Abstract
The zebrafish has been shown to be an excellent vertebrate model for studying the roles of specific genes and signaling pathways. The sequencing of its genome and the relative ease with which gene modifications can be performed have led to the creation of numerous human disease models that can be used for testing the potential and the toxicity of new pharmaceutical compounds. Many pharmaceutical companies already use the zebrafish for prescreening purposes. So far, the focus has been on ecotoxicity and the effects on embryonic development, but there is a trend to expand the use of the zebrafish with acute, subchronic, and chronic toxicity studies that are currently still carried out with the more conventional test animals such as rodents. However, before we can fully realize the potential of the zebrafish as an animal model for understanding human development, disease, and toxicology, we must first greatly advance our knowledge of normal zebrafish physiology, anatomy, and histology. To further this knowledge, we describe, in the present article, location and histology of the major zebrafish organ systems with a brief description of their function.
Hematopoietic Tissue
In contrast to mammals, the bones of the zebrafish have no medullary cavity. Hematopoietic tissue is located in the stroma of the spleen and the interstitium of the kidney (Figures 1 and 3 ). In the adult zebrafish, hematopoiesis occurs primarily in the interstitium of the anterior and posterior kidneys. The hematopoietic stem cells are situated within a stroma of reticuloendothelial tissue, similar to that of the bone marrow in mammals. Endothelial cells line numerous sinuses, through which blood from the renal portal vein is passed for filtration of effete cells, and for the addition of new blood cells to the circulation (Ellet and Lieschke 2010; Roberts and Ellis 2001).
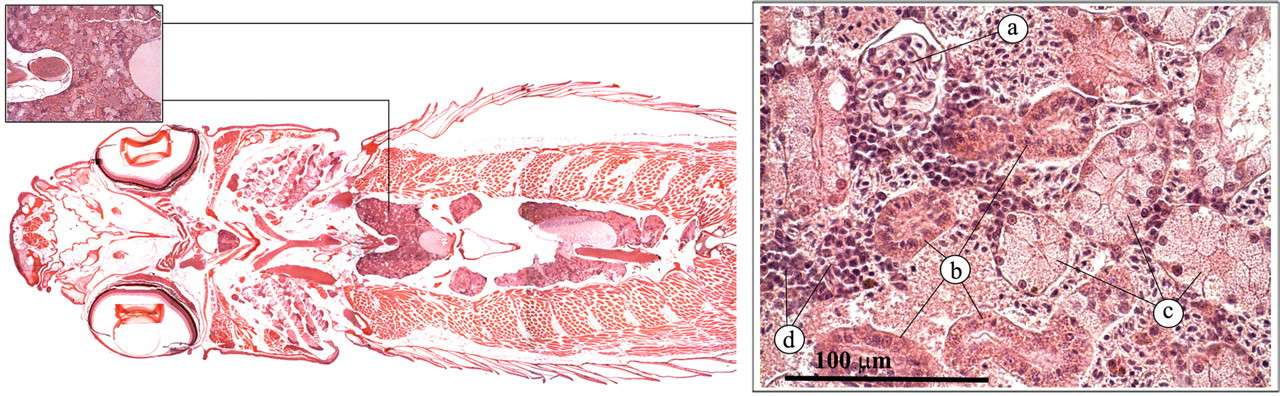
Kidney. (a) Glomerulus; (b) proximal or distal tubulus; (c) collecting duct; (d) hematopoietic tissue.
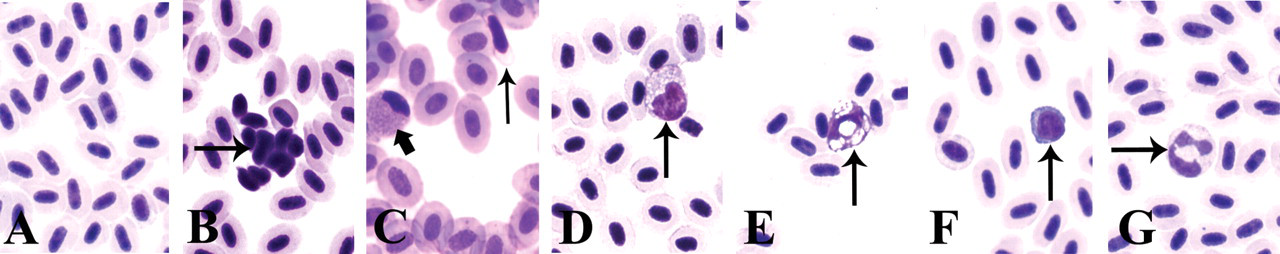
Peripheral blood. a) Erythrocytes, b) activated thrombocytes, c) unactivated thrombocytes (thin arrow) and eosinophilic granulocyte (fat arrow), d) monocyte, e) macrophage, f) lymphocyte, g) neutrophilic granulocyte.
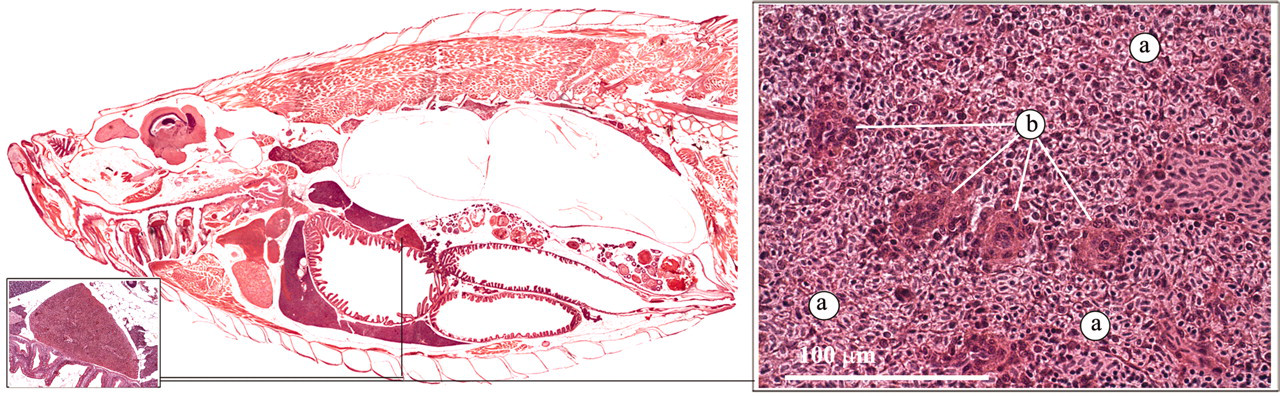
Spleen. a) Red pulp, b) Ellipsoid.
Peripheral Blood
Erythrocytes
Erythrocytes are the main vehicle of oxygen transport and to a lesser extent, of carbon dioxide (Roberts and Ellis 2001). In contrast to the mammalian erythrocyte, the teleost erythrocyte is oval and nucleated and uses aerobic metabolism instead of anaerobic metabolism to generate adenosine triphosphate (Figure 2a).
Thrombocytes
Thrombocytes play an important role in blood clotting (Roberts and Ellis 2001). In contrast to the mammalian thrombocyte, the zebrafish thrombocyte is nucleated (Figure 2a and c; Jagadeeswaran et al. 1999).
Leukocytes
As in mammals, leukocytes in the zebrafish play an important role in the defense against both infectious diseases and foreign materials. Two types of granulocytes have been identified, neutrophilic (heterophilic) granulocytes and eosinophilic granulocytes (Bennett et al. 2001; Crowhurst et al. 2002; Lieschke et al. 2001). The neutrophilic granulocyte is the most abundant and is characterised by a pale cytoplasm and a multilobed, segmented nucleus (Figure 2g). This cell has a morphology that is similar to the mammalian neutrophil.
The eosinophilic granulocyte is characterized by an eosinophilic cytoplasm and a small, nonsegmented, peripherally located nucleus (Figure 2c; Bennett et al. 2001; Lieschke et al. 2001). Their appearance differs considerably from that of mammalian eosinophils, and it remains to be elucidated whether the zebrafish eosinophil has a function that is similar to the mammalian eosinophil (Bennett et al. 2001). It has been postulated that this cell may represent a combined eosinophil/mast cell.
Monocytes form about 5–15 % of the circulating leukocyte population in the zebrafish (Murtha et al. 2003). Morphologically, they are very similar to mammalian monocytes (Figure 2d). Under the appropriate circumstances, the monocytes will develop into mature macrophages (Figure 2e). Macrophages are more obvious in kidney and spleen sections of the adult zebrafish (Bennett et al. 2001; Lieschke et al. 2001). These are large cells with large phagosomes, a high cytoplasm-to-nuclear ratio, diffuse nuclear chromatin, and an agranular, but vacuolated, cytoplasm. The vacuoles can contain phagocytosed material, including pigment and red cell carcasses.
Fish have the functional equivalents of B and T lymphocytes. They are present in the circulating blood (Figure 2f), lymph system, thymus, kidney, and spleen. Lymphocytes form about 71–92% of the circulating leukocyte population in the zebrafish (Murtha et al. 2003).
Spleen
Like other teleosts, the zebrafish lacks lymph nodes. Together with the kidney, the spleen forms the major filtering organ for the removal of foreign agents and defective blood cells (Ellis 2001; Noga 2006). Macroscopically, the spleen is a dark red organ, located in the peritoneal cavity, adjacent to one of the liver lobes.
The splenic parenchyma consists mainly of erythrocytes and thrombocytes (red pulp). In addition, ellipsoids can be distinguished (Figure 3). These are periarterial sheaths of macrophages and reticular cells, supported by reticulin fibers, that are formed at the termination of splenic arterioles. Foreign bodies, such as bacterial cells, are trapped by the ellipsoids.
Thymus
The thymus is a paired organ on the dorsomedial aspect of the brachial cavity (Figure 4 ; Ellis 2001; Noga 2006). Thin trabeculae extend into the thymic parenchyma, forming a three-dimensional lattice that supports the thymocytes and other immune cells, mainly macrophages. In young zebrafish, a cortex, with a higher density of thymocytes, can be distinguished, as well as a medulla, with more epithelial cells and less thymocytes. From the main dorsomedial body, the thymus extends and covers the whole branchial cavity. As in mammals, the thymus appears to function as a maturation site for lymphocytes.
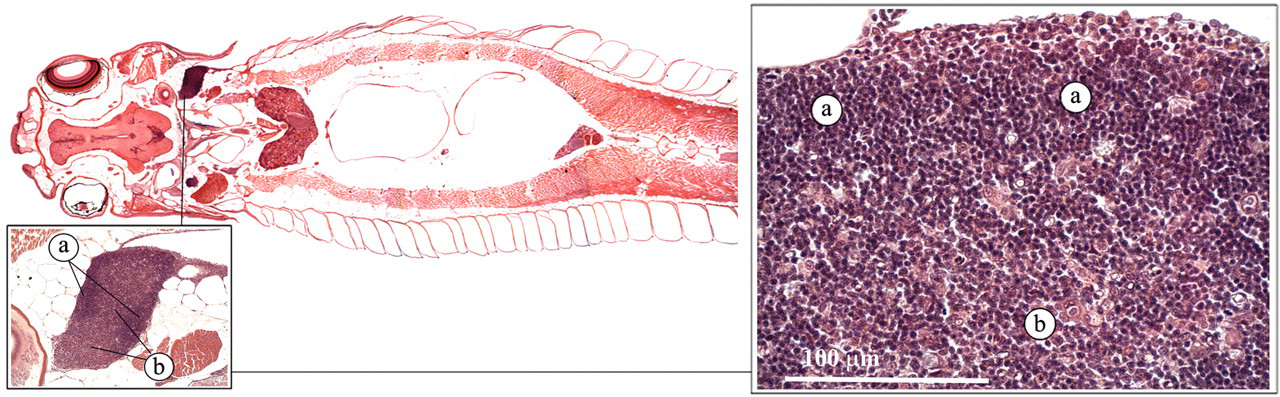
Thymus. (a) Cortex; (b) medulla.
Heart
In the zebrafish, the heart is situated anterior of the main body cavity and ventral to the esophagus (Poppe and Ferguson 2006; Roberts and Ellis 2001). Deoxygenated venous blood enters the sinus venosus (Figures 5 and 6 ). The wall of the sinus venosus is thin and is mainly composed of collagenous connective tissue (Figure 6a). The blood subsequently passes through the sino-atrial valve into the atrium. The atrium has a thin, muscular wall, and thin trabeculae form a loose meshwork in the lumen (Figure 6b). Contraction of the atrium and dilation of the ventricle forces the blood into the ventricle via the atrioventricular valve (Figure 6c).The ventricle has a much thicker wall than the atrium (Figure 6d). There is a compact outer layer of muscle and a spongy inner layer with numerous trabeculae. Contraction of the ventricle generates a relatively high pressure, and the blood is pumped into the onion-shaped bulbus arteriosus via the ventricular-bulbar valve (Figure 6e). The bulbus arteriosus has a thick wall consisting of fibro-elastic tissue and some smooth muscle fibers (Figure 6f). Because of its elasticity, it can distend considerably, thereby dampening the ventricular pulse pressure (Hu et al. 2001). From the heart, the ventral aorta distributes blood to the gills via the afferent branchial arteries.
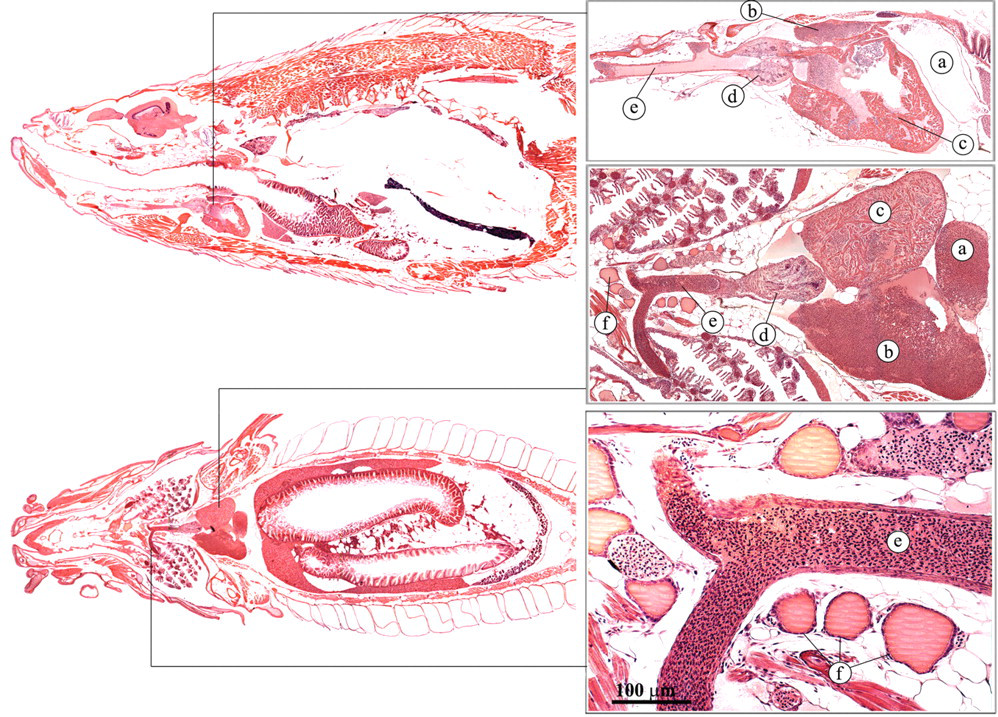
Heart and thyroid. (a) Sinus venosus; (b) atrium; (c) ventricle; (d) bulbus arteriosus; (e) ventral aorta; (f) thyroid follicles.
Heart. (a) Wall of the sinus venosus; (b) atrium; (c) leaflet of atrio-ventricular valve; (d) ventricle; (e) leaflet of bulboventricular valve; (f) bulbus arteriosus.
Thyroid
As in mammals, the thyroid follicles of the zebrafish are usually round to oval, with low cuboidal epithelium (Figure 5; Leatherland and Ferguson 2006; Roberts and Ellis 2001). In contrast to mammals, the follicles are not concentrated together but are diffusely distributed along the ventral aorta. The thyroid hormone is very similar to the thyroid hormone of higher animals and has a stimulatory effect on many metabolic processes.
Gills
The gills (Figure 7 ) play an important role in the oxygenation of the blood (Roberts and Ellis 2001; Speare and Ferguson 2006). Water is passed in, through the mouth, over the gills and out through the opercula. The waterflow is driven by alternate expansion and contraction of the buccal and opercular chambers. Blood flows via the afferent filament arteries of the primary lamellae into the blood spaces of the secondary lamellae, where CO2 is released into the water and O2 is taken up. The secondary lamellae consist of one layer of epithelial cells, supported, and separated, by pillar cells. The oxygenated blood leaves the secondary lamellae by efferent lamellar arteries to feed the dorsal aorta, from where it is distributed to all tissues. In addition to their role in the oxygenation of blood, the gills also play an important role in acid-base balance, osmoregulation, and the excretion of waste products.
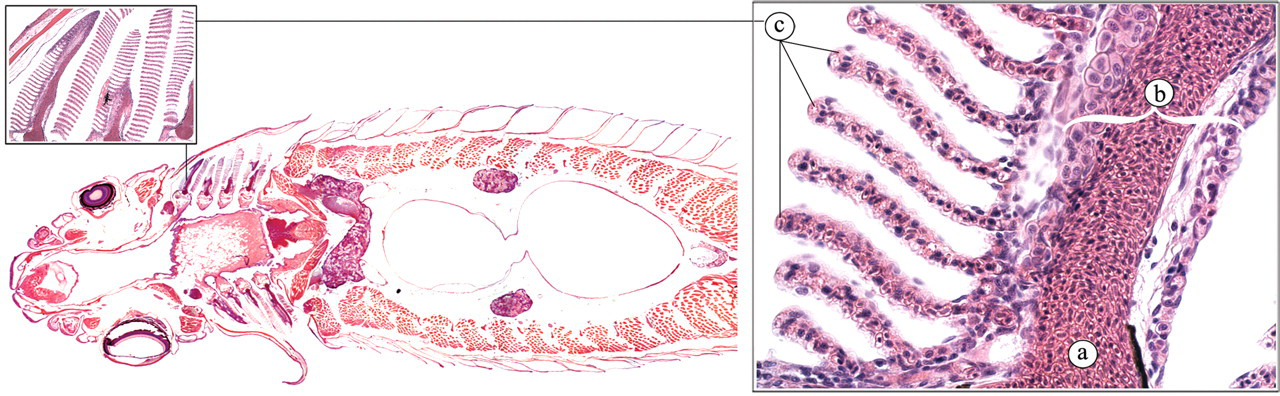
Gills. (a) Lamellar artery; (b) primary lamella; (c) secondary lamella.
Kidney
The zebrafish kidney lies in a retroperitoneal location, just ventral of the vertebral column. It has distinct head and trunk regions. Similar to the mammalian kidney, it has nephrons with a glomerulus, proximal tubules, distal tubules, and collecting ducts (Figure 1). However, the distal tubules are difficult to distinguish from the proximal tubules with routine hematoxylin and eosin (H&E) staining (Reimschuessel and Ferguson 2006; Roberts and Ellis 2001). The renal interstitium contains hematopoietic cells. Endocrine cells (interrenal and chromaffin cells) can be found along the major blood vessels in the anterior part of the kidney (Figure 8 ).
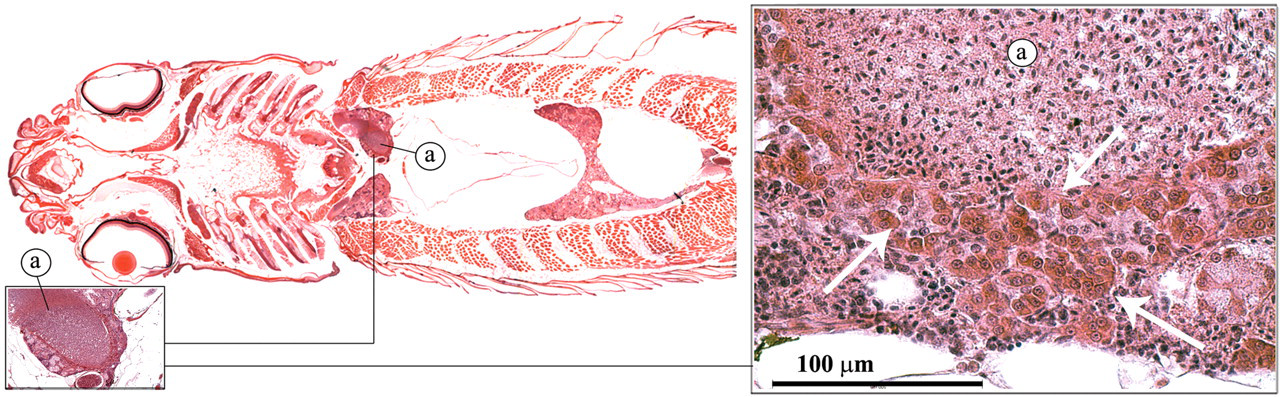
Interrenal and chromaffin cells (arrows). Posterior cardinal vein (a).
Interrenal and Chromaffin Cells
Interrenal cells represent the equivalent of the mammalian adrenal cortex (Leatherland and Ferguson 2006). These cells line the major blood vessels (posterior cardinal veins) of the anterior head of the kidney (Figure 8). They have a strong eosinophilic granular cytoplasma and produce corticosteroids, including cortisol. Cortisol has a potent effect on the intermediary metabolism and is an important factor in stress response.
Chromaffin cells are homologous to the adrenal medulla of mammals (Leatherland and Ferguson 2006). They are mixed among the interrenal cells and cannot be clearly distinguished with H&E staining in zebrafish. The chromaffin cells sythesize and secrete catecholamines such as epinephrine and norepinephrine. Catecholamines play a role in the regulation of the rate and the force of the cardiac constriction. In addition, catecholamines have been shown to bring about systemic vasoconstriction and branchial lamellar vasodilatation in teleosts. Furthermore, they can alter the respiratory functions of blood and the activity of chloride cells in the gills (Leatherland and Ferguson 2006).
Corpuscle of Stannius
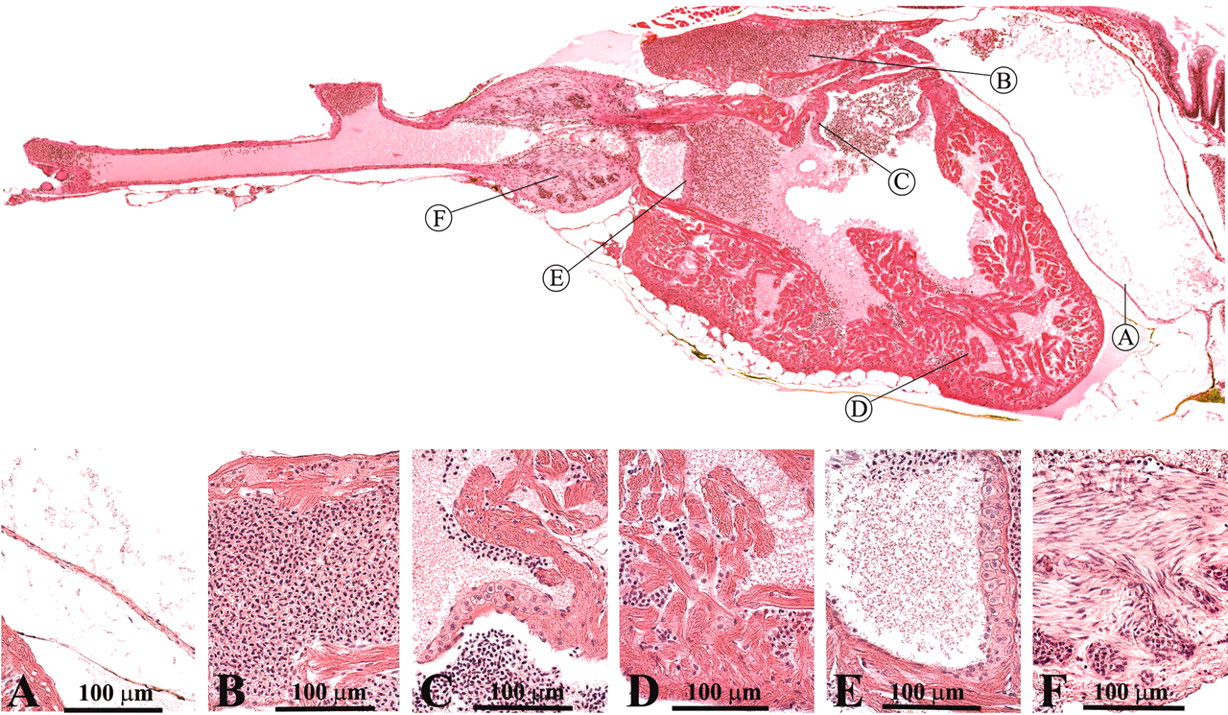
The corpuscle of stannius is a paired organ of endocrine tissue that is located in the caudal part of the renal parenchyma (Figure 9 ; Leatherland and Ferguson 2006; Roberts and Ellis 2001). The endocrine cells secrete hypocalcin, a hormone that blocks calcium uptake by the gills. In addition, the organ secretes substances that elevate the arterial blood pressure and may play a role in osmoregulation.
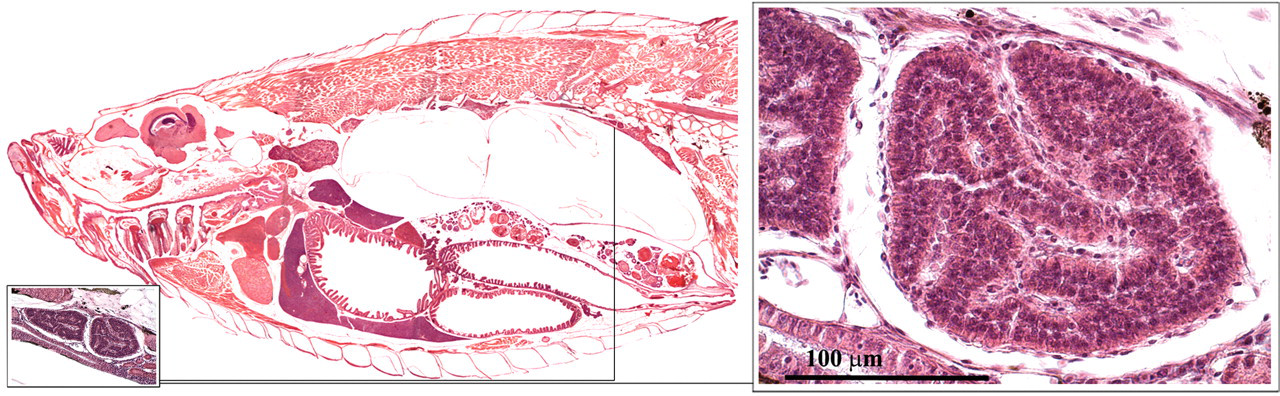
Corpuscle of stannius.
Ultimobranchial Gland
The ultimobranchial gland is a paired organ that is derived from the last branchial pouch in the embryonic fish (Leatherland and Ferguson 2006; Roberts and Ellis 2001). In the adult zebrafish, it is located just ventral of the esophagus, in the transverse septum, separating the heart from the abdominal cavity (Figure 10 ). It comprises small follicles of columnar cells that produce calcitonin, which plays a role in the regulation of calcium levels.
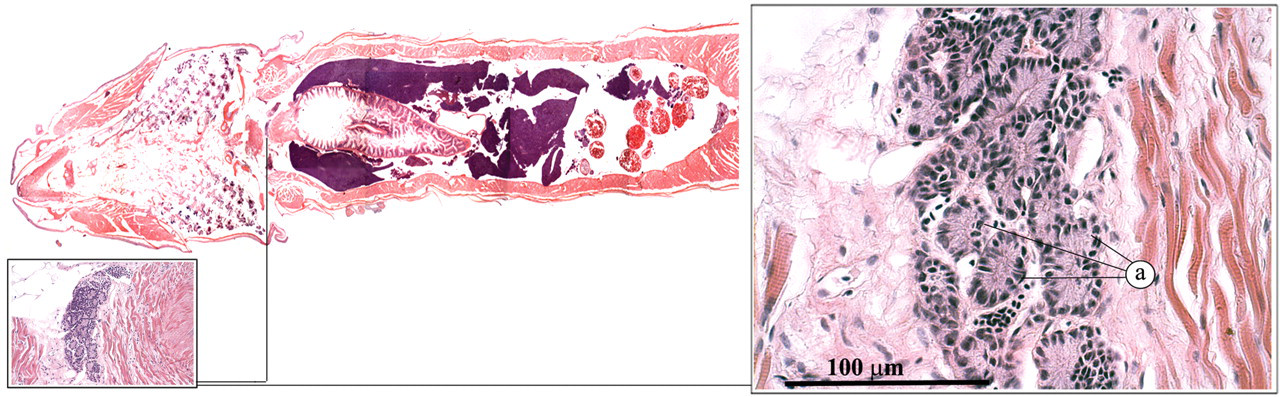
Ultimobranchial gland: follicles (a).
The Gastrointestinal System
Mouth and Buccal Cavity
The mouth and buccal cavity are shared by the respiratory and digestive systems (Roberts and Ellis 2001). The digestive function is confined to selection, seizure, and orientation of food for transfer to the intestines. In the zebrafish, the mouth and the perioral regions have many taste buds. The lining of the buccal cavity consists of mucoid epithelium on a thick basement membrane with numerous goblet cells.
Pharyngeal Pad and Teeth
In the zebrafish, the buccal cavity leads into the esophagus, which encompasses blind diverticula (esophageal sacs), a pharyngeal pad, and teeth (Figures 11 and 12 ) where food can be ground (Roberts and Ellis 2001). The teeth consist of an enamel coating, a dentine layer, and a pulp core.
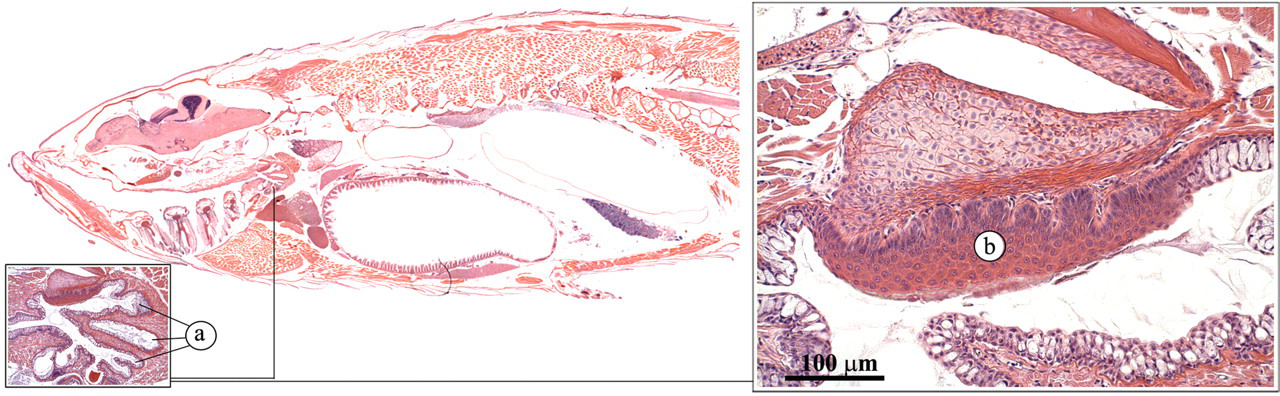
Pharyngeal pad. (a) Esophageal sacs; (b) pharyngeal pad.
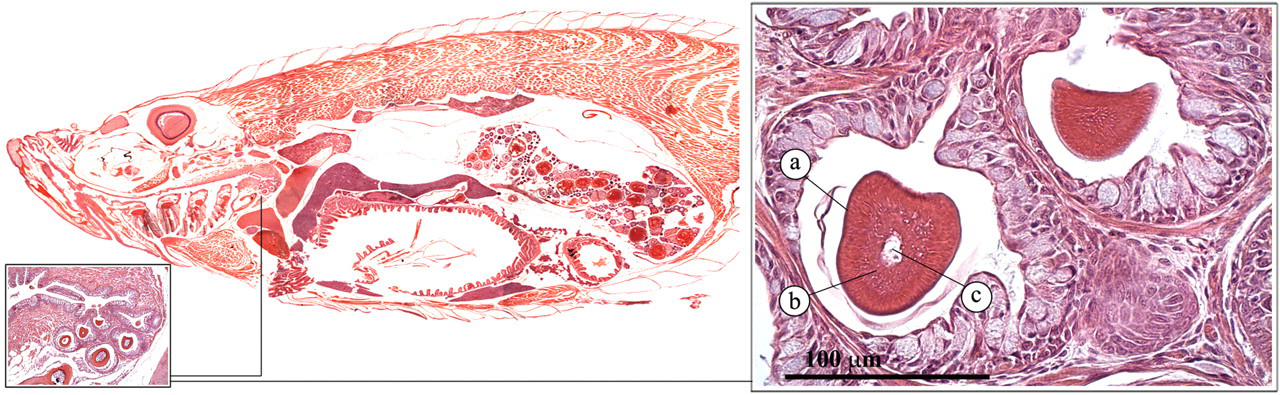
Teeth. (a) Enamel coating; (b) dentine layer; (c) pulp core.
Intestine
The intestine of the zebrafish consists of one long tube that folds twice in the abdominal cavity. The intestine starts with a wide lumen that progressively becomes smaller in a rostral-to-caudal direction. No stomach, small intestine, or large intestine can be distinguished. However, differences can be found in the morphology of the mucosa columnar epithelial cells and the number of goblet cells, suggesting functional differentiation (Figure 13 ). The intestine has numerous folds that become progressively shorter in a rostral-to-caudal direction. Proportionally, these folds are significantly larger than the finger-like intestinal villi of mammals and other amniotes (Wallace et al. 2005). Columnar-shaped absorptive enterocytes are the most numerous in the zebrafish intestinal epithelium. Goblet cells are the second most populous epithelial cell type.
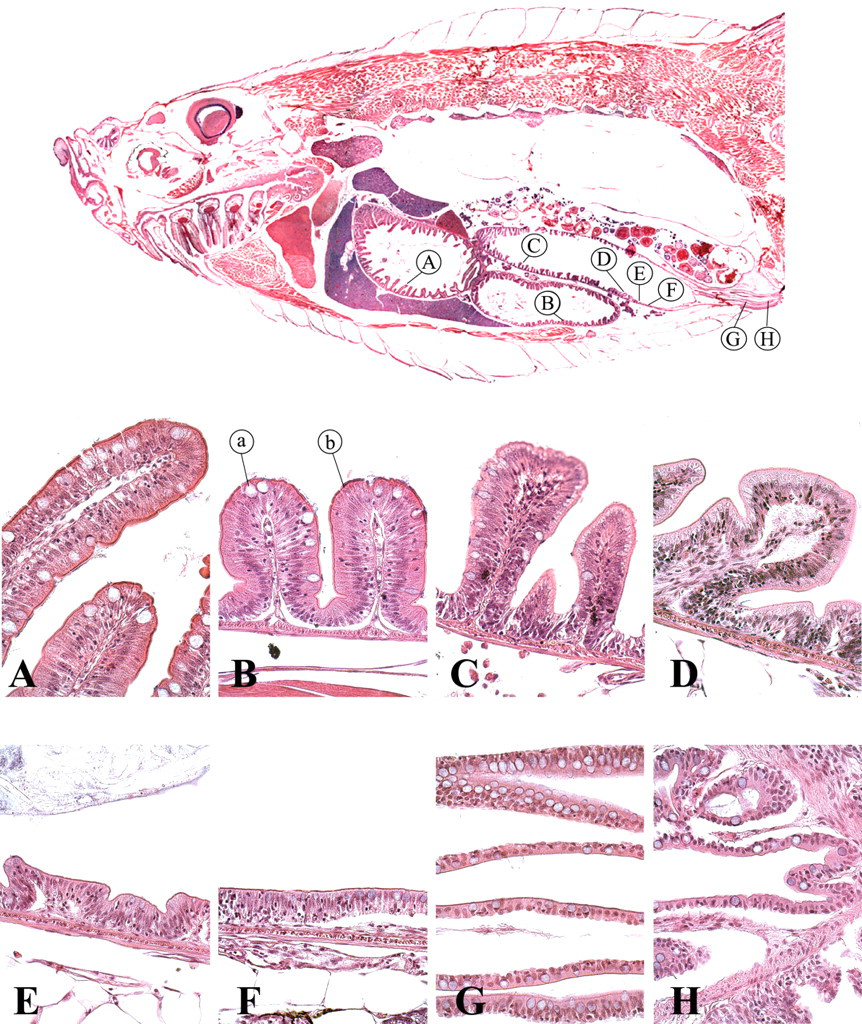
Intestine. (a) Goblet cell; (b) columnar-shaped absorptive enterocyte.
Gas Bladder
The gas bladder, or swim bladder, is derived embryologically from the upper digestive tract but has no digestive function (Lumsden 2006; Roberts and Ellis 2001). In the zebrafish, it plays a key role in buoyancy. The gas bladder is located ventrally of the kidney and is divided into two compartments (Figure 14 ). It is composed of a single layer of columnar, surfactant-producing epithelium that overlies a thin adventitia, a muscular layer composed of smooth muscle and a submucosal layer, where most blood vessels are found. In the columnar epithelium, numerous rodlet cells can be detected. Rodlet cells can be found in many species of teleost and are most commonly found within the gills, intestine, bile ducts, and bulbus arteriosis (Speare and Ferguson 2006). The rodlet cells are viewed by some as parasites, but since they fail to elicite any inflammatory response, most histologists view them as normal cells. In the zebrafish, a connection (pneumatic duct) is retained between the swim bladder and the esophagus, allowing the fish to fill up the swim bladder by gulping in air (Finney et al. 2006).
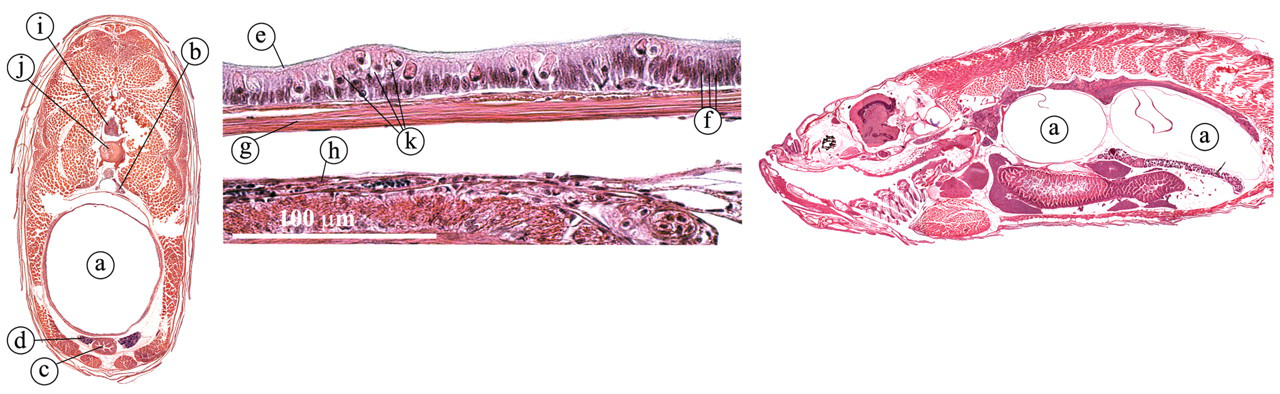
Gas bladder. (a) Gas bladder; (b) kidney; (c) intestine: (d) ductus deferens; (e) surfactant; (f) columnar epithelium cells; (g) smooth muscle layer; (h) submucosal layer; (i) spinal cord; (j) vertebral column; (k) rodlet cells.
Liver and Gall Bladder
The zebrafish liver encompasses three lobes that lie along the intestinal tract (Figure 15 ). Similar to the mammalian liver, the teleost liver plays an important role in the metabolic homeostasis of the body (Evensen 2006; Roberts and Ellis 2001). This role includes the processing of carbohydrates, proteins, lipids, and vitamins. In addition, it also plays a key role in detoxification and the synthesis of serum proteins such as albumin, fibrinogen, complement factors, and acute-phase proteins. The zebrafish liver differs from the mammalian liver in that the hepatocytes are not clearly organized in cords or lobules and the typical portal triads are not apparent. In addition, the zebrafish liver does not have Kuppfer cells. Furthermore, a clear distinction can be made between the male and female liver in the adult zebrafish. The female hepatocytes are very basophilic (Figure 15c) as a result of the production of vitellogenin (Van der Ven et al. 2003). The biliary system also differs from that of mammals in that intracellular canaliculi occur, which eventually anastomose to form bile ducts. The bile ducts fuse and ultimately form the gall bladder, which contains greenish bile that is conducted to the intestine via the common bile duct (Figure 16 ).
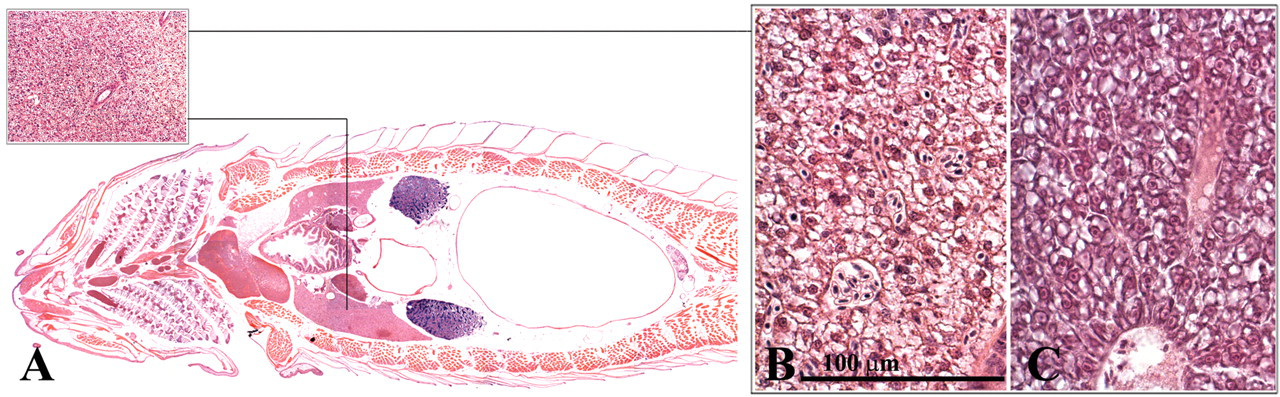
Liver. (a) Dorsal view, male; (b) male liver; (c) female liver.
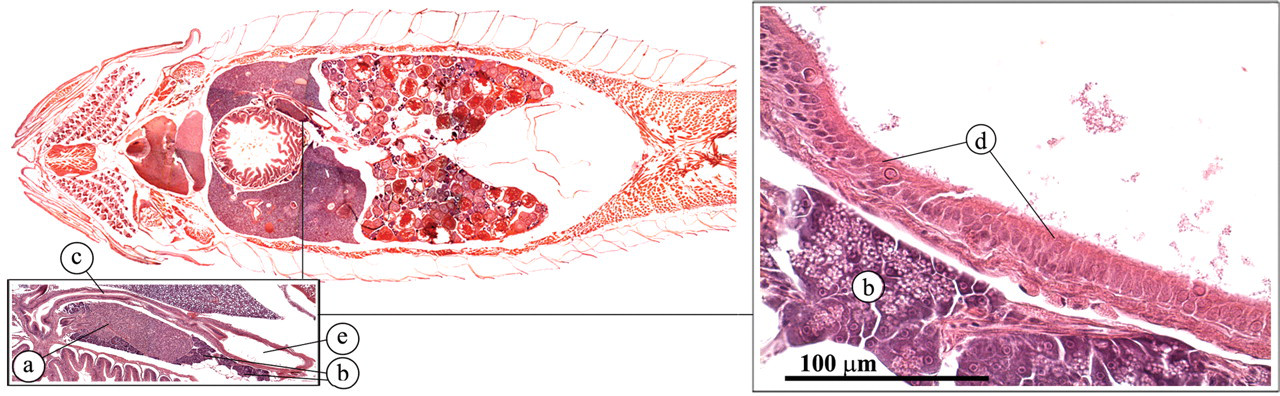
Gall bladder. (a) Endocrine pancreas (Brockman body); (b) exocrine pancreas; (c) common bile duct; (d) transitional gall bladder epithelium; (e) gallbladder.
Pancreas
The zebrafish does not have a discrete pancreas. Exocrine pancreatic tissue can be found scattered along the intestinal tract (Figures 16 and 17 ). The acinar structure of the exocrine pancreas is very similar to that of mammals and comprises cells with a very dark, basophilic cytoplasm (Roberts and Ellis 2001). In actively feeding fish, they contain large numbers of bright eosinophilic, secretory granules.
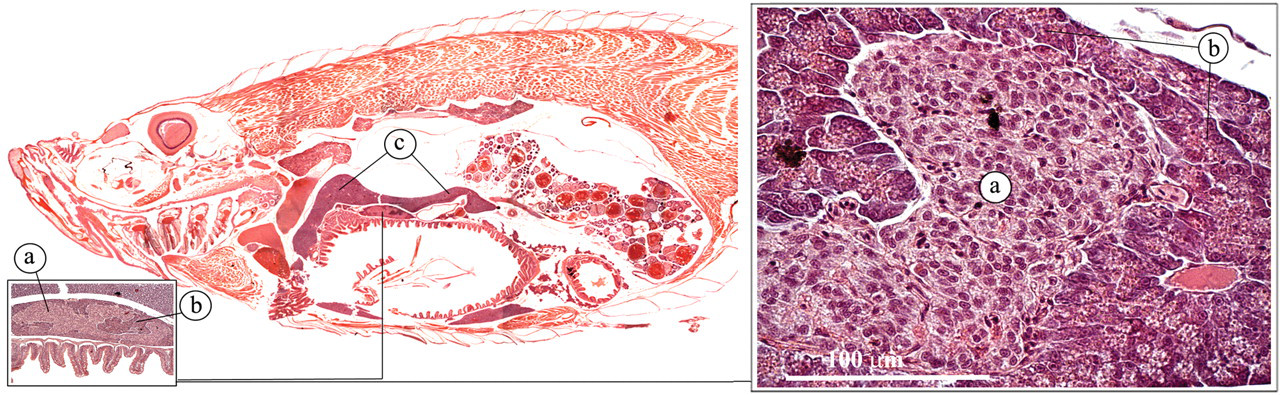
Pancreas. (a) Endocrine pancreas (Brockman body); (b) exocrine pancreas; (c) liver.
In the AB zebrafish strain that we analyzed, only one major islet of Langerhans could be distinguished, the Brockman body. The endocrine pancreatic tissue encompasses α-cells (producing glycagon-like peptide), β-cells (producing insulin), and δ-cells (producing somatostatin).
Reproductive Tract
Testes
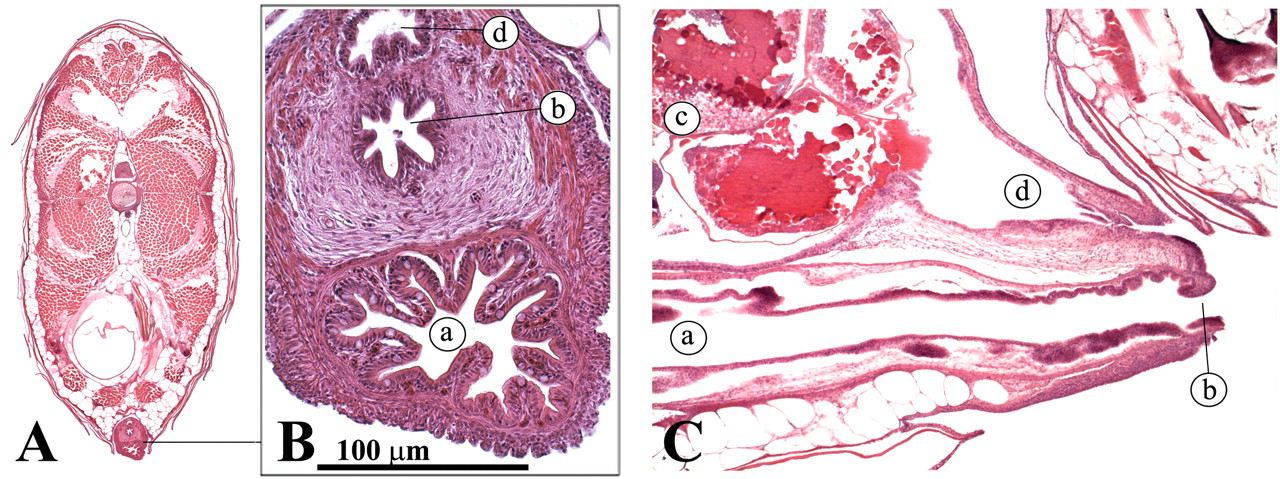
Zebrafish testes are lateral, paired organs that comprise a series of tubules or blind sacs, which are lined with spermatogenic epithelium (Roberts and Ellis 2001) (Figure 18 ). Spermatogenesis occurs in cysts that are formed when a single spermatogonium is completely surrounded by the cytoplasmic projections of one or two Sertoli cells. In contrast to mammalian spermatogenesis, a given Sertoli cell is in contact with only one germ cell clone (Leal et al. 2009; Schulz et al. 2010). Different germ cell cysts can be observed during the spermatogenic process. Mature spermatozoa are conducted to the genital orifice by two ducti deferens that merge caudally (Figure 19 ; Leatherland and Ferguson 2006).
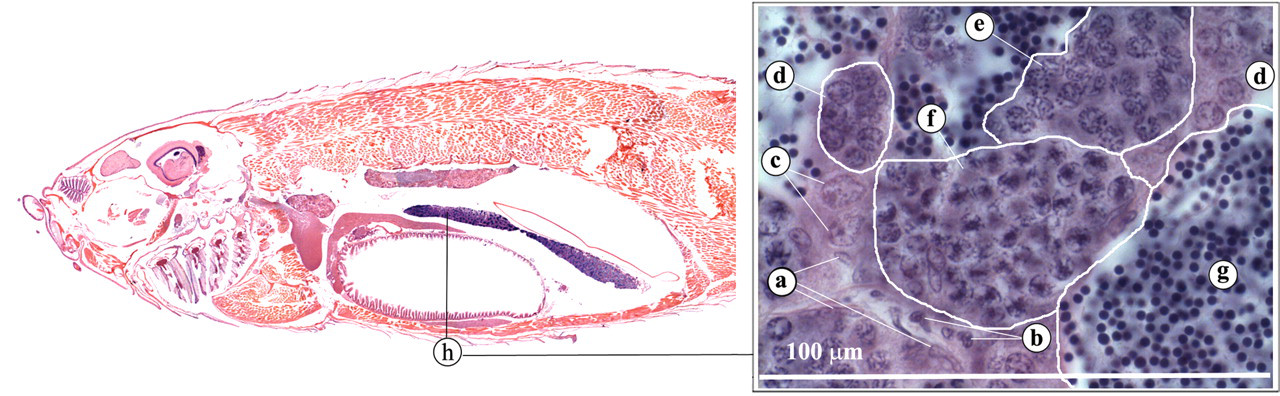
Testis. (a) Sertoli cells; (b) Leydig cells; (c) spermatogonia type A; (d) spermatogonia type B; (e) pachytenic spermatocytes; (f) dyplotenic spermatocytes and first meiotic division; (g) mature spermatids; (h) testis.
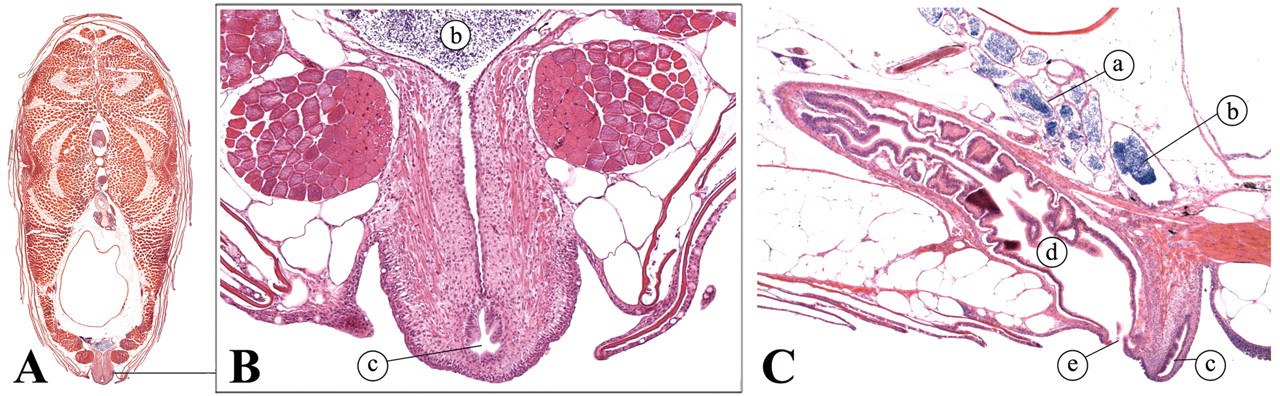
Male reproductive tract. (A) Transverse view; (B) Transverse view; (C) lateral view. (a) Ducti deferens; (b) fused ducti deferens; (c) genital orifice; (d) intestine; (e) anus.
Ovaries
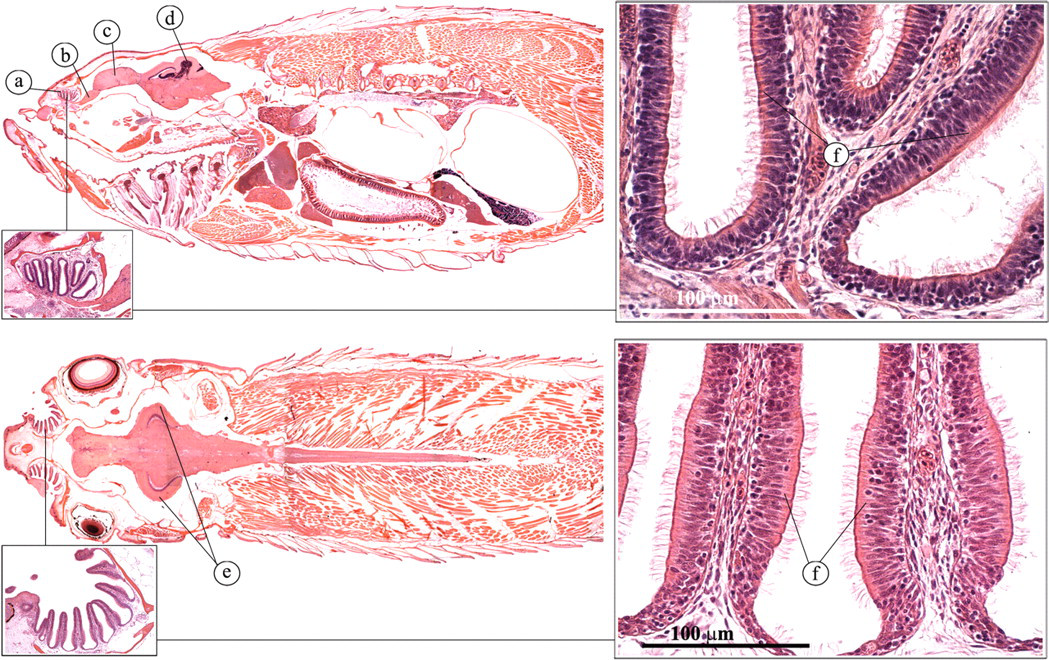
Ovaries are paired, elongated structures (Figure 20 ). Histological examination of ovarian tissue commonly reveals eggs at different stages of development (Wang and Ge 2004). A short oviduct conducts the eggs to the outside (Figure 21 ).
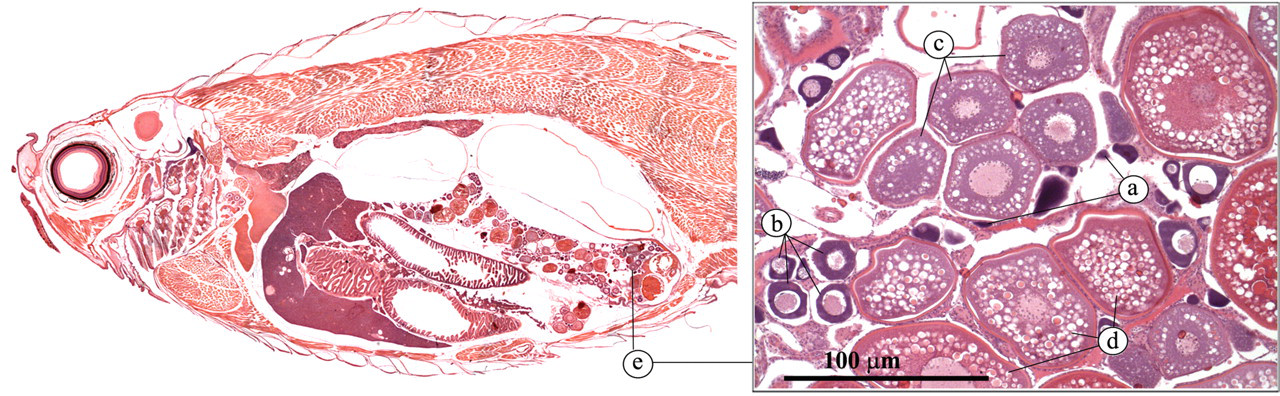
Ovary. (a) Oogonium; (b) pre-vitellogenic follicle; (c) vittelogenic follicle; (d) pre-ovulatory follicle; (e) ovary.
Female reproductive tract. (A) Transverse view; (B) transverse view; (C) lateral view. (a) Intestine; (b) anus; (c) ovary; (d) oviduct.
Brain and Specialized Sensory and Endocrine Organs
The zebrafish brain has been described in detail by Wullimann et al. (1996). It is very similar in its basic components to the brain of higher animals and can be divided into five regions: the telencephalon, the diencephalons, the mesencephalon, the metencephalon, and the myelencephalon (Speare and Frasca 2006). The telencephalon is responsible for olfaction and for aspects of memory, reproductive behavior, feeding behavior, and color vision (Roberts and Ellis 2001). The olfactory organ is directly connected to the telencephalon via the olfactory bulb (Figure 22 ).
Olfactory organ. (a) Olfactory organ; (b) olfactory bulb; (c) telencephalon; (d) cerebellum; (e) corpora bigemia; (f) ciliated columnar epithelium.
Olfactory Organ
The olfactory organ consists of paired pits on the snout with a single opening, transversed by a cusp of skin that divides it into an anterior inlet and a posterior outlet (Roberts and Ellis 2001). Swimming allows passage of water through the sacs and over the olfactory epithelium. The latter is composed of folds or fingers, which increase the surface area of the sensory tissue (Figure 22). The actual olfactory tissue consists of ciliated columnar epithelium cells. Axons from the olfactory bulb collect from the bases of these receptor cells to form the olfactory tracts to the telencephalon.
Pineal Gland
The diencephalons can be subdivided into three components: the epithalamus, the thalamus, and the hypothalamus. The epithalamus encompasses the pineal gland (Figure 23A) and the saccus dorsalis (Figure 23B). The pineal gland is a light-sensitive neuroendocrine structure that projects from the roof of the telencephalon (Leatherland and Ferguson 2006).
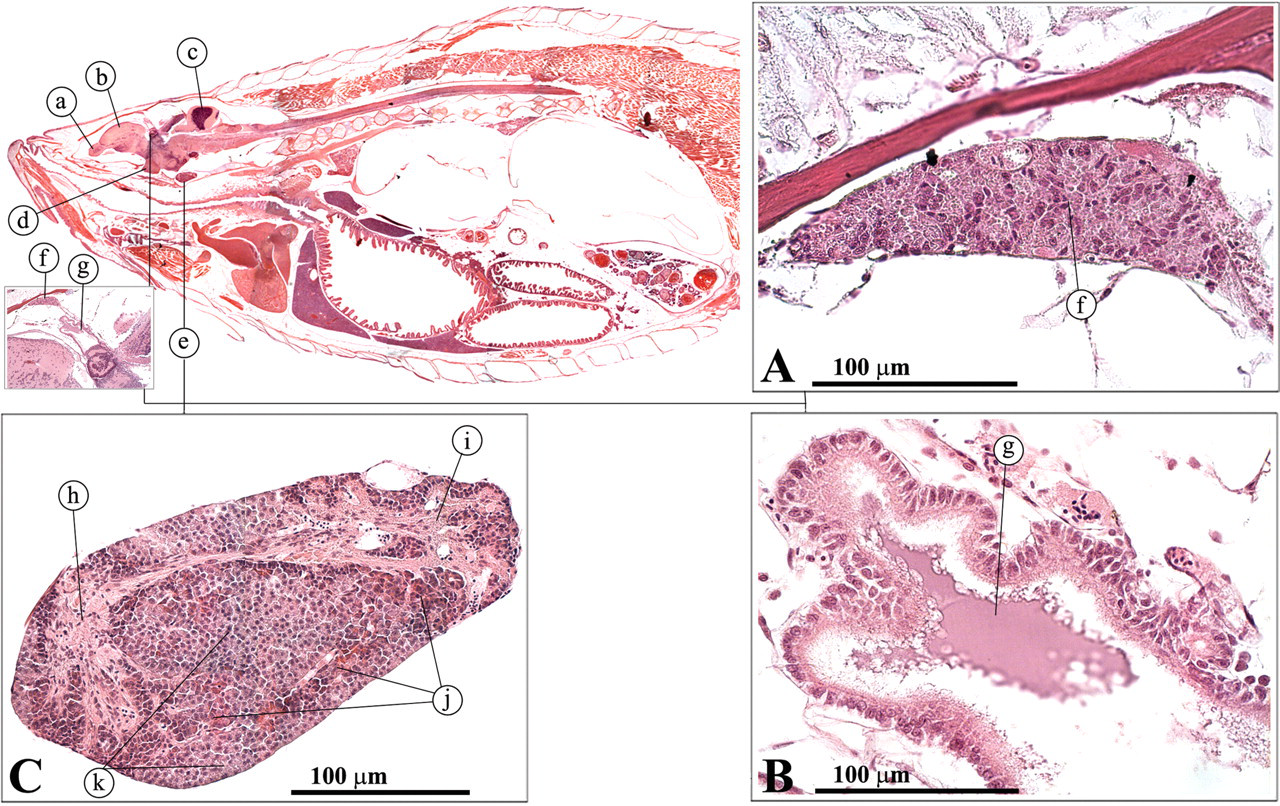
(A) Pineal gland; (B) saccus dorsalis; (C) pituitary gland. (a) Olfactory bulb; (b) telencephalon; (c) cerebellum; (d) optic nerve; (e) pituitary gland; (f) pineal gland; (g) saccus dorsalis; (h) anterior pars nervosa; (i) posterior pars nervosa; (j) pars intermedialis; (k) pars distalis.
As in mammals, the pineal gland secretes the hormones indolamine and melatonin, as well as neurotransmitter substances. It appears to play a key role in the control of daily and seasonal rhythms such as the regulation of reproduction, growth, and seasonal migration. The thalamus and the hypothalamus function mainly as correlation centers for sensory inputs such as gestation and olfaction.
Pituitary Gland
The pituitary gland is a complex neuroepithelial structure (Figure 23C). As in other vertebrates, it is derived embryologically from a down push of the floor of the hypothalamus (forming the pars nervosa) and an upgrowth of the roof of the mouth, forming the pars distalis and pars intermedia of the adult (Leatherland and Ferguson 2006; Roberts and Ellis 2001). The pars nervosa comprises glial cells and neurosecretory axons, which originate from neurons lying in the hypothalamus. Two pars nervosa hormones, isotocin and arginine vasotocin, are known in the teleost, but their exact function remains to be elucidated. Prolactin and corticotrophic hormone–secreting cells can be found in the rostral pars distalis. Thyroid-stimulating hormone–secreting cells can be found in the proximal pars distalis. The pars intermedia contains melanotrophic cells that secrete α−MSH, β−endorphin, and β−hypotrophic hormone.
Eye
The mesencephalon is relatively large and anatomically subdivided into the optic tectum, which forms the roof of the third ventricle, and the tegmentum, which is its floor (Roberts and Ellis 2001). The optic tectum is relatively large and is divided by a longitudinal furrow into two globular structures, the corpora bigemina (Figure 22). These structures in particular are concerned with the reception and the coordination of optic nerve inputs (Figure 23).
The zebrafish eye is similar to the eye of all other vertebrates (Figure 24 ) (Koppang and Bjerkås 2006; Roberts and Ellis 2001). It consists of three layers: (1) the tunica fibrosa, which encompasses the cornea and the sclera; (2) the tunica vasculosa, which encompasses the choroid, the choroid rete, and the iris; and (3) the retina.
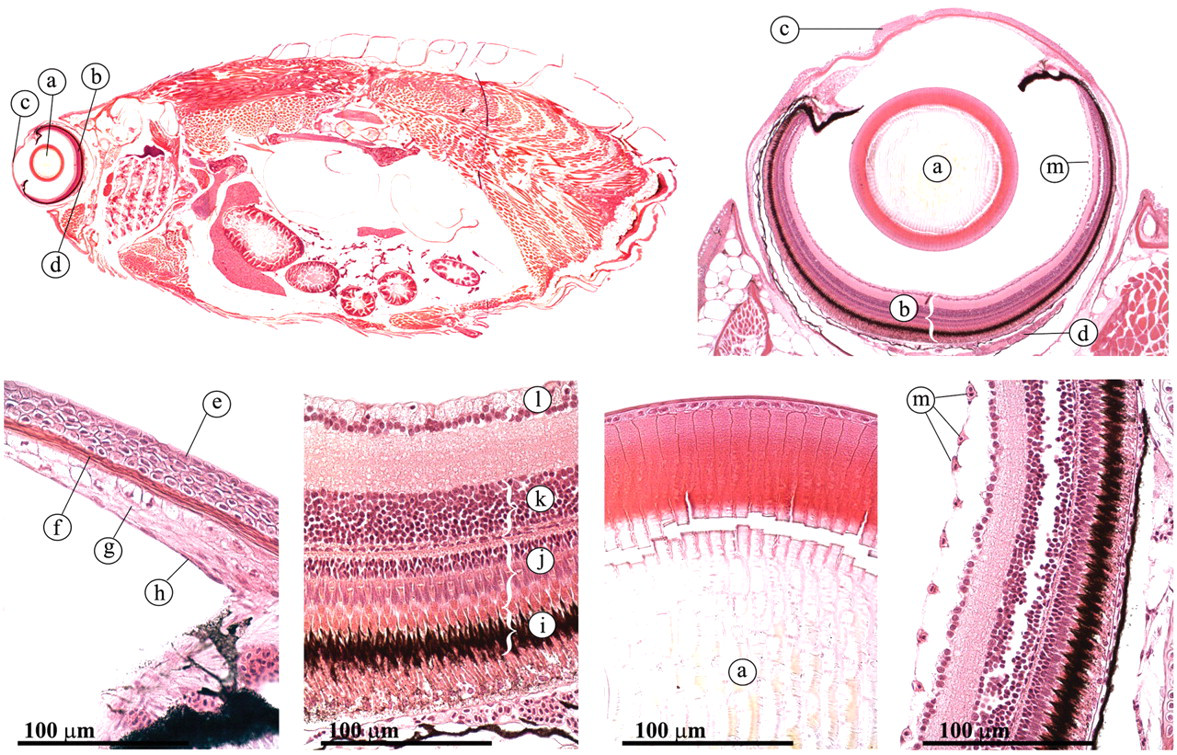
Eye. (a) lens; (b) retina; (c) cornea; (d) choroid rete; (e) nonkeratinizing epithelial cells; (f) Bowman’s membrane; (g) substantia propria; (h) endothelium cells; (i) pigment epithelium; (j) cones and rods; (k) bipolar cells; (1) ganglion cells; (m) preretinal vessels.
Cornea
Compared to terrestial animals, the cornea is relatively flat. It consists of nonpigmented, stratified squamous nonkeratinizing epithelial cells, attached to a thick basement membrane that is considered to be analogous to the Bowman’s membrane in mammals (Figure 24). The stroma rests on a distinct Descemet’s membrane, which is difficult to detect with H&E staining. The innermost layer of the cornea, the endothelium, serves to maintain corneal transparency through its dehydrating pump function.
Lens
The teleost lens is not lenticular, but completely spherical (Figure 24). It protrudes partially through the iris, providing a wide-angle of view.
Retina
Several layers can be distinguished in the retina (Figure 24): (1) the retinal pigment epithelium, in which photoreceptor cells (rods and cones) embed their outer segments; (2) the external nuclear layer, which contains the nuclei of the rods and cones; and (3) the bipolar cells that connect the rods and cones to (4) the ganglion cells, which send their axons through optic fibers that fuse into the optic nerve, which is connected to the brain. Whereas the retina is avascular in most species of teleost, the zebrafish has pre-retinal vessels.
Pseudobranch and Choroid Rete
The pseudobranch is derived from the first gill arch (Roberts and Ellis 2001; Speare and Ferguson 2006). It is composed of similar arrays of capillaries, alternating with rows of slender fibroblast cells (Figure 25 ). The opthalmic artery receives its blood from the pseudobranch and branches out in the capillaries of the choroid rete. The choroid rete is a capillary countercurrent system formed by the ophthalmic artery and vein.
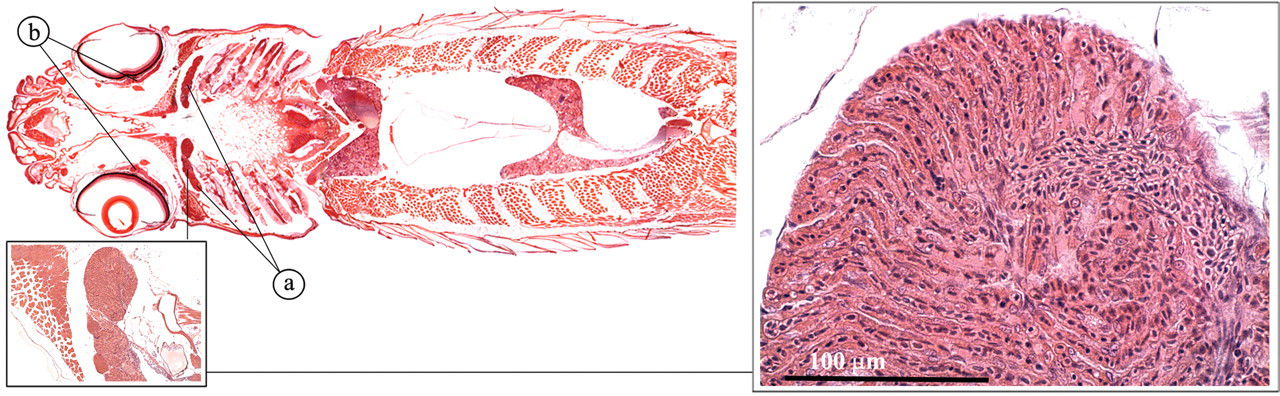
Pseudobranch and choroid rete. (a) Pseudobranch; (b) choroid rete.
The pseudobranch and the choroid rete appear to elevate the arterial oxygen pressure by enzymatically acidifying the arterial blood via the action of carbonic anhydrase.
In addition, the pseudobranch may have sensory properties and play a role in osmoregulation. The choroid rete may also have immune capabilities, since it filters both afferent and efferent blood and contains cells that seem to be involved in antigen trapping.
Otolith Organ
The cerebellum, or metencephalon, is associated with reception and coordination of proprioceptive and balance stimuli (Roberts and Ellis 2001). It has two components: a vestibulolateralis lobe, which receives stimuli from the otolith organ and lateral line inputs; and the corpus cerebelli, more dorsally situated, which receives sensory stimuli via the spinal cord from extremities and proprioreceptors.
The otolith organ is very important in the perception of sound, acceleration, and change in equilibrium (Figure 26 ; Roberts and Ellis 2001). It consists of three interconnecting chambers (utriculus, sacculus, and lagena) that are filled with endolymph. In each chamber lies a calcified “stone,” the otolith, on sensory epithelium cells. Movement of the otolith by force of gravity or low-frequency sound vibrations stimulates these sensory cells.
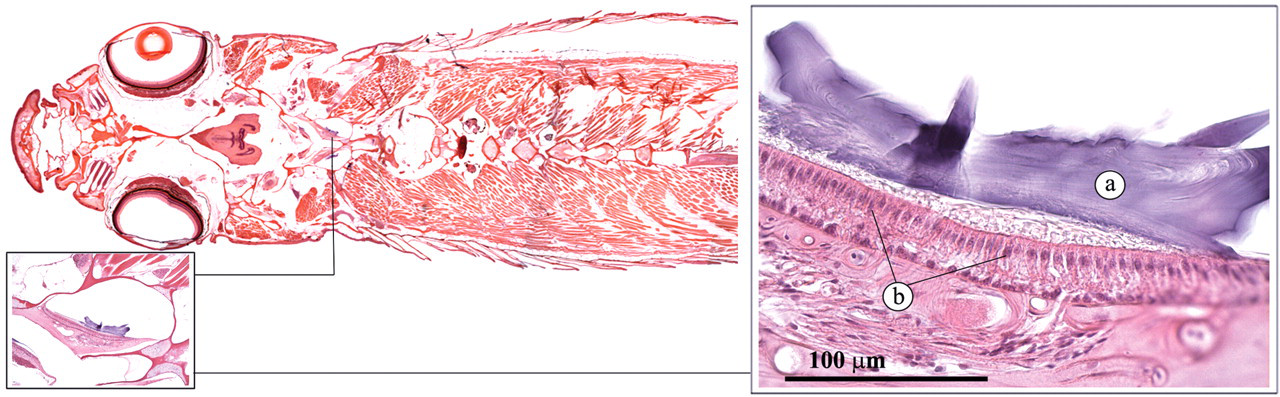
Otolith organ. (a) Otolith; (b) sensory epithelium.
Musculoskeletal System and Skin
The main vertebral column develops from the notochord into a series of connected vertebrae (Figure 27 ; Roberts and Ellis 2001; Turnbull 2006). The vertebrae of the main body have ribs on the transverse processes. Throughout the spine, arches above the main vertebral bodies accommodate the spinal cord. The skeletal muscles bind to ribs and skin (Figure 28 ), thereby directing the energy of muscular contraction into a propulsive force. Unlike mammals, the entire surface of the fish is a mucous membrane except for a little keratinized epithelium on the jaw and fins.
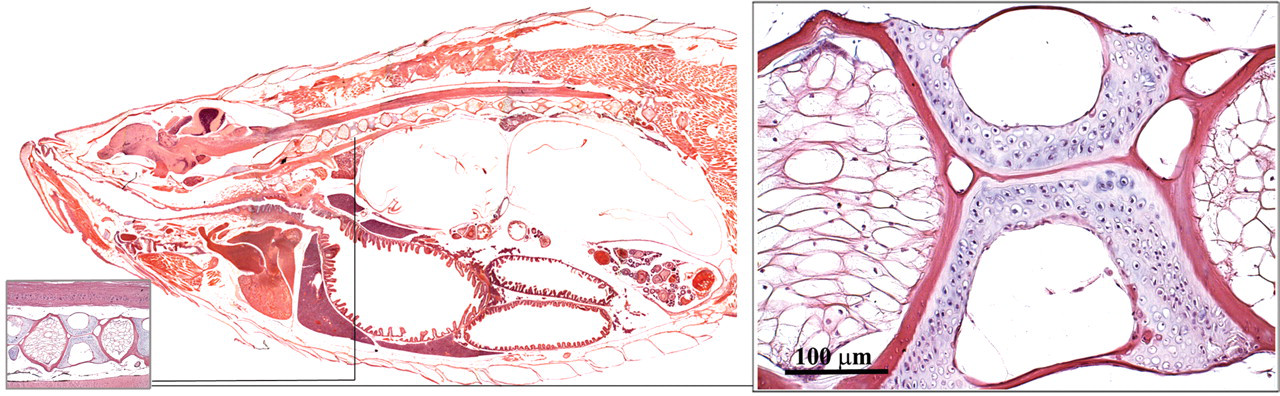
Vertebral column.
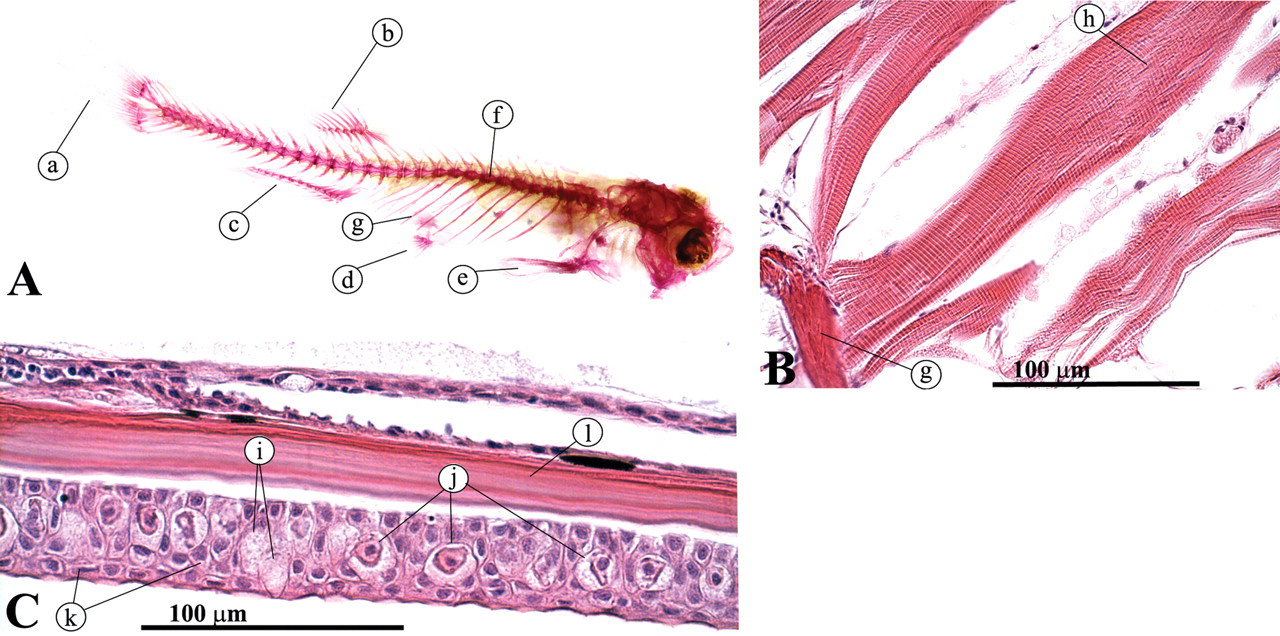
(A) Alizarin Red staining; (B) dorsal view of skeletal muscle attached to ribs; (C) lateral view, skin. (a) Tail fin; (b) dorsal fin; (c) anal fin; (d) pelvic fin; (e) pectoral fin; (f) vertebral column; (g) rib; (h) skeletal muscle; (i) goblet cells; (j) alarm cells; (k) keratinocytes; (1) scale.
Conclusion
The zebrafish has become an important animal model for understanding human development, disease, and toxicology. The identification of thousands of early developmental zebrafish mutants has established the zebrafish as a mainstream model in developmental biology (Lieschke and Currie 2007). The sequencing of its genome and the relative ease with which gene modifications can be performed have led to the creation of numerous zebrafish models for studying human diseases such as cancer, heart disease, Alzheimer’s, Parkinson’s, and muscular dystrophy (Best and Alderton 2008; Guyon et al. 2007; Lieschke and Currie 2007; Zon and Peterson 2005). In addition, the zebrafish is increasingly used for assessing drug toxicity and safety (Eimon and Rubinstein 2009; McGrath and Li 2008). However, before we can use the full potential of the zebrafish model, we must advance our knowledge of zebrafish diseases and pathology (Spitsbergen and Kent 2003). In this context, it is essential to understand normal zebrafish physiology, anatomy, and histology. Hopefully the current article will serve as a first roadmap to further this knowledge.
Footnotes
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The author(s) received no financial support for the research and/or authorship of this article.
Acknowledgments
We would like to thank E. C. M. van Oostrum, J. D. Jansen, T. J. S. van Olmen, S. T. van den Broek, D. Leijdekkers, R. Jansen, M. Stoepker, and S. van Essen for their technical assistance.
