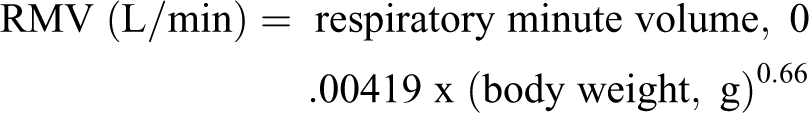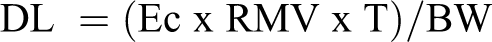Abstract
Afrezza delivers inhaled insulin using the Gen2 inhaler for the treatment of patients with type 1 and type 2 Diabetes. Afrezza was evaluated in long-term nonclinical pulmonary safety studies in 2 toxicology species. Chronic inhalation toxicology studies in rat (26 weeks) and dog (39 weeks) and an inhalation carcinogenicity study in rats were conducted with Technosphere insulin (Afrezza) and with Technosphere alone as a vehicle control. Respiratory tract tissues were evaluated by histopathology and cells expressing proliferating cell nuclear antigen (PCNA) were quantified in lungs of rats. Microscopic findings in rats exposed to Afrezza were attributed to the Technosphere particle component, were confined to nasal epithelia, and consisted of eosinophilic globules and nasal epithelial degeneration. There were no Afrezza-related changes in pulmonary PCNA labeling indices in alveoli, large bronchioles, or terminal bronchioles. Microscopic findings in rats exposed to Technosphere particles included eosinophilic globules, mucus cell hyperplasia, and epithelial degeneration in the nasal cavities. PCNA labeling indices were increased in large bronchioles and terminal bronchioles but not in alveoli. There were no Technosphere particle-related findings in the dog study. Afrezza did not exhibit carcinogenic potential in the 2-year study in rats. These nonclinical inhalation studies support the use of Afrezza in humans over extended periods.
Introduction
A wide range of injectable insulin products, used by millions of patients with type-1 and type-2 diabetes, is available globally for the treatment of diabetes. Of the rapid-acting insulins with an onset of action in 15 to 20 minutes and mean peak action in 1 to 2 hours (Humalog, Novolog, and Apidra), 1 –3 all are administered by subcutaneous injection. Afrezza, administered by inhalation, 4 has an onset of action of less than 15 minutes with mean peak action at approximately 50 minutes. In addition, despite advances in the development of patient-friendly, pen-injector devices to allow better tolerability of subcutaneous administration, injection of insulin is still viewed as an unacceptable procedure by many, 5 resulting in avoidance to self-inject, even in the absence of overt needle phobia. 6,7 Therefore, attempts to develop noninvasive routes for insulin administration emerged soon after the introduction of insulin. 8
In early studies, aerosolized insulin was effectively delivered to the alveolar region of the lung with absorption rates and decreases in glucose levels similar to those achieved with subcutaneous-delivered insulin during the fasting state in human volunteers, 4 and postprandial glucose levels were effectively controlled. 9,10 In these earlier studies with aerosolized insulin administered over short periods of time, there was no evidence of irritation, hypoglycemia, or changes in pulmonary function. 8 –15 Patient satisfaction and quality of life were found to be significantly greater in those receiving inhaled insulin versus injectable insulin. 16,17
Background of Previous Inhaled Insulin Products
Companies such as Aradigm Corporation/Novo Nordisk and Alkermes, Inc/Eli Lilly in clinical development with pulmonary administration systems for inhaled insulin announced the termination of their product’s development following the approval and subsequent withdrawal of the first US Food and Drug Administration (FDA)-approved inhaled insulin product, Exubera. Exubera was voluntarily withdrawn from the market because of low sales, possibly resulting from high costs, the large size of the device and an FDA warning regarding the potential for primary lung cancer. 12
Afrezza Safety and Efficacy
The novel excipient in Afrezza is Technosphere particles or fumaryl diketopiperazine (FDKP), which adsorbs to monomeric insulin to form ultrarapid acting Technosphere insulin or Afrezza inhalation powder. Afrezza inhalation powder administered using the Gen2 Inhaler is uniformly distributed throughout the lungs, where approximately 40% of the initial cartridge load reaches the lungs (disposition fraction). Afrezza is quickly cleared from the lungs (approximately 1 hour), mainly by absorption into the systemic circulation. Since there is an overnight washout period with use of the product, the potential for accumulation following chronic administration is minimal. 12 Insulin exposure following Afrezza administration was found to be linear over the dose range of 25 to 100 U. 18,19
Afrezza lowers postprandial glucose levels when added to current therapy in patients with inadequately controlled type 2 diabetes and results in significant A1C reduction, increase in time-in-range on Continuous Glucose Monitoring System with no significant increase in hypoglycemia and no severe hypoglycemic episodes. 20,21 Observed changes in lung function with Afrezza are small, occur early after therapy initiation, remain nonprogressive over 2 years, and were unlikely to be clinically meaningful. 22,18,19 Changes in lung function include a small decrease in Forced Expiratory Volume in 1 second (FEV1, 40 mL 95% CI). Forced Expiratory Volume in 1 second decline was noted within the first 3 months and persisted for the entire duration of therapy (up to 2 years of observation). The annual rate of FEV1 decline did not appear to worsen with increased duration of use. The observed changes in FEV1 were similar in patients with type 1 and type 2 diabetes. Current recommendations are that pulmonary function (ie, spirometry) should be assessed at baseline, after 6 months of therapy, and annually thereafter, even in the absence of pulmonary symptoms.
Afrezza is contraindicated in patients with underlying lung disease, including chronic obstructive pulmonary disease (COPD) and asthma. Afrezza is not cytotoxic in an in vitro human lung cell model and does not function as a penetration enhancer. Furthermore, Afrezza does not appear to affect the transport of insulin across cellular barriers. 7 Afrezza has been associated with less weight gain and a lower incidence of hypoglycemia compared to subcutaneous insulins, which may enhance patient satisfaction and acceptability of inhaled insulin therapy. 19,23
Potential Side Effects of Inhaled Insulin
Concern has been raised regarding possible insulin-induced carcinogenesis. Insulin-like growth factor 1 (IGF-1) has characteristics of both a circulating hormone and a tissue growth factor. The circulating IGF-1 levels vary considerably among normal individuals, and there is limited evidence from epidemiological studies that individuals with levels at the higher end of the normal range may have increased cancer risk. 21,24 –29 In the lung, IGF-1 receptors are expressed most notably in macrophages and to a lesser extent in bronchiolar and alveolar epithelia. 30 The human insulin concentrations required to bind the IGF-1 receptor are usually 50 to 100 times higher than the IGF-1 concentrations required to bind to its own receptor. 31,32 Based on the Afrezza therapeutic dose range, insulin concentrations high enough to bind the IGF-1 receptor are unlikely and provide a robust calculated safety margin of exposure.
The purpose of this research was to evaluate the respiratory safety profile of Afrezza inhaled insulin and the novel excipient, Technosphere particles, in animals by evaluating histopathology and proliferation indices from proliferating cell nuclear antigen (PCNA) labeling in respiratory tissues. These studies will be discussed with respect to the potential clinical significance of pulmonary changes in these nonclinical chronic toxicology, and carcinogenicity studies, as well as, demonstrate the importance of utilizing a non-biased technique to quantify cellular proliferative events above background levels or secondary to non-pathologic, adaptive responses to physical deposition of Technosphere particles on respiratory airways.
Materials and Methods
Summary
Technosphere Insulin or Afrezza is inhaled as a dry powder. Afrezza is comprised of insulin and a novel excipient, FDKP, the primary component of Technosphere particles. The insulin in Afrezza is human insulin (rDNA origin; manufactured by N.V. Organon). Fumaryl diketopiperazine undergoes intermolecular self-assembly in water to form Technosphere particles, and insulin is adsorbed to those particles to form Technosphere insulin. Both Technosphere particles and Afrezza were tested separately in long-term inhalation studies in rats (26-week toxicology study and 104-week carcinogenicity study) and in dogs (39-week toxicology study). Technosphere particles were tested as the vehicle control in these studies.
All long-term toxicology studies were conducted by inhalation at Charles River Laboratories (chronic rat toxicity study) and ITR Laboratory (chronic dog toxicity study and rat carcinogenicity study). These nonclinical safety studies provided a characterization of the potential adverse effects on the respiratory system using histopathology evaluations and a proliferation marker, PCNA, of both insulin, administered as Afrezza, and Technosphere particles, administered separately as the novel excipient. An air only negative control group was included in all nonclinical studies to evaluate safety of Afrezza and separately, Technosphere particles.
Animals
In the rat studies, male and female Sprague-Dawley rats, Rattus norvegicus, from Charles River Canada Inc, were evaluated. The rats were Crl; CD (SD) BR rats in the toxicity study and Crl: CD (SD) IGS BR rats in the carcinogenicity study. At the start of treatment in the 26-week toxicity study, animals were approximately 9 to 10 weeks of age with body weight ranges of 309 to 393 g and 153 to 258 g for males and females, respectively. At the start of treatment in the carcinogenicity study, the animals were approximately 6 weeks old and ranged in weight between 143 to 226 g (males) and 125 to 170 g (females). In both studies, rats were housed individually in stainless steel wire mesh-bottomed cages equipped with an automatic watering system. Targeted conditions for animal room environment and photoperiod were as follows: temperature 22 ± 3 °C (toxicity study) or 21± 3 °C (carcinogenicity study); humidity 50% ± 20%; 12 air changes per hour (toxicity study) or 10 to 15 air changes per hour (carcinogenicity study); light cycle 12 hours light and 12 hours dark. In the toxicity study, animals had free access to standard certified pelleted commercial laboratory diet (PMI Certified Rodent Chow 5002: PMI Nutrition International Inc) except during designated procedures. In the carcinogenicity study, animals were subjected to a restricted feeding regimen to enhance survival. Accordingly, 5 and 4 food pellets per day were provided to males and females, respectively, of a certified, commercial laboratory rodent diet (Certified Rodent Diet 4.2 grams-RPR 5L35, PMI Nutrition International LLC).
In the 39-week toxicity study, male and female Beagle dogs (Canis familiaris) were evaluated from Marshall Farms, North Rose, New York. At the start of treatment, animals were between 7 and 8 months old and ranged in weight between 6.8 to 9.9 kg (males) and 5.4 to 8.0 kg (females). The animal room environment was controlled (targeted ranges: temperature 21 ± 3 °C, relative humidity 50% ± 20%, 10-15 air changes per hour). Lighting was regulated to provide 12 hours light, 12 hours dark per 24-hour period (except during ophthalmological examinations). A standard certified commercial dog chow (400 g of Teklad certified 25% Lab Dog Diet #8727C) was made available to each dog once daily during a feeding period of approximately 2 hours throughout the study. In addition, any animals showing signs of hypoglycemia were allowed access to food for an additional ca 30 minutes prior to inhalation exposure. Municipal tap water (which had been purified by reverse osmosis, treated by ultraviolet light and further filtered through a 0.2-µm filter) was provided to the animals ad libitum, except during designated procedures.
Dose Administration
Aerosol exposures by nose-only inhalation chamber (rats) or oronasal facemask (dogs), in addition to exposing the lungs, results in powder deposition in the nasal cavity, oropharyngeal tissues, or upper airways, where it is subject to mucociliary clearance and/or swallowing to the gastrointestinal tract. Therefore, a portion of the administered dose was not systemically bioavailable because insulin was not absorbed orally.
In rats, actual chamber concentrations of aerosol were measured each day from a sampling port from the animal breathing zone using a gravimetric method. Chamber concentrations of insulin and Technosphere particles were determined analytically from the deposit collected on the gravimetric filters. Particle size distribution analysis was performed once weekly from each vehicle and test article chamber using a 9-stage cascade impactor followed by gravimetric analysis for each size range. In the rat toxicity and carcinogenicity studies, the achieved dose of Technosphere particles or Afrezza for each treatment level was determined as follows:
Where:
Active concentration (mg/L) = aerosol concentration of Technosphere or insulin determined by chemical analysis
T (min) = exposure duration (up to 180 minutes)
D (%) = pulmonary aerosol deposition fraction, according to the particle size
BW (kg) = mean body weight per sex per group during treatment
Achieved inhalation doses during the exposure period in animal studies were estimated regularly through the entire dosing interval. This was done to ensure that body weight changes, and therefore, respiratory minute volumes, over the study duration, were accounted for in the dosimetry calculations. Achieved doses were calculated using the following parameters; mean aerosol concentration delivered to the animal (mg/L air, as determined by chemical analysis of insulin or FDKP via HPLC), mean body weight, minute volume (L/min), and duration of daily exposure (minutes).
The mass median aerodynamic diameter and the geometric SD (MMAD ± GSD) were calculated for each group on the basis of impactor results using a log-probit transformation. Particle size distribution measurements were evaluated to confirm that exposure aerosols were respirable for the test species and comparable across groups. The estimation of total inhaled dose assumed 100% deposition within the respiratory tract. The MMAD ± GSD for Afrezza and Technosphere particles determined gravimetrically was 2.4 to 2.8 µm ± 1.9. The MMAD ± GSD for insulin determined by chemical analysis was 2.4 to 2.5 µm ± 1.9.
Afrezza doses were limited by the pharmacology of insulin; however, substantially higher doses of Technosphere particles alone were utilized to maximize FDKP exposure. During the acclimation period, the animals were conditioned to the laboratory setting and restraint tubes. Acclimation to restraint tubes was performed for increasing periods of time prior to initiation of treatment up to the duration of exposure (up to 180 minutes).
In dogs, prior to dosing, animals were accustomed to the animal restraint procedure, the muzzle mask and aerosol delivery tube and the sensation of air passing through the mask. The acclimation was carried out over a period of approximately 3 weeks to ensure that the animals were fully habituated to each element of the dose administration procedure (up to 15 minutes). Achieved dose levels in the chronic dog toxicity study during the exposure period were estimated using the following formula:
DL = Achieved dose levels (mg/kg/d)
Ec = Actual concentration delivered to the animals (mg/L air)
RMV = Respiratory Minute Volume (L/min); RMV (L) = 0.499 × W (kg)0.809
T = Time, duration of daily exposure (min.)
BW = Mean body weight (kg) during exposure period
Twenty-Six-Week Toxicity Study in Rat: Design and Histopathology Evaluations Including Respiratory Tissues
The study design for the 26-week rat toxicity study is outlined in Tables 1A and 1B. This study was designed to assess the toxicity and toxicokinetic (TK) profiles of Afrezza and Technosphere particles following administration to the rat by inhalation for 180 minutes daily for up to 26 weeks. Additionally, the reversibility of any effects in the high-dose Afrezza group versus air control group was assessed following a 4-week recovery period. A comprehensive list of tissues (Table 1C) was prepared and examined by light microscopy. Respiratory tissues examined in H&E-stained slides included lungs, larynx, pharynx, bronchi, trachea, nasal cavities including nasopharynx and sinuses, and tracheobronchial lymph node.
Study Design: Targeted Dose in 26-Week Inhalation Toxicity Study in Rat.a,b
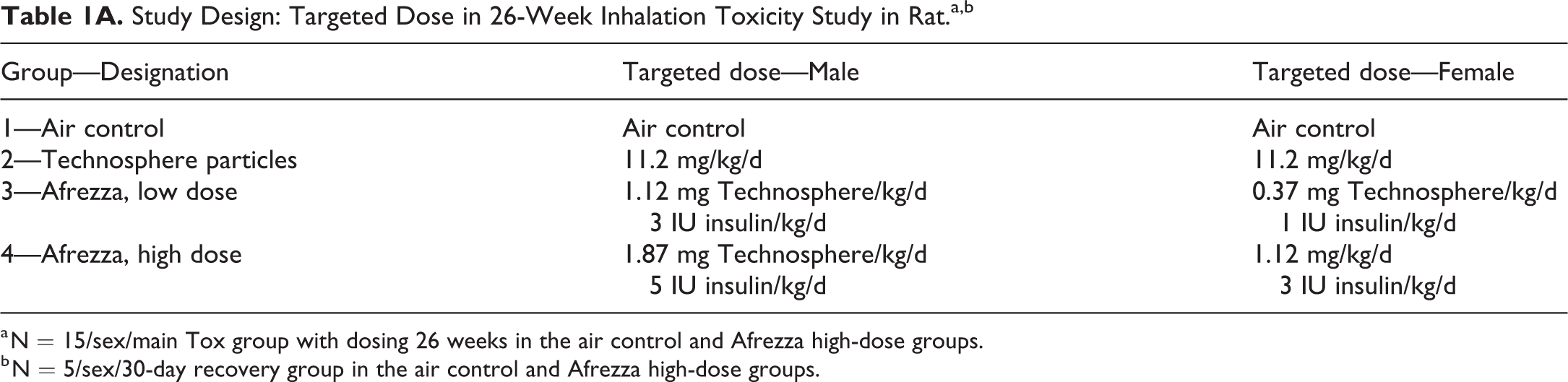
a N = 15/sex/main Tox group with dosing 26 weeks in the air control and Afrezza high-dose groups.
b N = 5/sex/30-day recovery group in the air control and Afrezza high-dose groups.
Study Design: Theoretical Achieved Dose in 26-Week Inhalation Toxicity Study in Rat.a,b
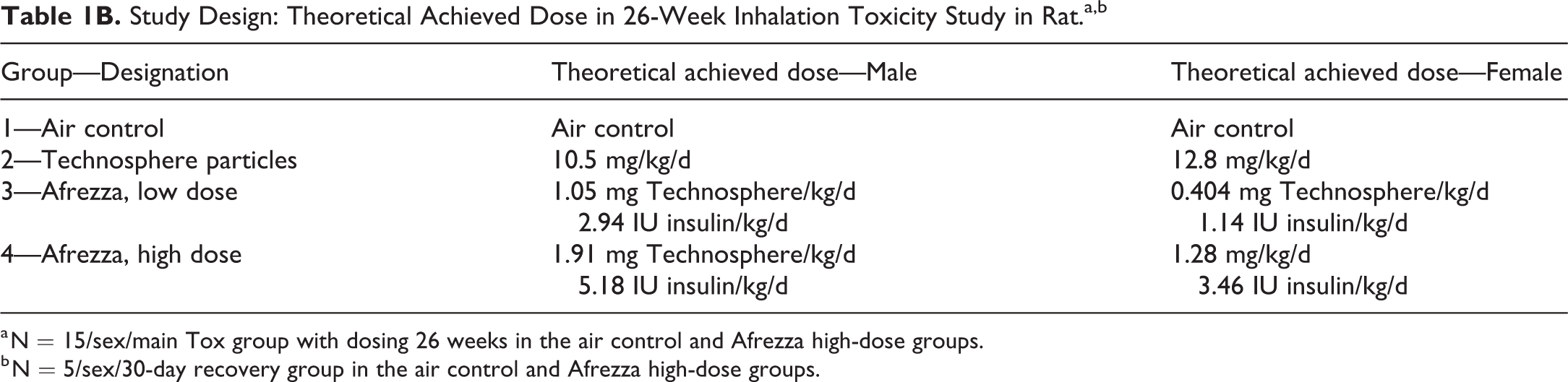
a N = 15/sex/main Tox group with dosing 26 weeks in the air control and Afrezza high-dose groups.
b N = 5/sex/30-day recovery group in the air control and Afrezza high-dose groups.
Histopathology Tissue Lista: 26-Week Inhalation Toxicity Study in Rats.

a Groups 1, 2, and 4: All tissues listed above. Group 3: respiratory tissues (lungs, larynx, pharynx, bronchi, trachea, and nasal cavities and sinuses, tracheobronchial lymph nodes) and abnormal findings.
Thirty-Nine-Week Toxicity Study in Dogs, Design, and Histopathology Evaluations Including Respiratory Tissues
The study design for the 39-week dog toxicity study is outlined in Tables 2A and 2B. This study was designed to assess the toxicity and TK profiles of Afrezza and Technosphere particles following administration to the dog by inhalation once daily for up to 39 weeks. Additionally, the reversibility of any effects was assessed following an 8-week recovery period. The animals were exposed to aerosols of Afrezza or Technosphere particles alone for 15 minutes, once daily for up to 39 weeks. A comprehensive list of tissues (Table 2C) was prepared and examined by light microscopy. Respiratory tissues examined in H&E-stained slides included lungs, larynx, pharynx, bronchi, trachea, carina, and nasal cavities including nasopharynx and sinuses and bronchial lymph nodes.
Study Design: Target Dose Level—39-Week Inhalation Toxicity Study in Dog.
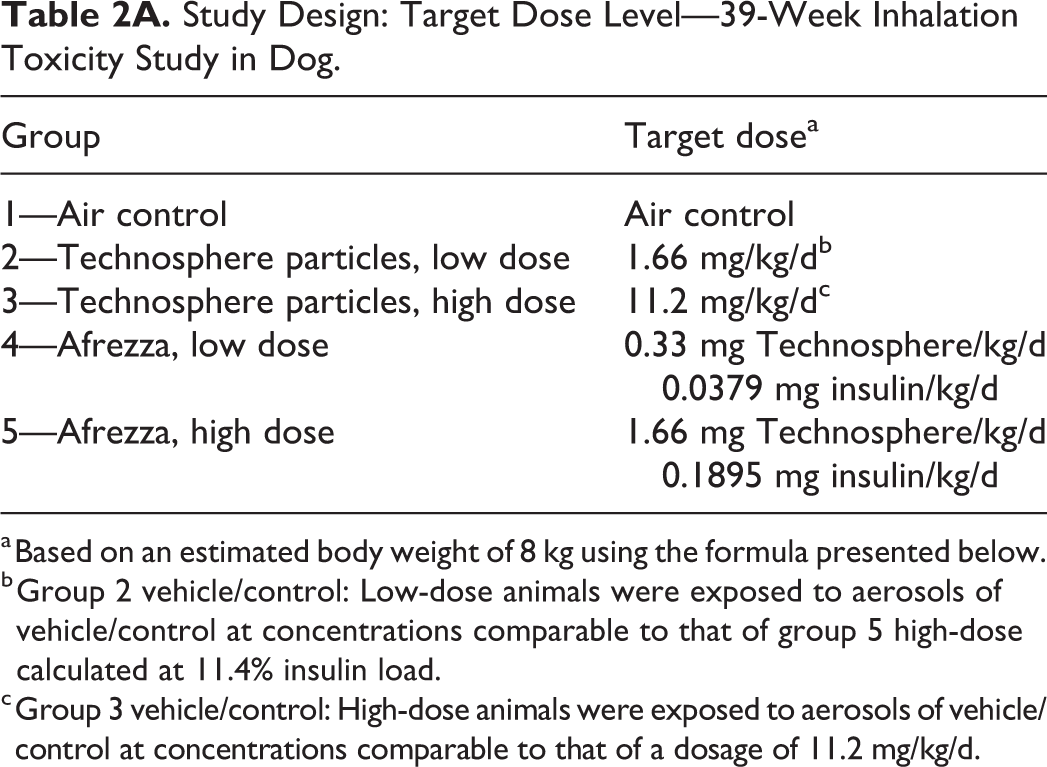
a Based on an estimated body weight of 8 kg using the formula presented below.
b Group 2 vehicle/control: Low-dose animals were exposed to aerosols of vehicle/control at concentrations comparable to that of group 5 high-dose calculated at 11.4% insulin load.
c Group 3 vehicle/control: High-dose animals were exposed to aerosols of vehicle/control at concentrations comparable to that of a dosage of 11.2 mg/kg/d.
Study Design: 39-Week Inhalation Toxicity Study in Dog.a,b,c
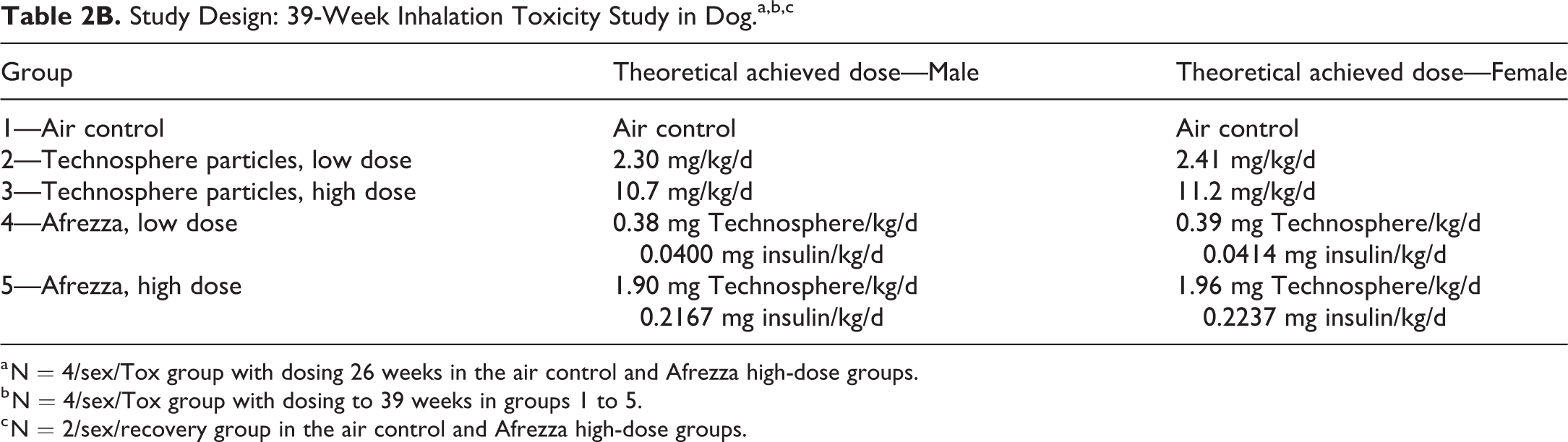
a N = 4/sex/Tox group with dosing 26 weeks in the air control and Afrezza high-dose groups.
b N = 4/sex/Tox group with dosing to 39 weeks in groups 1 to 5.
c N = 2/sex/recovery group in the air control and Afrezza high-dose groups.
Histopathology Tissue List: 39-Week Inhalation Toxicity Study in Dog.

Study Design and Histopathology Methods: 104-Week Oral Carcinogenicity Study in Rat
The study design for the 104-week rat carcinogenicity study is outlined in Tables 3A and 3B. This 104-week study was designed to assess the carcinogenicity potential for Afrezza and Technosphere particles following administration to the rat by inhalation once daily for up to 104 weeks. The animals were exposed to aerosols of Afrezza or Technosphere particles alone for up to 60 minutes, once daily for up to 104 weeks. The animals underwent a full necropsy and tissues were retained for histopathological evaluation at the completion of the study. A comprehensive list of tissues (Table 3C) was prepared and examined by light microscopy. Respiratory tissues examined in H&E-stained slides included lungs, larynx, bronchi, trachea, carina, nasal cavities including nasopharynx and sinuses, and tracheobronchial lymph node.
Study Design: Target Dose—104-Week Carcinogenicity Study in Rat.a

a N = 60/sex/group.
Study Design: Theoretical Achieved Dose—104-Week Carcinogenicity Study in Rat.a

a N = 60/sex/group.
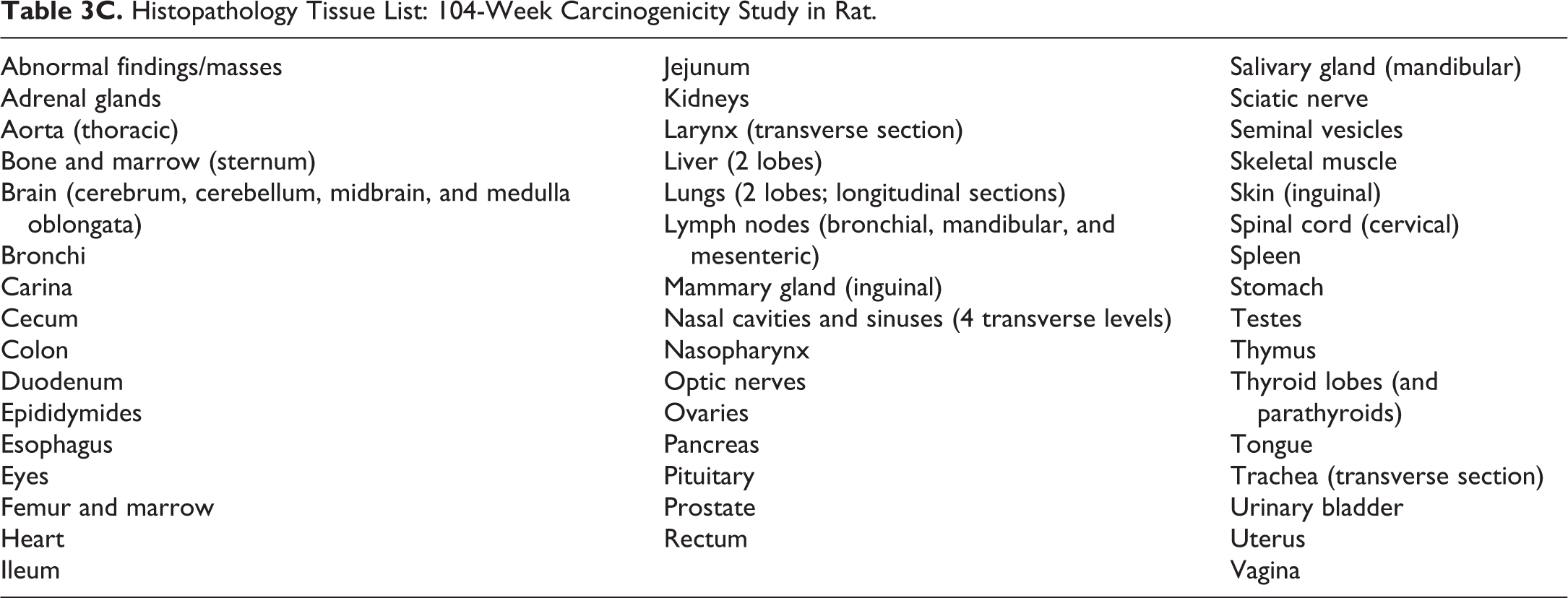
Histopathology Tissue List: 104-Week Carcinogenicity Study in Rat.
Assessment of Cellular Proliferation Using PCNA
Initial or investigational immunostaining and assessment of PCNA labeling was conducted in all 3 safety studies, but these early labeling studies were considered inadequate in retrospect due to issues with the methodology employed including; sampling bias, inadequate numbers of cells evaluated or imprecision in the number of cells evaluated (counts of labeled cells/fixed microscopic field in variably inflated lungs), low microscopic magnification that precluded detection of labeled cells or classification of cell types, and grouping of counts as all lung cells, regardless of whether they were in alveoli or bronchioles or grouping of all bronchioles regardless of size, low numbers of animals (4/sex/group) in the case of the dog study, and lack of masked (“blinded”) evaluation. Consequently, these initial method development challenges resulted in the evolution of a more robust method for PCNA assessment of cellular proliferative events in pulmonary tissue.
There are inherent difficulties in conducting retrospective labeling studies in chronic inhalation studies. Instillation of fixative in the lung typically is not done at constant hydrostatic pressure and airways are not dissected, which precludes assessment of proliferation along precise branching pathways in exactly the same bronchiolar generation in all animals. However, these studies represented a valuable resource to further understand the potential effect of inhaled insulin on lung cell proliferation. Therefore, the 26-week rat study was selected for rigorous, unbiased evaluation of PCNA labeling indices. The chronic rat study was chosen over the chronic dog or rat carcinogenicity studies because of the larger number of animals compared to dog and because lung inflation and sampling were considered most uniform in the 26-week rat study.
Immunohistochemically stained lung slides from the 80 rats sacrificed after 26 weeks of exposure were provided by Charles River Laboratories to Seventh Wave Laboratories. These slides had been reacted with an antibody to PCNA, diaminobenzidine (DAB) was used as the chromogen, and hematoxylin provided the counter stain. This staining technique caused nuclear staining that varied from blue (hematoxylin) to weakly blue-brown or light brown to intensely rusty brown (DAB) within each slide. This variation in staining intensity without a clear threshold to identify cells in S-phase is a common finding with PCNA immunohistochemistry because although PCNA is necessary for DNA synthesis and is elevated during the G1/S phase of the cell cycle, quiescent cells (G0) also have low levels of PCNA. 33 Additionally, PCNA degrades slowly, with a median life of at least 20 hours in tissues. 34 However, all of the lung slides contained bronchiole-associated lymphoid tissue (BALT), and this BALT provided a positive control tissue type to calibrate the staining. Bronchiole-associated lymphoid tissue contains lymphocytes in G1/S phase, and thus the most intense, rusty brown (DAB chromogen) nuclear staining could be identified for each slide to provide an internal control for G1/S-phase staining. The approach taken was to examine BALT from each slide when the slide was first placed on the microscope stage. Staining that was interpreted as G1/S-phase staining within BALT was identified. Cells within airway or alveolar epithelium with similar intense staining were considered PCNA-labeled cells during the counting procedure.
Randomized, masked immunohistochemically stained histologic slides were examined microscopically for the PCNA labeling evaluation by the Seventh Wave Principal Investigator (PI; KJN) without knowledge of the animal’s identification, sex, or treatment group. Counting consisted of classifying cells as PCNA-labeled or unlabeled and recording the number of labeled cells and total cells counted in each of 3 compartments: respiratory epithelium of large bronchioles (proximal airways), respiratory epithelium of terminal bronchioles, and alveoli. The epithelial labeling index in each compartment was calculated as the number of PCNA-positive nuclei divided by the total number of nuclei counted (multiplied X 100% to express the data as a percentage). Following collection and locking of these data, animal identifications and treatment groups were unmasked for sorting and subsequent generation of individual animal data tables and summary tables. These unmasked data were provided to Charles River Laboratories for statistical analysis and to the PI (KJN) for interpretation of the results.
Proliferating Cell Nuclear Antigen Methods: Large Bronchioles
Counts of unlabeled and labeled airway epithelial nuclei were obtained from the pseudostratified mucociliary epithelium lining the large bronchioles in the left and right lung lobe sections. The main axial airway (large bronchiole) of the left lung was sectioned longitudinally on the slide. Counts were obtained from the epithelium starting at the most proximal portion of the large airway that appeared in longitudinal orientation. Using the 40X objective lens and 10X ocular lenses (final magnification of 400X), the PI counted cells proceeding from proximal to distal along each side of the axial airway until 100 total cells were counted from each side of the airway for a total of 200 bronchiolar epithelial cells from the left lung.
The main axial airway (large bronchiole) of each right lung lobe was sectioned transversely or approximately transversely. For each of the 4 right lung sections, the main axial airway was identified using a 2.5X objective lens. The axial airway was identified as the largest bronchiole appearing in transverse (cross) section or approximate transverse section. Further aids in identification of the axial airway included position within the lung lobe and size of the pulmonary artery alongside the axial airway. The starting point for counting was defined using a clock-face approach, while the airway was viewed using the 2.5X objective. If the random animal identification number was even, the PI started counting cells, using the 40X objective (final magnification of 400X), at the 12 o’clock or highest position and proceeded in a clockwise direction until 200 cells from the mucociliary epithelium were counted. If the random animal identification number was odd, the PI started counting cells, using the 40X objective, at the 6 o’clock or lowest position and proceeded in a clockwise direction until 200 cells from the mucociliary epithelium were counted. This rigid sampling scheme determined at a low magnification provided a stratified random sampling that precluded bias toward selecting areas of low or high labeling.
Counts were not obtained from the lymphoepithelium lining BALT or epithelium with artifacts such as folds or tears or portions of the airway that were cut so tangentially that the normal orientation of the pseudostratified epithelium with subepithelial layers was obscured; instead, the PI moved to the next useable segment of epithelium lining the airway. In rare instances when the main axial airway in appropriate orientation could not be identified, there were no counts for the corresponding proximal airway in that lobe. If there was tangential sectioning or there were artifacts such that 200 cells could not be counted from an airway, then the number of cells (less than 200) that could be counted from that airway was used to calculate labeling indices. In most animals, a total of 1000 cells from large bronchioles were counted.
Proliferating Cell Nuclear Antigen Methods: Terminal Bronchioles
Counts of unlabeled and labeled airway epithelial nuclei were obtained from the simple epithelium lining the terminal bronchioles in the left and right lung lobe sections. Terminal bronchioles were defined as the last (terminal) segment or branch of conducting airway immediately proximal to the alveolar duct. They were identified when an approximately longitudinal section cut through the terminal bronchiole and the alveolar duct subtending that bronchiole. Lung slides were examined using a random starting point on the glass slide, a Z-raster approach, and the 2.5X objective and 10X ocular lens to identify terminal bronchioles. When a terminal bronchiole was identified, the objective lens was switched to 40X (final magnification of 400X) and epithelial cells were counted from each side of the terminal bronchiole from the distal end proximally until a branch point was reached. The identification of terminal bronchioles and counting proceeded until 400 cells had been counted from the left lung and 200 cells had been counted from each of the right lung lobes for a total of 1200 cells. Counts were not obtained from epithelium with artifacts such as folds or tears. If there were too few correctly oriented terminal bronchioles in the lung section such that 400 cells could not be counted from the left lung terminal bronchioles or 200 cells could not be counted from the terminal bronchioles in a right lung lobe, then the number of cells (less than 400 or 200) that could be counted from the lobe was used to calculate labeling indices. In most animals, a total of 1200 cells from terminal bronchioles were counted.
Proliferating Cell Nuclear Antigen Methods; Alveoli
Counts of unlabeled and labeled alveolar epithelial cells were obtained from the alveoli in the left and right lung lobe sections. Lung slides were examined using a random starting point on the glass slide, a Z-raster approach, and the 40X objective and 10X ocular lens. From the random starting field, every other microscopic field was used for counting. An ocular lens piece containing a grid was used during these counts. This grid aided the pathologist by providing boundaries (inclusion and exclusion lines) within the field for counting. The counting proceeded until 1000 cells were counted from the left lung and 1000 cells were counted from the right lung lobes for a total of 2000 cells. Counts were not obtained from areas of lung with technical artifacts that precluded identification of alveolar epithelium or interpretation of staining. Examples of such artifacts included marked lack of inflation of the lobe by fixative, folds, or tears. In rare instances when the size of the left lung section and the sampling of every other field resulted in less than 1000 cells counted from the left lung, then the number of cells (less than 2000 total) that was counted was used to calculate alveolar labeling indices.
Statistics
Proliferating cell nuclear antigen numerical data was subjected to calculation of group mean values and standard deviations. Group variances were compared using Levene test at the .05 significance level. When differences between group variances were not found to be significant, a parametric 1-way analysis of variance (ANOVA) was performed. If significant differences among the means were indicated by the ANOVA (P ≤ .05), then Dunnett t test was used to perform the group mean comparisons between the air control group 1 and groups 2 to 4 as well as between the vehicle control group 2 and groups 3 and 4. Whenever Levene test indicated heterogeneous group variances (P ≤ .05), the nonparametric Kruskal-Wallis test was used to compare all considered groups. If the Kruskal-Wallis test was significant (P ≤ .05), then the significance of the differences between the air control group 1 and groups 2 to 4 as well as between the vehicle control group 2 and groups 3 and 4 were assessed using Dunn test. For each pairwise group comparison of interest, significance was reported at the .05, .01, and .001 levels.
Research Involving Animals
All studies reported in this manuscript were conducted in accordance with the US FDA Good Laboratory Practice Regulations (21 CFR Part 58) and following institutional IACUC guidelines and SOPs.
Results
Twenty-Six-Week Toxicity Study in Rat
There were no test article–related changes in absolute lung weights or lung-to-brain weight ratios. Lung-to-body weight ratios were increased in rats in the Technosphere particle group and male rats in the high-dose Afrezza group. However, these increases in lung-to-body weight ratios were not clearly test article–related effects on lung as they only occurred in groups with body weights that were slightly or significantly (females) lower than controls.
Other than findings in the nasal cavities, there were no Technosphere particle- or Afrezza-related microscopic findings in H&E-stained tissues in the rat. All rats in the Technosphere particle group or the high-dose Afrezza groups exhibited cytoplasmic eosinophilic globules in the nasal or nasopharyngeal respiratory and/or olfactory epithelium at 26 weeks. Olfactory epithelium lining the ethomoturbinates and caudal dorsal meatus was most involved, followed by respiratory epithelium lining the dorsal meatus and septum, and to a lesser extent, respiratory epithelium lining turbinates and the nasopharynx. The incidence was 8/10 and 5/10 in low-dose Afrezza males and females, respectively (Table 4). The severity of eosinophilic globules ranged from moderate to marked in Technosphere particle animals and was clearly lower in those animals exposed to Afrezza (minimal to moderate in low-dose males and minimal in low-dose females; mild to marked in high-dose males and minimal to mild in high-dose females). Eosinophilic globules were considered a non-adverse response to irritation. Higher incidences and severity of eosinophilic globules were due to the Technosphere particle component of the exposures as particle exposure was highest in the Technosphere particle only group, followed by the Technosphere particle components of the high and low Afrezza groups, respectively. Minimal to mild degeneration of the nasal epithelium was present in 2/10 males and 2/10 females in the Technosphere particle groups and 1/10 high-dose Afrezza males and degeneration was attributed to irritation due to Technosphere particle exposure (Table 4). The severity of the eosinophilic globules decreased in recovery group females, but there was no clear evidence of recovery in males during the 8-week recovery period. Epithelial degeneration completely recovered.
Incidence and Severity of Eosinophilic Globules and Epithelial Degeneration in Nasal Cavity in Rats at 26 Weeks.

a Severity range shown in parentheses; dash used when the incidence was 0.
Thirt-Nine-Week Toxicity Study in Dog
All doses were well tolerated in the 39-week inhalation toxicity study in dogs. Microscopically, there was a low incidence (1/8 and 3/8 animals) of minimal to mild alveolar and/or bronchial–interstitial neutrophil infiltrates in the low- and high-dose Afrezza groups, respectively. However, due to the presence of this finding in 2/6 control animals and 2/6 high-dose Afrezza animals at 26 weeks (these were the only groups examined in a limited 26-week sacrifice), the relationship of this finding to Afrezza inhalation for 39 weeks cannot be ruled-out but is unlikely. This finding completely resolved during the 8-week recovery period (Table 5). There were no other microscopic findings that were considered Technosphere particle- or Afrezza-related.
Neutrophil Infiltration in Lung at 26 Weeks, 39 Weeks, and Recovery in Dogs.
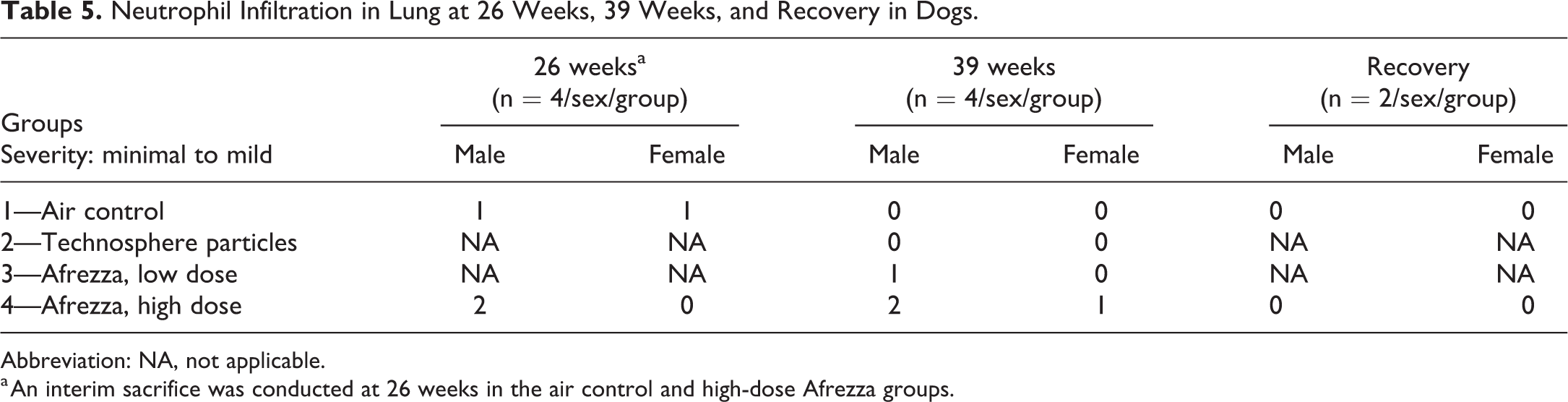
Abbreviation: NA, not applicable.
a An interim sacrifice was conducted at 26 weeks in the air control and high-dose Afrezza groups.
Incidence and Severity of Eosinophilic Globules and Goblet Cell Hyperplasia in Nasal Cavity in Male Rats in the Carcinogenicity Study.

a Average severity grade of finding in affected animals: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
Incidence and Severity of Eosinophilic Globules and Goblet Cell Hyperplasia in Nasal Cavity in Female Rats in the Carcinogenicity Study.
a Average severity grade of finding in affected animals: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
Rat Carcinogenicity Study
Inhalation of Technosphere particles or Afrezza was not associated with a carcinogenic effect in any tissue. There were no Afrezza-related microscopic findings in any tissue and Technosphere particle–related microscopic findings were restricted to the nasal cavity. Eosinophilic globules in olfactory and nasal respiratory epithelia and goblet cell hyperplasia primarily involving the respiratory epithelium of the nasal septum and nasopharyngeal duct were present in all groups, including air controls. Eosinophilic globules were characterized by enlarged epithelial cells containing homogeneous, brightly eosinophilic globules, and goblet cell hyperplasia was characterized by an increase in the number and size of goblet cells and these cells occasionally formed small gland-like clusters within the epithelium. The incidence and severity of eosinophilic globules and goblet cell hyperplasia were increased in the high-dose Technosphere animals (Tables 6A and 6B).
Assessment of Cellular Proliferation Using PCNA
There was no indication that the insulin component of Afrezza caused or exacerbated epithelial cell proliferation in the bronchioles or alveoli. There was an increase in cell replication in the large and terminal bronchioles, but not the alveoli, of rats exposed to Technosphere particles only (Tables 7 and 8; Figure 1A-C and Figure 2A-C). This finding, at 10.5 and 12.8 mg/kg/d Technosphere particles in male and female rats, respectively, was judged to be due to the physical load of powder depositing on the conducting airway epithelium.
Mean Percent PCNA-Labeled Epithelial Cells in Male Rats at 26 Weeks.

Abbreviation: PCNA, proliferating cell nuclear antigen.
a P < .01 compared to group 1.
b P < .05 compared to group 2.
Mean Percent PCNA-Labeled Epithelial Cells in Female Rats at 26 Weeks.
Abbreviation: PCNA, proliferating cell nuclear antigen.
a P < .05 compared to group 1.
b P < .01 compared to group 1.
c P < .05 compared to group 2.
d P < .01 compared to group 2.
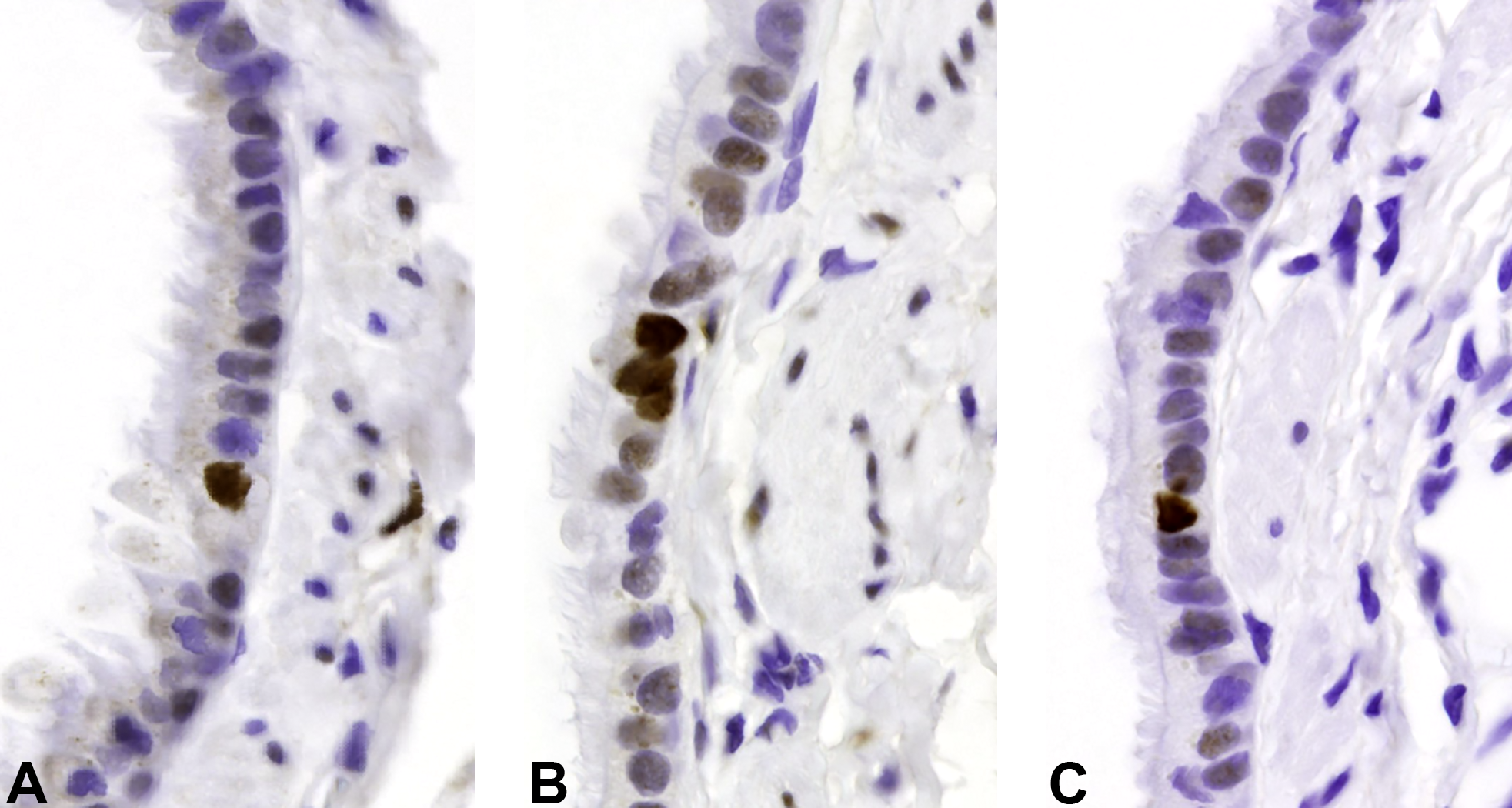
PCNA immunostaining of large bronchioles from female rats in 26-week toxicity study illustrating the increased labeling index in large bronchiolar epithelium from Technosphere particle-exposed animals. A, Air control. B, Technosphere particles. C, Afrezza. PCNA indicates proliferating cell nuclear antigen.
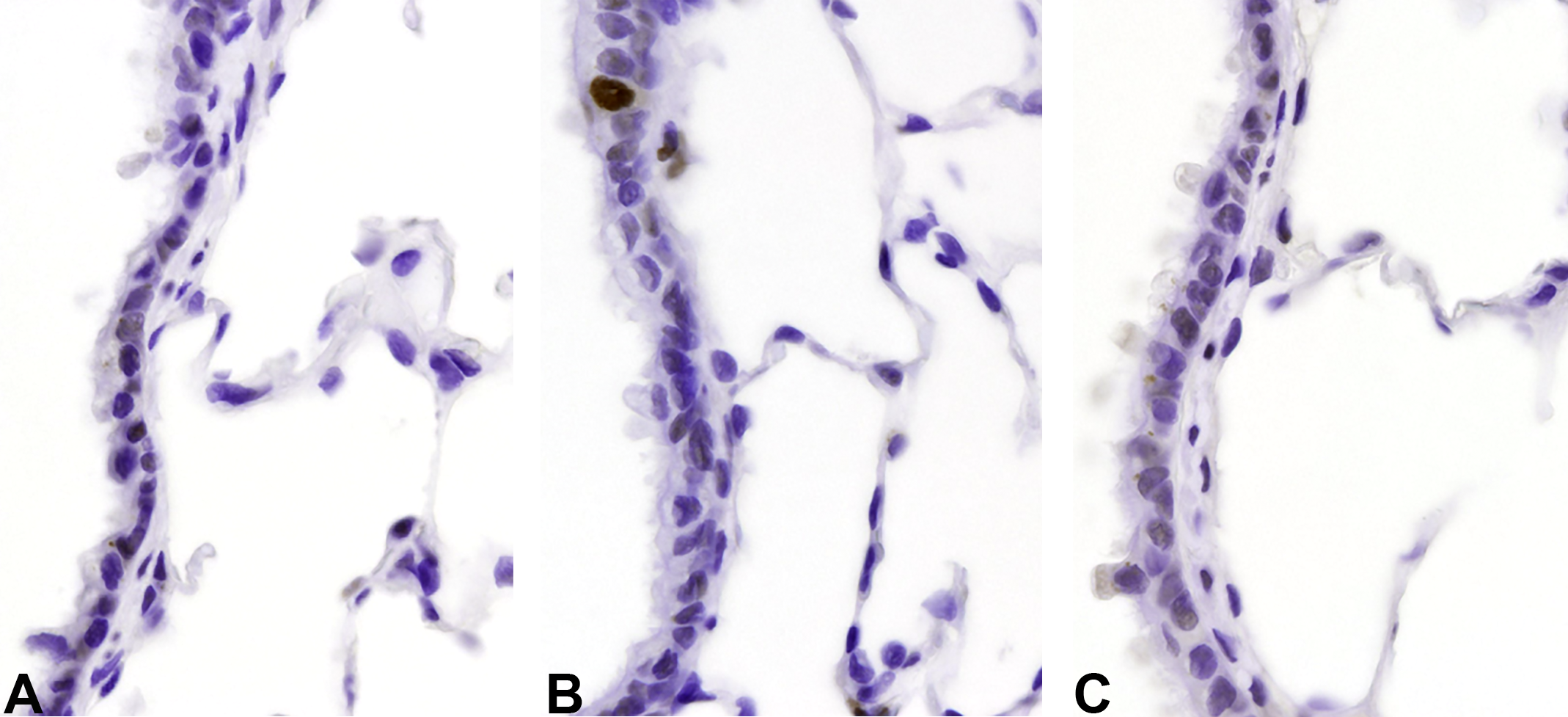
PCNA immunostaining of terminal bronchioles from male rats in 26-week toxicity study illustrating the increased labeling index in terminal bronchiolar epithelium of Technosphere particle-exposed animals. A, Air control. B, Technosphere particles. C, Afrezza. PCNA indicates proliferating cell nuclear antigen.
Discussion
Rat Studies—Histologic Evaluations of Pulmonary Tissues
Technosphere particle–related findings
Histopathologic nasal cavity findings in both rat studies were related to the Technosphere particle dose and consisted of an increased incidence and/or severity of (1) minimal to marked eosinophilic globules in nasal olfactory and respiratory epithelium, (2) minimal to mild goblet cell hyperplasia in nasal respiratory epithelium (carcinogenicity study), and (3) a low incidence of minimal to mild nasal respiratory epithelial degeneration (Chronic Tox study). These findings were considered a response to irritation as a result of impaction of particles on the epithelium. Eosinophilic globules are commonly observed in aging rats with age-related increases in incidence and severity. 35 In chronic inhalation studies, the incidence and severity of this change is often exacerbated in an exposure-dependent manner and is thought to be induced by irritation. 36,37 Eosinophilic globules and goblet cell hyperplasia were observed with comparable frequency and severity in air control and Afrezza-treated groups in the rat carcinogenicity study (up to 104 weeks). Nasal lesions in rats were not considered relevant for human risk assessment because Afrezza is not administered to the nose of humans whereas rats are obligate nose-breathers, and Afrezza was administered to rats in nose-only inhalation chambers.
Evaluation of proliferation in pulmonary tissues (Chronic Tox)
Evaluation of the effects of Technosphere particles and Afrezza on pulmonary epithelial labeling indices was conducted using a rigorous, randomized and masked, microscopic evaluation of airways and alveoli in sections from the 26-week rat toxicity study.
Technosphere-related Findings
There were no findings that would suggest increased epithelial proliferation in the alveoli of Technosphere particle groups. Proliferating cell nuclear antigen labeling assessment demonstrated that exposure to Technosphere particles at high doses caused an increase in cell proliferation in the epithelium of large bronchioles in female rats and terminal bronchioles in male and female rats compared to the corresponding airway labeling in air-exposed rats. This increase in cell replication in the large and terminal bronchioles, but not the alveoli, of rats exposed to Technosphere particles may be due to the physical load of particles depositing on the conducting airway epithelium. A portion of inhaled Technosphere particles would be expected to deposit in the conducting airways. In addition, due to the relatively small surface area of the conducting airways compared to the large surface area of the alveoli, the load of Technosphere particles per unit surface area would be much larger in the conducting airways than in the alveoli.
Afrezza-related Findings
There were no findings that would suggest increased epithelial proliferation in the conducting airways or alveoli in the Afrezza groups. It is clear there was no indication that the insulin component of Afrezza caused or exacerbated pulmonary epithelial cell replication.
Tumorigenicity evaluation
There were no neoplastic findings in any tissues in rats administered Technosphere particles or Afrezza for up to 104-weeks.
Dog Toxicity Study: Evaluation of Pulmonary Tissues
There was no evidence of a hyperplastic or proliferative effects due to Technosphere particles or Afrezza in the chronic dog toxicity study. There was a low incidence of neutrophilic infiltrates in the lung of animals exposed to Afrezza for 39 weeks. However, at 26 weeks, neutrophilic infiltrates were present in the lungs of control animals at a comparable incidence. Hence, a relationship between Afrezza exposure and neutrophilic infiltrates was considered to be equivocal but cannot be dismissed.
Taken together, these data demonstrate (1) no histopathology changes in H&E-stained sections in lung or respiratory tissues other than the nasal cavity in rats administered Technosphere particles or Afrezza for up to 104 weeks; (2) nasal eosinophilic globules and a low incidence nasal epithelial degeneration in rats administered Technosphere particles or Afrezza for 26 weeks and eosinophilic globules and goblet cell hyperplasia in rats administered Technosphere particles for up to 104 week; these findings were interpreted as a response to irritation due to Technosphere particle exposure; (3) no evidence of carcinogenicity in rats administered Technosphere particles or Afrezza for up to 104-weeks in the carcinogenicity study; (4) a small, equivocal, increase in pulmonary neutrophilic infiltrates without evidence of cell damage, epithelial hyperplasia, or fibrosis in dogs exposed to the high dose of Afrezza in the 39-week dog study; and (5) unbiased assessment of PCNA labeling in 3 major lung compartments in animals from the 26-week rat toxicity study demonstrated that exposure to Technosphere particles at 10.5 and 12.8 mg/kg/d in male and female rats, respectively, caused an increase in cell proliferation in the epithelium of large bronchioles in female rats and terminal bronchioles in male and female rats compared to the corresponding airway labeling in air-exposed rats. Although this increase in cell proliferation was detected by PCNA labeling, it did not result in morphologic epithelial hyperplasia in H&E-stained sections. There were no findings that would suggest increased epithelial proliferation in the conducting airways or alveoli in the Afrezza groups or in the alveoli of Technosphere particle groups.
Evaluation of Carcinogenic Potential of Inhaled Insulin
With the review and subsequent approval of 2 native human insulin products for administration by the pulmonary route, there has been considerable discussion and debate regarding the potential carcinogenicity of human insulin on respiratory tissue. This study discussed in the current manuscript, in addition to the rigorous review of the insulin hexamer Exubera, has clearly demonstrated the absence of any in vivo carcinogenic signal, even after long-term pulmonary administration. 38 Even though both of these products differ substantially in their drug product formulations, that is, Exubera is a molecular hexamer of homologous human insulin that disassociates locally to release the active insulin monomer and Afrezza is a monomer of homologous human insulin that is adsorbed to a completely aqueous soluble nanoparticle that locally releases the adsorbed insulin molecule, both products after extensive review and critiques by regulatory agencies and academics and have been approved for human use for adult and pediatric diabetes mellitus types 1 and 2. The extensive battery of preclinical evidence coupled with the decades and multimillion patient years of combined patient utilization of exogenously administered insulin demonstrates quite convincingly the absence of human carcinogenic risk of insulin use, either parenterally or by pulmonary administration. 39,33
Both the concern and the confusion for the potential carcinogenic activity of “insulin” are a result of a lack of precision in the utilization of nomenclature and a lack of scientific rigor in discussing the cellular, that is, in vitro activity of “insulin.” This lack of rigor in terminology is an important factor especially in discussing the 2 major categories of “insulin” therapies: (1) products like Afrezza and Exubera which are comprised of native human insulin homologues and (2) other “insulin” therapies that are comprised of active insulin analogues, that is, they are not equivalent in structure to native human insulin and therefore vary in their primary, secondary, and tertiary structure compared to native insulin. 34
Numerous papers have erroneously attributed direct, in vitro “mitogenic” or proliferative responses to insulin without regard to the specific type of insulin that was used or the experimental conditions utilized in the cited experiments. Minute quantities of insulin are required for the maintenance of many, if not most in vitro, long-term cell cultures and after decades of use under these conditions, a cellular proliferative effect of insulin has not been reported. However, mitogenic, that is, proliferative effects of insulin have been reported when (1) extremely high, nonphysiologic exposure levels of insulin have been used in the cell culture and/or (2) when the insulin in the experiment is not human insulin, but rather an insulin analogue, and neither of these conditions are relevant to the risk assessment of the human therapeutic use of native human insulin homologues at tolerable human dose levels of exposure. 32
The reported mitogenic effects of insulin have sometimes been reported as proliferative or potentially carcinogenic in vitro effects, 40,41 but a critical review of the literature reveals these effects are (1) observed only at suprapharmacologic in vivo exposure levels and associated with the high-dose insulin cross reactivity with the known mitogenic receptor cascade mediated by “IGF-1-like” activity or (2) mediated by insulin analogues that demonstrate low-dose activity for the IGF-1 receptor. 42,43 Importantly, in the discussion of a worst-case risk assessment, even if there were a slight potential that an insulin therapeutic might trigger an analogous in vivo action, there are reported counter balancing or moderating in vivo mechanisms that would preclude an evolving carcinogenic risk to humans. 44
Certainly, several insulin analogue products are approved and widely used in the treatment of diabetes. There have been no carcinogenic or tumor incidence-related safety events, relabeling or recalls of these products to date. The achievement of suprapharmacologic exposure levels of human insulin by the pulmonary route that might even theoretically trigger a pro-carcinogenic response is simply not attainable due to the very real pharmacologic effects of high-dose insulin, which not only is dose-limiting in human prescribing practices, but was in fact the dose-limiting criterion in all of the pre-clinical, high-dose toxicity and carcinogenicity studies as well.
Contemporary regulatory practices and guidance documents take into consideration much of the controversies and nuances briefly highlighted above in their recommendations for assessment and expected review criteria for all potential insulin therapies. Those considerations can be summarized as (1) in vitro cell concentrations of compounds, (2) differential receptor binding assays for IR and IGF-1, and (3) the definitive in vivo assessment of cellular proliferative activity in relevant tissues. 40 It is reasonable to conclude that in any development strategy to assess the potential carcinogenic potential effects of an insulin therapy, whether by the parenteral or pulmonary route of administration, the final step in this preclinical evaluation is the in vivo assessment and evaluation of any lesion(s) that may signal a potential carcinogenic risk. 43 Consequently, the role of the Toxicologic Pathologist is central to this process. The inadequate or scientifically deficient study design or evaluation methods for carcinogenic effects can otherwise derail acceptable drug development programs or, in a worst-case, could result in missing an important signal of an undesirable effect.
In clinical trials, 2 cases of lung cancer, 1 in controlled trials and 1 in uncontrolled trials (2 cases in 2750 patient-years of exposure), were observed in participants exposed to Afrezza while no cases of lung cancer were observed in comparators (0 cases in 2169 patient-years of exposure). 4 In both cases, a prior history of heavy tobacco use was identified as a risk factor for lung cancer. Two additional cases of lung cancer (squamous cell) occurred in nonsmokers exposed to Afrezza and were reported by investigators after clinical trial completion. These data are insufficient to determine whether Afrezza has an effect on lung or respiratory tract tumors.
For clinical use of Afrezza, current warnings and precautions include (1) risk of acute bronchospasm in patients with chronic lung disease; (2) Afrezza is contraindicated in patients with chronic lung disease such as asthma or COPD; (3) before initiating AFREZZA, perform a detailed medical history, physical examination, and spirometry (FEV1) to identify potential lung disease in all patients. 4 In patients with active lung cancer, a prior history of lung cancer, or in patients at risk for lung cancer, considers whether the benefits of AFREZZA use outweigh this potential risk.
Conclusion
In conclusion, there were no adverse pulmonary effects with chronic administration of Afrezza or Technosphere particles in nonclinical safety studies. These studies also demonstrated no increase in epithelial proliferation in the conducting airways or alveoli in animals administered Afrezza or in the alveoli of animals administered Technosphere particles. Effects of Afrezza or Technosphere particles were primarily isolated to nasal passages and were considered due to impaction of particles on these airways at high doses. In comparison, Technosphere particle dose in humans using the Afrezza Gen2 inhaler is much less. There were no preneoplastic or neoplastic effects of Afrezza or Technosphere particles.
Assessment of insulin safety margins were not possible due to adverse effects associated with exaggerated pharmacology. However, based on Technosphere particle (FDKP) exposure in the chronic animal toxicity studies, estimated safety margins are 2- to 5-fold over the maximum clinical Afrezza dose. In these studies, the no observable adverse effect level was the highest Technosphere particle dose, and therefore, safety margins are likely much higher than estimated.
Footnotes
Acknowledgments
The authors would like to thank David Thomson, PhD, JD for his support in the writing of this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Jack Reynolds and Dr. Stephanie Greene are preclinical development consultants and Dr. Kristen Nikula is a pathologist employed by a contract research organization. Dominic Poulin and Dr. Kevin McInally are Study Directors employed by contract research organizations. All 5 authors contributed to the Afrezza program at various times. Dr. Stephanie Greene is a former employee of Mannkind Corp. Dr. Jack Reynolds and Dr. Stephanie Greene own stock in Mannkind.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.