Abstract
This paper presents a review of the nature, range, and incidences of background pathology findings in the respiratory tract of cynomolgus monkeys and rats. Data were collected from 81 inhalation studies and 133 non-inhalation studies evaluated at 3 geographically distinct contract research organization facilities. The inhalation studies were comprised of 44 different small molecule pharmaceuticals or chemicals which were also analyzed in order to understand the patterns of induced changes within the respiratory tract. The lung was the most frequently affected organ in both species, with increased alveolar macrophages being the most common background and test article–related finding. In the upper respiratory tract (URT), inflammatory cell infiltrates were the most common background findings in the nasal cavity in monkeys. Induced URT findings were more frequent in rats than monkeys, with squamous metaplasia in the larynx, and goblet cell hyperplasia in the nasal cavity being the most common. Overall, the data revealed a limited pattern of response to inhaled molecules in the respiratory tract, with background and test article–related findings often occurring in the same regions. It is hoped that these data will assist in the interpretation of findings in the respiratory tract induced by novel inhaled small molecule entities.
Keywords
Introduction
For the clinical development of inhaled drugs, it is mandatory that the drug candidates are tested in relevant nonclinical species via the inhalation route, to assess both local and systemic effects of the drugs. 1 -3 Histopathological examination of the respiratory tract tissues from the inhalation toxicity studies in animals is regarded as one of the primary end points for assessing toxicity effects in order to predict the potential risk for human health. 4 -7 However, the evaluation of the respiratory tract and the interpretation of histopathology findings present many challenges. As a system, the respiratory tract often presents with relatively high incidences, and a wide range of both background and drug-induced lesions, 8,9 some of which show a characteristic distribution pattern that is both site- and species-specific. 10 The differentiation of background from treatment-related changes is further hampered by a paucity of information on the comparative pathology and the relevant incidence (historical control data [HCD]) of the more commonly occurring background or incidental pathology findings in some laboratory animal test species. 11 -13 Rodents have been extensively used in inhalation studies, and therefore, the response of the rodent respiratory tract to a wide variety of inhaled toxicants is well documented. 7,9,14 -16 Much less information is published on dogs and primates. In particular, for the cynomolgus monkey (Macaca fascicularis), which is the most widely used purpose-bred nonhuman primate in nonclinical safety testing, very little published information on spontaneous 12,13,17 and induced 8,9 respiratory tract lesions is readily available. Thus, there is a need for more information on the range, incidence of specific changes, and the most commonly affected sites/location of both incidental and drug-induced lesions in the respiratory tract following inhalation exposure by different drug compounds, in various laboratories.
This study was therefore designed to provide reference material documenting the nature and incidence of the background (including vehicle-induced) and test article–related nonneoplastic pathology findings in the respiratory tract of the cynomolgus monkey and rat in studies up to 13 weeks in duration for the nonhuman primates and 26-week duration for the rat. These findings represent the first multi-institutional effort that we are aware of to establish respiratory HCD and were derived from 3 distinct contract research organization (CRO) facilities. In addition, the relative frequency of induced findings, representing 44 different NCE (New Chemical Entity/small molecule) test articles, is presented. It is hoped that these data will aid in the identification and interpretation of test article–related findings, and their differentiation from background changes. Since data from vehicle controls were included in the analysis of background findings, and some findings classified as “background lesions” were considered vehicle-related, it must therefore be clarified that in this study “background” and “spontaneous” are not necessarily synonymous.
Materials and Methods
Study Design
Two separate retrospective analyses were carried out to collect background and induced pathology data. Studies were conducted at 3 geographically distinct CRO facilities referred to as 1, 2, and 3.
The first survey was carried out to compile background pathology findings in the respiratory tract of control animals in inhalation studies in cynomolgus monkeys (29 studies of 10 days to 13 weeks duration, evaluated between 2007 and 2019), Han Wistar (HW) rats (22 studies of 4 to 13 weeks duration, evaluated between 2013 and 2019) and Sprague Dawley (SD) rats (30 studies of 4 to 26 weeks duration, evaluated between 2011 and 2018). The vehicles used to dose the control groups in the inhalation studies were air, nebulized vehicle, or powdered vehicle. As a comparison, data were also compiled from non-inhalation controls (primarily from oral gavage studies) in cynomolgus monkeys (48 studies of 2- to 39-week duration, evaluated between 2012 and 2018), HW rats (22 studies of 4- to 26-week duration, evaluated between 2015 and 2018), and SD rats (63 studies of 4- to 13-week duration, evaluated between 2013 and 2018).
The second survey collected pathology data on the commonly encountered induced changes seen in inhalation studies in cynomolgus monkey and rats exposed to NCE/small molecule test articles via the inhalation route. Data were gathered from 10 cynomolgus monkey studies (representing nine different small molecule test articles) of 2- to 13-week duration evaluated between 2013 and 2019. Data were also collected from inhalation studies conducted in HW and SD rats and evaluated between 2013 and 2019, representing 28 test articles comprising 18 studies of 4-week duration and 11 studies of 13-week duration in the HW rats, and 9 test articles comprising 9 studies of 4-week duration in the SD rats. Overall, our review included 44 different NCE small molecule test articles, as 2 of them were administered to both species.
Data were collected by reviewing a total of 48 pathology reports for test article–related effects and extracting relevant data from various pathology data capture systems. Synonymous terms were merged (see Appendix 1) to harmonize nomenclature across different studies, using current INHAND (International Harmonization of Nomenclature and Diagnostic Criteria) terms where possible.
Criteria for inclusion into the study survey where possible were: small molecule test article (NCE) inhalation route of administration study duration of between 2 and 26 weeks full tissue list including the recommended panel of respiratory tissues to be evaluated in inhalation studies (OECD guidance).
All test articles included in these studies were administered as liquid or dry powder aerosol. Administration was by face mask for cynomolgus monkey and from a chamber for tube restrained or nose (snout)-tubes for the rats. The length of exposure was typically 1 hour per day (ranging from 20 minutes to 6 hours per day in rats). For studies that included a recovery period, only the main phase of the study was included in the analysis, and the potential for recovery from test article–related effects was not evaluated.
Pathology reports of the selected studies were reviewed to identify findings considered to be test article–related, based on the interpretation and judgment of the original study pathologist. There were no attempts to reinterpret the findings. Microscopic findings in the respiratory tract as reported in the original report were merged (to reduce the number of synonymous terms and modifiers and/or use INHAND terminology if considered appropriate) and tabulated to generate incidence data. The severity grade (or score) of the microscopic findings was not included in the tables to simplify the reporting of the large data set. The key anatomic sites assessed were lungs, bronchial lymph node, nasal cavities, pharynx, larynx, trachea, and the tracheal bifurcation (carina).
Animal Management
All studies and procedures were conducted in accordance with study plans, the principles of Good Laboratory Practice and local legislation for conducting scientific procedures on animals, respectively, the UK Home Office Legislation (Animals [Scientific Procedures] 1986 Act (Hollands, 1986), which conforms to the European Convention for the Protection of Vertebrate Animals Used for Experimental or Other Scientific Purposes (Strasburg, Council of Europe) for studies conducted in the United Kingdom, and in accordance with Health Canada Legislation for studies conducted in Canada. The following strains of laboratory species were used in this review: cynomolgus monkeys sourced from Vietnam or China and aged between 27 and 40 months; HW rats aged between 36 and 83 days, from the following suppliers: Charles River limited, Envigo RMS limited, and Harlan Ltd; SD from the following suppliers: Charles River (United Kingdom) limited and Charles River Canada Inc.
Necropsy and Histology Procedures
Necropsies were conducted according to local standard operating procedures. Lung tissues were inflated via the trachea with neutral buffered 10% formalin to approximately the same volume as the functional residual capacity of the lungs by visual observation and fixed by immersion in 10% neutral buffered formalin. All the other tissues were fixed by immersion in 10% neutral-buffered formalin. For the lung, nasal cavities, larynx, trachea, and tracheal bifurcation, the trimming strategies and published guidelines for inhalation studies at each independent laboratory are summarized in Table 1. Tissues were conventionally processed, embedded in paraffin wax, sectioned at 4 to 6 μm, and stained with hematoxylin and eosin. All light microscopic evaluations were subjected to peer review by a second pathologist.
Trimming Strategies in Inhalation Studies at Each CRO’s Site in the Rat and Cynomolgus Monkey.
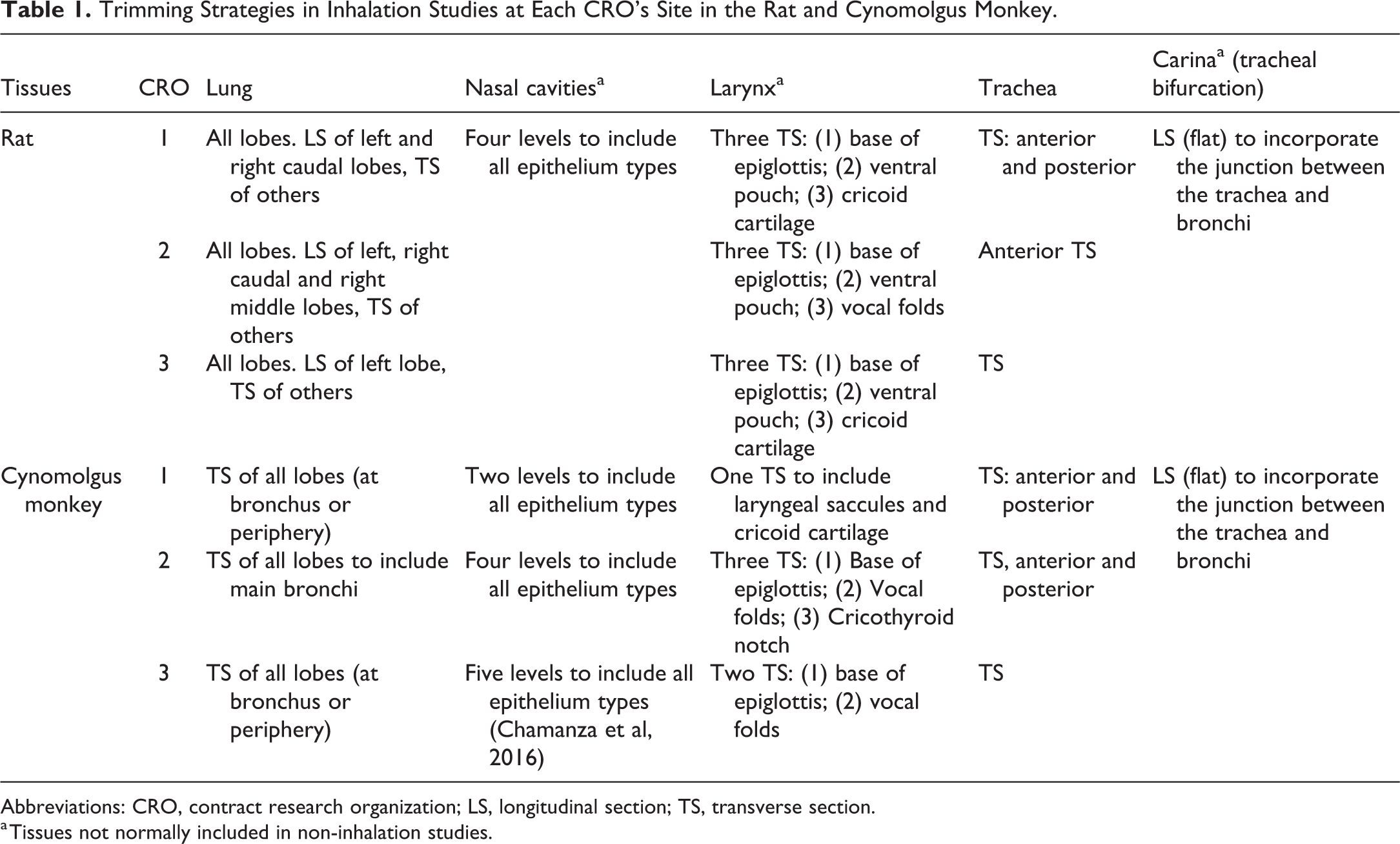
Abbreviations: CRO, contract research organization; LS, longitudinal section; TS, transverse section.
a Tissues not normally included in non-inhalation studies.
Percentage Background Lung Findings in Control Cynomolgus Monkeys—Inhalation Versus Non-Inhalation Studies.
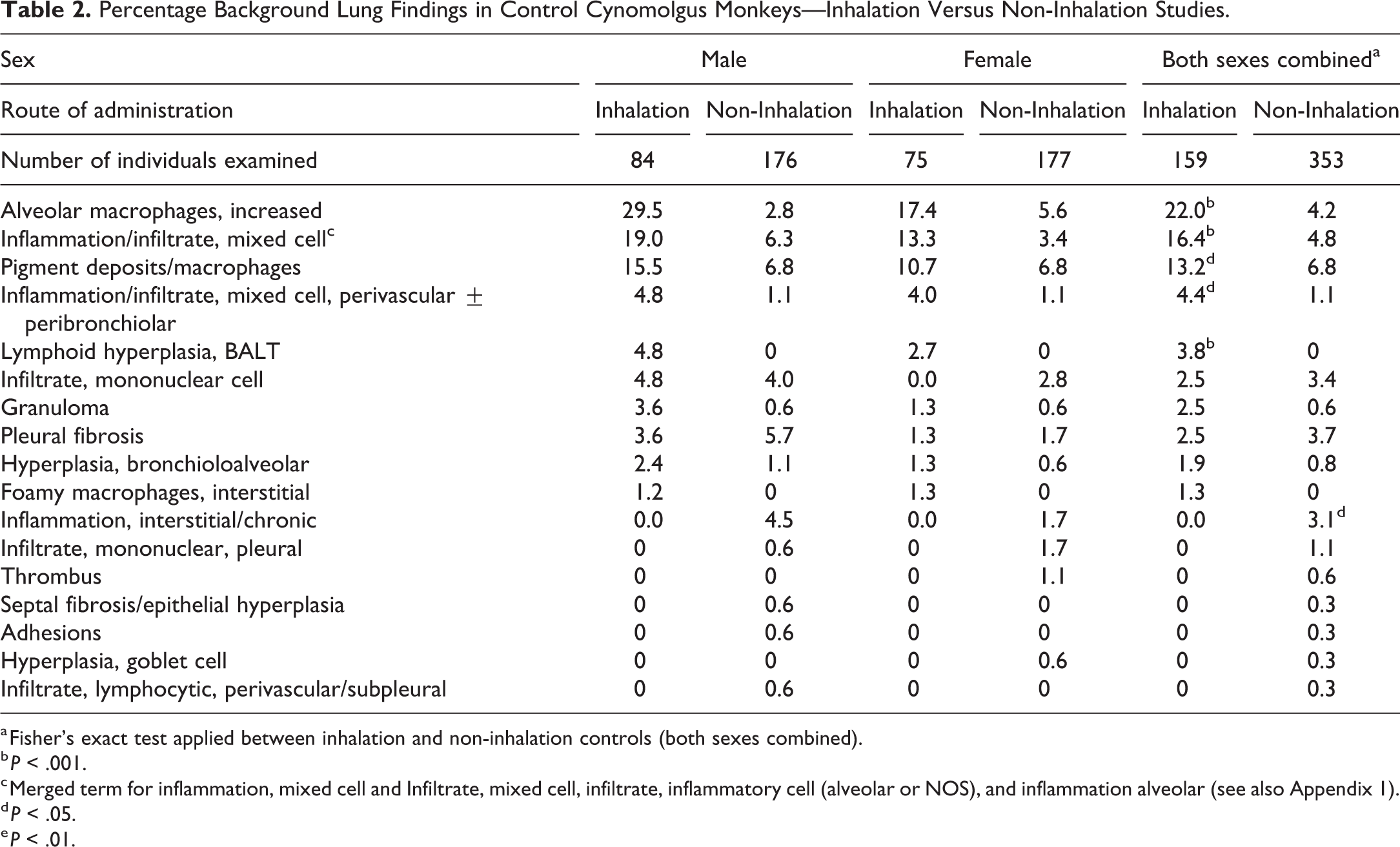
a Fisher’s exact test applied between inhalation and non-inhalation controls (both sexes combined).
b P < .001.
c Merged term for inflammation, mixed cell and Infiltrate, mixed cell, infiltrate, inflammatory cell (alveolar or NOS), and inflammation alveolar (see also Appendix 1).
d P < .05.
e P < .01.
Statistics
Pairwise comparison by Fisher exact test (2-tailed) was applied to the incidence of findings in controls, comparing non-inhalation and inhalation control values of both sexes combined. For rats, the 2 strains were compared separately (Tables 3 and 4) and combined (Table 5B). Within inhalation controls, powdered vehicle and nebulized (liquid) vehicle values were compared against air control values (both sexes, and for rats both strains, combined).
Percentage Background Lung Findings in Control Han Wistar Rats—Inhalation vs Non-Inhalation Studies.
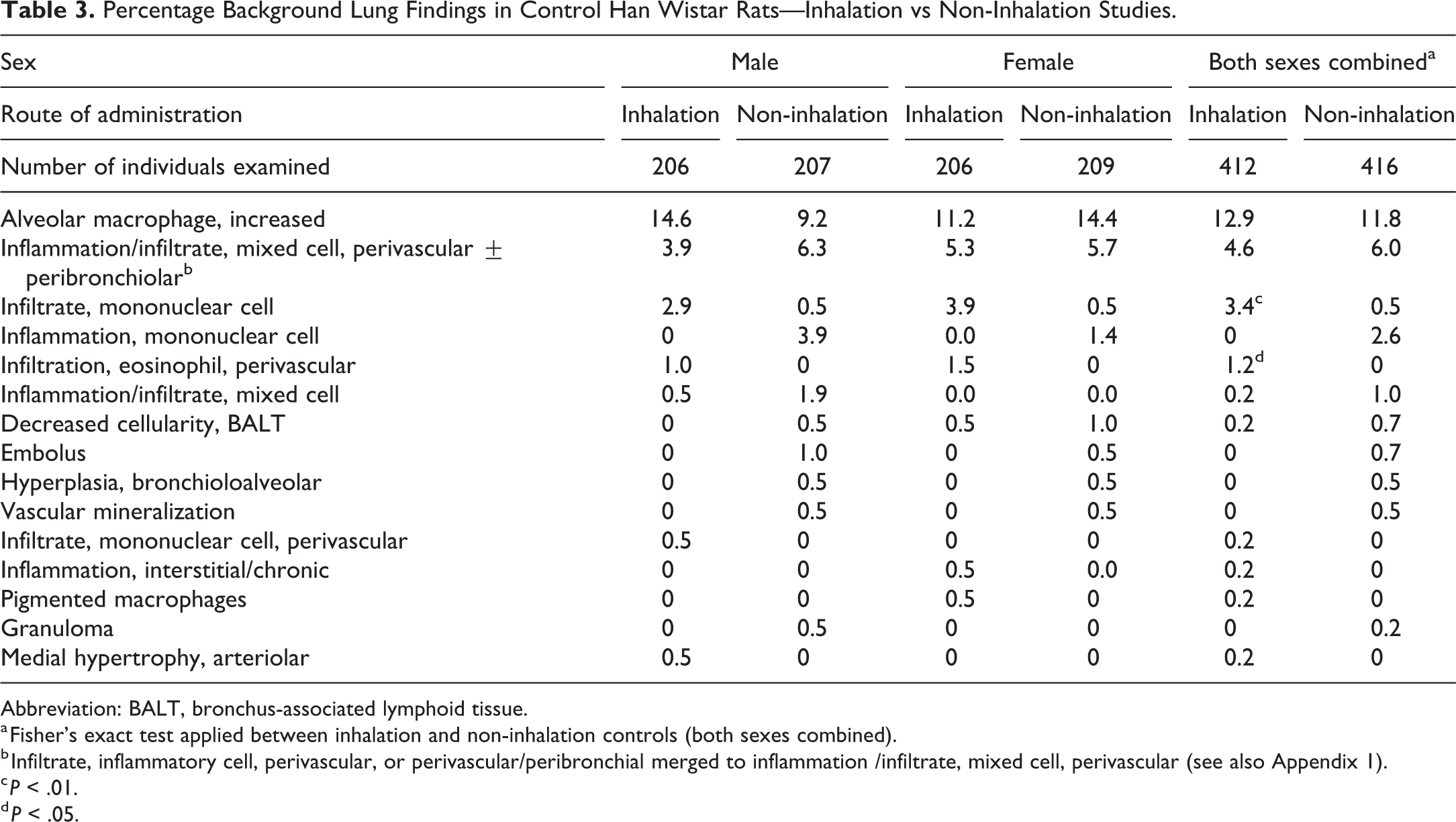
Abbreviation: BALT, bronchus-associated lymphoid tissue.
a Fisher’s exact test applied between inhalation and non-inhalation controls (both sexes combined).
b Infiltrate, inflammatory cell, perivascular, or perivascular/peribronchial merged to inflammation /infiltrate, mixed cell, perivascular (see also Appendix 1).
c P < .01.
d P < .05.
Percentage Background Lung Findings in Control Sprague Dawley Rats—Inhalation vs Non-Inhalation Studies.
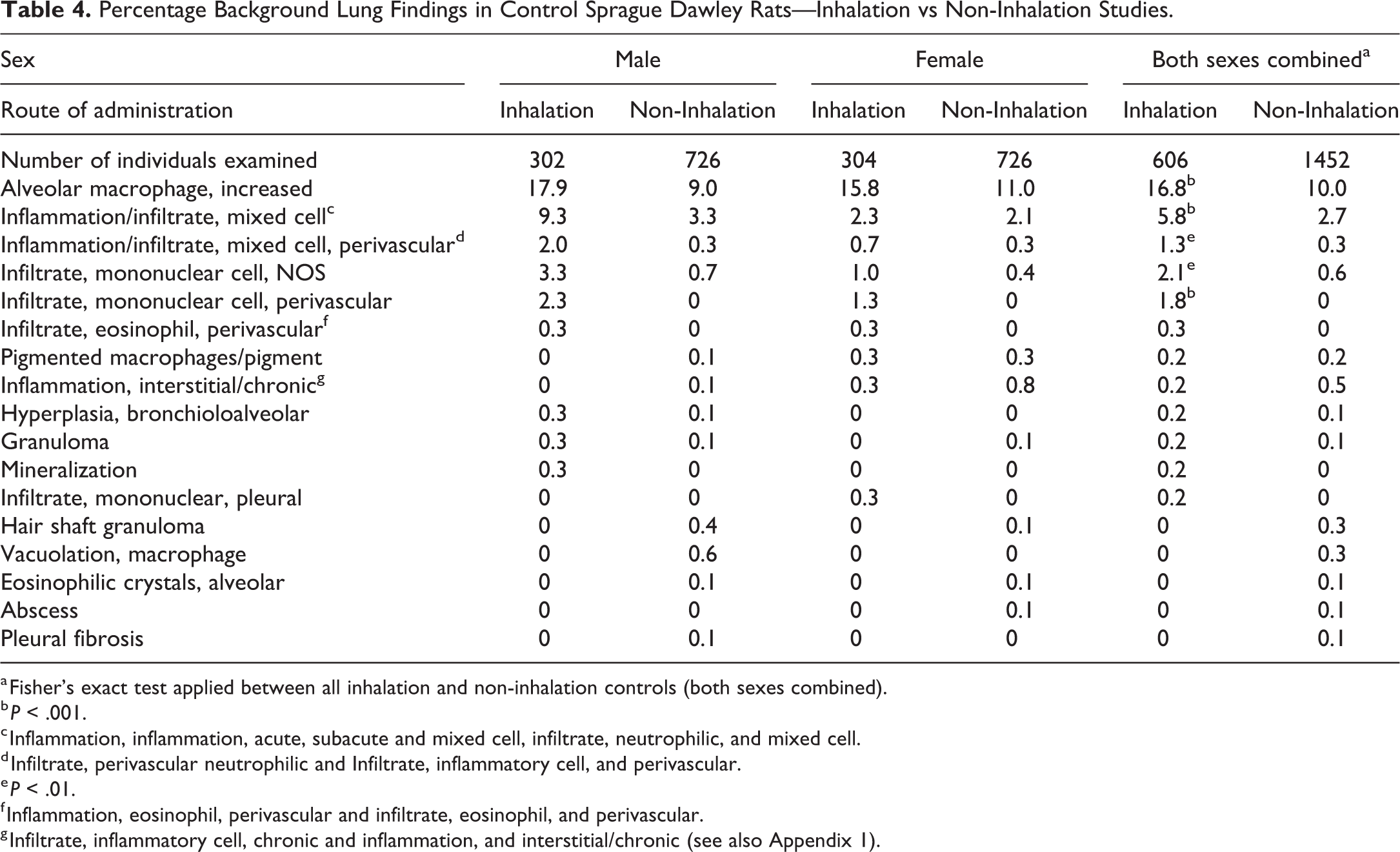
a Fisher’s exact test applied between all inhalation and non-inhalation controls (both sexes combined).
b P < .001.
c Inflammation, inflammation, acute, subacute and mixed cell, infiltrate, neutrophilic, and mixed cell.
d Infiltrate, perivascular neutrophilic and Infiltrate, inflammatory cell, and perivascular.
e P < .01.
f Inflammation, eosinophil, perivascular and infiltrate, eosinophil, and perivascular.
g Infiltrate, inflammatory cell, chronic and inflammation, and interstitial/chronic (see also Appendix 1).
Results
Overall Incidence and Patterns of Background Findings
In both cynomolgus monkeys and rats, the most commonly recorded background findings in the respiratory tract were seen in the lung and occurred more frequently in inhalation compared to non-inhalation controls (Tables 2-5B). After the lung, the second most commonly affected organ was the nasal cavity in the monkey and SD rat and the larynx in the HW rat.
In both species, the most commonly recorded background changes in the respiratory tract were minimal or mild inflammatory cell infiltrates of various types (increased macrophages and mononuclear cell infiltrates, Figures 1 and 2). Inflammation, which was consistently used by study pathologists as a compound term representing an inflammatory cell infiltrate plus indicators of tissue damage (such as edema, hemorrhage, or necrosis), was also relatively frequently encountered. Other background findings occurred at lower incidences in both species and study types, with the exception of pigment deposits/macrophages which was relatively commonly observed in monkeys in the lungs at 1 facility. Further details of the background findings in each species and study type are described below, and the incidences presented in Tables 2 to 4.
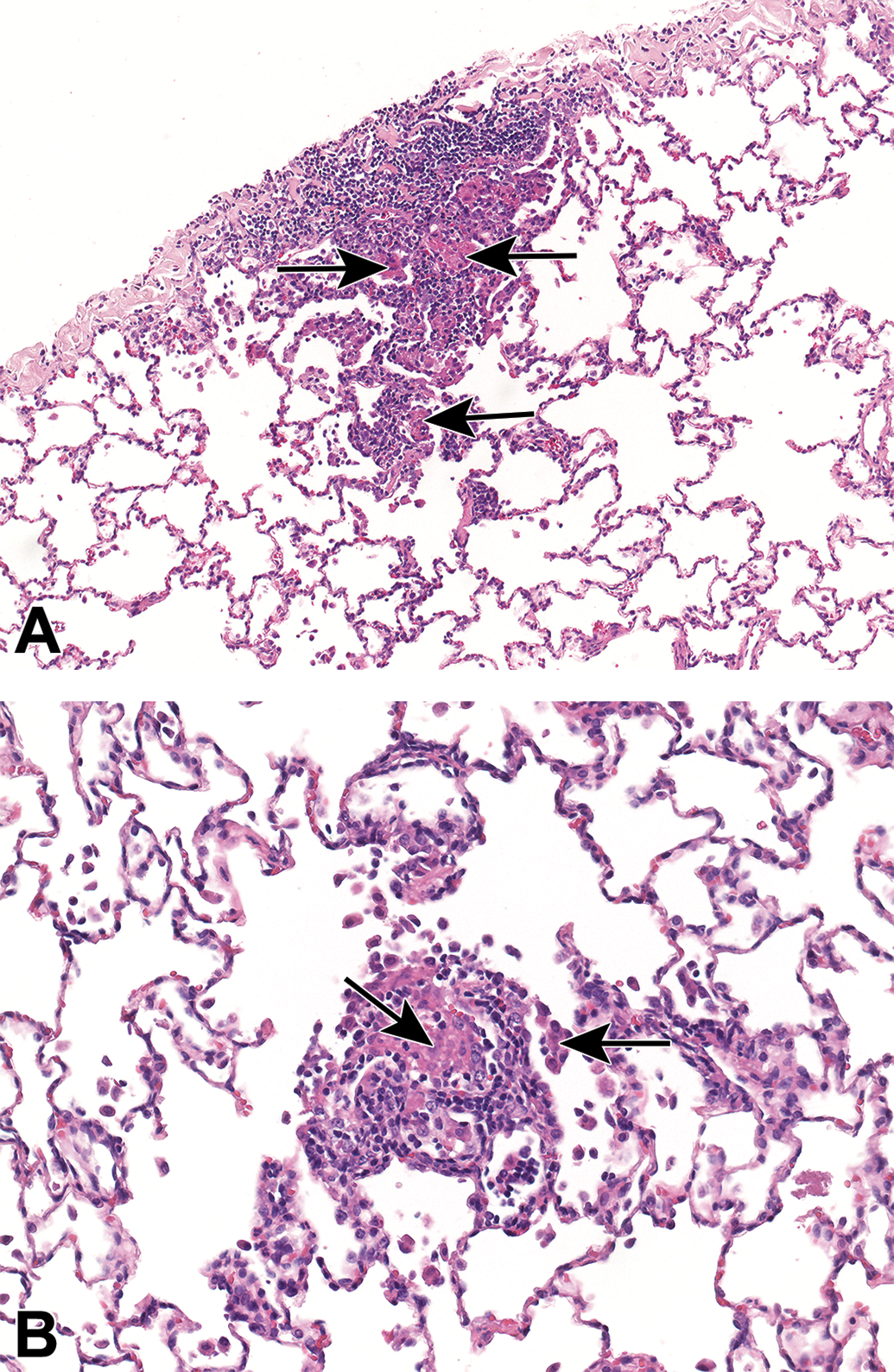
Background findings in the lungs of cynomolgus monkeys. Mononuclear cell infiltrates including macrophage aggregates (arrows), with a subpleural (A) and alveolar distribution (B).
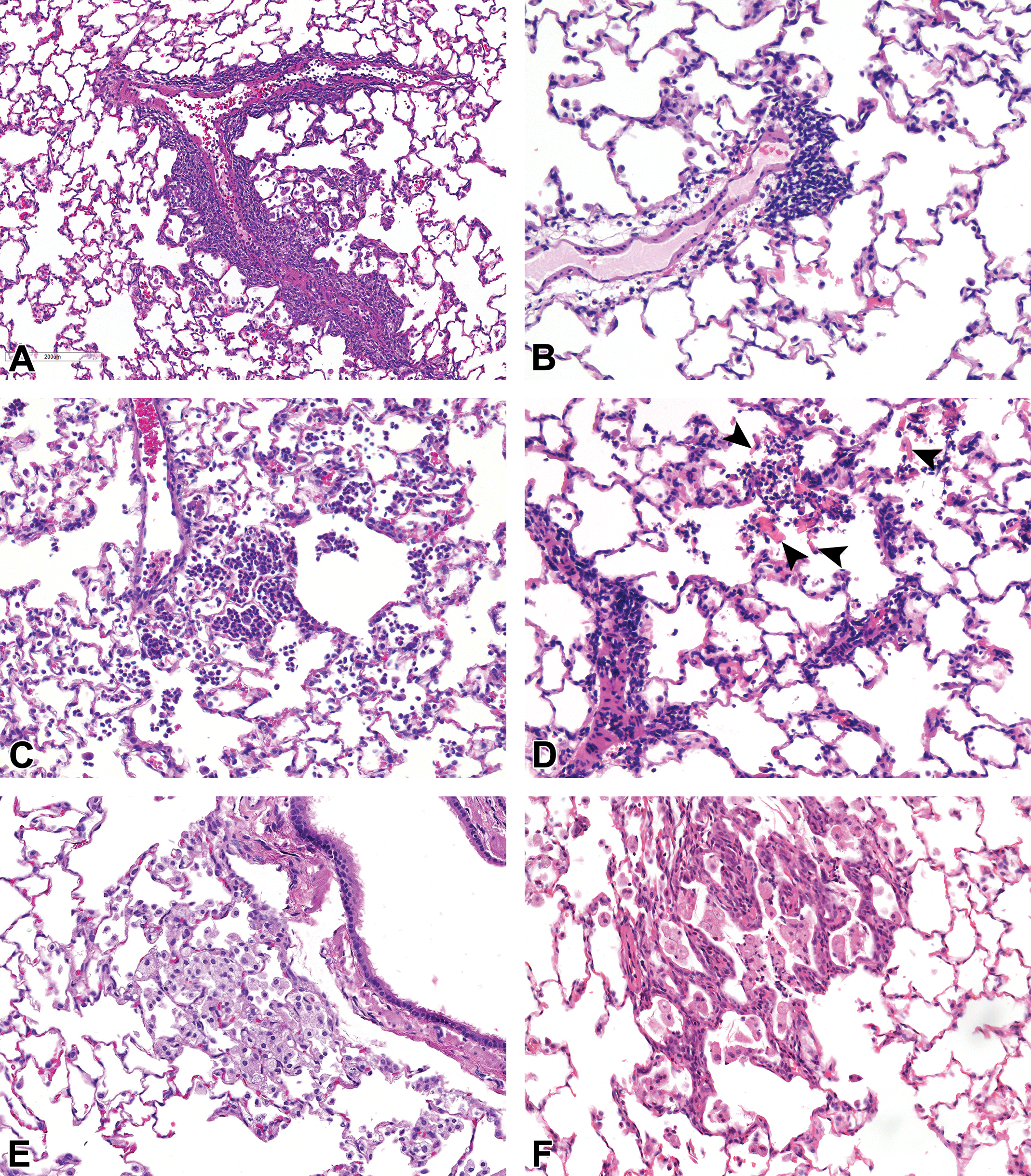
Background findings in the lungs of rats. A and B, Perivascular mononuclear cell infiltrate, (C) alveolar mixed cell infiltrate, (D) mixed cell infiltrate and eosinophilic crystals (arrowheads), (E) alveolar macrophages, and (F) alveolar macrophages and bronchioloalveolar hyperplasia.
Common background findings in lungs
In the lung, which was the most affected tissue, increased alveolar macrophages were the most commonly recorded background change in both species from inhalation studies, occurring more commonly in monkeys (22.0%) compared to rats (16.8% and 12.9% SD and HW respectively, Figures 1 and 2). However, in non-inhalation studies, increased alveolar macrophages occurred at a higher incidence in rats (11.8% and 10% HW and SD, respectively) compared to cynomolgus monkeys (4.2%). In both species, increased alveolar macrophages in control animals were usually recorded as minimal or mild focal changes, distributed either in the subpleural location or at the bronchioloalveolar junction (centriacinus; personal observations), and often without substantial inflammatory changes (Figure 2E), but may also be associated with mononuclear cell infiltrates (Figure 1A and B). In monkeys, most alveolar macrophages, were not foamy, and occasionally contained pigment (personal observations). Inflammatory or mononuclear inflammatory cell infiltrates, which were often distributed in a PV location (Figures 2A and B), were the second most frequently observed finding in control rats and monkeys on inhalation studies. The incidences of inflammatory cell infiltrates and inflammation were generally similar between monkeys and rats, although inhalation monkeys had a noticeably higher level (16.4%). Increased lymphoid cellularity of bronchus-associated lymphoid tissue (BALT) hyperplasia was only recorded in monkeys.
Comparison of incidences of background lung findings between inhalation and non-inhalation controls
A comparison of the incidences of background respiratory tract findings between inhalation and non-inhalation studies is presented in Table 2 to 5B. The 5 most common background findings in the lungs of control cynomolgus monkeys showed statistically significantly (using a 2-tailed Fisher exact test) higher incidences in inhalation compared to non-inhalation controls (Table 5B). The incidence of increased alveolar macrophages (P < .001; 22.0% vs 4.2%); inflammation/infiltrate, mixed cell (P < .001; 16.4% vs 4.8%; Figure 2C); inflammation/infiltrate, mixed cell, perivascular (PV) ± peribronchiolar (PB; P < .05; 4.4% vs 1.1%); and BALT lymphoid hyperplasia (P < .001; 3.8% vs 0%) were significantly higher than the respective incidences recorded in non-inhalation controls (Table 5A).
Summary of Selecteda Lung Findings in Cynomolgus Monkeys.
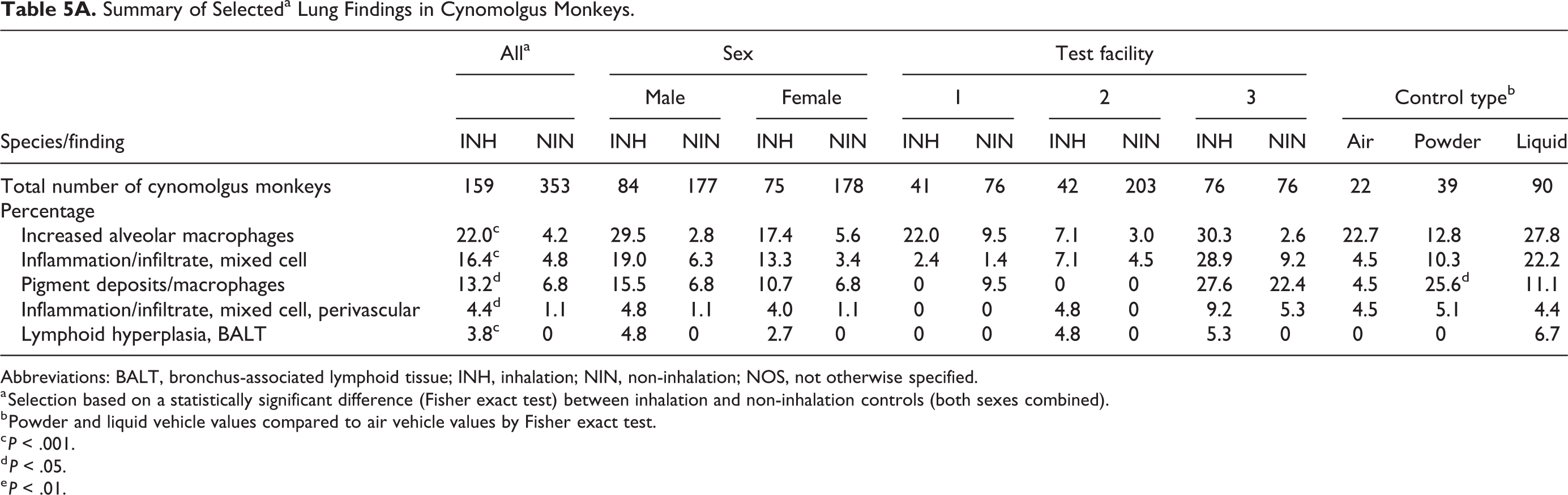
Abbreviations: BALT, bronchus-associated lymphoid tissue; INH, inhalation; NIN, non-inhalation; NOS, not otherwise specified.
a Selection based on a statistically significant difference (Fisher exact test) between inhalation and non-inhalation controls (both sexes combined).
b Powder and liquid vehicle values compared to air vehicle values by Fisher exact test.
c P < .001.
d P < .05.
e P < .01.
Summary of Selecteda Lung Findings in Rats.

Abbreviations: INH, inhalation; NIN, non-inhalation; NOS, not otherwise specified.
a Selection based on a statistically significant (P < .05) difference (Fisher exact test) between inhalation and non-inhalation controls (both sexes/strains combined).
b Powder and liquid vehicle values compared to air vehicle values by Fisher exact test.
c P < .001.
d P < .05.
e P < .01.
A comparison of the incidences of background respiratory tract findings between inhalation and non-inhalation rat studies is presented in Tables 3, 4, and 5B. As was seen with cynomolgus monkeys, increased alveolar macrophages occurred at a higher incidence in inhalation compared to non-inhalation studies in SD (P < .001; 16.8% vs 10.0%) but not HW rats (12.9% vs 11.8%). There was also a higher incidence of inflammation/infiltrate, mixed cell in inhalation compared to non-inhalation studies in SD rats (P = .008; 1.3% vs 0.3%) but not HW rats (4.6% vs 6.0%). The incidence of mononuclear cell infiltrates (NOS) was higher in inhalation compared to non-inhalation controls in both SD (P = .002; 2.1% vs 0.6%) and HW (P = .002; 3.4% vs 0.5%) rats. The incidence of PV mononuclear cell infiltrates in SD rats was also higher in inhalation studies (P < .001; 1.8% vs 0%).
Comparison of incidences of background lung findings between air and vehicle controls
In addition to the comparison between inhalation and non-inhalation control cynomolgus monkeys, in the lung a further comparison was made between inhalation controls dosed with air, nebulized vehicle, or powdered vehicle (Table 5A-5B). Higher incidences were observed in nebulized and/or powdered vehicle controls for several findings, relative to air controls, but the only statistically significant differences were a higher incidence of pigment in the lungs in powdered vehicle controls (25.6%) compared with air controls (4.5%; P = .045). Notably, there was a similar difference in the bronchial lymph node, 35.9% in powdered vehicle controls compared with 4.5% in air controls (P = .002). However, as explained in the following section, this was not a true effect of the control type. In the rat, there were higher incidences of increased macrophages in nebulized controls (15.3% vs 9.9%, P = .043) and powder controls (15.4% vs 9.9%, P = .064) versus air controls.
Lung pigment in cynomolgus monkeys
In cynomolgus monkeys, there were trends in the incidences of pigmented macrophages or pigment deposits in the lung and pigment deposits in the bronchial lymph node. Notably, these changes nearly all occurred in studies from one distinct CRO site and were considered to be linked to the geographic region of their supplier’s facilities (South East Asia). Similar findings were rarely observed in animals supplied from other geographic regions (primarily Mauritius). These findings were therefore considered to be the result of carbon particle inhalation at the supplier’s facility due to variations in air quality. Interestingly, when the data from inhalation studies using monkeys from South East Asia suppliers were analyzed in isolation, a comparison between different control methods, revealed that powder controls had higher incidences of pigment than air controls in the lung (38.5% vs 6.3%, P = .021) and bronchial lymph node (53.8% vs 6.3%, P = .016). However, this was likely the result of the low number of air control studies (3 studies with a total of 16 individuals). This was supported by the incidence of pigmented macrophages in the lung of non-inhalation controls from the same source (26.7%), which was not statistically significant different from the powder controls (P = .19).
Common background findings in larynx, nasal cavity, and trachea
The larynx was sampled in only a small proportion of cynomolgus monkeys on non-inhalation studies. Of the 355 control monkeys from non-inhalation studies, the larynx was evaluated in only 42 animals (12%, from 36 studies). Background findings in the larynx of cynomolgus monkeys generally occurred at a very low incidence in all studies, but in inhalation controls, lymphoid hyperplasia and inflammation (3.9 and 1.9%, respectively) were observed. Other isolated findings recorded in inhalation studies included epithelial degeneration/regeneration, goblet cell hyperplasia, and inflammatory cell infiltration (each recorded in single animals). No findings were recorded in the larynx from the non-inhalation studies in cynomolgus monkeys included in this review. In monkeys, there were no statistically significant differences in any finding with respect to the type of control employed.
In control rats on inhalation studies, the larynx was the second most affected respiratory tissue and squamous metaplasia/hyperplasia (5.6% and 2.5% in HW and SD rats, respectively) and mixed cell infiltration (1.4% in SD rats) were the most commonly recorded findings. Other findings occurred at lower incidences and/or were observed at a similar incidence in non-inhalation animals. In the larynx, in addition to the comparison between inhalation and non-inhalation controls, a further comparison was made between findings in inhalation controls dosed with air, nebulized vehicle, or powdered vehicle (data not shown). In rats (using combined strain data), higher incidences were observed in nebulized and/or powdered vehicle controls for several findings, relative to air controls. These were, squamous metaplasia in nebulized controls (6.9% vs 1% in air controls, P < .001), epithelial alteration in nebulized controls (1.4% vs 0% in air controls, P = .004), and mixed cell infiltrate in powder controls (1.7% vs 0% in air controls, P = .012). However, these were likely to be a consequence of the specific vehicles rather than the administration method, as the majority of squamous metaplasia and all epithelial alteration cases received benzalkonium chloride, 18 and all mixed cell infiltration cases received a sugar or sugar alcohol (lactose or mannitol).
The nasal cavities were sampled and evaluated in almost all the control cynomolgus monkeys from inhalation studies (96%, 152/159 animals) and in approximately 21% (73/355 animals) of those from non-inhalation studies (data not shown). In inhalation controls, the most common finding was mononuclear cell infiltrate in the lamina propria (data not shown). There were no findings recorded in nasal cavities from animals on non-inhalation studies. The nasal cavities were evaluated in all rats from inhalation studies, but only in 35% of SD rats and 4.8% of HW rats from non-inhalation studies (data not shown). In SD rats on inhalation studies, the only notable finding was exudate in the lumen (6.1% compared vs 0% in the non-inhalation controls). A few findings were recorded in the non-inhalation controls, but since they were all present in a single study, they were not considered to be representative of general background findings. No notable findings were noted in HW rats.
In the trachea of cynomolgus inhalation controls, there were 2 findings of note, mononuclear cell infiltrate and inflammation, that occurred at a low incidence and were not present in non-inhalation controls (data not shown). Other findings were observed with isolated or very low incidence. In rats, there were a few findings present in inhalation controls that were not present in non-inhalation controls, but all occurred at a low incidence such as mucosal hyperplasia, mixed cell infiltration and mononuclear cell exudate in SDs, and loss of cilia and mixed inflammatory cell infiltrate in HWs.
Part 2: Induced Findings in the Respiratory Tract
Overall Frequency of Induced Findings
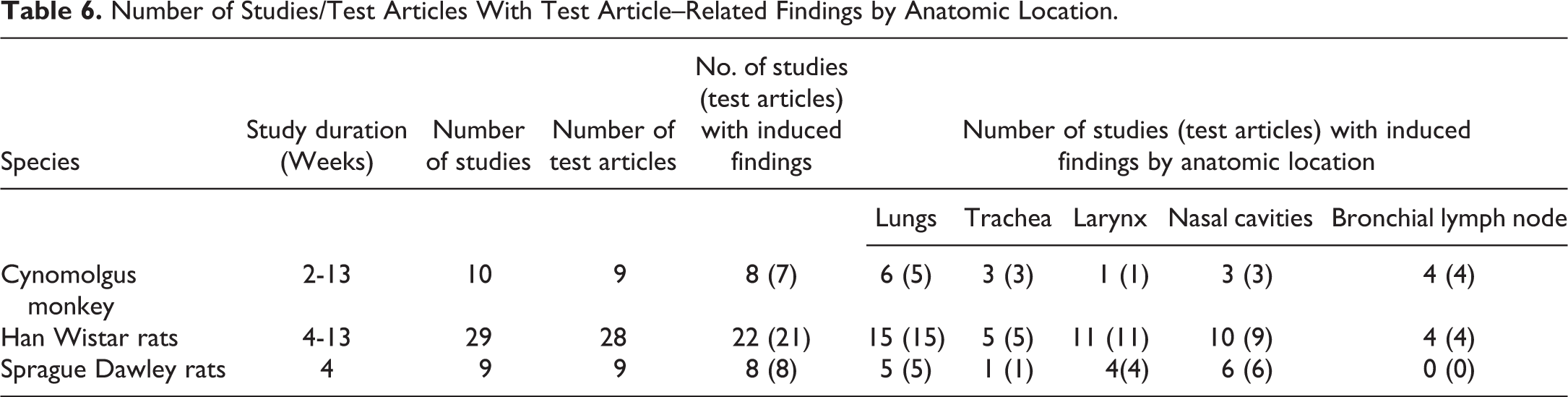
The overall incidences and frequency of the most commonly recorded test article–induced findings in the respiratory tract of cynomolgus monkeys and rats from inhalation studies of 4- to 13-week duration are presented in Table 6. Test article–related effects were identified in 80% (8 of 10) of the studies in the cynomolgus monkey (representing 7 test articles), in 76% (22 of 29) of the studies in HW rats (representing 20 test articles) and 88.8% (8 of 9) of the studies in SD rats (representing 8 test articles). The most frequently affected organ in both species was the lung, followed by the larynx and the nose in the rat, and the trachea and nose in the monkey.
Number of Studies/Test Articles With Test Article–Related Findings by Anatomic Location.
Test article–induced lesion in the lungs and draining lymph node of Cynomolgus monkeys
The summary of incidences of the most commonly recorded test article–induced findings in the respiratory tract of cynomolgus monkeys are presented in Table 7 (Figure 3). In monkeys, test article–induced lesions in the lungs were observed in 60% (6 of 10) of the studies, representing 5 different test articles. The most common treatment-related pathology findings in the lungs were increased alveolar macrophages (often described as alveolar macrophage aggregations by the original study pathologists) and were observed in 50% (5 of 10) of the studies with 5 different test articles. The lesion was characterized by multifocal accumulations of macrophages that were occasionally described as foamy. The accumulations were generally reported as having a bronchiolar distribution (Figure 3A) and localized in the terminal airways (junction of respiratory bronchioles and alveolar ducts) and adjacent alveoli or in the interstitial space. In 2 of the studies, increased macrophages were described as being associated with a mixed inflammatory cell infiltrate containing granulocytes and lymphocytes in the terminal airways/alveoli, while in one other study, they were associated with acute bronchoalveolar inflammation. The severity of these changes was variable from minimal to marked. Macroscopic correlates were inconsistently reported as multifocal pale areas or areas of discoloration in the lungs. Increased individual and or mean organ weights was reported in only 2 studies, which showed a high severity of these changes, graded as moderate to marked.
Incidence of Pathology Findings in the Respiratory Tract of Cynomolgus Monkeys Treated With 7 Inhaled New Chemical Entities/Small Molecules for 2 to 13 Weeks.
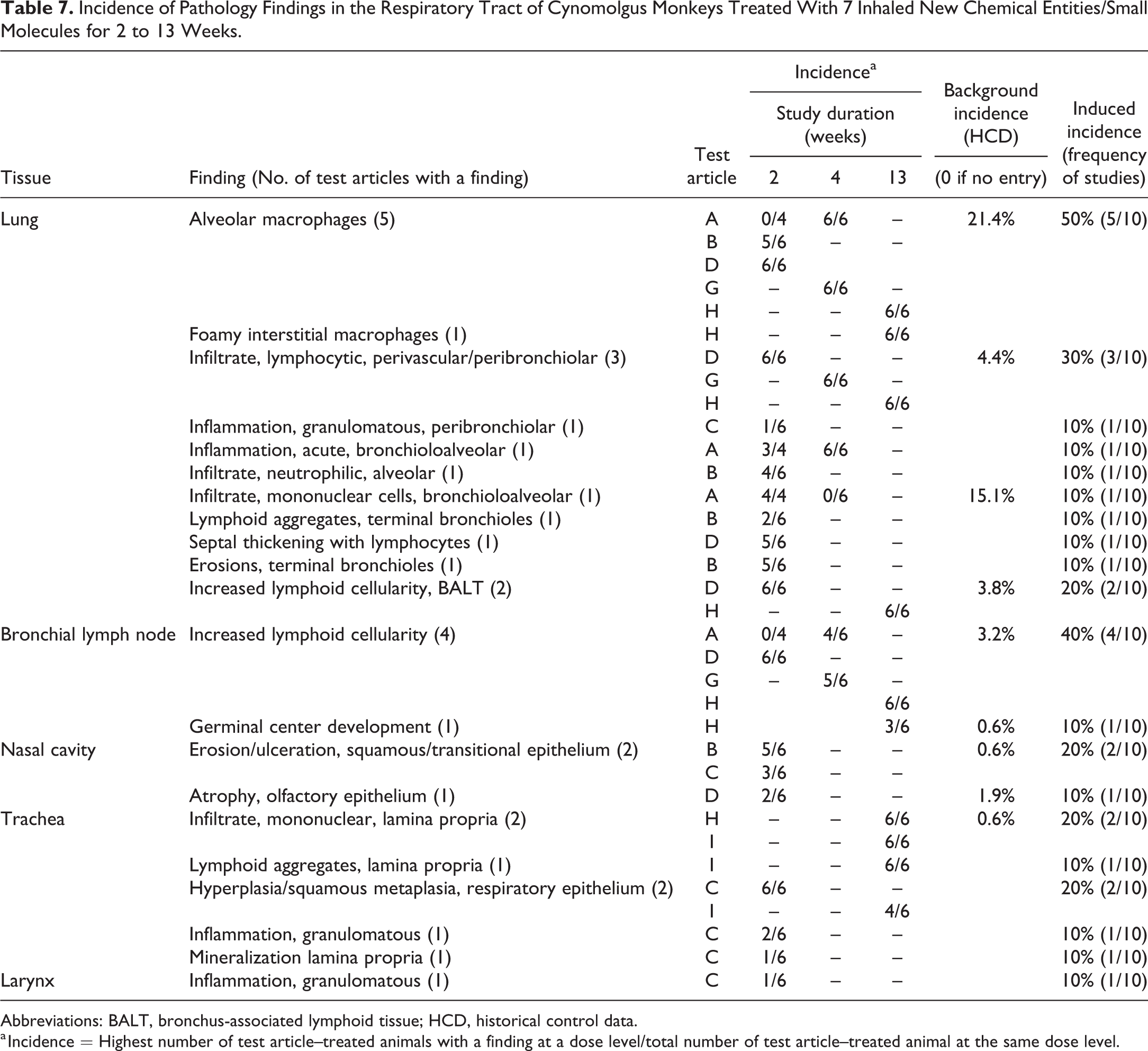
Abbreviations: BALT, bronchus-associated lymphoid tissue; HCD, historical control data.
a Incidence = Highest number of test article–treated animals with a finding at a dose level/total number of test article–treated animal at the same dose level.
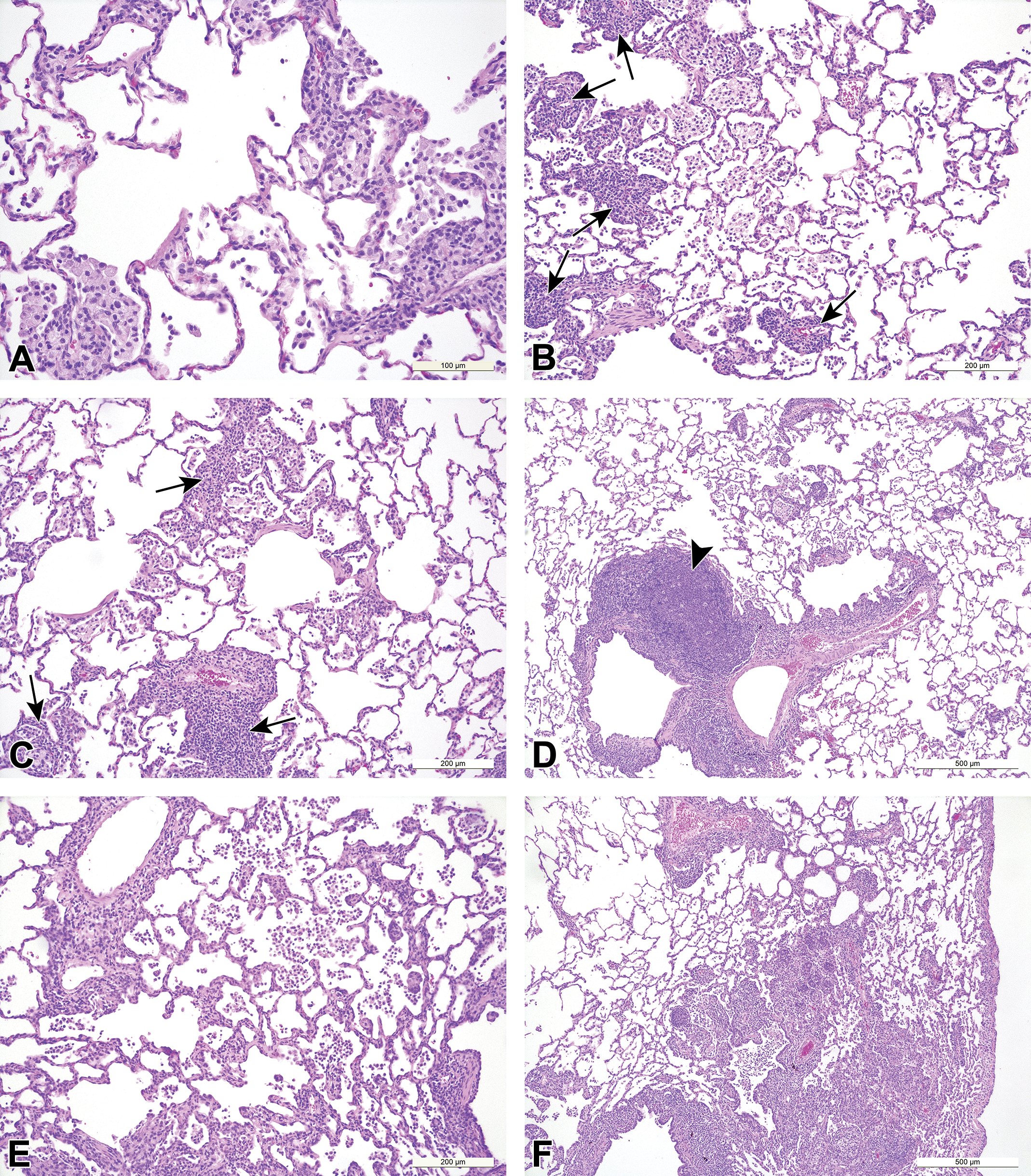
Small molecule-induced findings in the lungs of cynomolgus monkeys. A, Alveolar macrophage accumulation with a bronchioloalveolar distribution, (B, C, and D) alveolar macrophages and peribronchiolar/perivascular mononuclear cell infiltrate (arrows), accompanied by increased BALT cellularity (D, arrowhead), (E) increased alveolar macrophages, alveolar mononuclear cell infiltrate and septal thickening, and (F) increased alveolar macrophages and mononuclear cell inflammation.
The second most commonly recorded induced finding in the lungs of monkeys was mononuclear or lymphocytic/lymphoplasmacytic inflammatory cell infiltrates, which presented, was most often recorded as “PV/ PB mononuclear cell infiltrates,” and was reported in 30% (3 of 10) of studies, representing 3 different test articles (Figure 3B-D). These changes were graded as minimal and were mainly composed of lymphocytic infiltrates with a PV and/or PB distribution and occurred in association with BALT hyperplasia in 2 studies. Notably, the combination of PV/PB mononuclear cell infiltrate and BALT hyperplasia were only recorded in studies where the test article was a small molecule immuno-modulator. In the 3 studies where PV/PB mononuclear cell infiltrates was recorded, there was a concurrent enlargement/lymphoid hyperplasia of the draining bronchial lymph nodes, indicating immune stimulation. Test article–induced lymphoid hyperplasia/increased cellularity in the bronchial lymph nodes was overall observed in 40% or 4 of 10 studies, representing 4 different test articles. All the test articles were immunomodulators, and in one of these studies, germinal center development was also observed.
Test article–induced lesion in the lungs and draining lymph nodes of rats
The summarized incidences of the most commonly recorded test article–induced findings in the respiratory tract of HW and SD rats are presented in Table 8 and Table 9, respectively (Figure 4). Test article–induced changes in the lungs were observed in 51.7% (15 of 29) of studies in HW rats, which represented 15 different test articles, and in 30% (3 of 9) of studies in SD rats which represented 2 different test articles. As in the monkey, increased alveolar macrophages were the most common findings and were observed in 51.7% (15 of 29) of studies, representing 15 different test articles in HW rats and in 22.2% (2 of 9) of studies, representing 2 test articles, in SD rats (Tables 8 and 9, respectively). These changes were characterized as a multifocal aggregation of foamy macrophages often distributed in the centriacinar region and recorded as a single entry (Figure 4B), but in a few studies, they were recorded together with the concurrent diffuse increase in the alveolar macrophages (Figure 4A), as 2 separate entries. The severity was reported as minimal to marked and a distinct dose response was observed in most of the studies. In 4 studies, diffuse increases in alveolar macrophages together with the PB aggregates resulted in increases in mean organ weight of the lungs compared to controls. Macroscopic correlates were inconsistently reported as multifocal pale areas. In HW rats, unlike in monkeys, alveolar macrophages aggregations, there was a trend toward an association with bronchioloalveolar hyperplasia, which was observed in 6.9% (2 of 29) of studies representing 2 test articles; acute/subacute alveolar inflammation (6.9% or 2 of 29 studies) and mononuclear cell infiltrate in the terminal airways and alveolar spaces (6.9% or 2 of 29 studies). The concurrent presence of eosinophilic crystals in the alveolar spaces or alveolar proteinosis in 2 of these studies was reported to be associated with the macrophage aggregations and considered to be suggestive of an impairment or overwhelming of the clearance mechanisms. In SD rats, macrophage aggregations were reported to be associated with bronchioloalveolar hyperplasia in 11.1% (1 of 9) of the studies. Induced lymphoid hyperplasia/increased cellularity in the tracheobronchial lymph nodes were observed in 10.3% or 3 of 29 studies (representing 3 test articles) in HW rats. In one of these studies, the hyperplastic changes observed in the tracheobronchial lymph node were considered to reflect the pharmacological action of the test article (immunomodulator) and occurred in parallel with BALT hyperplasia in the lungs. In the other 2 studies, hyperplasia of the tracheobronchial lymph nodes was associated with acute/subacute alveolar inflammation in the lungs and increased alveolar macrophages. No induced changes of the tracheobronchial lymph nodes were observed in the SD rat studies.
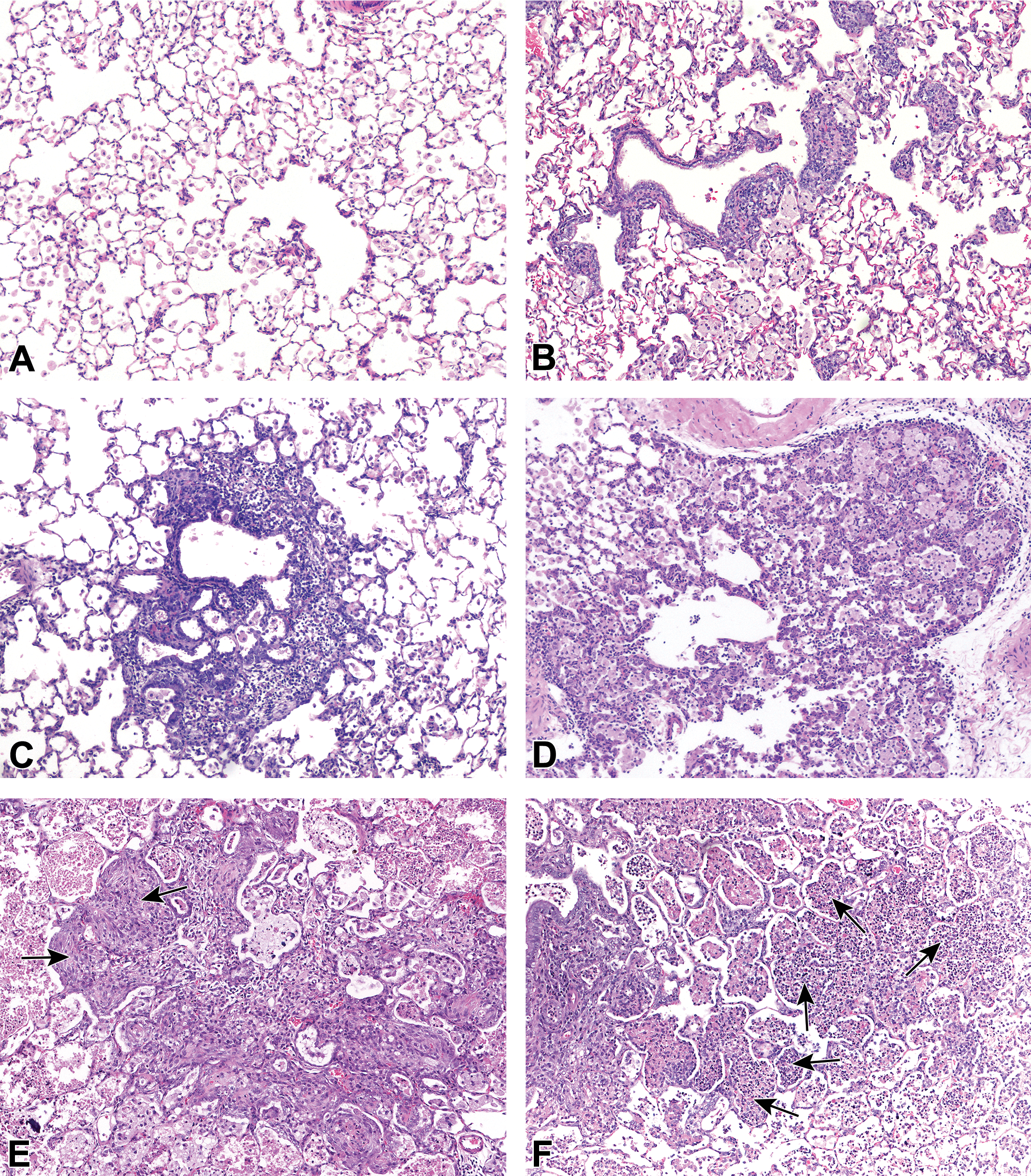
Small molecule–induced findings in the lungs of rats. A, Diffusely increased alveolar macrophages, (B) alveolar macrophages and peribronchiolar mononuclear cell infiltrate, (C) centriacinar inflammation and bronchioloalveolar hyperplasia, (D) alveolar mixed cell inflammation and bronchioloalveolar hyperplasia, (E) inflammation and fibrosis (arrows), and (F) inflammation and alveolar necrotic cellular debris (arrows).
Incidence of Pathology Findings in the Respiratory Tract of Han Wistar Rats Treated With 21 Inhaled New Chemical Entities/Small molecules for 4 to 13 Weeks.
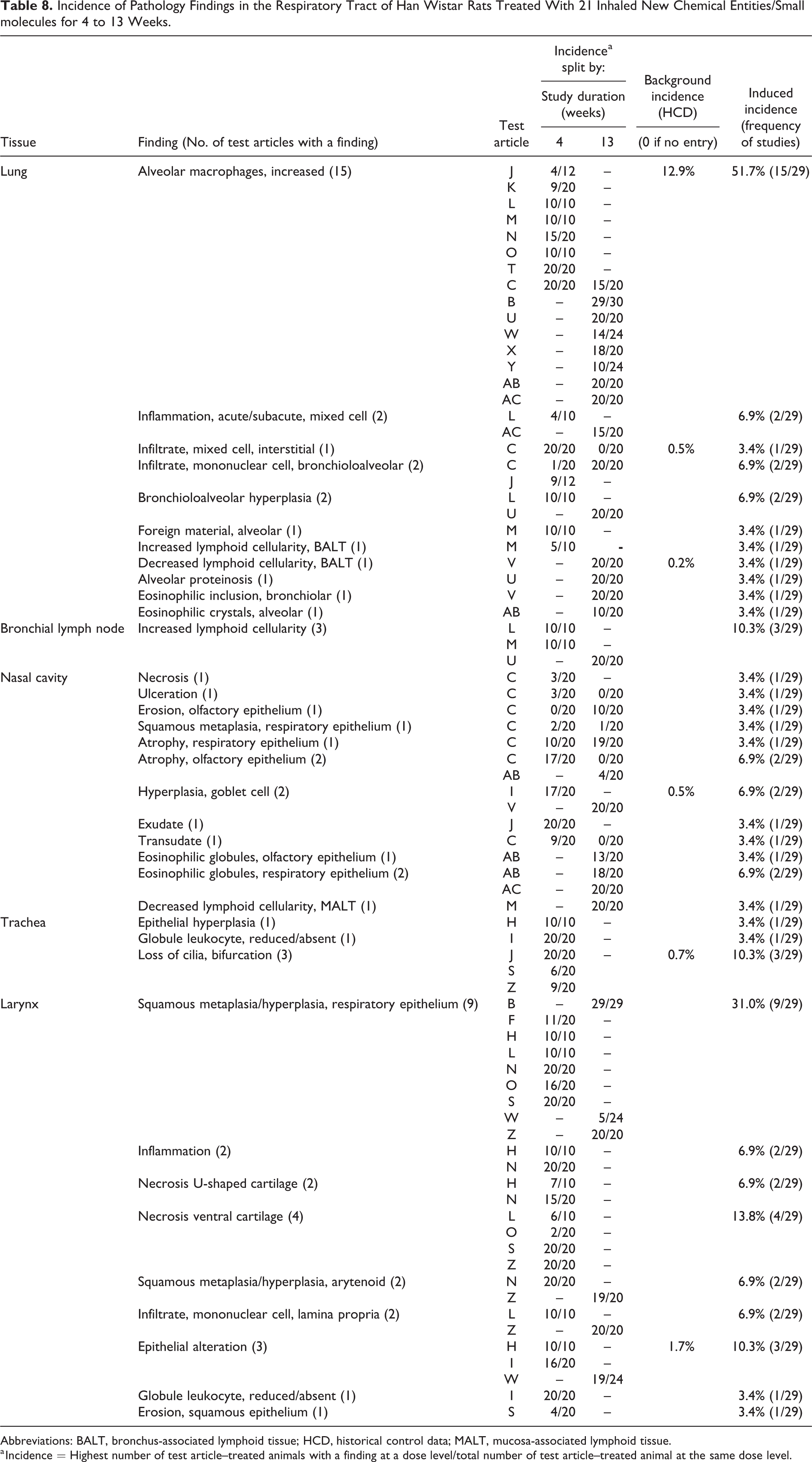
Abbreviations: BALT, bronchus-associated lymphoid tissue; HCD, historical control data; MALT, mucosa-associated lymphoid tissue.
a Incidence = Highest number of test article–treated animals with a finding at a dose level/total number of test article–treated animal at the same dose level.
Incidence of Pathology Findings in the Respiratory Tract of Sprague Dawley Rats Treated With 8 Inhaled New Chemical Entities/Small Molecules for 4 Weeks.
Abbreviation: HCD, historical control data.
a Incidence = Highest number of test article–treated animals with a finding at a dose level/total number of test article–treated animal at the same dose level.
Test article–induced lesion in the trachea, nasal cavity, and larynx
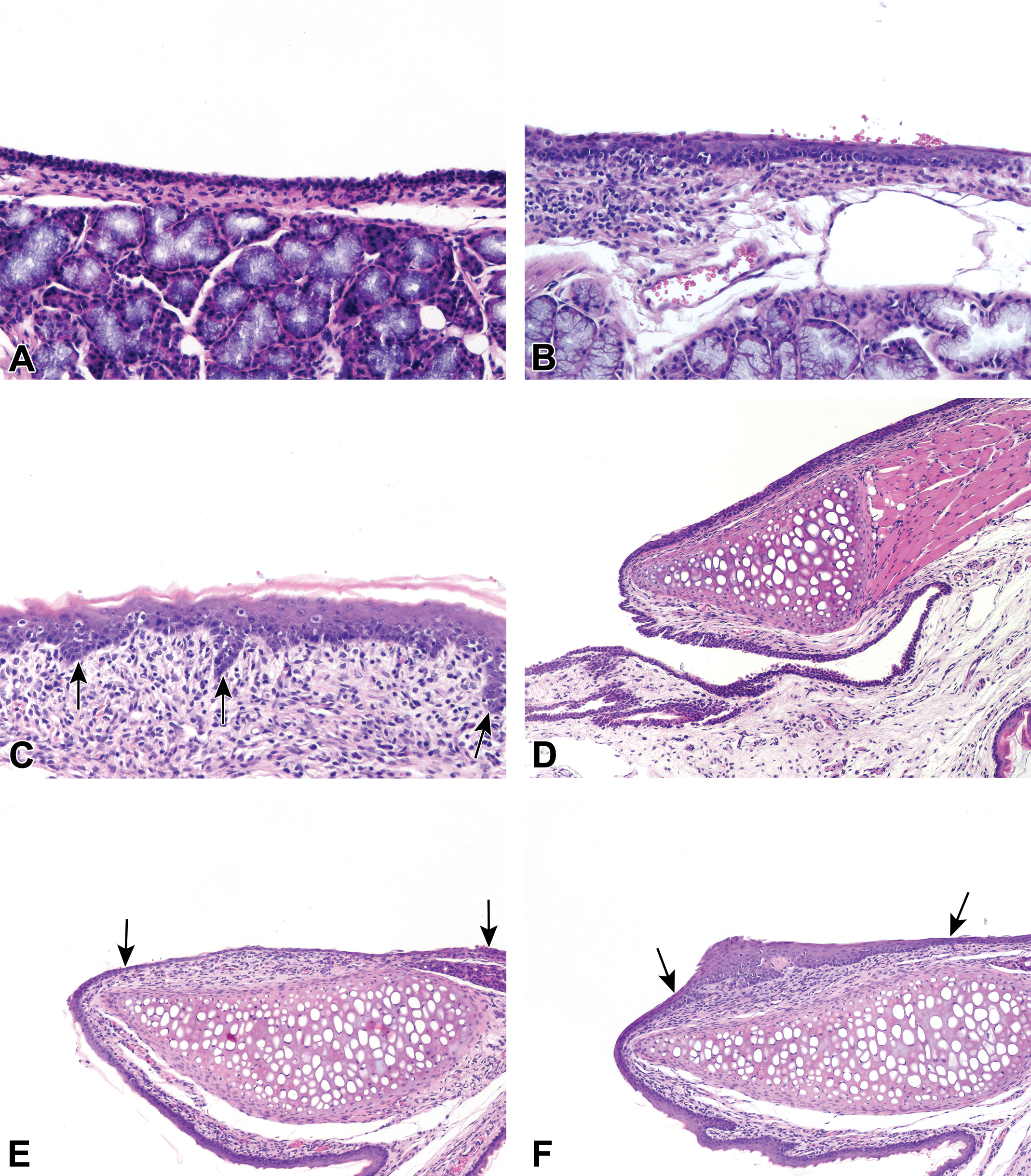
In cynomolgus monkeys, induced tracheal lesions were observed in 30% (3 of 10) of studies representing 3 different test articles (Figures 5 -7). Hyperplasia/squamous metaplasia of the respiratory epithelium was the most common finding and was observed in 2 of 10 studies and was associated with inflammatory cell infiltration including granulocytes or inflammation in the lamina propria. In rats, induced tracheal changes were more commonly observed at the point of tracheal bifurcation (carina) and were recorded in 17.2% (5 of 29) of studies in HW rats (representing 5 test articles). Loss of cilia was the most common finding and was observed in 10.3%, 3 of 29 studies in HW rats. Other changes associated with loss of cilia included epithelial hyperplasia of the respiratory epithelium (3.4%, 1 of 29 studies in HW rats). In SD rats, mixed (granulocytic) inflammatory cell infiltrate in the lamina propria was the only induced finding recorded in the trachea and was observed in 11.1% or 1 of 9 studies.
Induced lesions of the upper respiratory tract (URT) were reported more frequently in rats compared to monkeys. In the larynx, findings were recorded in 44.4% (4 of 9) of the studies in SD rats and 37.9% or 11 of 29 studies in HW rats, compared to only 10% or 1 of 10 studies in cynomolgus monkeys. Epithelial hyperplasia and squamous metaplasia of the respiratory epithelium at the base of the epiglottis were one of the most frequent induced lesions in both the strains (31%, 9 of 29 studies in HW rats and 22.2% or 2 of 9 studies in SD rats; Figure 5C). Concurrent squamous metaplasia of the arytenoids was also observed in HW rats (6.9%, 2 of 29 studies; Figure 5F). These epithelial changes were more commonly observed in association with necrosis of the ventral cartilage in 13.8% of studies in HW rats, necrosis of the U-shaped cartilage (6.9% in HW and 22% in SD rats), or inflammation (6.9%, 2 of 29 studies in HW rats; and 11%, 1 of 9 studies in SD rats). Granulomatous inflammation in the larynx was observed with isolated incidence in cynomolgus monkey (10%, 1 of 10 studies) which was reported with concurrent granulomatous changes in the lungs and trachea.
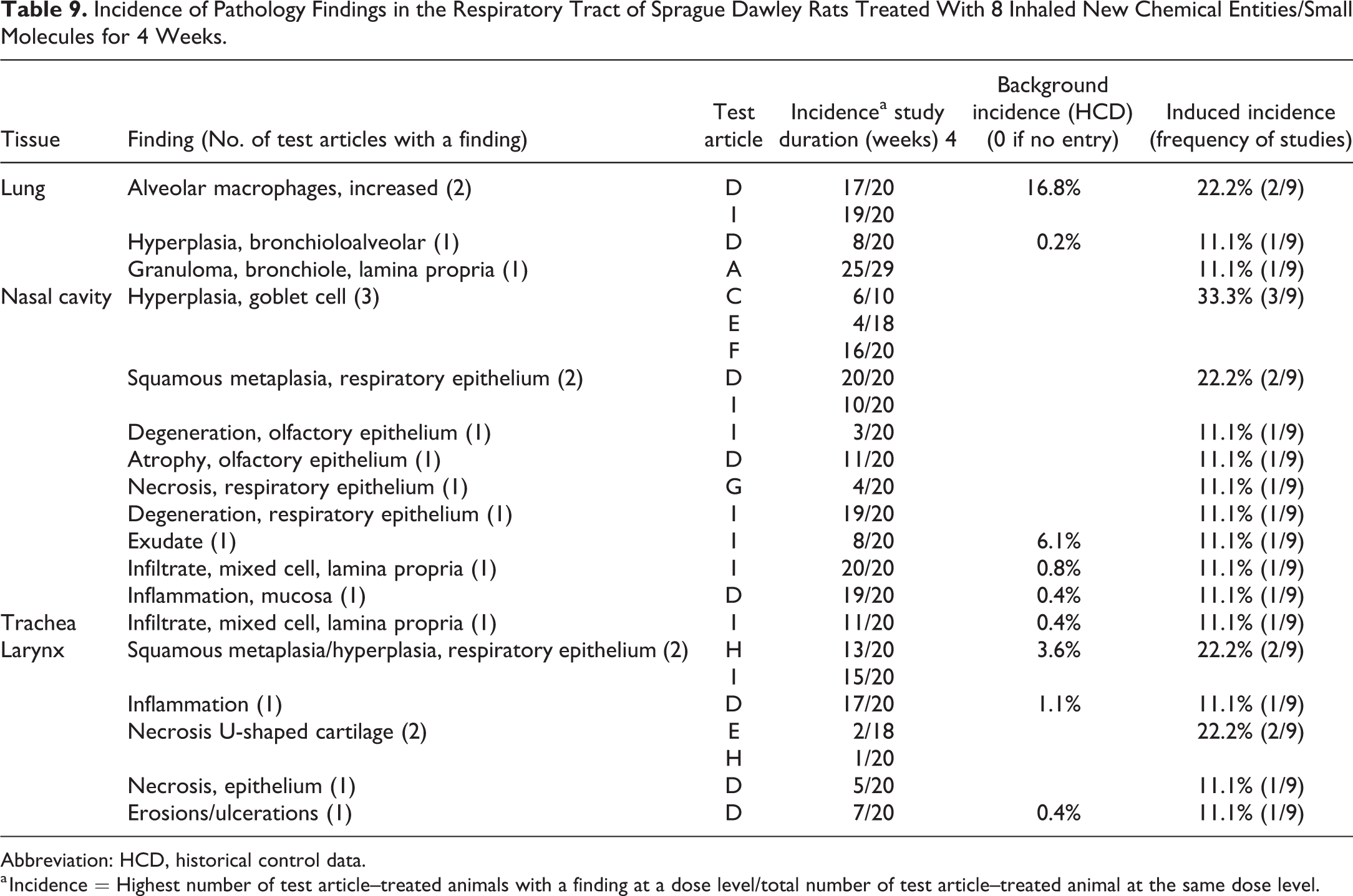
Small molecule induced findings in the larynges of rats. A, The base of the epiglottis with normal epithelium, (B) epithelial alteration and mononuclear cell infiltrate, and (C) squamous metaplasia with rete pegs (arrows) and submucosal inflammation. (D) The medial arytenoid process showing normal epithelium, (E) epithelial atrophy and erosion (between arrows) and submucosal inflammation, and (F) squamous hyperplasia (between arrows).
Induced lesions in the nose were also observed more frequently in rats, particularly in SD rats (66.6%, 6 of 9 studies), when compared to monkeys (30%, 3 of 10 studies). In HW rats, induced nasal lesions were recorded in 34.5% or 10 of 29 studies. Goblet cell hyperplasia of the septum was one of the most common findings in both strains of rats (6.9%, 2 of 29 studies in HW rats and 33%, 3 of 9 studies in SD rats). Other more commonly induced changes in the nasal cavities of rats were atrophy of the olfactory epithelium (6.9%, 2 of 29 studies in HW rats; Figure 7B and D), eosinophilic globules of the respiratory epithelium (6.9%, 2 of 29 studies in HW rats), and squamous metaplasia of the respiratory epithelium (22%, 2 of 9 studies in SD rats). In monkeys, erosions/ulceration of the squamous epithelium lining the midlateral or midseptal regions of the vestibule, or at the transition from squamous to transitional epithelium at level 1, were the most commonly recorded changes and were observed in 20% or 2 of 10 studies (Figure 6).
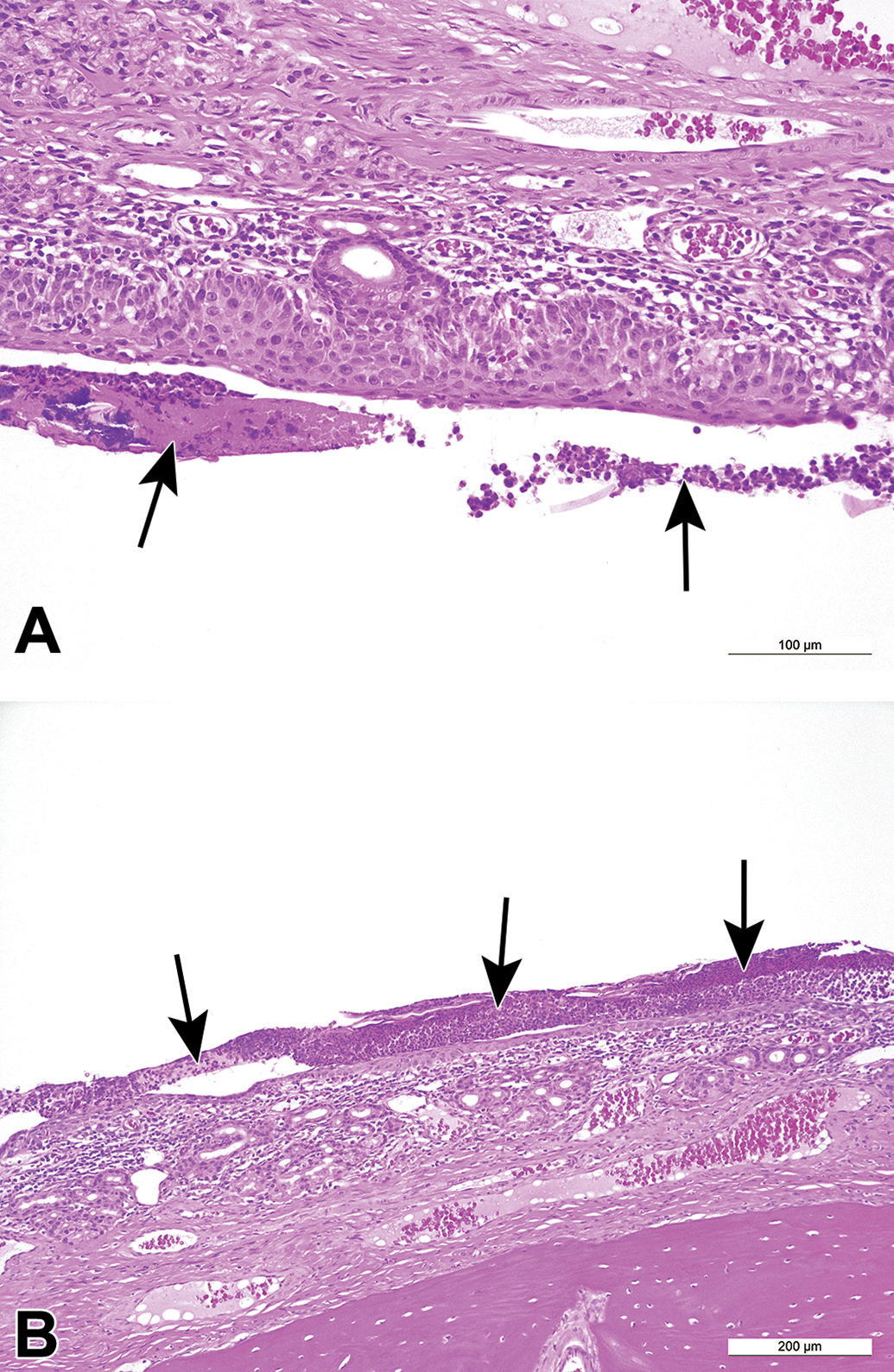
Small molecule-induced findings in the nasal cavity of cynomolgus monkeys. A and B, Inflammation with mononuclear cells, edema, attenuation of the epithelium, and luminal exudate (arrows).
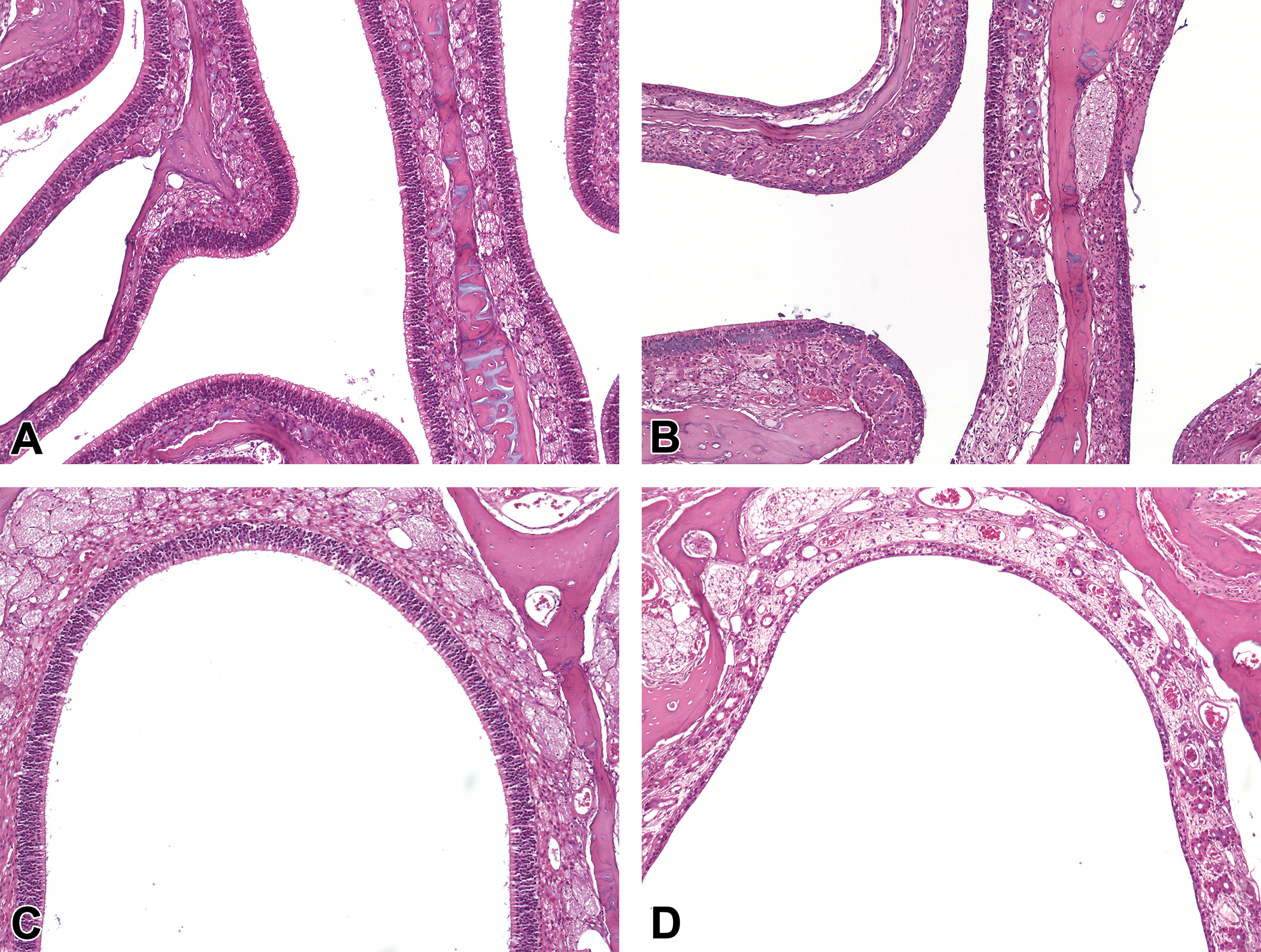
Small molecule-induced findings in the nasal cavity of rats. A, Normal olfactory epithelium on the medial septum and ethmoid turbinates of a control rat and (B) atrophy of the olfactory epithelium at the same location in a small molecule dosed rat. C, Normal olfactory epithelium at the dorsal meatus of a control rat and (D) atrophy of the olfactory epithelium at the same location in a small molecule-dosed rat.
Discussion
The purpose of this manuscript was to compile and generate an extensive database of reference material which documented the nature and incidence of the nonneoplastic lesions in air, vehicle control, and small molecule-treated animals in the respiratory tract of the cynomolgus monkey and rat. It is hoped that this will help identify and interpret test article–induced findings in toxicologic studies and act as a data set comparator for other inhalation modalities. Historical control data from 3 CRO facilities were compiled from inhalation studies and compared with that from non-inhalation dose routes. Secondly, an analysis of the relative frequency of induced findings, following inhalation treatment with 44 small molecule test articles was performed and documented. The relatively large number of animals used was considered likely to increase the chances of identifying trends otherwise not evident within individual studies and minimize confounding factors such as different recording levels between pathologists. However, several potentially confounding factors were still considered inherent and unavoidable and should be taken into consideration when interpreting these results. Foremost is the possibility that pathologists may be more sensitized to recording findings in respiratory tissues when evaluating inhalation studies, due to the anticipation of subtle test article–related effects. When entering data within a study, pathologists are often presented with various options with respect to how complex findings are recorded in data capture systems. Multiple findings may be combined into a single term to facilitate tabulation and analysis, often with an explanation in the report narrative. Alternatively, study pathologists may elect to enter and grade each component separately. Collating data from several studies, as in the present paper, variation in how such findings are entered will influence their incidence as only the terms entered in the study data are transcribed. There may be also some differences in the trimming strategies, number and area of respiratory tissues sampled and evaluated between inhalation and non-inhalation studies, depending on CRO and species. The method of fixation of the lungs should also be considered as a potential confounding factor. In rodents in particular, intratracheal instillation of formalin followed by immersion fixation has been shown to have an impact with respect to the macrophage appearance and distribution in the lungs. 19 Further, relatively high incidences of findings such as foamy alveolar macrophages in the lung, distributed in a bronchioloalveolar (centriacinar) location, may occur in a given study as a result of inadvertent aspiration of the vehicle material in oral gavage studies, or reflux of gavage material leading to inflammation or exudate in the various areas of the respiratory tract. The geographical origin, genetics, age, sex and health status of the animals, other study procedures, and laboratory management practices as well as environmental factors may also affect the incidence of background respiratory findings on a study.
In this study, the tissue with the most commonly recorded findings in air, vehicle-control animals, and in animals exposed to inhaled small molecules of both species, was the lung, and the most common finding was increased alveolar macrophages. Although we could not find a previous study that directly compared the incidences of background findings across all respiratory tissues or compared background findings between monkeys and rats, our results are consistent with those in the published literature. 5,20,21 In both the rat and monkey, background or incidental alveolar macrophages are generally distinguished from treatment-related effects due to their random distribution, minimal or mild severity grades, and a tendency for a subpleural distribution. 5,20,21 Although the severity assessment of the lesions was not documented in our study, this is recognized to be useful in order to differentiate between spontaneous or background findings and test article–related lesions. Compared to spontaneous lesions, lesions induced by exposure to small molecules test articles tend to be multifocal or diffuse, follow a pattern of distribution (eg, centriacinar, PV, and interstitial) and show a dose response in the incidence and severity. 11 Other common incidental findings in rats that may be observed in association with increased alveolar macrophages include focal hemorrhage, hemoglobin crystalline material, other cell types such as neutrophils, pyknotic debris, and type II pneumocyte hyperplasia. 5,22 Focal hemorrhage and crystalline material may be associated with inhalation anesthesia, whereas alveolar macrophages with inflammation or granuloma may increase due to histiocytic inflammation or granulomas secondary to aspiration of feed or bedding materials. 5,22
The incidence, nature, and distribution of the most common lung findings in air and vehicle-control cynomolgus monkeys in our study were also consistent with what has been reported in the literature. 21 Alveolar macrophages, PV or PB inflammatory cell infiltration, bronchial-associated lymphoid tissue (BALT) hyperplasia, focal interstitial inflammation or focal subpleural fibrosis, focal foreign body granuloma, emboli, and lung pigment were some of the more common findings observed in air and vehicle controls.
The incidences of most findings in the lung were higher in inhalation controls (air controls or exposed to nebulized or powdered vehicle) compared with non-inhalation controls (exposed to various oral gavage vehicles) which is consistent with what has been reported by other authors. 5 Although it is possible that these results may reflect recording or sampling bias inherent to inhalation studies and previously discussed, they may also reflect the possibility that some nebulized or powdered vehicle may induce phagocytic clearance by alveolar macrophages. 11 The presence of potential recording or sampling bias inherent to inhalation studies could also explain differences in the incidence of URT findings related to irritation between inhalation and non-inhalation studies.
In the URT and the conducting airways, the most common findings observed in air and vehicle-control animals were in the nasal cavity in the monkey, and the larynx in the rat, although the SD rat also had high incidences of findings in the nasal cavity, consisting of goblet cell metaplasia, olfactory atrophy, and exudate in the nasal cavity. The most common background findings observed in inhalation control monkeys were inflammatory lesions including mononuclear cell infiltrate in the lamina propria, inflammation, and increased lymphoid cellularity. Inflammatory lesions of the nasal cavities have been previously reported as the most common background pathological findings in the cynomolgus monkey. 17 The incidence of background findings in the rat larynx in our study was higher than those reported in the literature, 15,16 which could be a reflection of the increased awareness among pathologists of the significance of the larynx as a target tissue in rat inhalation studies. However, the range of the most common findings, consisting of squamous metaplasia, and mixed inflammatory cell infiltration is consistent with those reported in the literature. 7,14 -16 Our data showed a higher incidence of laryngeal epithelial changes in nebulized and/or powdered vehicle controls compared to air controls suggesting that some vehicles may be responsible for causing a mild local irritant effect in this organ. 7,23
Analysis of test article–related findings from inhalation data with small molecules in both rats and monkeys, revealed increased alveolar macrophages as the most commonly observed pathology change, similar to the control data. The lesion was described as aggregates of foamy macrophages of either diffuse or multifocal distribution, typically located at the bronchioloalveolar (centriacinar) junction and occasionally in the interstitium in monkeys and mainly in the centriacinar region in rats. Since most inhaled small molecule drugs are specifically designed to be of low solubility in order to ensure limited systemic exposure, and thereby reducing undesirable side effects, the majority will cause accumulation in the alveoli. 4 -6 In our study, a higher frequency of pathological changes associated with increased alveolar macrophages was observed in rats compared to monkeys exposed to inhaled small molecules. This is in agreement with several reports that describe rats as being more susceptible to developing alveolar macrophages in response to inhaled test articles than other species such as the dog and monkey. 4,5 Increased alveolar macrophages in this area are most likely a nonspecific response to the deposition of poorly soluble test article and represents a physiological clearance mechanism. 5 Clearance is via the mucociliary escalator or by migration to the pulmonary interstitium and, subsequently, to the local lymph nodes. 4,6,24,25 Accumulation of undissolved material may prompt a local adaptive macrophage response that increases clearance of the material 26 and may lead to macrophage activation and recruitment of other inflammatory cells with secondary tissue damage, regeneration, and possibly fibrosis. 24,27 In some cases, clearance by macrophages may be overwhelmed leading to “lung overload” which occurs when the amount of particle deposited exceeds pulmonary clearance mechanisms. Although monkeys have higher fractional deposition and a slower rate of clearance than rats, 24 lung overload occurs more frequently in rats and is rare in monkeys and humans. 6 Our data also showed that in a few studies in rats, a mixed inflammatory reaction possibly in response to particle load was observed and was characterized by accumulation of neutrophils and other leukocytes in the alveolar space and/or acute to subacute bronchioloalveolar inflammation and bronchioloalveolar or type II pneumocyte hyperplasia. Rats are even more sensitive to lung overload than other rodent species such as mice and hamsters and develop more prominent alveolar epithelial hyperplasia, chronic active inflammation, and focal fibrosis to particles such as those in diesel exhaust or dust, whereas monkeys tend to accumulate macrophages in the interstitium. 28 This can be explained in part by the presence of lymphatics adjacent to the respiratory bronchioles and alveoli in primates, decreasing the amount of luminal particles which initiate the inflammatory response. 29 Our study showed no differences in the incidence of induced inflammatory lung changes, including increased alveolar macrophages, in relation to the duration of exposure, which suggests that the occurrence of induced lung pathology by undissolved material depends on the total quantity (volume) of the material in the lung, which is greatest for poorly soluble particles, rather than the duration of exposure per se. 26
Induced changes in the larynx of animals were observed much more frequently in rats compared to the cynomolgus monkey, which is consistent with reports in the literature. 8,9 Due to differences in anatomy of the upper airways and different airflow characteristics, the rat is considered to be more sensitive to the effects of inhaled irritants than other species, including monkeys and dogs. 7 -9,14,30 Squamous metaplasia or epithelial alteration of the ventral larynx, often associated with squamous hyperplasia of the epithelium overlying the arytenoid cartilages, were the most common findings seen in rats exposed to inhaled small molecules, consistent with other reports. 15,16 The distribution of induced lesions is reported to show a distinct predilection for the areas of transition between stratified squamous epithelium and respiratory epithelium. 8,16,31 Laryngeal squamous metaplasia induced by inhalation exposure was occasionally associated with necrosis of the ventral or U-shaped cartilages and inflammation. It has been reported that the commonly observed induced laryngeal changes depend to some extent upon the duration of exposure and presumably the dose level employed. 8,16 However, our data did not show any differences in the incidence of the laryngeal changes based on duration of exposure between 4- and 13-week studies in rats. Laryngeal lesions were however uncommon in monkeys, either as background findings in inhalation controls or as induced changes in animals exposed to inhaled small molecules, consistent with very few literature reports of inhalation exposure-induced laryngeal lesions in this species. 8 Similarly, induced changes in the trachea/carina are considered infrequent in cynomolgus monkeys, and less common than in rats, 9 although drug-induced changes consistent with hyperplasia/squamous metaplasia may still occur in the trachea/carina in monkeys, as was the case in our study. Consistent with our study, loss of cilia, epithelial hyperplasia, inflammation, and squamous metaplasia are reported as the most commonly reported induced findings in rats. 9 The most commonly observed induced findings in the nasal cavity included, goblet cell hyperplasia of the septum, squamous metaplasia of the respiratory epithelium, and atrophy of the olfactory epithelium, consistent with other reports. 8 They were observed more frequently in the rat, and were uncommon in cynomolgus monkeys, which is consistent with the published literature. Numerous reports describe nasal lesions induced in rodents by inhaled chemicals with no corresponding nasal effects in dogs or monkeys exposed to similar concentrations. Interspecies differences between rodents and nonrodents in the anatomy and therefore the air flow and type and distribution of nasal epithelium have been described in detail 12,17,32 -34 and result in different susceptibility, occurrence, and distribution of lesions following exposure to inhaled nasal toxicants. However, in 2 monkey studies in our review, erosion/ulceration of the squamous epithelium was reported and considered to represent a local response to inhaled irritants.
Conclusions
These data which were compiled from 3 different CROs sites and from a very large number of animals represent the most comprehensive multi-institutional study on the background and induced pathology of the respiratory tract in the cynomolgus monkey and rat. The study objective was to present and document the range and incidences of common induced lesions in the respiratory tract of laboratory animals exposed to inhaled small molecules drugs to act as a data set for comparison with other treatment modalities including inhaled biologics as well as those reported in the published literature. We also aimed to provide updated reference material on the incidences and distribution of common background changes in the respiratory tissues of control cynomolgus monkeys and rats which could be used as an aid when evaluating inhalation studies. Finally, it is hoped that these data will aid in the identification and interpretation of test article–induced findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
