Abstract
The safety of 2 single domain antibodies (dAbs) was evaluated by inhalation toxicology studies in the cynomolgus monkey. In the first case study, a 14-day repeat-dose study evaluating an anti-thymic stromal lymphopoietin (anti-TSLP) dAb resulted in minimal mononuclear inflammatory cell infiltrates in the lungs, increases in lymphocytes in bronchoalveolar lavage fluid, and development of antidrug antibodies (ADAs). In a 6-week inhalation study, there was an increase in incidence and/or severity of mononuclear cell infiltrates in the lung, increased cellularity in the tracheobronchial lymph node (TBLN), and development of ADA. The second case study evaluated a change in duration of inhalation dosing, a different route of exposure (intravenous or IV), and recovery following an off-dose period with an anti-tumor necrosis factor receptor 1 dAb. A 7-day repeat-dose inhalation study and a 14-day IV study produced no microscopic effects in the lung, whereas a 14-day inhalation study resulted in moderate increases in pulmonary perivascular/peribronchiolar/alveolar lymphocytic infiltrates and increased cellularity in the TBLN, with partial and full recovery, respectively, after 14 days. The lung and lymph node findings seen after inhalation of either dAb were considered secondary to the immunogenic response to a human protein and were considered nonadverse.
Introduction
Delivering therapeutic proteins by inhalation offers the prospect of treating respiratory diseases with fewer side effects and lower doses relative to systemic delivery. Nonclinical safety evaluation requires testing in a pharmacologically relevant species. In the case studies presented here, the cynomolgus monkey was selected based on sequence homology for the target when compared with the human target and in vitro demonstration of inhibition of the target following addition of the therapeutic proteins. The published literature on the toxicology of inhaled antibodies or antibody-like biopharmaceuticals is limited, 1 and the case studies presented in this article are intended to increase the understanding of nonclinical safety findings arising from inhalation of one specific type of antibody subunit: domain antibodies (dAbs). Domain antibodies are the smallest functional binding units of human antibodies (10-13 kDa) and are derived from the variable regions of either the variable heavy (VH) or variable light (VL) chains of human immunoglobulin G (IgG). 2
Targets
Two case studies are presented, which explore the effects on the respiratory system of dAbs: one inhibiting thymic stromal lymphopoietin (anti-TSLP) given by inhalation dosing and one inhibiting tumor necrosis factor receptor 1 (anti-TNFR1) given by inhalation and intravenous (IV) dosing. The objectives of both case studies were to determine the toxicity and toxicokinetics (TK) of the inhaled test article when administered to cynomolgus monkeys in support of developing these medicines for clinical use. The first case study enabled comparison of 2 or 6 weeks of inhalation dosing of the anti-TSLP dAb on study end points. The second case study evaluated shorter term dosing (7 or 14 days) of the anti-TNFR1 dAb by inhalation, while also addressing questions on its effect on the respiratory system and other study end points when administered by IV dosing in a 14-day study.
Anti-Thymic Stromal Lymphopoietin
Thymic stromal lymphopoietin, an epithelial cell-derived cytokine, causes upregulation of costimulatory molecules on dendritic cells (DC), priming them to direct naive CD4+ T cells to differentiate into Th2 cells. 3 This cytokine is overexpressed in patients with asthma and has been negatively correlated with lung function (FEV1). 4 Data suggest increased expression in epithelial cells and in cells in the submucosa in asthmatics compared to healthy controls and expression generally increases with greater severity of disease. 5 -7 Blocking TSLP, therefore, has the potential for beneficial effects, which may include improved asthma control, improved response to or reduced use of steroid therapy, and reduction of frequency and severity of exacerbations.
Tezepelumab, which is an anti-TSLP monoclonal antibody (mAb), dosed IV, attenuated both the early and late asthmatic response in an allergen challenge study in mild, allergic asthmatics 8 and reduced clinically significant asthma exacerbations. 9 In addition, the mAb significantly reduced the number of eosinophils in the sputum and blood and decreased the fraction of exhaled nitric oxide both before and after allergen challenge. This suggests that TSLP plays a nonredundant role with respect to the other innate cytokines (interleukin-25 [IL-25] or IL-33) in driving the type 2 response. An anti-TSLP antibody fragment, formulated as a PulmoSol engineered powder in hard capsules for delivery to the lungs via dry powder inhaler, recently resulted in a significantly higher minimum FEV1 during the late response in a randomized, double-blind, placebo-controlled study of 28 patients with mild, atopic asthma. 10
GSK3191812 is an anti-TSLP dAb that was under development as a dry powder inhaled product for the treatment of moderate-to-severe asthma. This fully human Vκ dAb, consisting of 107 amino acids, selectively binds human and cynomolgus monkey TSLP and prevents TSLP signaling (unpublished data). GSK3191812 has demonstrated dose-dependent inhibition of type 2 cytokines (IL-5 and IL-13) and TARC (thymus- and activation-regulated chemokine) production from human peripheral blood mononuclear cells and nasal polyp tissue following complex innate cytokine stimulation (TSLP, IL-33, and IL-25; data not shown). In in vitro assays, cross-reactivity (binding and inhibition of TSLP) could not be demonstrated in the mouse, rat, or dog, but was successfully confirmed in the cynomolgus monkey.
Anti-Tumor Necrosis Factor Receptor 1
Tumor necrosis factor-α is a highly pleiotropic cytokine produced by macrophages/monocytes during acute inflammation and is responsible for a diverse range of signaling events within cells, leading to necrosis or apoptosis. 11 The protein is also important for resistance to infection and cancers. 12 TNF-α exerts many of its effects by binding, as a trimer, to either a 55-kDa cell membrane receptor termed TNFR1 or a 75-kDa cell membrane receptor termed TNFR2. The TNFR1 signaling mediates many of the pro-inflammatory and cell death-mediated effects of TNF; in contrast, TNFR2 signaling is primarily associated with immune modulation and tissue regeneration. Tumor necrosis factor receptor 1 is ubiquitously expressed on cells; indeed, no cell type in the body has yet been found that does not express TNFR1, while TNFR2 is restricted to T lymphocytes and other cells. 12,13
Anti-TNF mAbs (etanercept, infliximab, adalimumab, golimumab, certolizumab) have been marketed for several decades for the treatment of autoimmune diseases. Currently, TNF inhibitors are mainly used to treat rheumatoid arthritis, psoriasis, Crohn’s disease, and ankylosing spondylitis. Additional potential indications of TNF inhibitors, the majority of which are mAbs, include neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease.
Clinical studies and practice have shown that approximately one-third of patients demonstrate poor responses (primary failure) to current TNF inhibitors and more patients have shown decreased efficacy after sustained use (secondary failure). 13,14 The mechanisms of poor responses and decreased efficacy are unclear. In addition, application of current TNF-α mAb therapies shows increased risk of infection (tuberculosis), malignancies (lymphomas), central nervous system demyelinating disorders (multiple sclerosis), and additional autoimmune disease (type 1 diabetes, systemic lupus erythematosus, and psoriasis). 14 15 The possible reason for the side effects is that current TNF-α inhibitors block the interaction of TNF-α to both TNFR1 and TNFR2 receptors, which leads to the loss of regulatory function of TNFR2 signaling.
GSK1995057 is a fully human dAb that binds specifically to the extracellular domain of human TNFR1 (hTNFR1) and inhibits the binding of human TNF-α. 15 GSK1995057 showed specific and high affinity binding to cynomolgus TNFR1 and has been shown to inhibit TNF-α-induced IL-8 secretion in cynomolgus monkey cells, but does not bind TNFR1 from other species (mouse, rat, rabbit, or dog) nor does it bind to other related human TNF superfamily receptors (eg, TNFR2, NGFR, or TRAILR; data not shown). GSK1995057 binds to the same domain of TNFR1 as the natural ligand, TNF-α, and is, therefore, a competitive antagonist of TNFR1. Targeting TNFR1 alone with GSK1995057 may preserve interactions between TNF-α and TNFR2 and potentially reduce the abovementioned side effects.
Materials and Methods
Animals
These data were assembled based on samples from cynomolgus monkeys (
Housing and Environmental Conditions
Compatible pairs/triples, by sex, were housed in elevated, stainless steel cages. Pairs/triples remained together for group assignment, and animals in the same dose group were housed together to avoid cross-contamination. Animals were provided with environmental enhancement in accordance with the Animal Welfare Standards, Final Rule (9 CFR part 3) effective March 18, 1991—following the testing facility standard operating procedures (SOPs). Light, temperature, humidity, and air supply were monitored in accordance with testing facility SOPs. All primates were fed Certified Primate Diet No. 2050C (Envigo Teklad). The amount fed per day was determined by the approximate body weight average of all the animals and according to the testing facility SOP. Diets were supplemented with fruits and/or vegetables, and treats were presented as appropriate. Water was available ad libitum.
Study Designs
Anti-TSLP dAb 14-day inhalation toxicity study
GSK3191812 was given to cynomolgus monkeys (1/sex/group) once daily at gender-averaged estimated inhaled doses of 880, 2272, or 7046 µg/kg/d for 1 hour by inhalation (face mask) for 14 days (Table 1). These doses were selected to provide lung deposition multiples of 2-, 6-, and 20-fold above a projected human clinical dose of 10 mg, respectively. The exposure system consisted of a dust generator, an aerosol conditioning chamber, an aerosol expansion chamber fitted with multiple takeoff points and tubing for administration to the primate by inhalation via face mask, a collapsible aerosol reservoir, with further tubing to a particulate filter trap and extract. Aerosols were generated using a Wright Dust Feed (WDF) mechanism. The WDF mechanism was attached to the aerosol conditioning chamber for the delivery of powder into the exposure system. Achieved doses for all inhalation studies were calculated using the formula recommended by Alexander et al. 16 GSK3191812 was supplied as a spray-dried powder and was subsequently administered as a dry powder formulation suitable for inhaled administration. The control article matched the levels of the vehicle constituents of the GSK3191812 formulation. The following end points/parameters were evaluated: mask concentration, particle size distribution, clinical observations, body weights, food consumption estimates, hematology, coagulation, clinical chemistry and urinalysis results, lung weights, bronchoalveolar lavage fluid (BALf), and macroscopic and microscopic observations. Electrocardiograms were collected pretest and on days 1, 6, and 13 following the test and control article administrations. Toxicokinetic evaluation was performed on samples collected on days 1 and 14; whole-blood immunophenotyping was performed on samples collected at pretreatment and day 14; tissue immunophenotyping was performed on tissue samples collected at necropsy; and anti-GSK3191812 antibody evaluation was performed on samples collected at pretreatment and necropsy. Additional samples were collected from the lungs at necropsy: BALf, bronchial brushings, and lung lobe biopsies. Analysis and reporting of some of these samples, which were not related to the objectives of this study, were conducted outside the scope of the study and are not included in the results.
Toxicology Studies Conducted With GSK3191812 and GSK1995057.
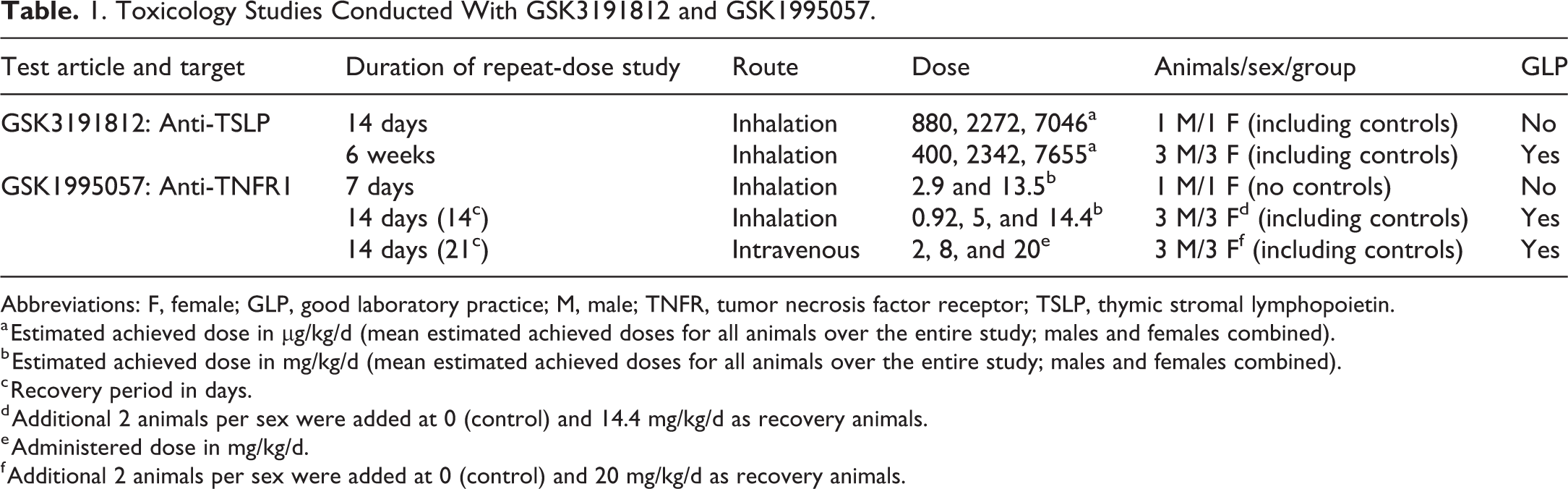
Abbreviations: F, female; GLP, good laboratory practice; M, male; TNFR, tumor necrosis factor receptor; TSLP, thymic stromal lymphopoietin.
a Estimated achieved dose in µg/kg/d (mean estimated achieved doses for all animals over the entire study; males and females combined).
b Estimated achieved dose in mg/kg/d (mean estimated achieved doses for all animals over the entire study; males and females combined).
c Recovery period in days.
d Additional 2 animals per sex were added at 0 (control) and 14.4 mg/kg/d as recovery animals.
e Administered dose in mg/kg/d.
f Additional 2 animals per sex were added at 0 (control) and 20 mg/kg/d as recovery animals.
Anti-TSLP dAb 6-week inhalation toxicity study
GSK3191812 was given to cynomolgus monkeys (3/sex/group) once daily at gender-averaged estimated inhaled doses of 400, 2342, or 7655 µg/kg/d for 1 hour by inhalation (face mask) for 6 weeks (Table 1). The formulation and vehicle were the same as for the 14-day study. The control article matched the levels of the vehicle constituents of the GSK3191812 formulation. The exposure system consisted of a dust generator, an aerosol conditioning chamber fitted with multiple takeoff points and tubing for administration to the monkey by inhalation via a face mask, a collapsible aerosol reservoir, with further tubing to a particulate filter trap and extract. Aerosols were generated using a WDF mechanism. The WDF mechanism was attached to an aerosol conditioning chamber for the delivery of powder into the exposure system. The following end points/parameters were evaluated: mask aerosol concentration, particle size distribution, clinical observations, body weights, visual food consumption, electrocardiography, hematology, coagulation, clinical chemistry and urinalysis, ophthalmoscopic observations, organ weights, and macroscopic and microscopic observations. Toxicokinetic evaluation was performed on samples collected on days 1 and 14 and week 6. Serum cardiac troponin I evaluations were conducted on samples taken twice pretreatment and during week 6. Immunophenotyping of peripheral blood (pretreatment and week 6) and of samples of the spleen, mesenteric, and tracheobronchial lymph nodes (TBLNs; collected at necropsy) were also undertaken. Anti-drug antibodies were assessed pretreatment, day 15, and week 6. Bronchoalveolar lavage fluid collected at necropsy was also analyzed for GSK3191812 concentration. Immunohistochemistry (IHC) analysis of inflammatory cell infiltrates on sample of lung and TBLN from selected animals was also conducted.
Anti-TNFR1 7-day inhalation toxicity study
GSK1995057 was administered to cynomolgus monkeys (1/sex/group) once daily as a liquid droplet aerosol at estimated inhaled doses of 2.9 or 13.5 mg/kg/d for 1 hour by inhalation (face mask) for 7 days (Table 1). The exposure system consisted of 2 Pari LC Plus nebulizers, both fitted to a single subchamber that included multiple takeoff points and tubing for administration to the monkey by inhalation via face mask. Also included in the exposure system was a collapsible aerosol reservoir, with further tubing to a particulate filter trap and extract. Formulations were fed to the nebulizers using syringe pumps. Compressed air was provided to the nebulizers to generate the aerosol for delivery to the face masks. The following end points/parameters were evaluated: mask aerosol concentration, particle size distribution, in-life animal observations, body weight, food consumption, hematology, coagulation and clinical chemistry, and macroscopic and microscopic observations. Anti-GSK1995057 antibody analysis was performed on samples collected on day 1 (prior to dosing) and day 7 (postdosing). Toxicokinetic evaluation was performed on samples collected on days 1 and 7. Additionally, separate sections of the kidney from the female given 2.9 mg/kg/d were processed for IHC evaluations and for transmission electron microscopy (results not included here).
Anti-TNFR1 14-day inhalation toxicity study
GSK1995057 was administered to cynomolgus monkeys (n = 3 or 5/sex/group) once daily as a liquid droplet aerosol at estimated inhaled doses of 0 (vehicle), 0.92, 5.0, or 14.4 mg/kg/d for 1 hour by inhalation (face mask) for 14 days, followed by a 14-day recovery period (Table 1). The dose selection was based on the exposure measured in the 7-day inhalation study, in which the high dose (13.5 mg/kg/d) provided approximately 13-fold cover over the highest anticipated clinical dose to humans on an mg/kg basis. The target low dose of 1 mg/kg/d was expected to be at parity with the highest anticipated clinical dose to humans on an mg/kg/d basis. The exposure system consisted of Hudson RCI Up-Draft II Opti-Neb nebulizers, syringe pumps, a central acrylic chamber to provide initial aerosol conditioning, 2 acrylic subchambers each fitted with multiple aerosol takeoff points, clinical hose providing aerosol delivery to the monkey facemasks, a collapsible air reservoir (small meteorological balloon), and further aerosol tubing to a droplet trapping system and extract. Test article aerosols were generated using 2 Hudson RCI Up-Draft II Opti-Neb nebulizers. A third nebulizer was located to deliver a low flow of diluent air to the output of the 2 test article nebulizers and was used to provide additional humidity to the system. The 2 test article containing nebulizers were attached directly to the central acrylic chamber. The following parameters were evaluated: mask aerosol concentration, particle size distribution, and clinical observations; body weights; food consumption measurements; hematology, coagulation, clinical chemistry, and urinalysis results; ophthalmoscopic observations, electrocardiography, neurobehavioral assessment, and organ weights; and macroscopic and microscopic observations. Assessment of spermatogenesis could not be made owing to the sexual immaturity of the majority of males. Toxicokinetic evaluation was performed on samples collected on days 1 and 7 (4 and 23 hours after completion of dosing only) and day 14; anti-GSK1995057 antibody analysis was performed on samples collected on days 1 and 14 and at the end of the recovery period (day 29), flow cytometry analysis was performed on samples collected pretest and on days 1 and 14 and at the end of the recovery period (day 29), and cytokine level analysis was performed on samples collected twice pretest, day 14, and at the end of the recovery period (day 29).
Anti-TNFR1 14-day study IV toxicity study
To support infusion dosing in the clinic, the toxicity potential of GSK1995057 was evaluated in cynomolgus monkeys following IV (infusion) administration for 14 days, followed by a 21-day recovery period (Table 1). GSK1995057 was administered once daily in a suitable IV formulation (3-hour infusion) to cynomolgus monkeys (n = 3 or 5/sex/group) at doses of 0 (vehicle), 2, 8, or 20 mg/kg/d. Based on the findings from the earlier inhalation study, a target dose of 20 mg/kg/d was selected as the high dose for the current study, which provided approximately 10-fold cover over the highest anticipated clinical dose to humans on an mg/kg basis. A target low dose of 2 mg/kg/d was expected to be at parity with the highest anticipated clinical dose to humans on an mg/kg/d basis. The following parameters were evaluated: clinical observations; body weights; food consumption; hematology, coagulation, clinical chemistry, and urinalysis results; ophthalmoscopic observations, electrocardiography, and organ weights; and macroscopic and microscopic observations. Toxicokinetic evaluation was performed on samples collected on days 1, 4, 7, 10, 12, and 14 and at the end of the recovery period (day 36); anti-GSK1995057 antibody analysis was performed on samples collected prior to dosing on days 1, 7, and 14 and at the end of the recovery period (day 36), flow cytometry analysis was performed on samples collected pretest and on days 1 and 14 and at the end of the recovery period (day 36), and cytokine level analysis (IL-1β, TNF-α, IL-6, and IL-8) was performed on samples collected twice pretest and on days 1 and 14 and at the end of the recovery period (day 36).
Toxicokinetic Analysis
Anti-TSLP dAb
Monkey plasma and BAL samples from the 2- and 6-week studies were analyzed for GSK3191812 using an electrochemiluminescent immunoassay (ECLIA). GSK3191812 in diluted plasma and BAL samples was captured with immobilized human TSLP and detected using a ruthenium-labeled α-Vκ detection mAb. The validated range for GSK3191812 in both monkey plasma and BAL was 100 pg/mL to 100 ng/mL.
GSK3191812 concentrations in epithelial lining fluid (ELF) were estimated by measuring urea concentrations in BAL and plasma following necropsy. Monkey plasma and BAL samples were analyzed for urea using a colorimetric assay. The validated range for urea in monkey plasma was 5 to 100 mg/dL, and in monkey BAL 0.5 to 10 mg/dL. The volume of ELF contained in the BAL samples was estimated as previously described. 17
Anti-TNFR1 dAb
Monkey plasma samples were diluted in assay buffer and analyzed for GSK1995057 using a chemiluminescent immunoassay. GSK1995057 was captured with hTNFR1 and detected using a biotinylated mouse idiotypic antibody against GSK1995057 followed by an HRP-labeled goat antibiotin antibody conjugate. The validated range for GSK1995057 was 0.35 to 3.5 ng/mL.
Anti-drug antibody analysis for anti-TSLP dAb
Serum samples were analyzed for antibodies against GSK3191812 using a validated qualitative bridging ECLIA. Samples defined as potentially positive after screening were titrated and confirmed by drug competition testing. Ruthenium functioned as the label necessary for electrochemiluminescence detection using the Meso Scale Discovery (MSD) Sector Imager 6000 (MSD Discovery Workbench software, version 4.0). Two concentrations of an affinity-purified rabbit anti-Vk dAb, prepared in 10% monkey serum, as well as a negative control (10% monkey serum without anti-GSK3191812), were used as the quality controls. Confirmation testing was conducted on potentially positive samples by adding excess unlabeled GSK3191812 (100 µg/mL) to determine competitive inhibition of the signal in the anti-GSK3191812 bridging ECLIA.
Anti-drug antibody analysis for anti-TNFR1 dAb
Serum samples from all GSK1995057-treated monkeys were analyzed for anti-GSK1995057 antibodies by ECLIA. Ruthenium functioned as the label necessary for electrochemiluminescence detection using the MSD Sector Imager 2400 (MSD Discovery Workbench software, version 3.0). An affinity-purified rabbit anti-GSK1995057 antibody, prepared in 10% normal cynomolgus monkey serum in assay buffer, was used as the reference standard. For specificity testing, samples were mixed with unlabeled GSK1995057 or a VH dummy dAb prior to inclusion in the assay. The VH dummy dAb shares similar framework sequences and overall ∼83% homology with GSK1995057 but does not bind TNFR1.
Clinical Pathology
Following an overnight fast, blood samples were obtained from all animals from the femoral vein or other suitable site for routine hematology, clinical chemistry, and coagulation testing. Samples were collected twice pretreatment and close to or at termination of the study. Whole blood was collected in ethylenediaminetetraacetic acid (EDTA)-containing tubes (hematology), sodium citrate (coagulation), or tubes without anticoagulant (clinical chemistry), and analyses performed using automated analyzers and appropriate reagents and controls according to manufacturer’s instructions.
Immunophenotyping of Peripheral Blood
In the anti-TSLP studies, blood samples were collected into EDTA anticoagulant from fasted animals pretreatment (week-2) and on day 14 or during week 6 (24 hours postdose, fasted) from the femoral vein or other suitable site. Samples were stained using commercially available antibodies and acquired on the BD FACSCanto II flow cytometer with BD FACSDiva software version 6.1.3. The following data were collected in the 14-day dose range finding (DRF): total T cells (CD3+CD20−), B cells (CD3−CD20+), natural killer (NK) cells (CD3−CD20−CD16+), T helper cells (CD3+CD4+CD8−), cytotoxic T cells (CD3+CD4−CD8+), and CD4+ regulatory T cells (CD3+CD4+CD8−CD25+FoxP3+). Data collected and evaluated in the 6-week study included those in the 14-day study, with the exception of B and NK cells. Appropriate FMO (Fluorescence Minus One) controls and single stained tubes for each fluorochrome were stained and acquired using one control animal or an animal from the testing facility’s colony at every time point the regulatory T-cell panel was run. The FMO controls were used for setting gates. Absolute lymphocyte counts were derived from the hematology analyzer (ADVIA 120). Absolute numbers (cells/µL) for each respective lymphocyte subset population were derived from the cell subset percentage of lymphocytes derived from the flow cytometer (%) multiplied by the lymphocyte concentration derived from the ADVIA hematology analyzer.
In the 14-day anti-TNFR1 studies, the following immunophenotyping on whole blood was conducted at pretest, day 1 (predose), day 14 (4 and 23 hours postdose), and at the end of the recovery period: T cells (CD3+), B cells (CD20+), NK cells (CD3−CD16+), helper T cells (CD3+CD4+CD8−), cytotoxic T cells (CD3+CD4−CD8+), and monocytes (CD14+). In the 14-day inhaled and IV studies, Beckman Coulter EPICS XL-MCL flow cytometer with manufacturer’s System II software were used.
Study Termination and Pathological Examination
After administration of ketamine/xylazine-induced sedation and sodium pentobarbital, animals were exsanguinated and humanely euthanized. Comprehensive necropsy was performed on all animals. Tissues were fixed by immersion in 10% neutral-buffered formalin. Eyes, optic nerve, and testes were placed in modified Davidson solution initially, followed by 10% neutral-buffered formalin. Tissues were embedded in paraffin, and the sections were stained with hematoxylin and eosin for microscopic examination. Microscopic data from all studies were recorded by direct computer entry by the study pathologist. All studies underwent pathology peer review, and all data generated on the GLP-compliant studies were reviewed by the quality assurance department prior to the release of the final pathology report.
Tissue Immunophenotyping
In the anti-TSLP studies, samples (∼1 cm3 each) were collected from the spleen, mesenteric, and TBLNs at necropsy. Tissues samples were dissociated into single-cell suspensions and further processed. The following data were collected using a validated method: percentages of lymphocytes, total T cells (CD3+CD20−), helper T cells (CD3+CD4+CD8−), cytotoxic T cells (CD3+CD4−CD8+), B cells (CD3−CD20+), NK cells (CD3−CD20−CD159a+), and, in the 14-day study only, CD4+ regulatory T cells (CD3+CD4+CD8−CD25+FoxP3+).
Bronchoalveolar Lavage
In the 6-week anti-TSLP study, at necropsy, after the lung was weighed, the accessory lung lobe for all animals was isolated, removed, and used for BAL with an isotonic saline solution. The BALf was centrifuged (400 g for ∼8 minutes at 2 °C-8 °C). The cell-free supernatant was transferred to sterile polystyrene tubes and stored at −80 °C (±10 °C) and used to measure the concentration of test article.
Immunohistochemistry
Formalin-fixed, paraffin-embedded sections of lung and TBLN from 2 high-dose animals given 7655 µg/kg/d and 1 control animal from the anti-TSLP 6-week inhaled toxicity study were prepared using a microtome at a nominal thickness of 4 to 5 µm and stained for CD3 (Dako, catalogue no. A0452), CD20 (Abcam, catalogue no. ab78237), and CD25 (Abcam, catalogue no. ab128955). Positive control tissue (tonsil) was included in every run for each test antibody, which yielded specific membranous staining at the optimized test antibody concentration.
Positive staining was recorded semiquantitatively for both intensity (graded negative, weak positive, moderate positive, or strong positive) and frequency (graded minimal, slight, moderate, marked, or very marked). Cell type, pattern of staining, and distribution of positively staining cells within the tissue were also recorded. All tissue sections were reviewed by 2 pathologists and a consensus score reached.
Results
Animal Fate, Clinical Observations, Body Weight and Food Consumption Ophthalmology, Electrocardiography
In all studies, all animals survived until their scheduled necropsy, and there were no test article–related clinical observations, nor effects on body weight or food consumption. In the 7-day inhaled toxicity study with the anti-TNFR1 dAb, there were isolated incidences of decreased activity and reduced food consumption noted in females given GSK1995057; however, in the absence of concurrent controls and of a clear dose-related pattern, the significance of these findings is uncertain. In the GLP-compliant studies (Table 1), which included ophthalmology and electrocardiography, there were no test article–related effects on these end points.
Anti-TSLP dAb
14-day inhaled toxicity study
Toxicokinetics
GSK3191812 plasma concentrations were quantifiable for the entire 24-hour sampling period after dosing at all dose levels on days 1 and 14 (Table 2). The maximum plasma concentrations (
Mean Systemic Exposure Following Dry Powder Inhaled Administration of GSK3191812.
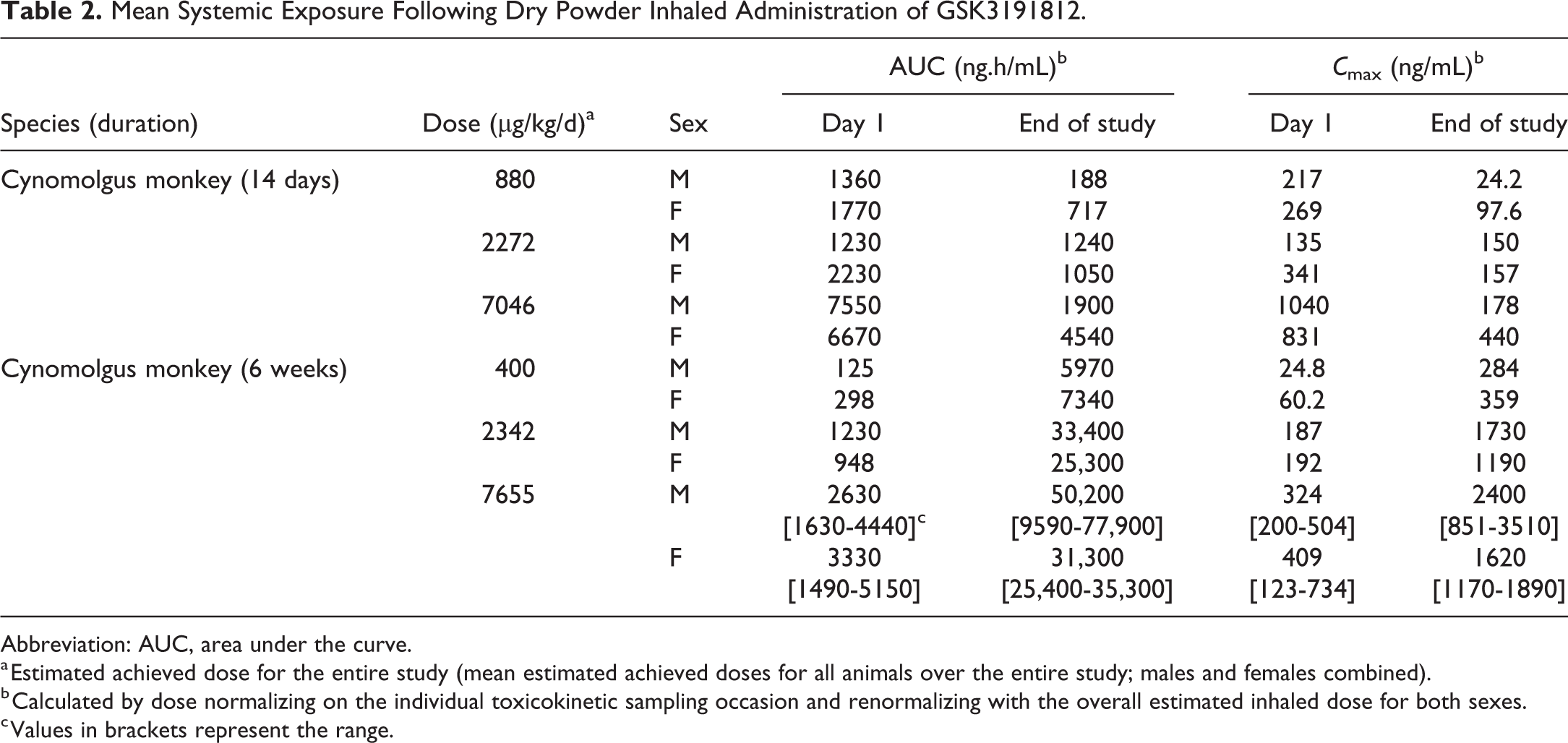
Abbreviation: AUC, area under the curve.
a Estimated achieved dose for the entire study (mean estimated achieved doses for all animals over the entire study; males and females combined).
b Calculated by dose normalizing on the individual toxicokinetic sampling occasion and renormalizing with the overall estimated inhaled dose for both sexes.
c Values in brackets represent the range.
Immunophenotyping
There were no apparent test article–related changes in total T cells, T helper cells, cytotoxic T cells, B cells, NK cells, and monocytes in peripheral blood, spleen, and lymph nodes of male and female cynomolgus monkeys administered GSK3191812. Additionally, there were no apparent test article–related changes on circulating CD4+ regulatory T-cell numbers (both percentages and absolute counts) and no dose-dependent test article–related changes in lymphocytes (CD45+) and CD4+ regulatory T cells in the spleen or lymph nodes, respectively.
Clinical and anatomic pathology
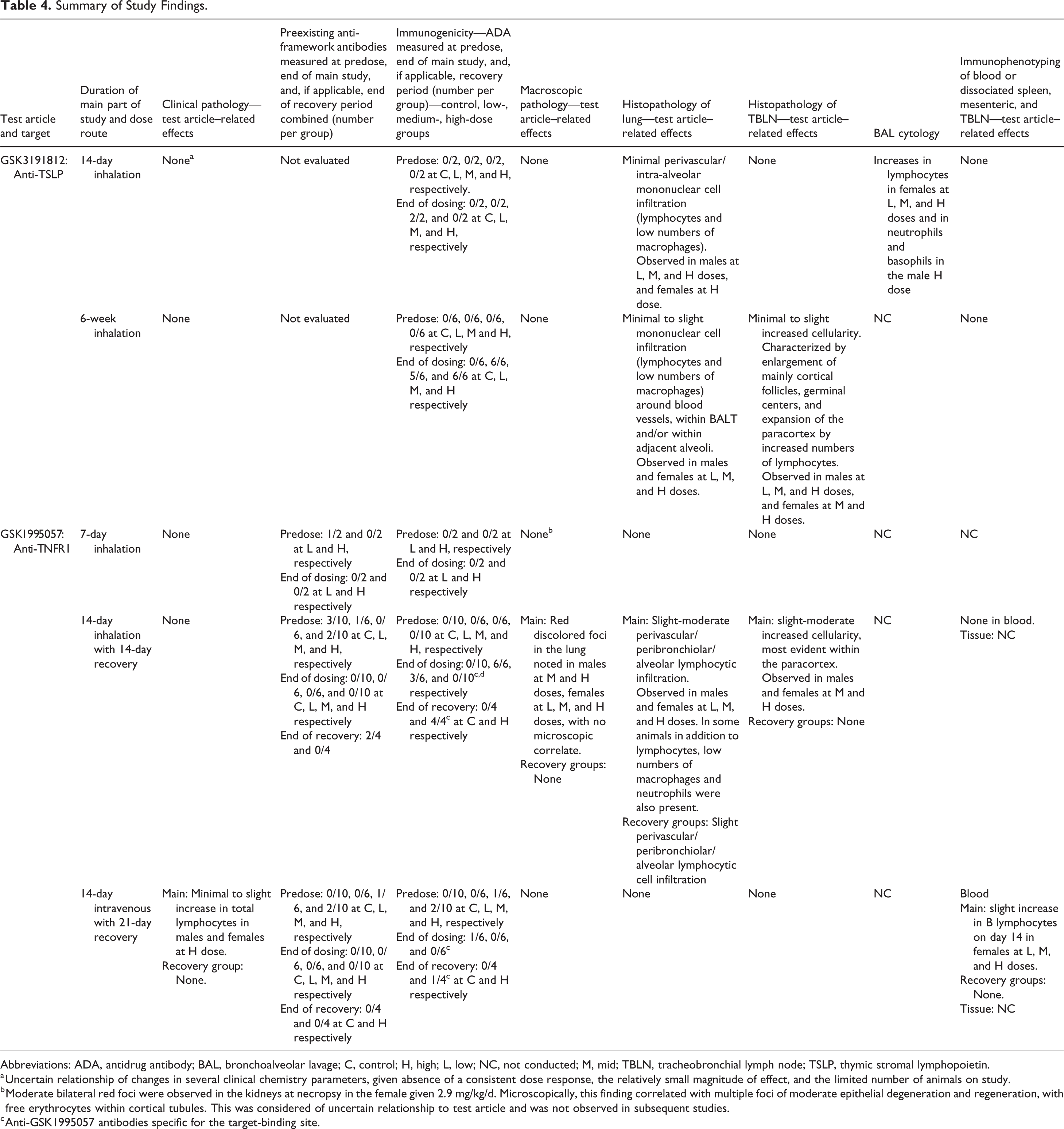
Minimal mononuclear inflammatory cell infiltrates were present in the lungs of males at ≥880 µg/kg/d and the female at 7046 µg/kg/d. Infiltrates were oriented around blood vessels and within the adjacent alveoli and were composed of lymphocytes and low numbers of macrophages (Figure 1). The finding did not occur in the controls and was considered to be possibly associated with GSK3191812. Changes in several clinical chemistry parameters were possibly related to treatment with GSK3191812, but the relationship was uncertain, given the absence of a consistent dose response, the relatively small magnitude of effect, and the limited number of animals on study. In BALf, there were increases in lymphocytes in females given ≥880 µg/kg/d and in neutrophils and basophils in the male given 7046 µg/kg/d (Table 4).
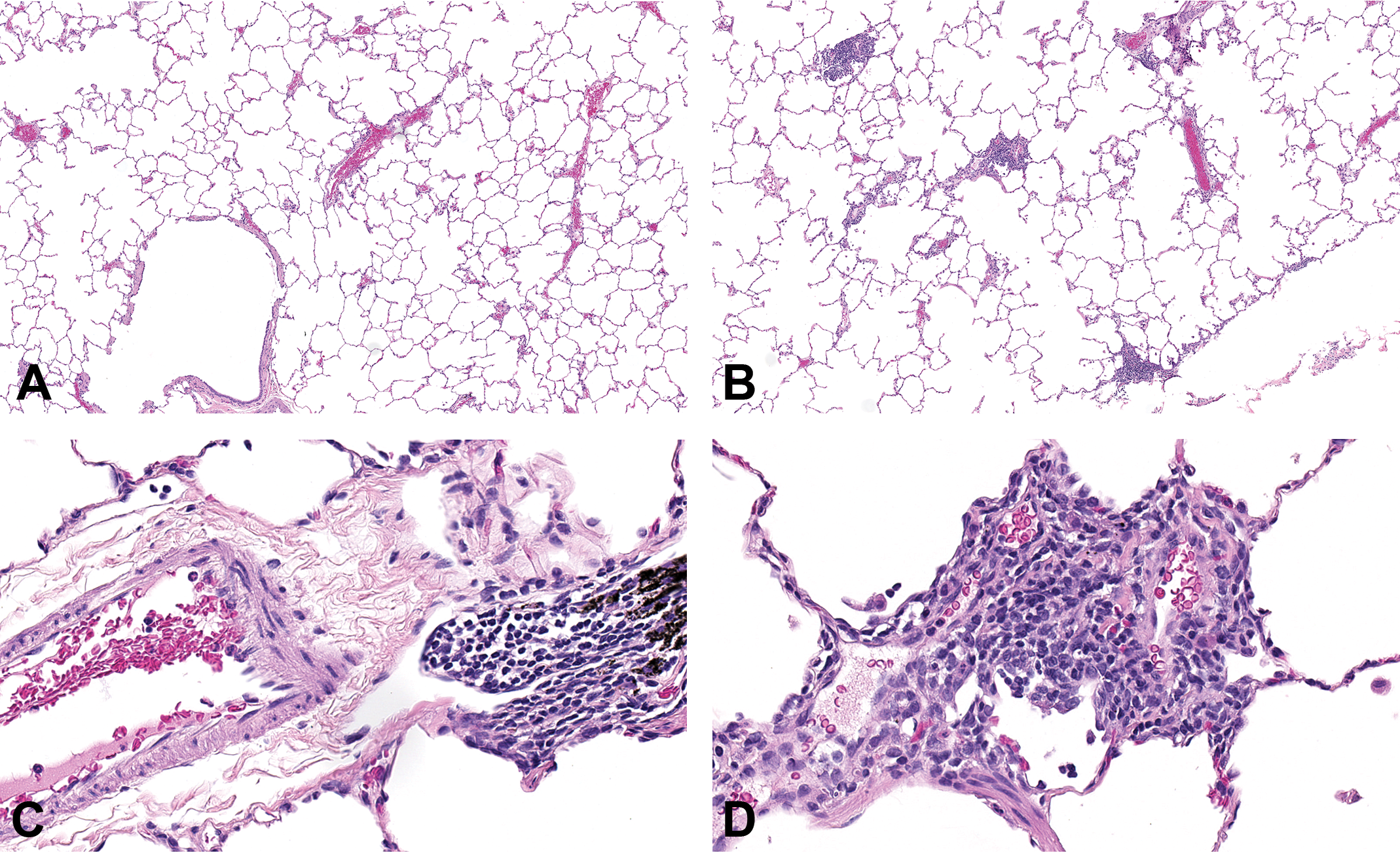
Lung sections from cynomolgus monkeys in 14-day inhaled anti-thymic stromal lymphopoietin dose range finding study. A, Control male (1410) terminal on day 15. Normal appearance of small airway and adjacent alveoli. Original Objective ×5. B, High-dose male (4410) given 7046 µg/kg/d terminal on day 15. Multifocal perivascular/Intra-alveolar distribution of the minimal, mononuclear cell inflammatory cell infiltrate ×5. C, Control male (1410). Blood vessel with adjacent normal BALT containing pigmented macrophages. Original Objective ×40. D, High-dose male (4410) given 7046 µg/kg/d terminal on day 15. The perivascular/intra-alveolar mononuclear cell infiltrates were oriented around blood vessels and within the adjacent alveoli and were composed of lymphocytes and low numbers of macrophages. Original Objective ×40.
Comparison of the Mean Systemic Exposure of GSK1995057 Following a 14-Day Intravenous (Infusion) Study and a 14-Day Inhalation Study in the Cynomolgus Monkey.
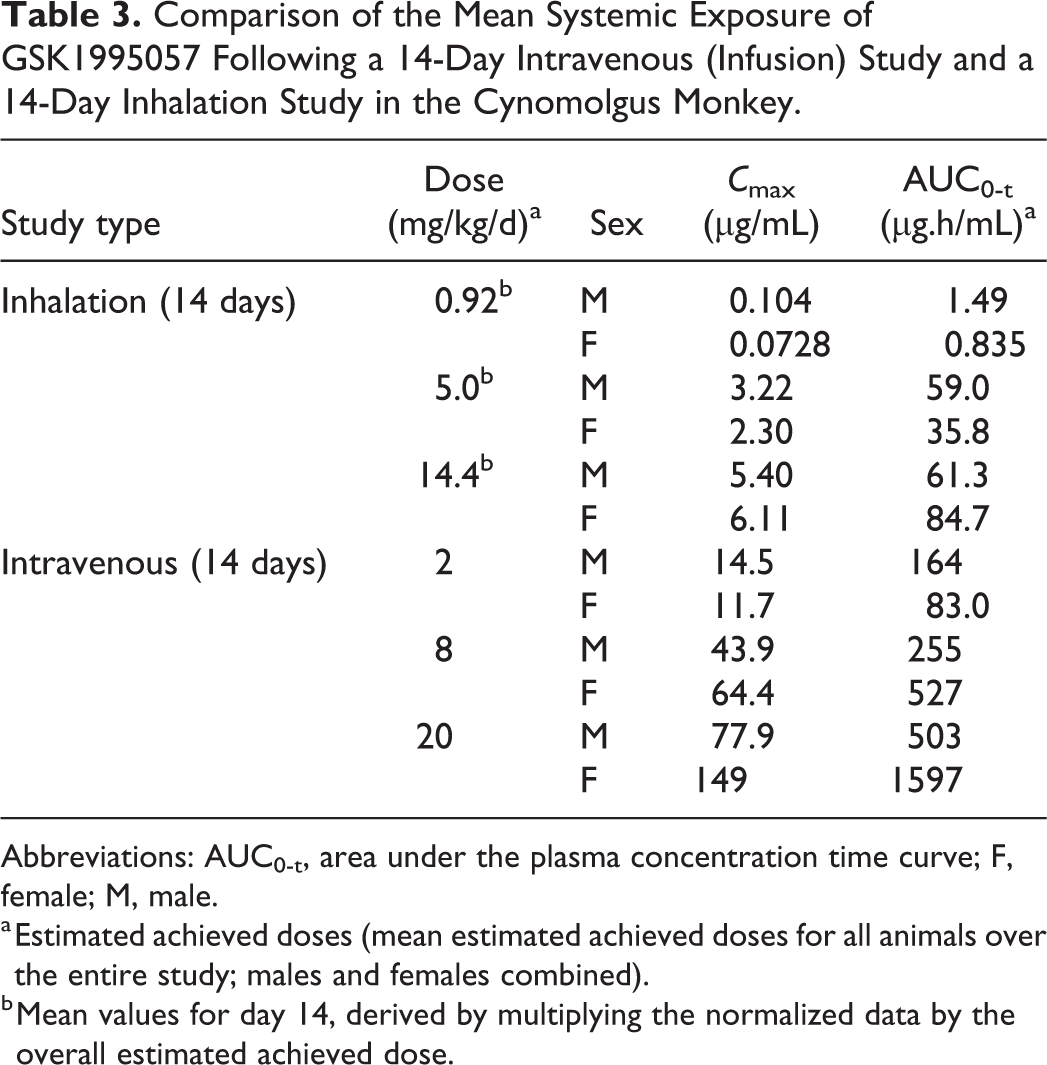
Abbreviations: AUC0-t, area under the plasma concentration time curve; F, female; M, male.
a Estimated achieved doses (mean estimated achieved doses for all animals over the entire study; males and females combined).
b Mean values for day 14, derived by multiplying the normalized data by the overall estimated achieved dose.
Summary of Study Findings.
Abbreviations: ADA, antidrug antibody; BAL, bronchoalveolar lavage; C, control; H, high; L, low; NC, not conducted; M, mid; TBLN, tracheobronchial lymph node; TSLP, thymic stromal lymphopoietin.
a Uncertain relationship of changes in several clinical chemistry parameters, given absence of a consistent dose response, the relatively small magnitude of effect, and the limited number of animals on study.
b Moderate bilateral red foci were observed in the kidneys at necropsy in the female given 2.9 mg/kg/d. Microscopically, this finding correlated with multiple foci of moderate epithelial degeneration and regeneration, with free erythrocytes within cortical tubules. This was considered of uncertain relationship to test article and was not observed in subsequent studies.
c Anti-GSK1995057 antibodies specific for the target-binding site.
Anti-drug antibodies
Immunogenicity analysis detected anti-GSK3191812 antibodies on the day of necropsy in both the male and female animals at 2272 µg/kg/d, with titers of 320 and 20, respectively (Table 4).
6-Week inhaled toxicity study
Toxicokinetics
In the 6-week inhaled repeat-dose toxicology study, there was generally no marked (>2-fold) difference in systemic exposure between male and female animals on day 1, except at 400 µg/kg/d (Table 2) where the mean AUC0-t and
GSK3191812 was quantifiable in all terminal BAL samples from groups given the test article. The standardized individual apparent concentrations of GSK3191812 in ELF overlapped across the dose groups ranging from 1200 to 13700, 135 to 10200, and 1590 to 15300 ng/mL for the animals dosed at overall estimated inhaled doses of 400, 2342, and 7655 µg/kg/d, respectively.
Immunophenotyping
There were no test article–related changes in percentage or absolute number of T cells, T cell subsets, or CD4+ regulatory T cells in whole blood. There were no test article–related changes in percentage or absolute number of T cells, T-cell subsets, B cells, or NK cells in spleen, mesenteric, and TBLNs.
Clinical and anatomic pathology
Microscopically, GSK3191812-related findings at ≥400 µg/kg/d included minimal-to-slight mononuclear cell infiltrates in the lung and minimal-to-slight increased general cellularity in the TBLN (Table 4). The mononuclear cell infiltration in the lung was characterized by lymphocytes and low numbers of macrophages oriented around blood vessels, within bronchus-associated lymphoid tissue (increased BALT cellularity), and/or within the adjacent alveoli (Figure 2). The increase in general cellularity in the TBLN was characterized by enlargement of mainly cortical follicles, germinal centers, and expansion of the paracortex by increased numbers of lymphocytes (Figure 2).
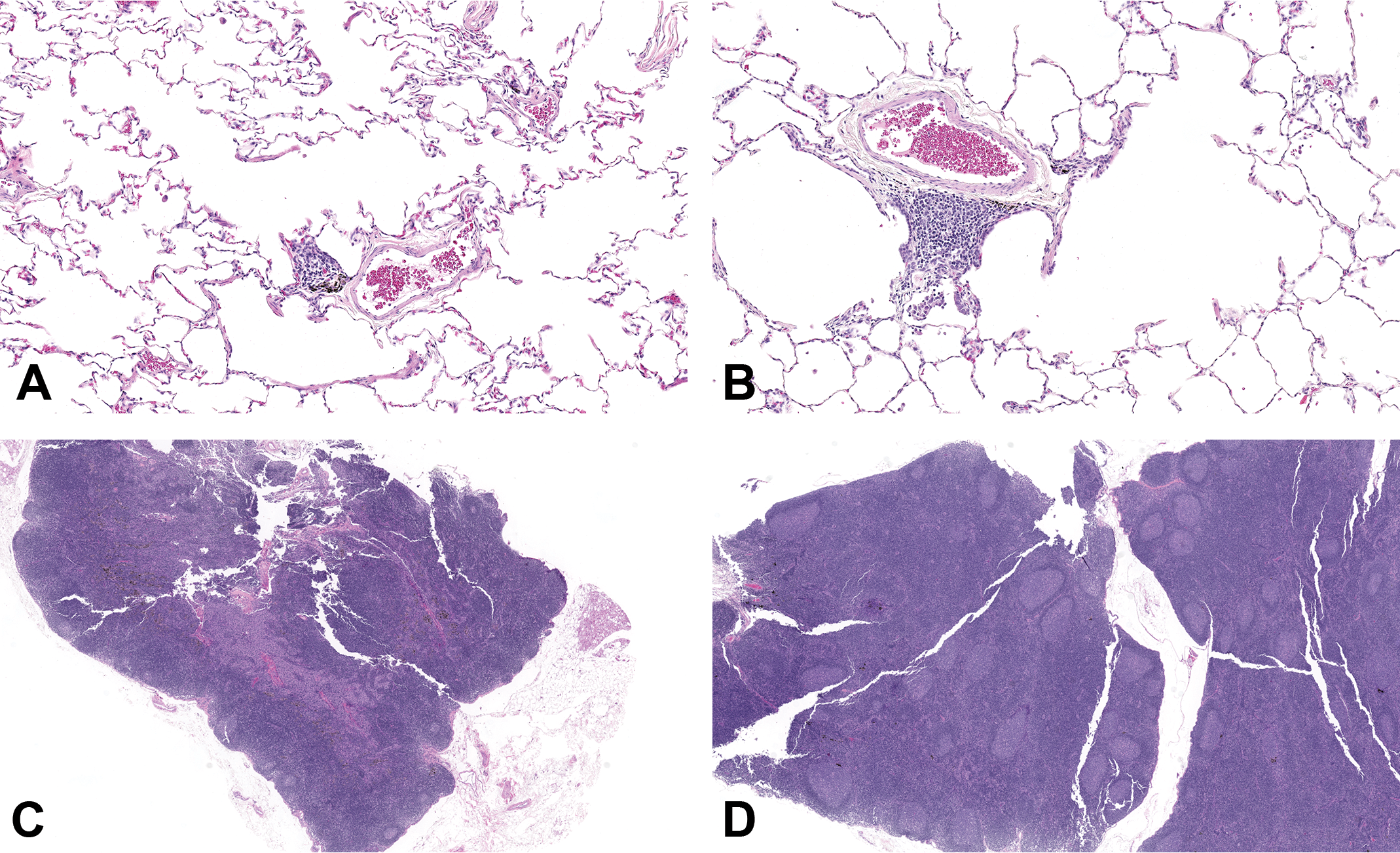
Lung and tracheobronchial lymph node (TBLN) sections from cynomolgus monkeys in a 6-week inhaled anti-thymic stromal lymphopoietin study. A, Control female (1945) terminal on day 43. Lung. Normal appearance of terminal airways, adjacent alveoli, blood vessel, and normal bronchiolar-associated lymphoid tissue with pigmented macrophages. Original Objective ×10. B, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. Lung. Minimal mononuclear inflammatory cell infiltrate, consisting of lymphocytes and low numbers of macrophages oriented around blood vessels, within bronchus-associated lymphoid tissue and/ adjacent alveoli. Original Objective ×10. C, Control female (1945) terminal on day 43. TBLN. Normal appearance with few germinal centers visible within cortical follicles in the lower part of the image. Original Objective ×2. D, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. TBLN. Slight increased cellularity with enlargement of mainly cortical follicles, germinal centers, and expansion of the paracortex by increased numbers of lymphocytes. Original Objective ×2.
Immunohistochemistry of lung and TBLN infiltrates
A small increase in the frequency of CD3- and CD20-positive cells was seen in the alveolar walls of the lungs in treated animals compared to the control animal (Figure 3; Table 5). CD25-positive cells were not detected within the alveolar walls in treated or control animals. The perivascular/peribronchiolar mononuclear cell infiltrates seen in treated animals only were composed of marked numbers of CD20-positive cells and slight-to-moderate numbers of CD3- and CD25-positive cells (Figure 3).
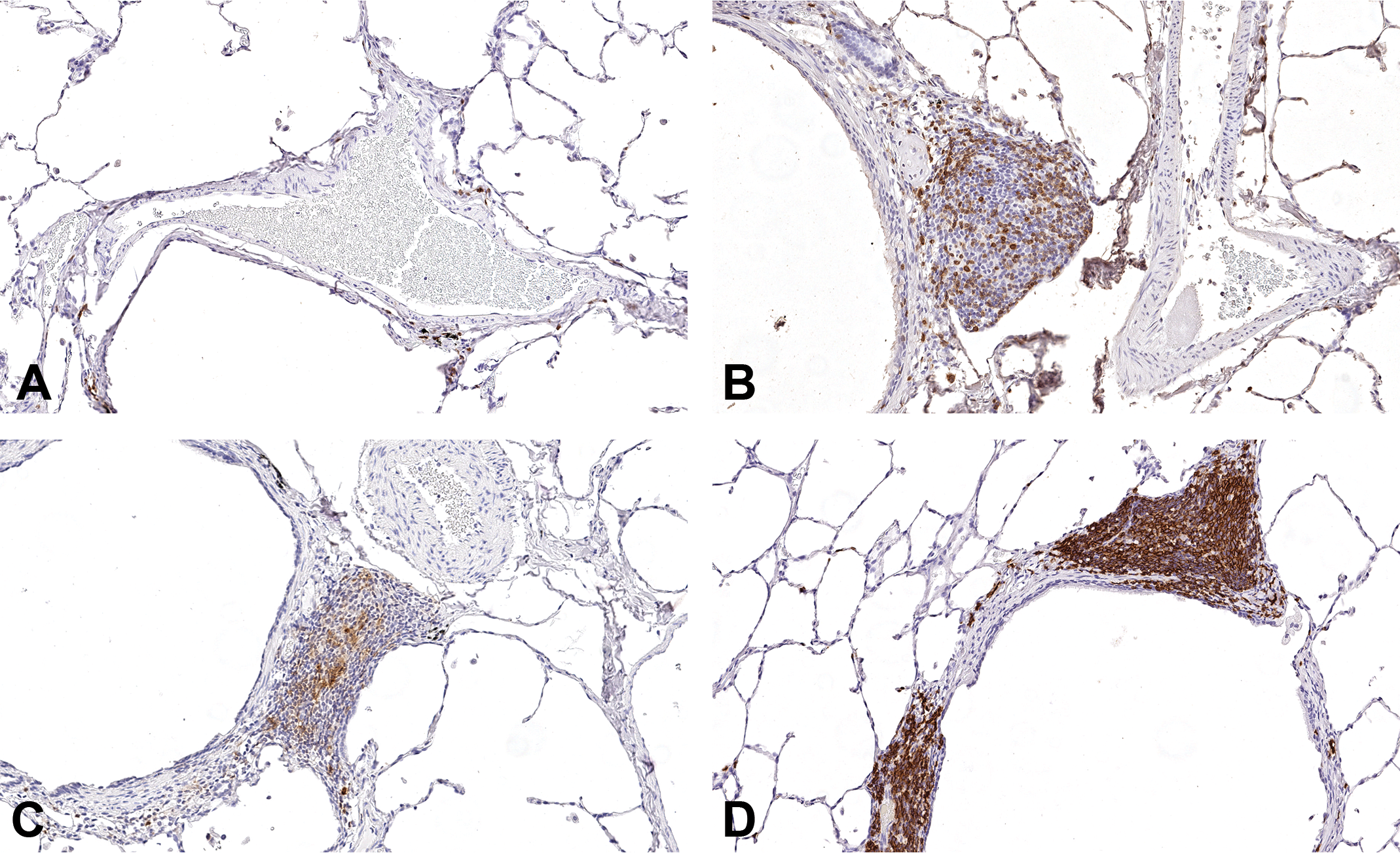
Immunohistochemistry of lung sections from cynomolgus monkeys in a 6-week inhaled anti-thymic stromal lymphopoietin study. A, Control female (1945) terminal on day 43. CD3. Minimal numbers of positively stained round cells scattered with in the alveolar walls adjacent to the blood vessel in the center of the image. Original Objective ×20. B, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. CD3. Slight numbers of positively stained round cells scattered within the alveolar walls and moderate numbers of positively stained round cells within the peribronchiolar mononuclear cell infiltrate. Original Objective ×20. C, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. CD25. Slight numbers of positively stained large irregular cells and fewer small round cells with indistinct cell boundaries scattered within mononuclear cell infiltrates situated perivascularly/peribronchiolarly. Original Objective ×20. D, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. CD20. Slight numbers of positively stained solitary round cells scattered throughout the alveolar walls. Marked numbers of positively stained round cells within mononuclear cell infiltrates situated perivascularly/peribronchiolarly (BALT). Original Objective ×20.
Immunohistochemistry of the Lung in the Anti-TSLP 6-Week Inhaled Toxicity Study.a,b
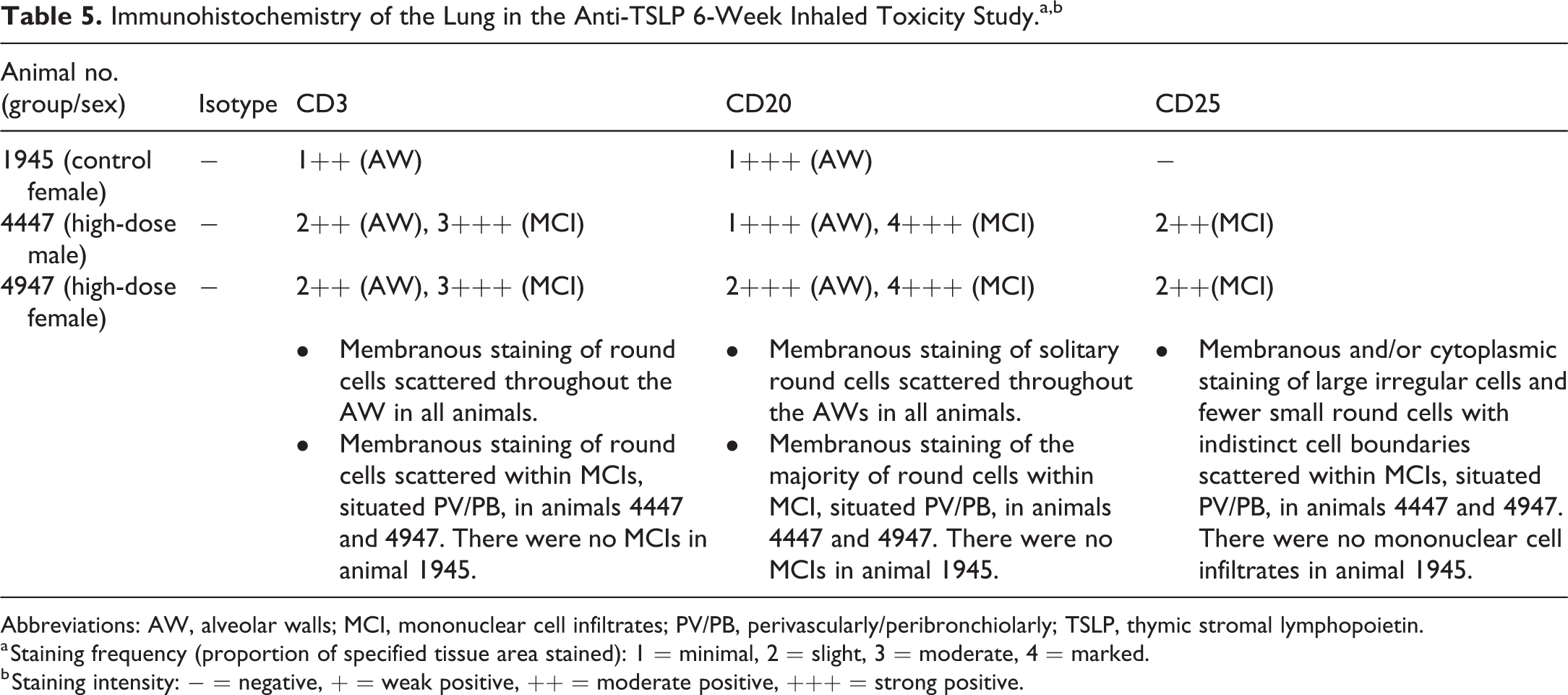
Abbreviations: AW, alveolar walls; MCI, mononuclear cell infiltrates; PV/PB, perivascularly/peribronchiolarly; TSLP, thymic stromal lymphopoietin.
a Staining frequency (proportion of specified tissue area stained): 1 = minimal, 2 = slight, 3 = moderate, 4 = marked.
b Staining intensity: − = negative, + = weak positive, ++ = moderate positive, +++ = strong positive.
The staining frequency and intensity of CD3- and CD20-positive cells in the TBLNs were very similar between the treated animals and the control animal, although slight staining for CD3 was seen in the germinal centers of treated animals, with no such staining seen in the control animal (Table 6). Increased frequency and intensity of staining for CD25 was seen in the medullary cords and sinuses of treated animals compared to the control animal. CD25 staining was also seen in the germinal centers, follicles, and paracortex of treated animals, whereas it was absent in these locations in the control animal (Figure 4).
Immunohistochemistry of the TBLN in the Anti-TSLP 6-Week Inhaled Toxicity Study.a,b
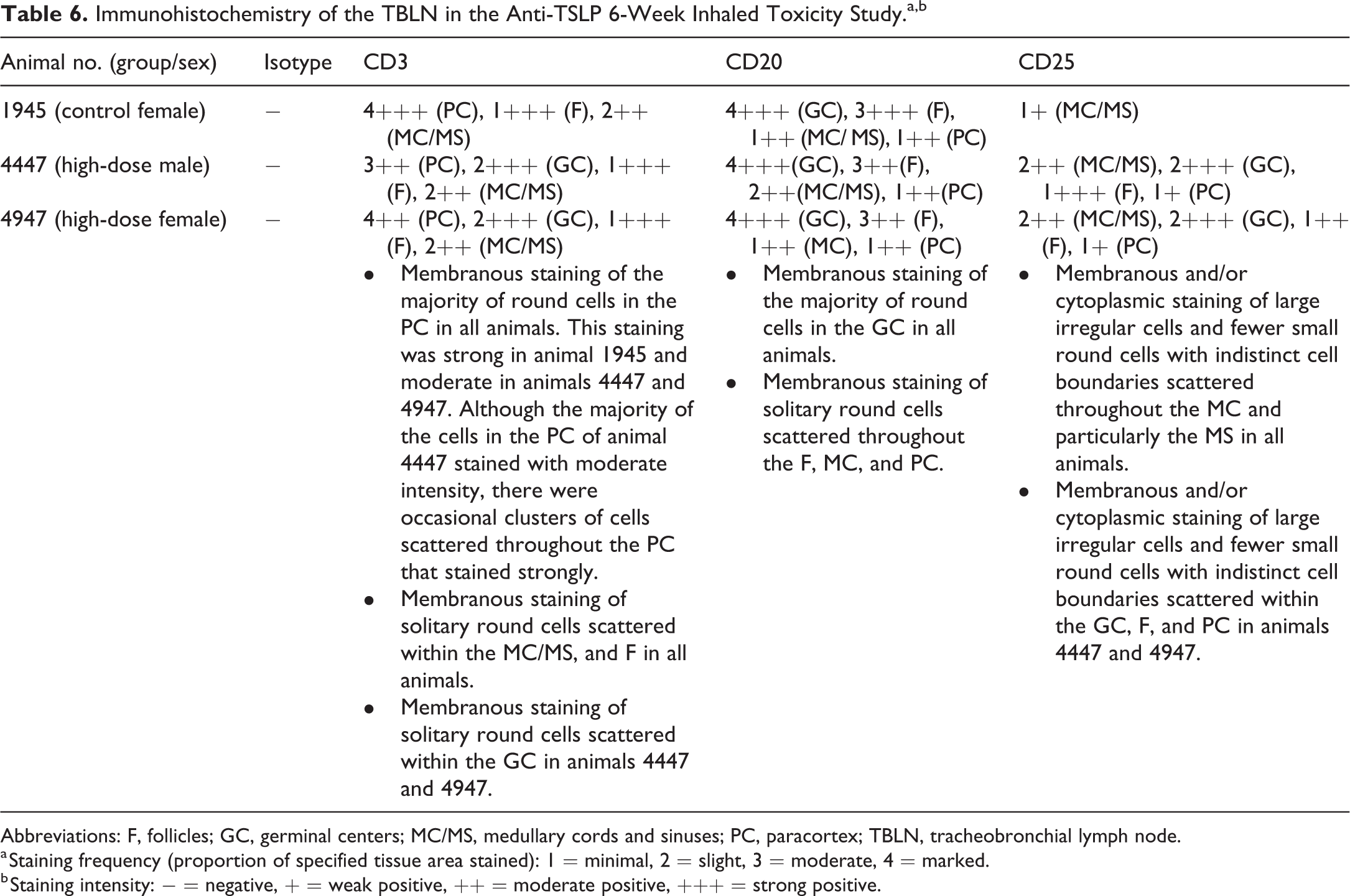
Abbreviations: F, follicles; GC, germinal centers; MC/MS, medullary cords and sinuses; PC, paracortex; TBLN, tracheobronchial lymph node.
a Staining frequency (proportion of specified tissue area stained): 1 = minimal, 2 = slight, 3 = moderate, 4 = marked.
b Staining intensity: − = negative, + = weak positive, ++ = moderate positive, +++ = strong positive.
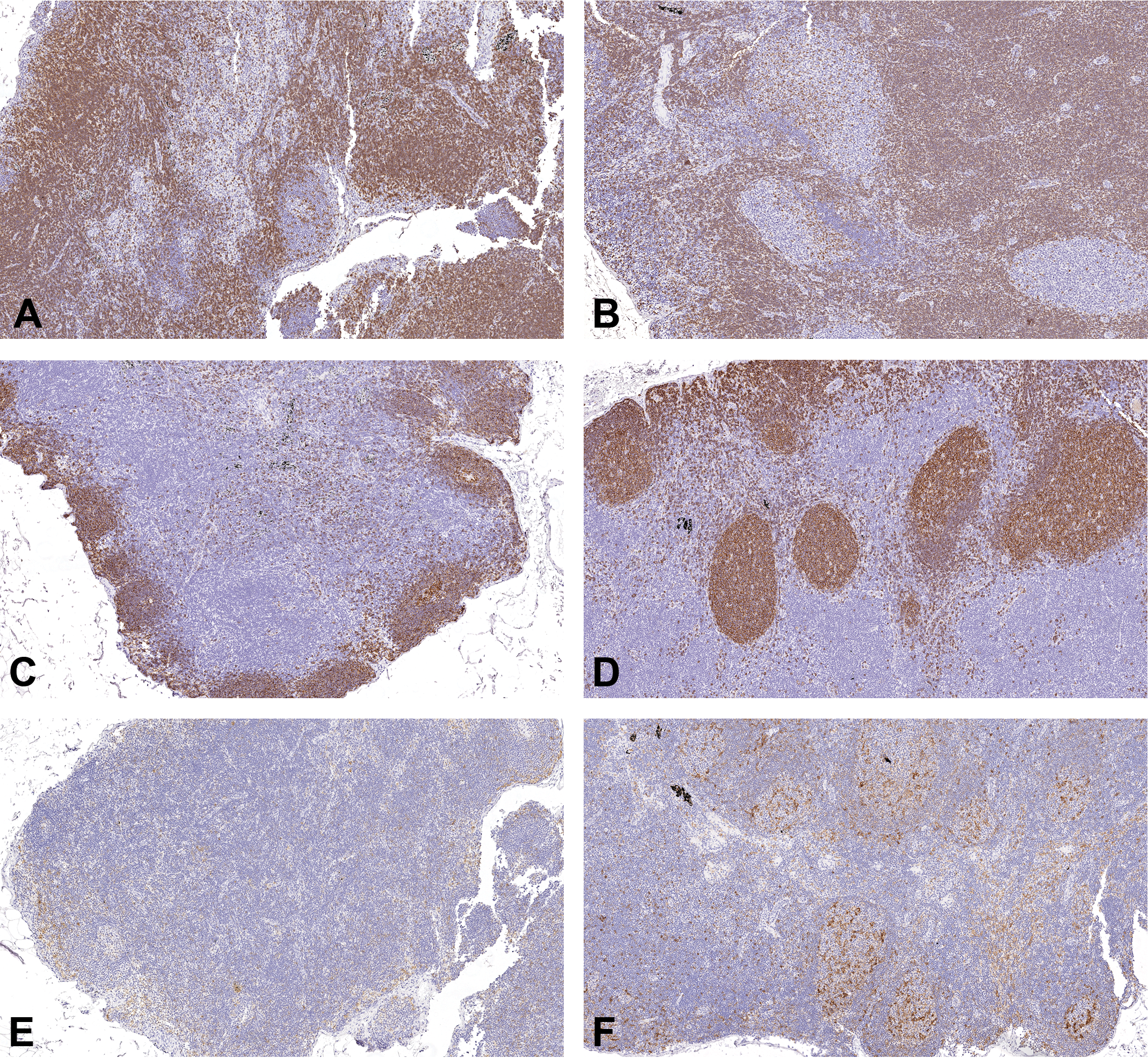
Immunohistochemistry of tracheobronchial lymph node sections from cynomolgus monkeys in a 6-week inhaled anti-thymic stromal lymphopoietin study. A, Control female (1945) terminal on day 43. CD3. Marked numbers of positive round cells in the paracortex with fewer positive solitary round cells scattered within the medullary cords, sinuses, and follicles. Original Objective ×10. B, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. CD3. Marked numbers of positive round cells in the paracortex with fewer positive solitary round cells scattered within the medullary cords, sinuses, and follicles and also germinal centers. Original Objective ×10. C. Control female (1945) terminal on day 43. CD20. Marked numbers of positive round cells in the germinal centers, moderate numbers in the follicles, and fewer positive solitary round cells scattered within the medullary cords, sinuses, and paracortex. Original Objective ×10. D, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. CD20. Marked numbers of positive round cells in the germinal centers, moderate numbers in the follicles, and fewer positive solitary round cells scattered within the medullary cords and paracortex. Original Objective ×10. E, Control female (1945) terminal on day 43. CD25. Minimal numbers of large irregular cells and fewer small round cells with indistinct cell boundaries scattered throughout the medullary cords and sinuses. Original Objective ×10. F, High-dose female (4947) given 7655 µg/kg/d terminal on day 43. CD25. Slight numbers of large irregular cells and fewer small round cells with indistinct cell boundaries in the germinal centers, medullary cords, and sinuses and fewer positive cells scattered within the follicles and paracortex Original Objective ×10.
Anti-drug antibodies
No preexisting anti-GSK3191812 or VH nonspecific antibodies were detected prior to administration of the first dose. However, anti-GSK3191812 antibodies were detected either at day 15 or at the end of the dosing period in all monkeys given GSK3191812, with titers ranging from 10 to 1000 (Table 4). In the case of 2 males given 400 µg/kg/d, there were no quantifiable GSK3191812 plasma concentrations across the entire 24-hour TK profile during week 6. This was attributed to the presence of ADAs at greater levels than other animals.
Anti-TNFR1 dAb
7-day inhaled toxicity study
Toxicokinetics
GSK1995057 plasma concentrations were quantifiable for the entire 24-hour sampling period after dosing at both dose levels on days 1 and 7 and
Clinical and anatomic pathology
Moderate bilateral red foci of the kidneys were noted in the female given 2.9 mg/kg/d. This macroscopic finding correlated microscopically with multiple foci of moderate epithelial degeneration/regeneration and free erythrocytes seen within the lumen of the cortical tubules. Minimal thinning of the tracheal epithelium was also seen microscopically at ≥2.9 mg/kg/d. The findings in the kidney and trachea were considered of uncertain relationship to test article.
Anti-drug antibodies
Although treatment-naive monkeys were used in this study, the low-dose (2.9 mg/kg/d) female monkey had measurable preexisting anti-GSK1995057 antibodies prior to dosing on day 1, but they were not detected on day 7 (Table 4). Both unlabeled GSK1995057 and a VH nonspecific dAb (with high homology to GSK1995057 but no binding capacity for TNFR1) could completely inhibit the anti-GSK1995057 antibody detection signal in the ECLIA, suggesting that the preexisting antibodies recognized common, framework-specific regions on the dAb rather than those associated with target binding.
14-Day inhalation toxicity study
Toxicokinetics
Systemic exposure to GSK1995057 generally increased in a greater than dose-proportional manner at 0.92 or 5 mg/kg/d and less than dose proportionally at 5 or 14.4 mg/kg/d, with no gender differences noted (Table 3). After 14 days of administration, there was a notable but variable increase in plasma concentrations of GSK1995057 relative to day 1 (∼20-fold mean change, range 0.5- to 138-fold) where comparable time points had been collected (5 and 24 hours after the start of dosing).
Clinical and anatomic pathology
In the lungs, test article–related slight-to-moderate red discoloration was seen macroscopically at doses ≥0.92 mg/kg/d but could not be correlated with any microscopic findings. Slight-to-moderate increases in perivascular/peribronchiolar/alveolar lymphocytic infiltrate were seen in the lungs of the majority of animals given GSK1995057 (Figure 5; Table 4). Also, slight-to-moderate increased lymphocytic cellularity was noted in the TBLN of all animals given ≥5mg/kg/d. There were no test article–related effects on clinical pathology parameters, lymphocyte subset counts, or systemic concentrations of IL-1β, IL-6, IL-8, TNF-α, and C-reactive protein.
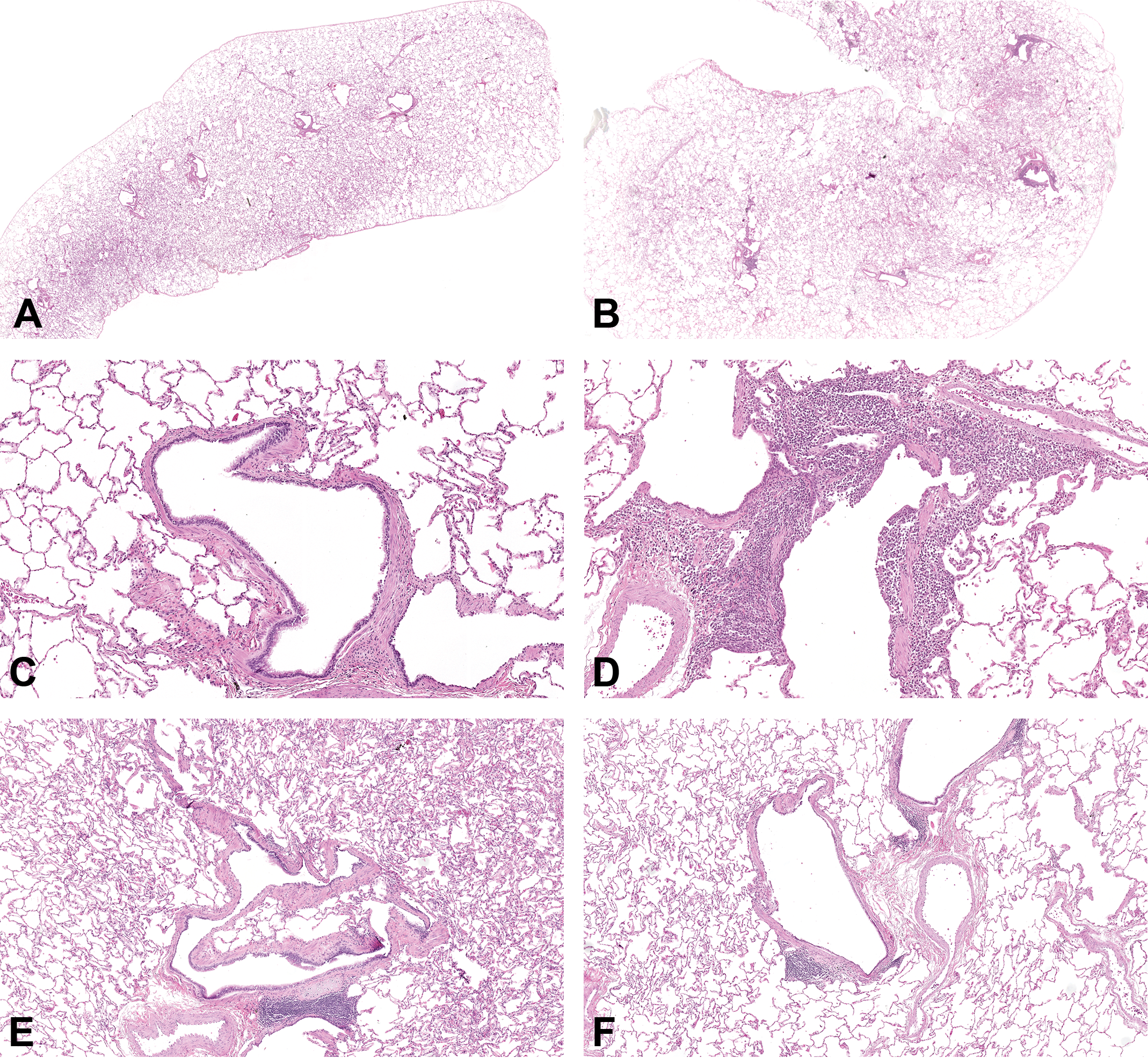
Lung sections from cynomolgus monkeys in 14-day inhaled anti-TNFR1 dAb study with a 14-day recovery period. A, Control male (1376) final-phase termination. Original Objective ×0.5. Normal appearance of lung. B, High-dose male (4376), final-phase termination, given 14.4 mg/kg/d. Moderate perivascular/peribronchiolar/alveolar lymphocytic infiltrate extends into alveolar interstitium and space. Original Objective ×0.5. C, Control male (1376), final phase termination. Normal appearance of small airway and adjacent alveoli. Original Objective ×10. D, High-dose male (4376), final phase termination, given 14.4 mg/kg/d. Moderate perivascular/peribronchiolar/alveolar lymphocytic infiltrate extends into alveolar interstitium and space ×10. E, Control female (1877), recovery phase. Normal appearance of small airway, adjacent alveoli, and BALT at the bottom of the image. Original Objective ×5. F, High-dose female (4877) recovery phase, previously given 14.4 mg/kg/d. Slight perivascular/peribronchiolar/alveolar lymphocytic infiltrate extends into alveolar interstitium and space. Original Objective ×5. dAb indicates domain antibody; TNFR1, tumor necrosis factor receptor 1.
At the end of the recovery period (day 29), full reversibility of the findings in the TBLNs and partial reversibility of the pulmonary lymphocytic infiltrates were observed.
Anti-drug antibodies
Although treatment-naive monkeys were utilized in this study, preexisting anti-GSK1995057 antibodies were present in 3 of 10, 1 of 6, 0 of 6, and 2 of 10 monkeys in the 0 (vehicle), 0.92, 5, and 14.4 mg/kg/d groups, respectively (Table 4). In the ECLIA, both free GSK1995057 and a VH nonspecific dAb (with high homology to GSK1995057 but no binding capacity for TNFR1) could completely inhibit the anti-GSK1995057 antibody detection signal in all day 1 and vehicle control samples tested, suggesting that the preexisting antibodies recognized common framework specific regions on the dAb rather than those associated with target binding. At the end of the dosing period (day 14), anti-GSK1995057 antibodies specific for the target binding site of GSK1995057 were detected in 0 of 10, 6 of 6, 3 of 6, and 0 of 10 monkeys in the 0 (vehicle), 0.92, 5, and 14.4 mg/kg/d dose groups, respectively. On day 14, high levels of circulating GSK1995057 in the animals given 14.4 mg/kg/d likely prevented detection of anti-GSK1995057 antibodies due to assay interference since at the end of the recovery period (day 29), GSK1995057-binding site-specific antibodies were detected in all 4 recovery animals given 14.4 mg/kg/d.
14-Day IV toxicity study
Toxicokinetics
GSK1995057 plasma concentrations were quantifiable during the entire 24-hour sampling period on days 1, 7, and 14 and in the plasma samples collected on days 4, 10, and 12 at all doses (Table 3). Additionally, GSK1995057 was quantifiable (0.486-0.751 ng/mL) in the 20 mg/kg/d dose group following the 21-day recovery period on day 36. There were no marked (>2-fold) differences in systemic exposure (AUC0-t and
Systemic exposure increased approximately dose proportionally for male and female monkeys on days 1 and 7. However, day 14 systemic exposure in male monkeys increased less than dose proportionally, whereas for female, monkeys systemic exposure increased in a greater than dose-proportional manner.
Following repeat dosing, there was no change in systemic exposure between day 1 and day 7; however from day 7 to day 14, both mean AUC0-t and
Clinical and anatomic pathology
In females, dose-dependent slight increases in B cells (up to 2.37× controls) were observed at all doses on day 14 (4 hours postdose). At doses of 2 and 8 mg/kg/d, B-lymphocyte counts returned to baseline levels at 23 hours postdose on day 14. In monkeys administered 20 mg/kg/d, B-cell counts had returned to baseline by day 36 (end of the recovery phase). In addition, a minimal-to-slight increase in total lymphocyte counts was noted in males and females given 20 mg/kg/d (1.26× and 1.35× controls, respectively). The total lymphocyte counts were comparable with concurrent controls by the end of the recovery period. At the end of the dosing period, nondose-related decreases in thymus weights were noted for animals given ≥2 mg/kg/d; by the end of the recovery period, weights in animals previously given 20 mg/kg/d were generally comparable with controls. There were no GSK1995057-related effects on systemic concentrations of IL-1β, IL-6, IL-8, and TNF-α. There were no test article–related macroscopic or microscopic findings.
Anti-drug antibodies
Although treatment-naive monkeys were utilized in this study, preexisting anti-GSK1995057 antibodies were noted in 3 of 32 animals (1/3 males given 8 mg/kg/d and 2/5 males given 20 mg/kg/d) prior to the commencement of dosing (Table 4). These antibodies were found to be framework-specific and not specific for the target-binding site of GSK1995057. Antibodies specific for the target-binding site of GSK1995057 were found in 1 female given 2 mg/kg/d on day 14 and in 1 female previously given 20 mg/kg/d at the end of the off-dose recovery period (day 36). The presence of preexisting framework-specific antibodies was not associated with the detection of binding site-specific anti-GSK1995057 antibodies during the treatment and recovery periods. Also, the presence of anti-GSK1995057 antibodies is not considered to have impacted TK data.
Discussion
Delivering biopharmaceuticals by inhalation is attractive when the intent is to maximize drug delivery to the lung and/or systemic circulation because of the large surface area of the lung and close proximity of the alveolar and vascular systems. 1 However, administration of a protein intended for human therapy to nonhuman laboratory animal species often results in an immune response, and the question arises to what extent the route of administration can exacerbate this response. Inflammatory cell infiltrates in the lung or draining lymph nodes may represent a test article–related immune response, chronic inflammation unrelated to test article administration, or a combination thereof. Nonclinical findings of lymphocytic infiltrates in the lungs following inhaled administration of human proteins with diverse modes of action have previously been reported. 18
In this summary of 5 studies, inhalation of 2 single dAbs was well tolerated in cynomolgus monkeys. The recruitment of lymphocytes to locations around the pulmonary vessels, bronchioles, and alveoli was accompanied by development of ADAs in many animals exposed to GSK3191812 anti-TSLP. To characterize the cellular phenotype of the lung and TBLN mononuclear cell infiltrate, IHC using leukocyte markers was applied to tissues from the 6-week inhalation study with GSK3191812. Increased numbers of T cells (CD3 positive) and B cells (CD20 positive) were observed in the alveolar walls and perivascular/peribronchiolar mononuclear cell infiltrates of animals given GSK3191812. Staining frequency was significantly higher for CD20 than CD3, indicating a predominance of B cells in the infiltrate, with fewer T cells. At least 2 morphologically different cell types exhibited positive CD25 staining. The small round cells were considered to be regulatory T cells and the larger irregularly shaped cells were considered to be DCs, as both cell types have been shown to express CD25. 19 -23 In the TBLN, B cells (CD20 positive) were the majority cell type within germinal centers. Positive staining was seen in the medullary cords and sinuses of all animals; however, it was noticeably stronger and more frequent in animals given GSK3191812. Positive CD25 staining was also seen in the germinal centers, follicles, and paracortex in the treated animals, whereas it was absent in these areas in the control animal. Similar to the mononuclear cell infiltrates in the lungs, CD25 staining was observed in 2 cell types that were consistent with regulatory T cells and DCs. These cellular changes observed within the lung and TBLN are compatible with the induction of an adaptive humoral and cellular immune response to the inhaled protein. 24 Luminal antigens, which in this case are components of the inhaled dAbs, are sampled by DCs, located within the surface epithelium of the bronchial mucosa or in the alveoli, which then migrate through the afferent lymphatics to the TBLN and present antigenic peptides to naive T cells. 25 Activated T cells then proliferate and migrate through the efferent lymphatics into the blood and then exit into the bronchial mucosa through postcapillary venules in the lamina propria or capillaries into the lung parenchyma. This is observed histologically as perivascular/peribronchial lymphoid infiltrate or cuffing.
Interestingly, immunophenotyping of blood or dissociated spleen, mesenteric, and TBLN, which was conducted in both studies evaluating GSK3191812, demonstrated no test article–related effect. The sampling of the TBLN for immunophenotyping was not specific to a particular tissue subcompartment and therefore the resulting single-cell suspension used for the flow cytometry would be anticipated to include the regions of cortex, paracortex, and medulla. This tissue immunophenotyping is not intended to measure characteristic changes in distribution of B and T cells, which are observed microscopically on tissue sections as enlargement of cortical follicles/germinal centers and paracortex, which provide evidence of an ongoing immune response. Therefore, the tissue and blood immunophenotyping appear as relatively insensitive methods for detecting this response. The BAL cytology was only conducted in the 14-day inhalation study with GSK3191812, and although this showed some changes in cell populations in some animals, interpretation is limited since this study consisted of a single animal per sex at each dose group and there is inherent variability in the method.
An adaptive immune response results in the expansion and selection of antigen-specific T and B cells that are recruited to sites of antigen exposure. B cells, in the form of plasma cells, secrete antibodies (including secretory IgA at mucosal sites) that bind to the antigen target in an attempt to eliminate it. The skin and the mucosal membranes make up the primary surface barriers to pathogens, and just beneath these lies the primary machinery to protect the body when these barriers are breached: abundant antigen-presenting cells. Routes of administration that involve the skin or mucosa thus may carry the greatest potential for an immunological response. Clinical evidence of this hypothesis is provided by patients with type 1 and type 2 diabetes who switched from subcutaneous dosing to inhaled delivery of insulin and showed larger ADA responses. 26,27
An ADA response against the humanized mAb therapy GSK3191812 was observed in both of the cynomolgus monkey studies following inhalation of GSK3191812, indicating that they can develop within 14 days of dosing, their induction is not dose-dependent, and, in the 6-week study, they were observed at high incidence. The detection of these anti-GSK3191812 antibodies is consistent with an immune response to the test article, which is a single dAb with a human sequence, which in this case has been recognized as foreign by the cynomolgus monkeys. The source of these antibodies is likely to be the B cells that were demonstrated to be the predominant lymphocyte located in the perivascular/peribronchial areas of the lung and the B-cell-rich germinal centers in the TBLN. The production of ADA due to a host immune response against foreign epitopes present in large molecule therapeutics occurs frequently. 28,29
The 3 studies presented here evaluating GSK1995057, using similar doses, enable investigation of the role of duration and route of exposure on the generation of an immune response against a humanized protein. For example, the 7-day inhalation study produced no test article–related effects in the lung and TBLN and, as would be expected, is of insufficient duration to enable the production of ADAs through an adaptive immune response. In contrast, a study with 14 days inhalation exposure provides sufficient time for the development of an immune response against the administered dAb protein, as evidenced by ADAs within the blood and development of lymphocytic infiltrates in the lungs and increased lymphocytic cellularity in the TBLN. Increased lymphocytes at the site of antigen exposure in the lung or in draining lymph nodes, as a consequence of immune cell recruitment or proliferation, have long lives and homeostatic mechanisms to maintain their presence, sometimes in organized structures in the lung termed inducible BALT, once the initial allergenic stimulus is no longer present. 30 Therefore, it is not surprising that following a 14-day recovery period, perivascular/peribronchiolar mononuclear cell infiltration was still present.
The absence of test article–related findings in the lung or TBLN following IV infusion of GSK1995057 in a 14-day repeat-dose study at doses up to 20 mg/kg/d highlights the importance of route of exposure in eliciting microscopic cellular infiltrates associated with the host immune response to the administered foreign protein. In this study, although there was evidence of an adaptive immune response to the dAb by the production of binding site-specific ADA, the lack of direct inhalation exposure of the lung mucosa to the dAb resulted in no microscopic findings at this site or the draining lymph node. In summary, the effects in the lung and TBLN observed in the 14-day inhalation study are a consequence of an adaptive immune response arising from respiratory exposure to a human protein (GSK1995057) and were not observed in the 14-day IV study, which had higher systemic exposures (AUC) than the inhalation study. This conclusion of an immune mechanism is also supported by the very similar microscopic observations in the lung and TBLN following inhalation dosing with the anti-TSLP dAb. In both cases, there was no histologic evidence of tissue damage, inflammation, or perceivable alteration in the function of the respiratory system and therefore the lung findings were considered nonadverse. There was no evidence of the multi-organ vasculitis or mural fibrinoid necrosis associated with immune-complex disease. 31,32
One intriguing difference between the ADA data on the 2 dAbs was the observation in all 3 studies evaluating GSK1995057 of endogenous, preexisting, framework-specific ADA in a minority of naive monkeys prior to the commencement of dosing. These antibodies bound common, framework-specific regions on the dAb rather than those associated with target binding. In studies previously described by Cordy et al, 33 the anti-framework antibodies were shown to be specific for the C-terminus of the GSK1995057. Indeed, a simple C-terminal alanine extension reduced binding of the preexisting antibodies to GSK1995757 in patients. 33 Natural antibodies to cytokines can be found in the sera of normal healthy nonhuman primates and healthy individuals in the absence of specific immunostimulation. However, the function, impact, and purpose of natural antibody development have yet to be fully elucidated. 33 These framework-specific antibodies were not detected at the end of the dosing period in any of the repeat-dose cynomolgus monkey studies. When GSK1995057 progressed into phase I clinical trials, preexisting, naturally occurring anti-immunoglobulin autoantibodies were discovered in the serum of approximately 50% of drug-naive, normal, healthy humans. 34 These preexisting ADAs were shown to bind to an epitope close to the C-terminal region of VH dAbs, including GSK1995057. In the case of GSK1995057, the complex of these endogenous non-neutralizing human anti-VH (HAVH) autoantibodies with framework sequences of GSK1995057 led to activation of TNFR1 signaling and the occurrence of mild-to-moderate infusion reactions in human with high HAVH autoantibody titers. In the presence of HAVH autoantibodies, GSK1995057-induced cytokine release in vitro due to binding of HAVH autoantibodies to a framework region of the dAb. Subsequently, it was discovered that a single amino acid extension of the C-terminal framework sequence is sufficient to reduce binding of GSK1995057 to HAVH autoantibodies, while retaining comparable pharmacology and biophysical properties. 33 These observations parallel findings of naturally occurring/preexisting autoantibodies to Fab or F(ab′)2 fragments of immunoglobulins in humans. 35,36 The absence of preexisting antibodies in the cynomolgus monkey to GSK3191812 probably reflects differences in epitopes arising from critical regions of the framework of the 2 dAbs.
The cynomolgus monkey was selected as the most relevant toxicology species for these 2 development projects on the basis of pharmacodynamic responsiveness to the test articles. Since both of the pharmacological targets are cytokine or cytokine receptors, this raises the question as to whether exaggerated inhibition of either the TSLP or TNF-α pathway could have contributed to the test article–related lung and TBLN microscopic changes. Thymic stromal lymphopoietin is a key orchestrating cytokine produced by the epithelium (and by cells such as fibroblasts, mast cells, and DCs) in response to allergen exposure, viral infections, and other inflammatory and environmental insults. It acts at the interface of the airway epithelium and the immune system and is upstream of type-2 effector cytokines. Thymic stromal lymphopoietin causes upregulation of costimulatory molecules such as OX40L and CD80 on DCs, priming them to direct naive CD4+ T cells to differentiate into inflammatory Th2 cells, which produce IL-4, IL-5, IL-13, and TNF-α, but not IL-10. Thymic stromal lymphopoietin can also amplify and maintain the allergic response by recruiting (via DC-derived MDC and TARC) and directly activating immune cells capable of producing cytokines involved in type 2 inflammation (eg, Th2 cells, mast cells, eosinophils, and type 2 innate lymphoid cells). 37 Therefore, inhibition of TSLP would be expected to reduce the host immune response to inhaled allergens rather than increase the cellular response at the site of administration.
Turning to the target pathway for GSK1995057, disease models using transgenic mice have provided insights to the role of TNFR1. Compared with wild-type mice, TNF-α−/− mice exhibit an enhanced susceptibility to pathogen invasion. 38 They also exhibit a deficiency in TNFR1. These TNF-α KO models have shown that many of the pro-inflammatory functions of soluble TNF are mediated by TNFR1 signaling. Therefore, for reasons shared with anti-TSLP, exaggerated inhibition of TNFR1 would seem most likely to reduce the cellular response to an inhaled immunogen, rather than enhance it. In addition, no specific immunostaining was noted in lung tissue samples in the GLP tissue cross-reactivity study with either GSK1995057 (human and cynomolgus monkey) or GSK3191812 (only human tissue assessed, data not shown), indicating that the lung findings in the repeat-dose inhalation studies were not related to GSK1995057- or GSK3191812-specific or nonspecific target binding. The absence of any treatment-related increase in IL-6 or IL-8 levels, which were measured in the 14-day inhalation toxicity study with GSK1995057, suggests that no receptor agonism/activation occurred in this in vivo study, even in the presence of preexisting framework-specific ADAs.
The lung changes seen after inhaled administration of the dAbs in these case studies are similar to those seen after inhaled administration of other biologicals. 39 The dAb used in these studies contained human protein sequences. In these studies, human proteins were recognized as foreign “non-self” by cynomolgus monkeys and elicited lung and TBLN responses and ADA. If these human-derived dAbs were administered to humans, the lung and TBLN responses seen in the monkeys in these studies would not be expected to occur, as the proteins would not be expected to be recognized as foreign. Overall, ADA and immunogenicity in laboratory animal species induced by human proteins are not considered to be predictive of similar responses in humans who have been administered human-derived “self” proteins. 40,41 In summary, the lung and lymph node findings seen after inhalation of either dAb were considered secondary to the immunogenic response to a human protein.
Footnotes
Acknowledgments
The authors thank all our colleagues at Covance and GSK in the United Kingdom and the United States for their diligent study direction, scientific contributions, study monitoring, and pathology evaluation. The authors also thank Aili Lazaar, Tracey Wright, Curtis Maier, Maggie Dempster, and Nicola Aston for their helpful review comments. Some of the data were presented at the 17th European Congress of Toxicologic Pathology held on September 20, 2019, in Cologne, Germany.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Richard Haworth, Rajni Fagg, George Karantabias, and Mark Price are employees of GlaxoSmithKline and hold stock and/or stock options in GlaxoSmithKline. Molly Boyle, a Covance employee, declared no potential, real or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
