Abstract
A host of novel renal biomarkers have been developed over the past few decades which have enhanced monitoring of renal disease and drug-induced kidney injury in both preclinical studies and in humans. Since chronic kidney disease (CKD) and acute kidney injury (AKI) share similar underlying mechanisms and the tubulointerstitial compartment has a functional role in the progression of CKD, urinary biomarkers of AKI may provide predictive information in chronic renal disease. Numerous studies have explored whether the recent AKI biomarkers could improve upon the standard clinical biomarkers, estimated glomerular filtration rate (eGFR), and urinary albumin to creatinine ratio, for predicting outcomes in CKD patients. This review is an introduction to alternative assays that can be utilized in chronic (>3 months duration) nonclinical safety studies to provide information on renal dysfunction and to demonstrate specific situations where these assays could be utilized in nonclinical drug development. Novel biomarkers such as symmetrical dimethyl arginine, dickkopf homolog 3, and cystatin C predict chronic renal injury in animals, act as surrogates for GFR, and may predict changes in GFR in patients over time, ultimately providing a bridge from preclinical to clinical renal monitoring.
Introduction
This review will discuss current and novel biomarkers used to monitor chronic renal dysfunction including some biomarkers of acute kidney injury (AKI) that have also been evaluated in the chronic setting. Through a weight of evidence approach, gleaning information from the published literature regarding biomarkers utilized to monitor chronic renal injury and disease in humans and animals, the intent of this review is to highlight the potential application of some chronic renal injury biomarkers in nonclinical toxicity studies of 4 months or longer duration where they may provide clinical translation, aid pharmaceutical development, and help resolve regulatory hurdles. One of the primary causes for the attrition of promising therapeutic agents from the drug development pipeline is the observation of treatment-related microscopic injury to the kidney in animal toxicology studies. 1 Therefore, identifying, evaluating, qualifying, and utilizing relevant biomarkers of renal injury has greatly benefited the drug development process in recent years. 2 The inclusion of novel urinary biomarkers and other investigative techniques in nonclinical toxicity studies has successfully identified nephrotoxic signals in animals and, due to their clinical translatability, helped predict and monitor renal toxicity in humans. 3 This has helped inform clinical risk assessment and provide enhanced clinical safety monitoring through the establishment of defined entrance and stopping criteria in clinical trials based on information provided by changes in these analytes in laboratory animals in nonclinical studies. 4 For many decades, the measurement of blood urea nitrogen (urea nitrogen [UN]) and creatinine in serum or plasma have been the basis for assessment of renal function in both animals and humans. The advantage of these 2 biomarkers is that they progressively increase in both acute and chronic renal toxicity, and the magnitude of response correlates with decreases in renal function as assessed by glomerular filtration rate (GFR). 5,6 Unfortunately, both UN and creatinine lack sensitivity in detecting renal disorders as they only begin to increase once approximately 50% to 60% of nephrons are damaged and/or a similar percentage of renal function is compromised, depending on the species. 7 –9 This is unacceptable for a clinical biomarker since the goal of any monitoring paradigm in a clinical trial would be to detect the earliest nephrotoxic signal prior to significant kidney injury in order to initiate stopping criteria for kidney injury before damage occurs. There are also a host of extrarenal factors, including dehydration, which can interfere with interpretation of UN and/or creatinine by overestimation of renal injury. In addition, several drugs can compete or interfere with the renal creatinine transporter system resulting in spurious changes. 5,10 –13 For animals such as dogs that are generally on meat diets, creatinine is vulnerable to diet-related variability (eg, higher creatinine with meat vs casein-based diets) and other factors including muscle mass and body weight (ie, higher serum creatinine levels with larger muscle mass), housing, circadian and seasonal variation, hydration status, physical activity, drug treatment, and age. 14
In humans, AKI is defined as damage or loss of kidney function for a duration of 7 to 90 days after an AKI initiating event which would parallel renal injury occurring during the acute/subacute nonclinical toxicity study time frame. 15 In contrast, chronic renal failure or chronic kidney disease (CKD) is defined by the persistence of kidney disease for greater than 90 days, which would translate to renal injury that persists in nonclinical toxicity studies of greater than 4 months’ duration. 15 Chronic kidney disease staging in humans is based on biomarkers of glomerular function (estimated GFR [eGFR]) and tubuloglomerular injury associated with saturation of the megalin/cubulin resorptive capacity (eg, urinary albumin to creatinine ratio [UACR]). 16 Since CKD and AKI share similar underlying mechanisms of functional and structural injury, exist on the same pathophysiologic continuum, and because the tubulointerstitial compartment has a functional role in the progression of CKD, 17 –19 urinary biomarkers of AKI may also provide predictive information for chronic renal injury/disease. Recent efforts have focused on identifying urine or serum biomarkers of renal tubular injury that are either produced by the kidney or accumulate secondary to tubular cell dysfunction. Most of the kidney’s energy expenditure supports the maintenance of non-glomerular functions (ie, tubular functions), so biomarkers that correlate with tubular injury may act as indicators of kidney pathology or health. In addition, studies have shown that the prognosis of even glomerular origin kidney disease is dependent on the extent of tubulointerstitial fibrosis. 19 Renal tubule injury biomarkers also provide information about kidney tubular injury in early CKD where the “renal filtration reserve” is able to compensate for filtration deficits and serum creatinine levels remain unchanged. 20 However, once renal filtration reserve is lost, additional renal insults result in further decreases in glomerular filtration and subsequent increases in serum creatinine, limiting the utility of tubular injury biomarkers in patients with diminished renal reserve. 21
Numerous studies have explored whether the recent AKI biomarkers (especially kidney injury molecule 1 [KIM-1] and neutrophil gelatinase–associated lipocalin [NGAL]) could improve upon eGFR and UACR for predicting outcomes in CKD patients (reviewed in the studies 21 –32 ). Even though many studies have linked KIM-1 and NGAL to deterioration of renal function, there is conflicting evidence that use of KIM-1 or NGAL improves upon risk assessment beyond the standard renal biomarkers. 21,22,25 One possibility is that tubular injury biomarkers may have limited utility in patients with diminished renal reserve because of nephron loss. 29 Chronic kidney disease is not a discrete disease entity but rather the common result of diverse disease processes that can occur simultaneously, and it is unlikely that one biomarker will capture all processes. 25 Instead, biomarker panels or disease-specific biomarkers may provide better prognostic utility for CKD. 30,31 In the future, the use of urinary biomarkers in CKD may involve a more personalized medicine approach, for example, by targeting patients undergoing therapy for renal fibrosis prevention or by identifying AKI patients with preserved kidney function who are at risk of progression to CKD. 22,30
Over the past 10 to 20 years, many novel renal biomarkers with increased sensitivity and specificity for renal disease have been developed and used to monitor drug-induced kidney injury (DIKI) in both preclinical studies and human trials. 33 Although multiple renal biomarkers are available, currently there is no single defined battery of tests which will identify the pathophysiologic basis for every potential mechanism of renal injury, and most of the established analytes in animals are best suited for acute DIKI because these biomarkers were developed and evaluated for detection and monitoring of renal dysfunction in the setting of AKI. Urinary assays such as KIM-1, albumin, total protein, clusterin (CLU), cystatin-C (CysC), β2 microglobulin (B2M), and renal papillary antigen have their place in renal investigative studies and have achieved qualification by the Food and Drug Administration (FDA) for defined contexts of use in rat preclinical studies. 34 –39 In addition, urinary NGAL and osteopontin (OPN) have also received a letter of support by the FDA for further evaluation in rat preclinical studies. 40 Many of these biomarkers provide nephron segment injury localization and have even shown utility as sensitive AKI immunohistochemical reagents. 33,41 In 2018, the FDA qualified the use of a single composite measure of 6 urine biomarkers (KIM-1, NGAL, CLU, OPN, N-acetyl-beta-D-glucosaminidase [NAG], and CysC) in conjunction with traditional measures of kidney function (creatinine, UN, serum CysC, urine albumin, and urine total protein) to aid in the detection of kidney tubular injury in phase 1 trials in healthy volunteers when there is a prior concern that a drug may cause tubular injury in humans. 42 –44 However, KIM-1, albumin, and NGAL have demonstrated the most translational applicability and appear to be the most commonly used in acute or subacute nonclinical studies. 2,45 –52
The purpose of this manuscript is (1) to provide a comprehensive literature review of current human and animal renal biomarkers of AKI and CKD and how they are being utilized in chronic renal injury/disease (Table 1), (2) to provide a review of alternative assays (Table 2 and Figure 1) that can be utilized in nonclinical safety studies to evaluate chronic renal dysfunction (>4 months duration), and (3) to identify specific situations where these assays could best be utilized. However, only with increased use of these chronic renal injury biomarkers by multiple institutions and in many nonclinical studies will enough information be generated to establish industry-wide confidence in their use and interpretation. A list of abbreviations used in this review can be found in Supplemental Table 1.
Human Clinical Research Biomarkers Evaluated in Chronic Kidney Injury and Disease in Animals.
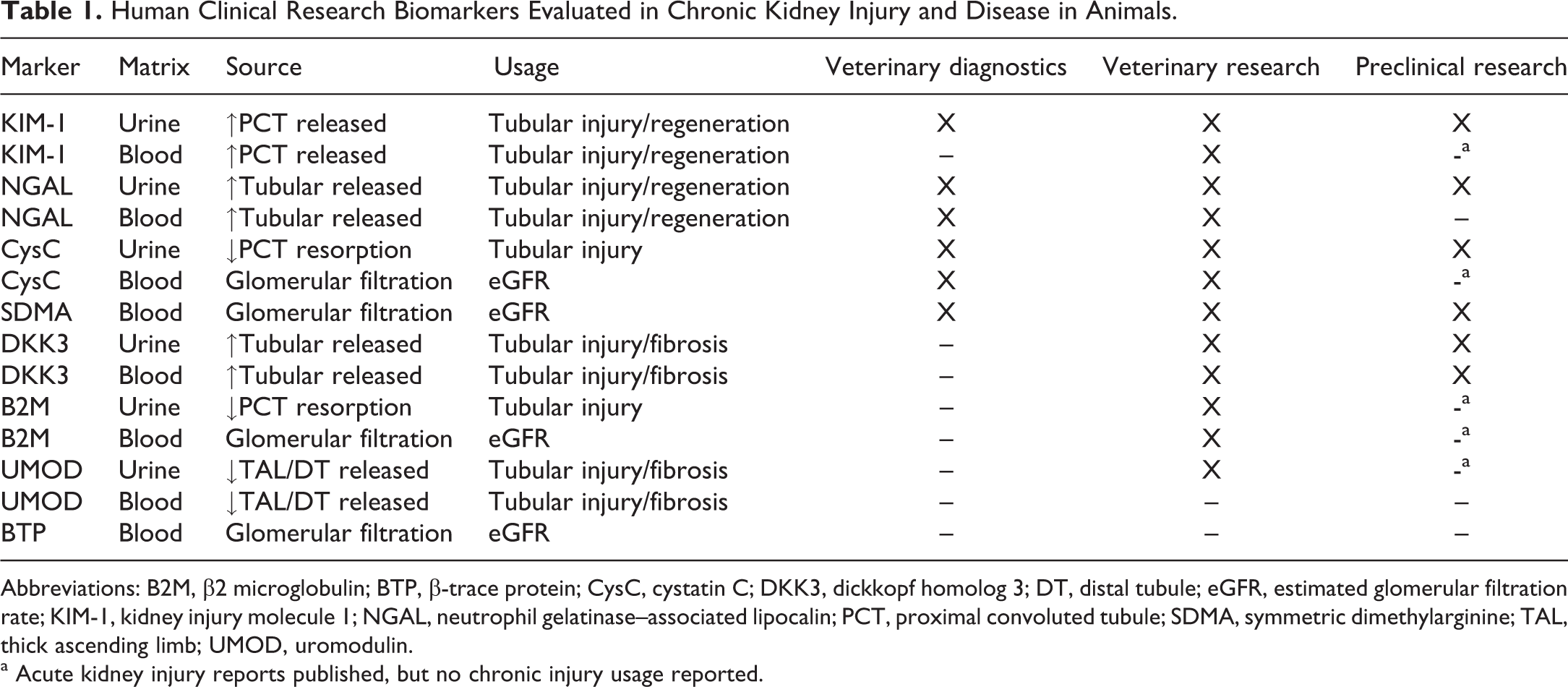
Abbreviations: B2M, β2 microglobulin; BTP, β-trace protein; CysC, cystatin C; DKK3, dickkopf homolog 3; DT, distal tubule; eGFR, estimated glomerular filtration rate; KIM-1, kidney injury molecule 1; NGAL, neutrophil gelatinase–associated lipocalin; PCT, proximal convoluted tubule; SDMA, symmetric dimethylarginine; TAL, thick ascending limb; UMOD, uromodulin.
a Acute kidney injury reports published, but no chronic injury usage reported.
Summary of Biomarkers of Acute and Chronic Renal Injury Evaluated in Chronic Kidney Injury and Disease.
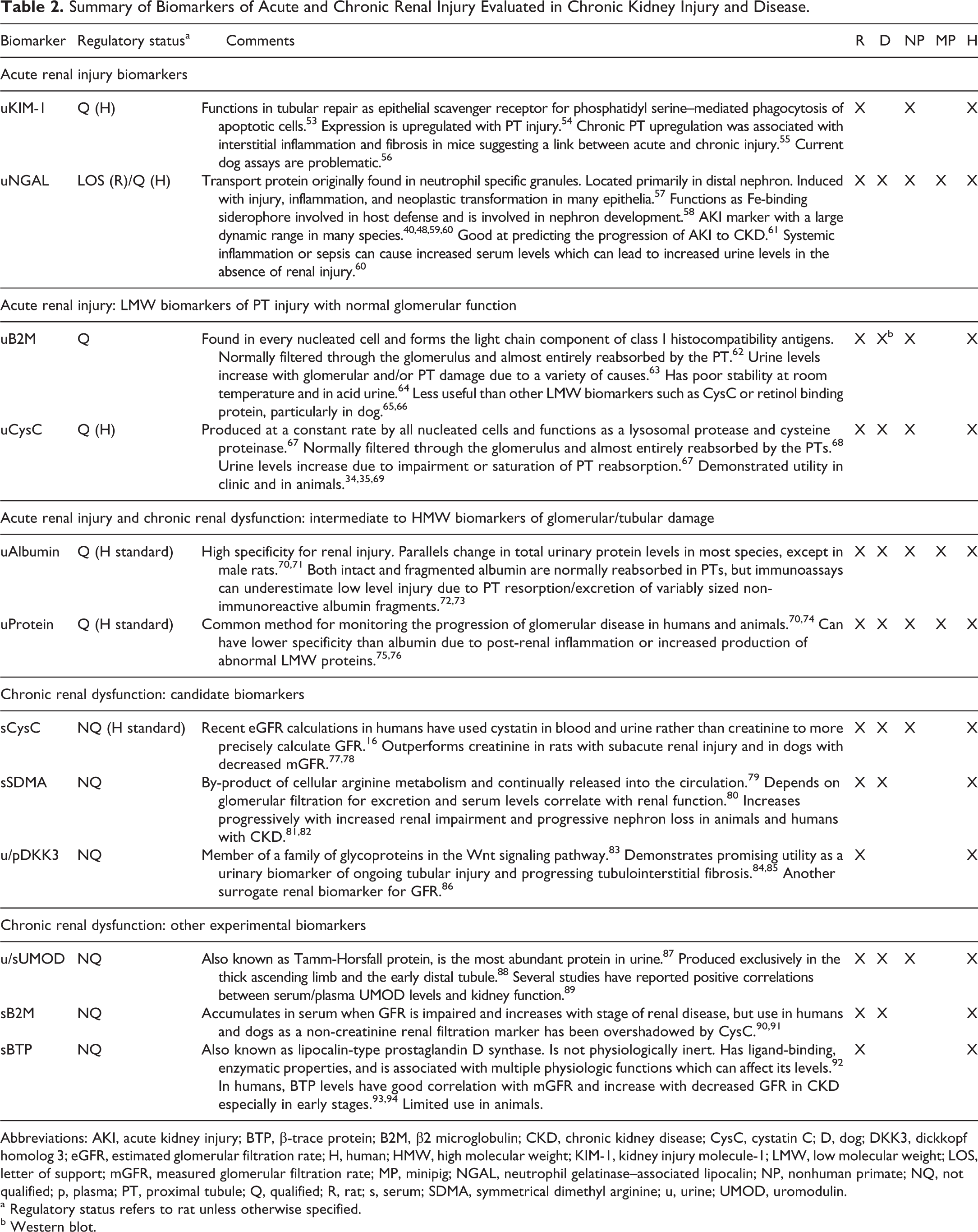
Abbreviations: AKI, acute kidney injury; BTP, β-trace protein; B2M, β2 microglobulin; CKD, chronic kidney disease; CysC, cystatin C; D, dog; DKK3, dickkopf homolog 3; eGFR, estimated glomerular filtration rate; H, human; HMW, high molecular weight; KIM-1, kidney injury molecule-1; LMW, low molecular weight; LOS, letter of support; mGFR, measured glomerular filtration rate; MP, minipig; NGAL, neutrophil gelatinase–associated lipocalin; NP, nonhuman primate; NQ, not qualified; p, plasma; PT, proximal tubule; Q, qualified; R, rat; s, serum; SDMA, symmetrical dimethyl arginine; u, urine; UMOD, uromodulin.
a Regulatory status refers to rat unless otherwise specified.
b Western blot.
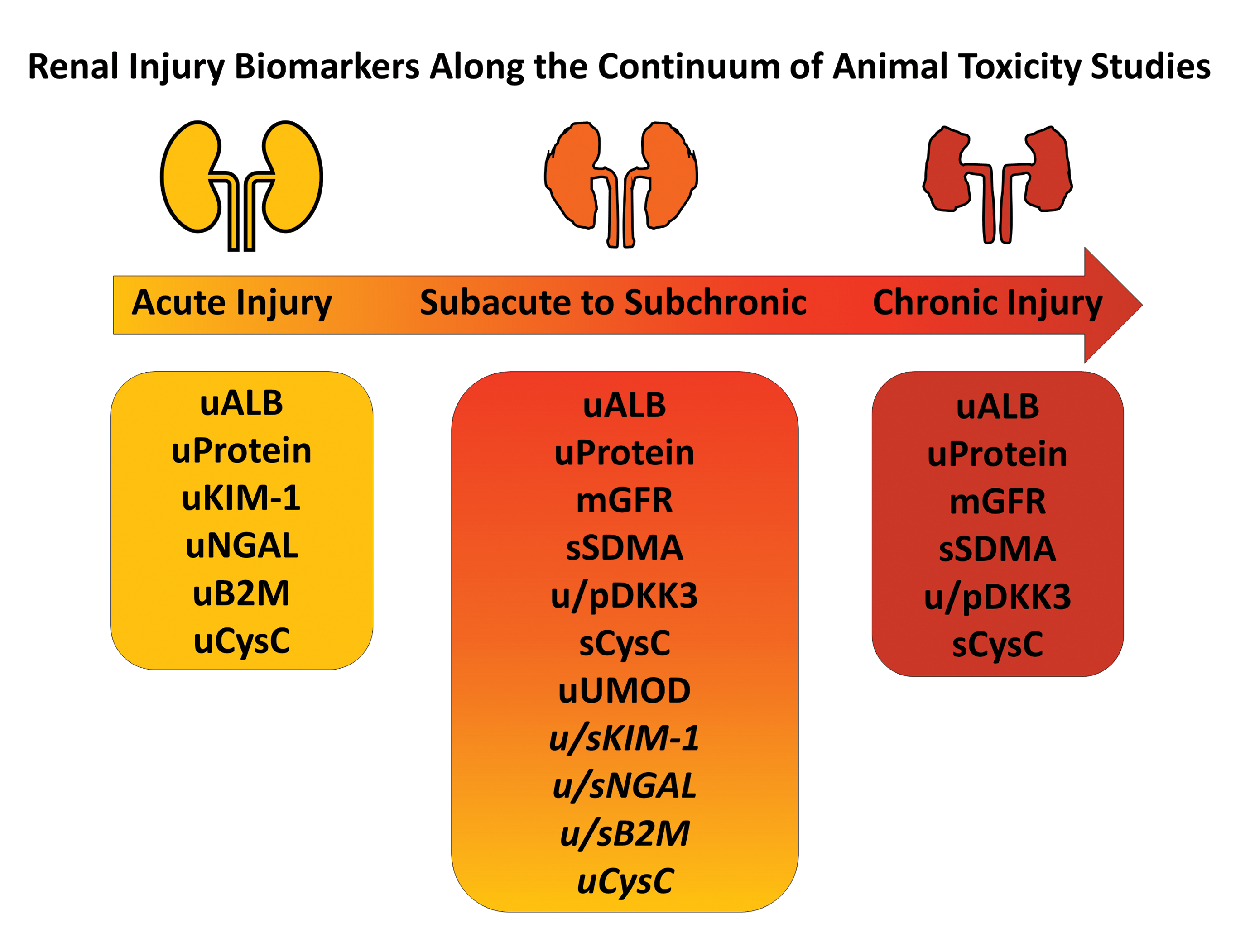
Renal injury biomarkers along the continuum of animal toxicity studies. Acute renal injury biomarkers (KIM-1, NGAL, B2M, urine CysC) have shown some utility in monitoring the progression of acute to chronic injury, but often their successful usage in subacute to subchronic renal injury has been dependent upon type of injury and/or species (italicized). In addition to the standard renal functional biomarkers (ALB, protein, mGFR), other biomarkers have shown promise in predicting and monitoring subacute to subchronic (SDMA, DKK3, serum CysC, UMOD) or chronic renal injury (SDMA, DKK3, serum CysC) in animal toxicity studies. ALB indicates albumin; B2M, β2 macroglobulin; CysC, cystatin C; DKK3, dickkopf homolog 3; KIM-1, kidney injury molecule 1; mGFR, measured glomerular filtration rate; NGAL, neutrophil gelatinase–associated lipocalin; p, plasma; s, serum; SDMA, symmetrical dimethyl arginine; u, urine; UMOD, uromodulin.
The following sections first discuss the current clinical standard biomarker of renal function (eGFR) and highlight 3 established biomarkers of AKI (urine albumin, KIM-1, NGAL) that may also be utilized in the chronic setting. Subsequent sections introduce 3 promising novel biomarkers of chronic renal dysfunction (CysC, symmetrical dimethyl arginine [SDMA], dickkopf homolog 3 [DKK3]) as well as 3 other candidate biomarkers (uromodulin [UMOD], B2M, β-trace protein [BTP]) that hold promise for detecting chronic renal injury.
Estimated GFR
Glomerular filtration rate is the gold standard used to diagnose, manage, and predict risk of end-stage renal disease (ESRD) in humans and animals. It is also used clinically to adjust dosing of renally excreted medications in patients with renal disease. 16 Glomerular filtration rate often serves as an outcome measure in clinical trials and is used to determine prevalence of CKD in epidemiologic studies. 95,96,97 Unfortunately, GFR is not readily measured directly and is generally assessed using the clearance of bolus-injected surrogates including creatinine, inulin, iohexol, or radiolabeled molecules at specific timepoints. 14,98 However, this is rarely done in the clinic due to cost, availability, difficulty of measurement, and inconvenience. 99,100 Likewise, it is logistically difficult and expensive to include in animal studies. To the authors’ knowledge, measured GFR (mGFR) has not been included routinely in toxicologic and safety pharmacology studies 101,102 and certainly would not be a practical end point to add to a routine good laboratory practice (GLP) study. Additionally, for regulatory toxicology studies, there could be a hypothetical concern for potential toxicity or drug–drug interactions with these surrogate molecules. 103 Technological advances in imaging equipment (optoacoustic tomography and transcutaneous fluorescent-sinistrin decay) have enabled use of portable units for more direct measurement of GFR in investigational experiments in animals and in institutional diagnostic applications, but these techniques are equally difficult to incorporate into nonclinical toxicology studies where large numbers of animals and strict time-sensitive protocols are in place. 3,104,105,106 This instrumentation requires advanced training and practice to effectively standardize or equilibrate between animals, and the time it takes to measure even 1 animal requires prospective planning for staggered dosing if multiple dose groups are to be measured on study. Even in human nephrology practice, it is extremely uncommon to measure GFR directly, and in clinical trials, GFR is routinely estimated using endogenous biomarkers in a variety of specific formulas that estimate urinary clearance (eGFR). 107,108 -110 All endogenous filtration biomarkers have determinants other than GFR (non-GFR determinants), which include production, tubular secretion or reabsorption, and extrarenal clearance that could affect the accuracy of GFR estimates. 111,112 Since these processes are usually not measured, surrogate variables to adjust for characteristics such as age and sex have been incorporated into eGFR equations. 113 Additionally, non-GFR determinants in one population might differ from the source population used to derive the equation, so estimating equations should be used in the populations in which they were developed. 114 For example, since urea and creatinine are eliminated through the dialysis treatment due to their small molecular weight, other low-molecular-weight (LMW) proteins (CysC, B2M, BTP) that have higher molecular mass are more useful to estimate residual kidney function in ESRD patients. 114
Some of the more commonly used eGFR equations include Cockcroft–Gault (CG), Modification of Diet in Renal Disease (MDRD) study, and the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. 115 -117 The CG is no longer recommended for clinical use since it does not use standardized creatinine values, does not account for body surface area (obese and elderly individuals), lacks inclusion of racial diversity, and overestimates kidney function. 117 –119 In contrast, the MDRD study and CDK-EPI equations use creatinine measurement procedures that are traceable to an isotope dilution mass spectrometry reference and contain variables that adjust the eGFR to account for creatinine differences due to age, sex, and ethnicity. 120 The CKD-EPI equation is currently recommended by Kidney Disease Outcomes Quality Initiative for estimation of GFR 70 and is more accurate and less biased than the MDRD study equation in populations with eGFR near and above 60 mL/min/1.73 m2 or older than 70 years of age. 118,119,121 In addition, the CKD-EPI equation more accurately predicts adverse kidney, cardiovascular, and mortality outcomes for patients with CKD. 122 However, the CKD-EPI equation does not work well for all patient populations including renal transplant patients, whereas some studies have shown better performance in Asians vs the MDRD. 122,123 All estimating equations have errors relative to mGFR, and errors will be greater for people who have non-GFR determinants that differ from the populations used to derive the equations (eg, vegetarians vs bodybuilders). 120 Attempts to adapt human eGFR equations for use in veterinary species have been unsuccessful, and currently, there are no widely accepted eGFR formulas for preclinical species. 124 –127
One of the measured parameters that are included in the estimation of eGFR in both animals and humans has historically been creatinine; therefore, creatinine clearance is a major determinant of eGFR. However, many other non-GFR determinants can influence creatinine levels, including age, sex, race, liver function, and intestinal flora (since creatinine is metabolized by intestinal bacteria). 11,12,112,128 –134 While the majority of creatinine is filtered unchanged through the glomerulus into the urinary filtrate, over 40% may be secreted via renal transporters depending on the species, and this transport may be affected by drugs. 12,135 –137 Tubular secretion can be increased to up to 50% of total renal elimination as GFR declines and can be supplemented with increased extrarenal elimination via the gastrointestinal tract. 11,130,138,139 Creatinine production differs between patients with stable CKD and those with progressive disease due to comorbidities, further limiting its utility. 30 In addition, patients with glomerular disease have progressive fractional hypersecretion of creatinine by renal tubules as the disease worsens leading to overestimation of GFR, so the use of creatinine as a filtration biomarker is inaccurate in these individuals. 132 Another gap in clinical monitoring during early GFR decline referred to as the “creatinine blind region” occurs when there is increased creatinine secretion and decreased creatinine generation. 140
Even though all current eGFR equations in humans include demographic adjustments of creatinine concentrations, GFR estimation remains imprecise for both under and overestimation of mGFR. 141 In order to improve GFR estimation, several alternate LMW biomarkers (CysC, BTP, and B2M) have been proposed and studied. Since these LMW proteins are mainly cleared from the blood through glomerular filtration, are almost completely reabsorbed by proximal tubular cells, and are degraded to smaller peptides and amino acids that are reabsorbed into peritubular circulation with minimal urinary excretion, 142 they were proposed as potential serum biomarkers of impaired GFR, and their increased urinary excretion was considered to be an indicator of tubular damage. 138,143,144 Several GFR estimation equations have incorporated these molecules alone or in combination. 145 The 2012 Kidney Disease: Improving Global Outcomes (KDIGO) CKD guidelines recommend the use of the CKD-EPI CysC or creatinine/CysC equation in certain circumstances where eGFR accuracy is critical or when CKD diagnosis using creatinine alone is uncertain. 16 Recently, the CKD-EPI collaboration has developed equations using the additional serum biomarkers BTP and B2M. Although BTP and B2M slightly underestimate mGFR in the higher eGFR range, combining these biomarkers has similar accuracy as that of the combined CKD-EPI creatinine and CysC equation. 99,146 In a recent study that evaluated the performance of 6 CKD-EPI GFR estimation equations (eGFR-EPI) against the gold standard urinary inulin clearance in patients with CKD, the combination creatinine/CysC equation provided the best overall estimate of mGFR with the lowest bias, highest precision and accuracy, and greatest classification. 145 The BTP and B2M equations displayed the worst performance with significantly less precision at all levels of GFR, and averaging the creatinine/CysC and BTP/B2 M equations did not improve or worsen the performance compared to the creatine/CysC equation alone. 145
Estimated GFR is absolutely critical to chronic renal functional assessment in human patients, but it is clear that to date mGFR is impractical to institute in nonclinical safety assessment studies, and eGFR calculations and interpretation in laboratory animals are still in their infancy. 124,125,147 Thus, surrogate indicators of chronic renal function are essential for providing a basis for a reliable nephrotoxic signal in animal trials that can be translated into clinical patients.
Established Biomarkers of Acute Kidney Disease That May Also Be Utilized in the Chronic Setting
Urinary Protein and Urinary Albumin
Urine protein excretion is frequently assessed in nonclinical toxicity studies and even in many clinical trials using rapid dipstick techniques that are qualitative, but only semi-quantitative. However, in the chronic setting, a more quantitative approach is often necessary to properly assess renal function. Both urine total protein and urine albumin are typically represented as concentrations and, like other urine analytes, are subsequently normalized to creatinine as a ratio. In humans, spot urine samples are usually taken from a first morning void, but in laboratory animals, urine is collected in a metabolism cage and the total volume collected is recorded for a defined time period of up to 16 hours depending on species or experimental design. 148 Thus, in laboratory animals, urine creatinine or urine volume can be used as normalization parameters. Urinary protein to creatinine ratio (UPCR) and UACR are 2 of the few biomarkers that are used to monitor the progression of chronic renal failure in humans, generally in association with calculation of eGFR. The 2012 KDIGO clinical practice guideline for the evaluation and management of CKD includes proteinuria in the staging of CKD. 16 Proteinuria can be differentiated on the basis of amount of protein (nephrotic [>3.5 g protein excreted over 24 hours] vs non-nephrotic), type of protein (albuminuria or LMW proteins), and underlying pathology (glomerular vs non-glomerular). 72,75 Proteinuria of renal origin can be classified into one or more of the following categories: tubular, overflow, or glomerular. 75,76 Tubular proteinuria results from tubulointerstitial disease affecting the proximal tubules and interstitium resulting in decreased proximal tubule reabsorption of proteins, especially LMW proteins (<25,000 Daltons) which under normal conditions are completely reabsorbed in the proximal tubules. 75,76 Overflow proteinuria is mostly associated with increased production or circulation of large quantities of proteins in disease states (eg, light chains in multiple myeloma, myoglobin in rhabdomyolysis) that exceed the reabsorption capacity of the proximal tubule, leading to excessive urinary protein which can be toxic to the tubules and cause AKI. 75,76 Glomerular proteinuria is associated with damage to the glomerulus, and more significant glomerular disease results in more severe proteinuria. Post-renal proteinuria can occur with inflammation, hemorrhage, and/or mucosal secretions from the genitourinary tract (eg, infections, nephrolithiasis, tumors) and resolves with resolution of the underlying condition. 75 Urinary protein levels can fluctuate considerably in both acute and chronic renal failure and may vary considerably even in patients with relatively normal renal function. 149 Changes in urinary protein levels remain the most reliable method for monitoring the progression of glomerular disease in human patients and animals. 70,74 In general, urinary albumin levels tend to parallel changes in total protein in most species. 70,150 The rat is an exception, as rat urine is protein-rich due to the presence of large quantities of proteins known as “major urinary proteins” including α2 urinary globulin. 71 This is especially true in male rats. 71 Consequently, urinary albumin excretion can be more specific as a biomarker of renal disease than urinary protein. 71,151 –154 Based on a rat renal micropuncture study, albumin absorption is distributed almost equally across the 3 segments of the proximal tubule. 155 Albumin in the filtrate occurs as both intact (immunoreactive) albumin and variably sized albumin fragments. However, an elegant study using 125I-albumin intravenously administered to rats demonstrated that only 1% to 2% of the albumin recovered in urine was the intact form and that the vast majority of recovered albumin (98%-99%) was fragmented and exocytosed both apically and basolaterally from proximal tubule cells. 73 Thus, contrary to past dogma, more albumin than previously thought crosses the glomerular barrier in health, and not all urinary albumin is completely resorbed into the blood stream. 73 This observation also has clinical implications in that immunoturbidimetric assays can underestimate severity of albuminuria since only intact albumin is detected by these assays. 72,156 In addition, there are situations, particularly in chronic renal disorders, where urine albumin does not parallel total protein in the filtrate. Plasma albumin levels can decrease with chronic loss, and with a higher percentage of renal uptake, changes in urinary albumin may be overshadowed by large protein spillage. Therefore, in these situations (eg, certain tubulointerstitial and vascular nephropathies including polycystic kidney disease), changes in urinary albumin levels may not detect progressive renal damage or dysfunction with advanced chronic renal disease. 21,22 Thus, monitoring urinary protein and albumin levels is of clear benefit in the analysis of acute, subchronic, or chronic nonclinical toxicity studies where renal toxicity is predicted or suspected. However, there remains a substantial need for biomarkers which can provide more sensitive and specific data on the progressive renal dysfunction that may accompany drug or chemical administration with more chronic duration.
Kidney Injury Molecule 1
KIM-1 is a type 1 transmembrane glycoprotein on the cell surface of epithelial and immune cells that serves as a scavenger receptor for oxidized low-density lipoproteins and phosphatidylserine-mediated phagocytosis of apoptotic cells. 53,157 –160 It is virtually absent in healthy kidneys, but its expression is upregulated in proximal tubule cells following acute injury. This is especially true after ischemia–reperfusion injury and drug-related renal toxicity. 55,161 -166 The upregulation of KIM-1 following an ischemic insult is thought to be associated with the renewal of functional and morphological integrity of kidneys. 161 Kidney injury molecule-1 upregulation has an anti-inflammatory effect by mediating epithelial cell phagocytosis of apoptotic cells leading to downregulation of innate immunity and inflammation through effects on nuclear factor-κB signaling. 167 In addition to its role in acute tubular damage, glomerular KIM-1 expression was increased in parallel with proteinuria and podocytopenia (decreased podocytes in the glomerular tuft) in a diabetic animal model supporting its use as a potential biomarker for glomerular injury in proteinuric kidney disease. 168 Kidney injury molecule-1 has also been shown preclinically to be upregulated in the later phases of AKI suggesting a role in renal repair. 169 However, chronic proximal tubule overexpression of KIM-1 in a mouse model caused interstitial inflammation and fibrosis linking acute and recurrent injury in the progression of chronic renal disease. 55 Thus, KIM-1 can be used as a biomarker of tubular damage, but it can also cause tubular damage by promoting interstitial fibrosis, so chronic elevations in KIM-1 may warrant additional monitoring for ongoing renal damage.
Following acute injury, the membrane bound domain of KIM-1 is cleaved via a metalloproteinase-dependent process releasing it into the urine and extracellular space. 170 Urinary KIM-1 has been shown to be a good predictor of acute renal injury prior to detectable changes in eGFR. 161,171 Numerous recent studies in humans have evaluated the biomarker potential of urinary KIM-1 in CKD. 22 Kidney injury molecule-1 was useful in the detection of early CKD of uncertain etiology and was better than conventional biomarkers. 28 Urinary KIM-1 was also higher in patients with proteinuria and normal renal function, suggesting that urinary KIM-1 should be evaluated in conjunction with urine protein excretion in patients with preexisting nephropathy. 172,173 Antihypertensive therapy in a rat model and in patients decreased urinary KIM-1 in parallel with lowering urinary protein excretion suggesting an improvement of proteinuria-induced tubular injury; however, there was no change in creatinine clearance despite decreased KIM-1 excretion. 174,175 Kidney injury molecule-1 may also be a potential biomarker of CKD caused by tubulointerstitial damage since its expression correlates with the degree of interstitial fibrosis and inflammation in affected patient kidneys and in a rodent model. 54,176,177 –179 In addition, several studies in various patient disease cohorts including diabetics demonstrated the ability of urinary KIM-1 levels to predict the development and progression of CKD. 21,179 –186 A meta-analysis of 10 studies found that urinary KIM-1 had borderline significance as an independent predictor of stage 3 CKD. 187 Kidney injury molecule-1 in combination with urinary NGAL improved the ability to predict renal outcomes in other human chronic renal disease cohorts (glomerulonephritis, vasculitis, lupus nephritis). 27,188 In a longer term study, plasma KIM-1 predicted future eGFR decline and CKD risk in healthy middle-aged patients. 189 Serum/plasma KIM-1 was also elevated in patients with CKD of various etiologies. 190 In longitudinal studies in normoalbuminuric diabetic patients, serum/plasma KIM-1 was found to be strongly associated with renal decline (eGFR) and/or onset of CKD stage 3 or higher. 190 –192
Numerous rodent DIKI studies demonstrated the utility of urinary KIM-1 for detection of AKI, 47,52,77,193 –199 but performance of urinary KIM-1 in nonrodent species has been less consistent. Early increases in urine KIM-1 were seen in some AKI studies in monkeys 45,48,196,200 but not in others. 201 In the dog, some authors describe diagnostic utility for KIM-1, 202,203 while others, including the current authors, report poor accuracy of KIM-1 in dog DIKI studies. 3,4,56,198,204
Urinary KIM-1 is rarely used in veterinary clinical practice due to questionable single analyte assay performance in dogs. Also, canine-specific KIM-1 enzyme-linked immunosorbent assay (ELISA) 198,202,203,204 and/or multiplex assays with KIM-1 205 have underperformed other biomarkers including NGAL in dogs. Nevertheless, in dogs, urine KIM-1 has been shown to be increased in AKI and in conditions such as babesiosis, 202 but not in CKD. 203 In cats, KIM-1 was detected by a qualitative urine immunoassay in those at risk of AKI but not in cats with CKD. 206
In rat investigational studies of DIKI, elevated KIM-1 levels have persisted during regeneration phases following dosing with gentamicin and carbapenem, a reflecting ongoing repair. 77 However, in other investigational studies in rodents, peak values of KIM-1 in DIKI have occurred at 3 to 6 weeks, with values steadily declining afterward despite microscopic evidence of ongoing renal degeneration or fibrosis. Kidney injury molecule-1 urine and serum levels rose initially in a mouse ischemia–reperfusion injury model, but steadily declined from day 7 post injury to day 28 despite ongoing tubulointerstitial fibrosis. 207 Additionally, in a rat model of adriamycin nephrotoxicity, urinary KIM-1 was elevated at 12 weeks and reversed following 6 weeks antiproteinuric treatment (renin–angiotensin system blockade) but was not accompanied by reversal of interstitial fibrosis. 175 Kidney injury molecule-1 has also been less consistent when utilized in chronic nonclinical drug studies and has often been undetectable in rodents when assessed in toxicity studies of 6 months or longer duration (unpublished data). In some rodent subchronic studies, KIM-1 levels increased following toxicant exposure concordant with renal histopathology and declined to normal or near normal levels during the recovery phases. 194,208 In other rodent subchronic studies, KIM-1 values remained elevated after 90 days of Ochratoxin A dosing where it outperformed other biomarkers, 52 and at 12 weeks following adriamycin administration, 209 suggesting it could be a biomarker of subchronic nephrotoxicity. Interestingly, in a subclinical adenine or aristocholic acid-induced CKD model where AKI was induced following a recovery period by low-dose cisplatin, 210 KIM-1 responses to AKI were lower and delayed in CKD rats compared to non-CKD rats and were inversely linked to the extent of prior cortical damage. 210 These data suggest that in the presence of known CKD in rats, urinary KIM-1 levels may vary indicating the need for thresholds to be lowered, combined with delaying and/or including serial sampling. The totality of these findings indicate that urine KIM-1 alone may not be ideal for detecting chronic renal injury in longer term animal toxicity studies but may add value as part of a panel of other biomarkers.
Neutrophil Gelatinase–Associated Lipocalin
Neutrophil gelatinase–associated lipocalin, also known as lipocalin-2, is a glycoprotein bound to matrix metalloproteinase-9 in human neutrophils and is involved in transport of hydrophilic substances through membranes to maintain cellular homeostasis. 211 Neutrophil gelatinase–associated lipocalin is involved in the early stages of nephron development 212 and is widely expressed in adult tissues (lung, gastrointestinal tract, liver, and kidney), but its main sites of expression are white blood cells and kidney loop of Henle and collecting ducts. 57,213 Neutrophil gelatinase–associated lipocalin inhibits bacterial growth via binding bacterial siderophores and sequestering iron. 58,214 Tubule cells process the NGAL-Fe-siderophore complex leading to increased cytoplasmic iron concentrations, activation of iron-dependent processes, and decreased oxidative stress. 215,216 Neutrophil gelatinase–associated lipocalin is markedly induced in epithelial cells in response to injury, inflammation, and neoplastic transformation. 57,216,217 Neutrophil gelatinase–associated lipocalin was identified as one of the earliest and most upregulated genes in rodent tubular and ischemic renal injury models, especially in the distal nephron. 218,219 However, in a mouse model of cisplatin AKI, NGAL was induced predominantly in proximal tubules. 220 Neutrophil gelatinase–associated lipocalin was also shown to be an early biomarker of tubular injury in dogs with experimentally induced AKI 196,204,221 –223 or naturally occurring AKI. 59,224 –228 In an experimental model of septic AKI in pigs, both serum and urine NGAL increased in association with tubular histopathology in septic animals compared to controls. 60 Even though most research supports a protective role for NGAL in AKI, studies in mice revealed that NGAL upregulation is involved in the progression of chronic renal injury in humans and certain strains of mice by modulation of epidermal growth factor–mediated tubular mitogenesis and cyst formation. 217 In a mouse model of ischemia–reperfusion injury, NGAL increased continuously during the AKI-CKD transition (from day 3 to day 28 post injury), whereas KIM-1 declined gradually in the chronic phase suggesting that NGAL may be able to dynamically monitor AKI-CKD progression. 207 Similarly, in a rat model of adenine-induced tubulointerstitial injury, urinary NGAL levels were increased during the AKI phase and remained elevated for 4 to 8 weeks in the CKD phase. 229
Neutrophil gelatinase–associated lipocalin exists in multiple forms, with 25 kD monomers considered to be of renal tubular origin and dimers of the 25 kD monomer of neutrophilic origin. 230 It should also be noted that certain NGAL forms of systemic origin can cross the glomerular filtration barrier, contributing to urinary NGAL excretion. In a study comparing the utility of serum NGAL in AKI and chronic kidney injury models in rats and mice, serum NGAL had variable success in detection of kidney injury across all of the models, which is not surprising given that serum NGAL can lack specificity for renal injury due to production and release by neutrophils and nonrenal epithelia (liver, bladder, lung, stomach, small intestine, pancreas, prostate gland, mammary gland, adrenal gland, thymus) in response to injury. 231,232 In a mouse ischemia–reperfusion kidney injury model, serum and urine NGAL increased continuously during progression of AKI to CKD compared with serum and urine KIM-1 which peaked in the acute phase and declined during the chronic phase. 207 Similarly in humans, serum and urine NGAL were good prognostic biomarkers for predicting the progression of AKI to CKD in sepsis-associated AKI patients. 61 Because of potential nonrenal sources of serum NGAL, the association of serum NGAL changes with renal injury is ideally informed by effects on biomarkers of systemic inflammation as well as changes in other renal injury biomarkers.
In humans, urinary NGAL has been shown to be a good predictor of renal injury prior to detectable changes in eGFR 28,179,233 and has also been shown to be inversely correlated with eGFR while being directly correlated with interstitial fibrosis and tubular atrophy. 179,234 Investigators have also shown in CKD patients that increased urine NGAL reflects residual renal function and is directly correlated with serum creatinine and proteinuria, and inversely with GFR. 235 As GFR decreases and urine protein and serum creatinine increase in early CKD, urine NGAL increased proportionately in the majority of patients until late in disease progression when advanced stages of CKD and severe renal tubular degeneration and atrophy resulted in diminished renal reserve. 235 Unlike KIM-1 and NAG, urine NGAL was shown to be an independent predictor of ESRD risk and of mortality for patients with CKD. 187 Similarly, in elderly CKD patients, increased serum NGAL reflected renal impairment and was an independent risk biomarker for progression of ESRD. 236 However, other studies have shown that urine NGAL did not provide additional information beyond the standard renal biomarkers in predicting outcomes in patients with ESRD but did improve prediction of CKD progression in elderly patients with low-grade proteinuria 237 and in diabetic patients. 238 Similarly, plasma/serum NGAL improved upon serum creatinine and eGFR in predicting the severity of tubulointerstitial infiltrates and fibrosis in patients with early CKD and predicted renal dysfunction in patients with early diabetes and children with CKD. 239 –241
In the clinical veterinary setting, urinary and, to a lesser extent, serum NGAL have shown promise as biomarkers of CKD progression. In a study of naturally occurring CKD in dogs, higher urine and serum NGAL were associated with death, and serum NGAL was better than serum creatinine at predicting clinical outcomes. 226 In another study in dogs with naturally occurring CKD, serum NGAL was a good indicator of CKD severity and correlated with serum creatinine, but the serum NGAL values in dogs with advanced CKD had a wide range of variation suggesting that dogs with more stable CKD may not have on-going renal tubular injury leading to serum NGAL increases and that urine NGAL may be a more sensitive disease biomarker in these dogs. 242 Likewise, in dogs presenting with azotemia, plasma NGAL levels were able to differentiate between those with AKI (higher NGAL) versus CKD. 227 Increased urinary NGAL was shown to occur early in the development of CKD, correlate with glomerular and tubulointerstitial lesions, and/or be associated with shorter survival in dogs. 65,225 –227,243 –247 In dogs with x-linked hereditary nephropathy, urinary NGAL differentiated affected from unaffected dogs early in the disease process, with increases during early to mid-stages of disease, but it was not helpful for monitoring mid- to late-stage renal failure. 65 Similarly, cats with higher urinary NGAL concentrations have more rapid CKD progression. 248
Urinary NGAL to creatinine ratios have been quite successful in detecting DIKI in animals including mice, rats, dogs, and nonhuman primates. 3,4,40,45,47,51,52,194 –196,223,236,249,250 However, the ability of NGAL to detect minimal kidney lesions in monkeys was inferior to KIM-1 and CLU. 45 In addition, NGAL was the most nonspecific biomarker evaluated in a series of rat studies comparing renal and nonrenal toxins, and increased urine and plasma NGAL were observed in a variety of different end-organ toxicities in the absence of renal toxicity. 40,47 In bacterial nephritis, NGAL was elevated up to 6 weeks post infection in rodents. 251 Similarly, dogs with lower urinary tract infections or diseases and pyuria can have increased urinary NGAL. 225,252,253 In cats, 3 forms of urinary NGAL were detected and 2 were found to be associated with renal diseases of either the upper (monomeric form) or the lower (dimeric form) urinary tract. 254 Increased NGAL concentrations in the urine of animals with lower urinary tract diseases having inflammation could be due to the presence of neutrophils, and as with sepsis or systemic inflammation, determining the molecular forms of urine NGAL may allow for more specific and precise detection of urinary tract disease. 245
In chronic animal safety assessment studies of 3 months or longer, urinary NGAL values tend to wane over time similar to KIM-1. Therefore, urinary NGAL values appear to be ineffective in monitoring the progression of chronic renal injury or fibrosis as NGAL levels have been inconsistently elevated despite sustained injury (unpublished data). Both KIM-1 and NGAL were not increased at any time point throughout a 36-week study tracking the progression of nephropathy in hypertensive and obese rats. 255 One explanation for this negative result is the relatively more severe glomerular damage than tubular injury in these rats, 255 but transiently increased NGAL expression may have been missed. The poor performance of NGAL in longer term nonclinical safety studies has led to the search for other renal biomarkers that may augment current biomarkers used in chronic animal studies. In addition to KIM-1 and NGAL, UMOD (discussed later) has also been shown to predict AKI before renal functional decline. 161,179,235
Promising Novel Biomarkers of Chronic Renal Dysfunction That Can Be Added to Nonclinical Studies
Cystatin C
As the authors noted, currently utilized biomarkers generally have limitations in the chronic setting, but there are some potential alternatives available to fill this important niche. Cystatin C is one of the promising new analytes that may make the transition from AKI to CKD biomarker and, in the context of nonclinical safety, work as both an acute and chronic renal biomarker. 45,47,51,69,198,236 As noted above, recent eGFR calculations in humans have utilized cystatin in blood and urine rather than creatinine to more precisely calculate GFR, but this has not generally been an accepted practice in nonclinical safety assessment due to limited availability of CysC testing and the overall difficulty in validating eGFR calculations in animals.
Cystatin C is an LMW protein produced at a constant rate by all nucleated cells and functions as a lysosomal protease and cysteine proteinase. 67 It is freely filtered at the glomerulus with almost complete reabsorption and catabolism in the proximal tubules and it is not secreted by the tubules, so it is minimally present or absent in urine under normal circumstances. 68,256 However, similar to other LMW proteins, impairment or saturation of proximal tubule reabsorption can lead to marked increases in levels of urinary CysC in both animals and humans and thus is biomarker of acute tubular injury when glomerular function is not affected. 257,258 Since the sieving coefficient (measure of equilibration of a substance through a semipermeable membrane) of CysC is close to unity, the renal clearance of CysC approximates GFR, and serum CysC has been considered to be an effective alternative to creatinine as a biomarker of GFR. 259,260 In addition, CysC-based GFR estimates do not require correction for muscle bulk, diet, age, gender, and race. 143,261 –263 However, some researchers found that older age, male gender, greater weight and height, cigarette smoking, thyroid disease, malignancy, corticosteroid therapy, pregnancy, inflammation, and/or higher C-reactive protein levels are associated with increased serum CysC values. 99,264 –268 In addition, the nonrenal clearance of CysC in both healthy humans and rats was found to be substantial and approximately 15% of the total clearance. 259,269 The relative proportion of CysC eliminated extrarenally increases with decreasing GFR and was shown to overestimate renal clearance in conditions such as dialysis patients with advanced renal failure. 269,270 Despite these limitations, serum CysC has been shown to be useful for detecting slight decreases in renal function in early renal failure in various patient groups 120,271 –273 and to predict mortality in CKD independent of GFR. 274,275 Serum CysC also correlated better with mGFR than creatinine concentration in stage 3 CKD patients and was a good predictor of kidney dysfunction in patients with portal hypertension. 112,276 In addition, serum CysC was better able to predict adverse outcomes compared to serum creatinine. 277,278 Equations utilizing CysC combined with creatinine improve the accuracy of GFR estimation compared to equations based on single biomarkers. 99,113,122,145,275,279 Serum CysC has also been shown to be independently associated with cardiovascular disease, ESRD, and mortality. 280,281 Even though no causal relationship has been established to date, these associations may provide a better understanding of the common pathways of morbidity and mortality in cardiovascular and renal disease. 282 As the 2012 KDIGO CKD guideline recommends assessment of CysC based on evidence that eGFR with CysC instead of creatinine may better classify CKD in human patients, 16,62,265,275,279,280 there is promise for using CysC in chronic animal safety studies.
In the rat, serum CysC was shown to be more sensitive, specific, and reliable than serum creatinine and was able to detect different types of acute drug-induced lesions in various nephron segments (proximal and distal tubules) independent of the mechanisms of nephrotoxicity and even at levels of minimal injury. 77 In addition, in rat acute and subacute nephrotoxicity studies, urine CysC levels were increased, often earlier than other urine biomarkers. 47,52,197,198,208,283,284 In dogs with renal disease, serum CysC outperformed serum creatinine when compared to mGFR, and it was a more sensitive indicator of decreased GFR. 78,285 Serum CysC has also been proposed as an early biomarker for kidney dysfunction in critically ill dogs and in dogs with leishmaniasis, chronic nephritis, and diabetes mellitus. 286 –288 In addition, serum CysC had better sensitivity and specificity for detection of early stage I CKD in dogs than serum creatinine or SDMA. 289 Serum CysC was also increased in an AKI study in monkeys given gentamicin. 201 In dogs 244,290 and cats, 291,292 urinary CysC was found to be higher in animals with renal disease than in healthy animals or animals with nonrenal disease, but it was only increased in the urine of dogs with leishmaniasis during the azotemic stages of CKD. 293 In dogs with gentamicin-induced AKI, urinary CysC was found to be the most sensitive indicator of both structural and functional kidney injury relative to traditional and novel urinary biomarkers including urinary albumin, Kim-1, and NGAL. 204
Although these results are encouraging, considerable biological variability of serum CysC concentrations in dogs has been reported, suggesting that serum CysC may not be superior to serum creatinine for GFR determination, and further studies will be needed to confirm the value of serum CysC compared to serum creatinine. 294,295 As CysC analysis becomes more routine in the assessment of chronic renal disease in human practice, and eGFR estimation is improved in animals, there is the potential that nonclinical use of CysC in both urine and serum could also become routine.
Symmetrical Dimethyl Arginine
Symmetrical dimethyl arginine, one of the most promising candidates for a chronic renal biomarker, was discovered over 40 years ago. 296 The essential amino acid arginine is metabolized within mitochondria into 2 structural isomers: SDMA and asymmetrical dimethyl arginine (ADMA). 297 Symmetrical dimethyl arginine and ADMA are released into the cytoplasm and subsequently transported out of the cell and into the circulation. 79 Symmetrical dimethyl arginine is excreted primarily through the kidney (>90%) and undergoes minimal metabolism, 298,299 but ADMA is highly protein-bound, extensively metabolized enzymatically, and only 20% is excreted in the urine making it unsuitable as a renal biomarker. 299,300 In contrast, SDMA depends almost entirely on glomerular filtration for excretion, thus its serum levels correlate directly with renal function (GFR). 80,301 In addition, SDMA is not affected by nonrenal factors that influence creatinine and/or CysC such as lean body mass, food intake, inflammation, diabetes, or estrogen therapy. 302,303 Most importantly, SDMA levels have been demonstrated to rise progressively with increased renal impairment and progressive nephron loss in animals and humans with chronic renal disease. 80,81,82,304 –310 Chronic SDMA infusion does not affect renal function or kidney histology, so SDMA is not a direct cause of kidney injury. 311 In the absence of renal impairment, SDMA is not affected by acute inflammatory responses, liver and cardiovascular disease, or by diabetes mellitus. 312,313
Multiple studies in human CKD patients have demonstrated significant and progressive increases in SDMA relative to control populations with normal renal function. 82,301,302,314 In humans with impaired renal function, there was a strong correlation between SDMA and mGFR, and SDMA outperformed creatinine but was equivalent to CysC and eGFR equations. 80,302 Interestingly, in a study in healthy dogs of various ages fed prescription diets designed to maintain lean body mass, SDMA concentrations decreased significantly from baseline at 3 months and decreased further at 6 months, suggesting a dietary effect on renal function, and that SDMA could have clinical advantages over serum creatinine in monitoring responses to nutritional interventions. 315 Based on total biological variability (analytical, intra-, and interindividual) in healthy dogs, the use of SDMA as a single measurement is not recommended to screen for early renal functional loss, rather serial monitoring and use of the critical difference (CD) is recommended for monitoring kidney function instead of comparison with population-based reference values. 316 In renal ischemia models in both rats and mice, and in nephrectomized dog and rat models, serum SDMA levels increased progressively as renal mass was reduced. 311,317 –320 This increase in SDMA associated with nephron loss is not significantly diminished over time in contrast to NGAL, KIM-1, or OPN for which responses wane over time.
Symmetrical dimethyl arginine has been routinely used in companion veterinary practice for over 10 years as a reliable biomarker of progressive kidney disease in dogs and cats with a variety of chronic conditions including Lyme nephropathy, familial amyloidosis, renal dysplasia, and polycystic kidney disease. 304,321,322 One of the advantages of this assay is that it is performed in plasma or serum rather than urine, eliminating the need for urine collection or use of a metabolism cage. Venipuncture can be easily scheduled in GLP studies at specific timepoints weeks or months apart. In animals, SDMA increases compare favorably with declines in GFR. An investigative study in dogs comparing SDMA to mGFR demonstrated 90% sensitivity and specificity, similar to creatinine and much better than CysC. 323 In a separate study in dogs, similar sensitivity (90%) and specificity (83%) were noted compared with GFR as assessed by iohexol, but this depended upon the threshold used. 324 It has been noted that the optimal SDMA cutoff for detecting greater than a 40% decrease in GFR was serum SDMA >18 μg/dL. 324 Symmetrical dimethyl arginine has begun to be incorporated in target animal safety studies in dogs for detection of drug-induced nephrotoxicity by drugs intended for veterinary use in this species (personal communication). Symmetrical dimethyl arginine has been evaluated in human studies and in rare clinical trials, but the liquid chromatography–mass spectrometry analytical method required for the assay in humans has limited its availability and utility. 80 In animals, the liquid chromatography–mass spectrometry method is still the gold standard, but a more recent proprietary ELISA assay appears to outperform the older DLD ELISA. 325 While there is extensive experience in dogs and cats, this assay has only recently become commercially available in rodents and is scaled to be able to be run on large numbers of animals with a required serum volume of 50 to 100 uL. 326,327
Dickkopf Homolog 3
Another surrogate biomarker for GFR that has received increased attention in the human literature is DKK3. Dickkopf homolog 3 belongs to a family of glycoproteins that modulate the Wnt signaling pathway and has promising utility as a urinary biomarker of ongoing tubular injury and progressing tubulointerstitial fibrosis in animals and humans. Dickkopf homolog 3 has been shown to be a stress-induced, pro-fibrotic molecule of renal tubular origin with dual roles in promoting repair/regeneration acutely or facilitating progression to CKD after AKI, depending on the magnitude and duration of activation. 83,328 Although regenerative processes may occur in the early stages of renal injury via the Wnt pathway, continuous Wnt activation is detrimental and induces epithelial–mesenchymal transition and tubulointerstitial fibrosis. 329,330 Dickkopf homolog 3 is expressed in the developing kidney, is suppressed in adult life, and reexpressed in tubular epithelium under pathological conditions, eliciting a profibrotic T cell response. 85,328
Acute kidney injury is a known and common complication in patients after cardiac surgery. In preoperative cardiac patients at risk of AKI and subsequent loss of renal function, DKK3 has been shown to be an effective biomarker of renal tubular stress.
331
In a study of 733 patients, preoperative urinary DKK3 was found to significantly improve prediction of risk of AKI and kidney function loss after cardiac surgery (
Dickkopf homolog 3 also appears to be important in progression of renal disease in animals. Using DKK3-deficient mice, tubular damage and renal interstitial fibrosis were significantly reduced compared with wildtype mice after unilateral ureteral ligation as well as in a mouse adenine nephropathy model. 328 This study also used antibody-mediated blockade of DKK3 to elicit similar findings. Urinary levels of DKK3 in rodents follow disease progression, tubule atrophy, and interstitial fibrosis. 85 In an adriamycin nephropathy model in mice, DKK3 expression was upregulated in the tubule epithelium and interstitium. 332 Taken together, these findings suggest that DKK3 may be a useful diagnostic biomarker for both acute tubular injury and chronic renal disease involving tubulointerstitial fibrosis. Dickkopf homolog 3 is just beginning to be assessed in rodent and nonrodent toxicology studies (unpublished data), so it has yet to be determined whether this potential biomarker will be successful for prediction and monitoring of chronic renal disease progression in nonclinical studies, but since the biology of the Wnt pathway is similar in humans and animals, the future for this analyte is promising. An ELISA assay is available in humans that apparently will cross-react with most species of laboratory animals (personal communication), although other immunoassays and mass spectrometry have also been used for analysis of urinary and plasma DKK3. 84,86,333
In summary, DKK3 is a stress-induced, profibrotic, tubular epithelium-secreted glycoprotein that may be a useful marker for ongoing tubular injury and progressing tubulointerstitial fibrosis as it has dual roles in the promotion of acute repair/regeneration and facilitating progression to CKD.
Other Experimental Biomarkers That May Have Potential in Chronic Renal Dysfunction
Uromodulin
Uromodulin, also known as Tamm-Horsfall protein, is a glycosylphosphatidylinositol (GPI)-anchored glycoprotein. It is the most abundant protein excreted in urine and is the predominant protein in hyaline casts. 87,334 Uromodulin is produced exclusively in the thick ascending limb and early distal tubule and is released via proteolytic cleavage of the GPI-anchored ectodomain into the tubule where it coats the tubular lumen. 88 In addition to this canonical apical targeting, UMOD also sorts, to lesser degree, to the basolateral domain of the tubular epithelial cells and is released into the interstitium where it enters the circulation. 335 Uromodulin has been implicated in the pathophysiology of multiple human kidney diseases including polycystic kidney disease and nephrolithiasis and may have a protective role in urinary tract infections. 336 –340 It has a tendency to form gels in aggregates, and this is increased by the presence of urinary constituents including albumin, sodium, and calcium ions as well as acidic pH. 341 In patients with chronic renal failure, urinary UMOD has been shown to be decreased suggesting that serum UMOD levels may somehow reflect the amount of intact nephrons being linked to the “functional renal mass.” 342,343 Long-term studies in elderly adults demonstrated that low urine UMOD may identify persons at risk of progressive kidney disease and mortality. 344,345 In a study comparing urine and plasma UMOD levels in patients with and without CKD, decreases in plasma UMOD paralleled decreases in eGFR and outperformed urinary UMOD in the identification of early stages of CKD, discrimination between non-CKD versus advanced CKD, and correlation with eGFR. 8 Several studies have reported direct correlations between serum/plasma UMOD levels and kidney function. 89,346 –348 Because serum UMOD often correlates significantly with albuminuria, which is itself a strong predictor of CKD progression, UMOD may also play an important role in the prediction of CKD progression. 349 One prospective study showed that low serum UMOD concentrations in CKD patients was associated with an increased risk of ESRD, independent of the traditional risk factors for progression of CKD. 350 On the other hand, higher serum UMOD levels have been associated with better mortality outcomes for cardiovascular disease. 349,351 Thus, the evidence suggests that high levels of urine and serum UMOD are associated with better kidney, cardiovascular disease, and mortality outcomes. 338 Recent investigations in humans and transgenic mice demonstrated that UMOD inhibits the generation of reactive oxygen species in the kidney and systemically by suppressing transient receptor potential cation channel, subfamily M, member 2 activity, which may explain why lower UMOD levels are linked to poor outcomes and increased mortality. 352
Both animal and human studies suggest that UMOD may be involved in inflammation and progression of chronic urinary disease by entering the renal interstitium, either through basolateral secretion or urinary back-leakage in damaged tubules, and stimulating cells of the immune system. 353 Rare mutations in UMOD can result in autosomal dominant tubulointerstitial kidney disease, leading to CKD. 354 Another study showed that tubulointerstitial nephritis could be produced in rabbits by intravenous injection of urine or UMOD and was the result of a predominately cellular immune response directed against UMOD. 355 Uromodulin also has renoprotective effects, as shown in a mouse model of ischemia–reperfusion injury where UMOD knockout mice had more severe renal injury and increased toll-like receptor 4 (TLR4) expression that was localized on the basolateral rather than apical membrane of the proximal tubule S3 segment where it is normally expressed in wild-type mice. 356 These findings suggest that UMOD protects the outer medulla during renal injury by reducing inflammation through decreased interaction between interstitial proinflammatory TLR4 ligands and the TLR4 receptor. 356 However, in a rat model of pyelonephritis-induced AKI, urinary UMOD levels decreased, but paradoxically increased after 3 weeks, and the authors concluded urine UMOD was not a suitable biomarker in this model. 283 In a few AKI studies in primates, urine UMOD was shown to decrease early following injury, but its use in chronic studies or CKD has not been reported for this species. 191,357
In veterinary clinical practice, UMOD performance has been inconsistent in CKD. A few reports in dogs with chronic renal failure or urolithiasis have shown reduced renal excretion of UMOD. 358 –361 In dogs with stage 4 CKD, UMOD was undetectable or lower than controls in a long-term prospective study. 362 In dogs with babesiosis, UMOD did not distinguish between diseased and normal dogs and did not correlate with urine protein increases, in contrast with other biomarkers including KIM-1 and SDMA. 202,363 In a study comparing dogs with various degrees of CKD, only the most azotemic and proteinuric animals showed significant decreases in urine UMOD compared with controls. 364 However, in a recent study, decreased UMOD was observed by sodium dodecyl sulfate polyacrylamide gel electrophoresis in stage 1 non-proteinuric CKD dogs, suggesting that UMOD may be an early biomarker of renal dysfunction in dogs. 365 In a study of familial glomerular nephritis in Doberman dogs, the levels of UMOD changed throughout the course of disease such that UMOD levels increased above normal during the early course of nephropathy but declined as disease progressed. 366 Similar observations have been made in patients with early diabetes without GFR impairment, animal models of diabetes, and patients prior to the onset of CKD. 8,349,367 –369 However, as CKD progresses and GFR declines, continued fibrosis causes additional nephrons to drop out leading to decreased UMOD production. 8,89,369
Various animal knockout and transgenic studies have been done to further characterize UMOD and its role in CKD. 370 Recently, the use of urinary UMOD as a diagnostic biomarker of chronic renal disease in humans has been questioned since an analysis of UMOD in 77 CKD patients demonstrated that as many as 22% had normal urinary levels. 370,342 Although more studies are required to fully understand the biological function of UMOD, the fact that UMOD levels may either increase or decrease following injury makes use of UMOD as a biomarker of CKD challenging. At present, UMOD alone may not be a candidate chronic renal biomarker for nonclinical toxicity studies, but it might add value in a panel of chronic analytes.
In summary, decreases in plasma UMOD have been shown to parallel decreases in eGFR and outperform urinary UMOD in the identification of early stages of CKD, discrimination between non-CKD versus advanced CKD, and correlation with eGFR. Because UMOD is produced exclusively in the thick ascending limb and early distal tubule, it may be decreased in chronic renal disease and aid in the identification of early stages of CKD.
β2 Microglobulin
Early in the search for renal biomarkers of AKI and tubule injury, urinary B2M was proposed as an effective biomarker in humans. β2 microglobulin is an LMW protein that is found in every nucleated cell and forms the light chain component of class I histocompatibility antigens. It is normally filtered through the glomerulus and then almost entirely reabsorbed by the proximal tubules where it is subsequently catabolized into its constituent amino acids. 66 β2 microglobulin renal handling studies in rats, 371 dogs, 372 and humans 373 suggested that extraction of B2M may also occur from the peritubular capillary circulation into proximal tubules; however, another study in rats was unable to show that the extraction rate of B2M exceeded the rate of filtration. 374 β2 microglobulin urine levels increase following proximal tubular damage due to causes ranging from cardiac surgery, renal transplantation, or drug-induced renal injury. 63 In a prospective study of 252 children, urine B2M, NGAL, and KIM-1 all demonstrated good accuracy in predicting AKI in the emergency room situation. 375
In rats, urine B2M is sensitive to functional alterations of the proximal tubules, and albumin to B2M ratios may be used to determine the origin of proteinuria since glomerular injury is expected to have a higher albumin to B2M ratio than tubular injury. 376 Because urinary albumin levels increase 10-fold more than B2M in rats between the ages of 2 and 20 months, B2M may be of benefit in evaluation of renal injury in rats with chronic progressive nephropathy (CPN) since tubular function is thought to be preserved more than glomerular permeability in CPN. 376 Over the past decade, urine B2M has been studied extensively and qualified for preclinical use in tubular and glomerular DIKI in rat. 47,193,197 In a few cases, urine B2M was better able to detect glomerular injury than tubular damage in rats, 193 paralleling findings in human renal diseases with predominantly glomerular lesions (IgA nephropathy and systemic lupus erythematosus). 377,378 Thus, caution is warranted when interpreting high urine B2M levels as increased filtered protein load may also be due to glomerular injury and competitive tubular uptake. 357,379 β2 microglobulin assays are not widely available for nonrodent laboratory species, and even in the rat, values can be highly variable, 47,193,197 with normal values in males approximately 3 times those in females. 71 β2 microglobulin is degraded in acidic urine or after short periods at room temperature. 380,64 A few studies have demonstrated increased concentrations of urine B2M via Western blot in dogs with X-linked hereditary nephropathy and progressive renal disease, but B2M (as well as urine NGAL) was not helpful for monitoring mid- to late-stage renal failure, which was best monitored using retinol binding protein (RBP), another LMW protein. 65,381 For these reasons, B2M has not been very successful or widely used in nonrodent laboratory animals or veterinary nephrology. Other LMW biomarkers such as α 1 microglobulin or RBP can be substituted to assess proteinuria of tubular origin in AKI; however, many have not been evaluated in CKD. Despite these caveats, there is some evidence, mostly from the human literature, that urine B2M might provide some support for monitoring renal injury as an additive parameter in the clinic, for example, with lithium-treatment, cadmium toxicity, and human immunodeficiency virus patients treated with tenofovir. 382 –384
β2 microglobulin accumulates in the serum when GFR is impaired 90 ; however, serum B2M concentrations can be increased in conditions other than CKD such as neoplastic and inflammatory conditions. 385 Systolic blood pressure, age, weight, gender, total cholesterol, smoking, and urine protein excretion have been shown to be associated with increased serum B2M independent of GFR. 112,386 In humans, serum B2M has shown good correlation with eGFR, especially in genetic kidney diseases, but poor correlation with urine protein or albumin. 387 In human nephrology, its use as a non-creatinine renal filtration marker has been surpassed by CysC. Similarly, serum B2M was outperformed by serum CysC in the detection of renal damage in dogs with CKD and other kidney diseases. 91 This is because there are many nonrenal diseases that can affect serum values and there are conflicting data about the relationship between B2M and disease progression. 388,389 For example, catabolism in the kidney results in a very small percentage of B2M left in the filtrate under normal conditions, and altered cell binding from interaction with proteins that B2M chaperones in serum may cause fluctuations of serum B2M during periods of inflammation. 388,389 Since it has been known for over 50 years that B2M can be catabolized in rat proximal tubules, there are additional reasons that it is not a good substitute for creatinine or CysC in GFR equations. 371 These poorly understood factors may therefore affect both serum and subsequently urine levels, 390 and the degree of correlation between B2M and GFR varies greatly with the nephrotoxin studied. 388,391
In human CKD, B2M increased with the stage of disease 392 and could reliably predict GFR in many situations. 393,394 However, in a recent meta-analysis by the CKD Biomarkers Consortium, it was determined that B2M and BTP (discussed below) did not provide “substantial additional prognostic information over eGFR-creatinine or albuminuri, but might be appropriate in certain circumstances where eGFR was inaccurate or albuminuria was unavailable.” 395 Given the current data, we believe other biomarkers are better suited for use in monitoring chronic kidney injury in nonclinical safety studies although B2M may have added value in a panel of biomarkers in certain situations.
Beta Trace Protein
β-trace protein is also known as lipocalin-type prostaglandin D synthase. Glycoforms of varying molecular weights exist due to differences in posttranslational glycosylation that occur between tissues. 140,396 –398 The smaller “brain” origin glycoform predominates in the cerebrospinal fluid (CSF); whereas, the larger “plasma” type glycoform predominates in serum and urine. 397 β-trace protein was identified first in human CSF 399 and in rat brain. 92,398,400,401 β-trace protein represents almost 3% of the total CSF protein making it a useful diagnostic biomarker of CSF leakage. 399,402 It is also found in smaller concentrations in serum, breast milk, aqueous and vitreous humor, seminal fluid, and amniotic fluid. 403 In monkey, BTP was localized via in situ hybridization and immunohistochemistry to the proximal tubules, loop of Henle, and glomerulus. 404 Similarly in a diabetic rat model, BTP was detected in the proximal tubules but was not present in glomeruli. 405 Unlike serum creatinine, BTP is not physiologically inert and has both ligand-binding and enzymatic properties. β-trace protein binds with high affinity to a number of lipophilic substances including thyroid hormones, retinoids, β amyloid, and bilirubin. 92,406,407 It catalyzes the conversion of prostaglandin H2 to prostaglandin D2. 92 Prostaglandin D2 has several important physiologic functions including sleep regulation, nociception, inflammation, vasodilation, bronchoconstriction, platelet aggregation, bone remodeling, and adipogenesis. 140 Thus, there are many clinical conditions that could influence BTP levels. 408 –410 In addition, many of these non-GFR determinants of BTP are not included as variables in the eGFR equations. 112,140,408 However, unlike serum creatinine which can be decreased with hepatic dysfunction due to reduced hepatic production of the precursor creatine, BTP concentrations are not affected by liver dysfunction. 411
β-trace protein is almost completely excreted by the kidneys. It is freely filtered by the glomeruli, reabsorbed and metabolized in the proximal tubule, with minimal tubular secretion. 390,396,401,412,413 Unlike CysC and B2M, BTP is not completely reabsorbed by tubular cells and is detectable in urine of healthy individuals, which argues against tubular dysfunction as the sole cause of increased urinary levels. 144,396,397 In humans, 90% to 100% of radiolabeled BTP was recovered in urine indicating almost total renal elimination. 414 In a similar study in dogs, only 10.3% of intravenous radiolabeled BTP was recovered in the urine, suggesting that BTP was primarily degraded in the tissues. 415 However, both studies demonstrated rapid elimination of BTP from the serum, with a half-life of <1 hour. 414,415 The reason for the discordance between these 2 studies is not known. β-trace protein is highly lipophilic with a distribution volume in the dog comparable to the volume of canine plasma indicating primarily distribution within the blood, 415 but different BTP isoforms used in the 2 studies could have had different filtration properties. 140 Others speculated that BTP could be metabolized by tubular cells or other cell types, so additional studies are needed to understand renal BTP handling. 416
Serum BTP levels in humans were shown to have a good correlation with mGFR based on inulin clearance and nuclear medicine methods. 93 β-trace protein serum levels were found to be elevated in patients with decreased kidney function suggesting that this protein could be used as a diagnostic biomarker in kidney disease. 397,417 Multiple studies showed that increased BTP in serum and urine correlated well with decreased GFR in patients with CKD and that the increase was observed in the early stages of the disease. 94,418 –422 Studies of diverse patient groups including kidney transplant recipients, children, and patients with CKD found that BTP was better for detecting reduced GFR than serum creatinine, 423 –425 whereas others did not. 418,420,426,427 β-trace protein has been reported to be associated with higher mortality in patients with CKD and ESRD. 395,428 –430 Studies suggest age, 390,418,427,431,432 muscle mass, 433 gender, 134 ethnicity, 434 serum albumin levels, 112 urine protein excretion, 112 and body weight 408 may have an effect on BTP serum concentrations. β-trace protein was not considered to be an appropriate biomarker of GFR in pregnancy, 435 –437 but serum BTP for estimating GFR was reliable for assessment of kidney function in neonates. 438 β-trace protein is less affected by glucocorticoids than CysC as a biomarker of GFR in patients, especially after renal transplantation. 398 However, high-dose (but not low dose) steroid therapy has been shown to lower serum BTP. 426,439 In studies of elderly patients comparing the novel eGFR equations to mGFR, the BTP equation was less precise and accurate than the creatinine/CysC equation. 432,440 In other studies, the BTP equation alone or added to the creatinine/CysC equation did not improve upon mGFR. 90,441,442 White et al 140 found that the BTP equations provided no clear advantage over the traditional creatinine-based equations in adults due to imprecision of the equations and substantial non-GFR-dependent influences on serum BTP concentrations. As predicted, in a recent study comparing multiple CKD-EPI-GFR equations against the gold standard of inulin clearance, the BTP and B2M equations displayed the worst performance with significantly less precision at all levels of GFR and did not improve GFR estimation when added to the creatinine/CysC equation. 145 Taken together, these data suggest that increased serum BTP may occur due to nonrenal pathophysiologic processes or biological functions of BTP beyond its association with GFR; therefore, the utility of BTP for evaluation of chronic renal disease in patients is questionable. 140
Limited information is available regarding serum, urine, or tissue BTP levels in normal or diseased animals. 400,404,405,415,443 To the authors’ knowledge, neither urine nor serum BTP has been assessed in nonclinical studies. However, given the abundant human literature demonstrating its many issues, BTP is not likely as useful as CysC for GFR estimation. Based on current information, its use in animals for GFR estimation or as a biomarker of chronic DIKI cannot be recommended until further data regarding its utility in chronic renal toxicity in nonclinical species become available.
Other Alternative Methods for Assessing Chronic Renal Function
A variety of other biomarker modalities have been considered for noninvasive monitoring of CKD and chronic renal diseases including microRNA assessment, 444 –446 metabolomics, 365,447 and peptidomics 448 –452 as well as biomarkers originally intended for detection of glomerular diseases (eg, slit diaphragm proteins 453 –455 ). Unfortunately, while these approaches may have some utility in some chronic renal diseases in humans, they do not lend themselves to the high throughput and rapid analysis required for nonclinical GLP studies, nor can they be extrapolated to GFR estimation. Also, they provide no translational advantages in the nonclinical safety domain to help monitor potential DIKI in clinical trials. Such approaches, despite their promise in clinical CKD, are not discussed in this review.
Applications for Biomarkers of CKD
When it comes to renal biomarker implementation in nonclinical safety, the most frequent question from toxicologists and clinical pathologists is “In what situation should I utilize these biomarkers where they are most appropriate, and can they provide clinically translatable information without encumbering study logistics?” It is inappropriate to include these or any other analytes in a toxicity study simply because they are available. It is likely that these parameters will be part of a panel of renal biomarkers that include acute biomarkers such as urinary KIM-1, NGAL, and albumin to cover both acute and chronic renal events. However, there are specific situations where the judicious use of the SDMA, CysC, DKK3, or other assays discussed above may be valuable to clinicians overseeing patients in clinical trials. The first situation is probably the most obvious and common but potentially has the most potential for overuse. This occurs when a potentially progressive tubulointerstitial kidney lesion is noted in 2- to 4-week acute or subacute toxicity studies in animals, and there is concern that the lesion could progress to chronic renal failure. If the lesion initiated in the acute period, these chronic biomarkers can be evaluated in studies of 6 months or longer where NGAL or KIM-1 might no longer be increased since these biomarkers tend to wane over time. In other cases, a renal lesion is found that is very subtle when it is noted in 2- to 4-week studies, and there is no meaningful increase in biomarkers such as urinary KIM-1, NGAL, or albumin. It is possible that progression could result in a more severe lesion by 3 or 6 months of dosing and that a panel of renal biomarkers which would include both acute and chronic biomarkers could provide some confidence for clinicians that the lesion could be monitored in the clinic, particularly if the safety margin was small and the lesions occurred at or near clinical parity. It should be stressed that the clinical safety margin, nature of the lesion, and potential clinical translatability are all important factors in the decision to add chronic biomarkers in subsequent studies. If a nephrotoxic signal occurs only at animal doses that represent 20-fold or higher multiples of clinical exposure or was limited to non-tolerated doses resulting in morbidity in animals, there is little to be gained by adding any renal biomarkers in longer term animal studies. If the renal toxicity is represented by recurrent tubule degeneration and/or necrosis typical of AKI, then acute renal biomarkers are likely to be as effective in detecting renal injury as the novel biomarkers discussed above and may be useful in the clinic to monitor for potential nephrotoxicity as additional nephrons undergo degenerative changes. In contrast, a very subtle lesion or changes that are distinct from AKI (eg, fibrosis or atrophy as noted below) may be better candidates for inclusion of chronic renal functional biomarkers than acute injury biomarkers which are less likely to demonstrate modest structural changes.
As another example, a problematic gap in long-term safety evaluation occurs when the lesion is not typical of AKI and necrosis or degeneration are not the primary features. In this circumstance, most acute renal biomarkers would not be expected to change significantly. Examples include animal studies where drugs that have induced minimal interstitial fibrosis and tubular atrophy after several weeks of dosing may cause regulatory or clinical concern for progressive renal fibrosis in humans or inflammatory conditions that raise concern for potential chronic interstitial nephritis in patients. The authors are familiar with 2 different proprietary drug candidates that induced mild fibrosis in definitive GLP studies in rats or dogs, but while the lesions were still present after 3 months, neither progressed in severity in longer term chronic studies based on histopathology. However, regulatory concern over the lesions resulted in clinical holds and a project cancellation due to a lack of a reliable translatable biomarker in the clinic to augment eGFR alone. There are also examples of drugs which have induced transient vascular ischemia in the kidney causing very small infarcts at irregular and unpredictable intervals. In these cases, acute renal biomarkers could miss the lesion if sampling is not performed near the time of injury, and the long-term decline in renal function would be difficult to monitor or prevent due to minimal severity, renal compensatory changes, or sampling at a time that is distant from the peak injury or functional defect. With the trend among pharmaceutical companies for developing highly immunomodulatory molecules/biologics as anticancer therapeutics, we have noted a large number of compounds (proprietary information) which induce enhanced inflammatory infiltration into the renal interstitium replacing parenchyma as a function of suprapharmacology. There are similar examples in the literature, including lymphohistocytic inflammation reported in the kidneys of monkeys treated with an anti-cytotoxic T-lymphocyte-associated protein 4 antibody, 456 and there are several clinical reports of acute interstitial nephritis associated with check point inhibitor administration. 457 –461 In many of these examples, urinary biomarkers have not been effective in identifying lesions except in the most severe cases, and regulatory concern has extended to the possibility that these may mimic the syndrome of drug-induced interstitial nephritis, which is life-threatening in humans (albeit very rare) and generally not predicted in animals. 462 Antisense oligonucleotide therapies, particularly of the 2’-O-methyl or 2’-O-methoxyethyl class, have often been associated with perivascular inflammatory infiltrates, and the kidney is one of the more common locations for this lesion. 463,464 There have been several highly publicized regulatory issues in the kidney with these compounds, and the practical use of chronic renal biomarkers would help alleviate some of this regulatory concern. 465
Applications for Biomarkers of Glomerular Injury
Another relatively common situation where the use of chronic renal biomarkers in nonclinical studies would be extremely beneficial to safety scientists overseeing clinical trials involves nephrotoxic compounds that affect the glomerulus rather than the tubules. While by far the most common glomerular toxicity encountered by nonclinical toxicologic pathologists is related to immune complex disease associated with the administration of humanized antibodies and peptides to nonhuman primates, this is not a situation that lends itself to the use of chronic renal biomarkers. 465 –467 The rapidly progressive glomerulonephritis (RPGN) produced by anti-drug antibody-related immune complexes is too fulminant to require chronic dosing to observe a signal, and these preclinical immune reactions are not predictive in humans. 465 Also, most cases of immune-mediated glomerulonephritis due to drug or chemical treatment may have a rapid onset and fulminant course typical of human RPGN, thus human trials would not benefit from the addition of chronic renal biomarkers in longer term animal studies. In contrast, there are other types of glomerular disease in toxicologic studies that have a more protracted progression and where the clinical translatability is less clear, including glomerulosclerotic lesions and/or those that are related to intracellular accumulation of xenobiotic substances. 465,468 In these situations, serial UPCR measurement along with surrogate biomarkers of GFR such as SDMA, CysC, or DKK3 could track the decline in renal function that accompanies glomerular injury enabling assessment of the development of the lesion in individual animals over time, instead of comparing microscopic lesions across dose groups at the end of dosing. While these analytes may be utilized directly as human biomarkers in clinical trials, the information derived from animal data can also be directly compared to alterations in eGFR in humans, which assesses the same factors. Thus, direct comparison of biomarker data or clinical translation would not be necessary.
Applications in Discovery Pathology
The final area of unexplored utility for these chronic renal biomarkers is in discovery pathology. Clinical pharmacology studies of renal impairment in human patients are often required in phase II or III trials when the intended target population for a drug includes patients with below normal renal function. In general, supportive studies in animal models of renal disease are not necessary to begin these trials. However, in early phases of candidate selection of potential drugs that may be intended for these renally impaired populations, it is quite common to compare candidate compounds or to run investigational studies with lead candidates in rodent renal models looking for efficacy in restoring or maintaining renal function as well as eliminating candidates with unacceptable nephrotoxic profiles. In these situations, SDMA, DKK3, CysC, or even GFR measurement are an excellent means of determining potential effects on renal function over weeks or months. For some drug indications such as lysosomal storage diseases or polycystic kidney disease, serial use of these biomarkers in the same animal is probably the most critical area where information might be obtained that could have a direct effect on the prospects of drug success. Currently, histopathology and urine protein are the main parameters utilized in these long-term animal model studies to compare between drug candidates, but the variability of morphologic changes between individual animals often makes histopathologic assessment difficult and inadequate.
Summary and Conclusion
Estimation of GFR is a critical component in assessing chronic renal function in human patients and along with UPCR and UACR is the cornerstone of monitoring potential kidney adverse events during clinical trials. Unfortunately, GFR measurement is often logistically impractical to implement in nonclinical safety assessment studies, and eGFR calculations in animals are not yet available. 124,125,147 Thus, surrogate analytes of chronic renal function are needed to provide a basis for reliable translation of potential nephrotoxic signals in chronic animal safety studies to clinical trials and for monitoring potential adverse renal events.
Biomarkers that have promising potential applicability in chronic nonclinical studies which may directly parallel changes in GFR currently include SDMA, CysC, and DKK3 alone or in combination in a panel of analytes along with proven AKI biomarkers such as KIM-1 or NGAL. These individual biomarkers and/or biomarker panels may provide substantial advantages over using KIM-1 or NGAL alone in studies >3 months’ duration, increasing the sensitivity and specificity of current approaches. Other biomarkers such as UMOD or BTP may be included in a panel as additional parameters to support a weight-of-evidence approach in certain situations. This additional information on renal function may highlight potential clinical risk and provide a direct bridge to predict potential adverse declines in GFR in patients. Therefore, although there is a translational aspect to their use and they may be added to clinical trials to monitor changes observed in nonclinical studies, the fact that they can act as surrogate GFR biomarkers indicates that significant changes in SDMA, DKK3, CysC, KIM-1, and/or NGAL (with changes in urine protein) in chronic animal studies may reflect or predict possible changes in GFR in patients over time. Therefore, routine eGFR and UPCR measurements in clinical trial patients may be sufficient to provide confidence that chronic renal signals in animals are not occurring in clinical patients, even if the same biomarkers are not used in both animal and human studies. Additional toxicity studies with these exploratory biomarkers will demonstrate which of the alternative chronic renal biomarkers hold the most promise or, like acute renal biomarkers, if a panel of biomarkers will best monitor progression of chronic renal injury. The use of several of these chronic renal biomarkers can be implemented almost immediately in drug discovery or animal model studies and should provide a great benefit over current renal biomarkers for assessment of safety and efficacy of drugs intended to target CKD and inherited chronic renal diseases.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320985045 - A Review of Specific Biomarkers of Chronic Renal Injury and Their Potential Application in Nonclinical Safety Assessment Studies
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320985045 for A Review of Specific Biomarkers of Chronic Renal Injury and Their Potential Application in Nonclinical Safety Assessment Studies by Leslie A. Obert, Susan A. Elmore, Daniela Ennulat and Kendall S. Frazier in Toxicologic Pathology
Footnotes
Acknowledgments
The authors thank Rick Adler (GSK), Karen Lynch (GSK), John Kreeger (Inotiv), Greg Travlos (NTP), and Michelle Cora (NTP) for their helpful review and suggestions concerning the manuscript.
Declaration of Conflicting Interests
The authors declared no potential, real or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences (SE).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
