Abstract
In nonrodent toxicity studies that are usually conducted in cynomolgus monkeys or beagle dogs, the added value of examining all tissues from all dose groups (current practice) versus all tissues in only control and high-dose groups and target tissues in intermediate-dose groups by default, is a subject of debate. A previous retrospective review of 325 nonrodent toxicity studies that included a limited number of biotherapeutics suggested that the evaluation of all tissues from all groups was not justified as a routine practice and recommended the examination of all tissues in control and high-dose groups and only target tissues in intermediate-dose groups. In contrast, the present retrospective review which examined 213 nonrodent studies (212 in cynomolgus monkeys and 1 in dog) from 4 multinational pharmaceutical companies (Bristol-Myers Squibb, Novartis, Pfizer Inc, and Roche) conducted only with biotherapeutics showed that restricting the microscopic examination in intermediate-dose groups to target tissues has the potential to miss findings in 6.6% of studies, possibly impacting the overall study interpretation and conclusion. In conclusion and in the opinion of the authors, all tissues from all dose groups should be examined in toxicity studies with biotherapeutics conducted in nonrodent species.
Introduction
Pharmaceutical companies are constantly pressured to decrease costs, increase research and development (R&D) productivity, and shorten cycle times in their efforts to develop new therapies/medicines. In nonclinical safety studies, postmortem microscopic examination of tissues is an important end point in the identification of test article-related findings with respect to dose. Many pharmaceutical companies examine all protocol tissues in all dose groups in nonrodent toxicity studies for the following reasons: the European Medicines Agency (EMA) guideline for repeated-dose toxicity studies encourages that histopathology “should be conducted in all animals at all dose levels” in nonrodent studies 1 ; bias due to the small group size used in nonrodent studies is exacerbated by interanimal variability resulting from the outbred status of nonhuman primates and dog species, the different geographical origins, and broad age and weight ranges dictated by the animal supply of non-human primates. However, the microscopic examination of tissues is often the rate-limiting step in generating the study report, and in contrast to the EMA guideline, the International Conference for Harmonization (ICH) M3(R2) guideline: Guidance on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals 2 and ICH S6 (R1) Guideline: Preclinical safety evaluation of biotechnology-derived pharmaceuticals 3 do not provide a specific recommendation to conduct a histopathologic evaluation of all tissues from intermediate-dose groups in nonrodent studies. Consequently, the value of examining all tissues from all animals from all dose groups has been the subject of debate among toxicologic pathologists.
To investigate the impact of evaluating all organs in all dose groups versus all organs in just control and high-dose animals and target organs in intermediate-dose groups on risk assessment, 3 pharmaceutical companies (Abbott Laboratories, Eli Lilly, and Pfizer Inc.) performed a retrospective review of anatomic pathology data from nonrodent toxicity studies and published their results in a Regulatory Forum Opinion Piece in 2012. 4 Results of this review identified only 1 of 325 nonrodent studies in which test article-related findings were observed in an intermediate-dose group but not in the high-dose group. The findings demonstrated no impact on the overall risk assessment since the no-observed-adverse-effect-level (NOAEL), no-observed-effect-level (NOEL), and overall study interpretations were not affected, suggesting that the evaluation of all tissues from all animals might not be justified as a routine practice. However, the only study with test article-related findings restricted to the intermediate-dose group involved nonhuman primates receiving a biotherapeutic test article. The number of studies with biotherapeutics in this retrospective review was limited while most of the studies involved small molecules.
Due to the limited number of biotherapeutics in the retrospective review by Weddle et al, 4 our objective was to perform a similar retrospective review focusing only on studies with biotherapeutic entities and to determine the incidence of test article-related findings lacking a dose relationship in these studies. Our review evaluated the incidence of test article-related findings that were: (1) present in the intermediate-dose groups and absent in the high-dose group, and (2) present in the high- and low-dose groups and absent in the intermediate-dose group.
Materials and Methods
A total of 213 preclinical toxicity studies conducted by 4 different companies (Bristol-Myers Squibb, Novartis, Pfizer Inc, and Roche) were included in this retrospective review. All the studies involved biotherapeutic entities and, except for 1 dog study, all involved nonhuman primates (cynomolgus monkeys). Only studies with at least 1 control group and 2 test article-dosed groups were included in this retrospective analysis, which involved a review of pathology reports and microscopic diagnostic tabular data from each study. Three of the 4 companies participating in the study used the same comprehensive tissue list for Good Laboratory Practice (GLP) and exploratory (non-GLP) studies, and the fourth company only used a limited list of tissues for exploratory/dose range finding studies. Consequently, 14 (6.6%) of the 213 studies had a limited tissue list, but the majority of the studies (199 studies, 93.4%) had a comprehensive list of tissues that comprised adrenal glands, aorta, bone and bone marrow (only histopathology evaluation), brain, cervix, epididymis, esophagus, eyes, gallbladder, gut-associated lymphoid tissue, heart, kidneys, large intestine (cecum and colon), liver, lung, lymph nodes (subcutaneous/peripheral and mesenteric), mammary gland, optic nerves, ovaries, pancreas, parathyroid glands, peripheral nerves, pituitary gland, prostate gland, salivary glands, sciatic nerve, seminal vesicles, skeletal muscle, skin, small intestine (duodenum, jejunum, and ileum), spinal cord, spleen, stomach, testes, thymus, thyroid gland, tongue, tonsil, trachea, urinary bladder, ureter, uterus, vagina, and injection site(s). The biotherapeutics evaluated included peptides, cytokines, antibodies (monoclonal, polyclonal, fusion, and/or bispecific), and proteins (recombinant, fusion or PEGylated). The mechanism of the findings was undisclosed in most of studies, except for some studies with PEGylated compounds (usually proteins) characterized by having a polyethylene glycol (PEG) molecule attached, and some studies with antibody-drug conjugates (ADCs). In studies showing more than one finding per tissue attributable to 2 or more different mechanisms (even those undisclosed), the findings were noted separately. An example of this were studies with PEGylated compounds where there might be changes attributed to target engagement as well as accumulation of PEG in vacuoles within mainly phagocytes in the same tissue. 5 Another example was studies with ADCs in which toxicity can be mediated via any of the components of the drug, although most of the toxicity is usually thought to be derived from the payload. 6
Only the data from the animals euthanized at the end of dosing were reviewed. The data from the recovery phase were excluded from review in all the studies examined.
All procedures performed on the animals in these studies were in accordance with regulations and established guidelines and were reviewed and approved by the respective company’s Institutional Animal Care and Use Committee or equivalent.
Results
Overall, 213 studies comprising 212 studies conducted in nonhuman primates (cynomolgus monkeys) and 1 study conducted in dog were reviewed (Table 1). The most common dosing route was intravenous injection (149 studies, 70%), followed by subcutaneous injection (59 studies, 28%), with a few studies using intravitreous injection (4 studies, 2%), and 1 study using intra-articular injection. The study types included GLP and non-GLP studies, and the duration of the studies was either single dose or ranged from 5 days to 52 weeks with most studies being between 4 and 9 weeks. Most of the studies had 3 or 4 animals per group (see Tables 1 and 2).
Nonrodent (Cynomolgus Monkeys and Beagle Dogs) Studies With Biotherapeutics Revieweda by Route and Duration, and Percentage of These Studies With Test Article-Related Findings on Histopathology.b
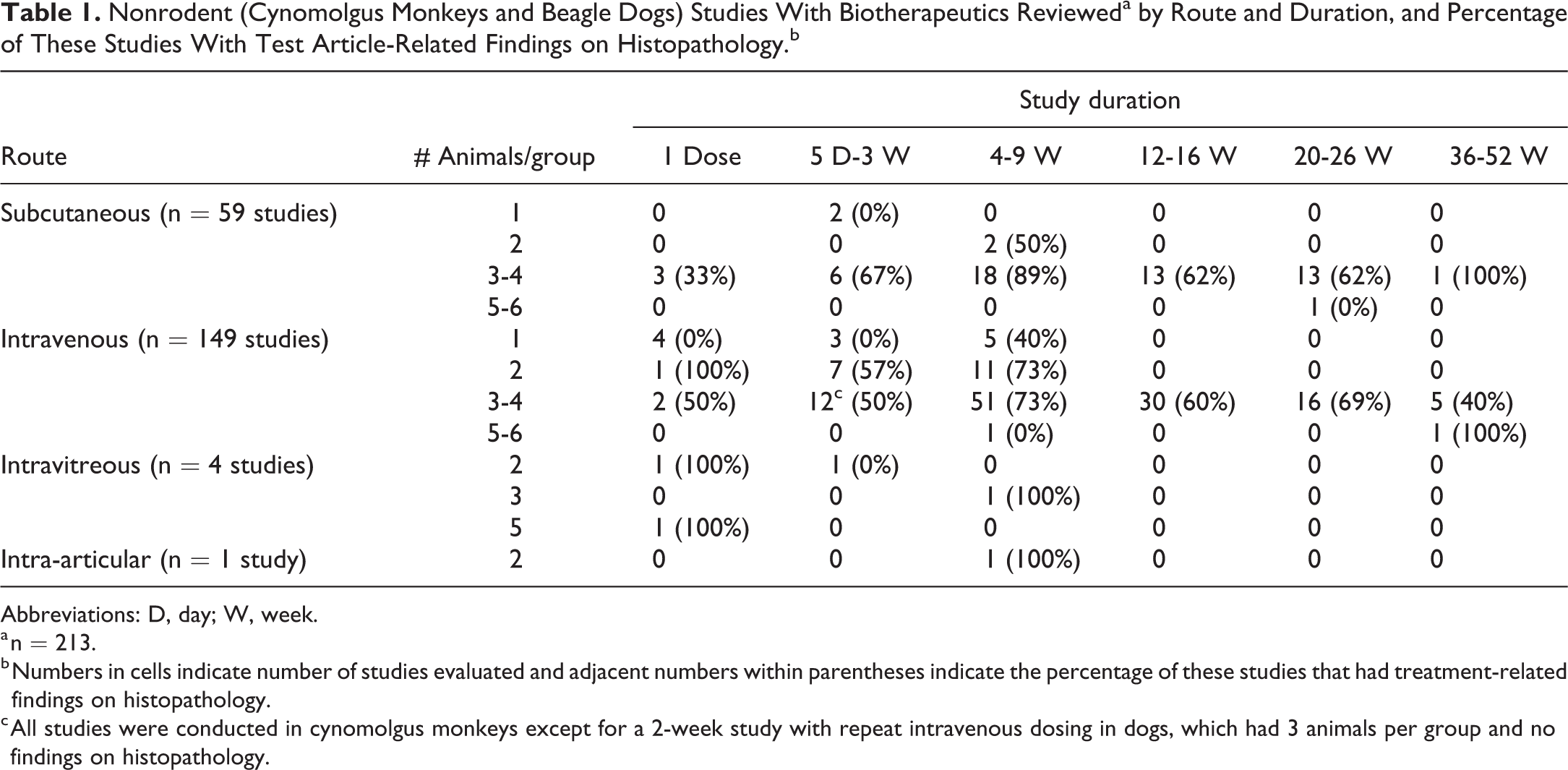
Abbreviations: D, day; W, week.
a n = 213.
b Numbers in cells indicate number of studies evaluated and adjacent numbers within parentheses indicate the percentage of these studies that had treatment-related findings on histopathology.
c All studies were conducted in cynomolgus monkeys except for a 2-week study with repeat intravenous dosing in dogs, which had 3 animals per group and no findings on histopathology.
Number of Nonrodent Studies With Biotherapeutics Revieweda and Incidence of Number of Test Article-Related Findings on Histopathology Per Study.b

a n = 213.
b Numbers in cells indicate number of studies evaluated without findings on histopathology, and number of studies with different number of findings.
Of the 213 studies reviewed, 174 (82%) had test article-related microscopic findings (Table 2). In these studies, there were a total of 601 test article-related microscopic findings (Table 3). There were 14 studies in cynomolgus monkeys where findings lacked a dose relationship (Table 4). Nine of these 14 studies were GLP and 5 were non-GLP studies, although 4 of the 5 non-GLP studies had an extensive tissue list similar to GLP studies. The animal number per group was usually 3 per sex, except in 3 studies that had 4/sex animals per group. In 9 of these 14 studies, the findings were present in an intermediate-dose group, but absent in the high-dose group, and in the remaining 5 studies, the findings were present in the lowest- and highest-dose groups, but absent in one or more intermediate-dose groups. Findings lacking dose relationship were considered adverse in 5 of the 14 studies and set the NOAEL in 1 of these studies. The organs affected in each of the 14 studies varied (eg, brain, spleen, male reproductive organs, gallbladder, lymph nodes, kidneys, etc) without specific organ system prevalence. The duration of the 14 studies varied from single dose to 6-month duration, and the most common route of administration was intravenous injection followed by subcutaneous injection, reflecting the higher prevalence of intravenous injection (70%) as the route of administration in the 213 studies reviewed. Five of these 14 studies involved ADCs, and 1 study involved a PEGylated compound. The findings that lacked a dose relationship in the 5 studies with ADCs were attributed to the cytotoxic payload, and in the study with a PEGylated compound, the findings were attributed to the accumulation of PEG in vacuoles. The remaining 9 studies involved peptides, proteins (including antibodies), and biotherapeutics of an undisclosed nature, where the mechanism responsible for the findings lacking a dose relationship, although undisclosed or unclear, was considered most likely related to the pharmacology of the test article.
Incidence of Test Article-Related Microscopic Findings Per Tissue and Organ System in Nonrodent Studies With Biotherapeutics Reviewed.a,b

Abbreviation: GALT, gut-associated lymphoid tissue.
a n = 213.
b There were 601 test article-related findings in 174 of 213 studies. The numbers within parentheses after each tissue indicate the number of studies with findings in a specific tissue across the 174 studies, although some studies had more than one finding per tissue.
Number of Nonrodent Studies With Biotherapeutics With Nondose-Related Test Article-Related Findings on Histopathology.a

Abbreviation: GLP, Good Laboratory Practice.
a n=14.
b May represent one or more intermediate dose levels between the low and high dose.
c Only 1 study had a limited tissue list, the remaining 4 studies had an extensive tissue list similar to GLP studies.
Discussion
In this retrospective study, we reviewed the data from 213 studies involving biotherapeutics in nonrodent species where there was at least 1 control group and 2 dosed groups. The majority of the studies were conducted in cynomolgus monkeys (212 studies), except for 1 study performed in dogs. In contrast to the retrospective study by Weddle et al, 4 where there was only 1 of 352 studies with test article-related microscopic findings lacking a dose relationship, there were 14 (6.6%) of 213 studies in our study that showed test article-related histopathological findings lacking a dose relationship. There was no organ system prevalence or correlation with study duration identified in these 14 studies.
These findings indicate that the examination of only the control and high-dose groups and target organs in studies with biotherapeutic entities could potentially misrepresent or mislead crucial safety findings that could impact the NOEL and NOAEL in a study. The data support the conclusion and recommendation that all tissues from all dose groups should be examined in toxicity studies with biotherapeutics conducted in nonrodent species. This conclusion should be relevant in studies with therapeutics performed in nonhuman primates, but the results are likely valid for other large animal species like dogs and minipigs as well.
The differences in the incidence of nondose-related findings in this analysis versus that of Weddle et al 4 was likely due to this review focusing solely on biotherapeutics, while that of Weddle et al 4 focused mainly on small molecules. The reasons for the higher incidence of test article-related findings lacking a dose relationship in biotherapeutics versus small molecules were not fully explored in this review.
In the present review, immunogenicity was determined to potentially have played a role in 6 of 14 studies lacking dose relationship. This is commonly attributed to the fact that immunogenicity leads to the generation of antidrug antibodies that bind to the active site or alter the tertiary structure of a protein via steric effects 7,8 and thereby reduce the biological activity of the test article (ie, neutralizing antibodies). Antidrug antibodies can also be non-neutralizing but alter the pharmacokinetics by either increasing or decreasing the clearance of the protein therapeutic and therefore alter the pharmacodynamic effect. 9 Lastly, antidrug antibodies can result in inflammatory lesions 9 or hypersensitivity reactions 10 as a result of immune complex deposition.
Notwithstanding the causes for the absence of a dose relationship in 6.6% of the studies with biotherapeutics examined in this review, the results indicate that the examination of all protocol tissues from all dose groups in such studies is important due to the potential to miss test article-related findings and affect the NOAEL/NOEL and consequently impact the overall study interpretation as well as safety risk assessment.
Footnotes
Authors’ Note
Cecilia Parrula performed the work while working at Drug Safety Evaluation, Bristol-Myers Squibb, New Brunswick, NJ, USA. This is an opinion article submitted to the Toxicologic Pathology Forum. It represents the views of the author(s). It does not constitute an official position of the Society of Toxicologic Pathology, British Society of Toxicological Pathology, or European Society of Toxicologic Pathology, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
