Abstract
Intracellular inclusions were observed in urinary bladder epithelium of male Wistar rats, following oral treatment with high doses of the α2A-adrenoceptor agonist tasipimidine for 28 days. No cell death or inflammation was associated with the brightly eosinophilic inclusions. Electron microscopy (EM) studies showed that the inclusions represented intact or fragmented red blood cells (RBC) resulting from erythrophagocytosis, further supported by the presence of iron in urothelial cells. In addition, scattered iron-positive macrophages were observed in the submucosa and muscle layer, indicating microvascular leakage, as no major hemorrhage was evident. Despite the presence of inclusions, the urothelium showed normal uroplakin III distribution, normal cell turnover, and an absence of α-2u-globulin. It is, therefore, concluded that the inclusions were not associated with urothelial damage or increased renewal of the epithelium. This finding shows also that urothelial cells have the capability to phagocytize and break down RBCs originating from submucosal microvascular leakage. Similar changes were not observed in tasipimidine-treated beagle dogs (28 days), suggesting these findings were rat specific. The leakage of RBCs into the urothelium is suggested to be a consequence of exaggerated pharmacology leading to vasoconstriction of submucosal blood vessels in combination with transient increased bladder distension and pressure.
Introduction
The epithelium of the urinary bladder, urothelium, consists of 3 distinct cell types, the umbrella cells facing the lumen, intermediate cells, and basal cells. A unique feature of the umbrella cells are the transmembrane proteins, uroplakins, which are assembled into plaques and play a crucial role in allowing membrane deformation during bladder filling and voiding. 1 –3 The deformation of the cell membrane is associated with a machinery of exocytosis and endocytosis, which maintain the optimal cell surface area of umbrella cells, a process that involves a balanced trafficking of cell membrane components between cytoplasmic storage vesicles, cell surface, and lysosomes. 4 Although lysosomal degradation of membrane components is a normal process in umbrella cells, there are only a few reports describing phagocytosis and lysosomal breakdown of extracellular material in urothelial cells. Wakefield and Hicks 5 found that in rats, urothelial cells can engulf RBCs following intravesical administration of a number of urothelial cytotoxins. The erythrophagocytosis appeared to be a secondary/regenerative response to the initial lesions, urothelial necrosis, and rupture of suburothelial capillaries. Also, Romih et al 6 noted phagocytosis of neighboring apoptotic urothelial cells during the regeneration of cyclophosphamide-induced urothelial cell loss. Thus, it appears that phagocytosis by urothelial cells is mainly a response to remove effete cells and hemorrhage from the local environment.
In general, the result of phagocytosis can be seen as intracellular accumulation of material in vacuoles or lysosomes or as unspecific inclusions. In the present study, we describe urothelial cell inclusions observed in Wistar rats treated orally (PO) with a new α2A-adrenoceptor agonist, tasipimidine, in a standard regulatory 28-day toxicity study. Light microscopy and EM studies revealed that the inclusions contained phagocytized RBCs. However, opposite to studies mentioned above, no apparent damage to the urothelium or to the urinary system in general was observed. No cell inclusions were observed in dogs treated with tasipimidine.
Pharmacologically, tasipimidine is a selective and potent agonist on the human α2-adrenoceptor with specificity for the α2A-subtype, developed for the treatment of situational anxiety and fear in dogs. 7 In the rat ortholog α2D-adrenoceptor, the potency of tasipimidine is slightly weaker, showing >8 times less potency than on the corresponding human α2A-adrenoceptor. 7 Typically, α2-adrenoceptor activation in brain and vasculature induces changes in blood pressure and heart rate. These effects reflect the balance between central (reduced heart rate and blood pressure) and peripheral (increased systemic vascular resistance and blood pressure) effects. Activation of pre- and postsynaptic α2-adrenoceptors in the brain leads to inhibition of sympathetic activity, causing decreases in blood pressure and heart rate together with decreased arousal, sedation, and relief of anxiety. 8 –11 Peripheral postjunctional α2-adrenoceptors in blood vessels mediate vascular smooth muscle contraction, causing an increase in blood pressure. 10,12,13 The overall hemodynamic effect depends on the dose, mode of administration, the animal species, and the sympathetic tone of the animal.
In the bladder, the contraction and relaxation of the detrusor smooth muscle is under the control of cholinergic, purinergic, and adrenergic nerves. 14 α-Adrenoceptor–mediated effects are most significant in the urethra where α1-adrenoceptor stimulates sphincter muscle contraction. 14 –16 α2-Adrenoceptors are present in smooth muscles of the bladder and urethra, but functional studies have not revealed any conclusive role of the receptor in these locations. 15 However, α2-adrenoceptors are involved in the volume-evoked micturition reflex, as signals between the micturition center in the brainstem and the sacral parasympathetic nerves innervating the bladder are mediated by these adrenoceptors. 17
Mechanisms leading to the urothelial inclusions in rats are discussed in context of pharmacological actions of tasipimidine on blood vessels and bladder function, and with respect to species differences.
Material and Methods
Compounds
Tasipimidine was synthesized at Orion Corporation, Orion Pharma, Espoo, Finland, following Principles of Good Manufacturing Practices.
In Vivo Pharmacological Methods in Rats and Dogs
Pharmacological properties of tasipimidine were studied in male Wistar rats and in male and female beagle dogs. Anaesthetized and pithed rat models were used to assess the effects of tasipimidine on centrally and peripherally mediated blood pressure, respectively. In isoflurane-anaesthetized rats (n = 5), tasipimidine was injected 0.5 mL/kg, at cumulative doses from 0.1 to10 µg/kg with 20-minute intervals. In the pithed rat method, 18 cumulative doses of 0.3 to 30 µg/kg were used with 15-minute intervals (n = 5). The left femoral vein was cannulated for intravenous (IV) administration of tasipimidine and the left femoral artery was cannulated to measure blood pressure signals which were amplified with a 2-channel-bridge-amplifier (Hugo Sachs Elektronik), and data collected with Notocord-hem system (NOTOCORD Systems SAS). The pharmacology in beagle dogs was assessed in telemetered conscious dogs. Tasipimidine was administered by single oral gavage at dose levels of 0.03, 0.1, and 0.3 mg/kg according to crossover design. Telemetric measurements of blood pressure, heart rate, body temperature, and electrocardiogram in freely moving animals (n = 6) started at least 2 hours before each administration and continued for at least 24 hours after dosing using telemetry acquisition system (Data Sciences International). In another dog study, tasipimidine at single oral doses of 0.01, 0.03, and 0.1 mg/kg (n = 4) was administered for restrained animals and the grade of sedation was scored by an experienced observer up to 6 hours after dosing. Blood pressure and heart rate were monitored from 20 minutes before dosing up to 6 hours after dosing using a telemetry acquisition system (Data Sciences International).
28-Day Toxicity Study Designs in Rats and Dogs
A 28-day toxicity study was performed in Wistar Han rats (Charles River Deutschland) and beagle dogs (Marshall BioResources) by daily oral administration of tasipimidine diluted in Elix water (vehicle). In the rat study, 80 animals were allocated to 4 dose groups, each containing 10 males and 10 females. At start of dosing the rats were approximately 6 weeks old, and the weight ranges were 132 to 164 g (males) or 111 to 133 g (females). Tasipimidine was administered once daily by gavage at doses of 0 (vehicle), 1.5, 5, and 15 mg/kg (dose groups 1, 2, 3, and 4, respectively) for 28 days. In the dog study, 24 beagle dogs were allocated to 4 dose groups, each containing 3 males and 3 females. The dogs were approximately 7 to 8 months old at study start. Tasipimidine was administered once daily by gavage at doses of 0 (vehicle), 0.03, 0.15, and 0.51 mg/kg (dose groups 1, 2, 3, and 4 respectively) for 28 days. For urine analyses, overnight urine was collected from rats and dogs at the end of the 28-day treatment period. The urine samples were studied both biochemically with a Clinitek 500 Urine Chemistry Analyzer (Siemens Healthcare Diagnostics B.V.) and microscopically.
Plasma and Urine Concentrations of Tasipimidine
To determine the oral bioavailability of tasipimidine in rats and dogs, animals were treated with single doses, IV or PO. In the rat study, the dose levels were 0.037 (IV) and 3 mg/kg (PO) and, in the dog study 0.01 (IV) and 0.03 mg/kg (PO). Plasma samples were collected at 8 to 10 time points until 24 hours (rats, n = 4) or 10 hours postdosing (dogs, n = 8).
In the rat and dog 28-day toxicity studies, plasma concentrations were determined on the first day of dosing and in week 4. In both studies, a full toxicokinetic profile was calculated based on plasma samples collected at 6 time points until 24 hours after dosing. In all studies, plasma tasipimidine concentrations were determined by a liquid chromatography tandem mass spectrometric method and the plasma kinetic parameters were calculated using the WinNonlin Professional version 5.2 (rat bioavailability study and the 28-day toxicity studies) or Phoenix WinNonlin 6.3 program (dog bioavailability study).
To evaluate the urinary excretion of tasipimidine and the main metabolite (ORM-18662), male Wistar rats (n = 4) and male beagle dogs (n = 4) were treated with a single dose of tasipimidine. The dose levels were 15 (rat) or 0.51 mg/kg (dog). Animals were kept in individual metabolic cages, and urine fractions were collected on ice up to 24 hours after dosing. A liquid chromatography-electrospray ionization tandem mass spectrometric method was used for the determination of tasipimidine and ORM-18662 in each fraction.
Guidelines
In vivo pharmacological and urinary excretion studies were performed at Orion Corporation, Orion Pharma, and at Centre de Recherches Biologiques (CERB), Baugy, France. The studies were approved by the Finnish animal experiment board (license numbers ESLH-2009-08809, ESLH-2010-02974, ESAVI-2012-5535, and ESAVI-6605) and by the internal ethics committee of the CERB. The 28-day toxicity studies were performed at Charles River Laboratories Den Bosch B.V. (previously NOTOX B.V.), and the test protocols were reviewed and agreed by the animal welfare officer and the ethical committee of NOTOX (DEC 07-73, DEC 06-121, and DEC 97-03-15) as required by the Dutch Act on Animal Experimentation (February 1997).
All study procedures were based on the following guidelines: ICH M3 (R2): Note for guidance on nonclinical safety studies for the conduct of human clinical trials and marketing authorization for pharmaceuticals, December 2009. CHMP Guideline on the evaluation of control samples in nonclinical safety studies: Checking for contamination with the test substance, March 2005. ICH S3A: Note for Guidance on Toxicokinetics: The assessment of systemic exposure in toxicity studies, December 2002. ICH 7A guidelines: Safety Pharmacology Studies for Human Pharmaceuticals, June 2001.
The studies were performed according to The Organisation for Economic Co-operation and Development Principles of Good Laboratory Practice (as revised in 1997) ENV/MC/CHEM (98) 17 or in accordance with the Directive 86/609/EEC European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes.
Histology
Formalin-fixed, paraffin-embedded sections of a standard full list of tissues (according to the guidelines) were stained using hematoxylin and eosin (H&E), and in addition, a number of special stains and immunohistochemical (IHC) stains were applied to urinary bladder of rats as depicted in Table 1. Urinary bladder sections were cut and stained with periodic acid–Schiff (PAS) or Perls Prussian blue (PPB) or IHC stained using antibodies against Ki67, uroplakin III, or α-2u-globulin according to Table 1. For IHC, biotinylated secondary antibodies, combined with streptavidin-horseradish peroxidase, were used, and staining was visualized using 3,3′-diaminobenzidine. Positive controls were small intestine (Ki67), kidneys with hyaline droplets (α-2u-globulin), and urinary bladder (uroplakin III). Also, isotype controls and negative controls were included. Detailed antibody information is presented in Table 2.
Special Staining Methods for Light Microscopy Employed in the Rat 28-Day Toxicity Study.a
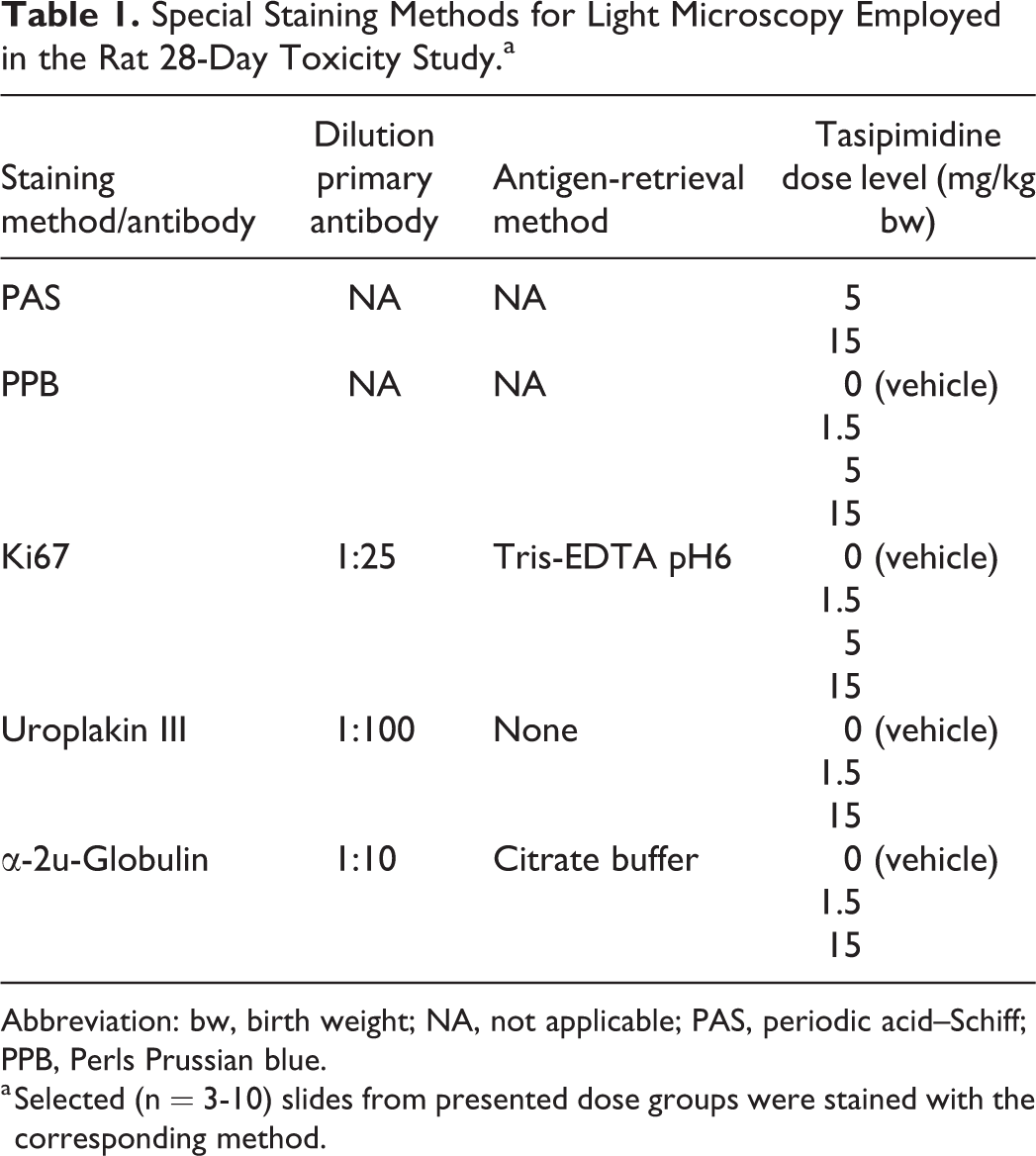
Abbreviation: bw, birth weight; NA, not applicable; PAS, periodic acid–Schiff; PPB, Perls Prussian blue.
a Selected (n = 3-10) slides from presented dose groups were stained with the corresponding method.
Antibodies Used in Immunohistochemical Staining of Rat Urinary Bladders.
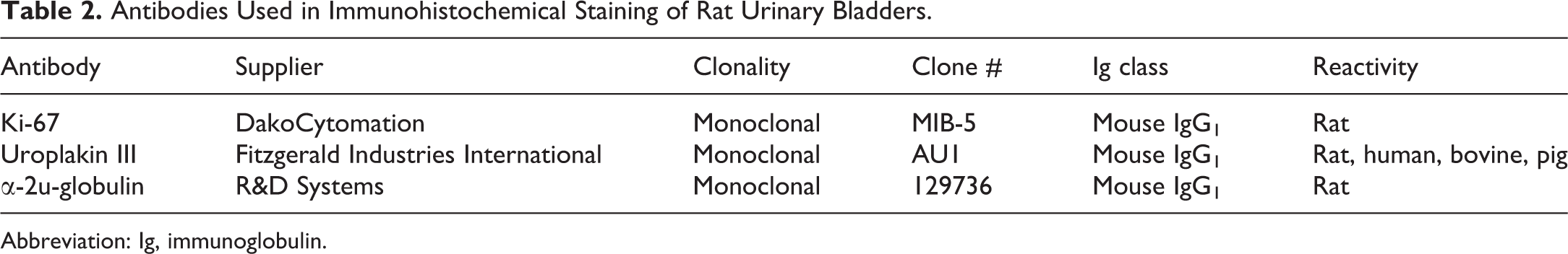
Abbreviation: Ig, immunoglobulin.
Electron Microscopy
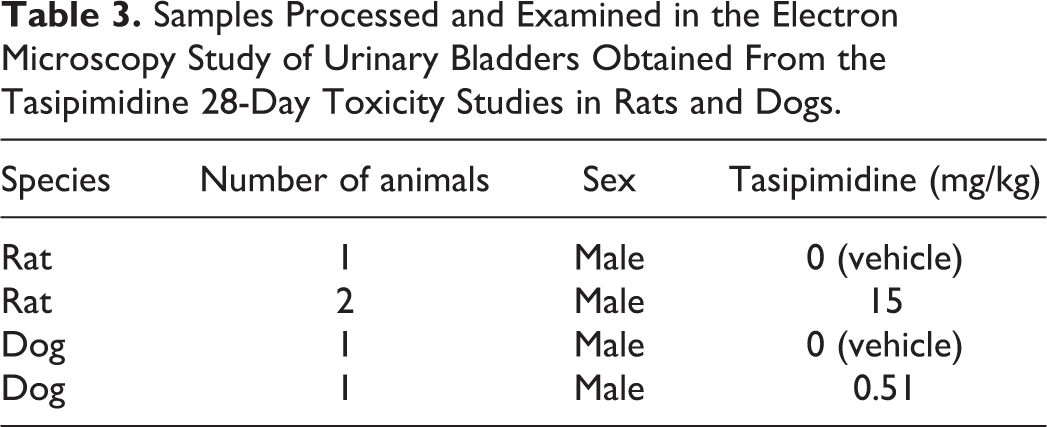
Electron microscopy was performed on the urinary bladder epithelium (urothelium) of 3 rats and 2 dogs treated with vehicle or high-dose tasipimidine (see Table 3 for detailed information). Specimens were retrieved from formalin, processed, and embedded at the University of Nijmegen, The Netherlands. In short, formalin-fixed tissues were cut and fixed in 2% glutaraldehyde solution. The samples were rinsed, post fixed in osmium tetroxide, rinsed, and dehydrated in increasing ethanol steps (up to 100%) and propylene oxide before embedding in Epon resin 812. After polymerization at 30-45-60 ºC, sections of about 1 µm were cut and stained with methylene blue for evaluation by light microscopy. Ultra-thin sections (60 nm) were cut from the blocks selected, stained with lead citrate and uranyl acetate, and examined using a transmission electron microscope and camera.
Samples Processed and Examined in the Electron Microscopy Study of Urinary Bladders Obtained From the Tasipimidine 28-Day Toxicity Studies in Rats and Dogs.
Results
In Vivo Pharmacology in Rats and Dogs
Tasipimidine dose dependently induced typical α2-adrenoceptor agonist symptoms such as sedation-related effects, vascular effects, decrease in heart rate, and decrease in rectal temperature in rats and dogs (Table 4). In anesthetized rat, centrally mediated α2-adrenoceptor agonist induced hypotension, whereas in pithed rat model, binding to peripheral postsynaptic α2-adrenoceptor in vasculature induced hypertension due to vasoconstriction of vascular smooth muscle. A similar cardiovascular profile of tasipimidine was also observed in dogs. In conscious freely moving dogs, tasipimidine induced a dose-dependent increase in arterial blood pressure, whereas in restrained dogs, centrally mediated hypotension was observed. In both dog studies, tasipimidine caused decrease in heart rate.
Tasipimidine Pharmacological Dose Levels In Vivo (Single-Dose Studies).
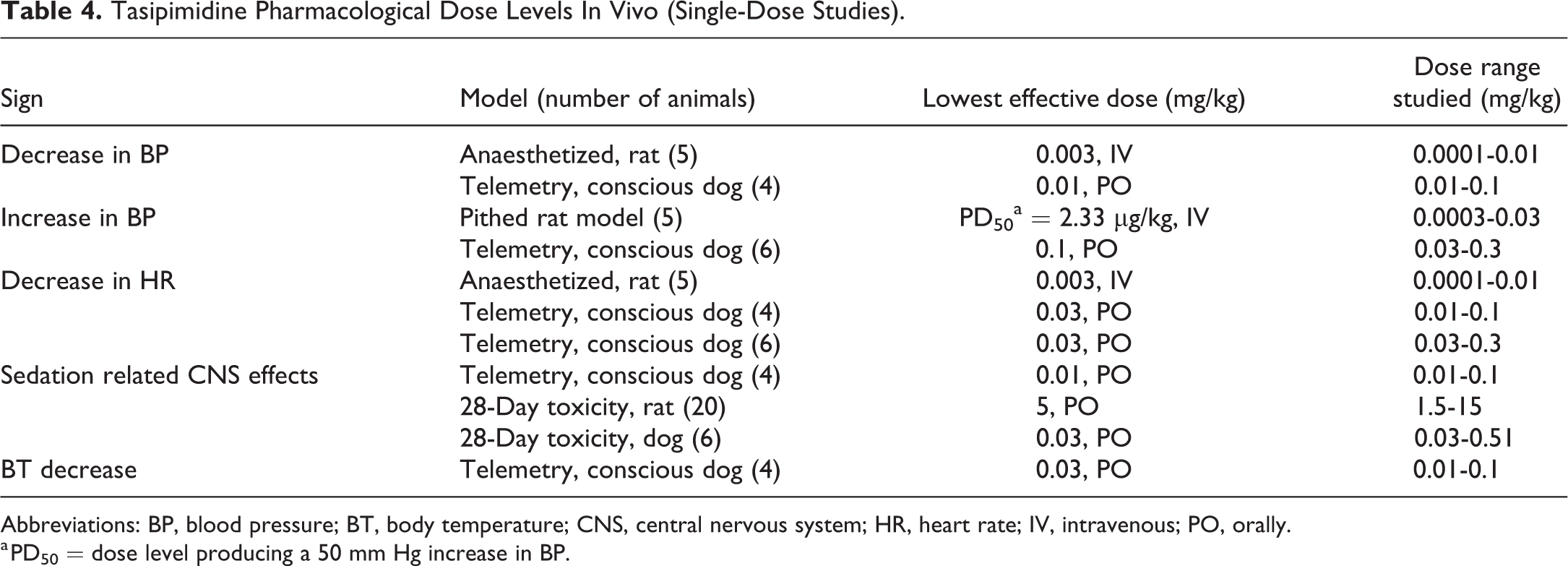
Abbreviations: BP, blood pressure; BT, body temperature; CNS, central nervous system; HR, heart rate; IV, intravenous; PO, orally.
a PD50 = dose level producing a 50 mm Hg increase in BP.
28-Day Toxicity Studies in Rats and Dogs
No mortality was observed in the studies. Tasipimidine was well-tolerated at the used dose levels, with a dose-dependent sedation-related lethargy being the most prominent clinical sign in rats at 5 and 15 mg/kg and in dogs at all tested dose levels. Microscopic and biochemical urine analyses in either species did not reveal any abnormalities, including any sign of hematuria, at the end of the study.
Urinary Tract Histopathology: H&E
Wistar Han rats showed tasipimidine-induced microscopic findings in the urinary bladder (Table 5), but not in the kidneys, ureters, or in the prostatic part of the urethra (data not shown). Urothelial hypertrophy was present in male and female rats treated at 15 mg/kg/d. Furthermore, urothelial eosinophilic inclusions were noted in male rats, at 5 and 15 mg/kg/d (Figure 1D). Microscopically, these inclusions were bright eosinophilic, often globular and sometimes appeared to be aggregated into larger clusters. These larger aggregates were present at the apical layer of the urothelium, while the smaller globular inclusions were scattered throughout the intermediate and basal cell layer of the urothelium. Tasipimidine did not induce any changes in the dog urinary bladder (Figure 2) or urinary tract.
Incidence Table of Histopathology Findings in the Urinary Bladder of Rats Treated With Tasipimidine, Once Daily for 28 Days.
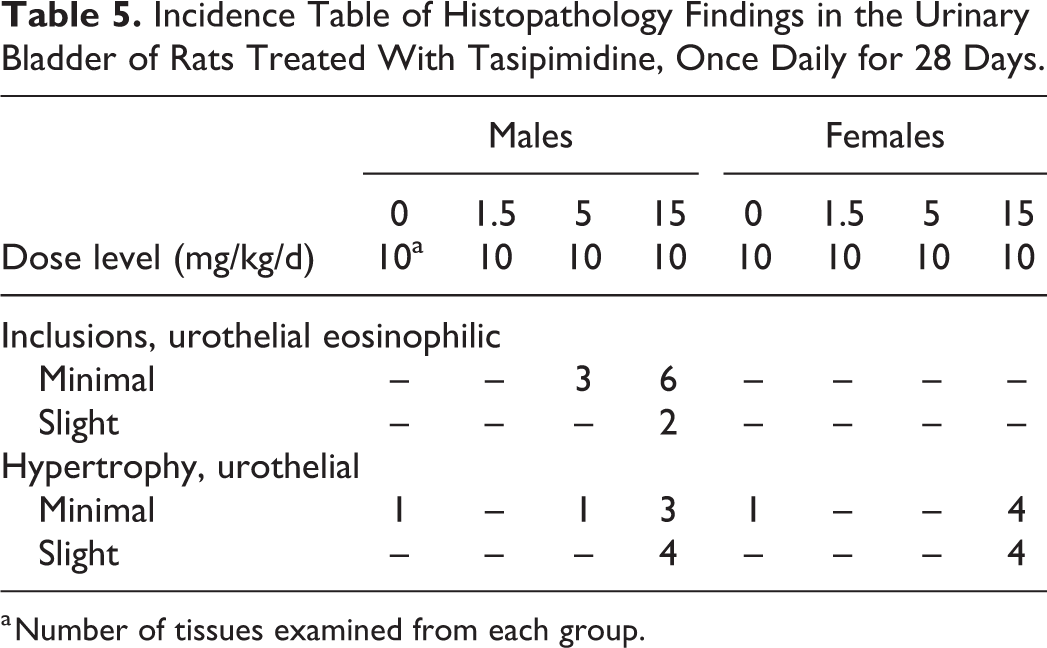
a Number of tissues examined from each group.
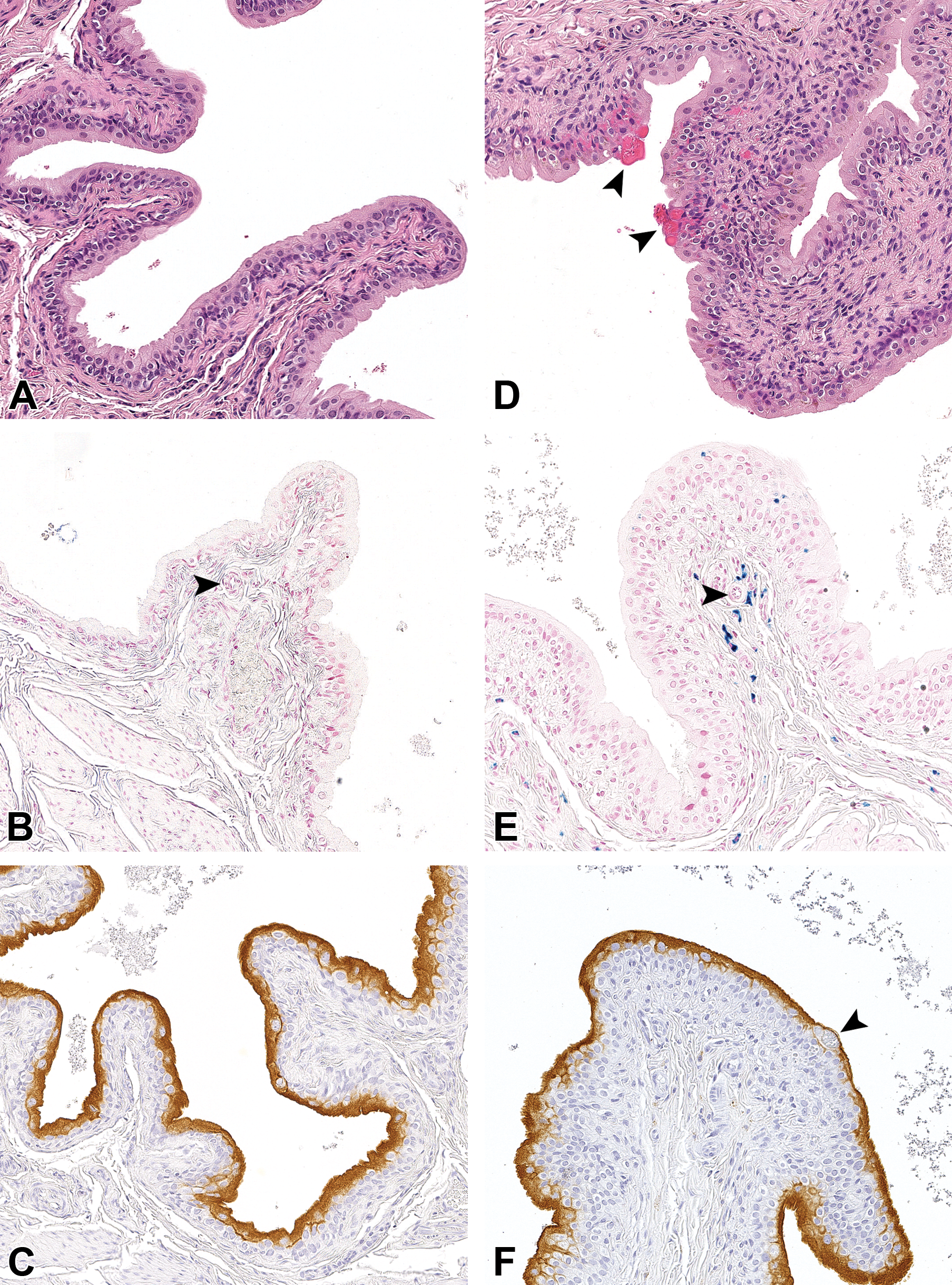
Urinary bladder of a control group (A-C) and of a 15 mg/kg/d tasipimidine treated (D-F) male Wistar Han rat. In (A), the normal rat urothelium is shown using hematoxylin and eosin, while in (D), the bright eosinophilic inclusions are abundantly present, the aggregated eosinophilic inclusions are indicated using the big arrowhead, with the smaller inclusions scattered throughout the urothelium and the underlying submucosa; in (B), a control group Perls Prussian blue (PPB; to detect “nonheme” iron) stained urinary bladder is shown, while in (E), the PPB stain shows the presence of bright blue-staining iron pigments in the urothelium and in the submucosa and surrounding a small blood vessel (small arrowhead in E, also used to show a small blood vessel in B) of treated rats; and, finally, in (C), the control immunohistochemical stain for uroplakin III is shown. Here the normal presence of this transmembrane protein can be seen present at the luminal surface of superficial umbrella cells, this pattern is the same in (F), with negative staining of the larger aggregates of inclusions (big arrowhead).
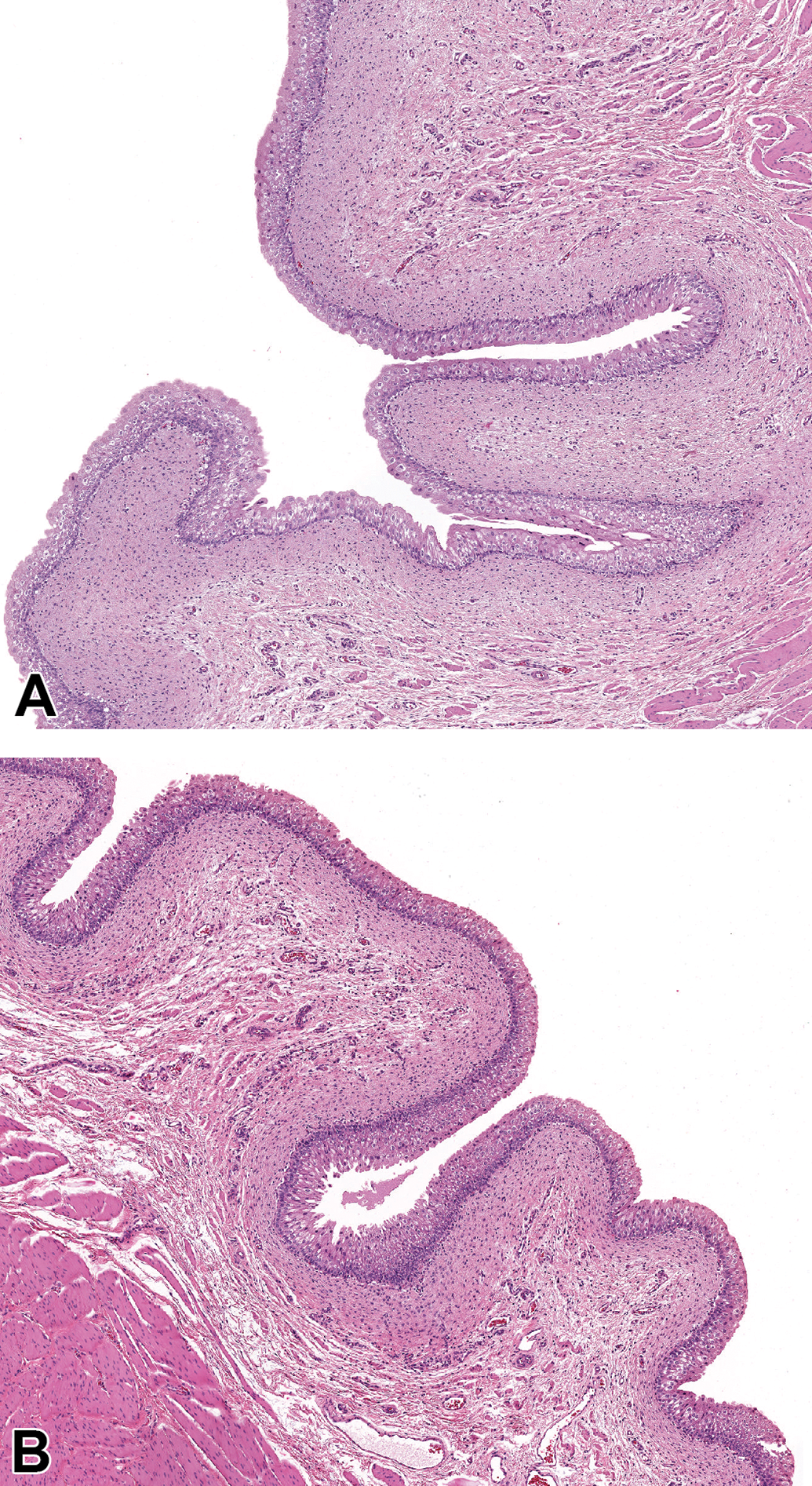
Urinary bladder of a control group (A) and of a 0.51 mg/kg/d tasipimidine treated (B) male beagle dog. There is no difference in hematoxylin and eosin staining between control and high-dose dogs, and there are no eosinophilic urothelial inclusions present.
Urinary Bladder Histopathology: Special Stains
To further examine the urothelial inclusions, the following special and IHC stains were applied: PAS, PPB, uroplakin III, α-2u-globulin, and Ki67. The PAS reaction was negative for the eosinophilic inclusions, indicating an absence of carbohydrates and mucous substances. The PPB staining showed the presence of “nonheme” iron in some urothelial cells of male and female rats treated with 5 and 15 mg/kg of tasipimidine (Figure 1E). Since PPB does not stain heme-bound iron present in hemoglobin, it did not stain large aggregates with intact erythrocytes. At the same dose levels, staining was also present in perivascular macrophages in the submucosa and the muscle layer. This suggests an extravasation of erythrocytes, which are subsequently phagocytized and broken down by macrophages. No positive staining was seen in the vehicle and in the 1.5 mg/kg treated rats.
Immunohistochemical staining with the proliferation marker Ki67 did not reveal any discernible differences between tasipimidine-treated and vehicle-treated male rats. There was a similar steady-state cell renewal visible in all animals. This suggests that tasipimidine did not increase urothelial cell turnover/renewal. The transmembrane protein uroplakin III was present in all tasipimidine- and vehicle-treated animals analyzed with a similar apical location of positive staining (umbrella cells), indicating that the urothelium was intact in tasipimidine-treated rats. The inclusions were negative for the uroplakin III antibody (Figure 1F). α-2u-Globulin immunostaining did not reveal any positive cells in any of the urinary bladder sections examined. This protein was not present in the urothelial cells, and the urothelial inclusions were negative.
Electron Microscopy: Urothelium
As the urinary bladder samples for EM were derived from formalin-fixed samples, some artifacts were visible. In general, urothelium was better preserved for EM processing in rat samples than in dog samples, as can be seen in the vehicle-treated animals (Figure 3A and E). In the urothelium of male rats administered with 15 mg/kg/day tasipimidine, there were infiltrates of erythrocytes (Figure 3B and D). These erythrocytes were characterized by the absence of a nucleus and organelles and had a uniform granular density and appeared to be very easily deformable (elastic). In Figure 3C, a normal erythrocyte (RBC) with its typical biconcave disc shape is shown in a control rat. In tasipimidine-treated rats, the typical biconcave shape is still partly present in some intracellular erythrocytes (see Figure 3D next to asterisk). The erythrocytes were mainly intracellularly, but some urothelial cells also showed remnants of phagocytized and internalized erythrocytes with lysosomes. There was a general impression that the erythrocytes traveled upward (toward the luminal side) and originated from the submucosa. In some samples, erythrocytes were actively phagocytized (Figure 3B and D). The umbrella cell layer appeared to be intact despite the presence of large erythrocyte accumulations. The phagocytized erythrocytes likely corresponded to the urothelial eosinophilic inclusions, which were noted by light microscopic histopathologic evaluation. There were no abnormalities noted in the urothelium of either the control dog sample or the 0.51 mg/kg/d treated dog sample (Figure 3E and F).
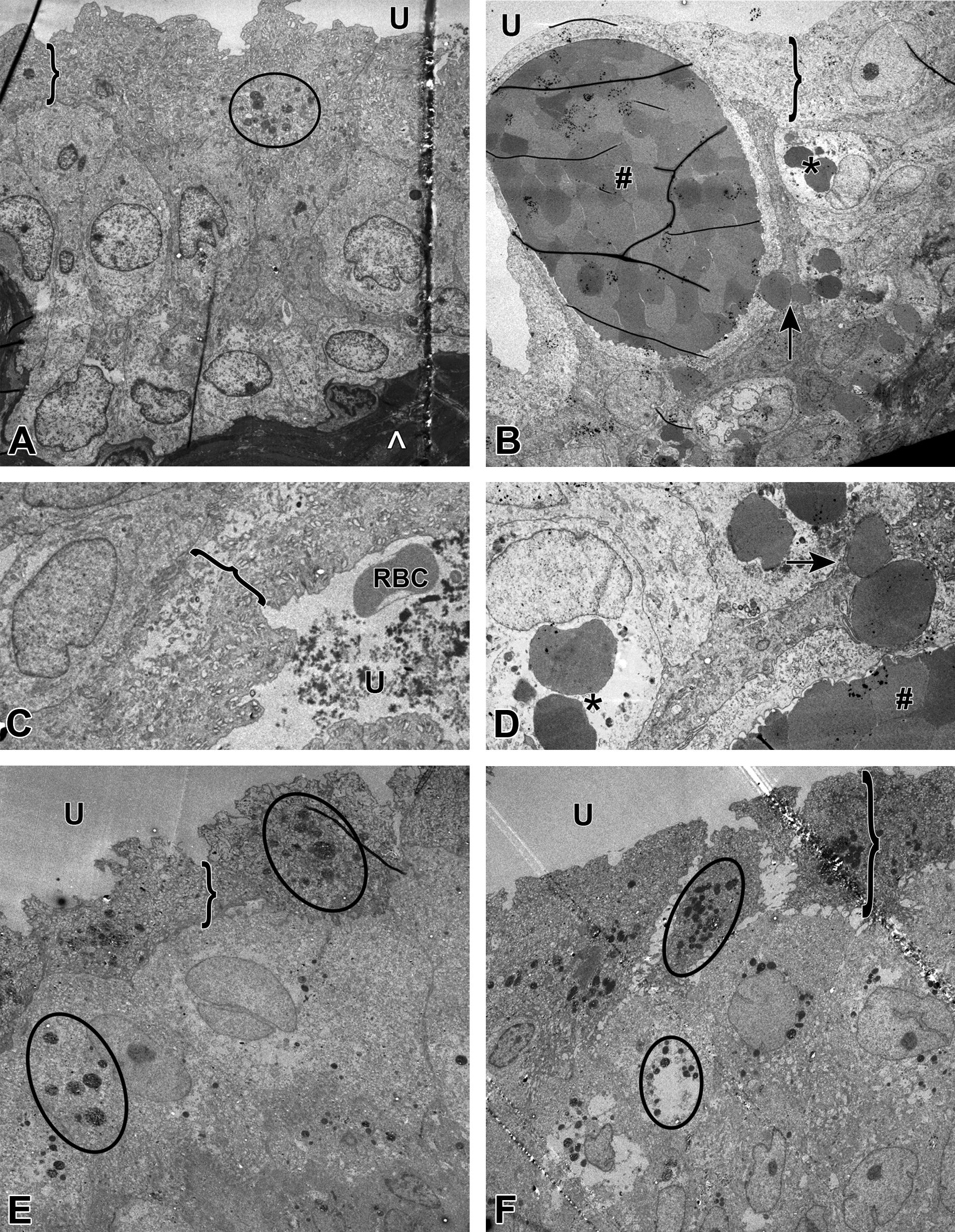
Electron microscopic (EM) images of male Wistar Han rats (A-D) and beagle dogs (E, F). In (A, C), the EM pictures show the normal control rat urothelium, with a normal erythrocyte (red blood cell) in (C) (likely present in the urinary space as a necropsy-related artifact). In (B, D, higher magnification of B), the EM of a high-dose rat shows intracellular erythrocytes in intermediate cells (*), large erythrocyte aggregates (#), and erythrophagocytosis (↑). E, F, EM of a control and high-dose beagle dog, respectively, with no differences between the 2. U indicates urinary space; }: umbrella cells (with the basal and intermediate layers underneath); encircled: lysosomes; ∧: lamina propria (collagen).
Tasipimidine Plasma Exposure
Oral bioavailability was significantly higher in dogs (60%) than in rats (2.2%). In the 28-day toxicity studies, tasipimidine showed a dose-dependent increase in plasma exposure in both species (Table 6). No significant differences in plasma exposures were observed between rats and dogs. The high-dose level in the dog study (0.51 mg/kg/d) achieved comparable plasma concentrations (C max) or slightly higher total exposure (AUC0-24 h) as the mid-dose level (5 mg/kg/d) in the rat study. Male and female rats, treated with the high-dose level (15 mg/kg/d), showed accumulation of tasipimidine in the plasma upon repeated dosing. In dogs, no consistent accumulation was observed.
Tasipimidine-Unbound Plasma Concentrations in Rat and Dog 28-Day Toxicity Studies as C max (nM) and Total 24 Hours Exposure AUC0-24 h (h × nM).a
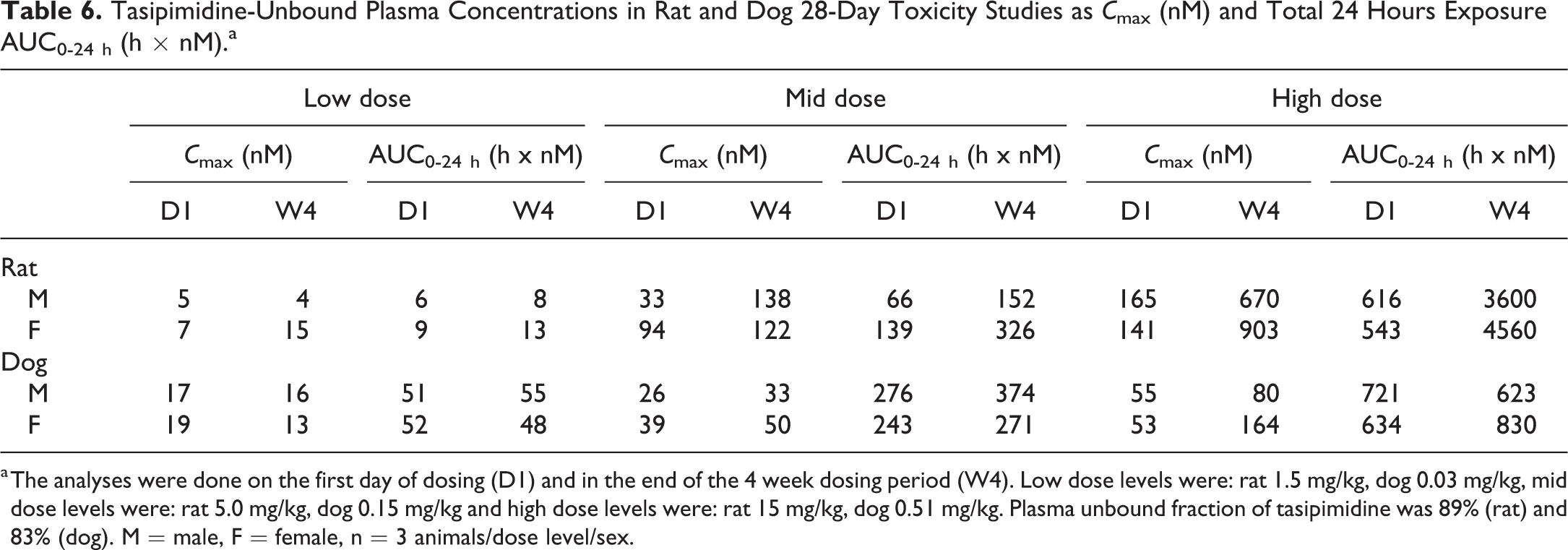
a The analyses were done on the first day of dosing (D1) and in the end of the 4 week dosing period (W4). Low dose levels were: rat 1.5 mg/kg, dog 0.03 mg/kg, mid dose levels were: rat 5.0 mg/kg, dog 0.15 mg/kg and high dose levels were: rat 15 mg/kg, dog 0.51 mg/kg. Plasma unbound fraction of tasipimidine was 89% (rat) and 83% (dog). M = male, F = female, n = 3 animals/dose level/sex.
Urinary Excretion
Following oral administration of tasipimidine, both parent compound and the main metabolite ORM-18662 were found in the urine of rats and dogs (Table 7). Related to the amount administered, tasipimidine and ORM-18662 showed slightly higher urinary excretion in dogs; however, rats showed occasional urine fractions with significantly higher concentrations of both tasipimidine (10×) and ORM-18662 (37×) compared to the highest concentrations measured in dog samples.
Urinary Excretion of Tasipimidine (A) and the Metabolite ORM-18662 (B) in Male Rats and Dogs Treated With a Single Dose of 15 or 0.51 mg/kg, Respectively.a
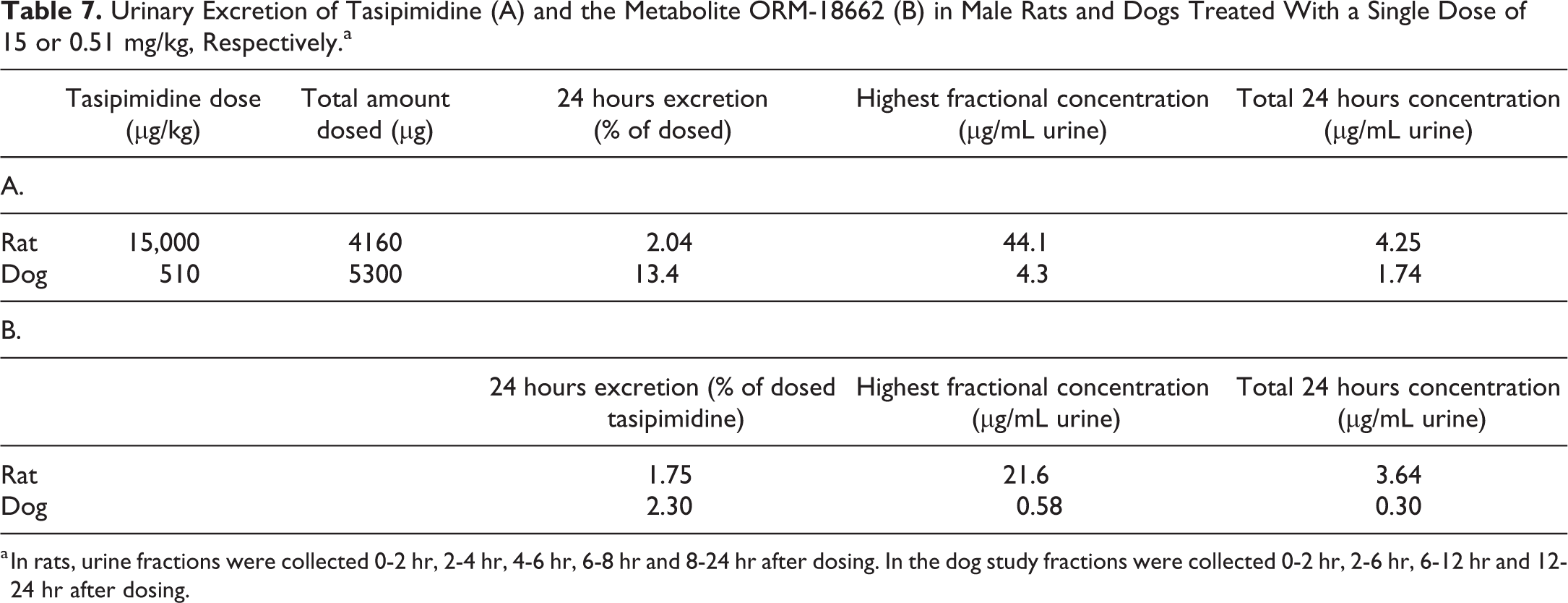
a In rats, urine fractions were collected 0-2 hr, 2-4 hr, 4-6 hr, 6-8 hr and 8-24 hr after dosing. In the dog study fractions were collected 0-2 hr, 2-6 hr, 6-12 hr and 12-24 hr after dosing.
Discussion
In this work, we have described and characterized intracellular inclusions in the urothelial cells of rats treated with the α2A-adrenoceptor agonist, tasipimidine. The inclusions were most prominent in the umbrella cells, but small inclusions were also present in intermediate and basal cells. No apparent inclusions were noted elsewhere in the urinary tract. The normal urothelial apical surface is covered with a mucous layer, 19 and umbrella cells are known to store uroplakin in specific cytoplasmic vesicles. 4 However, the inclusions were negative for the PAS reaction and uroplakin III immunostaining, indicating that the inclusions did not represent any disturbances in these normal cellular functions. Likewise, α-2u-globulin, typically seen in kidneys of male rats, 20 was absent in the urothelium, including the inclusions. Electron microscopy showed that the inclusions consisted of phagocytized RBCs, resembling that reported by Wakefield and Hicks. 5 In their study, the findings were clearly associated with necrosis of urothelial cells and ruptures of suburothelial capillaries. However, in the present study, we were unable to recognize any obvious tissue damage on light and EM level. The umbrella cell layer was intact with a normal uroplakin III staining and urothelial cell proliferation was unchanged, indicating an absence of increased cell renewal and regeneration. As in the work by Wakefield and Hicks, 5 the origin of RBC was most likely submucosal as light microscopy showed the presence of perivascular hemosiderin-like pigments that were stained positive for iron (PPB stain). Also, the absence of hemorrhage or other pathological findings in the upper urinary tract and the absence of blood in the urine support this interpretation.
The test compound described in the present work (tasipimidine) showed high potency and efficacy on human and rat adrenergic receptors, being most potent on the α2A/D-adrenoceptor. 7 No dog receptor data are available; however, canine α2A-adrenoceptor has been shown to be similar to the corresponding human receptor 21 –23 and tasipimidine showed good in vivo efficacy in dogs, with symptoms consistent with α2A-adrenoceptor agonist reported previously. 8 –13 Based on well-known vascular effects of centrally acting α2-adrenoceptor agonists, 11 an expected hypotensive effect was observed in both rats and dogs treated with tasipimidine. In addition, the pithed rat model and telemetry study in freely moving dogs revealed that tasipimidine is also able to induce hypertension at higher doses due to the activation of α2-adrenoceptors in peripheral blood vessels. This is in good agreement with the general action of α-adrenoceptors on the arterial smooth muscles as mediators of vasoconstriction. 13 Also, in the urinary bladder, α-adrenoceptors are heavily involved in the regulation of arterial blood flow. A distinctive feature of the bladder vasculature is the network of arterioles, capillaries, and venules located in the lamina propria immediately beneath the urothelium. 24,25
The underlying mechanisms leading to inclusions and submucosal erythrocyte extravasation in tasipimidine-treated rats are not fully understood, but exaggerated pharmacology, partly on the micturition reflex, partly on the bladder blood vessels, is a likely explanation. Urodynamics was not a part of the present toxicity studies, but dexmedetomidine, another selective α2-adrenoceptor agonist, inhibits the micturition reflex in rats, resulting in bladder over-filling and dribbling. 26 A healthy bladder adapts to abnormal stretching by different morphological changes, especially thickening of the detrusor muscle. 27 Muscle hypertrophy was not observed in the present study, but urothelial hypertrophy was noted in tasipimidine-treated male and female rats. In the absence of an overall urothelial regeneration (indicated by a normal cell proliferation rate), the hypertrophy could be interpreted as a marker for abnormal filling and voiding rhythm in tasipimidine-treated rats.
Under normal conditions, the bladder blood flow increases during the filling phase, but shows a transient drop prior to and during voiding. 28,29 In rat models with partial obstruction, Mizuno et al 30 showed a reduced blood flow, whereas others 31,32 found an increased blood flow and minor extravasation of RBC in the submucosa. Simultaneously with putative bladder overfilling and pressure-related changes in the blood flow, tasipimidine would likely induce an α2-adrenoceptor–mediated vasoconstriction of the bladder arteries and the suburothelial arterioles. A sum effect could be a transient congestion and microleakage of RBCs, similar to that reported in partially obstructed rat bladders. 31,32 Especially congestion of the suburothelial microvessels could explain the presence of RBC remnants in the urothelial cells. However, this would also indicate transport of RBC across the basal lamina. Again, a phenomenon that could be forced by a putative short-lasting, abnormal stretching, and pressure of the over-filled bladder.
In the 28-day toxicity studies with tasipimidine, the bladder findings appeared to be rat specific, as the urothelial inclusions, RBC extravasation, and urothelial hypertrophy were absent in dogs. The dose levels used were about 30 times higher in the rat study than in the dog study, on a mg/kg basis. However, this cannot explain the species differences as tasipimidine showed about 30× higher oral bioavailability in dogs compared to rats and plasma exposures in high-dose dogs were comparable with plasma exposures in rats associated with urothelial inclusions and hypertrophy. The distribution of α-adrenoceptor subtypes in the smooth muscles and blood vessels in the urinary bladder is also similar in both species. 33 –35 Tasipimidine and the main metabolite were present in the urine of both species, indicating no significant species differences in the urinary excretion.
Despite similar plasma exposures, male rats appeared to be more susceptible than female rats, as females showed only urothelial hypertrophy and PPB-positive cells, but no inclusions. Therefore, it appears that submucosal erythrocyte extravasation occurred in both genders, but no erythrocytes penetrated the urothelial basal lamina in females. Assuming that the penetration is forced by abnormal stretching of the urothelium as discussed above, this would indicate that the effect of tasipimidine on bladder function is more pronounced in male rats than in females. Male rats can be more susceptible to produce bladder over-filling due to urethral plugs 36 and stronger urethral smooth muscle layer. 37 The latter is especially interesting as α2-adrenoceptors agonist may cause urethral muscle contraction. 15 Male rat might, therefore, be more sensitive to micturition reflex inhibition induced by α2A/D-adrenoceptor agonism. Male bladder is also more susceptible to reperfusion damages, 38 and a number of urodynamic gender differences have been observed in voiding, as well as neurochemical differences in brain regions regulating bladder function. 39 It is likely that anatomical and neurophysiological differences also contribute to the species differences between rats and dogs. In addition, it is noteworthy that the urinary concentrations of tasipimidine and the metabolite were higher in rats and a potential leakage of the compounds through the urothelium would lead to higher local concentrations immediately beneath the urothelium in rats compared to dogs. The current study does not provide any evidence of such leakage, but tasipimidine penetrates other tissue barriers well (brain-to-plasma ratio is about 2.5 in rats). Bladder overdistension is associated with increased urothelial permeability in rats, 40 and the urothelium is thicker in dogs than in rats 41 ; thus, rat urothelium might be more prone to stretch-increased permeability. Also, dogs treated with tasipimidine for 28 days did not show any histological signs (urothelial or muscle hypertrophy) of overdistension.
In conclusion, this study showed that rat urothelial cells have the capability to phagocytize intact erythrocytes, likely originating from small microvessels located immediately beneath the urothelium. In this case, the proposed underlining mechanisms are related to α2-adrenoceptor agonist by the test compound tasipimidine. The leakage of erythrocytes is proposed to be a result of (1) bladder overfilling with stretch-related additive effects, such as changes in the blood flow, together with (2) local vasoconstriction of suburothelial blood vessels. Despite similar plasma exposures, urinary excretion, and pharmacological effects in rats and dogs, dogs did not develop urothelial changes after once daily treatment with tasipimidine for 28 days, indicating that the findings were a rat-specific phenomenon.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MHH, SK, TK, JL, and TL are employees of Orion Corporation, which hold tasipimidine patent and marketing rights.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Orion is the sponsor of the study which was performed on a fee for service basis by Charles River. No financial support for authorship or publication.
