral follicles. (C) BTZ2 group, follicular cyst (asterisk). BTZ indicates bortezomib; H&E, hematoxylin and eosin.
Furthermore, follicles were examined in terms of apoptosis. Each follicle containing more than 3 pyknotic granulosa cells was considered as atretic.28,29 BTZ1 and BTZ2 groups showed an increase in the number of atretic developing follicles (secondary and antral follicles) as compared with control groups. Follicles with various degrees of atresia showed apoptosis on follicular granulosa cells. Few ovarian sections from BTZ1 and BTZ2 group also exhibited degenerative granulosa cells in some preovulatory follicles and large follicular cyst appearing as a distended space that lacks an oocyte (Figure 3C).
Immunohistochemical Findings
Immunohistochemical evaluation and semi-quantitative scoring of ovarian sections of the control and treated groups stained with ERα, PR, caspase 3, and Ki67 antibodies is summarized in Table 2. We tested for immunoreactivity differences for ERα, PR, Cas-3, and Ki67 antibodies. No differences were detected between the BTZ1 and BTZ2 groups. Furthermore, no differences were noted when the C1 and C2 control groups were compared with each other. Sections were stained with Cas-3 to evaluate the occurrence of granulosa cell apoptosis leading to follicular atresia. Immunoreactivity of granulosa cells with Cas-3 antibody was mainly cytoplasmic with occasional nuclear staining. Follicles containing more than 3 strongly positive granulosa cells were considered atretic and the number of atretic follicles was counted per each section.28,29 According to this evaluation, we detected that the number of atretic follicles increased in BTZ1 compared with C1 and BTZ2 compared with C2 groups, respectively. The granulosa cells of primary, secondary, and especially antral follicles were positively stained with Cas-3; however, there was no notable positivity in primordial follicles. We observed that Cas-3 staining remarkably increased in the experimental groups versus control groups. When comparing the two experimental groups, a slight increase in the apoptosis scores was noticed in BTZ2 group (Figure 4A and C).
Semi-quantitative immunohistochemical scoring of ERα, PR, Cas-3, and Ki67 antibodies in control (C1, C2) and bortezomib treatment groups (BTZ1, BTZ2).

Abbreviations: BTZ, bortezomib; ERα, estrogen receptor alpha; PR, progesterone receptor; +, mild; ++, moderate; +++, strong.
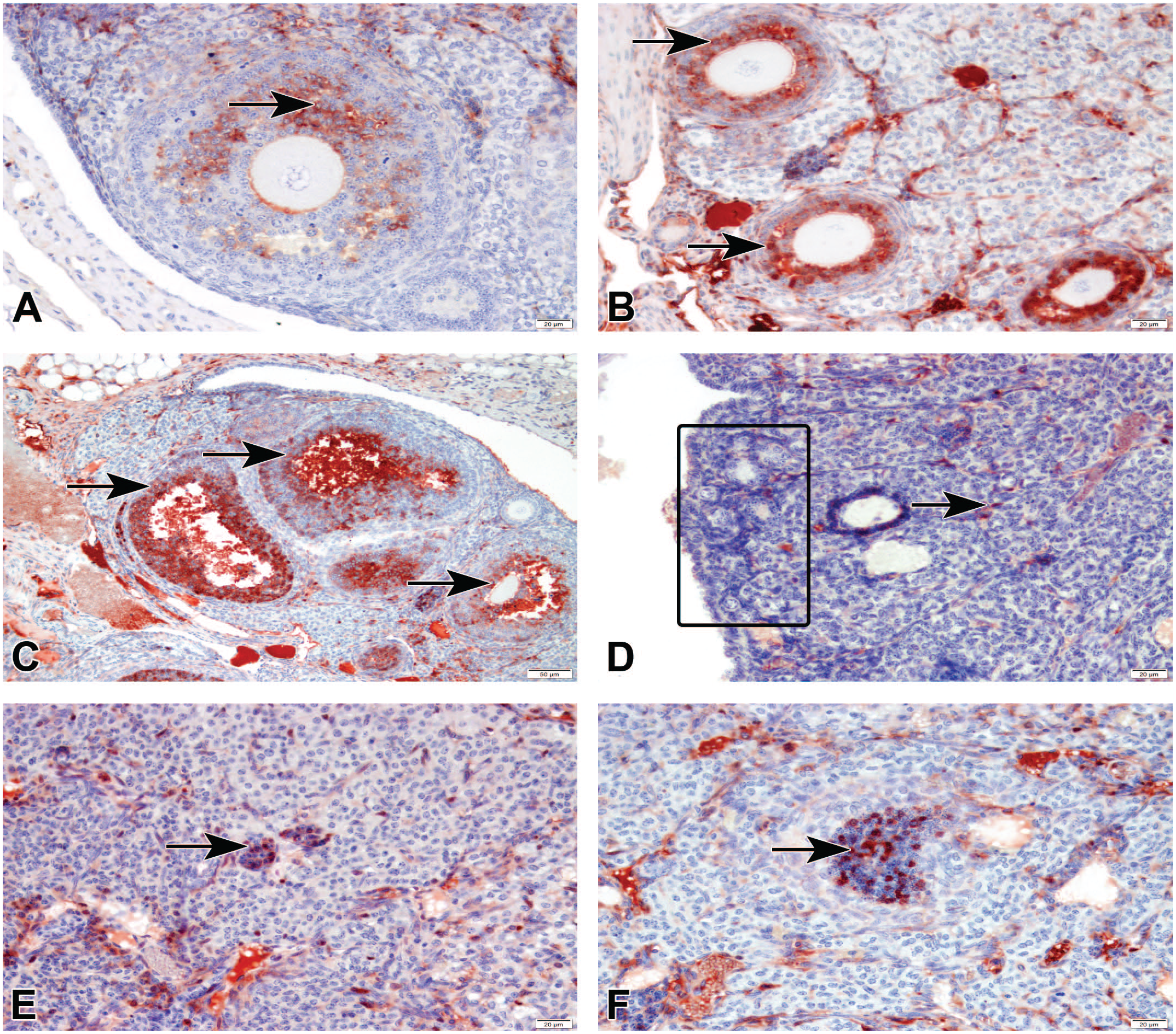
Ovarian immunohistostaining for Cas-3 (A-C) and Ki67 (D-F) in different groups, StreptABC-P. (A) Control group, mild expression of Cas-3 in granulosa cells (arrow). (B) BTZ1 group, moderate expression of Cas-3 in granulosa cells (arrows). (C) BTZ2 group, strong expression of Cas-3 in granulosa cells (arrows) of antral follicles. (D) Control group, no positive staining of Ki67 in primordial follicles (rectangle) and positivity in theca cell (arrow). (E) BTZ1 group and (F) BTZ2 group moderate expression of Ki67 in granulosa cells (arrow). BTZ indicates bortezomib.
A comparative evaluation of Ki67 staining was performed to assess the follicular growth and proliferation of granulosa cells in the control and treatment groups. In control groups, Ki67 was very weakly expressed with one or two nuclear positive granulosa cells in developing follicles (primary and secondary follicles) and a few scattered positive theca cells were also observed. Moreover, no detectable expression of Ki67 was noted in primordial follicles of C1 and C2 groups. In BTZ1 and BTZ2 groups, Ki67-positive cell number was higher in granulosa cells of primer and secondary follicles. Primordial follicles were positively stained differently from the control groups and Ki67 expression was detected in spindle-shaped interstitial cells as well. The expression of Ki67 in primordial, primary, and secondary follicles showed no notable difference between BTZ1 and BTZ2 groups (Figure 4E and F).
To evaluate the impact of bortezomib on hormone levels, we assessed ERα and PR immunoreactivity in follicles. ERα protein revealed a moderate nuclear staining on interstitial and thecal cells in various follicular stages of the ovarium in control groups as expected. Some positive cells were also observed in germinal epithelium. The same cell localization of ERα was detected in bortezomib-treated groups with a generally mild expression of ERα, suggesting that suppression of ERα expression caused notable induction in thecal and stromal cells immunoreactivity compared with control groups (Figure 5A and C).
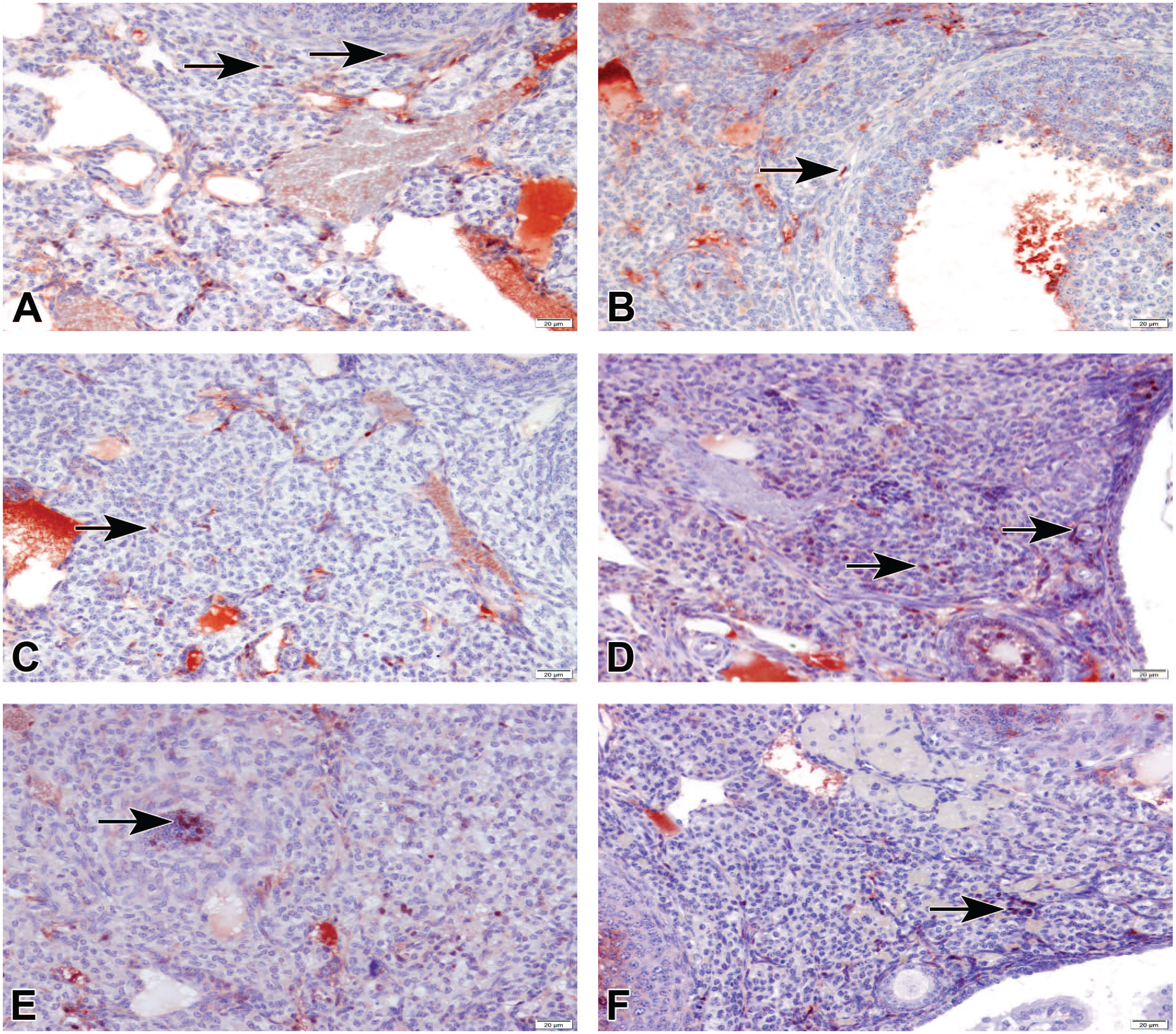
Ovarian immunohistostaining for ERα (A-C) and PR (D-F) in different groups, StreptABC-P. (A) Control group, moderate expression of ERα in theca and interstitial cells (arrows). (B) BTZ1 group and (C) BTZ2 group, mild expression of ERα in theca and interstitial cells (arrow). (D) Control group, moderate expression of PR in granulosa and theca cells (arrows). (E) BTZ1 group, mild expression of PR in granulosa and theca cells (arrows). (F) BTZ2 group, mild expression of PR in granulosa and theca cells (arrows). BTZ indicates bortezomib; ERα, estrogen receptor alpha; PR, progesterone receptor.
Ovarian sections from control and experimental groups were stained with anti-PR antibody, which can detect both PR-A and PR-B immunoreactivity. Nuclear immunolabelling with PR was detected in granulosa, theca, and interstitial cells at primordial, primary, secondary, and preovulatory stages of folliculogenesis. While this staining pattern was moderate in C1 and C2 groups, mild expression was observed in BTZ1 and BTZ2 groups indicating a decrease in its immunoreactivity (Figure 5D and F).
Discussion
In this study we explored the possible effects of commonly used chemotherapeutic agent bortezomib on mouse ovaries. The treated dose was similar to the standard dose that is used in cancer patients. 12 Different chemotherapy agents have various effects on the different cell types of the ovary depending on their mechanisms of action on cancer cells. 30 Therefore, it is important to investigate the long-term effects of cancer drugs on nontarget tissue such as the ovary for MM patients, which is one of the most affected organs by these agents generally. 30
At birth, the ovary has a certain number oocytes contained within the follicles. These primordial follicles are formed prenatally and represent the entire complement of germ cells present for the duration of woman’s reproductive lifespan. Primordial follicle remains in a dormant state for months in mice and for years in humans until they receive signals for activation to enter the growing pool. 27 We preferred to use mice in this study because mouse data can correlate with the human data. It has been known that there are pivotal structural and functional similarities between mouse and human ovaries. In both species, they reserve oocytes in primordial follicle and follicles have the same localization in the ovary cortex. We observed a decrease in the number of primordial follicles after bortezomib treatment. This finding can be associated with the depletion of primordial follicles which accelerated after cancer treatment in female ovaries. We also suggested that this drug may cause early menopause after long-time treatment. Moreover, both humans and mice have the same follicle development stages and growth with a different time schedule. The hormonal and molecular regulation in the ovary is similar for mouse and human, 31 so we also showed the effects of bortezomib on estrogen expression.
Studies showed that2,31 cancer therapy causes a decrease in primordial follicle count and a reduction in ovarian weight and size. Primordial follicles, which constitute the ovarian reserve, are created during the intrauterine life and continuously decrease until menopause. 30 They can be the direct targets of chemotherapeutic agents. 32 The main structural and morphological changes were observed both in cortex and medulla of the ovaries in control and treatment groups. Our histopathological examinations showed that bortezomib treatment results in loss of follicular reserve with a significant reduction in primordial and antral follicle count after three cycles of chemotherapy treatment, suggesting that bortezomib may accelerate ovarian reserve depletion. Our study also showed that cancer therapy induced primordial loss in accordance with the other studies.2,31,33 In our work, we detected a significant decrease in the antral follicle count, which is an important ultrasonographic marker of ovarian reserve. 30 Antral follicle number is related to the remaining primordial follicle number and supports our primordial and antral follicle depletion data.
In our study, to assess the impact of BTZ on apoptosis, we performed IHC staining for Cas-3 on ovaries. Antineoplastic drugs that act as proteasome inhibitors stimulate apoptosis by upregulating the functions of the proapoptotic factors and suppressing the antiapoptotic factors to kill the cancer cells. 3 BTZ induces apoptosis by the intrinsic and extrinsic pathways through upregulation of p53, caspase 8, and Cas-3. 16 Our observation of increased Cas-3 expression in BTZ1 group compared with the C1 group was consistent with these findings. The granulosa cells of primary, secondary, and especially antral follicles were positively stained with Cas-3; however, no expression of Cas-3 protein was found in the primordial follicles in any experimental groups. Our data suggest that bortezomib did not adversely affect primordial follicles by directly inducing apoptosis. Lopez and Luderer 34 showed increased apoptosis in granulosa cells of prenatal and antral follicles, whereas no sign of apoptosis was present on primordial follicles. Kalich-Philosoph et al. 31 explained the same situation with the “follicular burnout theory.” In this theory, chemotherapeutic drugs indirectly lead to depletion of primordial follicles by injuring developing follicles such as secondary and antral follicles. We detected that BTZ specifically targets granulosa cells by inducing granulosa cell apoptosis. Furthermore, to assess the effect of chemotherapy, we used an important proliferation marker Ki67. Studies reported that Ki67 increases in growing follicles after treatment of the chemotherapeutic agents such as cyclophosphamide31,35 and cisplatin. 36 Similarly, in our study, Ki67 was not observed in primordial follicles in the control groups; however, expression of Ki67 was increased in primary and secondary follicles in the BTZ1 and BTZ2 groups. Both increased immunoreactivity of Cas-3 and Ki67 in primary, secondary, and especially antral follicles in BTZ1 compared with C1 and BTZ2 compared with C2 groups may suggest that those follicles are more vulnerable to chemotherapy-induced damage. 33 We also detected slightly increased Cas-3 positivity in BTZ2 group compared with C2 group, suggesting that ovarian follicles cannot recover in 4 weeks and bortezomib may have long-term effects on follicles.
To evaluate whether bortezomib can cause any change in hormone levels, we assessed ERα and PR immunoreactivity in follicles which may affect fertility and physiology of the ovary. ERα is an essential hormone for healthy reproductive development since ERα knockout impairs sexual maturation of the whole female reproductive tract. 19 Immunohistochemical studies showed that ERα is exclusively distributed in the theca and interstitial cells. 37 Theca cells are known as special producer of androgens, which are necessary as substrates for estradiol production in the granulosa cells. 38 A main regulatory pathway leading to ERα expression is the UPS. This pathway controls the stability of ERα protein through the attachment of ubiquitin parts to the receptor that then targets it to the 26S proteasome for degradation. This mechanism results in increased ubiquitination of ERα and its proteolysis. 39 In our study, we detected decreased ERα expression in BTZ1 and BTZ2 groups suggesting that bortezomib may suppress ERα protein expression by inhibition of proteasome through this mechanism. In addition, ERα expression was specifically decreased in thecal and stromal cells in bortezomib-treated groups, suggesting that this inhibition may affect regulation of ovarian function adversely. As reported previously, 40 patients who were administered chemotherapy have lower in vitro estradiol levels related to altered ovarian stromal function regardless of the anticancer drug including alkylating agent and size of the germ cell damage. We detected a decreased PR expression in ovarian follicles of BTZ treatment groups compared with control groups. We also suggest that this decrease in ERα and PR expression may be a result of impaired follicle structures and induced apoptosis. Studies indicated that ERα and PR play essential roles in various physiological processes in the ovary and hormone levels need to be stable to function properly and maintain fertility. 41 Moreover, Lopez and Luderer 34 showed a temporary decrease in estradiol and progesterone levels in granulosa cells consistent with granulosa cell dysfunction two hours after the cyclophosphamide injection.
Bortezomib, a proteasome inhibitor, induces apoptosis in tumor cells and one hypothesis about this action was that these agents can inhibit nuclear factor kappa B (NFкB) signaling by blocking degradation of the IкB (an inhibitor of NFкB), therefore it can prevent nuclear translocation of NFкB. 4 It has been previously shown that ERα has functions in mediating gene transcription through heterodimerizing with other transcription factors like NFкB. 42 We are suggesting that the inhibition in ER expression after bortezomib treatment may correlate with the proteasome inhibitor-NFкB mechanism.
In conclusion, this study indicated that BTZ treatment was associated with depletion of ovarian reserve and impairment of ovarian follicles by inducing granulosa cell apoptosis. Inhibition of ERα and PR expression may affect fertility and regulation of ovarian function adversely. We did not detect any potential recovery after four weeks, revealing that bortezomib may have long-term effects on the ovary. Based on our results, patients should be informed about the potential negative effects of this chemotherapeutic agent and therapeutic agents need to be used to prevent ovarian dysfunction during chemotherapy.
Supplemental Material
sj-tif-1-tpx-10.1177_01926233221083527 – Supplemental material for Bortezomib-Induced Ovarian Toxicity in Mice
Supplemental material, sj-tif-1-tpx-10.1177_01926233221083527 for Bortezomib-Induced Ovarian Toxicity in Mice by Duygu Mutluay, Gözde Yücel Tenekeci and Yanad Abou Monsef in Toxicologic Pathology
Footnotes
Acknowledgements
The authors thank Dr Mustafa Agah Tekindal for helping with statistical analysis and MSc. Cennet Sinem Coban for her help with animal work and tissue collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
