Abstract
Fetal examinations in embryo-fetal developmental (EFD) studies are based on macroscopic and dissecting microscopic evaluations, and histopathology is rarely performed other than to confirm macroscopic findings. Fetal lens examination is therefore generally limited to the presence, size, shape, and color of any abnormality. In a Sprague-Dawley rat EFD study with the fatty acid amide hydrolase (FAAH) inhibitor JNJ-42165279, an unusually high incidence of macroscopic granular foci was noted within the lens of gestation day 21 fetuses across all groups including controls, with higher incidence in the high-dose group. On histological evaluation of the lenses from fetuses with/without gross findings, primary lens fiber hypertrophy (swelling) and degeneration were observed across vehicle- and JNJ-42165279-exposed fetuses. In a follow-up study to investigate the progression or resolution of the fetal lens changes, animals exposed to suprapharmacological doses of JNJ-42165279 in utero had higher incidence of nuclear cataracts as detected via slit-lamp ophthalmic examinations on postnatal days 18 to 21 and 35 to 41. No histologic correlates for these cataracts were identified. We conclude that fetal primary lens fiber hypertrophy and nuclear cataracts at ophthalmology, are common background changes in this rat strain that are exacerbated by in utero exposure to the FAAH inhibitor JNJ-42165279.
Keywords
Introduction
Embryo-fetal development (EFD) studies are routinely performed during safety assessments to evaluate the effect of test compounds on organogenesis in both rodent and nonrodent models (usually rats and rabbits). In a typical study design, the test compound is therefore dosed during the period of organogenesis: from gestation day (GD) 6 to GD17 in rats and from GD6 to GD19 in rabbits. One of the structures developing during organogenesis is the lens of the eye, which is formed on GD12 in both rats and rabbits. On GD13, the lens vesicle detaches from the optic cup and the posterior lens vesicle cells elongate to form primary lens fiber cells leading to narrowing of the vesicle lumen. By GD14, the lens vesicle lumen has disappeared, and the primary lens fibers have contacted the anterior lens vesicle cells which form the lens epithelium. These primary fibers undergo organelle degradation, including nuclear loss, to become transparent. After proliferation of anterior epithelial cells in the germinative layer above the lens equator (nuclear bow), the daughter cells migrate outward into the equatorial region, where they differentiate into secondary fiber cells, which form the cortex. 1,2
Continuous proliferation and differentiation of secondary fiber cells result in lens growth by the formation of multiple layers in an onion-like morphology and this process occurs throughout life, although at a slower rate in the adult. 1,3 In humans, the entire prenatal lens (mainly composed of the lens nucleus) has diameter of about 6.5 mm at birth and reduces in size over the years due to compaction by the secondary lens fibers. 3 –5
JNJ-42165279 is a potent, highly selective, and orally bioavailable inhibitor of fatty acid amide hydrolase (FAAH), 6 which is the main degrading enzyme for the endocannabinoid anandamide. Inhibition of FAAH results in accumulation of the anandamide substrate. The estimated half-maximal inhibitory concentration (IC50) for rat FAAH is 313 nM (128 ng/mL). 7
To assess the potential toxic effects of orally administered JNJ-42165279 on maternal and embryo-fetal parameters, an EFD study (study 1) was conducted in pregnant Sprague-Dawley rats. Histopathological evaluation of fetal lenses was included to address fetal lens gross observations. Fetal examinations in EFD studies are based on macroscopic and dissecting microscopic evaluations, and histopathology is rarely performed other than to confirm a macroscopic finding. Fetal lens examination is therefore generally limited to the presence, size, shape, and color of any abnormality.
At histology, primary lens fiber hypertrophy (swelling) and degeneration were observed in the lens nucleus. Hypertrophic lens fibers can vary from scattered individual fiber profiles that are increasing in size, to numerous, enlarged fusiform cells. Swollen lens fibers may not produce opacities identifiable on ophthalmic examination and may potentially be reversible. Degeneration results from biochemical changes that alter the homeostasis of the lens, denature, and coagulate the crystalline proteins of the lens and/or disrupt the integrity of the lens fiber. 8 Irreversible microscopic changes that likely correlate with lens opacity include bladder cells (enlarged fusiform cells with foamy cytoplasm), clefts, vacuoles, liquefaction, lens fiber fragmentation, Morgagnian globules, collapse of the lens capsule, and dystrophic mineralization. 9 A subsequent study (study 2) was designed to allow a cohort of pregnant animals dosed with JNJ-42165279 during the period of organogenesis to deliver, and the juvenile offspring were evaluated for evidence of cataracts (opacities) at ophthalmic examination.
Materials and Methods
All experiments were carried out in the Janssen Research & Development, Beerse test facility and were compliant with the current Good Laboratory Practice (GLP) principles of the Organization for Economic Co-operation and Development (OECD) GLP guidelines. 10 The study design was in accordance with the ICH Harmonized Tripartite Guideline for Detection of Toxicity to Reproduction for Medicinal Products. 11 Outsourcing and monitoring of toxicokinetics were performed according to the OECD principles of GLP to the organization and management of multisite studies. The test facility was approved by the Association for Assessment and Accreditation of Laboratory Animal Care International. All animals were treated humanely and cared for in accordance with the European 12 and Belgian guidelines 13 and with the principles of euthanasia as stated in the Report of the American Veterinary Medical Association Panel. 14 The studies were performed in accordance with Janssen Research & Development ethical protocols, which were approved by the local ethical committee.
Specific pathogen free Sprague-Dawley (Crl: CD) female, young and healthy virgin rats from the same breeding facility at Charles River, Sulzfeld, Germany, were used for all studies. Stock males of the same strain and supplier were used for mating only. The animals were group housed in polysulfone cages with floor area of 800 cm 2 and Corn Cob (size 12, Eurocob) was provided as bedding material. Environmental enrichment consisted of a rodent retreat (Bio-Serv) and aspen wood blocks (Datesand). The animal room was air-conditioned (own supply of filtered fresh air) and had a 12-hour light cycle (300 Lux at 1 m height). The animals were fed ad libitum with irradiated R/M-H pelleted maintenance diet from Ssniff.
JNJ-42165279 was formulated as an aqueous suspension containing 0.5% (wt/vol) MethocelTM and was stored at room temperature in a closed and labelled container protected from (white) light. The formulation or the vehicle (0.5% [wt/vol] MethocelTM) was administered once daily by oral gavage at a volume of 10 mL/kg body weight.
Study 1: Design of EFD Study in Rats
JNJ-42165279 was administered orally to pregnant Sprague-Dawley rats (n = 22, main study group and n = 3, toxicokinetic group) during the period of organogenesis (from GD6 to GD17, inclusive) at daily doses of 10, 30, or 100 mg/kg. Females were observed at least once a day for body weight, food consumption, illness, abnormal behavior or unusual appearance, unexpected clinical effects, toxic or pharmacological response, moribund state, or mortality for all dose levels. Females were anaesthetized by inhalation of an isoflurane/oxygen mixture and killed on GD21 by exsanguination via the carotid artery. During necropsy, the uterus and ovaries were dissected out, weighed, and examined for numbers of corpora lutea of pregnancy, implantations, resorptions, live fetuses, and intrauterine death. The fetuses were weighed, sexed, and examined for external, visceral, and skeletal abnormalities. Fetuses were kept in Bouin's fixative for 2 weeks, after 2 weeks they were transferred to denatured alcohol (2% industrial methylated spirit). Visceral examination was performed on fetuses fixed in Bouin's for 2 weeks, while skeletal examination was performed after staining with Alizarin red-S. For visceral examination by Wilson technique, 15 free hand serial sections of Bouin-fixed fetuses, including the heads, were examined using a dissecting microscope. After visceral examination, the head sections containing the eyes were processed to paraffin blocks, and sections were stained with hematoxylin and eosin (H&E) for histopathological evaluation of the eyes. The microscopic findings were graded (1: minimal histological change, 2: mild, 3: moderate, 4: marked, and 5: severe/massive histological change) per eye. Toxicokinetics were studied in additional satellite rats (groups of 3 pregnant females) at 0.5, 1, 2, 4, 7, and 24 hours postdose on GD6 and GD16 by using a validated liquid chromatography–tandem mass spectrometry (LC-MS/MS) method. Additionally, blood samples were collected from all toxicokinetic rats in the vehicle group at 0.5, 7, and 24 hours postdose.
Study 2: Design of EFD Study in Rats With Postpartum Recovery
JNJ-42165279 was administered daily to 3 groups of 44 females (F0) each during the period of organogenesis (from GD6 to GD17, inclusive) at doses of 10, 30, or 100 mg/kg; and the vehicle control group received 0.5% (wt/vol) MethocelTM. Half of the females (22/group; 88 total) were allocated to the EFD phase, euthanized on GD21, and necropsied to assess pregnancy parameters. Since histological evaluation was determined to be more sensitive than gross examination in study 1, the heads/eyes of study 2 fetuses and pups were not examined with the dissecting microscope. All live fetuses were decapitated, heads placed in Bouin's fixative, processed to paraffin blocks, and sections stained with H&E for histopathological evaluation of the eyes. The remaining females (22/group; 88 total) were allocated to the postnatal phase and allowed to litter and raise their offspring (F1 generation) to weaning. Ophthalmic examination was performed on these pups between postnatal days (PND) 18 to 21 and again between PND 35 and PND 41 after induction of mydriasis with 0.5% tropicamide eye drops. The eyes were examined by the same veterinary ophthalmologist with a slit-lamp biomicroscope and by indirect ophthalmic examination using a focused source of light and an aspherical 28 D lens. Following completion of the ophthalmic examinations, the animals were euthanized, the eyes fixed in Davidson's fixative, sectioned (12 levels were processed and examined for eyes with cataract or prominent lens nucleus and 6 levels for lenses without cataract/prominent lens nucleus at ophthalmic examination), and stained with H&E for histopathological evaluation. The microscopic findings were graded (1: minimal histological change, 2: mild, 3: moderate, 4: marked, and 5: severe/massive histological change) per eye. Toxicokinetics were studied in additional satellite rats (groups of 3 pregnant females) at 0.5, 1, 2, 4, 7, and 24 hours postdose on GD6 and GD16 by using a validated LC-MS/MS method. Ophthalmological evaluations were performed predose for the 44 maternal females/group as well as for the untreated paternal males.
Study 3: Tissue Distribution of JNJ-42165279 in Pregnant Rats
In this study, pregnant Sprague-Dawley rats were dosed orally with 30 mg/kg JNJ-42165279 daily for 5 days, from GD15 to GD19. On the last day of dosing, a mixture of JNJ-42165279 and 14 C-JNJ-42165279 was administered. Dam blood, amniotic fluid, fetal eye, fetal blood, and whole fetuses were collected at 0.5, 2, 7, and 24 hours after the last dose, on GD19. Blood samples of the dams were taken from a carotid artery under isoflurane anesthesia at necropsy. Amniotic fluid was collected and pooled per dam. Fetal blood samples were taken from the carotid artery. Fetal eyes were collected and pooled per dam. Remaining whole fetus was collected individually. Within 1 hour of sampling, the blood samples were centrifuged and within 1 hour after the start of centrifugation, plasma was stored in a freezer. Tissues and amniotic fluid were stored on dry ice until storage in a freezer.
JNJ-42165279 was measured in plasma by high performance liquid chromatography–tandem mass spectrometry, and total radioactivity in amniotic fluid, whole fetus, pooled eyes, and individual plasma by liquid scintillation counting.
Mean plasma and tissue concentration–time profiles were subjected to a pharmacokinetic analysis using validated pharmacokinetic Automated Workflow R1.0 software. A noncompartmental analysis using the linear up/log down trapezoidal rule was used for all data.
Statistical Analysis
Incidences of fetal abnormalities are always evaluated on a litter basis in reproduction studies as this is taken as the experimental unit. This removes some of the bias that could be created due to genetic factors and individuals within a single litter are more likely to respond in a similar way to any insult.
In studies 1 and 2, for pairwise comparison of the JNJ-42165279 dosage group with the vehicle control group, the following statistical methods were used: Fisher exact probability test (1-tailed) for clinical observations, pregnancy rate, incidences of fetal abnormalities including histopathology, mortality rates, and ophthalmic examinations. Mann-Whitney U test (2-tailed, correction for ties) was used for body weight gain and corrected maternal weight gain, food consumption, weight of the gravid uterus, number of live and dead fetuses, mean litter size, number of resorptions (early, late, and total), number of implantations, number of corpora lutea of pregnancy, pre- and postimplantation loss, and body weight and sex ratio of live fetuses. The P value <.05 was considered significant.
Results
Study 1
Maternal and fetal safety outcomes
There were no adverse maternal effects of JNJ-42165279 treatment at 10 mg/kg dose. A reduction in body weight and body weight gain (P < .01) was observed during the first few days of the treatment period in the group receiving 30 mg/kg dose compared to the vehicle control group, while at 100 mg/kg dose, there was an initial slight loss in body weight followed by a reduced body weight gain (P < .01) throughout the treatment period. The corrected mean maternal weight gain at 30 and 100 mg/kg was lower than that of the vehicle controls. These changes correlated with reductions in food intake.
At terminal necropsy, 20 females were confirmed pregnant in the vehicle controls and 22, 21, and 22 females were pregnant in groups receiving 10, 30, or 100 mg/kg JNJ-42165279, respectively. Fetal survival and growth in utero were unaffected by treatment with JNJ-42165279 at 10 and 30 mg/kg doses. A slightly lower fetal weight at 100 mg/kg was associated with slight increases in the incidence of observations indicative of delayed fetal development, such as incomplete descent of the thymus and an increase in the incidence of parameters associated with the extent of fetal ossification. These findings were considered to be a consequence of lower maternal body weight gain during pregnancy, maternally mediated, transitory, and not a direct JNJ-42165279-related adverse effect on fetal development.
At free-hand serial sectioning of the head (Wilson's technique 15 ), a dose related increase in the incidence of granular foci (referring to the granular texture of these foci) in the lens, was observed as a single focus per lens in all groups (Figure 1). The incidence of litters containing one or more fetuses exhibiting this finding was 10% in vehicle control and 9.1%, 19.0%, and 68.2% in the 10, 30, and 100 mg/kg dose groups, respectively (Table 1). To further understand this gross finding, histological assessment of the affected lenses was conducted. Other pregnancy and fetal parameters assessed were unaffected.
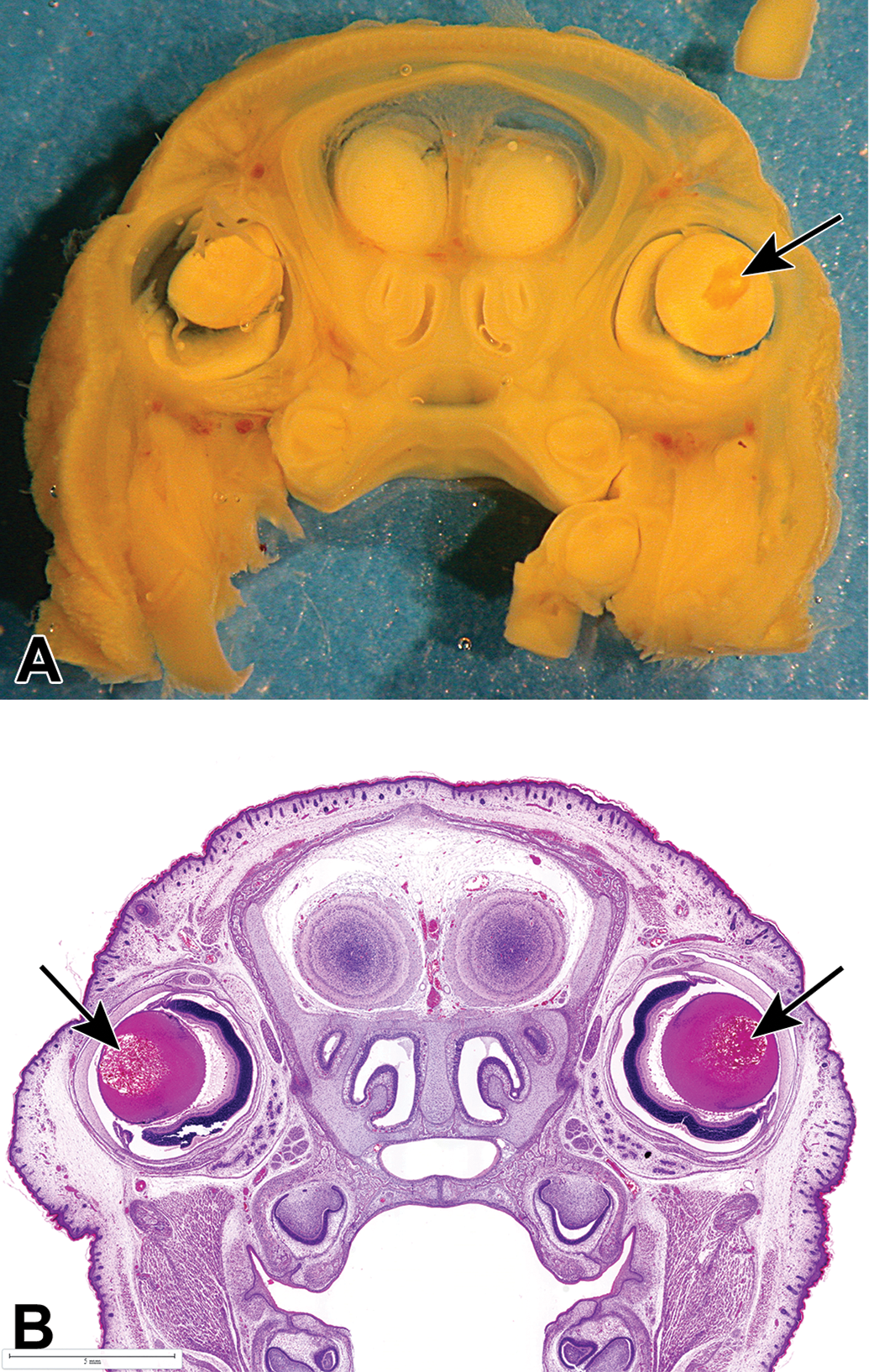
Head section of Bouin's fixed GD21 fetuses from dams administered 100 mg/kg/d. Study 1: A, Unilateral gross (granular) focus in right fetal lens (arrow). B, Bilateral lens changes (arrows) at histology (H&E). GD indicates gestation day; H&E, hematoxylin and eosin.
Incidence of Gross Granular Foci in Fetal Lens: Study 1.
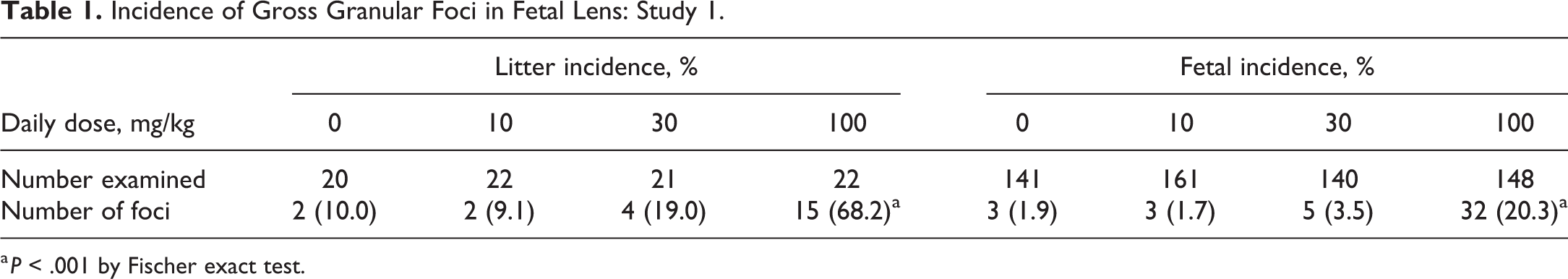
a P < .001 by Fischer exact test.
Histological examination
The lens is composed of 2 cell types: a single layer of cuboidal anterior lens epithelium with lens capsule and the primary and secondary lens fibers. In the fetal lens (GD21), the majority of lens fibers were the primary lens fibers (lens nucleus), with only a small outer rim of secondary lens fibers indicated by the equatorial nuclear bow. The tunica vasculosa lentis, the vascular supply of the developing lens, still surrounds the fetal lens during pregnancy (Figure 2) and undergoes apoptosis from PND 18 to 21 and even for weeks after birth. 16,17
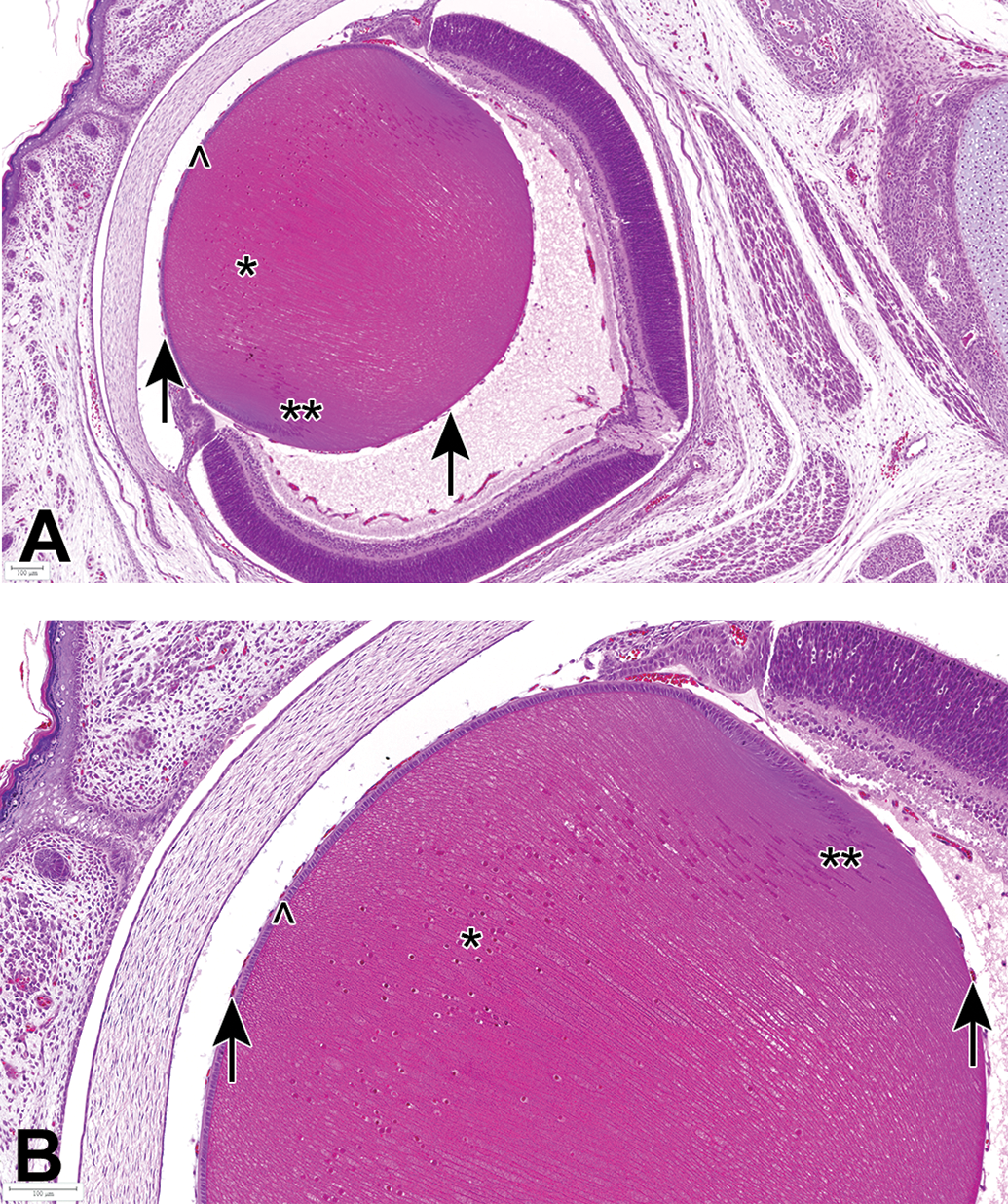
Eye from a control fetus (GD21). A, Overview and B, higher magnification demonstrate histology of the normal fetal lens, which is mainly composed of the primary lens fibers (nucleus). Primary lens fibers (*) are not yet denucleated, indicating that their final differentiation process is still ongoing. Secondary lens fibers start to develop at the equatorial region (**, formation of nuclear bow). A single layer of cuboidal lens epithelium is present below the lens capsule (^). Arrows indicate the tunica vasculosa lentis, the vascular supply of the developing lens. Note the high cellularity of the cornea and the presence of fused eyelids. Bouin's fixation, H&E stain. GD indicates gestation day; H&E, hematoxylin and eosin.
Morphological changes in fetal (GD21) lenses of vehicle- and JNJ-42165279-treated rats were mainly noted in the anterior part of the lens nucleus. The main features observed at 100 mg/kg were moderate hypertrophy (swelling) of primary lens fibers with or without degeneration, fragmentation of primary lens fibers, and occasional formation of fluid-filled clefts (liquefaction; Figure 3A and B), which correlated to the granular foci noted at visceral evaluation of fetuses. The lens epithelium showed focal degenerative changes (apoptosis, flattening, and loss of epithelial cells) and subcapsular proliferation of epithelial cells (epithelial hyperplasia) adjacent to the degenerative lens fibers (Figure 3C and D). The secondary lens fibers were unaffected. Focal degeneration and hypertrophy of primary lens fibers were also noted, although of lower severity, in vehicle-treated rats at the same location (Figure 4).
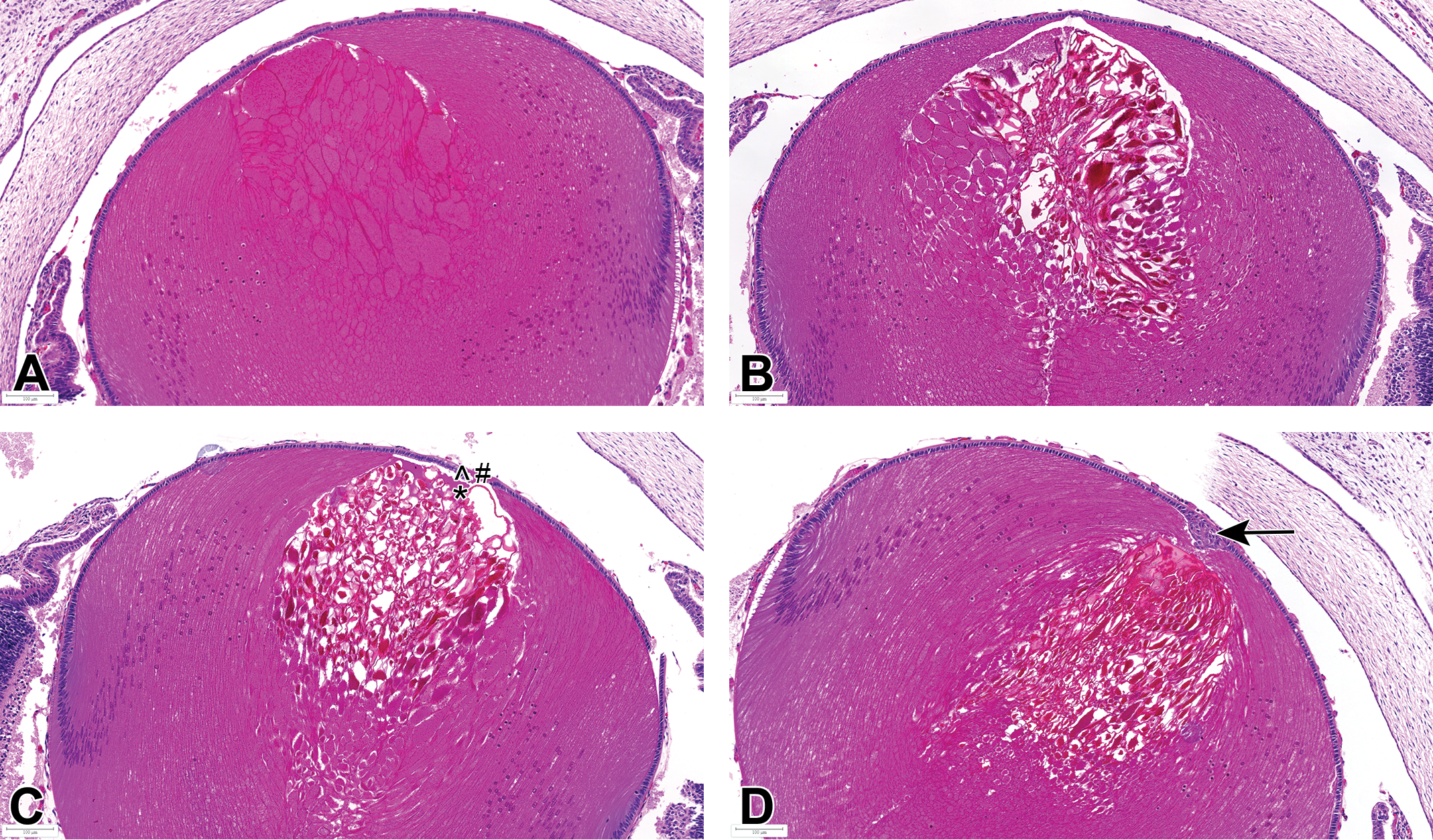
Representative lens findings in eyes of GD21 fetuses from dams administered 100 mg/kg/d. Study 1: A, Mild to moderate hypertrophy of anterior primary lens fibers. B, Hypertrophy of anterior primary lens fibers with degeneration and fragmentation. C, Degeneration and fragmentation of anterior primary lens fibers, with liquefaction (*, replacement of fibers by proteinaceous fluid) that extends to the anterior epithelium and secondary degenerative changes (^, apoptosis and #, loss) of lens epithelium. D, Degeneration and fragmentation of anterior primary lens fibers, with focal hyperplasia of subcapsular epithelial cells (arrow). Bouin's fixation, H&E. GD indicates gestation day; H&E, hematoxylin and eosin.
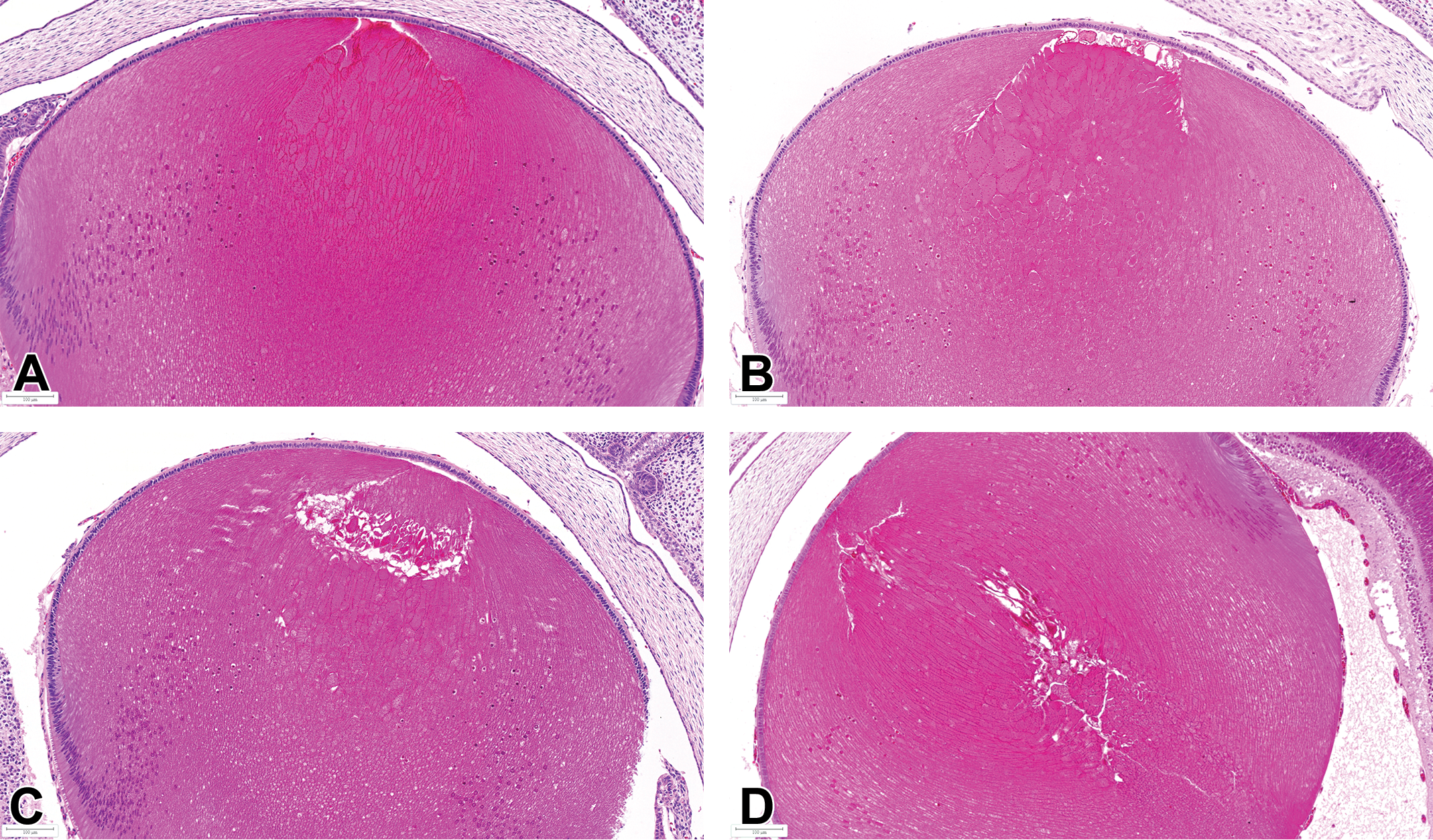
Representative lens findings in eyes of GD21 fetuses from vehicle control dam. Studies 1 and 2: A, Minimal hypertrophy of anterior primary lens fibers. B, Mild focal hypertrophy and focal degeneration of anterior primary lens fibers. C, Hypertrophy and degeneration/fragmentation of anterior primary lens fibers. D, Degeneration and fragmentation of central and anterior primary lens fibers. Bouin's fixation, H&E. GD indicates gestation day; H&E, hematoxylin and eosin.
Degeneration of lens fibers was often focal, mainly affecting the primary lens fibers adjacent to the anterior epithelium and recorded as minimal to slight in severity across all dose groups except for 6 to 8 litters at the high dose with moderate to severe changes compared to only 1 to 2 at the low and mid doses. There was a significant (P < .01) increase in the fetal incidence of lens fiber hypertrophy at the 30 mg/kg (46.4%) and 100 mg/kg (85.8%) and focal degeneration at the high dose of 100 mg/kg (53.4%; Table 2).
Incidence of Microscopic Findings in Fetal Lens: Study 1.
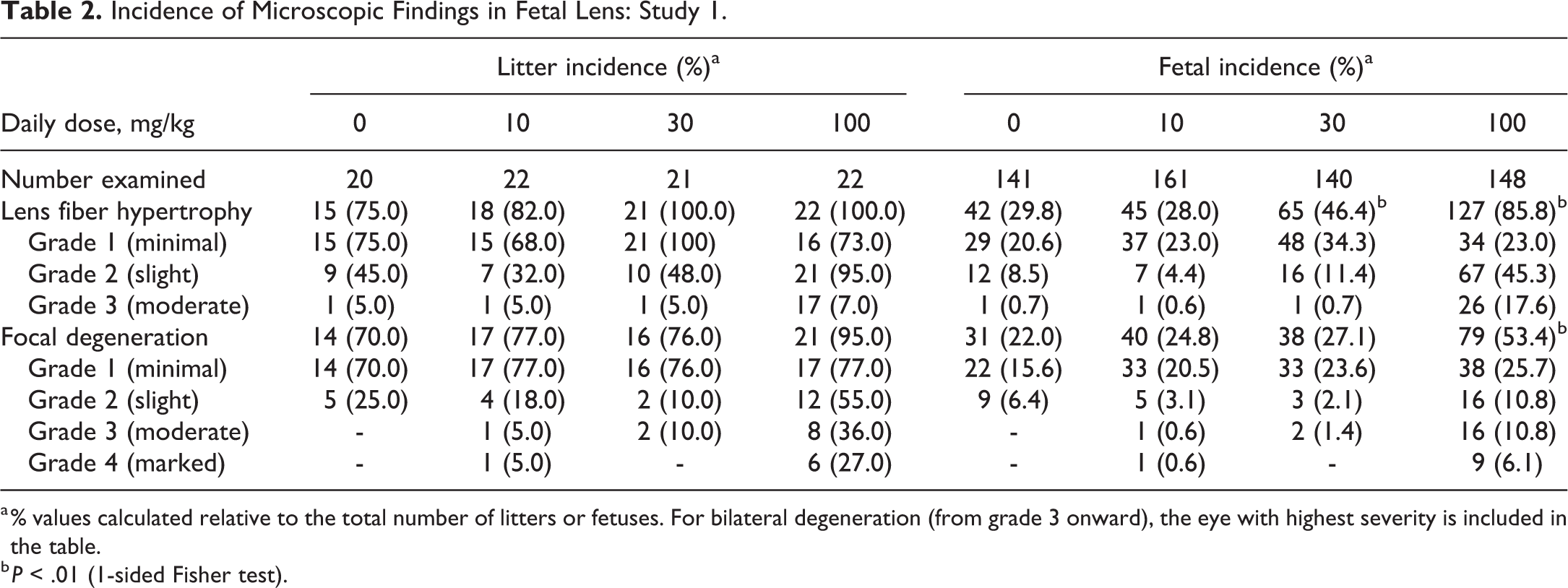
a % values calculated relative to the total number of litters or fetuses. For bilateral degeneration (from grade 3 onward), the eye with highest severity is included in the table.
b P < .01 (1-sided Fisher test).
There was a rather high litter incidence of primary lens fiber hypertrophy (75.0%, 82.0%, 100.0%, and 100.0%) and focal degeneration of the lens nucleus (70.0%, 77.0%, 76.0%, and 95.0%) for the vehicle control and groups treated at 10, 30, or 100 mg/kg, respectively (Table 2). The incidences of these findings were not statistically different between vehicle control and any JNJ-42165279-treated group; however, they tended to be of a higher severity grade at the high dose of 100 mg/kg (Table 2). The relatively high incidences observed in the control rats and at 10 and 30 mg/kg JNJ-42165279 suggested that at 100 mg/kg, JNJ-42165279 was exacerbating rather than inducing the lens changes.
Study 2
Fetal evaluations
At terminal necropsy, 20 females were pregnant in the vehicle controls and 21, 20, and 20 females were pregnant in groups receiving 10, 30, or 100 mg/kg JNJ-42165279, respectively. Fetal weight was unaffected by treatment at 10 or 30 mg/kg dose but was slightly reduced at 100 mg/kg dose, reflecting the lower maternal weight gain during the dosing period. Other pregnancy and fetal parameters assessed were unaffected.
Eyes (head sections) were evaluated histologically. The litter incidence of fetal (GD21) lens fiber hypertrophy and degeneration was similar to controls at ≤30 mg/kg dose; however, at 100 mg/kg dose, the incidences were significantly higher (P < .05). Lens fiber hypertrophy was seen in 100% of the litters and unilateral lens fiber degeneration in 85.0%. (Table 3). The severity of primary lens fiber degeneration was higher in the 100 mg/kg group.
Incidence of Microscopic Findings in Fetal Lens: Study 2.
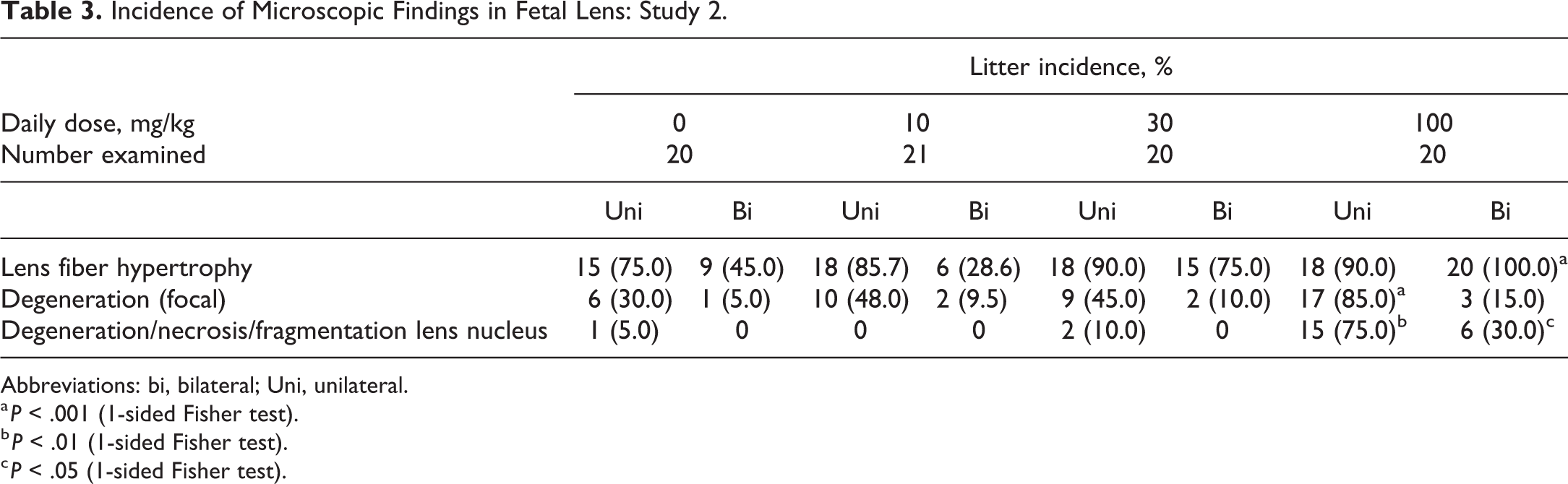
Abbreviations: bi, bilateral; Uni, unilateral.
a P < .001 (1-sided Fisher test).
b P < .01 (1-sided Fisher test).
c P < .05 (1-sided Fisher test).
Litter incidence for primary lens fiber hypertrophy and degeneration/necrosis of fibers in the lens nucleus, which resulted in fragmentation and occasionally focal degeneration or hyperplasia of the overlying anterior lens epithelium, was seen in 75.0% of the litters in 100 mg/kg dose group (Table 3). A female pup found dead at day 1 in the 30 mg/kg dose group showed bilateral hypertrophy of primary lens fibers with moderate degeneration/necrosis and moderate fragmentation.
Postpartum evaluation with focus on ophthalmology
In the groups allowed to litter, there was a slight delay in the onset of parturition at 100 mg/kg with only 4 females completing parturition by the end of GD22; the remaining females completed on GD23 (with 1 on GD24). This delay correlated with an increase in pup mortality at birth (live birth index of 86.7% vs 99.7% in controls, P < .05), although subsequent survival to weaning was unaffected by maternal treatment.
Ophthalmic evaluations were performed premating on the untreated males (n = 32) and parental females (n = 88 each for the EFD and postnatal phases), and then postnatally on the F1 pups when they were 19 to 21 days old and again at 35 to 40 days of age.
Thirty-two stock male rats (about 1 year old) were used to mate with the females. All but 4 of these untreated males had some ophthalmological finding. Small unilateral or bilateral nuclear cataracts with a slight opacity (ie, loss of transparency and obscuration of underlying structures) were diagnosed in 17 of 32 males (53%), and a medium-sized bilateral nuclear cataract of moderate opacity was recorded in 2 animals. For the maternal rats, small unilateral or bilateral nuclear cataracts of a slight opacity were observed in 17 of 88 (19%) mated females used for the EFD phase and in 12 of 88 (14%) used in the postnatal phase. These findings once again confirm that nuclear cataracts are common background changes in this rat strain with up to 19% of the parental females and 53% of the males affected premating in this study.
The F1 generation pups from all litters (556 males and 487 females) were evaluated by ophthalmic examinations. The lenses of pups from dams dosed with JNJ-42165279 were fully formed and no abnormal lens shapes were present. The observed nuclear cataracts were opacities in the central part of the lens nucleus, which were mostly small and of slight opacity (Table 4). In male pups dosed at 30 mg/kg, a slightly increased incidence of unilateral focal nuclear cataract was seen after both 19 to 21 days (9.6% vs 4.6%) and 35 to 40 days (18.4% vs 10.8%) compared to male vehicle controls, accompanied by a slightly higher severity grade in both sexes on days 35 to 40. No relevant change in incidence was noted in females but an increased severity was noted compared to control female pups. In addition to the increased incidence (males) and severity (both sexes) of unilateral focal nuclear cataract, a slight increase in the incidence of bilateral prominent lens nucleus (all with a slight degree) was noted in both sexes after 35 to 40 days (16/264 or 6.1% for both sexes combined compared to 8/263 or 3.0% in controls). A prominent lens nucleus was recorded when the nucleus of the lens was slightly less transparent compared to a normal lens and when the opacity is less prominent as seen for cataracts. The slightly lower lens opacity of a prominent lens nucleus can evolve to nuclear cataract over time. At 100 mg/kg, unilateral cataract was noted in 11.8% of the males and 23.1% of the females after 19 to 21 days, compared to 4.6% in vehicle males and 4.5% in vehicle females. In addition, bilateral cataract was seen in 12.5% of the male and 12.0% of the female pups while none were seen in vehicle-dosed males and 1 (0.8%) in females. During the day 35 to 40 period, 19.9% of the males and 26.9% of the females had unilateral cataracts compared to 6.6% in vehicle males and 12% in vehicle females, while bilateral cataract was present in 19.1% of the male and 14.8% of the female pups during the day 35 to 40 period but absent in vehicle pups. Furthermore, an increased Incidence of bilateral prominent lens nucleus was seen in pups of both sexes at the age of 35 to 40 days.
Incidence of Slit-Lamp Biomicroscopic Findings in F1 Pup Lens Nucleus: Study 2.
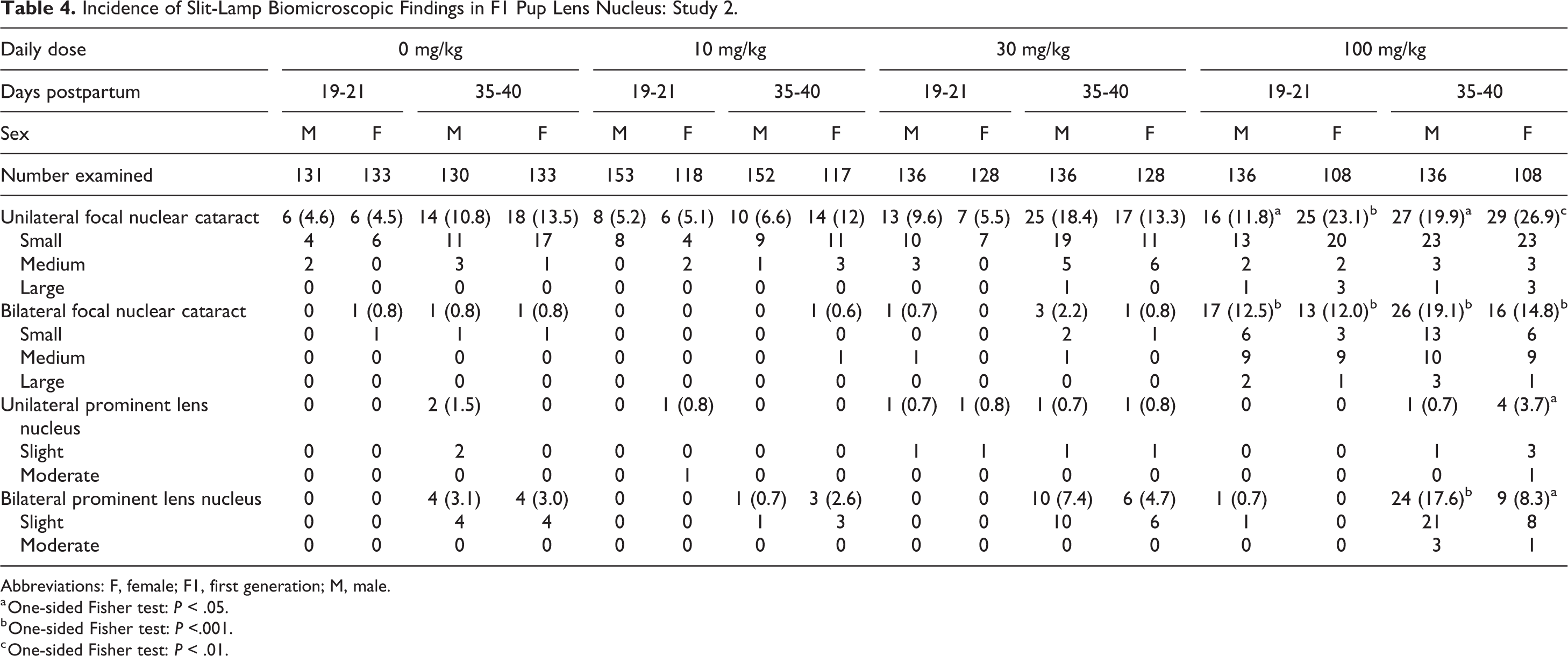
Abbreviations: F, female; F1, first generation; M, male.
a One-sided Fisher test: P < .05.
b One-sided Fisher test: P <.001.
c One-sided Fisher test: P < .01.
The F1 litter and pup incidences of ophthalmic changes across all study groups, including vehicle controls, demonstrated that the incidence of nuclear cataracts tended to increase from PND 19-21 to 35-40, with the percentage increase showing no relationship to treatment (Table 5). This apparent increase was probably related to the increased ease of detection due to the eyes being larger in the older animals, making it easier to detect changes, but the possibility of the appearance of new opacities could not be entirely excluded. As evidenced in the older male rats used as breeders in this study, where nuclear cataracts were observed in 53% of the animals, the incidence of lens opacities, including nuclear cataracts, increases in aging rats.
Litter Incidence of Focal Nuclear Cataracts Identified Via Slit-Lamp Biomicroscopy: Study 2.
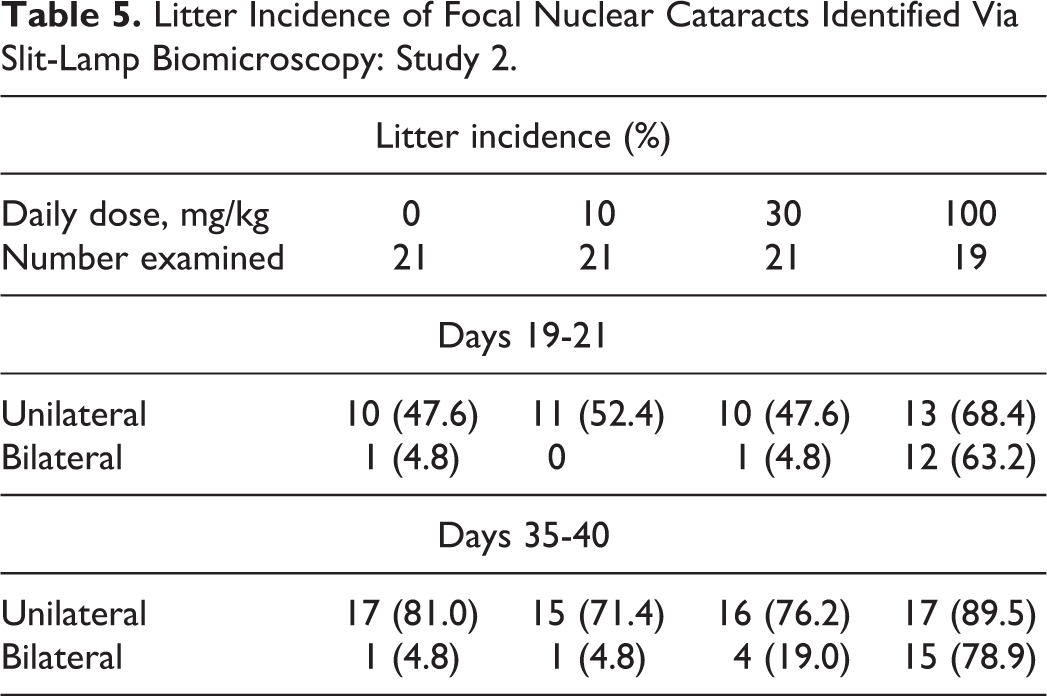
The majority of cataracts at the
There was no correlation in the occurrence of unilateral or bilateral nuclear cataracts between litters when one or both parents showed unilateral or bilateral nuclear cataracts or where both parents showed no abnormalities.
The overall incidences of primary lens fiber hypertrophy with degeneration/necrosis observed by histopathology at GD21 and nuclear cataracts recorded following ophthalmic examination in juvenile pups were generally comparable. The high histological incidence of fetal primary lens fiber hypertrophy, in the absence of degeneration, did not correlate with nuclear cataracts in pups as assessed by ophthalmology, confirming that this variation is likely a normal developmental process not previously characterized since histopathology is rarely a part of EFD or pre- and postnatal development studies.
In the pups, at PND 35 to 41, there was no direct histological correlation for nuclear cataracts diagnosed in ophthalmological evaluations when histological lens changes were compared with ophthalmological lens changes at an individual animal level. However, certain lens changes were observed histologically (degeneration or hyperplasia of lens epithelium and vacuolation or hypertrophy of secondary lens fibers) and ophthalmologically. The lenses of pups at PND 35 to 41 showed compression of primary lens fibers and a decrease in the relative size of the lens nucleus.
Toxicokinetic Results
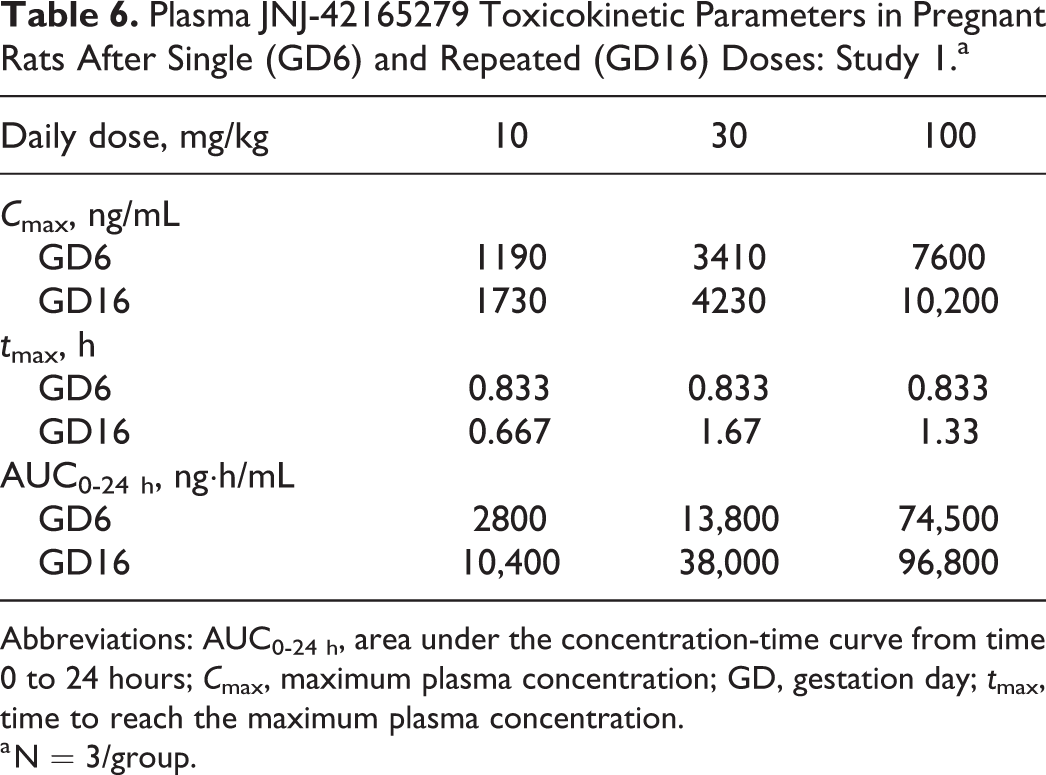
In study 1, following single and repeated oral dosing of JNJ-42165279 (10, 30, or 100 mg/kg), mean peak plasma concentrations were observed between 0.667 and 1.67 hours after dosing, suggesting rapid absorption. JNJ-42165279 demonstrated a dose-proportional increase in mean maximum concentration (C max) on GD6 and GD16 for the 10 and 30 mg/kg doses, whereas the increase in mean area under the concentration-time curve (AUC0 24 h) was more than proportional to dose (Table 6). From 30 to 100 mg/kg JNJ-42165279, mean C max and AUC0-24 h increased less than dose proportionally except for the mean AUC0-24 h at GD6, which increased more than dose proportionally. The mean C max and AUC0-24 h values were generally higher (1.2-1.5× and 1.3-3.7×, respectively) on GD16 relative to GD6 (Table 6).
Plasma JNJ-42165279 Toxicokinetic Parameters in Pregnant Rats After Single (GD6) and Repeated (GD16) Doses: Study 1.a
Abbreviations: AUC0-24 h, area under the concentration-time curve from time 0 to 24 hours; C max, maximum plasma concentration; GD, gestation day; t max, time to reach the maximum plasma concentration.
a N = 3/group.
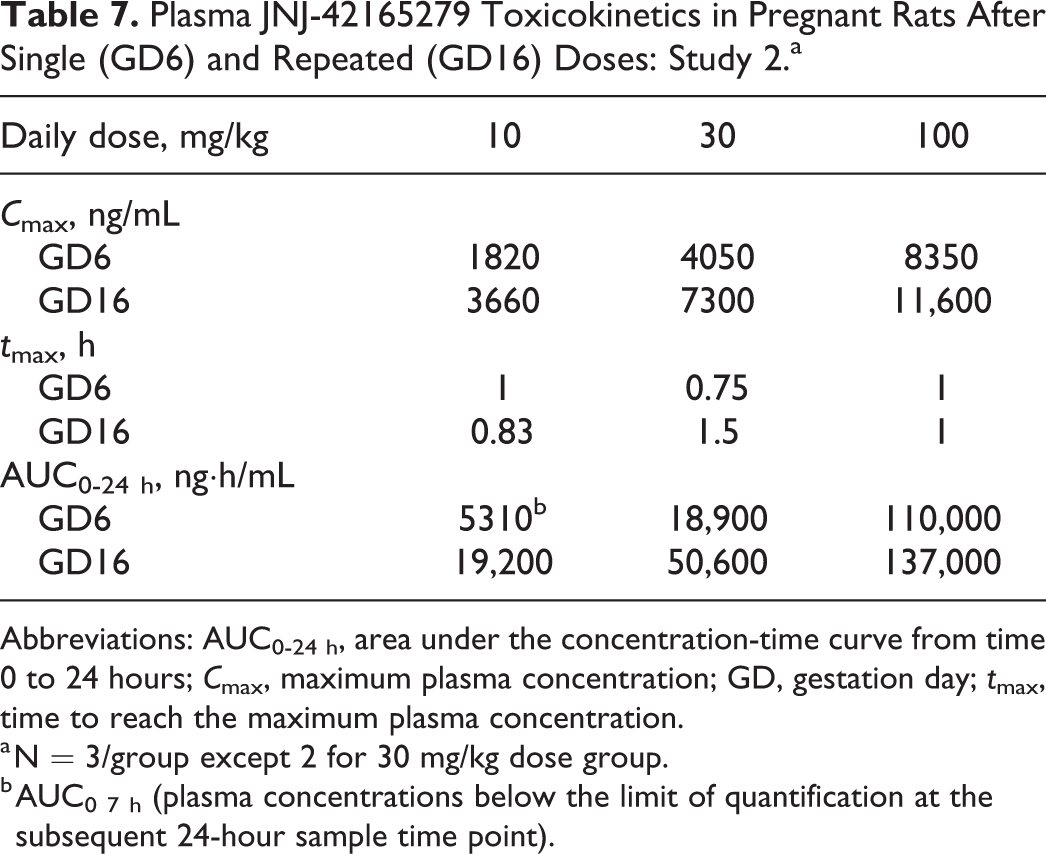
In study 2, toxicokinetic analysis revealed that, overall, the increase in exposure (AUC0-24 h) within the dose range 10 to 100 mg/kg tended to be greater than dose proportional on GD6 and less than dose proportional on GD16. Following repeated administration at 10 and 30 mg/kg, GD16 exposures were higher than GD6, with accumulation ratios ranging from 2.54 to 4.06. At 100 mg/kg, no notable difference in systemic exposure was observed between GD6 and GD16. The exposures in this study tended to be slightly higher than in study 1 (Table 7). Day 16 C max plasma exposures at 10 mg/kg were about 10× above the IC50 of JNJ-42165279 in rats and close to 80-fold at the high dose of 100 mg/kg.
Plasma JNJ-42165279 Toxicokinetics in Pregnant Rats After Single (GD6) and Repeated (GD16) Doses: Study 2.a
Abbreviations: AUC0-24 h, area under the concentration-time curve from time 0 to 24 hours; C max, maximum plasma concentration; GD, gestation day; t max, time to reach the maximum plasma concentration.
a N = 3/group except 2 for 30 mg/kg dose group.
b AUC0 7 h (plasma concentrations below the limit of quantification at the subsequent 24-hour sample time point).
In study 3, after oral administration of JNJ-42165279 and 14 C-JNJ-42165279 (last day of dosing) at 30 mg/kg for 5 days in pregnant rat, at GD19, AUC0-24 h values of JNJ-42165279 for plasma and whole fetuses were 1.8- and 1.1-fold higher than dam plasma AUC0-24 h values, respectively (Table 8). The fetal eye was exposed to JNJ-42165279 but to a lesser extent than in fetal plasma (fetal eye to plasma AUC0-24 h ratio of 0.7). The distribution of JNJ-42165279 to the amniotic fluid was limited with an amniotic fluid to dam plasma AUC0-24 h ratio of 0.4. The AUC values of total radioactivity were much higher than JNJ-42165279 AUC0-24 h values in all the matrices (total radioactivity to JNJ-42165279 AUC0-24 h ratios: 2.5-4.9), indicating that a substantial proportion of the radioactivity was related to metabolites.
Tissue Distribution of JNJ-42165279 and Total Radioactivity at GD19 in Study 3.
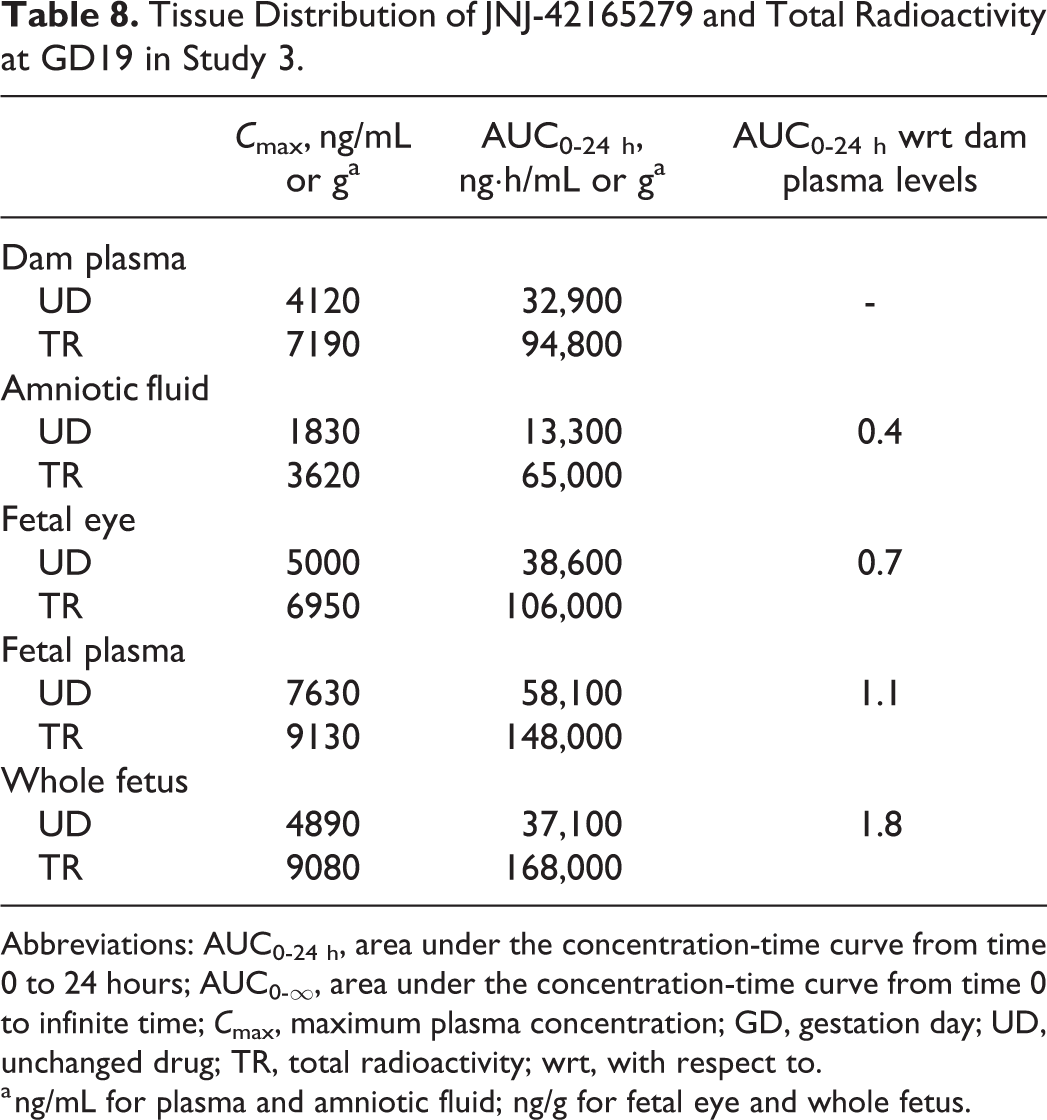
Abbreviations: AUC0-24 h, area under the concentration-time curve from time 0 to 24 hours; AUC0-∞, area under the concentration-time curve from time 0 to infinite time; C max, maximum plasma concentration; GD, gestation day; UD, unchanged drug; TR, total radioactivity; wrt, with respect to.
a ng/mL for plasma and amniotic fluid; ng/g for fetal eye and whole fetus.
Discussion
The definition of a cataract is “opacification of the normally transparent crystalline lens.” 18 Congenital cataracts present at birth and therefore must be caused by changes that occur during embryonic development. Here we describe the findings of EFD studies in rats where primary nuclear lens fiber hypertrophy and degeneration were observed in developing lenses of rat fetuses, both in control- and JNJ-42165279-treated animals. A follow-up study was performed to evaluate the effects of these changes on the optical clarity of the lens in offspring exposed in utero. The studies demonstrated that JNJ-42165279, at suprapharmacological doses (and exposures—80-fold over IC50 at 100 mg/kg), exacerbates the incidence and/or the progression of background nuclear cataracts in Sprague-Dawley rats, a susceptible rat strain that already has a high background incidence of nuclear cataracts. 19 Overall, JNJ-42165279 is not teratogenic and does not induce cataracts in adult rats (data not included).
In most EFD studies in rats, the examination of Bouin-fixed heads of the rat is done by serial free-hand sectioning, and structures are assessed for presence, position, size, shape, and color using a dissecting microscope. Histopathology is rarely performed other than to confirm or characterize a macroscopic observation. As no routine histopathology is performed, very little data are available on the background changes, incidence, and interstudy variations of “normal” findings in fetal lens. Also, there are no data on background ophthalmoscopic changes/variations in juvenile rat lenses.
Assuming that many of the focal lens degeneration with fragmentation changes are irreversible and will be retained in adult rat as nuclear cataracts in ophthalmological evaluations, the fetal incidence in the control, 10, and 30 mg/kg dose groups in this study, although high, is within our in-house historical control data range of animals ordered for various safety studies. In a review of our in-house prestudy ophthalmological findings in young adult rats (∼6-8 weeks), the incidence of nuclear cataracts ranged from 0% to 37%. In most cases, a small-sized focal nuclear cataract with slight opacity was observed. Therefore, it is established that the rat strain used in the current study already has a high background incidence of congenital nuclear cataracts which is further accelerated or exacerbated in offspring of pregnant rats exposed to high exposures of JNJ-42165279 in utero. Additionally, ophthalmoscopic examination of rat pups of the age of PND 18 to 21 and again between PND 35 and 41 is not regularly performed in preclinical studies.
In study 1, granular foci were noted in the lens at a high incidence in fetuses dosed at 100 mg/kg JNJ-42165279 as well as at lower dose levels and the control group. The fact that granular foci correlated with degeneration of primary lens fibers in the treated fetuses but not in the controls was attributed to the greater number, size, and severity of these degenerative foci noted at the high dose and the greater sensitivity of histological evaluation, since several severe lesions were noted histologically in vehicle controls. Microscopic examination of the free-hand serial head sections with eyes from all fetuses revealed that the high incidence of changes in fetal primary lens fiber hypertrophy (and degeneration) is a common spontaneous developmental change in fetal rats and does not necessarily lead to cataracts. This finding may be related to a spontaneous delay or defect in elongation and/or differentiation of primary lens fibers. We speculate that if primary fibers don’t reach the anterior epithelium in time, this could lead to degeneration or hypertrophy of the lens fibers to fill up the central anterior cavity. Due to the mainly anterior location, there might be an effect on elongation of primary lens fibers (lens development, Figure 5). Upon development of secondary lens fibers, compression of primary lens fibers occurs with a decrease in the relative size of the lens nucleus. Presumably the hypertrophic and degenerated nuclear fibers gradually became so compressed as to be undiscernible in study 2 at PND 35 to 41. Additionally, eyes were fixed in Davidson solution for variable periods and not transferred to ethanol or formalin, which is recommended when rodent eyes cannot be processed within 24 to 48 hours; prolonged fixation in Davidson solution can cause artifactual hardening of the lens.
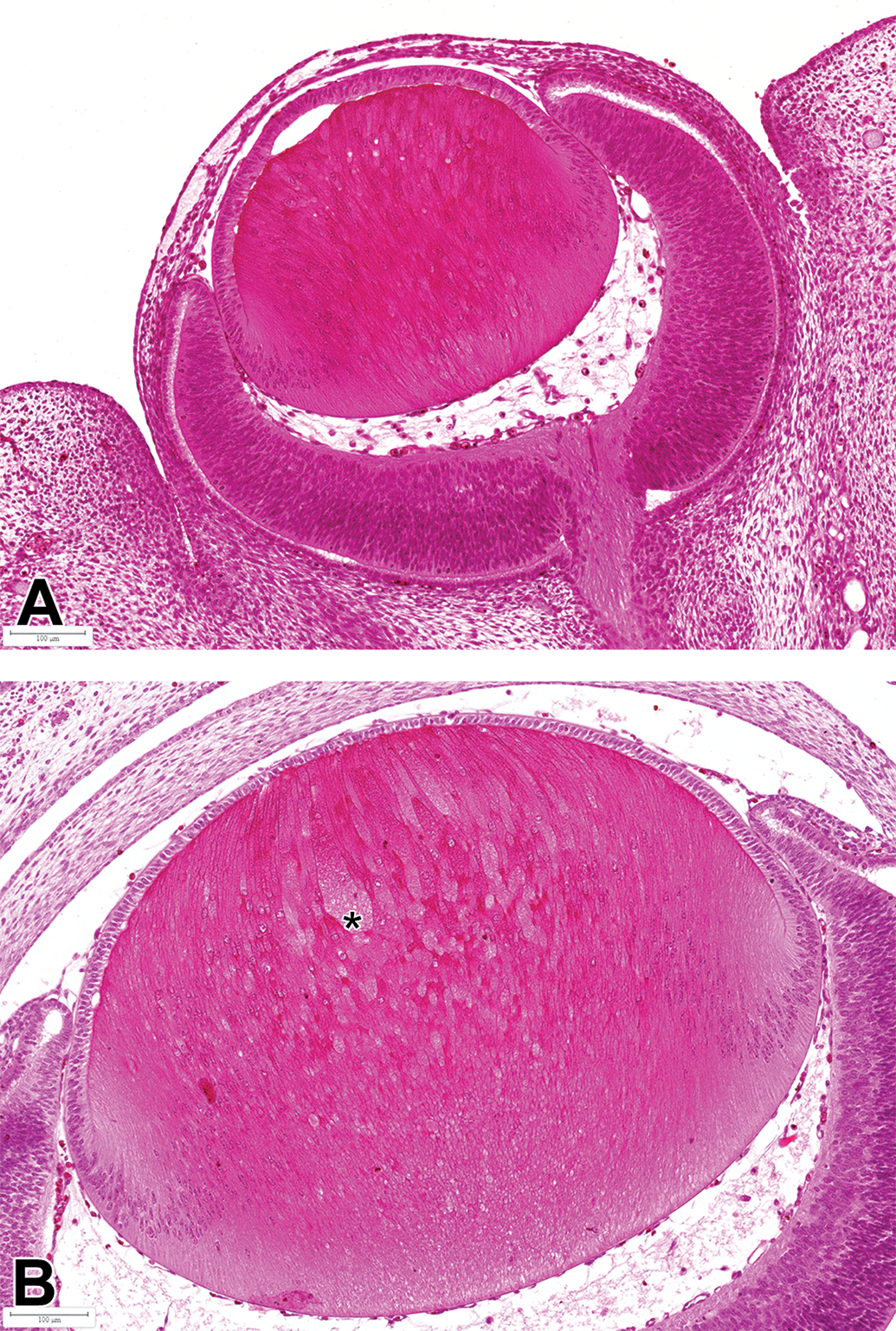
A, Eye of GD15 fetus from untreated control dam (additional pregnant rat from study 2): Normal elongation of posterior lens vesicle cells to form primary lens fibers. B, Eye of GD18 fetus from untreated control dam (additional pregnant rat from study 2): Focal hypertrophy (*) of anterior portion of primary lens fibers. Bouin's fixation, H&E. GD indicates gestation day; H&E, hematoxylin and eosin.
Given that a clear exacerbation of the lens degeneration was only noted at the high dose, over 80-fold above the IC50 for FAAH inhibition, we conclude that the mechanism is unlikely to be related to FAAH inhibition but may be associated with an off-target disturbance of the lens crystallin—the main structural proteins in the lens linked to most cataracts. 20 During pregnancy, rat fetuses were well exposed to JNJ-42165279 and the compound distributed to the eyes. Comparing total radioactivity to JNJ-42165279 levels, it is evident that JNJ-42165279 was also metabolized in rats.
One of the well-established mechanisms of cataracts induction is related to disruption of the cholesterol biosynthesis in the lens. The literature evidence on the use of statin cholesterol-lowering agents 21 and the development of cataracts is still conflicting, but this is largely due to strain and species differences in susceptibility, with dogs considered most sensitive. However, JNJ-42165279 given to rats and dogs in general toxicity studies induced slight increases in serum cholesterol levels. Notably, JNJ-42165279 did not induce lens changes in adult animals dosed for up to 3 months (data not included). As secondary lens fibers are continuously formed throughout life, the absence of lens changes in adult animals point toward a persistent effect on the transparency of the lens nucleus that lacks an identifiable histologic correlate due to compression of the nucleus by the secondary lens fibers.
In a similarly designed EFD study in rabbits, no changes were noted in fetal lenses even at greater maternal and fetal exposures (data not shown). Sprague-Dawley rats were therefore considered more sensitive to the development of these nuclear opacities. Except for the finding in the fetal lens, no other treatment-related fetal abnormalities were recorded. It can be concluded that JNJ-42165279 is not acting as a teratogen in pregnant rats but is triggering an enhanced or exacerbated effect on the progression of background nuclear cataracts in this susceptible rat strain.
Ophthalmic examination is conducted on long-term repeat dosing studies and so data are available for the general background incidence in this strain of rat, but ophthalmoscopic examination of rat pups of the age of PND 18 to 21 and again between PND 35 and 41 is not regularly performed in preclinical studies. In most cases, small-sized focal nuclear cataract of slight opacity was observed. In our facility, the prevalence of this type of nuclear cataract in young adult rats at the start of regular toxicology studies ranged from 0% to 37% (unpublished observation). The fetal incidence observed in the control and JNJ-42165279-treated groups in this study, although high, is within our in-house historical control data range and is in line with a publication of Morita et al, 19 where nuclear cataract was present in 30.6% of the evaluated male Sprague-Dawley rats and 33.7% of the female rats in the age range of 4 to 7 weeks old. Published data from other laboratories on older animals (not juvenile rats) indicate an incidence range of 1.6% by Taradach and Greaves 22 and up to 13.1% by Kuno et al. 23 Kuno et al 23 mentions that male and female Sprague-Dawley rats of 4 to 6 weeks of age showed nuclear cataracts at ophthalmoscopical examination in 8.4% of the males and 13.1% of the females, which is in line with the incidences observed in the control, 10, and 30 mg/kg dose groups in this study. Although the overall pup incidence of cataracts at 100 mg/kg was within our historical background levels (0%-37%) and within the published data of Morita et al, 19 the high dose of 100 mg/kg of JNJ-42165279 clearly caused an increase in incidence and severity over controls in this study and was consistent with the higher incidence of focal degeneration and degeneration/necrosis with fragmentation of the primary lens fibers observed histologically in the fetuses. The high histological incidence of fetal primary lens fiber hypertrophy, in the absence of degeneration, did not correlate with nuclear cataracts at an individual animal level in pups as assessed by ophthalmology, but the incidences and severities were comparable at group level. Our hypothesis is that this degree of hypertrophy (swelling) of fetal primary lens fibers is either reversible or no longer detectable in the juvenile rats (age of 5-6 weeks), as the lens epithelium proliferates to form secondary lens fibers throughout the life of the animal, resulting in compression of the lens nucleus (Figure 6) and potentially also of the hypertrophic fibers of the fetal lens nucleus. Therefore, this variation is likely a normal developmental process not previously characterized since histopathology is rarely a part of EFD or pre-and postnatal development studies. The litter incidences of the lens findings (nuclear cataract and prominent lens nucleus, both signs of a disturbed lens opacity) clearly indicate that these were distributed widely across the litters and showed no influence of the parental animals. This further supports the conclusion that the effects seen were a direct effect on the fetus rather than a parentally mediated response. The statistically significant increased litter incidences in bilateral cataracts and bilateral prominent lens nucleus at 100 mg/kg on days 35 to 40 reflect the significantly increased pup incidences compared to vehicle rats and considered to be related to the pup effects and not the litter effects.
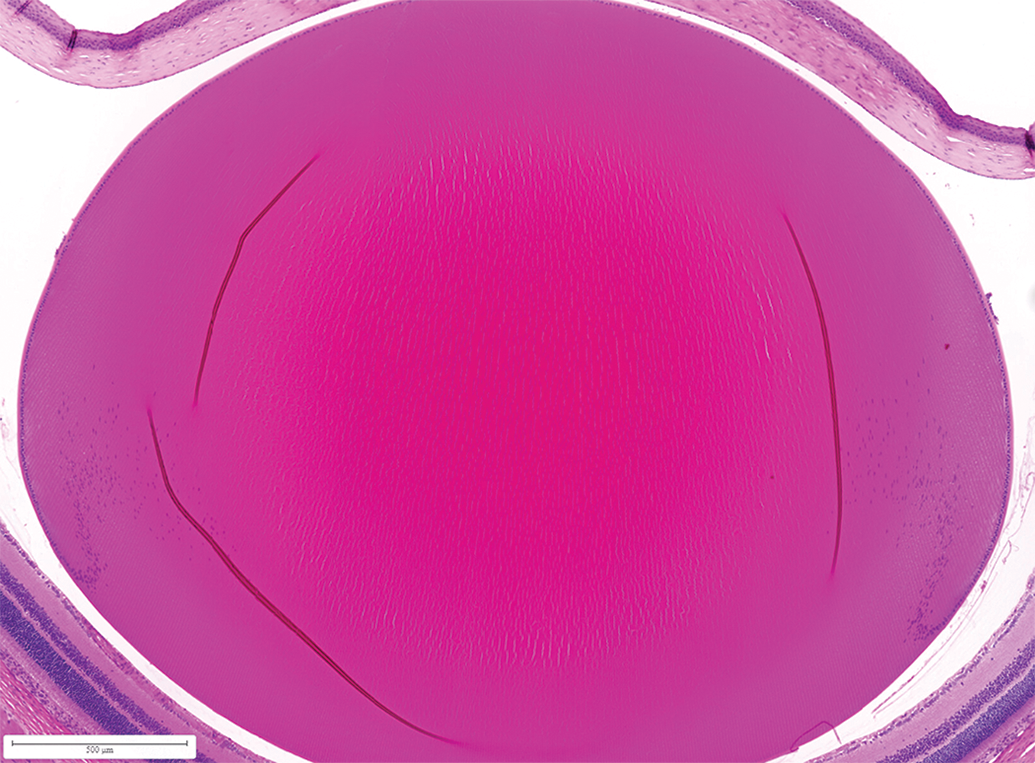
Lens of PND 37 pup (without abnormalities at ophthalmology) from vehicle treated dam. Study 2: Illustrating compression of primary lens fibers and the decrease in the relative size of the nucleus compared with the cortex over time. Davidson fixative, H&E. H&E, hematoxylin and eosin; PND, postnatal day.
Conclusion
Fetal primary lens fiber hypertrophy and focal degeneration as well as juvenile focal nuclear cataract at ophthalmic examination are common background occurrences in Sprague-Dawley rats. The overall incidences of primary lens fiber hypertrophy or degeneration/necrosis observed by histopathology at GD21 and nuclear cataracts recorded following ophthalmic examination in young adult pups were generally comparable. The high histological incidence of fetal primary lens fiber hypertrophy, in the absence of degeneration, did not correlate on an individual animal level, but the incidences were comparable at group level with nuclear cataracts in pups as assessed by ophthalmology, indicating that this variation in primary lens fiber hypertrophy is likely a normal developmental process not previously characterized since histopathology is rarely a part of EFD or pre-and postnatal development studies, and ophthalmology is seldomly performed in juvenile rats. Treatment of pregnant rats with the FAAH inhibitor JNJ-42165279 at suprapharmacological doses appears to exacerbate both the incidence and severity of the fetal and juvenile background nuclear lens changes in this susceptible rat strain.
Footnotes
Authors’ Note
Marjolein van Heerden and Wendy Roosen were the investigators. Graham Bailey had primary roles in the study design, results assessment, and data interpretation as study toxicologist for the study. All authors contributed to the data interpretation for the results. All authors met ICMJE criteria and all those who fulfilled those criteria are listed as authors. All authors had access to the study data and made the final decision about where to publish these data and approved submission to this journal. Graham Bailey is now retired from Jansse.
Acknowledgments
Himabindu Gutha, PhD, and Vaibhav Deshpande, PhD (SIRO Clinpharm Pvt. Ltd., India), provided writing assistance and Ellen Baum, PhD (Janssen Global Services, LLC), provided editorial support for this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of Janssen Research & Development, LLC and are shareholders in the parent company (Johnson & Johnson). Mr Graham Bailey is now retired from Janssen.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies presented in this report were sponsored by Janssen Research & Development, LLC, USA.
