Abstract
Squamous metaplasia is a nonspecific adaptive response to chronic irritation in the larynx and is often diagnosed as a test item-related change in rat inhalation studies. Investigating scientists are frequently asked to assess the adversity of laryngeal squamous metaplasia and to interpret its relevance to human risk. One factor in predicting relevance to human risk is the kinetics (degree and speed) of recovery following the cessation of exposure to the test item. Most reports describing recovery from squamous metaplasia in the rat larynx discuss the more severe end of the spectrum of metaplastic change (moderate to severe) and include relatively long (6 weeks or more) recovery periods. We conducted 2 studies to evaluate the toxicity and recovery from any potential effects of 4-(Chloro-2-methylphenoxy) butyric (MCPB) acid, a herbicide, when administered by inhalation to young male Sprague Dawley rats for 3 to 4 weeks. The studies resulted in minimal to moderate laryngeal squamous metaplasia for which we describe the kinetics of recovery over 1 to 4 weeks. We found that the microscopic change epithelial alteration, which is normally considered to be a precursor in the development of squamous metaplasia, can occur as a transitional stage between squamous and normal epithelium during recovery.
Keywords
Introduction
Regulatory requirements dictate the use of rodents as animal models in nonclinical toxicology studies. 1 As a result, rats are often used in inhalation toxicity studies. The upper respiratory tract, particularly the larynx, is a common target organ for many pharmaceuticals, industrial chemicals, agrichemicals, and environmental pollutants 2 –9 and many regulatory risk assessments for potential inhalation exposure to toxicants are based on lesions observed in the upper respiratory tract of rodents. 10
The larynx’s response to insult is limited. Most toxicant-associated changes are nonspecific and consist of inflammation, necrosis, degeneration, and regeneration similar to other areas in the respiratory tract. 11,12 Neoplasia is uncommon. 12 These changes depend to some extent on the dose and duration of the study rather than the specific test item. 10,11 Squamous metaplasia is a common test item-related change in the larynx in inhalation studies. 13 It is an adaptive response to repeated irritation in which the more sensitive respiratory epithelium is replaced by a more resistant squamous epithelium. 14 Transformation from normal to squamous epithelium can occur within 3 days of commencement of exposure, whereby the damaged respiratory epithelial cells are “lifted” from the basement membrane by a squamous epithelium. 11 The severity of squamous metaplasia depends on the concentration of the test item and duration of exposure. 10 The epithelium at the base of the epiglottis, located anterior to the ventral pouch, is the most sensitive region of the rodent larynx for the development of squamous metaplasia. It is often the first location for squamous metaplasia to occur and may be the only site where low-grade irritation changes occur. 10,11,15,16 Squamous metaplasia may be accompanied by inflammatory cell infiltrates or inflammation. Epithelial alteration is the initial stage of a generally concentration-dependent transformation from normal respiratory epithelium to laryngeal squamous metaplasia and is characterized by a focal loss of cilia and flattening of epithelial cells at the base of the epiglottis. 5 Criteria for grading epithelial alteration and squamous metaplasia and the assessment of adversity are based on the extent of the lesion, number of cell layers, and the degree of hyperplasia and keratinization described by Kaufmann et al. 5 Kauffman et al further state that epithelial alteration and minimal to slight squamous metaplasia, in the absence of inflammation, should be considered as nonadverse as laryngeal dysfunction is not expected. On the other hand, per authors, moderate to severe squamous metaplasia should be considered as adverse as dysfunction due to reduced mucociliary clearance and decreased tendency to cough cannot be ruled out. Keratinization, seen with moderate to severe squamous metaplasia, is also closely related to the risk of aspiration and animals may die of airway obstruction.
The epithelium recovers for both epithelial alteration and squamous metaplasia following removal of the inciting stimulus. Epithelial alteration completely regresses 6 to 13 weeks after removal of the inciting stimulus. 5 Recovery from epithelial squamous metaplasia is variable and depends on the time scale of exposure and severity of the initial lesion. 11 Removal of the inciting stimulus for a period of 6 of 13 weeks following subchronic (13 weeks) exposure results in regression of the lesion and return of the affected epithelium to normal morphology. 4,10 With shorter exposures, regression of squamous metaplasia can occur within 1 week of removal of the inciting stimulus. 6,17
Studies in this laboratory involving a synthetic herbicide that induced squamous metaplasia in the larynx of rats provided additional insights into the early changes associated with regression and recovery from squamous metaplasia. We found that epithelial alteration, which is normally considered to be a precursor in the development of squamous metaplasia, can occur as a transitional stage between squamous and normal epithelium during recovery and can be seen within 1 week following cessation of exposure to an inciting stimulus.
Materials and Methods
Two studies (a preliminary study, study 1, and a definitive study, study 2) evaluated the toxicity and toxicokinetic profile of 3 selected exposure levels of MCPB acid (a synthetic plant auxin herbicide) when administered via nose-only inhalation to rats and evaluated the degree and time dependency of recovery from any potential effects during an up to 4-week test item-free recovery period.
Animals
Male Sprague Dawley rats (approximately 9-10 weeks old at the start of exposure) were obtained from Envigo (Fredrick, Maryland in study 1, Indianapolis, Indiana in study 2). Animals were housed, 2 or 3 per cage, in polycarbonate cages with stainless steel lids and were provided with food (Teklad Global 16% Protein Rodent Diet 2016C; certified, non-autoclavable; Envigo, Madison, Wisconsin) and water ad libitum. Cages were maintained in humidity and temperature-controlled, high-efficiency particulate air-filtered mass air displacement rooms in a facility accredited by the American Association for Accreditation of Laboratory Care. A 12-hour light/dark cycle was provided via automatic timer. Animals were acclimated to the method of restraint for 3 (study 1) or 7 (study 2) days within the 7 days preceding their initial test item exposure. The duration of restraint during the habituation phase was increased on each day to minimize restraint-associated stress during subsequent MCPB acid exposures. Both studies were conducted in a facility which operates in accordance with Good Laboratory Practice principles and both complied with all appropriate parts of the Animal Welfare Act Regulations: 9 CFR Parts 1 and 2 Final Rules, Federal Register, Volume 54, No. 168, August 31, 1989, pp. 36,112- 36,163, effective October 30, 1989, and 9 CFR Part 3 Animal Welfare Standards; Final Rule, Federal Register, Volume 56, No. 32, February 15, 1991, pp. 6426-6505, effective March 18, 1991.
Test Item
4-(4-chloro-2-methylphenoxy) butyric (MCPB) acid (CAS Number 94-81-5) (Nufarm, Wyke, Bradford, United Kingdom). Purity 96.33%. The test item was supplied for use as a micronized powder.
Control
Control animals received compressed air only from the same source used for the test groups. The compressed air used for aerosol generation was supplied from an in-house system and was of breathing quality.
Exposure System
The test item exposure system consisted of separate modular 40 L flow through exposure towers (ADG Developments, Codicote, Hertfordshire, United Kingdom), each with two 20-port animal exposure levels. The particulate aerosol was generated using a rotating brush generator (RBG 1000; Palas GmbH, Karlsruhe, Germany) fitted with a stainless steel brush. The brush was operated at a constant speed of 650 revolutions per minute. The micronized MCPB acid powder was packed into 20-mm-diameter (group 2) or 28-mm-diameter (groups 3 and 4) powder canisters at a target pressure of 5 psi using a mechanical press. Aerosol was generated into an airflow of 35 L/min from the house compressed airline. An additional 3 L/min of compressed air was supplied from the house air system and 2 L/min drawn into each tower directly from the room environment. Extract air was drawn from the base of each chamber at a flow of 40 L/min and filtered locally before passing into the house vacuum line. All airflows were calibrated before the start of each study, and the inline flows displayed continuously and confirmed at intervals throughout each exposure. The control and test item exposure systems were each housed in a separate ventilated containment cabinet.
On each day of exposure, the RBGs were located onto the top section of each exposure system and the exhaust airflow established. The rats were placed into their restraint tubes and the restraints located on the designated exposure tower. After establishing the aerosol generation and diluent air supplies, all remaining unused exposure ports were fitted with blanking stoppers. Exposure system temperature, humidity, and airflows were documented at 30-minute intervals throughout each exposure. At the end of the exposure period, the generators were switched off and a single blanking stopper at the top level of each system was removed to allow room air to enter. Animals remained on the exposure tower for a minimum of 7 minutes to allow decay of the residual test item in the exposure system atmospheres. Each animal was examined as it was removed from its restraint and again following return to the home cage.
Atmosphere Sampling and Analysis
Aerosol concentration samples were collected at a flow of 2 L/min directly onto Quartz fiber filters (37 mm; Millipore Sigma, Burlington, Massachusetts). Sample volume was precisely measured using an inline wet type gas meter (Type W-NK, Shinagawa, Tokyo, Japan). On each occasion of exposure, a minimum of 3 samples were collected from representative animal exposure ports on each exposure system and the achieved concentration assessed gravimetrically from the filter weight gain. Additionally, all samples collected on days 1 of 4 and on 1 occasion in each subsequent week were submitted for high-performance liquid chromatography (HPLC) determination of the MCPB acid collected. The particle size distribution (PSD) of the MCPB acid presented in the breathing zone was determined at weekly intervals. The PSD sampling was performed using a Marple 290 series cascade impactor (296 configuration; Thermo Fisher Scientific, Waltham, Massachusetts) operated at 2 L/min and the volume of each sample determined by wet type gas meter. Both gravimetric and HPLC determination of the material collected on the cascade impactor stages were performed for all PSD samples collected.
Inhalation Exposures and Recovery Period
Study 1
This was a preliminary study with the test item. It was conducted in advance of study 2 (the definitive study) in order to confirm dose level selection and recovery periods. It utilized fewer animals and had a single recovery period. Animals in this study were exposed by nose-only inhalation as a powdered aerosol once daily, 5 days/week, 6 hours/day at nominal concentrations of 0 mg/L (group 1, air only, n = 10), 0.02 mg/L (group 2, n = 5), 0.05 mg/L (group 3, n = 5), or 0.20 mg/L (group 4, n = 10) MCPB acid for 3 consecutive weeks. Mean achieved aerosol analytic concentrations were 0.000, 0.021, 0.055, and 0.175 mg/L, respectively, and were consistent with the concentrations determined gravimetrically. The mean achieved analytically determined mass median aerodynamic diameter (MMAD) in the presented atmospheres were 3.7, 3.8 and 3.4 µm equivalent aerodynamic diameter (EAD) for groups 2, 3, and 4, respectively, with a range of geometric standard deviation (sg) across all samples of 1.94 to 2.41. At the end of the treatment period, 5 animals/group were euthanized and necropsied. The remaining 5 animals in the control and 0.20 mg/L groups were held for a 1-week treatment-free recovery period and then euthanized by exsanguination following isoflurane inhalation and necropsied.
Study 2
Animals were exposed by nose-only inhalation as a powdered aerosol once daily, 5 days/week, 6 hours/day at target concentrations of 0 mg/L (group 1, air only, n = 28), 0.02 mg/L (group 2, n = 28), 0.05 mg/L (group 3, n = 28), or 0.20 mg/L (group 4, n = 28) MCPB acid for 4 consecutive weeks. Exposure levels for this study were the same as for study 1 since there were no clinical signs at these exposure levels and the histopathology findings in study 1 were not considered dose limiting. Mean achieved aerosol analytically determined concentrations were 0.000, 0.025, 0.063, and 0.196 mg/L, respectively. The mean achieved analytically determined MMAD of MCPB acid in the presented atmospheres were 2.9, 3.1, and 3.4 µm EAD for groups 2, 3, and 4, respectively, with a range of geometric standard deviation (sg) across all samples of 1. 66 to 2.16. At the end of the treatment period, 10 animals/group were euthanized and necropsied. After a 1-week, 2-week, and 4-week treatment-free recovery period, 6 animals/group/time point were euthanized by exsanguination following isoflurane inhalation and necropsied. Animals did not have access to food and water during the exposures and observations for signs of reaction to treatment were made on a group basis once during each exposure for both studies.
Histopathology (Both Studies)
At necropsy, complete macroscopic examinations were performed on all animals and study plan-specified organs were weighed. Multiple tissues including the entire respiratory tract were collected and fixed in 10% neutral-buffered formalin. Fixed tissues were embedded in paraffin, sectioned, and mounted on slides. Slides were then stained with hematoxylin and eosin (H&E) and evaluated by light microscopy.
Larynx
Three anatomic levels of the larynx were evaluated per Registry of Industrial Toxicology Animal data Revised Guides for Organ Sampling and Trimming in Rats and Mice. 18 Level 1 sampled the anterior larynx at the base of the epiglottis and included the ventral seromucinous glands. Level 2 sampled the mid-larynx and included the ventral pouch and arytenoids. Level 3 sampled the posterior larynx and included the cricoid cartilage (Figure 1A-D). The 3 levels were examined microscopically utilizing a total of 6 tissue sections; 3 tissue sections from level 1, 1 tissue section from level 2, and 2 tissue sections from level 3. This was done to ensure adequate visualization of all relevant structures. Epithelial alteration and squamous metaplasia were diagnosed and graded according to criteria established by Kaufmann et al 5 (Table 1).
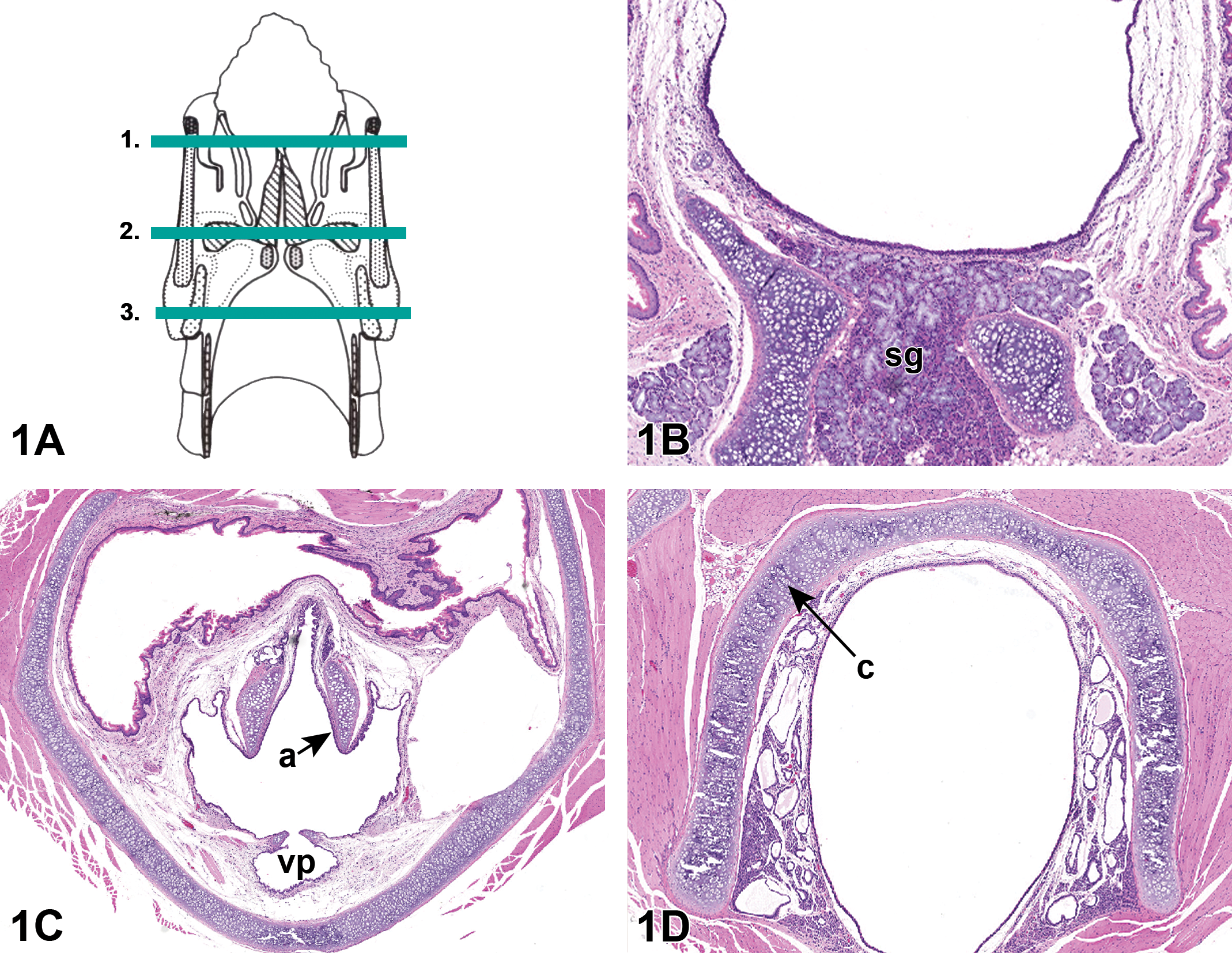
A, Trimming procedure of the rodent larynx (according to Kittle et al 18 ). B, Cross section of level 1 of the rat larynx, seromucinous glands (sg). C, Cross section of level 2 of the rat larynx, process of the arytenoid cartilages (a), ventral pouch (vp). D, Cross section of level 3 of the rat larynx, cricoid cartilage (c). Tissues stained with H&E.
Diagnosis and Grading of Epithelial Alteration and Squamous Metaplasia in the Rat Larynx.

a Adapted from Kaufmann et al. 5
Review of Slides
Slides were evaluated independently by 2 different pathologists (study 1: C.M.K., study 2: N.P.M.).
Results
No unscheduled deaths, test item-related organ weight changes, or test item-related macroscopic changes occurred in either study.
Histopathology
Test item-related microscopic findings were present in the larynx in both studies.
Study 1
Terminal necropsy treatment phase
Epithelial alteration or squamous metaplasia occurred in the larynx of nearly all animals at all MCPB acid exposure levels following 3 consecutive weeks of exposure to the test item. Minimal to slight squamous metaplasia of the laryngeal respiratory epithelium occurred at ≥0.02 mg/L and exhibited an exposure-related increase in incidence and severity. Most or all animals at 0.05 and 0.2 mg/L had squamous metaplasia and most animals at 0.02 mg/L had epithelial alteration (Table 2). Epithelial alteration did not occur concurrently in the same animals as squamous metaplasia. The findings showed a concentration-dependent shift in character from epithelial alteration to squamous metaplasia. Squamous metaplasia was located in the anterior larynx, both ventrally and occasionally in the lateral wall. Epithelial alteration was located in the anterior larynx at or near the base of the epiglottis. Neither epithelial alteration nor squamous metaplasia occurred in the control group. Examples of epithelial changes seen at the end of the treatment phase in studies 1 and 2 are given in Figure 2.
Study 1 Test Item-Related Findings in the Larynx in Male Rats Exposed to MCPB Acid for 3 Weeks and Followed by a 1-Week Recovery Period.
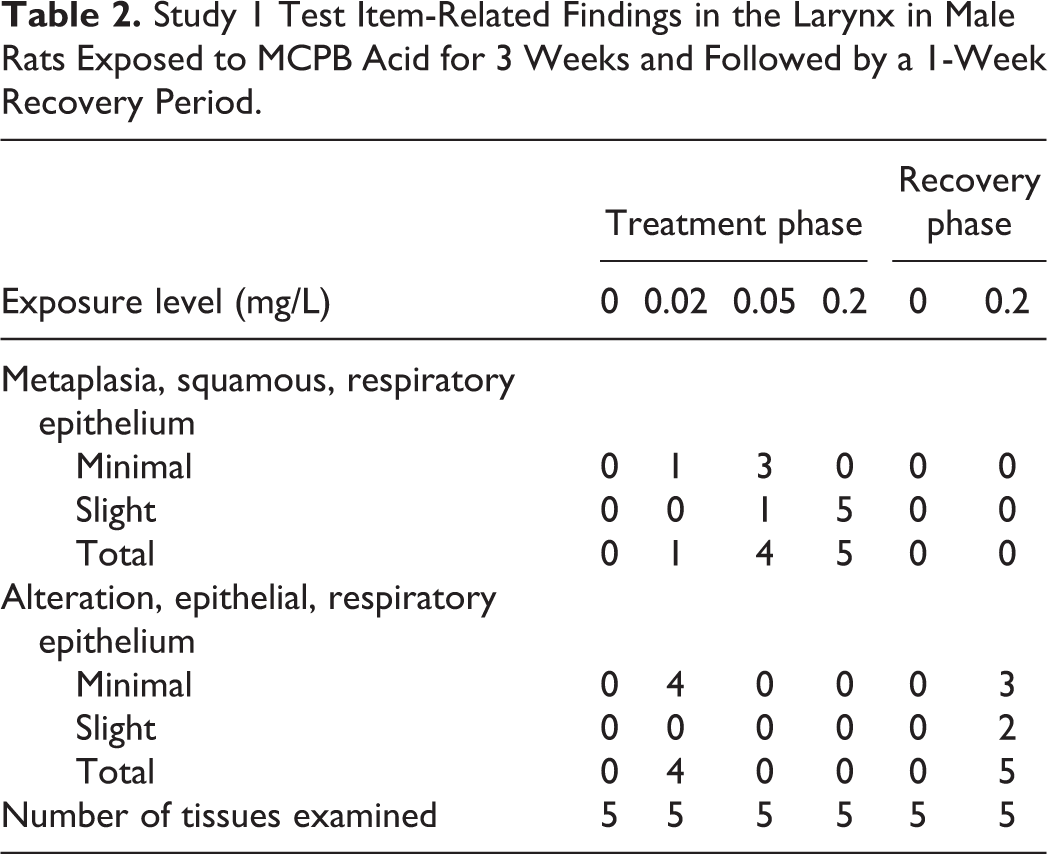
Examples of epithelial alteration and squamous metaplasia found in rats in studies 1 and 2. End of treatment phase. A, Normal epithelium (arrows); (a) higher magnification. B, Minimal epithelial alteration (arrow); (b) higher magnification. C, Slight epithelial alteration (arrow); (c) higher magnification. D, Minimal squamous metaplasia (arrow); (d) higher magnification. E, Slight squamous metaplasia (arrow); (e) higher magnification. Tissues stained with H&E.
Terminal necropsy recovery phase
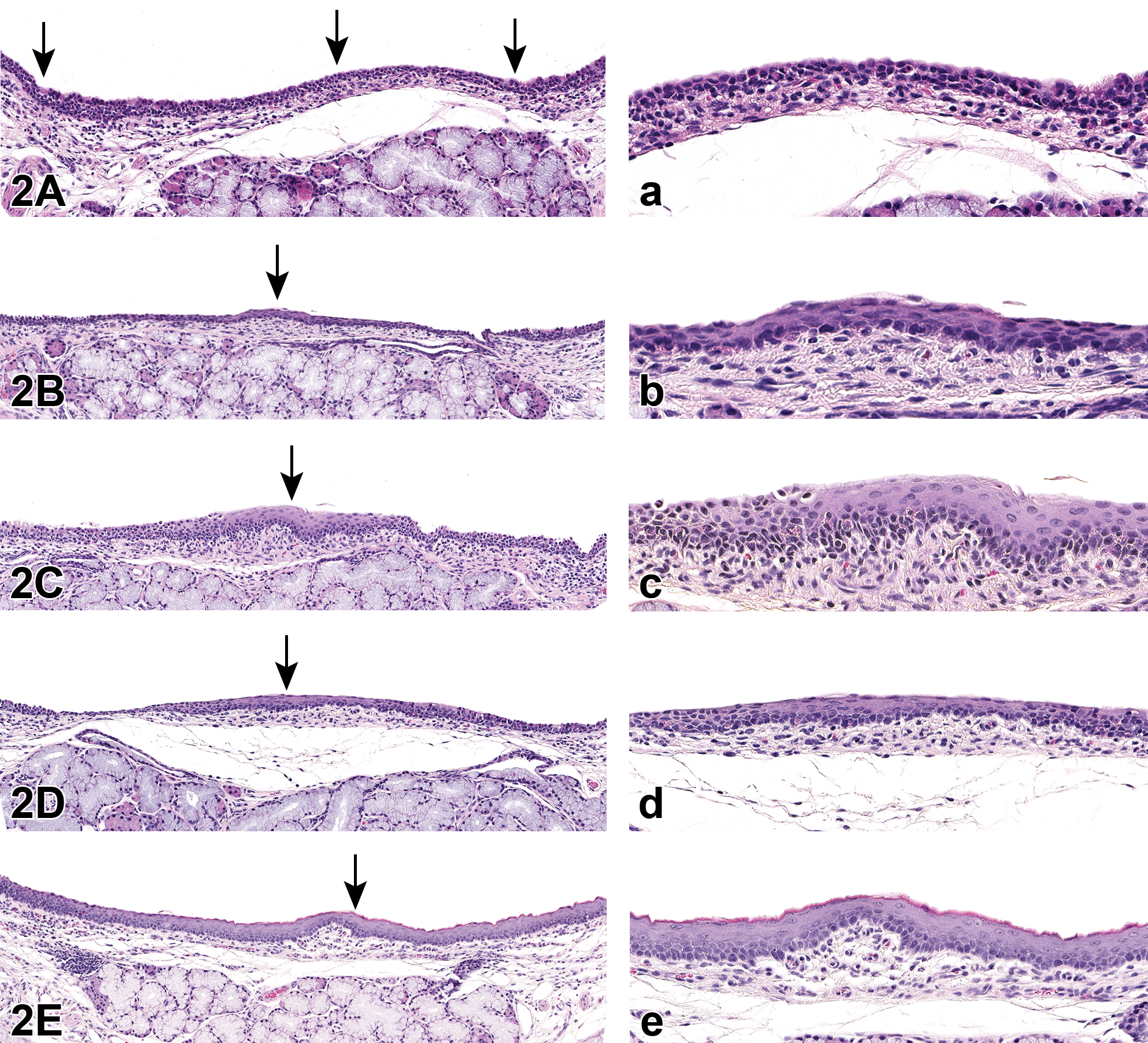
All animals in the 0.20 mg/L recovery group exhibited only minimal to slight epithelial alteration following a 1-week treatment-free recovery period. Squamous metaplasia was notably absent (Table 2). Examples of epithelial changes seen at the end of the 1-week recovery phase in studies 1 and 2 are given in Figure 3.
Examples of epithelial alteration and squamous metaplasia found in rats in studies 1 and 2. End of 1-week recovery phase. A, Normal epithelium (arrows); (a) higher magnification. B, Minimal epithelial alteration (arrow); (b) higher magnification. C, Slight epithelial alteration (arrow); (c) higher magnification. Tissues stained with H&E.
Study 2
Terminal necropsy treatment phase
Changes in the larynx at the end of the treatment phase in study 2 were similar to those in study 1. Epithelial alteration or squamous metaplasia occurred in all animals at all MCPB acid exposure levels and again there was an exposure-related shift in character from epithelial alteration at 0.02 mg/L to squamous metaplasia at ≥0.05 mg/L (Table 3). Both changes were located in the anterior larynx at or near the base of the epiglottis. Neither epithelial alteration nor squamous metaplasia occurred in the control group.
Study 2 Test Item-Related Findings in the Larynx in Male Rats Exposed to MCPB Acid for 4 Weeks and Followed by a 1-Week, 2-Week, and 4-Week Recovery Period.
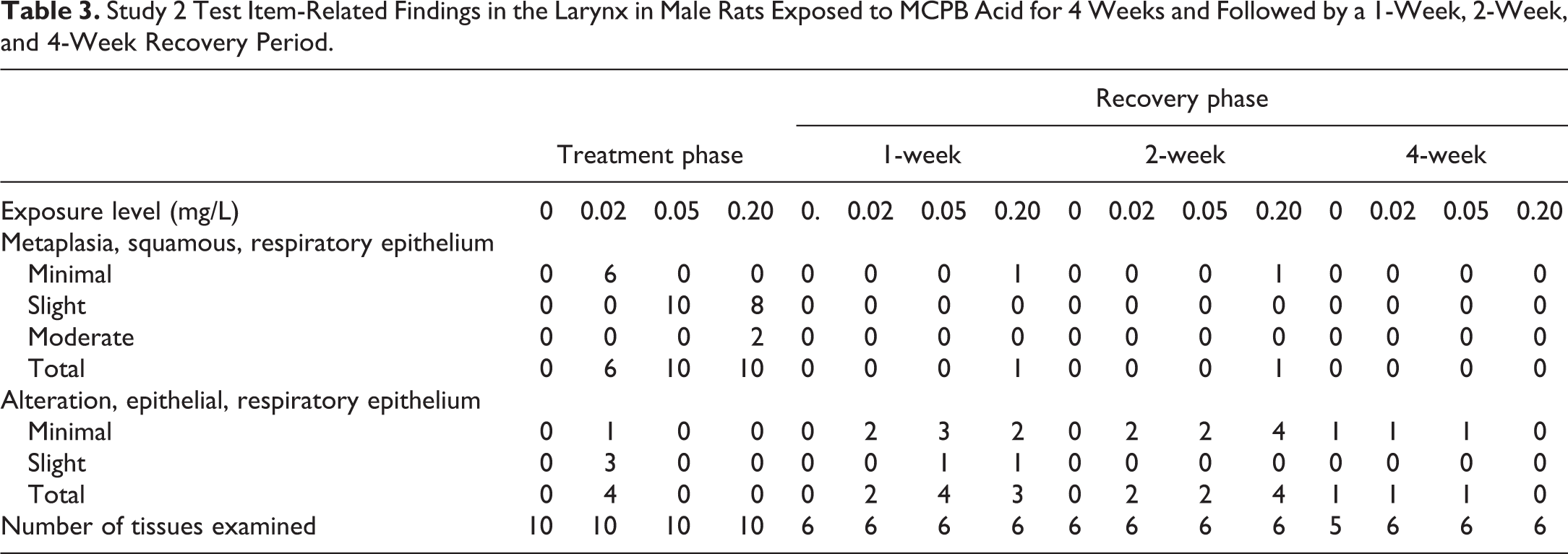
Terminal necropsy recovery phase
Whereas squamous metaplasia was present in all animals in the 0.05 and 0.20 mg/L groups at the end of the dosing period, epithelial alteration was present in many animals in these exposure groups at the end of the 1 and 2 week recovery periods. This shift in character, amelioration from squamous metaplasia to epithelial alteration, was similar to that seen in the 0.20 mg/L recovery group in study 1. Epithelial alteration in the 0.05 and 0.20 mg/L groups was decreased in severity between the end of the 1-week (minimal to slight) and 2-week (minimal) recovery periods and was also indicative of progression to normal epithelium. Squamous metaplasia was absent in the 0.020 mg/L recovery group beginning at 1 week. Complete recovery from both changes in all exposure groups occurred by the end of 4 weeks. Epithelial alteration occurred with a similar incidence (1 animal) and severity (minimal) in the control 0.02 and 0.05 mg/L recovery groups and was, at the 4-week recovery time point, considered be a background change that was unrelated to exposure (Table 3).
Discussion
In both studies, exposure to the test item was associated with epithelial alteration and squamous metaplasia in the larynx and had a concentration-dependent shift from epithelial alteration to squamous metaplasia during the exposure period. This shift was consistent with the concept that epithelial alteration is a precursor in the development of squamous metaplasia from normal epithelium. 5 During the recovery period, a pattern of change was noted in both studies in which squamous metaplasia that was observed at a given concentration during the exposure period was absent and was replaced by the less severe change, epithelial alteration, in the corresponding group during the recovery period. In study 1, this was seen in the 0.2 mg/L exposure group at the end of the 1-week recovery period. In study 2, this was seen in the 0.05 and 0.2 mg/L exposure groups at the end of the 1- and 2-week recovery periods. In both studies, the replacement of squamous metaplasia by epithelial alteration was interpreted as being indicative of ongoing recovery and suggested that epithelial alteration can occur as a transitional stage in the regression of squamous metaplasia to normal epithelium. Further transition from epithelial alteration to normal epithelium, complete recovery, was observed in all exposure groups at the end of the 4-week recovery period in study 2.
The term epithelial alteration was used for the microscopic changes observed during the recovery period in both studies due to the similarity in appearance to the eponymous change described by Kaufmann et al. The finding likely resulted from squamous epithelial cells and keratin, on the surface of areas with squamous metaplasia, sloughing away and leaving behind underlying cells that were transitioning to normal respiratory epithelium. These remaining cells met the diagnostic criteria for epithelial alteration. It is acknowledged that the pathogenesis of this change is not exactly the same as for epithelial alteration as described by Kaufmann et al. 5 The cells observed during the recovery period were not transforming (becoming altered) in response to a stimulus but rather were responding to a lack of stimulation and were returning (transforming) to normal morphology. An alternative, albeit awkward, term for the finding seen during recovery from squamous metaplasia could be “epithelial alteration-like change.” Regardless of terminology, both studies showed that the presence of this finding in the larynx of rats during the recovery period from test item-associated squamous metaplasia was indicative of ongoing recovery from squamous metaplasia and that this can occur within 1 week of removal of the inciting stimulus. This has ramifications for risk assessment as it is an early indicator of recovery which should be a fundamental part of assessing risk. The finding of epithelial alteration during recovery from squamous metaplasia is not unique to this test item. The authors (N.P.M. and C.M.K.) have seen and described it in other inhalation studies with different chemicals unrelated to MCPB acid. The ability to differentiate this change in these studies was most likely related to the short recovery periods (1 to 2 weeks) which allowed it to be detected before complete recovery to normal respiratory epithelium.
In conclusion, epithelial alteration can occur during recovery from laryngeal squamous metaplasia. The change can occur within 1 week after removal of the inciting stimulus of squamous metaplasia and can serve as an early indicator of ongoing recovery.
Footnotes
Acknowledgments
The authors wish to thank the members of MCPB acid Task Force for their contribution to the MCPB acid studies and their permission to share the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
