Abstract
Fusion of biologic therapeutics to hyaluronic acid binding proteins, such as the link domain (LD) of Tumor necrosis factor (TNF)-Stimulated Gene-6 (TSG-6), is expected to increase vitreous residence time following intravitreal injection and provide for long-acting delivery. The toxicity of a single intravitreal dose of free TSG-6-LD and fusion proteins of TSG-6-LD and a nonbinding rabbit antibody fragment (RabFab) were assessed in New Zealand White rabbits. Animals administered free TSG-6-LD exhibited extensive lens opacities and variable retinal vascular attenuation, correlated with microscopic findings of lens and retinal degeneration. Similar but less severe findings were present in animals dosed with the RabFab-TSG-6-LD fusion proteins. In-life ocular inflammation was noted in all animals from 7-days postdose and was associated with high anti-RabFab antibody titers in animals administered fusion proteins. Inflammation and retinal degeneration were multifocally associated with evidence of retinal detachment, and hypertrophy and migration of vimentin, glial fibrillary acidic protein, and glutamine synthetase positive Müller cells to the outer nuclear layer. Further assessment of alternative hyaluronic acid binding protein fusions should consider the potential for retinal degeneration and enhanced immune responses early in development.
Keywords
Introduction
Diseases of the retina, such as age-related macular degeneration (AMD), are best treated via intraocular delivery of therapeutics. Indeed, anti-vascular endothelial growth factor therapies, such as Lucentis and Eylea, have proved to be effective therapies for neovascular AMD when administered every 4 to 8 weeks via intravitreal injection. 1,2 However, intravitreal injection is not an innocuous procedure, requiring a visit to the ophthalmologist for each injection and carrying the risk of endophthalmitis, traumatic cataracts, and retinal detachment. 3 As a result many pharmaceutical companies are investing in potential long-acting delivery (LAD) platforms, which will allow reduced frequency of intravitreal injections while maintaining intraocular drug levels above therapeutic concentrations. 4
Hyaluronan/hyaluronic acid (HA) is a mucopolysaccharide that is ubiquitously expressed in all mammals and is a major component of the vitreous gel. 5 Consequently, the fusion of a therapeutic molecule to an HA-binding moiety is expected to increase the ocular residence time of a therapeutic via binding to the vitreous gel components and reduction of passive clearance. Tumor necrosis factor (TNF)-stimulated gene-6 (TSG-6) is a 35 kDa multifunctional, anti-inflammatory, secreted protein. 6 TSG-6 is constitutively expressed in tissues such as the central nervous system, lung, endocrine pancreas, spleen, and skin and is inducible in several tissues, leukocytes, and in mesenchymal stem cells during inflammation. 6 The link domain (LD) of TSG-6 (TSG-6-LD) binds to several components of extracellular matrices, its major ligand being HA. 6 Therefore, it has been postulated by ourselves and others that therapeutic antibody fragments (Fabs) fused to TSG-6-LD will have extended ocular residence compared to Fabs alone due to their binding to HA in the vitreous gel. 7 TSG-6-LD alone is not expected to provide therapeutic benefit in ocular diseases.
This article describes early tolerability and pharmacokinetic (PK) studies in rabbits with a tool Fab-TSG-6-LD fusion. Unexpected retinal and lens lesions developed alongside a robust antidrug antibody (ADA) response despite the use of a species-matched Fab component. Investigative studies subsequently identified the lens and retinal lesions to be a consequence of TSG-6-LD interactions with ocular components and not secondary to the immune response. The potential relevance of these findings to patients and to other HA-binding domain Fab fusions is discussed.
Materials and Methods
Animals
All in vivo studies were reviewed and approved by the Genentech Institutional Animal Care and Use Committee and were conducted in accordance with the Animal Welfare Act and regulations and the Guide for the Care and Use of Laboratory animals. Naive New Zealand White (NZW) rabbits were purchased from Charles River, Canada, or Covance Lts at approximately 2.5 to 2.8 kg weight. Following an acclimatization period of 7 to 16 days, they were assigned to study.
Test Items
One test item consisted of TSG-6-LD (residues 36-133 of TSG-6 [TNFAIP6], molecular weight 11.8 kD) expressed by secretion from Escherichia coli with a hexa-histidine C-terminal purification tag. Two additional test items were a 1:1 (molecular weight 57.7 kD) or 1:2 (molecular weight 69.8 kD) C-terminal fusion of TSG-6-LD to G10rabFab (RabFab-TSG-6-LD), a species-matched rabbit antibody fragment. 8 These 2 items were produced by secretion from stably transfected Chinese hamster ovary (CHO) cells. G10rabFab was derived from a parent monoclonal antibody raised in rabbits against a phosphorylated peptide derived from the intracellular domain of the human hepatocyte growth factor (cMET) receptor, and as such does not bind any target in the rabbit eye. The 1:1 fusion comprised the LD fused to the C-terminus of the Fab heavy chain using a GGGGS polypeptide linker. A his-tag at the end of the LD was included for purification. Similarly, the 1:2 fusion added a second TSG-6-LD module to the C-terminus of the light chain, also with a GGGGS linker, and with an antiflag epitope peptide at the end. These fusion proteins were purified from CHO supernatants using 3 column chromatography steps consisting of (1) capture on an antigen-affinity column as described in Shatz et al, 8 (2) isolation of His-tagged material on a Nickel-NTA column followed by (3) cation exchange chromatography on SP-Sepharose. AlexaFluor-488 labeling of RabFab and 1:2 RabFab-TSG-6-LD was conducted as per Dickmann et al. 9
The isolated (free) TSG-6-LD was purified from E coli cells using a denaturant extraction and refolding procedure adapted from that described by Kahmann et al. 10 Briefly, E coli cells were extracted using a buffer consisting of 7M guanidine-HCl, 50 mM Tris-HCl, 100 mM sodium tetrathionate, and 20 mM sodium sulfite. After homogenization using a Polytron, centrifugation and filtration of the supernatant, the his-tagged protein was captured on a Ni-NTA column (GE Healthcare) equilibrated with 6M guanidine-HCl, 25 mM Tris-HCl, pH 8.6. The column was washed with 25 mM Tris-HCl pH 8.6, 0.1% Triton X-114 and eluted with buffer containing 250 mM imidazole. TSG-6-LD eluted from the column was refolded by dilution to 1.5 mg/mL followed by overnight dialysis at a temperature of 4 °C versus a solution of 0.5M guanidine-HCl, 0.5 M l-arginine, 1 mM reduced glutathione and 1 mM oxidized glutathione. After buffer exchange into 25 mM sodium acetate, pH 5.0, the refolded material was purified by cation exchange chromatography on SP-Sepharose (GE Healthcare).
All proteins were confirmed to have expected binding to HA by using surface plasmon resonance (SPR) measurements on a Biacore T200 instrument (GE Healthcare) to determine the affinity of the interaction (Table 1). Briefly, the molecules were injected for 80 or 120 seconds onto a Series S CM5 chip (GE Healthcare Life Science Solutions) indirectly coated with biotin-HA (Creative PEGWorks) through immobilized streptavidin. Injection concentrations ranged from 600 nM to 10 µM. The dissociation phase was monitored for 300 to 600 s. Subsequently, the surface was regenerated by injecting 1M MgCl2 for 15 seconds. The derived curves were fitted to a 1:1 Langmuir binding model using the BIAevaluation software in order to determine the binding affinity. All experiments were performed at 25 °C using phosphate-buffered saline (PBS; 10 mM Na2HPO4, 1 mM KH2PO4, 137 mM NaCl, 2.7 mM KCl pH 7.4). AlexaFluor-488 labeling did not significantly affect HA binding. The isolated TSG-6-LD and 1:1 RabFab-TSG-6-LD gave similar HA-binding affinities, the difference being less than the expected 2-fold range in KD values determined by SPR measurements. As expected, the 1:2 RabFab-TSG-6-LD having 2 HA-binding modules per molecule, harnesses an avidity effect to produce an apparent higher affinity for HA.
HA-Binding Affinity.
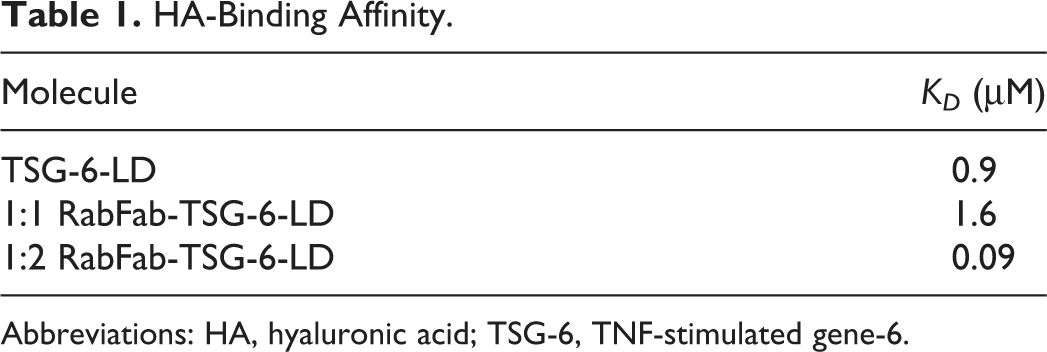
Abbreviations: HA, hyaluronic acid; TSG-6, TNF-stimulated gene-6.
Proteins for animal studies were formulated in PBS, pH 7.4 via dialysis. Formulations were isotonic with Osmolality measured by freezing point method between 300 and 340 mOsm/kg. Analysis by size exclusion chromatography indicated that all test articles were ≥95% monomeric in these formulations. For all animal studies, endotoxin levels were reduced so as to be less than 0.1 endotoxin unit (EU) per eye at the final dosing concentration. Materials for study 3 had endotoxin levels at 0.08 EU/eye (1:1 RabFab-TSG-6-LD) and 0.01 EU/eye (free TSG-6-LD).
Dosing and In Vivo Assessment
Topical anesthetics were applied to the ocular surface of anesthetized rabbits prior to aseptic preparation for intravitreal injection. Single doses of test item were delivered bilaterally to the inferior vitreous, in a total volume of 50 µL/eye. Following the dosing procedure, eyes received topical antibiotics.
Data are presented from 3 separate studies (Table 2). Study 1 was a tolerability study of 1:1 RabFab-TSG-6-LD at the maximum feasible dose of this early stage material. Study 2 was originally designed as a PK study of AlexaFluor-488 (AF-488) labeled 1:2 RabFab-TSG-6-LD to understand the dose exposure relationship when a Fab had 2 TSG-6 LDs fused. This study examined a range of concentrations to explore the potential effect of saturating vitreous HA concentration. Finally, study 3 was an investigative toxicity study designed to compare the onset and severity of lesions induced by approximately equimolar concentrations of free TSG-6-LD and 1:1 RabFab-TSG-6-LD.
Study Design for Toxicity (Studies 1 and 3) and Pharmacokinetic (Study 2) Studies of RabFab-TSG-6-LD Fusions.
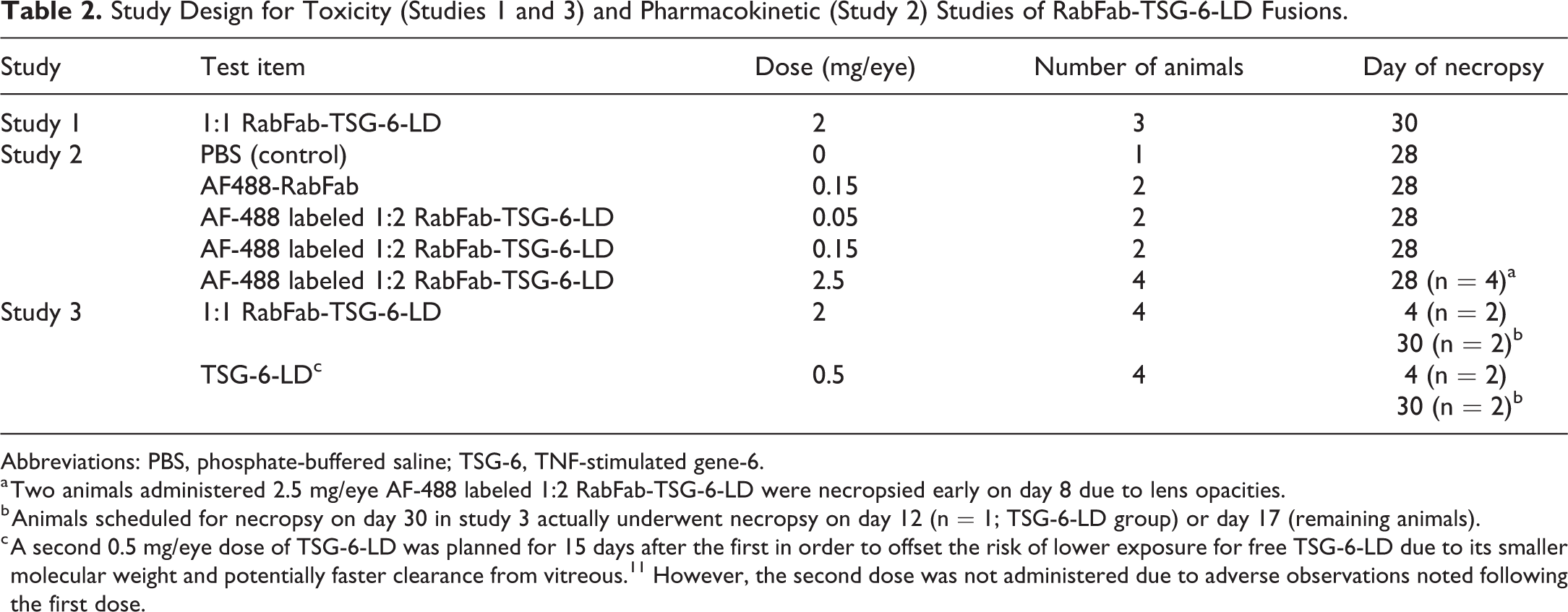
Abbreviations: PBS, phosphate-buffered saline; TSG-6, TNF-stimulated gene-6.
a Two animals administered 2.5 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD were necropsied early on day 8 due to lens opacities.
b Animals scheduled for necropsy on day 30 in study 3 actually underwent necropsy on day 12 (n = 1; TSG-6-LD group) or day 17 (remaining animals).
c A second 0.5 mg/eye dose of TSG-6-LD was planned for 15 days after the first in order to offset the risk of lower exposure for free TSG-6-LD due to its smaller molecular weight and potentially faster clearance from vitreous. 11 However, the second dose was not administered due to adverse observations noted following the first dose.
Tolerability and toxicity were assessed by ophthalmic examination (OE) and intraocular pressure assessments conducted prior to dosing, on the day of dosing (postdose), and then on days 3, 5/6 (study 1), 8, 15, 22, and end of study following dosing. OEs consisted of slit-lamp biomicroscopy and indirect ophthalmoscope examination of the adnexa, anterior portion, aqueous humor, vitreous humor, and fundus. Ocular findings were graded using a modified McDonald-Shadduck grading system. 12 Additionally, there were daily clinical observations and weekly body weight assessments. Pharmacokinetic/toxicokinetic (TK) and ADA evaluations were made as described in Table 3. Serum was collected via the central auricular artery at the times specified for analysis of test item as described below. Test article concentration in vitreous and aqueous humor was measured at specified time points using fluorophotometry in animals administered AF488-labeled test item as described previously. 9 Intravitreal injections and ophthalmic examinations were conducted by a board-certified veterinary ophthalmologist.
Pharmacokinetic, Toxicokinetic, and Antidrug Antibody Assessment Schedule.
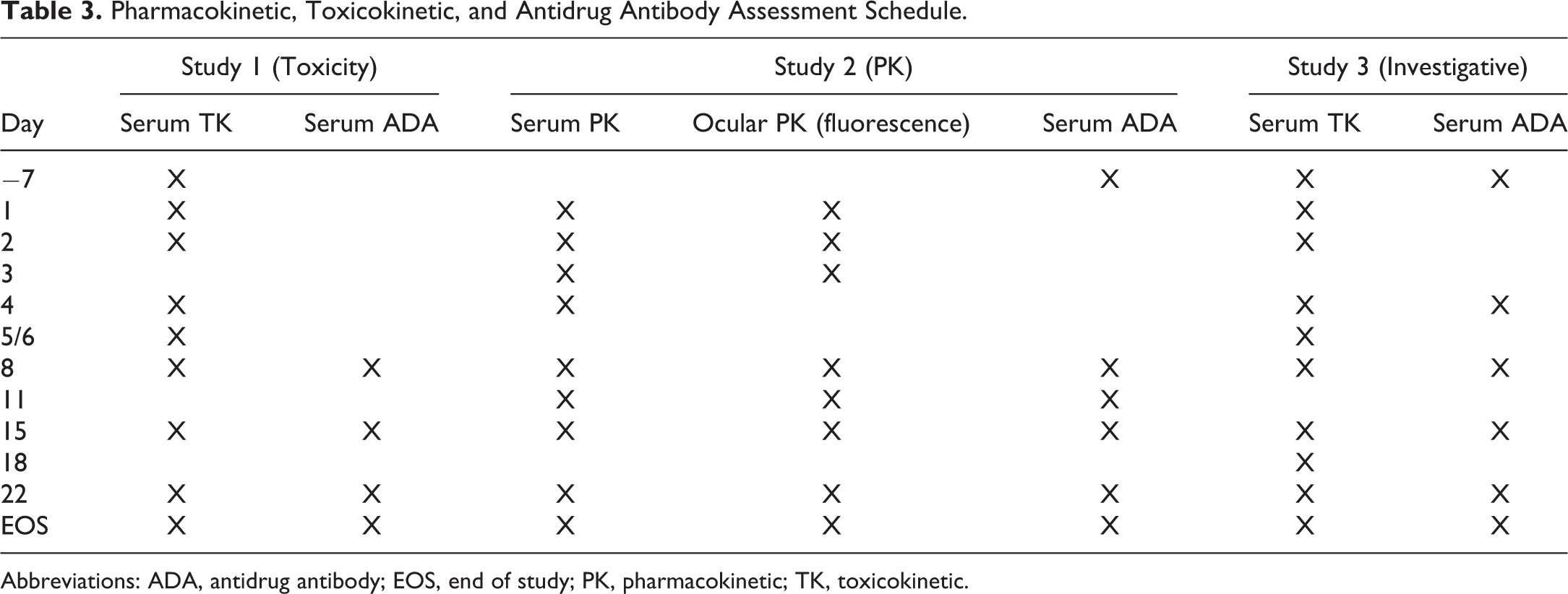
Abbreviations: ADA, antidrug antibody; EOS, end of study; PK, pharmacokinetic; TK, toxicokinetic.
Bioanalytical Assessment
For the determination of the Rabbit Fab molecules in rabbit samples, anti-RabFab ID antibody #9 (Genentech) was diluted to 2 µg/mL in coating buffer (50 mM sodium bicarbonate, pH 9.6) and coated onto 384-well Maxisorp plates (Thermo Scientific) overnight at 4 °C. Plates were washed with washing buffer (PBS 0.05% Tween-20) and blocked with blocking buffer (0.5% bovine serum albumin [BSA], 15 ppm Proclin in PBS) for 1 to 2 hours. The blocking incubation and all subsequent incubations were performed at room temperature with gentle agitation. Standards were made from the source material of each different Rabbit Fab molecule. The standards and samples were diluted in the standard/sample dilution buffer (PBS, 0.5% BSA, 15 ppm Proclin, 0.05% Tween 20, 0.25% CHAPS, 5 mM EDTA, 0.35M NaCl, pH 7.4) with 10% normal rabbit serum and added to the blocked plate for 2 hours. The standard range was 0.156 to 10 ng/mL. Serum samples had an initial dilution of 1:10. The plate bound RabFab molecules were detected with biotinylated anti-Rabbit Fab ID antibody #6 (Genentech) diluted to 1 µg/mL in assay buffer (PBS, 0.5% BSA, 15 ppm Proclin 0.05% Tween 20) for a 1 hour incubation. The plate was washed, followed by an incubation with Fitzgerald StreptAvidin-polyHRP diluted to 25 ng/mL in assay buffer for 1 hour. After a final wash, the plate was developed with tetramethyl benzidine (TMB) peroxidase substrate system (KPL #5120-0077) and the reaction was stopped with 1M phosphoric acid. The plates were read at 450 nm with a 620 nm reference using a plate reader. The concentrations of the Rabbit Fab molecules were calculated from a 4-parameter fit of the respective standard curve.
An ADA ELISA was used to qualify the presence of antidrug antibodies in rabbit serum. RabFab, RabFab-TSG-6-LD, or TSG-6-LD were diluted to 1 µg/mL or 0.5 µg/mL in coating buffer and incubated on 384-well Maxisorp plates overnight at 4 °C. Plates were washed then blocked for 2 hours. This and all subsequent incubations were performed at room temperature with gentle agitation. Naive rabbit serum samples were employed as a negative control, both negative and positive serum controls reflected the same initial dilution as samples. Serum samples were diluted to either a minimum of 1:50 or 1:100 and serially diluted 1:3 in sample buffer either with or without 2% naive rabbit serum, respectively. The controls and samples were incubated on the blocked plates for 2 hours. Bound ADA were detected using HRP-conjugated goat-anti-rabbit Fc antibody (Jackson Immuno Research) at 20 or 40 ng/mL in assay buffer for one hour. After a final wash, TMB was added, color developed, and the reaction was stopped with 1M phosphoric acid. The plates were read at 450 nm with a 620-nm reference using a microplate reader. A cut point (3 times the negative or average predose control) was used to determine ADA-positive and ADA-negative samples. Samples with a signal above the cut point were considered ADA-positive and samples with a signal below the cut point were considered ADA negative. Titer was defined as log10 of the last dilution of serum that has an optical density (OD) above the cut point or was determined with an equation that took into account the 2 dilutions where the OD crosses the cut point:
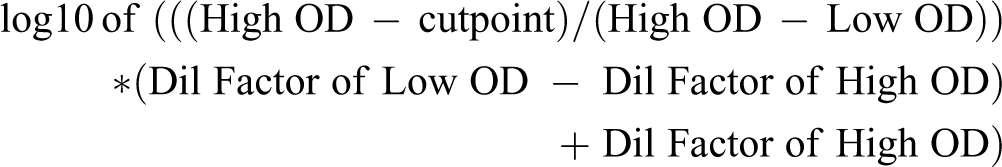
The lowest titer was either 1.7 or 2 since the minimum dilution was 1:50 or 1:100.
Ocular PK Assessment
Concentration-time profiles were used to estimate PK parameters using noncompartmental analysis using Phoenix WinNonlin (Certara Inc). For concentration-time profiles generated using fluorophotometric approaches, sampling in the first 48 hours postdose was excluded from PK analyses due to high variability, likely attributable to interindividual variation in the site of administration and subsequent diffusion of test article through the vitreous. 9 Clearance was calculated as CL = dose/AUC, where dose is known and AUC is measured using the linear trapezoidal method. The volume of distribution at steady state was calculated as V = CL/kel, using the clearance value and the elimination rate constant obtained from the slope of the terminal phase. Elimination half-life was calculated as t1/2 = ln(2)/kel.
Histopathology
At necropsy, the animals underwent a gross examination and eyes were collected for microscopic examination. In study 1, eyes were fixed for 24 hours by submerging the entire eye into modified Davidson’s fixative prior to being transferred to neutral buffered formalin. In study 2, the right eye was processed similarly, while the left eye was frozen for cryosectioning. In study 3, the lateral calotte was removed and the eyes were submerged in Karnovsky’s fixative to allow for potential electron microscopy examination (not conducted). For fixed eyes, 3 superioinferior callotes (lateral, medial, and central) of the globes were routinely processed to paraffin, and 9 hematoxylin and eosin (H&E) stained step sections throughout the globe were examined. The eyes collected for cyrosections were placed temporal side down into Shandon Peel-A-Way Disposable Embedding Molds (Thermo-Fisher) and surrounded with Tissue-Tek OCT (Optimal Temperature Cutting) Compound (Sakura Finetek, USA INC 4583) cryoembedding medium before being placed into a CB-60 cryobath unit (NESLAB 23700) filled with 2-Methylbutane at −45 °C to solidify. The frozen blocks were cryosectioned on a Leica CM 3050S cryostat at −20 °C. Two 7-µm thick sections were collected at 2 levels, one at one-third into the globe and the other at midline of globe containing optic nerve and lens, onto Superfrost Plus positively charged microscope slides (Thermo Scientific) charged slides. Cryosections were mounted with Invitrogen ProLong Gold with DAPI (Thermo Fisher Scientific, P36931) and VWR micro cover glass 24 × 50 mm coverslips (VWR). They were scanned at ×20 on a Hamamatsu XR whole slide scanner using DAPI/FITC filters from Semrock.
Immunohistochemistry
A representative eye (modified Davidson’s fixed) was assessed for expression of vimentin, glial fibrillary acidic protein (GFAP), glutamine synthetase (GS), and Ki67. The antibodies and methods used are provided in Table 4. The tissue positive controls were normal human skin for vimentin, normal rabbit brain for GFAP, normal human tonsil for Ki67, and normal rabbit liver for GS. Isotype controls were used in each immunohistochemistry experiment.
Immunohistochemistry Methods.
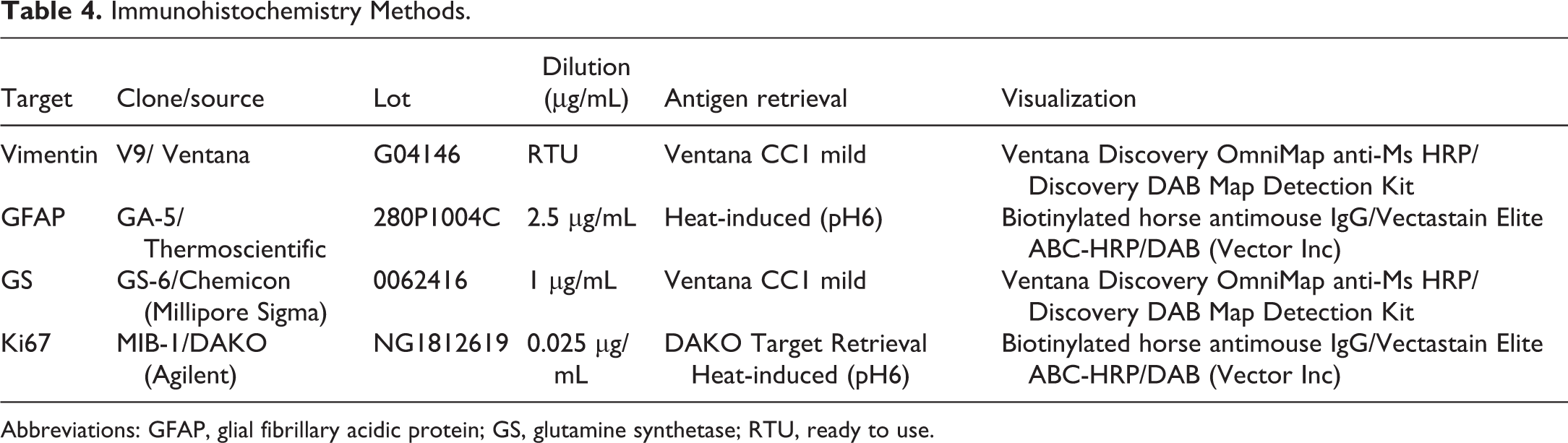
Abbreviations: GFAP, glial fibrillary acidic protein; GS, glutamine synthetase; RTU, ready to use.
Results
Studies 1 and 2: Lens opacities, inflammation, and retinal degeneration in rabbits administered 1:1 RabFab-TSG-6-LD and AF488 labeled-1:2 RabFab-TSG-6-LD.
In study 1, 3 of 3 rabbits administered 2 mg/ eye 1:1 RabFab-TSG-6-LD developed significant (grade 3-4) anterior and vitreal cell infiltrates by day 15. In study 2, 4/4 animals administered 2.5 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD developed multifocal posterior lens opacities by day 3. Subsequently, dose-dependent incidence and severity of aqueous/vitreous cells developed in animals administered ≥0.15 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD from day 15, although visualization of the vitreous chamber was hampered by the lens opacities in the high-dose group. In comparison, only trace vitreal cells and no other ocular findings were observed in animals administered AF488 labeled-RabFab, and there were no significant findings in the animal administered PBS. Vitreal cell scores from animals in both study 1 and study 2 are shown in Figure 1. Topical atropine and prednisone were administered to all affected rabbits and they remained comfortable till scheduled necropsy.
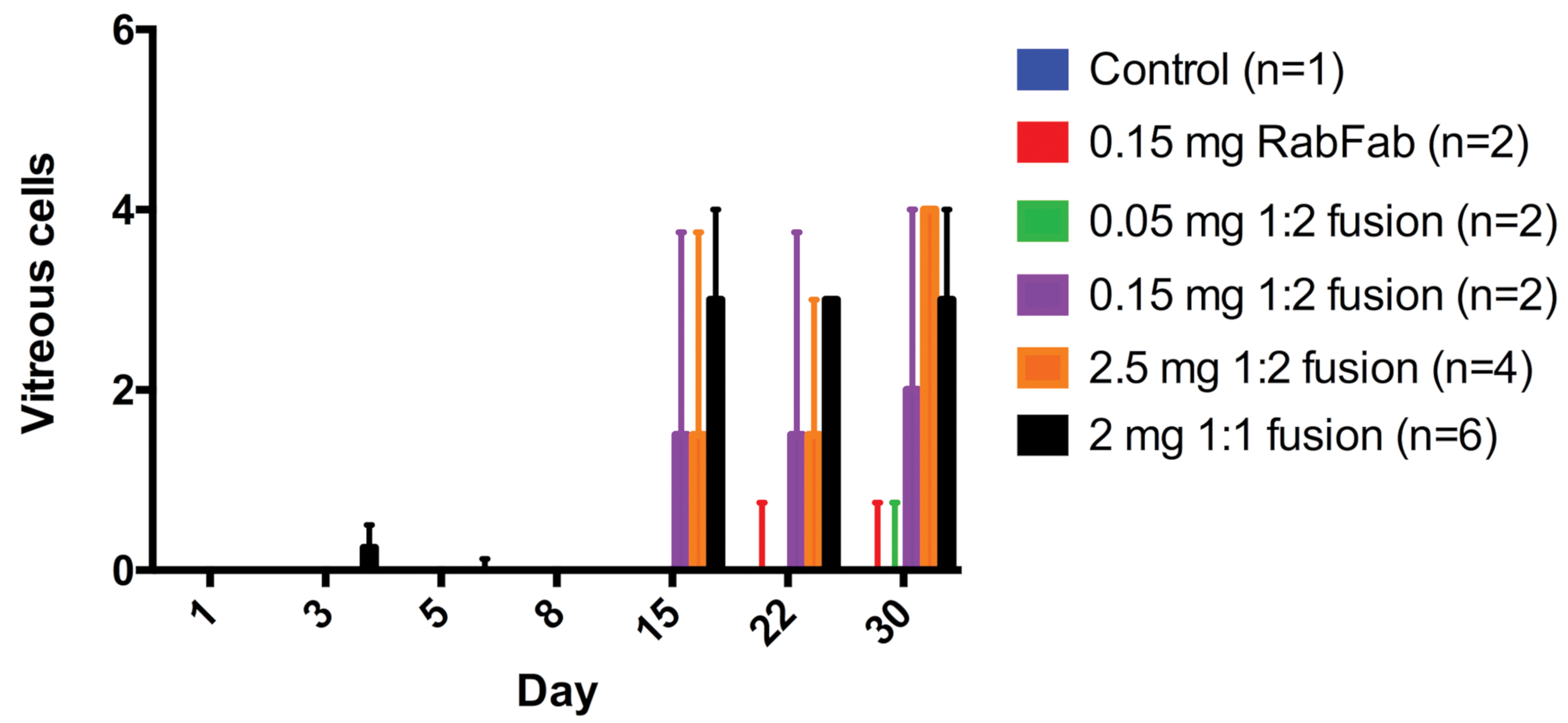
Study 1 and 2. Vitreous cell score based on ophthalmic examinations following intravitreal administration of PBS (control), AF488 labeled-RabFab, AF488 labeled-1:2 RabFab-TSG-6-LD, or 1:1 RabFab-TSG-6-LD to NZW rabbits. 2/4 animals administered 2.5 mg/eye AF488 labeled-1:2 RabFab-TSG-6-LD were euthanized ahead of schedule on day 8. Significant inflammation was only noted from day 15 postdose in animals administered ≥0.15 mg/eye 1:2 RabFab-TSG-6-LD and 2 mg/eye 1:1 RabFab-TSG-6-LD. Bars represent the median score per eye and bars the interquartile range. NZW, New Zealand White; PBS, phosphate-buffered saline; TSG-6, TNF-stimulated gene-6.
Microscopic findings from both studies 1 and 2 are presented in Table 5. In-life inflammation was correlated with minimal to mild, predominantly mononuclear cell infiltrates in the vitreous, retina, optic nerve head (perivascular), and ciliary body (Figure 2). Lens opacities present in animals administered 2.5 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD were correlated with multifocal lens fiber swelling and degeneration, subjacent to the posterior capsule and close to the axis, which was observed in 1 eye. The multifocal nature of the in-life lens opacities suggests that lesions in other lenses may have been missed in section. Lens degeneration was not present in animals administered 0.05 or 0.15 mg/eye of AF-488 labeled 1:2 RabFab-TSG-6-LD or 2 mg/eye 1:1 RabFab-TSG-6-LD. Eyes administered PBS and 0.15 mg/eye AF-488 labeled RabFab were within normal limits and animals administered 0.05 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD exhibited only minimal mononuclear infiltrates in 1 of 2 eyes.
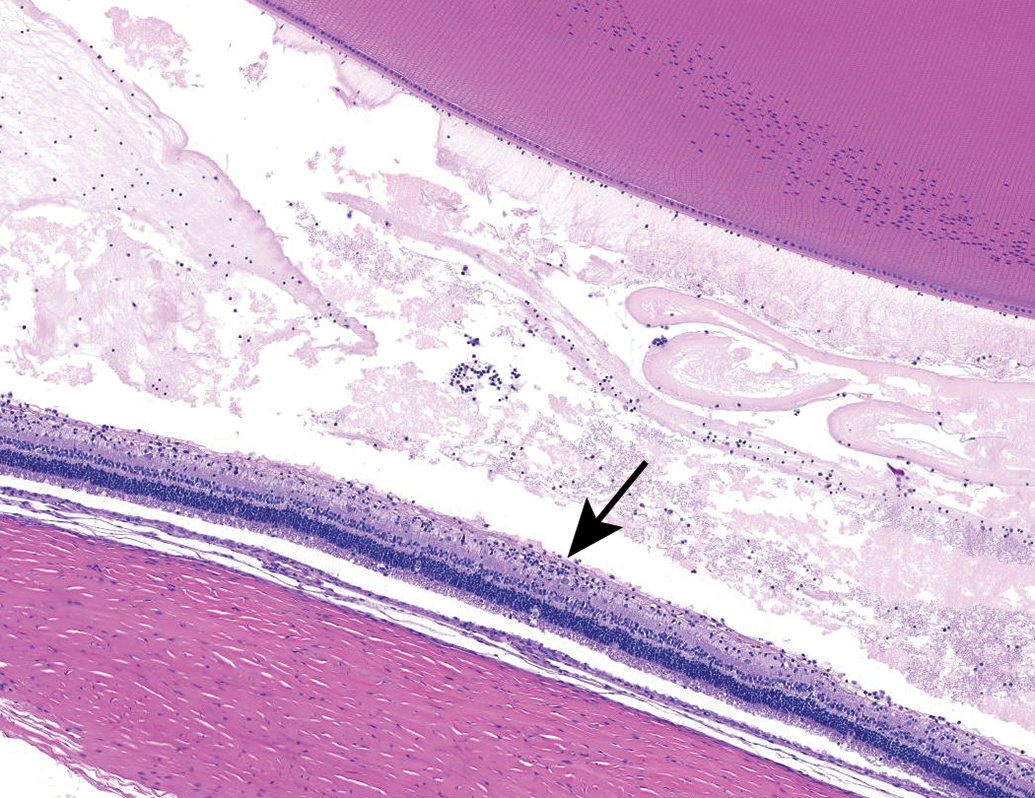
Mononuclear inflammatory cells and increased protein (eosinophilic material) in the vitreous chamber. Clusters of inflammatory cells multifocally infiltrate the retina (arrow). NZW Rabbit, Eye, 2 mg/eye 1:1 RabFab-TSG-6-LD, 30 days post intravitreal dose. Hematoxylin and eosin (H&E). NZW, New Zealand White; TSG-6, TNF-stimulated gene-6.
Unexpectedly, outer retinal degeneration and detachment was noted in animals administered 2 mg/eye 1:1 RabFab-TSG-6-LD and 0.15 or 2.5 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD (Figures 3 and 4). Retinal degeneration presented with variable severity ranging from minimal vacuolation and hypercellularity of the photoreceptor (PR) layer (consistent with photoreceptor swelling and nuclear displacement), to severe necrosis and detachment with regional loss of the PR layer, outer nuclear layer (ONL), and to a lesser extent the inner nuclear layer. When present in either study 1 or 2, retinal findings were multifocally present in all sections of the globe examined and affected the peripheral retina and posterior retina equally. In study 2, the severity of the retinal degeneration was in general dose-dependent, but there was significant variability in severity between animals and between 2 eyes of the same animal. Severe retinal degeneration was identified at 8 days postdose in 2 animals administered 2.5 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD. Frozen sections of eyes collected on study day 8 were examined to assess sublocalization of AF-488 labeled 1:2 RabFab-TSG-6-LD. Test item could clearly be identified in the vitreous chamber, but there was no significant signal within the layers of the retina (Figure 5).
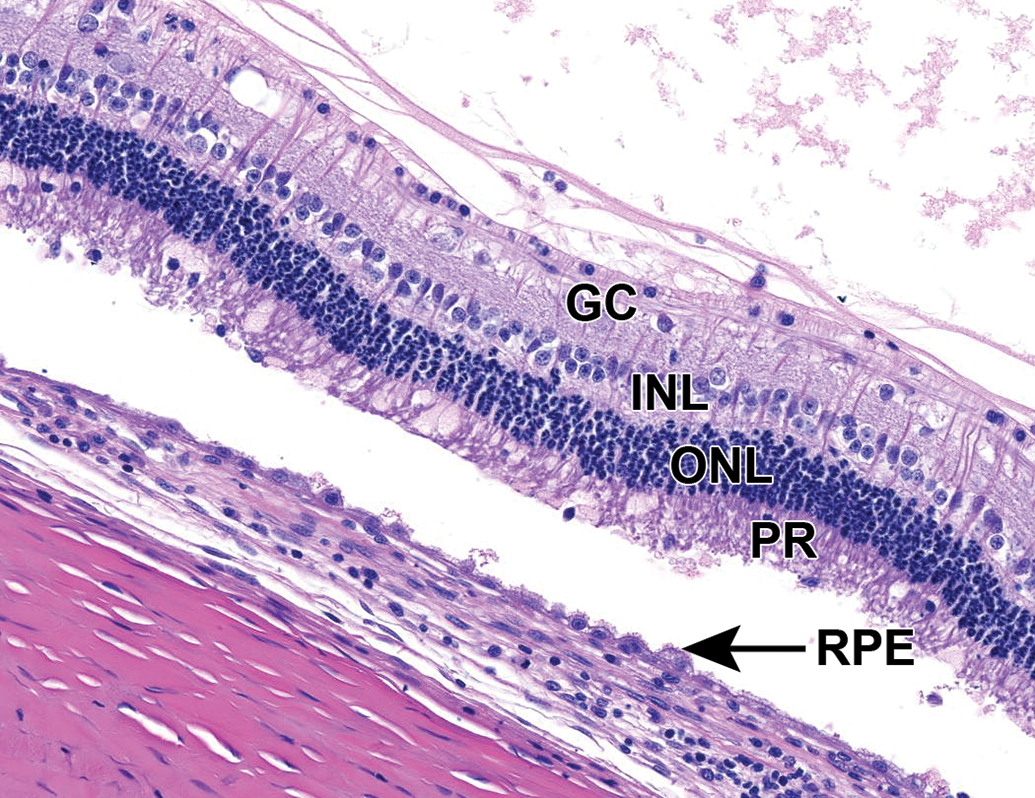
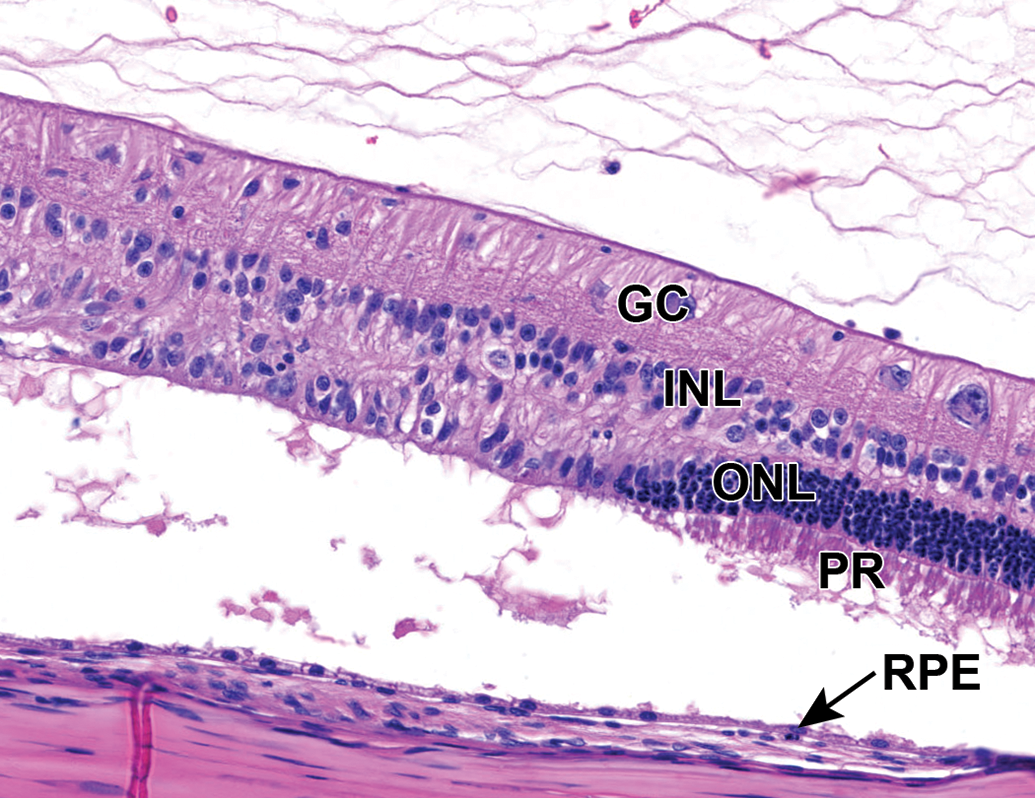
Mild multifocal outer retinal degeneration, characterized by vacuolation and hypercellularity of the photoreceptor layer (interpreted as swelling of photoreceptors and nuclear displacement, respectively) and retinal detachment. Retinal pigment epithelial cells are multifocally hypertrophied. NZW Rabbit, Eye, 2 mg/eye 1:1 RabFab-TSG-6-LD, 30 days post intravitreal dose. Hematoxylin and eosin (H&E). GC indicates ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer; RPE, retinal pigment epithelium. NZW, New Zealand White; PR, photoreceptor layer; TSG-6, TNF-stimulated gene-6.
Marked multifocal retinal degeneration and detachment with regional loss of the photoreceptor layer and substantial loss of the outer and inner nuclear layer. NZW Rabbit, Eye, 2.5 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD, 8 days post intravitreal dose. Hematoxylin and eosin (H&E). GC indicates ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer; RPE, retinal pigment epithelium. NZW, New Zealand White; PR, photoreceptor layer; TSG-6, TNF-stimulated gene-6.
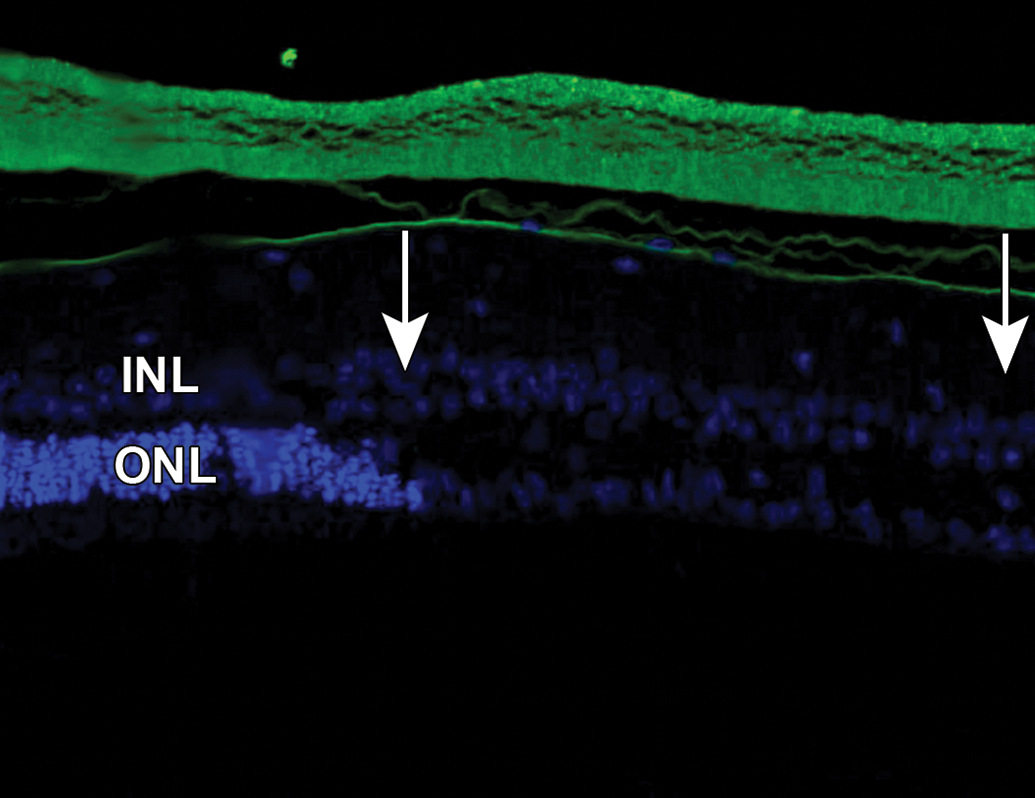
AF-488 labeled 1:2 RabFab-TSG-6-LD is present within the vitreous chamber and abuts the inner limiting membrane. However, no significant AF-488 signal was noted within the retina. The region to the right of the image (between arrows) shows significant degeneration as characterized by the decreased nuclei in the outer nuclear layer. NZW Rabbit, Eye, 2.5 mg/eye AF-488 labeled 1:2 RabFab-TSG-6-LD, 8 days post intravitreal dose. Green = AF-488 labeled 1:2 RabFab-TSG-6-LD. Cryosection, DAPI counterstain. INL indicates inner nuclear layer; NZW, New Zealand White; ONL, outer nuclear layer; TSG-6, TNF-stimulated gene-6.
Microscopic Findings in Study 1 and Study.a
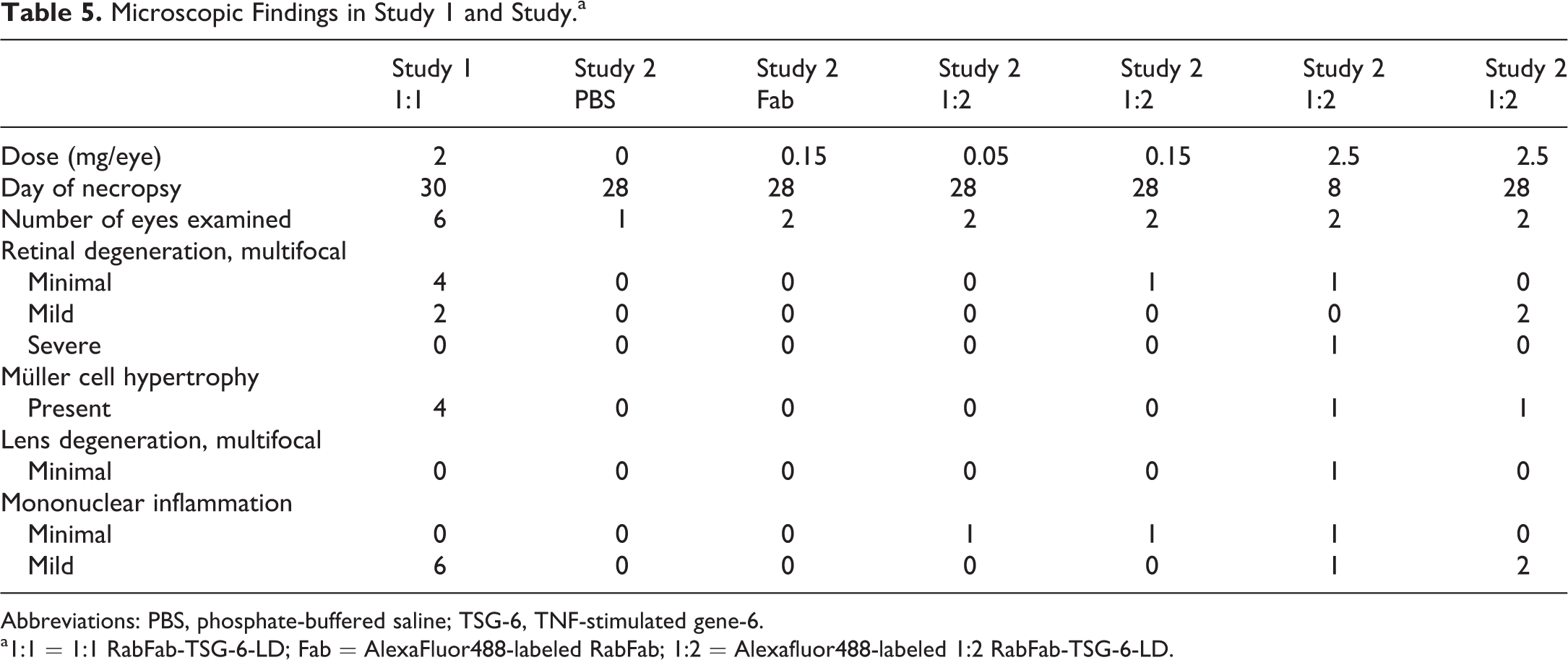
Abbreviations: PBS, phosphate-buffered saline; TSG-6, TNF-stimulated gene-6.
a1:1 = 1:1 RabFab-TSG-6-LD; Fab = AlexaFluor488-labeled RabFab; 1:2 = Alexafluor488-labeled 1:2 RabFab-TSG-6-LD.
In 4 of 6 eyes administered 2 mg 1:1 RabFab-TSG6-LD and 2 of 4 examined eyes administered 2.5 mg AF488-labeled 1:2 RabFab-TSG-6-LD there were conspicuous, swollen cells in the ONL with large nuclei exhibiting unique chromatin formation (Figure 6). Due to their location and morphology, these cells were suspected to be Müller glia and a panel of antibodies were selected to confirm this hypothesis. Immunohistochemical staining of an eye administered 2 mg 1:1 RabFab-TSG6-LD revealed them to have strong cytoplasmic reactivity for vimentin, GFAP, and GS (Figures 7 -9). Müller cells are expected to be positive for vimentin, but this is not specific and may also react with other ocular structures. Glial fibrillary acidic protein may be expressed in astrocytes and is upregulated following activation of Müller cells. Glutamine synthetase is expressed in both Müller cells and astrocytes, but astrocytes are predominantly limited to the medullary rays of the rabbit globe. 13 In addition, the nuclei of the Müller cells were variably Ki67 positive, a marker of cell proliferation (Figure 10). The staining pattern and localization of these cells in the ONL is consistent with them being activated Müller glia cells.
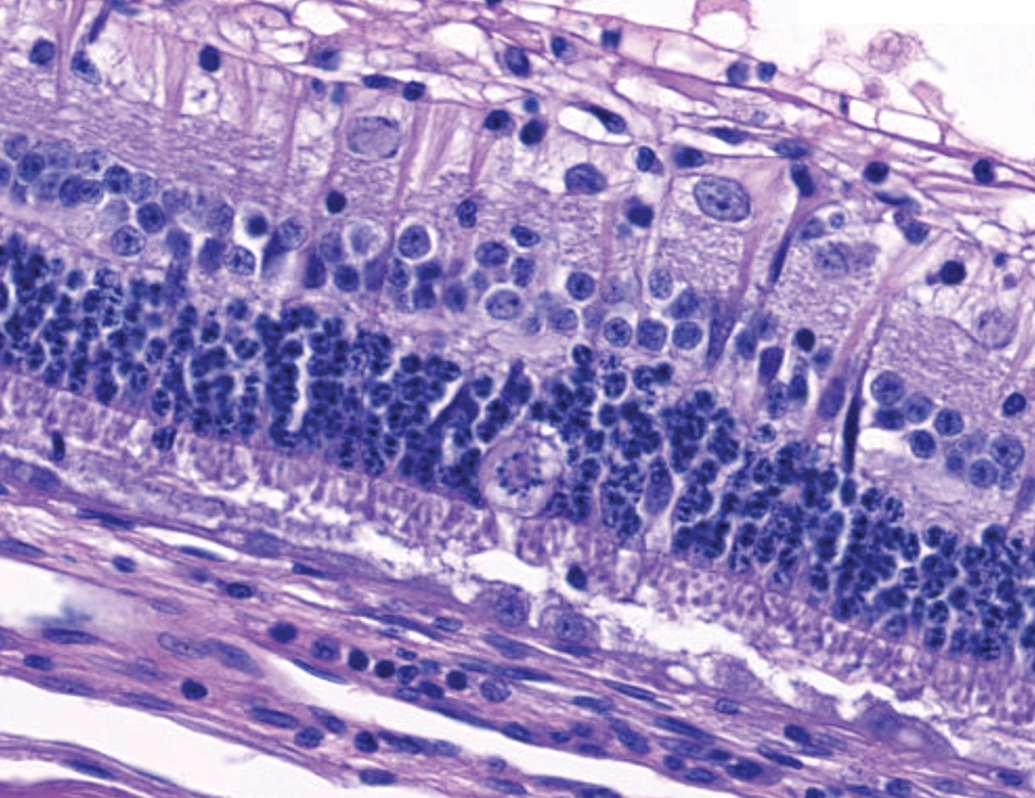
Swollen cell bodies with unique chromatin arrangement in the ONL. Immunohistochemical assessment (see subsequent figures) identified these cells as Müller glia. Retinal pigment epithelial cells are multifocally hypertrophied consistent with the photoreceptor damage and retinal detachment described. NZW Rabbit, Eye, 2 mg/eye 1:1 RabFab-TSG-6-LD, 30 days post intravitreal dose. Hematoxylin and eosin (H&E). NZW, New Zealand White; ONL, outer nuclear layer; TSG-6, TNF-stimulated gene-6.
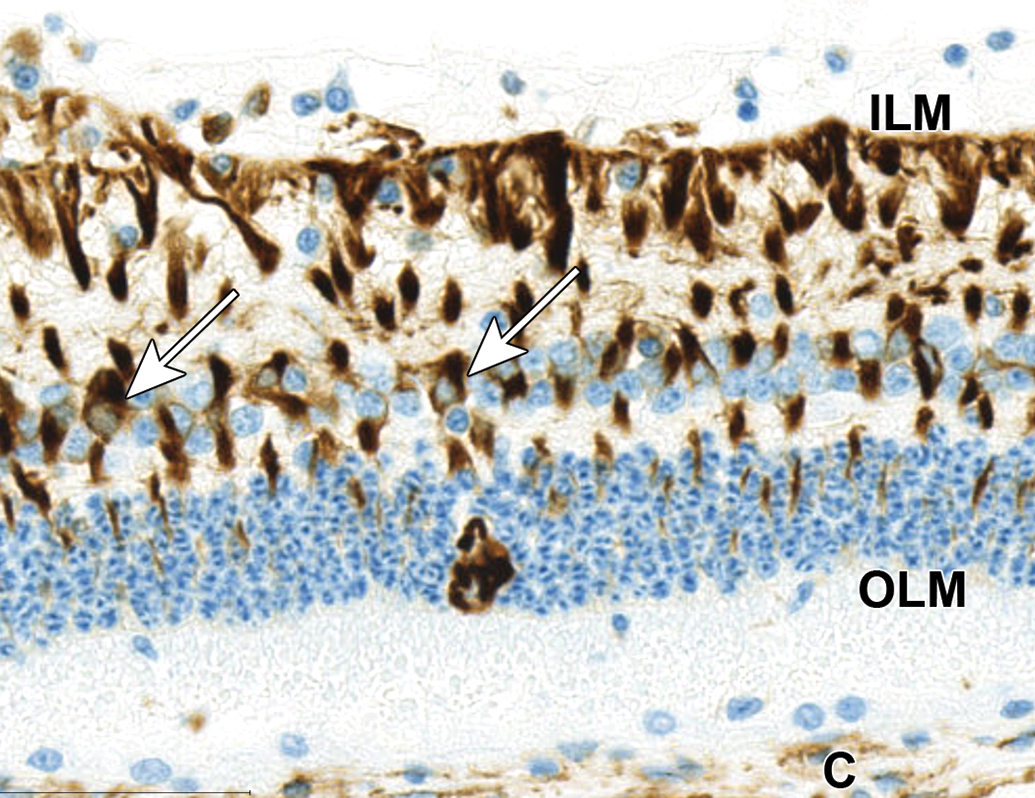
Swollen cell bodies with unique chromatin arrangement in the ONL exhibit cytoplasmic reactivity for vimentin. Surrounding normal Müller cells are also immunoreactive for vimentin. The cell bodies (arrows) are in the inner nuclear layer and processes extend from the inner limiting membrane (ILM; dark staining) to the outer limiting membrane (OLM; lighter staining). Stromal cells of the choroid (C) are also immunoreactive for vimentin. NZW Rabbit, Eye, 2 mg/eye 1:1 RabFab-TSG-6-LD, 30 days post intravitreal dose. Anti-vimentin DAB, hematoxylin counterstain. NZW, New Zealand White; ONL, outer nuclear layer; TSG-6, TNF-stimulated gene-6.
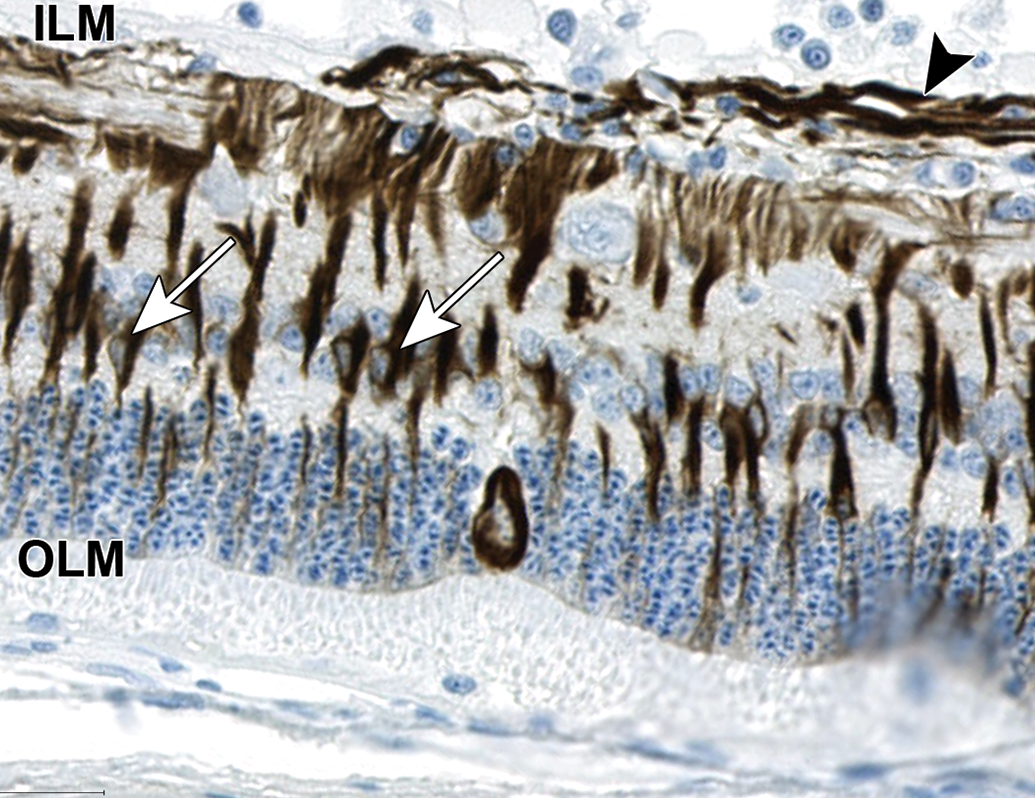
Swollen cell bodies with unique chromatin arrangement in the ONL exhibit cytoplasmic reactivity for glial fibrillary acidic protein (GFAP). Surrounding normal Müller cells are also immunoreactive for GFAP. The cell bodies (arrows) are in the inner nuclear layer and processes extend from the inner limiting membrane (ILM; dark staining) to the outer limiting membrane (OLM; lighter staining). In the upper part of image, horizontal GFAP immunoreactive fibers (arrow head) likely represent astrocytic processes in the nerve fiber layer. NZW Rabbit, Eye, 2 mg/eye 1:1 RabFab-TSG-6-LD, 30 days post intravitreal dose. Anti-GFAP DAB, hematoxylin counterstain. NZW, New Zealand White; ONL, outer nuclear layer; TSG-6, TNF-stimulated gene-6.
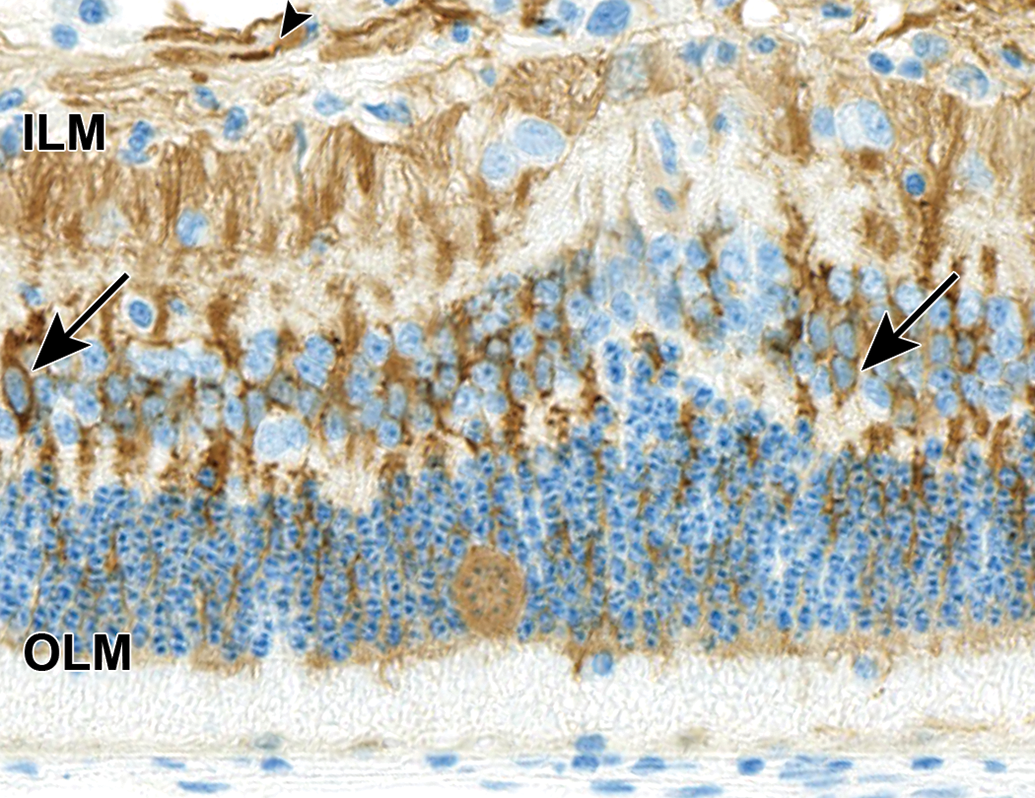
Swollen cell bodies with unique chromatin arrangement in the ONL exhibit cytoplasmic reactivity for glutamine synthetase (GS). Surrounding normal Müller cells are also immunoreactive for GS. The cell bodies (arrows) are in the inner nuclear layer and processes extend from the inner limiting membrane (ILM) to the outer limiting membrane (OLM). In the upper part of image, horizontal GS immunoreactive fibers (arrow head) likely represent astrocytic processes in the nerve fiber layer. NZW Rabbit, Eye, 2 mg/eye 1:1 RabFab-TSG-6-LD, 30 days post intravitreal dose. Antiglutamine synthetase DAB, hematoxylin counterstain. NZW, New Zealand White; ONL, outer nuclear layer; TSG-6, TNF-stimulated gene-6.
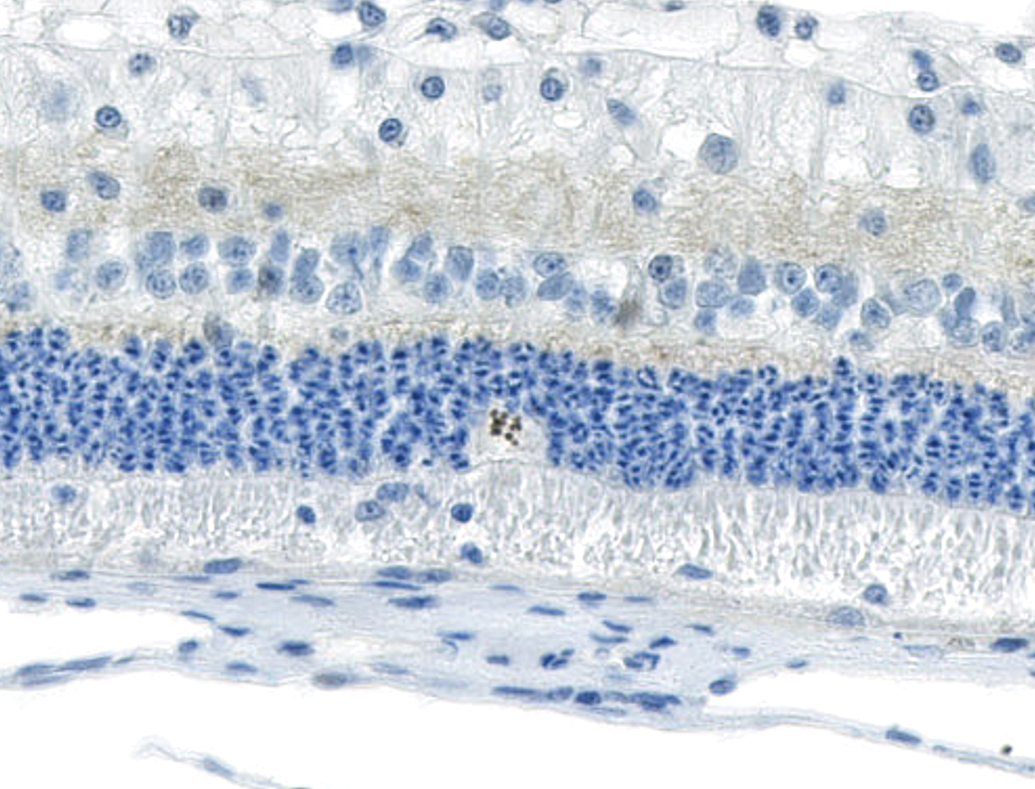
Swollen cell bodies with unique chromatin arrangement in the outer nuclear layer exhibit nuclear reactivity for Ki67, a marker of cell proliferation. NZW Rabbit, Eye, 2 mg/eye 1:1 RabFab-TSG-6-LD, 30 days post intravitreal dose. Anti-Ki67 DAB, hematoxylin counterstain. NZW, New Zealand White; TSG-6, TNF-stimulated gene-6.
The timing and severity of the in-life cell infiltrates were reminiscent of immune-mediated inflammation directed against the test item. Analysis of ADA and TK data from both study 1 and 2 confirmed very high serum ADA titers from day 8 in animals administered 1:1 or AF488 labeled-1:2 RabFab-TSG-6-LD (Figure 11). However, in the authors’ experience, similarly high ADA titers have been noted in rabbits administered biologics by intravitreal injection without similar inflammation and/or retinal and lens lesions. There was a ∼3- to 4-fold increase in the vitreal half-life of AF488 labeled-1:2 RabFab-TSG-6-LD compared to AF488 labeled-RabFab observed in study 2, with no apparent dependence on dose over the range evaluated (Figure 12). However, the 28-day study duration was not long enough for reliable determination of PK parameters, with approximately 40% of the administered 1:2 RabFab-TSG-6-LD estimated to be remaining in vitreous at the end of the study (based on comparison of AUClast to the total administered dose). Toxicokinetic analysis of serum concentration-time data from studies 1 and 2 suggest exposure was maintained for the duration of the study, and observed terminal half-lives in serum are consistent with prolonged clearance of 1:1 and 1:2 RabRab-TSG-6-LD from vitreous humor relative to Fab alone (data not shown).
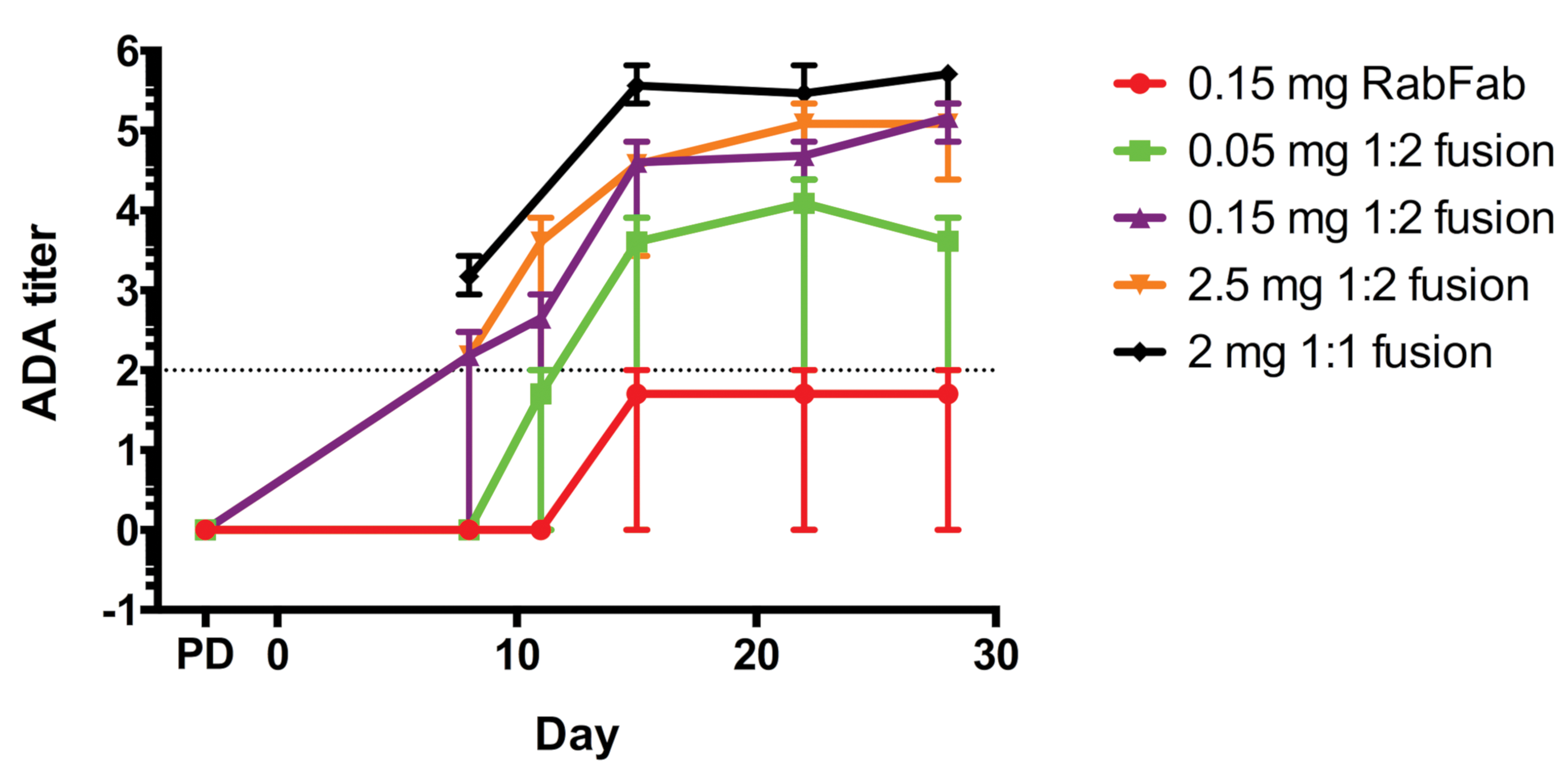
Study 1 and 2. Serum anti-RabFab antibody titers (ADA) following administration of AF488 labeled-RabFab, AF488 labeled-1:2 RabFab-TSG-6-LD, or 1:1 RabFab-TSG-6-LD to NZW rabbits. The ADA titer is the log of the dilution at which the sample optical density (OD) crosses the cut point OD. The minimum dilution for this assay is 1:100, thus the minimum individual titer is 2 (indicated by dotted line) and negative ADA animals that are below the cut point at the minimal dilution (<2 log ADA titer) are denoted as 0. Positive ADA titers were noted from day 8 postdose in animals administered RabFab-TSG-6-LD fusions with titers >4 in most animals from day 15 post dose. In contrast, animals administered RabFab had ADA titers at or below the cut point. Data points represent mean values and error bars the range. ADA, antidrug antibody; NZW, New Zealand White; TSG-6, TNF-stimulated gene-6.
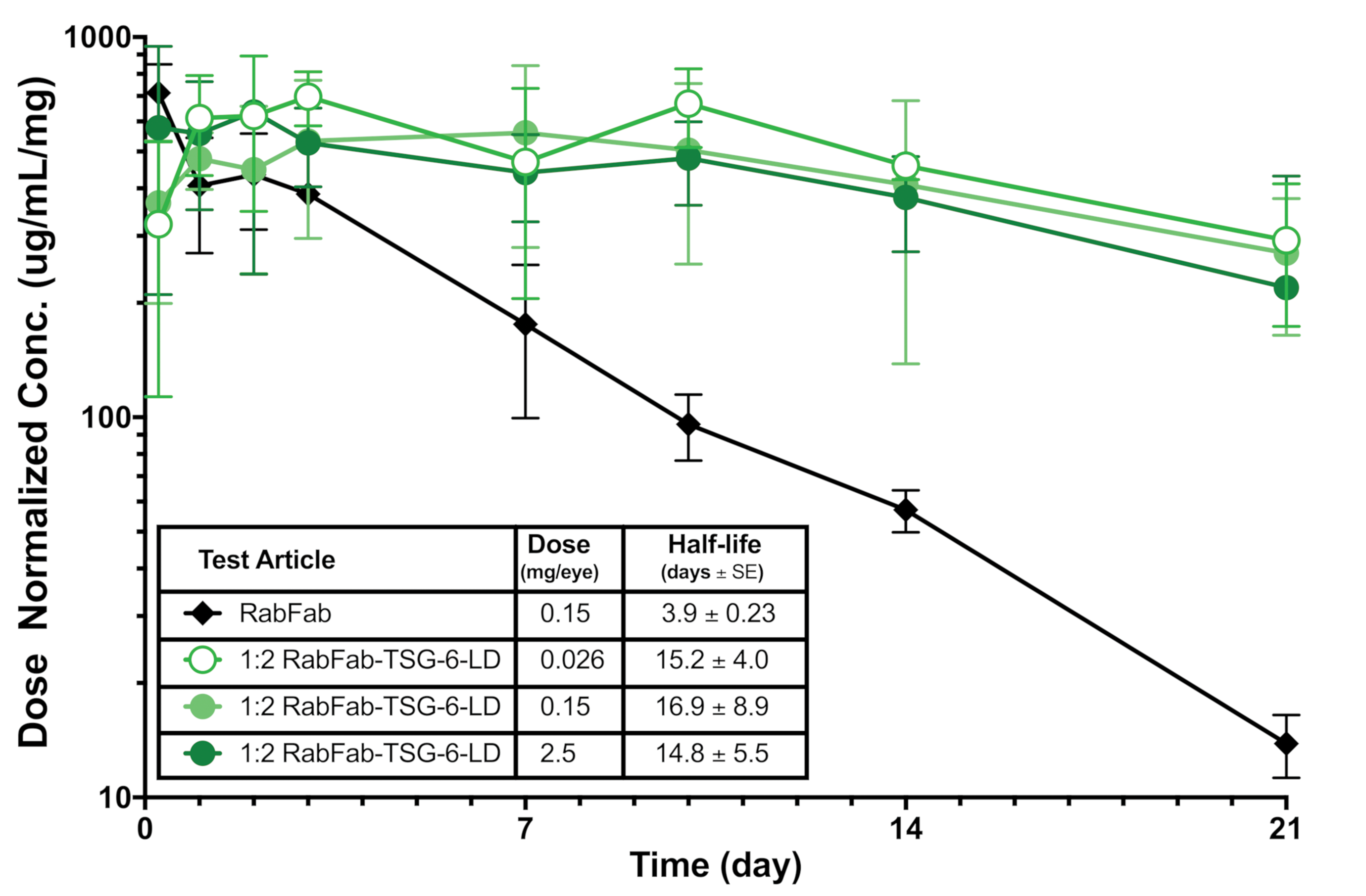
Measured vitreal concentration of RabFab versus 1:2 RabFab-TSG-6-LD. The concentration of 1:2 RabFab-TSG-6-LD is maintained for longer than that of RabFab. Note that approximately 40% of the administered doses of 1:2 RabFab-TSG-6-LD have not yet been eliminated during the PK sampling period, which is too short for reliable determination of PK parameters for this longer acting test item. PK, pharmacokinetic; TSG-6, TNF-stimulated gene-6.
Retinal and Lenticular Degeneration Within 4 Days of Administration of Free TSG-6-LD
Based on studies 1 and 2, there were several outstanding questions relating to the pathogenesis of the lesions noted with RabFab-TSG-6-LD fusions. In particular, the rapid and marked serum anti-RabFab ADA response noted in animals administered the fusion peptides was in contrast to the minimal or lack of ADA response in animals administered RabFab in study 2 and expected from historical data. 8 This raised the possibility that these lesions were an aberrant immune mediated-response rather than a direct effect of binding HA in the rabbit eye; however, it should be noted that retinal degeneration is not typically seen as a manifestation of an antitest item immune reaction. Furthermore, the role of the AF-488, higher binding affinity to HA of 1:2 versus 1:1 RabFab-TSG-6-LD, and inferred increased ocular exposure in the more severe lesions noted in study 2 was unknown.
Study 3 was designed to more clearly understand the role for TSG-6-LD in the pathogenesis of the lens and retinal degeneration. Male NZW rabbits were dosed either with the maximum feasible dose of 1:1 RabFab-TSG-6-LD or the approximate molar equivalent of free TSG-6-LD. Additionally, a cohort of animals in each group were euthanized 4 days postdose, which was expected to be an insufficient amount of time to generate an adaptive immune response against the test item.
Animals administered free TSG-6-LD developed bilateral, posterior lens opacities that covered the majority of the lens by the time of the ophthalmic examination on day 3. Additionally, one animal, scheduled for necropsy on day 4, had retinal vascular attenuation noted on fundoscopic examination, consistent with retinal degeneration by day 3. These findings correlated with microscopic evidence of lens fiber swelling and degeneration (Figure 13) and multifocal retinal degeneration in the affected animals (Figure 14), respectively. Retinal degeneration was variably severe in individual eyes and appeared to affect all layers of the retina. The distribution of the lesions, while multifocal, was extensive in 2 eyes administered free TSG-6-LD, affecting approximately 30% to 40% of the retina in section including both peripheral and more posterior regions. Animals scheduled for necropsy on day 30 had continuing worsening of lens opacities that hampered assessment of the posterior chamber. However, moderate to marked vitreal and aqueous cells and flare were present in at least one animal by day 8. Due to the severity of findings, these animals were euthanized ahead of schedule on day 12 and 17 and did not receive a second dose of free TSG-6-LD.
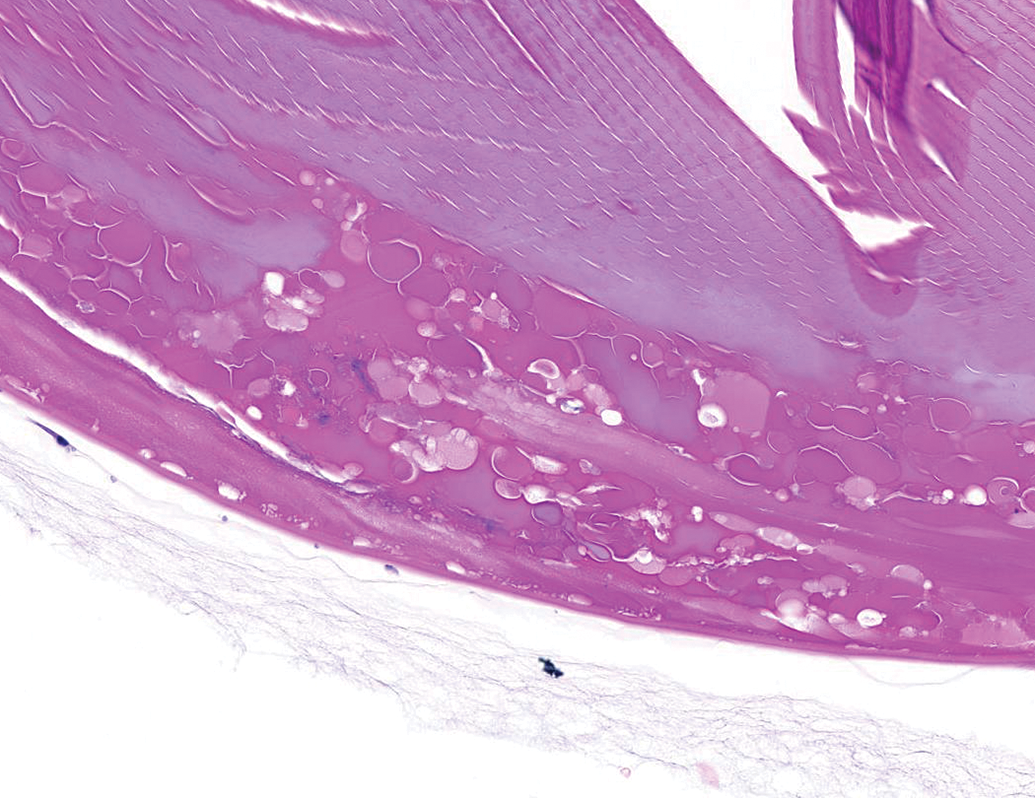
Posterior lens degeneration characterized by swollen lens fibers, liquefaction, and clefting. NZW Rabbit, Eye, 0.5 mg/eye free TSG-6-LD, 4 days post intravitreal dose. Hematoxylin and eosin (H&E). NZW, New Zealand White; TSG-6, TNF-stimulated gene-6.
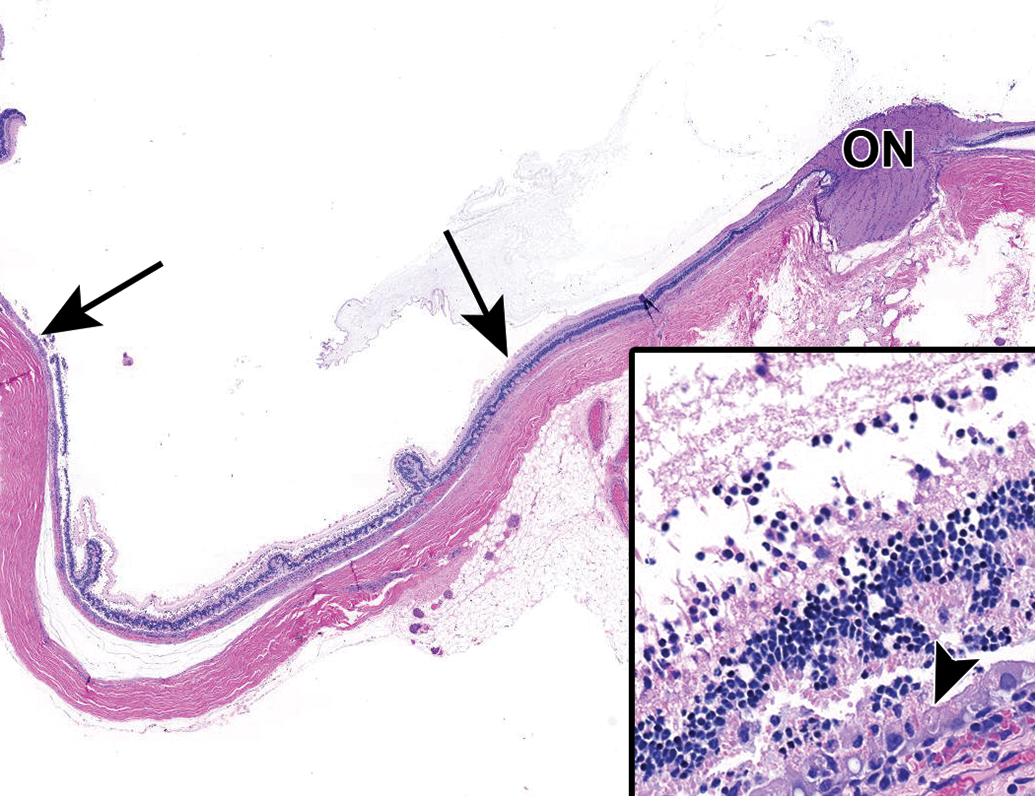
Extensive retinal degeneration (between arrows) affecting all layers of the retina. Inset: Acute necrosis of all layers of the retina. Note hypertrophy and vacuolation of the retinal pigment epithelium (arrowhead) and necrotic debris and inflammatory cells replacing the photoreceptor layer. NZW Rabbit, Eye, 0.5 mg/eye free TSG-6-LD, 4 days post intravitreal dose. Hematoxylin and eosin (H&E). ON indicates optic nerve head. NZW, New Zealand White; TSG-6, TNF-stimulated gene-6.
In general, animals that received 1:1 RabFab-TSG-6-LD had less severe findings than those that were administered free TSG-6-LD (Table 6). Lens opacities were present in individual animals that received 1:1 RabFab-TSG-6-LD but were punctate in nature and a correlate was not identified in microscopic sections. There was no clinical evidence of retinal degeneration, but microscopic evidence of minimal to mild outer retinal degeneration was present in individual eyes. Notably, hypertrophic Müller cells were not present in this study. Similar moderate to severe vitreous and aqueous cells were present from day 8 onward. The animals were euthanized on day 4 and day 17.
Microscopic Findings in Study 3.a
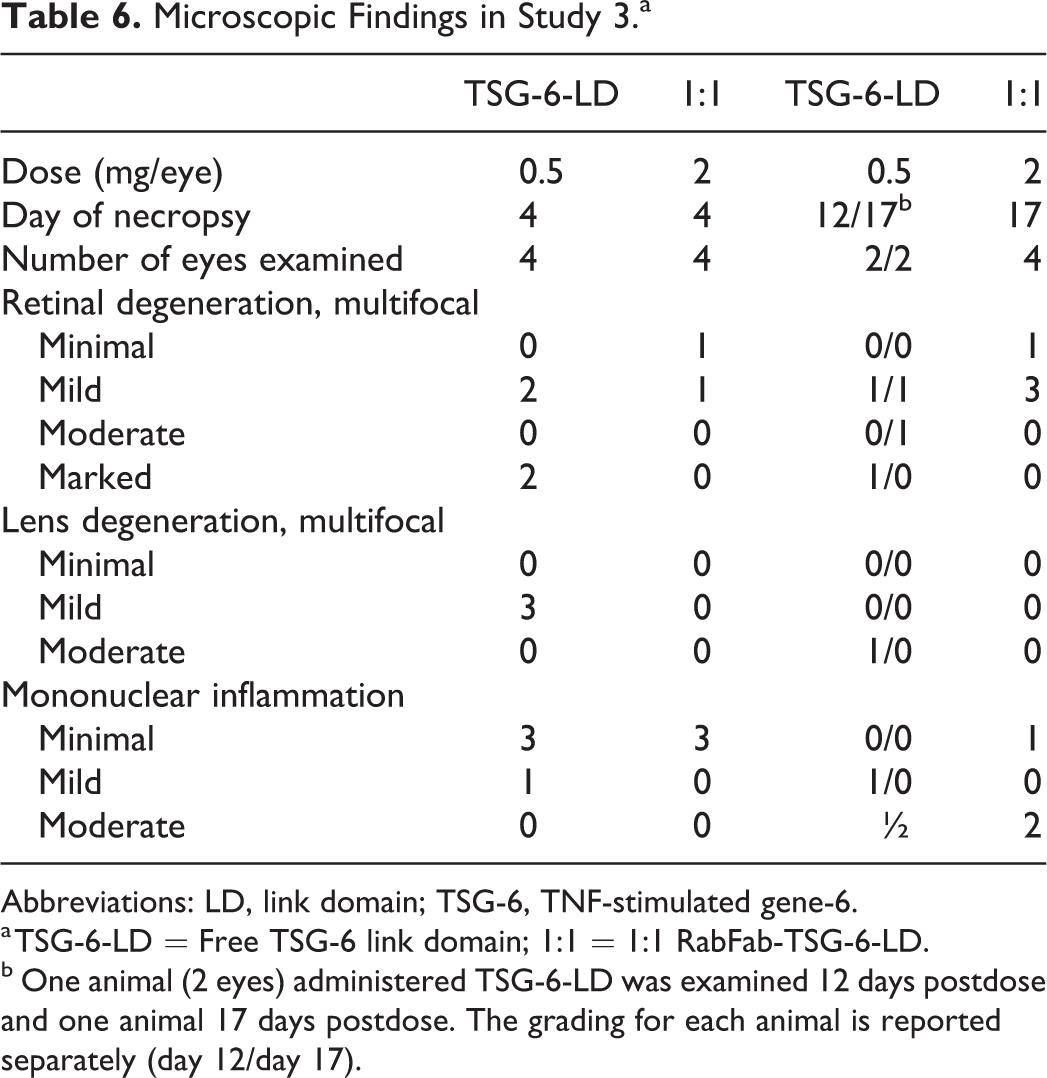
Abbreviations: LD, link domain; TSG-6, TNF-stimulated gene-6.
a TSG-6-LD = Free TSG-6 link domain; 1:1 = 1:1 RabFab-TSG-6-LD.
b One animal (2 eyes) administered TSG-6-LD was examined 12 days postdose and one animal 17 days postdose. The grading for each animal is reported separately (day 12/day 17).
Assessment of anti-RabFab responses was complicated by the presence of values above the cut point for 1/8 animals prior to dosing (1 animal administered 1:1 RabFab-TSG-6-LD). However, following administration of the test items, 3/4 animals administered 1:1 RabFab-TSG-6-LD had emergent or increasing serum ADA titers: 1 animal euthanized on day 4 and both animals euthanized on day 17. In contrast, only animal 3 (necropsy day 17) administered free TSG-6-LD had an elevated serum ADA titer compared to predose on days 15 and 17 (Table 7).
Serum Antidrug Antibodies (ADA) in Study 3.a

Abbreviations: ADA, antidrug antibody; LD, link domain; PD, predose; TSG-6, TNF-stimulated gene-6.
a Data are presented as ADA titers for each sample, which is the log of the dilution at which the sample optical density (OD) crosses the cut point OD. The minimum dilution for this assay is 1:50. A log titer of <1.7 means it is below the cut point at the minimum dilution, and therefore, the animal is ADA negative. Only 1 of 4 animals administered free TSG-6-LD developed anti-TSG-6-LD antibodies and not until day 15, after the development of notable in-life and microscopic lesions. All animals administered RabFab-TSG-6-LD developed anti-RabFab-TSG-6-LD antibodies, within 15 days. Animal 5 had evidence of anti-RabFab-TSG-6-LD antibodies prior to dosing and titers did not rise between the predose assessment and the last assessment on day 4.
Discussion
Fab-TSG-6-LD fusions were being investigated as potential LAD therapeutics for posterior segment disease such as AMD. Although the data presented suggests a significant increase in vitreal retention, retinal degeneration and/ or lens degeneration were observed following a single intravitreal dose of RabFab-TSG-6-LD to NZW rabbits, both of which are considered adverse and are expected to be irreversible. The subsequent investigative study demonstrated that these findings were related to the TSG-6-LD moiety and were not a consequence of an unexpectedly robust immune response in rabbits administered RabFab-TSG-6-LD. These findings resulted in the discontinuation of development of Fab-TSG-6-LD fusions at Genentech.
The use of the rabbit as a toxicology species for humanized biologic therapeutics administered via intravitreal injection is not advised due to severe antidrug immune responses. 14 However, the rabbit has several advantages over rodent models due to the larger size of the globe and vitreous chamber, which allow for easier intravitreal injection and intraocular PK that more closely resemble those of human. 15 In the studies described herein, a species matched Fab, RabFab, was utilized to minimize immune responses in the rabbit. This allowed early assessment of PK and safety of TSG-6-LD fusion LADs, while reducing the need for higher species such as monkey or mini-pig. A drawback of the rabbit as a model for this particular LAD is that the vitreous gel of the rabbit contains only 14 to 52 μg/mL of HA compared to 100 to 400 μg/mL in humans. 5 Therefore, the retention of Fab-TSG-6-LD fusion in the vitreous chamber of rabbits is predicted to be less than in humans. Given the octasaccharide minimal binding site on HA for TSG-6-LD 5 and rabbit vitreous volume (∼2 mLs), an upper limit of 30 µM for the molar vitreous binding site concentration, at 50 µg/mL HA concentration, can be calculated. Since it is unclear if all binding sites in the vitreous humor are freely accessible, the initial vitreous concentrations in study 3 (∼20 µM for TSG-6-LD and ∼17 µM for 1:1 RabFab-TSG-6-LD) could have approached this limit. This may have allowed excessive distribution to the retina and lens. In our study, AF-488 labeled 1:2 RabFab-TSG-6-LD was not visualized in the retina at 8 days postdose; however, this is likely due to fluorescent microscopy being inadequately sensitive to detect the labeled test item. We propose that the retinal toxicities described required direct interaction between TSG-6-LD and the retina. Free TSG-6-LD is approximately 5-fold smaller than 1:1 RabFab-TSG-6-LD, on the basis of their relative molecular weights, and thus is expected to diffuse more quickly and distribute more rapidly throughout the eye. This may explain the more rapid onset of severe clinical signs in animals administered free TSG-6-LD versus 1:1 RabFab-TSG-6-LD. It is tempting to hypothesize that the higher HA content of the human vitreous would limit distribution and damage to the retina and lens. However, it should be noted that the overall composition of the human vitreous as well as its homogeneity vary with age. Specifically, the vitreous liquefies and while total HA remains constant above the age of 20, concentration of HA in the liquid phase mildly increases. 16,17
TSG-6 is a secreted protein composed of Link and complement component Clr/Cls, Uegf, and bone morphogenetic protein 1 (CUB) modules. It is constitutively expressed in several tissues including astrocytes of the central nervous system, epidermal cells of the skin, islets cells in the pancreas, and in the granules of neutrophils and mast cells. TSG-6 is also induced in inflammatory processes in several tissues, leukocytes, and mesenchymal stem/stromal cells. 5 Expression in the normal eye has not been assessed. In general, TSG-6 is thought to be anti-inflammatory via incompletely elucidated mechanisms that may include binding of pro-inflammatory HA fragments, binding of chemokines including CXCL8, effects on leukocyte migration and rolling utilizing CD44 receptors, and inducing macrophage polarization to the anti-inflammatory M2 phenotype. 5 Intravitreally administered recombinant TSG-6 has been suggested to be protective in a mouse model of retinal degeneration. 18
In the studies described in this article, the link domain of TSG-6 alone (TSG-6-LD) was investigated purely for its HA-binding properties. However, TSG-6-LD is promiscuous and binds not only HA, but several other glycosaminoglycans (GAG), matrix proteins, chemokines, and growth factors. 6 Many of these GAG binding substrates, including HA, are present throughout the eye including the retina and in the lens capsule of mammals. 19 –22 The consequences of TSG-6 binding are varied, context-specific, and include crosslinking and condensation of GAGs and modulation of HA interactions with its receptors CD44 and lymphatic vessel endothelial hyaluronan receptor-1 (LYVE-1). CD44 is a ubiquitous cell surface glycoprotein that is involved in cell adhesion, migration, and polarization. In the retina, CD44 is expressed on Müller cells, amacrine cells and in astrocytes. 23 –25 The LYVE-1 is another membrane glycoprotein that is expressed on lymphatic endothelial cells. The TSG-6-LD has a higher binding affinity for HA than CD44 and so may competitively inhibit these interactions. However, there is also evidence that conformational changes in HA following TSG-6 binding may increase HA-CD44 interactions. 26,27 Based on our current data set, it is impossible to ascertain if the lesions noted are a result of TSG-6-LD interactions with HA or another binding partner. However, the potential to affect HA interactions with CD44 is particularly intriguing: these interactions are involved in attachment of the neuroretina to the interphotoreceptor matrix, and CD44 signaling is involved in the regulation of cell proliferation, migration, differentiation, and apoptosis. 5,19,23,24,28,29
Müller cell hypertrophy, nuclear migration to the ONL, and proliferation are an expected response to retinal injury and have been followed in rabbits with induced retinal detachment. 30 The striking Müller cells noted in study 1 and 2 were likely secondary to damage to the photoreceptors and/ or retinal detachment, which may have been a direct consequence of disruption of normal CD44–HA interactions. The process of Müller cell hyperplasia with nuclear migration was demonstrated to peak approximately 3 days following experimental retinal detachment in rabbits by Lewis et al. 30 Nuclear migration and hypertrophy are transient processes and may have been missed in study 3. In study 3, retinal degeneration occurred in all layers of some animals administered free TSG-6 by day 4 suggesting a direct toxic effect to the retina, perhaps in addition to outer retinal degeneration subsequent to detachment. Müller cell hyperplasia has also been noted by the author in rabbit eyes with retinal detachment secondary to marked inflammatory processes, suggesting this phenomenon is not specific to TSG-6-LD-induced lesions. Müller cell hyperplasia following retinal injury has been proposed as an attempt at neuroprotection and neuroregeneration but is also known to lead to subretinal membranes that prevent reattachment of the neuroretina. 13,30,31
The robust immune response noted in rabbits administered RabFab-TSG-6-LD and associated inflammation was likely a consequence of the modification of the RabFab by the fusion of TSG-6-LD. An immune reaction to released retinal or lens proteins is a possible differential but was considered less likely as the severity of inflammation was not clearly associated with the severity of retinal and lens lesions. Furthermore, minor damage to the lens was not associated with significant inflammation in the rabbit. 32 Although it is accepted that immunogenicity in nonclinical toxicology species is not predictive of similar responses in patients, it is concerning that any species matched Fab will become inherently more “foreign” to the immune system of that species following addition of a peptide domain. 33,34 However, given the propensity of rabbits to generate immune responses, it is possible that this is a rabbit-specific phenomenon. 14 Prediction of immunogenicity in patients is currently difficult to achieve and it remains to be seen if modified biologics, such as Fab-HA binder fusions, will prove to be more immunogenic than their unmodified counterparts.
The potential risk of the TSG-6-LD-induced lesions in the rabbit eye translating to the clinic resulted in discontinuation of Fab-TSG-6-LD fusion development. However, as discussed, there are several unanswered questions relating to the precise mechanism of toxicity and the appropriateness of the rabbit as a model for these toxicities. Of note, another company has pursued similar Fab-TSG-6 fusions into the clinic, but development was halted due to undisclosed safety issues in Ph1. 7 The relevance of the findings in rabbits following intravitreal administration of RabFab-TSG-6-LD to other HA-binding peptide-Fab fusions, which remain intriguing LAD candidates, is uncertain and will require further investigation.
Footnotes
Acknowledgments
Michelle McDowell and Cynthia McCaughey are thanked for their assistance with animal studies. Charles Havnar is thanked for tissue sectioning. Neeraj Sharma, Linda Rangell, and Meredith Sagolla are thanked for immunohistochemistry and fluorescence microscopy support. Pamela Chan is thanked for her PK and ADA assay support. Ryan Abraham and Phil Hass are thanked for assistance with test article purification.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
