Abstract
The histopathology slide seminar “Classic Examples in Toxicologic Pathology XXVII” was held on February 21 and 22, 2020, at the Department of Pathology at the University of Veterinary Medicine in Hannover, Germany, with joint organization by the European Society of Toxicologic Pathology. The goal of this annual seminar is to present and discuss classical and actual cases of toxicologic pathology. This article summarizes the presentations given during the seminar, including images of representative lesions. Ten actual and classical cases of toxicologic pathology, mostly induced by a test article, were presented. These included small intestine pathology and transcriptomics induced by a γ-secretase modulator, liver findings in nonhuman primates induced by gene therapy, drug-induced neutropenia in dogs, device-induced growth plate lesions, polycystic lesions in CAR/PXR double knockout mice, inner ear lesions in transgenic mice, findings in Beagle dogs induced by an inhibitor of the myeloid leukemia cell differentiation protein MCL1, findings induced by a monovalent fibroblast growth factor receptor 1 antagonist, kidney lesions induced by a mammalian target of rapamycin inhibitor in combination therapy, and findings in mutation-specific drugs.
Keywords
Introduction
The seminar “Classic Examples in Toxicologic Pathology” has 2 goals: (1) as part of the PhD training program of the University of Veterinary Medicine in Hannover, it aims to give postgraduate students in pathology insight into toxicologic pathology and (2) as one of the major scientific activities of the European Society of Toxicologic Pathology (ESTP), it offers focused training for both inexperienced and experienced colleagues. Participants can examine representative lesions by use of scanned whole slide images prior, during, and after the seminar. In addition, the university provides microscopes for each participant for examination of glass slides (usually 60 recuts per lesion), which are made available during the seminar for most presentations. The discussion during and after slide examination by the participants is an integral part of this seminar.
Ten presentations of 45 minutes duration, including discussion, were made on 2 half days. Several cases presented were from ongoing research or development projects. This was possible only as it was not required to disclose chemical structures or compound names. Cases of particular morphological interest were presented without disclosure of the pharmacological target. Eight speakers received employer/sponsor permission to publish their case in this article. The remaining 2 are described here in brief:
Ingrid Pardo from Pfizer, Groton, Connecticut, presented liver findings in nonhuman primates induced by gene therapy. She illustrated the degenerative/regenerative morphological findings, made correlations to clinical signs and clinical pathology parameters, and presented a plethora of mechanistic investigations. In addition, she gave a brief overview of induced pathologies in other organs. Finally, she proposed a mode of action of the gene therapy leading to the morphological and functional effects described. Dr Pardo is going to publish this case separately in a comprehensive original article.
Michael Winter and Barbara Lenz from Roche Innovation Center in Basel, Switzerland, presented a case of drug-induced neutropenia in dogs. Dr Winter described in detail the hematological changes including morphological alterations as evidenced by examination of blood smears and the associated increase in inflammatory parameters. He paid special attention to their time course, including reversibility. Dr Lenz presented the complementary histopathology findings, before both colleagues showed results of mechanistic investigations. The presentation was concluded with demonstration of the mode of action, alternative modes, and their human relevance. Also, this case will be published separately as original article.
Drug-Induced Small Intestine Pathology
The first case of the 2020 ESTP Classical Examples series was presented by Dr Marielle Odin (Roche, Basel, Switzerland) and introduced the added value of molecular techniques to standard pathological assessments to further understand the potential mechanism of drug-induced toxicity, adding perspective to the risk assessment.
The study case came from a 4-week Good Laboratory Practice (GLP) oral toxicity study in Göttingen minipigs with compound A, a small molecule targeting the processing of amyloid β subsequent to secretase-mediated amyloid precursor protein cleavages. The presentation focused on the challenges and opportunities to establish the transcriptomics profile of the major target organ, the small intestine, from formalin-fixed paraffin-embedded (FFPE) tissue blocks, and to identify potential alterations in Notch-related gene expression in these tissues.
Compound A belongs to the class of second generation γ-secretase modulators. It was shown in vitro to not interact with Notch pathways and revealed no relevant histomorphological changes in a 4-week GLP oral toxicity study in mice. In the 4-week oral toxicity study in minipigs, dose levels were reduced from day 14 onward due to physical signs and body weight loss. All animals survived until scheduled sacrifice. Antemortem findings included emesis at ≥100 mg/kg/d, decreased activity, subdued behavior, soft/watery feces, reduced food intake and body weight gain at ≥ 80 mg/kg/d. In histopathology, goblet cell hyperplasia was observed in the small intestine at ≥ 60 mg/kg/d. This finding was more prominent in the duodenum (Figure 1A-D) than in the jejunum and not seen in the ileum; alcian blue staining confirmed the presence of many mucus-filled intestinal goblet cells in crypts and in villi and the abundance of mature mucus in the intestinal lumen (Figure 1E and F).
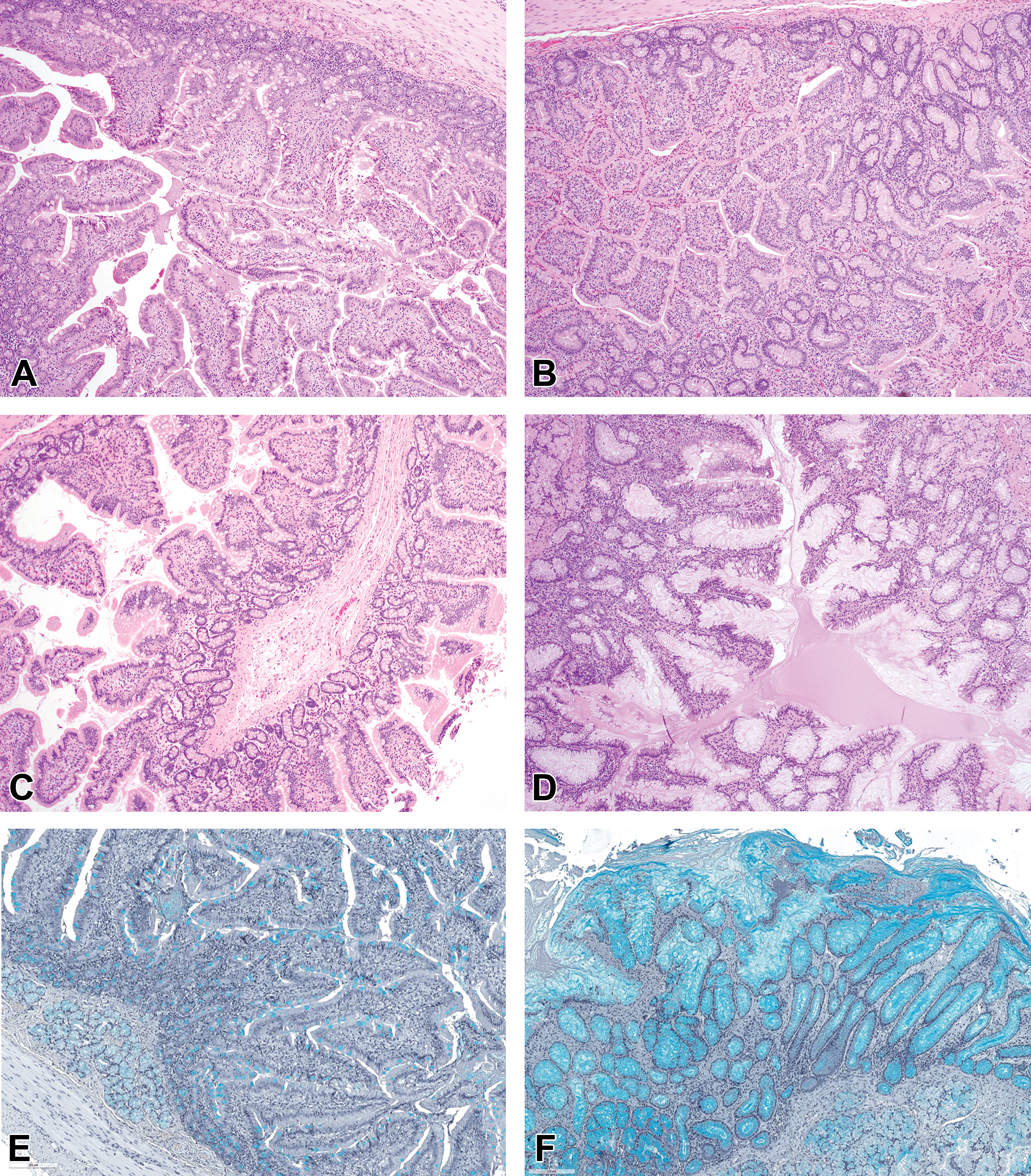
A-D, Duodenum from control (A and C) and treated (B and D) animals. Note the increased number of mucus-secreting goblet cells in the crypts and villi of the mucosal epithelium in the treated animal (B and D) compared to control animal (compare the lower magnifications B to A and the higher magnifications D to C). E and F, Duodenum from control (E) and treated (F) animals. Increase of mucus secreting goblet cells in the mucosal epithelium of crypts and villi, with goblet cells in juxtaposition in crypts and on villi. Alcian blue stain. (Continued). G, Gene-set enrichment analysis of the minipig tissue samples against small intestinal epithelial cell signature using BioQC computational tool. 1 Each 1 column represents the signature of 1 animal. Left, crypts: Slight goblet cell enrichment in treated animals (right columns, 80) when compared to control animals (left columns, 0). Minimal proximal absorptive enterocyte signature (most likely indicating sampling close to the distal potion of the crypt), minimal tuft cell signature, and no detectable entero-endocrine signature in both treated and control animals. Right, villi: Goblet cell enrichment in treated animals (right columns, 80) when compared to control animals (left columns, 0). Overt proximal and distal absorptive enterocyte signature. Minimal Tuft cell signature and no detectable entero-endocrine signature in both treated and control animals. H, Differential regulation related to controls of Notch-related genes in the duodenum (statistically significant changes highlighted in yellow) and the jejunum (statistically significant changes highlighted in pink) of treated animals. Left, crypts: Upregulation of ATOH-1 and SPDEF, and, to a lesser extent, KLF4 and downregulation of OLFM4 in the duodenum. No notable change in HES-1. Marginal or no meaningful differences in the jejunum crypts. Right, villi: Overall similar profile as in crypts, with additional clear upregulation of HES-3 and downregulation of HES-5. No meaningful expression differences in the jejunum villi.
Based upon the nature of the change, Dr Odin hypothesized that compound A administration led to alterations in downstream Notch-signaling pathways in the small intestine. To investigate this hypothesis, the transcriptomic profile of the minipig small intestine was established from FFPE tissue blocks, using laser capture microdissection to isolate intestinal epithelium from the crypts and villi of the duodenum and jejunum, and the SMART-Seq Stranded Kit (ref. 634443) from Takara (adapted to low-yield RNA samples) for library preparation prior to RNA sequencing. The analysis was based on the model of intestinal differentiation published by Noah et al. 2
Although transcriptomic analysis was performed on samples collected after 4 weeks of repeat dosing, the investigation was considered appropriate to detect meaningful signals because of the high cell turnover in small intestine. The results could be summarized as below: Tissue signature correlated with histopathology (Figure 1G). Downstream Notch-signaling pathways were affected in treatment-group samples, with noteworthy increased SPDEF and ATOH-1 expression in the duodenum, while the transcription factor HES-1 remained unchanged (Figure 1H). Other variations in signaling and metabolic pathways corresponded to physiopathological responses (data not shown). More details on the investigations will be part of a future publication (in preparation).
Besides the technical challenges of preanalytical steps, from tissue collection, preservation and further processing to tissue preparation for RNA sequencing, and the discussion around the transcriptomics data, results, and limitations, the discussion evolved around the terminology used to report an increased number of goblet cells in the small intestine. Two schools challenged each other: * Those favoring the use of “goblet cell hyperplasia”, that is, increased number of a cell type that is already existing in the given location in the tissue. * Those favoring the use of “goblet cell metaplasia”, that is, occurrence of a cell type that normally would not be present in such a number and/or such a location in the tissue.
While both terminologies can be found in the literature to report such a finding, 3,4 the author opted for hyperplasia to focus on the morphological aspect and the fact that there was no new cell type in the given location (ie, small intestine). Although goblet cells are most prominent at the isthmus of the crypts in the small intestine and scattered along the villi, their presence in the intestinal epithelium does not indicate an abnormal differentiation (dysplasia) or an abnormal location for such a differentiation (metaplasia). Yet, the transcriptomics results indicate an alteration in the differentiation paradigm with a push toward mature goblet cells (increased SPDEF/ATOH1) over absorptive enterocytes in the duodenum. 2,5,6 If we consider the molecular mechanism of differentiation, the change could potentially fit with a definition of metaplasia. This point of discussion around nomenclature exemplifies the challenges faced by the toxicologic pathologist when recording findings in preclinical toxicity studies: Should he or she use pure morphological diagnosis(es) or should he or she consider a potential pathogenesis behind the morphological change? The increasing use of quantitative molecular techniques on top of histopathological examination will surely contribute to the transformation of the pathology contributions.
Device-Induced Growth Plate Changes
Dr Nehrbass (AO Research Institute Davos [ARI], Davos, Switzerland) presented results of a study on a new pediatric medical device for growth modulation using the alloy nitinol. Nitinol is currently used for orthopedic devices in adults in the form of staples to treat deformities at fore and midfoot (hallux) and as intramedullary nails. It is also used for vascular/cardiac devices (eg, stents, balloons, catheters, atrial meshes, wires). Due to its unique physical properties, it could potentially be used in mechanical growth modulation to treat skeletal deformities in infants/juveniles (eg, at spine [scoliosis] and femur/tibia [leg length discrepancy, LLD]).
Nitinol is a nickel-titanium alloy (Ni and Ti in roughly equal atomic percentages) named by the laboratory who invented it (Nickel Titanium Naval Ordnance Laboratory). It is a non-ferro-magnetic (advantageous for magnetic resonance imaging), superelastic metal with shape memory properties which are based on reversible, temperature-dependent (martensitic) phase changes of the crystalline grid. To produce a device with shape memory, manufacturing undergoes several steps: Preshaping (under heat). Initially, the device is bent applying a defined force (= preload). Straightening (under deep-frozen conditions). The device is bent back. Storage (at low temperature). For example, at 20 °C. Implanting (at body temperature). Under the influence of the warm temperature of the bone, the straight implant tends to regain its original preshaped form (= shape memory) applying a defined compression force.
The goal of the study was (A) to develop an animal model 7,8 for mechanical growth modulation to treat LLD at its preferential site in infants being femur and/or tibia and (B) applying a compression force 9 which avoids a complete physeal growth arrest. Due to the complex, nonsymmetric anatomical shape of the sheep’s knee joint (in which the rudimentary fibula causes a convex contour at the lateral side 10 ), for the initial study a different site was chosen (distal growth plate of the metatarsus III + IV: L = left leg untreated [internal control] and R = right leg treated). Animals used were skeletally premature, male-neutered Texel sheep (27-37 kg body weight), with an age of 5 months (at device implantation surgery) plus 2 months (at revision surgery for the device removal) plus 2 months (for recovery) resulting in an age of 9 months at euthanasia. For visualization of the temporal processes of bone formation, 3 different calcium-seeking fluorochromes were sequentially applied subcutaneously at different time points near to implant removal (calcein green: 2 weeks prior to implant removal [week 22], xylenol orange: at implant removal [week 24], oxytetracycline: 4 weeks after implant removal [week 28]). This method allows observation of the changes “over time” at the sequentially formed bone in a single slide of only one animal without the need of additional animals at interim necropsies, as these fluorescent dyes form chelate bonds with Ca2+ depositions in calcifying bone tissue that is newly formed at the short time period of their in vivo application. After necropsy and removal of the devices, contact radiographs (CRs) of undecalcified full thickness bone samples were made (Figure 2A and B). Afterward, samples were fixed in 70% ethanol, dehydrated, embedded in poly-methylmethacrylate resin and subsequently cut in a coronary plane at a thickness of approximately 350 μm, using a diamond blade band saw. Subsequently, CRs were also made of the unground sections (Figure 2C). Two consecutive sections were ground and polished down to a thickness of 105 to 265 µm. One section was surface-etched and stained with Safranin O/Fast Green and investigated by brightfield microscopy (Figure 2D-G). The other section was kept unstained to qualitatively investigate the fluorochrome labels by fluorescence microscopy (Figure 2H-I).
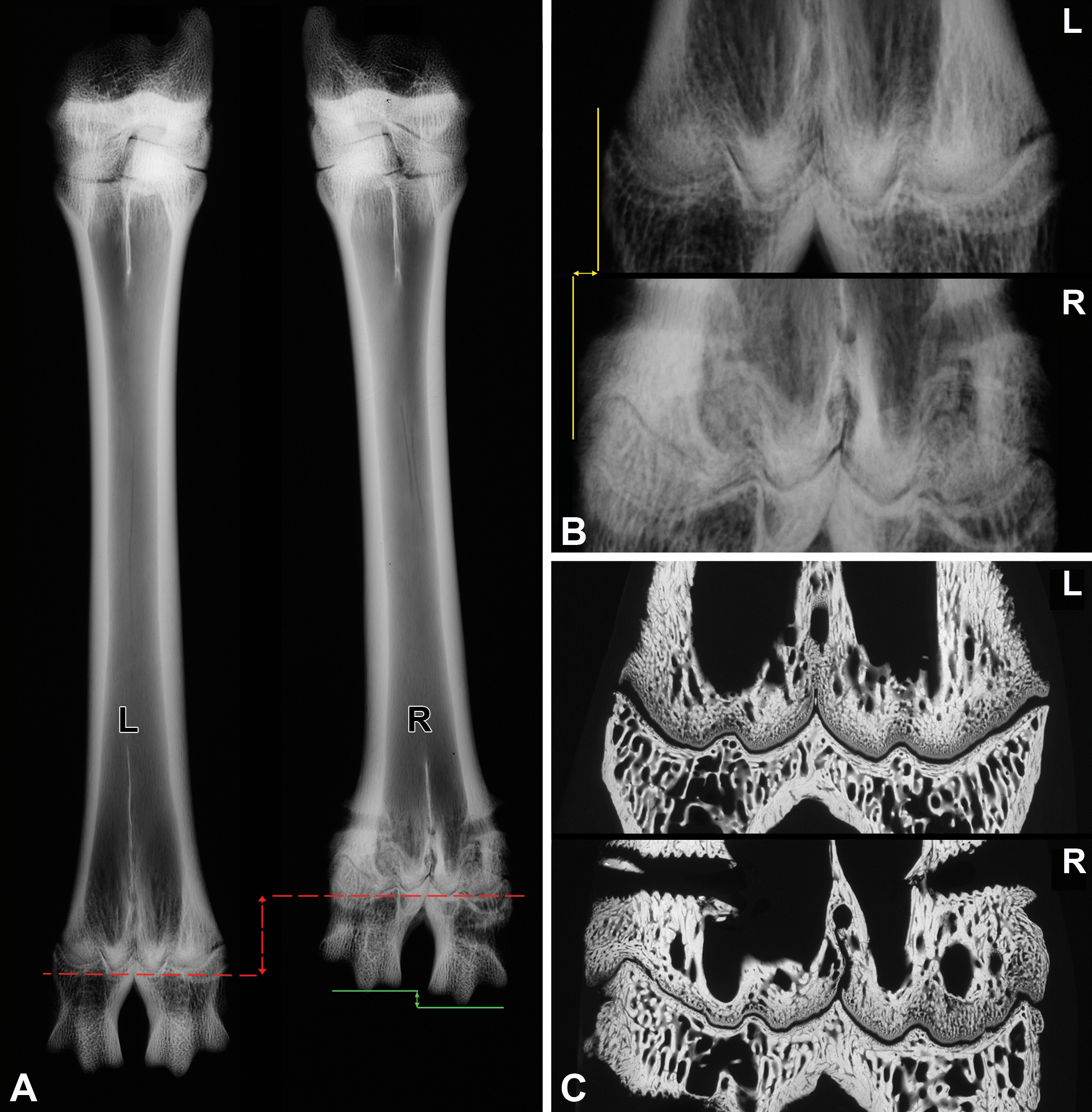
A-C, Contact radiographs (CR, device removed) of metatarsus III + IV (L: left-sided, untreated control; R: right-sided, treated). A, CR of full thickness bone (“3D”), fixed, nondecalcified, nonembedded sample. Note the intended length shortening (red arrow) of the treated right bone compared to the untreated left bone, as well as the step formation at the right metaphalangeal joint (green arrow). B, Higher magnification of distal growth plate and epiphysis of (A). Note the widening of the right, treated bone (at the level of the distal physis) compared to the untreated one (yellow arrow). C, CR of slides (“2D”) after coronary cutting of the distal area. Note the alteration of the trabecular bone structure directly proximally of the growth plate (for more details see [G]). (Continued). D-G, Brightfield (BF) images of Safranin O/Fast Green (SOFG) stained slides. D and E, The identical slides as shown in (C). L: left-sided, untreated control; R: right-sided, treated; p: peripheral, c: central. D, Acute phase (immediately after device removal). Left untreated side with physiologic growth plate morphology characterized by symmetric undulation of the 2 fused metatarsal bones; right treated side with distinct asymmetry. E, Higher magnification of areas shown in (D). Left-sided, untreated sample (upper row) demonstrating the physiologic difference between wider peripheral areas (p) and thinner central areas (c). Both areas with highly ordered cell stacking. The right-sided, treated sample (lower row) with discontinuity, thinning, and slight perturbation of the physis. F, Recovery phase (2 months after device removal), peripheral areas of other right-sided, treated samples (R). Growth plate changes of different degree and morphology (thickening, step/spike formation perturbation/distortion). G, In contrast to the left-sided, untreated sample (L), the right-sided, treated samples (R) with alterations of the trabecular meshwork, in particular shortened or fused trabeculi, reduced number and increased density of trabeculi, which went in parallel to the growth plate changes. Thick-sections, fixed, PMMA-embedded, cut, ground, Safranin O/Fast Green stained, 2D, scale bar 5 mm (overview image composed of stitched tiles and extended focus imaging applied). E, Scale bar 500 µm; F and G: scale bar 200 µm. (Continued). H and I, Fluorescence images visualizing of the temporal processes of sequential bone formation (images of consecutive sections shown in C and D). H, Overview showing highly orchestrated bone formation in the untreated control sample on the left side in contrast to the uncoordinated bone formation in the treated sample on the right side. I, Details of peripheral areas shown in (A). Note the highly ordered sequence of bone trabeculae of different ages in the untreated sample on the left side, compared to the chaotic bone formation in the treated sample on the right side. Thick sections, fixed, PMMA-embedded, cut, ground, unstained, labeled; green label: calcein green (week 22 = 2 weeks prior to implant removal), red label: xylenol orange (week 24 = at implant removal), and blue label: oxytetracycline (week 28 = 4 weeks after implant removal). H, scale bar 5 mm; I, scale bar 200 µm. PMMA indicates poly-methylmethacrylate.
At necropsy, gross pathology revealed a length shortening of the right, treated side (Figure 2A, red arrow). This intended temporary growth reduction should clinically lead to an alignment with the altered, contralateral side. Side effects were shape changes of the affected bone areas: For example, widening 11 of the bone circumference in both planes, sagittal and coronary (Figure 2B, yellow arrow), and step formation at bone ends/joints (Figure 2A, green arrow). 12 Mechanically induced growth plate changes consist of a plethora of malformations arranged in parallel in growth plate cartilage and trabecular bone. As a consequence, dysplasia of the physis was found and differed in morphology from the acute phase (thinning; Figure 2D and E) to the recovery phase (thickening, step/spike formation, perturbation/distortion, changes in the cellularity of the layers; Figure 2F). Additionally, dysplasia of the trabecular bone was found. This meshwork which is contiguously formed by the ossification of the growth plate cartilage showed trabecular fusion, trabecular shortening, increased trabecular density, and decreased trabecular number (Figure 2G). Finally, and most prominent, were the temporal changes in bone deposition. Instead of a highly orchestrated bone formation resulting in highly ordered bone trabeculae of different age, as seen on the untreated leg, the treated leg showed a completely uncoordinated, chaotic bone formation (Figure 2H and I), leading to macroscopically visible shape deformations. Due to continuous bone remodeling processes after implant removal, it is very likely that these deformations would potentially be, at least in part, reversible.
In the discussion, Dr Nehrbass pointed out that the exact pathogenesis of mechanical growth modulation is unknown. 13 Potential alterations are highly dependent on which zone is affected 8,14 –16 and are seen as either (A) the growth plate cartilage with its germinal, proliferative, hypertrophic, or ossification zone (growth in length) and/or (B) the bone with its perichondral zone of bone apposition (growth in width) and its metaphyseal remodeling zone (shape modulation). Potential mechanisms consist of changes in cell division or death of germinal cells, disturbed matrix production (especially collagen II and aggrecan), maturation of chondrocytes, altered chondrocyte apoptosis versus transdifferentiation from chondrocytes to osteocytes, 11 disturbed matrix calcification, or disturbed vascularization. Regarding translatability, the results of this study can be transferred to the situation in children in which the method of mechanically induced growth modulation (epiphysiodesis) by unilateral positioning of conventional implants (eg, steel, titanium) is a common surgical method, for example, by using screw/plate combinations (like eight Plate, Hinge Plate, Peanut Plate, Butterfly 8 Plate, or with staples). 17,18 In contrast, the bilateral use, necessary for therapy of leg length discrepancies, reflects an off-label use and is not (yet) approved by authorities. The maximal force of 200 Newtons (N) applied to the growth plate (correlating to a mass of approximately 20 kg) is in the physiological range of the proximal tibial physis of growing children with LLD. 19 This means it inhibits the growth but does not permanently stop the physeal growth. Although a nonorthotopic model was used, the results have a high predictivity for the human situation. The next steps would be adaptation of the device to the use at the clinically preferential site at the knee joint (epiphysis of distal femur and or proximal tibia). In this context, the testing of new methods and/or new materials using animal models is an indispensable mean to improve patient treatment. Moreover, more research is needed to understand the changes observed and the consequences thereof.
After the presentation, participants discussed the diagnostic term for the changes at growth plate and adjacent trabecular bone and came to the consent that “dysplasia” is more appropriate than “malformation.”
Findings in Kidneys and Livers of CAR/PXR Double Knockout Rats and Their Influence on Liver Cell Proliferation
Dr Treumann and Dr Marxfeld (both BASF SE, Ludwigshafen, Germany) presented cases of polycystic kidney disease (PKD) in CAR/PXR double knockout (CARKO/PXRKO) rats bred on a Sprague Dawley (SD) background. The CARKO/PXRKO rats as well as wild-type (WT) rats were included in a cell proliferation study (Table 1) to test the usefulness of the knockout model with phenobarbital (PB); animals were 7 weeks old at the beginning of the study. As expected, only in WT animals receiving 500 ppm PB in the diet, a minimal to slight centrilobular hypertrophy was observed in all livers. Two control and 3 treated CARKO/PXRKO rats revealed enlarged kidneys at necropsy. Hematoxylin and eosin (H&E)-stained slides of the kidneys of these grossly affected animals showed high numbers of tubular cysts, predominantly in the outer stripe of the outer medulla and to lesser extent in the cortex (Figure 3A and B). In addition, a slight fibrosis was observed with mild infiltration of inflammatory cells. In the livers of the same animals, there were marked biliary cysts with fibrosis and inflammatory cell infiltrates, diagnosed as hyperplasia bile duct, cystic (Figure 3C and D).
Design of a Study With Phenobarbital for Demonstration of the Usefulness of CAR/PXR Double Knockout Rats When Compared to Wild-Type Rats in the Assessment of Cell Proliferation.
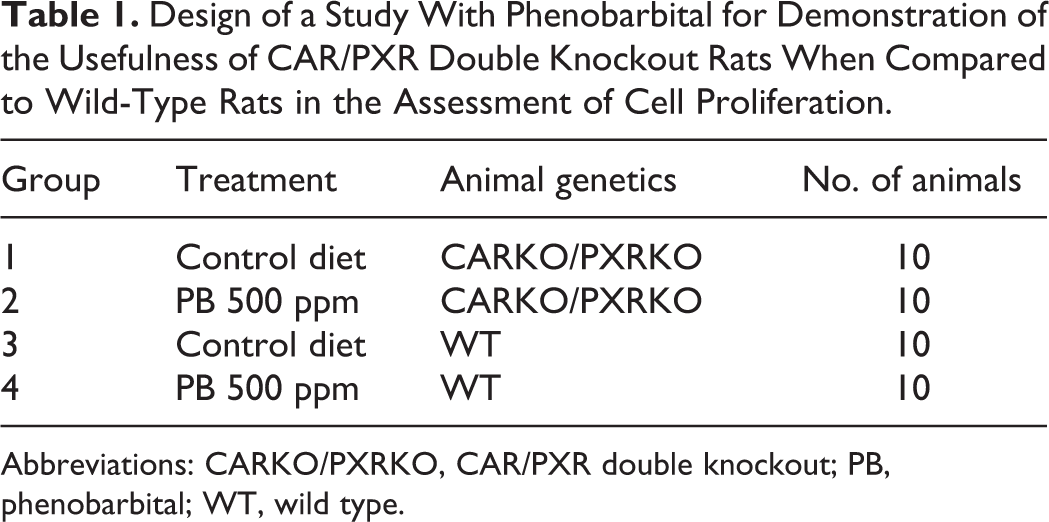
Abbreviations: CARKO/PXRKO, CAR/PXR double knockout; PB, phenobarbital; WT, wild type.
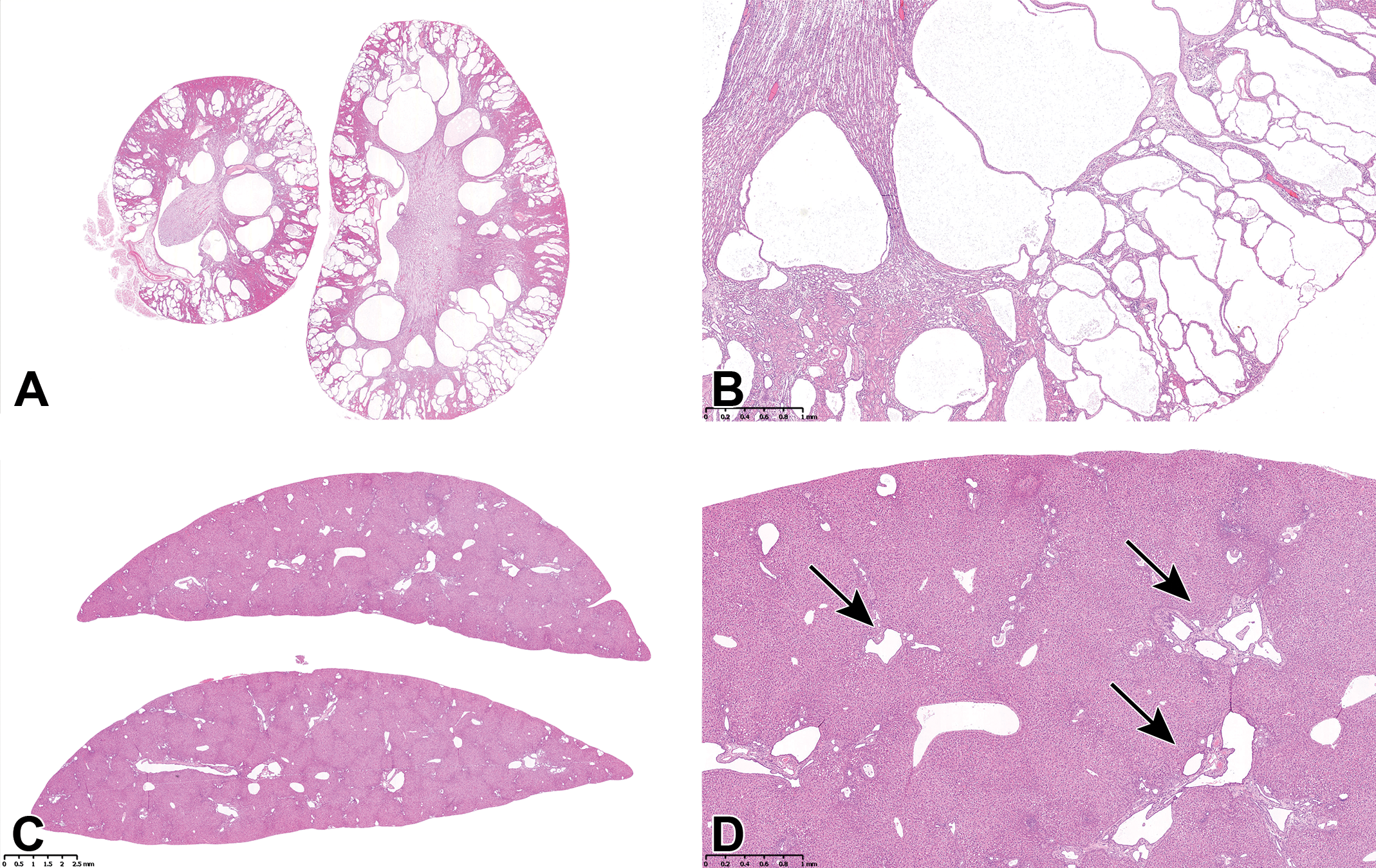
Examples of cystic kidneys and liver bile duct proliferation from a CAR/PXR double knockout rat. A, Low magnification of affected kidneys with severe polycystic kidney disease (PKD) characterized by multiple large tubular cysts, in the cortex and outer stripe of the outer medulla (OSOM). B, Higher magnification of an area of cystic tubules in (A). C, Low magnification of a liver with cystic bile duct proliferation. D, Higher magnification of an area in (B) showing greater detail of the cystic bile ducts (arrows).
Constitutive androstane receptor (CAR) and pregnane X receptor (PXR) are important elements in the mechanism for development of hepatic carcinogenesis in the rodent following exposure to some xenobiotics. Many nongenotoxic chemicals such as PB cause liver tumors in rats and mice. 20 This liver tumor formation is often associated with the selective induction of hepatic microsomal cytochrome P450 (CYP) enzymes. These induction responses are normally mediated through the activation of nuclear receptors (as CAR and PXR) that lead to enhanced gene transcription. 20 The CAR activation results in changes in the expression of a wide range of genes, including genes involved in phase I and II xenobiotic metabolism as well as regulation of genes associated with various physiological processes such as cell proliferation, apoptosis, and metabolism. 21,22 Many of the molecules that can activate CAR may also activate PXR, producing a combined response pattern of gene expression and functional changes. 23
Polycystic kidney disease is a cystic genetic disorder of the kidneys which is typically associated with cystic bile duct dilation in the liver of humans as well as domestic and laboratory animals. In animals, familial/hereditary PKD has been recognized in many species including dogs, goats, mice, and rats. 23 It is known that PKD with cystic bile duct dilation in the liver can be an inherited genetic disorder in SD rats. 24 -26 In this case, it occurred in genetically modified animals only, namely CARKO/PXRKO rats and not in WT animals. The affected animals in this study were siblings (information from the breeder). A genetic analysis revealed the same mutation as is spontaneously occurring in SD rats. Therefore, it was assumed to most likely be a spontaneous mutation rather than a mutation forced by the knockout.
Bromodeoxyuridine-stained slides were evaluated for cell proliferation according to Bahnemann and Mellert. 27 As expected, there was a statistically significant (3-fold) increase in cell proliferation in WT rats treated with PB, whereas CARKO/PXRKO rats without cystic bile duct proliferation revealed labeling indices (LI) comparable to control animals. In the control group of CARKO/PXRKO rats, there were 2 of 10 animals with cystic bile duct hyperplasia. These 2 animals had a 10-fold higher LI in the periportal area (zone 1) when compared to unaffected rats. In CARKO/PXRKO rats receiving 500 ppm PB, the same phenomenon was observed in animals with cystic bile duct hyperplasia, revealing a 7-fold higher LI compared to animals without bile duct hyperplasia.
Dr Treumann and Dr Marxfeld concluded that when performing cell proliferation studies, it is especially important to review the organs on H&E stained slides to ensure they do not exhibit potentially confounding spontaneous findings before evaluating the LI. As shown in this study, some (spontaneous) findings can interfere with the study results. In this case, bile duct hyperplasia was associated with an increase in hepatocellular cell proliferation in the periportal area.
Otoconial Dysplasia in Mice
Dr Andreas Popp (Abbvie GmbH & Co. KG, Ludwigshafen, Germany) presented this case from a breeding colony of an unspecified transgenic mouse line. The colony started with 3 animals (1 male and 2 females). After expansion of the colony, 16 of 133 offspring showed an unexpected severe clinical phenotype: stereotypic behavior (circling), ataxia, and small size. Most of the animals with phenotype had to be euthanized for ethical reasons. Three mice harboring this phenotype were killed and brain and skull were processed for histopathological evaluation. After decalcification, the skull was trimmed transversally close to the external auditory canal (Figure 4A).
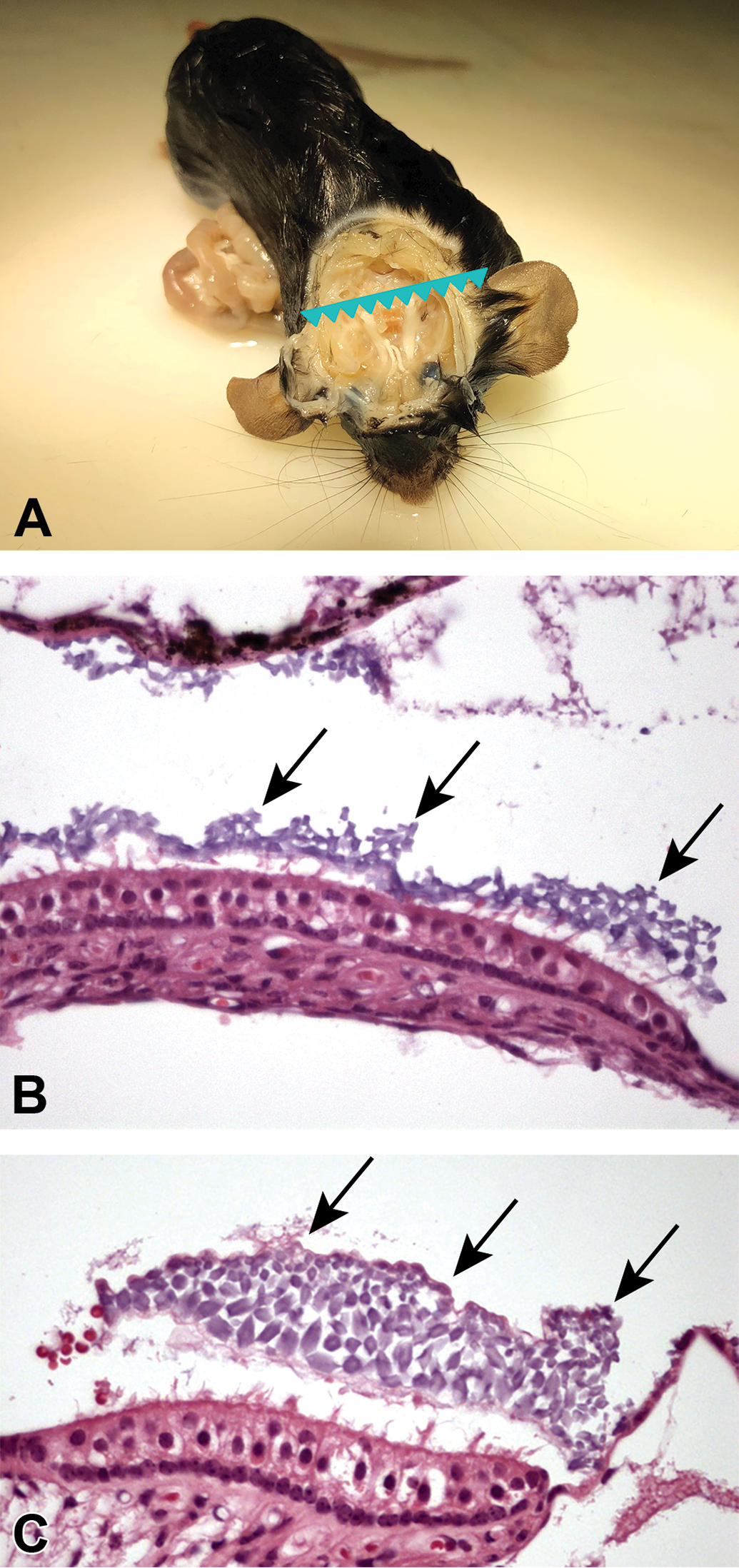
A, Trimming level for evaluation of the inner ear, indicated by the serrated line. B and C, Saccule of mice with normal (B) and altered (C) clinical phenotype. Clusters of enlarged and irregularly shaped otoliths are present on the surface of hair cells in C (arrows) compared to the hexagonal and evenly sized otoliths in B (arrows).
The inner ear is the innermost part of the vertebrate ear. It is mainly responsible for detection of auditory signals and balance. 28 In mammals, it consists of the bony labyrinth; a hollow cavity in the temporal bone of the skull with a system of passages comprising 2 main functional parts. 29 The cochlea is dedicated to hearing, converting acoustic pressure patterns from the outer ear into electrochemical impulses which are passed on to the brain via the auditory nerve. The vestibular system of the inner ear is responsible for the sensation of balance and motion. It uses the same kinds of fluids (perilymph and endolymph) and sensory cells (hair cells) as the cochlea and sends information to the brain about the posture, rotation acceleration, and linear acceleration of the head. The type of acceleration or posture detected by a hair cell depends on its associated mechanical structures, such as the curved tube of a semicircular canal or the calcium carbonate crystals (otoliths) of the saccule and utricle.
An otolith, also called statoconium, otoconium, or statolith, is a calcium carbonate structure in the saccule or utricle of the vestibular system. The saccule and utricle, in turn, together make the otolith organs. These organs enable an organism, including humans, to perceive linear acceleration, both horizontally and vertically. In mammals, otoliths are small particles, composed of calcium carbonate crystals embedded in the gelatinous otolithic membrane and surrounded by the viscous fluid endolymph of the saccule and utricle (Figure 4B). The inertia of these small particles causes them to stimulate hair cells when the head moves. The hair cells are made up of 40 to 70 stereocilia and 1 kinocilium and are connected to an afferent nerve. Stereocilia and the kinocilium extend into the gelatinous matrix of the otolithic membrane. When the body changes position or begins a movement, the weight of the membrane bends the stereocilia and stimulates the hair cells. Hair cells send signals down sensory nerve fibers, which are interpreted by the brain as motion. The brain interprets the orientation of the head by comparing the input from the utricles and saccules from both ears to the input from the eyes, allowing the brain to discriminate a tilted head from the movement of the entire body.
Investigation of the brain did not give any hint for histopathological changes related to the phenotype. Deeper analysis of the inner ear, specifically the vestibulum, resulted in a very distinct change of the morphology of the otoliths. Presence of abnormal crystal morphology of the otoliths correlated with the expression of the abnormal phenotype (Figure 4B and C). Compared to controls, otoliths in affected animals were irregular, enlarged, and lacked the even cross-sectional hexagonal shape. Hair cell morphology was not changed. There was also no hint for any inflammatory response.
Mice expressing a strong behavior phenotype had a vestibular phenotype consisting of otoconial dysgenesis and no structural changes in sensory hair cells. The phenotype was not related to the introduction of the transgene, as most transgenic animals behaved normal. An analysis of the genetic basis of the changes in this specific case was not possible since the breeding strategy excluded all phenotype expressing animals from the colony.
In the literature, several mouse strains with selective peripheral vestibular deficits have been described and the genes responsible for these deficits have been identified: Mouse mutants carrying null mutations in the cadherin 23 (Cdh23), myosin VIIa, and myosin VI genes are described. 30,31 C57 and A/J strains both carry a G to A transition at nucleotide position 753 of the Cdh23 gene, known as the ahl allele (Cdh23ahl), which can lead to the phenotype of head tilt. 32 -34 Altered incorporation of organic components into otoconia alone (eg, Oc90 and otolin) could also contribute because organic matrix has been shown to regulate the growth, morphology, and stability of each otoconium. 35 -38 Genes on chromosome 17 (Nox3 and Noxo1) play an important role, leading to complete otoconial agenesis when inactive. 39
MCL1 Inhibitor-Induced Lesions
Dr Florian Colbatzky (Boehringer Ingelheim Pharma GmbH & Co. KG, Biberach [Riß], Germany) presented a plethora of histopathological changes in Beagle dogs exposed to an inhibitor of the induced myeloid leukemia cell differentiation protein MCL1.
The lesions were observed in a repeat dose (30-minute slow infusion) exploratory toxicity study. The study comprised 2 phases: In the escalating dose phase, consecutive dose levels of 5 mg/kg (3 days [days 1-3]), 15 mg/kg (4 days [days 4-7]), and 30 mg/kg (2 days [days 8-9]) were administered to 1 male and 1 female dog. The fixed dose phase was started with 1 drug naive male and 1 drug naive female at a dose level of 20 mg/kg. Due to moribundity of the male animal resulting in its premature sacrifice, the remaining female was administered the reduced dose of 15 mg/kg for an additional 6 days (days 2–7). Besides the standard study parameters including gross and histopathological examination, ultrastructural examination of the heart and an messenger RNA profiling (toxicogenomics) of heart samples using RNA sequencing (Illumina NextSeq 500) were conducted to characterize the toxicological profile of the compound.
Clinical signs were apathy, ataxia, and muscle rigidity. All animals showed moderate signs of local intolerability (ie, histopathology of infusion sites) and temporary dark red discoloration of the urine after each injection due to hemoglobinuria. Besides large amounts of proteins in general, the urine contained a variety of different crystals (tyrosine, calcium phosphate, other crystals with high potassium, sodium, chloride, sulfur and/or phosphorus). In addition, the animals showed exposure-dependent increases in plasma enzyme activities of alanine transaminase, aspartate transaminase, alkaline phosphatase, creatine kinase, γ-glutamyl transferase, glutamate dehydrogenase, and lactate dehydrogenase as well as in total bilirubin, blood urea nitrogen, and packed cell volume and significant decreases in white blood cells and lymphocytes.
At necropsy, 1 dog with exceptionally high systemic exposure to the drug showed hemorrhages in various tissues, especially in fasciae and mesenteric fat tissue, fast rigor mortis, and yellow discoloration of the liver. But there was no evidence for jaundice.
Histopathological target organs of toxicity included the exocrine pancreas, the liver, the kidneys, the esophagus, the gastrointestinal tract, the bone marrow, the thymus, the lymph nodes, the testes, the epididymides, and the infusion sites. The pancreas (exocrine) showed vacuolization and diffuse apoptotic cell death of acinus cells which resulted in a distorted architecture of the exocrine tissue (Figure 5A). Only in the liver of the male, necrosis/apoptosis of hepatocytes was present primarily in the midzonal region (Figure 5B and C). In the mucosa of the stomach and all segments of the small and large intestines including the rectum, there were many apoptotic epithelial cells (Figure 5D-G). Changes in the small intestine were further characterized by villous atrophy and cell debris in the crypts (Figure 5E and F). In the large intestine, pseudodilatation of crypts containing cell debris was observed in addition to apoptoses of epithelial cells (Figure 5G). Only in the male, the epithelial layer of the esophagus was almost completely effaced (not shown). The bone marrow (Figure 5H), the thymus (Figure 5I), the spleen (not shown), and the lymph nodes (not shown) showed cellular depletion and numerous apoptotic cells. Formation of giant sperm cells, loss of round spermatids, and vacuolization of Sertoli cells were present in the testes (Figure 5J and K). The epididymides showed the presence of cellular debris and a reduced number of sperm cells in the duct lumen (not shown). At the injection sites, thrombophlebitis was observed (not shown). In the kidneys (not shown), tubular degeneration, accumulation of hyaline droplets in the tubular epithelium, proteinaceous casts, and vacuolization of tubular epithelial cells were noted.
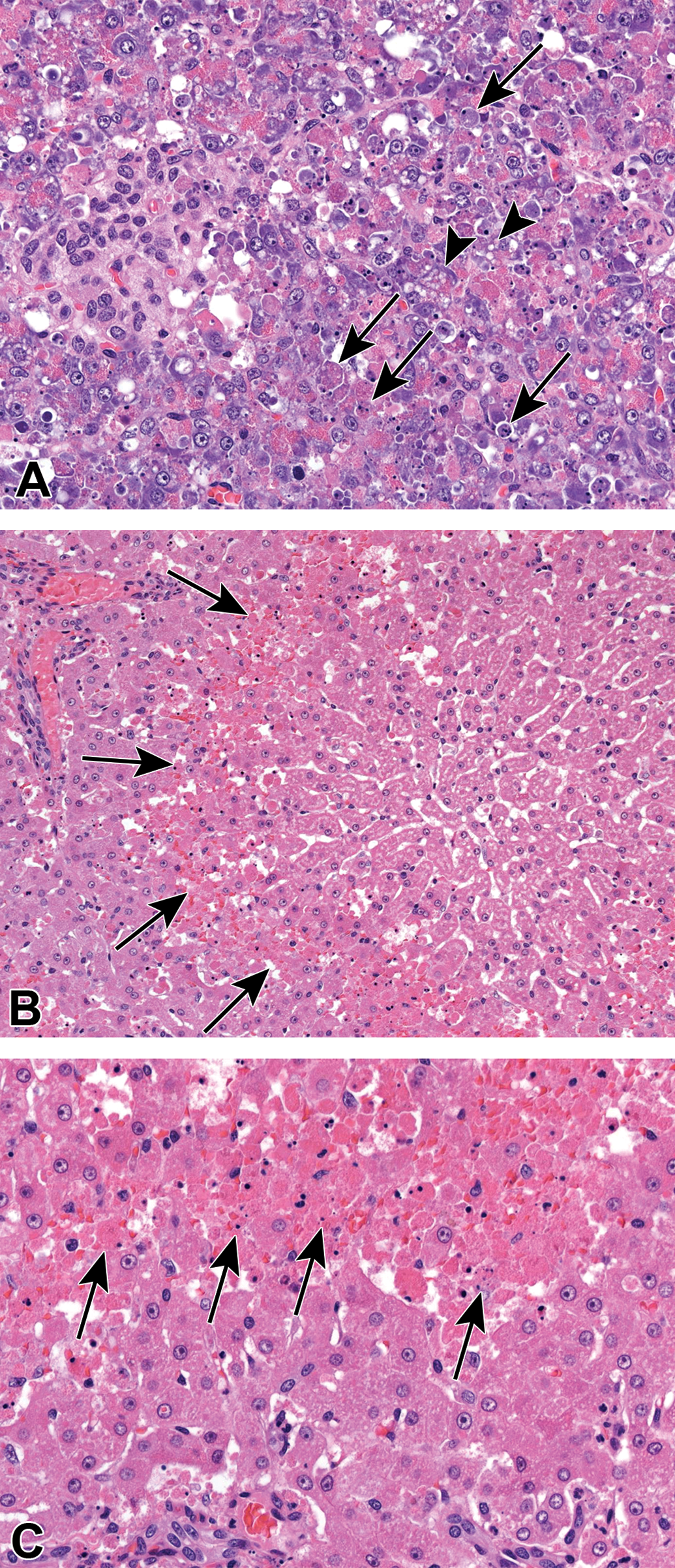
A-C, Representative images from pancreas (A) and liver (B and C) of a male dog killed 1 day after receiving a single dose of 20 mg/kg. The pancreas is severely affected by apoptosis (arrows) and vacuolization (arrowheads) of acinar cells, resulting in a distorted architecture of the exocrine tissue. B, Hypereosinophilia in midzonal localization of the liver (arrows). C, Higher magnification of (B) shows that the hypereosinophilia is caused by hepatocellular apoptosis and congestion of sinusoids which are widened due to the loss of hepatocytes. (Continued). D-G, Representative images from the gastrointestinal mucosa of a male dog treated in the escalating dose phase at daily doses of up to 30 mg/kg for an overall duration of 9 days. D, The gastric pyloric mucosa with scattered apoptoses in the glandular epithelium. Duodenum (E), ileum (F), and colon (G) are affected by villous atrophy (duodenum, ileum), apoptosis, cell debris in pseudodilated glands (arrows), atrophy of crypt, and villous (ileum) epithelium as well as a diffuse reactive inflammatory cell infiltrate. (Continued). H and I. Representative images from bone marrow (H) and thymus (I) of a male dog treated in the escalating dose phase at daily doses of up to 30 mg/kg for an overall duration of 9 days. Low cellular density characterizes the bone marrow and thymus. Apoptotic bodies are visible in the thymus only (arrows). J and K. Representative images from the testes of a male dog killed 1 day after receiving a single dose of 20 mg/kg. Formation of multinucleated spermatids (arrows), absence of early germ cell generations, in particular spermatogonia and spermatocytes (asterisks), and vacuolization of Sertoli cells (arrowheads).
In none of the animals, unequivocal histopathological changes of the heart were detectable, although toxicogenomic analyses provided evidence for the altered expression of multiple genes considered related to the cardiac hypertrophy signaling pathways. 40 Additionally, electron micrographs of the heart did not reveal morphological changes in the mitochondria.
Next, Dr Colbatzky discussed possible mechanisms of pathogenesis for the lesions. Apart from its role in the regulation of mitochondrial apoptotic cell death, the involvement of MCl-1 in various other cellular/mitochondrial functions was shown. These other functions are fulfilled by an alternative (shorter) splice form, which is localized in the mitochondrial matrix. These functions are mitochondrial fusion and fission, 41,42 mitochondrial long chain fatty acid β-oxidation, 43 and interference with the electron transport chain. 44
A critical role for normal cardiac function was assigned to MCl-1, because, for example, loss of MCL-1 caused cardiac failure. 45 -47 The 3 papers by Thomas et al, 45 Thomas and Gustafsson, 46 and Wang and colleagues 47 were the reason for the toxicogenomic analyses conducted in the study presented here. However, neither histopathological changes nor ultrastructural alterations of mitochondria were detectable in the 4 dogs administered an MCL-1 inhibitor as described for various cell types including human stem cell-derived cardiomyocytes. 48 -50
Toxicogenomic analyses of heart of these 4 dogs showed that the expression of multiple genes related to cardiac hypertrophy and related cardiac disorders 40 was significantly altered and provides further evidence that MCL1 may be critical for normal cardiac function.
Structure and function of MCL1 in humans are comparable to many animal species including dogs. It is, therefore, expected that treatment of patients with MCL-1 inhibitors may also cause adverse effects on various organs depending on treatment schedule, dosage, kinetics, and their capability to inhibit also the shorter splice forms of the protein within the mitochondrial matrix.
Monovalent FGFR1-Antagonist Induced Metastatic Mineralization in Rats
Dr Knippel (Merck KGaA, Darmstadt, Germany) presented the results of 2 preclinical toxicity studies in Crl:WI (Han) rats, investigating a novel monovalent fibroblast growth factor receptor 1 (FGFR1) antagonist which shall be used for oncology indications. The FGFR1 is expressed by a certain number of normal tissues but it is highly amplified and overexpressed in a subset of solid tumors, such as lung and breast cancer. 51,52 Amplification and overexpression of FGFR1 have been described as potential oncogenic drivers in lung squamous cell carcinoma and small-cell lung cancer.
The development of a bivalent antibody structure as an anticancer drug is challenging as these antibodies are reported to cause severe hypophagia and weight loss in rodents and monkeys. 53 Therefore, an immunoglobulin G-like monovalent fully human FGFR1 binder with enhanced antibody dependent cellular cytotoxicity effector function was developed. This monovalent binder shows high affinity and selectivity for FGFR1 and is sparing other FGFR family members. It blocks the dimerization of the receptor as well as ligand-mediated activation and thereby inhibits the downstream signaling such as receptor autophosphorylation and ERK1/2 activation. Concentration-dependent inhibition of FGFR1 autophosphorylation was demonstrated in FGFR1-overexpressing lung cancer cells both in vitro and in vivo. Strong antitumor activity in specific FGFR1-expressing cell line- and patient-derived xenograft models was shown. In these xenograft models, it was well tolerated with no signs of body weight loss.
A dose-range finding (DRF) study in rats was conducted as follows: Rats were treated once weekly by intravenous (iv) infusion with the vehicle or 50, 100, or 200 mg/kg (maximum feasible dose) of the test article. Each group consisted of 5 male and 5 female rats. Animals were killed 24 hours after the last (fifth) dosing on day 30. Afterward, an investigative follow-up study was conducted to gain insight into the temporal occurrence of lesions observed in the DRF study. Each of the 10 male rats per dose group were treated once weekly by iv infusion with the vehicle or 10, 25, or 50 mg/kg of the test article. Rats were killed 24 hours after either the first infusion (2 rats/group), the second infusion (4 rats/group), or the fifth infusion (4 rats/group), that is, on days 2, 9, or 30.
Hematology and clinical chemistry investigations were performed at the time of necropsy for both studies. Urinalysis was done after the last dose in the DRF study and after the second and last dose in the follow-up study. Additionally, toxicokinetic analyses were performed by using satellite animals assigned to each dose group. After macroscopic examination, standard tissues were weighed and a guideline conform panel of tissues was processed for histopathology examination.
Dr Knippel then gave an overview on the main results of both rat studies. In the DRF study, clinical chemistry revealed a dose-unrelated increase in serum inorganic phosphorus (IP; group mean value up to +68% vs controls) and a minimal increase in calcium (group mean value up to +8%). Urinalysis showed a slight decrease of pH and protein in males at 100 and 200 mg/kg (urinary Ca and IP were not evaluated in this study). At ophthalmology investigation, focal or diffuse corneal opacity was detected in most animals of all treatment groups. Toxicokinetic analysis showed that the exposure increased roughly proportionally to the increasing dose from 50 to 100 mg/kg and slightly subproportionally from 100 to 200 mg/kg. After repeated dosing, an accumulation was observed. The half-life was in the range of 76 to 129 hours. At gross pathology, no relevant observations occurred. Histopathology investigation revealed metastatic mineralization (calcification proved by von Kossa staining) in various organs of animals at all doses, with male rats clearly more affected than females and without a clear dose-dependency. Mineralization was seen in the intima, subintima, and muscularis of the aorta (Figure 6A) and in various other arteries/arterioles, especially within the heart (Figure 6B), kidney (Figure 6E), stomach, and skeletal muscle. Further tissues/subtopographies showing mineralization were the lung alveolar ducts (Figure 6C), kidney tubular cells (Figure 6E), stomach fundic mucosa, (Figure 6D) and lamina muscularis, basal layer of the corneal epithelium, spinal meninges, lamina propria of the trachea, and the periosteum of the tibia and/or femur. In the bone marrow, woven bone formation (Figure 6F) or fibrosis was visible in males of all dose groups whereas females were not affected.
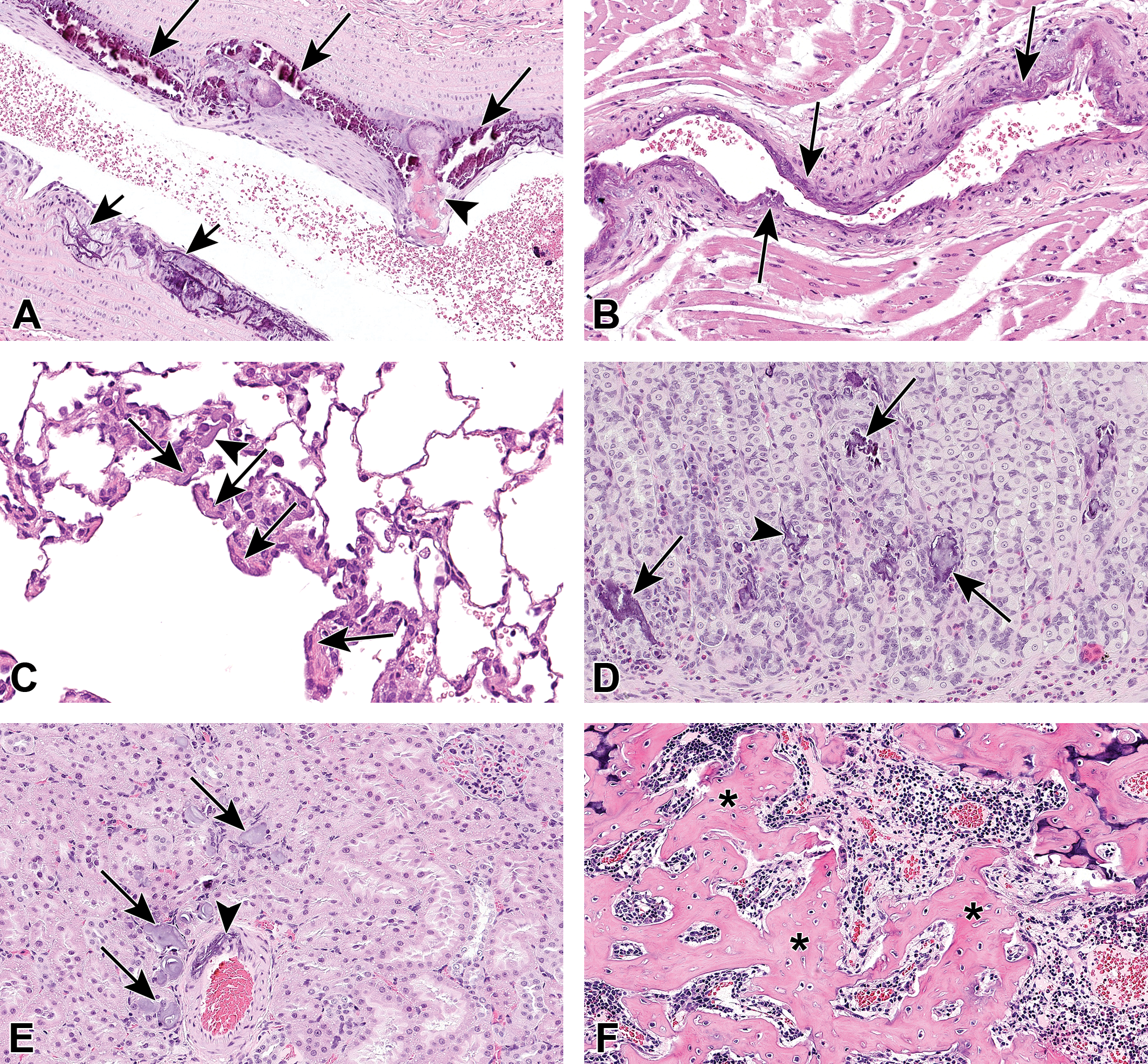
A-F, Representative images taken from animals which were killed on day 30 of the dose-range finding study or the follow-up study in male Wistar rats. A, Aorta close to its origin from the heart (dose 50 mg/kg) with linear mineralization along elastic fibers (short arrows) in the lower part of the picture, whereas the upper part shows a more profound accumulation of mineral deposits (long arrows) with an area of osseous metaplasia (arrowhead) bulging into the lumen. B, Large artery in the right ventricle of the heart (dose 50 mg/kg) with linear basophilic mineralization (arrows) of intima and inner parts of the media resulting in stiffening of the vascular wall. C, Alveolar ducts of the lung (dose 50 mg/kg) with thin layers of basophilic material (mineral deposits, arrows) along the elastic fibers which are located closely underneath the alveolar duct epithelium. In the middle upper part of the image, a mineral spicule is within an airway (arrowhead). The overall thickness of the alveolar walls is increased in this affected area, possibly also due to an adaptive increase of smooth musculature. D, Mineral deposits in the fundus mucosa of the stomach (dose 100 mg/kg) with a more linear to garland-like pattern of mineral deposits following the basement membranes/elastic fibers (arrowhead) or more extensive crumbly deposits located within the gastric acid producing parietal cells (arrows). E, Mineral deposits in a kidney (dose 100 mg/kg). Both tubular structures (arrows, intraluminal and intracellular) and the tunica media of a large artery (arrowhead) are affected. Often mineral deposits in the kidney are characterized by onion-like layers. F, Bone marrow of the tibia in the subphyseal area (dose 200 mg/kg). The original lamellar bone is characterized by its bluish cartilage core and regular parallel bands of collagen, whereas the centrally located woven bone (asterisks) lacks both features which makes it far less resilient.
Results of the follow-up study showed an increase in serum IP (group mean value about +30% to +50% vs controls) in all dose groups at all recording times. At urinalysis, a dose-related increase in diuresis (group mean value up to +23%) and in urinary calcium (group mean value up to 3-fold) and a dose-related decrease in urinary phosphorus (group mean value up to −35%) was seen on day 9. No relevant effects on urinalysis were detected on day 30. At ophthalmology investigation, 1 animal at 25 mg/kg presented with unilateral multifocal corneal spots. Toxicokinetic investigations showed comparable results as described for the DRF study, and at gross pathology, no relevant observations were made. Histopathology showed a pattern of soft tissue mineralization and woven bone formation as already described for the DRF study. A clear dose- and time-dependency regarding the number of involved organs and the severity of findings could be established with the used lower doses. The mineralization was first observed within the periosteum of the bone (minimal) at 24 hours after the first treatment with the highest dose (50 mg/kg). After the second dose, mineralization was seen in the aorta, bone, spinal meninges, and fundic mucosa of the stomach at 50 and 25 mg/kg. The latter was also found in 1 rat of the 10 mg/kg dose group. After the last dose, mineralization affected additionally the kidneys, lung, eyes, and heart, again with a clear dose-dependency regarding incidence and severity. Moreover, moderate to marked woven bone formation and fibrosis were found in single rats of each dose group with 1 rat at 50 mg/kg showing subphyseal fibrosis already on day 2.
Next, Dr Knippel described the possible mechanism for the observed metastatic mineralization. The fibroblast growth factor (FGF) family of ligands bind to FGF receptors (FGFRs). They are involved in a myriad of biological functions, including cell growth, differentiation, angiogenesis, embryonic development, wound healing, and metabolic regulation. Fibroblast growth factors are classified into the following 3 subgroups: (1) an autocrine/paracrine FGF subgroup consisting of FGF1-10, FGF16-18, FGF20, and FGF22; (2) an endocrine FGF subgroup consisting of FGF19, FGF21, and FGF23; and (3) an intracellular subgroup consisting of FGF11-14. Paracrine FGFs transduce signals through cell-surface FGFRs and heparan-sulfate proteoglycans, whereas endocrine FGFs transduce signals through cell-surface FGFRs and Klotho family proteins. FGFR1-4 are encoded by 4 genes, and alternative splicing (b and c isoforms of FGFR1-3) results in tissue and ligand binding specificity. FGF23, mainly synthesized from osteoblasts, is the physiological regulator of phosphate and vitamin D serum levels. The proximal tubules of the kidneys are the site of most renal phosphate reabsorption and 25(OH) vitamin D-1α-hydroxylase activity and they express FGFR1, FGFR3, and FGFR4 but not FGFR2. 54 FGF23 increases urinary phosphate excretion by decreasing renal brush-border expression of the sodium phosphate cotransporters 2a and 2c (NaPi-2a and NaPi2c) mainly by interaction with FGFR1. 54 In addition, FGF23 reduces serum 1,25(OH)2 vitamin D3 levels by decreasing the expression of 25(OH) vitamin D-1α-hydroxylase (CYP27B1) and increasing the expression of 24-hydroxylase (CYP24) 55 mainly by FGFR3 and FGFR4. 56 Taken together, FGF23 prevents both hyperphosphatemia and hypervitaminosis D. Hyperphosphatemia as seen in both FGF23- and α-klotho-deficient animals can promote senescence, ectopic calcification, and chronic kidney disease (CKD). 57 Dr Knippel emphasized that although FGF23 plays a central role in mineral homeostasis, the overall interactions are much more orchestrated with various factors involved. 58
The antagonistic activity of our monovalent FGFR1 binder likely led to decreased FGF23 signaling in the kidneys, thereby decreasing urinary phosphate excretion by increasing renal brush-border expression of the sodium phosphate cotransporters 2a and 2c (NaPi-2a and NaPi2c). As we did not measure 1,25(OH)2 vitamin D3 levels, any test article-related effect on this hormone remains speculative and, taking into account the high specificity of our antagonist, would seem unlikely as published data indicate that FGF23 triggers its effects on vitamin D metabolism via FGFR3 and FGFR4. 56
Hyperphosphatemia, in the presence of normo-or hypercalcemia, results in an increased calcium-phosphorous product, which is associated with ectopic tissue mineralization. Ectopic mineralization can be metastatic or dystrophic. Metastatic mineralization is defined as a systemic mineral imbalance associated with widespread ectopic mineralization, whereas dystrophic mineralization occurs in the absence of systemic mineral imbalance at sites of tissue alteration and/or necrosis. The classically held view that mineralization is a passive, degenerative process must be dispelled nowadays as it turns out to be actively regulated akin to bone mineralization. 59,60
Regardless of the primarily altered blood electrolyte(s) in rats (Ca, IP, or both), the resulting histomorphological picture remains the same and is in accordance with the pattern described above.
The presence of woven bone formation indicates that bone resorption exceeds bone formation rates. Accumulation of fibroblastic osteoprogenitors, which are not in the osteoblastic differentiation program, results in collagen deposition (fibrosis) in the peritrabecular and marrow space. 61 Multiple mechanisms are likely accountable for woven bone formation/fibrosis: (1) elevated phosphorus levels promote parathormone (PTH) secretion directly (independent of changes in serum calcium or calcitriol).When PTH is elevated, bone turns over with excessive rapidity. (2) As the FGF23 activity in the kidneys is partly blocked (at the FGFR1 receptor), an adaptive increase of FGF23 might be expected and FGF23 is an inhibitor of bone formation. (3) Finally, a sustained increase of phosphorus leads to ectopic mineralization with hydroxyapatite deposition for which the consumed calcium is provided from the bone reservoir.
Dr Knippel finally concluded on the possible relevance to humans. The described effects of FGF23 have both been confirmed by mouse knockout studies and by the reciprocal phenotypes of humans with 2 syndromes caused by mutations in FGF23: familial tumoral calcinosis with hyperphosphatemia (inactivating mutations in FGF23) and autosomal-dominant hypophosphatemic rickets (activating mutations in FGF23). The FGF signaling pathway (FGFR signaling) is an evolutionary conserved signaling cascade, 62 and therefore an effect of the investigated monovalent antibody in humans cannot be excluded. Interestingly, different susceptibilities of various toxicology animal species toward ectopic mineralization were detected during the preclinical development of this monovalent FGFR1 antagonist. Only rats (especially male rats) were affected. Mice developed hyperphosphatemia but no mineralization, and Cynomolgus monkeys neither showed hyperphosphatemia nor mineralization. It is likely that variably efficient compensatory mechanisms are accountable for these species differences. There might also be a connection to the chronic progressive nephropathy syndrome in male rats which possibly enhances the inorganic phosphate imbalance/inefficient compensation, although we were not able to find more detailed information/publications supporting this hypothesis. Despite the apparent species specificity for mineralization and/or hyperphosphatemia, it is not appropriate to discount the risks toward humans, especially as hyperphosphatemia is the most commonly reported adverse finding in clinical studies with pan-FGFR inhibitors; and uncertainty remains regarding the relative contribution of individual FGFR subtypes leading to hyperphosphatemia in men. 63 Hyperphosphatemia in men has also been linked to vascular calcification and in CKD, it contributes to the high rates of mortality. 64
Kidney Lesions Associated With mTOR Inhibitor in Combination Therapy
Johannes Zellmer (Department of Nuclear Medicine, Ludwig Maximilians University, Munich, Germany) and Dr Hsi-Yu Yen (Comparative Experimental Pathology, Institute of Pathology, Technical University of Munich, Germany) presented kidney lesions of Lewis rats from a 16-week repeated dose oral gavage toxicity study of a combined treatment with the mammalian target of rapamycin (mTOR) inhibitor everolimus and [177Lu-DOTA0, TYR 3 ]-octreotate (177Lu-DOTATATE) in Lewis rats.
Everolimus is a signal transduction inhibitor targeting mTOR or, more specifically, mammalian “target of rapamycin” complex 1 (mTORC1). Mammalian target of rapamycin is a key serine-threonine kinase which plays a central role in the regulation of cell growth, proliferation, and survival. 65 The regulation of mTORC1 signaling is complex, being modulated by mitogens, growth factors, energy, and nutrient availability. 66 Everolimus binds to its protein receptor FKBP12 (12-kDa FK506-binding protein), which is an ubiquitous and abundant protein that acts as a receptor for the immunosuppressant drug FK506, binds tightly to intracellular calcium release channels and to the transforming growth factor β type I receptor and interacts directly with mTORC1, inhibiting its downstream signaling. The resulting complex prevents mTOR activity, leading to inhibition of cell cycle progression, survival, and angiogenesis. 67 As a consequence, tumor growth is inhibited.
DOTATATE is a compound containing tyrosine 3 -octreotate, a somatostatin receptor (SSTR) agonist, and the bifunctional chelator DOTA (tetraxetan). Somatostatin receptors are found with high density in numerous malignancies, including central nervous system, breast, lung, and lymphatic tumors. 68 The use of SSTR agonists (ie, somatostatin and its analogs such as octreotide) in treatment of neuroendocrine tumors (NETs) is well established. 69 Most NETs are characterized by overexpression of SSTR, mainly subtype 2. Targeting these receptors by administration of somatostatin analogs radiolabeled with 177Lu (β particle-emitting radionuclide) allows peptide receptor radionuclide therapy (PRRT) of patients with NET. 70
Everolimus is known to increase the sensitivity of solid tumor to external radiotherapy. Therefore, an additive or even over-additive therapeutic effect of everolimus combined with 177Lu-DOTATATE is expected due to their mechanisms of action. However, both compounds adversely affect the kidneys and the hematopoietic system. It was the aim of the present study to investigate the associated nephrotoxicity of this combination therapy.
This study involved 62 female Lewis rats, divided into 4 groups of approximately 16 animals each. Group 1 received 5% glucose solution, ∼0.5 mL/animal weekly; group 2 received 5 mg/kg body weight everolimus once weekly; group 3 received the combination of 5% glucose solution, ∼0.5 mL/animal once weekly and 200 MBq 177Lu-DOTATATE as a single dose 112 days before necropsy; and group 4 received the combination of 5 mg/kg body weight everolimus once weekly and 200 MBq 177Lu-DOTATATE as a single dose 112 days before necropsy.
The rats belonging to each group were again divided and used in one of the 2 parts of the study. In the first part, blood levels of creatinine and urea were assessed weekly to monitor nephrotoxicity. In the second part of the study, the renal function was analyzed by sequential 99mTc-Mercaptoacetyltriglycine (MAG3) scintigraphs. MAG3 is a compound almost exclusively secreted by the proximal renal tubules and commonly used in routine clinical scintigraphic measurements of renal function. As a measure of renal clearance, the fractional uptake rate (FUR) of MAG3 was calculated from its activity in the kidneys and the whole body. The renal clearance of MAG3 over time was visualized as relative Tc activity in the renal compartment over time.
Both kidneys of 32 animals (4 animals per group from the first part and 4 animals per group from the second part) were preserved at necropsy and further processed for histopathological examination.
Mr Zellmer presented the in-life findings including renal serum parameters and renal function testing: All rats survived until scheduled necropsy. Groups that received everolimus showed a slower gain in weight than the vehicle group. No significant differences in serum urea level were found between the groups. The increase of creatinine level was significantly lower in rats that received everolimus (P = .023). No significant differences were found for 177Lu-DOTATATE (P = .185) or the combination of everolimus and 177Lu-DOTATATE (P = .308).
In the scintigraphy, the MAG3 clearance of group 1 (vehicle) was almost unchanged compared to baseline. The initial slope and late excretion of group 2 (everolimus) was also comparable to baseline, whereas the peak was slightly higher (P = .063) and delayed (P = .621). The initial slope of both PRRT groups (group 3, vehicle + 177Lu-DOTATATE and group 4, everolimus + 177Lu-DOTATATE) was less steep compared to baseline and to groups 1 and 2. This was reflected by significantly lower FUR values on day 112 (P = .003 for group 3 and P = .002 for group 4 vs baseline). Compared to vehicle, the administration of everolimus induced a later and higher peak, as already demonstrated between groups 1 and 2. The late excretion appeared to be preserved (Figure 7A).
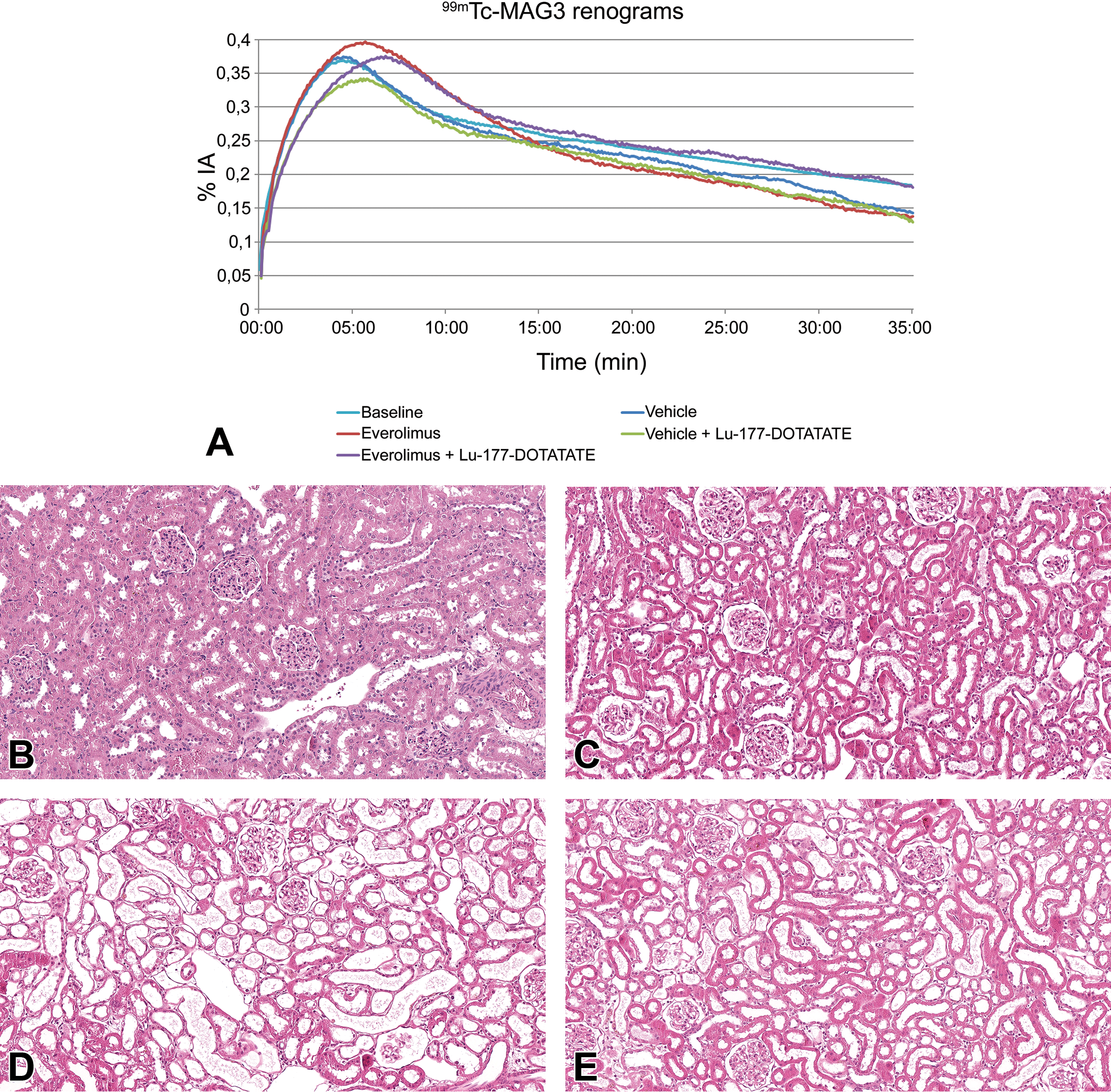
A, Renograms at follow-up examination 16 weeks after the beginning of each treatment. The baseline renogram was extrapolated to 35 minutes using a monoexponential fit of the excretion phase. For clarity, error bars are not shown. Reduced steepness of the initial slope reflects impairment of the renal function after PRRT treatment. The constant decrease in the excretion phase shows the transfer of the tracer to the urinary bladder. Its absolute value depends on renal plasma flow rather than renal function. B-E, Kidney lesions from female Lewis rats of a 16-week toxicity study of a combined treatment with the mTOR-inhibitor everolimus and [177Lu-DOTA0, TYR3]-octreotate. B, Minimal tubular dilatation in the vehicle control was potentially caused by perfusion at necropsy. C, Everolimus at a dose of 5 mg/kg once weekly caused tubular dilatation and loss of tubular epithelium. Similar findings were induced by a single dose of 177Lu-DOTATATE 200 mBq (D) or the combination of everolimus 5 mg/kg once weekly and a single dose of 177Lu-DOTATATE 200 mBq (E). mTOR indicates mammalian target of rapamycin; PRRT, peptide receptor radionuclide therapy
Dr Yen presented the lesions in the kidneys. Histopathologically, multifocal atrophy of tubular epithelium and tubular dilatation were observed in all animals. Minimal lesions were detected in the tubules in the vehicle group, whereas more prominent lesions were found in all test article groups (Figure 7B-E). Additionally, minimal to slight multifocal mononuclear inflammation was detected in almost all animals in group 3 (vehicle + 177Lu-DOTATATE) and in group 4 (everolimus + 177Lu-DOTATATE).
After the case was presented, Mr Zellmer and Dr Yen discussed the possible causes of the minimal kidney lesions detected in the vehicle group. The repeated and/or long anesthesia during the blood samplings and scintigraphs as well as the preliminary perfusion at necropsy were considered as the potential causes.
In summary, the study shows the potential toxicity of a combined treatment with the mTOR-inhibitor everolimus and 177Lu-DOTATATE, especially serious side effects like nephrotoxicity, using a preclinical rat model. In the context of the present study, the combined treatment of rats with a therapeutic dose of everolimus and 177Lu-DOTATATE did not result in additive renal toxicity.
Unexpected Intolerability of Mutation-Specific Drugs
In his second presentation, Dr Florian Colbatzky (Boehringer Ingelheim Pharma GmbH & Co. KG, Biberach [Riß], Germany) gave insight into effects induced by 3 compounds, which have been designed to be rather specific for certain mutations of their target molecules.
The first case was from 2-week exploratory toxicity studies in male mice and male rats exposed to a fourth generation epithelial growth factor receptor (EGFR) inhibitor specific for mutated variants of the receptor (del19, del19 T790M, del19 C797S). In these 2 exploratory toxicity studies as well as in those of the 2 other cases, 8 male animals were used per dose group. The dose levels were intended to induce toxicity starting with minimal adverse effects at the low dose and increasing up to dose-limiting toxicity at the high dose. The animals had to be killed within the first 2 to 7 days of the studies because of severe body weight loss and no food consumption. Study parameters did not show any changes, which would provide evidence for the low tolerability of the compound. There were no histopathological changes. Toxicogenomics analyses also did not reveal alterations of gene expression, which could explain the intolerability of the compound.
The second case was from a 2-week exploratory toxicity study in male rats, which were exposed to graduated daily dose levels of 0, 40, 130, and 420 mg/kg of a compound targeting HER2 exon 20 insertion oncoproteins. The WT EGFR is almost completely spared by the compound. The animals did not show pertinent clinical signs of ill-being apart from reduced spontaneous activity, closure of eyelids, and diarrhea in the high-dose group.
At the dose level of 420 mg/kg, body weight gain and absolute and relative organ weights of the testes, the prostate, and the thymus were reduced. Clinical chemistry showed mild increases in total bilirubin and decreases in total protein, creatinine, glucose, and chloride. At necropsy, gross abnormalities were recorded for the high-dose males: The cecum contained brown fluid, the thymus, the prostate, the seminal vesicles, the testes, and the epididymides were all decreased in size.
Histopathological findings were mainly noted in the males given 420 mg/kg. In these animals, apoptoses and vacuolization of acinar cells of the pancreas (not shown) were present. In the stomach, hyperkeratosis of the epithelium of the nonglandular mucosa (not shown), in some animals accompanied by the formation of erosions and ulcers, was noted. Increased apoptoses of thymocytes/lymphocytes and marked cortical cellular depletion were observed in the thymus (Figure 8A). There was also cellular depletion of the marginal zones and, less pronounced, of lymph follicles as well as accumulation of foamy macrophages in the adjacent red pulp of the spleen (Figure 8B) and increased accumulation of foamy macrophages in lymph nodes (Figure 8C). Beginning testicular atrophy due to degeneration and loss of spermatocytes and spermatids was present in the testes (Figure 8D). It was accompanied by vacuolization of Sertoli cells and presence of cell debris in the ducts of the epididymides (Figure 8E). The secretory activity of the prostate and seminal vesicles was reduced (not shown).
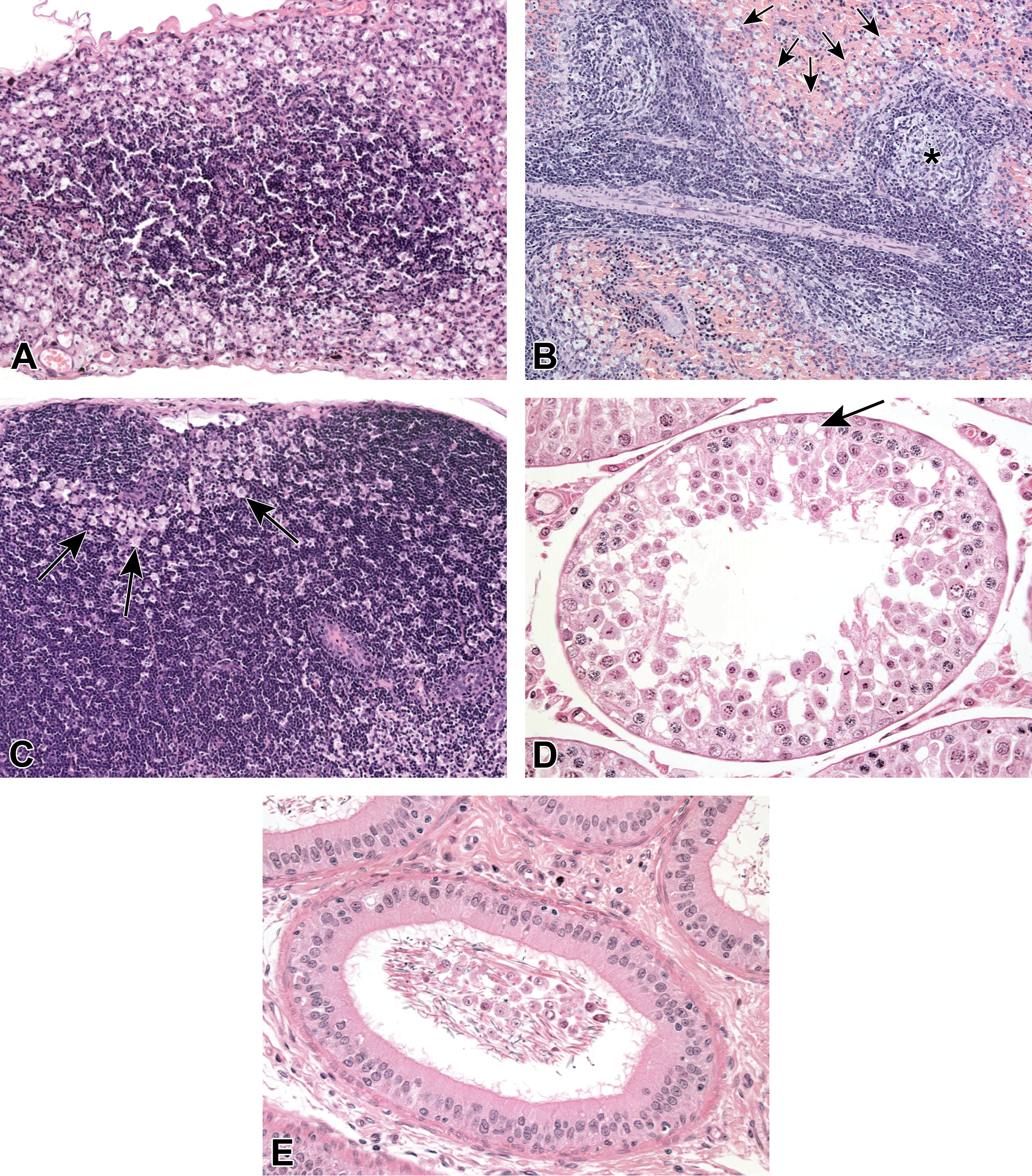
A-C, Examples of lesions in the thymus, spleen, and mesenteric lymph node of a male rat treated with 420 mg/kg of compound B. A, Marked cellular depletion in the thymus with accumulation of foamy macrophages in the cortex and an increased number of apoptotic thymocytes in the medulla. B, Cellular depletion of splenic marginal zone (arrows) and follicles (asterisk) with an accumulation of foamy macrophages. C, Foamy macrophages within the cortical region of a mesenteric lymph node. D and E, Examples of lesions in the testes and epididymides of a male rat treated with 420 mg/kg of compound B. D, Degeneration and loss of spermatocytes and spermatids in the testes. Occasional vacuolization of Sertoli cells (arrow). E, Cell debris in the lumen of an epididymal duct.
Toxicogenomics analyses of liver samples collected at necropsy showed prominently altered expression of a variety of genes related to testicular function and steroid hormone biosynthesis such as androgen receptor, estrogen receptor 1, 17β-hydroxysteroid dehydrogenase (17β-HSD5), sulfotransferase family 1E member 1, and inhibin. In general, expression of genes related to the synthesis and secretion of estrogen was upregulated, whereas expression of genes related to synthesis and secretion of androgens was downregulated.
The third case was from a 2-week exploratory toxicity study in male mice given an inhibitor of a mutated variant of KRAS showing a substitution of glycine 12 by cysteine 12 (G12C). The animals were given graduated daily dose levels of 0, 50, 150, and 450 mg/kg. Pertinent clinical signs were severe bloating of the gastrointestinal tract resulting in premature decedents of animals of the mid- and high-dose group in the first 7 days of the study. Surviving animals showed mild dose-dependent increases in aspartate transferase, alanine transferase, and potassium as well as decreases in white blood cell and lymphocyte counts. Due to gastric reflux, minor inflammatory processes were noted in the mucosa of the esophagus and the larynx. The mild cellular depletion of the thymus of high-dose animals was considered stress related.
Next, Dr Colbatzky discussed possible mechanisms of pathogenesis for the outcomes of the 4 exploratory toxicity studies. Despite substantial additional efforts, it was not possible to elucidate the pathomechanism of the low tolerability of the selective fourth generation EGFR inhibitor (first case).
For the second case with the selective HER2 exon 20 inhibitor, alterations of gene expression, which would help to understand the histopathological changes in lymphatic organs (thymus, lymph nodes, and spleen) were not detected. But the changes in the male reproductive organs may be explained by the consistent alterations of expression genes related to the synthesis, secretion, and function of androgens as well as estrogens. Connections/interactions between Her2 (=ErbB2) signaling and androgen receptor expression have been described for humans. 71 The involvement of genes such as estrogen- and androgen-converting enzymes 17β-HSD5 in cancer types such as breast cancer is well known. 72 It is unclear, however, whether these would result in effects on the therapeutic outcomes of treatment of sexual hormone-sensitive cancer types such as breast and prostate cancers in general or only interfere with sexual hormone-dependent gene signatures.
Like the first case, the mechanism of action for the severe clinical signs of gastrointestinal bloating was not elucidated for the third case with the highly selective KRAS G12C inhibitor. Especially, there is no evidence, based on its inhibitory profile (including the Cerep and PanLabs screens), on histopathology and on toxicogenomics analyses, for compound-related effects on the interstitial cells of Cajal.
In summary, mutation-specific drugs may show unexpected intolerability, due to off-target effects, even when careful profiling of these compounds has been conducted.
Footnotes
Acknowledgments
The authors acknowledge image editing by Beth Mahler (EPL) and organization and formatting work by Elisabeth Schmid (Boehringer Ingelheim Pharma GmbH & Co. KG). Key to the success of the seminar was the local organization by Lukas Mathias Michaely and his colleagues from the Institute of Pathology of the University of Veterinary Medicine, Hannover, Germany. Dr Odin (drug-induced small intestine pathology) acknowledged the key contributions of Dr Gitte Jeppesen (Pathologist, CRL, Denmark), Isabelle Wells (BioInformatician), Solveig Badillo (Statistician), Marion Richardson (Molecular Pathology Scientist), and Nicolas Giroud (Genetics & Genomic Scientist). Dr Nehrbass (AO Research Institute [ARI], Davos, Switzerland) acknowledged scientific contributions to the investigations he presented (device-induced growth plate lesions) by Daniel Cheney (R&D DePuy Synthes, West Chester, Pennsylvania), Dominic Gehweiler (ARI, Davos, Switzerland), Jim Hearn* (Royal Hospital for Sick Children, Bristol, United Kingdom) Maria Hildebrand (ARI, Davos, Switzerland), John Mukhopadhaya* (Paras Hospital, Patna, India), Unni Narayanan* (Hospital for Sick Children, Toronto, Canada), Matias Sepulveda-Oviedo* (Universidad Austral de Chile, Valdivia, Chile), Theddy Slongo* (University Children’s Hospital, Bern, CH), Stephan Zeiter (ARI, Davos, Switzerland), Boyko Gueorguiev (ARI, Davos, Switzerland), Jonathan S.M. Dwyer* (University Hospital of North Staffordshire, Stoke-on-Trent, United Kingdom). Members of the AO Technical Commission Pediatric Expert Group, Davos, Switzerland, are marked above by *.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The case of drug-induced small intestine pathology (Dr. Odin) was presented as a poster at the 2019 STP meeting in Raleigh, NC. The investigations presented by Dr Nehrbass were the result of a cooperation of AO Research Institute Davos (ARI), AO Technical Commission (AOTC), and DePuySynthes. The other authors declared no potential, real or perceived conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
