Abstract
Scoring demyelination and regeneration in hematoxylin and eosin–stained nerves poses a challenge even for the trained pathologist. This article demonstrates how combinatorial multiplex immunohistochemistry (IHC) and quantitative digital pathology bring new insights into the peripheral neuropathogenesis of the Twitcher mouse, a model of Krabbe disease. The goal of this investigational study was to integrate modern pathology tools to traditional anatomic pathology microscopy workflows, in order to generate quantitative data in a large number of samples, and aid the understanding of complex disease pathomechanisms. We developed a novel IHC toolkit using a combination of CD68, periaxin-1, phosphorylated neurofilaments and SOX-10 to interrogate inflammation, myelination, axonal size, and Schwann cell counts in sciatic nerves from 17-, 21-, 25-, and 35-day-old wild-type and Twitcher mice using self-customized digital image algorithms. Our quantitative analyses highlight that nerve macrophage infiltration and interstitial expansion are the earliest detectable changes in Twitcher nerves. By 17 days of age, while the diameter of axons is small, the number of myelinated axons is still normal. However, from 21 days onward Twitcher nerves contain 75% of wild-type myelinated nerve fiber numbers despite containing 3 times more Schwann cells. In 35-day-old Twitcher mice when demyelination is detectable, nerve myelination drops to 50%.
Keywords
Introduction
The histopathological hallmarks of Krabbe disease (KD), also known as globoid cell leukodystrophy, consist of demyelination of the nervous system in association with massive infiltration of giant multinucleated cells termed globoid cells, which can be as large as 40 µm in diameter.
1,2
Krabbe disease is a rare autosomal recessive disorder that affects approximately 1 per 244,000 newborn infants.
3
Infants with KD have a point mutation in the
The pathogenesis of KD has been best studied in the Twitcher (TWI) mouse model, 7 –14 in which several treatment strategies including gene therapy, substrate reduction therapy, chemical chaperones, and enzyme replacement therapy are currently under investigation. 15 –23 Twitcher mice show clinical signs at around 21 days of age (coinciding with the onset of the active myelination period) 23,24 and histopathological features 14,24,26 similar to human KD, and die prematurely at postnatal day 45. 13,23
Although the direct association between GALC deficiency and psychosine accumulation is the disease cause, the downstream pathomechanisms leading to demyelination remain unsolved. 3,11,26 –28 Demyelination occurs as early as 10 and 20 days of age in the nerve and spinal cord, respectively, and it progresses with macrophage infiltrates due to the presence of myelin debris. 24,25,29 –31 Most data have been quantitatively gathered from a couple of morphological, electron microscopy-based studies. 31,32
To date, there are no published studies in this demyelinating disease that underscore peripheral myelin-related abnormalities at the nerve fiber spatial resolution using a combined immunohistochemistry (IHC) and digital imaging quantitative modality. This anatomic pathology-based study using a digital pathology workflow shows that combinatorial use of multiplex fluorescent IHC paired with computational digital imaging algorithms can generate robust and accurate data sets that aid in the understanding of diseases with complex pathogenesis.
Materials and Methods
Twitcher Mice and Tissue Processing
C57 black 6J strain (C57BL/6J; stock# 000664) and B6.CE-
At study termination, the mice were intracardially perfused with saline buffer at room temperature (21 ± 3 °C) while under isoflurane anesthesia, then cervically dislocated. Sciatic nerves were dissected and collected proximal to its peripheral branches, immersed in 4% paraformaldehyde in phosphate-buffered saline (1× PBS, pH 7.4, Thermo Fisher Scientific, CAS 30525-89-4) for 48 hours at 4 °C. Nerves were cross-sectioned in the middle and transferred to 70% ethanol at 4 °C for 3 to 5 days prior to paraffin embedding. When manually embedding, nerve segments were placed on the mold with a vertical orientation carefully ensuring to maintain a 90° angle to the mold while fully embedding in paraffin. Additionally, representative sciatic nerve segments were placed flat in the mold and embedded parallel to the plane of the mold. Generated slides containing several cross-sections of both sciatic nerves were sectioned at 5 µm thickness and further processed for routine hematoxylin and eosin (H&E) to assess nerve lesions morphologically while a Masson’s trichrome stain was used to visualize intranerve fibrous connective tissue. In addition, the severity of nerve lesions at 35 days of age was qualitatively assessed by IHC. Moreover, one sciatic nerve each from a 35-day-old healthy and from a TWI mouse were processed for examination under transmission electron microscopy. Nerves were trimmed to generate multiple 1 mm3 pieces using a razor blade (American Safety Razor Company) and placed in 2% paraformaldehyde (Sigma Aldrich, reagent grade) and 2.5% (wt/vol) glutaraldehyde (Sigma Aldrich, grade 1) in 150 mM sodium cacodylate buffer (Sigma Aldrich) at pH 7.2 for 4 days at 4 °C and post-fixed in 1% (wt/vol) osmium tetroxide (Electron Microscopy Sciences [EMS]), in 0.1M sodium cacodylate for 60 minutes at room temperature, then stained with 2% (wt/vol) uranyl acetate (EMS) for 30 minutes at room temperature. Dehydrated and EMbed-812 (EMS) infiltrated samples were embedded in EMbed-filled BEEM (EMS) capsules at 60 °C for 72 hours. Samples were trimmed on a MT-7000 ultramicrotome at a 1-µm-thick setting and were stained with toluidine blue and observed under transmitted light to determine the appropriate depth for collection of ultrathin sections which were then cut at a thickness of 60 nm. Sections were collected on copper 200 mesh grids (EMS) and were imaged on a Phillips Tecnai 12 transmission electron microscope at 80 kV.
Strategic Selection of IHC Markers to Interrogate Age-Related TWI-PNS Pathologic Phenotypes
Based on histopathological evaluation from 35-day-old TWI mice, pathophysiologic mechanisms underlying TWI-PNS degeneration include a combination of inflammatory, degenerative, and regenerative responses. Accordingly, we further focused our effort on designing and developing a targeted immunohistochemical toolkit that combined with digital pathology algorithms could successfully dissect each tissue response at a given specific age. Ideally, such an approach would investigate hypercellularity mainly due to infiltration of macrophages and Schwann cell proliferation, and demyelination and axonal damage in peripheral nerves at preclinical and clinical stages of the disease. Our targeted immunofluorescent-IHC (IF) approach, which includes commercially available antibodies against specific proteins (Table 1), was as follows: to analyze nerve inflammation, in particular infiltrating macrophages, we used CD68. 34 Phosphorylated neurofilaments (pNF), which are enriched in axons, were used also to assess axonal size and/or axonal breakdown. Finally, the positive regulator of myelination Sry-related HMg-Box gene 10 (Sox-10) 35 and periaxin-1 (PXN), one of the earliest myelin proteins expressed during myelinogenesis and remyelination, 36 were used to detect nonmyelinating and myelinating-Schwann cells, and peripheral myelin, respectively. Various combinations of the above-described markers were applied to freshly acquired paraffin sections and run in the Discovery Ultra Autostainer (Ventana) as automated IFs using prior manually IF-optimized protocols. Microscopic qualitative examination by a pathologist ensured marker specificity in single chromogenic IHCs (cIHCs) and in multiplex IF before the execution of automated runs. Consistency on specificity and sensitivity between IHC runs was monitored by using a shared positive control tissue of spinal cord from a terminally sick TWI mouse in each run.
List of Optimized Antibodies to Assess Nerve Histopathology.
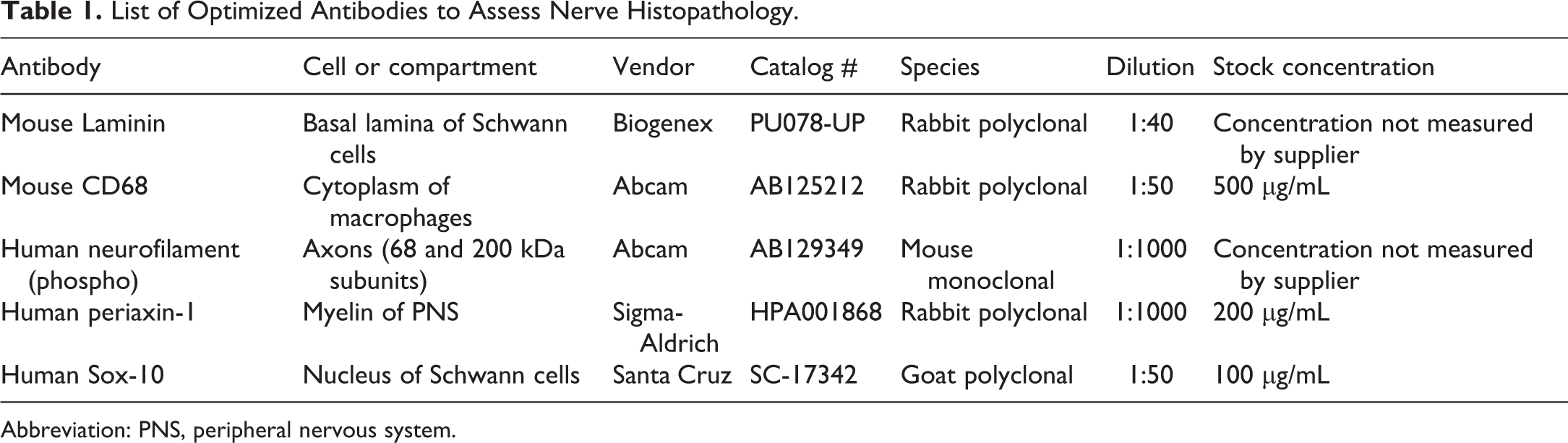
Abbreviation: PNS, peripheral nervous system.
Chromogenic and Fluorescence IHC
Various antibodies (Table 1) were optimized first manually in a subset of nerves, and subsequently transferred to an automated closed IHC system (Ventana Discovery Ultra Autotimer). Both cIHC and fluorescence (IF) IHC were run as per manufacturing instructions. Briefly, tissue sections on Superfrost plus slides (Thermo Fisher Scientific) were deparaffinized in EZ Prep (Ventana Discovery). Heat-induced epitope retrieval in Tris-EDTA buffer pH 7.8 at 95 °C for 44 minutes (standard CC1, Ventana Discovery) was used to retrieve antigen at 95 °C. Nerves were incubated for 45 minutes in 1% TBS (Tris-buffered saline) blocking solution containing 10% normal donkey serum (Jackson ImmunoResearch), 0.1% bovine serum albumin (Sigma-Aldrich), and 0.3% Triton (Sigma Triton-X-100). All primary antibodies were diluted (Table 1) in Ventana Reaction Buffer (Ventana Medical Systems; cat. # 950-300) and slides were incubated overnight at 4 °C. For cIHC, respective OmniMap secondary antibodies conjugated to horseradish peroxidase were applied for 16 minutes at 37 °C. Finally, the 3,3′-diaminobenzidine (DAB) reaction was performed by applying one drop of DAB CM and one drop of H2O2 CM with 8-minute incubation followed by a drop of Copper CM for 5 minutes. Slides were counterstained with hematoxylin for 8 minutes, incubated for another 8 minutes with the Bluing reagent. For automated IF, slides were washed in 1× TBS. For IF, the secondary antibodies (Alexa Fluor 488 (1:125), 555 (1:500), 594 (1:500), or 647 (1:500)) matching the host species of the primary antibodies were prepared in Ventana reaction buffer and applied to the tissues in a light-blocking staining tray for 1 hour at room temperature. Primary antibodies were omitted in negative control slides, which only received each of the combination of secondary antibody with fluorophores. The tissues were again washed 3 times on a shaker with 1× TBS for 5 minutes for each wash. DAPI (4′,6-diamidino-2-phenylindole) Fluoromount-G (17984-24, EMS) was applied to tissues and the slides were coverslipped. Primary antibodies were replaced by the IgG subtype and applied to negative control sections. In addition, slides with only secondary antibodies were included in order to assess nonspecific staining. For IF, a blank slide with no primary or secondary antibodies was included to test tissue autofluorescence under the different channels.
Morphometric and Statistical Analyses
Microscopy, quantitative analysis, and digital imaging protocol design
A minimum of 3 mice per combination of time-point (age) and genotype were included for digital analyses except for counts of nuclei (DAPI) and Schwann cells (Sox-10) in 17- and 25-day-old WT mice, and for counts of CD68-positive cells in the 17-day-old WT group, in which only 2 animals were included due to the small size and fragile nature of those sciatic nerves sections (Supplementary data). Note that 2 to 5 replicates representative of the same nerves were analyzed in 28% of the analyses (Supplementary data) in an attempt to gather more data for quantitative analysis (Supplementary data). From each animal, fluorescent images from all optimal transverse sections of sciatic nerves were taken at 20× and /or 40× and acquired using the Leica Application Suite X (version 3.0.2) computer program connected to a Leica DM4 B (Leica Microsystems) episcope equipped with a Leica DFC 9000 GT (Leica Microsystems VSC06236) and a Lumencor SOLA-SM light engine. The 20× or 40× objective enabled the full cross-sectional area of the nerves to be captured and prevented sample bias in subsequent digital imaging analyses. The blue, green, red, and far-red channels were used to image all tested samples as well as its corresponding secondary antibody-only controls. Adjusted-exposure histograms were optimized for each combination of primary antibodies and corresponding fluorophore-tagged secondary antibodies to achieve desired sensitivity and specificity. Final histograms were consistent across all examined animals in order to be able to compare differences in immunosignal.
Our workflow for combined IHC and digital image platform is illustrated in Figure 1. In order to interrogate disease status of sciatic nerves, we focused specifically on image quantification of (1) hypercellularity and nerve enlargement, (2) macrophage infiltration, and (3) PNS myelination, myelin loss, axonal damage, and remyelination. In the PNS, increased cell numbers due to macrophage infiltration or Schwann cell proliferation were calculated as percentages of CD68+ or Sox-10+ cellular counts, respectively. Demyelination was first calculated by assessing how many axons positive for pNF were surrounded by a PXN+ myelin sheath. Finally, the enlargement of the extracellular space, also known as the interstitium of the nerve, was calculated by subtracting all positive components of the nerve fiber (pNF+ and PXN+ structures) from the total area. Tissue artifacts were manually removed from images prior to analyses. For such digitized quantification, self-developed protocols with custom macros using the digital image processing software Volocity Version 6.3.0 (Quorum Technologies) were optimized and applied to full cross-sections of nerves. Specific details on custom macros and protocols are provided as supplementary data.
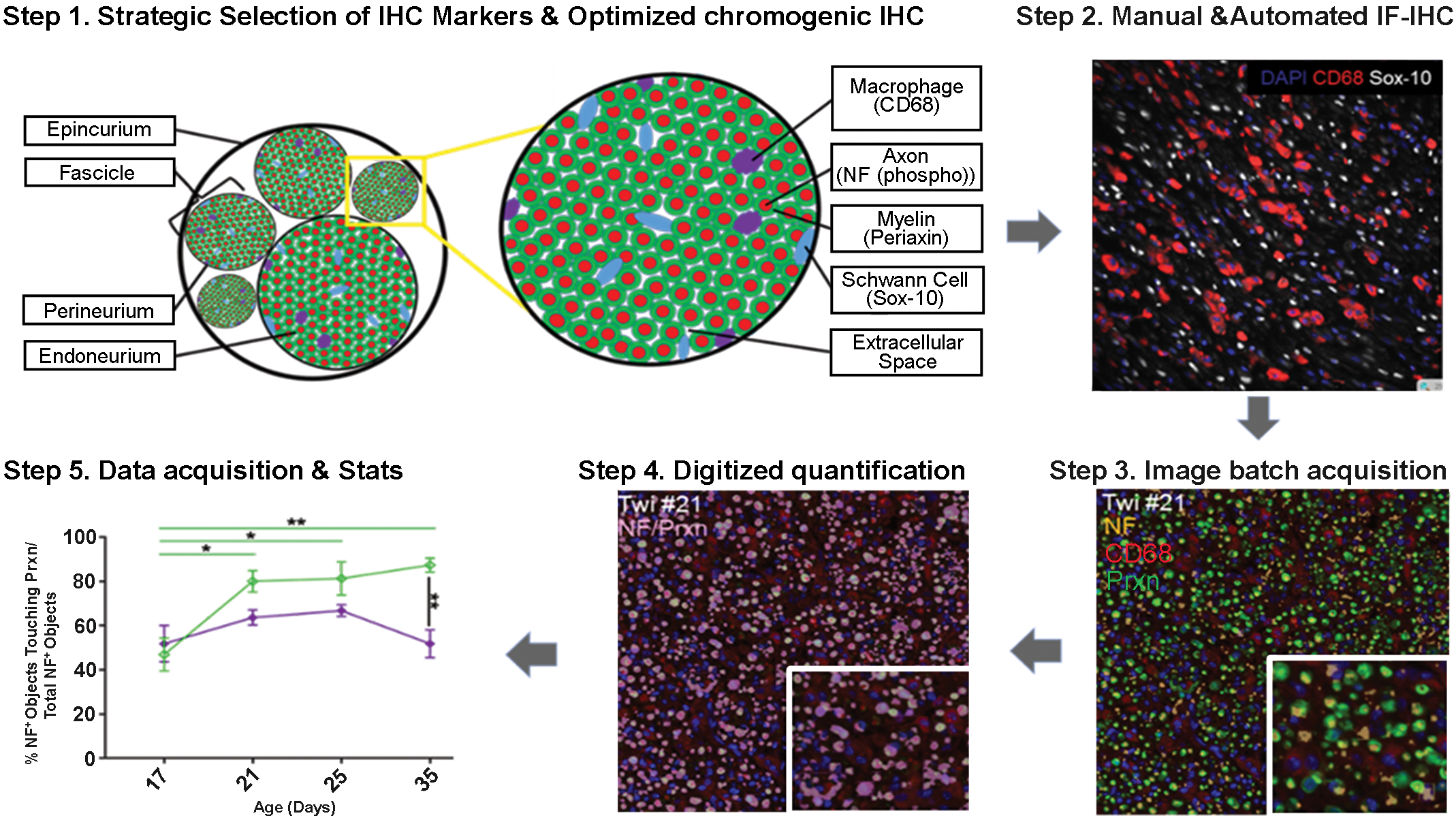
Workflow to quantitative digital pathology in the peripheral nerve when using multiplex fluorescence immunohistochemistry (IHC). First, we identified our peripheral nerve targets to be quantified as macrophages, myelin sheaths, axons, Schwann cells, and extracellular space. Next, we developed and optimized a novel multiplex IHC toolkit to those targets using best combinations of CD68 (for macrophages), periaxin-1 (Prxn for myelin), phosphorylated neurofilaments (NF for axons), and SOX-10 (for Schwann cells). Step 2 shows the presence of numerous red CD68+ macrophages and white Schwann cell SOX-10+ nuclei in a 35-day-old Twitcher (TWI) nerve as an example of an optimized IHC. Next and prior to any image batch acquisition, the pathologist revised and optimized adjusted-exposure histograms for each combination of primary antibodies and corresponding fluorophore-tagged secondary antibodies to ensure optimal sensitivity and specificity. Step 3 shows red CD68+ macrophages and many yellow NF+ axons surrounded by a green Prxn+ myelin sheath in a 35-day-old TWI nerve. Note a few denuded axons identified by yellow structures devoid of green (inset). Next, targets’ immunosignals are transformed into spatial objects by the development of self-customized digital image algorithms using image analytical softwares (step 4). Digitized numerical data are acquired and ready to be analyzed, plotted, and interpreted. Step 5 shows the temporal myelination of wild-type (WT) nerves (green lines) compared to TWI nerves (purple lines) as nerves age. Plotted counts (mean and SEM) represent the percent of NF+ objects that were embedded in a Prxn (periaxin myelin) object out of the total NF+ objects present in those nerves. Statistical analyses highlight changes that are significantly different between the age groups and/or the genotype.
Statistical Analysis
All statistical analyses were performed using R (version 3.6.2; https://www.r-project.org/). Differences between 2 groups of samples (TWI and WT for different age groups) were tested by Wilcoxon rank-sum tests. For groups in which animals had repeated measurements, mean value was used for the given animal in group comparison. All measurements were checked for normal (Gaussian) distribution using Shapiro-Wilk test for normality and Fligner-Killeen test for homogeneity of variance between different groups of samples. Data were also inspected visually for deviation from normal distribution using density plot. The following experimental measurements passed normality test using all samples and homogeneity of variance tests between genotype groups (
Results
Twitcher Mouse Peripheral Neuropathy
At terminal stage of the disease, 35-day-old TWI mouse sciatic nerves were larger and had an abnormal tissue architecture. Microscopically (Figure 2), nerves showed hypercellularity with severe macrophage infiltration and a few lymphocytes and neutrophils as the main lesion. Apparent at 25 days, macrophages expanded the interstitial and perineural space, thus contributing to the increase in nerve size and to the disorganization of the nerve architecture. Nerve damage such as axonal loss or myelin breakdown was not prominent features of H&E stained sections; however, evidence of fibrous deposition was clearly demonstrated in Masson’s trichrome-stained sections confirming underlying nerve damage. Ultrastructurally, nerves from 35-day-old mice were highly disorganized, with expanded extracellular space and overrepresentation of unmyelinated axons. They displayed reminiscent features of onion bulb formation, a term that refers to the observation of imbricated layers of supernumerary Schwann cell processes forming rings around individual axons in cross sections. 1 Onion bulb formation is the classical peripheral nerve lesion in the TWI or the Trembler mouse 38 that reflect repetitive cycles of demyelination and remyelination. Figure 2 shows supernumerary branching large processes from Schwann cells surrounding numerous nonmyelinating axons and fibroblasts around variably myelinated axons intermingled with the deposition of polymerized collagen bundles.
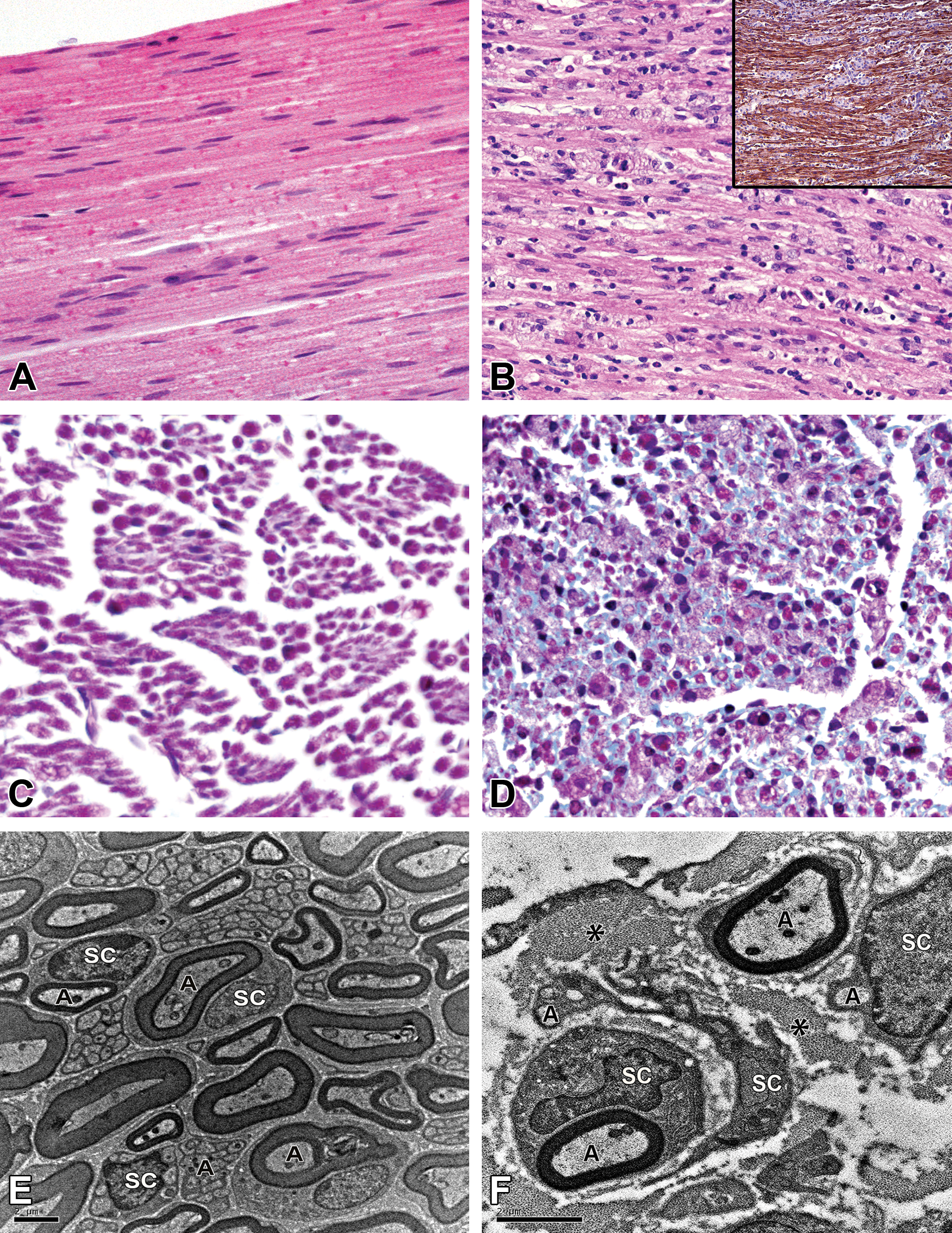
Nerve histopathology in the 35-week-old Twitcher (TWI) mouse. While wild-type (WT) nerves are uniformly assembled with tightly packed nerve fibers (A), TWI nerves were disorganized with dispersed fibers and with large numbers of infiltrating mononuclear cells (B). Large arrays of foamy macrophages (blue DAB-negative) disrupt the boundaries of brown laminin-DAB-positive nerve fibers (B, inset). When comparing to WT mice (C), the Masson’s trichrome stain reveals an increased amount of pale blue endoneurial fibrous connective tissue in nerves from the TWI mice (D). Ultrastructural examination of the sciatic nerve from a WT mouse (E) and a TWI mouse (F) confirms that in disease there is endoneurial expansion due to supernumerary Schwann cells processes and collagen deposition visible between those Schwann cell processes. Note that the couple of Schwann cells that surround nonmyelinated nerve fibers also extend their supernumerary branching large processes around myelinated fibers forming rings (onion bulbs). A, axon; DAB, 3,3′-diaminobenzidine; SC, Schwann cell; *, collagen.
Inflammation Is the Earliest Detectable Change and Major Contributor to Nerve Pathology
Histopathological observations on H&E and trichrome-stained nerve sections guided our strategy to quantify digitally nerve size and hypercellularity (Figure 3). Consistently, the average nerve area was larger in TWI mice at all ages when compared to WT nerves. At terminal disease (35-day-old mice), the average cross-sectional area of TWI sciatic nerve was close to 100,000 µm2, which is a 2-fold increase from its healthy (WT) counterpart. Of note, the interstitial space of healthy nerves increased progressively with age and doubled in size by 35 days reaching an area close to 55,000 µm2. This result confirms that TWI nerves are larger mainly due to the increase in extracellular space. Hypercellularity in healthy nerves as determined by counts of DAPI+ nuclei/mm2 may inform about nerve maturation and not necessarily disease. In that respect, immature 17-day-old WT nerves had similar DAPI+ nuclear counts as their age-matched TWI nerve counterpart. However, as WT nerves aged to 21 days, the nuclear counts decreased by approximately half and remained low throughout maturation. Despite TWI nerves did not show a significant decrease in cell counts until 25 days of age, yet 35-day-old TWI nerves maintained similar intranerve cell counts as their WT counterparts.
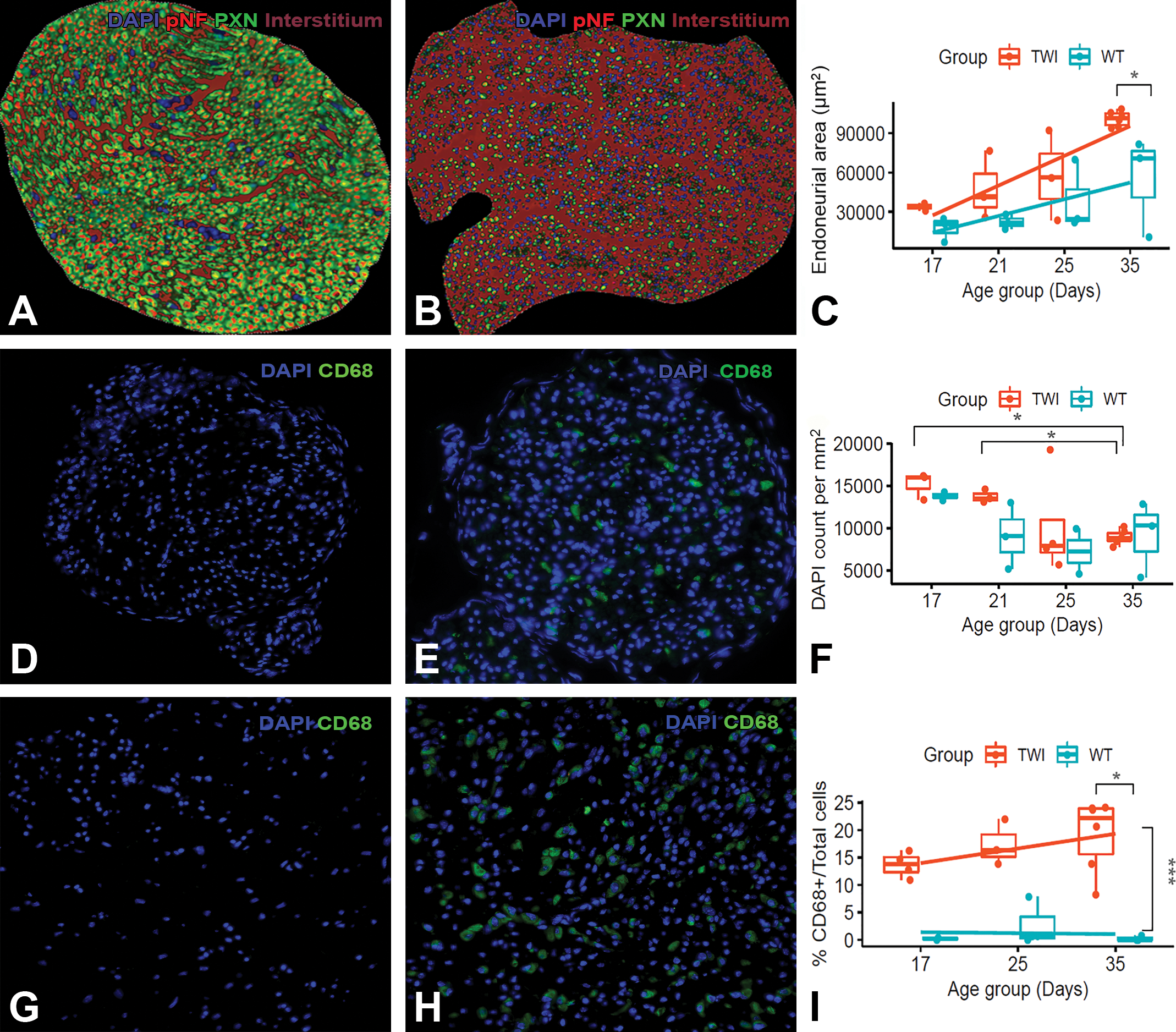
Inflammation of sciatic nerves from 35-day-old Twitcher (TWI) mice. The area of empty space (interstitium in brown) for the shown wild-type (WT) nerve is 58,520 µm2 (A) while 109,602 µm2 for the TWI nerve. Note that the image magnification in panel A is at ×40, while in B is at ×20 to allow visualization of the entire tissue cross-section. Boxplot representation of the area (in µm2) of the interstitial thickening (C) as denoted by digital quantification of area left between nerve fibers positive for neurofilaments and periaxin (A, B). Comparative intranerve DAPI+ (in blue) and CD68+ (in green) cells in 17- and 35-day-old healthy and diseased sciatic nerves (D-I). Boxplot representations of DAPI+ and CD68+ cell counts used mean per animal if multiple measures were taken (F, I). Significant differences between TWI and WT nerves at specific time-point are shown as an asterisk (
Next, in order to dissect the inflammatory component from hypercellular nerves, CD68+ cells in full nerve cross sections were counted. Already at 17 days of age, the earliest evaluated time-point, TWI nerves had a distinct 15% macrophage infiltration contributing to their nerve hypercellularity. Moreover, a significant and progressive increase in CD68+ cells at 35 days of age contributed in average up to 20% of the total nerve cellularity (Figure 3). As expected, there were minimal CD68+ cells in WT nerves, and the number remained constant throughout maturation.
Evidence of a Degenerative Phase With Minimal Regeneration in TWI Sciatic Nerves
Because degenerative changes affecting axons or myelin were scarce based on H&E examination, the degenerative and regenerative phases were immunohistochemically explored by studying the relationship between the onset of sciatic nerve damage and the number of Schwann cells. Selected markers of nerve fibers including axons (pNF), myelin (PXN), and Schwann cells (SOX-10) interrogated the relationship between nonmyelinated axons, axonal diameter, and Schwann cell counts in TWI nerves (Figure 4). Qualitatively, pNF+ axons appeared smaller in diameter in the TWI mice as early as 17 days of age. However, those thin pNF+ axons would appear mostly myelinated since PXN cIHC demonstrated thinner and smaller myelin sheaths. Wild-type nerves showed homogenous myelin sheath and axonal sizes across the entire nerve cross section based on cIHC for PXN and pNF, respectively. However, same cIHCs in TWI nerve highlighted the loss of size homogeneity with a wide spectrum from small to very large nerve fibers (Figure 4). The next paragraphs described our strategic combinatorial multiplex IF approach with quantitative digital imaging data sets targeting specific pathogenetic findings.
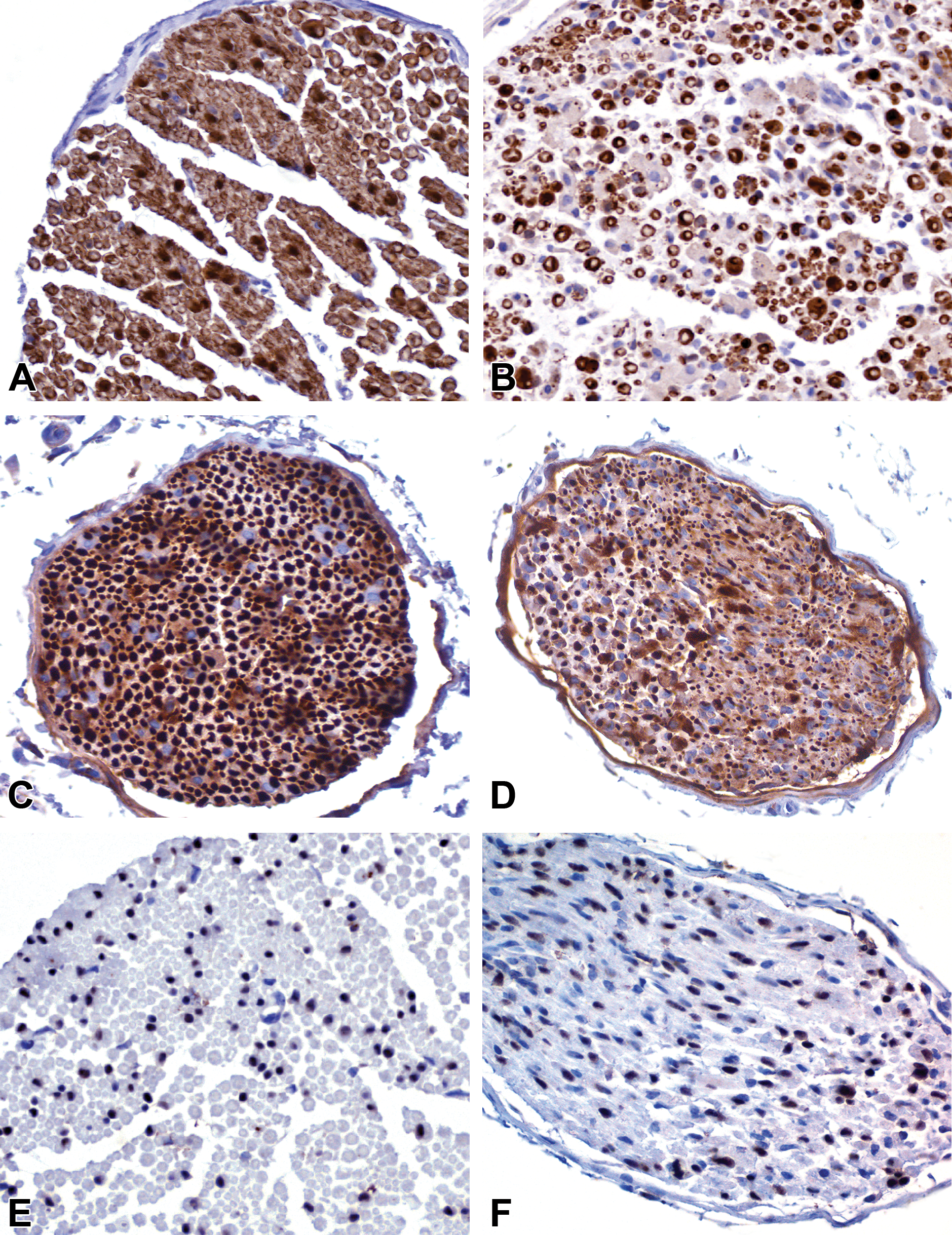
Selection of markers to dissect components of the nerve fiber in wild-type (WT) (A, C, E) and Twitcher (TWI) (B, D, F) 35-day-old mice. Chromogenic single immunohistochemistry (IHC) for periaxin-1 (A, B) to label myelin sheaths, phosphorylated neurofilaments (p-NF; C, D) to label axons, and SOX-10 (E, F) to label Schwann cells reveals loss of nerve fiber architecture, diameter heterogeneity of myelin sheaths and axons, and suspect increased number of Schwann cells in terminally sick TWI mice when compared with healthy nerves.
First, we wanted to investigate whether or not TWI nerves had delayed development and/or from demyelination and ultimately axonal breakdown. We initially interrogated whether or not TWI nerves had increased numbers of nonmyelinated axons compared to their WT counterpart. Following a duplex IF, digitized image analysis counted individual pNF+ axons either surrounded by or lacking a PXN+ sheath of myelin. Such analyses, which are summarized in Figure 5, confirmed that TWI nerve contained similar myelinated nerve fibers at 17 days of age but fewer myelinated nerve fibers than WT nerves from 21 days onward. Postnatal myelination of healthy nerves was half-completed at 17 days of age, and fully completed by 21 days of age when approximately 80% of the axons are myelinated. Instead, TWI mice followed different kinetics of myelination with a myelination peak of only 67% occurring later between 21 and 25 days of age, which suggests not only a delay but also an overall decrease in axonal myelination. Distinctly at 35 days of age, TWI nerves had a dramatic 22% drop in axonal myelination disclosing that only 50% of the total intranerve axons are myelinated, similarly to the initial myelination recorded at the 17-day time-point (Figure 5). Altogether, these data are suggestive of TWI nerves having dysmyelination at early clinical stages and from demyelination only at terminal disease. Since the kinetics of myelin growth are tightly regulated and in concordance with that of axonal growth, we hypothesized that unmyelinated axons due to growth retardation would remain smaller whereas fully grown axons that subsequently had demyelination would have a larger size. In order to assess axonal growth retardation, we next analyzed the cross-sectional area of all nonmyelinated axons (pNF+ not surrounded by PXN) in 17 versus 35-day-old sciatic nerves. We decided to replace the standardized axonal diameter measurement by the axonal area measurement since it was a more robust computational algorithm. Distinctively, our quantification data highlight that the largest proportion of nonmyelinated axons from TWI nerves at 35 days of age have a larger area when compared with their healthy age counterpart or at a younger age (Figure 5).
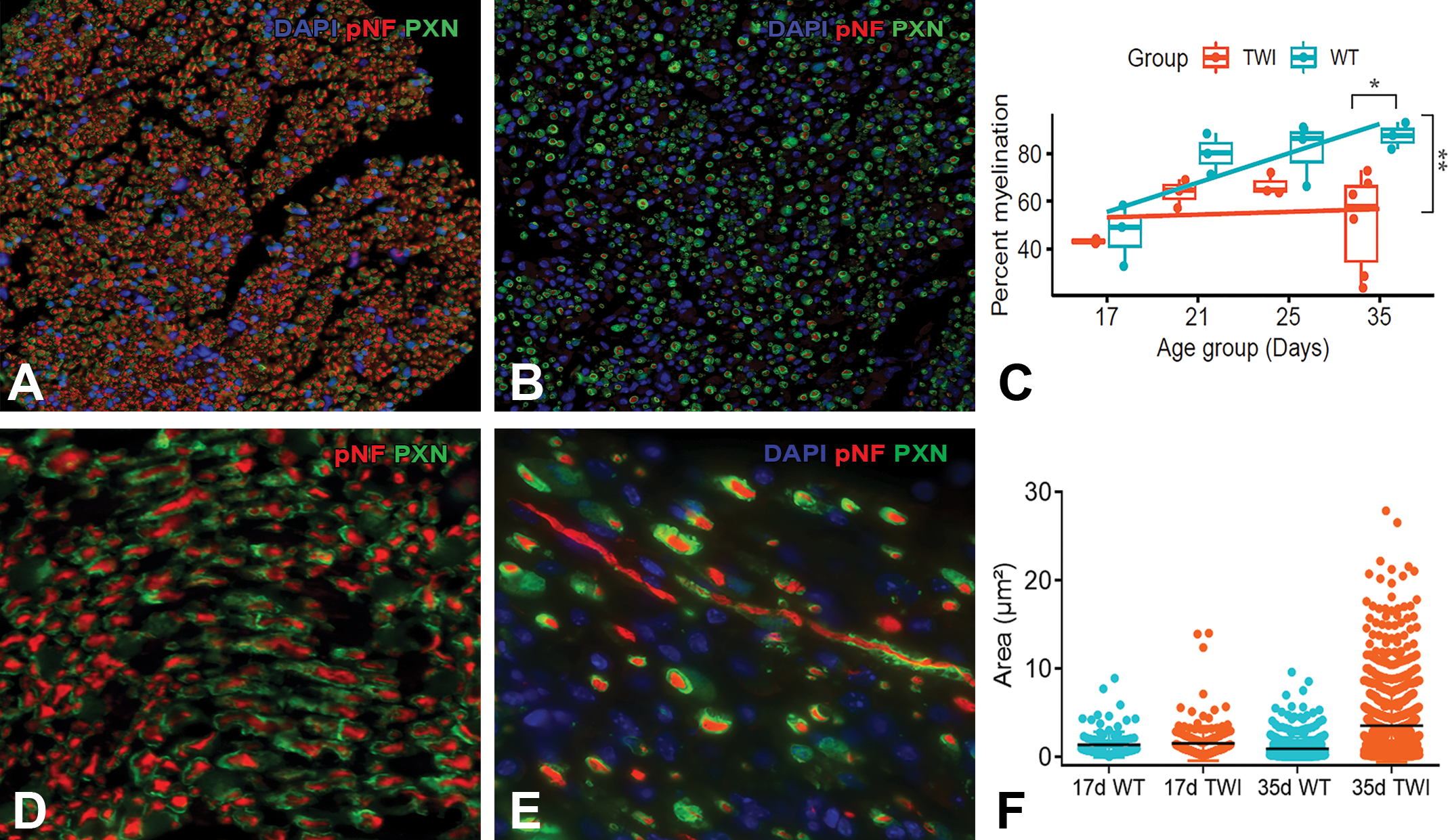
Myelinated axons in sciatic nerves from wild-type (WT) (A, D) and Twitcher (TWI) (B, E) 35-day-old mice. Quantitative analyses from a combined immunohistochemistry (IHC) for phosphorylated neurofilaments (pNF, in red) and periaxin-1 (PXN, in green) applied on sciatic nerves from mice at 17-, 21-, 25-, and 35 days of age confirmed that TWI nerves overall contained less myelinated axons. Boxplot representations of nerve myelination used mean per animal if multiple measures were taken (C). Significant drop of 20% myelination in the TWI nerve from 35-day-old mice is shown as an asterisk (
Next, we attempted to interrogate the regenerative capacity of nerves. The reaction to a demyelinating injury in a segment of a nerve fiber results in the transient proliferation of Schwann cells to remyelinate the denuded segment. TWI nerves contained higher Sox-10+ Schwann cells at all time-points; despite not reaching significance, even at 35 days of age. Similarly in WT and TWI groups, the number of Sox-10+ cells per mm2 decreased significantly by approximately 50% by 35 days of age (Figure 6). However, the onset of Schwann cell depletion occurred in WT nerves between 17 and 21 days of age while in TWI nerves did not occur until 35 days.
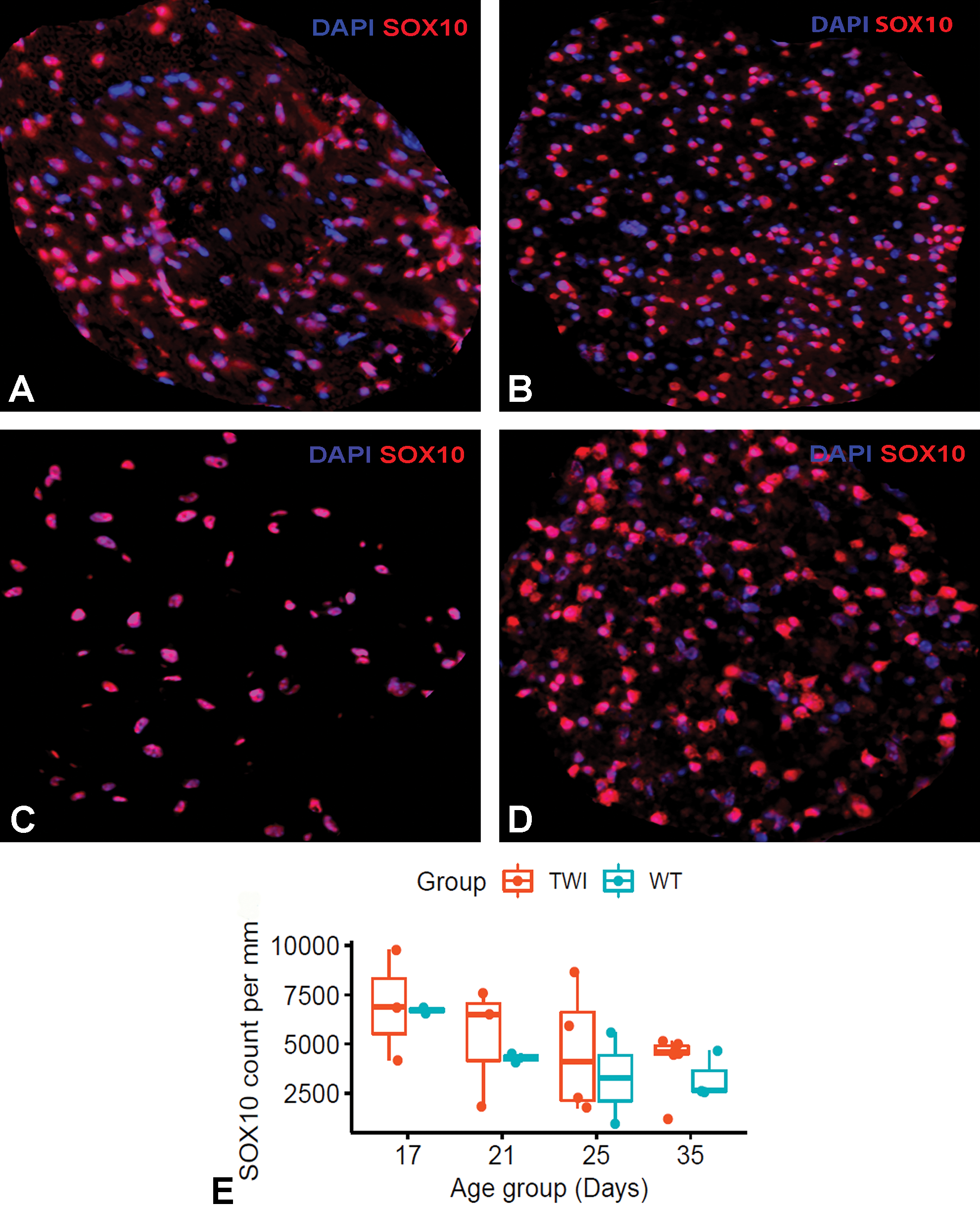
Schwann cell density in the sciatic nerve over time. Images of sciatic nerves fluorescently labeled with DAPI (blue) and Sox-10 (red) from wild-type (WT) (A, C) and Twitcher (TWI) (B, D) mice at 17 (A, B) and 35 (C, D) days of age. Boxplot graphical representation of the number of Schwann cell nuclei within 1 mm2 (E). Linear mixed model to test age-dependent changes confirm that Schwann cell counts significantly decrease with nerve maturation within the measurement period in both healthy and disease mice (
Discussion
Herein, we examined sciatic nerves from a TWI mouse natural history study to elucidate spatiotemporal pathomechanisms underlying the peripheral neuropathy associated with psychosine toxicity. Gathered knowledge on TWI neuropathy has mostly benefited from transmission electron microscopy (TEM) examinations describing mostly qualitative and a few quantitative changes in a limited number of both TWI mice and human KD patients. 30,31,36,39 The overall goal of this current investigational study was to integrate modern pathology tools to traditional anatomic pathology microscopy workflows, in order to generate quantitative data in a large number of nerve samples and aid to the interpretation and understanding of complex disease processes in a timely efficient manner.
The pathogenesis of KD is still a matter of investigation and an unresolved debate in regard to the temporal sequence of demyelination, macrophage infiltration, and axonal damage. Despite demyelination is described as the hallmark feature of TWI nerves, 40 preliminary neuropathological examinations in our own TWI colony based on H&E stained sections revealed macrophage infiltration as the most prominent feature, in particular, in the PNS. These observations led our focus on confirming or refuting our hypothesis that early activation of macrophages is a key event in the pathogenesis of experimental KD and may occur prior to nerve demyelination. Our IHC data argue that demyelination might distinctly be a cardinal feature of TWI nerves only at 35 days of age, challenges previous data reporting demyelination as early as 10 days of age 24,25,31 and suggests that inflammation is the major early pathogenetic event. One could argue that mild demyelination could be present earlier in our mouse model but below detectable levels using our approach since measurements in teased nerve fiber preparations reported the low incidence of demyelinating events in 2-week-old TWI mice. 24 But instead, at 5 weeks of age, quantitative analyses from montages of electron micrographs of peroneal nerves from TWI mice detected 10% of myelinated fibers as demyelinated. 25 Our analyses counting pNF+ axons devoid of a PXN+ myelin sheath confirmed an increase of 10% of nonmyelinated fibers in TWI sciatic nerve as early as 21 days of age. In addition, nerve quantitative analyses confirmed that the number of nonmyelinated axons involving larger size axons was a distinct feature of 35-day-old mice. And last, our quantitative approach was sensitive enough to detect 1- to 2-fold increases in nonmyelinating fibers from 21- to 35-day-old in TWI sciatic nerves when compared with age-matched nerves from WT mice. Altogether these results provide clear-cut evidence of ongoing demyelination at the terminal stage of the disease. Importantly, our observations are supported by ultrastructural G-ratio changes detected at day 30 but not at day 9. 39 Future digital analyses could measure myelination status by means of calculating the G-ratio, defined as the ratio of the axon diameter to the total nerve fiber (axon plus myelin) diameter. The onset of demyelination at 35 days of age coincided with a significant further increase in intranerve CD68+ macrophages, representing 20% of the total cells within the nerve. These macrophage cell counts correlate well with the similar 30% increase in Iba1+ area of monocytes and infiltrating macrophages previously reported in 35-day-old TWI nerves. 41 Since early cycles of demyelination and remyelination do not necessarily result in extensive macrophage infiltration, 26 the question remains whether the early macrophage infiltration in the TWI nerve is the reaction to postnatal psychosine build-up in an attempt to maintain the homeostatic environment of the peripheral nerve.
Our global PXN, pNF, and Sox-10 quantitative digital image analyses confirmed that TWI nerves have suboptimal axonal size and lower myelination despite larger number of Schwann cells. Altogether, these data argue that TWI mice may suffer first from peripheral hypomyelination in association with inefficient remyelination most likely due to Schwann cell dysfunctionality for most part of their life span. Both cIHC and IF for pNF highlighted that many 17-day-old TWI axons, while myelinated, do not reach the averaged mature WT size and remain small even at 35 days of age. The reduction in size of myelinated and unmyelinated axons is a well-described feature of the TWI nerve. 30,31,36 In fact, TWI nerves can show reduced axonal density in semi-thin sections as early as 9 days of age. 39 As part of nerve maturation, reduction of Schwann cell numbers in healthy nerves occurred progressively after 17 days of age with the lowest count at 35 days of age, when 80% of the axons contain mature myelin. Twitcher nerves followed a similar trend but Schwann cell counts remained 2- and 3-fold higher than WT levels during nerve development (3-4 weeks of age) and at final maturation (5 weeks of age), respectively. It remains unknown why despite having larger numbers of myelinating cells, TWI nerves contained significantly less mature myelin than healthy nerves by 21 days, yet in the absence of demyelination. This observation, which is supported by previous ultrastructural work, 36 may point toward a Schwann cell dysfunction and limited plasticity to switch function to a repair phenotype. Early pathological accumulation of psychosine and macrophages could affect the proliferative capacity of Schwann cells 42,43 and nerve myelination due to loss of orchestration between phagocytosis and Schwann cells. 44,45
Platform Considerations When Applying Quantitative Digital Pathology to Immunostained Sections
We believe that applying similar IHC-based histopathological workflows in the current setting of modern pathology with access to digitized quantitative tools will aid the understanding of complex pathological processes and provide the investigational, toxicologic and diagnostic pathologist with new data-driven hypothesis. As one may expect there are multiple important considerations when designing this type of workflow. Increasing the number of animals per combination of age and genotype would have increased statistical power facilitating data interpretation and improved the robustness of our data. Standardization of tissue fixation and sample processing across samples processed in different batches (ie, cohorts of different age) is key since one of the variables of working with formalin-fixed paraffin-embedded (FFPE) is differences in tissue shrinkage during fixation or paraffin processing. Accordingly variation in artificial shrinkage of nerve structures after immersion fixation or suboptimal fixation could play a confounding factor to increasing or reducing the quantified intercellular space, or the total area of a structure. As one would anticipate, digital imaging analyses on IHC-stained sections of suboptimal quality and inaccurate computational algorithms will not provide robust data due to the overrepresentation of false-positive and/or false-negative measurements, and similarly it will not allow other laboratories to reproduce results reliably. The role of the pathologist is also to ensure that the final data are robust and quantitates tissue immunolabeling accurately.
One of the biggest limitations and drawback when performing multiplex immunolabeling is that antibodies need to be raised on different host species. Anticipating the questions that you need to answer might help elect specific antibodies and save time. For examples in order to quantify myelinated axons, we combined a well-established clean mouse pNF antibody with a robust rabbit PXN antibody. In addition, nuclear markers might prove beneficial over cytoplasmic or membranous once since it is easier to count nuclei that are positive for the antibody of interest from a total of DAPI+ nuclei.
Concluding Remarks
This study provides several novel examples on how a digital pathology workflow on immunolabeled slides can generate robust and accurate data sets that aid to the understanding of pathogenetic studies. Overall, our data support a primarily and progressive defect on myelinogenesis detectable in TWI mice after 17 days of age but with evidence of segmental demyelination and axonal enlargement only at 35 days of age. Future studies could characterize the onset of axonopathy in TWI nerves since a large set of studies have reported features such as fast axonal transport impairment, poor axonal development, or reduced motor conduction velocities in TWI nerve, all in the absence of demyelination. 31,46 –50
Of great importance, this workflow detects and quantifies complex features such as changes in myelination status of nerve fibers or axonal size, providing a fast and high-throughput ancillary approach to the gold standard H&E histopathological examination or TEM examination while overcoming the limitations of semiquantitative subjective pathology scoring.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623321991469 - Quantitative Assessment of Neuroinflammation, Myelinogenesis, Demyelination, and Nerve Fiber Regeneration in Immunostained Sciatic Nerves From Twitcher Mice With a Tissue Image Analysis Platform
Supplemental Material, sj-docx-1-tpx-10.1177_0192623321991469 for Quantitative Assessment of Neuroinflammation, Myelinogenesis, Demyelination, and Nerve Fiber Regeneration in Immunostained Sciatic Nerves From Twitcher Mice With a Tissue Image Analysis Platform by Irene Wilson, Cathy Vitelli, Guoying Karen Yu, Glenn Pacheco, Jon Vincelette, Stuart Bunting and Sílvia Sisó in Toxicologic Pathology
Footnotes
Acknowledgments
The authors would like to thank the faculty from Dominican University of California and Biomarin, in particular Dr Meredith Protas and Kathryn Davidson, for their unconditional support to the Masters of Science Biology program. In addition, we are thankful to Stella Breslin from the Morphologic Imaging Core (Buck Institute, Novato, CA) who provides us with excellent histological and ultrastructural support, and to our colleagues at the Buck Institute who take extreme care of our rodent colonies at the Biomarin animal vivarium. Lastly, the authors would also like to acknowledge the important work of the Biomarin ARC members for critical revision of in vivo studies to ensure ethical use of animals.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of BioMarin Pharmaceutical Inc.
Funding
The author(s) received financial support from BioMarin Pharmaceutical Inc. for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
