Abstract
The most common target organ for toxicity in the endocrine system is the adrenal gland, and its function is dependent upon the hypothalamus and pituitary gland. Histopathologic examination of the adrenal glands and pituitary gland is routinely performed in toxicity studies. However, the function of the adrenal gland is not routinely assessed in toxicity studies. Assessment of adrenal cortical function may be necessary to determine whether a histopathologic finding in the adrenal cortex results in a functional effect in the test species. As juvenile toxicity studies are more commonly performed in support of pediatric indications for pharmaceuticals, it is important to establish historical control data for adrenal gland function. In this study, adrenal cortical function was assessed in control neonatal and weanling beagle dogs as part of an ongoing juvenile toxicology program. Measurements of serum adrenocorticotropic hormone (ACTH), cortisol prior to and following administration of exogenous ACTH, and aldosterone were conducted beginning at 2 weeks of age continuing through 26 weeks of age. Serum electrolyte concentrations were determined at 4, 13, and 26 weeks of age. Dogs as young as 2 weeks of age synthesize and secrete adrenal cortical hormones and exhibit a functional hypothalamic pituitary adrenal axis.
Keywords
Introduction
The endocrine system is network of glands and organs that synthesizes and secretes hormones to maintain homeostasis, regulate metabolism, reproduction, growth and development, and respond to injury and stress. The adrenal gland, as part of the hypothalamic pituitary adrenal (HPA) axis, is critical in response to stress and regulation of metabolism. 1 The adrenal cortex consists of the zona fasciculata, zona reticularis, and zona glomerulosa and synthesizes glucocorticoid and mineralocorticoid hormones via cytochrome P450 enzymes. The adrenal medulla synthesizes and secretes the catecholamines, epinephrine, and norepinephrine. The chromaffin cells of the adrenal medulla synthesize neuropeptides and catecholamines in a stepwise manner from the precursor, tyrosine. Glucocorticoids induce the activity of phenylethanolamine N-methyltransferase which converts norepinephrine to epinephrine, which is released in response to stress.
The adrenal gland is the most common target organ of toxicity in the endocrine system. 2,3 This is believed to be due to the high vascularity, various receptors, and numerous enzymes involved in the stepwise synthesis of steroid hormones present in the adrenal gland. Xenobiotics may be metabolized by the enzymes in the adrenal gland resulting in production of toxic metabolites and/or free radicals. Other adverse effects include enzyme inhibition. The high content of unsaturated fatty acids in adrenocortical cell membranes makes the adrenal cortex susceptible to lipid peroxidation, while the lipophilicity due to the cholesterol and steroid content favors accumulation of lipophilic xenobiotics. Furthermore, the function of the adrenal gland is dependent on the function of the other organs of the HPA axis; therefore, adverse effects in the hypothalamus or pituitary gland can disrupt the normal function of the adrenal gland. The adrenal cortex, specifically the zona fasciculata and zona reticularis, is affected most frequently by xenobiotics, while fewer findings have been reported in the zona glomerulosa and adrenal medulla following exposure to various compounds.
Adrenal cortical hormones are not stored in significant amounts in the adrenal gland; therefore, hormone synthesis must be continuous to maintain homeostasis. Circulating adrenocorticotropic hormone (ACTH), which is synthesized and secreted by the pituitary gland, regulates the secretion of cortisol, which is synthesized predominantly by the zona fasciculata and to a lesser extent by the zona reticularis. Both cortisol and aldosterone are vital to life. Cortisol is released in response to stress and is involved in glucose homeostasis and immune function. Synthesis of aldosterone, which occurs in the zona glomerulosa, is regulated by angiotensin II via the renin–angiotensin system. Aldosterone acts upon the kidney resulting in conservation of sodium, chloride, and water and loss of potassium with consequent effects on blood volume and blood pressure. Effects on aldosterone synthesis and secretion can lead to water and electrolyte imbalance and alterations in blood volume and blood pressure, which may result in a rapid life-threatening condition. Therefore, xenobiotic-related effects on the adrenal gland can have a profound effect on maintenance of body homeostasis and life.
The aim of this study was to include assessment of adrenal cortical function in a subchronic toxicity study in neonatal and weanling laboratory beagle dogs. The dog is a relevant species in which to evaluate adrenal function because, like the human, the dog adrenal cortex contains CYP17 which is required for production of cortisol, the major circulating glucocorticoid in dogs and humans. The enzyme CYP17 is not present in the rat and mouse which secrete corticosterone as the dominant glucocorticoid. Moreover, the dog is a domesticated species and is less likely to be stressed during laboratory procedures such as blood collection; therefore, reliable measurements of adrenal hormones can be made. The stress of blood collection, especially in rodents, can lead to alterations in adrenal hormones making assessment of potential xenobiotic-related changes in adrenal cortical function in these species challenging. Lastly, the blood volumes required for accurate measurement of adrenal hormones are more easily obtained in the dog versus rodent species.
Materials and Methods
Animals
The data collected and reported in this publication are from control animals in a juvenile toxicology study. Three- to 5-year-old pregnant beagle dogs, with a prior breeding history of 3 to 6 litters, were obtained from Covance (Cumberland, VA) and were received by Charles River Laboratories (Senneville, Quebec, Canada). At arrival, the females were between gestation days 38 and 44. Each pregnant dog and the subsequent litter were housed in an individual double stainless steel cage equipped with modified floors and nesting boards. Nesting boxes were provided to pups after birth. Prior to parturition up to the time when the youngest litter in the room reached 6 weeks of age, the room temperature was maintained at 21°C to 27°C and then reduced to 17°C to 23°C. On postnatal day (PND) 1, litters were culled randomly, if necessary to 8 pups per litter (4 males and 4 females, if possible), and subsequently culled to 6 pups per litter (3 males and 3 females, if possible) on PND 4. On PND 6, litters of healthy pups were randomly assigned to a group such that the entire litter was assigned to the same dose group. Each litter had 1 to 6 pups assigned to the study. Beginning on PND 7, as part of a 13-week toxicity study with a 13-week recovery period, 10 control pups/sex were dosed via oral gavage with vehicle (0.5% [wt/vol] carboxymethylcellulose in ultrapure water, 5 mL/kg) for 90 days; 6 pups/sex were necropsied on PND 98 (14 weeks of age) following administration of the last dose on PND 97, and the remaining pups (4 males and 3 females) were necropsied at the end of a 13-week recovery period on PND 189 (27 weeks of age). During the recovery period, 1 female was removed from the study for humane reasons. A total of 6 litters was assigned to the control group.
Beginning on PND 28, pups were offered dry- or moistened-certified Eukanuba Premium Performance formula. Weaning for increasing durations per day occurred during PND 49 to 55 until complete separation from the dam on PND 56. During PND 49 to 55, pups were offered electrolyte solution (Calf-Lyte II).
Following weaning, animals were housed within the same dose group in stainless steel cages equipped with a vinyl-coated mesh floor and an automatic watering valve. At approximately 4 months of age, dogs were acclimated to standard pelleted food using a stepwise escalation from Eukanuba Premium Performance Formula to PMI Nutrition International Certified Canine Chow No. 5007 (25% protein) for 6 days. Once the 6 days of acclimation to new diet was completed, all dogs had access to 400 g PMI Nutrition International Certified Canine Chow No. 5007 once daily. The food ration was offered after dosing and was left in the cage for a period of 4 to 6 hours and then removed.
Animals were vaccinated as follows. At 8 weeks of age, dogs received a combined vaccine of distemper, adenovirus type 2, parainfluenza, parvovirus, and Bordetella intranasal. For dogs assigned to the recovery portion only, a combined vaccine of distemper, adenovirus type 2, parainfluenza, parvovirus, and leptospirosis was administered at 12 weeks of age. At 16 weeks of age, dogs assigned to the recovery portion of the study received a combined vaccine of distemper, adenovirus type 2, parainfluenza, parvovirus, leptospirosis, and rabies.
Animals were observed for mortality twice daily and for general observations daily. Body weights were obtained from individual pups daily from birth to PND 21 and then twice weekly thereafter. Food consumption was measured daily in weaned pups.
The study was approved by an Institutional Animal Care and Use Committee and was conducted in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited institution in accordance with current guidelines for animal welfare 4 and in compliance with Good Laboratory Practices.
Sample Collection
At 4 and 13 weeks of age during the dosing period (all animals, 10 males and 10 females), and at 26 weeks of age during the last week of the 13-week recovery period (animals designated for recovery, 4 males and 3 females), serum samples were collected for determination of electrolytes using the Roche Modular P800 chemistry analyzer. Animals were not fasted for blood collection during the dosing period. During the recovery period, blood was collected prior to offering of food. When applicable, blood collection for adrenal cortical function was performed at least 1 day after blood collection for serum electrolytes.
At 2, 4, 8, and 13 weeks of age during the dosing period (all animals, 10 males and 10 females), and at 26 weeks of age during the last week of the 13-week recovery period (animals designated for recovery, 4 males and 3 females), serum samples were collected for determination of ACTH, cortisol (pre- and 1 hour post exogenous ACTH stimulation), and aldosterone. Blood was collected via the jugular vein; samples for measurement of ACTH, aldosterone, and cortisol were collected prior to ACTH stimulation, and samples for measurement of cortisol also were collected at 1 hour post ACTH stimulation.
For the ACTH stimulation test, pups were administered Cortrosyn® (Amphastar Pharmaceuticals) intramuscularly at a dose of 5 µg/kg between 8:00 and 9:00 in the morning. Each lyophilized vial of Cortrosyn® containing 0.25 mg of cosyntropin was reconstituted as per package insert instructions under clean conditions, by adding 1.0 mL of 0.9% sodium chloride for injection, United States Pharmacopeia (USP) on each day of administration to achieve a concentration of 250 µg/mL. Reconstituted vials were gently swirled to ensure mixing and stored at room temperature for no longer than 24 hours; any residual volumes were discarded.
Samples were allowed to clot at room temperature for 1 to 1.5 hours. Samples were centrifuged at 1800 x g for 15 minutes. The resultant serum was aliquoted separately for each hormone to be measured, frozen immediately on dry ice, and transferred to a −80°C freezer until shipment on dry ice to Antech Diagnostics (Morrisville, NC) for analysis.
Hormone Analysis
Serum samples were shipped on dry ice to Antech Diagnostics for analysis of ACTH, cortisol, and aldosterone using validated assays. Analysis of hormones was performed within the established stability for each validated assay. Briefly, ACTH was determined using an immunoradiometric method (kit no. 3KG011; Scantibodies Laboratory, Inc). The method utilized polyclonal antibodies directed against ACTH 1-16 and ACTH 24-39. The anti-ACTH 24-39 was labeled with I125 and the antibody directed against ACTH 1-16 was coated onto the tubes. Adrenocorticotropic hormone in the dog samples was bound both to the tubes and to the I125-anti ACTH 24-39 antibody. After incubation, free I125-anti ACTH 24-39 and bound I125-anti ACTH 24-39 antibody fractions were separated by discarding the supernatant. Simple washing reduced the nonspecific binding to a minimum. The concentration of ACTH was directly proportional to the radioactivity bound to the tubes after separation. The concentration of ACTH in the dog samples was determined by interpolation using a calibration curve. The measurement of radioactivity was performed using a Packard Cobra II Series Gamma Counter (Perkin Elmer). The lower limit of quantitation (LLOQ) was 1.9 pg/mL for ACTH. Values below the LLOQ were assigned a value of 1 pg/mL for calculation of mean values.
The cortisol assay was a solid-phase chemiluminescent enzyme immunoassay (Immulite® kit no. LKCO1; Siemens Healthcare Diagnostics). The bound complex, and thus also the photo output, was measured by a luminometer (Siemens Healthcare) and was inversely proportional to the concentration of cortisol in the sample. The LLOQ was 0.2 µg/dL for cortisol. Values below the LLOQ were assigned a value of 0.1 µg/dL for calculation of mean values.
The aldosterone assay was a solid-phase enzyme-linked immunosorbent assay, based on the competitive binding principle (kit no. 11-AD2HU-E01; Alpco). Each sample was placed in a microplate well with a known concentration of aldosterone conjugated to horseradish peroxidase, which competed for binding sites of aldosterone antiserum coated on the microplate well walls (solid phase). After incubation and a wash to remove any unbound aldosterone, a substrate solution (3,3,5,5’-tetramethylbenzidine) was added to bind with the conjugated aldosterone. A stop solution (1M sulfuric acid) was added, and the optical density at 450 nm was read using a microplate reader (Molecular Devices). The concentration of aldosterone was inversely proportional to the optical density. The LLOQ was 15.0 pg/mL for aldosterone.
Necropsy, Histology, and Histopathologic Examination
Animals were fasted overnight prior to the scheduled necropsies at the end of 13 weeks of dosing on PND 98 (14 weeks of age) and at the end of the 13-week recovery period on PND 189 (27 weeks of age). A full list of tissues, including the adrenal glands and pituitary gland, was collected from each dog at the scheduled necropsies. The adrenal and pituitary glands were weighed. The adrenal and pituitary glands were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at a thickness of 5 µm, and stained with hematoxylin and eosin. The tissue section slides were examined by a board-certified veterinary pathologist (DVM, DACVP; A.A.).
Results
Hormone Analyses and Electrolytes
Adrenocorticotropic hormone concentrations in males and females were relatively low at 2, 4, and 8 weeks of age but increased at 13 and 26 weeks of age (Table 1). In neonatal beagle dogs through 8 weeks of age, all ACTH values were below the LLOQ of 1.9 pg/mL in 7/10 males, and in the remaining 3/10 males, each had a single value above the LLOQ at 2 or 4 weeks of age. In neonatal females through 8 weeks of age, all ACTH values were below the LLOQ in 6/10 females, and in the remaining 4/10 females, each had a single value above the LLOQ at 2 or 4 weeks of age. The values above the LLOQ were 2.3 to 5.8 pg/mL and 2.0 to 5.6 pg/mL in neonatal males and females, respectively.
Serum ACTH Concentrations (pg/mL) in Male and Female Juvenile Beagle Dogs.a
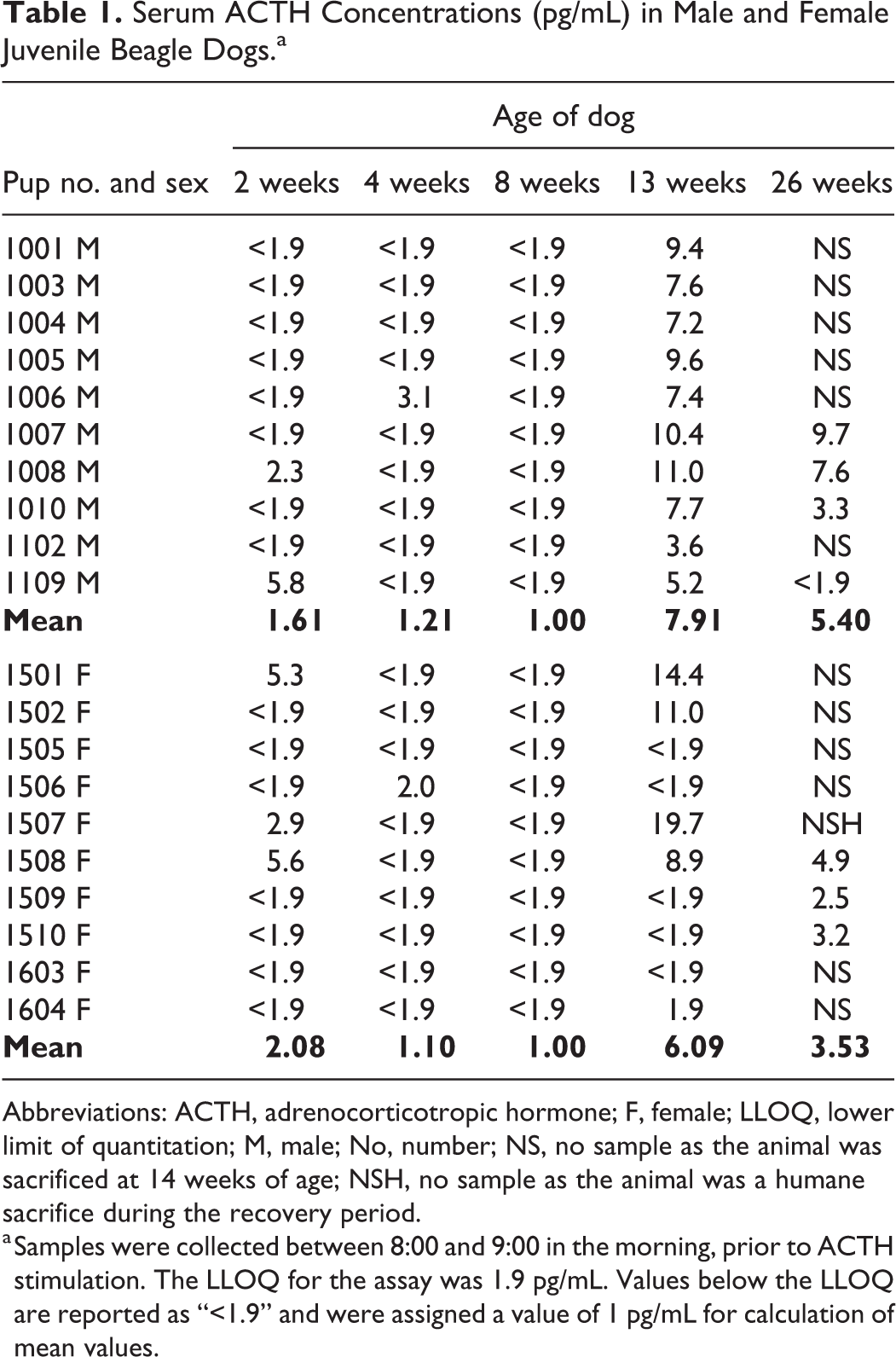
Abbreviations: ACTH, adrenocorticotropic hormone; F, female; LLOQ, lower limit of quantitation; M, male; No, number; NS, no sample as the animal was sacrificed at 14 weeks of age; NSH, no sample as the animal was a humane sacrifice during the recovery period.
a Samples were collected between 8:00 and 9:00 in the morning, prior to ACTH stimulation. The LLOQ for the assay was 1.9 pg/mL. Values below the LLOQ are reported as “<1.9” and were assigned a value of 1 pg/mL for calculation of mean values.
Beginning as early as 2 weeks of age, all males and females responded to exogenous ACTH (Figure 1, Supplemental Tables 1 and 2). Baseline cortisol concentrations were above the LLOQ of 0.2 µg/dL in the majority of samples collected. Four of 10 males and 1/10 females each had a single baseline cortisol value below the LLOQ at 2 or 4 weeks of age. Throughout the study, mean cortisol values increased 7.8- to 19-fold in males and 6.3- to 11-fold in females following ACTH stimulation. When baseline cortisol values were above the LLOQ, individual increases in cortisol concentrations at 1 hour following ACTH stimulation were 3.7- to 43-fold in males and 2.6- to 41-fold in females.
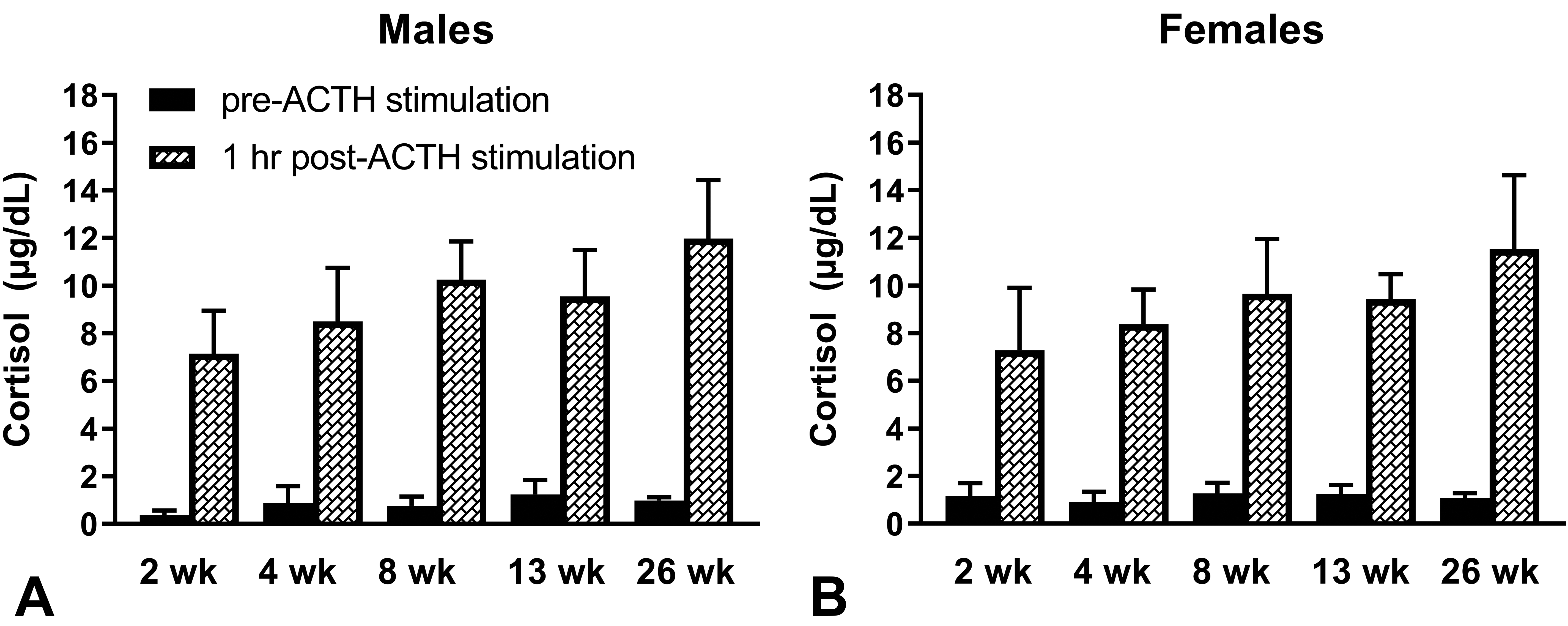
Serum cortisol concentrations in male (A) and female (B) juvenile beagle dogs prior to and at 1 hour following intramuscular administration of 5 µg/kg ACTH (Cortrosyn®). The ages of the dogs are shown on the x-axis. Cortrosyn® was administered between 8:00 and 9:00 in the morning. Immediately prior to and at 1 hour following administration of Cortrosyn®, serum was collected for measurement of cortisol. The LLOQ was 0.2 µg/dL. Values below the LLOQ were assigned a value of 0.1 µg/dL for calculation of mean values. Values are mean + SD. At 2, 4, 8, and 13 weeks of age, n = 10/sex. At 26 weeks of age, n = 4 males and n = 3 females. ACTH indicates adrenocorticotropic hormone; LLOQ, lower limit of quantitation.
All aldosterone values were above the LLOQ of the assay. Aldosterone concentrations were quite variable in males and females and for individual animals between intervals (Figure 2, Supplemental Table 3). Mean aldosterone values in males were lowest at 2 weeks of age and were highest at 26 weeks of age. In females, mean values of aldosterone were relatively consistent from 2 weeks to 26 weeks of age.
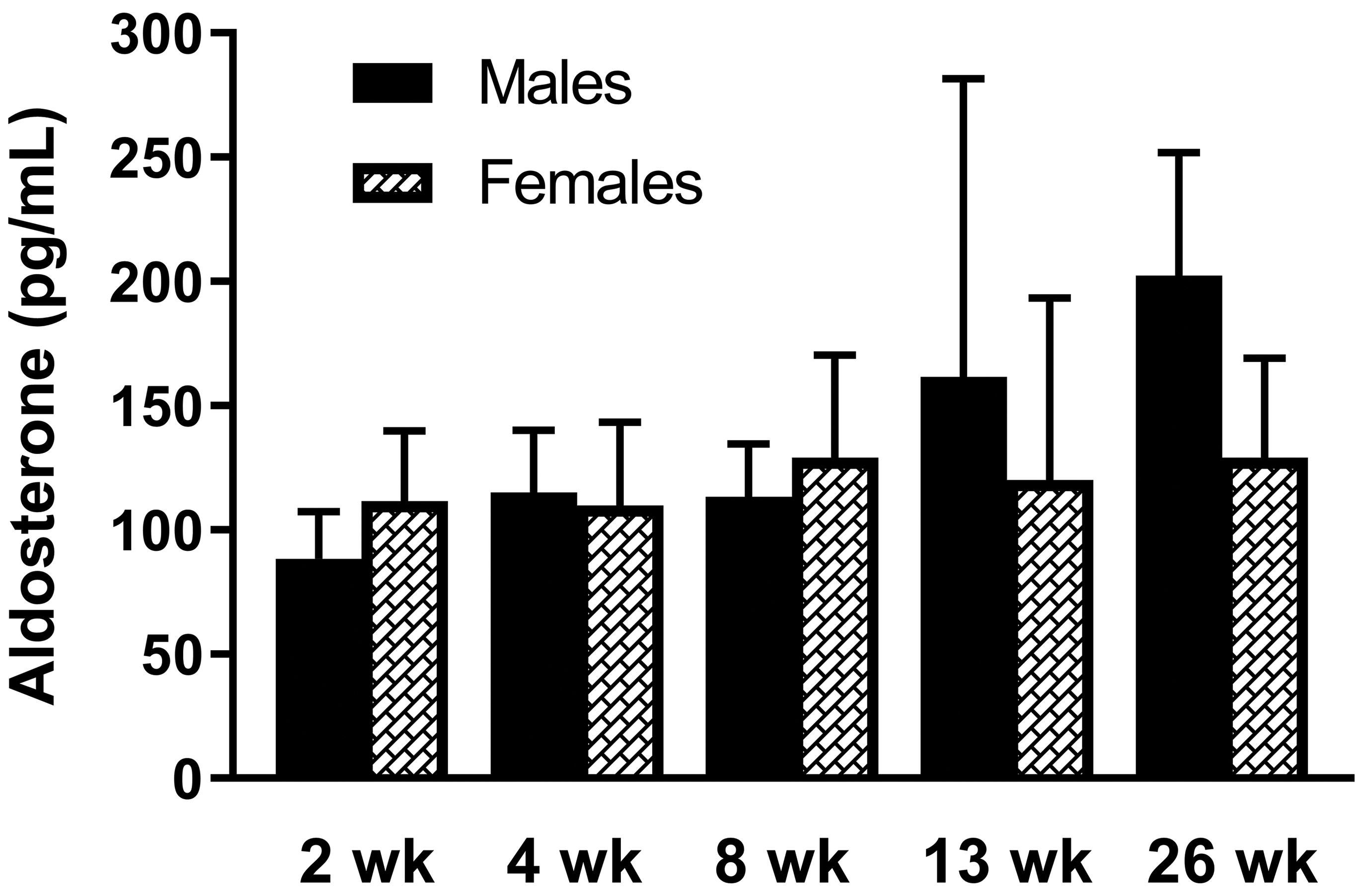
Serum aldosterone concentrations in male and female juvenile beagle dogs. The ages of the dogs are shown on the x-axis. Samples were collected between 8:00 and 9:00 in the morning, prior to ACTH stimulation. The LLOQ was 15.0 pg/mL. Values are mean + SD. At 2, 4, 8, and 13 weeks of age, N = 10/sex. At 26 weeks of age, N = 4 males and N = 3 females. ACTH indicates adrenocorticotropic hormone; LLOQ, lower limit of quantitation.
Mean serum concentrations of chloride and sodium increased slightly from 4 to 26 weeks of age (Figure 3A and B). Mean serum potassium concentrations decreased slightly with age (Figure 3C). At each interval, serum values for each electrolyte were similar in both sexes.
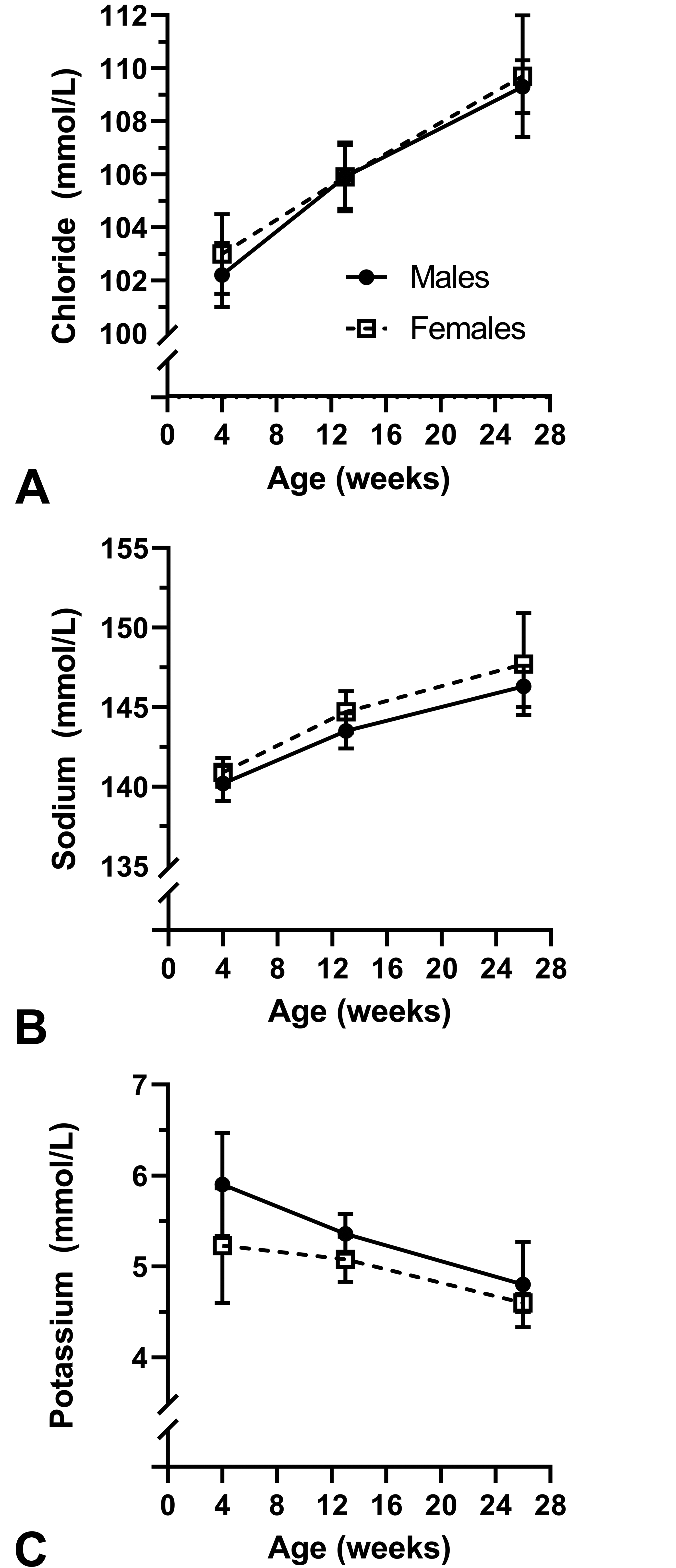
Serum chloride (A), sodium (B), and potassium (C) concentrations in male and female juvenile beagle dogs. Animals were not fasted for blood collections at 4 and 13 weeks of age and were fasted for the blood collection at 26 weeks of age. Values are mean ± SD. At 4 and 13 weeks of age, n = 10/sex. At 26 weeks of age, n = 4 males and n = 3 females.
Organ Weights
Absolute weights of the adrenal glands and pituitary glands increased slightly from 14 to 27 weeks of age in males and females (Figure 4A and C). This correlated with the increase in body weights as the pups grew, and when the adrenal gland and pituitary gland weights were normalized to body weight, the differences in these organ weights between 14 and 27 weeks of age were unremarkable (Figure 4B and D). In males the slight difference noted in pituitary gland weights when normalized to body weight was insignificant and related to individual body weight variability.
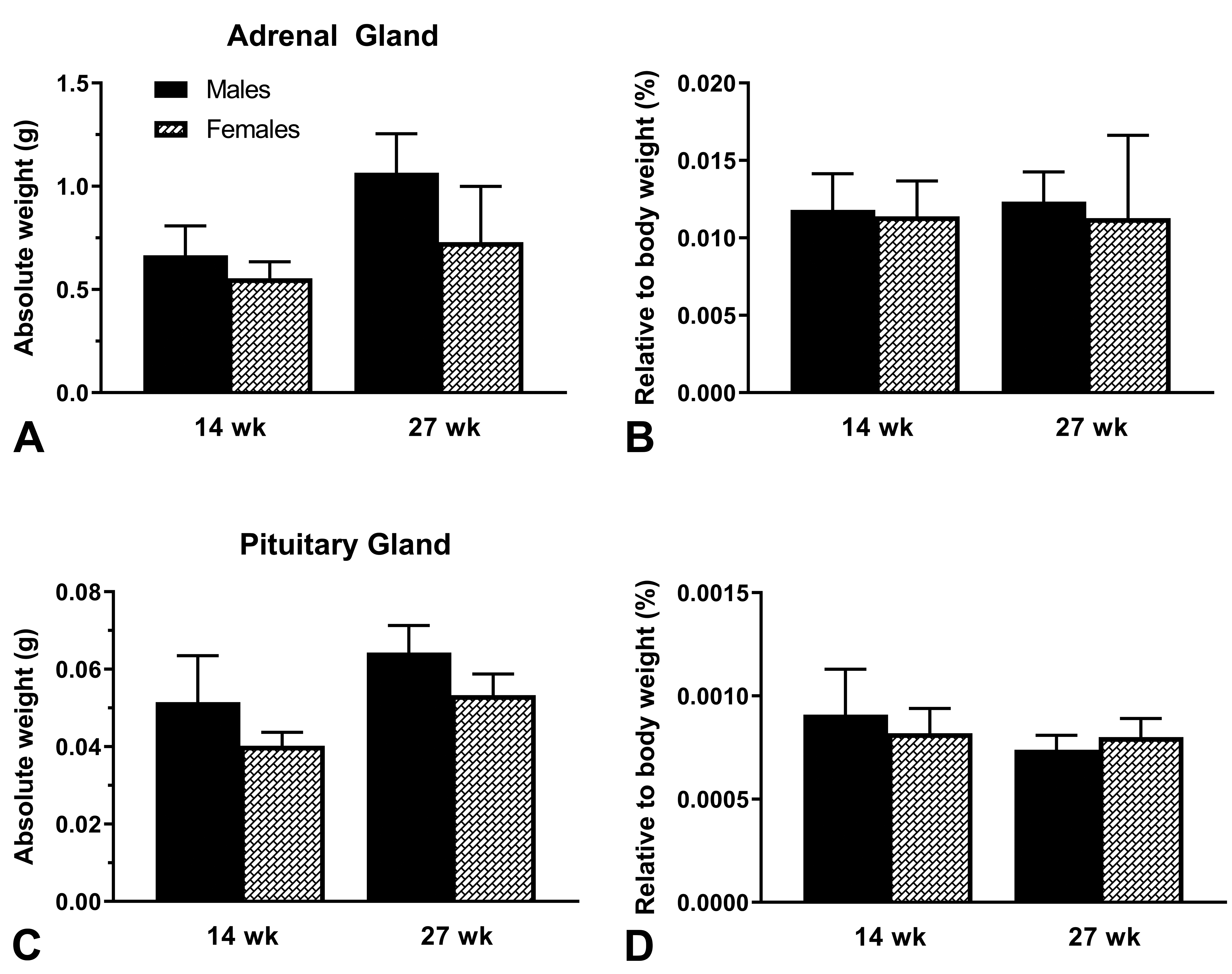
Absolute (A) and relative (B) weights of adrenal glands and absolute (C) and relative (D) weights of pituitary gland in juvenile beagle dogs sacrificed at 14 and 27 weeks of age. Animals were fasted overnight prior to necropsy. Values are mean + SD. N = 6/sex for 14-week-old beagle dogs. N = 4 males and n = 3 females for 27-week-old beagle dogs.
Histopathology
Histopathological findings in the adrenal and pituitary glands at scheduled necropsies were limited to an incidental subcapsular cyst in the adrenal gland of 1/6 males at 14 weeks of age (Figure 5) and an incidental cyst in the pituitary gland of 1/4 males at 27 weeks of age. There were no microscopic findings in the adrenal and pituitary glands in females at 14 and 27 weeks of age.
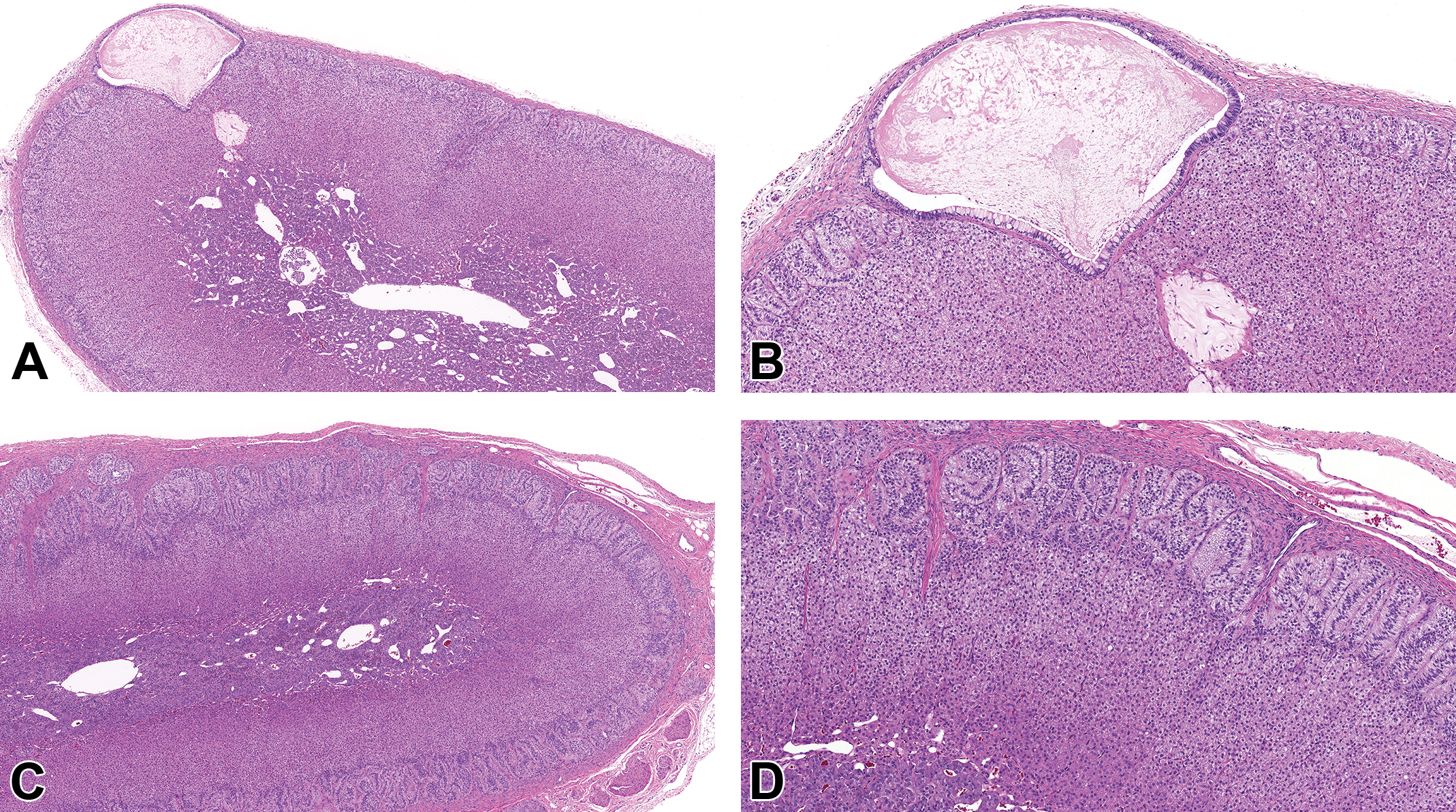
Adrenal gland from a 14-week-old beagle dog showing a subcapsular cyst lined by cuboidal to columnar pseudostratified epithelium with mucus cells and filled with amphophilic to eosinophilic flocculent material (A and B). A, The original scan ×4 and (B) the original scan ×10. C and D, Low and high magnification of normal adrenal gland from a 27-week-old beagle dog. C, The original scan ×4 and (D) the original scan ×10. All sections stained with hematoxylin and eosin.
Discussion
The beagle dog is a common laboratory species for toxicity testing of small molecule xenobiotics, including pharmaceuticals. In support of dosing in adult humans, initiation of dosing in laboratory beagle dogs typically occurs at 4 to 6 months of age. 5 Juvenile toxicology studies are performed to support dosing of pharmaceuticals in children, including progressive and fatal diseases that may preclude children from reaching adulthood and diseases that affect both adults and children. In the latter case, it is standard practice first to determine that the drug can be dosed safely in adults and then perform additional clinical trials enrolling children. Due to concerns regarding the potential of a pharmaceutical agent to affect growth and development, juvenile toxicology studies in the more sensitive species in a toxicology program are routinely conducted to support administration in the intended age range of the pediatric population to be treated. 6 –8 Recently, the International Council for Harmonization (ICH) finalized guideline S11 to provide clarification of the ICH M3(R2) guideline recommendations 9,10 on when juvenile toxicology studies are necessary, important points to consider in the design of toxicity studies in juvenile animals, and development of drugs for pediatric-only indications. Generally, juvenile toxicology studies are performed on a case by case basis and are often requested by regulatory agencies. Due to the increased awareness of the need to dose pediatric patients safely, there has been an increase in the number of juvenile toxicology studies performed. These studies are done in juvenile animals, whereas most general toxicology studies are initiated in young adult animals in which historical control data are readily available. Therefore, it is important for testing facilities to establish historical control data in juvenile animals in which changes are seen during development. As animals mature, there may be differences in various clinical pathology parameters, including adrenal cortical hormones, and in parameters regulated by these hormones, including electrolytes. When a new xenobiotic is tested in a juvenile species, there may be limited data from concurrent controls, and comparison to historical control or reference range data may be necessary to discern whether there is a test article–related effect on adrenal function.
The aim of this study was to monitor adrenal cortical function in control neonatal and weanling dogs in a juvenile toxicology program. The adrenal gland comprises two embryologically and functionally distinct endocrine tissues. The adrenal cortex is derived from the mesoderm and synthesizes and secretes hormones derived from cholesterol, and the adrenal medulla is derived from the neural crest and synthesizes and secretes catecholamines derived from tyrosine. 11 Beginning at 2 weeks of age in laboratory beagle dogs, the three major zones of the adrenal cortex, namely the zona glomerulosa, zona fasciculata, and zona reticularis, are distinguishable upon histopathologic examination. 12 In all ages of dogs, the zona fasciculata is the largest region of the adrenal cortex, while the zona glomerulosa and zona reticularis are smaller and approximately equal in size. 12 The results of this study show that beagle dogs as young as 2 weeks of age have a functional adrenal cortex and have a reserve of cortisol that is released in response to ACTH. The ACTH stimulation test is commonly performed to assess adrenal cortical function 13 and is optimally performed with an intramuscular or intravenous injection of 5 µg/kg synthetic ACTH, 14 with evaluation of cortisol concentrations at 1 to 2 hours post ACTH administration. In this study, measurement of cortisol concentrations at 1 hour post ACTH stimulation was optimal for evaluation of adrenal cortical function. Moreover, consistent with previous observations that the zones of the adrenal cortex are present at 2 weeks of age, 12 in this study, 2-week-old dogs did synthesize and secrete cortisol and aldosterone. The zona fasciculata and, to some extent, the zona reticularis are responsible for production of the glucocorticoid, cortisol. The zona glomerulosa is responsible for production of the mineralocorticoid, aldosterone. Also at 2 weeks of age, the HPA axis was functional, as dogs did respond to exogenous ACTH, with resultant secretion of cortisol from the adrenal gland. 15 In comparison to 13 and 26 weeks of age, the neonatal dogs at 2, 4, and 8 weeks of age had relatively lower levels of endogenous ACTH.
The observations in this study are supported by a study in which neonatal beagle pups had evaluations of cortisol, ACTH, aldosterone, and electrolytes every 2 weeks from 2 weeks to 12 weeks of age. 16 Many of the ACTH values in dogs ≤ 8 weeks of age in the current study were below the LLOQ of 1.9 pg/mL. In the current study through 26 weeks of age, endogenous ACTH values were consistently lower than those observed in the study conducted by Randolph et al. 16 These differences in ACTH may be due to differences in the assays used to measure this hormone between the two studies. The reference range for ACTH in normal dogs is 10 to 90 pg/mL. 13,17,18 Based on the results of the previous study, 16 as well as reference ranges in normal dogs, 13,17,18 the lower limit of quantitation of 1.9 pg/mL for the current study seemed appropriate. Adrenocorticotropic hormone is a labile peptide hormone, and in the current study, care was taken to preserve this peptide during collection, processing, storage, and shipment to the analytical laboratory. Although possible, it is not believed that ACTH degraded and that the values are correct using a validated assay and may reflect the immaturity of the dogs in this study as well as the tightly controlled laboratory conditions to minimize stress to the animals. As evaluation of adrenal hormones is not routinely performed in neonatal dogs, reference ranges for ACTH reflect mature dogs in which a diagnosis of possible adrenal disease is under evaluation.
Baseline and post-ACTH cortisol values in this study and the previous study 16 were similar. There were two cortisol samples per time point for each animal: one sample collected prior to and a second sample collected 1 hour following ACTH stimulation. Therefore, the total number of cortisol values in this study was 174. Of the 174 values, 5 were below the LLOQ of 0.2 µg/dL; these 5 samples were collected prior to ACTH stimulation. Overall reference ranges for baseline cortisol concentrations are 0.5 to 5 µg/dL, and for cortisol concentrations post ACTH stimulation are 5.5 to 20 µg/dL. 13,14,17 –21 Baseline cortisol values in 2- and 4-week-old dogs were slightly below or within the reference ranges, and at ≥ 8 weeks of age, baseline cortisol values were within the reference range. Almost all cortisol concentrations obtained at 1 hour post ACTH administration were within the reference range for cortisol measurements obtained following ACTH stimulation. More importantly, all dogs had an appropriate response to exogenous ACTH challenge with a several-fold increase in cortisol demonstrating a functional HPA axis as young as 2 weeks of age. The organ weight and histopathology data for the pituitary gland and adrenal glands reflect normal healthy dogs at 14 and 27 weeks of age.
Similar to that seen in the previous study, 16 aldosterone values were highly variable. In addition, electrolyte concentrations were similar here and in the previous study. 16 Reference ranges for adult beagles (8-12 months old) for serum chloride, sodium, and potassium are 105 to 118 mmol/L, 140 to 154 mmol/L, and 4.2 to 5.3 mmol/L, respectively. 22 Other reported reference ranges for serum chloride, sodium, and potassium in dogs are 105 to 115 mmol/L, 141 to 152 mmol/L, and 4.37 to 5.35 mmol/L, respectively. 23 At 4 weeks of age, dogs in this study had serum chloride and sodium values that were slightly lower or within these reported reference ranges and serum potassium values that were slightly higher or within these reported reference ranges. At 13 and 26 weeks of age, electrolyte values were within the reported reference ranges for adult dogs.
In conclusion, this study in neonatal and weanling beagle dogs substantially augments the limited published adrenal hormone data in juvenile beagle dogs. Dogs as young as 2 weeks of age synthesize and secrete adrenal cortical hormones and exhibit a functional HPA axis.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211009492 - Evaluation of Adrenal Cortical Function in Neonatal and Weanling Laboratory Beagle Dogs
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211009492 for Evaluation of Adrenal Cortical Function in Neonatal and Weanling Laboratory Beagle Dogs by Janet M. Petruska, Maria Adamo, Jeffrey McCartney, Ahamat Aboulmali and Thomas J. Rosol in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211009492 - Evaluation of Adrenal Cortical Function in Neonatal and Weanling Laboratory Beagle Dogs
Supplemental Material, sj-docx-2-tpx-10.1177_01926233211009492 for Evaluation of Adrenal Cortical Function in Neonatal and Weanling Laboratory Beagle Dogs by Janet M. Petruska, Maria Adamo, Jeffrey McCartney, Ahamat Aboulmali and Thomas J. Rosol in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-docx-3-tpx-10.1177_01926233211009492 - Evaluation of Adrenal Cortical Function in Neonatal and Weanling Laboratory Beagle Dogs
Supplemental Material, sj-docx-3-tpx-10.1177_01926233211009492 for Evaluation of Adrenal Cortical Function in Neonatal and Weanling Laboratory Beagle Dogs by Janet M. Petruska, Maria Adamo, Jeffrey McCartney, Ahamat Aboulmali and Thomas J. Rosol in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declare the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Janet Petruska is a former employee of and owns stock in PTC Therapeutics. Maria Adamo, Jeffrey McCartney, and Ahamat Aboulmali are employed by Charles River Laboratories. Thomas Rosol served as a paid consultant to PTC Therapeutics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by PTC Therapeutics, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
