Abstract
The INHAND Project (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) is a joint initiative of the Societies of Toxicologic Pathology from Europe (ESTP), Great Britain (BSTP), Japan (JSTP), and North America (STP) to develop an internationally accepted nomenclature for proliferative and nonproliferative lesions in laboratory animals. The purpose of this publication is to provide a standardized nomenclature and differential diagnosis for classifying microscopic lesions observed in the male reproductive system of laboratory rats and mice, with color microphotographs illustrating examples of some lesions. The standardized nomenclature presented in this document is also available for society members electronically on the Internet (http://goreni.org). Sources of material included histopathology databases from government, academia, and industrial laboratories throughout the world. Content includes spontaneous and aging lesions as well as lesions induced by exposure to test materials. A widely accepted and utilized international harmonization of nomenclature for lesions of the male reproductive system in laboratory animals will decrease confusion among regulatory and scientific research organizations in different countries and provide a common language to increase and enrich international exchanges of information among toxicologists and pathologists.
Keywords
General Introduction
For the purposes of background information and references, each section begins with a brief review of normal function, anatomy, and histology. Particularly for the testis, a comprehensive knowledge of normal morphology, spermatogenesis, and cell associations is important for detecting abnormalities. Since it is beyond the scope of this document to provide an in-depth review of normal structure, references are provided to guide the reader to more comprehensive reviews wherever possible.
Correct fixation and appropriate sampling of the tissues are important for a detailed and consistent evaluation. The reader is referred to the trimming protocols in goRENI (Kittel et al. 2004) and Boorman, Chapin, and Mitsumori (1990); Boorman, Elwell, and Mitsumori (1990); Foley (2001); and Suwa et al. (2001, 2002).
Testis
Introduction: Testis
Histopathologic evaluation of the testis is an important component of drug safety assessment and evaluation of environmental toxicants. Proper microscopic assessment of testicular tissue in toxicity studies requires use of sexually mature animals, appropriate fixation, sampling and processing (Foley 2001; Lanning et al. 2002; Latendresse et al. 2002; Kittel et al. 2004), and an understanding of spermatogenesis and its histologic presentation in the species under consideration (Russell et al. 1990).
Histology: Testis (Figures 1–3)
The testis comprises compactly arrayed convoluted loops of seminiferous tubules separated by an interstitium containing Leydig (interstitial) cells, vasculature, macrophages, a protein- and testosterone-rich ultrafiltrate, and supporting stroma. The seminiferous epithelium is formed by basally located Sertoli cells supporting successive synchronized populations of maturing germ cells, namely spermatogonia, spermatocytes, round spermatids, and elongating spermatids. Seminiferous tubules are ensheathed by contractile myoid cells and converge, through tubuli recti, on the rete testis, which is continuous with the efferent ductules and epididymis (Boorman, Chapin, and Mitsumori 1990).
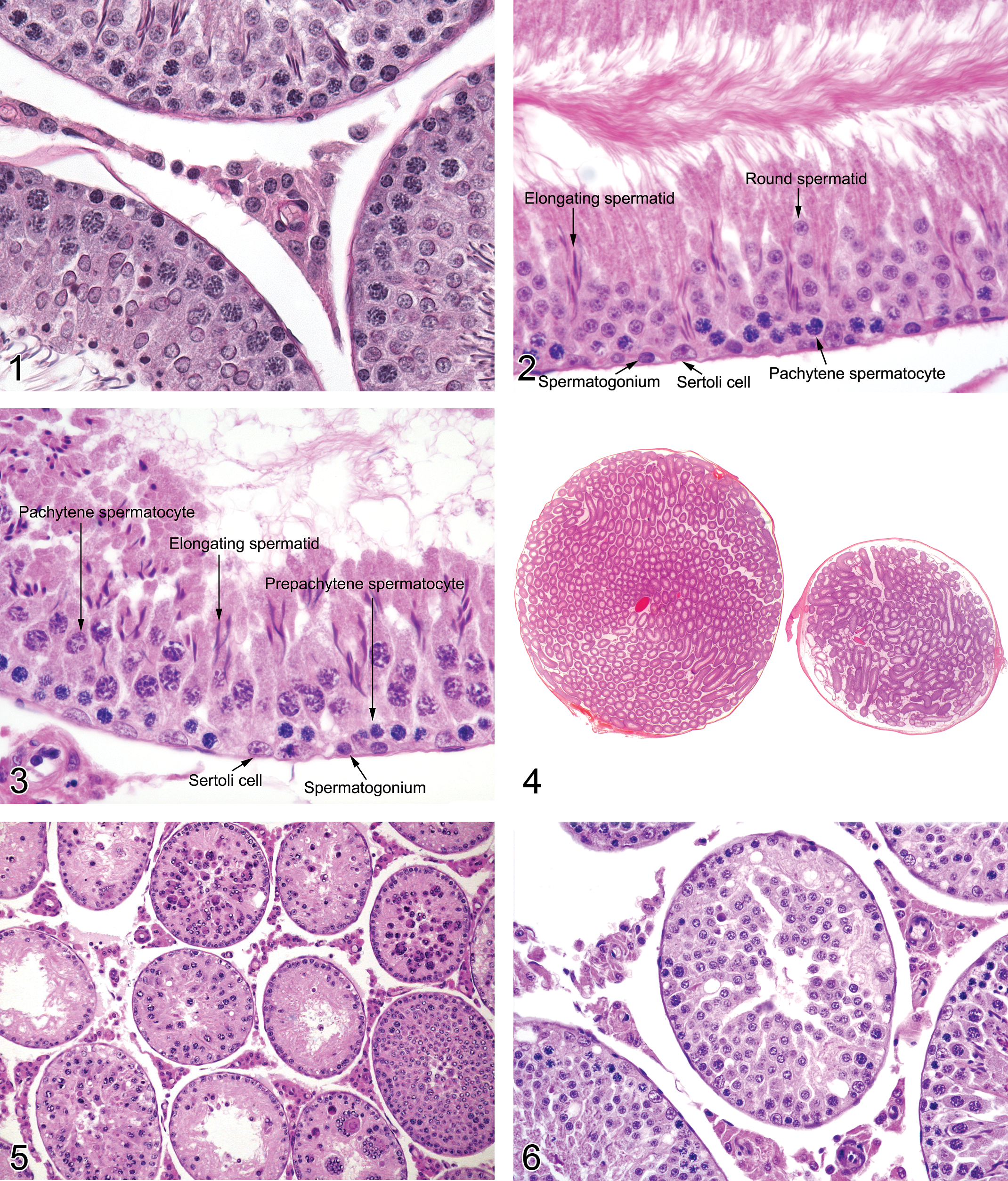
Rat testis. Normal tubules and interstitial tissue.
Germ Cell Types
The germ cell types are distinctive in ploidy, morphology, and susceptibility to harmful influences, including certain chemical and pharmaceutical compounds. Spermatogonia are unique both in constituting the sole proliferative cell population within the seminiferous epithelium and in residing outside of the protective “blood–testis barrier” formed by tight junctions between Sertoli cells. These diploid cells are thus particularly vulnerable to cytotoxic agents, because of their mitotic activity and due to direct exposure to the interstitial ultrafiltrate supplying the seminiferous tubules. Spermatocytes are the largest germ cells, are mostly tetraploid, and represent the meiotic phase of germ cell differentiation. Like the other germ cell types, spermatocytes are susceptible to the variety of circumstances that may trigger apoptosis (including physiologic attrition as well as androgen deficiency and cytotoxic effects); additionally, they can be targets of agents interfering with cytokinesis. Spermatids, round and elongating, are the haploid products of meiotic division. With maturation, which involves progressive loss of cytoplasm and organelles, spermatids are dependent on the supporting Sertoli cell for successful terminal differentiation and release. Due to DNA exchange during the process of meiosis, spermatids and spermatozoa become antigenically foreign, and these “immune-privileged” cells (due to their location within the blood–testis barrier) incite an inflammatory response when the seminiferous epithelium is disrupted.
Sertoli Cells
Sertoli cells are large, post-proliferative cells that are essential to spermatogenesis. They serve multiple complex roles, including simultaneous support of synchronous differentiation among several cohorts of germ cells of differing maturity, maintenance of the blood–testis barrier, secretion of seminiferous fluid, release of matured spermatids, and phagocytosis of residual bodies and apoptotic germ cell remnants (Clermont 1990). Sertoli cells subject to toxicant effects may manifest a variety of morphologic changes, including effects on the germ cells they sustain. Sertoli cells are much less subject to the apoptosis or necrosis in comparison to germ cells and are often the sole survivors within seminiferous epithelium in the face of a variety of chronic degenerative processes.
Leydig (Interstitial) Cells
Intratesticular androgen levels, crucial to germ cell maintenance, are sustained by Leydig cells. These endocrine cells are outside of the protective blood–testis barrier. In addition to direct toxic effects on steroidogenesis, Leydig cell function may be subject to indirect effects impacting gonadotropin release.
Cellular Relationships and Stage-aware Examination
The relationships among germ cells as they mature coordinately have been recognized and described in terms of stages (represented by Roman numerals I to XIV in the rat; I to XII in the mouse), which constitute a useful tool for testicular microscopic examination (Creasy 1997). Knowledge of the cellular relationships occurring during the spermatogenic cycle (stage-aware evaluation) aids in recognition of absent cells and detection of subtle changes restricted to specific points in spermatogenesis. Periodic Acid Schiff reaction and use of plastic embedding to generate thin sections aid in the identification of subtle features of rodent spermatid acrosomes (Russell et al. 1990) but are not essential for stage recognition. It is possible to recognize the most common morphologic effects of toxicity in routine paraffin-embedded hematoxylin- and eosin-stained sections through the ability to recognize two morphologically distinctive cellular associations in the rat: stage VII/VIII, immediately prior to release of mature elongating spermatids (spermiation), and stage XIV, representing division of meiotic spermatocytes. Recognition of where a given tubular cross section falls temporally in relation to stages VII/VIII and XIV informs the examiner of which cell layers should be represented: two layers of spermatids (one layer of round spermatids and one layer of elongating spermatids) in stages I through VIII (Figure 2) versus a single layer of elongating spermatids in stages IX through XIV (Figure 3). This is in addition to spermatocytes and spermatogonia in all tubular cross sections. In many cases, testicular toxicity is cell-specific and stage-specific during the early stages of its development, but with continued dosing, cell degeneration becomes more generalized and nonspecific. In this respect, examination of spermatogenesis with stage awareness is generally most valuable for compound exposures of 1 month or less (Lanning et al. 2002). Due to the variable appearance of seminiferous tubular changes in short-term versus chronic studies, terminology that deals with cell- and stage-specific changes and terminology that can be used for nonspecific changes have both been presented. Test article related changes may be cell- and stage-specific or nonspecific, whereas incidental background changes are generally nonspecific.
Congenital Lesions: Testis
Agenesis: Testis
Synonym: Aplasia
Pathogenesis: developmental abnormality of the gonadal (urogenital) ridge, mesonephros
Diagnostic feature One or both testes are absent as a gross finding
Differential diagnoses Fibrosis, testis: if a testis becomes infarcted resulting in contraction and replacement by fibrotic tissue, it may be mistaken for agenesis, but there will generally be residual tubular tissue and/or evidence of inflammation and fibrosis
Comments: This is a rare congenital condition, but it could be a test article–related finding if exposure occurred during
Hypoplasia (Figure 4): Testis
Pathogenesis: failure of sex cords to develop in the fetal testis
Diagnostic features Small testis or testes Reduced organ weight Unilateral or bilateral Reduced number of tubular profiles
Differential diagnoses Sampling artifact (caused by sampling the testis at the cranial or caudal pole, giving the impression of reduced numbers of tubules): normal testis weight Atrophy, tubular: the number of tubules in the testis is normal but the tubules lack all or most of their germ cells Fibrosis, testis: if a testis becomes infarcted resulting in contraction and replacement by fibrotic tissue, it may be mistaken for hypoplasia, but there will generally be residual tubular tissue and/or evidence of inflammation and fibrosis Immaturity: the number of tubules in the testis is normal but the tubules lack varying numbers of germ cells
Comments: It is recommended that the term hypoplasia only be used for situations where there has been a developmental abnormality resulting in an obvious reduction in the number of tubules. The term hypoplasia is often more widely defined and in reality, it is possible for there to be a failure of germ cell migration into entire tubules or into segments of tubules, resulting in hypoplastic tubules (similar to the segmental hypoplasia observed in the beagle dog testis). However, since it is impossible to morphologically distinguish this condition from tubular atrophy, where germ cells were once present, but have since undergone degeneration and depletion, it is recommended that in the rodent, hypoplasia be restricted to a reduction in the number of tubules only. Testicular hypoplasia is an uncommon developmental abnormality in rodents that may be unilateral or bilateral. It may also be related to test article administartion if exposure occurred during
Cryptorchidism: Testis
Synonyms: undescended testis, failure of testicular descent
Pathogenesis: failure of gubernaculum-facilitated passive descent of testis through the inguinal canal into the scrotal sac
Diagnostic features One or both testes are intra-abdominal Small testis/testes with decreased organ weights Tubules with degenerating or missing germ cells When unilateral, tubular degeneration may be present in the contralateral descended testis
Differential diagnoses Necropsy artifact: normal testicular histology Tubular degeneration/atrophy: degenerating and/or missing germ cells in a testis that is descended
Comments: Cryptorchidism is a gross observation made when one or both testes are present in the abdominal cavity due to failure of normal descent (which occurs at approximately PND 22 in rats). Testes are frequently found at necropsy in the abdominal cavity of non-cryptorchid animals due to the ability of rodents voluntarily to retract the testis through the inguinal canal, which remains patent in the adult rodent (Boorman, Chapin, and Mitsumori 1990). Because there are no specific histopathologic changes that characterize a cryptorchid testis, other than tubular degeneration and atrophy, it is critical that the gross observation of intra-abdominal testes has been accurately made. The tubular degeneration/atrophy present in undescended testes is thought to be due to exposure of the seminiferous epithelium to the increased temperature of the abdominal cavity relative to the scrotal sac. The reasons for the degenerative changes in the contralateral, descended testis are unknown. Cryptorchidism occurs spontaneously at a high frequency in Long Evans rats (Barthold et al. 2006) or in unilateral urogenital anomalies in rats (Amakasu, Suzuki, and Suzuki 2009).
Seminiferous Tubular Changes, Nonspecific: Testis
Introduction
The nomenclature associated with spermatogenic disturbance in the testis will depend on whether germ cell changes are cell- and/or stage-specific (generally related to test article administration and seen after exposures of 28 days or less) or the changes reflect a more generalized, nonspecific germ cell degeneration and depletion. The nonspecific terminology, which may be applied to spontaneous or compound-related effects in animals of any age, is presented first.
Degeneration/Atrophy, Tubular (Figure 5): Testis
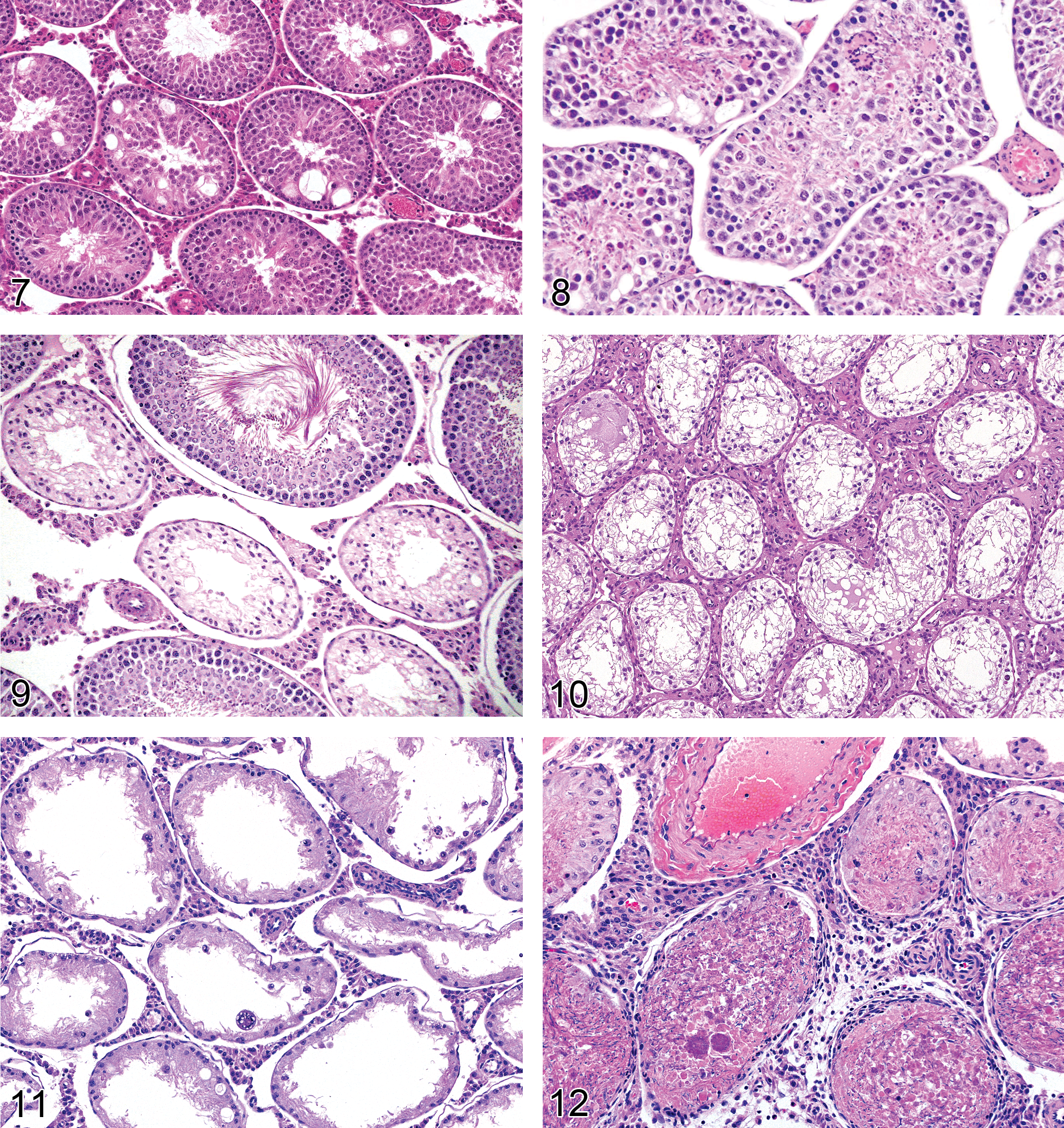
Rat testis. Degeneration, tubular.
Pathogenesis: consequence of germ cell degeneration and depletion, which may be mediated through Sertoli cell injury, primary cytotoxicity, hypoxia, inflammation, or other effects
Diagnostic features Some tubules depleted of all germ cells and lined only by Sertoli cells. Some tubules with partial germ cell depletion Some tubules containing actively degenerating or apoptotic germ cells A mixture of the features described for “degeneration, tubular” and “atrophy, tubular” (see below) Reduced organ weight
Differential diagnoses See “Degeneration, tubular” and “Atrophy, tubular”
Comments: It is highly recommended that “degeneration/atrophy, tubular” be used as the summative diagnosis when there are combinations of changes that include atrophy, degeneration, vacuolation, exfoliation, and so on. This is preferred over listing individual diagnoses. In most cases of tubular degeneration/atrophy, there will be a proportion of tubules that are undergoing active germ cell degeneration (tubular degeneration) and a proportion of tubules that have lost all of their germ cells (tubular atrophy). Since degeneration and atrophy form a continuum, there is generally no reason to distinguish between degenerating and atrophic tubules, hence, the recommendation to use the term degeneration/atrophy. Severity gradings can be used to provide an indication of the numbers of germ cells and tubules affected. Individual use of the terms “degeneration, tubular” and “atrophy, tubular” may be justified for more specific, test article–related changes if there is a clear separation of the two processes. This might be particularly useful to distinguish differences between end of dosing and recovery phases, or in a time course study. Unilateral and bilateral tubular degeneration/atrophy may be seen as a low incidence, background finding in rats and mice. A common incidental finding in rat and mouse testes is the presence of a few (e.g., 1–5) atrophic tubular profiles lined only by Sertoli cells (Foley 2001). If these are recorded, it is important that they be distinguished from the more generalized distribution of tubular degeneration/atrophy described above. This may be done with the use of modifiers (e.g., focal).
Degeneration, Tubular (Figures 6–8): Testis
Pathogenesis: the consequence of germ cell degeneration, which may be mediated through Sertoli cell injury, primary cytotoxicity, hypoxia, inflammation, or other effects
Diagnostic features (may include any combination of the following) Germ cell degeneration (individual germ cells with eosinophilic cytoplasm and nuclear condensation), not restricted to a specific germ cell type or stage Multinucleated germ cells Spermatid retention Sertoli cell cytoplasmic vacuolation Disorganization of germ cells Exfoliation of germ cells into tubular lumen Segmental partial germ cell loss Degenerate germ cells and debris present in the epididymis Generally associated with reduced organ weight
Differential diagnoses Atrophy, tubular: majority of tubules depleted of most or all germ cells, leaving tubules mostly lined by Sertoli cells Degeneration, germ cell: largely restricted to a specific cell type and/or a specific tubular stage; generally seen as a compound-related change in studies less than 28 days duration Fixation artifact: germ cell dissociation and sloughing of cells into the lumen without seminiferous epithelial architectural disorder and without cell debris or sloughed cells in the epididymis; generally subcapsular Handling artifact: architectural disorder and/or sloughing of germ cells into the lumen without germ cell degeneration; generally affects few tubules in the subcapsular region; no sloughed germ cells or debris in the epididymis (Foley 2001) Necrosis, tubular: coagulative necrosis of seminiferous epithelium including Sertoli cells; inflammation may be present Autolysis: generalized dissociation of the Sertoli and germ cells with clumping and margination of nuclear chromatin; no cell debris or sloughed cells in the epididymis. (Bryant and Boekelheide 2007). Dilation, tubular: increased tubular diameter, thinned epithelium containing normal complement of germ cells
Comments: Tubular degeneration and its sequel, tubular atrophy, are common manifestations of toxicologic injury to the testis (Greaves 2012a; Yuan and McEntee 1987), encompassing effects mediated through Sertoli cell injury, germ cell injury, hormonal disruption, or vascular effects. In the early stage of lesion development, it may be possible to identify changes specific to a single cell type (Sertoli cell or specific germ cell types), in which case, it is encouraged to use one of the cell/stage specific diagnoses, but with continued dosing, a more generalized, nonspecific tubular degeneration or tubular atrophy usually develops. The distinction between tubular degeneration and tubular atrophy (see below) depends on the number of germ cells remaining in the affected tubules. If there is a mixture of atrophic and degenerating tubules (which is generally the case), the term “degeneration/atrophy, tubular” is recommended. Tubular degeneration (generally bilateral) also can be seen as a low incidence background finding in rats and mice. The finding generally presents as a small number of tubules with partial germ cell depletion and occasional degenerating germ cells.
Atrophy, Tubular (Figures 9–11): Testis
Synonym: Sertoli cell–only tubules.
Pathogenesis: consequence of prolonged or severe germ cell degeneration, which may be mediated through Sertoli cell injury, primary cytotoxicity, hypoxia, inflammation, or other effects
Diagnostic features Absence of most or all germ cells from affected tubules Tubules lined only by Sertoli cells Decreased tubular diameter Focal (segmental) or diffuse distribution Interstitial cells may be relatively or actually increased in size and/or number A small number of residual germ cells may be present in affected tubules Decreased testis size and weight possible (depending on number of tubules affected)
Differential diagnoses Degeneration, tubular: tubules have significant numbers of degenerating germ cells present Hypoplasia: reduced numbers of tubules Tubuli recti: normal structures lined only by Sertoli cells, focal and adjacent to rete testis (subcapsular). These should not be recorded as a diagnosis nor mistaken for atrophic tubules Dilation, tubular: increased tubular diameter, thinned epithelium containing normal complement of germ cells
Comments: Tubular atrophy is an end-stage lesion where there are no germ cells left within a tubule. It can result from progressive degeneration and phagocyosis/exfoliation of germ cells or cumulative depletion of germ cells (e.g., prolonged maturation depletion). It is a nonspecific change. If there is a mixture of atrophic and degenerating tubules, the term “degeneration/atrophy, tubular” is recommended.
Necrosis, Tubular (Figure 12): Testis
Pathogenesis: intermittent ischemia or hypoxia (e.g., caused by disturbances in blood flow) impacting germ cells and Sertoli cells
Diagnostic features Coagulative necrosis of germ cells and Sertoli cells Generally focal or multifocal (segmental) Disruption of normal tubular architecture Often associated with inflammatory infiltrate surrounding and invading affected tubules
Differential diagnoses Autolysis: no inflammatory response; cell dissociation without cytoplasmic and nuclear changes of necrosis; Sertoli cells intact Degeneration/atrophy, tubular: degeneration and/or depletion of germ cells; Sertoli cells intact; Leydig cells intact, no inflammatory response Necrosis, testicular: a more diffuse change with necrosis of Leydig cells and interstitial structures in addition to tubular elements
Comments: Tubular necrosis represents coagulative necrosis of the seminiferous epithelium, in contrast to apoptosis of germ cells, represented by germ cell degeneration. Sertoli cells are generally extremely resistant to cell death and their tight junctions (forming the blood–tubular barrier) are rarely breached by injurious agents. Ischemia, however, can cause Sertoli cell death, and in such cases an inflammatory response generally accompanies the injury. Due to the loss of Sertoli cells, tubular necrosis is not reversible; the affected area is replaced by scar tissue. Tubular necrosis appears after compound-induced ischemia, due to endothelial cell toxicants (cadmium) or vasoactive agents (e.g., serotonin, histamine, and epinephrine; Creasy 2001; Creasy and Foster 2002; Lanning et al. 2002).
Necrosis, Testis (Figures 13–15 and 43): Testis
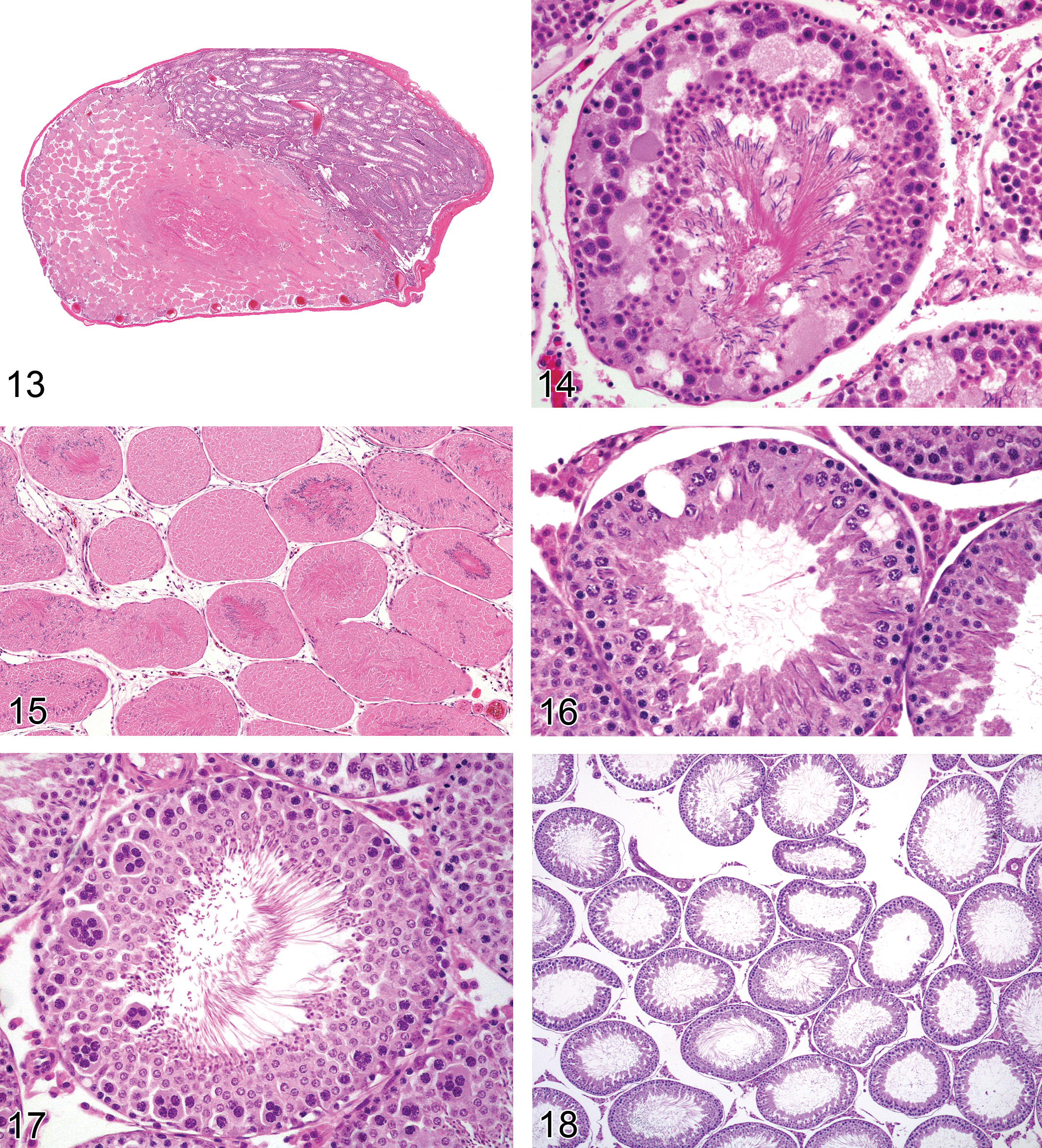
Rat testis. Necrosis, testis, partial.
Pathogenesis: prolonged ischemia (e.g., caused by torsion, thrombi, or prolonged vasoconstriction) impacting all testicular elements
Diagnostic features Coagulative necrosis of all structures within the testis (infarct) Possible acute to chronic inflammation
Differential diagnoses Autolysis: no inflammatory response; cell dissociation without cytoplasmic and nuclear changes of necrosis Tubular degeneration/atrophy: degeneration and/or depletion of germ cells; Sertoli cells intact; Leydig cells intact, no inflammatory response Tubular necrosis: degeneration of seminiferous epithelium, including Sertoli cells, with persistence of interstitial structures
Comments: Testicular necrosis is generally the result of ischemia, which is most often caused by torsion of the testis resulting in prolonged obstruction of the blood flow. Torsion is a gross (rather than microscopic) term. This spontaneous finding is uncommon in rats and mice and is usually unilateral. The extent of tubular injury depends on the duration and the severity of the torsion (Becker and Turner 1995). Chemically induced testicular necrosis has also been described in rodents administered cadmium, which is an endothelial toxicant in the testis of rodents (Aoki and Hoffer 1978) and can be caused by agents causing vascular thrombosis in the testis. A characteristically focal necrosis at the frontal lower part of the rat testis has been described following a single administration of human chorionic gonadotropin (hCG) to rats. The necrosis was considered to be due to local ischemia caused by prostaglandin release from Leydig cells (Chatani 2006). Testicular necrosis is not reversible, due to loss of Sertoli cells and tubular structure, and the affected area is replaced by scar tissue. Less severe forms of anoxia or ischemia are likely to result in tubular necrosis (see above). Extensive necrosis of the testis can also be caused by accidental i.p. injection directly into the testis (which can occur because the rat is able to retract its testes into the abdominal cavity).
Vacuolation, Tubular (Figure 16): Testis
Rat testis. Dilation, tubular.
Pathogenesis: vacuolation of the seminiferous epithelium may result from a number of degenerative changes including accumulation of fluids, lipids, or phospholipids. Vacuoles can also result from the physical loss of embedded germ cells
Diagnostic features Macro-vacuolation: single, large vacuoles within the tubular epithelium at any level of seminiferous epithelium Microvacuolation: multiple, small, vacuoles within the basal Sertoli cell cytoplasm
Differential diagnosis Fixation artifact: caused by hyperosmotic fixatives and restricted to a line of vacuoles adjacent to the basement membrane and below the level of the Sertoli cell tight junctions
Comments: Occasional solitary vacuoles can be seen as an incidental finding in most control testes. This diagnosis should only be used when the number of vacuoles is above control levels. Vacuolation is generally an early morphological indicator of disturbance to the Sertoli cell (Creasy 2001). The vacuoles may be intracellular or intercellular and in both cases probably reflect a disturbance in fluid balance of the Sertoli cell. Microvacuolation of the basal Sertoli cell cytoplasm occurs with some Sertoli cell toxicants (Hild et al. 2001) and with some phospholipidosis-inducing chemicals. Significant germ cell degeneration and necrosis are often accompanied by vacuolation of the tubular epithelium, secondary to the loss of the space occupying germ cell from between the Sertoli cell processes (Kerr et al. 1993). Tubular vacuolation should only be used as a diagnosis when the vacuolation is considered a primary or separate event.
Multinucleated Giant Cells (Figure 17): Testis
Synonyms: symplasts, syncytial cells
Pathogenesis: impaired Sertoli cell maintenance of cytoskeletal bridge closure among cohorts of spermatids (and less frequently spermatocytes) or germ cell degeneration (Greaves 2012a)
Diagnostic features Large cells having multiple germ cell nuclei of the same maturity May be present within the seminiferous epithelium, within the tubular lumen, or within epididymal lumen Often present with evidence of tubular degeneration or atrophy
Differential diagnosis Immature testis (pre- or peripubertal): small numbers of affected cells without other concurrent degenerative changes; present among control animals
Comments: A feature of spermatogenesis is that the dividing spermatogonia and spermatocytes maintain stable cytoplasmic bridges between their progeny, resulting in the interconnection of groups of cells. Giant multinucleated spermatids or spermatocytes are probably the result of spermatid or spermatocytes degeneration and widening of the intercellular bridges. They are found in association with age-related focal testicular atrophy, following efferent duct ligation, exposure to gamma irradiation, or as a result of administration of many xenobiotics (Hild et al. 2007). These symplasts finally die and are phagocytized by Sertoli cells or are sloughed into the tubular lumen. Because multinucleated giant cells are frequently associated with tubular degeneration, they are usually considered a component of that entity, and as such, are usually not diagnosed separately. However, when they are the only change present, this specific diagnosis is appropriate. Multinucleated giant cells may be more prevalent among immature animals; only frequency levels occurring noticeably beyond those among concurrent control animals should be recorded. In the
Dilation, Tubular (Figures 18–20): Testis
Pathogenesis: impaired fluid reabsorption in the efferent ducts/caput epididymis, distal obstruction in excurrent duct system (rete testis, efferent ducts, or initial segment), increased secretion of seminiferous tubule fluid by Sertoli cells, or inhibition of peritubular contractions, resulting in increased seminiferous tubular fluid
Diagnostic features Increased luminal diameter of seminiferous tubules Accompanied by thinning of the seminiferous epithelium (but usually with normal number of germ cell layers present) Increased testis weight usually present May be accompanied by dilated rete testis, dilated efferent ducts, or sperm stasis/sperm granulomas in the efferent ducts May be associated with germ cell degeneration as a progressive consequence of increased pressure
Differential diagnosis Tubular degeneration/atrophy: degeneration and/or depletion of germ cells without increase in tubular luminal diameter
Comments: Tubular dilation can be seen as a background incidental lesion or may be related to treatment. In either case, it can be unilateral or bilateral. If it is unilateral, the probable cause is efferent duct blockage. The efferent ducts are the major site of reabsorption of seminiferous tubule fluid, which is secreted by the Sertoli cells (Hess 2002). Excessive resorption can result in sperm stasis and blockage; inadequate resorption can result in efferent duct dilation and fluid backpressure (Hess 1998). Resorption of fluid involves active transport mediated through a sodium chloride ion exchange mechanism and removal of fluid by an adequate blood supply. Estrogen is an important regulator of fluid resorption and endothelin is also involved in the process (Harneit et al. 1997; Hess 2002). Estrogen receptor α knockout (ERKO) mice develop tubular dilation with subsequent pressure atrophy and infertility at an age when seminiferous fluid starts to be secreted (Eddy et al. 1996). Tubular dilation has also been described following administration of endothelin antagonists (Creasy 2001), the fungicide carbendazim (Nakai et al. 1992), and a 5HT agonist, where the pathogenesis was considered to be due to reduction in fluid resorption caused by vasoconstriction of the blood vessels in the mediastinal plexus overlying the rete testis (Piner et al. 2002).
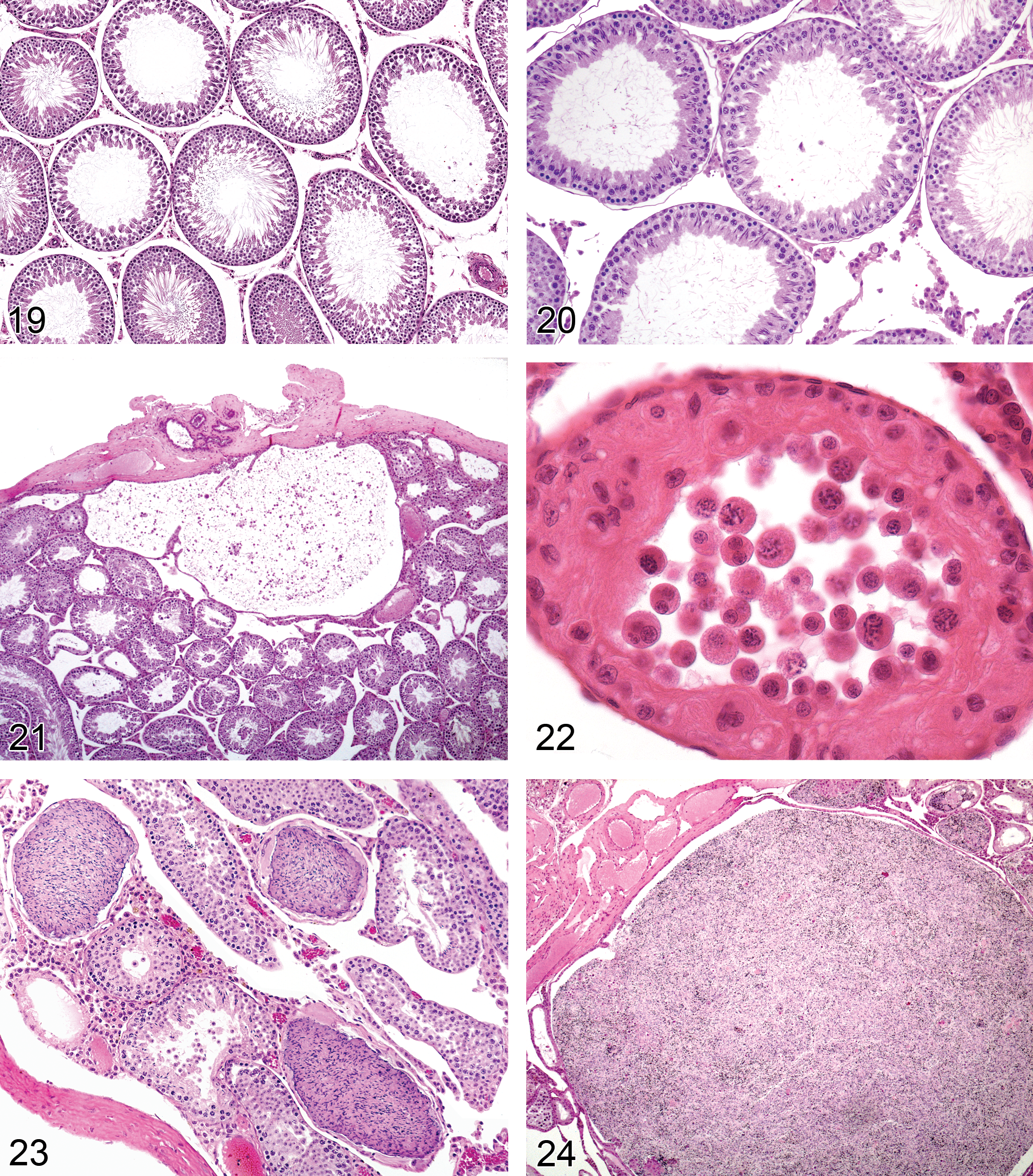
Dilation, Rete Testis (Figure 21): Testis
Pathogenesis: impaired fluid reabsorption in the efferent ducts/caput epididymis, distal obstruction in excurrent duct system (rete testis, efferent ducts, or epididymis), inhibition of peritubular contractions, or increased secretion of seminiferous tubule fluid by Sertoli cells, resulting in increased seminiferous tubular fluid
Diagnostic features Increased lumen size of rete testis May be associated with seminiferous tubular dilation
Comments: Rete testis dilation may be associated with sperm stasis or sperm granuloma of the rete testis or efferent ducts and decreased or absent epididymal sperm. Dilated rete testis was an early event associated with seminiferous tubular dilation caused by a 5-HT agonist (Piner et al. 2002).
Exfoliation, Germ Cell (Figure 22): Testis
Pathogenesis: disturbance of Sertoli–germ cell intercellular junctions leading to loss of germ cell adherence to Sertoli cell
Diagnostic features Presence of nondegenerated germ cells in the seminiferous tubular lumen Accompanied by sloughed germ cells in the rete testis or epididymal lumen Accompanied by depletion of germ cells from the seminiferous epithelium
Differential diagnoses Necropsy artifact: no exfoliated cells in epididymis lumen and no obvious depletion of germ cells from the seminiferous epithelium Immature and prepubertal animal: accompanied by absence/reduction of sperm in the tail of the epididymis Abnormal residual bodies: hypereosinophilic, apoptotic like bodies; stage-restricted
Comments: Exfoliation of germ cells can be a specific response to certain Sertoli cell toxicants (e.g., colchicine, vinblastine, and phthalate esters; Creasy and Foster 2002; Lanning et al. 2002) or it can be a secondary event associated with nonspecific germ cell degeneration. In the case of colchicine and carbendazim, extensive sloughing of the adluminal germ cells and Sertoli cell processes is due to effects on the microtubules that form the Sertoli cell cytoskeleton. In the case of phthalate esters, retraction of the Sertoli cell cytoplasmic processes has been demonstrated. In these cases, the seminiferous tubules contain large numbers of normal appearing germ cells in the seminiferous tubular lumen and an obvious depletion of germ cells from the seminiferous epithelium. There will also be large numbers of normal appearing germ cells in the epididymis. Sloughed germ cells and cell debris will also be present in the epididymis with most other forms of tubular degeneration/atrophy, but this generally reflects the sloughing of degenerating germ cells rather than a specific effect on the Sertoli–germ cell junctions. The finding of “exfoliation, germ cell” should only be used in the testis when it is considered a primary event. The term “cell debris, intraluminal” can be used in the epididymis to record the more common occurrence of degenerating germ cell debris within the epididymis that occurs secondary to tubular degeneration/atrophy (see Epididymis/Efferent duct terminology).
Stasis, Sperm (Figure 23): Testis
Synonym: sperm impaction
Pathogenesis: decreased intratubular fluid or tubular propulsion, impaction of released sperm inside the lumen of seminiferous tubules or rete
Diagnostic features Luminal aggregation of released sperm, generally within an atrophic tubule Frequently associated with mineralization of the sperm Commonly near or within the rete testis
Differential diagnoses Spermatocele: sperm stasis with expansion of the tubule to more than two times the normal Sperm granuloma: sperm stasis with infiltration of macrophages
Comments: Sperm stasis can be associated with increased fluid reabsorption or decreased fluid secretion in efferent ducts (benomyl; Hess 1998). Sperm stasis is occasionally seen as an incidental finding in rats and mice, particularly in atrophic tubules of aged mice. When sperm stasis occurs within the rete and causes obstruction of outgoing seminiferous tubule fluid, it can lead to tubule dilation and tubule atrophy, as described following administration of theophylline (Foley 2001).
Spermatocele (Figure 24): Testis
Synonym: sperm cyst
Pathogenesis: decreased intratubular fluid or tubular propulsion, impaction of released sperm inside the lumen of seminiferous tubules or rete
Diagnostic feature Sperm filled tubule that is greater than two times the normal tubular diameter
Differential diagnoses Sperm granuloma: sperm stasis with infiltration of macrophages Sperm stasis: luminal aggregation of released sperm; increase of the lumen size less than two times the normal tubular diameter
Comments: May result in proximal dilation of tubules or sperm granuloma formation. Spermatoceles and sperm granulomas are often spontaneous changes; however, they may be compound related due to effects on intratubular secretion.
Seminiferous Tubular Changes, Cell and/or Stage-specific: Testis
The following terms are recommended for changes of the testis related to specific germ cell types and/or stages, usually associated with shorter duration (up to 28 days) compound administration. They also can be seen as occasional background changes, but this is uncommon in the rat.
Degeneration, Germ Cell (Figures 25–28): Testis
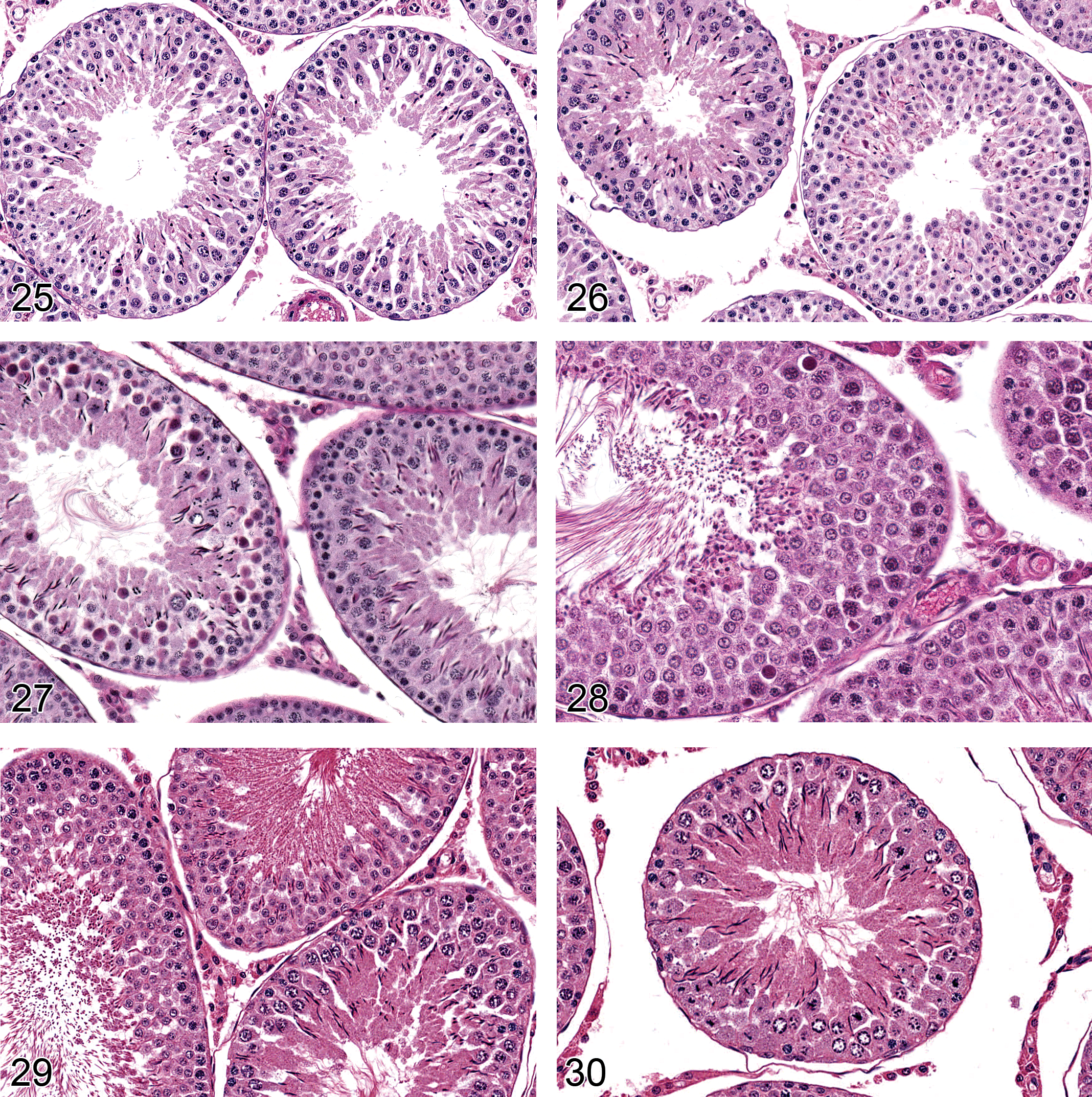
—Rat testis. Degeneration, germ cell, elongating spermatid. Stage XIV (left), stage XII (right).The heads are clubbed and condensed compared with normal.
Rat testis. Depletion, germ cells, spermatogonia, spermatocytes, spermatids up to step 6. Caused by maturation depletion of cells following dosing of a spermatogonial toxicant for 4 weeks.
Synonyms: apoptosis; single cell necrosis
Modifiers: spermatogonia, spermatocytes, round spermatids, elongating spermatids
Pathogenesis: Causes of cell-specific germ cell death may be multiple, including androgen deficiency, hypoxia, antimitotic agents, proapoptotic mediators (Yan et al. 2000), and cytokine inhibitors. Cell-specific germ cell death may also be mediated through disturbances in Sertoli cell functions that are critical to survival of a specific germ cell type
Diagnostic features Cytoplasmic eosinophilia/hyalinization/contraction Nuclear apoptotic bodies (generally spermatogonia) Chromatin condensation (generally spermatocytes) Chromatin margination (generally round spermatids) Clubbing and misshapen head (elongating spermatids) Phagocytosis of degenerate germ cells by Sertoli cells Subsequent germ cell absence/depletion Generally cell type–specific and stage-restricted
Differential diagnoses Background germ cell attrition: usually stage XII spermatogonia (occasionally other germ cells and stages are involved, e.g., stage XIV spermatocytes) in rat (Kerr 1992); limited numbers; present in control animals; more common among younger (peripubertal) animals Residual bodies: limited to stage VIII or IX (rat); no nucleus present; may appear at varying levels of the seminiferous epithelium during resorption by Sertoli cells Tubular degeneration/atrophy: not stage-specific; multiple germ cell types affected; in conjunction with multinucleated giant cells, vacuolation, disorganization
Comments: Although the term germ cell degeneration is recommended in this nomenclature system, most germ cell death (including chemical/pharmaceutical-induced changes, but excluding ischemic coagulative necrosis) occurs through triggering of programmed cell death (apoptotic) mechanisms (Boekelheide 2005; Boekelheide et al. 2000; Brinkworth et al. 1995; Shinoda et al. 1998). Unlike most other tissues, the apoptotic germ cells do not demonstrate the classic morphologic features familiar to pathologists. Therefore, the term degeneration has been recommended although it is not strictly accurate. Germ cell degeneration is morphologically transient and inconspicuous, due to rapid phagocytosis of affected cells by Sertoli cells. The term germ cell degeneration is most appropriately applied to compound-related changes observed in short duration studies (generally 28 days or less), referencing specific cell types and stages affected (when applicable) to provide insight into the mechanism of toxicity (Creasy 1997). The early effects of androgen deficiency are recognized by degeneration of round spermatids and pachytene spermatocytes in stage VII/VIII tubules of rats (Hikim, Leung, and Swerdloff 1995; Kerr et al. 1993; Russell et al. 1990). Mitotically active spermatogonia are affected by cytotoxic agents such as busulfan and bleomycin; pachytene spermatocytes by 2-methoxymethanol and dinitropyrroles; round spermatids by ethylmethane sulfonate and methyl chloride; and elongating spermatids by boric acid and dibromoacetic acid (Creasy 2001; Creasy and Foster 2002). Degeneration of occasional stage VII pachytene spermatocytes has been noted in young, food-restricted rats due to decreased testosterone (Rehm et al. 2008). Germ cell degeneration results in germ cell depletion (see below). These two findings may be evident concurrently, in which case the term germ cell degeneration/depletion is appropriate.
Depletion, Germ Cell (Figures 29–31): Testis
Modifiers: spermatogonia, spermatocytes, round spermatids, elongating spermatids
Pathogenesis: consequence of germ cell degeneration (see above)
Diagnostic features Partial or complete depletion of a single cell type (spermatogonia, spermatocytes, round spermatids, elongating spermatids)
or Partial or complete absence of two or three germ cell layers (spermatogonia, spermatocytes, round spermatids) but with the presence of more mature germ cell layers (round spermatids, elongating spermatids) Generally a diffuse change Concurrent germ cell degeneration may be evident but not prominent Mild decreases in testes weights possible (depending on numbers of cells lost)
Differential diagnosis Tubular degeneration/atrophy: not restricted to specific germ cell population/populations; disorganization of germ cells within affected tubules; often multifocal; often comprises tubules with complete absence of cells and others with partial absence as well as degenerating cells
Comments: Death of a specific target germ cell population, with subsequent progressive, duration-dependent loss of its descendant cells (maturation depletion) can cause depletion of multiple cell layers within the seminiferous epithelium. The type and number of germ cell populations lost (i.e., spermatogonia, spermatocytes, round spermatids, and/or elongating spermatids) will depend on the primary target population and the elapsed time between death of the affected cell and examination of the testis (Creasy 2001; Creasy and Foster 2002). As for “germ cell degeneration,” the germ cell type and/or the stage affected should be specified when the term germ cell depletion is used. This diagnosis should generally be reserved for studies of less than 1 month duration. When germ cell degeneration and germ cell depletion are both evident, the term germ cell degeneration/depletion is appropriate.
Retention, Spermatid (Figure 32): Testis
Synonym: delayed spermiation
Pathogenesis: functional disturbance in the process of spermiation, which may be due to abnormalities in the Sertoli cell or the mature spermatids or due to reduction in testosterone levels
Diagnostic features Persistence of the most mature elongating spermatids (step 16 mouse, step 19 rat) after the stage of physiologic release (stage VIII) Mature elongating spermatids present at the luminal surface of stages IX–XI tubules or present in the basal Sertoli cell cytoplasm (generally stage XII tubules), lying parallel to the basement membrane
Differential diagnosis Background retention of spermatids in a few tubules in maturing animals: present among control animals
Comments: Spermatid retention is a subtle but important change because it is frequently associated with abnormalities in sperm parameters (number, motility, and/or morphology) and may also be associated with decreased fertility. The change can only be detected by examining tubules in the appropriate stages (stages IX–XII in the rat). Occasional retained spermatids can be seen in normal rat testes; this finding should only be recorded when there is an obvious increase in the number of retained spermatids over control levels. The change can occur in isolation (e.g., boric acid, 2,5-hexanedione; Bryant et al. 2008) or can be one of many other degenerative changes (e.g., methylmethanesulphonate; Kuriyama et al. 2005). Because testosterone is an important regulator of spermatid maturation and spermiation, spermatid retention is also associated with androgen deficiency (Beardsley and O’Donnell 2003; Beardsley, Robertson, and O’Donnell 2006; D’Souza et al. 2009; Saito et al. 2000).
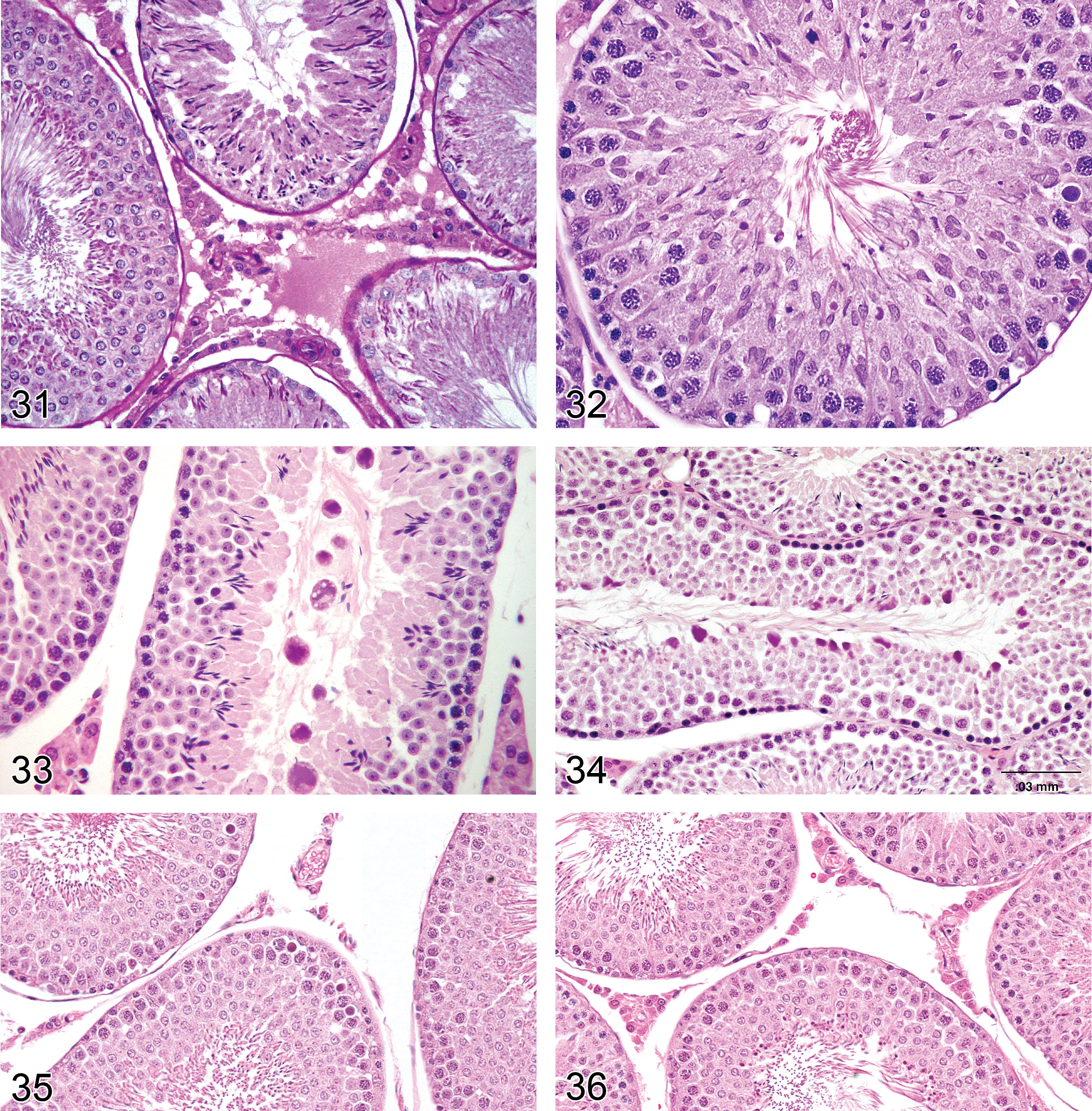
Residual Bodies, Atypical (Figures 33 and 34): Testis
Synonym: enlarged residual bodies
Pathogenesis: impaired maturation of elongating spermatids and/or processing of residual bodies by the Sertoli cell
Diagnostic features Abnormally large, misshapen, or clumped residual bodies Have the appearance of apoptotic like bodies Present at the luminal surface or resorbed into Sertoli cell cytoplasm (within seminiferous epithelium or basalar) May persist after stage XI May appear in the epididymal lumen
Differential diagnoses Luminal cellular debris: presence of nuclear material Germ cell degeneration: smoother, rounder, more homogeneous; within the seminiferous epithelium
Comments: Residual bodies represent the redundant cytoplasm and organelles discarded during maturation of elongating spermatids into spermatozoa. The cell debris is combined with lysosomes, mitochondria, and endoplasmic reticulum, and extruded in the form of membrane-bound residual bodies, which are shed from the mature spermatids at the time of spermiation (during stage VIII). These residual bodies are then phagocytized and transported into the basal Sertoli cell cytoplasm during the following stages (stages IX–XII) at which point they disappear due to phagocytosis. Abnormal residual bodies result from impaired Sertoli cell processing of these cytoplasmic remnants. They are generally larger than normal and present in stages where they are not normally seen (e.g., present in early stage tubules where they should never normally be seen). Abnormal residual bodies are a frequent background finding in mice but can also be a treatment-related finding in rats and mice. They have been described following administration of the water-disinfecting chemical dibromoacetic acid in rats (Linder et al. 1994, 1997) and mice.
Leydig Cell Changes: Testis
Vacuolation, Leydig Cell: Testis
Pathogenesis: probably due to disturbance in steroidogenesis
Diagnostic feature Leydig cells with pale vacuolated cytoplasm
Differential diagnoses Vacuolation, macrophage: macrophages stain Periodic acid Schiff (PAS) positive; Leydig cells are negative
Comments: Leydig cells of mice normally have a vacuolated appearance to their cytoplasm whereas rat Leydig cells normally have a dense eosinophilic cytoplasm. Testosterone is not stored in the Leydig cell; it is secreted as soon as it is produced. Although rare, cytoplasmic vacuolation of the Leydig cell can be seen as a test article–related change. Most likely it represents a disturbance in steroidogenesis.
Atrophy, Leydig Cell (Figures 35–36): Testis
Synonym: decreased size/number of Leydig (interstitial) cells
Pathogenesis: decreased steroidogenesis in the Leydig cell due to enzyme inhibition, decreased functional demand or reduced stimulation
Diagnostic feature Decreased number and/or size of Leydig cells
Comments: Leydig cell atrophy is detectable morphologically only when steroidogenesis has been severely decreased (Keeney et al. 1988). The resulting androgen deficiency causes decreased size and weight of the accessory sex glands and epididymides, which are androgen-dependent tissues (Creasy 2001). There may also be hypertrophy of gonadotropin-secreting cells in the pituitary and atrophy of the male mammary gland (Creasy 2008). Leydig cell atrophy will normally be accompanied by decreased spermatogenesis (depletion of elongating spermatids, stage VII/VIII germ cell degeneration, and spermatid retention). Decreased steroidogenesis may be caused by direct inhibition of steroid biosynthesis or via reduced stimulation from the hypothalamic pituitary gonadal axis. Estrogen administration, high doses of testosterone, or luteinizing hormone suppression can also result in Leydig cell atrophy (Greaves 2012a).
Necrosis, Leydig Cell: Testis
Pathogenesis: necrosis of the Leydig (interstitial) cell, reported as a chemically induced change (Jackson et al. 1986) or in association with ischemic necrosis of the testis
Diagnostic features Chromatin clumping and margination in Leydig cell nuclei Phagocytosis of Leydig cell debris by testicular macrophages Absence of Leydig cells
Comments: Necrosis of Leydig cells has been described following administration of the alkylating agent ethane dimethane sulphonate. The time sequence of Leydig cell necrosis has been described (Bartlett, Kerr, and Sharpe 1986; Jackson et al. 1986; Molenaar et al. 1985).
Miscellaneous Changes: Testis
Amyloid (Figure 37): Testis
Pathogenesis: age-related degenerative condition,
Diagnostic features Accumulation of pale eosinophilic extracellular material Perivascular, peritubular, or interstitial Green birefringence using polarized light with Congo Red stain May form thin bands or extensive sheets
Comments: Amyloid deposition is an age-related, incidental, spontaneous, often systemic disease characterized by the extracellular deposition of polypeptides (often serum amyloid-associated protein or immunoglobulin fragments) appearing in routine section as a lightly eosinophilic amorphous material. It is common in aged mice, and rare in rats. Confirmation of the deposits as amyloid can be accomplished with light microscopy using special stains such as Congo Red. Amyloid appears apple green under polarized light with this stain. Amyloid deposition in the interstitium of the testis may lead to atrophy of the Leydig cells.
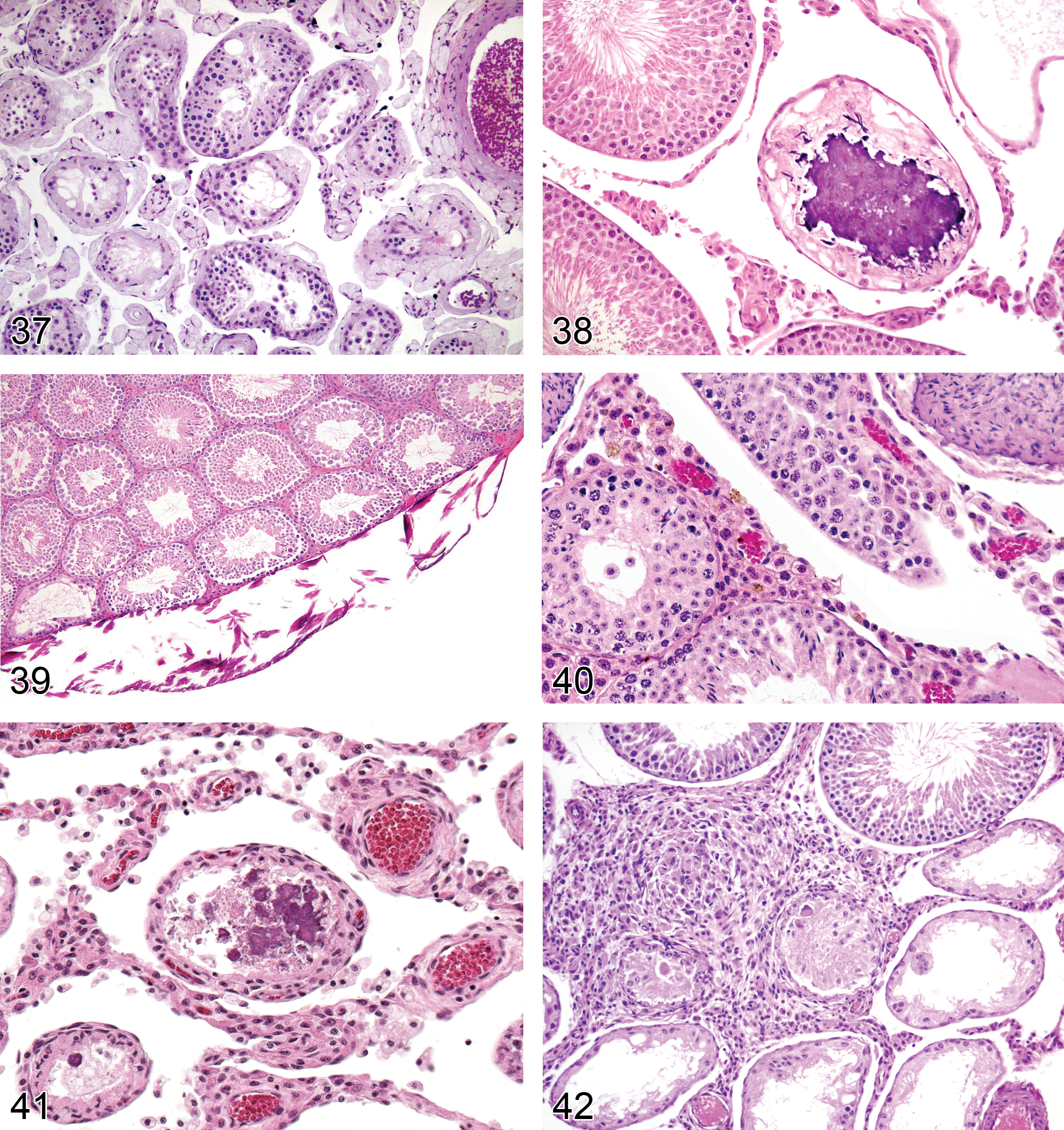
Mouse testis. Amyloid.
Fibrosis: Testis
Pathogenesis: collagen deposition by fibroblasts following inflammation, necrosis, or hemorrhage
Diagnostic features Interstitial, perivascular, and/or peritubular tissue replaced by collagen Seminiferous tubular diameter may be decreased and/or profiles distorted Testis diameter may be decreased Testis shape may be distorted
Comments: Fibrosis is usually secondary to inflammatory or degenerative processes associated with disruption of blood supply or damage to the blood–testis barrier. Fibrosis is induced by chronic cocaine or cadmium administration to rats (Barroso-Moguel, Méndez-Armenta, and Villeda-Hernàndez 1994; Bomhard, Vogel, and Loser 1987; Gouveia 1988; Jana and Samanta 2006).
Mineralization (Figures 38 and 39): Testis
Pathogenesis: deposition of mineral within degenerate tissue (dystrophic calcification)
Diagnostic features Basophilic amorphous or lamellar deposits May involve seminiferous tubular basement membranes, tubular epithelium, impacted sperm, tunica albuginea
Comments: Calcium salts are frequently deposited in areas of sperm stasis. If Bouin’s fixative is used, the mineral will be partially or totally disolved and may not be apparent.
Pigment (Figure 40): Testis
Synonyms: hemosiderosis, lipofuscinosis
Pathogenesis: accumulation of pigmented material with age (lipofuscin) or following hemorrhage (hemosiderin)
Diagnostic feature Cytoplasmic accumulation of yellow-brown material within Sertoli cells; Leydig cells; macrophages
Differential diagnosis Artifactual formalin pigment (acid hematin): extracellular, black
Comments: Lipofuscin pigment is the remnant of breakdown products of lipid oxidation and is often seen in aging rats and mice (Giannessi et al. 2005). Lipofuscin is positive for PAS and Schmorl reactions. Accumulation of hemosiderin pigment can be found at all ages as a consequence of hemoglobin breakdown following hemorrhage. The iron in hemosiderin stains positive with a Perl’s stain.
Rat testis. Inflammation, neutrophilic (associated with testicular necrosis).
Vacuolation, Macrophage (Figure 41): Testis
Pathogenesis: generally due to phospholipidosis
Diagnostic feature Macrophages with foamy cytoplasm, located within the interstitium
Differential diagnosis Vacuolation, Leydig cell: macrophages stain PAS positive; Leydig cells are negative.
Comments: Although interstitial macrophages form approximately 25% of the interstitial cells, they are generally difficult to distinguish from Leydig cells in normal testes. They can be distinguished using a PAS stain (Leydig cells are PAS negative and macrophages are positive). In some cases of drug-induced phospholipidosis, their cytoplasm becomes vacuolated, with a similar appearance to foamy histiocytes in other locations. It is important not to mistake this as Leydig cell vacuolation.
Inflammatory Changes: Testis
Inflammation (Figures 42 and 43): Testis
Synonym: inflammatory cell infiltrate
Modifiers: neutrophilic, lymphocytic, mixed cell, granulomatous
Pathogenesis: migration of leukocytes into extravascular spaces in response to foreign body, organisms, or necrosis
Diagnostic features Inflammatory cell infiltrate of variable composition Neutrophilic, e.g., when responding to early tubular necrosis Granulomatous, e.g., in response to foreign body (sperm) Lymphocytic, e.g., in response to autoimmune conditions May be accompanied by edema and/or hemorrhage Infiltration of the peritubular myoid cell layer of affected tubules May destroy and replace tubules
Comments: Uncommon, due to the immunologically protected nature of the seminiferous tubules. The presence of inflammatory cells generally indicates that the tight junctions between Sertoli cells have been breached and/or that Sertoli cells have been seriously damaged by a necrotic process. Most cases of tubular degeneration/atrophy, whether incidental or treatment related, will not incite an inflammatory response. An exception to this has been described with administration of di-n-pentyl phthalate to rats where a transient neutrophilic infiltrate occurs early in development of Sertoli cell changes and then disappears (Creasy, Foster, and Foster 1983).
Sperm Granuloma (Figure 44): Testis
Pathogenesis: Foreign body reaction due to access of immunologically competent cells to antigenically foreign sperm. Generally caused by disruption of the blood–tubular barrier and/or loss of tubular/rete integrity with sperm stasis
Diagnostic features Granulomatous inflammation surrounding a central core of aggregated sperm Expansion of affected tubule or rete with variable degrees of disruption and rupture Epithelioid macrophages and foreign body giant cells Possible peripheral fibrosis
Differential diagnosis Spermatocele: expansion of tubule to two times the normal diameter; no inflammation
Comments: Usually present in the rete testis and usually an incidental change, but it may be secondary to chemically induced obstruction of the efferent ducts. Sperm granulomas are much more common in the epididymis.
Vascular Changes: Testis
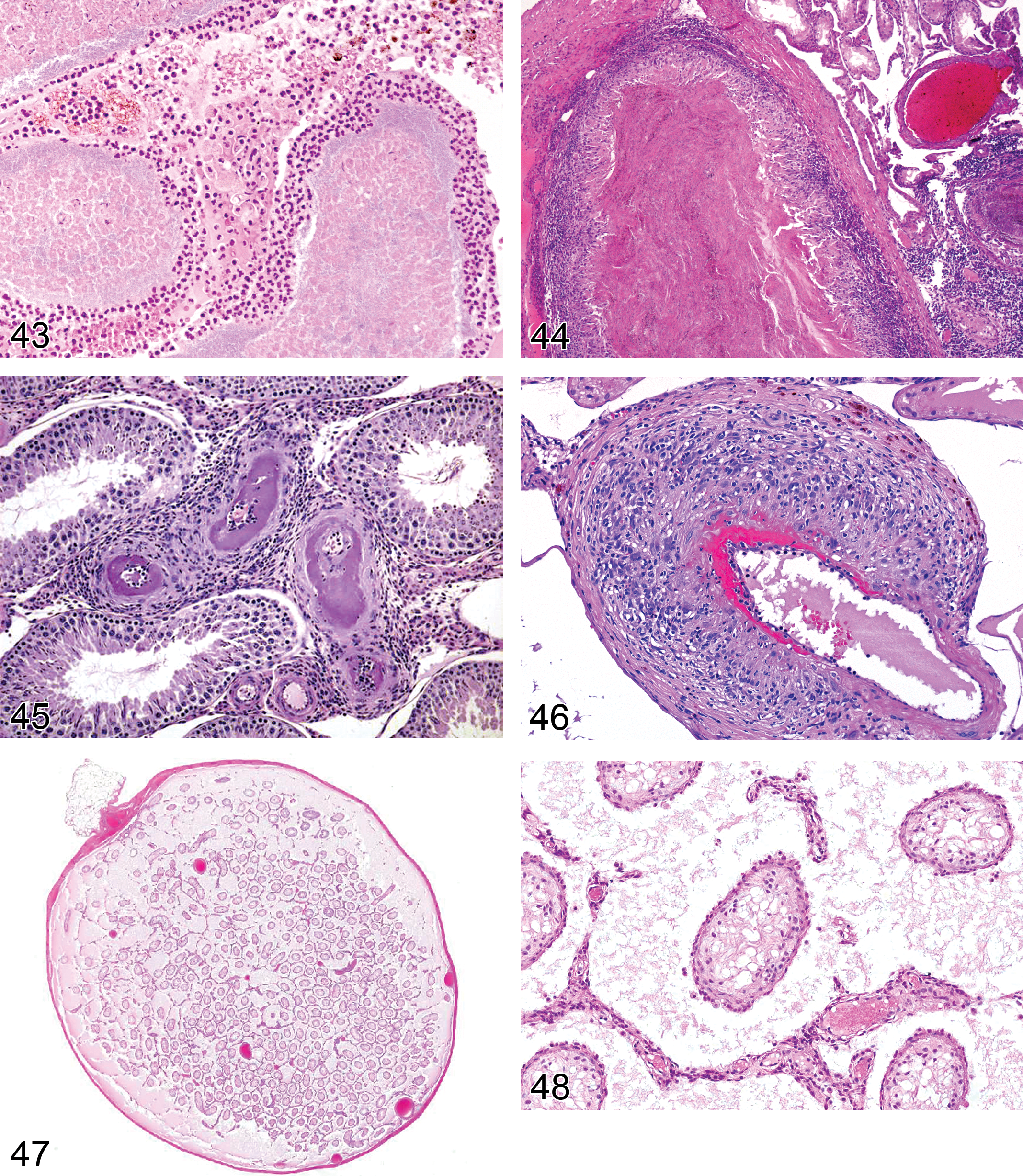
Necrosis/Inflammation, Vascular/Perivascular (Figures 45 and 46): Testis
Synonyms: vasculitis, arteritis, perivascular inflammation, periarteritis, polyarteritis nodosa
Pathogenesis: Generally a feature of spontaneous, age-related, systemic “polyarteritis nodosa,” for which the testis is a common site. It may also be caused by xenobiotics and hypertension
Diagnostic features Loss of smooth muscle cell nuclei Karyorrhectic debris Fragmentation of smooth muscle cells Expansion of the tunica media by hyaline eosinophilic amorphous material (fibrinoid change) May be accompanied by hemorrhage and/or inflammatory cell infiltrate
Comments: Spontaneous, age-related necrotizing arteriopathy is a common age-related lesion involving medium-sized vessels in many tissues, but the testis is a particularly common site for this change (Creasy 2012). The incidence can be affected by levels of fat and protein in mice, and food restriction reduces the incidences in rats (Greaves 2012b). However, a variety of procedures and pharmacologic agents can also initiate or exacerbate it. For a detailed discussion of this, see the INHAND nomenclature for the Cardiovascular System. Inflammation in or around the arteries in the testis is usually a reflection of systemic vascular disease such as systemic hypertension or immune complex deposition. In rats, the most commonly affected arteries in both spontaneous and induced lesions are the small muscular arteries of the mesentery, pancreas, and testis; in the mouse, renal vessels are frequently involved (Greaves 2012b; Mitsumori 1990). Lesions are prevalent in strains that develop spontaneous hypertension (Fawn-Hooded rat and stroke-prone spontaneously hypertensive rats; Saito and Kawamura 1999) in repeat breeder males and in rats with experimentally induced hypertension (Akagashi et al. 1996). Nitrofurantoin caused above-background incidence of perivascular inflammation in the rat testis (Mitsumori 1990).
Edema (Figures 47–49): Testis
—Rat testis. Fixation artifact caused by hypertonic fixative. Presence of proteinaceous interstitial fluid surrounding normal tubules in the center of a testis.
Pathogenesis: increased vascular permeability
Diagnostic features Increased eosinophilic fluid (interstitial fluid) within interstitium Generally accompanied by severe tubular atrophy (see comment below) or evidence of inflammation
Differential diagnosis Fixation artifact: commonly seen when testes are fixed in Bouin’s fluid, and less so with Modified Davidson's fluid. When due to fixation, the eosinophilic fluid is generally present surrounding normal appearing, contracted tubules and is characteristically more prominent in the center of the testis
Comments: It is important not to misinterpret artifactual change as edema in the testis. Accumulation of interstitial fluid (which is modified lymph) commonly occurs as a result of fixing the testes in hyperosmotic fixatives such as Bouin’s and Modified Davidson’s which cause tubular contraction and postmortem diffusion of proteinaceous fluid into the interstitial space surrounding the contracted tubules. When due to fixation, the fluid accumulation is typically more prominent in the center of the testis (Latendresse et al. 2002). In some cases of severe tubular atrophy, there appears to be a true edema, which likely existed ante mortem, but most accumulation of proteinaceous fluid in the interstitium is probably a postmortem fixation artifact. A potential way to distinguish real from artifactual edema is to examine the testis weight. If the edema was present prior to fixation and is present in association with normal seminiferous tubules, there should be an increase in testis weight.
Angiectasis (Figure 50): Testis
Pathogenesis: focal dilation of blood vessels
Diagnostic features Increased vascular diameter Possible compression of adjacent structures
Differential diagnoses Congestion: lacks compression of adjacent tissue Hemangioma: Increased number of vascular channels lined by hyperplastic endothelium. Disturbance of normal testicular architecture. Compression of adjacent structures
Comments: Uncommon background lesion seen in aged rats and mice.
Non-neoplastic Proliferative Lesions: Testis
Introduction
With the exception of Leydig cell hyperplasia and Leydig cell tumors, very few non-neoplastic or neoplastic proliferative lesions are seen in the rodent testis. Leydig cell hyperplasia can occur as a focal or diffuse lesion. Focal Leydig cell hyperplasia and Leydig cell adenomas form a continuum of change, making it difficult to separate hyperplasia and adenoma. In the absence of any significant morphological differences in cellular appearance, size is generally used as the main but arbitrary classification criterion. Diffuse Leydig cell hyperplasia is generally a physiological response to hormone imbalance. One other relatively common, non-neoplastic proliferative lesion in the mouse is hyperplasia of the rete testis epithelium, which is a common age-related finding.
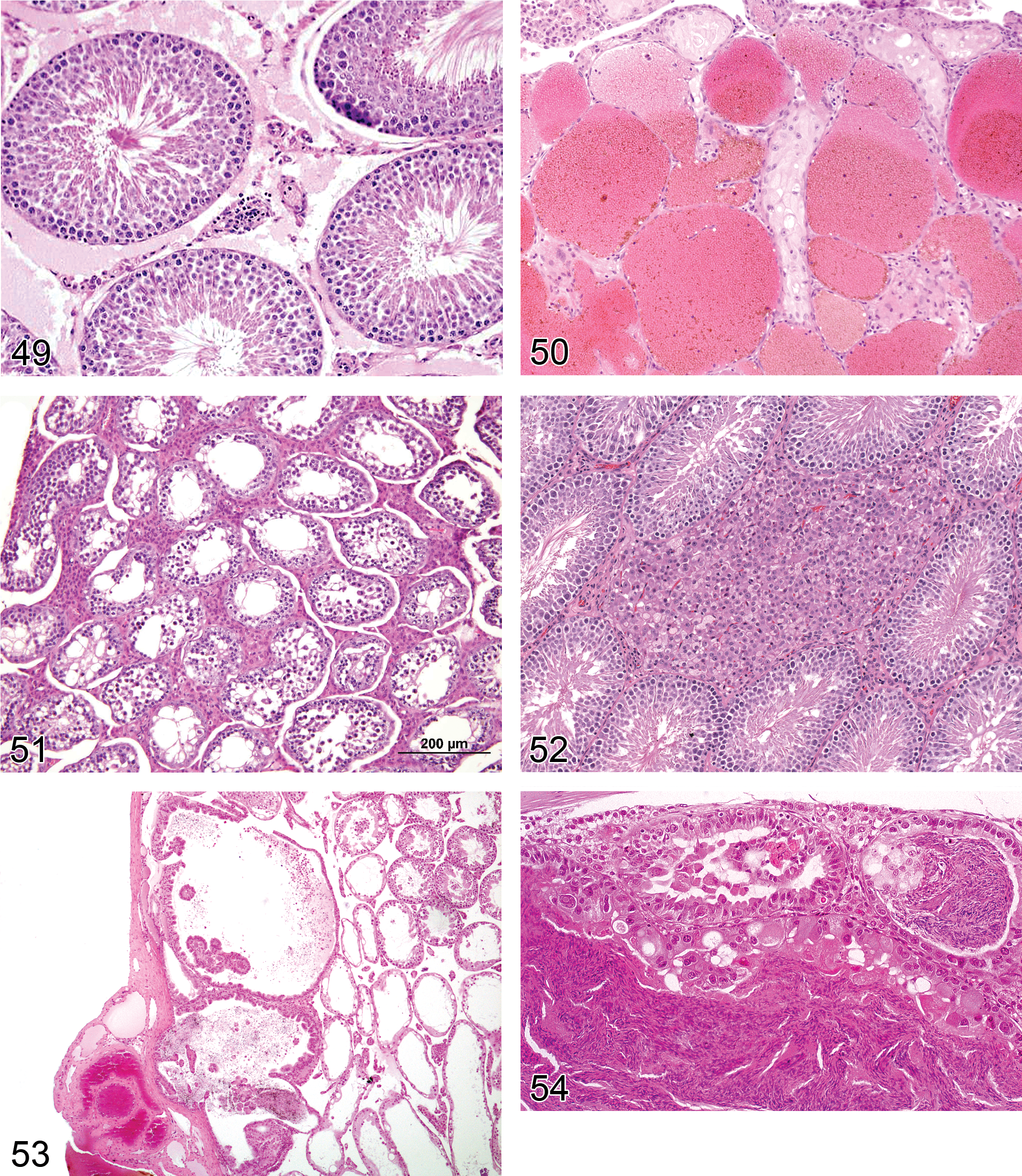
Hyperplasia, Leydig Cell (Figures 51 and 52): Testis
Species: mouse, rat
Synonyms: hyperplasia, interstitial cell
Pathogenesis: response to increased levels of luteinizing hormone from the pituitary or to release of stimulatory paracrine factors within the testis; compensatory response to decreased spermatogenesis
Diagnostic features Focal, multifocal, or diffuse, solid collections of Leydig cells between seminiferous tubules Cells with central nucleus and often prominent nucleolus, abundant eosinophilic cytoplasm, sometimes vacuolated Focal/multifocal Angular or rounded Leydig cell aggregation between seminiferous tubules No or only minimal compression of the surrounding tissue Diameter is smaller than or equal to three seminiferous tubules Diffuse Bridging strands of Leydig cells several layers thick between seminiferous tubules Lesion may be locally extensive involving a large portion of the testis Hyperplastic foci may also be present
Differential diagnoses Inflammation, granulomatous: presence of macrophages may resemble vacuolated Leydig cells but are admixed with other inflammatory cells Adenoma, Leydig cell: usually compression of adjacent tubules, cells and nuclei generally larger and rounder, may have some nuclear atypia. If differentiation between focal hyperplasia and adenoma cannot be made on the basis of these criteria, a proliferative lesion with a diameter larger than three seminiferous tubules is interpreted to be an adenoma Relative increase in Leydig cells: concurrent decreased tubular diameter
Comments: In mice, normal Leydig cells are larger and more numerous than in rats. In mice, Leydig cell hyperplasia has characteristically a diffuse distribution pattern and is often most prominent in the subcapsular region, while in rats the pattern is generally focal or multifocal. The diagnostic criteria for distinguishing Leydig cell hyperplasia from normal and for separating Leydig cell hyperplasia from Leydig cell adenoma are generally arbitrary, since most Leydig cell tumors begin as focal hyperplasia and represent a continuous spectrum from small collections of hyperplastic cells to large tumors. Care should be taken when diagnosing hyperplasia when associated with atrophic tubules since a decreased tubular volume may cause the impression of a higher density of Leydig cells. Since 1992, the Society of Toxicologic Pathologists (USA) recognized three tubules as the borderline between hyperplasia and adenoma (McConnell et al. 1992). Morphometric investigations of proliferative Leydig cell changes in Wistar rats have confirmed that Leydig cell hyperplasias smaller than 3 normal seminiferous tubules, and benign Leydig cell tumors larger than 3 normal seminiferous tubules differ cytologically: nuclei of hyperplastic Leydig cells are significantly smaller, more oval, and have more indentations. The larger neoplastic nuclei generally contain twice the amount of DNA found in normal or hyperplastic Leydig cells (Ettlin et al. 1992). The definition of criteria for differentiation between “normal” and Leydig cell hyperplasia may be useful. In general (for both rats and mice), focal Leydig cell accumulations with a diameter equal to or more than half of an average seminiferous tubule are considered focal hyperplasia. However, in the context of a given study and for a specific strain, this limit may be lower, or other criteria (e.g., demarcation from surrounding interstitial tissue) may be more appropriate. Leydig cell hyperplasia and neoplasia are particularly common in F344 rats but are less frequent and less severe in Wistar and Sprague-Dawley rats. Strain differences are also evident in mice, where Leydig cell hyperplasia and neoplasia are particularly common in some strains (e.g., NMRI) but less common in others. Leydig cell hyperplasia has been observed in mice with testicular feminization and associated with treatment by estrogenic compounds or 5α-reductase inhibitors. Granulomatous inflammation with many macrophages may resemble vacuolated Leydig cells. Leydig cell hyperplasia has been described in Boorman et al. 1987a; Boorman, Chapin, and Mitsumori 1990; Faccini, Abbott, and Paulus 1990; Frith and Ward 1988; Gordon, Majka, and Boorman 1996; Mitsumori and Elwell 1988; Prahalada et al. 1994; Rao and Reddy 1987; Reddy and Rao 1987; Rehm et al. 2001.
Hyperplasia, Rete Testis (Figures 53 and 54): Testis
Species: mouse (predominantly), rat (rare)
Pathogenesis: spontaneous, age-related lesion and can also be induced by xenobiotics
Diagnostic features Focal or multifocal lesion in the mediastinum testis No compression of adjacent structures Architecture is maintained: tubules are lined by a single layer of epithelial cells, focal crowding of up to three cell layers may occur Papillary knob- or finger-like projections into the lumen are frequently present in mice; cross sections of these projections may resemble rosettes. These projections grow with scant stroma. Cells are flattened to cuboidal in rats but tall columnar with abundant eosinophilic cytoplasm in mice Nuclei are round to oval vesicular with a single nucleolus Mitotic figures are rare Cystic structures filled with spermatozoa may be present
Differential diagnoses Adenoma, rete testis: extensive papillary structures with supportive stroma are present. Compression of adjacent tissue due to increased tissue mass is present Carcinoma, rete testis: presence of malignant characteristics such as invasion, atypia, high mitotic rate, and hemorrhage.
Comments: Hyperplasia of the rete testis does not lead to compression of the adjacent tissue by itself. However, an associated dilatation of rete testis tubules may compress adjacent structures. The epithelium is negative for vimentin, but may produce mucins, i.e., may be PAS and/or Alcian blue positive. Proliferative lesions of the rete testis are rare spontaneous lesions in aging rats but are more common in CD-1 mice. Proliferative lesions of the rete testis have been reported to occur following prenatal treatment with diethylstilbestrol (Bullock, Newbold, and McLachlan 1988; Newbold et al. 1985). Hyperplasia of the rete testis has been described by Alison et al. (1997); Boorman, Chapin, and Mitsumori (1990); Boorman, Eustis, and Elwell (1990); Frith and Ward (1988); Gordon, Majka, and Boorman (1996); Maekawa and Hayashi (1987); Mitsumori and Elwell (1988); Rehm et al. (2001); and Yoshitomi and Morii (1984).
Hyperplasia, Mesothelium (also see “soft tissue” nomenclature manuscript)
Species: rat
Pathogenesis: unknown
Diagnostic features Usually localized Focal thickening or villous projections covered by cuboidal cells with little or no stratification Lacks evidence of mitotic activity or cellular atypia May possess a small fibrovascular core or stalk Fibrosis or inflammation may accompany the changes
Differential diagnosis Malignant mesothelioma is highly cellular and shows extensive spread or infiltrates adjacent tissues Epithelioid malignant mesothelioma exhibits poorly formed glandular structures or ill-formed glands. Sarcomatoid malignant mesothelioma consists of spindle-shaped cells
Comment: These are often small incidental lesions seen on the tunica vaginalis (McConnell et al. 1992). Also see “Soft Tissue” nomenclature manuscript.
Neoplastic Proliferative Lesions: Testis
Introduction
With the exception of Leydig cell tumors, proliferative lesions of the testis and epididymis are uncommon or rare in rodents. Leydig cell tumors occur as an age-related tumor in rodents and are more common in the rat than the mouse, but there are significant inter-strain differences in incidence. In the Fischer 344 rat, the incidence of Leydig cell tumors approaches 100% in 18- to 24-month-old animals whereas it is generally <2% in Sprague–Dawley IGS and Hannover Wistar derived strains. In some other strains of Wistar rat, the incidence can reach 40%, but the source (breeder) has been identified as the main reason for the marked differences. In mice, the background incidence of Leydig cell tumors in the commonly used strains is <1% in B6C3F1 and <2% in CD1 mice. They can be readily induced in the rat by any treatment that causes luteinizing hormone (LH) levels to increase and induced in mice by hyperestrogenism (Clegg et al. 1997; Cook et al. 1999). Tumors of the rete testis are also seen at a low incidence in aging mice but are rare in rats.
Rete testis adenomas and carcinomas may also occasionally be seen in the mouse and can be chemically induced. Although not strictly arising from the testicular parenchyma, mesothelioma is a tumor that preferentially develops on the tunica vaginalis of the rat testis. It is particularly common in the Fischer 344 strain but has not been described in the mouse.
Other proliferative lesions of rodent testes have only rarely been observed. Some transgenic mouse models may develop unusual proliferative lesions at a relatively young age and high incidence, but these have not been included here.
The terminology and diagnostic criteria provided for the testicular tumors is largely based on those previously published by World Health Organization (WHO)/International Agency for Research on Cancer (IARC) and Standardized System of Nomenclature and Diagnostic Criteria (SSNDC) (Alison et al. 1997; McConnell et al. 1992; Mostofi and Bresler 1976; Mostofi, Davis, and Rehm 1994; Rehm et al. 2001). There are numerous additional reviews of testicular tumors in rodents, detailing their general features and incidence (Boorman, Chapin, and Mitsumori 1990a; Boorman, Eustis, and Elwell 1990c; Faccini, Abbott, and Paulus 1990; Frith and Ward 1988; Gordon, Majka, and Boorman 1996; Maekawa and Hayashi 1992; Mitsumori and Elwell 1988; Radovsky, Mitsumori, and Chapin 1999; Rehm et al. 2001; Squire et al. 1978).
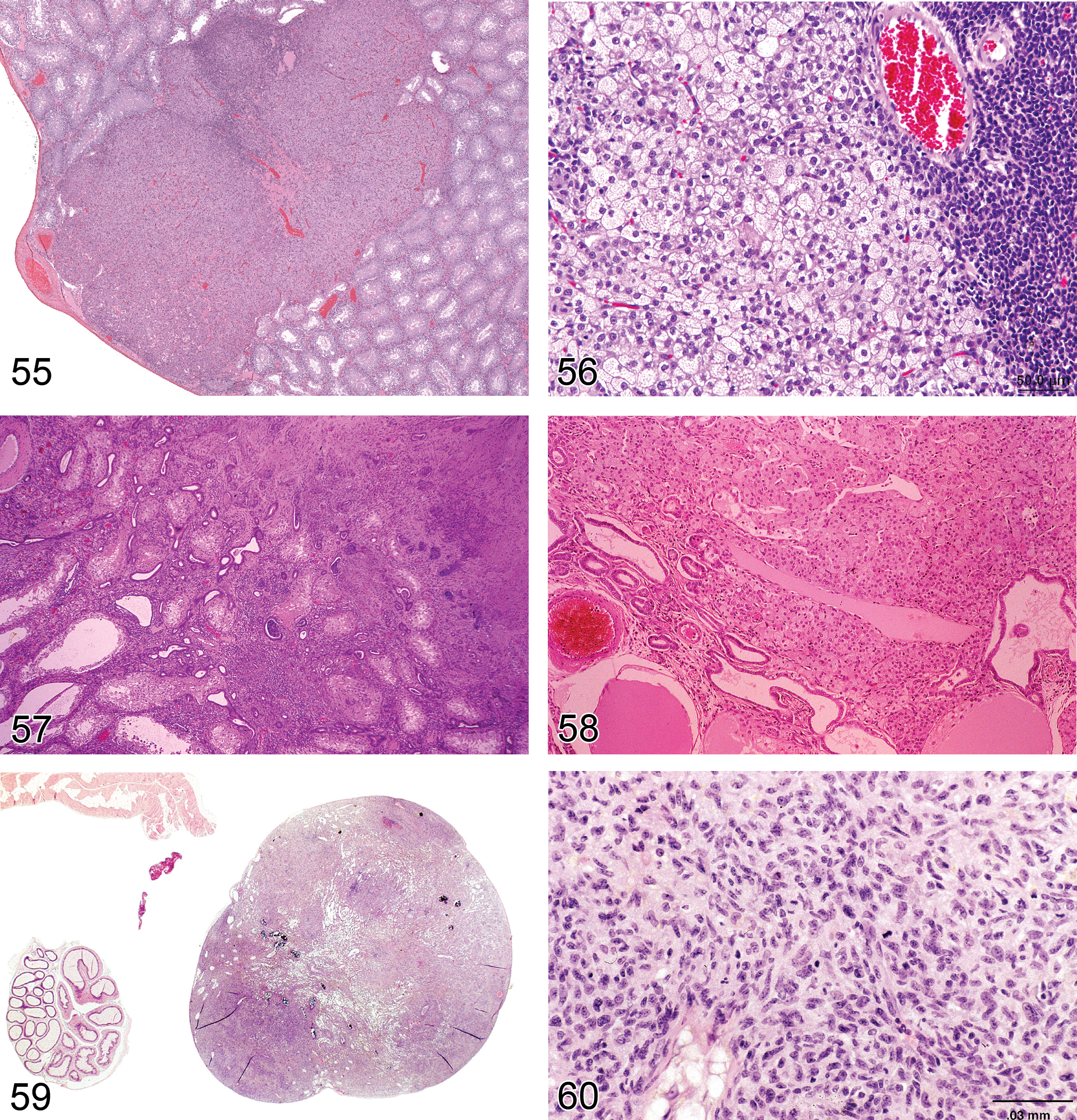
Adenoma, Leydig Cell (Figures 55–58): Testis
Species: mouse, rat
Synonyms: tumor, Leydig cell, benign; interstitial cell tumor, benign; interstitial cell adenoma
Pathogenesis: common spontaneous tumor, also common response to xenobiotics causing sustained increase in circulating LH levels in the rat or to estrogenic compounds in the mouse
Modifier: retiform (in rat)
Diagnostic features Mass with often circumferential peripheral compression of adjacent seminiferous tubules (may be unilateral or bilateral) Composed predominantly of uniform polyhedral cells with abundant eosinophilic, finely granular, or vacuolated cytoplasm Nucleus generally central, round with evenly distributed chromatin and single prominent nucleolus. Low nucleus to cytoplasm ratio. Polyploidy and larger nuclei may be found Large tumors may have areas of less well-differentiated basophilic cells with scanty cytoplasm or elongated spindle-shaped cells Mitotic rate usually low; no atypical mitotic figures Some tumors moderately vascular, containing dilated, thin-walled vessels and hemorrhage; focal areas of necrosis occasionally present Cystic areas contain proteinaceous material or blood Stroma usually scanty, areas of hyalinization and fibrosis of entrapped seminiferous tubules may be present Generally not encapsulated; adjacent seminiferous tubules often show variable degrees of atrophy If differentiation between focal hyperplasia and adenoma cannot be made on the basis of criteria listed above, a proliferative lesion with a diameter larger than three seminiferous tubules is interpreted to be an adenoma Retiform pattern (rat): Leydig cell tumor with embedded areas of glandular/tubular structures, lined by cuboidal to columnar cells with Alcian blue-positive brush borders, occasionally filled with PAS-positive substance
Differential diagnoses Hyperplasia, Leydig cell: usually no compression of adjacent seminiferous tubules, nuclei smaller (6–7 µm in diameter) with more infoldings. Diameter is smaller than or equal to three seminiferous tubules Carcinoma, Leydig cell: polymorphism, cellular atypia, and invasion of adjacent tissue (vessels, capsule) or formation of metastases. Hyperplasia or adenoma, Rete testis: glandular/tubular structures form the entire lesion, rather than being embedded within the Leydig cell mass Tumor, mixed Sertoli–Leydig cell, benign: features of both Leydig and spindle-shaped Sertoli cells forming tubules Tumor, granulosa cell, benign: composed of follicle-like nests or cords filled with small round cells with scant cytoplasm. Seminoma, malignant: large, deeply eosinophilic or basophilic cells with distinct cell boundaries, high nucleus to cytoplasm ratio, intratubular growth and atypical mitotic figures commonly present
Comments: The diagnosis of a Leydig cell adenoma versus focal hyperplasia is mainly based on the size of the Leydig cell mass exceeding three normal seminiferous tubules. Two or more distinct neoplastic nodules separated by tubular tissue are recorded as multifocal. Tubule-forming varieties of Leydig cell tumors have been observed in control Fischer 344 rats (Kanno et al. 1987) and in Wistar rats treated with a prolactin inhibitor (Qureshi et al. 1991). Serial sections showed no relationship with the rete testis. Immunohistochemical investigations suggest that the tubules are formed by metaplastic Leydig cells. Leydig cell tubules have to be differentiated from hyperplasia and adenoma of the rete testis (Maekawa and Hayashi 1987; Rehm and Waalkes 1988). The spontaneous incidence of Leydig cell adenoma in rats seems to be inversely correlated to the body weight (Nolte et al. 2010). Leydig cell hyperplasia and Leydig cell adenoma are readily induced in the rat by many xenobiotics from different chemical and therapeutic classes. In almost all cases, the underlying common mechanism appears to be through increasing LH stimulation of the Leydig cell (Clegg et al. 1997; Cook et al. 1999). Due to major differences between rodents and humans with respect to prevalence of different testicular tumor types, hormonal physiology and response and risk factors for Leydig cell tumors, chemical induction of Leydig cell tumors in rats is generally considered of limited relevance to humans (Alison, Capen, and Prentice 1994; Clegg et al. 1997; Cook et al. 1999). However, the mode of action of LH increase may be of toxicological relevance to humans. In the case of mice, the major risk factor for Leydig cell tumors appears to be hyperestrogenism (Huseby 1976, 1980; Juriansz, Huseby, and Wilcox 1988). This is also considered to have limited relevance to man (Clegg et al. 1997; Cook et al. 1999). Finasteride, a 5α- reductase inhibitor, has also been shown to produce Leydig cell tumors in mice (Prahalada et al. 1994; Zwieten 1994). Various aspects of Leydig cell adenoma have also been described by Ettlin et al. (1992); Qureshi et al. (1991); and Rao and Reddy (1987).
—Rat testis. Adenoma, Leydig cell.
Carcinoma, Leydig Cell (Figures 59 and 60): Testis
Species: mouse, rat
Synonyms: tumor, Leydig cell, malignant; interstitial cell tumor, malignant
Pathogenesis: generally a progression from Leydig cell adenoma. Usually caused by sustained increase in LH levels in the rat or estrogenic compounds in mice
Diagnostic features Mass showing invasion into the capsule or adjacent tissue or distant metastases Cellular pleomorphism is generally present such as poorly differentiated basophilic cells or spindle-shaped cells with scanty cytoplasm Mitoses are infrequent; mitotic figures are sometimes atypical Entrapped seminiferous tubules often seen Areas of necrosis and/or hemorrhage often present
Differential diagnoses Adenoma, Leydig cell: no invasion of adjacent tissues or metastases Seminoma, malignant: cells are large, clear, or eosinophilic with distinct cell boundaries and have a high nucleus/cytoplasmic ratio Tumor, granulosa cell, benign: composed of follicle-like nests or cords filled with small round cells with scant cytoplasm. Lack invasion. Due to the rarity of both granulosa cell tumors and Leydig cell carcinomas composed of small cells with scant cytoplasm, distinguishing features have not been delineated.
Comments: Invasion into the testicular tunica, the surrounding tissue or the spermatic cord, or metastases are the most important criteria for the distinction between carcinoma and adenoma. In man and dog, infiltration of the testicular tunica albuginea by Leydig cells can occur without neoplastic transformation. Identification of invasion into blood vessels and lymphatics is problematic. False positive diagnoses are easily made because of the blood and protein-filled spaces, which occur in Leydig cell tumors. Leydig cell carcinomas have been reported in CD-1 mice exposed to diethylstilbestrol
Adenoma, Rete Testis (Figures 61 and 62): Testis
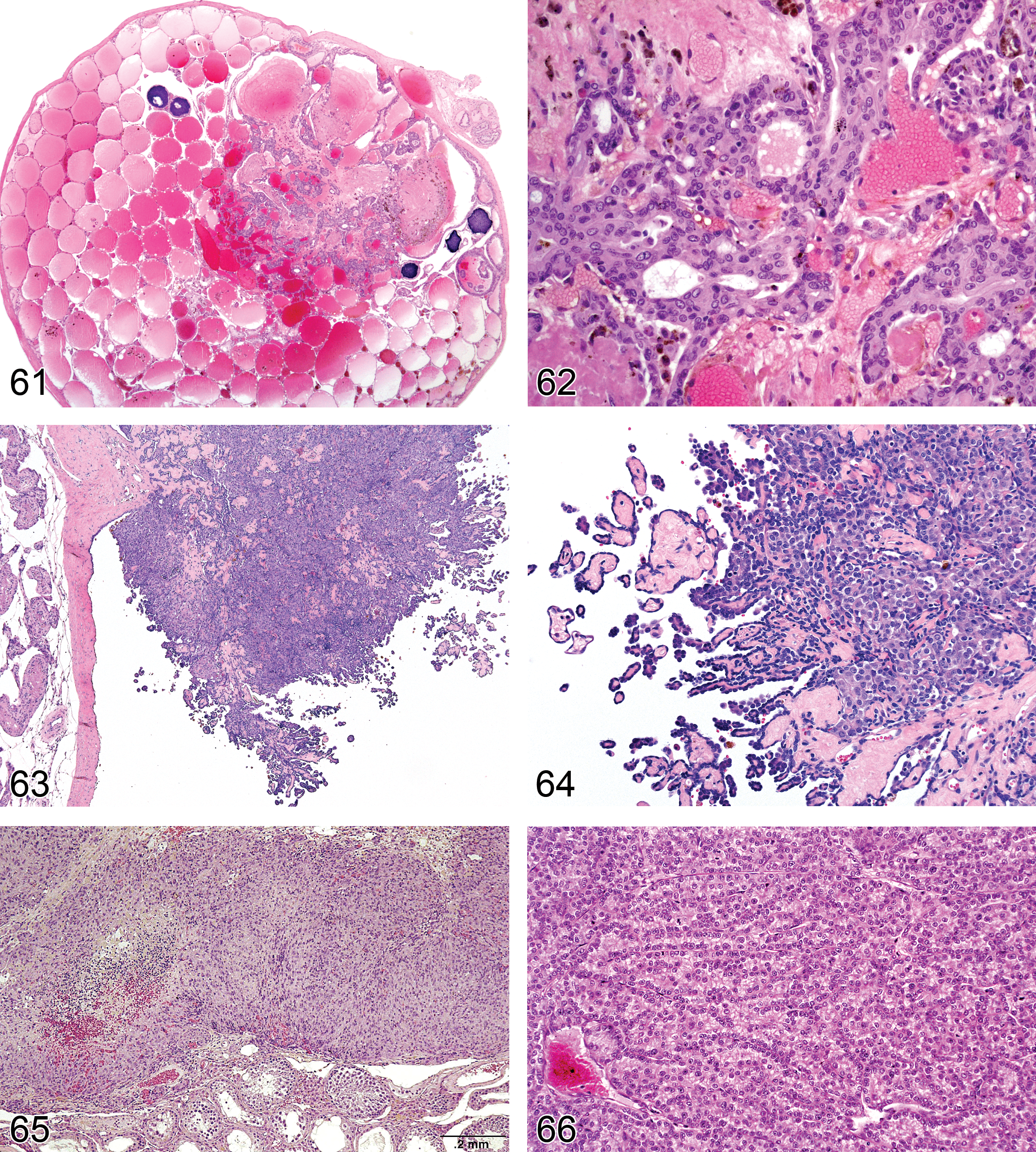
—Mouse testis. Adenoma, rete testis.
Species: mouse, rat
Pathogenesis: spontaneous, age-related lesion and can be induced by xenobiotics
Diagnostic features Tubulo-papillary tumor of rete testis structures localized in the mediastinum testis Shows compression of adjacent tissue Papillary structures and tubules generally lined with single or multiple layers of epithelial cells Epithelium is flattened to cuboidal in rats and cuboidal to tall columnar or pleomorphic with abundant eosinophilic cytoplasm in mice and nuclei are round to oval vesicular, with single nucleolus Mitotic activity is low Distension to cystic structures filled with spermatozoa is a common feature in CD-1 mice
Differential diagnoses Hyperplasia, rete testis: no compression of adjacent tissue, architecture maintained Carcinoma, rete testis: presence of malignant characteristics such as invasion, marked atypia, high mitotic rate, and hemorrhage
Comments: Rete testis adenoma is negative for vimentin but may produce mucins, i.e., may be PAS and/or Alcian blue positive. In contrast to rats, rete testis adenoma in mice shows some degree of cellular pleomorphism. Proliferative lesions of the rete testes are rare spontaneous lesions in aging rats but are more common in mice (Yoshitomi and Morii 1984) and have been reported to occur in both species following prenatal treatment with diethylstilbestrol and cadmium chloride (Bullock, Newbold, and McLachlan 1988; Newbold et al. 1985, 1986; Rehm and Waalkes 1988).
Carcinoma, Rete Testis: Testis
Species: mouse (predominantly), rat (rare)
Pathogenesis: spontaneous, age-related lesion and can be induced by xenobiotics
Diagnostic features Characterized by coalescence of rete testis tubules localized in the mediastinum testis expanded by masses of irregular papillary growths Cells are cuboidal in rats or pleomorphic in rats and mice, frequently tall columnar with eosinophilic cytoplasm and atypical, vesicular, or basophilic nuclei Mitotic figures may be common and atypical A marked scirrhous response, mucus, hemorrhage, and necrosis may be present Invasion of adjacent structures is present; may form extratesticular masses, noted in rats
Differential diagnoses Hyperplasia, rete testis: no compression of adjacent tissue due to increased tissue mass, architecture maintained. No invasion of adjacent structures Adenoma, rete testis: no invasion of adjacent structures. No cell atypia (in rats); absence of scirrhous response Mesothelioma: stains positive for vimentin
Comments: Proliferative lesions of the rete testes are rare spontaneous lesions of aging rats but are more common in mice (Yoshitomi and Morii 1984) and have been reported to occur following prenatal treatment with diethylstilbestrol and cadmium chloride (Bullock, Newbold, and McLachlan 1988; Newbold et al. 1985, 1986; Rehm and Waalkes 1988). Tumors of the rete testis are negative for vimentin but may produce mucins, i.e., may be PAS and/or Alcian blue positive.
Mesothelioma Malignant (Figures 63 and 64): Testis (also see “soft tissue” nomenclature manuscript)
Species: rat
Pathogenesis: spontaneous and xenobiotic induced tumors. Associated with mitogenic factor produced by Leydig cell tumors
Diagnostic features Characterized by fronds or papillary outgrowths from the serosal surface of the tunica vaginalis Generally arises in the region of the rete/pampiniform plexus Stains positive for vimentin Grows by local invasion along the mesothelial surface (often extending to the epididymis) or by local metastases into the peritoneal cavity
Differential diagnosis Hyperplasia, mesothelium: lack of evidence of mitotic activity, cellular atypia, or extension into adjacent tissues. Usually focal thickening or villous projections with cellular stratification. May be accompanied by inflammation Carcinoma, rete testis: tumor present in the mediastinum testis with invasion through capsule. Negative for vimentin
Comments: Mesotheliomas of the tunica vaginalis are relatively common tumors in the Fischer 344 rat and are also occasionally seen in other rat strains. They easily extend along the parietal mesothelial surface and will readily extend to the epididymis and also to other peritoneal mesothelial lined surfaces (McConnell et al. 1992). On the basis of their readiness to extend by local invasion, all are considered malignant. Spontaneous, as well as several, xenobiotic-associated tunica vaginalis mesotheliomas are causally associated with Leydig-cell tumors that lead to an autocrine growth factor–induced mesothelial mitogenesis (Maronpot et al. 2009). Also see “soft tissue” nomenclature manuscript.
Tumor, Mixed Sertoli–Leydig Cell, Benign: Testis
Species: mouse, rat
Pathogenesis: xenobiotic induced
Diagnostic features Composed of two different cell types reminiscent of Sertoli or Leydig cells Sertoli cells form tubules of highly variable size arranged perpendicular to the basement membrane; cells are pale and vacuolated, with elongated cytoplasm extending into a lumen; nuclei are cleaved and have a single prominent nucleolus; cells may show eosinophilic cytoplasmic inclusions and tubules may contain laminated mineralizations Pleomorphic Leydig cells similar to those of pure Leydig cell tumors are inseparably intermixed with neoplastic tubules; cytoplasm appears eosinophilic or vacuolated and cells have centrally located round nucleus
Differential diagnosis Adenoma, Leydig cell or Carcinoma, Leydig cell: only Leydig cells present. May also contain elongated and spindle-shaped Leydig cells, but not arranged in a palisading/tubular pattern as found in Sertoli cell tumors
Comments: A single mixed Sertoli–Leydig cell tumor has been described in a Wistar rat treated with cadmium chloride and is similar to neoplasms reported in men. The mixture of two cell types has been attributed to a possible common embryonic origin. Benign mixed Sertoli cell–Leydig cell tumors have been described by Rehm and Waalkes 1988 and Wakui et al. 2008, but in general they are rare tumors.
Tumor, Sertoli Cell, Benign (Figures 65–67): Testis
—Rat testis. Tumor, Sertoli cell, benign.
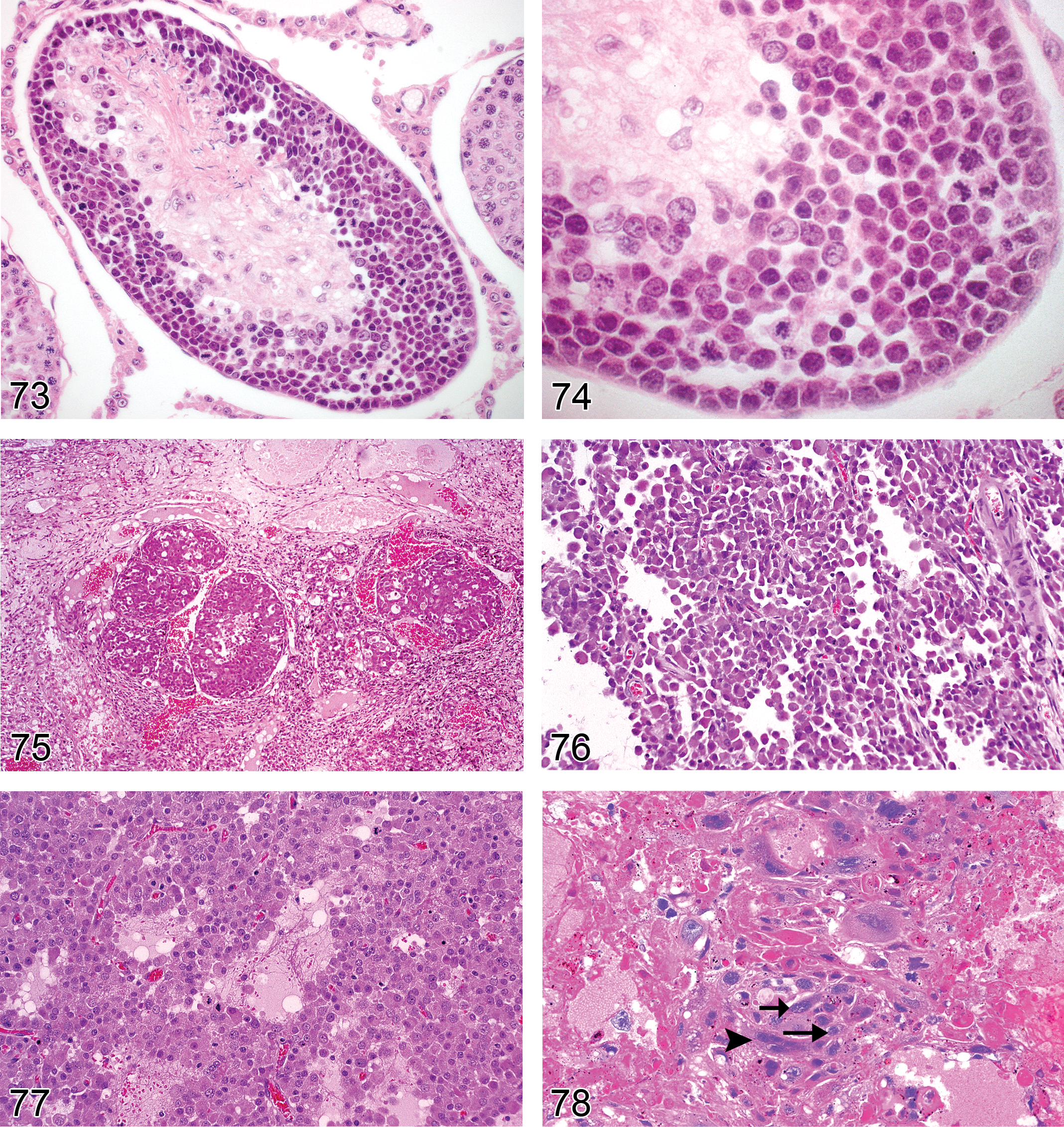
—Rat testis. Seminoma, benign.
Species: mouse, rat
Synonyms: tubular adenoma, sustentacular cell tumor, sex cord stromal tumor, gonadal stromal tumor, androblastoma, arrhenoblastoma
Pathogenesis: sex-cord stromal cells, Sertoli cells
Diagnostic features Cells generally arranged in palisades on a delicate fibrovascular stroma resulting in the formation of tortuous tubular structures without distinct lumina Tumor cells elongated with generally pale and vacuolated cytoplasm and finely stippled nuclear chromatin, cell borders indistinct Very low mitotic rate
Differential diagnosis Tumor, Sertoli cell, malignant: Evidence of invasion or poor differentiation. Increased mitotic rate Adenoma, Leydig cell or Carcinoma, Leydig cell: may also contain elongated and spindle-shaped cells but not arranged in a palisading/tubular pattern as found in Sertoli cell tumor
Comments: These tumors are extremely rare in rats and mice. The diagnosis is based mainly on the cellular arrangement and the nuclear orientation, reminiscent of Sertoli cells. Sertoli cell tumors have been described by Boorman et al. (1987c); Franks (1968); and Gordon, Majka, and Boorman (1996).
Tumor, Sertoli Cell, Malignant: Testis
Species: mouse, rat
Synonyms: tubular carcinoma; sustentacular cell tumor; sex-cord stromal tumor; gonadal stromal tumor; androblastoma; arrhenoblastoma
Pathogenesis: rare spontaneous tumors
Diagnostic features Similar features to Sertoli cell tumor, benign Local invasion of adjacent testicular structures Contains areas with poor differentiation, lacking pallisading Areas of necrosis may be present Mitotic figures quite common especially in less well-differentiated areas
Differential diagnoses Adenoma, Leydig cell or Carcinoma, Leydig cell: May also contain elongated and spindle-shaped cells but not arranged in a palisading/tubular pattern as found in Sertoli cell tumor Tumor, Sertoli cell, benign: well differentiated and no evidence of invasion
Comments: These tumors are extremely rare in rats and mice. Those that have been described appear largely benign and there is only one case (Abbott 1983) that was diagnosed as malignant based on local invasion and areas of poor differentiation. Bilateral Sertoli cell tumors with malignant features have been reported in transgenic mice (expressing the large T protein of polyoma virus in the seminiferous epithelium) (Paquis-Flucklinger, Rassoulzadegan, and Michiels 1994).
Tumor, Granulosa Cell, Benign (Figures 68 and 69): Testis
Species: mouse (predominantly), rat (rare)
Pathogenesis: mostly spontaneous
Diagnostic features Growth pattern reminiscent of ovarian granulosa cell tumors, e.g., forming follicle-like nests and nodules; additionally, in mice cords, sheets replacing seminiferous tubules and blood-filled sinuses may be present Small, basophilic cells similar to those of ovarian granulosa cell tumors with scant eosinophilic cytoplasm and nuclei with single nucleolus and stippled chromatin Cells usually round but may be spindle-shaped or pleomorphic; mitotic figures occasionally present
Differential diagnoses Seminoma, malignant: large, clear, or eosinophilic cells with distinct cell boundaries. Predominantly intratubular growth Tumor, Sertoli cell, benign: forms seminiferous tubule-like structures lined by highly elongated cells with abundant cytoplasm Adenoma, Leydig cell or Carcinoma, Leydig cell: small immature basophilic cells with scant cytoplasm may form part of Leydig cell neoplasm but lack follicle-like structures. Spindle-shaped cells usually are part of Leydig cell tumor composed otherwise of typical round cells with abundant cytoplasm
Comments: Rare tumor in rats occasionally seen in mice (Abdi 1995; Mitsumori and Elwell 1988; Mostofi, Davis, and Rehm 1994). Two unpublished cases of testicular granulosa cell tumors have been observed in F344 rats treated with a single dose of cadmium chloride. In the mouse, a single case has been documented in detail and seven cases of gonadostromal tumors with granulosa cell tumor features have been mentioned. Granulosa cell tumors have been induced in castrated rats by intrasplenic transplantation of testicular tissue (Kojima et al. 1984). These are also uncommon testicular tumors in men.
Seminoma, Benign (Figures 70–74): Testis
Species: mouse, rat
Synonyms: spermatocytoma, spermatocytic seminoma
Pathogenesis: rare spontaneous tumor of the germ cells
Diagnostic features Cells arranged in sheets and localized within the confines of one or a few tubular profiles Large, often polyhedral cells with distinct cell boundaries Cells are well differentiated and resemble spermatogonia, spermatocytes, and spermatids Occasional multinucleate cells may be present Mitotic figures can be numerous and may be atypical
Differential diagnosis Seminoma, malignant: Evidence of invasion through the tubular basement membrane. Invasion of tumor cells throughout the interstitial tissue. Seminoma cells less regular in shape and size, nucleus to cytoplasm ratio much higher.
Comments: Seminomas are extremely rare tumors in rats and mice, and there are very few documented examples. Most of the reported cases have been poorly differentiated and shown obvious evidence of invasion (Kerlin et al. 1998; Nyska et al. 1993; Kim, Fitzgerald, and De La Iglesia 1985; McConnell et al. 1992). Based on the features of these few tumors, it was suggested by McConnell et al. (1992) that all rodent seminomas should be considered malignant. However, recently, a number of well-differentied, intratubular seminomas, with features similar to those described for spermatocytic seminomas in humans (Aggarwal and Parwani, 2009; Emerson and Albright 2010) have been seen in young Sprague Dawley rats (Figures 70-74). Since the tumors appear confined to one or a few tubular profiles and are well differentiated, the diagnosis of benign seminoma is recommended for such lesions. The features of these benign variants resemble those illustrated by Boorman, Elwell, and Mitsumori (1987b).
Seminoma, Malignant (Figures 75–77): Testis
Species: mouse, rat
Synonyms: germinoma, spermatoblastoma, spermatocytoma
Pathogenesis: rare spontaneous tumor of the germ cells
Diagnostic features Cells arranged in sheets and lobules, may fill tubules and diffusely infiltrate stroma Large round to polyhedral cells with distinct cell boundaries Cells of different size, resembling primitive germ cells/spermatogonia Spermatocytic differentiation may occur Cytoplasm eosinophilic, amphophilic, or clear, usually containing glycogen, surrounding a large centrally located spherical nucleus with 1 to 2 basophilic nucleoli Mitotic figures can be numerous and may be atypical The tumor may have lymphocytic or granulomatous reaction in the stroma
Differential diagnoses Seminoma, benign: localized within one or a few tubules with no evidence of invasion. Well-differentiated germ cells resembling spermatocytes, spermatogonia, or spermatids Adenoma, Leydig cell or Carcinoma, Leydig cell: often uniform cells with abundant eosinophilic cytoplasm, sometimes also areas with spindle-shaped cells and small round nuclei. Small immature basophilic cells with scant cytoplasm may form part of Leydig cell neoplasm. Seminoma cells less regular in shape and size, nucleus to cytoplasm ratio much higher Carcinoma, embryonal: large and anaplastic epithelium-like tumor cells with primitive appearance Tumor, granular cell, benign: large or variably sized epithelioid or fusiform eosinophilic cells having PAS-positive cytoplasmic granules.
Comments: Seminomas are extremely rare tumors in rats and mice. The diagnosis is mainly based on the localization and cytological features of the tumor cells resembling spermatogonia, or at times spermatocytes. They stain positive for S-100 and vimentin. Anaplastic seminomas have also been described. “Classical seminomas” originate from undifferentiated germ cells. Seminomas have been described in several additional references, e.g., Boorman, Elwell, and Mitsumori (1987b); Boorman, Eustis, and Elwell (1990c); Faccini, Abbott, and Paulus (1990); Gordon, Majka, and Boorman (1996); Kerlin et al. (1998); Kim, Fitzgerald, and De La Iglesia (1985); McConnell et al. (1992); Mitsumori and Elwell (1988); Mostofi and Bresler (1976); Nyska et al. (1993); Radovsky, Mitsumori, and Chapin (1999); and Squire et al. (1978).
Carcinoma, Embryonal (Figures 78–80): Testis
Species: mouse, rat
Pathogenesis: rare spontaneous tumor arising from pluripotent germ cells, sex-cord stromal cells, cells of the visceral yolk sac
Diagnostic features Composed of large and anaplastic epithelium-like cells with a primitive appearance and indistinct cell boundaries Nuclei are large with coarse chromatin and prominent nucleoli Tumor cells may occur in solid sheets or have interspersed areas of acinar, papillary, or tubular structures Intratubular growth is considered to represent an early tumor stage Foci of yolk sac differentiation, choriocarcinoma, or well-differentiated tissues such as cartilage, bone, or skin may occur The stroma is scant and without inflammatory cell infiltrates Hemorrhage and necrosis are often present
Differential diagnoses Seminoma, malignant: large polyhedral cells with distinct cell boundaries and variably sized, spermatocytic differentiation may occur Teratoma, malignant: majority of the tumor tissue shows differentiation into various epithelial, mesenchymal, and neural tissues
Comments: Testicular germ cell tumors can be divided into two categories based on whether they display a single “pure” histologic pattern or more than one. In man, approximately 60% of testicular tumors display two or more different patterns, with the most common mixture being teratoma, embryonal carcinoma, yolk sac tumor, and elements of choriocarcinoma (Epstein 2010). Recently, such a tumor was described in a 12-week-old Swiss mouse (Jamadagni et al.
Choriocarcinoma (Figure 78): Testis
Species: rat
Pathogenesis: rare spontaneous tumor arising from pluripotent cells originating from displaced visceral yolk sac and deposited in gonads, placenta, mediastinum, or abdomen
Diagnostic features Presence of trophoblastic giant cells and smaller darker staining cytotrophoblasts is essential for the diagnosis. Trophoblastic giant cells are mononuclear and often appear irregular and bizarre Lacks an inherent vascular stroma and must receive nourishment by diffusion. Therefore, the tumors tend to grow at the periphery, while the center undergoes necrosis, often accompanied by hemorrhage. A fibrous capsule may be present Metastasizes at a very early stage of development
Differential diagnoses Seminoma, malignant: large polyhedral tumor cells with dark eosinophilic or amphophilic cytoplasm, may fill tubules and diffusely infiltrate stroma. Stroma present and usually infiltrated with mature lymphocytes Carcinoma, yolk sac: irregularly shaped tumor cells with a dark nucleus and a nucleus to cytoplasm ratio approaching 1:1. Tumor cells are suspended in a proteinaceous fluid Carcinoma, embryonal: large and anaplastic epithelium-like cells with a primitive appearance and indistinct cell boundaries. The tumor may contain areas of differentiation into any tissue type
Comments: The choriocarcinoma is a very rare tumor; a single case (as part of a mixed tumor) has been described for the mouse (Jamadagni et al.
Carcinoma, Yolk Sac, Malignant (Figure 79): Testis
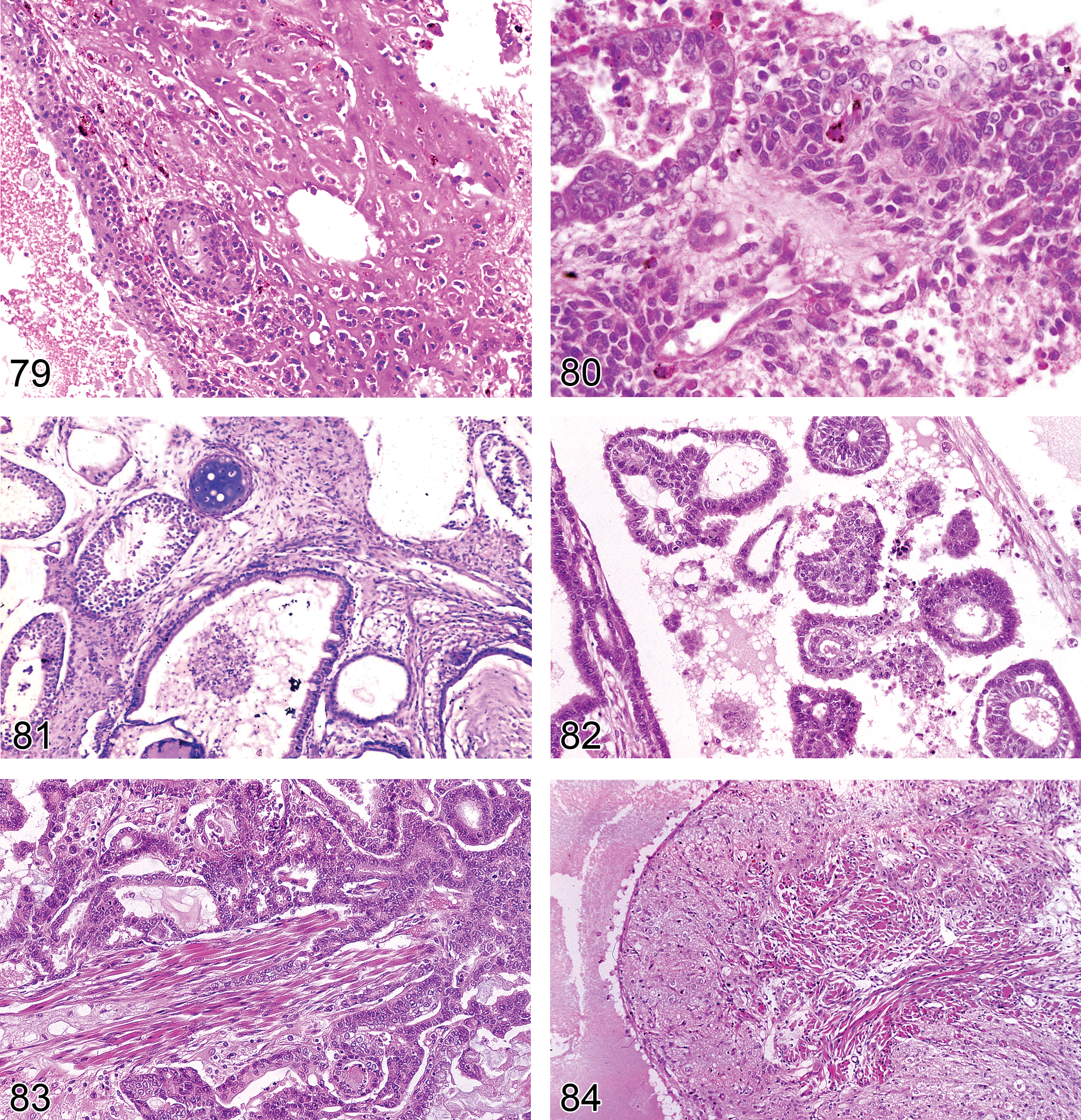
—Mouse testis. Carcinoma, yolk sac with presence of eosinophilic, extracellular matrix containing neoplastic cells. Part of a complex tumor. With permission from Jamadagni et al. (
Species: mouse, rat
Pathogenesis: rare spontaneous tumor arising from the extraembryonic portion of the egg cylinder or visceral yolk sac
Diagnostic features Most characteristic features are tumor cells producing abundant eosinophilic, PAS-positive matrix in which neoplastic cells are embedded Cell types and patterns that mimic the two layers of fetal membranes, i.e., the parietal and visceral yolk sac, may be observed Cells of the parietal yolk sac form disorganized clusters, sheets, rosettes, cords, or papillary structures Parietal yolk sac foci are composed of polygonal or cuboidal endodermal cells with amphophilic cytoplasm containing PAS-positive droplets Often the neoplastic cells are aligned on a conspicuous basement membrane Visceral yolk sac cells may form papillary structures surrounding capillaries, form cell clusters, or mingle with parietal cells Visceral yolk sac endodermal cells do not contain eosinophilic, PAS-positive droplets and may be columnar or large polygonal cells with pale, vacuolated cytoplasm and giant nuclei Cell nucleus is dark with closely clumped chromatin and contains one or two inconspicuous nucleoli The nucleus to cytoplasm ratio approaches 1:1 The tumor spreads aggressively to peritoneal surfaces and grows as nests or large colonies but seldom invades into the parenchyma of solid organs
Differential diagnoses Adenoma, Leydig cell or Carcinoma, Leydig cell: usually tumor cells have conspicuous eosinophilic cytoplasm. Usually no metastatic spread to peritoneal surfaces. Areas of necrosis are frequent, but suspension of tumor cells in proteinaceous fluid is not observed. Retiform variant of the rat may additionally show areas of glandular/tubular structures, lined by cuboidal to columnar cells but do not produce PAS-positive droplets Carcinoma, embryonal: may contain areas of yolk sac differentiation, but majority of tumor composed of large and anaplastic epithelium-like cells with a primitive appearance and indistinct cell boundaries Carcinoma, rete testis: lack abundant eosinophilic PAS-positive matrix Choriocarcinoma: central cavity necrotic, often with hemorrhage, but not filled with PAS-positive matrix; presence of trophoblast giant cells and smaller, darker staining cytotrophoblasts
Comments: Spontaneous yolk sac neoplasms are rare tumors in rats (Nakazawa et al. 1998) and mice. Only a single case of testicular yolk sac carcinoma (as part of a mixed tumor) has been reported in the mouse (Jamadagni et al.
Teratoma, Benign (Figures 81–84): Testis
Species: mouse, rat
Pathogenesis: rare spontaneous tumor arising from pluripotent germ cells
Diagnostic features Teratomas must contain tissue derived from the three germ layers Tissue components are generally well differentiated Benign teratomas often contain cysts lined by cuboidal, enteric, or respiratory epithelium. Smooth muscle may surround these cysts Other elements are pancreatic tissue, gastric epithelium, thyroid gland, well-differentiated neuronal tissue, cartilage, bone, and/or skeletal muscle
Differential diagnoses
Teratoma, malignant: shows peripheral invasion or metastases Carcinoma, embryonal: composed mainly of large and anaplastic epithelium-like cells with an immature morphology but foci of yolk sac differentiation or well-differentiated tissues such as cartilage, bone, or skin may occur
Comments: Teratomas are very rare tumors in rats and mice. Two cases of testicular teratoma were described in young Sprague-Dawley IGS rats by Sawaki et al. (2000). In both cases, the tumors appeared to be a combination of teratoma and embryonal carcinoma with transition of embryonal carcinoma cells into squamous epithelium, suggesting development of the embryonal carcinoma into teratoma. Similarly, a teratoma with elements of embryonal carcinoma (termed a teratocarcinoma) was described in a Swiss Albino mouse (Jamadagni et al. 2011) and a spontaneous teratoma was reported in an Institute of Cancer Research (ICR) mouse (Tani et al. 1997). In mice, teratomas are more commonly observed in strain 129 and its substrains. In this strain, a specific mutation (ter) has been identified, that markedly increases the incidence of teratomas from about 1% in wild-type mice to about 17% in ter heterozygous and more than 90% in ter homozygous male mice. Teratomas have also been described in the P53−/− transgenic mouse (Jacks et al. 1994). Teratomas may occur in any tissue but are most often encountered in the genital system. They originate from totipotent primordial germ cells and start to develop at about day 12 of fetal development (Alison et al. 1997; McConnell et al. 1992; Matin et al. 1998; Mostofi, Davis, and Rehm 1994; Rehm et al. 2001). Also, see comments under “embryonal carcinoma” relating to the potential for mixed embryonal tumors displaying two or more patterns of differentiation.
Teratoma, Malignant: Testis
Species: mouse, rat
Pathogenesis: rare spontaneous tumor arising fron pluripotent germ cells
Diagnostic features Teratomas must contain tissue derived from the three germ layers Nervous, epithelial, and mesenchymal tissues are poorly differentiated resembling embryonic tissue Peripheral invasion and tumors may metastasize Areas of necrosis and hemorrhage usually present
Differential diagnoses Teratoma, benign: no evidence of peripheral invasion or metastases Carcinoma, embryonal: composed mainly of large and anaplastic epithelium-like cells with an immature morphology but foci of yolk sac differentiation or well-differentiated tissues such as cartilage, bone, or skin may occur
Comments: See comments for “Teratoma, Benign.”
Excurrent Ducts (Efferent Ducts and Epididymis)
Introduction: Excurrent Ducts
The male excurrent duct system comprises the rete testis, efferent ducts, epididymis, and vas deferens (Foley 2001). Both ends of each seminiferous tubule empty into the rete testis, which serves as a small collecting reservoir for sperm as they exit the testis. In rodents, the rete testis is found at the cranial pole of the testis and connects to the efferent ducts via an extratesticular portion of the rete. The rete testis, efferent ducts, and epididymis form an integrated system that transports sperm from the testis to the vas deferens. This section will focus on the efferent ducts and epididymis.
One of the most common changes identified in the epididymis is the presence of sloughed germ cells and cell debris in the ductal lumen, originating from degenerative changes in the testes. Similarly, decreased sperm content (oligospermia/aspermia) will occur as a result of decreased testicular spermatogenesis. Although these are both secondary consequences of testicular changes, the epididymis is an important target tissue in its own right, playing a primary role in sperm maturation, sperm transport, and sperm storage as well as ejaculation (Foley 2001; Jones 2002; Olson, NagDas, and Winfrey 2002; Robaire and Hinton 2002)..
Function, Anatomy, and Histology: Excurrent Ducts (Figures 85–89)
Efferent Ducts
The efferent ducts link the rete testis to the epididymis and have been variably classified as part of the testis, a portion of the epididymal caput, or as a separate organ. Along with the rete testis, the efferent ducts reabsorb over 90% of the seminiferous tubular fluid that is secreted by the Sertoli cell and which serves to transport sperm from the seminiferous tubule to the epididymis (Hess 2002). The efferent ducts share many physiological similarities with renal tubules. If fluid reabsorption is disturbed in the efferent ducts, it can result in ductal dilation or ductal obstruction due to sperm stasis, both resulting in increasing back pressure of fluid, which can cause dilation and/or severe germ cell loss from the seminiferous tubules. In rodents, the efferent ducts form long, thin, tortuous ducts that are located within the epididymal fat pad and are generally trimmed off the epididymis and discarded in routine toxicity studies. In the dog, they are located within the initial segment of the epididymis and are readily available for examination, while in cynomolgus monkeys, the efferent ducts are enclosed within the same connective tissue capsule as the head of the epididymis but are located slightly proximal and may or may not be sampled in a routine section of epididymis. Although the efferent ducts are generally not collected in routine rodent toxicity studies, they are an important potential target for toxicity and may need to be examined.
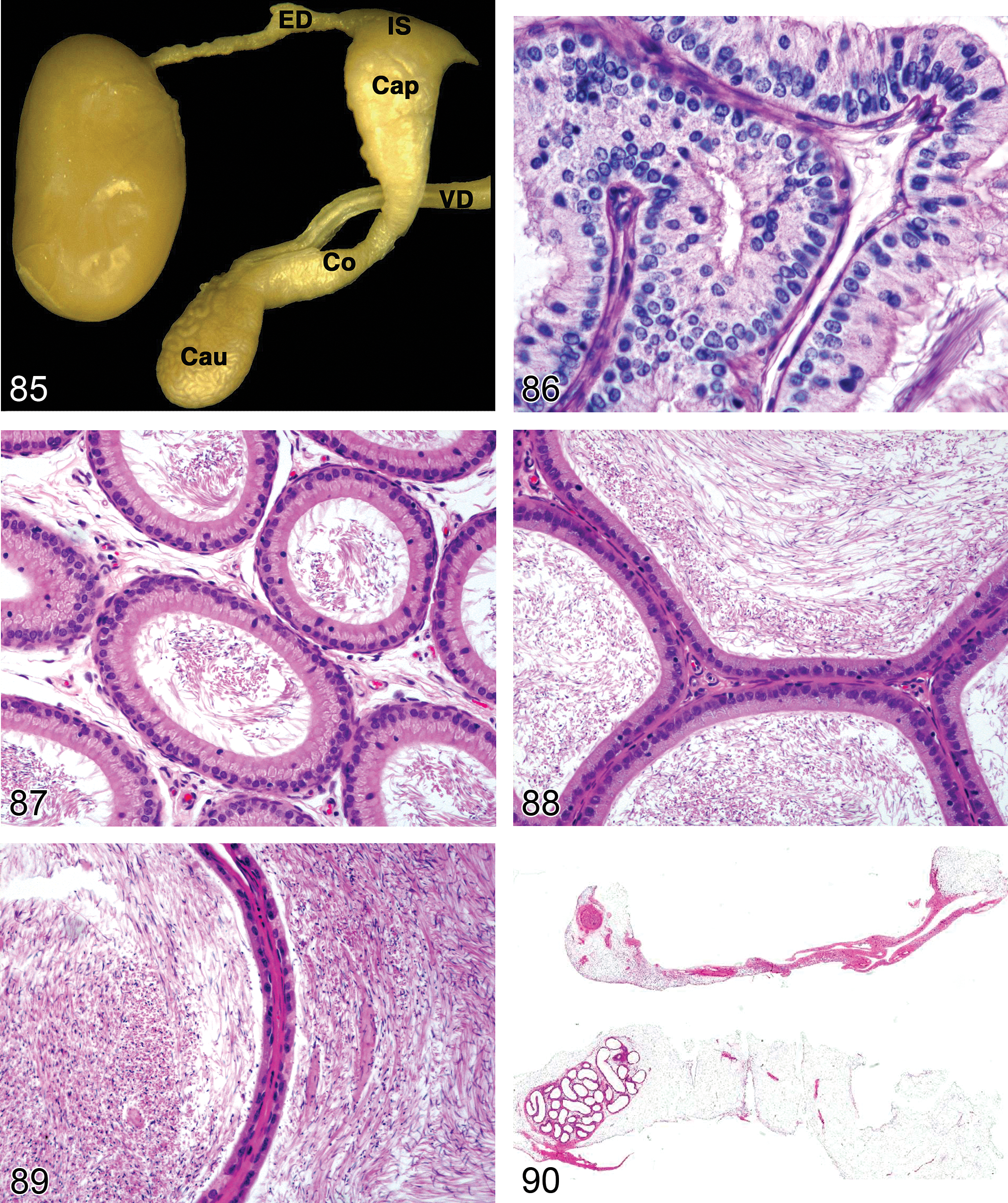
—Rat efferent ducts and epididymis. Normal anatomy. Efferent ducts (ED), initial segment (IS), Caput (Cap), Corpus (Co), Cauda (Cau), vas deferens (VD). With permission from Dr Rex Hess.
The efferent ducts are generally considered to develop from the mesonephric (Wolffian) duct or possibly the mesonephric tubules or glomeruli (Ilio and Hess 1994). In the rat, between 2 and 9 efferent ducts are most commonly reported, whereas in mice there are generally 3 to 5 ducts (Ilio and Hess 1994). In the rat, the efferent ducts can be divided into 3 zones or regions based on morphology: initial, conus, and terminal (Foley 2001; Hess 2002). In both mice and rats, the terminal portion of the efferent ducts forms a funnel in which the ductules anastomose and form a single tubule (common ductulus efferens) that abruptly transitions into the initial segment of the epididymis (Ilio and Hess 1994). The epithelium of the efferent ducts is composed of two main cell types in most species: principal and ciliated cells. A detailed review of the anatomy, function, and physiology of the efferent ducts is provided by Hess (2002) and Ilio and Hess (1994).
Epididymis
The epididymis is a highly coiled duct linking the efferent ducts to the vas deferens. Sperm leave the testis, largely immotile and unable to fertilize oocytes. By the time they reach the cauda epididymis, they are fully mature cells with progressive forward motility and the ability to recognize and fertilize an oocyte. In addition to mediating this maturation process, the epididymis concentrates, protects, and stores the sperm prior to ejaculation. Protein secretion and endocytosis are major functions of the epididymal epithelium and many of the proteins secreted and expressed are cell-specific and region-specific, underlining the importance of examination of the complete tissue. The luminal microenvironment of the entire epididymis is specialized and complex and varies along its length; it is maintained by secreted proteins, ions, and their transporters (Hermo and Robaire 2002). The ducts of the cauda epididymis are surrounded by prominent layers of contractile smooth muscle, which aids in the expulsion of sperm during ejaculation. The epididymis develops from the Wolffian duct under the influence of testicular testosterone (Byskov and Hoyer 1998; Rodriguez, Kirby, and Hinton 2002). Its position and dimensions vary with species. In rodents, the epididymis is relatively long and loosely attached to the testis compared to humans and domestic animals (Setchell, Maddocks, and Brooks 1988). It is generally subdivided into the initial segment and intermediate segment into which the efferent ducts empty, followed by three main parts (caput, corpus, and cauda), but it can be subdivided further by various histologic and/or functional schemes into as many as 6 to 12 distinct zones, comprising a variety of cell types with a variety of functions. In general, epithelial height decreases and luminal diameter increases from the caput to the cauda, with some regional exceptions. Cell types that are present throughout the epididymis include principal cells with apical stereocilia (the predominant cell type), basal cells, and halo cells (a subset of T lymphocytes and monocytes). In addition, the initial and intermediate segments contain apical and narrow cells, while clear cells are present in the caput, corpus, and cauda (Abou-Haila and Fain-Maurel 1984; Foley 2001; Hermo and Robaire 2002; Reid and Cleland 1957; Robaire and Hermo 1988; Setchell, Maddocks, and Brooks 1988).
Fixation and Trimming
Unlike the testis, conventional formalin fixation is optimal for the efferent ducts and epididymis. It is important to examine the entire length of the epididymis because many of its functions are cell-specific and region-specific and epididymal toxicity is frequently region-specific. This is best achieved by preparing a longitudinal section as described by Kittel et al. (2004).
Due to the difficulty in locating and identifying the efferent ducts in rodents, they are generally not routinely sampled, but if the profile of testicular changes (tubular dilation and/or severe atrophy, which is often unilateral) suggests the efferent ducts as a target, then they may need to be sampled (Hess 2002; Ilio and Hess 1994; La et al. 2012).
Congenital Lesions: Excurrent Ducts
Aplasia (Figure 90): Efferent Ducts, Epididymis
Synonym: agenesis
Pathogenesis: congenital abnormality resulting in absence of an organ due to failure of the primordium to develop during embryogenesis. Epididymal development is dependent on normal testosterone levels during the critical window of organogenesis
Diagnostic feature Complete absence of one or both epididymides on macroscopic and microscopic examination
Differential diagnosis Hypoplasia: partial absence or reduced size of the epididymis
Comments: Complete agenesis of the epididymis and efferent ducts is relatively uncommon in rodents but can be chemically induced by administration during gestation of compounds that disrupt testosterone biosynthesis or testosterone binding (e.g., dibutyl phthalate and flutamide) during the critical period of epididymal development. (Mylchreest et al. 1998, 1999).
Hypoplasia: Efferent Duct, Epididymis (Figure 90)
Synonyms: segmental aplasia, segmental agenesis
Pathogenesis: incomplete development of the epididymis or efferent ducts during embryogenesis. Normal epididymal development is dependent on adequate testosterone levels during the critical window of organogenesis
Diagnostic features Segmental loss or rudimentary development of segments of the epididymis or efferent ducts Segments proximal to the aplastic segment may be grossly distended and/or discolored Histologically, these distended areas often show evidence of sperm stasis, spermatoceles, and/or the formation of sperm granulomas Epididymis may be present but smaller than normal and have reduced numbers of ductular profiles
Comments: Complete agenesis or hypoplasia of the epididymis has been reported in rats due to
Ectatic Changes: Excurrent Ducts
Spermatocele (Figure 91): Efferent ducts, Epididymis
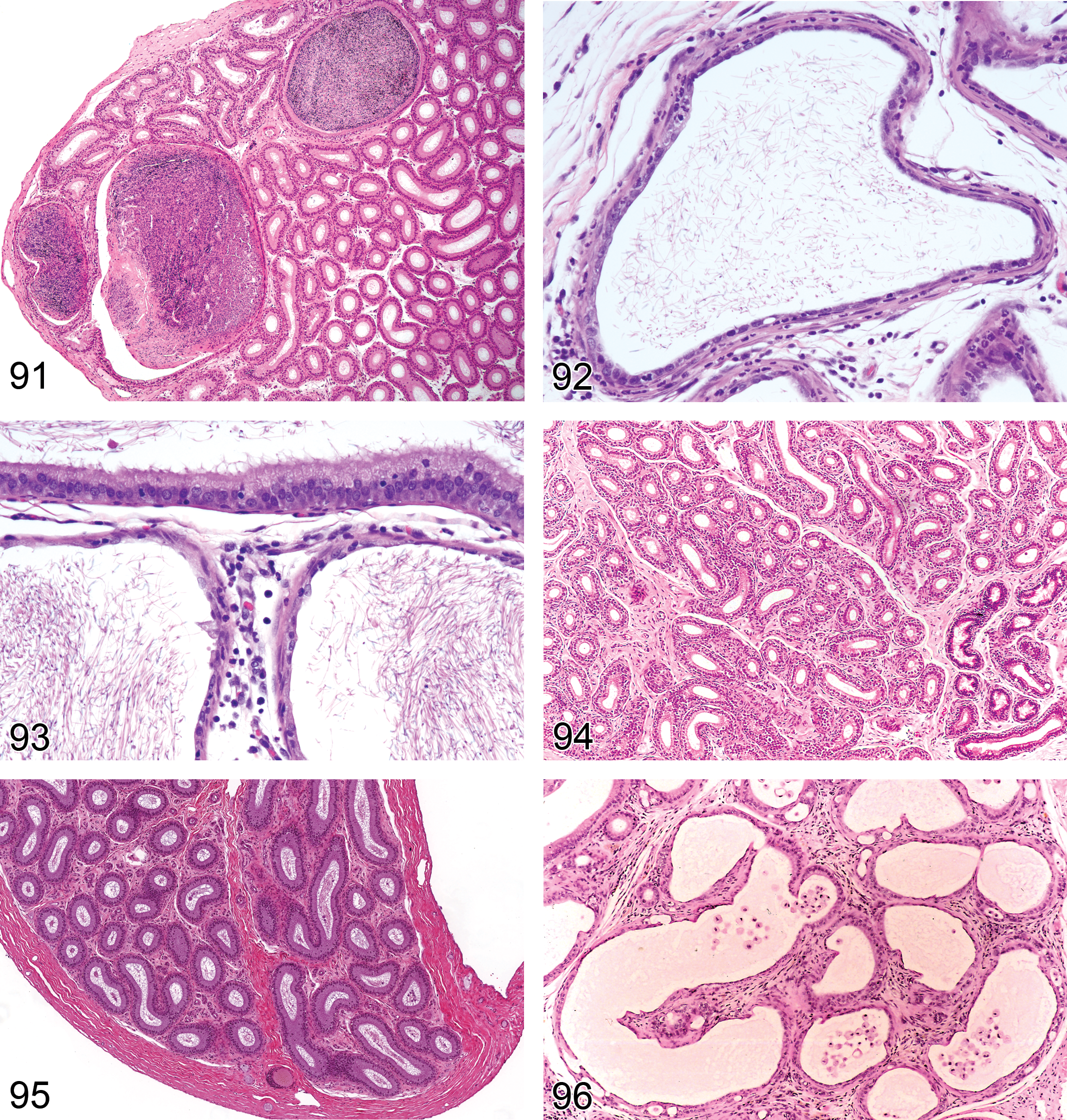
—Mouse epididymis, spermatocele (initial segment).
Synonyms: cyst, sperm retention cyst
Pathogenesis: congenital or acquired occlusion of the duct resulting in sperm accumulation and dilation of the duct
Diagnostic features Dilated ducts (≥3× diameter of normal duct lumen) filled with varying amounts of sperm May have hypertrophic or normal lining epithelium Generally present in the initial segment or caput epididymis
Differential diagnoses Sperm granulomas (accompanied by inflammation) Cystic atrophy: epithelium is flattened and luminal sperm are reduced or absent
Comments: In the mouse epididymis, spermatoceles may be seen as a background lesion, and they generally occur in the initial segment (Frith and Ward 1988; Radovsky, Mitsumori, and Chapin 1999). Chemically induced spermatoceles or cysts have also been reported with a number of compounds (for a review, see Hess 1998), including a single intraperitoneal dose of the alkylating agent ethylenedimethanesulphonate (EDS). With EDS, the spermatoceles occurred throughout the epididymis and most resolved with time although some developed into sperm granulomas (Cooper and Jackson 1973).
Degenerative Changes: Excurrent Ducts
Introduction
Degenerative changes in the epithelium of the efferent ducts or epididymis such as vacuolation, or atrophy generally reflect functional disturbances. Toxic injury that results in sufficient damage to the epithelium to result in breakdown or leakage of the blood–epididymal barrier will be accompanied by inflammation. Two of the most common findings in the epididymis are the presence of sloughed testicular germ cells admixed with luminal sperm and decreased numbers or density of sperm within the epididymal ducts. Both of these changes usually reflect testicular damage and are not primary degenerative conditions of the epididymis. They are therefore listed under “Miscellaneous Lesions.”
Degeneration, Epithelial (Figures 92 and 93): Efferent Ducts, Epididymis
Pathogenesis: degeneration of the epididymal epithelium is caused by cellular dysfunction leading to breakdown of the blood–epididymal barrier and generally accompanied by inflammation.
Diagnostic features Generalized or segmental thinning or loss of the epithelial lining Generally associated with an inflammatory response due to breakdown of the blood–epididymal barrier Generally leads to formation of sperm granulomas due to rupture of sperm into the interstitium
Differential diagnosis Fixation/handling artifact: cells appear normal but dislodged from the basement membrane. Autolysis frequently results in apparent breaks in the lining, not accompanied by inflammatory infiltrate
Comments: Epithelial degeneration as a morphological finding is very uncommon either as a background lesion or as a chemically induced finding (see Klinefelter 2002 for review). The best example is provided by the effects of methyl chloride inhalation in the rat (Chapin et al. 1984; Chellman et al. 1986), where epithelial degeneration was accompanied by inflammation and progressed to sperm granuloma formation. Although epithelial vacuolation can be considered evidence of epithelial degeneration, it has been dealt with separately (see below).
Atrophy, Ductal (Figures 94 and 95): Efferent Ducts, Epididymis
Pathogenesis: Epididymal atrophy is generally caused by decreased androgen support or decreased fluid and sperm output from the testis
Diagnostic features Generalized or segmental narrowing of ductal lumina with normal appearing epithelium, or slightly lower epithelial height When segmental, commonly seen at the corpus–caudal junction Generally associated with reduction or absence of sperm in the affected region/regions Intraductal folding of epithelium (cribriform change) may be present
Differential diagnosis Peripubertal status: the epididymis of the peripubertal rat (≤8–10 weeks of age) or mouse (≤6–8 weeks of age) may be only partially expanded
Comments: Epididymal atrophy is commonly associated with severe tubular degeneration/atrophy in the relevant testis. Expansion of the epididymal ducts relies on continuous output of seminiferous tubular fluid from the Sertoli cells and normal sperm production from spermatogenesis. Reduced diameter of the epididymal ducts is a normal response to a decrease in output of either product. The epididymis is an androgen-dependent tissue and it will undergo atrophy if androgen stimulation is decreased. This can occur through decreased testosterone production from the testis, decreased metabolism of testosterone to dihydrotestosterone (the major effective androgen for epididymal function) or androgen receptor blockade. Inhibitors of steroid biosynthesis, such as ketoconazole, will result in testicular changes as well as epididymal atrophy, but androgen receptor antagonists such as flutamide or inhibitors of 5α-reductase (which converts testosterone to dihydrotestosterone) cause epididymal atrophy in the absence of any discernable morphological effects on the testis (Creasy 1999). Segmental atrophy of the epididymis at the junction between the corpus and cauda is sometimes seen as a background finding in rodents and can also be a chemically induced change. The significance is unknown. Epididymal atrophy is a common finding in aged rodents secondary to testicular degeneration.
Dilation, Ductal (Figure 96): Efferent Ducts, Epididymis
Pathogenesis: generally due to increased fluid pressure within the ducts caused by disturbance in fluid reabsorption or obstruction distal to the dilation
Modifier: cystic
Diagnostic features
Dilation of ductal lumens May be lined by normal or flattened epithelium Affected ducts may appear empty or contain cellular debris
Differential diagnosis Spermatocele: focal dilation of the duct to ≥3× normal diameter and filled with sperm
Comment: Dilation of efferent ducts is an early indication of disturbed fluid reabsorption which can rapidly lead to secondary effects of tubular dilation and atrophy in the testis (La et al. 2012). Dilation of the epididymal ducts can also be a test article–related effect. Cystic dilation with atrophy of the epithelial lining of the epididymis is occasionally seen as a background incidental finding in rodents.
Cribriform Change (Figure 97): Efferent Ducts, Epididymis
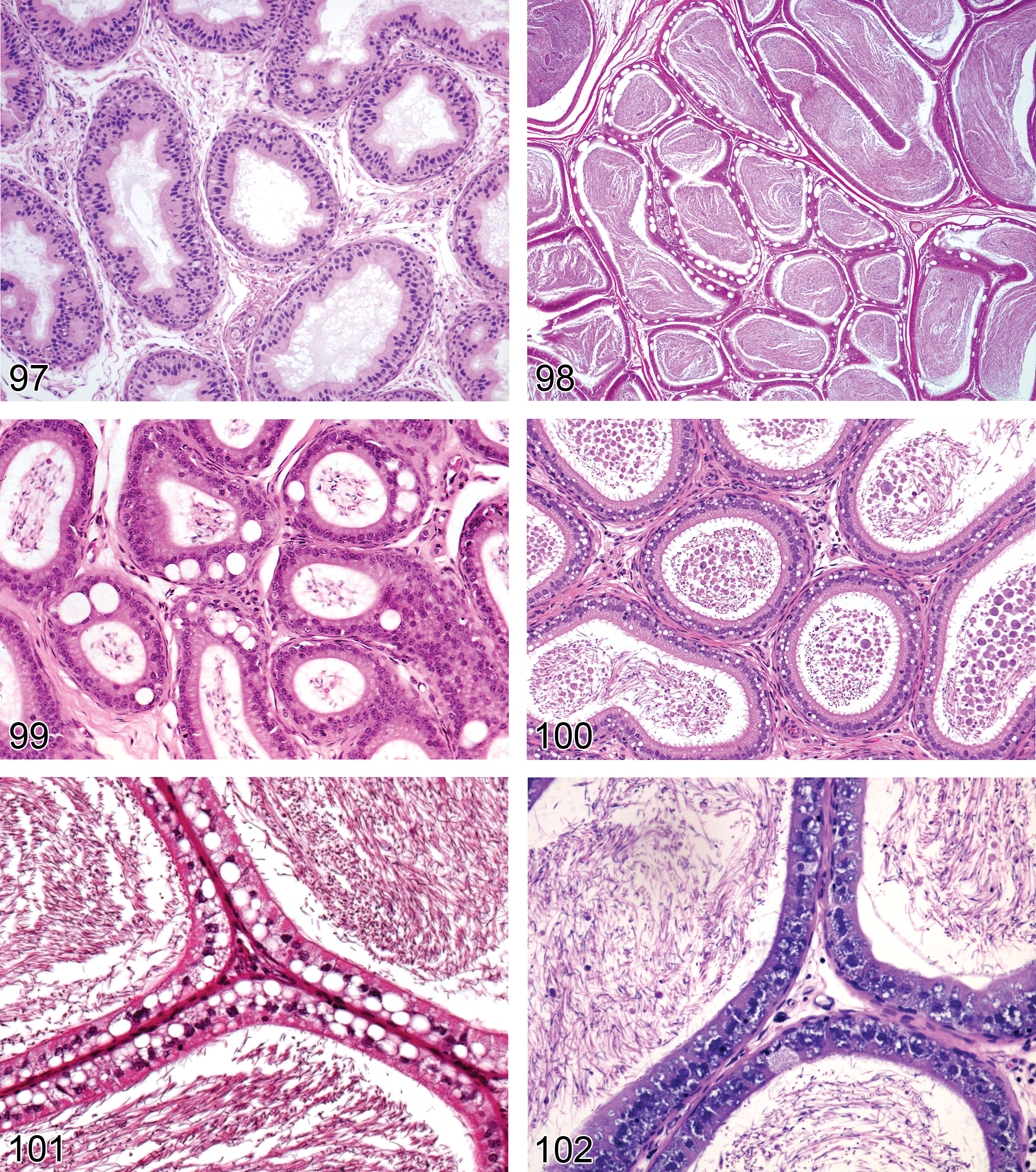
—Rat epididymis. Cribriform change, caput.
Synonyms: (Intra) epithelial lumina formation (Foley 2001; Ladds 1993), epithelial hyperplasia (Foley 2001), pseudoglandular formation
Pathogenesis: due to infolding and bridging of the epithelium in segments of duct that have undergone contraction
Diagnostic features Infolding of the epididymal epithelium resulting in “scalloping” of the luminal surface Apparent formation of pseudoglandular structures with lumens within the epithelium May be present in the caput region when associated with blocked efferent ducts (see comments) Often seen in the distal corpus/proximal caput in association with decreased/absent sperm and ductular atrophy
Comments: Cribriform change is generally seen at the junction between the distal corpus and the cauda epididymis in association with decreased sperm content and probably represents infolding of the epithelium secondary to ductal contraction (Foley 2001). More rarely it is seen as a segmental change in the caput epididymis and in this case it is usually associated with and secondary to efferent duct blockage.
Vacuolation, Epithelial (Figures 98–102): Efferent Ducts, Epididymis
Synonyms: vacuolar degeneration, vesicular degeneration, hydropic degeneration, basophilic degeneration
Modifiers: macrovesicular, microvesicular, basophilic
Pathogenesis: vacuolation of the epididymal epithelium may result from a number of degenerative changes including accumulation of fluids, lipids, phospholipids, and glycoproteins
Diagnostic features Microvacuolation is characterized by small, intracytoplasmic vacuoles often in the apical aspect of the cell Macrovacuolation is characterized by the presence of large, clear vacuoles within and between epithelial cells that may disrupt the epithelial alignment and displace the cytoplasm and nucleus to the periphery of the cell. Often present in the proximal caudal region and a common age-related change Basophilic vacuolation is characterized by basophilic, foamy vacuolation, which is generally restricted to a specific segment of the caput epithelium (common age-related change in rats) Vacuolation may be diffuse, but more often is localized to a specific segment of the epididymis
Differential diagnosis Fixation artifact: suboptimal fixation can result in variable numbers of vacuoles in the epididymal epithelium, particularly in the subcapsular ducts
Comments: Chemically induced epithelial vacuolation has been described with a number of compounds including drugs that cause phospholipidosis (Creasy 2001; Rudmann et al. 2004). Phospholipidosis generally appears as microvacuolation and may be caused by a variety of cationic amphiphilic compounds and not all produce vacuolation in the epididymis. When they do, the vacuolation is frequently restricted to a specific region (which will vary with the compound). If phospholipidosis is suspected, special procedures such as examination of plastic-embedded thin sections stained with toluidine blue; immunohistochemistry (IHC) using LAMP-2 (Obert et al. 2007) or electron microscopy should be performed for confirmation. Epididymal phospholipidosis has been reported in rats and mice given spinosad (Stebbins et al. 2002; Yano et al. 2002), and in rats given a selective dopamine 3 antagonist (Rudmann et al. 2004). Macrovacuolation is often seen in normal (control) rats as a spontaneous change in the proximal caudal region of the epididymis. It has also been reported to increase with age, with the vacuoles thought to represent disturbances in intracellular trafficking in the clear cells (Robaire 2002). Vacuolation of clear cells is also often caused by increased uptake of cellular debris from the epididymal lumen when associated with spermatogenic disturbances in the testis (Figure 99). Clear cells can be demonstrated using a PAS stain, which strongly stains the lysosomal complexes. Extensive vacuolation may occur spontaneously in aged mice, for example, as has been reported in B6;129 mice (Haines, Chattopadhyay, and Ward 2001). In aging rats, a specific segment of the caput epididymis commonly develops a basophilic foamy vacuolation (Boorman, Chapin, and Mitsumori 1990). It probably represents degeneration of the secretory product (proteins and glycoproteins) that are secreted by this portion of the epididymis.
Single cell necrosis, Epithelial (Figures 103 and 104): Efferent Ducts, Epididymis
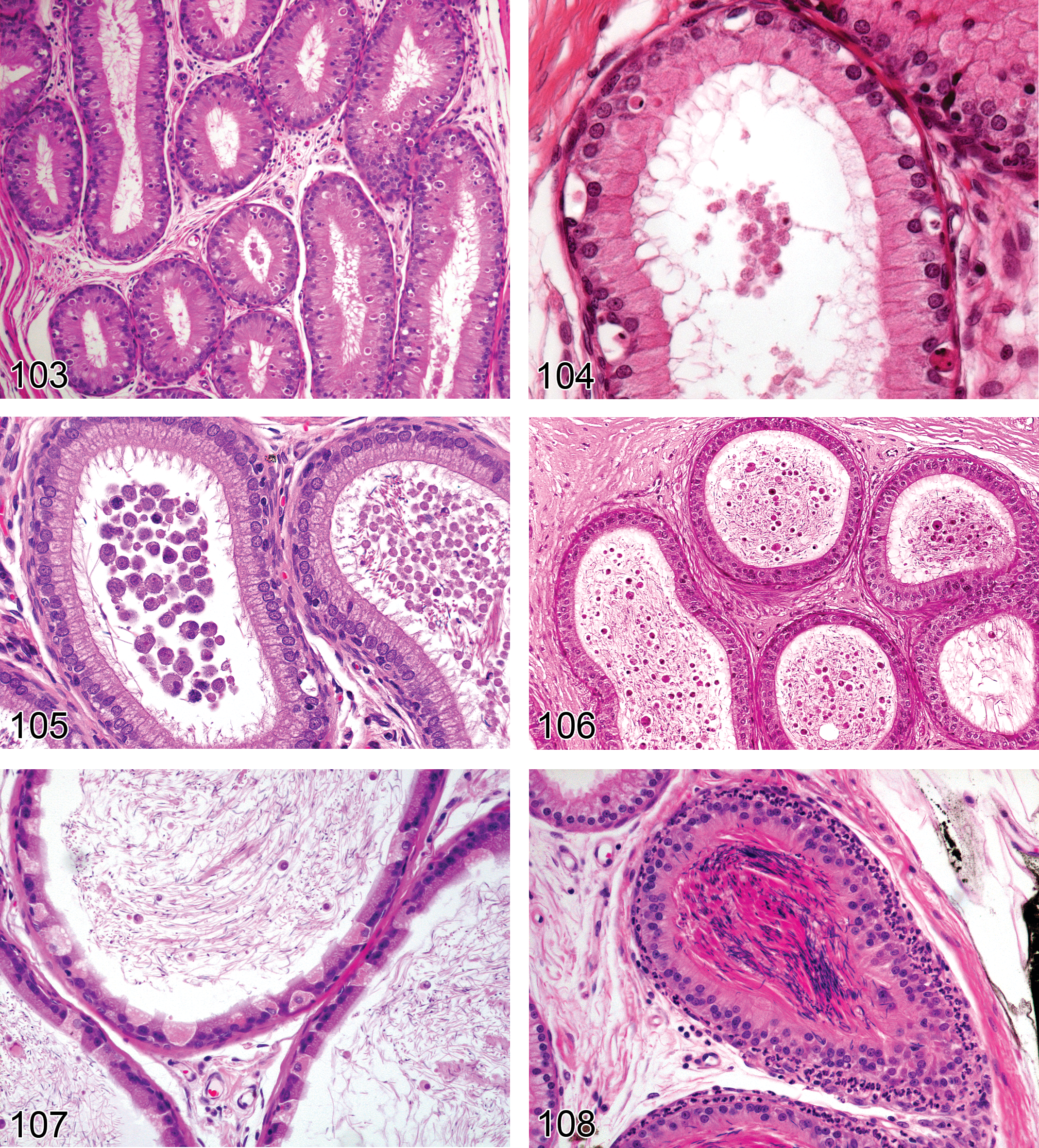
—Mouse epididymis. Single cell necrosis, epithelial, caput.
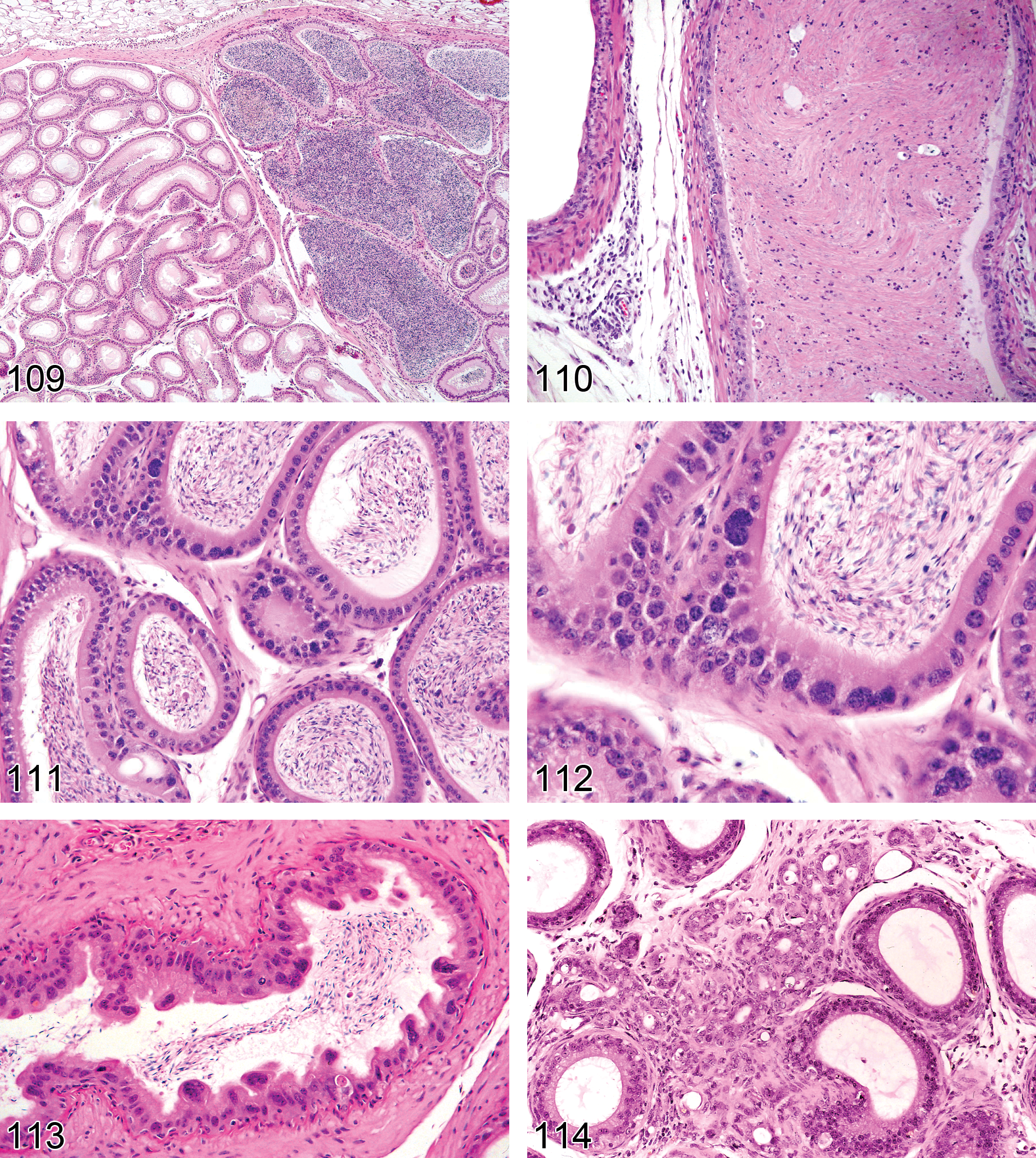
—Mouse epididymis. Sperm stasis, caput.
Synonym: apoptosis
Pathogenesis: may be caused by decreased androgen stimulation and efferent duct occlusion
Diagnostic features Increased numbers of epithelial cells having decreased size, shrunken, eosinophilic cytoplasm, and condensed or fragmented nucleus Generally localized to a specific region of the epididymis and most commonly seen in the initial segment or proximal caput epididymis, especially when related to androgen deficit
Differential diagnosis Normal background levels of single cell necrosis: occasional necrotic cells
Comments: There is a low background level of apoptosis and cell division in the normal epididymis but decreased androgen status causes rapid and prominent apoptosis in the principal cells. When caused by androgen depletion, epithelial apoptosis moves in a wave-like manner over time through different segments of the epididymis, starting in the initial segment (within 18 hrs of androgen withdrawal) and then, to the caput, corpus, and finally to the cauda by day 5 after withdrawal. Efferent duct ligation (or occlusion) also results in apoptosis of principal cells, but the apoptosis is limited to the initial segment (Ezer and Robaire 2002).
Metaplasia, Squamous Cell: Efferent Ducts, Epididymis
Pathogenesis: adaptive response resulting in replacement of the normal epididymal or efferent duct epithelium, with stratified squamous epithelium. May be secondary to tissue damage, modified differentiation in transgenic animals, or retinoid deficiency (Slausson and Cooper 1990)
Diagnostic features Replacement of the normal epididymal epithelial cells with flattened, attenuated to multiple layers of stratified squamous epithelium Affected ducts may contain laminated keratin whorls or debris May be associated with severe inflammation or sperm granulomas
Comments: Squamous metaplasia may be seen secondary to inflammation of the epididymis and/or sperm granuloma formation. Squamous metaplasia of the epididymal epithelium has been reported in mice secondary to experimental dietary hypovitaminosis (Bern 1952). Squamous metaplasia of the epididymides and vas deferens has been reported in transgenic mice expressing a dominant negative mutant of RARα driven by the murine mammary tumor virus promoter (Costa et al. 1997), as well as Ae2 mice deficient in anion exchanger 2 (Medina et al. 2003) or with combined Vhlh and Pten Mutation (Frew et al. 2008).
Miscellaneous Changes: Excurrent Ducts
Cell Debris, Luminal (Figures 105 and 106): Efferent Ducts, Epididymis
Synonyms: sloughed germ cells, germ cell debris, degenerate luminal germ cells, increased intraluminal germ cells
Modifiers: caput, cauda
Pathogenesis: exfoliation and sloughing of testicular germ cells into the epididymal lumen, either as part of the maturation process (peripubertal animals) or secondary to injury and/or sloughing of the testicular germ cells
Diagnostic features Presence of cell debris and/or recognizable degenerating germ cells originating from the testis May be accompanied by reduced density (or absence) of sperm May be throughout the epididymis or restricted to segments (caput or cauda) May be associated with increased number/vacuolation of clear cells in the cauda
Differential diagnosis Normal cell debris due to peripubertal/immature status
Comments: There are very few sloughed germ cells and cell debris in the normal adult rat epididymis, making this a very sensitive marker for detecting cell sloughing caused by testicular injury (Foley 2001). More are present in the normal adult mouse epididymis and even more are present in peripubertal rats and mice, due to the inefficiency of the first cycle of spermatogenesis. Increased numbers of sloughed, degenerate germ cells in the epididymal lumen of adult rodents generally reflect ongoing testicular degeneration/atrophy, spermatid retention, or germ cell exfoliation. The location of the cell debris can provide important information on when cells were sloughed from the testes. Cell debris in the caput would have been released from the testis within the previous 3 days whereas cells in the distal cauda were probably released more than a week previously. The presence of cell debris in the epididymis is often associated with increased prominence of clear cells in the distal corpus and cauda, probably due to increased endocytosis of particulate matter (Figure 107). If young animals (≤8 weeks) are used in short-term studies (≤2 weeks), it is common for there to be significant amounts of cell debris in the cauda epididymis.
Reduced Sperm, Luminal (Figures 106 and 107): Epididymis
Synonyms: hypospermia, oligospermia, aspermia, reduced spermatids
Pathogenesis: a reduction in sperm content is generally the result of reduced sperm output by the testis due to germ cell injury, decreased androgen support or rarely, secondary to congenital testicular hypoplasia/agenesis
Diagnostic features Reduced density or absence of sperm in the lumen of the epididymal duct The reduction may be throughout the epididymis or limited to specific regions (depending on the time after spermatogenic disturbance) Often accompanied by luminal cell debris from ongoing tubular degeneration/atrophy in the testis Often accompanied by ductal atrophy, caused by reduced sperm and fluid volume
Differential diagnosis Reduced sperm due to peripubertal status
Comments: A reduction in sperm content is an expected consequence of spermatogenic disruption in the testis (Radovsky, Mitsumori, and Chapin 1999). Sperm is present throughout the epididymis of the mouse and rat (from initial segment through to cauda), but the density varies in different regions (Reid and Cleland 1957). It is sometimes difficult to identify small reductions in density just by qualitative, subjective evaluation; quantitative sperm analysis is very useful to confirm such findings. The term aspermia could be used to denote complete absence of sperm, but since it is part of the continuum of sperm reduction, the use of severity gradings is recommended to convey degree of change. The number and density of sperm also vary with the age of the animal. Sperms are likely to be absent or few in number in the cauda epididymis of rats and mice younger than 8 or 5 weeks of age, respectively. They do not reach maximal numbers until approximately 12 (rats) or 9 (mice) weeks of age and content can vary significantly between peripubertal animals. Sperm numbers can also be reduced or increased through direct or indirect alterations in epididymal structure and function. Some reproductive toxicants increase or decrease transit time through the epididymis (Klinefelter 2002) or can alter epididymal function through decreasing androgen status (Ezer and Robaire 2002).
Sperm Stasis (Figures 108–110): Efferent Ducts, Epididymis
Pathogenesis: sperm stasis may occur in the efferent ducts as a result of increased fluid resorption or due to impaction of sperm in blind ending ducts. It may also occur in the cauda epididymis when sperm is not being actively produced by the testis.
Diagnostic features Increased density of luminal sperm in a segment of the epididymal or efferent ducts Recently impacted sperm appear dense and eosinophilic but may become basophilic with time due to the deposition of calcium salts The duct distal to the site of impaction is usually empty of sperm Sperm stasis generally incites an inflammatory response with time (Radovsky, Mitsumori, and Chapin 1999)
Differential diagnoses Spermatocele (duct is ≥3 times the normal size). Sperm granuloma (accompanied by inflammation)
Comments: Movement of sperm through the efferent ducts and epididymis is dependent on the correct fluid dynamics (fluid production by the Sertoli cell and fluid reabsorption by the efferent ducts) and also on smooth muscle contraction within the epididymis and vas deferens. Disturbances in either of these processes or obstruction of any part of the duct system can lead to sperm stasis, which in turn often leads to inflammation and/or sperm granulomas. Blind ending proximal efferent ducts often have sperm stasis but generally do not incite an inflammatory response (Foley 2001). Chemically induced increases in fluid reabsorption in the efferent ducts generally result in sperm stasis followed by sperm granulomas, which cause secondary tubular dilation and atrophy in the testis (for review, see Hess 1998, 2002; La et al. 2012). Sperm stasis can occur in the cauda epididymis from a variety of causes including blockade of adrenergic pathways by alpha-adrenergic antagonists such as prazosin and guanethidine (for review, see Klinefelter 2002). Sperm granulomas are frequent sequelae. Sperm stasis may also be present in the cauda epididymis if the testis is no longer producing sperm due to severe spermatogenic disturbances. In these cases, the rest of the epididymis is empty of sperm, but some residual, eosinophilic sperm and cell debris often remains in the cauda epididymis.
Amyloid: Efferent Ducts, Epididymis
Synonym: amyloidosis
Pathogenesis: age-related degenerative condition in mice
Diagnostic features Accumulation of pale eosinophilic extracellular material Perivascular or interstitial Green birefringence using polarized light with Congo Red stain May form thin bands or extensive sheets
Comments: Amyloidosis is an age-related, incidental, spontaneous, often systemic disease characterized by the extracellular deposition of polypeptides (often serum amyloid-associated protein or immunoglobulin fragments) appearing in routine section as a lightly eosinophilic amorphous material. It is common in aged mice and rare in rats. Confirmation of the deposits as amyloid can be accomplished with light microscopy using special stains such as Congo Red. Amyloid appears apple green under polarized light with this stain.
Karyomegaly (Figures 111–113): Epididymis (Generally Confined to Vas Deferens)
Synonym: karyocytomegaly
Pathogenesis: unknown
Diagnostic features Most commonly seen in the vas deferens Individual cells or groups of tall epididymal epithelium display enlarged, deeply basophilic, atypical, folded single or multiple nuclei These nuclei may show cytoplasmic invaginations and the cells may protrude into the lumen and/or show pseudostratification
Comments: Karyomegaly is usually seen in the vas deferens and is commonly seen in aged mice. For example, this finding has been reported in aged B6;129 mice (Haines, Chattopadhyay, and Ward 2001). Despite the presence of cellular atypia, this lesion is not known to progress.
Adenosis (Figures 114 and 115): Epididymis
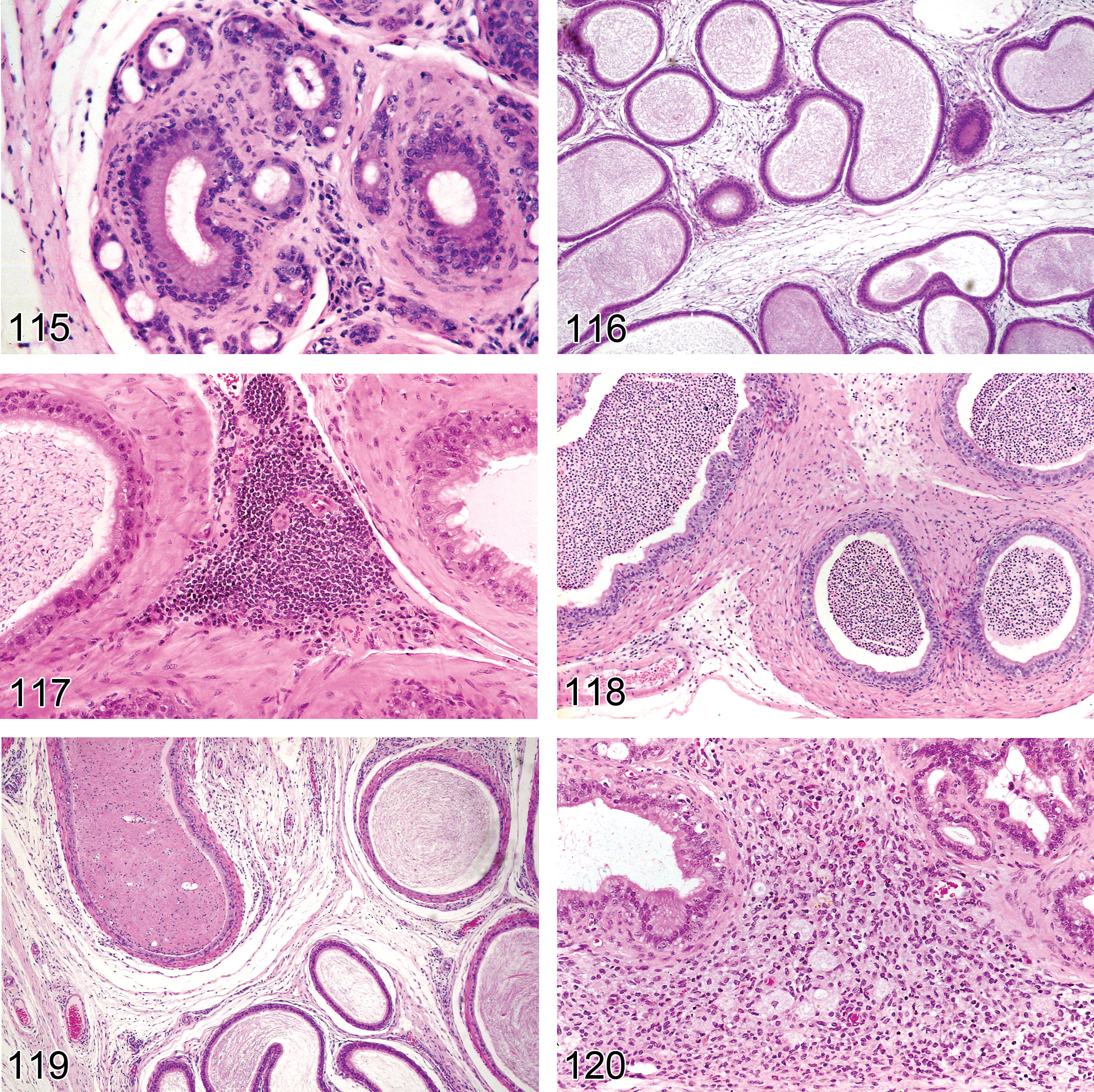
Mouse epididymis. Adenosis.
Pathogenesis: unknown
Diagnostic features Groups of epithelial cells in close association with epididymal ducts or located in between ducts Microlumens frequently present Maybe associated with periductal fibrosis and epithelial vacuolation
Comments: Adenosis may occur spontaneously in aged mice (Radovsky, Mitsumori, and Chapin 1999) or in mice exposed neonatally to diethylstilbestrol (Bullock, Newbold, and McLachlan 1988).
Inflammatory Changes: Excurrent Ducts
Introduction
A step-wise approach is recommended for diagnosing inflammatory conditions. Edema of the interstitial tissue can sometimes occur without any obvious accompanying inflammatory infiltrate. Minimal and/or focal aggregates of inflammatory cells should be referred to as “inflammatory cell infiltrates” with the additional use of modifiers to specify the predominant cell type involved (lymphocytic, neutrophilic, lymphoplasmacytic, etc.). If the inflammatory infiltrate is more diffuse and accompanied by other classic markers of inflammation such as edema, congestion, or cellular degeneration/necrosis, then the term inflammation should be used, and appropriate modifiers may be used to specify the character of the inflammation (e.g., lymphocytic, neutrophilic, purulent, etc.).
Edema (Figure 116): Efferent Ducts (Figure 116), Epididymis
Pathogenesis: extravasation of fluid into the interstitial tissue, occasionally seen in pre- or peripubertal rats as a background finding and may be test article–related in situations where fluid balance is disturbed
Diagnostic features Expansion of interstitial tissue by clear, slightly eosinophilic, or slightly basophilic appearing fluid May be accompanied by slight increase in mixed inflammatory cells
Differential diagnosis Inflammation: accompanied by diffuse inflammatory infiltrate and tissue damage Normal maturing epididymis: loose interstitial connective tissue without fluid accumulation
Comments: Edema in the cauda epididymis may sometimes be seen as a background finding, particularly in the prepubertal rat epididymis (Sawamoto et al. 2003). It should not be confused with the normal expansion of loose connective tissue in the cauda epididymis as the tissue undergoes maturation. Edema may also be seen as a test article–related change when fluid balance is disturbed (this may involve vascular mediated changes or altered fluid reabsorption from the ducts). Administration of
Infiltrate, Inflammatory Cell (Figures 117 and 118): Efferent Ducts, Epididymis
Synonyms: lymphocytic infiltrate, mononuclear cell infiltrate, inflammation
Modifiers: lymphocytic, neutrophilic, plasmacytic, mixed inflammatory cell
Pathogenesis: focal extravasation of inflammatory cells into the interstitial tissue, common background finding of unknown etiology
Diagnostic features Small, focal aggregates of inflammatory cells, generally lymphocytic, interstitial, and perivascular, may also be intraluminal Mixed inflammatory cell foci (microabscesses) are also commonly seen as a background finding in the adjacent adipose tissue surrounding the epididymis
Differential diagnosis Inflammation: more diffuse inflammatory infiltrate with additional features such as edema and tissue damage
Comments: Focal lymphocytic aggregates are commonly seen as a background incidental finding, particularly in the caput. Focal neutrophilic inflammatory infiltrates within the interstitium of the epididymis may represent an early stage of inflammation. Inflammatory infiltrates within the ductal lumen (admixed with sperm), generally indicate sperm stasis (see sperm stasis).
Inflammation (Figures 119 and 120): Efferent Ducts, Epididymis
Synonym: epididymitis
Modifiers: neutrophilic, lymphocytic, lympho-plasmacytic, mixed inflammatory cell. Other modifiers include purulent or granulomatous
Pathogenesis: generally a response to tissue injury or necrosis. Inflammation in the epididymis may occur secondary to breakdown of the blood epididymal barrier, which allows access of immunocompetent cells in the interstitium to the antigenically foreign sperm.
Diagnostic features Focally extensive or diffuse accumulation of inflammatory cells May be within the interstitium, epithelium, and/or within the ductular lumens Associated with evidence of tissue damage and/or edema
Differential diagnoses Inflammatory cell infiltrate: focal, minimal, and without evidence of tissue injury Sperm granuloma: granulomatous inflammation admixed or surrounding degenerating sperm within the interstitium
Comments: Inflammation of the epididymis is an uncommon lesion. As with the testis, sperms are protected against immunologically competent cells by tight junctions between adjacent ductal epithelial cells. However, the barrier is not as efficient as the one in the testis and is more easily compromised. Relatively few chemicals have been reported to cause inflammation in the epididymis as a primary effect. The best example is the administration of methyl chloride in Fischer rats, which results in destruction of the ductular epithelium and inflammation, which is generally segmental and limited to the cauda, with progressive development of sperm granulomas over time (Chapin et al. 1984; Chellman et al. 1986). Inflammation in the epididymis and vas deferens has also been reported in adult rats secondary to neonatal administration of diethylstilbestrol (Atanassova et al. 2005). In rodents, the presence of prominent mast cells in the interstitium of the epididymis is a common background finding that should not be confused with inflammation. The number of mast cells reportedly varies with sexual maturation, reaching a peak at approximately 90 days of age (Jiménez-Trejo et al. 2007).
Sperm Granuloma (Figures 121–123): Efferent Ducts, Epididymis
Synonyms: spermatic granuloma, spermatogenic epididymitis
Pathogenesis: Sperm granulomas occur when immunologically competent cells gain access to sperm, which are antigenically foreign. This generally occurs following rupture of the luminal sperm into the interstitium of the epididymis (McGinn et al. 2000). This can occur following obstruction of the vas deferens, damage to the epididymal epithelium, rupture of the duct, or as a chronic sequel to sperm stasis. Most sperm granulomas are located in the interstitium
Diagnostic features Central accumulation of degenerating sperm surrounded by variable amounts of granulomatous (foreign body) inflammatory response Inflammatory response characterized by an inner layer of epithelioid (spermiophagic) macrophages, surrounded by a loose, vascular connective tissue rich in lymphocytes and plasma cells Sometimes surrounded by a fibrous capsule Sperm may form discrete “balled” clumps within the central cavity May have epididymal epithelial hypertrophy/hyperplasia (intraductal granulomas) surrounding impacted sperm Most commonly located in the body or cauda epididymis Also occur in the efferent ducts as a background or chemically induced lesion
Differential diagnoses Spermatocele: no inflammation Inflammation: absence of sperm associated with the inflammatory response
Comments: Sperm granuloma formation most likely begins with compromise of the epithelium lining the epididymal duct and leakage of sperm through the epithelium to gain access to the underlying lamina propria where immunocompetent cells mount a foreign body inflammatory response and attempt to wall off the extruded sperm. Compromise of the wall can be due to multiple causes, including sperm stasis, direct damage, or increased intraductal pressure. Sperm granulomas are more common in the epididymis than in the testis and may result from congenital, focal, or segmental aplasia of the ducts. In addition, sperm granulomas may occur as a chemically induced finding and have been reported throughout the epididymis. Examples of chemically induced sperm granulomas include caudal granulomas secondary to inflammation (e.g., methyl chloride); efferent ducts and caput granulomas following epithelial damage (e.g., dibromochloropropane, α chlorohydrin) in rats (Boorman, Chapin, and Mitsumori 1990; Creasy 1998); efferent ducts and caput granulomas in B6C3F1 mice given 2-methylimidazole (Tani et al. 2005); and granulomas in the corpus and cauda epididymis of rats given
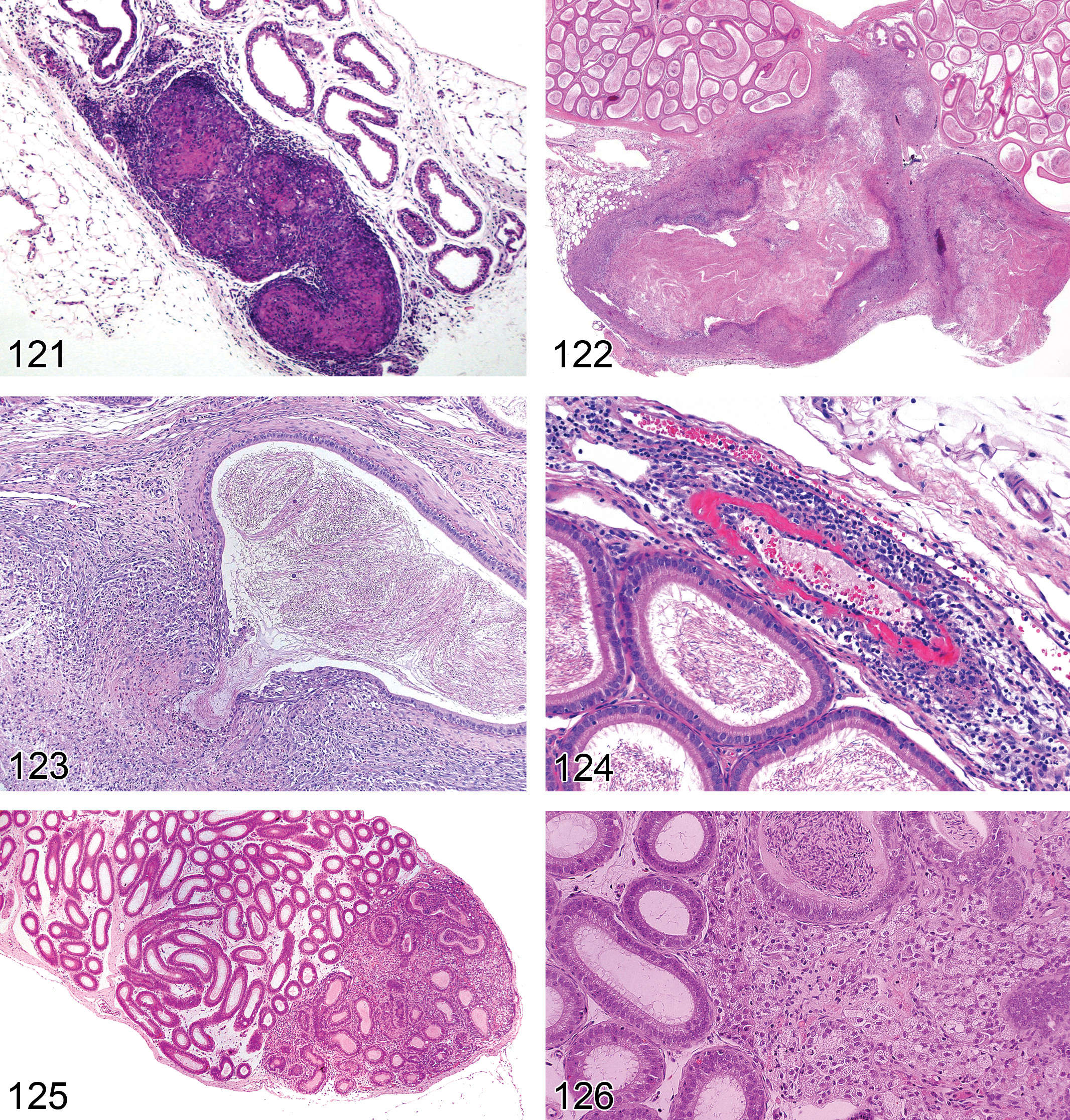
—Rat efferent ducts. Sperm granuloma.
Vascular Changes Excurrent Ducts
Necrosis/Inflammation, Vascular/Perivascular (Figure 124): Efferent Ducts, Epididymis
Synonyms: vasculitis, arteritis, perivascular inflammation, periarteritis, polyarteritis nodosa
Pathogenesis: generally a feature of spontaneous, age-related, systemic “polyarteritis nodosa,” which is a progressive degenerative lesion of the walls of small arterioles. May also be induced or exacerbated by vasoactive drugs and chemicals through over distension of blood vessel walls, or by systemic hypertension
Diagnostic features Inflammatory cells (usually lympho-plasmacytic) surrounding blood vessels (small arterioles) and often infiltrating vascular walls Generally accompanied by medial hypertrophy, fibrinoid necrosis (accumulation of fibrillar, eosinophilic material in the media), or hyalinization (necrosis) of affected vessels Commonly seen in the adjacent adipose tissue surrounding the epididymis or within the interstitium of the epididymis
Differential diagnoses Inflammatory cell infiltrate: the inflammatory infiltrate is generally lymphocytic, does not involve the blood vessel wall, and is not accompanied by hypertrophy or degenerative changes in the blood vessel wall Inflammation: the inflammatory infiltrate is more diffuse and not centered on the blood vessels
Comments: Age-related polyarteritis nodosa is much more common in the mouse than in the rat and much more common in the testis than in the epididymis. The blood vessels of the mesentery and testis are more sensitive to spontaneous and induced changes in the vascular bed, but the epididymis may also be affected. Vascular injury has been reported in the testis, epididymis, and accessory sex glands of the rat after administration of cadmium chloride (Waites and Setchell 1966). In most cases, medial necrosis is seen as part of the age related, systemic “polyarteritis nodosa” condition. Also see INHAND nomenclature of the Cardiovascular System.
Neoplastic Proliferative Lesions: Excurrent Ducts
Introduction
Neoplasms of the excurrent ducts are extremely rare in rats and mice. Tumors arising from the associated connective tissue matrix, such as leiomyomas/sarcomas, fibromas/sarcomas, and hemangiomas/sarcomas are occasionally seen but are not included here. Standard terminology and diagnostic criteria apply to these tumors. Two tumor types specific to the epididymis have been described in the mouse; these are histiocytic sarcoma and Leydig cell adenoma.
Adenoma, Leydig Cell (Figures 125 and 126): Epididymis
Species: mouse
Synonyms: tumor, Leydig cell, benign, tumor, interstitial cell, benign
Pathogenesis: spontaneous tumor, unknown pathogenesis
Diagnostic features Nodular or diffuse mass with peripheral compression, displacement, or replacement of adjacent epididymal tubules Composed of polygonal cells with abundant eosinophilic or vacuolated cytoplasm Nucleus generally central, round with evenly distributed chromatin and single prominent nucleolus. Low nuclear/cytoplasmic ratio Smaller hyperchromatic cells with scant cytoplasm and cells with yellow-brow pigment (lipofuscin) are less common Mitoses infrequent with no atypical mitotic figures
Differential diagnoses Granuloma: composed of vacuolated macrophages and other inflammatory cells with central accumulation of sperm Sarcoma, histiocytic: cells may be fusiform, multinucleated giant cells may be present. Definite differential diagnosis may require IHC (Leydig cell positive for calretin, inhibin; histiocytic sarcoma positive for Mac2 and F4/80)
Comments: Rare tumor described in B6C3F1 mice (Mitsumori et al. 1989; Mitsumori and Elwell 1988). There are no reports on epididymal Leydig cell hyperplasia or carcinomas, and since Leydig cells normally are not found in the epididymis, it has been suggested that these tumors may in fact be primary histiocytic sarcomas of the epididymis (Yano et al. 2008). Immunohistochemical markers are recommended to provide a definitive diagnosis.
Sarcoma, Histiocytic (Figures 127–129): Epididymis
Species: mouse
Synonyms: reticulum cell sarcoma, histiocytic lymphoma, histiogenic tumor
Pathogenesis: rare spontaneous tumor arising from undifferentiated mesenchymal or histiocytic cell
Diagnostic features Arise as nodules in the cauda of the epididymis Composed of sheets of pleomorphic cells ranging from round to fusiform in shape Multinucleated giant cells may be present Cytoplasm may be eosinophilic, foamy, with diastase-resistant PAS-positive deposits, erythrophagocytosis and may contain pigments such as hemosiderin, hematoidin, and lipofuscin Pleomorphic nuclei may display sharp cleavage, mitotic figures may be frequent Invasion of epididymal tissues and distant metastases, primarily to the peritoneum and liver, may be observed May be associated with spermatic granuloma due to obstruction
Differential diagnoses Spermatic granuloma: shows organization with central area of spermatozoa surrounded by granulomatous capsule composed of epithelioid giant cells and mixed inflammatory cell infiltrates, few mitotic figures. Hemorrhage, tissue invasion, and metastases are absent Adenoma, Leydig cell: composed of polygonal epithelial cells, rare fusiform cells. Hemorrhage, tissue invasion, and metastases are absent. Definite differential diagnosis may require IHC (Leydig cells positive for calretin, inhibin; histiocytic sarcoma positive for Mac2, F4/80)
Comments: Histiocytic sarcomas of the epididymis have only been observed in mice and considered equivalent to mouse uterine histiocytic sarcoma. There is some suggestion that lesions diagnosed as Leydig cell tumors may in fact be histiocytic sarcomas (Yano et al. 2008). Histiocytic sarcomas of the epididymis have been described by Baldrick and Reeve (2007); Itagaki, Tanaka, and Shinomiya (1993); Shiga (1994); and Yano et al. (2008).
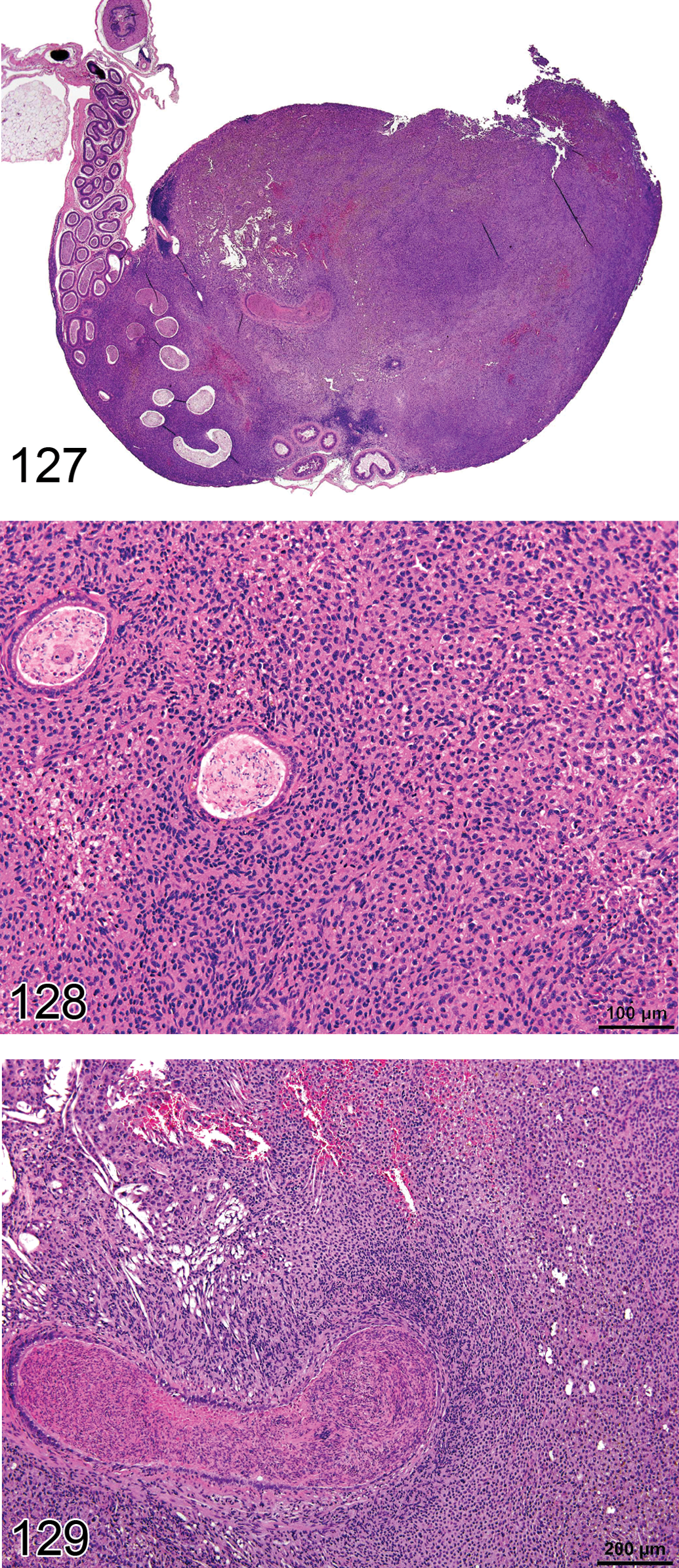
—Mouse epididymis. Sarcoma, histiocytic.
Accessory Sex Glands (Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland)
Introduction and Histology: Accessory Sex Glands (Figures 130–134)
There is marked variability in the anatomy, biology, function, and number of the male accessory sex glands among species. In mammals, the male accessory sex glands include the prostate, coagulating gland (anterior prostate), seminal vesicle, ampullary gland, the bulbourethral gland, the urethral gland, and the preputial gland. Although not all accessory sex glands are normally present in all species, with the exception of some transgenic rodents, all these glands are present in rats and mice. This article will be limited to the prostate, seminal vesicle, coagulating gland, and the bulbourethral gland. Evaluation of the accessory sex glands relies on adequate and consistent sampling of the various tissues and their different regions. This is particularly true in the mouse. Guidelines on dissection and trimming of these tissues can be found in Boorman, Elwell, and Mitsumori (1990); Kittel et al. (2004); and Suwa et al. (2001, 2002).
—Rat accessory sex organs. Normal anatomy and histology. Reprinted with permission from Suwa (2001).
—Rat seminal vesicle and coagulating gland. Dilation.
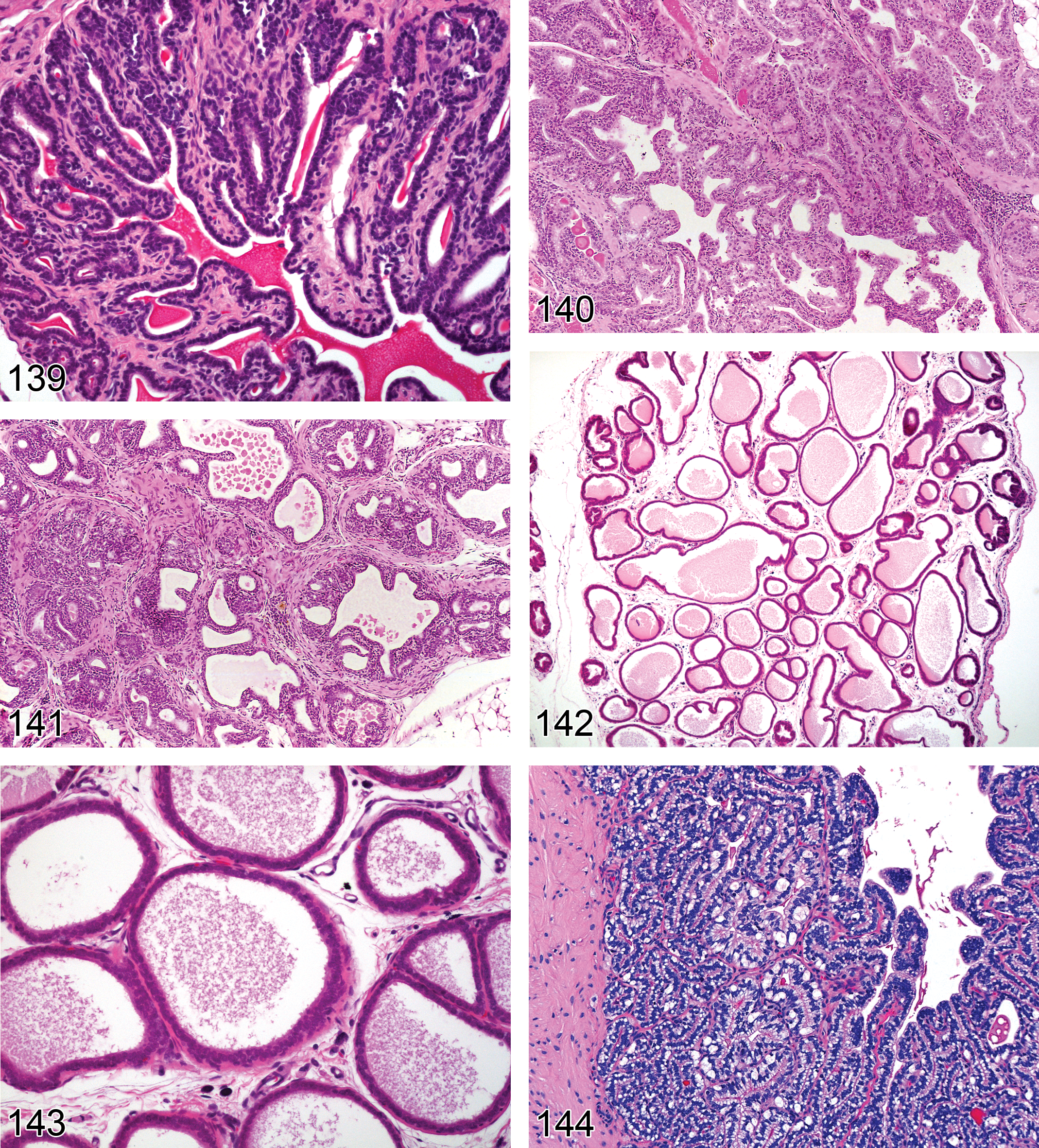
—Rat seminal vesicle. Atrophy. Higher magnification of Figure 138.
The excurrent duct system, comprising the rete testis, efferent ducts, epididymis, and the vas deferens, develops from the Wolffian (mesonephric) duct system. The seminal vesicle and ampullary gland also develop from lateral buds of this duct. However, the prostate, coagulating, and bulbourethral glands, along with the urethra and external genitalia, have a different embryological origin, developing from the urogenital sinus and urogenital tubercle. Hormonal regulation of the developing components of the tract also varies.
Prostate Gland
The prostate in rodents consists of a paired ventral portion and a paired dorsolateral portion, which together encircle the urethra. Each of these portions has distinct physiologic functions and responds differently to hormonal perturbations and toxic injury. The acini of the ventral prostate are lined by a single layer of cuboidal to tall columnar epithelium, which varies in its height depending on secretory activity. Additional cell types that are noted in low numbers in the prostatic epithelium include a nonsecretory basal cell, a macrophage-type cell, and a lymphocyte-type cell. The microscopic features of the dorsal portion of the dorsolateral prostate are similar to the coagulating gland, while the lateral portion most closely resembles the ventral prostate, with the addition of a distinctive brush border. Acini are surrounded predominantly by stromal smooth muscle cells and occasional fibrocytes. Depending on the secretory activity, all lobes are filled with secretory material that may appear flocculent. In the ventral lobes, the secretion is palely eosinophilic, while in the lateral lobes it stains intensely eosinophilic and in the dorsal lobes it stains intermediate between the other two lobes (Boorman, Elwell, and Mitsumori 1990; Lee and Holland 1987, Suwa et al. 2001, 2002).
Coagulating Gland
The coagulating gland is derived from the prostate and is sometimes referred to as the dorsocranial, cranial, or anterior prostate (Boorman, Elwell, and Mitsumori 1990; Creasy 1998; Radovsky, Mitsumori, and Chapin 1999). It comprises 5 to 6 tubular structures, which lie adjacent and parallel to the seminal vesicle and are often included in sections of seminal vesicle. The acini of the coagulating gland are lined by columnar epithelium that is arranged in branching, papillary projections, with a lumen containing eosinophilic, proteinaceous material similar to that seen in the dorsal prostate.
Seminal Vesicle
The seminal vesicle of rodents are paired, sac-like organs that have a muscular wall, surrounding a large central lumen. The mucosa has a honeycombed structure formed by complex folding to produce irregular anastomosing channels that communicate with the central cavity; thin primary folds of the mucosa also extend out into the vesicle lumen. The epithelium is composed of pseudostratified columnar cells in the mouse and simple columnar epithelium in the rat (Suwa et al. 2001, 2002). The secretory material from this gland is intensely eosinophilic and constitutes 50 to 80% of the ejaculate (Creasy and Foster 2002).
Bulbourethral Gland
The bulbourethral or Cowper’s gland is a small, paired organ that is difficult to evaluate grossly or dissect due to its location between the ischiocavernosus and bulbocavernosus muscles. In rats, the pear-shaped, tubuloaveolar gland has acini, lined by a single layer of basophilic pyramidal cells with closely packed secretory granules (Boorman, Elwell, and Mitsumori 1990) that may have a filamentous or reticular texture (Dünker and Aumüller 2002). The secretion stains palely basophilic and is made up of acidic and neutral mucosubstances. In mice, this gland is described as having a highly arborized arrangement of acini lined by tall columnar epithelium that fills the acinar lumen (Dünker and Aumüller 2002).
During ejaculation, secretions from the prostate, seminal vesicle, and coagulating gland are added to the sperm in the vas deferens. As the sperm enter the penile urethra, additional secretions from the bulbourethral gland are added (Creasy 1998). Together, the secretions from these glands form the seminal fluid. Although the composition of the seminal fluid varies between species, its main role is to extend the viability of sperm during their transit through the female genital tract (Luke and Coffey 1994). Species differences also exist in the coagulation of semen. Most muroid rodents form copulatory plugs (Hartung and Dewsbury 1978) derived from the secretions of the seminal vesicle, coagulating gland, and bulbourethral gland. For example, in rats, the semen is composed of a fluid phase, which contains the sperm, followed by a solid phase, which forms the copulatory plug, which functions to retain the sperm in the vagina and enhances fertilization. In the rat, the coagulation enzyme, vesiculase, is produced by the coagulating gland, converting procoagulase (secreted by the seminal vesicle) into coagulase. This process requires a cofactor produced by the bulbourethral gland (Creasy 1998).
Congenital Lesions: Accessory Sex Glands
Aplasia: Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Synonym: agenesis
Pathogenesis: failure of the primordium during embryonic development (Shappell et al. 2004)
Diagnostic feature Organ/organs are not evident on macroscopic or microscopic examination
Differential diagnosis Hypoplasia: incomplete absence of the organ
Comments:
Hypoplasia: Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Pathogenesis: incomplete development of the organ that differs from aplasia in that the organ is not completely absent, but there is inadequate development or underdevelopment of the organ (Shappell et al. 2004)
Diagnostic features Grossly small (taking into account body size and sexual maturity) Lobes are often malformed Decreased secretion
Differential diagnosis Atrophy: Overall structure is normal but the epithelium is atrophic
Comments: As with aplasia, hypoplasia of the accessory organs is rare. Prostatic hypoplasia has been reported in 5α-reductase type 2 knockout mice and other transgenic rodents (Li et al. 2001). Hypoplasia of the ventral and dorsolateral prostate, and seminal vesicles have been reported in male Sprague-Dawley rats exposed
Ectatic Changes: Accessory Sex Glands
Dilation, Acinar/Vesicle (Figures 135–137): Prostate, Coagulating Gland, Bulbourethral Gland, Seminal Vesicle
Synonyms: acinar distension, cystic dilation, cysts, distended vesicle
Modifier: cystic
Pathogenesis: glandular acini or seminal vesicle may become distended with secretion due to increased production of secretion (hyperandrogenism) or inhibited release of secretory product (anti-adrenergic drugs, age-related sexual inactivity). Cystic dilation may occur secondary to obstructive, fibrosing inflammation in the seminal vesicles and coagulating gland
Diagnostic features Grossly, the gland may have focal distension or the entire gland may be enlarged due to accumulation of secretory fluids May be associated with an increase in organ weight Diffuse or focal distension of acini/vesicles with secretory fluid May form mucinous cysts (prostate and bulbourethral gland) Associated with severe thinning of the acinar/vesicle walls (cystic dilation) Often accompanied by degradation of the secretory material within the acini/vesicles
Comments: Dilation of the seminal vesicle and coagulating gland, often associated with degradation of the secretory contents and severe thinning of the vesicle wall, occurs in aging rats and mice (Faccini, Abbott, and Paulus 1990; Frith and Ward 1988; Radovsky, Mitsumori, and Chapin 1999; Suwa et al. 2001, 2002; Yamate et al. 1990), and it has been suggested that the accumulation of secretory product may be due to sexual inactivity and declining testosterone levels (Suwa et al. 2001, 2002). The change is also sometimes associated with chronic inflammation of the vesicle wall and may be secondary to ascending urinary tract infections, which are relatively common in male mice. Mucinous cystic acini (cysts) have also been described in the prostate of aging rats and mice (Suwa et al. 2001, 2002). Similarly, dilation of the bulbourethral glandular acini or ducts, progressing to cystic atrophy may be observed in aging rats and mice where enlarged bulbourethral glands are often sampled as gross abnormalities (Kiupel, Brown, and Sundberg 2000; Wardrip et al. 1998). Diffuse acinar or vesicle dilation due to increased secretory product and accompanied by organ weight increase has been reported as a drug-induced finding, and has also been described with hyperprolactinemia and hyperandrogenism (Coert et al. 1985; Van Coppenolle et al. 2001).
Degenerative Changes: Accessory Sex Glands
Atrophy (Figures 138–143): Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Modifiers: acinar, vesicle, cystic
Pathogenesis: atrophy of the accessory sex organs is generally associated with decreased androgen status (Gordon, Majka, and Boorman 1996; Liu and Hurtt 1996; Stolte 1993; Sugimura, Cunha, and Donjacour 1986; Suwa et al. 2002). Decreased testosterone stimulation results in decreases in organ weight caused by decreased secretory activity (Creasy 1998)
Diagnostic features Reduction in gross size and weight Reduced acinar/vesicle lumina and crowding of intra-acinar/vesicle epithelial folds Reduced or absent luminal secretion Acini or vesicles are lined by lower or flattened, epithelial cells with decreased or no secretory droplets in them With severe atrophy the stroma is increased May be accompanied by increased epithelial pigment (lipofucsin) Cystic pattern Dilated acini or vesicles lined by mostly flattened epithelium Significant epithelial loss may be apparent Acini/ducts may contain proteinaceous fluid, minimal cellular debris or appear empty (Faccini, Abbott, and Paulus 1990; Gordon, Majka, and Boorman 1996)
Differential diagnosis Hypoplasia: glandular tissue shows normal structure but the entire organ is small and often malformed
Comments: Commonly seen with chronic androgen depletion, e.g., castration, aging. Atrophic alterations in accessory sex glands due to aging are generally not as prominent as those secondary to castration (Boorman, Elwell, and Mitsumori 1990). Epithelial apoptosis in an otherwise normal appearing epithelium may be evident in the early stages of glandular atrophy. It is important to evaluate organ size and secretory activity relative to sexual maturity and overall body size/weight. When evaluating secretory content and atrophy of the accessory sex glands, it is important to ensure consistent sampling of the glands (Suwa et al. 2001, 2002).
Vacuolation, Epithelial (Figures 144 and 145): Prostate, Seminal Vesicle, Coagulating Gland
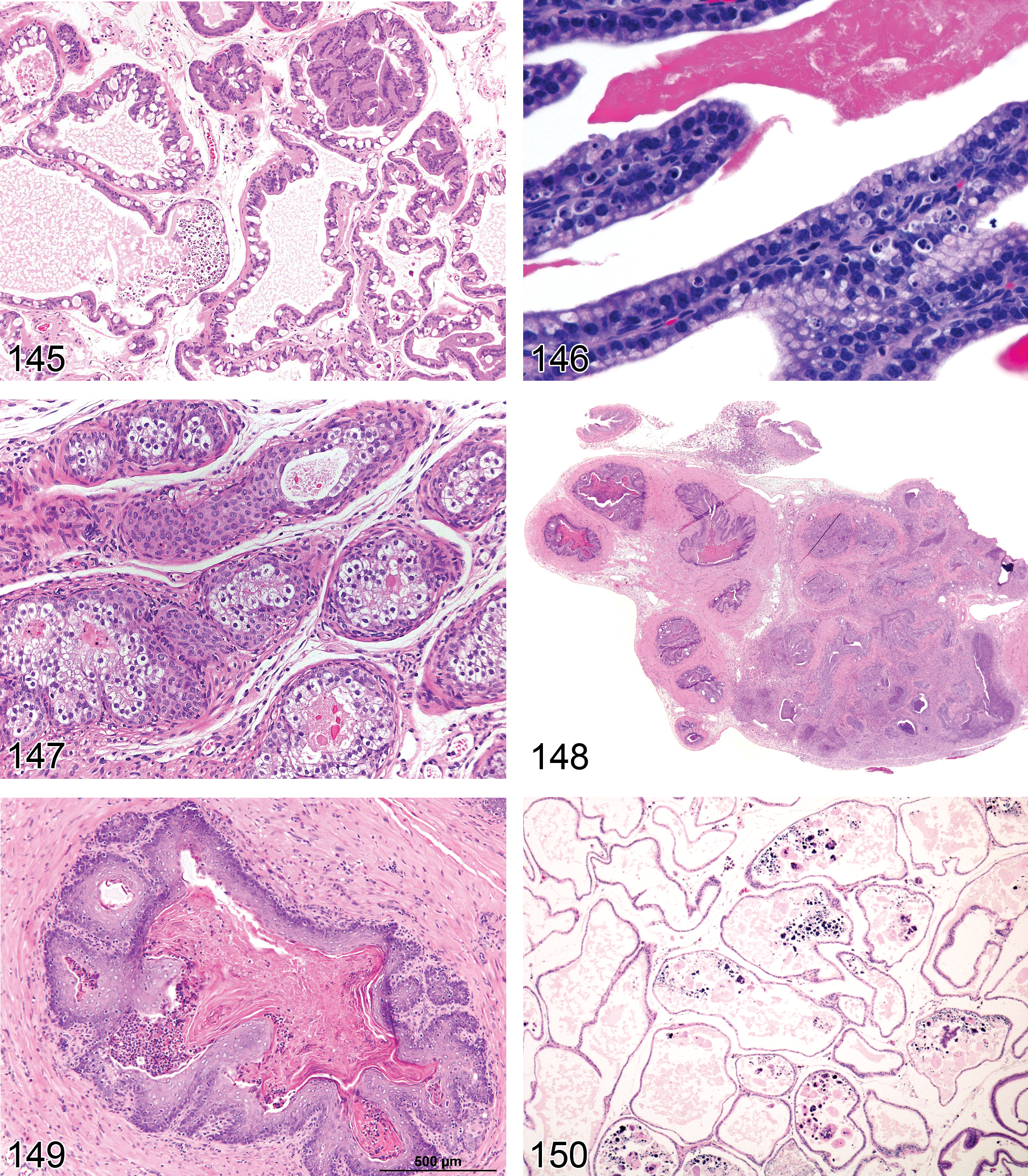
—Rat prostate. Vacuolation, epithelial.
Synonyms: vacuolar degeneration, vesicular or hydropic degeneration
Modifiers: macrovesicular, microvesicular
Pathogenesis: vacuolation of the epithelium may result from a number of degenerative changes including accumulation of fluids, lipids, phospholipids, glycoproteins
Diagnostic features Vacuolation may be micro or macrovesicular depending on the cause The vacuoles generally displace the nucleus and may distort cell shape; they may be apical, basal, or both
Differential diagnoses Fixation artifact: suboptimal fixation can result in variable numbers of vacuoles in the epithelium Apoptosis: apoptotic debris present within the vacuoles
Comments: Phospholipidosis can sometimes affect the epithelium of the prostate and seminal vesicles and appear as diffuse vacuolation
Single cell necrosis, Epithelial (Figure 146): Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Synonym: apoptosis
Pathogenesis: single cell necrosis (apoptosis) is considered a form of programmed cell death resulting from a cascade of intracellular signals and events (Shappell et al. 2004)
Diagnostic features Individual cells that exhibit nuclear fragmentation or condensation and have shrunken, eosinophilic cytoplasm Remaining epithelial cells appear normal No signs of inflammation May be accompanied by reduced secretory content
Comments: Apoptotic cell death of the sex organs may be seen with acute androgen depletion, but generally progresses to atrophy with persistent androgen depletion (Shappell et al. 2004). Apoptosis may be confirmed through the use of terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) and other apoptotic markers. When evaluating secretory content of the accessory sex glands, it is important to ensure consistent sampling of the glands (Suwa et al. 2001, 2002).
Necrosis: Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Pathogenesis: coagulative necrosis may be caused by hypoxia and/or various toxins (Shappell et al. 2004). May accompany purulent inflammation in a urogenital infection
Diagnostic features Hypereosinophilic cells that retain distinct cell outlines but have loss of nuclear detail (Shappell et al. 2004) Necrotic areas typically involve multiple cells and can sometimes span large areas of contiguous cells Necrotic areas may be bordered by inflammatory cell infiltrates, hemorrhage, and/or edema
Comments: Coagulative necrosis has been reported occasionally in the prostate and seminal vesicle of older transgenic animals, most likely secondary to ischemia (Shappell et al. 2004). Areas of necrosis may also be associated with purulent inflammation in urogenital infections and with tumors.
Metaplasia (Figures 147–149): Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Modifiers: mucinous, squamous cell, transitional cell
Pathogenesis: mucinous, squamous cell, or transitional cell differentiation of the normal secretory epithelium may result from inflammatory mediated tissue damage, nutritional deficiencies, and hormonal perturbations
Diagnostic feature
Replacement of normal prostatic epithelium (basal and luminal secretory cells) by: mucinous epithelium: tall columnar mucin producing cells squamous epithelium: stratified squamous, keratinizing cells transitional epithelium: transitional (urothelial) cells
Comments: Squamous metaplasia often accompanies severe inflammation of the sex glands. Metaplastic changes have also been noted in the prostate in transgenic mice and in mice treated with estrogenic compounds (Bierie et al. 2003). Treatment with estrogens in male mice results in squamous metaplasia of the coagulating gland (anterior prostate), dorsal prostate, and seminal vesicle. Proliferation is most extensive in the coagulating gland and seminal vesicle and often results in transformation of many of the alveoli into small keratinized nodules (Bern 1952). Similar changes occur in male rats treated with estrogen (Alison et al. 1997; Arai 1968; Arai, Suzuki, and Nishizuka 1977; Bern 1952; Bierie et al. 2003; Bosland 1992; Cunha et al. 2001; Heywood and Wadsworth, 1980; Kawamura et al. 2000; Rehm et al. 2001). Avitaminosis A in rats and mice (experimentally induced) also results in squamous metaplasia of the accessory sex glands, starting as a layer of squamous epithelium overlying the original luminal epithelium of the seminal vesicle, coagulating gland, ductus deferens, and dorsal prostate. Intraepithelial keratinization and the formation of “keratin pearls” are common (Bern 1952).
Miscellaneous Changes: Accessory Sex Glands
Amyloid: Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Synonym: amyloidosis
Pathogenesis: age-related degenerative condition in mice
Diagnostic features Accumulation of pale eosinophilic extracellular material Perivascular, interstitial Green birefringence using polarized light with Congo Red stain May form thin bands or extensive sheets
Comments: Amyloidosis is an age-related, incidental, spontaneous, often systemic disease characterized by the extracellular deposition of polypeptides (often serum amyloid-associated protein or immunoglobulin fragments) appearing in routine section as a lightly eosinophilic amorphous material. It is common in aged mice and rare in rats. Confirmation of the deposits as amyloid can be accomplished with light microscopy using special stains such as Congo Red. Amyloid appears apple green under polarized light with this stain.
Concretions (Figures 150 and 151): Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
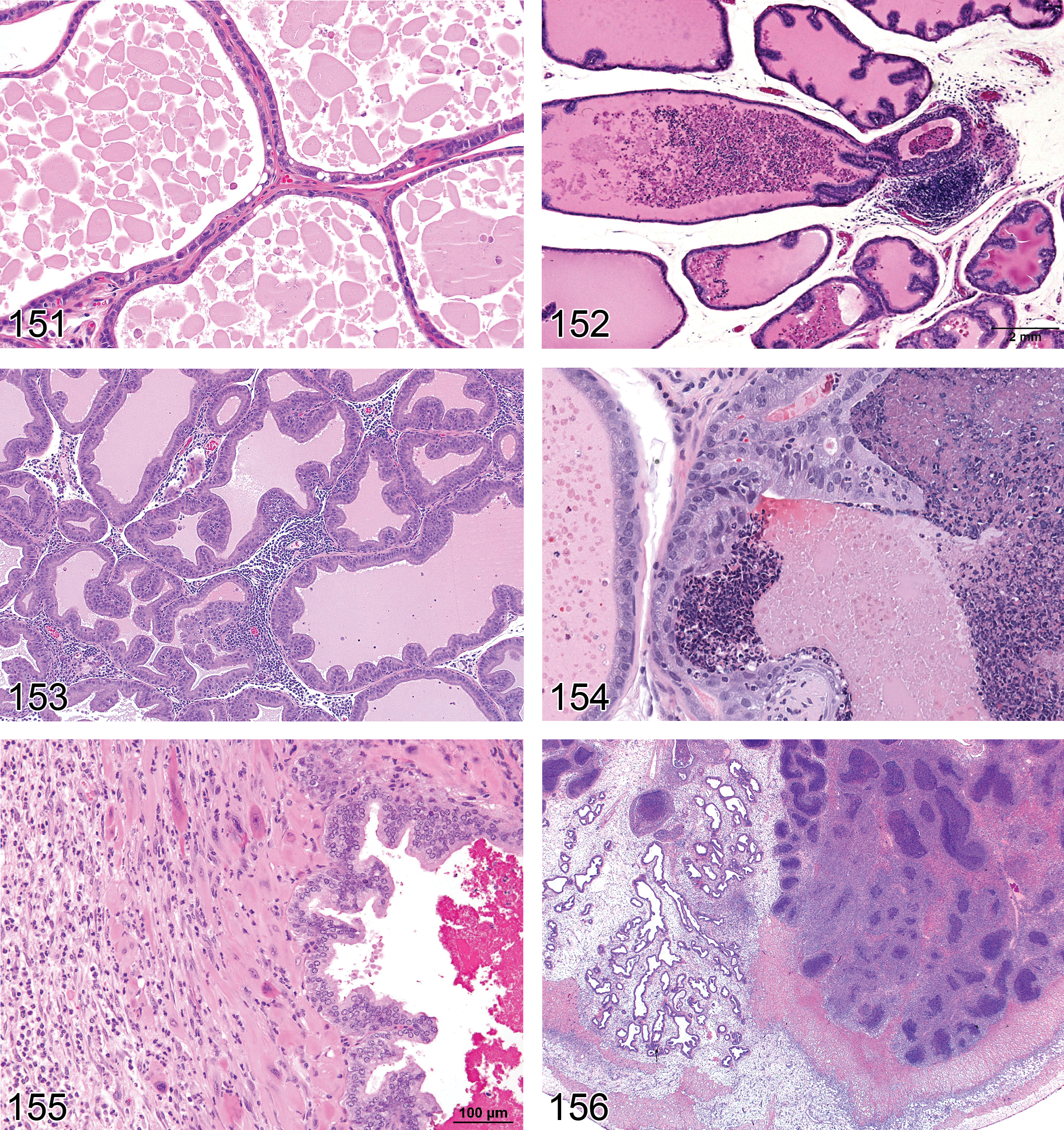
—Rat prostate. Concretions.
Synonyms: Mineralized deposits, corpora amylacea
Pathogenesis: Small hyaline masses of degenerated cells that develop into compact concretions due to pressure dehydration and “compaction” of dead cells. May appear mineralized
Diagnostic features Rounded, homogenous, or concentrically laminated bodies Usually eosinophilic but may have a basophilic tinge at the outer edges These bodies are found in the lumen of the prostate (Jones and Hunt 1983)
Comments: Concretions are a very common age related change. They are generally associated with acinar atrophy
Inflammatory Changes: Accessory Sex Glands
Introduction
A step-wise approach is recommended for diagnosing inflammatory conditions. Minimal and/or focal aggregates of inflammatory cells should be referred to as “inflammatory cell infiltrates” with the additional use of modifiers to specify the predominant cell type involved (lymphocytic, neutrophilic, lymphoplasmacytic, etc.). If the inflammatory infiltrate is more diffuse and accompanied by other classic markers of inflammation such as edema, congestion, or cellular degeneration/necrosis, then the term inflammation should be used and appropriate modifiers may be used to specify the character of the inflammation (e.g., lymphocytic, neutrophilic, purulent, etc.).
Infiltrate, Inflammatory Cell (Figures 152–154): Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Synonyms: lymphoid infiltrate, mononuclear cell infiltrate, inflammation
Modifiers: lymphocytic, neutrophilic, plasmacytic, mixed inflammatory cell
Pathogenesis: focal extravasation of inflammatory cells into the interstitial tissue or into the acinar lumen, common background finding of unknown etiology
Diagnostic features Small, focal aggregates of inflammatory cells, generally lymphocytic in the interstitium or neutrophilic in the acinar lumens Often accompanied by denuded cells or cellular debris when intraluminal
Differential diagnosis Inflammation: more diffuse inflammatory infiltrate with additional features such as edema, tissue damage, reactive hyperplasia, epithelial metaplasia
Comments: Focal lymphocytic aggregates are very common as a background finding in rats and mice, particularly in the prostate. In B6C3F1 mice, lymphocytic infiltrate was present in the prostate of over 30% of mice and in the coagulating gland of almost 20%, but the pattern of inflammatory lesions varies with the species and the lobe (Suwa et al. 2001, 2002). Focal neutrophilic inflammatory infiltrates within the lumen and/or interstitium are also relatively common.
Inflammation (Figures 155–157 ): Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
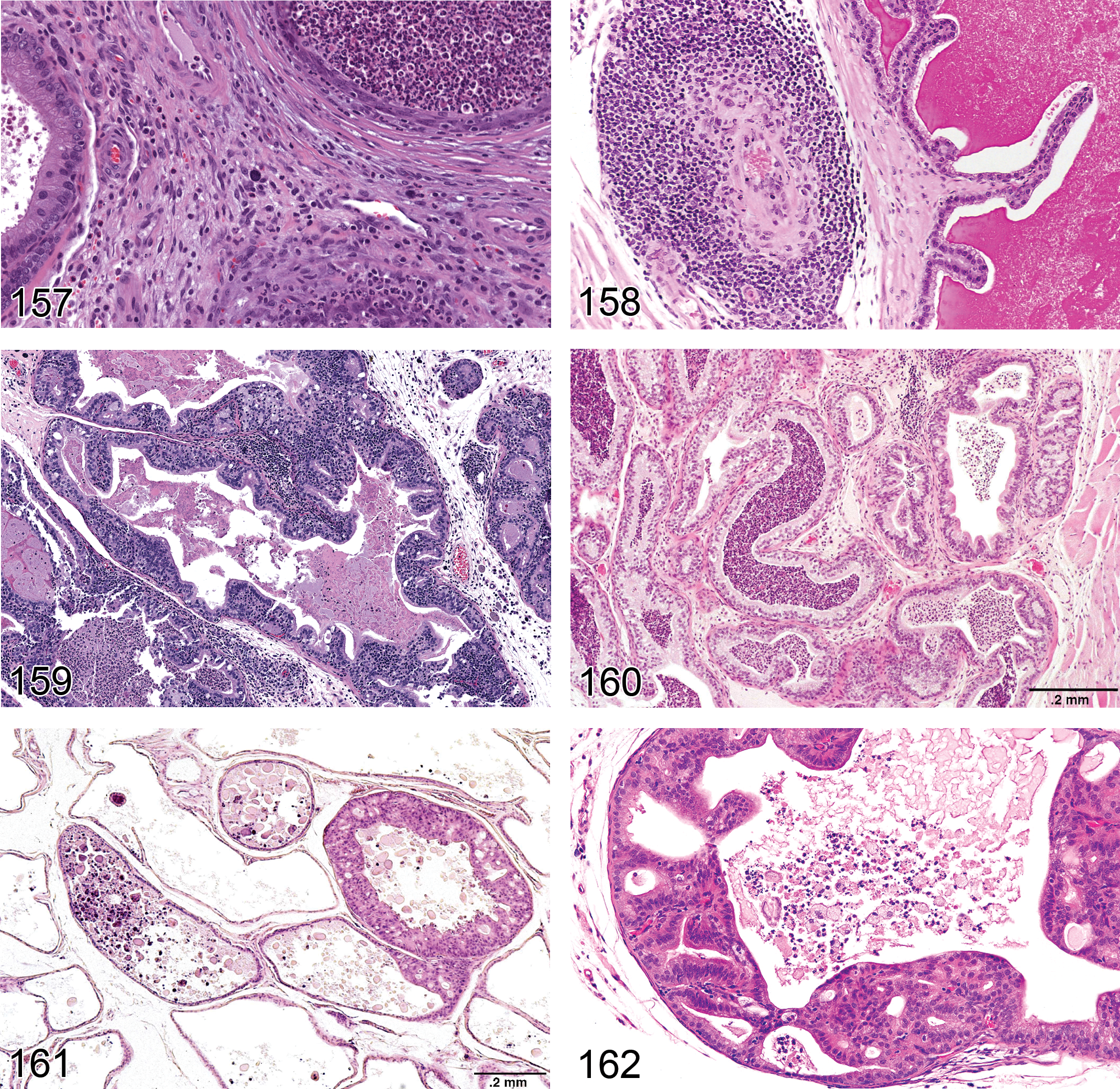
—Rat prostate. Inflammation.
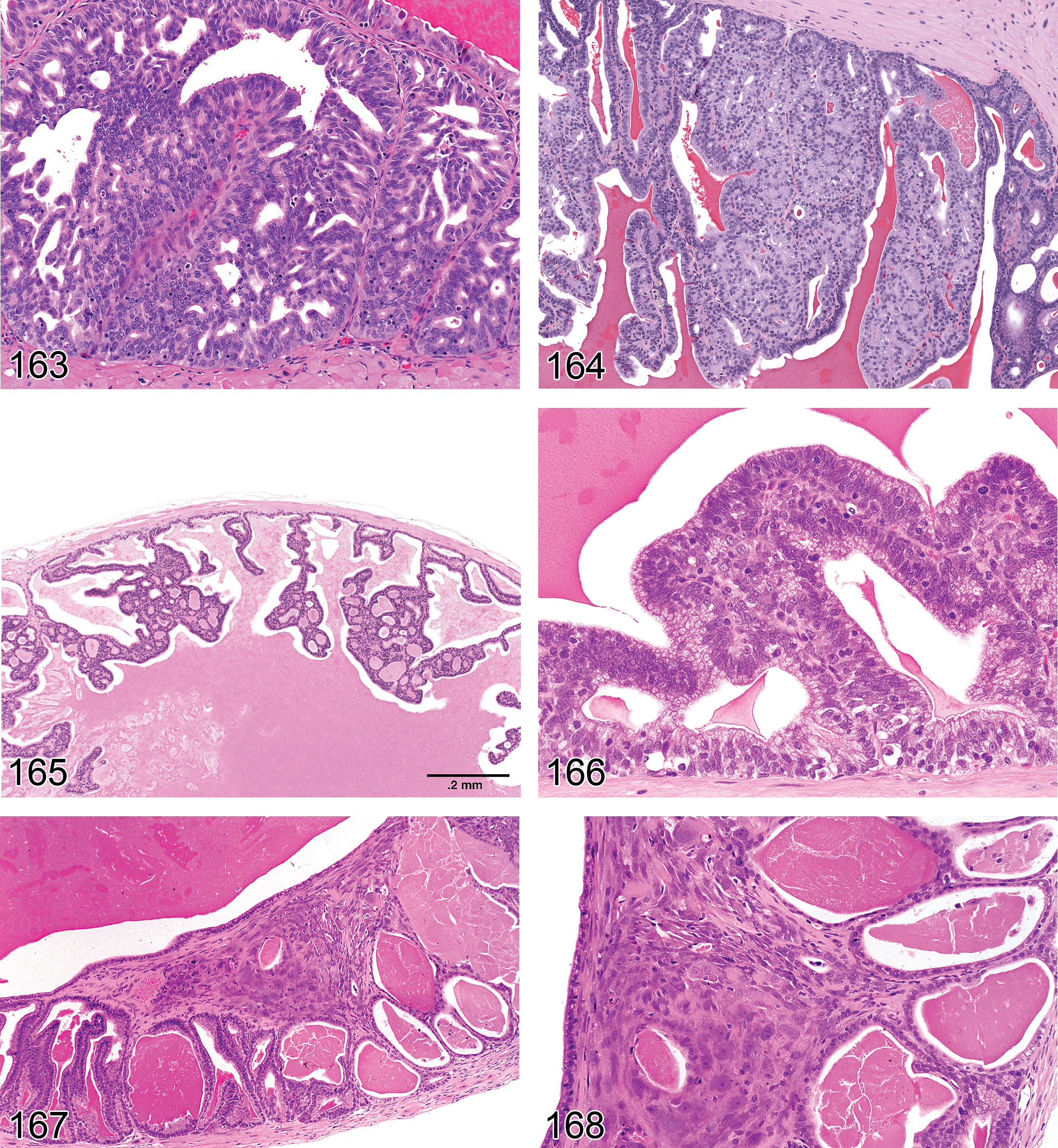
—Rat seminal vesicles. Hyperplasia, atypical.
Synonyms: prostatitis, abscesses, seminal vesiculitis, granulomatous inflammation
Modifiers: neutrophilic, lymphocytic, lympho-plasmacytic, mixed cell. Other modifiers include purulent, or granulomatous
Pathogenesis: generally a response to tissue injury or necrosis, which is often associated with urogenital bacterial infections
Diagnostic features Focally extensive or diffuse accumulation of inflammatory cells Evidence of tissue injury and often reactive hyperplasia May be associated with squamous metaplasia and vacuolar degeneration of the epithelium May progress to abscesses or pyogranulomatous response Increased connective tissue stroma and/or fibrosis with persistent inflammation Swelling or enlargement of the perineum may be noted grossly due to enlarged, abscessed bulbourethral glands (Kiupel, Brown, and Sundberg 2000; Sebesteny 1973)
Differential diagnosis Inflammatory cell infiltrate: focal, minimal and without evidence of tissue injury
Comments: Inflammation of the secondary sex organs is a common background finding in rats and mice, which increases with age (Boorman, Elwell, and Mitsumori 1990). In the prostate, active inflammation often includes a mix of neutrophils and small monocytes, may progress to abscessation, and may be accompanied by reactive epithelial proliferation and nuclear atypia and mineralized deposits (Boorman, Elwell, and Mitsumori 1990; Shappell et al. 2004). Severe inflammation may be secondary to ascending urogenital infections, which can sometimes be caused by injuries due to fighting with cage mates or following experimental infections. Bulbourethral abscesses have also been associated with bacterial infections in mice naturally and experimentally infected with
Fibrosis, Stromal: Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Pathogenesis: May be associated with administration of estrogenic compounds or loss of androgen support. Also secondary to persisting inflammation. In transgenic animals, stromal fibrosis may result from alterations in growth factors and/or extracellular matrix (Shappell et al. 2004)
Diagnostic features Relative increase in ratio of stromal tissue to glandular tissue (estrogenic stimulation) Discrete accumulation of interstitial collagen or wide bands of dense connective tissue Fibrosis is often associated with active or persisting inflammation The normal anatomy may be completely distorted, resulting in deposits of dense eosinophilic collagen
Differential diagnosis Amyloid: fibrillar eosinophilic, Congo red positive, material deposited in perivascular and periacinar locations
Comment: In both rats and mice, chronic estrogen administration results in decreased secretion, epithelial atrophy accompanied by hyperplasia of the prostatic stroma and squamous metaplasia of the prostate and coagulating gland (Heywood and Wadsworth, 1980). In addition, severe chronic inflammation of the accessory sex organs can result in large areas of fibrosis.
Vascular Changes: Accessory Sex Glands
Inflammation, Vascular/Perivascular (Figure 158): Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland
Synonyms: vasculitis, arteritis, perivascular inflammation, periarteritis, polyarteritis nodosa
Pathogenesis: generally a feature of spontaneous, age-related, systemic “polyarteritis nodosa,” which is a progressive degenerative lesion of the walls of small arterioles. May also be induced or exacerbated by vasoactive drugs and chemicals through over distension of blood vessel walls or by systemic hypertension
Diagnostic features Inflammatory cells (usually lympho-plasmacytic) surrounding blood vessels (small arterioles) and often infiltrating vascular walls Generally accompanied by medial hypertrophy, fibrinoid necrosis (accumulation of fibrillar, eosinophilic material in the media), or hyalinization (necrosis) of affected vessels
Differential diagnosis Inflammatory cell infiltrate: the inflammatory infiltrate is generally lymphocytic, does not involve the blood vessel wall, and is not accompanied by hypertrophy or degenerative changes in the blood vessel wall Inflammation: the inflammatory infiltrate is more diffuse and not centered on the blood vessels
Comments: Age-related polyarteritis nodosa is much more common in the mouse than in the rat. The blood vessels of the mesentery and testis are more sensitive to spontaneous and induced changes in the vascular bed, but the secondary sex organs may also be affected. Vascular injury has been reported in the testis, epididymis, and accessory sex glands of the rat after administration of cadmium chloride (Waites and Setchell 1966; also see INHAND Nomenclature of the Cardiovascular System).
Angiectasis: Prostate, Seminal Vesicle, Coagulating Gland, Bulbourethral Gland, Urethral Glands
Pathogenesis: angiectasis may be spontaneous or secondary to occlusion, thrombosis, or hypertension
Diagnostic feature Irregular, dilated vessels lined by a single layer of normal endothelium
Comments: Angiectasis occurs in the cavernous blood vessels surrounding the urethra and periurethral glands in aging mice and is often sampled as a gross abnormality or sometimes mistaken as enlarged bulbourethral glands.
Non-neoplastic Proliferative Lesions: Accessory Sex Glands
Introduction
The terminology and diagnostic criteria provided for proliferative lesions of the accessory sex organs are based largely on those previously published by WHO/IARC, SSNDC, and International Life Sciences Institute (ILSI) (Alison et al. 1997; Bosland et al. 1998; Mitsumori and Elwell 1994; Rehm et al. 2001). There are numerous additional reviews of prostatic, seminal vesicle, and coagulating gland tumors in rodents, detailing their general features and incidence (Boorman, Elwell, and Mitsumori 1990; Bosland 1992; Faccini, Abbott, and Paulus 1990; Frith and Ward 1988; Gordon, Majka, and Boorman 1996; Mitsumori and Elwell 1988; Radovsky, Mitsumori, and Chapin 1999; Rehm et al. 2001; Squire et al. 1978).
Since proliferative lesions of the bulbourethral gland have only been described in the SV40 TAG transgenic mouse (Shibata et al. 1996), they have not been included in this classification.
Hyperplasia, Reactive (Figures 159 and 160) Prostate, Coagulating Gland, Seminal Vesicle
Species: mouse, rat
Synonyms: hyperplasia, regenerative hyperplasia
Pathogenesis: reparative hyperplasia of acinar epithelium in response to degeneration and inflammation, which is generally caused by a urogenital infection. Particularly common in mice
Diagnostic features Accompanied by inflammation Mostly in dorsolateral and ventral prostatic lobes, less common in the anterior lobe (coagulating gland) or seminal vesicle Focal, multifocal, or diffuse lesion Simple thickening of the epithelium to 2 to 6 or more cell layers; may be accompanied by squamous metaplasia Cells are cuboidal to columnar with increased cytoplasmic basophilia Some cellular atypia may be present Pseudoglandular structures may be present
Differential diagnoses Hyperplasia, atypical: focal lesion without evidence of inflammation Adenoma: compression of adjacent tissue with disruption of normal glandular architecture
Comments: Reactive hyperplasia is the most common type of hyperplasia seen in the accessory sex organs. The presence of accompanying inflammation, which is often suppurative, is the most reliable differential diagnostic criterion to distinguish it from preneoplastic focal hyperplasia (Bosland 1987d; Faccini, Abbott, and Paulus 1990; Frith and Ward 1988; Gordon, Majka, and Boorman 1996; Mitsumori and Elwell 1988; Radovsky, Mitsumori and Chapin 1999; Rehm et al. 2001; Reznik et al. 1981; Reznik 1990).
Hyperplasia, Functional: Prostate, Coagulating Gland, Seminal Vesicle
Species: mouse, rat
Synonyms: adaptive hyperplasia, simple hyperplasia
Pathogenesis: enlargement of the organ caused by hypertrophy and hyperplasia of the epithelium in response to increased demand or hormonal stimulation
Diagnostic features Diffuse or (multi)focal simple thickening of the epithelium with cellular crowding Diffuse change is generally associated with increased organ weight and gross enlargement In the prostate, focal functional hyperplasia is commonly seen in the periphery of the ventral lobe, where it is often associated with decreased secretion Foldings of the lining epithelium may extend into alveolar lumina Cells are cuboidal to columnar with increased cytoplasmic basophilia No cellular atypia or disturbance of architecture Not obliterating the acinar lumen of prostatic acini
Differential diagnoses Hyperplasia, atypical: focal lesion with multilayered epithelium with cell atypia Hyperplasia, reactive: accompanied by inflammation Adenoma: Obliteration of at lease one acinar lumen (prostate). Compression of adjacent tissue with disruption of normal glandular architecture
Comments: Functional hyperplasia is often difficult to detect since the tissue appears essentially normal and there is just more of it. The diffuse change is most commonly seen in response to administration of exogenous androgens, which results in enlargement, weight increase, diffuse hyperplasia, and an increase in secretion in all the accessory sex organs (Creasy 2008). Multifocal functional hyperplasia of the ventral prostate can be seen as a spontaneous change (Bosland 1987d).
Hyperplasia, Atypical (Figures 161–166): Prostate, Coagulating Gland, Seminal Vesicle
Species: mouse, rat
Synonym: focal hyperplasia, prostatic intraepithelial neoplasia (PIN), adenomatous hyperplasia, dysplasia
Pathogenesis: altered proliferation of glandular epithelium, that may be classified as preneoplastic, if sufficient evidence exists, e.g., accompanied by an increased incidence of accessory sex gland tumors or demonstrated progression of the lesion
Diagnostic features Can be seen in the ventral prostate, dorsolateral prostate, coagulating gland (anterior prostate) or seminal vesicle, especially in genetically engineered mice (GEM) and some types of rats given testosterone Focal or multifocal lesion (not diffuse) involving one or a few adjacent acini or seminal vesicular epithelium Focal hyperchromatic lesion which has atypical cytological features compared with reactive hyperplasia. Usually without inflammation but in GEM can be accompanied by a proliferative stromal reaction Gradual transition from normal epithelium to focal hyperplastic areas or, at times, more abrupt changes No disruption of normal glandular architecture of the prostate but disturbance of architecture in the seminal vesicle Not obliterating the acinar lumen
Differential diagnoses Hyperplasia, reactive: accompanied by inflammation. Normal cellular features without cellular atypia Adenoma: generally larger than atypical hyperplasia, obliteration of at least one acinar lumen or compression of adjacent tissues with disruption of normal glandular architecture
Comments: Commonly seen in GEM prostate adenocarcinoma models and chemically induced rat models. Also occurs as spontaneous lesion in rats and mice (Bosland 1987d; Reznik et al. 1981; Shappell et al. 2004; Tamano et al. 1996.) The diagnosis of atypical hyperplasia is mainly based on the occurrence of multilayered normal epithelium in a few adjacent acini of otherwise normal glands without architectural disturbance. In the seminal vesicles, there is focal disturbance of architecture and multilayering of the epithelium. There appears to be a morphologic continuum between atypical hyperplasia and benign adenoma of the prostate and the distinction between the two is not always clear. If differentiation between atypical hyperplasia and adenoma cannot be made on the basis of other criteria, a proliferative lesion obliterating at least one acinar lumen is interpreted to be an adenoma. Atypical hyperplasia is considered a preneoplastic lesion in mouse and rat prostate cancer models and is sometimes described as PIN, especially in GEM (Park et al. 2002; Shappell et al. 2004; Shibata et al. 1996). Atypical hyperplasia of the dorsolateral prostate and ampullary gland is known to occur following treatment with carcinogens, usually in combination with testosterone. It differs somewhat from that of the ventral prostate: the lesion is less uniform and is characterized by small acini with a single layer of atypical cuboidal to somewhat flat cells with pale cytoplasm and hypochromatic nuclei, the cellular polarity is often disturbed and no sign of secretory activity is seen, some necrosis and thickening of periacinar fibromuscular tissue is often found.
Mesenchymal Proliferative Lesion (Figures 167 and 168): Prostate, Seminal Vesicle
Species: mouse
Synonyms: vegetative lesion, mesenchymal tumor, decidual-like reaction, undifferentiated sarcoma, leiomyosarcoma, leiomyoblastoma, carcinosarcoma
Pathogenesis: not established
Diagnostic features Generally subepithelial in location Cells of two types are present: large epithelioid cells in compact islands and spindle cells located more peripherally Epithelioid cells have distinct borders, eosinophilic, homogeneous, or fibrillar cytoplasm, and eosinophilic granules may be present Nuclei of epithelial cells are eccentric, often with pleomorphic or bizarre outline, and prominent nucleoli Spindle cells are fibrocyte-like or smooth muscle-like Mononuclear inflammatory cell infiltrate is invariably present, usually peripherally PAS-positive granules may be present within the large epithelioid cells
Differential diagnoses Aggregate, granular cells: granular cells without cellular atypia and not accompanied by spindle cells Tumor, granular cell, benign: large or variable-sized epithelioid or fusiform eosinophilic cells with PAS-positive cytoplasmic granules are present
Comments: The definitive nature of this lesion is still under debate (Karbe 1999). One view regards the lesion as a benign tumor based on the potential to be locally invasive. The other considers the lesion to be a decidual-like reaction because of the remarkable morphological similarities to the uterine decidual reaction, which also can appear to be locally invasive, but is still considered a hyperplastic lesion. A general review of the controversy is provided by Karbe et al. (1998). The lesion is primarily spontaneous and strain-specific, being found most frequently in Swiss-derived mice. Thus, it has been seen predominantly in Swiss Webster, NMRI, and CD1 strains, but not as yet in B6C3F1 mice. Identical bladder lesions have been described as spontaneous lesions (in association with surgically implanted glass or paraffin pellets, particularly in the vicinity of sutures used to close the bladder after implantation). Whether these lesions can be induced by chemical agents is not presently known, although an increased incidence has been noted with various combinations of endogenously administered estrogens and progestogens and with an insecticide (Butler, Cohen, and Squire 1997). Primary lesions of similar morphology have been identified in reproductive organs of both male and female mice, namely the prostate, seminal vesicle, and uterus. Coagulating gland has been included as a potential site with a similar developmental origin as prostate and seminal vesicle (Chandra and Frith 1991; Halliwell 1998; Karbe 1987; Karbe et al. 1998; Kaspareit and Deerberg 1987; Mitsumori and Elwell 1994; Rehm et al. 2001).
Neoplastic Proliferative Lesions: Accessory Sex Glands
Introduction
Spontaneous neoplasms of the accessory sex organs are uncommon, with the prostate being the most commonly affected. Prostatic tumors have been induced by a number of chemicals.
The terminology and diagnostic criteria provided for tumors of the accessory sex organs are based largely on those previously published by WHO/IARC and SSNDC (Alison et al. 1997; Bosland et al. 1998; Mitsumori and Elwell 1994; Rehm et al. 2001). There are numerous additional reviews of prostatic, seminal vesicle, and coagulating gland tumors in rodents, detailing their general features and incidence (Boorman, Elwell, and Mitsumori 1990; Bosland 1992; Faccini, Abbott, and Paulus 1990; Frith and Ward 1988; Gordon, Majka, and Boorman 1996; Mitsumori and Elwell 1988; Radovsky, Mitsumori, and Chapin 1999; Rehm et al. 2001; Squire et al. 1978).
Adenoma (Figures 169–175) : Prostate, Coagulating Gland, Seminal Vesicle
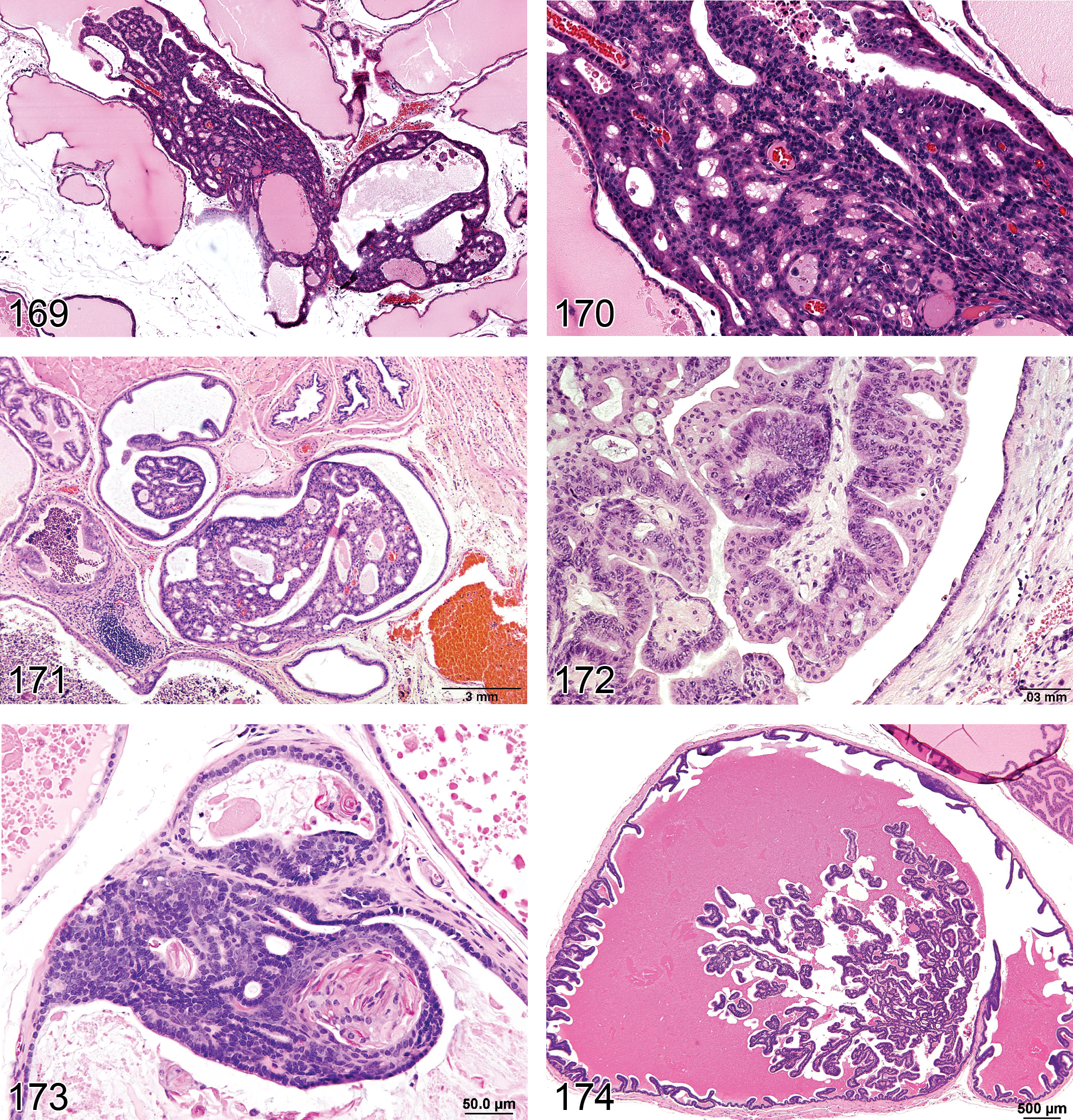
—Mouse prostate. Adenoma.
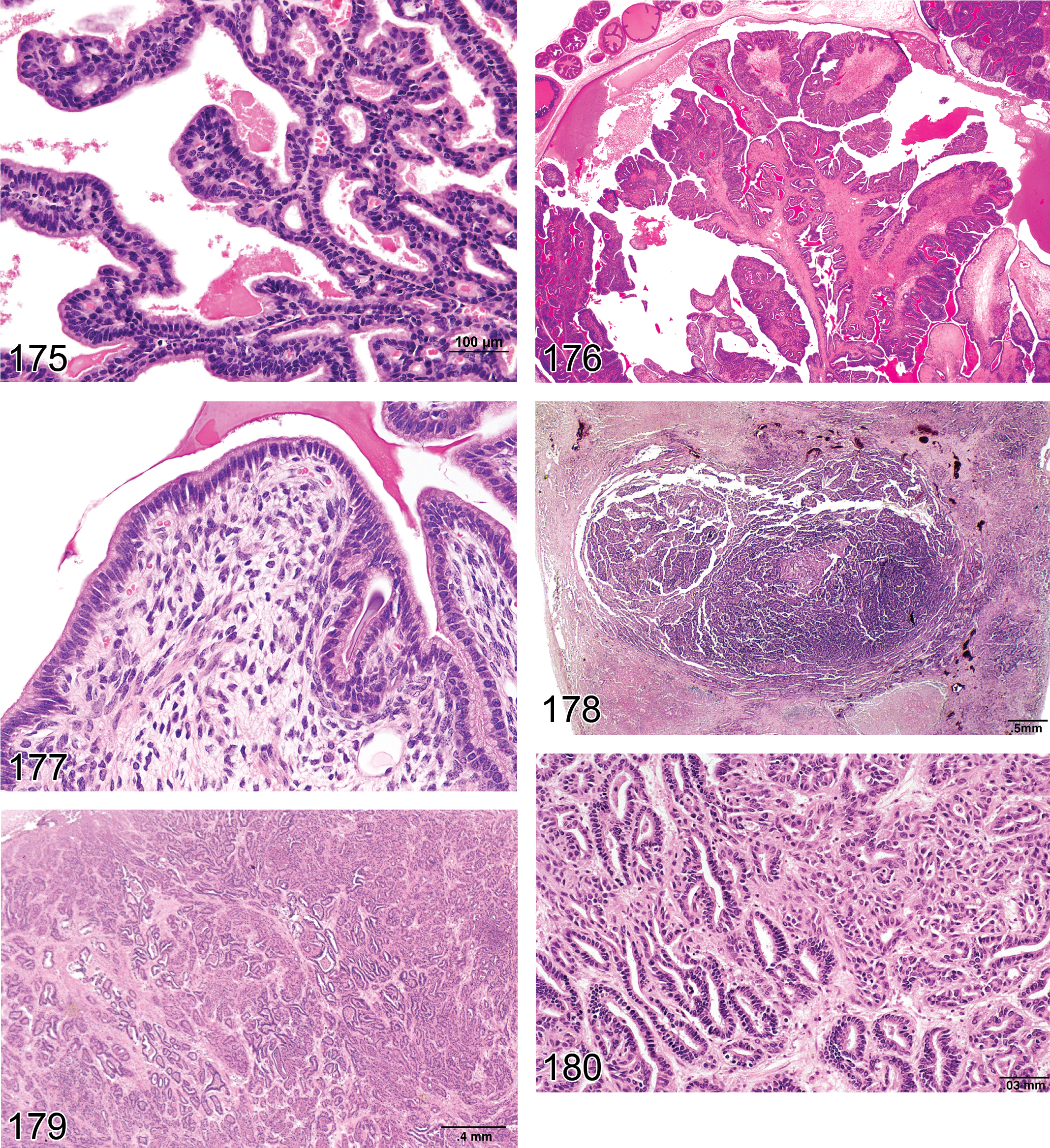
—Rat seminal vesicles. Adenoma. Higher magnification of Figure 174.
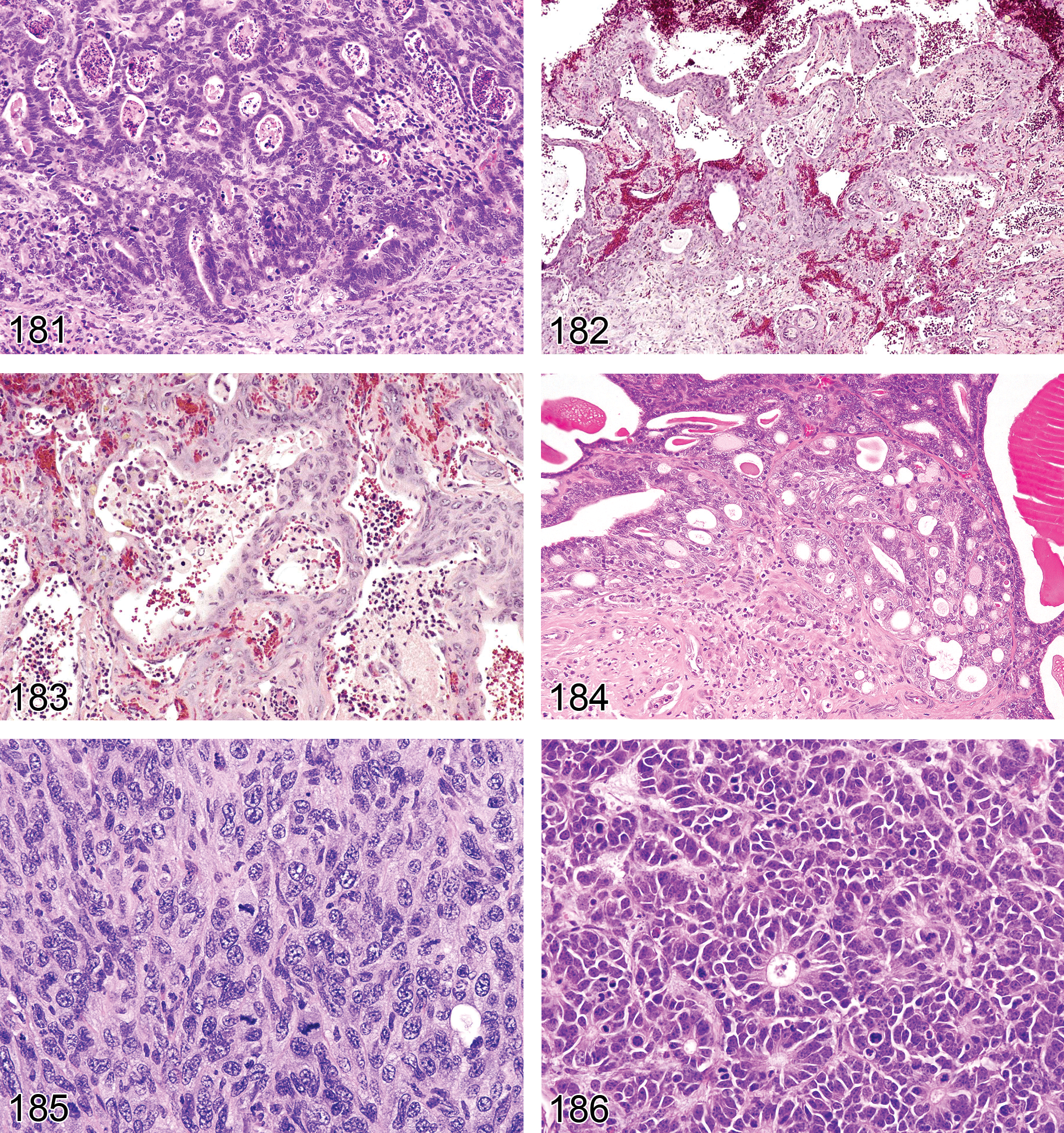
—Mouse prostate. Adenocarcinoma.
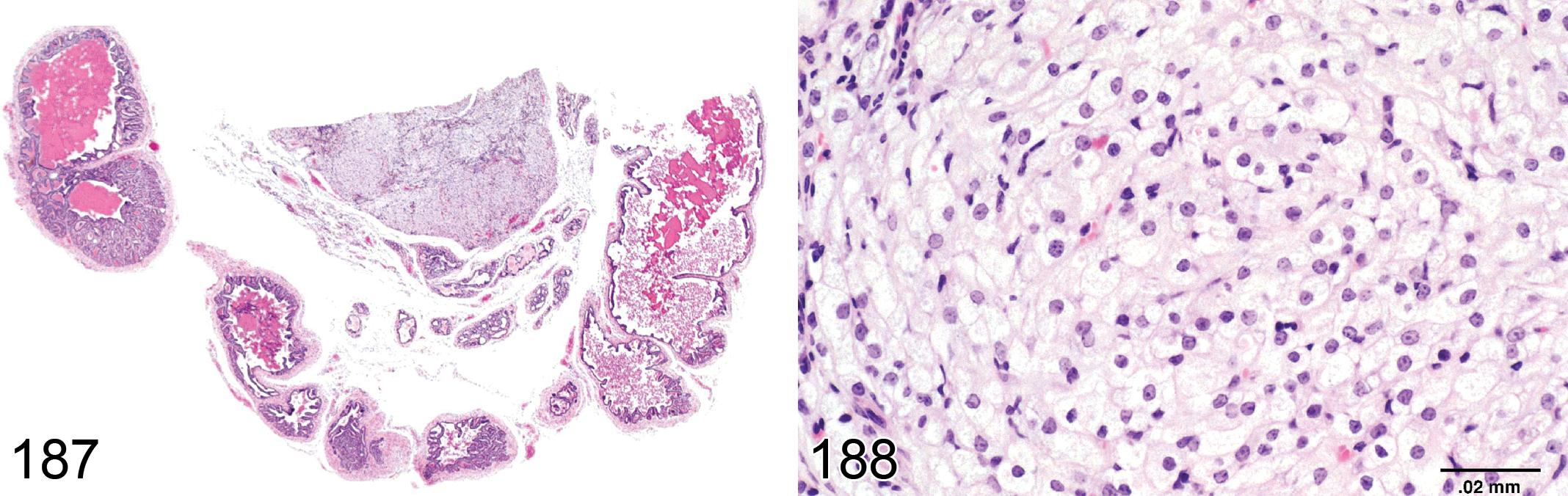
—Mouse seminal vesicles. Tumor, granular cell, benign.
Species: mouse, rat
Pathogenesis: spontaneous and induced by carcinogenic xenobiotics
Diagnostic features Located in the seminal vesicle, in the ventral prostate, rarely in the dorsolateral prostate or coagulating gland Generally involves more than one prostatic acinus and partially or completely obliterates the acinar lumen with distortion of the architecture In the seminal vesicle, it forms a well-delineated area, which may have a fibrous capsule and septa dividing tumor into pseudolobules Mild cellular atypia, cellular crowding, and loss of polarity generally present in the prostate and may be present in the seminal vesicle Cells are arranged in papillary, cribriform, comedo, or microglandular pattern Sometimes enclosed completely or partially by a fine fibrous capsule May compress surrounding tissue Mitotic figures may be present
Differential diagnoses Hyperplasia, atypical: generally smaller with no obliteration of the acinar lumen. Glandular architecture is maintained with no evidence of compression or capsule formation Tumor, granular cell, benign: large or variable-sized epithelioid or fusiform eosinophilic cells with weakly PAS-positive cytoplasmic granules are present Adenocarcinoma: disruption of the architecture, marked cellular atypia, invasive growth, or metastasis may be present
Comments: Coagulating gland adenoma may be grossly distorted by trapped colloid. The diagnosis of a prostatic/coagulating gland adenoma is mainly based on the occurrence of acinar proliferations obliterating the lumina of generally several acini, sometimes with compression and occasionally comedo growth pattern. There appears to be a morphologic continuum between focal atypical hyperplasia and benign adenoma of the prostate and the distinction between the two is not always clear. If differentiation between atypical hyperplasia and adenoma cannot be made on the basis of other criteria, a proliferative lesion obliterating at least one acinar lumen is interpreted to be an adenoma. Dorsolateral prostatic adenomas appear to be rare as spontaneous lesions but have been chemically induced (Bosland 1987c). The predominant growth pattern of the induced lesion was described as microglandular to tubular; cribriform and comedo patterns were not found. Spontaneous proliferative lesions of prostate lobes in mice are uncommon but some transgenic mice show a high incidence (Reznik et al. 1981; Shappell et al. 2004; Suttie et al. 2003).
Papilloma, Squamous Cell: Prostate, Coagulating Gland
Species: mouse, rat
Pathogenesis: rare spontaneous tumor arising from the acinar epithelium
Diagnostic features Marked papillary protrusion of the mucosa into the lumen is present with marked squamous metaplasia and possibly some keratinization No evidence of invasion into the lumen
Differential diagnoses Metaplasia, squamous cell: lesion is restricted to the regular mucosal surface without evidence of papillary protrusion in the lumen Carcinoma, squamous cell: invasion of the subepithelial layers is present
Comments: Squamous cell papilloma is extremely rare.
Tumor, Epithelial–Stromal, Benign: Seminal Vesicle (Figures 176 and 177)
Species
Pathogenesis: develops from mesenchymal and epithelial components of the seminal vesicle
Diagnostic features Papillary or polypoid tumor of the seminal vesicle Complex papillary growths with prominent stromal component Larger tumors may contain cystic and pseudoglandular structures May form “phylloides” pattern with myxomatous or myofibrous expansion of stromal elements Low mitotic rate with no evidence of invasion
Differential diagnoses Adenoma: predominance of epithelial component, lack of stromal prominence or expansion
Comments: Epithelial–stromal tumor has been described as one of the tumors in the TRAMP mouse (Tani et al. 2005).
Adenocarcinoma (Figures 178–184): Prostate, Coagulating Gland, Seminal Vesicle
Species: mouse, rat
Pathogenesis: spontaneous and following administration of carcinogenic xenobiotics. Also seen in GEM.
Diagnostic features Located in the seminal vesicle, in the ventral prostate, rarely in the dorsolateral prostate or coagulative gland Larger than 5 acini (prostate) A distinct fibrous capsule may be present in the prostate or coagulating gland Disruption of the architecture is generally present Growth is cribriform, comedo-type, papillary or glandular/tubular, or solid anaplastic Cellular atypia may often be marked with pleomorphic or anaplastic cells Cells may rarely be well differentiated with basophilic nuclei and abundant eosinophilic cytoplasm Pseudolobulation by fibrous septa, scirrhous stroma, or mucous secretion, focal necrosis and hemorrhage are often present Areas of squamous metaplasia may be present Mitotic figures are numerous Often associated with mixed inflammatory cell infiltrates Invasion of adjacent tissues is present Metastases to regional lymph nodes, lung, and kidney may be present
Differential diagnoses Adenoma: no evidence of invasive growth or metastasis. Disruption of architecture is minor. Cellular atypia is mild Tumor, granular cell, benign: large or variable-sized epithelioid or fusiform eosinophilic cells with weakly PAS-positive cytoplasmic granules are present Tumor, granular cell, malignant: large or variable-sized epithelioid or fusiform eosinophilic cells with weakly PAS-positive cytoplasmic granules are present
Comments: The diagnosis of an adenocarcinoma of the prostate or coagulating gland is mainly based on the presence of invasion and by a solid or adenomatous growth pattern with widespread necrosis. Cribriform adenocarcinoma of the ventral prostate and coagulating gland may lack clear invasive growth but is characterized by marked cellular and nuclear polymorphism. Adenocarcinoma of the prostate and seminal vesicle are rare spontaneous tumors in most strains of rat and mouse (Bosland 1987a, 1987b; Shoda et al. 1998). They have been observed in some strains, notably in ACI/segHapBR rats (Oshima et al. 1985; Ward et al. 1983). Transgenic mouse models of prostate cancer have also been described (Gingrich et al. 1996; Gingrich and Greenberg 1996; Maroulakou et al. 1994). Seminal vesicle and prostate adenocarcinomas have also been induced by a variety of carcinogens (Hoover et al. 1990; Pour 1981, 1983; Pour and Stepan 1987; Shirai et al. 1987, 1994; Slayter et al. 1994; Tamano et al. 1986). For large invasive neoplasms of the male accessory sex glands, it may be impossible to define exactly the gland of origin. These may therefore have to be classified as adenocarcinomas, origin unknown.
Carcinoma, Squamous Cell: Prostate, Coagulating Gland
Species: mouse, rat
Modifier: adenosquamous
Pathogenesis: following administration of carcinogenic xenobiotics
Diagnostic features Cells are epidermoid Arranged in cords, sheets, or irregular nests Keratinization may be present Evidence of invasion and metastasis Carcinoma, adenosquamous (variant): evidence of tubular or glandular structure in addition to extensive squamous differentiation
Differential diagnoses Papilloma, squamous cell: papillary protrusion into the lumen is present. No evidence of invasion of the adjacent tissues Adenocarcinoma: no evidence of significant squamous differentiation.
Comments: Squamous cell carcinoma of the prostate has not been described as a spontaneous tumor but can be induced by administration of a number of carcinogens (Bosland 1987a).
Carcinosarcoma (Figure 185): Prostate
Species: mouse (seen in several lines of GEM)
Synonyms: poorly differentiated adenocarcinoma, sarcomatoid carcinoma, epithelial mesenchymal tumor
Pathogenesis: develops from epithelial–mesenchymal transition in late stage adenocarcinomas of the prostate
Diagnostic features Areas of spindle cells, pleomorphic cells, or homogenous roundish large epithelial-like cells within an adenocarcinoma or as a mass by itself Cells stain positive for cytokeratin 8 or other keratins and vimentin or other mesenchymal cell marker Mitotic figures may vary in number Invasion into surrounding tissue and metastases can occur
Differential diagnoses Adenocarcinoma: lacks areas of proliferating mesenchymal cells Sarcoma, origin from prostate epithelium or epithelial tumor
Comments: Epithelial–mesenchymal transition is an important stage in the progression of some types of late stage adenocarcinoma in GEM models of prostate cancer (Martin et al. 2011). It may be seen within an adenocarcinoma or may progress to replace the entire epithelial component.
Tumor, Neuroendocrine, Malignant (Figure 186): Prostate
Species: mouse (GEM strains, especially TRAMP)
Synonyms: carcinoma with predominantly neuroepithelial differentiation
Pathogenesis: gene alteration in GEM strains
Diagnostic features Present in the ventral lobe of the prostate or submucosal glands of urethra Accumulations of small basophilic cells within acinar epithelium progressing to expansile masses Entrapment of acini within tumor mass Packets, pseudorosettes, or sheets of cells separated by fine fibrous stroma Marked cellular and nuclear pleomorphism and atypical mitoses may be found Angiocentric localization of cells separated by zones of necrosis Cells stain positive for synaptophysin and NSE Invasion into surrounding tissue and common metastases to lung and other tissues in some mouse models
Differential diagnoses Adenocarcinoma: glandular differentiation present Undifferentiated carcinoma: no evidence of neuroendocrine antigens by IHC
Comments: Neuroendocrine cells are present in small numbers between acinar and ductal cells of the normal prostate where they secrete a variety of growth factors. Neuroendocrine tumors have been reported in TRAMP mice (Suttie et al. 2005), occurring only in the ventral lobe. They have also been reported in FVB/N mice containing the SV40 T-Ag (Garabedian, Humphrey, and Gordon 1998). In TRAMP mice, neuroendocrine tumors are considered a subset of poorly differentiated carcinomas which undergo an epithelial to neuroendocrine shift (Martiniello-Wilkes et al. 2003.)
Tumor, Granular Cell, Benign (Figures 187 and 188): Prostate, Seminal Vesicle, Coagulating Gland
Species: mouse, rat
Synonyms: Abrikosoff’s tumor, myoblastoma, benign
Pathogenesis: not established, origin from Schwann cell or mesenchymal cell has been proposed. Spontaneous tumor
Diagnostic features Circumscribed, demarcated solid mass Composed of large, round to oval cells with large pale nuclei and abundant eosinophilic granular cytoplasm Expansive growth causes compression and atrophy of adjacent tissues Positive immunohistochemical reaction with S-100, only weakly PAS-positive
Differential diagnoses Tumor, granular cell, malignant: cellular pleomorphism
Comments: Granular cell tumor occurs as a single pink to pale yellow-gray mass. This tumor is frequently seen in the female genital tract and occasionally in the male (Karbe 1987; Mitsumori and Elwell 1994; Radovsky, Mitsumori, and Chapin 1999; Rehm et al. 2001; Suwa et al. 2002). It also occurs in other organs, particularly brain. Granular cell tumor reacts positively in immunocytochemical stains for NSE, S-100 protein, and peripheral myelin proteins. In the female, it has been suggested that lesions termed granular cell tumors may in fact be normal or reactive metrial glands (Picut et al. 2009).
Tumor, Granular Cell, Malignant: Prostate, Seminal Vesicle, Coagulating Gland
Species: mouse, rat
Pathogenesis: not established, origin from Schwann cell or mesenchymal cell has been proposed. Spontaneous tumor
Diagnostic features Solid mass composed of typical granular cells located in the periphery as well as of cells with decreased granularity and spindle cell morphology in the center Pleomorphism with increased nucleus:cytoplasmic ratio Mitoses are uncommon Necrotic areas are often present Positive immunohistochemical reaction with S-100, only weakly PAS-positive
Differential diagnoses Tumor, granular cell, benign: no pleomorphism, circumscribed, well demarcated, causing compression of adjacent tissue Adenocarcinoma: no evidence of weakly PAS-positive granular cells
Comments: The malignant granular cell tumor is a rare lesion described in the literature. It has the same biological behavior as the benign tumor and shows no regional or distant metastasis.
Footnotes
Acknowledgments
The group is deeply appreciative of the contribution of figures from the following sources: NTP archives—Figures: 34, 39, 51, 56, 59, 60, 65, 67, 111, 112, 127–129, 136, 148, 149, 152, 155, 160, 161, 165, 171-175, 178–180, 182, 183, 187, 188; Dr Jerrold Ward—Figures: 162, 166, 176, 177, 184–186; Dr. Shrirang Jamadagni, National Institute of Ayurvedic Drug Development—Figures 78–81; Dr. Noriyaki Ishigami, ONO pharmaceuticals—Figures 73 and 74; Dr. Rex Hess, University of Illinois—Figure 85; Suwa et al. 1981—![]() .
.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
