Abstract
Sexually mature nonhuman primates are often used in nonclinical safety testing when evaluating biopharmaceuticals; however, there is limited information in historical control databases or in the published literature on the spontaneous findings in the male reproductive system. This review evaluated digital slides from the male reproductive tract (testes, epididymides, prostate, and seminal vesicles) in sexually mature cynomolgus macaques (Macaca fascicularis; n = 255) from vehicle control groups in nonclinical toxicology studies and compared the observations with body weight, organ weight, and geographical origin. The most common microscopic findings were hypospermatogenesis and tubular dilatation in the testes; inflammatory cell infiltrate, cellular debris, and decreased sperm in the epididymides; inflammatory cell infiltrate and acinar dilatation in the prostate; and corpora amylacea and atrophy in the seminal vesicles. There were a few correlative observations in animals when grouped by weight or geographical origin: animals with lower terminal body weights (<5 kg) often displayed features of late puberty despite having sperm in the epididymis, while animals originating from Mauritius had a lower incidence of inflammatory cell infiltrates than those from Southeast Asia/China. This review provides incidence, descriptions, and photomicrographs of the common spontaneous microscopic findings in the reproductive system of mature male cynomolgus macaques.
Keywords
Introduction
Nonhuman primates (NHPs), most often the cynomolgus macaque, play a key role in the development of biopharmaceuticals as these therapeutics are often antigenic and/or are pharmacologically inactive in rodent and other large animal models.
1
When NHPs are the only species used in nonclinical safety testing, the main assessment for potential effects on the male reproductive system occurs in the context of a repeat dose general toxicity study due to the lack of a traditional rodent-based male fertility study.
2
This nonclinical safety strategy is defined in internationally accepted regulatory guidelines. The International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use: Preclinical Safety Evaluation of Biotechnology-derived Pharmaceuticals S6(R1) specifies It is recognized that mating studies are not practical for NHPs. However, when the NHP is the only relevant species, the potential for effects on male and female fertility can be assessed by evaluation of the reproductive tract (organ weights and histopathological evaluation) in repeat dose toxicity studies of at least 3 months duration using sexually mature NHPs.
Semen collection, testicular volume, and/or hormonal assessment may be added when there is specific cause for concern, but the anatomic pathologist’s light microscopic evaluation is a critical component of the assessment.
Adequate evaluation of the sexually mature reproductive tract by light microscopy requires the study and peer-reviewing pathologists to have a thorough understanding of the normal appearance and common spontaneous findings in the mature male reproductive system; however, there is limited published information available on these findings, particularly regarding incidence or frequency of occurrence.3,4 This paucity of information can be attributed to the relatively late onset of sexual maturation in the NHP, which increases the cost to source and house these animals. Hence, most of the nonclinical studies in NHPs are conducted using sexually immature animals or occasionally a mix of immature, pubertal, and/or mature animals. As a result, historical control data and published literature generally include younger NHPs. When spontaneous findings occur in animals administered a test article, these findings can be misconstrued as test article-related due to the lack of available information and the relatively small group sizes used in NHP studies. The objectives of this study were (1) to provide a comprehensive review of control animal tissues by a single observer using consistent terminology, thresholds, and grading to establish the overall incidence of microscopic findings in the male reproductive system of sexually mature cynomolgus macaques; and (2) to evaluate the potential impact/correlation of body weight, organ weight (OW), geographical origin, and other existing study data. These data can be used to better understand and evaluate the safety of therapeutic agents for which the cynomolgus macaque is the species selected for toxicologic evaluation.
Materials and Methods
Study and Animal Selection
Cynomolgus macaques (Macaca fascicularis; n = 255) from vehicle control groups from 88 nonclinical toxicology studies from 2001 to 2020 were included in this retrospective assessment. Only animals documented as sexually mature in the 88 nonclinical studies were included in this detailed review. Sexual maturity was defined in the 88 studies primarily by the microscopic presence of sperm in the tail of the epididymis at the end of the study. A subset of animals also had semen collection and/or testicular volume measurements obtained prior to study start. Animals that were not classified as sexually mature in the original study reports were excluded from our retrospective analysis. Studies were conducted in North America and Europe at Charles River or Covance Laboratories (now LabCorp) and ranged in duration from 7 days to 9 months. Animals originated from various breeders in Vietnam, Cambodia, Indonesia, or China (designated Southeast Asia [SE]/China) (n = 205) or Mauritius (n = 50). The studies had a mix of administration routes including oral gavage, oral capsules, intravenous injection, intravenous infusion, and/or subcutaneous injection. All studies were conducted in accordance with institutional and governmental guiding principles in the use of animals and all study procedures were approved by the Testing Facility’s Institutional Animal Care and Use Committees.
Digital Scanning, Slide Evaluation, Terminology, and Recording of Microscopic Findings
For each animal, standard tissue sections stained with hematoxylin and eosin of the male reproductive tissues (testes, epididymides, prostate, and seminal vesicles) were digitally scanned at 20× or 40× using Aperio (Leica Biosystems, Inc., Buffalo Grove, IL) or Hamamatsu (Hamamatsu Corporation, Bridgewater, NJ) slide scanners. Digital slide scans were reviewed by a single board-certified veterinary pathologist with experience in evaluation of the male reproductive system (JDV). Diagnostic terminology was established prior to evaluation based on available International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) 5 terminology for NHPs and severity graded (0 = no abnormalities detected, 1 = minimal, 2 = mild, 3 = moderate, 4 = marked, or 5 = severe). Embryonic remnants and ectopic tissues were not graded and were recorded as “present,” with a corresponding numerical grade of “6,” when identified. The information was recorded into a spreadsheet (Microsoft Excel, Microsoft, Redmond, WA). Microscopic data recorded in the original reports were not extracted or evaluated because evaluation of these data was outside the scope of our analysis.
Retrieval and Collation of Nonmicroscopic Study Data
Terminal body weight (TBW) and OW were retrieved from the original study reports. Occasional animals lacked individual endpoints. Total numbers available for review for each parameter are included in Tables 1 to 5. Additional information was extracted from study reports when available (age, semen parameters, testicular volume, hormonal measurements, supplier/vendor information, etc.); however, these data were outside of the scope of our evaluation, were not collected consistently across studies, and were not included as part of the overall assessment.
Terminal body weight (TBW) and organ weights (absolute, relative to TBW [%TBW], and relative to brain weight [%BrW]) for testes, epididymides, prostate, and seminal vesicles presented as means with standard deviation (parenthesis) and range [brackets].
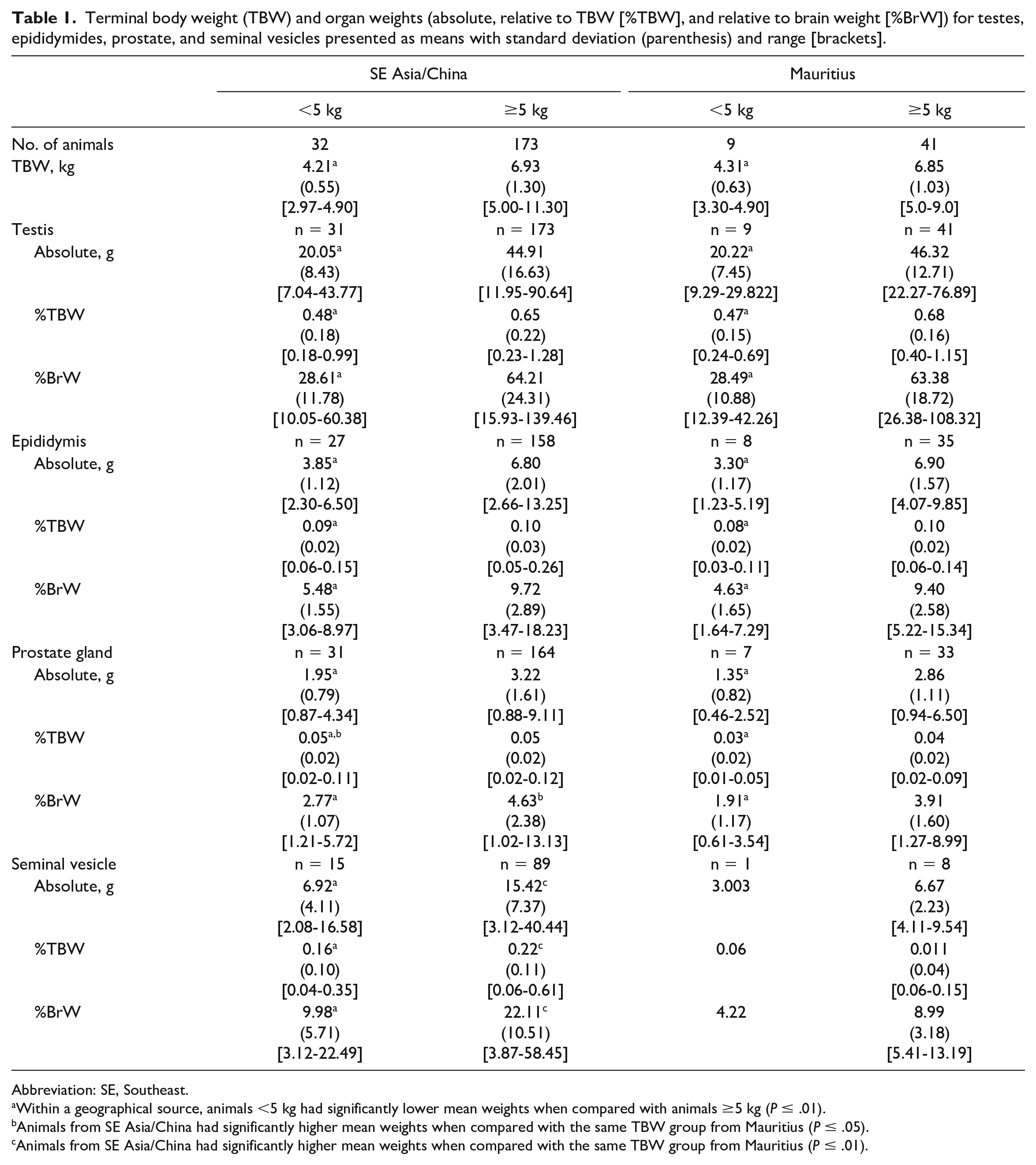
Abbreviation: SE, Southeast.
Within a geographical source, animals <5 kg had significantly lower mean weights when compared with animals ≥5 kg (P ≤ .01).
Animals from SE Asia/China had significantly higher mean weights when compared with the same TBW group from Mauritius (P ≤ .05).
Animals from SE Asia/China had significantly higher mean weights when compared with the same TBW group from Mauritius (P ≤ .01).
Incidence of microscopic findings in the testes separated by geographical source (SE Asia/China or Mauritius) and terminal body weight (<5 kg, ≥5 kg, or combined).
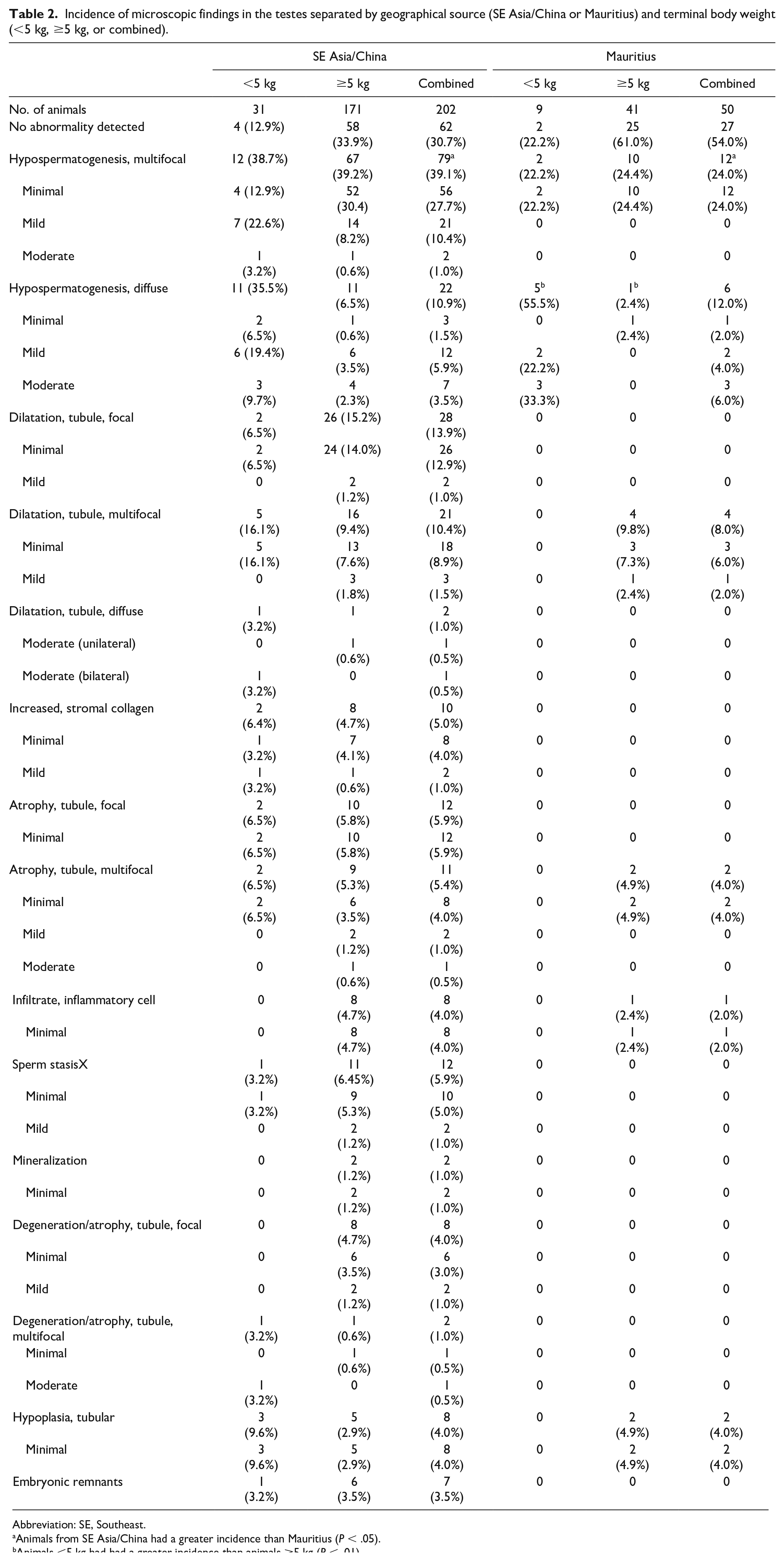
Abbreviation: SE, Southeast.
Animals from SE Asia/China had a greater incidence than Mauritius (P < .05).
Animals <5 kg had had a greater incidence than animals ≥5 kg (P < .01).
Incidence of microscopic findings in the epididymides separated by geographical source (SE Asia/China or Mauritius) and terminal body weight (<5 kg, ≥5 kg, or combined).
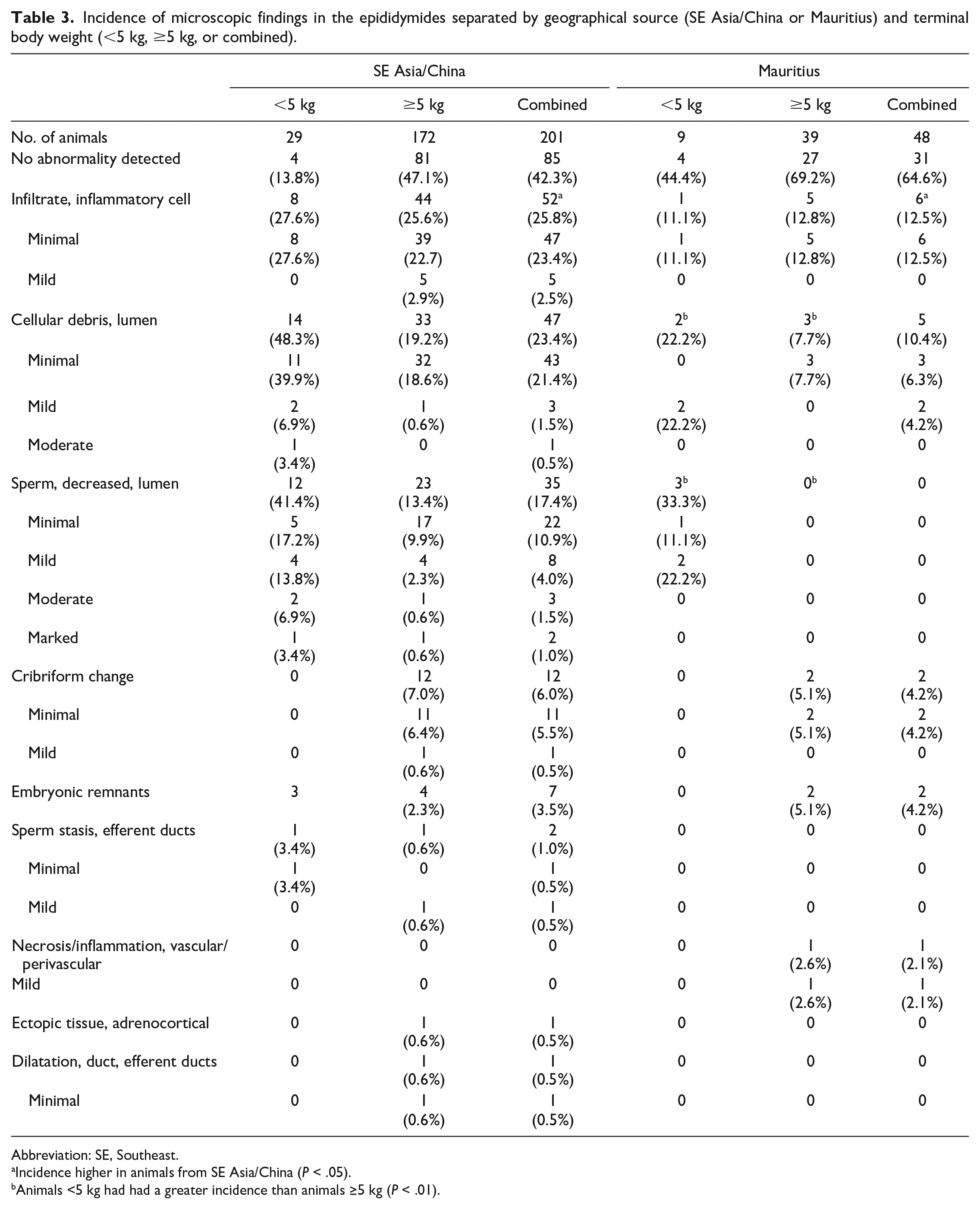
Abbreviation: SE, Southeast.
Incidence higher in animals from SE Asia/China (P < .05).
Animals <5 kg had had a greater incidence than animals ≥5 kg (P < .01).
Incidence of microscopic findings in the prostate separated by geographical source (SE Asia/China or Mauritius) and terminal body weight (<5 kg, ≥5 kg, or combined).
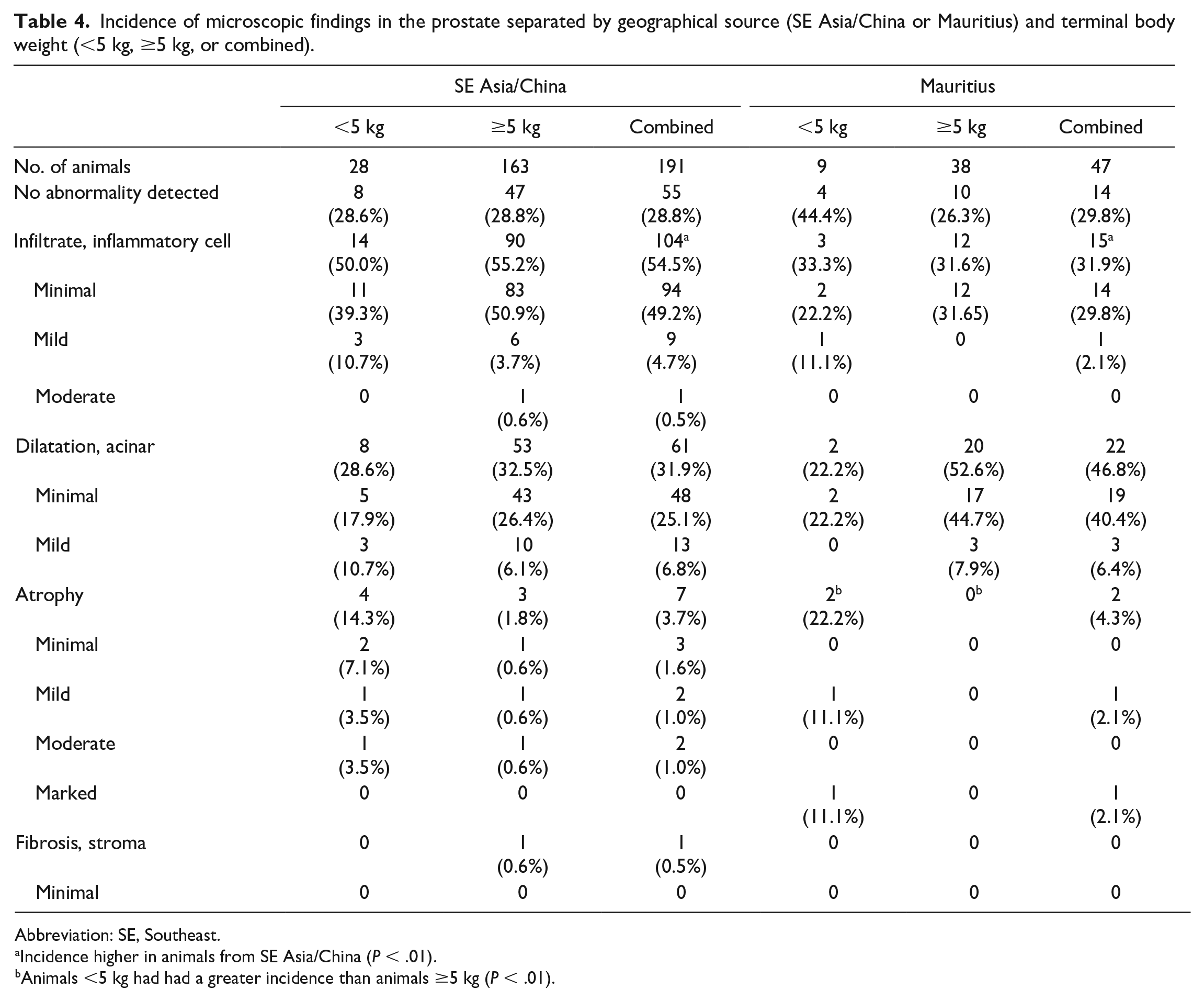
Abbreviation: SE, Southeast.
Incidence higher in animals from SE Asia/China (P < .01).
Animals <5 kg had had a greater incidence than animals ≥5 kg (P < .01).
Incidence of microscopic findings in the seminal vesicles separated by geographical source (SE Asia/China or Mauritius) and terminal body weight (<5 kg, ≥5 kg, or combined).
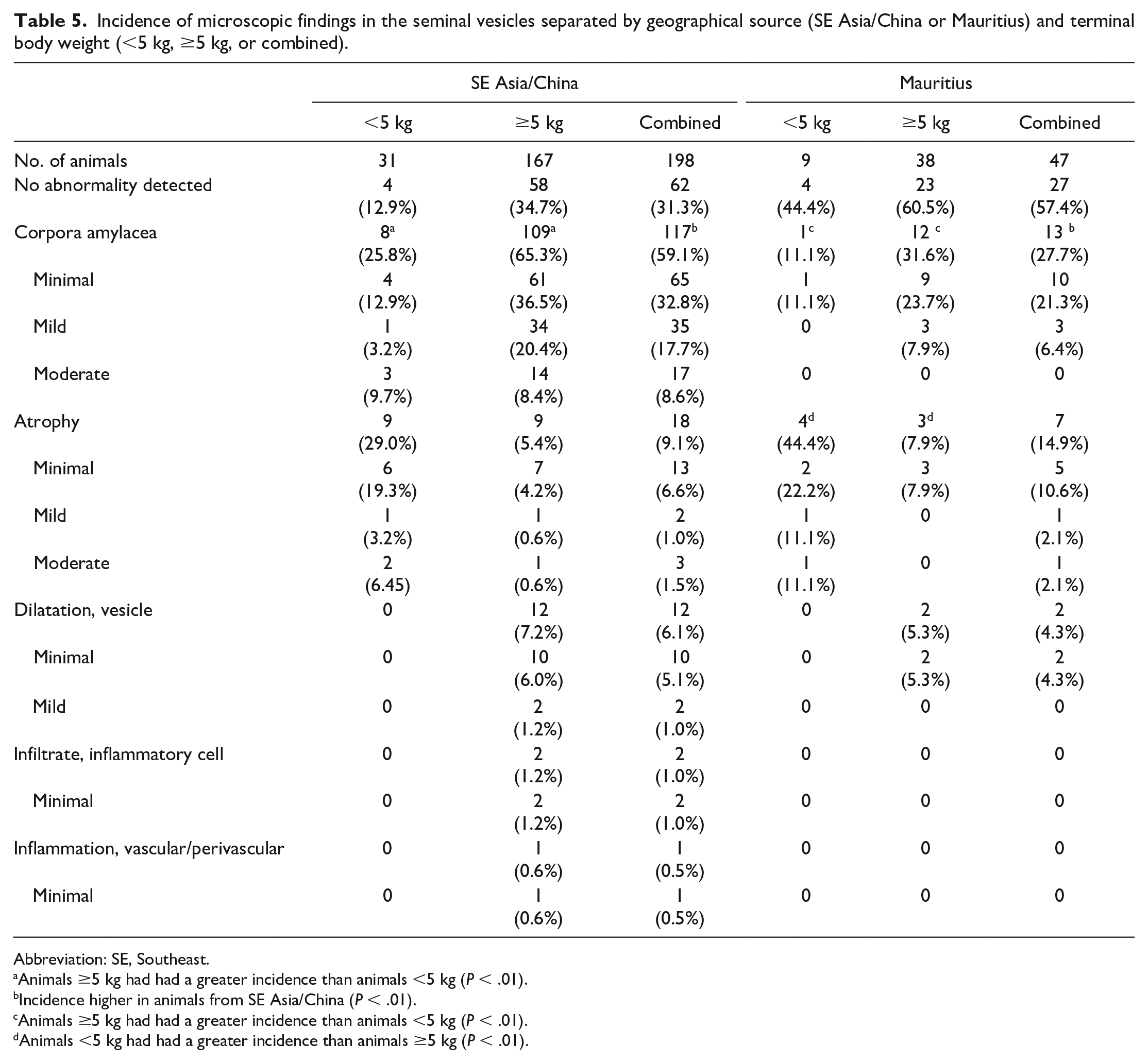
Abbreviation: SE, Southeast.
Animals ≥5 kg had had a greater incidence than animals <5 kg (P < .01).
Incidence higher in animals from SE Asia/China (P < .01).
Animals ≥5 kg had had a greater incidence than animals <5 kg (P < .01).
Animals <5 kg had had a greater incidence than animals ≥5 kg (P < .01).
Data Analysis
Terminal body weight and absolute and relative (to TBW and brain) weights of testes, epididymides, prostate, and seminal vesicles were analyzed. Animals were grouped by geographic source (SE/China or Mauritius) and by TBW (<5 kg or ≥5 kg). The results were analyzed using SAS 9.4 (TS1M6). Descriptive statistics were analyzed, and groups were excluded if standard deviation = 0 or N < 3. To check for normality and homogeneity of variance on the data set, Shapiro-Wilk and Levene tests were conducted. No variable met the assumptions of normality and homogeneity of variance; therefore, PROC GLIMMIX with a Gaussian distribution was conducted to account for nonconstant variability and non-normal data. 6 A generalized linear model was fit among each variable and the least squares means and simple effect comparisons were analyzed. Results describe means and 95% confidence limits for least squares means and simple effect comparisons. The linear model analyzed the differences between Mauritius animals <5 kg or ≥5 kg, differences between SE Asia/China animals <5 kg or ≥5 kg, differences between Mauritius animals and SE Asia/China animals regardless of weight, and differences between animals <5 kg or ≥5 regardless of source.
For the analysis of microscopic findings, animals were grouped by geographic source (SE Asia/China or Mauritius) and by TBW (<5 kg or ≥5 kg). Severity was coded as Minimal = 1, Mild = 2, Moderate = 3, Marked = 4, Severe = 5, Present = 6, and No Abnormalities Detected = 0. When captured, data were split between focal/multifocal vs. diffuse for individual findings except if the tissues had no abnormalities detected (i.e. severity score of “0”). The results from severity scoring were analyzed using SAS 9.4 (TS1M6). A frequency analysis was used to evaluate the Row Mean Differences of each diagnosis/modifier/severity by NHP Source, Weight, NHP Source*Weight, and Weight*NHP Source. 7 If columns or rows summed 0, statistics were not computed for that variable and the row mean difference score was the summary accumulation. If row mean difference were significant, the mean scores of the R rows were not equal.
Results
Terminal Body Weight and Organ Weights
A summary of TBW and OW (absolute, relative to TBW [%TBW], and relative to brain weight [%BrW]) for testes, epididymides, prostate, and seminal vesicles is presented in Table 1. Additional graphical representation with 95% prediction ellipses of each organ is presented in Figure 1. Animals from SE Asia/China or Mauritius with a TBW <5 kg had significantly lower mean TBW and absolute and relative OW and for testes, epididymides, prostate, and seminal vesicles when compared with animals ≥5 kg. There were no statistically significant differences in TBW when comparing geographical source. Animals from SE Asia/China had larger absolute and relative prostate and seminal vesicle weights when compared with animals from Mauritius (Figure 1). Statistical analysis for seminal vesicles for animals <5 kg was not performed due to the small group size (n = 1) for animals from Mauritius.
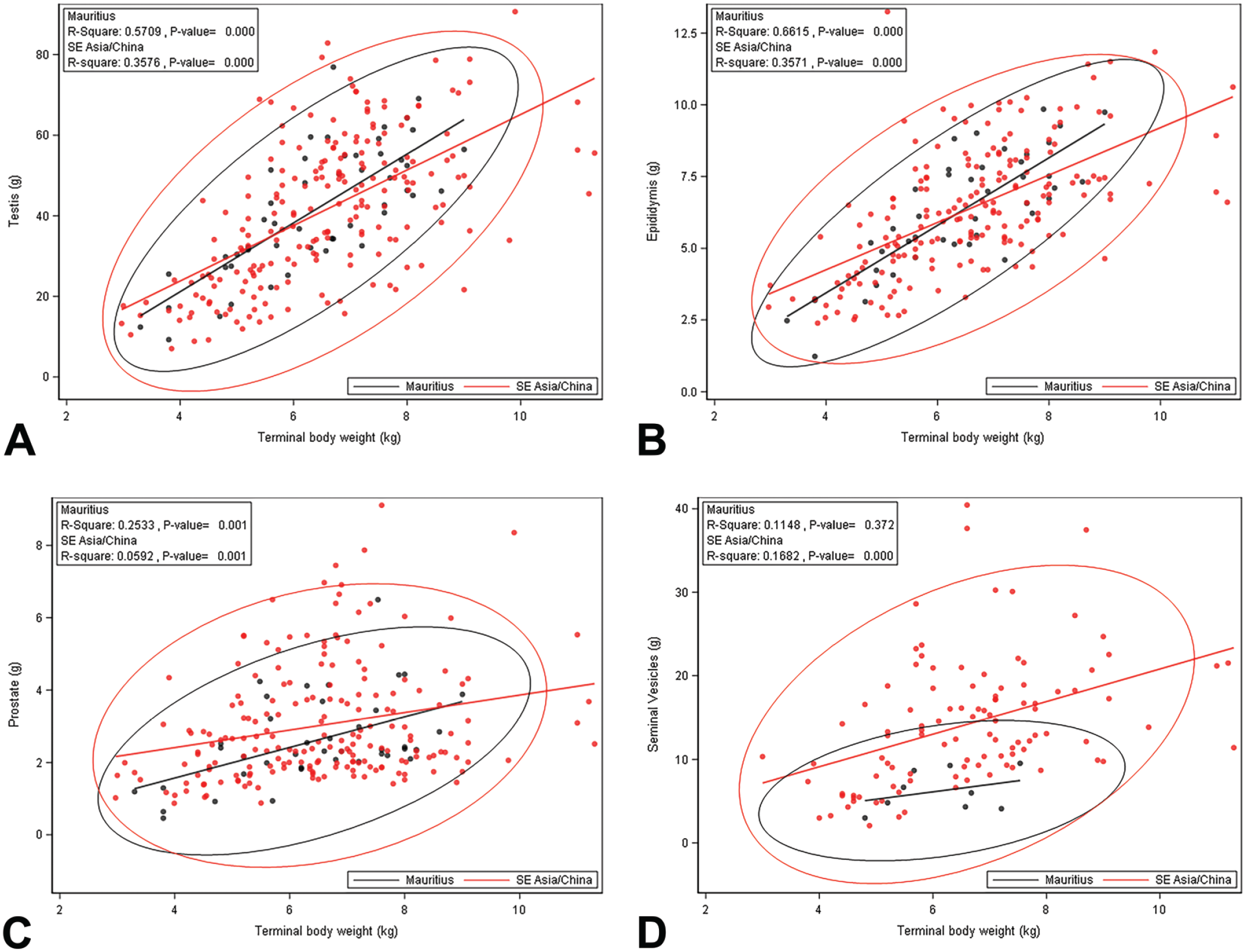
Regression of terminal body weight and absolute testis weight (A), epididymis weight (B), prostate weight (C), and seminal vesicles (D) with 95% prediction ellipse for animals from SE Asia/China and Mauritius. Note the considerable variability present across the weight range. SE indicates Southeast.
Microscopic Findings
The incidence of microscopic findings for the testes, epididymides, prostate, and seminal vesicles is presented in Tables 2 to 5.
Testes
The most common microscopic findings in the testes were hypospermatogenesis and dilatation, tubule (Table 2). Hypospermatogenesis, characterized by a partial or complete absence of one or more generations of germ cells (spermatocytes, round spermatids, and/or elongated spermatids), was present as either a multifocal or diffuse finding. Multifocal hypospermatogenesis was primarily minimal or mild and affected randomly distributed tubules and germ cell types (Figure 2A) and was present at a generally similar incidence regardless of TBW, although there was a trend toward increased severity in animals <5 kg from SE Asia/China. There was a significant difference between geographic groups of animals with multifocal hypospermatogenesis, where animals from SE Asia/China had a greater incidence of multifocal hypospermatogenesis than Mauritius (Table 2). Diffuse hypospermatogenesis was minimal to moderate and affected tubules and multiple germ cell types throughout both testes (Figure 2B). There was a significant difference (P < .0001) between TBW groups with diffuse hypospermatogenesis (all animals combined regardless of source), where animals <5 kg had a greater incidence of diffuse hypospermatogenesis than animals ≥5 kg. When evaluated within geographical source, there was a significant difference in animals from Mauritius where animals <5 kg had a greater incidence of diffuse hypospermatogenesis than animals ≥5 kg (Table 2). A similar difference was present in animals from SE Asia/China, but the difference was not statistically significant.
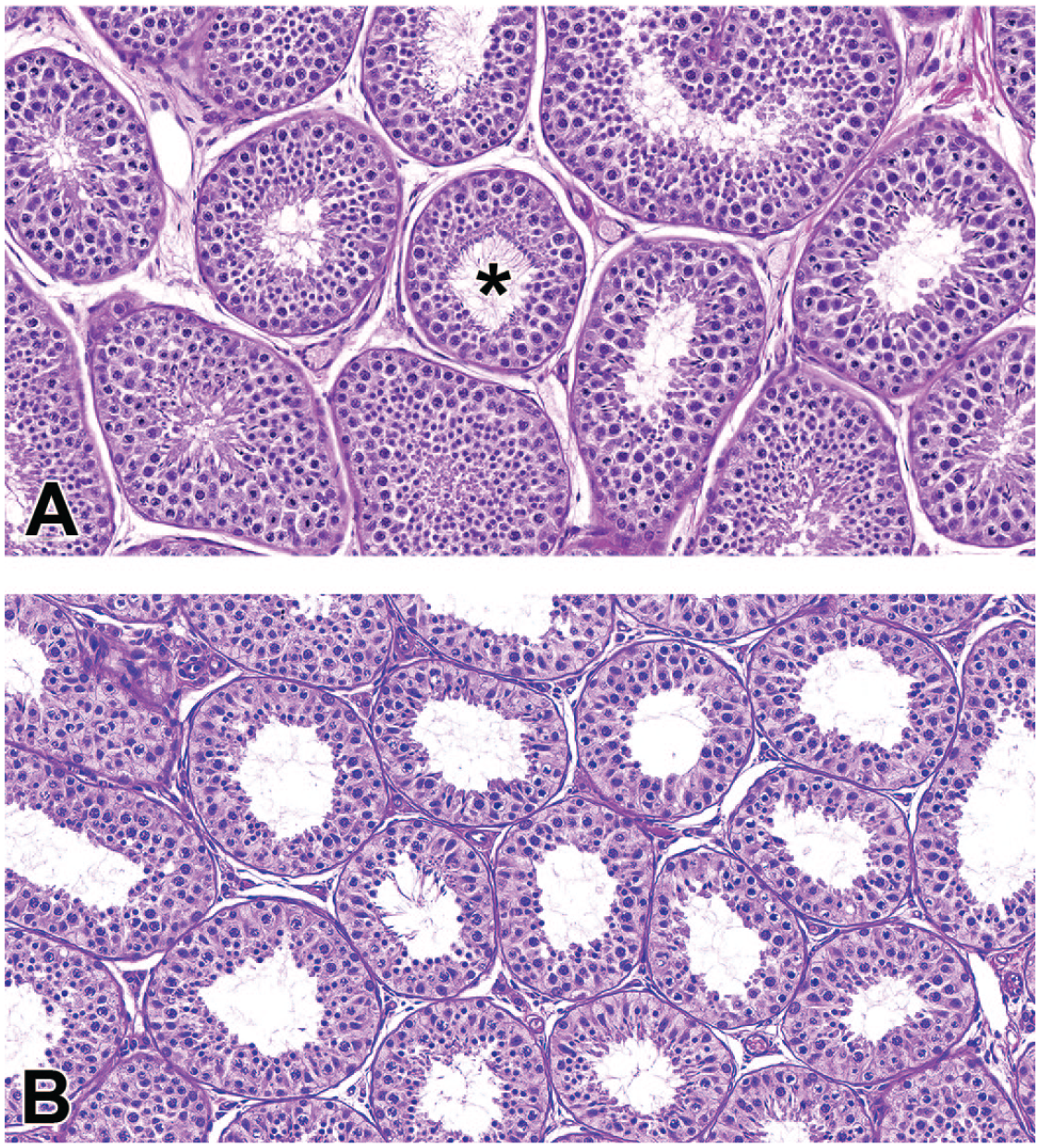
Photomicrographs (H&E) of testis demonstrating focal (A) and diffuse (B) hypospermatogenesis. Focal and multifocal hypospermatogenesis was commonly observed across all ages and sources of animals and typically consisted of scattered tubules lacking random germ cell types (*a superficial layer of mature spermatids is present, but the underlying layer of round spermatids is incomplete). In contrast, diffuse hypospermatogenesis was typically present in animals <5 kg and likely represents incomplete spermatogenesis as a feature of later pubertal development. Original magnification 40X (A and B).
Dilatation of seminiferous tubules was present as a focal, multifocal, or diffuse finding (Table 2). Focal and multifocal dilatation of seminiferous tubules was minimal or mild and despite the distribution difference had a similar microscopic appearance. Focal/multifocal dilatation consisted of segmental area(s) with increased luminal diameter of the seminiferous tubules (Figure 3A). There was typically thinning of the seminiferous epithelium, degeneration, sloughed germ cells, and/or sperm stasis (Figure 3B). Adjacent seminiferous tubules were normal. Diffuse dilatation of seminiferous tubules was observed rarely and consisted of a unilateral or bilateral increase in luminal diameter of seminiferous tubules across most of the tubules in the section (Figure 3C). Dilatation of seminiferous tubules was often present in animals with increased stromal collagen (Figure 4A) and was occasionally associated with an inflammatory cell infiltrate surrounding the dilated tubules (Figure 4B). Tubular dilatation lacked a clear association with TBW or geographic source.
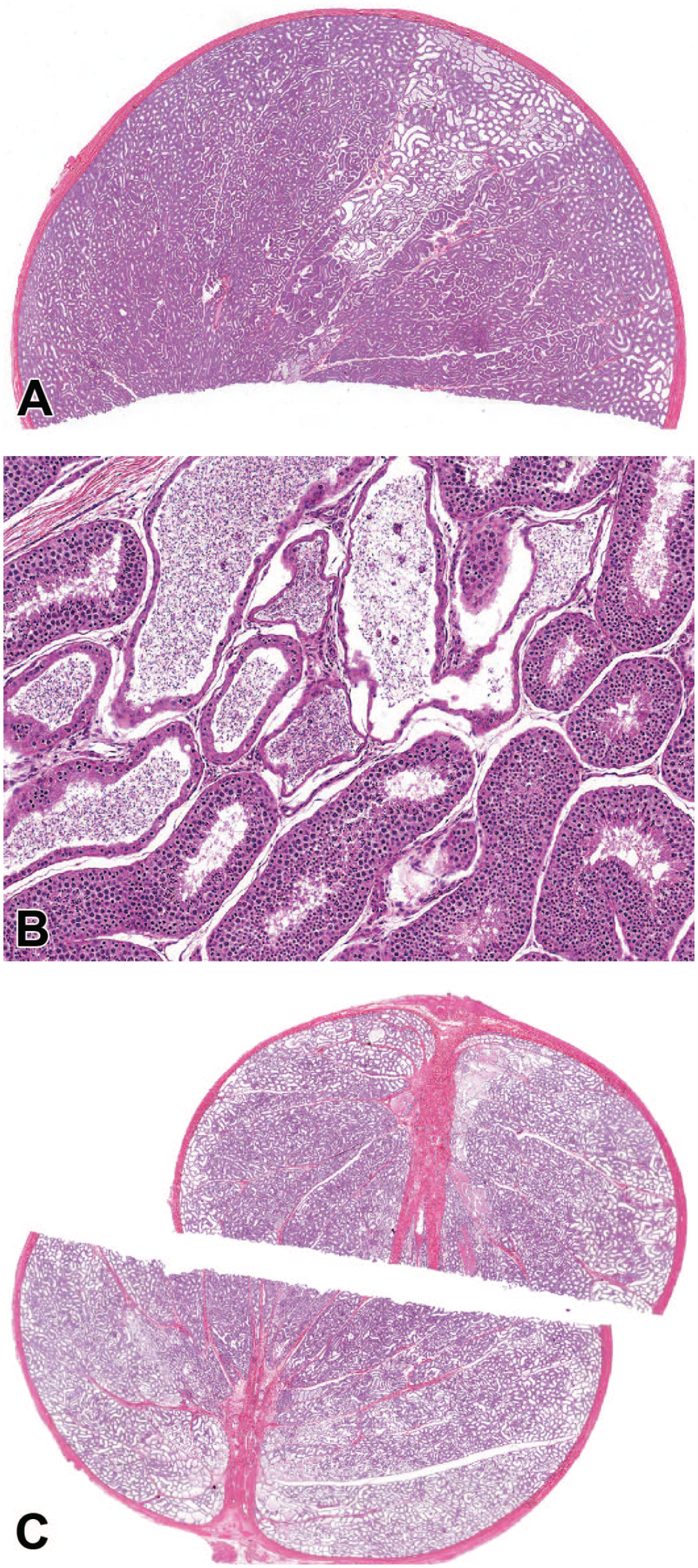
Photomicrographs (H&E) of testis demonstrating focal (A and B) and diffuse (C) tubular dilatation. Focal dilatation was common and most often present in a segmental pattern with varying degrees of degeneration, atrophy, and/or sperm stasis with adjacent seminiferous tubules displaying normal spermatogenesis. Diffuse tubular dilatation was rare but had a striking appearance that could be confused with a test article-related effect. Original magnification 0.5X (A), 8.0X (B), and 0.4X (C).
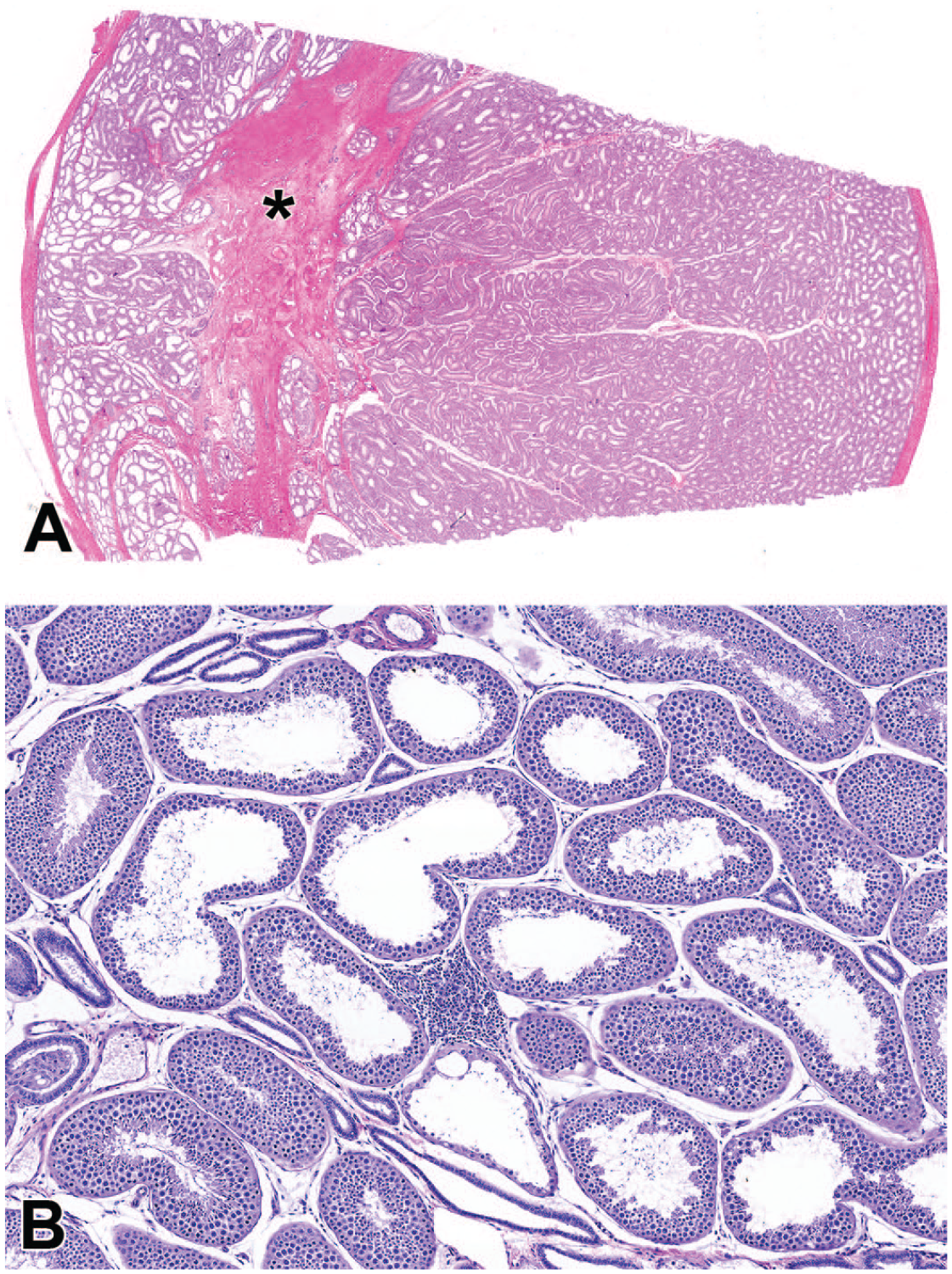
Photomicrographs (H&E) of testis demonstrating a focal increase in stromal collagen in the region of the rete testis (*) with focal tubular dilatation (A) and focal tubular dilatation with an associated focal mononuclear cell infiltrate (B). Original magnification 0.5X (A) and 15X (B).
Additional findings, including tubular hypoplasia and embryonic remnants, were present sporadically without an association with TBW or geographic source (Table 2 and Figures 5 and 6).
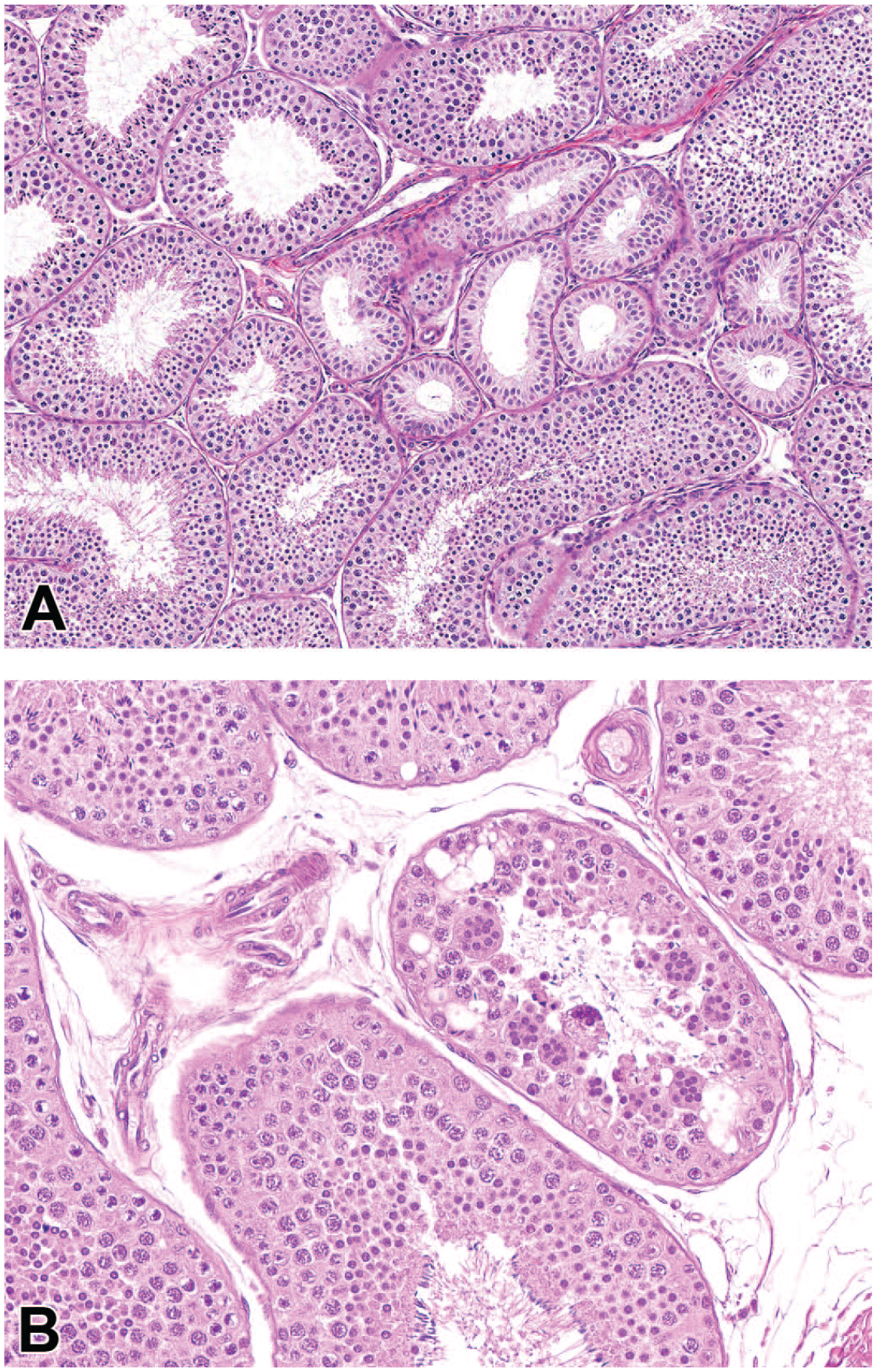
Photomicrographs (H&E) of testis demonstrating focal tubular hypoplasia (A) and focal tubular degeneration with multinucleated cells. Original magnification 20X (A and B).
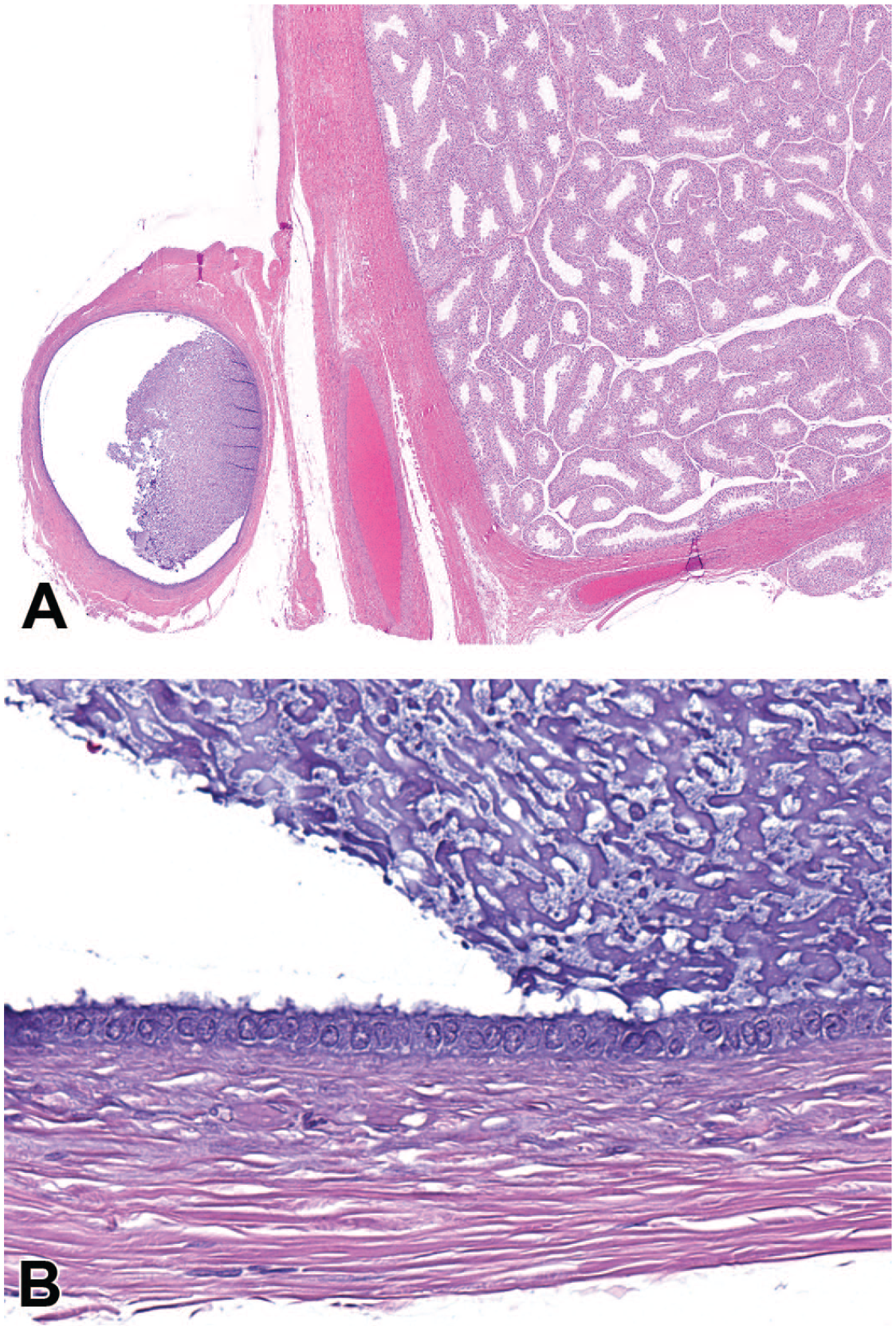
Photomicrographs (H&E) of testis with an attached embryonic remnant (consistent with appendix epididymis) consisting of a single cystic structure (A) with acellular basophilic material lined by a single layer of variably ciliated epithelial cells (B). Original magnification 4.0X (A) and 40X (B).
Epididymis
The most common microscopic findings in the epididymis were inflammatory cell infiltrate, luminal cellular debris, and decreased luminal sperm (Table 3). Minimal or mild inflammatory cell infiltrates were present within the epididymal interstitium as small focal to multifocal, often perivascular aggregates of predominately lymphocytes (Figure 7A). The incidence and severity of inflammatory cell infiltrates was higher in animals from SE Asia/China compared with animals from Mauritius (Table 3). The incidence of inflammatory cell infiltrates did not differ based on TBW.
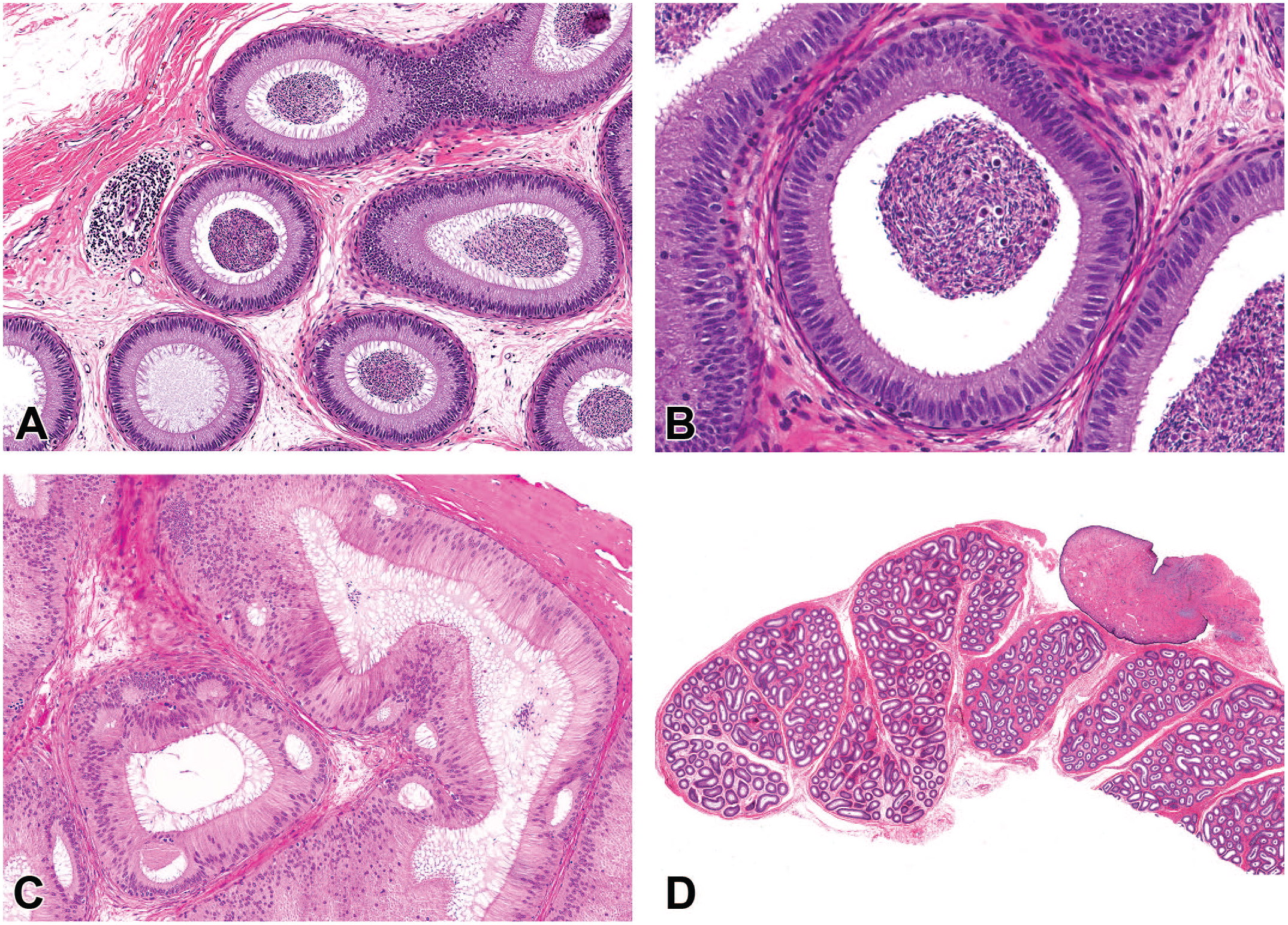
Photomicrographs (H&E) of epididymis demonstrating (A) focal area of perivascular mononuclear cell infiltrate, (B) cellular debris within the lumen of the epididymis, (C) cribriform change within the head of the epididymis consisting of multiple intraepithelial lumina, and (D) embryonic remnant consisting of a pedunculated stalk of fibrous connective tissue with an overlying surface epithelium (consistent with appendix testis). Original magnification 20X (A and B), 10X (C), and 0.8X (D).
Minimal to moderate cellular debris was characterized by the presence of degenerate germ cells intermixed with sperm within the lumen of the epididymis (Figure 7B). There was a significant difference (P < .001) between TBW groups with cellular debris (all animals combined regardless of source), where animals <5 kg had a greater incidence of cellular debris than animals ≥5 kg. When evaluated within geographical source, there was a significant difference in animals from Mauritius where animals <5 kg had a greater incidence of cellular debris than animals ≥5 kg (Table 3). A similar difference was present in animals from SE Asia/China, but the difference was not statistically significant. In addition, animals from SE Asia/China had an overall higher incidence of cellular debris than animals from Mauritius, but this difference was not statistically significant.
Minimal to marked decreased luminal sperm was characterized by a diffuse reduction in the number of sperm, often associated with a decreased luminal diameter of the epididymal duct. There was a significant difference (P < .001) between TBW groups with decreased luminal sperm (all animals combined regardless of source), where animals <5 kg had a greater incidence of decreased luminal sperm than animals ≥5 kg. When evaluated within geographical source, there was a significant difference in animals from Mauritius where animals <5 kg had a greater incidence of decreased luminal sperm than animals ≥5 kg (Table 3). A similar difference was present in animals from SE Asia/China, but the difference was not statistically significant.
Additional findings, including cribriform change and embryonic remnants, were present sporadically without an association with TBW or geographic source and are detailed in Table 3 and Figures 7 and 8.
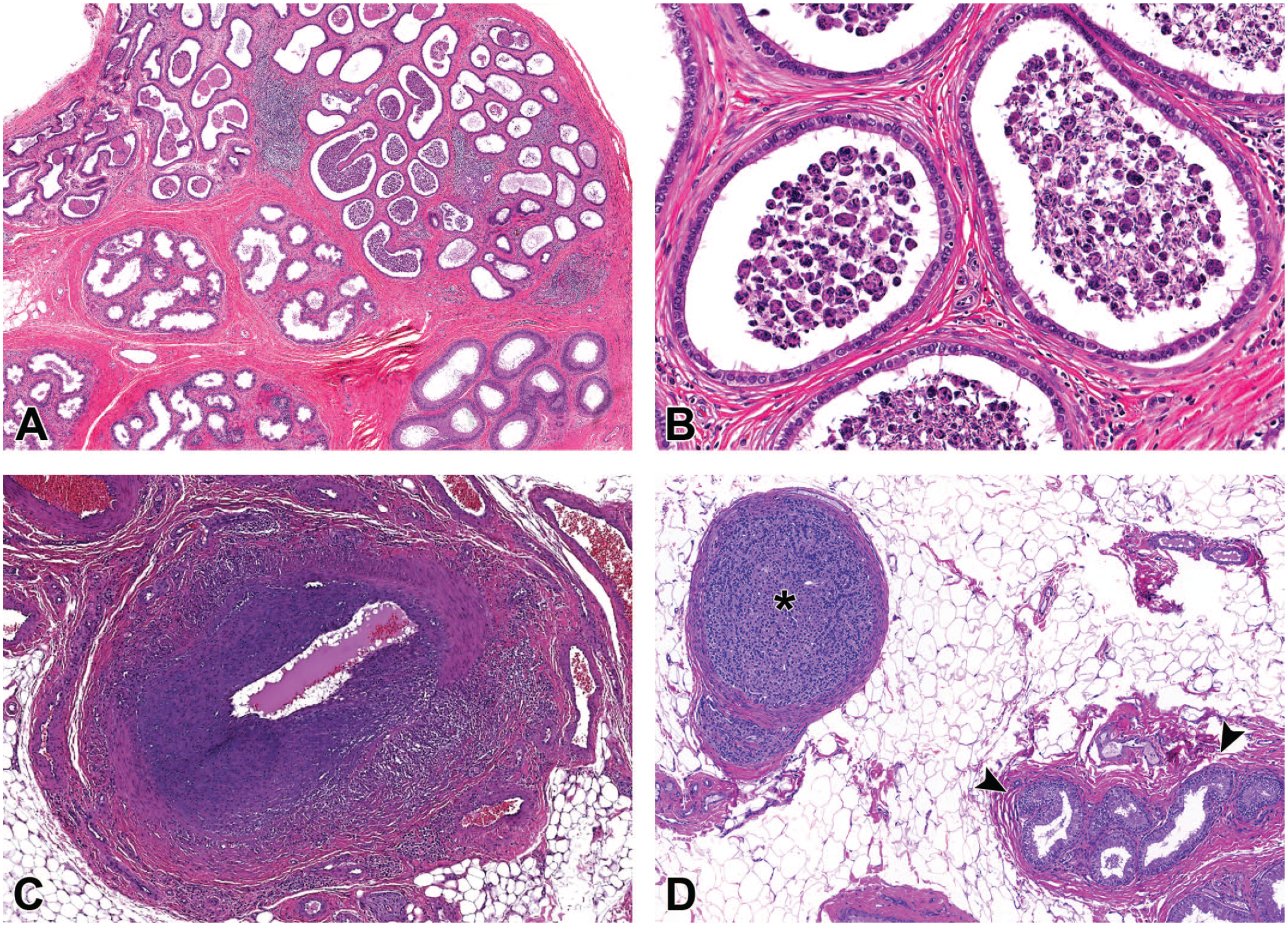
Photomicrographs (H&E) of epididymis demonstrating (A and B) sperm stasis and inflammatory cell infiltrate within the efferent ducts; (C) vascular necrosis and inflammation within an artery adjacent to the epididymis; and (D) ectopic adrenocortical tissue (*) within the fat adjacent to the efferent ducts (arrowheads). Original magnification 2.3X (A), 20X (B), 4.0X (C), and 10X (D).
Prostate
The most common microscopic findings in the prostate were inflammatory cell infiltrate and acinar dilatation (Table 4). Minimal or mild inflammatory cell infiltrates were present within the interstitium of the prostate as focal or multifocal aggregates of lymphocytes and occasional neutrophils (Figure 9A). There was a significant difference in the incidence and severity of inflammatory cell infiltrates where animals from Southeast Asia/China had a greater incidence than animals from Mauritius (Table 4). The incidence of inflammatory cell infiltrates did not differ based on TBW.
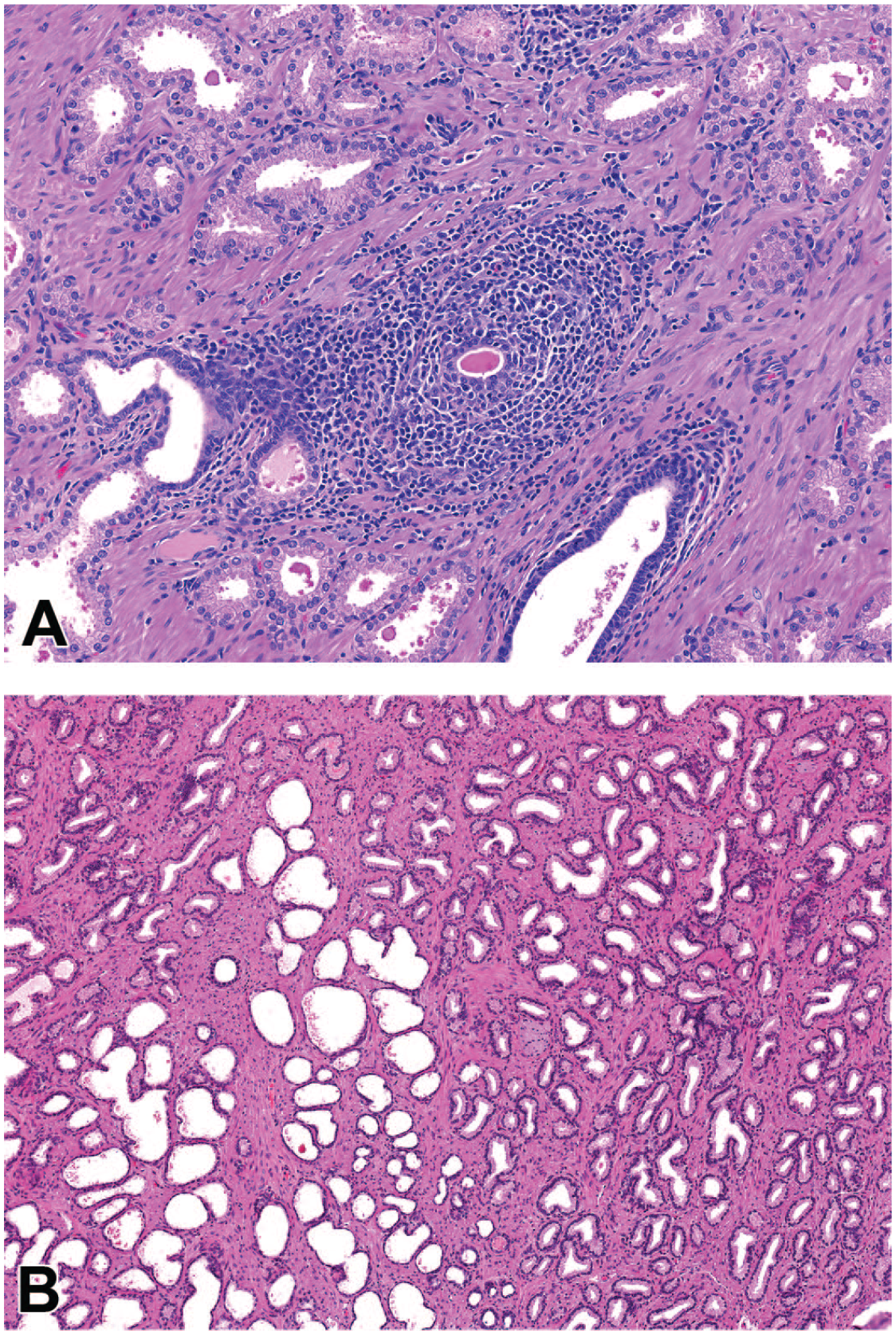
Photomicrographs (H&E) of prostate demonstrating a focal mononuclear infiltrate (A) and focal acinar dilatation (B). Original magnification 20X (A) and 6.6X (B).
Acinar dilatation was present as a minimal to mild, focal to multifocal areas of dilatation and increased luminal size of prostatic acini (Figure 9B). There was no significant difference in incidence between TBW or geographical source.
Less common findings in the prostate included atrophy and stromal fibrosis (Table 4). Minimal to moderate atrophy, characterized by an overall reduction in the size of the prostate with decreased epithelial height and limited secretory material within the cytoplasm, was present at a low incidence in the entire population of animals (3.8%). There was a significant difference (P < .001) between TBW groups with atrophy (all animals combined regardless of source), where animals <5 kg had a greater incidence of atrophy than animals ≥5 kg. When evaluated within geographical source, there was a significant difference in animals from Mauritius where animals <5 kg had a greater incidence of atrophy than animals ≥5 kg (Table 4). A similar difference was present in animals from SE Asia/China, but the difference was not statistically significant. Minimal stromal fibrosis was present in a single animal from SE Asia/China (Table 4).
Seminal vesicles
The most common microscopic findings in the seminal vesicles were corpora amylacea and atrophy (Table 5). Minimal to moderate corpora amylacea were present as multifocal intraluminal aggregates of eosinophilic matrix with/without mineralization (Figure 10A). There was a significant difference (P < .001) between TBW groups with corpora amylacea (all animals combined regardless of source), where animals ≥5 kg had a greater incidence of corpora amylacea than animals <5 kg (Table 5). Similar differences were observed between TBW groups when compared within a geographical source (Table 5). There was a significant difference between geographic source where animals from SE Asia/China had a greater incidence of corpora amylacea than animals from Mauritius (Table 5).
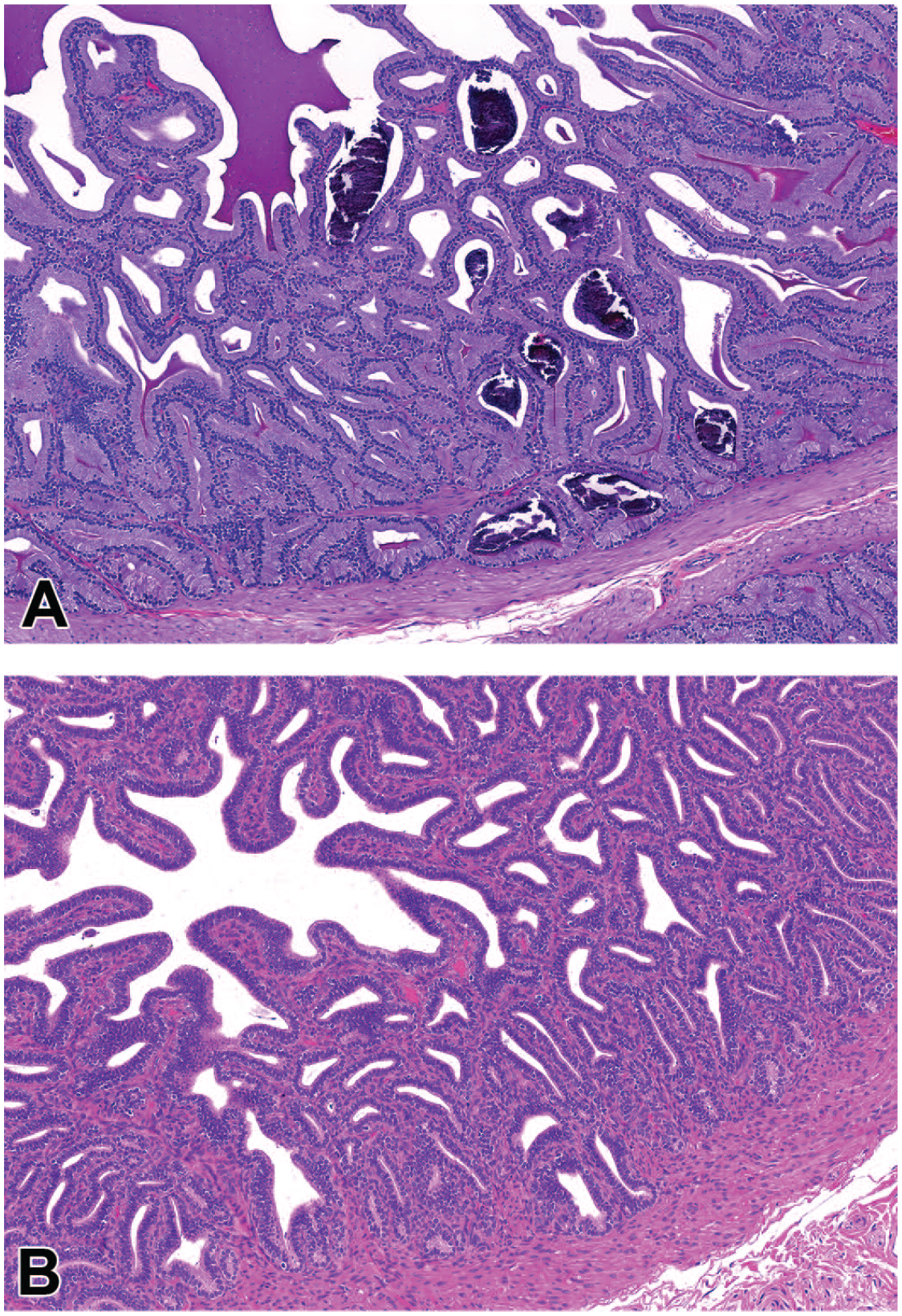
Photomicrographs (H&E) of seminal vesicles from a ≥5 kg male (A) and a <5 kg male (B). In the ≥5 kg male, there are multiple mineralized corpora amylacea within the glandular lumen along with secretory material, whereas in the <5 kg male, there is diffuse glandular atrophy characterized by decreased height of the epithelium and lack of lumen secretory material. As the <5 kg male is likely still peripubertal, atrophy may not be the most appropriate term and the observed lack of glandular development likely represents a feature of normal pubertal development. Original magnification 15X (A and B).
Minimal to moderate atrophy was present as an overall reduced size of the seminal vesicles along with reduced secretory material within the cytoplasm and in the lumen (Figure 10B). There was a significant difference (P < .0001) between TBW groups with atrophy (all animals combined regardless of source), where animals <5 kg had a greater incidence of atrophy than animals ≥5 kg. When evaluated within geographical source, there was a significant difference in animals from Mauritius, where animals <5 kg had a greater incidence of atrophy than animals ≥5 kg (Table 5). A similar difference was present in animals from SE Asia/China, but the difference was not statistically significant.
Additional findings, such as dilatation and inflammatory cell infiltrate, were present sporadically without an association with TBW or geographic source and are detailed in Table 5.
Miscellaneous observations
Several additional notable observations were present in different organs. While these were not tracked for incidence and likely represent features of normal histology, several examples are included for completeness. These include tubuli recti within the testes (Figure 11) and periurethral pigment (Figure 12) and Pacinian corpuscles (Figure 13) in the prostate.
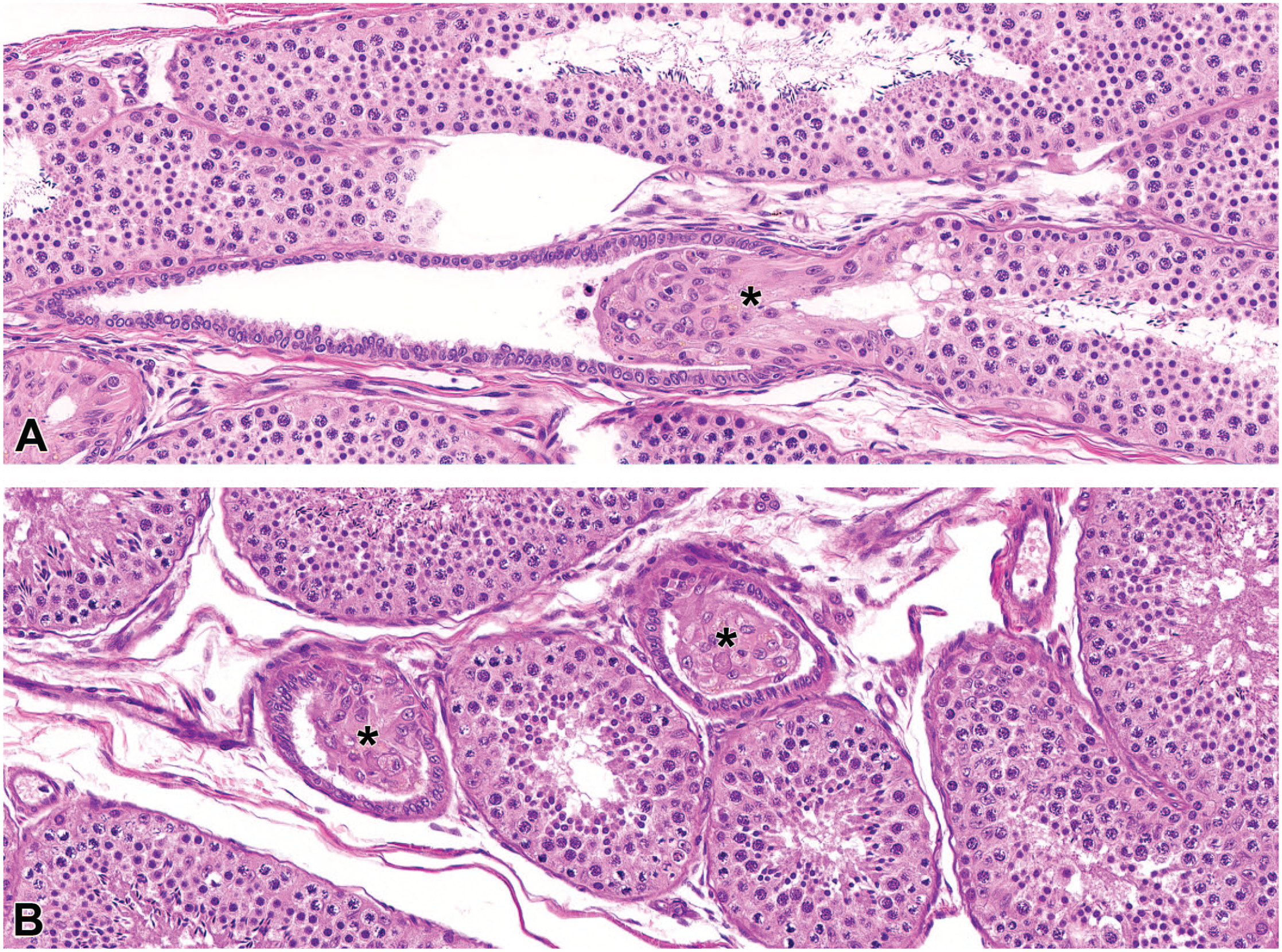
Photomicrographs (H&E) of testis at the region of the rete testis. At the junction of a seminiferous tubule and rete testis, a short portion of the seminiferous tubule is devoid of germ cells with exposed Sertoli cells (*). This region, termed the tubuli recti, is easy to identify in longitudinal section (A), but due to the protrusion of Sertoli cells into the rete testis there was often an unusual appearance in cross section (B) and should not be confused with a proliferative lesion. Original magnification 40X (A and B).
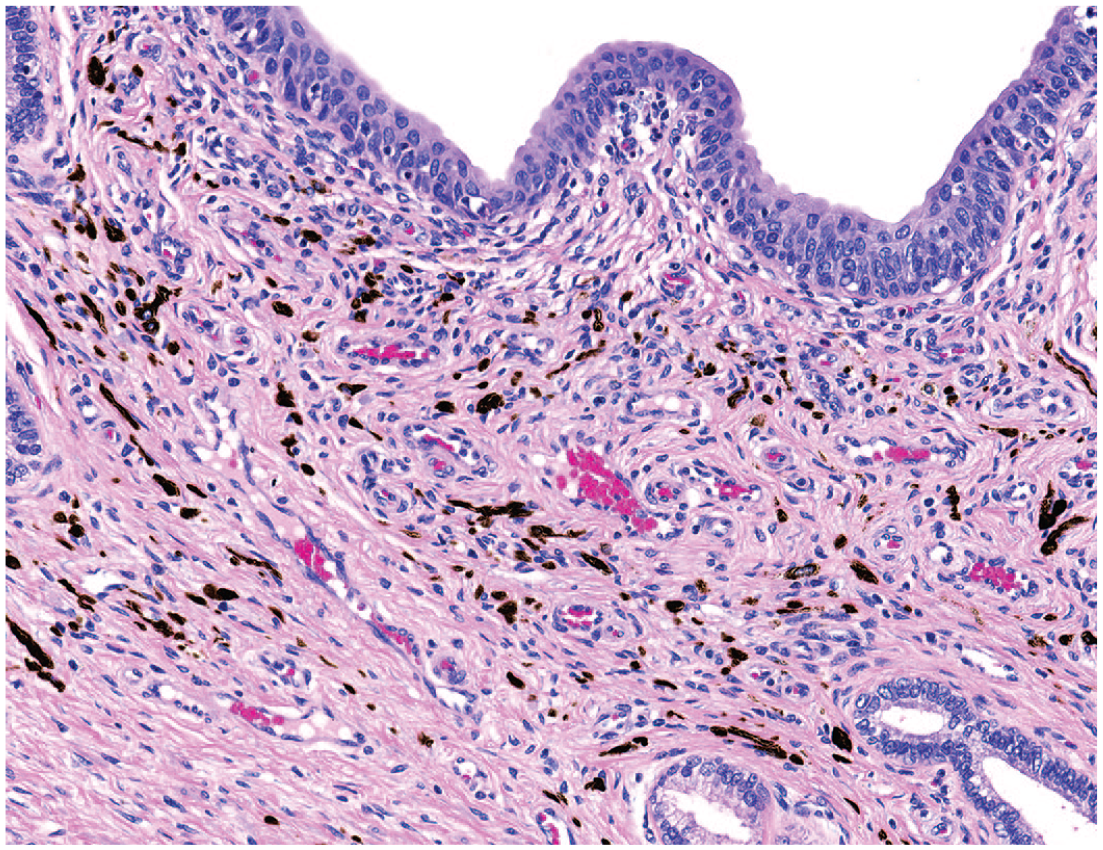
Photomicrograph (H&E) of the prostatic urethra with pigment in the surrounding connective tissue. Original magnification 40X.
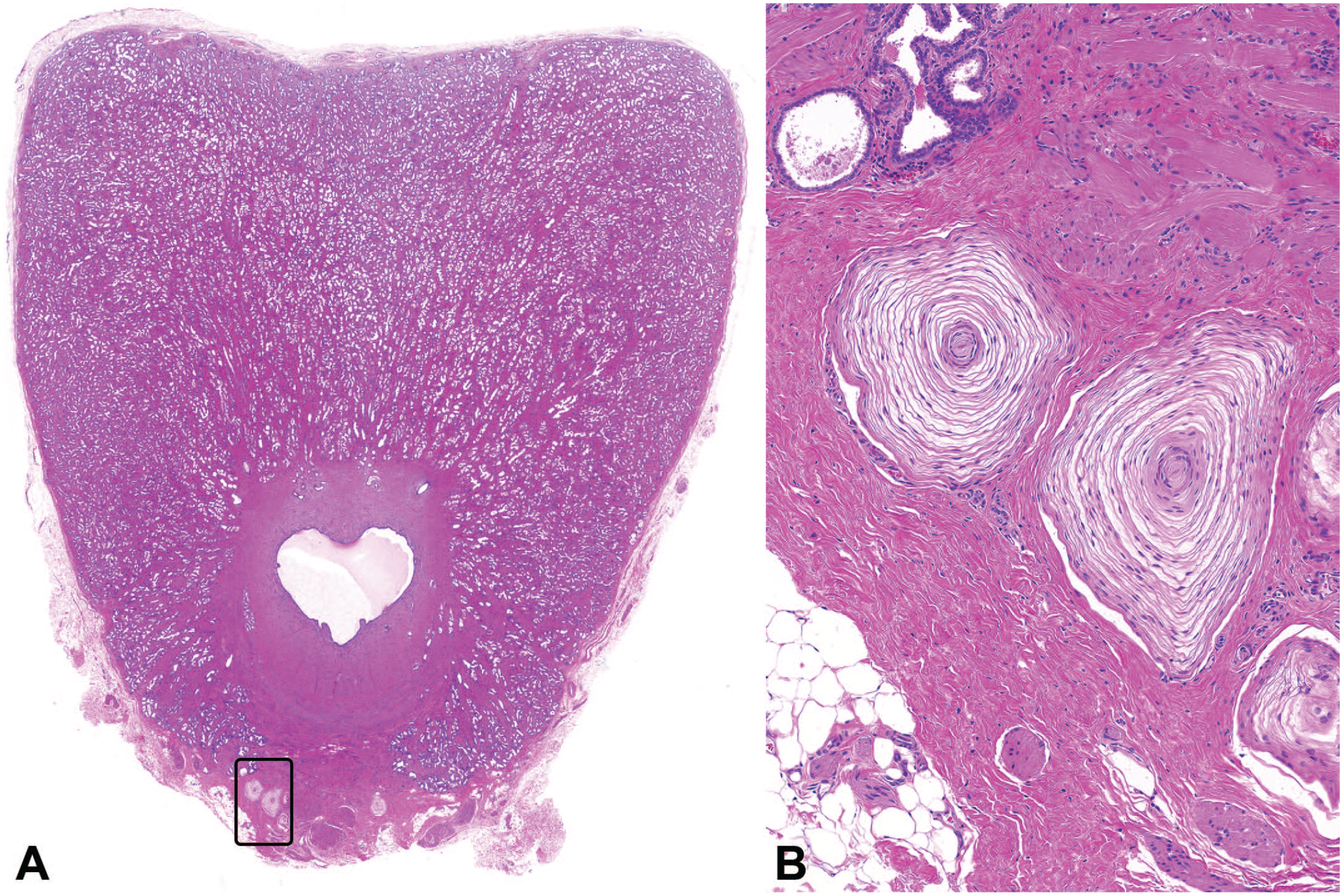
Photomicrographs (H&E) of caudal prostate with a low (A) and high (B) magnification of Pacinian corpuscles. Original magnification 0.7X (A) and 22X (B).
Discussion
Nonhuman primates are often the only species used when evaluating the nonclinical safety of biopharmaceuticals.1,2 As a result, the assessment of the male reproductive system occurs in the context of a repeated dose general toxicity study rather than a standard rodent-based male fertility study. 2 As a result, the pathology evaluation in these studies (OW and histopathology) serves as the primary data set for male fertility risk assessment. This requires the study pathologist to have a thorough understanding of the normal anatomy and physiology of the male reproductive system in NHPs as well as the common spontaneous findings. However, there is limited information in proprietary historical control databases at testing facilities and in the published literature on the common spontaneous findings in the reproductive system of mature male cynomolgus macaques. This study was conducted to address this knowledge gap and provide a comprehensive review, description, and incidence of the commonly observed spontaneous findings in the testes, epididymides, prostate, and seminal vesicles and potentially identify differences associated with TBW or geographical source (SE Asia/China vs. Mauritius).
The microscopic assessment in this study was conducted by a single pathologist with expertise in the male reproductive system using recently published INHAND terminology for the NHP. 5 This allowed for a consistent approach to terminology, thresholds, and grading and avoided the inter-pathologist variability that is inherent in historical control databases. As the goal of this study was to capture all possible spontaneous findings, it is recognized that the incidences described in this study may be higher than on typical nonclinical toxicity studies where thresholds are often applied.
A total of 255 cynomolgus macaques from vehicle control groups on repeated dose toxicity studies were included in this review. All animals were considered to be sexually mature as a requirement of inclusion in this retrospective evaluation. Sexual maturity was based on prestudy semen collection, testicular volume, and/or the presence of sperm in the epididymis during the histopathology assessment at the end of the study. The most common microscopic findings were hypospermatogenesis and tubular dilatation within the testes; inflammatory cell infiltrate, luminal cellular debris, and decreased luminal sperm within the epididymides; inflammatory cell infiltrate and acinar dilatation within the prostate; and corpora amylacea and atrophy within the seminal vesicles.
As stated above, one objective of this project was to identify potential differences in the type and incidence of spontaneous findings based on animal age or geographic source (SE Asia/China vs. Mauritius). Unfortunately, collection of accurate ages was not possible because most of the toxicology study reports did not have recorded ages or exact animal birthdates. To address this limitation, TBW was used as a surrogate for age because there is a strong correlation between age and body weight in cynomolgus macaques up to ~7 to 8 years of age and the age range listed in most study reports had an upper limit of 8 years of age. 8 Most cynomolgus macaques originating from SE Asia/China reach sexual maturity by 5 to 6 years of age and are generally >5 kg.9-11 NHPs originating from Mauritius are reported to reach sexual maturity approximately 1.5 to 2.0 years earlier than those from SE Asia/China, which is due to more rapid growth in animals from Mauritius. 9 Although animal ages were not available, there was no difference in mean TBW in NHPs from SE Asia/China and Mauritius in this review. Based on the requirement of sexual maturity, it was expected that most of the animals, regardless of source, would be >5 kg; however, 16% of the animals included in this review were 3 to 5 kg. It is recognized that body weight alone is not a guarantee of sexual maturity, but 5 kg was selected based on the available literature11-14 and the population of NHPs evaluated in this review were divided into those that were <5 kg and those that were ≥5 kg to compare potential impact of TBW on the parameters in this review. When compared with animals ≥5 kg, animals <5 kg had lower mean testicular weights with an increased incidence of diffuse hypospermatogenesis, lower mean epididymal weights with an increased incidence of cellular debris and decreased luminal sperm, and lower mean prostatic and seminal vesicular weights with an increased incidence of atrophy. These findings likely represent a spectrum of normal features that occur during the late stages of puberty transitioning to sexual maturity.14,15 Spermatogenesis in macaques does not begin in a synchronized fashion and sperm production often starts in a subset of tubules.10,16 As a result, some amount of sperm can be present in the epididymis without complete spermatogenesis in the testes, which likely correlated with diffuse hypospermatogenesis in this review. Following the first appearance of sperm in the epididymides, numbers will continue to increase over time before reaching full adult sperm count. 9
When a pathologist is evaluating male sexual maturity status, the presence or absence of sperm in the tail of the epididymis is generally a clear yes/no assessment of sexual maturity status but judging what constitutes a sexually mature-level complement of sperm in tissue sections can be challenging. This is further complicated when sections of epididymis are incomplete. As a result, animals may have sperm present in the tail of the epididymis during microscopic evaluation, while still being within the late pubertal to early adult phase of development.14,15 These late pubertal to early adult animals may be recorded as sexually mature by study pathologist(s), but these animals may be more consistent with late pubertal/early adult. Of note, most of the animals between 3 and 5 kg did not have semen collection performed prior to study start (data not shown); however, assessment of in life semen collection was not possible in this review as this was most often performed at the supplier prior to study start and data were not included as part of the study reports.
In animals approaching full sexual maturity, incomplete spermatogenesis, increased cellular debris and fewer sperm in the epididymides, and decreased accessory sex OW are expected.9,14,15 Therefore, the diagnoses of hypospermatogenesis in the testes, reduced sperm in the epididymides, and atrophy in the accessory sex organs are likely inappropriate as these are features of normal pubertal development and it is recommended to designate these animals as “peripubertal” in the individual animal data as described in Vidal et al. 17 As such, the study pathologist should use a weight of evidence approach when assessing sexual maturity including semen analysis, TBW, and OW along with the microscopic examination. Ideally, age would be a part of the weight of evidence as it is possible that some of the smaller animals in this project could be sexually mature subordinate animals with suppression of the endocrine axis due to social hierarchy-induced stress and subsequent decreased testicular size. 9 In addition, it is important to note that while animals in this review were separated into groups <5 kg or ≥5 kg as a means of comparing different parameters, 5 kg should not be considered a strict cutoff or definition of sexual maturity. There is a continuum and some individual NHPs <5 kg may have already attained sexually mature, whereas some ≥5 kg may still display peripubertal features. As a result of these challenges, when use of sexually mature animals is a study requirement, semen collection prior to study start is recommended vs. reliance on age, weight, or pathology assessment alone.
The only finding to occur at a higher incidence in animals ≥5 kg was corpora amylacea in the seminal vesicles. These often-mineralized concretions are commonly observed in mature NHPs 3 and the higher incidence in animals ≥5 kg is likely a function of full development of the seminal vesicles and a longer period of time as a sexually mature animal.
When comparing geographic source (SE Asia/China vs. Mauritius), several differences were identified. Multifocal hypospermatogenesis was present at a higher incidence in animals from SE/Asia than those from Mauritius. This differed from the diffuse change described above and consisted of focal to multifocal areas with incomplete spermatogenesis. This change in its more minimal version is subtle and may not be regularly diagnosed in routine toxicity studies, but it is generally similar to that described in the dog.18,19 Multifocal hypospermatogenesis generally had a random appearance without a germ cell specificity and most of the adjacent seminiferous tubules were normal. This pattern is critical to differentiating these changes from germ cell depletion. While a higher overall incidence and severity of multifocal hypospermatogenesis was present in SE Asia/China-sourced animals, there was no apparent difference when considering the minimal severity grade only. The differences across the geographical sources emerge at the higher severity grades in SE Asia/China-sourced animals. As these tended to occur more commonly in animals <5 kg, it is possible that the higher severity grades of multifocal hypospermatogenesis have overlap with diffuse hypospermatogenesis and may be a function of incomplete spermatogenesis during the later portion of puberty. Animals from Mauritius had a lower incidence of inflammatory cell infiltrate in the prostate and epididymides than those from SE/Asia. This is consistent with prior observations in other organ systems where animals of Mauritian origin generally have fewer spontaneous inflammatory cell infiltrates.20,21 Animals from Mauritius also had lower accessory sex gland weights; a lower incidence of germ cell debris and reduced sperm in the epididymides; and a lower incidence of corpora amylacea in the seminal vesicles. Lower prostate weights in Mauritian sourced animals have been reported previously 21 and the epididymal changes may be a function of the more rapid growth rate and attainment of sexual maturity at a younger age. However, in this review, there were fewer animals from Mauritius and studies with these animals were conducted at a limited number of facilities; therefore, the smaller group size and/or other study-related variables may have contributed to differences in OW and/or light microscopic observations.
Additional findings were observed without a clear association with TBW or geographic source. Tubular dilatation was often focal to multifocal and consisted of wedge-shaped areas of tubular dilatation. There was often degeneration and/or atrophy of the seminiferous epithelium with sperm stasis in the lumen. In fact, most of the degenerative and atrophic changes in the testes occurred as secondary changes in dilated seminiferous tubules. When the rete testis was present in the section, there was often inflammatory cell infiltration around tubules adjacent to the rete testis. Taken together, this spectrum of changes in the testes was interpreted to be due to local outflow obstruction at the level of the rete testis. In addition, tubular dilatation often occurred with increased stromal collagen in this review and in the literature, 22 which further implicates outflow obstruction as a cause of the tubular dilatation. The incidence of tubular dilatation and increased stromal collagen have been reported to occur at a higher incidence in SE Asia/China-sourced animals when compared with animals from Mauritius21,22 and while similar trends were present in this data set, clear differences could not be established. This may be a function of the generally lower number of animals from Mauritius or differences in growth rate and attainment of sexual maturity. Regardless, in most animals with focal/multifocal tubular dilatation, most of the adjacent seminiferous tubules were normal, and this pattern is critical to differentiating these changes from tubular degeneration/atrophy.
In the prostate, acinar dilatation was commonly observed and lacked any clearly detectable pattern or association with inflammatory cell infiltrates. It is important to note that macaques have two prostatic lobes (cranial and caudal) 23 and there was considerable variability in which lobe was present on the slide. The cranial prostate serves as a coagulating gland while the caudal lobe is more analogous to the human prostate.24,25 In some cases, it was unclear which lobe was collected and weighed. Cranial vs. caudal lobes were not tracked in any of the original studies and the reported weights of the prostate, lack of correlation with TBW, and overall incidence of microscopic findings in this review may be impacted by variable sampling across testing facilities. As these lobes have a different incidence of proliferative findings in aged animals and respond differently to hormonal agents,24,26 a standard approach to collection and evaluation is recommended. A reasonable approach would be to weigh and fix both prostatic lobes together and evaluate a single cross section of the caudal prostate. In cases where there is additional cause for concern, an additional section of cranial prostate could be included.
Additional findings were present sporadically in testes, epididymides, prostate, and seminal vesicles without an association with TBW or geographic source. In the testes, occasional small foci of tubular hypoplasia were present. These foci were small and lacked the wedge-shaped pattered described in the dog.18,19 Embryonic remnants were identified in both the testes and epididymides and are similar to those reported in NHPs and humans.27-29 In the human literature, these are often described as appendix testis or appendix epididymis and represent remnants of the paramesonephric or mesonephric ducts, respectively. Although there was a low number of embryonic remnants identified, the anatomic location appeared to vary in NHPs from that described in human with the appendix testis attached to the body of the epididymis and the appendix epididymis attached to the testis. Regardless of the embryologic origin or anatomic location, a collective term of “embryonic remnants” is sufficient for most routine nonclinical toxicity studies. 5 In the epididymides, cribriform change was occasionally identified in the head and most often appeared as intra-epithelial lumina. This finding is similar to that described in the dog 19 and likely represents normal microscopic anatomy and would not be expected to be diagnosed in the context of routine studies. Sperm stasis with an associated inflammatory cell infiltrate was occasionally present in the efferent ducts. The incidence of both cribriform change and sperm stasis/inflammatory cell infiltrate may be an underrepresentation as the head of the epididymis, with representation of efferent ducts, was not present in all sections. A single animal had vascular degeneration and inflammation in the epididymides and is similar to findings occasionally present in a variety of species.3,30,31
Finally, several features of normal microscopic anatomy, such as tubuli recti, Pacinian corpuscles, and periurethral pigment, were included. While these do not represent findings that warrant a morphologic diagnosis in a toxicology study, it is important for the pathologist to recognize these as normal features. In particular, the tubuli recti in NHPs are often large and depending on the plane of section can have an unusual appearance that could be confused with a finding.
This review provides a comprehensive description of the common microscopic findings in the male reproductive system of sexually mature NHPs and compared the incidence of these findings based on TBW and geographical source. Given the variety of spontaneous findings present, which could be confused with test article-related findings, and the impact of sexual maturation and geographic source, this review is intended to aid the toxicologic pathologist tasked with evaluating the male reproductive system in mature NHPs.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221082302 – Supplemental material for Spontaneous Findings in the Reproductive System of Sexually Mature Male Cynomolgus Macaques
Supplemental material, sj-docx-1-tpx-10.1177_01926233221082302 for Spontaneous Findings in the Reproductive System of Sexually Mature Male Cynomolgus Macaques by Ronnie Chamanza, Chidozie J. Amuzie, Jennifer Chilton, Jeffery A. Engelhardt, Justin D. Vidal, Manoj Bhaskaran, Mary Carsillo, Steve Denham, Olivia Dubay, Steven Laing, Balasubramanian Manickam, Stephanie Phillips, Jonathan Werner and Armando R. Irizarry Rovira in Toxicologic Pathology
Footnotes
Acknowledgements
The authors would like to thank Sarah Coe, Karyn Colman, Beth Mahler, Kevin McDorman, Justin Moghtader, Deanna Newcomb, Trung Ngyuen, Daniel Patrick, Marie-France Perron Lepage, Jared Slain, and Matthew Smith.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
