Abstract
This brief communication describes a previously unreported background lesion in the eye of a naive cynomolgus macaque. Inflammation of a posterior ciliary artery was, in this case, morphologically similar to vascular inflammation of other tissues described in naive cynomolgus macaques. However, the available literature does not describe this lesion at this anatomical site. The affected animal did not present with any abnormal clinical signs and ophthalmological examinations were within normal limits. Toxicologic pathologists should be aware of this finding in order to help differentiate it from a test item–related finding.
Cynomolgus macaques are frequently used to assess the safety of novel therapeutics, particularly biotherapeutics for which they may be the only pharmacologically relevant species. In order to accurately determine the test item-relatedness of findings in toxicity studies and their potential risk to patients, a thorough understanding of spontaneous and/or background findings in the test species is required. To date, descriptions of spontaneous or background lesions of the cynomolgus monkey eye are scarce. 1 –4 This communication adds to the previous literature by describing inflammation of a posterior ciliary artery (PCA) and choroidal vessels in a control animal included in a general toxicity study.
The affected animal was an approximately 3-year-old naive female cynomolgus macaque of Chinese origin that was included in a 4-month Good Laboratory Practice general toxicity study. The study was conducted at Covance Laboratories, Madison, in accordance with the IACUC, the USDA Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals. During prestudy screening, the animal was found to be clinically normal and tested negative for tuberculosis, macacine herpesvirus-1 (B-virus), Simian Immunodeficiency Virus, Simian type D Retrovirus, Simian T-cell Leukemia Virus type 1-1, and measles. The animal was administered 5 mL/kg of 40 mg/mL polyvinylpyrrolidone vinyl acetate/sodium dodecyl sulfate (7:1) dissolved in 0.5% (wt/vol) methylcellulose (20 to 30 cps) and 0.2% (wt/vol) polysorbate 80 (Tween 80) in reverse osmosis water (control article) by daily oral gavage for the duration of the study. The animal was group housed in an European Union style enclosure with one other control animal. It exhibited no significant clinical abnormalities for the duration of the study other than minor trauma to a digit during the predose phase that was treated with single subcutaneous injections of meloxicam (0.6 mg/kg) and ceftiofur (20 mg/kg). Hematology, chemistry, and coagulation were assessed in the predose phase and on study days 36, 70, and 108. There were no notable findings except a mild increase in neutrophils compared to baseline and the remainder of the control group on day 108. Ophthalmic examinations using an indirect ophthalmoscope following administration of a mydriatic agent were conducted by a board-certified veterinary ophthalmologist during the predose phase and on study days 36 and 100 and were within normal limits. On study day 113, the animal was euthanized and subjected to necropsy. There were no abnormal macroscopic observations.
Eyes were immersed in modified Davidson’s fixative for 24 hours before being fixed in neutral buffered formalin and processed to paraffin routinely. A single temporo-nasal section of each eye, that included the macula, was sectioned and stained with hematoxylin and eosin (H&E). In the right eye, the tunica media and tunica adventitia of a PCA and to a lesser extent the choroidal arteries temporal to the optic nerve head and subjacent to the macula were infiltrated and surrounded by an aggregates of lymphocytes and plasma cells with fewer histocytes and occasional neutrophils (Figure 1A and B). Endothelial cells were multifocally hypertrophied, exhibiting karyomegaly and an open chromatin pattern (Figure 1B). Mild hypertrophy of the media of a choroidal artery was present and the overlying retina was microscopically normal (Figure 1A and C).
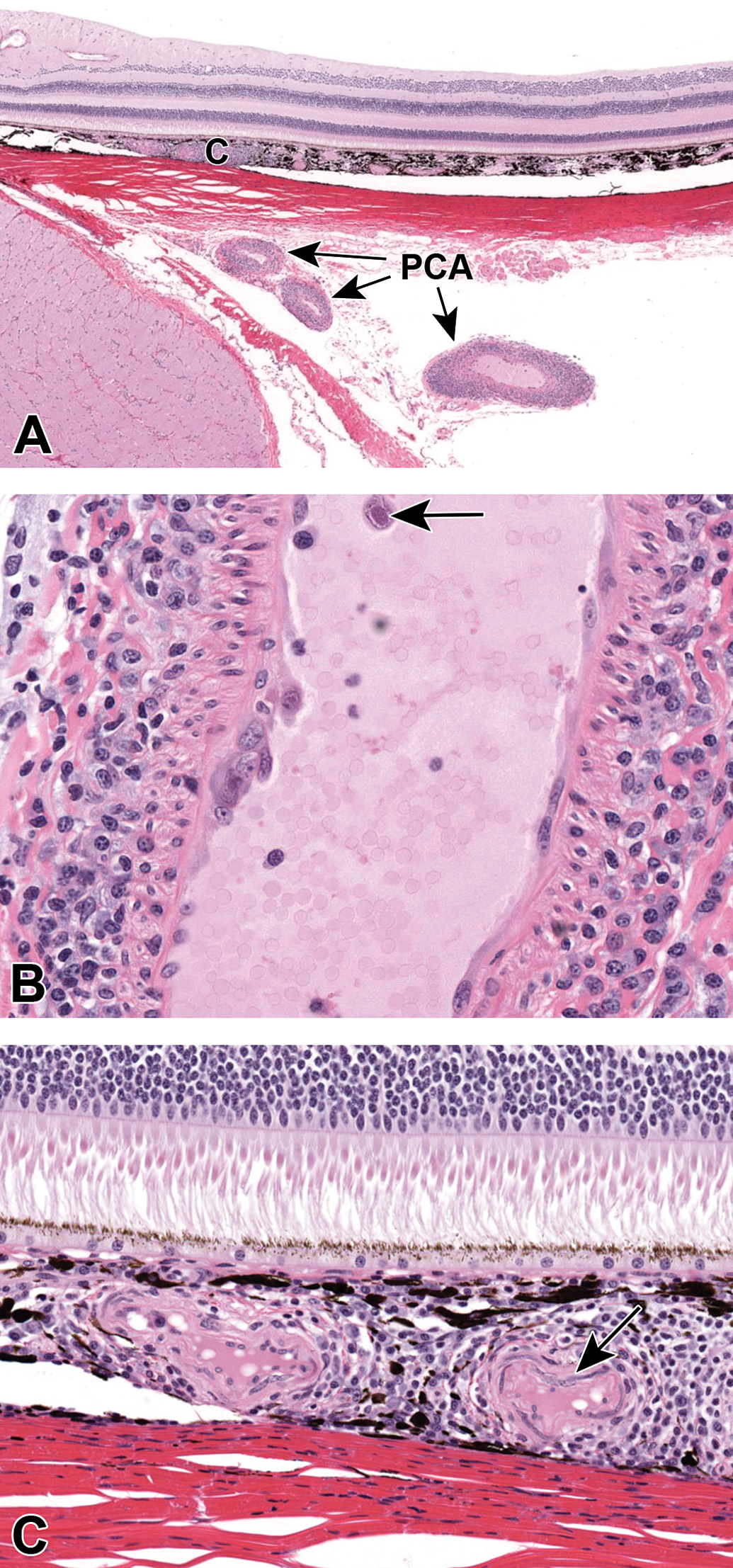
A, Inflammatory cells surround and infiltrate the tunica adventitia and media of a posterior ciliary artery (PCA) and choroidal blood vessels (C) temporal to the optic nerve and subjacent to the macula. B, Endothelial cells of the PCA are multifocally reactive with hypertrophic nuclei with an open chromatin pattern. Inflammatory cell infiltrates are predominantly mononuclear with scattered granulocytes. A single circulating mononuclear cell (arrow) exhibits an enlarged nucleus, with peripheral chromatin and a central basophilic inclusion. C, Mononuclear cells infiltrate the choroid and there is mild hypertrophy of the media of a choroidal vessel (arrow). The overlying retina (photoreceptors and outer nuclear layer shown) are within normal limits.
The anatomy of the PCAs is complex and difficult to ascertain from a single section. However, the distribution of the lesions in this case suggest that the lateral PCA and its short branches, which penetrate the sclera adjacent to the optic nerve head and supply the posterior choroid up to the equator, were primarily affected. 5 A single mononuclear cell in the lumen of the PCA exhibited an enlarged nucleus, with peripheral chromatin and a central basophilic inclusion, which may suggest an active viral infection. Given the lack of clinical signs, transient leukocytosis, and microscopic findings, cytomegalovirus (betaherpesvirus) infection was considered, but was not confirmed. Other findings in the eyes included minimal, bilateral, mononuclear cell infiltrates in the choroid. Microscopic observations in tissues other than the eye were limited to minimal, primarily mononuclear, inflammatory cell infiltrates in the adrenal medulla, large intestine, kidney, and mandibular salivary gland. Vascular inflammation was not identified in any other tissue or in other animals on study.
There are few reports describing background findings in the eyes of cynomolgus monkeys. The primary lesion described is lymphocytic or mononuclear cell infiltrates in the choroid and ciliary body, which may affect up to 75% of animals. 6 Less frequent background findings of the eye include lens degeneration, disorganization of the retina, cystic and degenerative changes in the ciliary epithelium and retina, and atrophy of the ganglion cell layer of the retina. Rare proliferative lesions include glial nodules and melanocytomas. 1,4,7
Spontaneous inflammation of arteries (synonyms: vasculitis/perivasculitis) in the eye of clinically normal, naive cynomolgus macaques has not been reported. However, spontaneous arterial changes including mural or medial degeneration, hemorrhage, inflammation, and perivascular infiltrates have been described in other tissues including kidney, lungs, meninges, heart, and sciatic nerve. 1 –3,8 Vasculitis is also a common finding in other toxicology species including the beagle dog (idiopathic canine polyarteritis) and rat (polyarteritis nodosa). 9 In both syndromes, small- to medium-sized arteries are primarily affected with coronary vessels being most often affected in the beagle; and testicular, pancreatic, and mesenteric vessels most often affected in the rat. There are no reports of vessels of the eye being affected. The mechanism of vascular injury in all of these spontaneous syndromes is incompletely understood, but may be immune-mediated and/or related to disturbances in vascular tone. 9
Giant cell arteritis (GCA), an immune mediated vasculopathy of man that affects medium- and large-sized vessels, frequently affects the PCA. 10 The PCAs supply discrete lobules of the choriocapillaris, the overlying retinal pigment epithelium and outer retina, and the optic nerve head. 5 Classic morphologic features of GCA consist of florid granulomatous inflammation including epithelioid cells and giant cells, which can affect all layers of the vessel wall but tend to predominate in the media. Thrombosis and vascular occlusion are common in GCA and lead to anterior ischemic optic neuropathy and vision loss in up to 20% of affected patients. 10,11 No ocular abnormalities were noted during the in-life assessment of the affected cynomolgus macaque. This is likely a consequence of minimal changes to the choriocapillaris, lack of evidence of thrombosis, and the subsequently unaffected overlying retina. Any changes in visual acuity were likely too subtle to have been noted using indirect ophthalmoscopy, which is best suited to assess changes in the retina and superficial retinal vessels. Indeed, even animals with histopathologic evidence of marked ganglion cell atrophy do not have notable abnormalities of vision in the controlled environment of a test-site enclosure. 7
Test item-induced vascular injury is a concerning toxicity that is difficult to monitor and must be differentiated from background lesions in toxicity species. Direct drug-induced vascular injury, such as is seen with vasoactive drugs (eg, phosphodiesterase III inhibitors or ergot 12 compounds), typically results in medial necrosis prior to induction of inflammatory infiltrates. 9,13 Thus, multifocal evidence of vascular necrosis in addition to evidence of a dose response will aid in determination of drug-relatedness. However, immune-mediated injury such as may be seen following dosing with immune modulatory therapeutics or following an antidrug antibody response may be morphologically similar to background lesions (typically predominantly inflammatory in nature), affect single or multiple small- to medium-sized vessels, and may not present with a clear dose response. 14 In the eye, this is particularly concerning for biotherapeutics that are administered intravitreally where robust immune-mediated inflammation is commonly encountered. 15 A weight of evidence approach must be used to determine the treatment-relatedness of such findings and requires a thorough understanding of spontaneous background lesions in addition to ancillary assays such as circulating antidrug antibody titers. 14
In conclusion, this communication describes a previously unreported ocular background lesion in the cynomolgus macaque. Inflammation of the PCA was not accompanied by correlative in-life or macroscopic findings and likely is a manifestation of the same spontaneous vascular inflammation that has been reported in other macaque tissues. Toxicologic pathologists should be aware of this entity in order to differentiate it from drug-induced retinal or vascular toxicity.
Footnotes
Acknowledgments
Covance Laboratories Inc., Madison, are thanked for their diligent conduct of the toxicology study described.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
