Abstract
Identification of test article–related microscopic findings in ocular toxicology studies requires a working knowledge of the artifacts and procedure-related or background findings commonly encountered in such studies. The objective of this article is to provide a mini-atlas of the artifacts and procedure-related or spontaneous background findings commonly observed in ocular tissues from animals in toxicology studies of ocular drug candidates. Artifacts in the eye are often related to collection or fixation procedures and include swelling and vacuolation of lens fibers, separation of the neuroretina from the retinal pigment epithelium (RPE), and vacuolation of the optic nerve. Common in-life procedure-related findings include intravitreal injection needle tracks in the sclera and ciliary body pars plana and foci of RPE hypertrophy and/or hyperpigmentation at subretinal injection sites. Common background findings include corneal mineralization, uveal mononuclear cell infiltrates, and peripheral displacement of photoreceptor nuclei in the retina. A few uncommon spontaneous background findings that may be confused with test article–related findings, such as bilateral optic atrophy in macaques, are also included.
Introduction
In all toxicology studies, the differentiation of microscopic findings induced by a test article from the spontaneous background findings that may occur in the tissues of a given species is critical. In ocular toxicology studies, additional potentially confounding factors include findings induced by the administration procedure itself or artifacts associated with the collection or fixation of the eye.
The objective of this article is to provide a mini-atlas of the artifacts and the procedure-related and background findings commonly encountered in ocular tissues from animals in toxicology studies of ocular drug candidates. We also include some uncommon background, species-specific findings that may be confused with test article–related findings if the study pathologist is unaware that these may occur in control animals.
Materials and Methods
Representative microscopic artifacts and background and procedure-related findings in ocular tissues were collected primarily from control animals in toxicity studies of (predominantly ocular) drug candidates. A few images were captured from the eyes of treated animals that lacked test article–related findings. The artifacts and findings presented were selected based on the frequency with which they occur (in the authors’ experience) and/or their resemblance to potential test article–related findings.
All animal procedures were performed in AAALAC International-accredited facilities following applicable animal welfare regulations. The local Institutional Animal Care and Use Committee approved all protocols. Following euthanasia, eyes with attached optic nerve were generally collected and fixed in modified Davidson’s fixative (5% glacial acetic acid, 15% ethanol, 30% [37%-40%] formaldehyde, and 50% water) or Davidson’s fixative (10% glacial acetic acid, 35% ethanol, 2% [37%-40%] formaldehyde, and 53% water). Although fixation protocols varied among studies, for most studies, the eyes were immersed in modified Davidson’s fixative (48-96 hours) or less commonly Davidson’s fixative (24-48 hours), then transferred to 10% neutral-buffered formalin or 70% ethanol (only following Davidson’s fixative) for up to 1 week prior to trimming and embedding. When vitreous was collected prior to fixation, it was replaced with an approximately equal volume of fixative; when vitreous was not collected, eyes were generally not injected or incised prior to fixation. Eyes of cynomolgus monkeys were usually trimmed horizontally in order to generate at least one section through the optic disc and fovea/macula, and eyes of all other species were usually trimmed sagittally in order to generate at least one section through the optic disc. Tissues other than the eye and optic nerve were fixed in 10% neutral-buffered formalin. Fixed tissues were embedded in paraffin, sectioned at 4 to 5 µm, and stained with hematoxylin and eosin.
Results
Artifacts and findings illustrated and/or discussed below are summarized in Table 1.
Artifacts and Procedure-Related or Spontaneous Background Findings in Ocular Tissues.
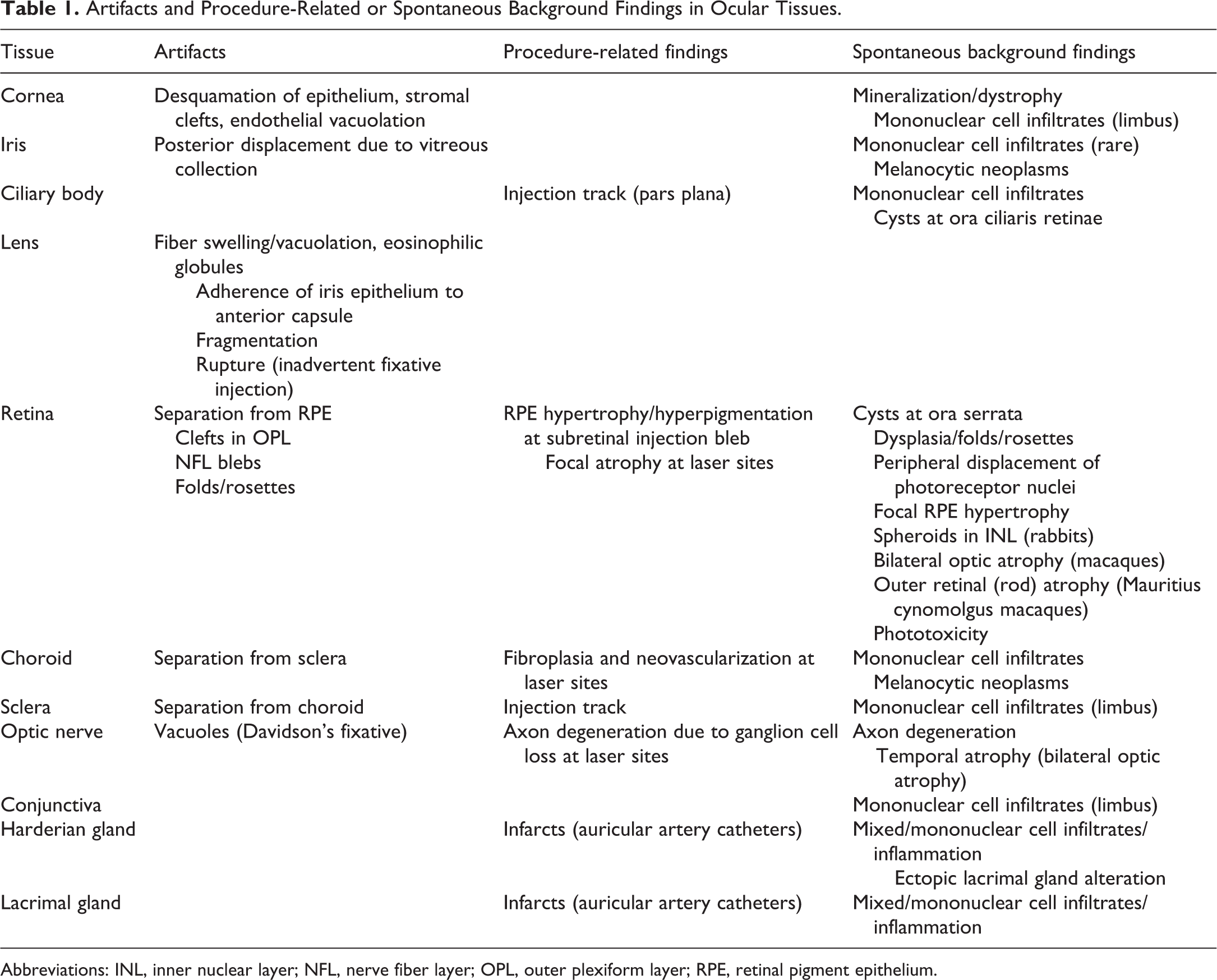
Abbreviations: INL, inner nuclear layer; NFL, nerve fiber layer; OPL, outer plexiform layer; RPE, retinal pigment epithelium.
Artifacts
Artifacts in the eye may be related to collection, fixation, trimming, and/or sectioning/mounting. Most toxicity studies utilize immersion in Davidson’s or modified Davidson’s fixatives for ocular fixation. 1 Each of these fixatives may produce artifacts in ocular tissues, some of which are species-dependent. Prolonged fixation times (longer than 48 hours for Davidson’s fixative or 96 hours for modified Davidson’s fixative) may exacerbate these artifacts. In the rabbit cornea, and less commonly in other species, modified Davidson’s fixation may result in desquamation and loss of the superficial epithelium. Desquamation may also result from touching the cornea at collection or using dull microtome blades. In all commonly utilized species (rabbit, monkey, dog, minipig, and rodents), fixation in Davidson’s or modified Davidson’s is associated with clefts in the stroma and cytoplasmic vacuolation in the endothelium (Figure 1). Stromal clefts may also result from prolonged floating of sections in the water bath. Modified Davidson’s fixation is frequently associated with lens artifacts that may resemble antemortem lens fiber degeneration; these are especially common in cynomolgus monkeys and include swelling and pallor, vacuolation, or eosinophilic globule formation, predominantly within subcapsular lens fibers, and less commonly in fibers in the deep cortex or nucleus (Figure 2A and B). Careful comparison of lenses from the control and treated animals may be required to differentiate such artifacts from the actual test article-related effects. Autolysis of lens proteins may also mimic degeneration (Figure 3). Artifactual adherence of posterior iris epithelium to the anterior lens capsule may occur in any species, regardless of fixative (Figure 2A). In all of the commonly utilized species, prolonged fixation in Davidson’s or modified Davidson’s results in hardening of the lens, which leads to shattering/fragmentation at microtomy.
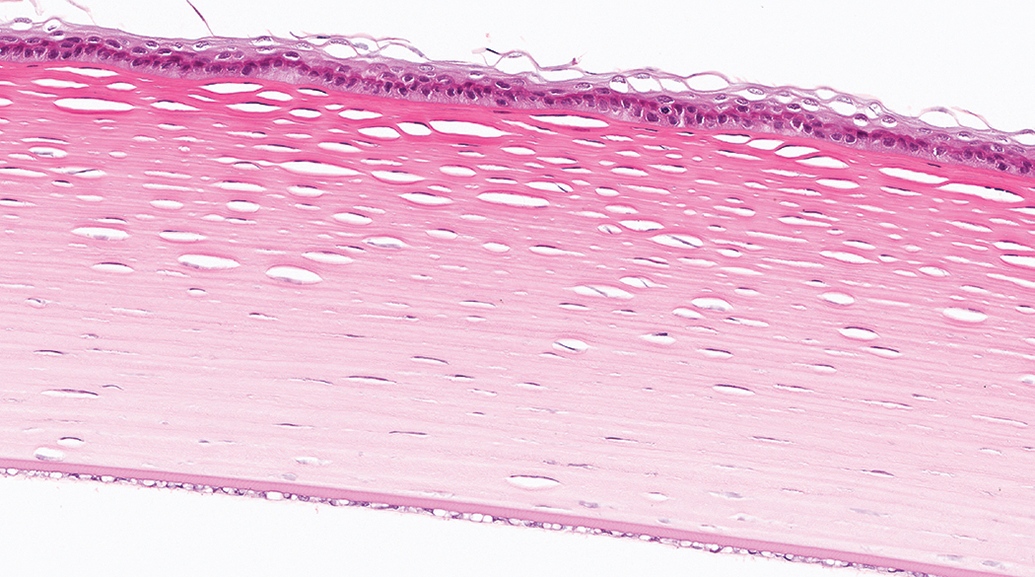
Cornea, Dutch-Belted rabbit. Artifacts of fixation in Modified Davidson’s fixative: ballooning and desquamation of superficial epithelium, clefts in the stroma, and cytoplasmic vacuolation of the endothelium.
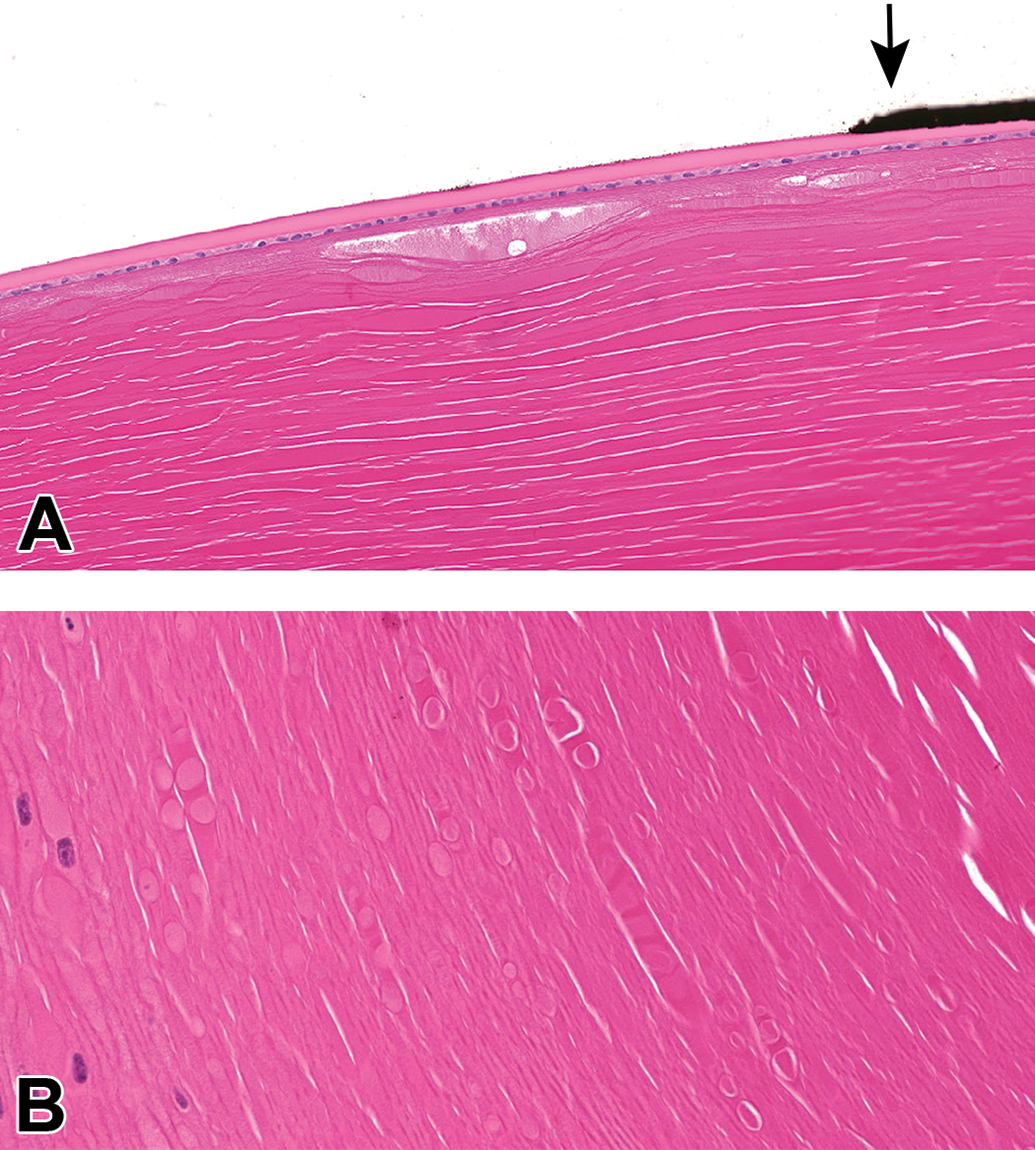
Lens, cynomolgus monkey. Common artifacts of fixation in Modified Davidson’s solution. A, Subcapsular anterior lens fibers are swollen and have clear to flocculent cytoplasm. Artifactual adherence of posterior iris epithelium (arrow) to anterior lens capsule may also occur with other fixatives. B, Lens fibers near the nuclear bow (equator) are variably swollen, and some contain eosinophilic globules.
Lens, Han-Wistar rat. Autolysis. Amorphous to granular eosinophilic material resulting from lens fiber dissolution.
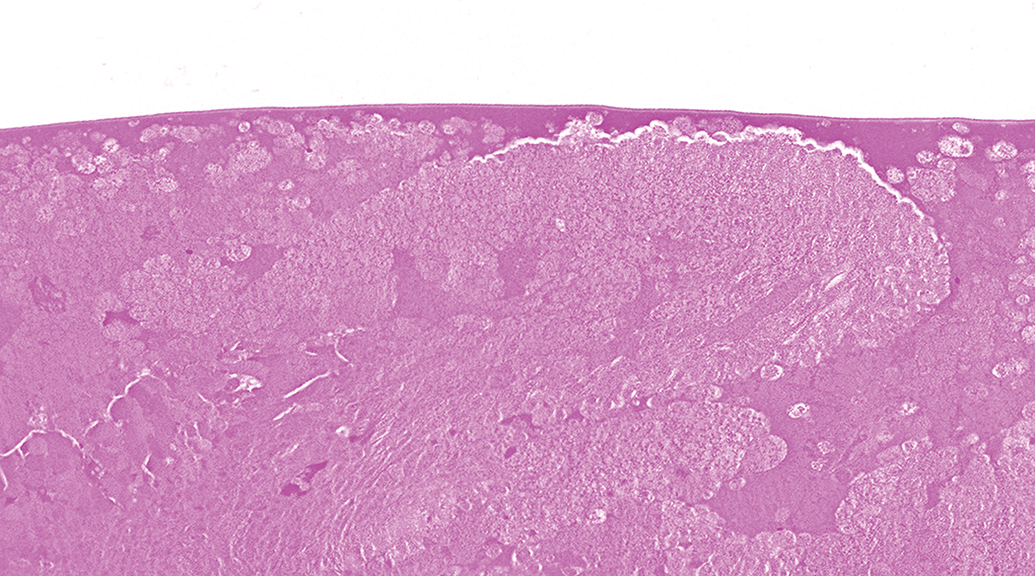
In many ocular toxicity studies, a sample of vitreous fluid is collected from one or both eyes immediately following removal of the globe from the orbit. This procedure often results in posterior displacement and distortion of the iris (Figure 4). Generally, the harvested vitreous is replaced with an approximately equal volume of fixative to maintain the normal contour of the globe and accelerate the fixation of the retina. In animals with large lenses, such as the rabbit, fixative may be inadvertently injected into the lens, resulting in rupture and release of lens protein (Figure 5). This postmortem artifact is not associated with inflammation; antemortem lens rupture may or may not result in an inflammatory response. 2

Anterior segment, cynomolgus monkey. Posterior displacement and distortion of iris leaflets due to the collection of vitreous at necropsy.

Lens, Dutch-Belted rabbit. Rupture of posterior lens capsule with loss and disruption of subcapsular lens fibers due to inadvertent injection of fixative following collection of vitreous at necropsy. Note the lack of an inflammatory response.
The most common artifact in the posterior segment is the separation of the neuroretina from the retinal pigment epithelium (RPE; Figure 6A and B), which may result from excessive traction on the globe during collection, 3 prolonged fixation, fixation in formalin instead of Davidson’s or modified Davidson’s, or rough handling (such as may result from using a dull blade) at trimming. Artifactual retinal separation is distinguished from antemortem retinal detachment (Figure 6C) by the absence of RPE hypertrophy, outer retinal atrophy, and/or fluid or cells in the subretinal space in the former. Artifactual separation may also occur in the choroid or between the choroid and sclera (Figure 6D). In rabbits, bleb-like protrusions of the nerve fiber layer into the vitreous are artifacts that may be the result of traction associated with vitreal condensation during fixation (Figure 6B). In monkeys, vacuolation or clefting in the outer plexiform layer is another common artifact, possibly the result of traction during collection or vitreal condensation during fixation (Figure 7).
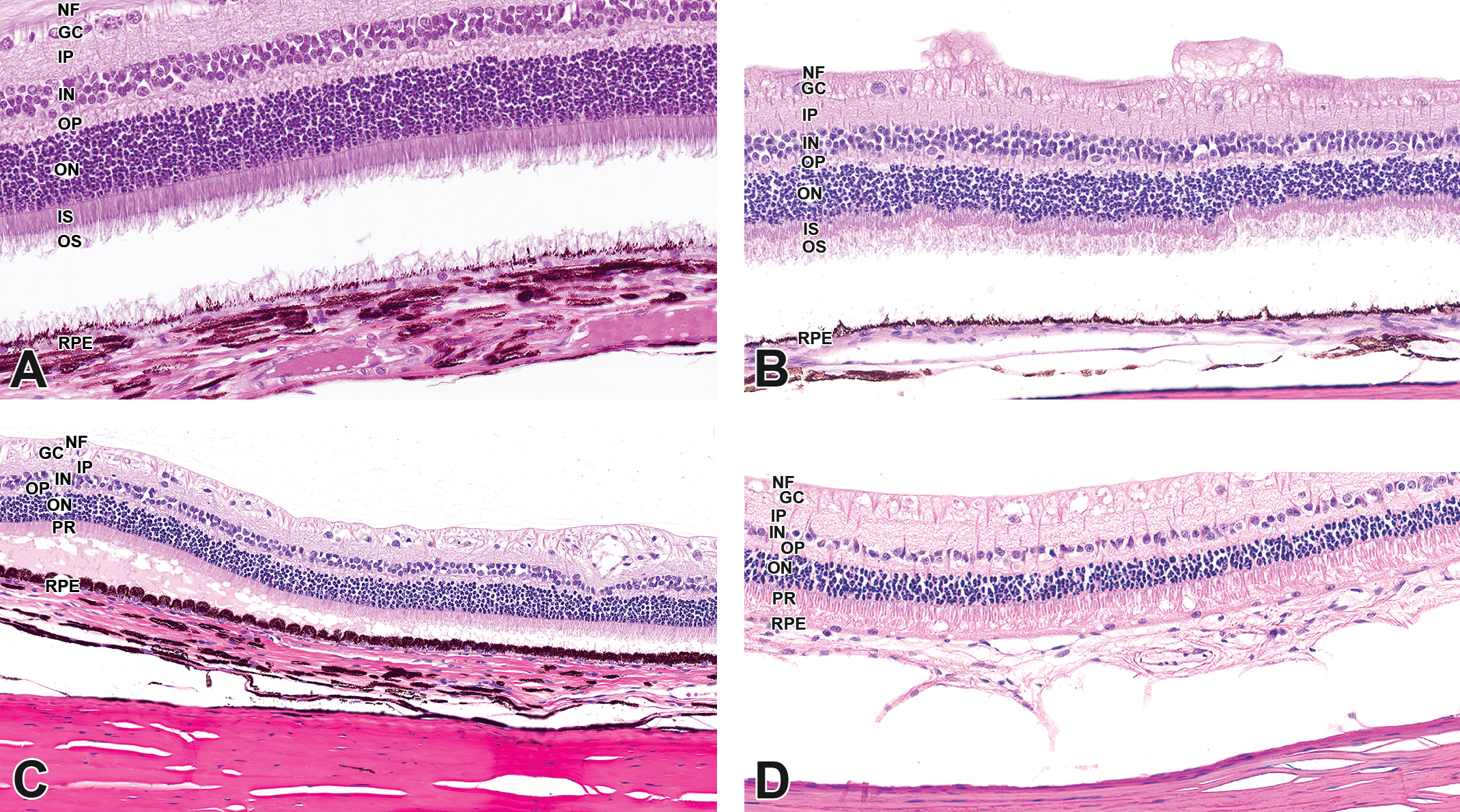
Retina/choroid. A, Artifactual separation of neuroretina from the RPE, beagle dog. Some photoreceptor outer segments are separated from the inner segments and remain attached to the RPE, which is morphologically unremarkable. B, Artifactual separation of neuroretina from the RPE, Dutch-Belted rabbit. Nerve fiber layer “blebs” at the vitreoretinal interface are artifacts that possibly result from traction associated with vitreal condensation during fixation. C, Retinal detachment, beagle dog. Hypertrophy of RPE and fluid in subretinal space on the left side of the image indicate that the detachment occurred antemortem. D, Artifactual separation of the choroid from the sclera, Dutch-Belted rabbit. Neuroretina remains attached to the RPE. GC, ganglion cell layer; IN, inner nuclear layer; IP, inner plexiform layer; IS, photoreceptor inner segments; NF, nerve fiber layer; ON, outer nuclear layer; OP, outer plexiform layer; PR, photoreceptor layer; OS, photoreceptor outer segments; RPE, retinal pigment epithelium.
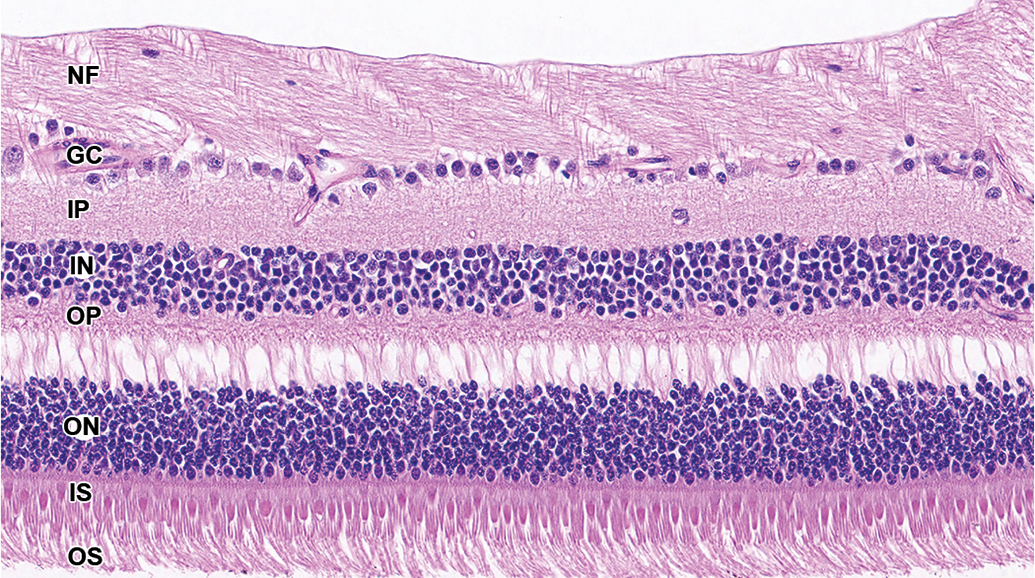
Retina, cynomolgus monkey. Artifactual large vacuoles or clefts in the outer plexiform layer are common in eyes fixed in Davidson’s or modified Davidson’s fixatives and possibly result from traction associated with vitreal condensation during fixation.
The relatively high (35%) ethanol content of Davidson’s fixative frequently results in artifactual vacuolation of the optic nerve (Figure 8). Examination of a portion of optic nerve fixed in 10% neutral-buffered formalin (transverse section), in addition to the Davidson’s fixed portion attached to the globe (longitudinal section), aids in distinguishing such artifacts from antemortem changes.
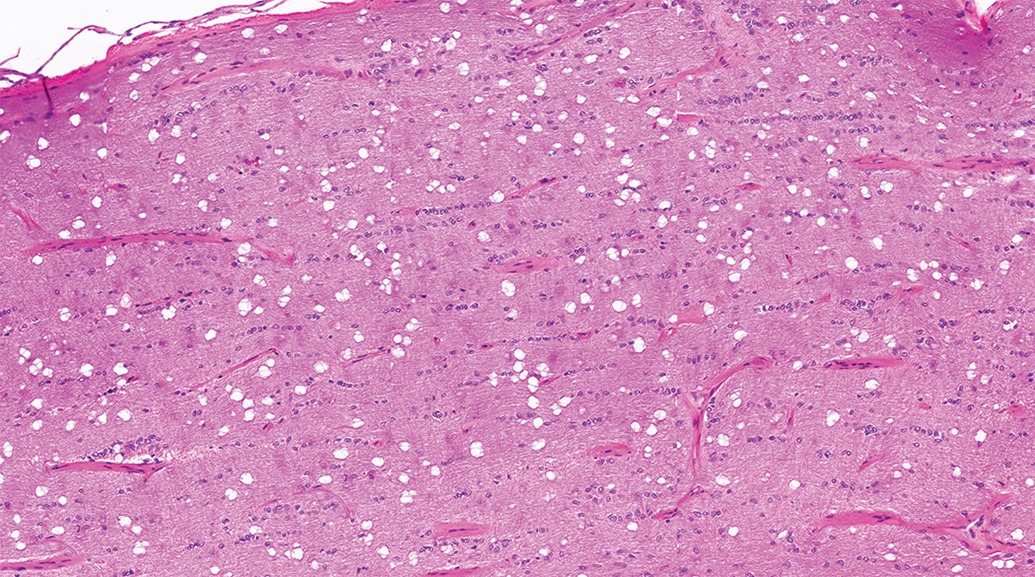
Optic nerve, Dutch-Belted rabbit. Artifactual vacuoles due to fixation in Davidson’s solution are common in multiple species.
In-Life Procedure-Related Findings
Drug delivery to the eye may occur via multiple routes. 4 The routes most commonly utilized in ocular toxicity studies are topical instillation and intravitreal or subretinal injection. The topical instillation procedure itself does not result in discernible microscopic findings in the eye; findings noted in control animals in instillation studies are generally attributable to the vehicle or considered spontaneous background findings. Intravitreal injections are typically performed in the superior temporal or inferior temporal quadrant of the eye; the needle enters the sclera posterior to the limbus and penetrates the ciliary body pars plana or peripheral retina, creating a track (Figure 9). At study termination (generally several weeks postinjection), these needle tracks are characterized by sharply demarcated foci of fibrosis with generally minimal mixed cellular infiltrate in the sclera and/or pars plana; the ciliary epithelium is focally absent or disrupted, and small amounts of fibrous tissue often extend a short distance into the vitreous, which may contain low numbers of macrophages or other inflammatory cells and extrude into the track. In addition, the sclera may contain small amounts of translucent refractile foreign material (likely silicone needle lubricant 5 ) that may be surrounded by macrophages. The presence of inflammation at intravitreal needle track sites of control animals that is of greater than minimal severity suggests an effect of the vehicle or microbial contamination. Intravitreal injection deposition sites (in the vitreous) are generally not identifiable at the end of a multiple week study if the vehicle is a solution. Depot formulations often localize in the inferior vitreous and may induce a granulomatous/foreign body response. 6,7
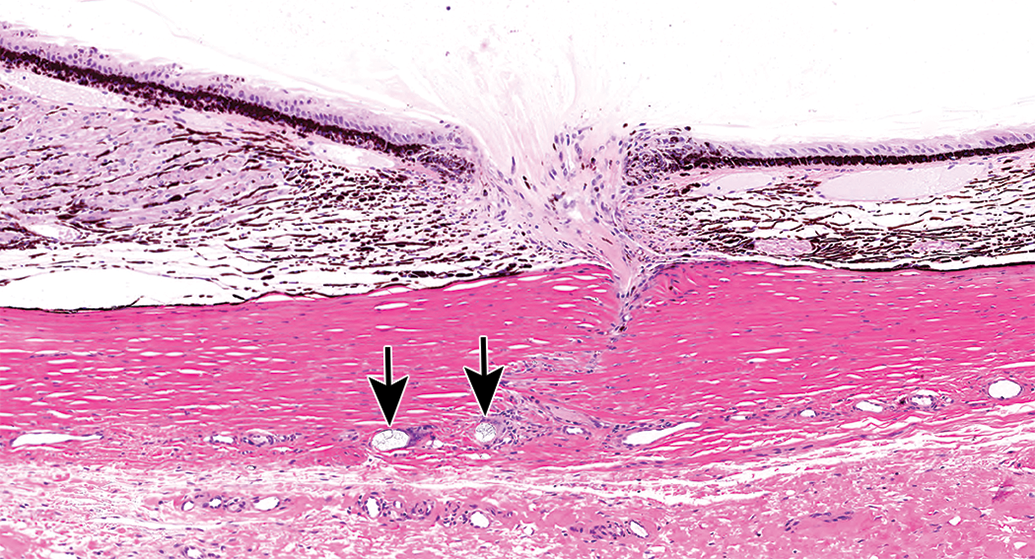
Ciliary body pars plana and sclera, cynomolgus monkey. Intravitreal injection track. Sharply demarcated focus of fibrosis and epithelial loss in the pars plana extends to the vitreous, and the vitreous (amorphous eosinophilic material) extends into the track. Note small amounts of translucent refractile foreign material (arrows) surrounded by low numbers of mononuclear cells in the sclera.
Subretinal injections are delivered into the potential space between the photoreceptors and the RPE and are most commonly used to deliver gene therapies targeting one or both of these cell layers. Vehicle injection into the subretinal space results in the formation of a subretinal “bleb,” that is, an accumulation of fluid in a focal area of retinal detachment. Such fluid may be absorbed and the photoreceptors reattached to the RPE as early as 2 days following injection. 8 At study termination (generally several weeks postinjection), the only evidence of the injection site/bleb is generally one or more foci of hypertrophy, hyperpigmentation, and/or loss of polarity of the RPE (Figure 10).
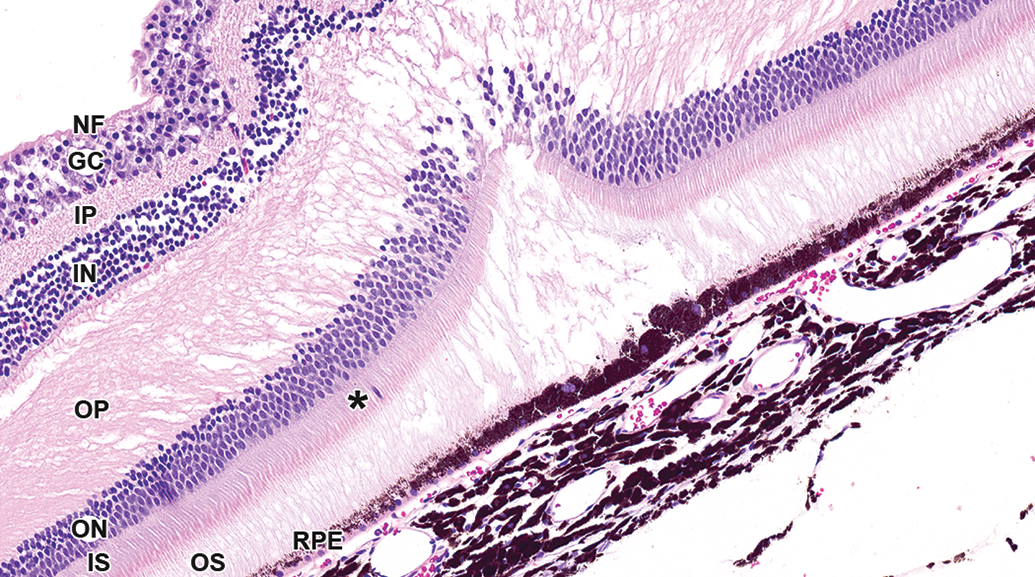
Retina/choroid at fovea, cynomolgus monkey. Subretinal injection site. Focal area of RPE hypertrophy and hyperpigmentation. Rarefaction of neuroretina is attributed to paraformaldehyde fixation. One photoreceptor nucleus is displaced into the inner segment of the photoreceptor layer (*).
In nonhuman primates and less often other species, laser-induced disruption of Bruch membrane to induce choroidal neovascularization is used as a model of neovascular age-related macular degeneration in which to evaluate the efficacy of potential inhibitors of neovascularization. 9,10 Microscopically, laser sites are characterized by sharply demarcated foci of full-thickness retinal atrophy; the atrophied retina and choroid are adhered by fibrovascular tissue (Figure 11). Varying numbers of individualized pigmented cells (displaced RPE and/or melanophages) are often noted within and around these sites.
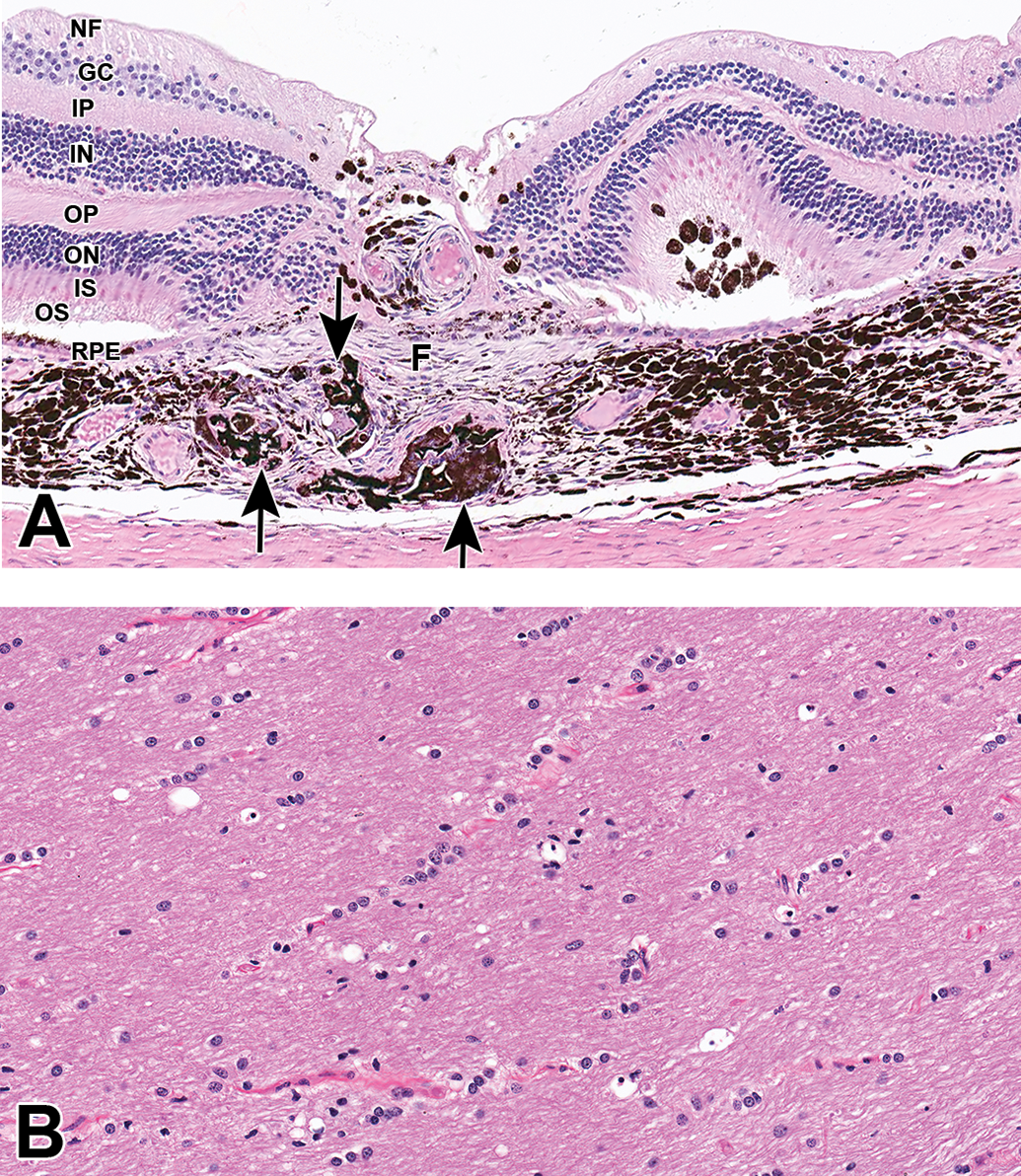
A, Retina/choroid, cynomolgus monkey. Laser site. A sharply demarcated zone of full-thickness retinal atrophy contains medium-sized blood vessels (choroidal neovascularization) and pigmented cells (displaced RPE and/or melanophages). The atrophied retina is adhered to the subjacent choroid, which exhibits fibroplasia (F) and contains pigment-laden multinucleate cells (arrows). Pigmented cells are also present within the subretinal space to the right of the laser site (focal retinal detachment). B, Optic nerve. Mild multifocal axon degeneration, presumably due to loss of ganglion cells at laser sites.
Findings have been noted in the paraocular (Harderian and lacrimal) glands in rabbits in ocular studies in which medial auricular artery catheters were used to facilitate the collection of multiple blood samples for toxicokinetic analysis. 11,12 These findings consist of sharply demarcated foci of coagulative necrosis in the Harderian and/or lacrimal glands (Figure 12A and B), less commonly in the mandibular salivary gland (Figure 12C) and rarely the brain (Figure 12D). Because these findings are consistent with infarcts and have not been noted when other methods of blood collection are utilized, 11 it is likely that microthrombi form in the catheter between blood collections and that flushing of the catheter with saline propels such thrombi retrograde into the transverse facial, maxillary, or other arteries via the auricular arteries.
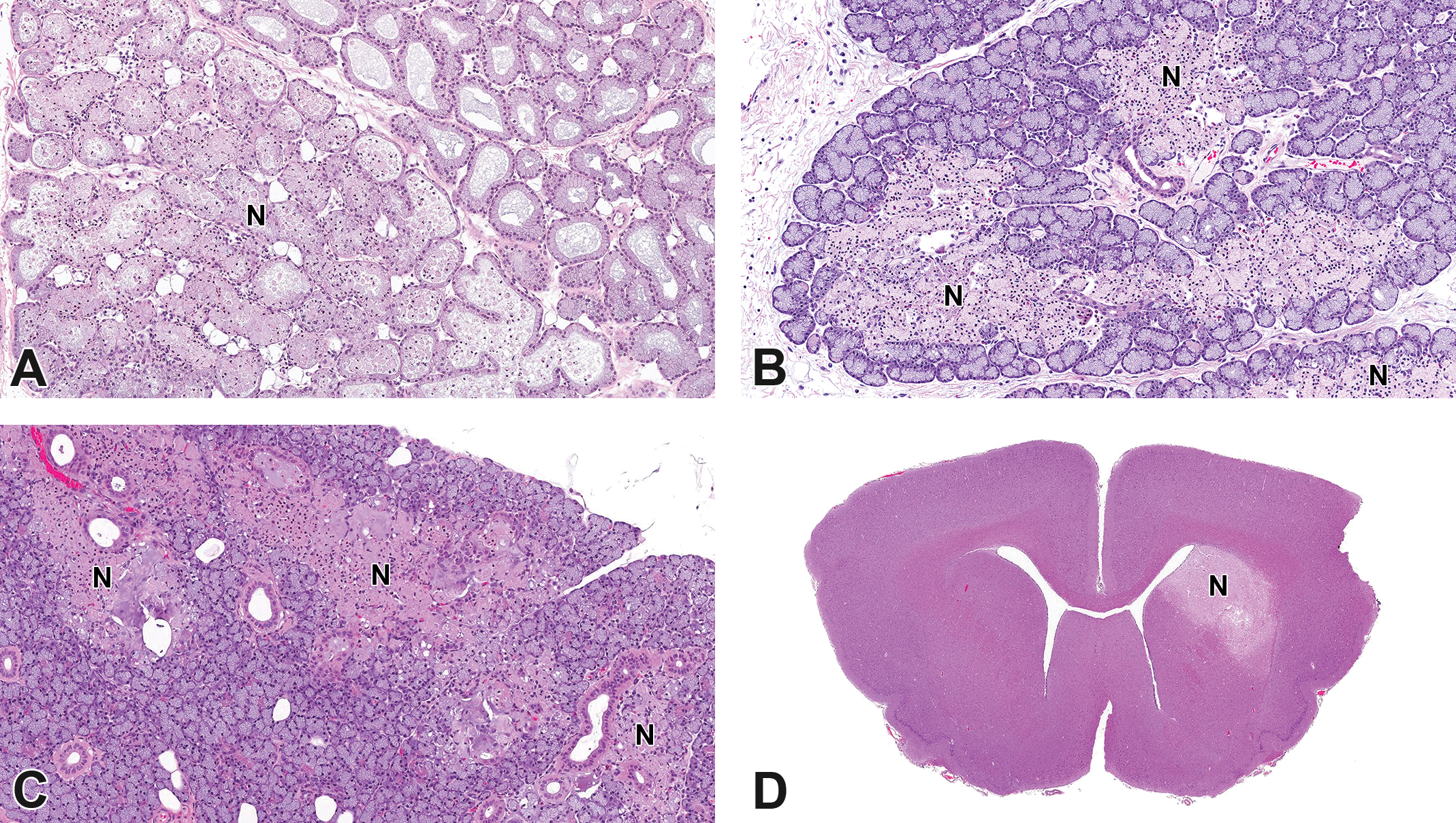
New Zealand white rabbits. Sharply demarcated foci of necrosis (N, presumptive infarcts) in the Harderian gland (A), lacrimal gland (B), mandibular salivary gland (C), and brain (D). Infarcts such as these occur sporadically in rabbits in which a catheter in the medial auricular artery was utilized to facilitate the collection of multiple toxicokinetic blood samples.
Spontaneous Background Findings
Spontaneous corneal mineralization (often termed corneal dystrophy) has been reported in multiple species, including multiple strains of rabbits, rats, and mice. 13 –15 Punctate corneal opacities are generally noted on slit-lamp examination; microscopically, these correlate with focal or multifocal punctate, linear, and/or coarse deposits of basophilic granular material in or along the epithelial basement membrane and/or sub-epithelial stroma (Figure 13). Additional features may include attenuation of the overlying epithelium, hypertrophy of subjacent keratocytes, or granulomatous inflammation. Calcium may or may not be demonstrable in the basophilic material. 16 Multiple sections may be required to identify lesions in mildly affected animals. Corneal dystrophy in humans and animals has been reviewed in detail by Schuh. 17
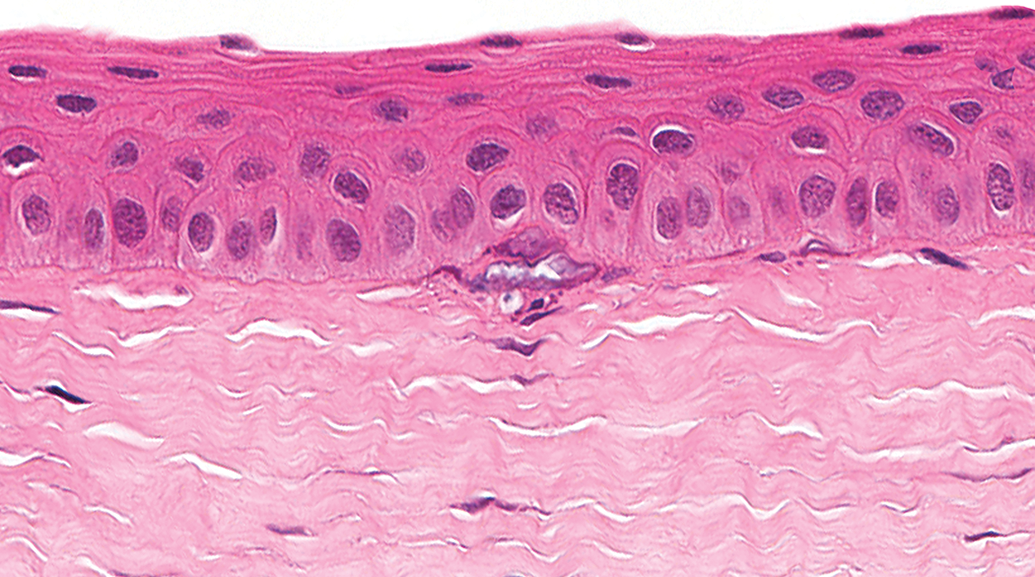
Cornea, Han-Wistar rat. Focal mineralization of the epithelial basement membrane.
Infiltrates of mononuclear cells occur sporadically in the uvea of cynomolgus monkeys. These are most common in the pars plana of the ciliary body (Figure 14), less common in the pars plicata of the ciliary body or choroid, and rare in the iris. 18,19 Similar infiltrates occur less frequently in the choroid of the rabbit. 20 An increase in the incidence and severity of uveal mononuclear cell infiltrates, especially if plasma cells are a prominent component, may indicate an immune response to an injected biotherapeutic. 21 Infiltrates of mononuclear cells may also be noted at the limbus in monkeys, dogs, and rabbits. 19,20,22 Mononuclear cell infiltrates in the conjunctiva 19,22 likely represent conjunctiva-associated lymphoid tissue. 23
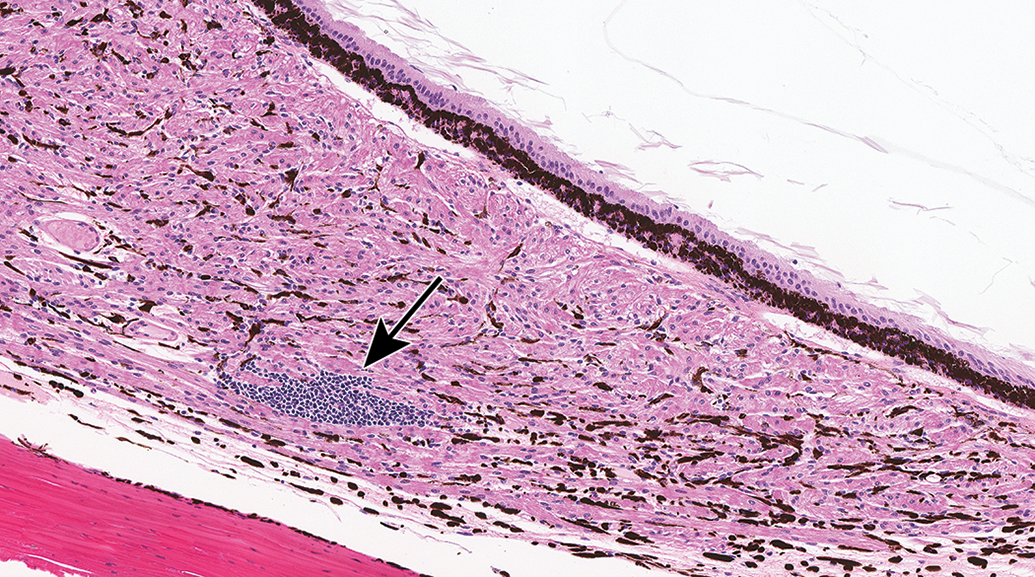
Ciliary body pars plana, cynomolgus monkey. Sporadic focal aggregates of mononuclear cells (arrow) are common at this location, less common in the choroid, and rare in the iris.
Another common finding in cynomolgus monkeys is the presence of small unilocular or multilocular retinal cysts at the ora serrata (Figure 15). These are rarely recorded in the microscopic data. Similar (uveal) cysts occasionally occur at the ora ciliaris retinae in dogs.
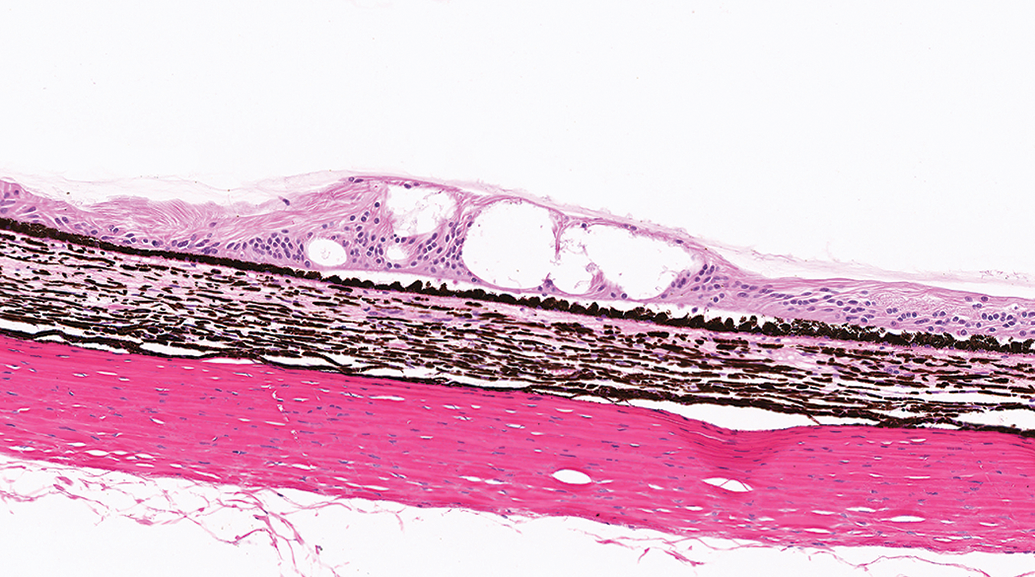
Ora serrata, cynomolgus monkey. Unilocular or multilocular cystic structures at the ora serrata are extremely common in cynomolgus monkeys and are generally not recorded.
Small foci of retinal dysplasia, including folds and rosettes, occur sporadically in many species (Figure 16). Since retinal dysplasia can only occur during differentiation of the retina, with the exception of juvenile or reproductive toxicity studies, such findings may be interpreted as spontaneous because they occurred prior to the onset of test article administration. Rosettes may also be artifacts that result from differential rate fixation of the vitreous and retina or the retina and sclera, 3 especially in mice and rabbits.
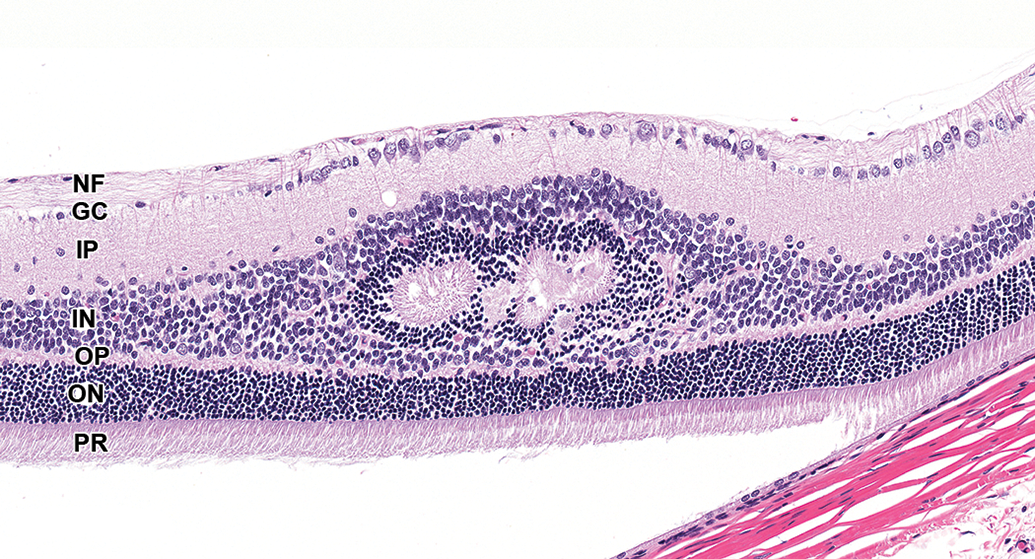
Retina, Han-Wistar rat. Retinal rosettes. Retinal folds and rosettes are developmental malformations (dysplasia) that occur sporadically in multiple species. Note artifactual retinal separation.
Peripheral displacement of low numbers of photoreceptor nuclei is a common finding in cynomolgus monkeys and less common in rabbits and rodents. This finding is characterized by the presence of a photoreceptor nucleus within the photoreceptor inner segments, external to the external limiting membrane (Figure 17), and is considered a degenerative change. 24 The presence of a few displaced nuclei is generally not recorded. Increased numbers of displaced nuclei may be a test article–related effect.
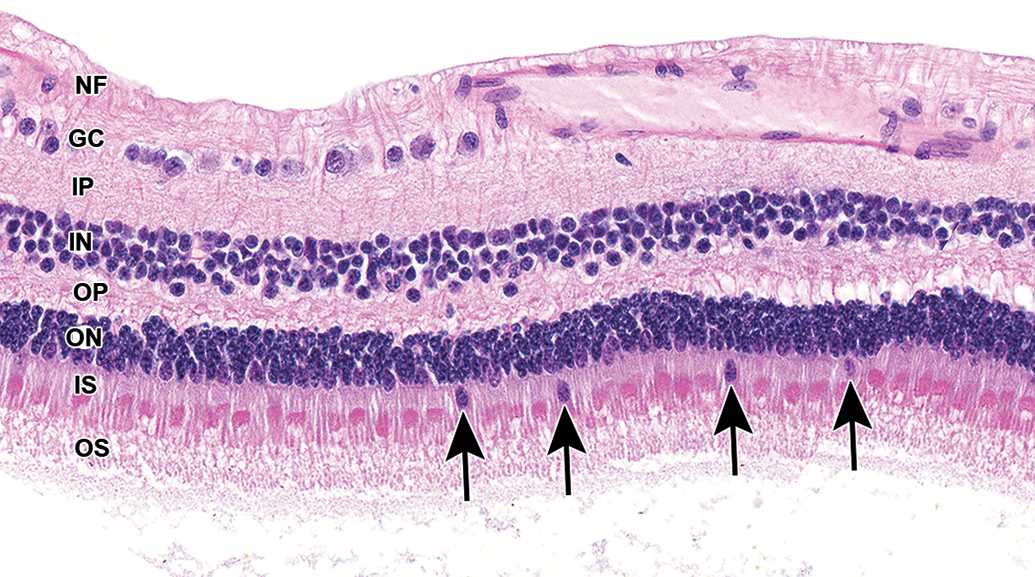
Retina, cynomolgus monkey. Peripheral displacement of photoreceptor nuclei. Low numbers of photoreceptor nuclei (arrows) are external to the outer limiting membrane, within photoreceptor inner segments. Low numbers of such displaced nuclei are common in cynomolgus monkeys and less common in rabbits.
A common spontaneous background finding in Dutch-belted rabbits is focal or multifocal hypertrophy of the RPE, characterized by small segments of the RPE in which cells have abundant cytoplasm that varies from homogenously gray to heavily pigmented. Such findings are most often noted adjacent to the optic nerve (peripapillary) or ora ciliaris retinae but may also occur at other locations. 25 Similar foci of RPE hypertrophy are occasionally noted in cynomolgus monkeys (Figure 18). Another occasional background finding in the rabbit retina is the presence of one or a few round eosinophilic structures in the inner nuclear layer (Figure 19). The identity of these structures is uncertain, although they are morphologically consistent with axonal spheroids.
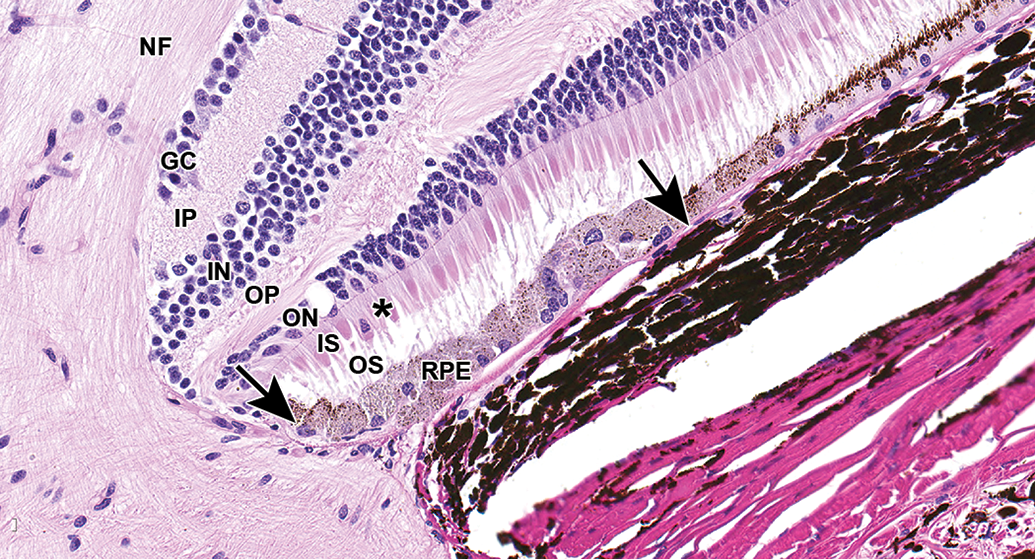
Peripapillary retina/choroid, cynomolgus monkey. Hypertrophied RPE (between arrows) with abundant cytoplasm is occasionally present adjacent to the optic disc. This background finding is more often noted in Dutch-Belted rabbits. Note displaced photoreceptor nucleus (*).
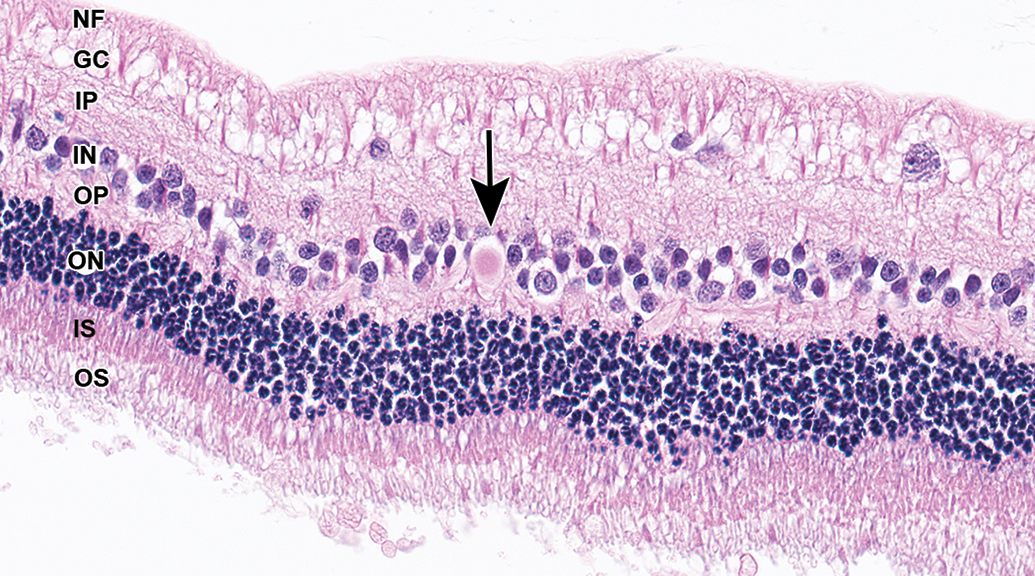
Retina, New Zealand white rabbit. Rare homogeneous eosinophilic round structures (arrow) such as this (consistent with a swollen axon/spheroid) are occasionally present in the inner nuclear layer in rabbits.
Bilateral optic atrophy is a well-known spontaneous finding in rhesus and cynomolgus macaques. 26,27 Affected animals are often identified via ophthalmoscopy (temporal pallor of the optic disc and thinning of the temporal retinal nerve fiber layer) and removed from ocular studies prior to initiation. Microscopically, optic atrophy is characterized by a reduction in the thickness of the ganglion cell and nerve fiber layers in the macula (Figure 20). In unaffected eyes, the ganglion cell layer is generally 6 to 8 cells thick at the macula; in affected animals, this is often reduced to a single layer of cells. There is a corresponding loss of axons in the temporal portion of the optic nerve. Vacuolation of the inner nuclear layer and a reduction in the number of neurons in the lateral geniculate nucleus may also be noted. Whether this lesion results from atrophy (or abiotrophy) or a form of hypoplasia has not been determined.
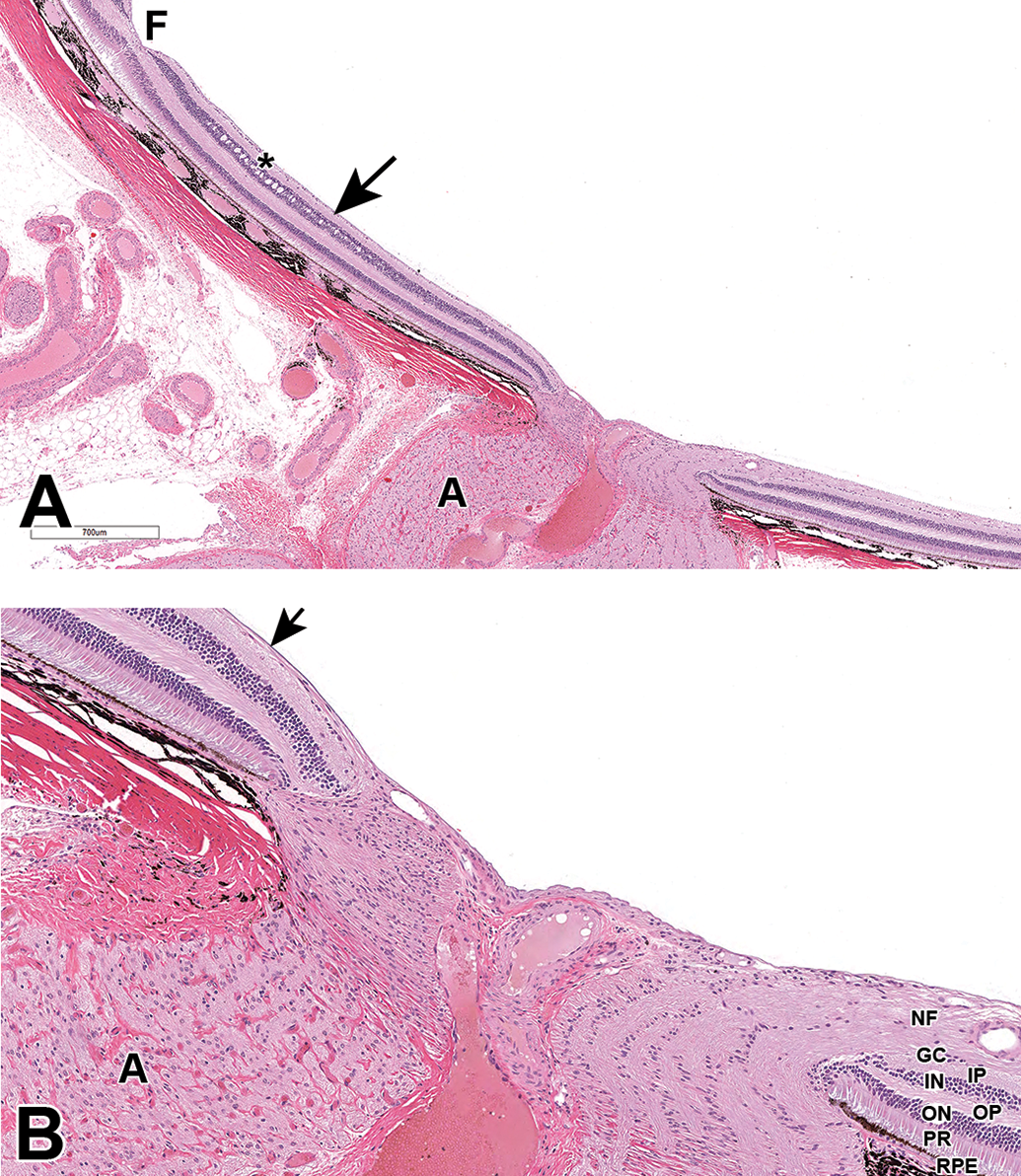
Posterior segment and optic nerve, cynomolgus monkey. Optic atrophy. A, At the macula, the nerve fiber and ganglion cell (arrow) layers are markedly reduced in thickness, and the inner nuclear layer is vacuolated (*). There is also atrophy (A) of the temporal portion of the optic nerve. B, Higher magnification of optic nerve and peripapillary retina. The retina nasal to the optic disc is unremarkable. F indicates fovea.
Atrophy of the outer retina is a rare background finding in cynomolgus macaques, which to date has only been reported in animals of Mauritius origin. 28,29 This form of outer retinal atrophy is characterized by a reduction in the thickness of the outer nuclear layer to a single layer of predominantly ovoid (cone) nuclei (Figure 21). There is also peripheral displacement of many (predominantly cone) nuclei, loss of photoreceptor outer segments, and vacuolation of the inner nuclear layer. This lesion is most prominent in the macula and elsewhere in the central retina. The peripheral retina and fovea are unaffected.
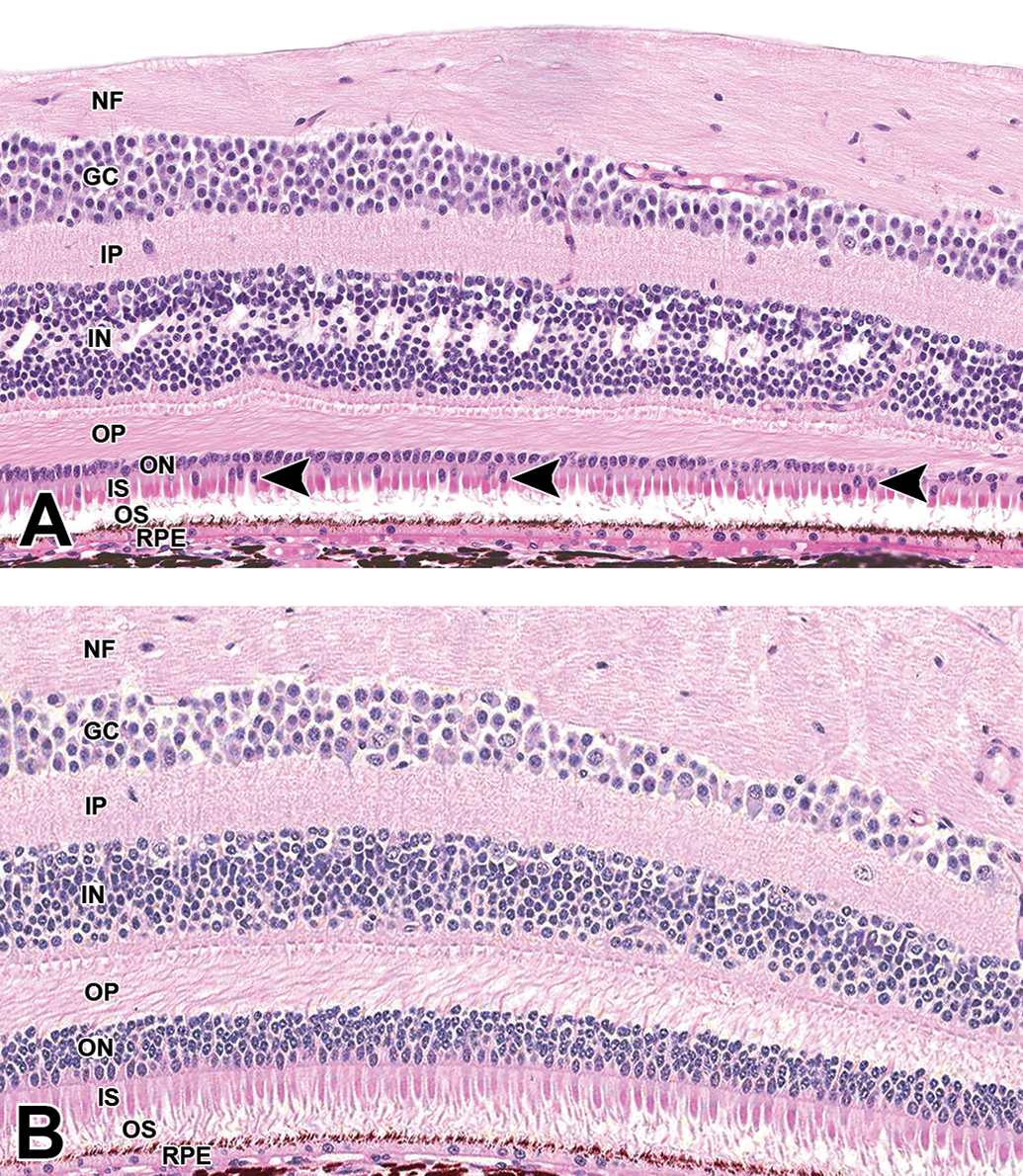
Macula, Mauritius cynomolgus monkey. Outer retinal (rod) atrophy (A). Macula from an unaffected animal for comparison (B). The outer nuclear layer is reduced to a single layer of predominantly ovoid (cone) nuclei, and there are multiple peripherally displaced nuclei among the photoreceptor inner segments, also predominantly cones (arrowheads). There is also loss of outer segments and vacuolation of the inner nuclear layer.
Atrophy of the outer retina due to phototoxicity is well characterized in albino rodents. 30 Retinal phototoxicity results in photoreceptor loss, characterized microscopically by a reduction in the thickness of the photoreceptor and outer nuclear and plexiform layers (Figure 22); these changes are initially limited to the central retina but progress to become diffuse. Test articles that induce mydriasis or otherwise increase the daily exposure of the retina to ambient light may exacerbate the incidence and severity of outer retinal atrophy.
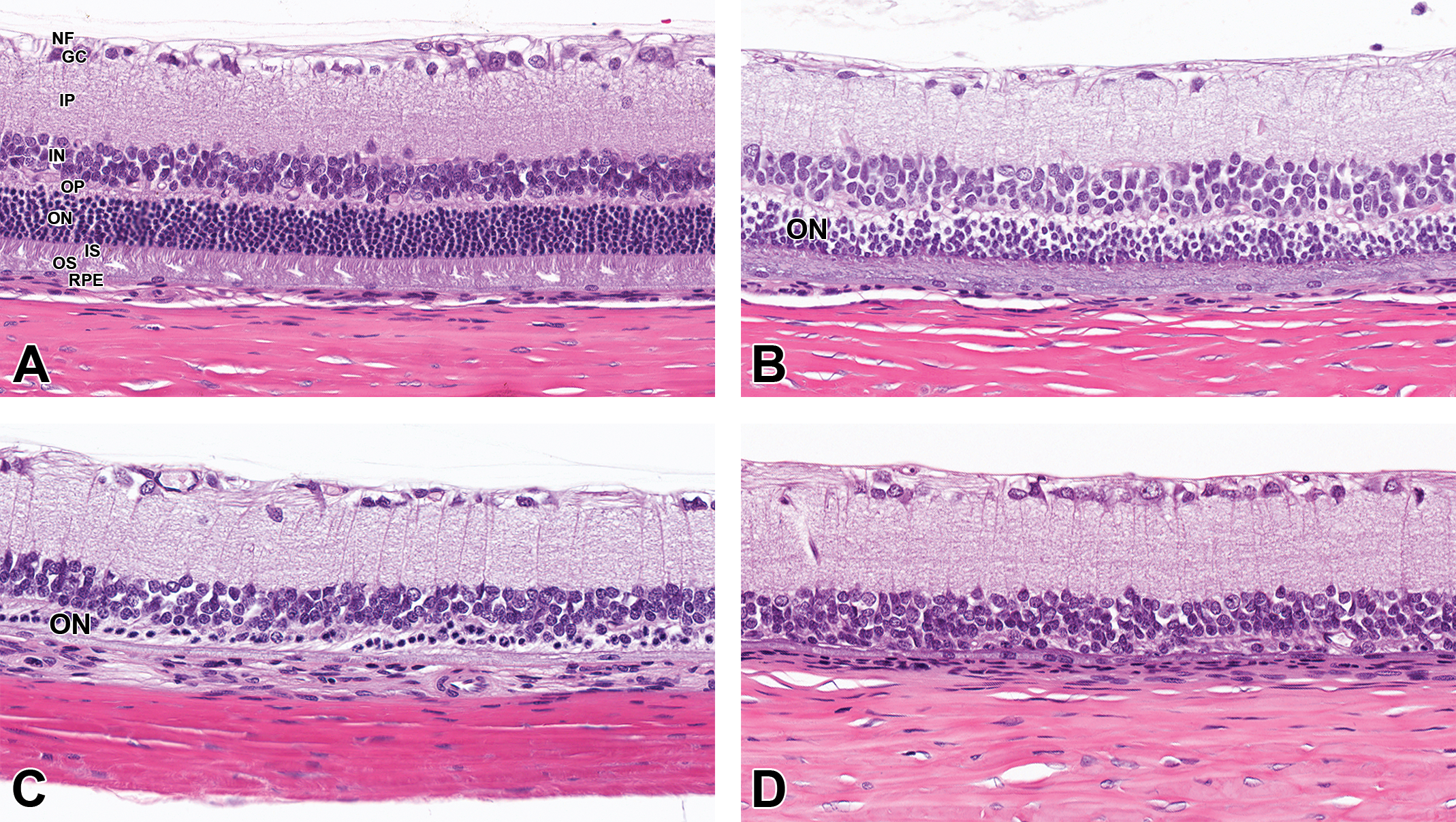
Retina, Han-Wistar rats. Retinal phototoxicity. Unaffected (A) and minimal (B), moderate (C), and marked (D) reduction in thickness (atrophy) of the outer plexiform, outer nuclear, and photoreceptor layers. Note that the outer nuclear layer is thicker than the inner nuclear layer in the unaffected animal (A).
Neoplastic lesions in ocular tissues are rare in ocular toxicity studies due to the young age of the animals utilized in most studies. However, proliferative lesions of uveal melanocytes are occasionally noted in control cynomolgus monkeys 28 (Figure 23) or dogs in toxicology studies.
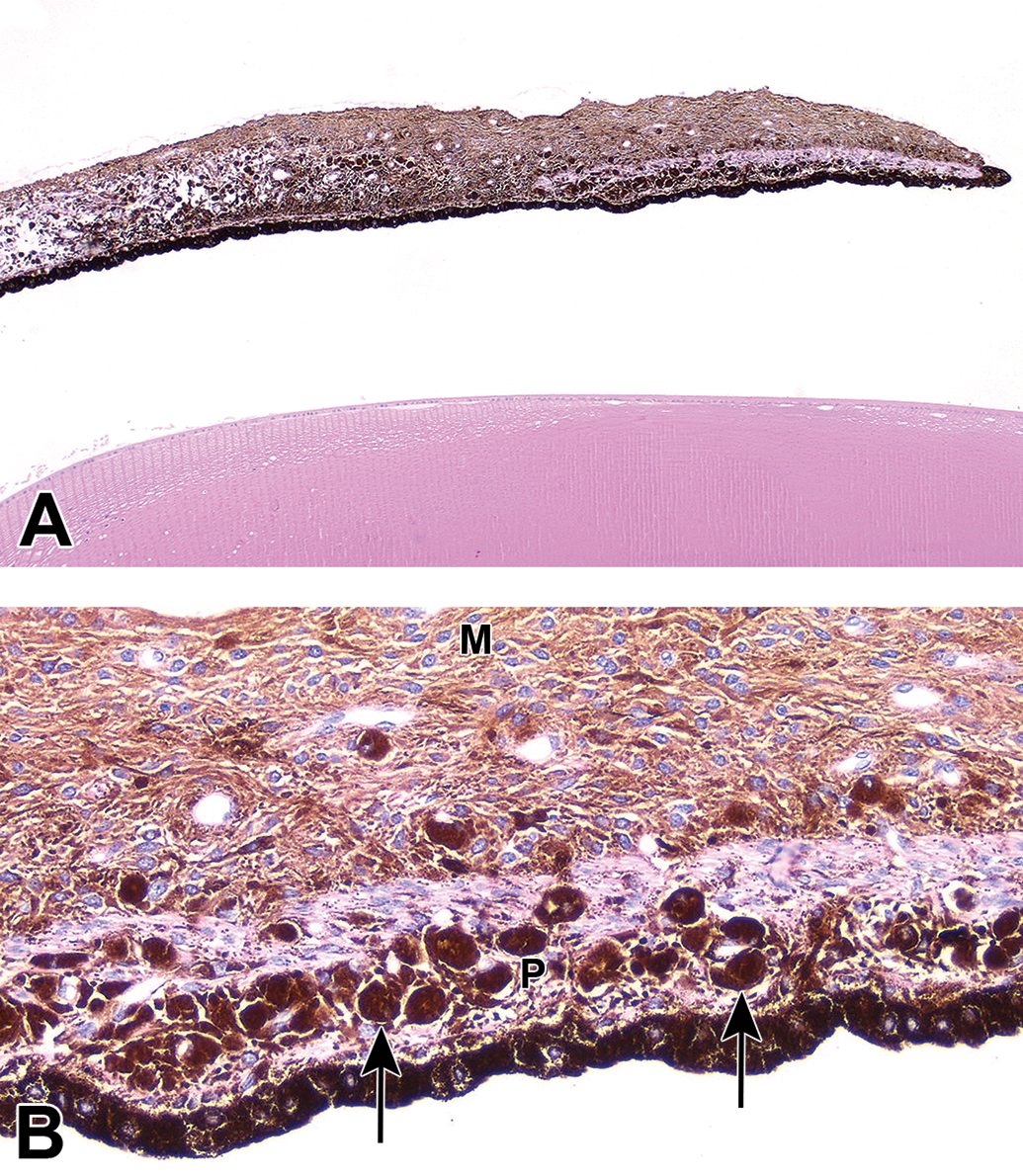
Melanoma, iris, cynomolgus monkey. A, The iris is expanded by neoplastic melanocytes. B, The anterior iris stroma is replaced by a monomorphic population of neoplastic melanocytes (M). Large densely pigmented cells (arrows) in the posterior stroma (P) may be melanophages or epithelioid neoplastic melanocytes.
In the optic nerve, minimal multifocal axon degeneration is occasionally noted in control animals, primarily in dogs (Figure 24) and cynomolgus monkeys. Axon degeneration in affected optic nerves is characterized by low numbers of swollen, eosinophilic axons (spheroids) and/or dilated axon sheaths that are empty or contain cellular debris and/or macrophages (gitter cells).
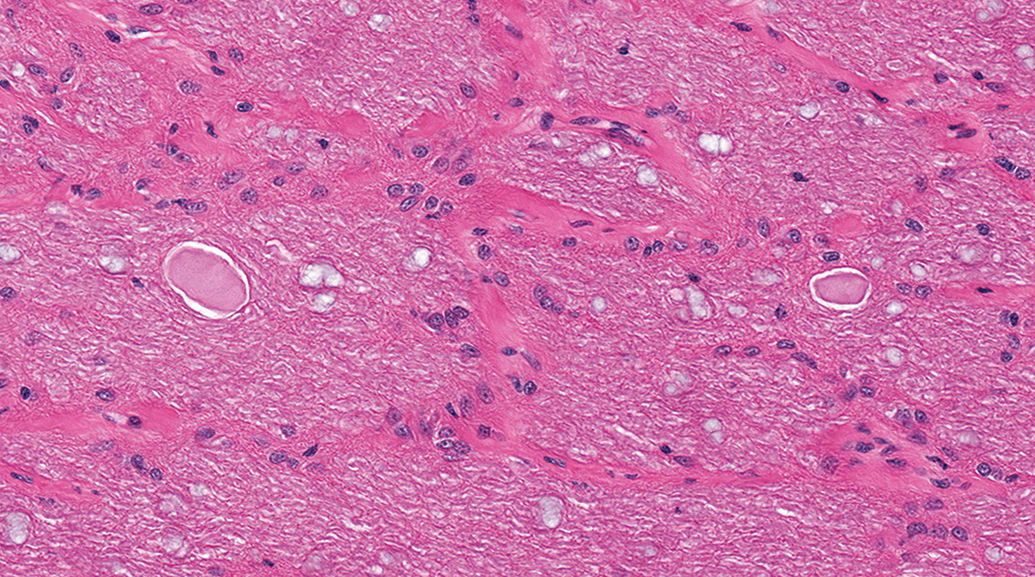
Optic nerve, beagle dog. Low numbers of swollen eosinophilic axons are occasionally present in control animals.
Small infiltrates of mixed or mononuclear cells are common incidental/background findings in the lacrimal and Harderian glands of most species. In the rabbit, foci of mixed or mononuclear cell inflammation (infiltrates within zones of acinar degeneration/atrophy) are frequently noted in the Harderian gland 20 (Figure 25). Rabbit Harderian glands also occasionally contain small foci of the ectopic lacrimal gland (alteration). 20
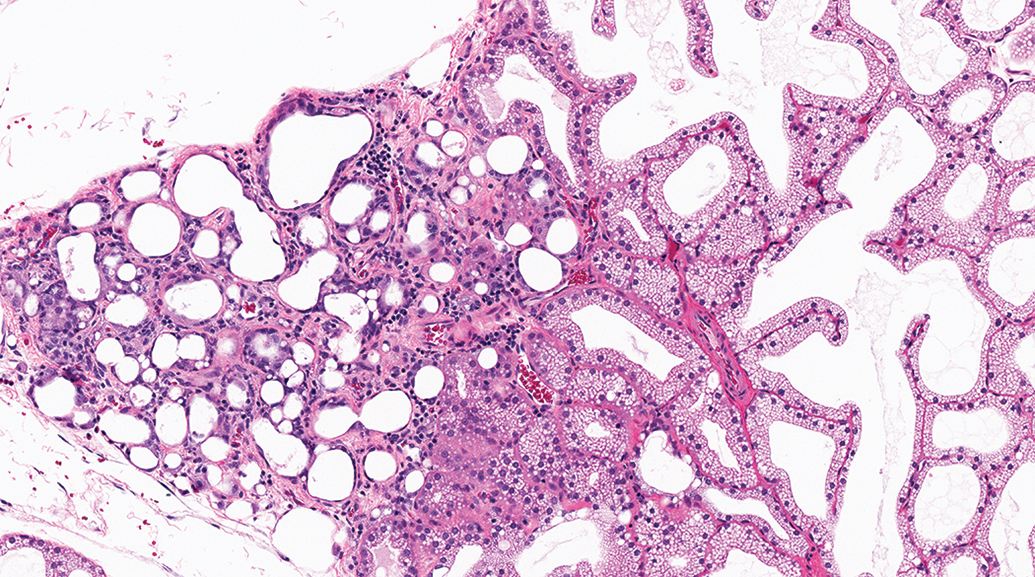
Harderian gland, Dutch-Belted rabbit. Focal chronic inflammation (left side of image) is characterized by interstitial infiltrates of mixed inflammatory cells in foci of acinar degeneration/atrophy.
Discussion
As noted above, most of the artifacts and findings presented here are those most commonly encountered in the ocular studies conducted at our laboratories. It is hoped that familiarity with these artifacts and findings will aid those who do not routinely read such studies and prevent the misinterpretation of artifacts or background findings as test article–related effects, which might lead to erroneous study conclusions. Regarding fixation, the choice of a particular protocol should be based on the experience of the laboratory and the objectives of the study; guidelines for selecting a fixation protocol are beyond the scope of this article.
In addition to familiarity with the changes presented here, knowledge of the study design is critical for study interpretation. In some studies, both eyes of each animal are treated identically; in others, the left or right may serve as untreated or vehicle controls. A unilateral finding in a study in which both eyes were treated identically is less likely to be test article related than a bilateral finding. Furthermore, the route of test article administration, the fixation protocol, the trimming and sectioning plan, and the time interval between dose administration and necropsy, among other factors, determine the nature of the findings present in a given section and how these findings are interpreted by the study pathologist. The fixation protocol and the trimming and sectioning plan should be developed or modified based on the in-life findings (slit-lamp biomicroscopy, indirect ophthalmoscopy, fundus photography, and optical coherence tomography, among others) in order to maximize the likelihood that microscopic correlates for the in-life findings will be identified.
Footnotes
Acknowledgments
The authors would like to thank Steve Van Adestine for figure preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
