Abstract
A Working Group of the Society of Toxicologic Pathology’s Scientific and Regulatory Policy Committee conducted a technical and scientific review of current practices relating to the fixation, trimming, and sectioning of the nonrodent eye to identify key points and species-specific anatomical landmarks to consider when preparing and evaluating eyes of rabbits, dogs, minipigs, and nonhuman primates from ocular and general toxicity studies. The topics addressed in this Points to Consider article include determination of situations when more comprehensive evaluation of the globe and/or associated extraocular tissues should be implemented (expanded ocular sampling), and what constitutes expanded ocular sampling. In addition, this manuscript highlights the practical aspects of fixing, trimming, and sectioning the eye to ensure adequate histopathological evaluation of all major ocular structures, including the cone-dense areas (visual streak/macula/fovea) of the retina for rabbits, dogs, minipigs, and nonhuman primates, which is a current regulatory expectation for ocular toxicity studies.
This Points to Consider article is a product of a Society of Toxicologic Pathology (STP) Working Group commissioned by the Scientific and Regulatory Policy Committee (SRPC) of the STP. It has been reviewed and approved by the SRPC, Executive Committee of the STP, the European Society of Toxicologic Pathology, and the British Society of Toxicological Pathology, but it does not represent a formal Best Practice recommendation of the Society; rather, it is intended to provide key “points to consider” for the toxicologic pathology community. The points expressed in this document are those of the authors and do not reflect views or policies of the employing institutions. Readers of Toxicologic Pathology are encouraged to send their thoughts on this article or ideas for new topics to the editor.
Introduction
Many ocular structures, particularly in the anterior segment, are present circumferentially around the eye. Because of this, specific orientation for trimming and sectioning of the eye (beyond capturing the optic disc in the section) has historically often not been considered in general toxicity studies (ie, studies with a nonocular route of administration) in most nonrodent species. Although histopathological examination of more than one section of the eye is standard for ocular toxicity studies at many laboratories, there are no existing recommendations pertaining to this topic. Moreover, the nonhuman primate (NHP) is the only nonrodent species for which the anatomical features of the posterior segment have been routinely considered, with a single section generally oriented horizontally to capture the macula/fovea in addition to the optic disc.
Recent developments in ocular drug discovery and safety programs have prompted re-evaluation of these traditional approaches. The increased use of newer clinical modalities (such as optical coherence tomography [OCT]) 1- 4 has resulted in far more detailed in-life ophthalmic observations in nonclinical ocular toxicity studies and in turn an increased need to capture correlating microscopic lesions. Methodological disparities in design and conduct of ocular toxicity studies between different laboratories could be problematic when studies performed at different institutions are compared (as is often the case when studies from multiple laboratories are bundled together in a single regulatory submission).
The current regulatory expectations for ocular toxicity studies include (1) adequate microscopic assessment of a full range of ocular tissues; (2) microscopic evaluation of a sufficient number of eye sections, including assessment of the retinal cone-dense areas; (3) consideration of in-life observations, including ophthalmologic examination findings, with regard to sectioning the globe; and (4) the justification for the section/assessment approach by the study pathologist. 5 Although it is a widely accepted requirement that evaluation of NHP ocular toxicity studies should include examination of the macula and fovea, requests for examination of the cone-dense areas of the posterior retina (ie, visual streak) in other nonrodent species are new, and the need for multiple sections is not aligned with current industry practices.
In response to these developments, the Society of Toxicologic Pathology (STP)’s Scientific and Regulatory Policy Committee established the Non-Rodent Ocular Trimming and Sectioning Working Group (NOTS-WG) to conduct a technical and scientific review of current practices relating to the fixation, trimming, and sectioning of the nonrodent eye, in order to identify key points to consider when preparing and evaluating the nonrodent eye and extraocular tissues in ocular and general toxicity studies.
This publication defines expanded ocular sampling and discusses the appropriate circumstances for its inclusion in a study. In addition, it describes the advantages and disadvantages of different fixation and trimming methods in the commonly utilized, nonrodent, toxicology species, that is, the NHP, minipigs, dogs, and rabbits. Rodents are not considered in this manuscript as their small globe size inhibits complex trimming and limits their utility for many ocular toxicity studies.
General Points to Consider
Anatomy and Histology Considerations
Rods and cone photoreceptors are the two main types of phototransductive cells in the retina. These are specialized neuroepithelial cells which contain visual pigments embedded within stacks of membranous discs or infolded surface membranes in their outer segments. Mammalian visual pigments are composed of a chromophore (11-cis retinal) bound to an opsin (rhodopsin in rods or a cone opsin). 6- 9 Light interacts with the visual pigments, triggering membrane hyperpolarization. The resultant electrochemical signals are transmitted through bipolar and ganglion cells, through the optic nerve and to the lateral geniculate nucleus and other visual processing centers in the brain. 10 There are specific functional differences between rod and cone photoreceptors. Rods are the predominant photoreceptor in the retina of most mammals, are highly light sensitive, and are responsible for achromatic (black-and-white) and scotopic (low light) vision. Cones are less light sensitive and confer photopic (color) vision. 11 Cones are far less prevalent (≤5% of total photoreceptors) than rods in both humans and the commonly used nonrodent laboratory species, and cone populations are concentrated in specific, cone-dense regions of the retina where they confer high resolution vision (Figure 1). 12- 18
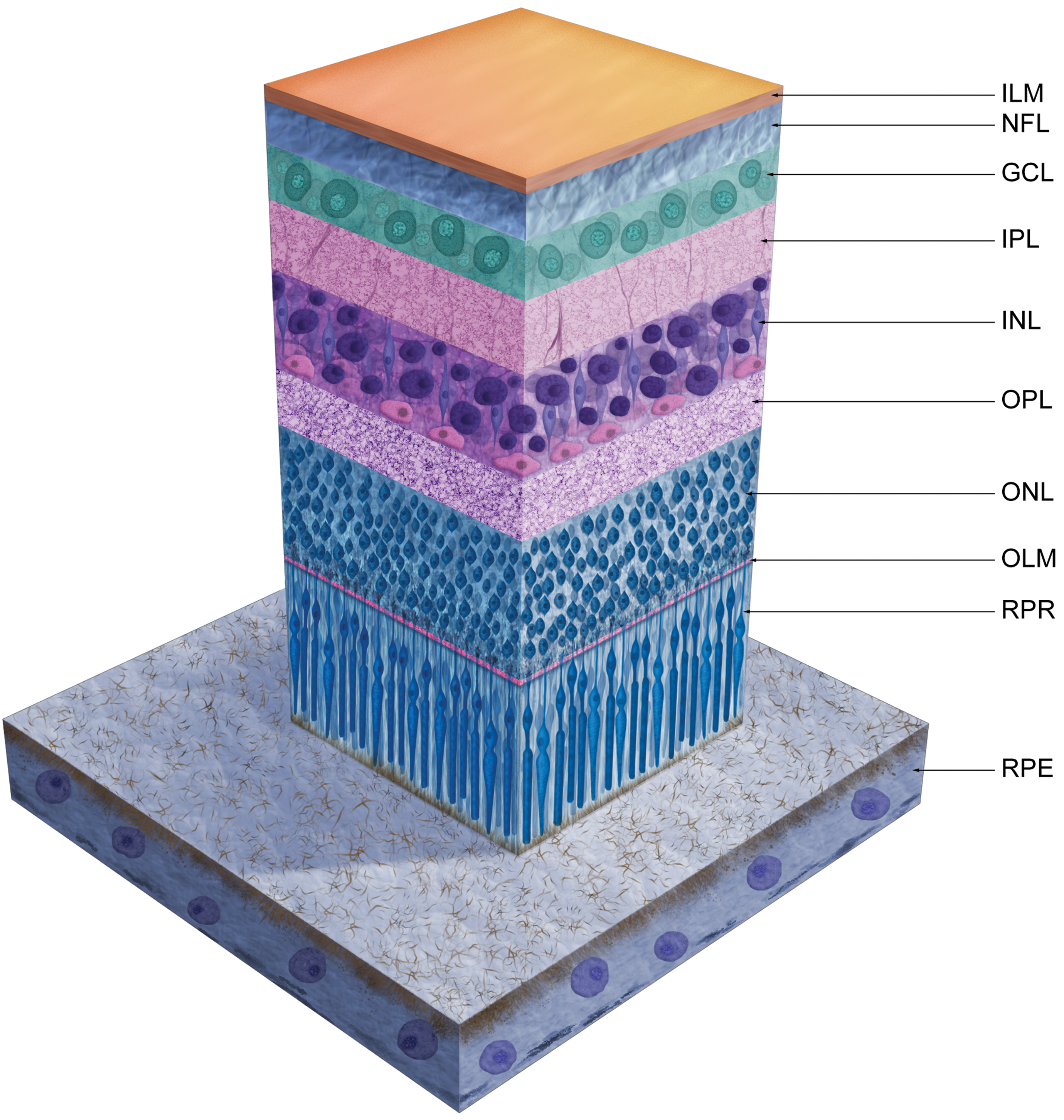
Schematic diagram of normal retina and retinal pigment epithelium. GCL indicates ganglion cell layer; ILM, inner limiting membrane; INL, inner nuclear layer; IPL, inner plexiform layer; NFL, nerve fiber layer; OLM, outer limiting membrane; ONL, outer nuclear layer; OPL, outer plexiform layer; RPE, retinal pigment epithelium; RPR, rod/cone photoreceptor processes extending into the subretinal space.
Cone-dense regions of the retina have developed due to evolutionary pressure to provide enhanced visual acuity in specific parts of the visual field. The character, extent, and location of these regions exhibit significant variability between species, driven from an evolutionary standpoint by ecological and behavioral features. 19
In foveate primates like macaques and humans, the cone-dense area is the macula/fovea. The macula is apparent on ophthalmoscopy or gross examination of the eye as a circular avascular area of the central temporal retina, with a distinctive yellow color imparted by carotenoid pigment (Figure 2A and B). 20 As the center of the visual field and the region of greatest visual acuity, it is a region of high cone density and includes the fovea in its center—an area almost completely devoid of rods. 21 The histologic hallmark of the macula is increased ganglion cell density (multiple rows; Figure 3I) in the ganglion cell layer, which results from centrifugal displacement of ganglion cells away from the central fovea (Figure 3J). This focal radial displacement of ganglion cells, and most other retinal nuclear and plexiform elements, creates a small circular to oval depression or pit known as the retinal fovea centralis (“fovea”). 22
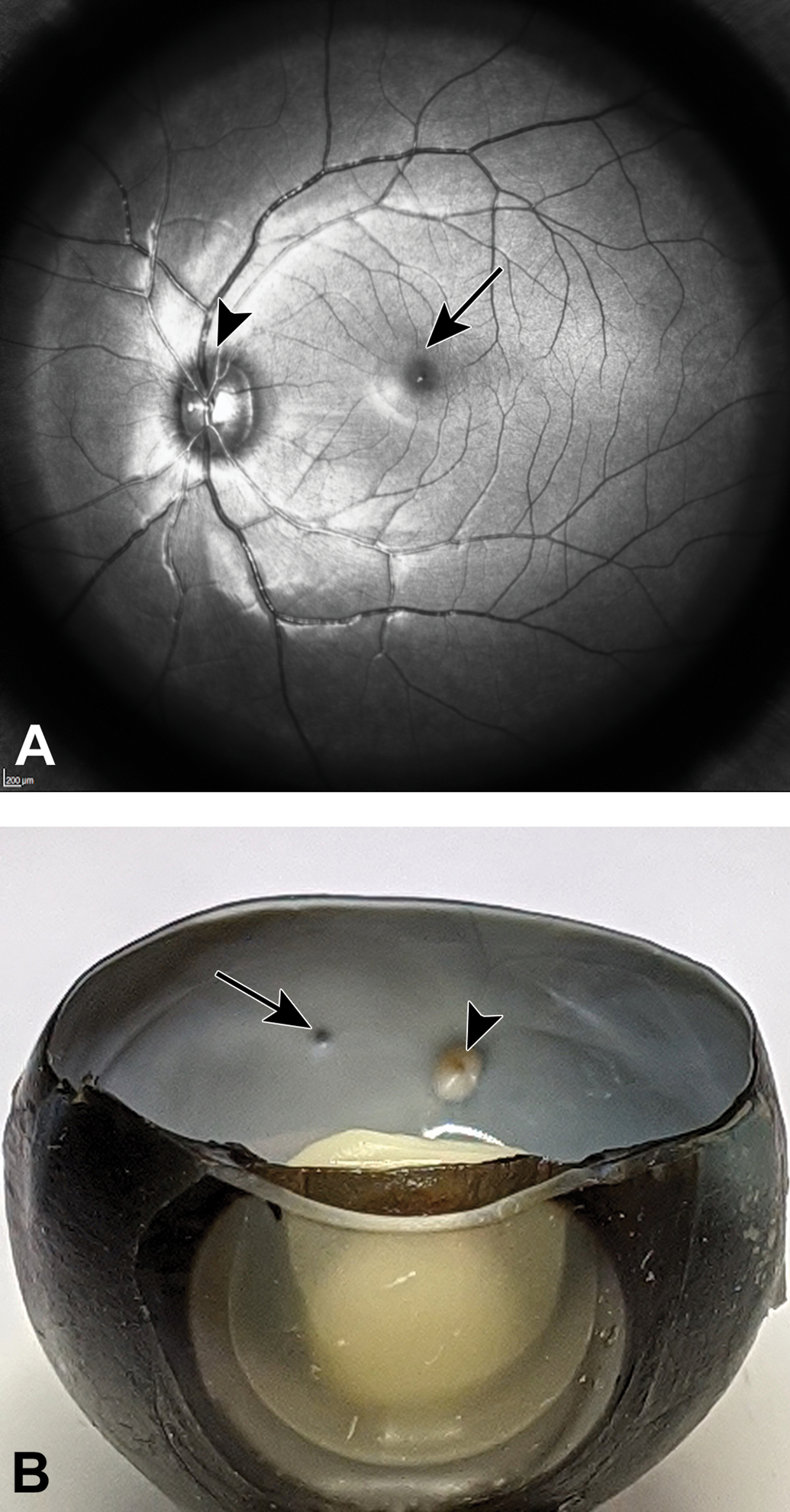
Macula and fovea of nonhuman primate on scanning laser ophthalmoscopy (SLO) and macroscopic examination. A, On an SLO view of the fundus (left eye), the macula/fovea appears as a circular avascular area in the central retina (arrow); arrowhead = optic disc. B, Following horizontal trimming of a right eye, the fovea is grossly apparent as a small pit (arrow) closely aligned horizontally with optic disc (arrowhead).
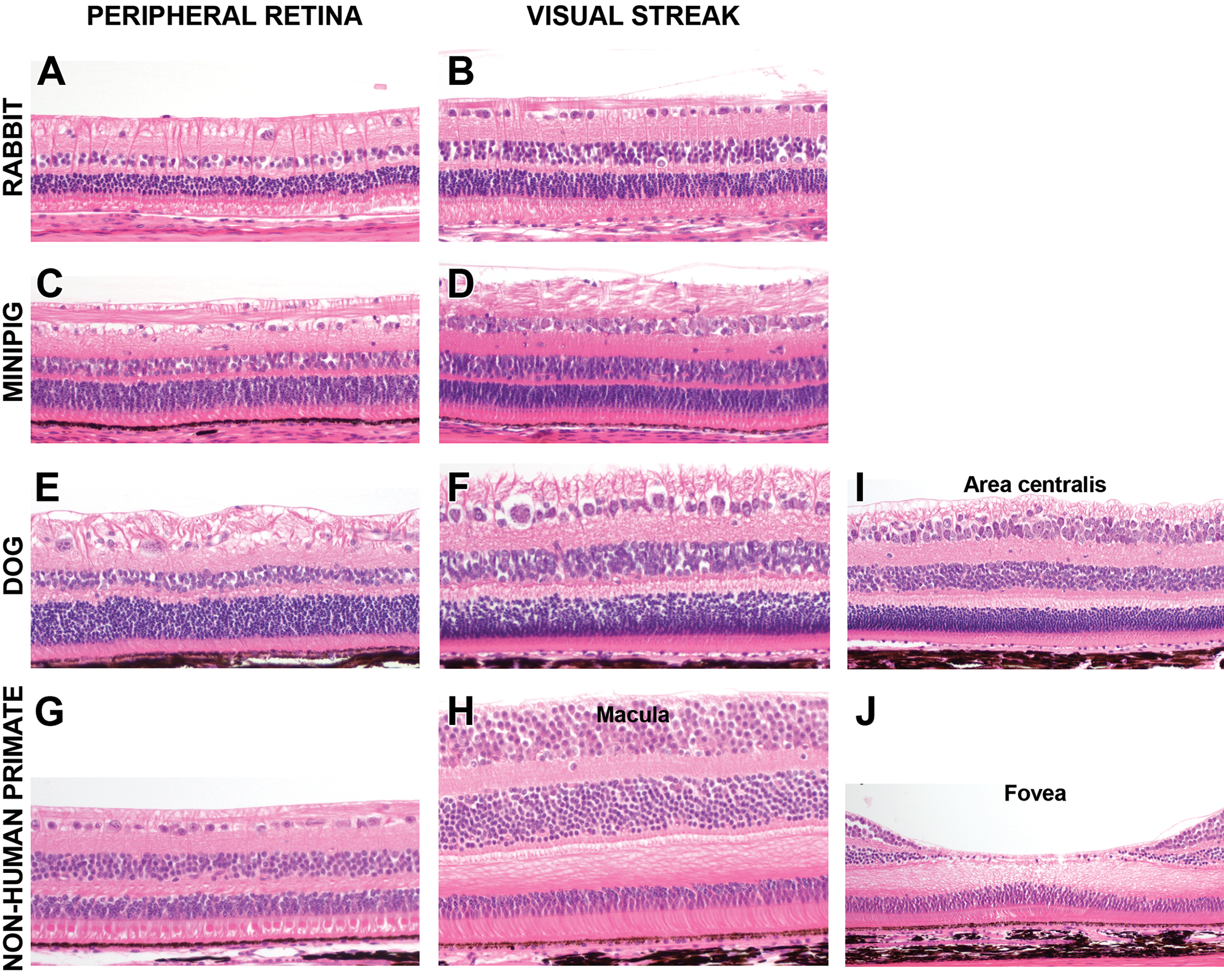
Peripheral retina versus cone-dense regions. Depending on species, visual streak/macula are characterized by tightly packed and/or multilayered ganglion cells in the ganglion cell layer and large numbers of cone nuclei in the outer nuclear layer. Fovea and area centralis are the center subregion of highest cone density within the nonhuman primate’s macula and dog’s visual streak, respectively. GCL indicates ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer. Hematoxylin and eosin stain, 20×.
Despite common misconceptions, primates are not the only species with a cone-dominant region in the retina. Cone-dense visual streaks have been identified in all commonly utilized nonrodent laboratory species—rabbits, dogs, and minipigs. 16,23- 27 In the past, there have been some discrepancies in the terminology used in nonprimate species; some publications refer to the visual streak as the area centralis. In this article, we use the term area centralis specifically to describe the small, focal subregion of the visual streak with the highest cone density, which (among the nonprimate species commonly used in ocular toxicity studies) is present only in the dog. 25,26,28 Despite the presence of a cone-dense visual streak in all of these species, NHPs are the only species for which attempts to sample and examine the cone-dense region have been routinely made in ocular and general toxicity studies.
Unlike the primate macula, the visual streak of rabbits, dogs, and minipigs is not visible on ophthalmoscopy or gross examination. In these species, the visual streak is a broad, horizontally orientated, gradually tapering, cone-dense region, which is only identifiable histologically, and can be recognized by an increased density of ganglion cells, which (depending on the species) may form multiple layers (Figure 3).
The location and size of cone-dense regions of the retina is species dependent, and even within a species there can be significant variability (Figure 4). In the dog, the visual streak is a horizontal band superior and predominantly temporal to the optic disc in the tapetal (superior) portion of the retina. The visual streak extends nasally to a variable extent, and ganglion cells are more numerous and densely packed in this region compared to the peripheral retina (Figure 3E and F). 25,29 Within the dog’s visual streak is the area centralis, a superior-temporal circular subregion approximately 200 to 300 µm in diameter (authors’ semiquantitative assessment) in which the ganglion cells are tightly packed and arranged in two to three layers, resulting in a focal retinal thickening/bulging (referred to by some authors as the “foveal bouquet”; Figure 3E). 28 Similar to the macula in the primate, the temporal portion of the canine visual streak is generally free of larger blood vessels, a feature which may be observed on fluorescein or OCT angiography. 28- 30 Interestingly, in the dog there is significant interindividual and interbreed variability in the horizontal, nasal extent of the visual streak, but the location of the area centralis (superior and temporal to the disc) remains consistent. 25
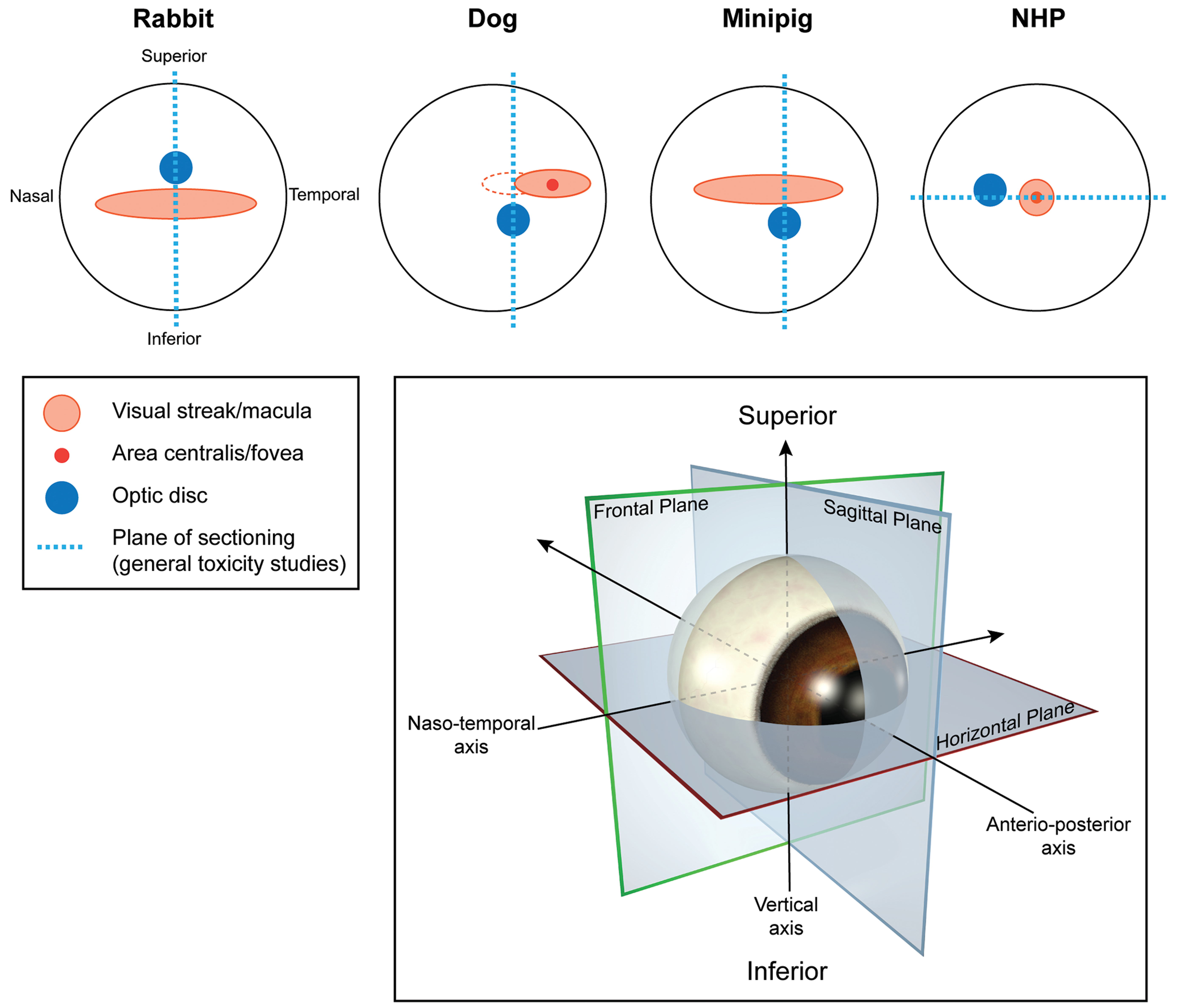
Topographical location of cone dense regions in nonrodent left eyes and location of standard eye sections examined in general toxicity studies. Visual streak in the rabbit and minipig is a broad horizontal band inferior (rabbit) or superior (minipig) to the optic disc. In the dog, the visual streak is a horizontal band, mainly superotemporal to optic disc and variably extending on the nasal side (dotted red line). Regardless of nasal extension of the visual streak, the dog’s area centralis position remains the same within the temporal visual streak. In the nonhuman primate, the cone-dense area is the circular macula with a central fovea composed predominantly of cones.
In the rabbit and minipig, the visual streak is much more extensive than in the dog and is characterized by a horizontal band of increased ganglion cell and cone density, which extends temporally and nasally. In the rabbit, the visual streak is located inferior to the optic disc and medullary rays (the horizontal bands of myelinated nerve fibers centered on the optic disc) and is characterized by a ganglion cell layer that is a single cell layer thick, but has increased cell density (Figure 3B). 24,31 In the minipig, the visual streak is superior to the optic disc, and ganglion cells are densely packed and form 1 to 2 layers (Figure 3D). 16,27 However, the visual streak of minipigs is not characterized by the focal retinal thickening/bulge that defines the area centralis as observed in dogs. Although the visual streak has been incorrectly referred to as the area centralis in some publications regarding the pig retina, 13,32- 34 the region of high ganglion cell density in this species should be designated as the visual streak, and area centralis terminology should not be used in microscopic evaluation of minipig eyes.
As described above, there are significant functional and anatomical differences between rods and cones as individual cell types. In addition, the retinal cone-dense regions have far greater photoreceptor and ganglion cell density compared with the peripheral retina, which places a high metabolic and phagocytic load on the subjacent retinal pigment epithelium. 35 Large blood vessels tend to skirt areas of high cell density (these regions being vascularized rather by dense beds of fine capillaries) 28,36,37 and both Bruch membrane and the inner limiting membrane are often thinner in these locations. 38 These differences in biochemical composition, function, and anatomy present the potential for retinal toxicity to be cone-specific. Despite this potential, such examples have not been reported in nonclinical studies and have only rarely been observed in humans. 39 It is important to explore the potential for cone-specific ocular toxicities by ensuring that the cone-dense regions of the retina are examined as cone-specific toxicities may be difficult to identify if these regions of the retina are not assessed.
In the context of general toxicity studies, ocular exposure is usually low compared to studies with direct ocular administration. Blood-ocular barriers formed by tight junctions between retinal pigment epithelial cells (RPEs), ciliary epithelial cells, and the retinal and iris capillary endothelial cells limit the exposure of the eye to agents administered systemically. Although small, lipophilic compounds can permeate blood-ocular barriers, permeation of hydrophilic compounds and large molecules is significantly restricted. 40 Within the setting of toxicologic pathology, organs and tissues cannot be examined in their entirety, and only representative samples are assessed histologically. 41 For this reason, examination of a single section of each eye, which encompasses all major ocular structures, is an STP recommendation for general toxicity studies. 42 The NOTS-WG believes this STP recommendation is sufficient in the context of a general toxicity study for nonocular routes of administration with molecules for which there is no known ocular liability. However, with ocular toxicity studies, there is greater local exposure and a higher incidence of test article-, vehicle-, and/or procedure-related findings is likely to be observed on ophthalmic examination and ocular histopathology. Likewise, occasionally, general toxicity studies may have a target, off-target, or class effect on the eye and/or key ophthalmic examination findings. In these situations, examination of a single section of the globe may not be sufficiently comprehensive and an expanded ocular sampling protocol which provides multiple sections of each eye for histopathological evaluation should be implemented.
Whenever possible, the pathologist should be consulted prior to protocol and study-specific procedure development, in order to ensure the best possible necropsy, trimming, and sectioning methods are used for any given study.
Considerations for Tissue Collection
Generally, the eyes are fully enucleated at necropsy. The globe should be marked with a suture or tissue ink at the 12 o’clock position to enable orientation during trimming. Left and right eyes should be clearly differentiated, preferably by collection into separate containers or by physical labeling with tissue ink or sutures.
If there is a particular interest in the interrelationship between lesions in the adnexa, conjunctiva, and cornea (eg, topical-administration studies requiring an intact fornical conjunctiva; subtenon or retrobulbar studies in which the relationship of injected drug depots and adjacent ocular tissues must remain intact), en bloc dissection, fixation, trimming, and sectioning of the eye and adnexal tissues may be performed.
Collection of aqueous humor and/or vitreous humor samples at necropsy can be valuable for many reasons, including the assessment of ocular pharmacokinetics and antidrug antibody levels, which may provide valuable context regarding in-life findings (such as inflammation associated with immunogenicity). 43- 45 In addition, maximizing the data obtained from each animal on study by collecting/using these samples may decrease the number of animals used overall by a development program. For nonrodent species, collection of up to 100 µL of aqueous humor is feasible at necropsy, via paracentesis of the anterior chamber with a 25-gauge needle/1 mL syringe, and should not induce artifactual changes that would impact the interpretation of the study. Vitreous humor samples of a similar volume may also be collected, however it is important to note that the vitreous of nonrodent species used for ocular toxicity studies is generally viscous and heterogeneous in composition (even in NHPs), and the sample collected may not therefore be representative of the entire vitreous. 46 Collection of terminal vitreous humor samples may produce some traction artifacts, particularly buckling of the iris or detachment of the retina 47 ; marking the site of collection with tissue ink may help in interpretation of any iatrogenic artifacts. Iatrogenic artifacts can be minimized by replacing the extracted vitreous with a similar volume of fixative; however, given that data obtained from vitreal samples are often inconsistent and difficult to interpret, the potential to compromise the histopathology should be weighed against the expected value of the vitreal sample.
Considerations for Fixation and Processing
Regardless of the study design, the eye is usually fixed whole to minimize artifactual changes. Since there is no “universally ideal” fixative for all species, tissues, or experimental procedures, selection of the most suitable fixative and fixation method(s) requires careful consideration of the study design and experimental goals, including possible downstream, investigational, and molecular pathology end points.
For light microscopy of paraffin embedded tissues, Davidson solution (DS: 10% glacial acetic acid, 35% ethanol, 2% [37%-40%] formaldehyde, and 53% water) or modified Davidson solution (MDS: 5% glacial acetic acid, 15% ethanol, 30% [37%-40%] formaldehyde, and 50% water) are generally the primary fixatives of choice for immersion fixation of the eye in most ocular and general toxicity studies. 48- 50 Immersion fixation in DS/MDS for about 24 to 48 hours is generally recommended for the large eyes of the nonrodent species; longer fixation times should be avoided as DS can cause the globe to over-harden as well as produce artifactual vacuolation of the corneal endothelium, RPEs, retina, and attached optic nerve. Fixation should be followed by thorough rinsing of the tissues under running water. The fixed tissues can then be transferred to either 10% neutral buffered formalin (10% NBF) or 70% ethanol for storage, although prolonged storage in either fixative is not optimal. Immersion fixation in DS was considered by the NOTS-WG to provide the best preservation of ocular tissues, particularly for topical studies, as desquamation of superficial corneal epithelium can sometimes be observed in the rabbit eye following fixation in MDS. 47 The preferred fixative for cornea, anterior chamber, and lens is 10% NBF. If the study is investigational, and there is no concern for or interest in the posterior segment, 10% NBF may be an appropriate fixative; however, 10% NBF is not preferred for general fixation of the eye due to the high incidence of artifactual retinal detachment and the less than optimal preservation of retinal morphological details. With DS/MDS fixation, the lens tends to become brittle and is predisposed to fracture during sectioning, which may result in significant artifact. Soaking the surface of the blocks in liquid phenol or glacial acetic acid immediately before sectioning can soften the lens and mitigate this effect, 49 in addition to sectioning slowly with a new microtome blade for each eye.
If the primary study objectives are immunohistochemistry (IHC) and/or in situ hybridization (ISH), different fixatives may also be required. Although many antibodies commonly used for IHC do work on tissues fixed in DS, 51- 53 an IHC/ISH technique that is developed and optimized for formalin-fixed, paraffin-embedded or frozen tissue is sometimes not directly transferable to DS-fixed tissue. Fixation of the eyes in 10% NBF or 4% paraformaldehyde or freezing in optimal cutting temperature compound may be necessary for certain IHC/ISH studies, although these methods will create artifacts that may compromise histopathologic evaluation of certain structures like the retina. In this case, injection of 10% NBF or paraformaldehyde (up to 100 µL) into the vitreous at collection may improve retinal preservation but will also cause iatrogenic artifacts.
For electron microscopy, perfusion fixation (generally limited to the upper body in nonrodent species) with either ½ strength Karnovsky or modified Karnovsky fixatives provides the best ocular (especially retinal) fixation. Care should be taken to minimize perfusion pressure in order to avoid artifactual vacuolation of the retina/subretinal space. If study conditions do not allow for perfusion, intravitreal injection of a 1:1 mix of glutaraldehyde:10% NBF or modified Karnovsky (up to 100 µL) followed by 5 hours immersion in the same fixative with subsequent trimming of the eye into smaller samples is recommended. If the retina is the tissue of interest, removal of the anterior segment to create an “eye cup” is recommended. Compared to perfusion, injection/immersion has the advantage of lower technical complexity but may result in a longer time delay for the fixative to reach the retina, early autolytic changes, and artifacts similar to those associated with collection of terminal vitreal samples.
If the study involves evaluation of (or delivery through) a device, depending upon the materials the device is composed of (and the existing information available regarding the local tolerability of the device), it may be necessary to trim the region of the eye containing the device following fixation, for embedding in plastic. This enables sectioning of the device and associated ocular tissues and allows easier evaluation of local reaction to the device. Plastic embedding is not recommended for the whole eye in these situations, as the sections are somewhat thicker than those usually examined. 54
Considerations for Trimming and Sectioning the Eye: An Introduction to Expanded Ocular Sampling
An expanded ocular sampling protocol is one which provides multiple sections of each eye for histopathological evaluation to allow comprehensive evaluation of ocular structures, including the cone-dense regions of the retina. Expanded ocular sampling should always be implemented for (1) ocular toxicity studies (defined as any study with an ocular or periocular route of administration), (2) general toxicity studies in which there is a known or suspected ocular safety liability associated with the target or class of molecules, or (3) general toxicity studies in which there are unexpected in-life test article-related ophthalmic examination findings. Expanded ocular sampling may also include periocular tissues, especially for ocular toxicity studies. The extent of the expanded sampling or sectioning protocol implemented for a given study may be adapted on a case-by-case basis but should be driven by a strong scientific rationale and tailored to fulfill the experimental objectives.
Considerations for the trimming and sectioning of nonrodent eyes are discussed below, illustrated in Figure 5 and summarized in Table 1, including advantages and disadvantages of each method.
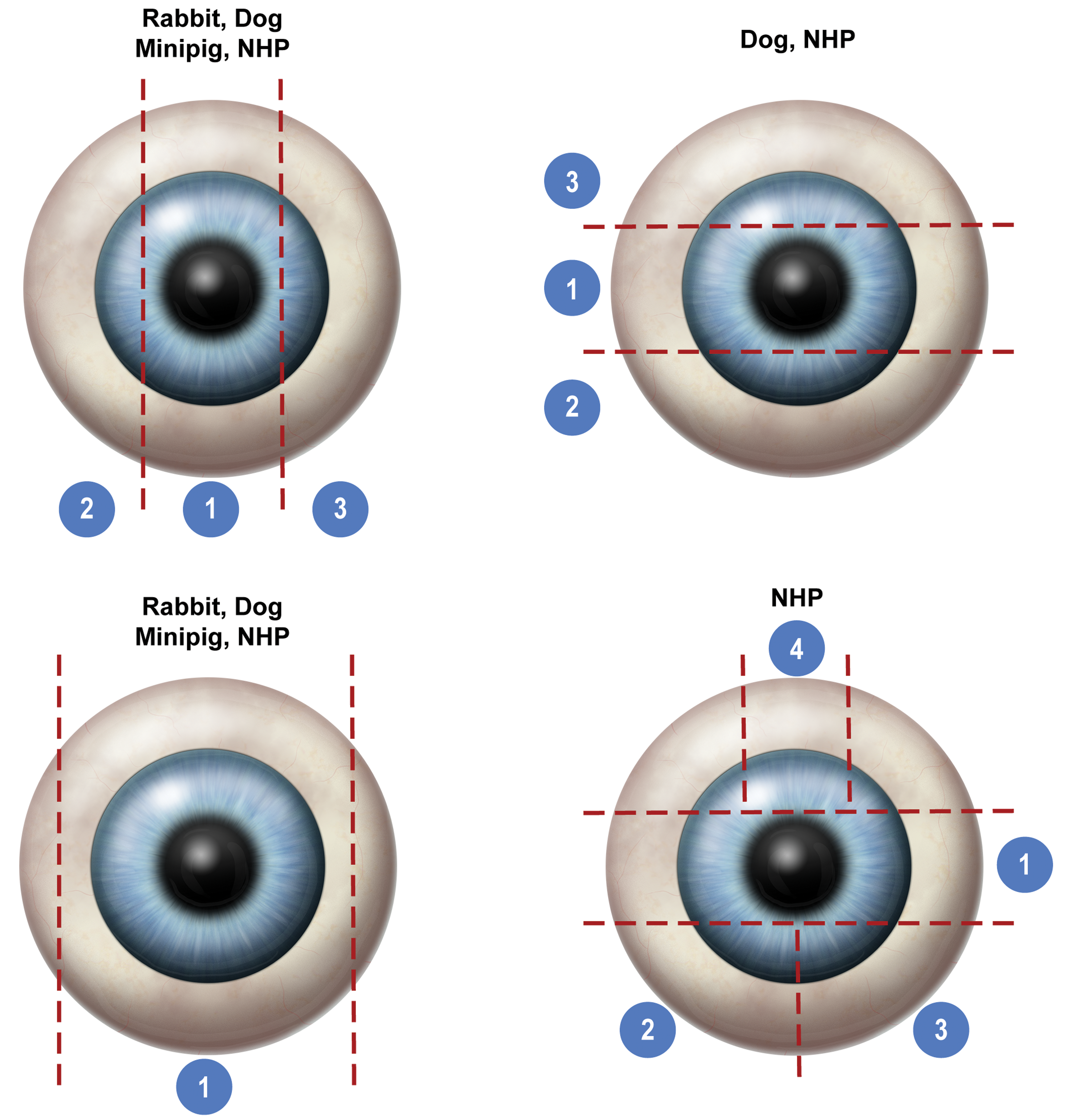
Expanded ocular sampling—example trimming guides. Red dotted lines = location of trimming; numbers = resultant blocks.
Advantages and Disadvantages of Different Eye Trimming Protocols for NHP and Dog.a
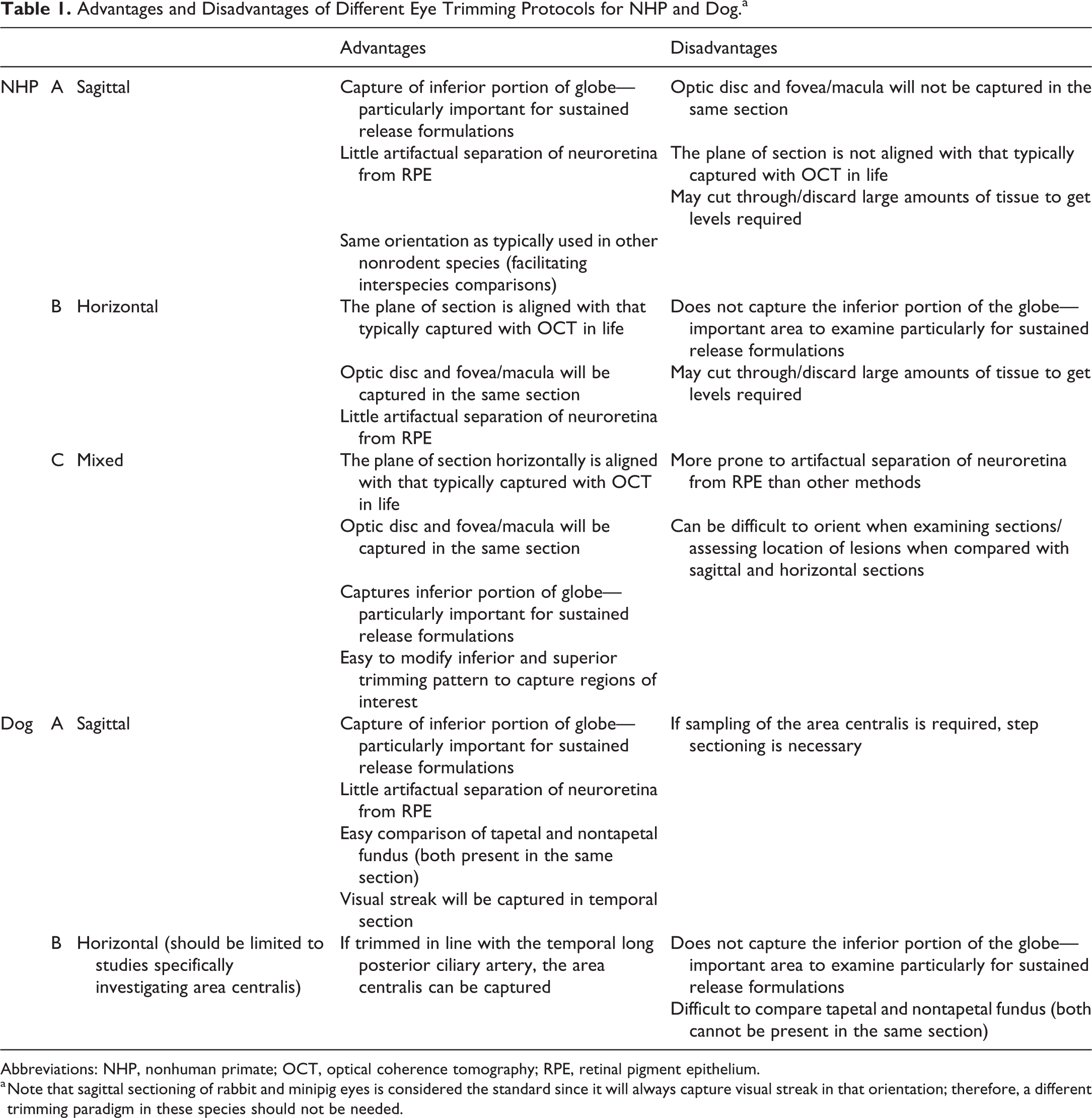
Abbreviations: NHP, nonhuman primate; OCT, optical coherence tomography; RPE, retinal pigment epithelium.
a Note that sagittal sectioning of rabbit and minipig eyes is considered the standard since it will always capture visual streak in that orientation; therefore, a different trimming paradigm in these species should not be needed.
Eye trimming and sectioning methods should account for species-specific anatomy and histology (including cone-dense areas) and ensure that key features such as administration sites, test item (delivery material), and ophthalmic examination observations are appropriately represented in the final sections on the glass slides. These methods can be adapted on a study-by-study basis to best meet the experimental objectives of ocular toxicity studies as well as general toxicity studies. Currently, the majority of ocular toxicity studies are either topical or intravitreal 55 ; however, several other routes may be utilized (Figure 6). 56 Communication between the in-life study team including technical personnel; the veterinarian and/or veterinary ophthalmologist; the necropsy and histology teams collecting, trimming, orienting, and embedding the eyes; and the pathologist is essential to enable thorough characterization of test article-related findings that are initially noted in-life and/or at trimming.
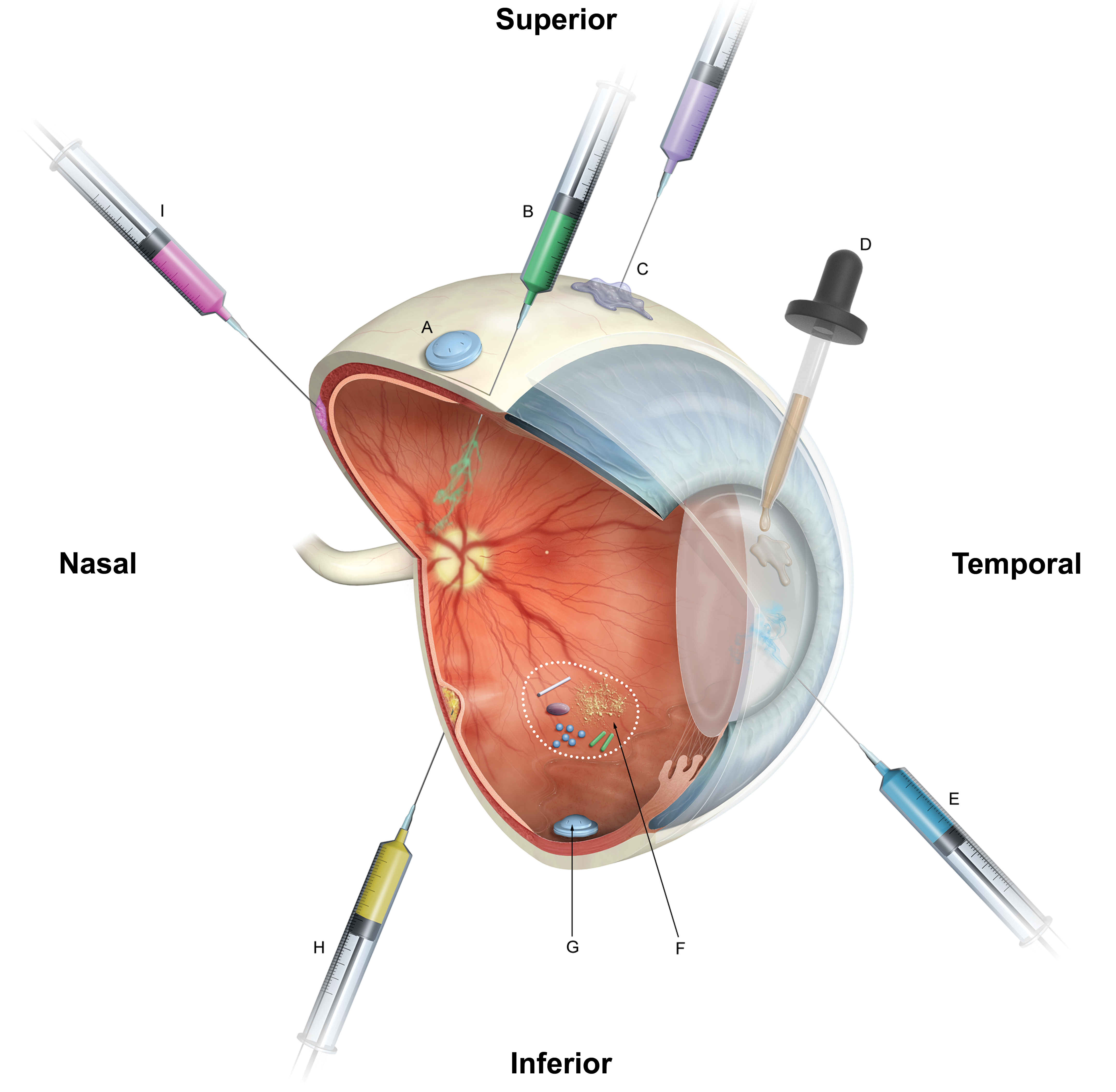
Cutaway representation of a foveate eye with common routes of ocular drug administration included*: A, Episcleral/subconjunctival implant; (B) intravitreal injection (eg, solutions, suspensions, implants [common injection site is superotemporal pars plana]; (C) periocular injection (eg, subtenon, subconjunctival, retrobulbar); (D) topical (eg, eye drops, ointments, gels, emulsions); (E) intracameral injection (eg, solutions, suspensions, free-floating implants); (F) schematic representation (within dotted circle) of the many types of materials that can be injected intravitreally, and in some cases, settle in the inferior floor of the vitreous cavity; (G) scleral-fixated intravitreal implant; (H) subretinal injection; (I) suprachoroidal injection. *Locations depicted are for illustration only and may not accurately reflect the clinical.
Expanded ocular sampling is indicated for all ocular toxicity studies and for toxicity studies with an ocular liability associated with the target or class of molecules or if there are test article-related ophthalmic examination findings. A minimum of 3 sections of each eye, at least one of which includes the cone-dense region of the retina, should be examined.
Species-Specific Points to Consider
Nonhuman Primate
Several different trimming strategies can be utilized for sampling the NHP eye—with horizontal (nasal to temporal), sagittal (superior to inferior), and mixed options appropriate in different situations. All these methods have, as a major consideration, the capture of the macula/fovea (“cone-dense” area). Regardless of the approach, in ocular toxicity studies, the fovea (and surrounding macula) should be present in at least one section, whereas in general toxicity studies, inclusion of the macula is generally considered sufficient.
Since the macula of the NHP is easy to visualize (both the macula and optic disc can be seen ophthalmoscopically and grossly at trimming; Figure 2B) and capture (because of its close horizontal alignment with the optic disc), the NHP eye is most commonly trimmed horizontally, which will produce an excellent single section containing optic disc and macula for examination in a general toxicity study (Figure 4). Additionally, an advantage of examining a horizontal section through the fovea/macula and optic disc is that this section correlates with commonly generated OCT images. 1 In the context of expanded ocular sampling, the remaining eye can also be trimmed horizontally with the choice of producing either three blocks (central, superior, and inferior) which will each be sectioned; or a single, larger block which will be step sectioned. In the latter case, additional, unstained, sections should be collected at each level to ensure material is available for any additional work needed for the study.
In certain contexts, sagittal trimming and sectioning of the NHP eye may be preferred. This provides the option to section primate eyes in the same orientation as other species (for comparison purposes), with the added benefit of more thoroughly evaluating the inferior region of the eye (an area which is a frequent location for depot-based intraocular sustained release formulations and which may exhibit localized changes resulting from this). 57,58 With careful sectioning, an experienced histology technician can locate the fovea in a sagittal block. In this case, however, the fovea and optic disc would need to be captured on separate slides.
“Mixed” trimming methods can also be used for the NHP eye (Figure 5)—that is, a horizontal central section (for optic disc and macula/fovea—and to correlate with OCT images as discussed above) with a resultant inferior calotte that can, in turn, be sectioned sagittally to capture depot material and a superior calotte that can be sectioned to examine the administration site (if present in this region).
Dog, Rabbit, and Minipig
As previously mentioned, for the dog, rabbit, and minipig, current standard ocular trimming schemes in general toxicity studies most commonly implement a single section through the optic disc in a sagittal plane. Sagittal sectioning is particularly important in the dog as this permits comparison of the choroidal tapetum lucidum and overlying retina in the superior globe, with nontapetal choroid and retina in the inferior globe. A sagittal section through the optic disc will always include the visual streak in the rabbit and minipig, however, it is important to note that it may not do so in a proportion of dogs due to individual variation in the extent of this region.
For expanded ocular sampling in these species, additional sagittal cuts, to either divide the eye into central (containing the optic disc), temporal, and nasal blocks, or produce a single larger block, should be made to produce a minimum of three sections/eye and to enable a more comprehensive evaluation of the globe. Additional sections from all blocks (to result in a total of five or more sections per eye) are encouraged to ensure that the visual streak, lens, and (if applicable) the site of dose administration are adequately captured (Figure 3).
Although the dog has a focused subregion of the visual streak with even greater cone density, the area centralis, this is very small (approximately 200-300µm in diameter) and often requires specialized sectioning to capture. 25,28 It is not practical to capture the canine area centralis in the context of toxicity studies, even those implementing an expanded ocular sampling protocol. The NOTS-WG considers the visual streak in the dog representative of the cone-dense area in this species and, therefore, examination of the visual streak is sufficient for histopathological evaluation in ocular toxicity studies in dogs.
Capture and examination of the canine area centralis may be required in the context of specialized investigational studies. Because the area centralis is very small, focal, and lacks a grossly visible depression, sectioning through a block to locate it for subsequent evaluation can be difficult and labor-intensive. In the course of preparing this manuscript, the NOTS-WG members noted that in sagittal sections of the canine eye temporal calotte, the temporal long posterior ciliary artery appears in cross sections within the sclera at approximately the horizontal meridian (along the circumferential line that divides the globe into superior and inferior hemispheres). 59 In sagittal sections of at least 120 Beagle eyes examined by several of the NOTS-WG members, the focal area centralis was invariably located in the retina directly adjacent or slightly inferior to the intrascleral artery cross section (results not published).
Exploiting the anatomic proximity of the area centralis to the temporal long posterior ciliary artery, using the artery as a landmark, the area centralis can be reliably captured in horizontal section. At trimming, a thin line of tissue-ink is carefully applied along the temporal long posterior ciliary artery to mark its course (Figure 7A). At sectioning, the ink mark remains visible and serves as a landmark for the histology technician. Upon reaching the ink mark, two to three step sections 100 µm apart are taken. The area centralis will typically be present in at least one of these step sections (Figure 7B). Since this method does not allow for easy comparison of the transition between tapetal and nontapetal fundus, or the inferior portion of the globe, it is not recommended for most ocular toxicity studies; however, this method may be useful for specific investigative studies.
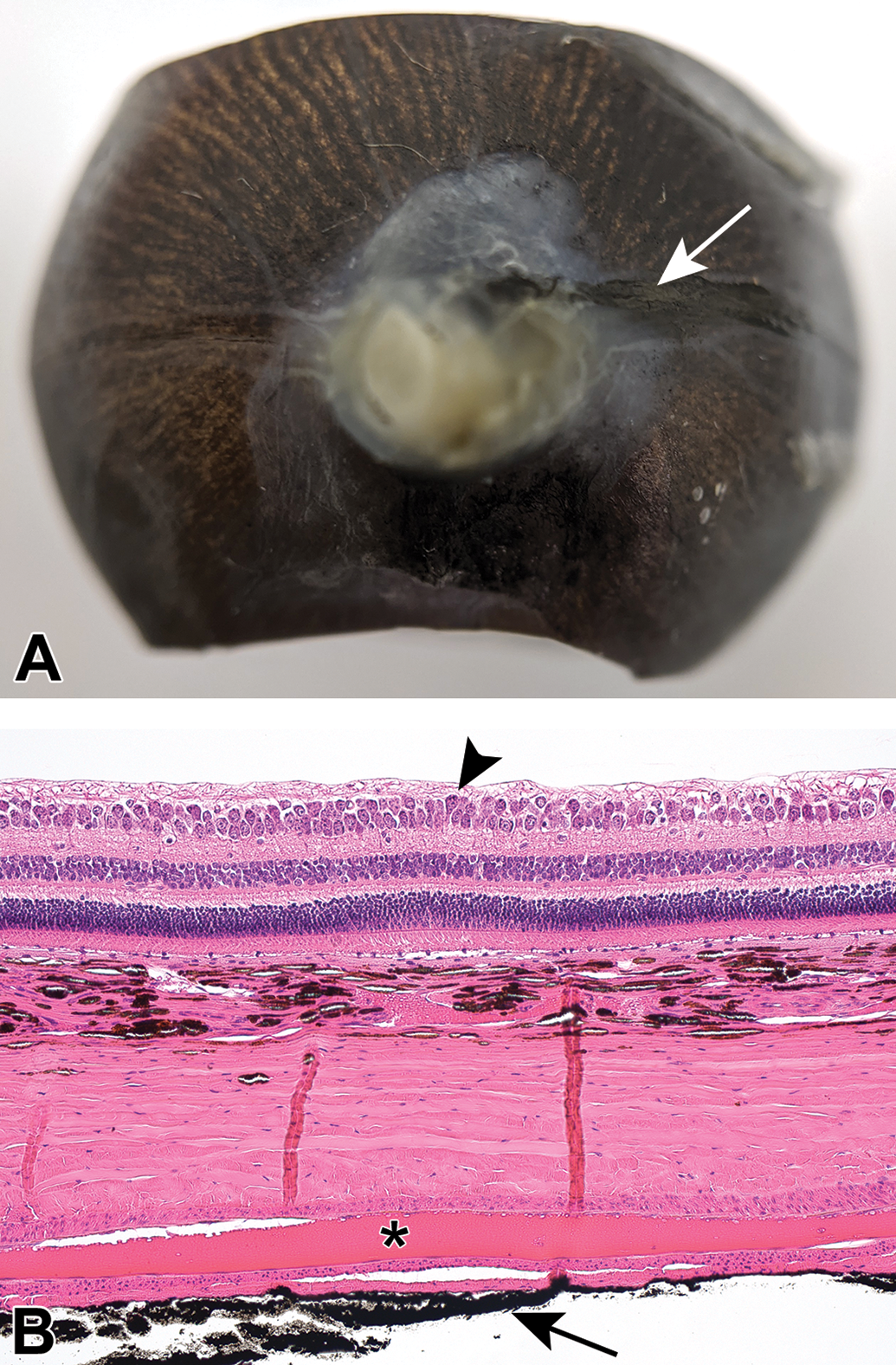
Trimming and sectioning of the dog eye for specific evaluation of the area centralis. If there is a specific interest in evaluating the area centralis, the dog eye can be trimmed horizontally. A, Trimmed right eye; prior to trimming, a thin ink line was applied along the long temporal ciliary artery (arrow) and the eye was trimmed parallel to that line (superior and inferior calottes are trimmed off). During sectioning, the ink mark will serve as a landmark for step-sectioning. B, H&E horizontal section with area centralis (arrowhead) overlying a longitudinal section of the long temporal ciliary artery within the sclera (asterisk) and the ink mark on the outside (arrow). Note that in this horizontal plane of section, the area centralis appears wider and lacks focal retinal bulging compared to the area centralis in a sagittal plane of section (Figure 3). H&E, 20×. H&E indicates hematoxylin and eosin stain.
Specific Trimming and Sectioning Considerations for Ocular Toxicity Studies
For ocular toxicity studies specifically, additional factors relating to the route of administration and the format of the test article (eg, solution, suspension, or sustained release formulation) should be considered.
Topical Ocular Studies
For topical ocular studies, three sections of the eye: central, temporal, and nasal (if the eye is trimmed in a sagittal plane) or central, superior, and inferior (if using horizontal or mixed trimming techniques) are usually considered sufficient to identify potential test article-related findings. Topical agents are most likely to induce findings in the cornea, conjunctiva, ocular adnexal glands and/or lacrimal apparatus. To date, topical agents have not been reported to induce retinal toxicity. Additional sampling (more than three sections) may be required if in-life ophthalmic examination findings will not be captured in the standard sections. In this case, additional sampling should be driven by the location of the findings observed in-life. Evaluation of the conjunctiva is important in topical ocular studies. Bulbar and palpebral conjunctiva are generally examined in sections of the eye and eyelids, respectively. If a more complete examination of the conjunctiva, to include the fornix, is desired, the eye and eyelids may be collected, trimmed, and sectioned en bloc.
Intravitreal Studies
For intravitreal studies, at least three sections of the eye should be examined. These may be central, temporal, and nasal (if the eye is trimmed in a sagittal plane) or central, superior, and inferior (if using horizontal or mixed trimming techniques). One section should include the region of the administration (injection) site (pars plana and adjacent sclera), although it is not usually necessary to section through the eye to capture the administration site itself. To adequately assess liabilities associated with sustained-release formulations, including implants or depot formulations, or local tolerability of the test-article, the inferior portion of the globe, or the location of the material as observed in-life on ophthalmic examination, ocular ultrasound or OCT, should be captured for histopathologic evaluation. For NHP eyes trimmed horizontally, this is particularly important to consider, as the inferior portion of the globe is easily overlooked during trimming/sectioning (Figure 3).
Intracameral Studies
Similar to topical and intravitreal studies, for intracameral studies (where administration occurs into the anterior chamber), at least three sections per eye should be examined. These may be central, temporal, and nasal (if the eye is trimmed in a sagittal plane) or central, superior, and inferior (if using horizontal or mixed trimming techniques). For intracameral studies, particularly if the injection is transcorneal, in-life examinations (including gonioscopy) should be used as guides to capture the injection site, injected materials, and any in-life findings. For those intracameral studies with sustained-release formulations, particularly implants, implants/depots are often located in the inferior iridocorneal angle. Sagittal step sections through this region may be necessary to fully evaluate the area.
Other Routes of Administration
In studies with other administration routes (eg, subretinal, suprachoroidal, subconjunctival, subtenon, retrobulbar), examination of the injection/deposition site(s) is also critical, since test article-related findings are most likely to occur at these locations.
Subretinal Studies
For subretinal injection studies, the location of the bleb (deposition site) will determine the trimming paradigm implemented. Ophthalmic examination results and/or other in-life findings (including diagrams, OCT, or fundus photographic images) should be used to identify landmarks for trimming and sectioning. Sagittal trimming of the globe should be considered in all species including NHP, and at least 2 sections through the bleb should be examined. Test articles in some subretinal injection studies are viral vectors for gene therapy, and these study protocols frequently include IHC and/or ISH assessment of the expression of the gene of interest or a reporter gene such as green fluorescent protein. Therefore, in such studies, sectioning plans frequently include collection of multiple sections at multiple locations/steps within the bleb, so that both morphology and gene expression can be evaluated. Capture or examination of the actual point of entry of the needle (administration/penetration site) in the retina is difficult and generally not worthwhile.
Suprachoroidal and Subconjunctival Studies
In suprachoroidal and subconjunctival studies, the injection/deposition site is often located in the superior temporal region of the globe. The injection site/region should be marked with ink at collection to aid in locating it at trimming, since examination of this region is critical in these studies.
Subtenon and Retrobulbar Studies
With subtenon and retrobulbar injection routes, the test article is administered outside of the globe. In such studies, it is recommended the eye, adnexa, and adjacent intraorbital structures (such as the orbital fat pad, lacrimal glands, etc) be collected, fixed, and trimmed en bloc to maintain the relationship of the injection/depot/implant to the globe and adjacent structures. Overtrimming of the associated tissues away from the eye at collection should be avoided.
Considerations for the Sampling and Examination of Extraocular Tissues
A number of extraocular tissues may be directly or indirectly impacted in the course of ocular toxicity studies or secondary to lesions in the eye. Reciprocally, primary effects in extraocular tissues may also have direct or indirect effects on the eye itself. For studies in which expanded ocular sampling is implemented, it is therefore important to consider which extraocular tissues should be examined.
Ocular Adnexa
The ocular adnexa (Figure 8) are critical to the protection of the eye and the production and maintenance of the ocular surface system. 60,61 The primary function of the ocular surface system is to provide, protect, and maintain a smooth corneal refractive surface. Because dysfunction in any of the components can significantly impact corneal health, histologic evaluation of the ocular surface system components is critical to differentiate primary and secondary corneal toxicities. Adnexal structures which should be collected and examined from ocular toxicity studies, or from studies in which there is a corneal or conjunctival finding in-life, include the lacrimal glands, upper and lower eyelids (including Meibomian glands), and bulbar conjunctiva (included in eye sections). The Harderian glands (rabbit and minipig) and the nictitating membrane (rabbit, minipig, and dog) should also be collected when present. For the rabbit Harderian gland, it is ideal to sample both the white and pink lobes, which differ microscopically. The eyelids should be sectioned perpendicular to the eyelid margin to allow evaluation of the skin, eyelid margin, palpebral conjunctiva, eyelashes, meibomian glands, and conjunctiva-associated lymphoid tissues. 62,63 Similar to the eyelid, the nictitating membrane should also be trimmed perpendicular to the free margin. The pathologist responsible for training technicians in collection of these tissues should carefully review the appropriate anatomy texts, in order to ensure that these tissues are collected and correctly identified.
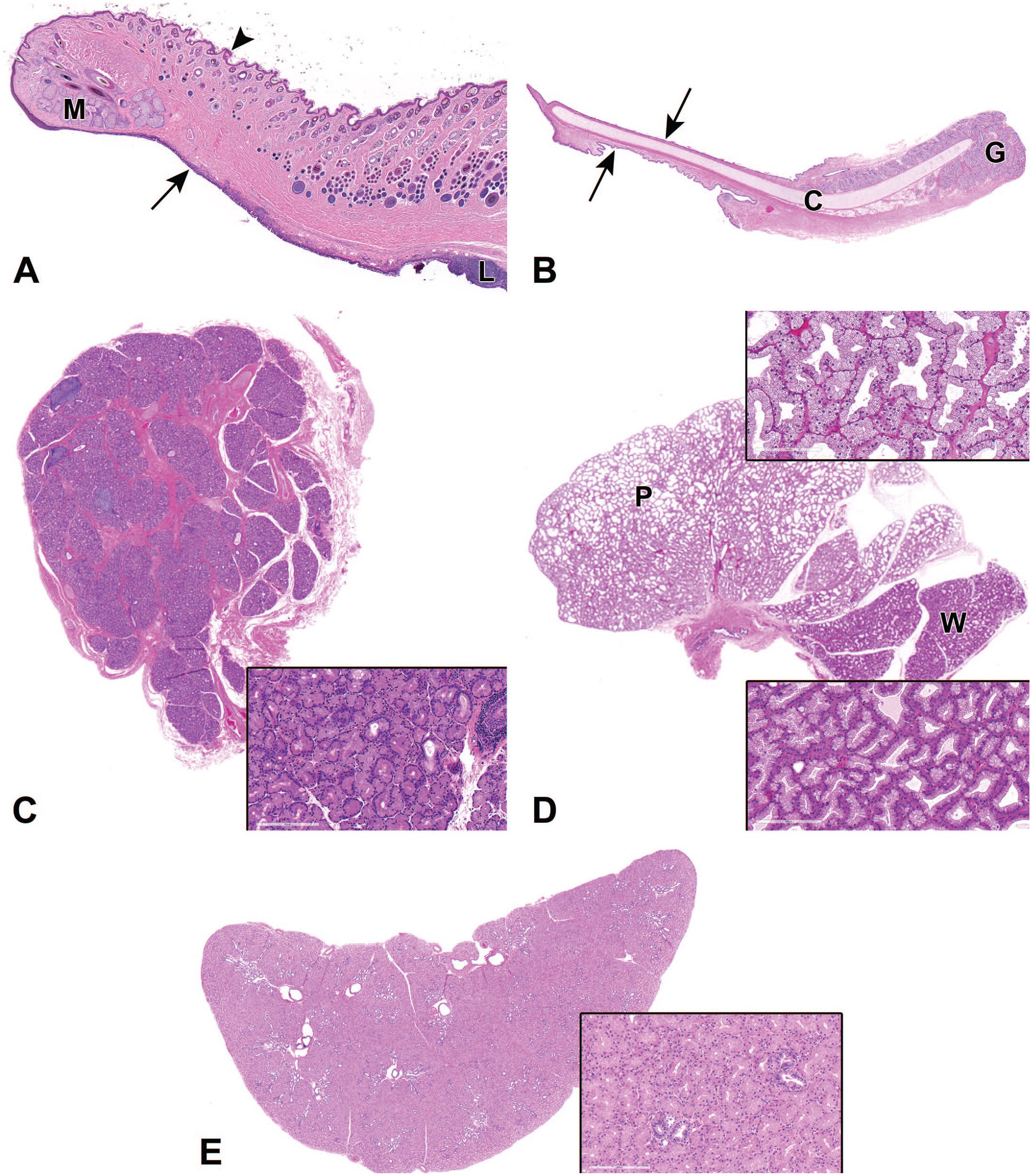
Example sections of adnexa (A) rabbit eyelid, (B) minipig nictitating membrane, (C) nonhuman primate lacrimal gland, (D) rabbit Harderian gland (pink and white lobes)*, and (E) minipig Harderian gland. *Note: color-based nomenclature of the rabbit Harderian gland refers to the gross rather than the histologic appearance.
The ocular adnexa (other than the bulbar conjunctiva, which is adherent to the globe) are generally collected separately from the eyes and fixed in 10% NBF. Adnexa from the left and right sides should be distinguished to enable correlation with any unilateral gross or clinical observations. If the interrelationship of the eye and various adnexal structures is of particular interest, the eye and adnexa may be collected en bloc at necropsy and fixed together in DS or MDS. The ocular adnexa for each species in which they are present, their function, and comments on their collection/trimming are summarized in Table 2.
Ocular Adnexa.
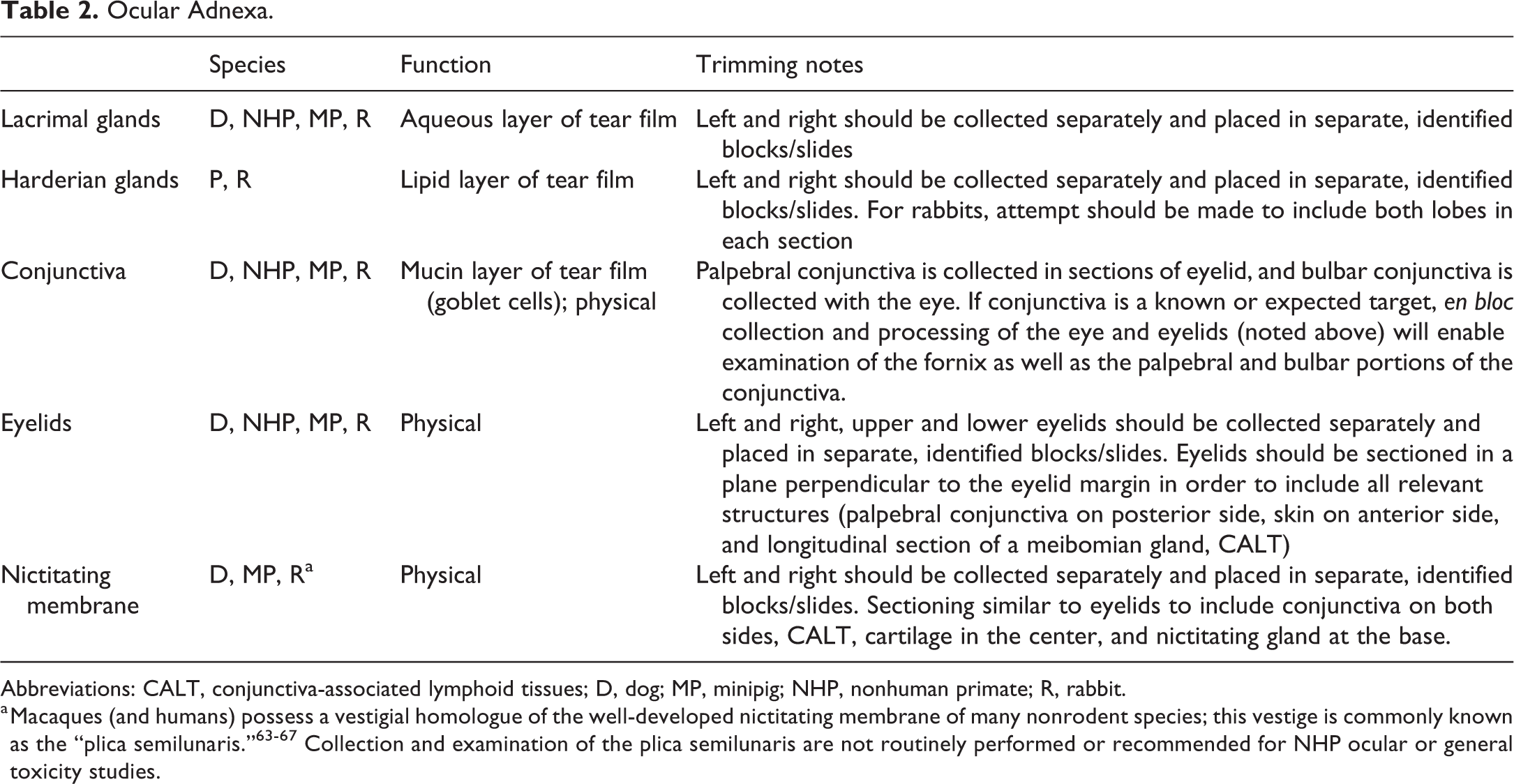
Abbreviations: CALT, conjunctiva-associated lymphoid tissues; D, dog; MP, minipig; NHP, nonhuman primate; R, rabbit.
a Macaques (and humans) possess a vestigial homologue of the well-developed nictitating membrane of many nonrodent species; this vestige is commonly known as the “plica semilunaris.” 63 -67 Collection and examination of the plica semilunaris are not routinely performed or recommended for NHP ocular or general toxicity studies.
Extraocular Muscles
Histological evaluation of extraocular muscles (EOMs) is not indicated for general or ocular toxicity studies unless there is a strong scientific rationale. For example, examination of EOM may be appropriate in studies with test articles having known muscle effects or target expression; test articles delivered by adeno-associated virus serotypes with known muscle tropism or when the route of administration (eg, retrobulbar, subtenon) is expected to result in high local concentrations of the test article in or around the EOM. When EOMs are examined, the muscles are usually blocked together for each eye (separating those associated with the left and right eyes). It is important to note that the individual EOM do vary in gross and histologic anatomy. 68,69 Therefore, identification, blocking, and evaluation of individual EOM may be warranted in certain investigational studies in which distinguishing potential test-article effects on a specific EOM is a study objective.
Optic Nerves, Optic Chiasm, and Optic Tracts
The optic nerves are central nervous system white matter tracts composed of the axons of retinal ganglion cells which, after exiting the globe, extend through the optic chiasm and optic tracts and ultimately terminate primarily in the lateral geniculate nucleus of the brain. 10 In addition to potentially being a primary target, the optic nerves/tracts may also manifest changes secondary to retinal lesions, including axonal degeneration as a consequence of retinal ganglion cell injury, and inflammation secondary to extension of intraocular inflammation.
As noted above, in most ocular and general toxicity studies, a portion of proximal (postlaminar) optic nerve is left attached to the globe at necropsy, fixed with the eye, and sectioned longitudinally as part of the standard eye section. Additional sections of the optic nerve can be collected and fixed in 10% NBF (to avoid DS/MDS artifactual vacuolation) and sectioned transversely as needed.
Examination of the optic tracts and/or lateral geniculate nuclei as well as the occipital cortex in coronal hemisections of brain is accomplished if the brain is trimmed as recommended for general toxicity studies with systemic evaluation. 10,42,70 Histopathological examination of these central nervous system structures is not specifically required for investigative ocular toxicity studies that do not evaluate systemic tissues but should be considered in situations where there is a known or suspected potential for toxicity. If the optic nerves and tracts are of particular interest, examination of the optic chiasm and both optic tracts and lateral geniculate nuclei (left and right brain coronal hemisections) should also be considered, as decussation of the left and right optic nerves at the optic chiasm occurs to varying degrees in the nonrodent laboratory species. 10 As such, lesions originating in one eye/optic nerve may extend into both the ipsilateral and contralateral optic tracts and visual pathway centers in the brain, depending on the species. 10
Nasolacrimal Duct and Nasal Turbinates
Nasal turbinates and nasolacrimal ducts should be collected and preserved for topically applied ocular formulations, if these are target tissues, or if there are relevant in-life observations or clinical signs.
For topically applied ocular formulations, drainage of the test-article via the nasolacrimal duct into the nasal cavity/turbinates and the nasopharynx can be a major route of clearance. 71 In view of the high local exposure with this route of administration, there is a strong scientific rationale for examination of nasolacrimal ducts, nasal turbinates, and nasopharynx in topical ocular toxicity studies, especially those with novel or little-studied test articles whose potential to cause histopathologic changes has not been previously investigated. At least two transverse sections of decalcified nose, encompassing left and right sides, to include each nasolacrimal duct, respiratory mucosa (nasal turbinates), and olfactory mucosa (ethmoid turbinates), should be examined. 72,73 A transverse section of nasopharynx should also be examined. Examination of these tissues may not be necessary in every study within a drug development program.
For ocular toxicity studies with an intraocular administration route, test article clearance is expected to be through the aqueous outflow routes (trabecular and/or uveoscleral) or the blood-ocular barriers (blood-aqueous and/or blood-retinal). 71 Test articles deposited in the vitreous that are particulate or relatively insoluble will be cleared by hyalocytes/macrophages that migrate to the optic nerve and exit via optic nerve vessels into the blood stream. 74 In either case, unless there is a known target or class effect of a particular test article, there is not a strong scientific rationale for the examination of nasolacrimal duct and other nasal tissues, and the examination of these tissues should be considered optional for studies with an intraocular route of administration.
Points to Consider for Recording Histologic Examination of the Eye
If expanded ocular sampling is implemented for a study, it is suggested that a general statement be made to this effect in the Materials and Methods section of the pathology report. It may be helpful to include the sectioning paradigm used and the number of sections examined per eye. Given recent communications between regulatory agencies and the authors, it may be advisable to include a specific statement that the sectioning technique was designed to capture the pertinent species-specific retinal “cone-dense” region (eg, visual streak, macula, fovea) of the eye. Consistent with this, it may also be helpful to record as a tissue comment (specifically, not a morphologic finding as this would not be Standard for Exchange of Nonclinical Data-compliant) if a normally examined component or structure, such as the lens or the macula, is not present in the sections examined.
In addition, in the context of ocular toxicity studies, the presence of the site of administration or implantation and the test article should be recorded if observed.
Conclusions
A number of factors should be considered in the fixation, trimming, and sampling of the nonrodent eye and extraocular tissues, and the processes and paradigms ultimately pursued should be driven by scientific rationale. A single section of the eye through the optic disc is usually adequate for most general toxicity studies. However, for all ocular toxicity studies, and those general toxicity studies with either test article-related ophthalmic examination findings or a target or class liability, histopathological examination of a single section of the eye is not considered sufficient.
Expanded ocular sampling requires evaluation of a minimum of three sections of each eye, at least one of which should include the cone-dense regions of the retina, in addition to the site/region of administration and the site of deposition of test-article for depot-based formulations. A specific conclusion of the NOTS-WG is that any requirement for evaluation of retinal cone-dense regions in ocular toxicity studies is adequately met by examination of the macula/fovea in NHP and of the visual streak in rabbits, pigs, and dogs. Sampling and evaluation of the focal area centralis of the dog are not considered necessary to fulfill this objective.
The NOTS-WG considerations for expanded ocular sampling take into account the fact that nonrodent ocular toxicity studies can vary widely in many respects including study objectives, regulatory status, test material class, administration route, institutional resources, and especially the many pertinent anatomic and histologic species differences between the rabbit, dog, minipig, and NHP (including differences in the retinal cone-dense subsites). Such a range of variation in ocular anatomy necessitates some flexibility in ocular toxicity study design and conduct (within the context of appropriate scientific principles). Many of the expanded ocular sampling guidelines include several options for practical techniques (ie, collection, fixation, trimming, sectioning) that may be used for a given study. These options should be carefully evaluated, selected, and even adapted on a case-by-case basis to best fulfill the scientific objectives of a given study. The accessibility of the eye allows longitudinal, in-life data acquisition. To best enable the correlation of in-life and histopathological findings, it is essential that the pathologist, ophthalmologist, and technical personnel (necropsy prosector, histology technician, etc) communicate effectively with one another.
Footnotes
Acknowledgments
The authors gratefully acknowledge the contributions of David Sabio (EPL, Inc.) who provided the illustrations for the manuscript, and Sonia Horth (CRL Montreal) for her technical contributions to the work on trimming and sectioning of the area centralis in the dog.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
