Abstract
Mucin-5AC (MUC5AC) is a major secreted mucin in pathogenic airways. To determine its role in mucus-related airway disorders, Muc5ac-deficient (Muc5ac−/− ) and wild-type (Muc5ac+/+ ) mice were compared in bleomycin-induced pulmonary fibrosis, respiratory syncytial virus (RSV) disease, and ozone toxicity. Significantly greater inflammation and fibrosis by bleomycin were developed in Muc5ac−/− lungs compared to Muc5ac+/+ lungs. More severe mucous cell metaplasia in fibrotic Muc5ac−/− lungs coincided with bronchial Muc2, Muc4, and Muc5b overexpression. Airway RSV replication was higher in Muc5ac−/− than in Muc5ac+/+ during early infection. RSV-caused pulmonary epithelial death, bronchial smooth muscle thickening, and syncytia formation were more severe in Muc5ac−/− compared to Muc5ac+/+ . Nasal septal damage and subepithelial mucoserous gland enrichment by RSV were greater in Muc5ac−/− than in Muc5ac+/+ . Ozone exposure developed more severe nasal airway injury accompanying submucosal gland hyperplasia and pulmonary proliferation in Muc5ac−/− than in Muc5ac+/+ . Ozone caused periodic acid-Schiff-positive secretion only in Muc5ac−/− nasal airways. Lung E-cadherin level was relatively lower in Muc5ac−/− than in Muc5ac+/+ basally and after bleomycin, RSV, and ozone exposure. Results indicate that MUC5AC is an essential mucosal component in acute phase airway injury protection. Subepithelial gland hyperplasia and adaptive increase of other epithelial mucins may compensate airway defense in Muc5ac−/− mice.
Introduction
Mucus forms a hydrophilic gel layer over mucosal tissue surface in respiratory, gastrointestinal, and urogenital tracts as well as in visual and auditory systems to provide nonspecific physical and biological barriers for innate defense. The respiratory track from nasal passages to conducting airways are lined by a complex epithelium containing mucus-producing surface mucous (goblet) cells. In addition, numerous submucosal glands in cartilaginous airways are either mucous (viscous, mostly mucus) or serous (watery, mostly with Cl− and HCO3 −) glands that are composed of mucous cells. 1 Club cells (formerly Clara cells) in small airways also known to produce and steadily secrete low amounts of mucin. 2,3 The increased abundance of goblet (mucous) cells (metaplasia or hyperplasia) is a common response to inflammation following exposure to microbial pathogens, particles, and toxicants, resulting in increased mucus production and secretion. 4 In addition, submucosal gland secretions have synergistic effects with surface secretion in airway defense against pathological insults. 5 However, overproduction and hypersecretion of mucus often obstruct airways and contribute to pathogenesis of chronic obstructive pulmonary disease (COPD), asthma, and bronchiectasis and are associated with recurrent infection and morbidity or mortality. 6,7
Mucin is the core glycoprotein of mucus, and MUC5AC and MUC5B are among 22 defined mucin proteins (16 mucins in mice) discovered. 8 Two subfamilies of mucins are membrane-bound mucins (eg, MUC1 and MUC4) and secreted mucin glycoproteins (eg, MUC2 and MUC5AC/B). The secreted mucins are further subdivided into insoluble gel-forming (eg, MUC2 and MUC5AC/B) and soluble mucins. 9,10 Goblet cells in nasal respiratory epithelium to large bronchial epithelium constitutively synthesize, store, and secrete MUC5AC and MUC5B. 2,3 In contrast, submucosal gland acini in large airways primarily synthesize MUC5B, 2,3 which is the most abundant secreted airway mucin in normal airways as the volume of the glands is about 50 times the volume of the surface goblet cells. 2,5,11 However, other evidence indicated that mucous and serous glands also express MUC5AC. 12,13 MUC5AC and MUC5B are also expressed predominantly by club cells in normal and asthmatic airways of mouse and human. 3,5,14 –16 Although the precise molecular mechanisms are not fully understood, investigations have demonstrated multiple molecular mechanisms of airway MUC5AC production. Interleukin (IL)13 increases expression of MUC5AC in human airway epithelial cells and murine models of asthma through sequential activation of signal transducers (eg, Janus kinase 1, signal transducer, and activator of transcription 6). 2,17 Epidermal growth factor receptor (EGFR) signaling also induced MUC5AC via phosphoinositide 3-kinase (PI3K)-transmembrane protein 16A signaling pathway. 18 In contrast, Foxa2 inhibited by IL-13 signaling negatively regulated Muc5ac. 17,19 We also demonstrated that MUC5AC expression is regulated, in part, by a cycloocygenase 2 and prostaglandin E2 pathway in human airway epithelial cells. 20 MUC5AC is abundant not only in airways but also in the superficial gastric epithelium. 21 It is also detected in pancreas, colon, gallbladder, and endocervix, and highly associated with various cancers. 22
The nasal epithelium is the first airway barrier between the host and the external environment and largely covered by a mucus gel for upper airway defense. The mucous membranes, or mucosa, lining the nasal airways consist of 2 layers: the surface epithelium and the underlying lamina propria which is connective tissue rich in subepithelial glands and blood vessels. 23 Goblet cells in the respiratory epithelium lining the proximal septum and subepithelial glands under respiratory (seromucous glands) and olfactory epithelium (Bowman glands) are cellular source of nasal mucus. 5 Gel-forming mucins in human nasal mucous glands are mainly MUC5B and MUC19, while MUC2 and MUC5AC are secreted from both mucous and serous glands. 12,13,24,25 Submucosal glands in tracheobronchi were increased in number and volume in smokers relative to nonsmokers and extended to the distal bronchioles in disease conditions such as chronic bronchitis which contributes to the formation of airway plugs. 5,26
Recent investigations have focused on molecular biology underlying single airway mucins. The Muc5ac transgenic mouse models demonstrated a protective role of against pulmonary influenza virus infection 16 or detrimental roles in allergic airway disease and ventilator-induced acute lung injury. 27,28 However, functions of MUC5AC in other mucus-engaged pulmonary pathogenesis or in nasal airway injury caused by airborne toxicants have not been investigated. The current study was designed to test the hypothesis that MUC5AC is protective in murine models of airway diseases which accompany increased mucus production and/or secretion and enhanced MUC5AC expression. For this purpose, we exposed wild-type mice (Muc5ac+/+ ) and mice genetically deficient in Muc5ac (Muc5ac−/− ) to a pulmonary fibrogen (bleomycin), respiratory syncytial virus (RSV), and subacute ozone (O3) and compared upper and lower respiratory injury, inflammation, and mucous production/secretion between 2 mouse genotypes.
Materials and Methods
Generation of Muc5ac Deficient (Muc5ac−/− ) Mice
Muc5ac −/− (B6; 129-Muc5actm1Unc /Ehs) mice were generated by targeted deletion of exons 21 to 31 encoding the MUC5AC protein region responsible for glycosylation. A targeting construct consisting of a neomycin-positive and herpes simplex virus thymidine kinase-negative selection cassette (NeoTKXho) in place of an 8 kilobases (8031 kb) spanning exon 21 to exon 31 of the Muc5ac gene (Supplemental Figure S1-A) was made at the National Institute of Environmental Health Sciences (NIEHS). The construct was introduced into embryonic stem cells (ES-E14TG2a) to generate targeted mice at the Mouse Model Core of the University of North Carolina at Chapel Hill (Supplemental Figure S1-A). The targeted embryonic stem cells were identified by Southern blot analysis and injected into C57BL/6J mice blastocysts which were transplanted into pseudopregnant females. The chimeric offspring obtained were then bred to C57BL/6J mice and the pups screened by Southern blotting. Heterozygous (Muc5ac+/− ) mice were transferred to the NIEHS animal care facilities and backcrossed into C57BL/6J mice for 8 generations to produce whole body knockout mice and wild-type controls (C57BL/6J-129Ola, Muc5ac+/+ ). Muc5ac+/+ and Muc5ac+/+ mice were identified by Southern blotting (Supplemental Figure S1-B), polymerase chain reaction (PCR; Supplemental Figure S1-C), immunohistochemical analyses (Supplemental Figure S2-A to C), and Western blotting (Supplemental Figure S2-C). Pathogen-free breeding colonies of Muc5ac+/+ , Muc5ac−/− , and Muc5ac+/− mice were maintained in sterilized, filter-topped cages and fed autoclaved food (NIH_31) at the NIEHS animal facilities. All animal use was approved by the NIEHS Animal Care and Use Committee.
Animal Experiments
Male, adult (6-8 weeks old) Muc5ac+/+ and Muc5ac−/− mice were used for all studies. Mice were randomly assigned to the experimental groups. For pulmonary fibrosis end points, mice were anesthetized with isoflurane (2% in air), and a single dose (0.5 U/kg body weight [bw]) of bleomycin sulfate (Sigma Chemical Co) or vehicle (PBS) was delivered by oropharyngeal aspiration (intratracheal). Mice were killed at 3, 7, or 14 days postbleomycin. For the RSV disease model, mice were anesthetized with isoflurane and intranasally infected with 106 plaque-forming units of human RSV-19 strain (ViraSource) in 50 µL Hanks balanced salt solution (HBSS). HBSS containing Hep-2 cell lysates (50 μL) was intranasally instilled into control mice as vehicle. Animals were killed at 1, 3, 5, or 7 days after intranasal instillation. To evaluate airway O3 toxicity, mice were placed in individual stainless-steel wire cages within a whole-body inhalation chamber (Hazelton 1000; Lab Products) with free access to water and foods and exposed continuously to 0.3 parts per million (ppm) O3 for 48 or 72 hours under constant chamber air temperature (72 °F ± 3 °F) and relative humidity (50% ± 15%) as described in detail previously. 29 Parallel exposure to room air was done in a separate chamber for the same duration. All mice were euthanized by sodium pentobarbital overdose (104 mg/kg bw) for necropsy immediately after the end of designated exposure. Body fat of mice was measured at 6, 9, and 12 months of age using a dual energy X-ray absorptiometry (Lunar PIXImus, GE Lunar Corp).
Bronchoalveolar Lavage Analyses
The right lung of each mouse was lavaged in situ, 4 consecutive times with HBSS (0.5 mL/25 g bw). The bronchoalveolar lavage (BAL) fluid returns were centrifuged (1000g, 10 minutes). An aliquot of the first BAL return supernatants (50 μL) was analyzed for total protein concentration (a marker of edema) in Bradford reagent (Bio-Rad) as indicated in the manufacturer procedure. Cell pellets from all lavage returns were combined and resuspended in 1 mL HBSS for epithelial and inflammatory cell counts as reported previously. 30 To detect cell lysis and toxicity, colorimetric assay for lactate dehydrogenase (LDH) was also performed in aliquots (25 μL) of BAL supernatants by determination of NADH production from added NAD+ (Sigma).
Sandwich Enzyme-Linked Immunosorbent Assay
The BAL supernatants (25 μL) were used to determine neutrophil myeloperoxidase (MPO) using a mouse-specific enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems) according to the manufacturer instructions. Secreted MUC5AC protein was also determined in BAL supernatants following the published method. 29 Briefly, an aliquot of BAL supernatant (20 μL) was loaded in each well of an ELISA plate containing a goat polyclonal anti-MUC5AC capture antibody (1:40 dilution of sc-16903; Santa Cruz Biotechnology Inc) in pH 9.5 bicarbonate–carbonate coating buffer (BD OptEIA Reagent; BD Biosciences). The plate was incubated at 48 °C until the reaction was dry (>5 hours). The wells were washed and blocked overnight with an assay diluent containing 10% fetal bovine serum (BD Opt EIA) at 4 °C. The samples were then incubated with a biotinylated monoclonal anti-MUC5AC detection antibody (1:100 of Clone 45M1, Thermo Scientific) for 1.5 hours at 37 °C. Following incubation with a peroxidase-conjugated secondary antibody (1:2500, goat antimouse-immunoglobulin G [IgG]-horseradish peroxidase), color change was developed by adding the 3,3′,5,5′-tetramethylbenzidine substrate solution (BD Biosciences). Optical density was measured at 450 nm after the stop buffer was added.
Lung and Nasal Airway Histopathology and Immunohistochemistry
The left lung from each mouse was inflated intratracheally in situ with 10% neutral buffered formalin and fixed following procedures published previously. 31 The fixed lung lobe was sectioned at proximal (generation 5) and distal (generation 11) levels of the main axial airway. 32 The excised heads of mice from RSV and O3 studies were flushed retrograde via a cannula through the nasopharyngeal orifice with 10% neutral buffered formalin. The fixed heads were processed for decalcification, and the proximal aspect of the nasal cavity section (ie, at the level of upper incisor teeth, T1) was cut following procedures published previously. 33 The lung and nasal tissue blocks were embedded in paraffin, and 5 μm-thick section cuts were stained with hematoxylin and eosin (H&E) for histopathological analysis, with Alcian blue (pH 2.5)/periodic acid-Schiff (AB/PAS) reagent for acidic and neutral mucus detection and with Mason trichrome for collagen localization. Severity of histopathologic lung toxicity in the bleomycin study was determined by blinded microscopic evaluation of H&E-stained slides for inflammation and injury and Masson-trichrome-stained slides for collagen associated with areas of inflammation (Experimental Pathology Labs). Tissues were also stained by immunohistochemical methods using mouse monoclonal (IgG1) anti-MUC5AC (1:100 dilution of Clone 45M1, Thermo Fisher Scientific), rabbit anti-MUC2 (1:667 dilution of GTX100664, GeneTex), goat anti-RSV (1:200 dilution of ab20745, Abcam), and proliferating cell nuclear antigen (PCNA; 1:200 dilution of PCNA; sc-56, Santa Cruz Biotechnology). Briefly, deparaffinized and hydrated tissue sections on microscope slides were treated sequentially with antigen unmasking solution (Vector Laboratories), 0.1% proteinase K, 1 N HCl (1 hour for PCNA only), and endogenous peroxidase quenching solution (5% H2O2) before blocking with 1.5% serum (Vectastain ABC kits, Vector Laboratories) at room temperature. Tissue sections were then incubated overnight at 4 °C with each specific primary antibody. After incubation with proper biotinylated secondary antibodies (1:200, Vectastain ABC kits) and avidin/biotin solution, antigens were detected by 3,3′-diaminobenzidine-peroxidase substrate solution (1 minute for PCNA, 5 minutes for RSV, 10 minutes for others, Sigma), and slides were mounted with cover glasses after counterstaining, if necessary, and dehydration. For immunofluorescence staining of RSV, micrograph slides incubated with primary antibody were incubated overnight with fluorochrome-conjugated goat IgG solution (1 μg/mL) for 1 hour and cover-slipped with prolong Gold Antifade reagent with 4′,6-diamidino-2-phenylindole (Thermo Fisher Scientific). Nasal airways and lungs from a subset of male mice (22 Muc5ac+/+ , 23 Muc5ac−/− ) at 11 to 12 months were processed for H&E and AB/PAS staining as indicated above. Occurrence of preinvasive alveolar epithelial proliferation was determined by percentage of mice with increased focal cellularity in alveoli including early adenoma on H&E-stained light micrographs. Nodular lymphocytic accumulation was determined by percentage of mice bearing lymphocyte nodules in perivascular-peribronchial pulmonary regions on H&E-stained light micrographs.
Morphometric Analysis of Nasal Airway Mucosubstances and Terminal Bronchial Proliferating Cells
The amount of AB/PAS-stained mucosubstances in nasal airways from RSV and O3 studies was estimated using computerized image analysis and standard morphometric techniques. 33,34 Briefly, images of AB/PAS-stained cross-sections from the proximal nasal airway (turbinate section T1) were taken using a light microscope with an attached camera and software (Olympus BX53 and cellSense, Olympus). The AB/PAS-positive areas within the surface epithelium lining mid-septum and ventral septum (from the tip level of nasoturbinate to the bottom of the ventral septum) were calculated from the semiautomatically circumscribed perimeter of the stained material using the NIH Image J 1.52a software (https://imagej.nih.gov/ij/index.html). The length of the basal lamina underlying the surface epithelium was concomitantly calculated from the contour length on digitized images using the same system. Similarly, stored PAS-positive substances in subepithelial glands (mucous glands) in the ventral septum (from the bottom level of maxilloturbinate to the end of the ventral septum) were quantitated and normalized to the length of basal lamina on top of the lamina propria. The volume of intraepithelial mucosubstances per unit surface area (volume density, nL/mm2 basal lamina) was determined as previously described in detail. 35 Areas of PAS-positive secretion in the ventral wall region of proximal nasal sections from the O3 study were also measured as indicated above.
In Situ Hybridization of Airway Mucins
Expression of Muc4 and Muc5b transcripts in lung and nasal tissues was detected by in situ RNA detection techniques in accordance with manufacturer instructions (FFPE assay kit; Advanced Cell Diagnostics). Briefly, formalin-fixed paraffin-embedded tissue sections on charged slides were baked at 60 °C (1 hour for lung, overnight for nasal) and deparaffinized and rehydrated. The sections were pretreated with hydrogen peroxide, incubated in boiling target retrieval solution for 15 minutes, and permeabilized with protease plus solution (40 °C, 30 minutes for lung, 15 minutes for nasal). Tissues were then hybridized with labeled double z RNA probe pairs specific for mouse Muc4 and Muc5b (40 °C for 2 hours). The target messenger RNA (mRNA) signals were sequentially amplified using a horseradish peroxidase-based signal amplification system, and color development was detected with 3,3′-diaminobenzidine. The slides were counterstained with hematoxylin for 10 seconds. Representative light photomicrographs were prepared by Olympus microscope BX53 and cellSense software.
Lung RNA Isolation and PCR
Total lung RNAs were isolated from lung homogenates (RNeasy Mini Kit) and reverse transcribed into complementary DNAs (cDNAs). Aliquots of cDNA (40 ng) were subjected to quantitative PCR with mouse-specific primers for Muc5ac (amplifying nucleotide 3547 to 3667 of NM_010844; 5′-TGCATGCGTACCTGCCAGAA-3′, 5′-CACACTGCATTGTGCCCTCA-3′), Muc5b, 36 Muc4 (RealTimePrimers), and Muc2, 36 and 18s ribosomal RNA, an internal control (amplifying nucleotide 324-507 of K1364; 5′-TACCTGGTTGATCCTGCCAG-3′, 5′-CCGTCGGCATGTATTAGCTC-3′) using CFX Connect Realtime System (Bio-Rad). Copy number of RSV nucleoprotein (N) was determined by droplet digital PCR. Briefly, an aliquot of lung cDNA (2 ng) in PCR mixture containing a SYBR Green dye (Bio-Rad) and 125 nM of RSV N gene primers (amplifying nucleotide 1137-1220 of M11486; 5′-AGATCAACTTCTGTCATCCAGCAA-3′, 5′-TTCTGCACATCATAATTAGGAGTATCAAT-3′) was partitioned into approximately 20,000 individual nanoliter-sized water-in-oil droplets by Bio-Rad QX200 Automated Droplet Generator followed by PCR reaction (C1000 Touch Thermal Cycler, Bio-Rad). The droplets were then read one-by-one in QX200 Droplet Reader (Bio-Rad) and assigned as positive (1) or negative (0). The copy number of the N gene per µg RNA was calculated by the positive droplets.
Western Blot Analyses
Protein level of epithelial cadherin (E-cadherin, 135 kDa for precursor and 120/80 kDa for matured), an intercellular adherens junction protein in epithelial barrier, was determined. Total lung proteins were isolated from individual mouse lung homogenate in radioimmunoprecipitation assay buffer (n = 4/group), and equal amounts of the lung proteins from 2 mice were pooled. Aliquots of pooled proteins (70-150 μg) were separated on Tri-HCl sodium dodecyl sulfate polyacrylamide gel electrophoresis genes (7.5% or 10%-20%, Bio-Rad) and analyzed by routine Western blotting using specific antibodies (Santa Cruz Biotechnology) against E-cadherin (sc-59778, 1:200 dilution) and pan-actin (sc-1616, 1:500 dilution). Blot images were scanned using the FluorChem HD2 System (ProteinSimple) and quantified by densitometry using Image J Gel analysis software (https://imagej.nih.gov).
Sircol Assay for Lung Collagen Quantitation
Total soluble collagen content in right lungs was determined using the Sircol collagen-dye binding assay kit (Biocolor). Briefly, whole right lung lobes from each mouse were homogenized in 1 mL of acetic acid solution (0.5 N with protease inhibitors) and incubated on a gentle shaker overnight at room temperature. The acid extracts were centrifuged (15,000g for 30 minutes), and aliquots of the supernatants (200 μL) containing acid-soluble collagens were mixed with 1 mL Sirius Red dye reagent for 30 minutes on a shaker (800 rpm). The pellets (bead-dye-collagen complex) acquired by centrifugation (10,000g for 10 minutes) were dissolved in 1 mL alkali reagent (0.5 M NaOH). Absorbance of alkali reagent in which collagen-dye complex was dissolved was measured at 550 nm. The amount of collagen was quantitated using a standard curve prepared from collagen standard.
Serum IgE Detection
From each mouse, an aliquot of serum (1 μL) was isolated from blood samples collected by cardiac puncture and was diluted in an assay diluent (1:100). The immunoglobulin E (IgE) levels were determined using a mouse-specific sandwich ELISA kit (BD Opt EIA) and similarly prepared serial dilution of IgE standards (BD Biosciences).
Statistics
Data were expressed as the group mean ± standard error of the mean. Two-way analysis of variance (ANOVA) was used to evaluate the effects of genotype (Muc5ac+/+ vs Muc5ac−/− ) and exposure (RSV vs vehicle, bleomycin vs vehicle, O3 vs air) at each time point. One-way ANOVA was used to assess MUC5AC secretion or Muc5ac mRNA expression in Muc5ac+/+ mice. Kruskal-Wallis ANOVA on Ranks test was applied to the histology score data. The Student-Newman-Keuls test was used for a posteriori comparisons of means for all multiple comparisons (P < .05). All statistical analyses were performed using SigmaPlot 13.0 program (Systat Software).
Results
Enhanced Responsivity of Muc5ac−/− Mice to Bleomycin-Induced Pulmonary Injury and Fibrosis
The bw gain at 14 days after bleomycin treatment was significantly lower in Muc5ac−/− mice compared to Muc5ac+/+ mice (Figure 1A). Relative to respective controls, bleomycin significantly increased the BAL protein concentration (a marker of protein edema), LDH activity (a marker of cell death), and the numbers of total cells and leukocytes (neutrophils and lymphocytes) in Muc5ac+/+ and Muc5ac−/− mice (Figure 1B-F). However, bleomycin-induced protein edema and lung cell death (7-14 days) as well as numbers of lung leukocytes (3-14 days) in Muc5ac−/− mice were significantly greater than those in Muc5ac+/+ mice (Figures 1B-F).
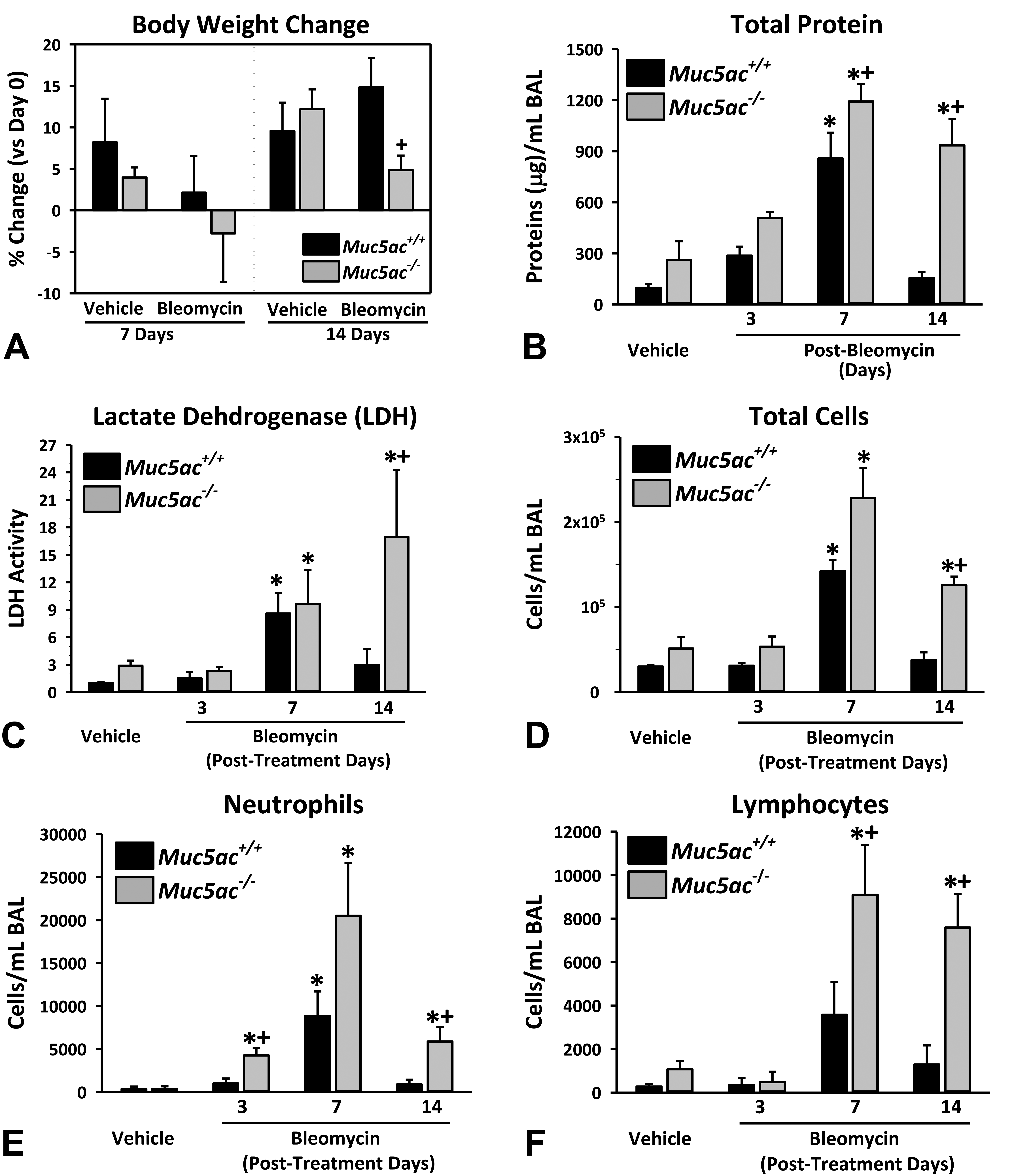
Enhanced bleomycin-induced lung injury in mucin-5AC-deficient (Muc5ac−/− ) mice: Body weight change (A) and bronchoalveolar lavage (BAL) analysis for protein concentration (B) and lactate dehydrogenase concentration (C) as well as the number of total cells (D), neutrophils (E), and lymphocytes (F) were determined in wild-type (Muc5ac+/+ ) and Muc5ac−/− mice after vehicle or bleomycin instillation. Data presented as mean ± standard error of the mean (n = 3-7 mice/group for body weight, 6-8 mice/group for vehicle-BAL, n = 3/group for 3-day bleomycin-BAL, n = 5-7 for 7- and 14-day bleomycin-BAL). Graph symbols: *P < .05 versus genotype-matched vehicle control mice; †P < .05 versus exposure-matched Muc5ac+/+ mice.
Histopathologic analysis found that bleomycin caused sporadic fibrotic patches in parenchyma with predominant fibrogenesis in perivascular–peribronchial regions from 7 days and more widespread fibrosis at 14 days postbleomycin in both genotypes (Figure 2). Relative to Muc5ac+/+ mice, the magnitude of the bleomycin-induced fibrosis was more severe and widespread in Muc5ac−/− mice at 7 to 14 days (Figure 2). Histopathologic scores of inflammation and fibrosis as well as lung soluble collagen contents (collagens type I-V) were significantly higher in Muc5ac−/− mice than in Muc5ac+/+ mice at 14 days (Figure 3A). In Muc5ac+/+ lung, Muc5ac mRNA abundance (3 days) and airway secretion (7 days) were significantly enhanced by bleomycin (Figure 3B). The E-cadherin levels determined as an intercellular junction marker were significantly lower in vehicle (PBS)-treated Muc5ac−/− mice than in vehicle-treated Muc5ac+/+ mice (Figure 3C). Bleomycin treatment significantly decreased amounts of mature lung E-cadherin (80kDa) in both genotypes of mice but more markedly in Muc5ac−/− mice than in Muc5ac+/+ mice at 3 and 14 days (Figure 3C).
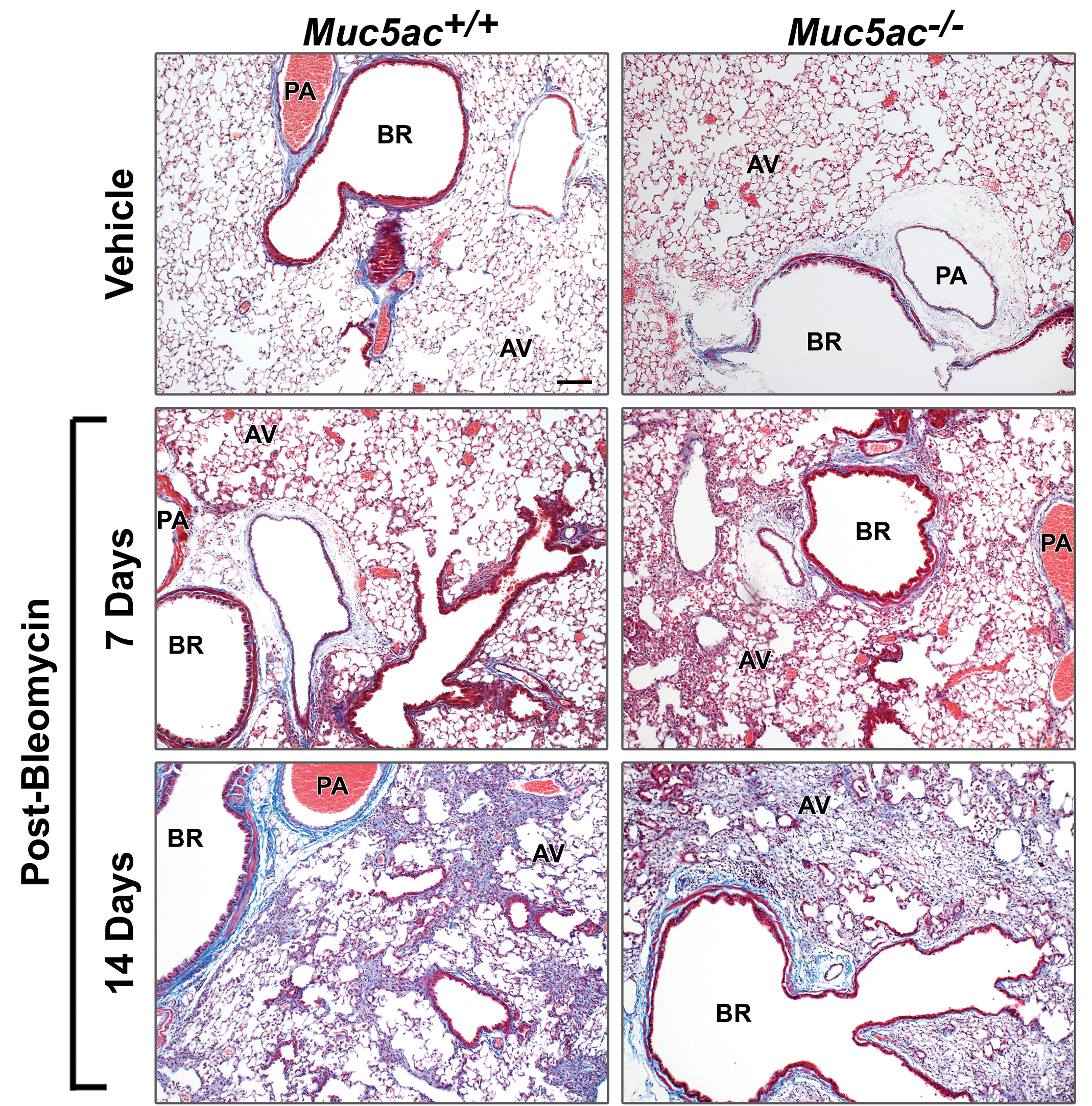
Augmented bleomycin-induced pulmonary histopathology in mucin-5AC-deficient (Muc5ac−/− ) mice. Masson trichrome staining exhibits differential progress and severity of pulmonary fibrosis and collagen accumulation between wild-type (Muc5ac+/+ ) and Muc5ac−/− mice at 7 and 14 days postbleomycin or at 14 days post vehicle. Representative light photomicrographs of lung sections (n = 6-7/group) were presented. Bar = 100 μm. AV indicates alveoli; BR, bronchi or bronchiole; PA, pulmonary artery.
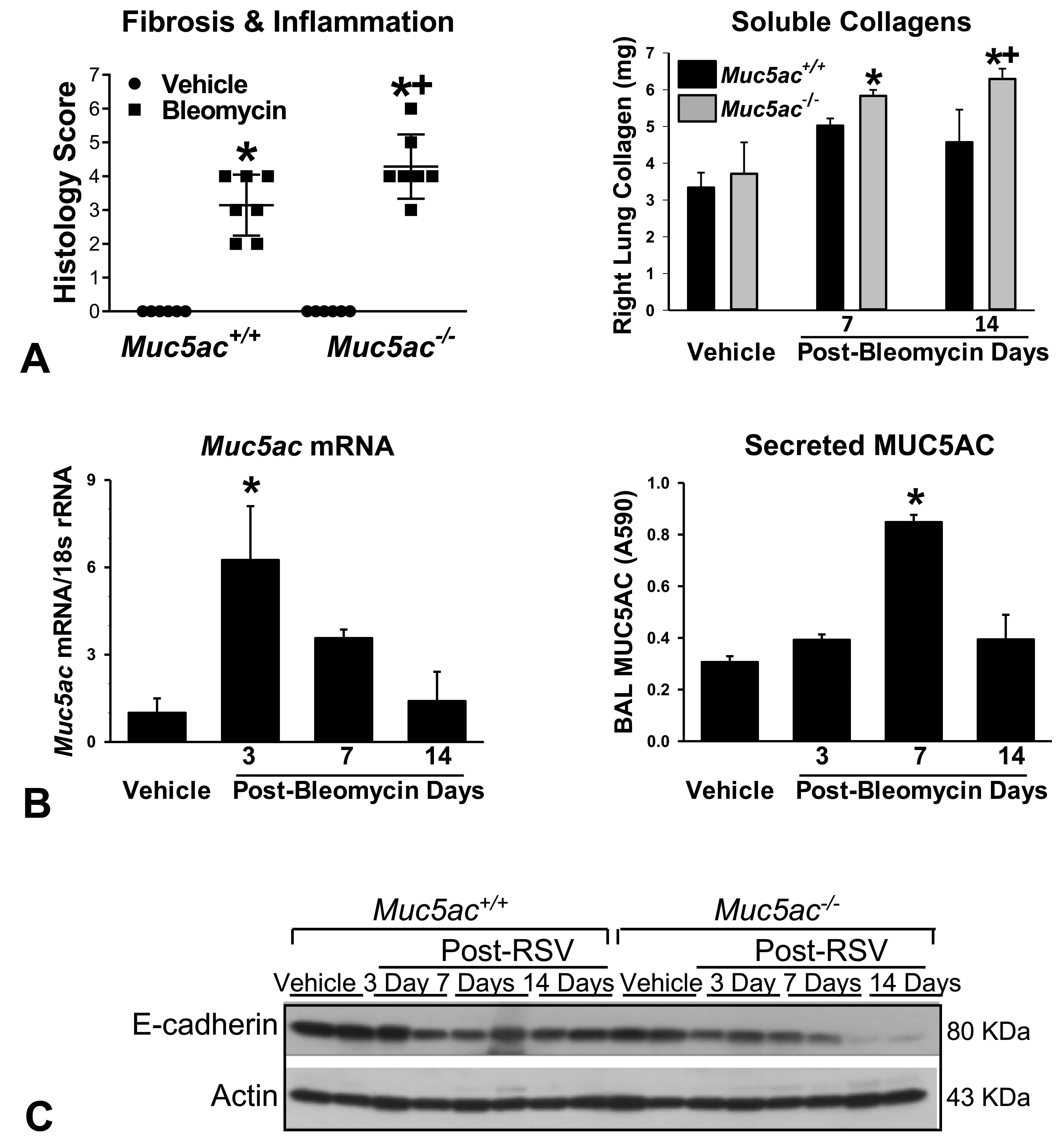
Bleomycin-induced changes in fibrotic lung injury, mucin-5AC (MUC5AC), and epithelial barrier expression in mucin-5AC-sufficient (Muc5ac+/+ ) and -deficient (Muc5ac−/− ) mice. A, Severity of histopathologic lung inflammation and fibrosis at 14 days postbleomycin was scored by microscopic evaluation of H&E and Masson trichrome-stained slides (n = 6-7/group). Total soluble collagen contents were determined in whole right lung lysates using a colorimetric Sirius Red dye-collagen binding assay. Mean ± SEM presented (n = 4/group). B, Pulmonary Muc5ac mRNA expression was determined by qRT-PCR using a mouse specific Muc5ac primer set in Muc5ac+/+ mice (n = 3/group). Secreted Muc5ac level was determined in the aliquots of bronchoalveolar lavage supernatants from Muc5ac+/+ mice using a sandwich enzyme-linked immunosorbent assay (n = 3-4/group). Mean ± SEM presented. C, Protein level of E-cadherin in aliquots of lung homogenates was determined by Western blot analysis using actin as an internal control (n = 2 pooled samples/group). Representative digitized images from duplicate assays are presented. Intensity of blot images was quantified by densitometry (n = 2/group, mean ± SEM presented). Graph symbols: *P < .05 versus genotype-matched vehicle control mice, †P < .05 versus exposure-matched Muc5ac+/+ mice. H&E indicates hematoxylin and eosin; mRNA, messenger RNA; qRT-PCR, quantitative real-time polymerase chain reaction; SEM, standard error of the mean.
Bleomycin caused mucous (goblet) cell metaplasia (MCM) in Muc5ac+/+ and Muc5ac−/− mice at 7 and 14 days (Figure 4A). Microscopic analysis indicated that MUC5AC proteins were localized in goblet cells of bronchi and bronchioles after vehicle or bleomycin in Muc5ac+/+ mice and were not detected in Muc5ac−/− mice (Figure 4A, insets). Although basal level of AB/PAS-stained bronchial intraepithelial mucus was relatively scarce in Muc5ac−/− lungs compared to Muc5ac+/+ lungs, the overall magnitude of bleomycin-induced MCM was greater in Muc5ac−/− lungs than in Muc5a+/+ lungs as MCM was more widely extended to smaller airways adjacent to fibrotic regions (at 14 days) in Muc5ac−/− mice compared to Muc5a+/+ mice (Figure 4A), suggesting a compensatory increase of non-MUC5AC mucin in Muc5ac−/− airways. Expression of MUC2 protein, Muc5b mRNA, and Muc4 mRNA were determined as these mucins were known to increase in pathologic airways 37 –39 and compensate for Muc5ac deficiency. 15,36,40,41 In situ hybridization (ISH) detected Muc4 and Muc5b mRNAs in vehicle-treated bronchial epithelium of both genotypes (Figure 4A). Bleomycin increased Muc4 and Muc5b transcripts in bronchial-to-bronchiolar epithelium of fibrogenic regions, and their increase was colocalized with MCM throughout the conducting airways (Figure 4A). High Muc4 expression was detected in honeycombing alveoli of Muc5ac−/− mice at 14 days postbleomycin and it was minimal throughout epithelium at 7 days postbleomycin (Figure 4A). Induced expression of Muc4 and Muc5b mRNA markedly in proximal-to-distal conducting airway epithelium was higher in Muc5ac−/− mice than in Muc5ac+/+ mice at 7 to 14 days, indicating compensatory overexpression (Figure 4A). Immunohistochemical staining found that MUC2 proteins were constitutively expressed in AB/PAS-positive bronchial-to-bronchiolar epithelium of both strains and it was increased by bleomycin in Muc5ac−/− mice at 7 days (Figure 4A). Quantitative PCR detected significantly greater expression of Muc5b (14 days), Muc4 (7 and 14 days), and Muc2 (3 days) messages in Muc5ac−/− mice than in Muc5ac+/+ mice after bleomycin treatment (Figure 4B). Upregulation of Muc2 mRNA by bleomycin treatment preceded (at 3 days) the increase of MUC2 proteins (at 7 days) in Muc5ac−/− mice (Figure 4A and B). Overall, these results indicated that MUC4 and MUC5B together with MUC2 contribute to bleomycin-induced MCM in Muc5ac−/− mice.
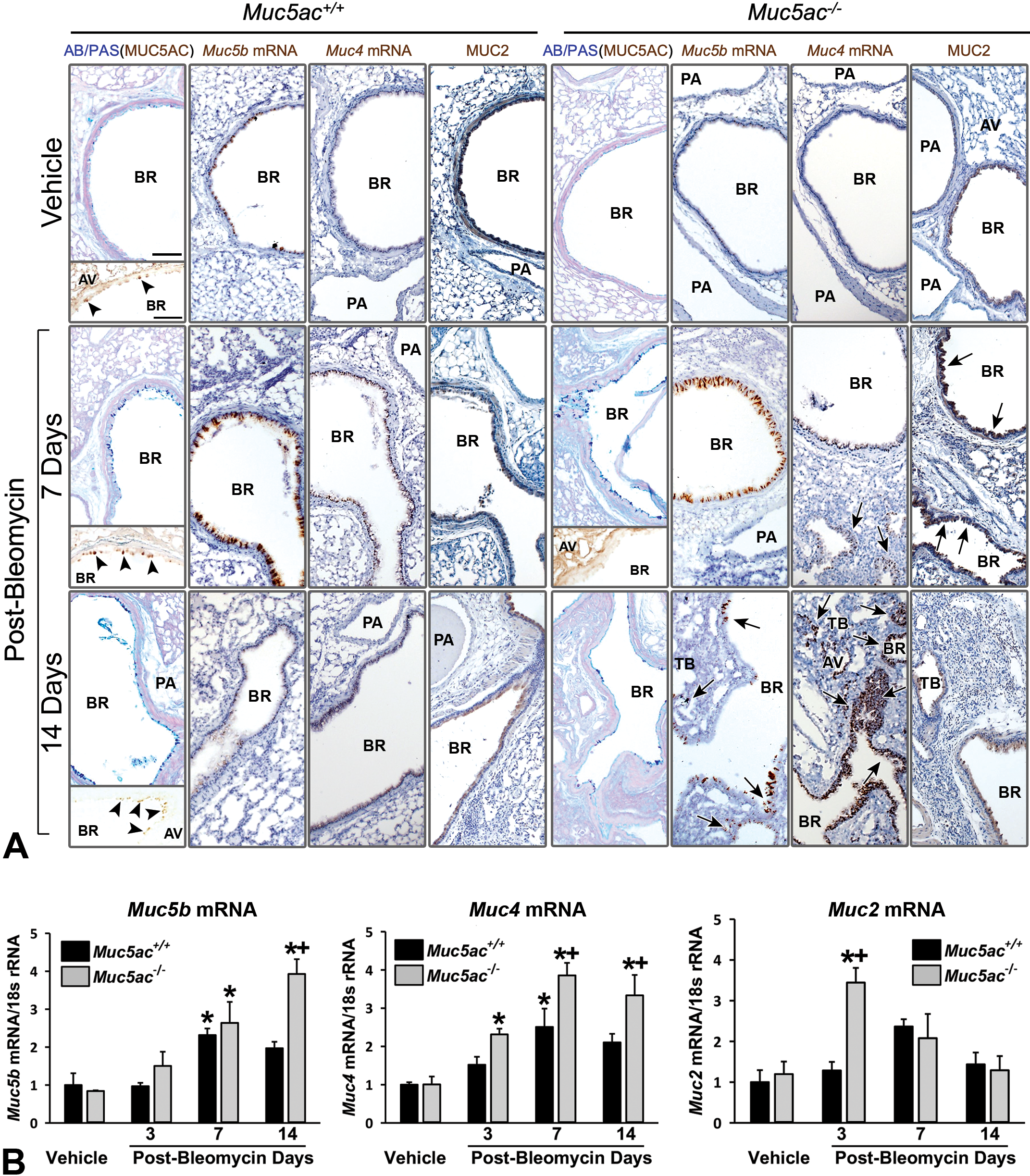
Bleomycin-induced compensatory mucin overexpression in mucin-5AC-deficient (Muc5ac−/− ) mouse lungs. A, Mucus in conducting airways was visualized by AB/PAS-stained proximal aspect of left lung sections from vehicle (14 days) and bleomycin (7 and 14 days)-treated mice. Expression and localization of single mucin were determined by immunohistochemical staining (IHC) using specific antibodies for MUC5AC (insets in AB/PAS) and MUC2 or in situ hybridization (ISH) using specific probes for Muc5b and Muc4 on the serial cuts of the same tissue blocks. Arrow heads indicate MUC5AC proteins localized in bronchial epithelium of Muc5ac+/+ mice. Arrows indicate more markedly increased Muc4 and Muc5b message or MUC2 protein in Muc5ac−/− mice compared to those in Muc5ac+/+ mice. Representative light micrographs presented. Bars = 100 μm. AV = alveoli, BR = bronchi or bronchiole, PA = pulmonary artery, TB = terminal bronchiole. (B, Pulmonary Muc5b, Muc4, and Muc2 mRNA expressions were determined by qRT-PCR using mouse-specific primer sets (n = 3/group). Mean ± SEM presented. Graph symbols: *P < .05 versus genotype-matched vehicle control mice; †P < .05 versus exposure-matched Muc5ac+/+ mice. AB/PAS indicates Alcian blue/periodic acid-Schiff; mRNA, messenger RNA; qRT-PCR, quantitative real-time polymerase chain reaction; SEM, standard error of the mean.
Enhanced Sensitivity of Muc5ac−/− Mice to RSV
Lung injury by RSV
Respiratory syncytial virus infection caused significant airway inflammation and bronchial and bronchiolar epithelial hyperplasia and hyperproliferation in Muc5ac+/+ and Muc5ac−/− mice (Figure 5A). Large syncytia (a multinucleated cell resulted from fusions of uninuclear cells by infected RSV) formation was more evident in lungs from Muc5ac−/− mice (Figure 5A, arrow heads). Compared to Muc5ac+/+ lungs, RSV-induced increases in the numbers of BAL epithelial cells and the concentration of neutrophilic MPO were significantly greater in Muc5ac−/− lungs at 1-day post-RSV (Figure 5B). Consistent with more severe airway epithelial hypertrophy/hyperplasia and smooth muscle thickening (Figure 5A, arrows), serum IgE level was significantly higher in Muc5ac−/− mice than in Muc5ac+/+ mice at 5 days post-RSV (Figure 5C).
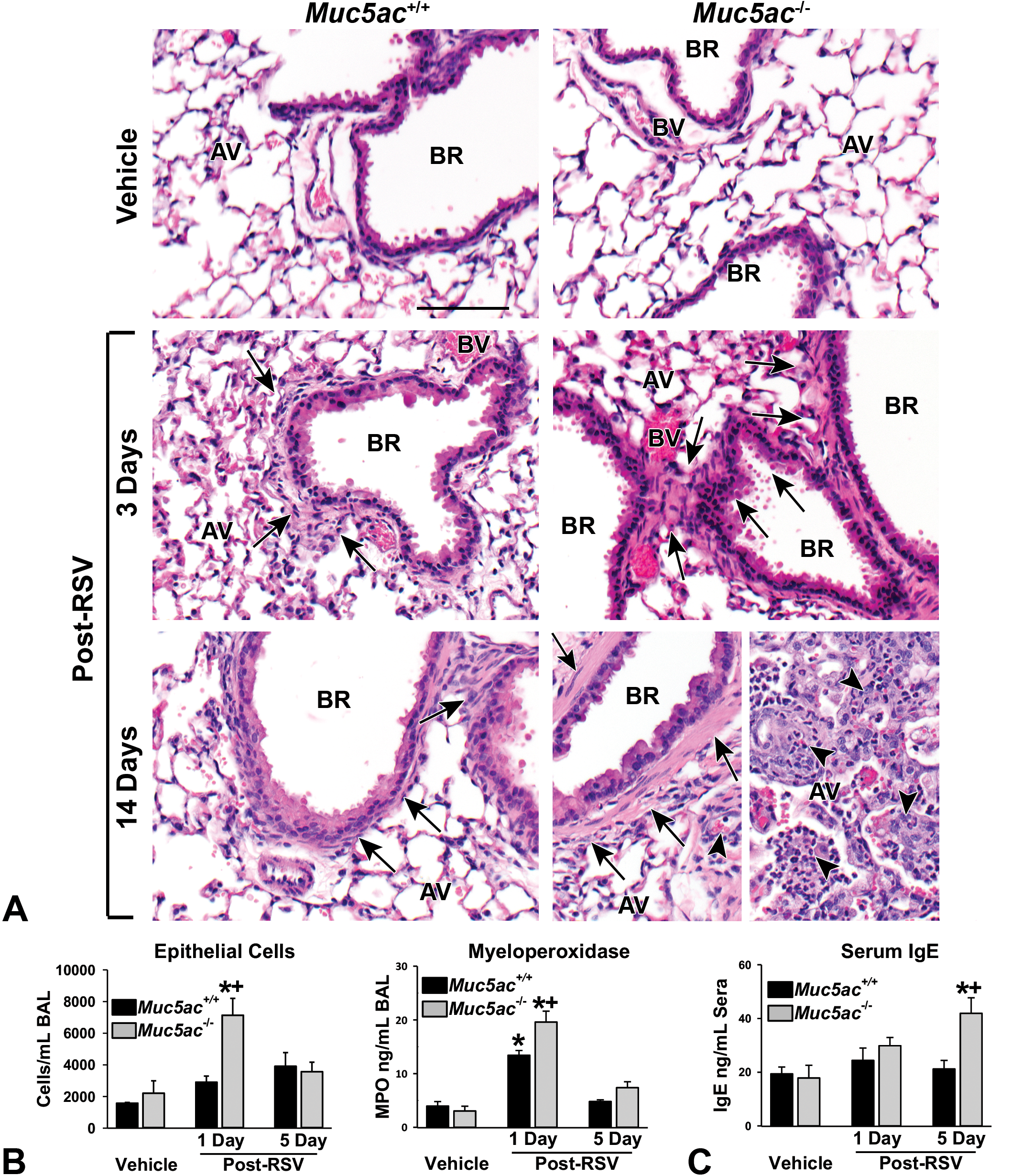
Exacerbated respiratory syncytial virus (RSV)-induced pulmonary injury and airway narrowing in mucin-5AC-deficient (Muc5ac−/− ) mice. A, Differential severe bronchial/bronchiolar hyperplasia and hypertrophy and smooth muscle thickening in Muc5ac−/− mice than in Muc5ac+/+ mice demonstrated by H&E staining of proximal aspect of left lung after 3 to 5 days post-RSV infection or 3 days post vehicle. Large syncytia (multinucleated cells) formation and lymphocytic inflammation in alveoli were detected more markedly in Muc5ac−/− mice at 5 days post-RSV. Representative light photomicrographs presented (n = 5-8/group). Arrows indicate perivascular-peribronchiolar edema and smooth muscle hyperplasia. Arrow heads indicate syncytia. AV = alveoli, BR = bronchi or bronchiole. Bar = 100 μm. B, Significantly higher number of epithelial cells and concentration of neutrophil myeloperoxidase (MPO) in bronchoalveolar lavage (BAL) fluids from Muc5ac−/− mice compared to those in Muc5ac+/+ after 1-day post-RSV infection. Mean ± SEM (n = 4-8/group for epithelial cells, n = 4-5/group for MPO) presented. C, Immunoglobulin E (IgE) level determined in aliquots of serum (1 μL) using a colorimetric enzyme-linked immunosorbent assay kit. Mean ± SEM (n = 3/group) presented. Graph symbols: *P < .05 versus genotype-matched vehicle control mice; † P < .05 versus exposure-matched Muc5ac+/+ mice. H&E indicates hematoxylin and eosin; SEM, standard error of the mean.
Immunohistologic localization of RSV indicated that viral proteins were predominantly in pleura, conducting airway epithelium, and blood vessels at 1-day post-RSV and more severe viral pleuritis was found in Muc5ac−/− mice than in Muc5ac+/+ mice (Figure 6A). By 3 days post-RSV, virus diffused throughout the lung parenchyma in both genotypes, but infection diminished by 5 days (Figure 6A). There was little difference in the magnitude of lung RSV localization between 2 genotypes at 3 and 5 days (Figure 6A), suggesting that Muc5ac may contribute to a barrier against RSV in early stages of viral entry and spread. Corresponding to RSV immunohistochemistry, viral nucleoprotein (N) gene copy number was significantly higher in Muc5ac−/− lung than in Muc5ac+/+ lung at 1-day post-RSV but not at later times (Figure 6B).
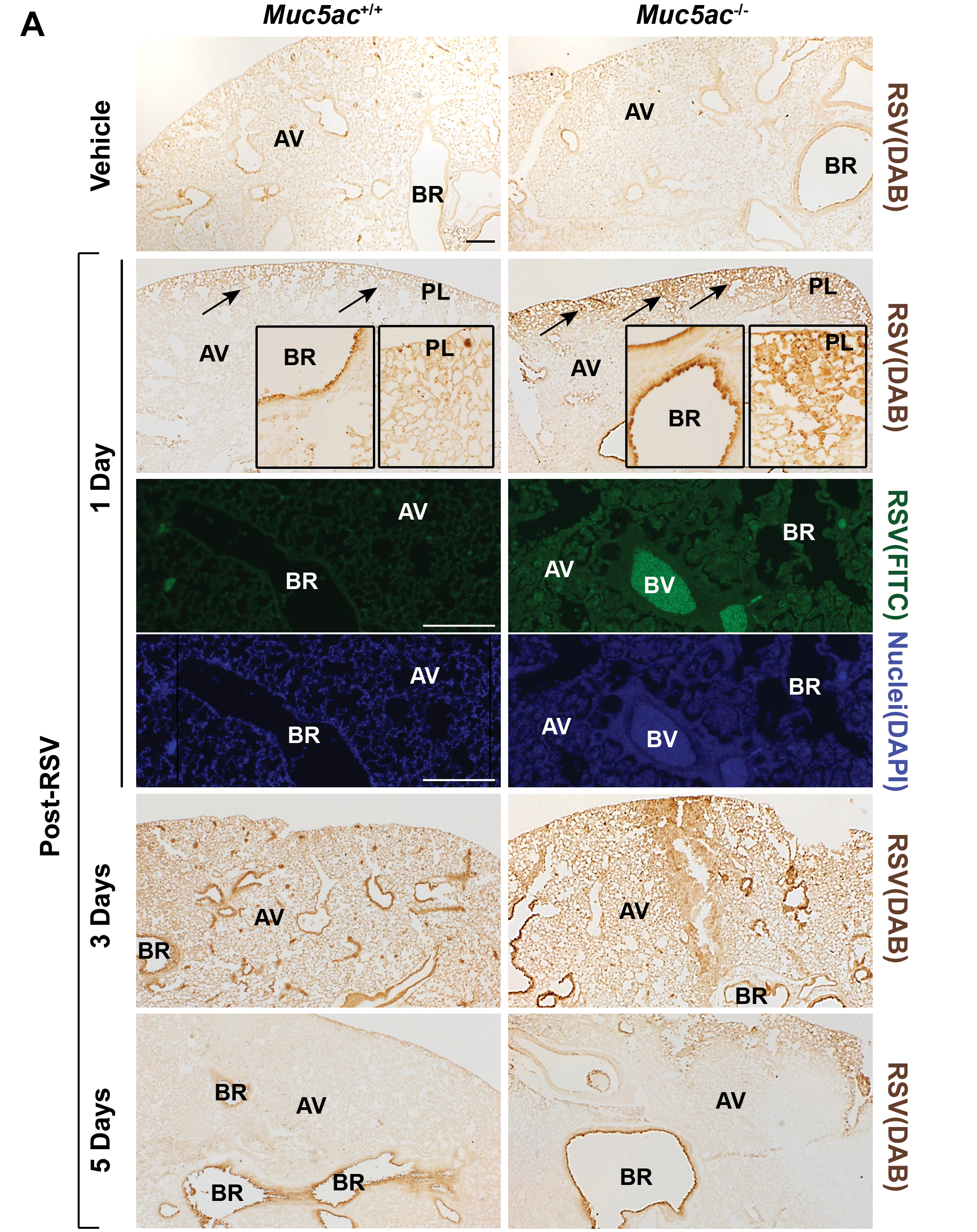
Differential respiratory syncytial virus (RSV) load and epithelial barrier expression in mucin-5AC-sufficient (Muc5ac+/+ ) and -deficient (Muc5ac−/− ) mouse lungs. A, RSV localization in lung sections detected by immunohistochemical (DAB) and immunofluorescent (FITC) methods using an anti-RSV antibody. Arrows indicate virus enrichment in pleurae (pleuritis) at 1-day post-RSV infection detected by DAB. Confocal images display more diffused RSV visualized with fluorescence (green, top) in Muc5ac−/− mouse lung than in Muc5ac+/+ lung. Nuclei were stained with DAPI (blue, bottom). Bars = 200 μm. AV = alveoli, BR = bronchi or bronchiole, PL = pleurae (pleural cavity), BV = blood vessel. Representative light photomicrographs presented (n = 5-8/group).
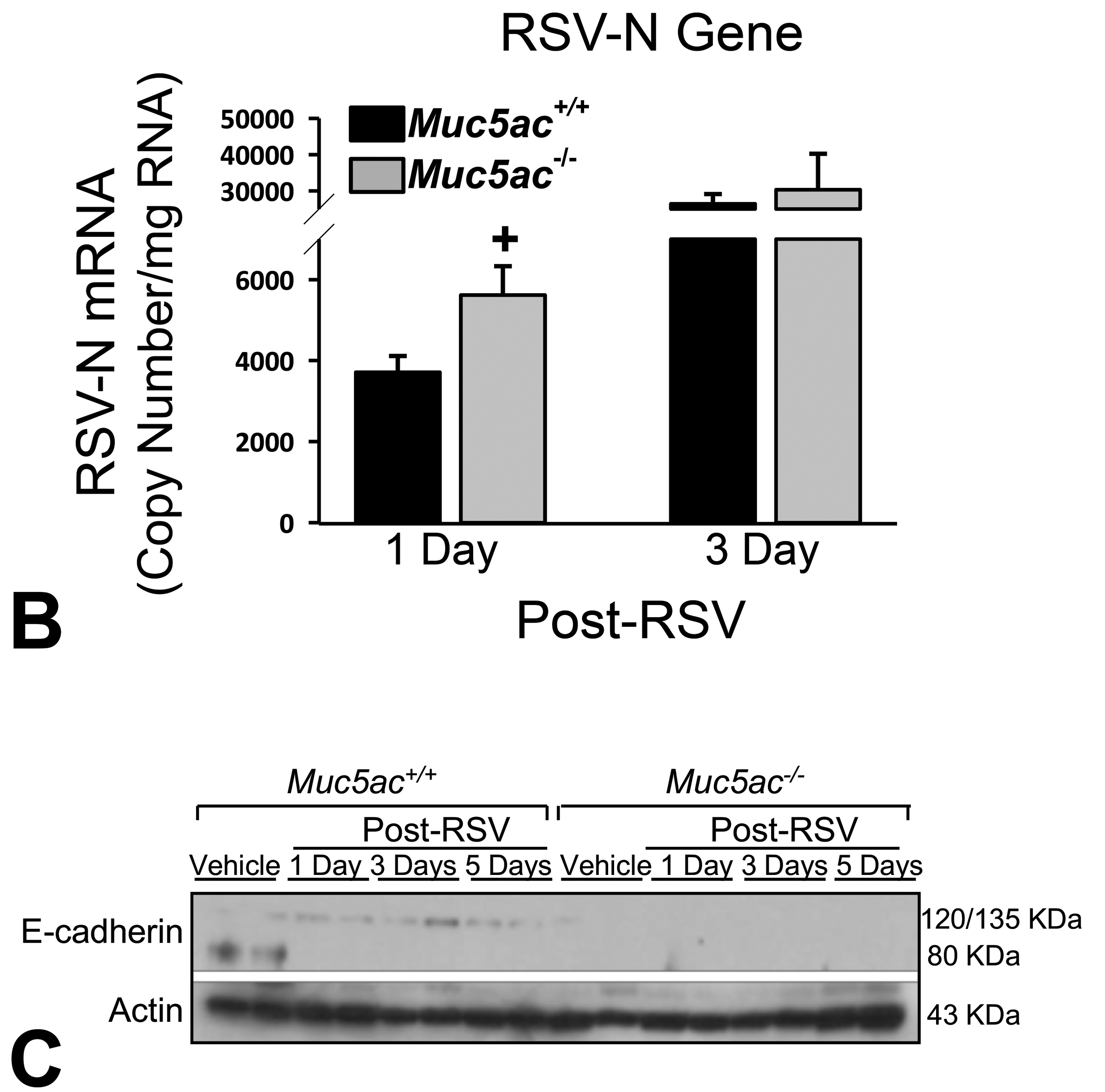
B, RSV infectivity was determined by quantification of viral nucleoprotein (N) gene expression using a digitized droplet PCR (ddPCR) method. Mean ± SEM (n = 3/group) presented. C, Protein level of E-cadherin in aliquots of lung homogenates was determined by Western blot analysis using actin as an internal control (n = 2 pooled samples/group). Representative digitized images from duplicate assays are presented. Graph symbols: *P < .05 versus genotype-matched vehicle control mice; †P < .05 versus exposure-matched Muc5ac+/+ mice. DAB indicates 3,3’-diaminobenzidine; FITC, fluorescein isothiocyanate; DAPI, 4′,6-diamidino-2-phenylindole; PCR, polymerase chain reaction; SEM, standard error of the mean.
The amount of mature E-cadherin protein (80 kDa) was basally lower in vehicle-treated Muc5ac−/− mice than in vehicle-treated Muc5ac+/+ mice (Figure 6C). After 1 and 3 days post-RSV, 120 kDa (mature)/135 kDa (precursor) E-cadherin levels were detected in Muc5ac+/+ lungs, which was declined at 5 days post-RSV in these mice (Figure 6C). E-cadherin was barely detected in Muc5ac−/− lungs after RSV infection (Figure 6C).
Nasal airway injury by RSV
Immunohistochemical localization in cross-sections of the proximal nasal turbinate (T1) indicated that RSV was mainly in surface epithelium lining septum, maxilloturbinate, blood vessels, and subepithelial glands in septum and walls, and walls at 1-day post-RSV (Figure 7). As found in the lung, RSV was most extensively detected at 3 days when nasal airway injury peaked, and virus propagation was relatively resolved at 5 days (Figure 7). Respiratory syncytial virus was more intensely and diffusively localized in Muc5ac−/− mice than in Muc5ac+/+ mice at 1 day, and nasal airway injury indicated by epithelial sloughing and hyperplastic changes in medial meatus region (medial wall, nasoturbinate, maxilloturbinate) was more prevalent in Muc5ac−/− mice than in Muc5ac+/+ mice at 1 to 5 days post-RSV (Figure 7).
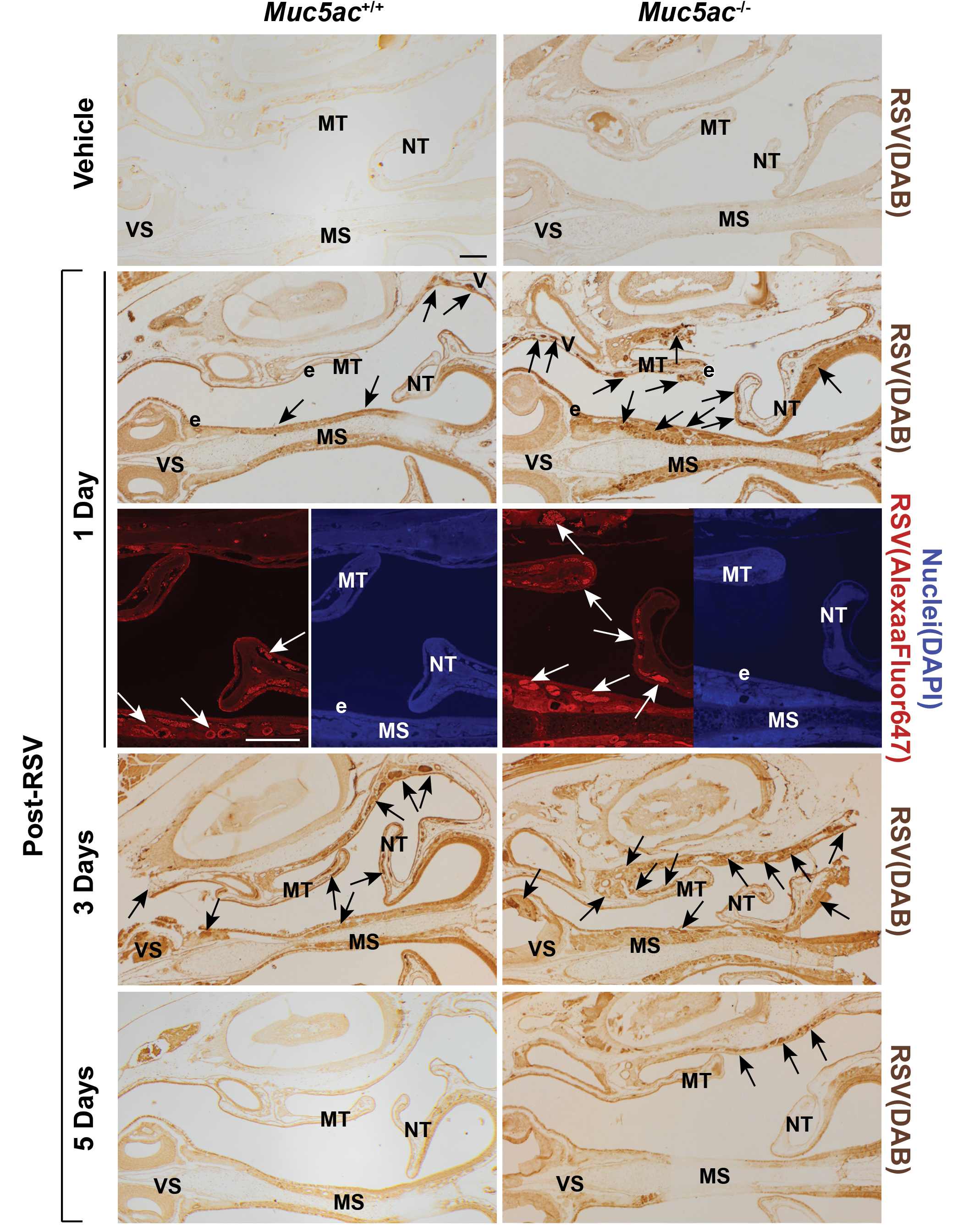
Heightened respiratory syncytial virus (RSV) load in nasal airway of Muc5ac-deficient (Muc5ac−/− ) mice. RSV detected by immunohistochemical (DAB) and immunofluorescent (Alexa Fluor 647) methods in proximal nasal airways of Muc5ac+/+ and Muc5ac−/− mice treated with vehicle (3 days) or RSV (1, 3, 5 days) using an anti-RSV antibody. Confocal images display more diffused virus visualized with fluorescence (left, red) in Muc5ac−/− turbinate than in Muc5ac+/+ turbinate at 1-day post-RSV. Nuclei were stained with DAPI (right, blue). Arrows indicate virus enriched in submucosal glands underneath septum and maxilloturbinate (MT). e = surface epithelium lining nasal mucosa, VS = ventral septum, NT = nasoturbinate. Bars = 200 μm. DAB indicates 3,3’-diaminobenzidine; DAPI, 4′,6-diamidino-2-phenylindole.
Alcian blue/periodic acid-Schiff staining of nasal airway epithelium revealed mucous cell hyperplasia in the mid-septum of Muc5ac+/+ mice from 1-day post-RSV (Figure 8A and B). Relative to vehicle control, RSV infection increased MUC5AC protein expression mostly in AB/PAS-positive nasal respiratory epithelium (mid- and ventral septum) and some in Bowman glands in Muc5ac+/+ mice (Supplemental Figure S3-A). The H&E staining found that RSV infection caused most morphological changes including epithelial sloughing and hypertrophy at 3 days postinfection largely in mid-septum, maxilloturbinate, and lateral meatus wall, and the injury was greater in Muc5ac+/+ mice than in Muc5ac−/− mice (Figure 8A, mid-septum shown). Although basal levels of AB/PAS-positive goblet cells were relatively fewer in Muc5ac−/− mice, RSV also caused significant goblet cell hyperplasia in Muc5ac−/− mice at later (7 days) postinfection days (Figure 8A and B). Interestingly, increase in PAS-positive substances in submucosal glands (mostly mucous glands) was more prevalent in Muc5ac−/− mice than in Muc5ac+/+ mice after RSV infection (Figure 8A). Respiratory syncytial virus-induced cellular proliferation and injury indicated by PCNA immunostaining were greater in Muc5ac−/− turbinates than in Muc5ac+/+ turbinates (Supplemental Figure S3-B). Consistent with the subepithelial gland hyperplasia (increased grand volume), PCNA localization in RSV-infected Muc5ac−/− turbinates was predominant in subepithelial gland cells in ventral septum (Figure S3-B). Immunohistochemical staining of MUC2 (Supplemental Figure S3-C) and ISH of Muc5b mRNA (Supplemental Figure S3-D) found that overall nasal expression of MUC5B and MUC2 may be increased by RSV infection similarly in both genotypes. Muc5b mRNA was detected mainly in hyperplastic/metaplastic respiratory epithelium (goblet cells and other cells) lining septum, nasoturbinate, and ventral wall and less frequently in Bowman glands (Supplemental Figure S3-D). It was, however, seldom detected in subepithelial glands. In contrast, MUC2 protein was increased in seromucosal glands in septum and lateral wall and in Bowmans glands with minor localization in septal surface epithelium (Supplemental Figure S3-C). Muc4 mRNA was detected only in cells surrounding maxillary sinus of the nasal airways (data not included). Therefore, compensatory septal goblet cell hyperplasia and submucosal gland hyperplasia manifest in Muc5ac−/− nasal airways may be at least in part contributed by upregulated MUC5B and MUC2, respectively.
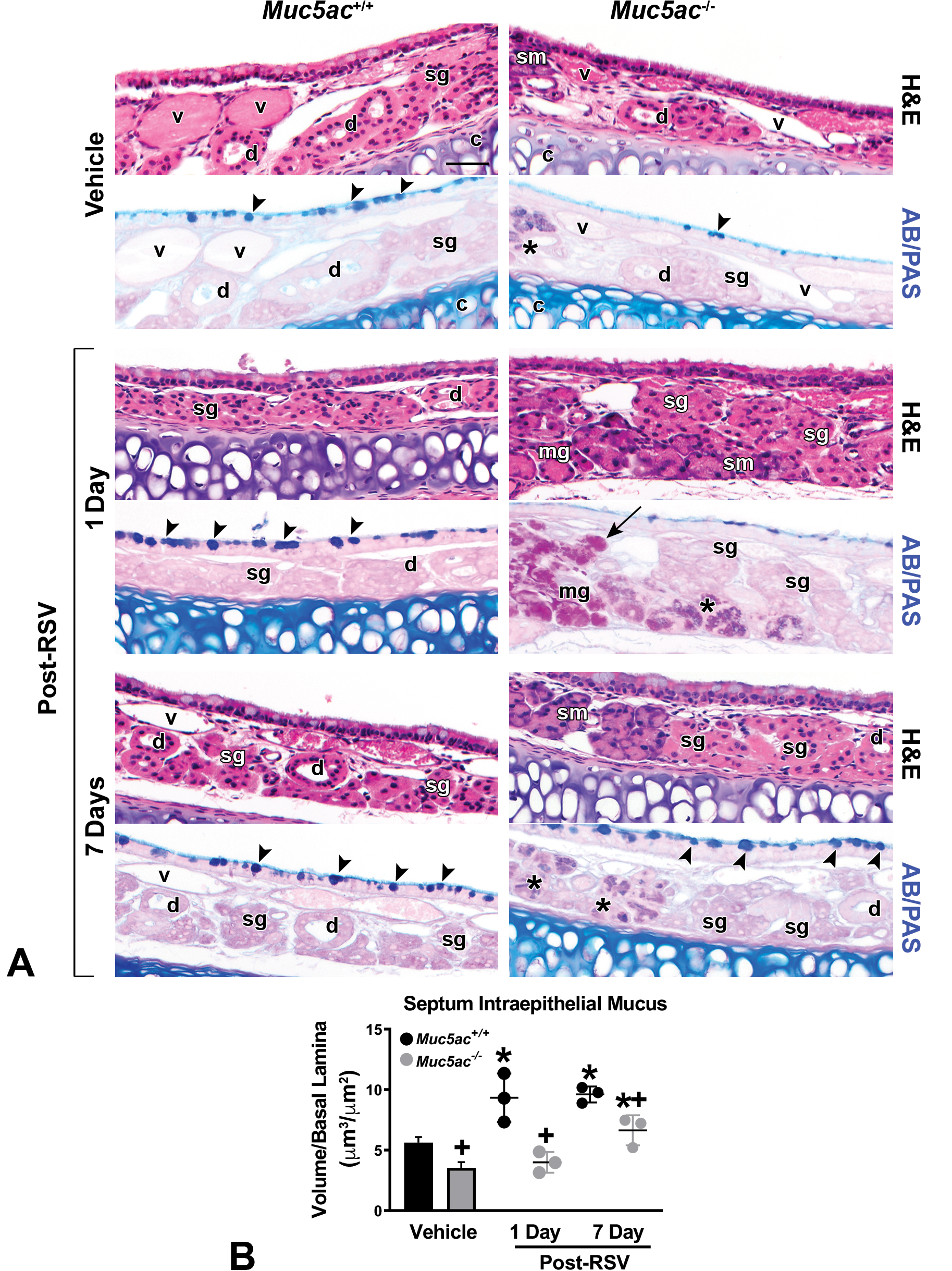
Differential changes of respiratory syncytial virus (RSV)-induced nasal airway mucosubstances in mucin-5AC-sufficient (Muc5ac+/+ ) and -deficient (Muc5ac−/− ) mice. A, Changes of intraepithelial and submucosal glandular mucosubstances in mid-septum (MS) of vehicle (1 day) and RSV (1 and 7 days)-treated mice. Representative light photomicrographs presented (n = 3-5/group). Arrow heads indicate stored intraepithelial mucus increased early (1 day) in Muc5ac+/+ mice and later (7 day) in Muc5ac+/+ and Muc5ac−/− vehicle after RSV infection. Arrow indicates PAS-positive mucus in hyperplastic mucosal glands. Asterisks indicate AB/PAS-stained mucus in newly developed glands (potentially submandibular) of Muc5ac−/− mouse septum. sg = serous gland; mg = mucous gland, sm = seromucous (submandibular) gland, d = gland duct, v = blood vessel, c = cartilage. Bar = 50 μm. B, RSV-induced increase in volume density (Vs) of AB/PAS-stained stored intraepithelial mucus in MS was morphometrically measured by Image J software. Mean ± SEM (n = 3/group) presented. Graph symbols: *P < .05 versus genotype-matched vehicle control mice; †P < .05 versus exposure-matched Muc5ac+/+ mice.

C, Transmission electron micrographs of nasal septum from vehicle-treated Muc5ac+/+ tissue with an intact periciliary layer, ciliated respiratory epithelium, and goblet cell vesicles filled with electron-dense materials (secretory mucins), while goblet cells in vehicle-treated Muc5ac−/− septum had relatively reduced granule storage. RSV-infected nasal epithelial cell surface with loss of cilia in both genotypes of mice. Goblet cells in Muc5ac+/+ mice had more frequent evidence of vacuoles (1 day), viral infection-associated debris trapped in fused granules (1 day), and granule exocytosis (3 days) against RSV indicating active antiviral activity. These goblet cell changes were relatively scarce in RSV-infected Muc5ac−/− mice and their secretory granule storage was decreased until 3 days post-RSV. In contrast, subepithelial glands in Muc5ac−/− septum contained uniformly stained granules connected with rich rough endoplasmic reticulum (rer) while subepithelial granules in Muc5ac+/+ septum had particularly electron-lucent outer rim (3 day). g = surface goblet cell with granules, c = cilia. Bar = 5 μm. AB/PAS indicates Alcian blue/periodic acid-Schiff; SEM, standard error of the mean.
Analyses of electron micrographs of the vehicle control epithelium from T1 found reduced mucus storage in goblet cell vesicles of Muc5ac−/− mice as indicated by relatively empty granules and electron-dense central core compared with the granule features in Muc5ac+/+ mice (Figure 8C). After 1 to 3 days of RSV infection, swollen granules, exocytosis, and formation of vacuoles were evident in goblet cells, and trapping of virus infection-associated debris in fused granules undergoing exocytosis were more commonly detected in wild-type mice (Figure 8C). However, Muc5ac−/− septum was less marked in these goblet cell activities after virus infection (Figure 8C). In contrast to the surface goblet cells, subepithelial glands in septum of Muc5ac−/− mice presented uniformly stained and electron-dense granules connected with rich rough endoplasmic reticulum at 3 days post-RSV, while RSV-infected glandular vesicles in Muc5ac+/+ septum had particularly electron-lucent outer rim (Figure 8C).
Increased O3-Induced Injury in Nasal Turbinate and Centriacinar Region of Muc5ac−/− Mice
Exposure to 0.3-ppm O3 caused epithelial exfoliation in mid- and dorsal-septum, mucous cell hyperplasia throughout the septal respiratory epithelium, mucoserous gland proliferation and hyperplasia, and blood vessel congestion in nasal airways of both genotypes from 48 hours (Figure 9A). Subepithelial gland expansion and congestion accompanying hypertrophy of basal lamina beneath septum, nasal wall, and turbinate were greater in Muc5ac−/− mice than in Muc5ac+/+ mice at 48-hour exposure (Figure 9A). Although O3 significantly increased AB/PAS-positive intraepithelial substances in mid-septum only in Muc5ac+/+ mice, PAS-positive stored mucoserous gland substances were significantly higher in Muc5ac−/− mice than in Muc5ac+/+ mice basally and after O3 (Figure 9B). In addition, these changes corresponded with the O3-induced secretion of PAS-positive mucosubstances in ventral wall detected only in Muc5ac−/− mice (Figure 9B). Using ISH, we found that O3 markedly increased Muc5b mRNA (peak at 48 hours) mainly in respiratory epithelium undergoing goblet cell hyperplasia (lining septum, nasoturbinate, and ventral wall) and less frequently in Bowman glands in both genotypes of mice (Figure 9C). However, Muc5b was seldom expressed in subepithelial glands, indicating other mucin genes/proteins (eg, Muc2, Muc7) may involve in PAS-positive subepithelial gland hyperplasia and secretion in Muc5ac−/− mice.
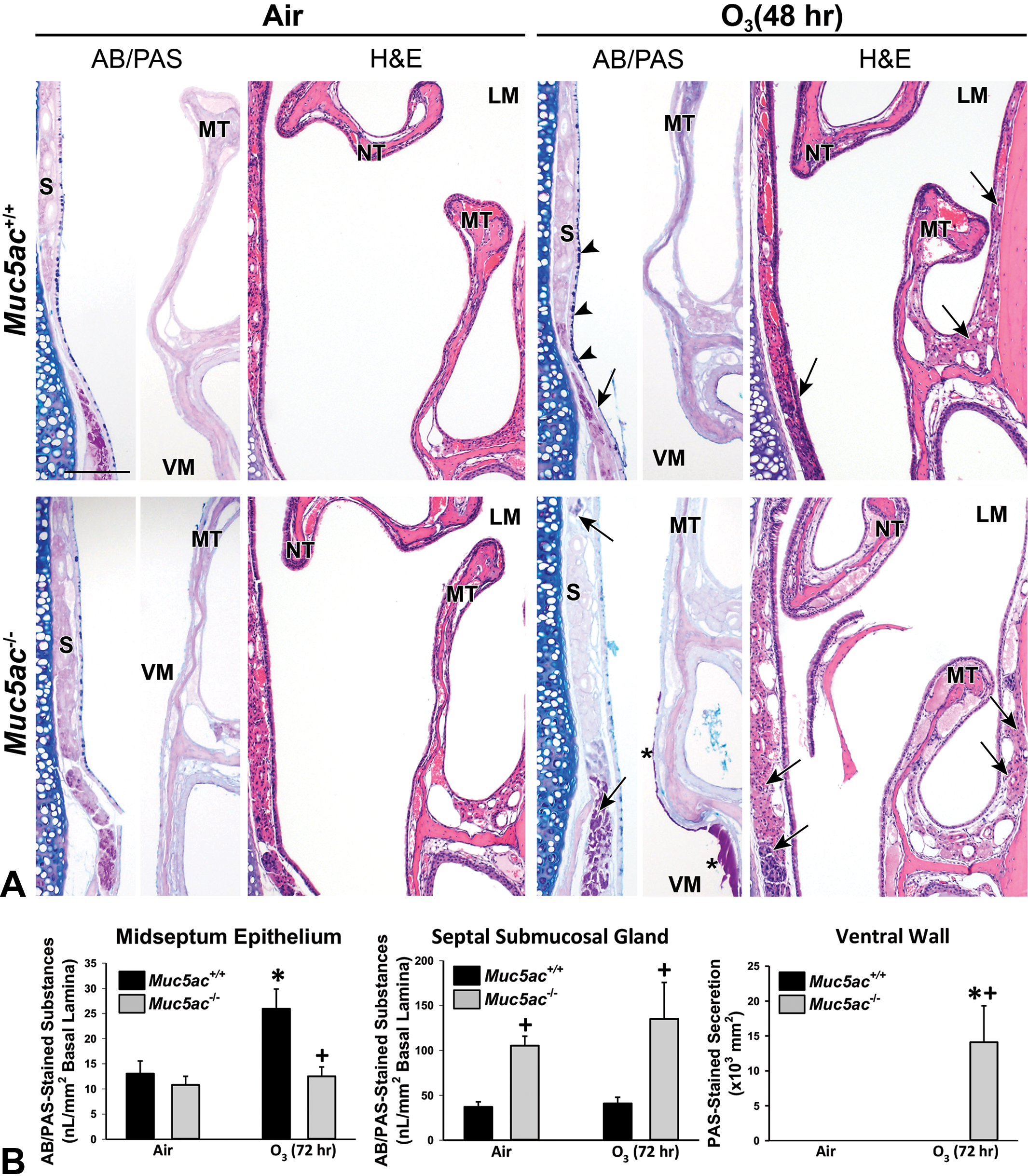
Exacerbation of nasal airway responses to ozone (O3) exposure in mucin-5AC-deficient (Muc5ac−/− ) mice. A, Increase in AB/PAS-stained septal intraepithelial mucosubstances (arrow heads) and PAS-stained septal and lateral wall subepithelial gland mucosubstances (arrows) in Muc5ac+/+ and Muc5ac−/− nasal airways after O3 exposure (48 hours). PAS-positive mucus-like secretion seen only in O3-exposed Muc5ac−/− terminates was indicated by an asterisk. S = septum, MT = maxilloturbinate, NT = nasoturbinate, LM = lateral meatus, VM = ventral meatus. Bar = 200 μm. B, Morphometric analyses identified O3-induced significant increase in AB/PAS-stained intraepithelial mucosubstances in Muc5ac+/+ turbinate (mid-septum) and PAS-stained subepithelial gland (mid-ventral septum) and PAS-positive mucus-like secretion (ventral meatus) in Muc5ac−/− turbinate. Mean ± SEM (n = 3/air group, n = 4/O3 group) presented. Graph symbols: *P < .05 versus genotype-matched air control mice; †P < .05 versus exposure-matched Muc5ac+/+ mice.
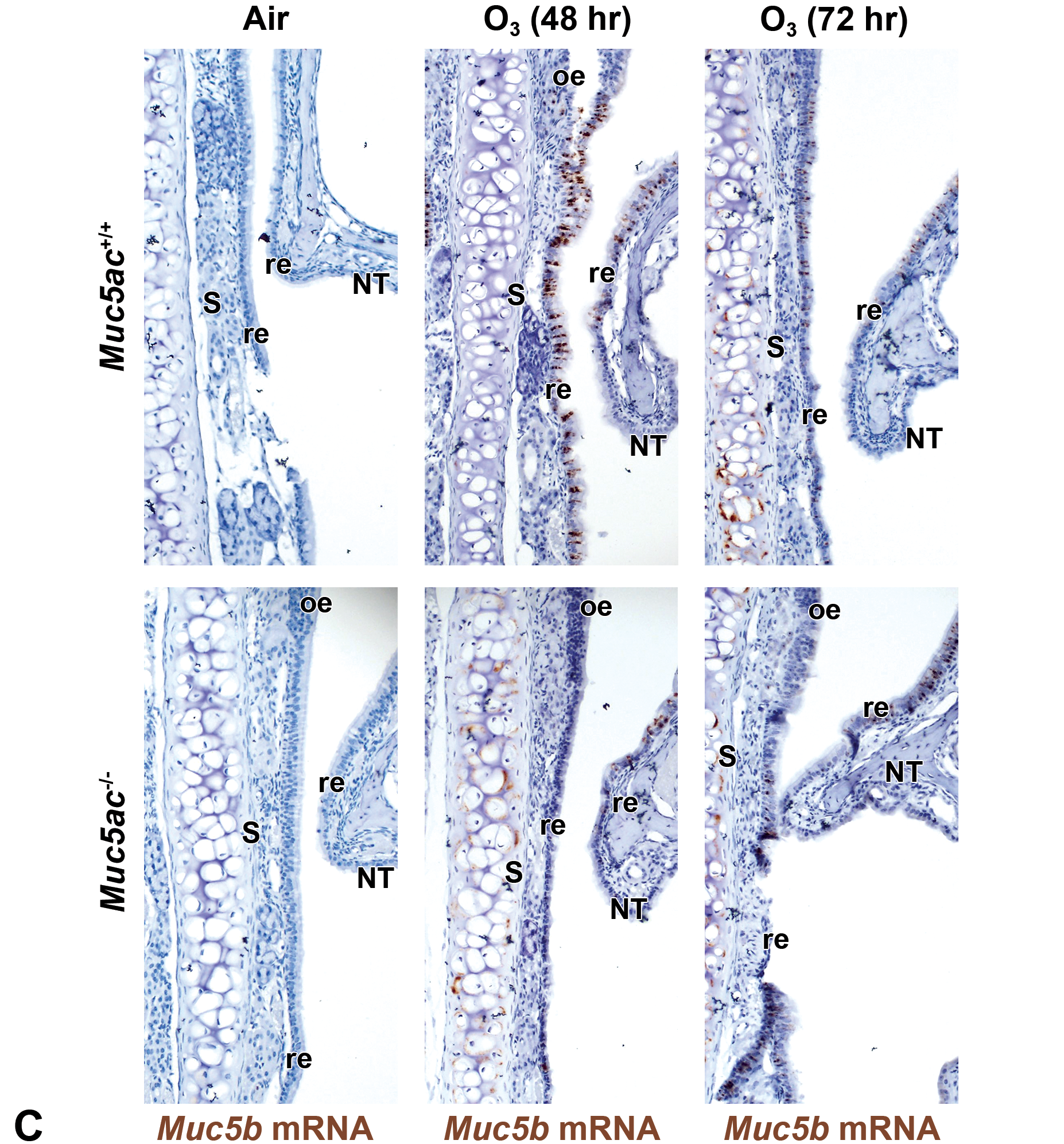
C, Nasal airway Muc5b mRNA expression (brown dots) determined by in situ hybridization (ISH) was markedly increased by O3 in respiratory epithelium throughout the septum and nasoturbinate undergoing mucous cell hyperplasia/metaplasia. Muc5b mRNA was also detected marginally after O3 exposure in Bowman glands. O3-induced Muc5b expression was higher in Muc5ac+/+ mice than in Muc5ac−/− mice, concurrent with AB/PAS-positive mucus increases (peak at 2 days) by O3 exposure. S = septum, NT = nasoturbinate, re = respiratory epithelium, oe = olfactory epithelium, BG = bowman glands. Bar = 100 μm. AB/PAS indicates Alcian blue/periodic acid-Schiff; mRNA, messenger RNA; SEM, standard error of the mean.
Mucous cell metaplasia developed in bronchial epithelium after O3 exposure (peaked at 48 hours) was predominant in Muc5ac+/+ mice relative to that in Muc5ac−/− mice (Figure 10A). In contrast, O3-induced injury and proliferation in the centriacinar region (junction of terminal bronchiole and alveoli) as determined by the abundance of PCNA-positive S phase cells were markedly higher in Muc5ac−/− mice than in wild-type mice at 48 hours (Figure 10B). Total protein concentration and total cells (Figure 10C) as well as inflammatory (macrophages, neutrophils, lymphocytes) and epithelial cell numbers in BAL were, however, not significantly different between 2 genotypes of mice after 48- to 72-hour exposure to O3. Lung E-cadherin amount detected at 120 (mature)/135 (precursor) kDa was basally lower in air-exposed Muc5ac−/− mice than in air-exposed Muc5ac+/+ mice (Figure 10D). O3 exposure decreased E-cadherin proteins in Muc5ac+/+ mice at 72 hours, and E-cadherin levels in Muc5ac−/− mice remained lower than in Muc5ac+/+ mice after O3 exposure (Figure 10D).
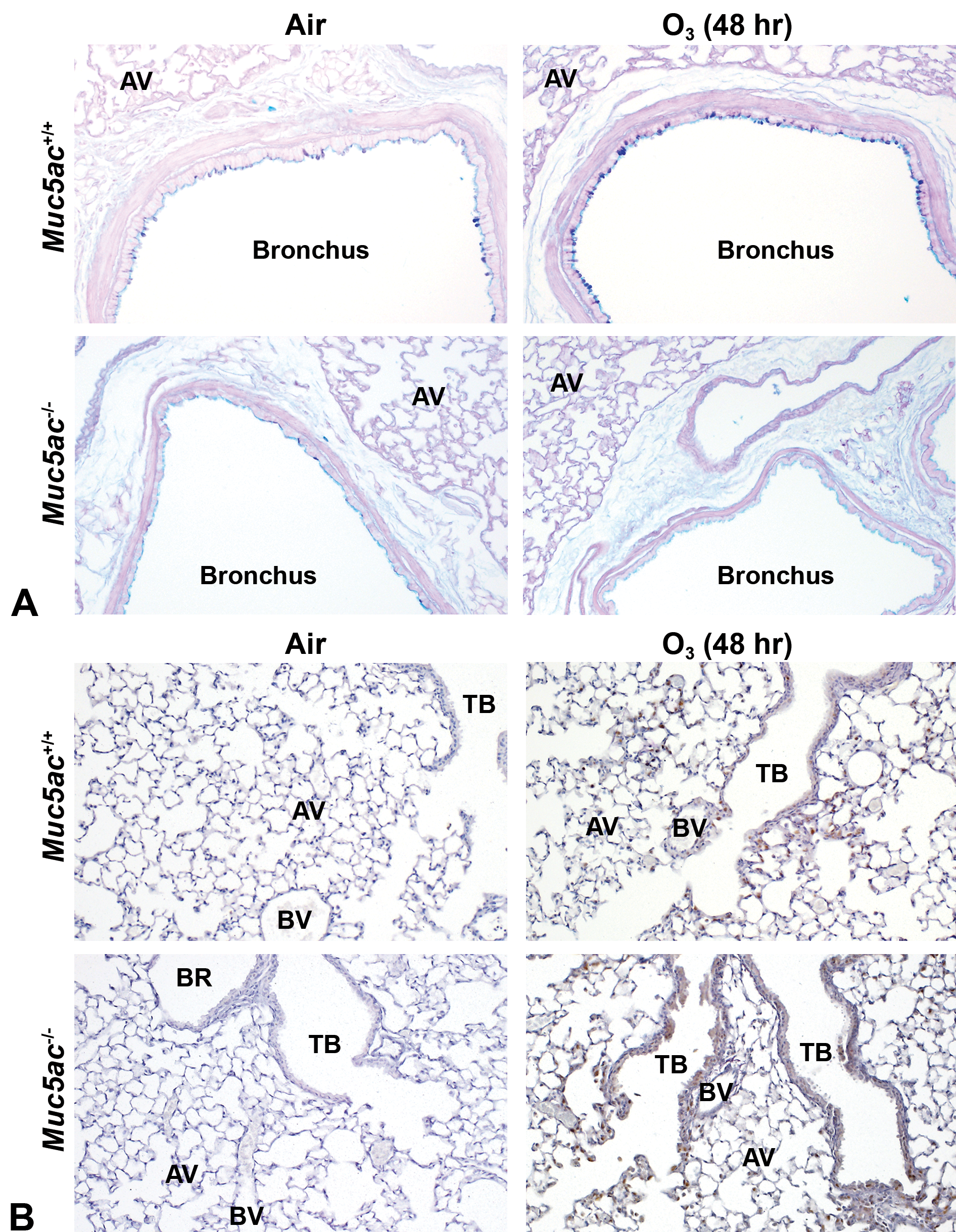
Exacerbation of pulmonary airway responses to ozone (O3) exposure in mucin-5AC-deficient (Muc5ac−/− ) mice. A, AB/PAS staining determined O3-induced bronchial epithelial mucous cell metaplasia in Muc5ac+/+ lung (peaked at 48 hours) but not in Muc5ac−/− lung. AV = alveoli. Bar = 100 μm. B, Epithelial cell proliferation in terminal bronchiole of centriacinal region, a marker of pulmonary O3 sensitivity, was determined by immunohistochemical staining of proliferating cell nuclear antigen (PCNA). Brown dots = PCNA. AV = alveoli, BR = bronchiole, TB = terminal bronchiole, BV = blood vessel. Bar = 100 μm.
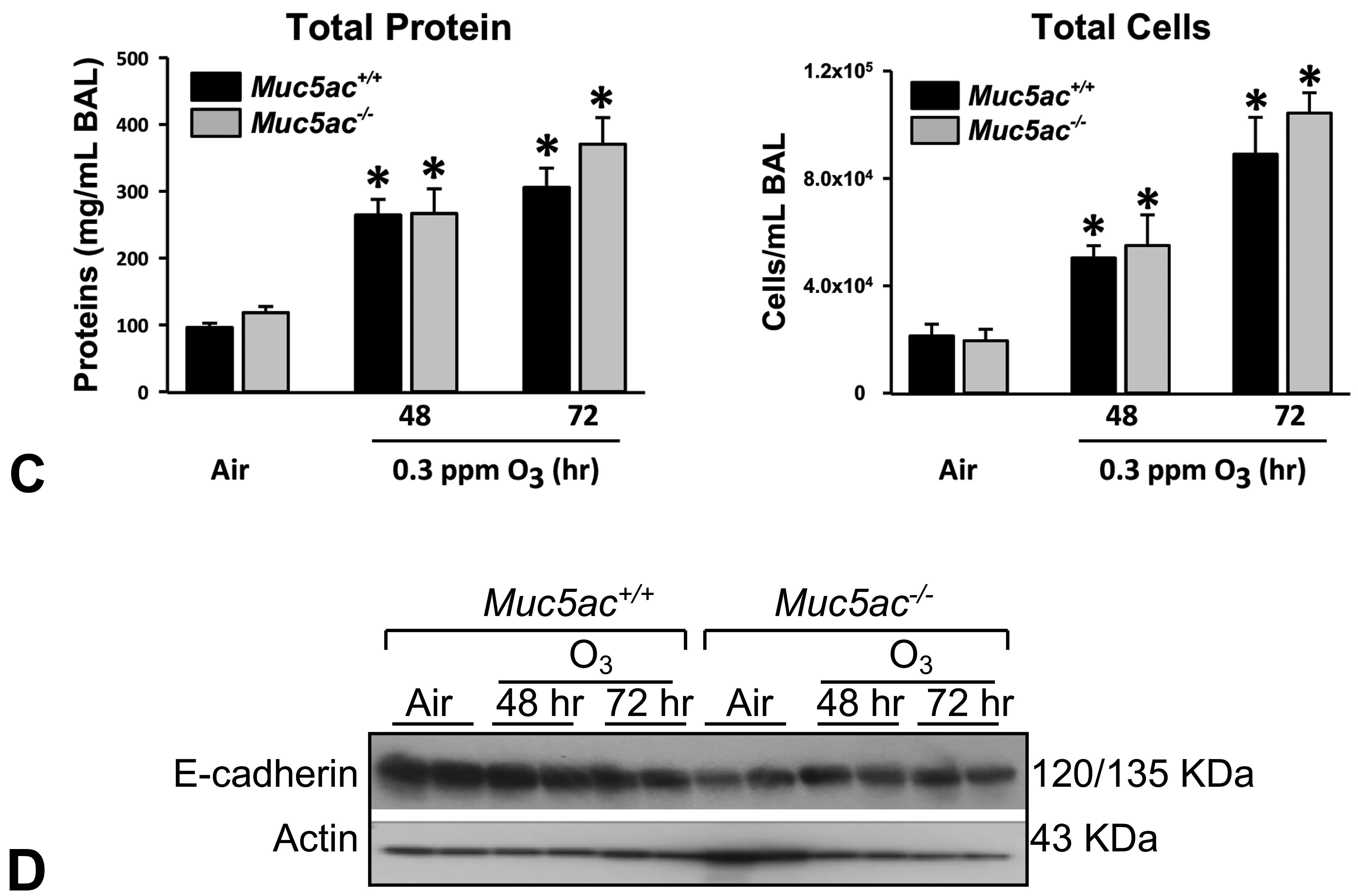
C, Bronchoalveolar lavage (BAL) analysis found no significant differences in lung protein concentration and cellular injury between Muc5ac+/+ and Muc5ac−/− mice after subacute exposure to 0.3 parts per million O3 exposure. Data presented as mean ± SEM (n = 4 for air groups, n = 6 for O3 groups). D, Protein level of E-cadherin in aliquots of lung homogenates was determined by Western blot analysis using actin as an internal control (n = 2 pooled samples/group). Representative digitized images from duplicate assays are presented. Intensity of blot images was quantified by densitometry (n = 2/group, mean ± SEM presented). Graph symbols: *P < .05 versus genotype-matched air control mice; †P < .05 versus exposure-matched Muc5ac+/+ mice.
Discussion
The current investigation demonstrated that deficiency of Muc5ac increased susceptibility to acute injury caused by virus and xenobiotics in nasal and pulmonary airways of mice. Compared to wild-type mice, significantly greater bleomycin-induced lung inflammation and fibrosis, early RSV infection and RSV-induced nasal and pulmonary injury, and O3-induced nasal mucosal and centriacinar pulmonary injury were found in Muc5ac−/− mice. Adaptive or augmented compensatory epithelial MCM in Muc5ac-deficient mice was concurrent with increased Muc4 mRNA, Muc5b mRNA, and/or MUC2 during pulmonary fibrogenesis. Lack of Muc5ac also led to subepithelial gland hypertrophy/hyperproliferation and/or hypersecretion in nasal airways after RSV and O3 exposure. A marker of epithelial barrier, E-cadherin, was suppressed in Muc5ac-deficient mice basally and after bleomycin, RSV, and O3 exposure. Results thus demonstrated requirement of MUC5AC for airway defense against acute phase injury and the compensatory adaptation of airway mucin system.
In disease conditions such as chronic bronchitis which contributes to the formation of airway plugs, submucosal glands in tracheobronchi increased in number and volume in smokers relative to nonsmokers and extended to the distal bronchioles. 26 In addition, total tracheobronchial gland volume in lungs from patients with severe COPD and chronic bronchitis or serous gland substance secretion in asthmatic lungs were 4-fold higher than those in healthy lung. 5,42 Our findings particularly in RSV- and O3-susceptible Muc5ac-deficient mice were consistent with these observations. Hyperplasia of the PAS-positive mucous glands and appearance of AB/PAS-positive submandibular-like glands (with mixed histologic feature of serous and mucous glands) were more distinct in Muc5ac−/− mice infected with RSV or exposed to O3. We postulate that Muc5ac−/− mice bearing decreased number of airway surface goblet cells and their granule contents may facilitate enrichment of mucus and/or serous substances in submucosal gland acini for their compensatory mucosal defence.
Although produced little in healthy bronchial airways, MUC5AC has been a marker of MCM 43,44 and is highly increased in asthma, allergic airway disorder, and acute lung injury. 28,45 –48 A MUC5AC single nucleotide polymorphism (rs1132440) was significantly associated with respiratory outcomes (bronchitis, wheeze, asthma, hay fever). 49 A direct correlation was also found between the polymorphism-related length of the MUC5AC variable number tandem repeat fragment and the severity of cystic fibrosis. 50 Our results are consistent with the hypothesis that lack of Muc5ac may lead to porous barrier during airway pathogenesis, which is not able to retain protective molecules in mucosa and in cells by trapping foreign bodies to inhibit acute phase of epithelial injury, viral entry into tissues, inflammation, and fibrogenesis. During RSV infection, the reduction in mucus levels in Muc5ac−/− mice may allow virus to reach the cell surface or viral products could diffuse through the mucus. Overexpression of Muc5ac provided resistance to pulmonary influenza virus infection and neutrophilia, 16 which was consistent with our current findings in the RSV disease model. Muc5ac-overexpressing mice had 20-fold increased lung MUC5AC protein but preserved mucociliary clearance function as the increased MUC5AC expanded (thicker), not concentrated, the mucus layer. 16 The anti-influenza role of MUC5AC was proposed to be at least in part by biochemical competition of viruses with host-derived materials for interaction with the oligosaccharide-attached terminal groups (eg, sialic acid, fucose, sulfate) of MUC5AC. 51,52 The sequence, conformation, and charges of these mucin sugar epitopes are known to modulate pathogen adhesion, and recognition and domains of the protein core can also serve as ligands. 53,54 Considering multiple antiviral substances including β-defensin, lactoferrin, and type I and III interferons against viral infection in airway mucus layer, 55 the more rapid and widespread effects of RSV infection in Muc5ac−/− mice are predicted due to reduced mucus during the early infection period. The functional role for the airway MUC5AC studied using transgenic animal models, however, has been debatable. For example, Muc5ac deletion improved survival time and decreased lung edema, inflammation, and gas-exchange reduction against ventilator-induced acute lung injury. 28 Muc5ac−/− mice had significantly reduced MCM and mucous plugging and airway hyperreactivity response in an allergic airway disease model, 27 consistent with the association of MUC5AC to bronchoconstriction of asthmatics. 45 Overall, the relationship between MUC5AC and airway pathogenesis is likely complex, and it may vary depending on the damaged area/cell type and the severity of mucus production/secretion in the diseases.
Basal, ciliated, and type 2 cells have been widely studied as potential stem/progenitors of metaplastic goblet cells during the pathogenesis of airway disorders. Signal transduction pathways including EGFR-PI3K/Akt (ciliated apoptosis inhibition), IL-13 recetptor-STAT6 (ciliated cell mucus production), and Notch2 (basal cell to goblet cell) have been demonstrated to involve the MCM, 56 –59 and trans-differentiation (lineage reprogramming) may underlie the cellular level of goblet cell formation in the fully differentiated respiratory epithelium (eg, club cell, ciliated cell). Recent investigations on progenitor cells have focused on club cells which are ubiquitous throughout the conducting airways of mice while restricted to the distal bronchioles in humans. 11 Club cells possess the entire secretory machinery to ensure post-translational maturation of mucins and populate the large and small murine airways. 14 Murine club cells are also found to be the major sources of MUC5B and MUC5AC. 15,60 Supporting these findings, mice that overexpress Muc5ac under control of club-cell secretory protein gene (Ccsp, Cc10, or Scgb1a1) produced more MUC5AC without goblet cell hyperplastic or metaplastic changes, and evidence of mucin granules was detected in the club cells. 16 Presence of cells sharing the characteristics of goblet cells and club cells in asthmatic mouse airways, which do not exist in normal murine epithelium, 61 and decrease in club cells in allergen challenged airway further supported club cells as progenitor. 62 We also determined marked increase of epithelial mucin messages (Muc5b, Muc4) or protein (MUC2) throughout the conducting airways including small bronchioles and terminal bronchioles, indicating production of these mucins in the club cells in response to environmental stimuli. However, lack of AB/PAS staining in the mucin-positive distal airway epithelium of the fibrotic lung in the current and previous studies 14,16 requires further investigation.
Previous work indicated that Muc5ac deficiency was compensated by an increase in Muc4 in the ocular surface of mice, 40 and MUC5AC alteration in cornea of allergic eye disorder was compensated by secretion of MUC1, MUC2, and MUC4. 41 Compensatory increase of MUC5B in Muc5ac−/− mice was also reported in eyes. 36 In tracheobronchial airways, Muc5b−/− mice had heightened Muc5ac in goblet cells. 15 MUC2 is a rarely detected mucin in healthy airways but it is associated with patients with asthma and COPD, 37 and MUC4 is involved in cell proliferation signaling and has been a marker of various cancers including lung adenocarcinoma. 38,39 We demonstrated that MUC5B and MUC2 (in lung and nasal airways) as well as MUC4 (in lung) may contribute in part to the compensatory increase of mucous cells in airways of injured Muc5ac−/− mice. Overall, our results and others indicate that single airway mucin deficiency may affect the composition and quantity of mucus in pulmonary and nasal airways for mucin compensation as an alternative defensive mechanism in airway mucosa.
Results of our experiments supported reduction of lung E-cadherin, an intercellular apical junctional complex (adherens junctions), by bleomycin, RSV, or O3 exposure observed in Muc5ac+/+ mice. Respiratory syncytial virus altered the molecular composition of tight junctions leading to airway epithelial hyperpermeability and decreased expression of tight-junction proteins and accumulation of cleaved extracellular fragments of E-cadherin in BAL. 63 During development of bleomycin-induced pulmonary fibrosis, E-cadherin was downregulated to promote accumulation of myofibroblasts. 64 In addition, O3 caused rapid disruption of lung epithelial barrier followed by increased neutrophilic inflammation and declined tight junction proteins and E-cadherin in mice. 65 A recent study determined that genetic loss of E-cadherin led to airway epithelial damage including epithelial denudation, decreased tight junction (zonula occludens-1) expression, loss of ciliated cells, and spontaneous goblet cell metaplasia in addition to eosinophilic inflammation and enlarged airspace in mice. 66 The authors postulated loss of E-cadherin as a critical step in the development of asthma or emphysema. 66
Although further examinations are warranted, we found several severe phenotypes in aged (11-12 months) Muc5ac−/− mice than in aged Muc5ac+/+ mice, which included the increased tendency of body fat (Supplemental Figure S4-A) and compensatory nasal septal mucous cell hyperplasia and nasal submucosal gland hyperplasia (Supplemental Figure S4-B). In addition, Muc5ac deficiency caused more severe and/or frequent nodular lymphoid aggregation and preinvasive benign neoplasm indicated by focal spontaneous alveolar hyperplasia or small adenomas (Supplemental Figure S4-C). The Muc5ac−/− mice also developed spontaneous gastric antropyloric adenoma by age 8 to 12 months. 67 MUC5AC is a commonly detected polymeric mucin in lung adenocarcinoma and was a significant determinant of poor prognosis, especially in patients with KRAS-mutant tumors. 68 Different from the enhanced spontaneous preinvasive changes in aged Muc5ac−/− lungs and stomachs, chemical-induced lung tumorigenesis was significantly lower in Muc5ac−/− mice (BALB/cJ background) compared with that in wild-type mice, 68 indicating different underlying molecular mechanisms.
In conclusion, ablation of Muc5ac is deleterious in the early response phase of airway inflammation and injury. Lack of Muc5ac affected the quantity of mucus and mucus-producing cells at baseline and the composition of mucins during airway pathogenesis. Muc5ac deficiency was compensated by other mucins followed by adaptive MCM and submucosal gland hyperplasia as the injury progresses, which explained an alternative defensive mechanism to avoid further mucosal injury in Muc5ac−/− mice. Although murine models have been useful to investigate MUC5AC in various disorders, differences remain between human and mouse airways in mucin composition and in distribution of potentially progenitor club cells. In addition, due to the adaptive mechanism of mucin compensation, genetic deletion of a single mucin gene may not be sufficient to address its role in airway pathogenesis. Although MUC5AC is found to be essential for host defense against airway toxicants, overproduction of MUC5AC has deleterious effects as well. Therefore, control of proper mucin induction and mucus secretion may enhance the intrinsic role for mucin in airway protection and defense.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211004433 - Role for Mucin-5AC in Upper and Lower Airway Pathogenesis in Mice
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211004433 for Role for Mucin-5AC in Upper and Lower Airway Pathogenesis in Mice by Hye-Youn Cho, Soojung Park, Laura Miller, Huei-Chen Lee, Robert Langenbach and Steven R. Kleeberger in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-zip-1-tpx-10.1177_01926233211004433 - Role for Mucin-5AC in Upper and Lower Airway Pathogenesis in Mice
Supplemental Material, sj-zip-1-tpx-10.1177_01926233211004433 for Role for Mucin-5AC in Upper and Lower Airway Pathogenesis in Mice by Hye-Youn Cho, Soojung Park, Laura Miller, Huei-Chen Lee, Robert Langenbach and Steven R. Kleeberger in Toxicologic Pathology
Footnotes
Acknowledgments
The authors thank Dr Daniel Morgan for coordinating O3 exposures at the NIEHS Inhalation Facility under contract to Alion Science and Technology, Inc. The authors highly appreciate Mrs T. Beth Mahler in NIEHS Comparative and Molecular Pathogenesis Branch for her efforts to format and finalize histology images and figures. Mrs Pamela Ovwigho and Natasha Clayton in the NIEHS Histology Core Laboratory provided excellent histology support, Mrs Deloris Sutton and Mr. Robert Keys in the NIEHS Electron Microscopy Core processed samples for electron microscopic analyses, and Mrs Ann Chavis in Experimental Pathology Lab evaluated pulmonary histopathology. Drs Stavros Garantziotis and Hideki Nakano at the NIEHS provided excellent critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Intramural Research program of the National Institute of Environmental Health Sciences, National Institutes of Health, Department of Health and Human Services.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
