Abstract
Long-acting delivery platforms for intravitreal therapies are an active area of research in ophthalmic drug development. The aim of these platforms is to decrease the burden of intravitreal therapies for patients, by increasing the period between intravitreal injections. This brief communication describes the in-life, histologic and immunohistochemical findings associated with repeat-dose intravitreal administration of poly D, L sustained lactide-co-glycolide polymeric rods, an intravitreal depot, in the cynomolgus monkey (Macaca fascicularis). These nonclinical investigations illustrate a pattern of foreign body reaction around intravitreal depots at the temporal pars plana and demonstrated the histopathologic and immunohistologic features of retinal degeneration and epiretinal membrane formation in the inferior retina.
Introduction
Retinal diseases, such as age-related macular degeneration, diabetic retinopathy, and diabetic macular edema, are increasingly prevalent causes of vision loss in developed countries. 1 Largely, treatments (either approved or in development) for these diseases are administered directly into the eye via intravitreal (ITV) injection. Although effective, for optimal efficacy many of the treatments require monthly or bimonthly injection, and this route of administration therefore places a significant burden on patients and caregivers. 2 -5 As such, in real-world scenarios, therapeutics are often administered less frequently than this, adversely impacting patient outcomes. 6
A number of strategies have been explored to help overcome the disconnect between patient outcomes obtained in clinical trials and those obtained with general clinical usage. Long-acting intravitreal delivery (LAD) systems have been explored for a number of years as a method of increasing the half-life of the drug in the eye, to provide therapeutic drug concentrations in the vitreous for an extended period, thereby increasing the dosing interval for patients, decreasing patient and caregiver burden, and provide optimal patient outcomes.
Poly D, L sustained lactide-co-glycolide (PLGA) polymer matrix rods were considered an attractive LAD option as, in addition to sustained delivery of the drug over a prolonged period, the Novadur (Allegan) solid polymer drug delivery system utilized by the long-acting dexamethasone formulation, Osurdex (Allergan) is an ITV PLGA rod formulation that is already successfully utilized clinically.
The data presented in this brief communication result from a nonclinical investigative study conducted in cynomolgus monkeys to explore the tolerability of 0.5 × 5 mm PLGA polymer matrix rods as a slow-release depot for LAD.
Materials and Methods
The study was conducted in a laboratory fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. All study procedures were compliant with applicable animal welfare acts and were approved by the local Institutional Animal Care and Use Committee.
Six cynomolgus macaques (Macaca fascicularis) of Chinese origin were utilized for the study. On day 1, animals were 35 months to 50 months old, and had body weights that ranged from 2.4 to 2.9 kg for males and 2.7 to 3.1 kg for females. Animals were socially housed (up to 3 per pen) in European Union–compliant housing (ETS 123), and supplied with species-appropriate environmental and dietary enrichment.
Animal were dosed on day 1 and day 79. Each dose comprised administration of two 0.5 × 5 mm sustained-delivery poly(lactic–glycolic acid) 88:12 (PLGA) rods (Figure 1), which were administered via an ITV cannula. Animals were dosed bilaterally. Animals were anesthetized (chemically restrained) with ketamine and dexmedetomidine, and the eyes were cleaned with a 1% povidone iodine solution and rinsed with sterile saline. An approximate 2.5% povidone-iodine solution was used at the dose site prior to injection. A 23G valved cannula (Alcon) was inserted into the eye approximately 4 mm posterior to the limbus in the superior temporal quadrant. The trocar was removed from the cannula, and the rod was inserted into the cannula valve. After the insertion of the second rod, a 32G needle was inserted to push the rod through the cannula into the vitreous. The cannula was then removed from the eye and the insertion site was closed with cauterization. Immediately post-dose, indirect ophthalmoscopy was performed to confirm and record the rod locations.
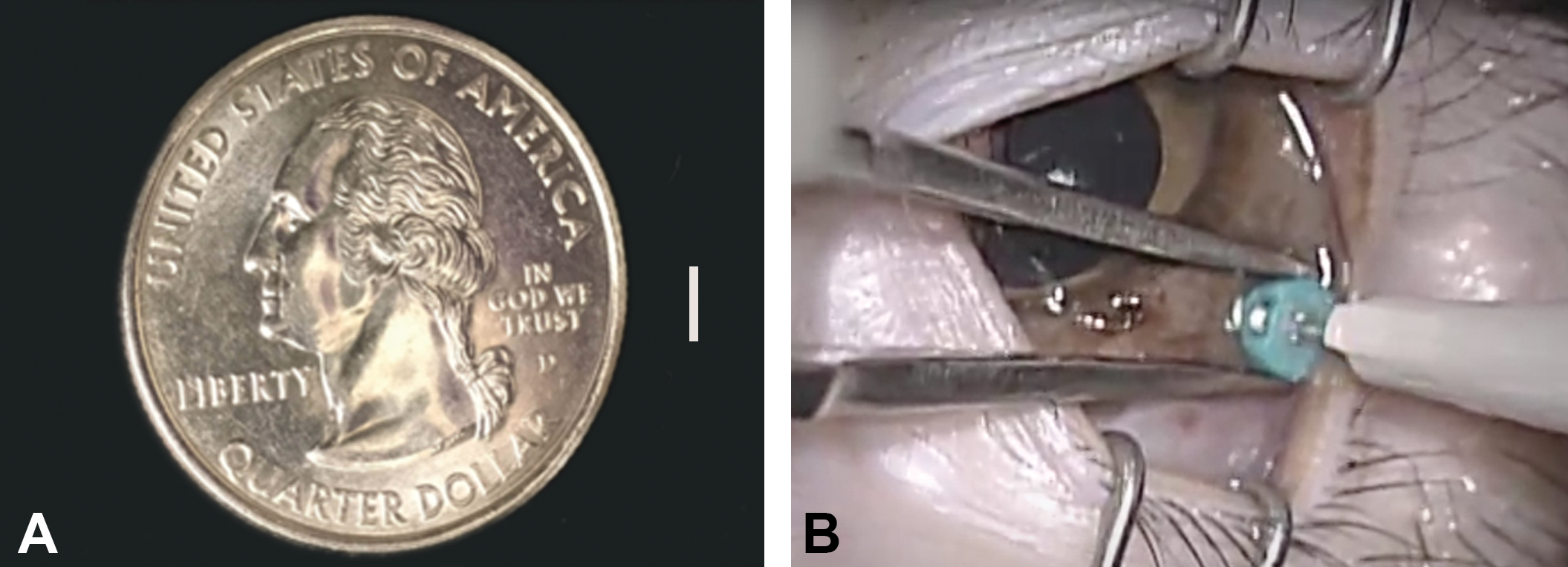
Poly D, L sustained lactide-co-glycolide (PLGA) rods were 0.5 × 5 mm in size and comprised of an 88:12 ratio of PLGA (A). Rods were inserted into the vitreous via a valved cannula (B).
At the time of surgical implantation of the PLGA rods, systemic flunixin meglumine (2 mg/kg) and buprenorphine (0.05 mg/kg) to provide analgesia, and topical Neo-Poly-Dex ointment were administered prophylactically. If significant inflammation was observed on ophthalmic examination (OE), or animals were considered to be demonstrating signs of discomfort, these medications, in addition to topical 1% atropine were administered as required.
Comprehensive OEs were conducted by a board-certified veterinary ophthalmologist and were performed periodically throughout the study. Findings were graded using the Modified McDonald-Shadduck scoring system, and the location of the PLGA rods within was recorded. 7,8
At the end of the study (day 131), all animals were euthanized and necropsied. The eyes and optic nerves were collected for histologic evaluation. Eyes were fixed in modified Davidson’s fixative for 48 to 96 hours and subsequently stored in 10% neutral-buffered formalin. Optic nerves were fixed in 10% neutral-buffered formalin. Eyes were trimmed to produce a horizontal section including the optic disc and macula. The remaining inferior calotte was divided into 3 parasagittal sections—temporal, central, and nasal (embedded temporal-side down). The superior calotte was trimmed and embedded to capture the implantation site. Tissues were processed and embedded in paraffin. Four micron sections were cut and stained with hematoxylin and eosin (HE), Masson’s Trichrome (MTC), and Periodic acid-Schiff (PAS) using standard methods. Immunohistochemical labeling of glial fibrillary acidic protein (GFAP Dako Z0334; 1:1000) was performed on a Ventana Discovery XT automated platform.
Results
In-Life Findings
In-life findings are summarized in Figure 2. Intravitreal insertion of PLGA rods on study day 1 was associated with a short-lived anterior segment inflammatory response that was, to a large extent, likely attributable to the surgical procedure itself and the physical disruption of the eye and resolved by day 19. Intravitreal insertion of a second set of rods on day 79 was associated with a more severe and prolonged anterior and posterior segment inflammatory response that did not resolve by the conclusion of the study (day 131) and frequently required veterinary intervention (anti-inflammatory therapy and analgesia).
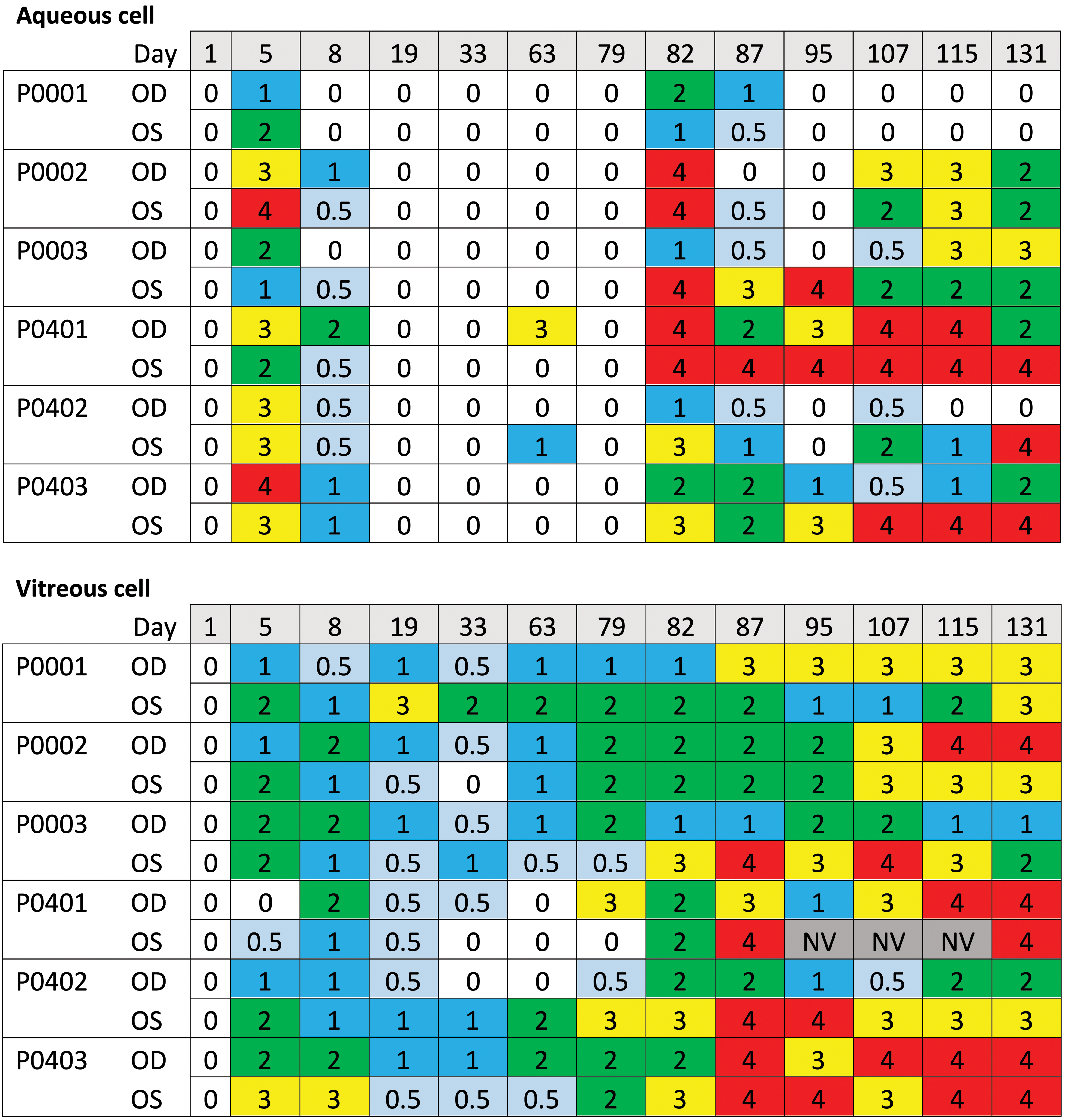
Heat maps summarizing relevant in-life ophthalmic examination (OE) findings (Modified McDonald-Shadduck scoring system). Grades: 0 = no finding; 0.5 = trace; 1 = minimal; 2 = mild; 3 = moderate; 4 = severe; NV = not visualized. OE’s were conducted by a board-certified veterinary ophthalmologist (Diplomate of the American College of Veterinary Ophthalmologists (DACVO)). Animals were administered two poly D, L sustained lactide-co-glycolide (PLGA) rods, bilaterally on day 1 and day 79. Sustained, moderate to severe ocular inflammation manifest here as increased aqueous and vitreous cell scores was observed following the second insertion procedure on day 79.
The PLGA rods were identified by indirect ophthalmoscopy during OE after insertion, and their location was recorded at each subsequent OE throughout the study. Rods were observed to be variably located in the vitreous. Frequently, not all rods were visible or their location had changed between examinations. In animals with more significant inflammation, the rods were difficult to visualize.
Histologic Findings
Numerous inflammatory, degenerative, and/or proliferative findings were observed, attributed to the presence of the PLGA rods. Changes predominantly affected the posterior segment of the eye, although large amounts of proteinaceous fluid, with occasional cells and inflammatory debris was also commonly observed in the anterior chamber. Insertion tracts, identifiable as foci of haphazardly organized collagen fibers admixed with small numbers of mononuclear inflammatory cells, which resulted from the placement of the trocar, were identified in the sclera of sections from the superior temporal quadrant.
Remnants of PLGA rods were visible histologically in the vitreous in the region of, or adherent to, the pars plana in all eyes examined. The rods were most commonly observed in the central, horizontal section through the globe (at the 3 or 9 o’clock positions) at the level of the pars plana, although they were not necessarily temporal (close to the insertion site)—in some instances they were observed nasally). The rods were clear to amphophilic to refractile and often outlined by foci of granulomatous inflammation or organized granulomas (Figure 3A and B).
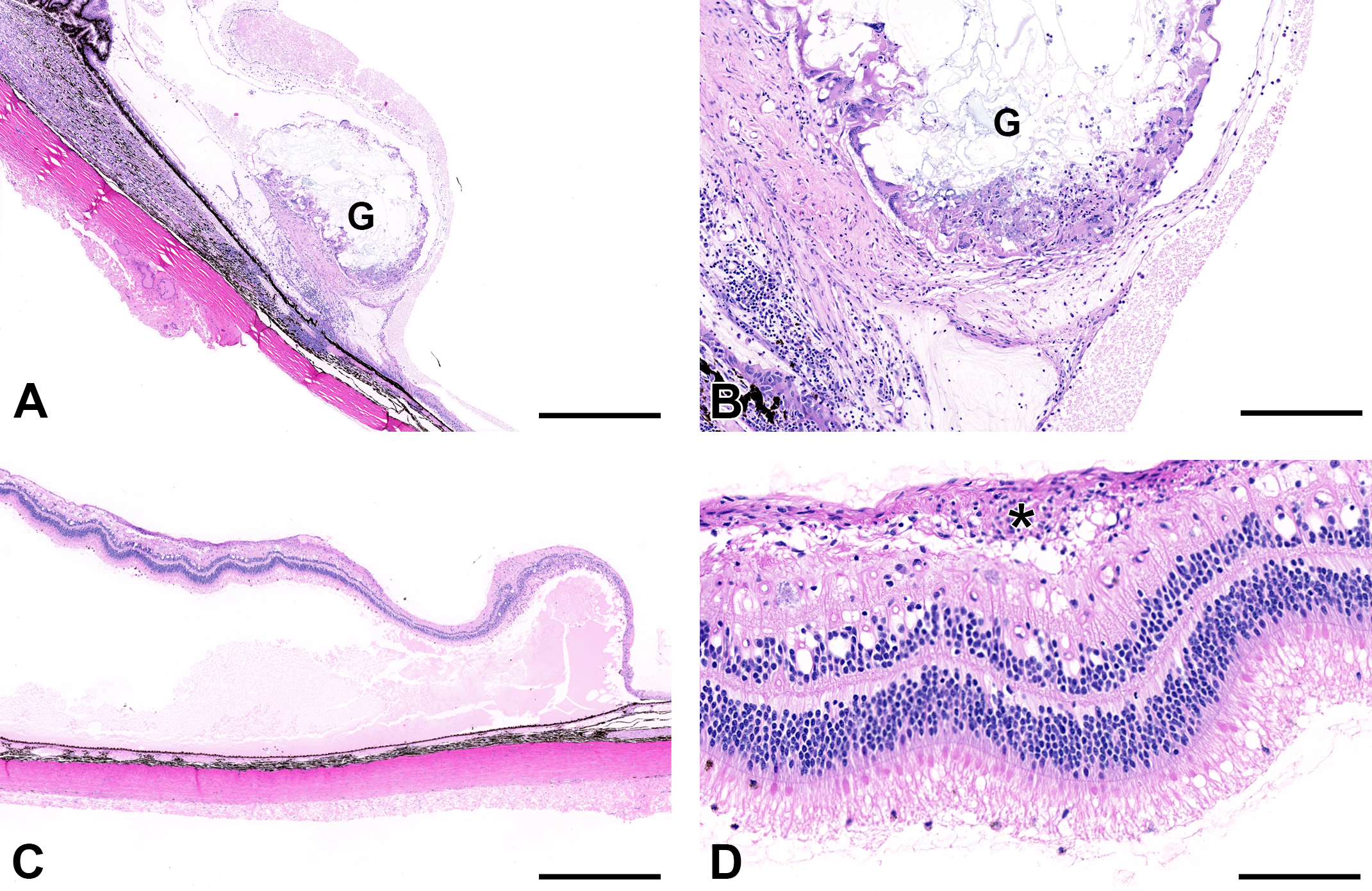
A granuloma (G), centered around degrading poly D, L sustained lactide-co-glycolide (PLGA) rods, attached to the pars plana (A, B). Large numbers of multinucleate giant cells surround the test article, delineated by fibrous connective tissue (B). A focally extensive area of retinal detachment in the inferior retina, with accumulation of subretinal fluid, retinal degeneration, and epiretinal membrane (*) formation (C, D). Original magnifications: (A, C) ×2.5 (scale bar—800 μm); (B) ×5 (scale bar—400 μm); (D) ×20 (scale bar—100 μm).
The foreign body reaction associated with the rods varied in severity and was observed in all eyes which had exhibited moderate to severe ocular inflammation in life (Figure 2). At the milder end of the spectrum, rods were surrounded by aggregates of moderate numbers of epithelioid macrophages, lymphocytes, and fine strands of fibrous connective tissue. At the severe end of the spectrum, there were granulomas in which the remnants of the rods were central, admixed with small to moderate amounts of fibrin and karyorrhectic debris, delineated by large numbers of epithelioid and multinucleate giant cells and lesser numbers of lymphocytes and encapsulated by fibrous connective tissue.
In all eyes with foreign body reactions, a layer of cells and cell processes formed a variably thick epiretinal membrane (ERM), superficial to the inner limiting membrane (ILM; Figure 3C and D; Figure 4). Epiretinal membranes were more commonly noted in the peripheral portion of the inferior sections of the eye and were often overlying areas of retinal detachment and significant retinal degeneration (Figure 3C and D). In minimal cases, ERMs were focal and less than 10 µm thick, whereas in more severe cases they were up to approximately 150 µm thick and overlying the majority of the inferior retina. Degenerative changes were observed in multiple retinal layers and were characterized by vacuolation, which was particularly prominent in the ganglion cell layer, inner nuclear layer, and photoreceptor layer; disorganization of the inner and outer nuclear layers; peripheral displacement of photoreceptor nuclei from the outer nuclear layer into the photoreceptor layer; and infiltration of small numbers of mononuclear inflammatory cells into the inner retina.
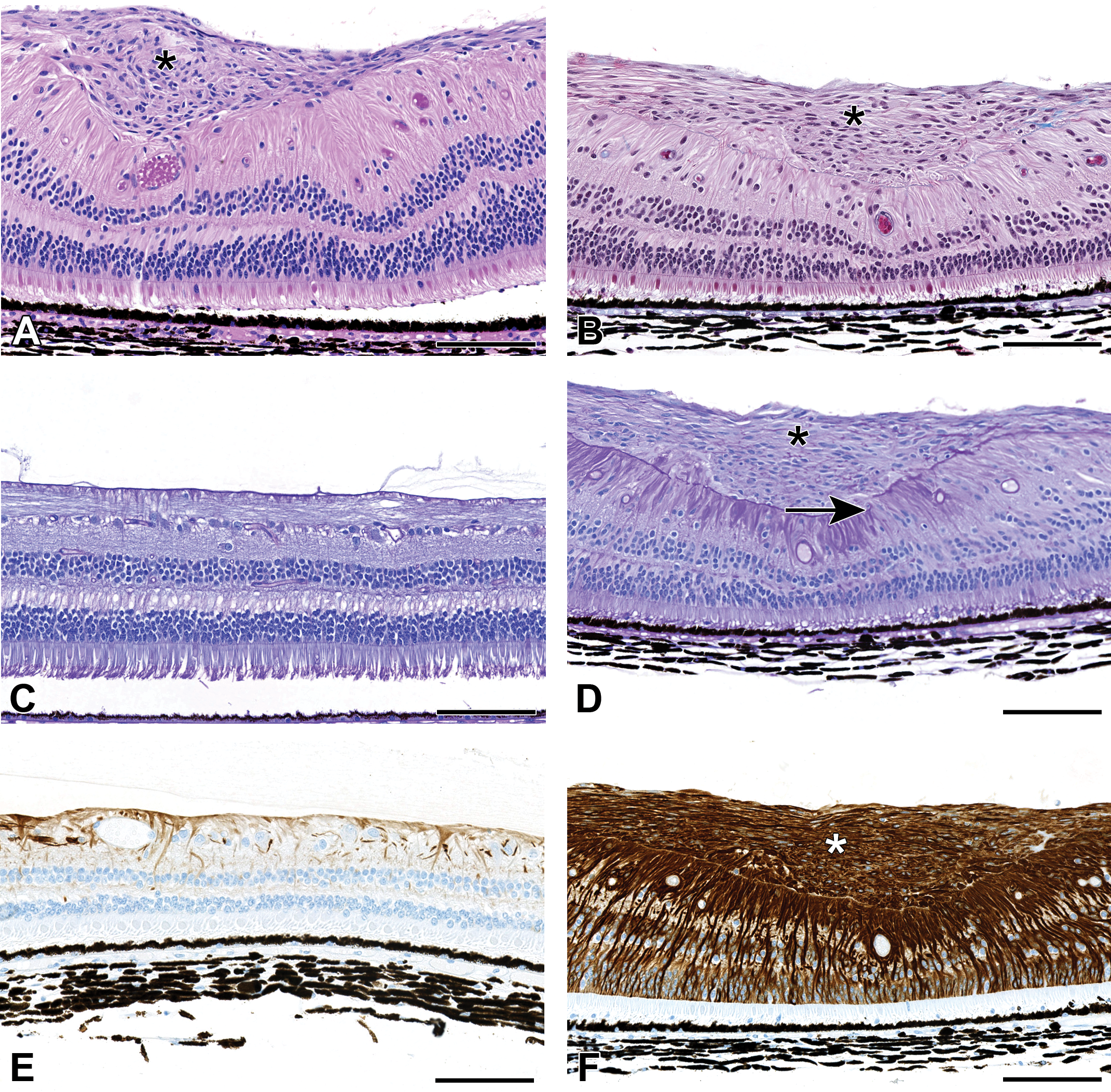
Epiretinal membranes (ERM) were often observed in the inferior retina and were characterized by a variably thick layer of spindle cells overlying the inner limiting membrane (ILM) (*) (A, hematoxylin and eosin [HE]). Labeling with a Masson’s Trichrome stain (B) revealed only very minimal collagen (blue fibrillar material) in the ERM (right side of figure near ILM). Periodic acid-Schiff (PAS) staining revealed focal defects in the ILM (arrow), with increased staining adjacent to the defects (D) when compared with the control (C). The ERMs were intensely immunopositive for glial fibrillary acidic protein (GFAP), suggesting glial origin (F), and there was also a diffuse increase in labeling of the underlying retina when compared with controls (E). Original magnification ×20 (scale bar 100 μm).
Masson’s trichrome and PAS histochemical stains and immunolabeling with an antibody to GFAP were conducted on eyes with ERMs, to further characterize their cellular origin. Masson’s trichrome revealed that the ERMs lacked collagen; however, they were intensely immunopositive for GFAP, suggesting they were of glial origin (Figure 4F). There were foci of disruption of the subjacent PAS-positive ILM (Figure 4D); with extension of GFAP-positive cells (Müller glia) through the defects (Figure 4F). Adjacent to areas of ILM disruption, there were multifocal to coalescing, discontinuous regions of increased PAS labeling, suggestive of abortive attempts at repair of the ILM (Figure 4D). Bacterial or fungal organisms were not identified in HE or PAS-stained sections. Collagen staining present in MTC stained tissues was characterized by thin wisps of light blue stained acellular material within the vitreous (interpreted as collagenous fibers) and/or surrounding or embedded within foci of inflammation and/or granulomas (not pictured).
Minimal axonal degeneration, characterized by the presence of dilated myelin sheaths, with or without minimal inflammatory cell infiltrates, was noted in the optic nerves of 6 of the eyes examined (not pictured). The findings in the optic nerve were considered secondary to inflammatory and degenerative changes in the retina.
Discussion
This brief communication documents a range of adverse ocular findings in cynomolgus monkeys, which included severe and sustained ocular inflammation, with foreign body reaction (including the formation of ITV granulomas) retinal degeneration, and ERMs, occurring following administration of 2 doses of PLGA rods into the vitreous.
Foreign body reactions that varied from granulomatous inflammation to discrete granulomas were observed, which were centered around the degrading PLGA rods. Ocular inflammation including localized foreign body reactions are an established nonclinical safety liability for ITV depot formulations and have been identified previously following ITV administration of some LAD platforms. 9,10 Although 20 to 100 µm PLGA microspheres and 62 to 66 µm PolyActive hydrogel copolymer microparticles have previously been shown to elicit foreign body responses in the eye following ITV injection, 9,10 ITV PLGA rods have been observed to be well tolerated. 9 This is consistent with the nonclinical data supporting the use and registration of Ozurdex, in which placebo PLGA rods were not associated with significant inflammation in the rabbit or the cynomolgus monkey. 11 Further, a single ITV dose of 2 PLGA rods of a similar composition to those administered in the current study by our group was well tolerated in both the cynomolgus monkey and the rabbit (unpublished data).
In the current study, in which PLGA rods were administered by ITV cannula on days 1 and 79, there appeared to be 2 distinct groups of findings—foreign body reactions (granulomatous inflammation and granulomas) which were focused around the degrading PLGA rods, and focally extensive retinal degeneration and ERM formation in the inferior retina.
Foreign body reactions are a chronic response of inflammation and wound healing, which occur subsequent to the surgical or iatrogenic implantation of foreign material. 12 Similar to the reactions described by Thackaberry et al 9 and Adamson et al, 10 the foreign body reactions (granulomatous inflammation and granulomas) observed in this study were present in the vitreous, adjacent (and in some instances adherent to) the pars plana. Although the inflammatory response was similar to those identified with microparticles in previous publications, the larger sizer of the PLGA rods seemed to prevent the anterograde movement into the anterior chamber, and ensured their retention in the vitreous. Granuloma formation has not previously been reported in association with PLGA rods and was not observed with a single dose (2 rods) of this formulation in the cynomolgus monkey or the rabbit (Booler et al, unpublished data, 2017).
The severity of the reaction in this study was considered likely to result from a combination of significant inflammation secondary to the surgical (trocar/cannula) insertion procedure, alongside the provision of a nidus for inflammation by the PLGA rods. The marketed ITV PLGA rod, Osurdex (Allergan), is inserted using a specifically designed applicator, much reducing the manipulation of the globe and the surgical trauma associated with the insertion procedure. The character of the inflammatory response, and the absence of evidence of bacterial or fungal organisms, suggested the response was sterile. The prophylactic, postoperative, anti-inflammatory treatment protocol used in this study was consistent with that followed for standard ITV injection, and it was considered, in retrospect, that this was not substantial enough for the degree of invasiveness/trauma associated with insertion of the rods via a trocar procedure. A more prolonged corticosteroid treatment protocol may have damped down postoperative inflammation. Suppression of the inflammatory response after insertion of the rods by steroids may also explain the difference in tolerability of the marketed dexamethasone-loaded PLGA rod, Osurdex (Allergan), for which this finding has not been reported.
In addition to the foreign body reaction, ERM formation, retinal detachment, and significant retinal degeneration were observed. These lesions were focally extensive, located in the inferior portion of the globe, and were not identified in-life. Interestingly, granulomas/granulomatous inflammation (and the associated PLGA rods) and areas of ERM formation were distinct, with the former present at the level of the pars plana in the peripheral retina, predominately visible on the horizontal section of the eye (in the 3 or 9 o’clock position), and the latter present in the inferior retina. Degenerative retinal changes and ERMs have not previously been reported with PLGA depots (microspheres or rods).
Epiretinal membrane is a clinical diagnosis and encompasses a number of diseases processes which culminate in the formation of sheets of extracellular matrix components and cells at the vitreoretinal interface. 13,14 In patients, they may be idiopathic (in an eye with no history of ocular disease) or secondary to ocular inflammation, retinal vascular disease, or retinal detachments. 13 ERMs appear to arise as the result of cells within the retina proliferating and migrating along the ILM. 14 Four distinguishable cell types have been identified in pathologic specimens of ERMs in humans—retinal pigment epithelial cells, macrophages, fibroblasts, and glial cells; although many cells may change their phenotype as the membrane develops. 15,16 The ERMs observed in this study were comprised of GFAP-positive cells and cell processes, characteristic of glial origin (most likely processes of Müller glia). Consistent with this, cell processes could be observed extending through defects in the ILM and extending along the vitreal aspect of the ILM. It cannot be determined whether disruption of the ILM occurred secondary to physical trauma (the number, location, or size/shape of PLGA rods within the eye), or as a result of inflammation within the globe.
The retina beneath the ERM was often detached and degenerated. Retinal detachment in this region is likely to have occurred as a result of traction of the ERM on the retina beneath it, producing a physical separation of the neurosensory retina from the underlying retinal pigment epithelium.
Despite the retinal detachments and overlying ERMs being extensive in some eyes, they were not identified in-life. The lack of an in-life, OE correlate most likely resulted from the degraded view of the posterior segment (secondary to moderate to marked ocular inflammation), and the location of these lesions in the inferior, peripheral retina—a region difficult to visualize with indirect ophthalmoscopy.
In the current study, given the slow dynamics of degradation of the PLGA formulation used, at the time of onset of significant ocular inflammation, 4 PLGA rods were present in each eye. It could not be determined whether the degeneration of the inferior retina and ERM in the eyes examined was associated with the increase in surface area provided by this number of rods (a potential mechanism discussed by Thackaberry et al 9 ), or simply a function of the number of objects in the globe increasing propensity for physical trauma.
A fundamental aspect of nonclinical drug development is to utilize the most relevant species and a clinically relevant route of administration to characterize potential liabilities and provide a risk assessment to patients—including, for relevant and potentially translatable adverse findings, providing a time course, an indication of clinical monitorability and potential for reversibility. The findings from this study indicated significant further work is required to enable this platform for repeat ITV dosing.
In conclusion, we observed a number of significant and potentially translatable adverse findings, comprising severe ocular inflammation, foreign body reactions, retinal degeneration, and ERM formation, which were associated with the repeat administration of 2 × PLGA rods by ITV cannula to cynomolgus monkeys. The precise mechanisms could not be established, but the potential role of postoperative inflammation (and different implantation technique), increased implant surface area, and physical trauma by the implants to the inferior retina, were discussed as hypotheses for the difference in tolerability between the ITV PLGA rods used in this study, and marketed ITV PLGA rods.
Footnotes
Acknowledgments
The authors thank Peter Sonnentag (Covance, Inc), Susan Lynk (Covance, Inc), Paul Miller (OSOD), Karthik Rajagopal (Genentech), Matthew Holdren (Genentech), Rami Hannoush (Genentech), and Mike Elliot (Genentech).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
