Abstract
Middle ear administration has numerous applications, including antibiotherapy and gene therapy, and is increasingly used to target the auditory and vestibular systems. In animal studies, investigating repeated exposure that mimics clinical dosing regimens has remained a challenge due to the lack of suitable models. Intratympanic injections are not suitable for long-term studies due to the increased risk related to tympanic membrane rupture or scarring and repeat anesthesia events. Surgical models of middle ear catheterization previously used have not been reliable for longer than 4 weeks, resulted in elevated stress levels, and have been associated with significant changes related to the surgery and/or the presence of the catheter such as local trauma and inflammatory and degenerative processes. These complications cause decreased hearing/deafness and greatly diminish the value and accuracy of ototoxicity studies. We describe here a procedure that permits repeat dosing into the middle ear of guinea pigs and can be used to produce a model of aminoglycoside-induced hair cell injury. The innocuity of the procedures and the efficacy of the ototoxicity model were confirmed using auditory brain stem response assessment, histopathological evaluation, and cytocochleograms. Procedure-related changes were limited to minimal inflammation in the middle ear.
Introduction
Historically, and despite the importance of hearing and balance for human well-being, the middle ear and inner ear have been largely ignored during drug development and safety assessment, particularly during preclinical testing. 1 In recent years, there has been an increased interest in exploring the ototoxic potential of new therapeutic agents administered directly to the middle and/or inner ear with the goal of providing otoprotection or treating conditions such as infectious and genetic diseases, loss of hearing, tinnitus, and balance disorders.
Drug delivery by intratympanic injection (injection into the middle ear through the tympanic membrane, also referred to as transtympanic injection) can be performed in humans for drugs such as gentamicin, corticosteroids, and others to the middle ear to treat conditions including otitis media 2,3 or the inner ear to treat Ménière disease, 4,5 tinnitus, 6 and sensorineural hearing loss. 7 Drugs injected into the middle ear diffuse into the adjacent inner ear, 8 mainly through the round window membrane. 9 In many instances, it is necessary to repeat the intratympanic injections, and complications are likely to occur, including pain, tongue numbness, transient dizziness, vertigo, tinnitus, and persistent tympanic perforation. 10
Animal studies that involve repeated middle ear administration present several major challenges. First, it is important to avoid complications resulting from repeated intratympanic injections that could mimic or mask eventual drug-related effects. These consist of inflammation of the outer ear canal due to its small diameter relative to humans, increased risk of tympanic membrane perforation and scarring secondary to multiple needle insertions, infection, and middle ear inflammation. Changes in the outer and middle ear can result in shifts in hearing thresholds, vestibular or behavioral changes, and microscopic findings. Second, while intratympanic injections can be performed in nonanesthetized human patients, they must be performed under general anesthesia in animals to prevent movement during the procedure and minimize stress. Combined, these challenges limit the frequency of administration in order to allow sufficient healing of the tympanic membrane between injections and recovery between anesthetic events. Further, assessment of in vivo hearing evaluation (eg, auditory brain stem response [ABR]) requires additional anesthetic events, which must be taken into consideration. Finally, histological preparation, including adequate fixation of the hair cells and decalcification of the temporal bone, must be done immediately after collection using optimum reagents to avoid postmortem changes in the hair cells that interfere with histopathological examination. Hair cells are very sensitive to decalcification solutions, and the usual formic acid method damages them greatly. Thus, a chelating agent, such as EDTA, is often the preferred choice for temporal bone decalcification. 11 While this method is easy and efficient when working with small rodents such as mice and rats, it becomes very lengthy and tedious when applied to larger and thicker temporal bones found in bigger animals such as dogs and monkeys, necessitating up to several months for complete decalcification in many cases. To avoid this issue, an alternate method was used to accelerate decalcification while preserving cellular morphology. 12 –14
The guinea pig is a preferred species in ototoxicity testing for several reasons. Guinea pigs are relatively easy to handle and house. Anatomically, their temporal bone, cochlea, and vestibulocochlear nerve are similar to those of humans. 15 Their ear structures are large enough to be relatively easily accessed for dose administration and tissue collection at necropsy but small enough to allow minimal decalcification time. Finally, this species demonstrates reproducible sensitivity to ototoxicity and appears to be the most reliable model for neurosensory deafness. 16
To the authors’ knowledge, no successful long-term animal models of bilateral drug delivery via middle ear catheterization through the temporal bone have been described in the literature. In our experience, such models have often failed because of significant complications associated with surgery model and/or reaction to the catheter material. Here, we describe a refined model of long-term, bilateral middle ear catheterization through the temporal bone in guinea pigs, which can be used in toxicology and safety assessment studies as well as to produce models of hearing loss and hair cell injury for efficacy studies.
Materials and Methods
Animals
Ten (10) male Hartley albino guinea pigs (Cavia porcellus; Crl: HA from St-Constant, Quebec, Canada) weighing approximately 400 g were group housed in open-top plastic children’s pools (purchased at Wal-Mart) according to densities and housing conditions listed by the Canadian Council on Animal Care. The pool floors were covered with a commercially available corn cob bedding, and opaque roofed structures were provided as group shelters. Animals received ad libitum water, irradiated hay, and pelleted diet as well as fresh produce daily.
Preoperative Procedures
Animals were fasted for 1 to 3 hours prior to anesthesia. Preparation for surgery included prophylactic administration of antibiotics (trimethoprim sulfa at a dose of 30 mg/kg orally) at least 30 minutes prior to surgery, and multimodal analgesia included the administration of a nonsteroidal anti-inflammatory and an opioid (meloxicam, 0.5 mg/kg orally, and sustained release buprenorphine, 0.5 mg/kg subcutaneous). A subcutaneous injection of isotonic crystalloid solution, lactated Ringer’s, was administered prior to surgery at 10 mL/kg. The animals then received an intramuscular administration of a preanesthetic cocktail of ketamine (64 mg/kg) and xylazine (9.6 mg/kg), and anesthesia was achieved with concomitant isoflurane set at 0.5% to 1% in oxygen and set at 500 mL/minute. Once sufficiently sedated, a liberal application of eye lubricant (ie, Natural Tears, Alcon Canada Mississauga, Ontario) was performed. The head, cheeks, interscapular, and dorsal thoracic regions were shaved. The shaved areas along with the inside of the pinna were cleaned with a water diluted chlorhexidine 4% solution. The entire region, except the inside of the pinna, was then further prepared by 1 wipe of sterile water for irrigation, followed by 3 wipes of iodine 10%. A pulse oximeter was used for monitoring of blood oxygen saturation and heart rate. In addition, visual observation of breathing and mucosa color was done regularly along with pedal/palpebral reflexes verification.
Surgical Procedures
The animals were positioned in sternal recumbency with rolled gauze under the chest giving a slight forward tilt to the head. The animals’ temperature was maintained using water circulating heating pads throughout the anesthesia. The animals were then draped in sterile fashion. As a landmark, the angular process of the mandible was palpated knowing the temporal bone is almost in line ventrally immediately above. An elliptical skin incision was performed along most of the medial side of the pinna to the base at the cartilage and muscle junction. The ear pavilions were retracted laterally using Alm retractors and stay sutures (4-0 Monosof, Medtronic Canada, Kirkland, Quebec) and hemostats clamped to the surgical drape, thus gaining better visualization of the area of interest. Using a combination of Metzenbaum, iris scissors, and/or periosteal bone elevators, the cleidomastoid, cleidooccipital, sternomastoid, and cleidocephalic muscles were gently and bluntly dissected exposing the temporal bone. The tissues were continuously kept moist throughout the surgery with saline irrigation. Once the dissection was completed and the temporal bone ready for cannulation, an incision in the interscapular region was made and a subcutaneous pocket made to accommodate the placement of an Instech Laboratories (Plymouth Meeting, PA) dual channel Vascular Access Button for Rats (VAB). The VAB, with catheters preattached, was filled with saline solution and the catheters meticulously tunneled, to avoid damage to the catheters, from the interscapulum incision site using a trocar to the bilateral ear sites. The VAB was positioned snug with an interrupted absorbable subcuticular suture at the end of the incision line (4-0 PDS, Ethicon, Somerville, NJ) without further anchoring necessary. This permitted a snug incision and prevented damage to the catheters. A Stryker Core Impaction electric drill with a 1-mm drill bit was used to breach the middle ear cavity via a hole in the ventral portion of the temporal bone (Figure 1A). Saline solution was used to irrigate the drilling area avoiding heat damage during drilling, and the bone dust was removed as much as possible in order to avoid cavity contamination. A custom 3F, bulbed silicone catheter was introduced through the drill holes. The bulb was press fitted through and past the bone cortex. Once inside, it was pulled back toward the endosteum and a drop of surgical glue (Vet bond, 3M, Saint Paul, MN) was used to anchor the catheter in place. A moveable disk on the catheter was advanced to the temporal bone and glued in place preventing movement either way (Figure 1B). Patency checks, using saline solution, were made to ensure there was no reflux. The muscles were repositioned naturally over the disk and temporal bone using single interrupted sutures of absorbable suture material (4-0 PDS). The surgical site was gently flushed with warmed saline followed by a local anesthesia field block of a lidocaine/bupivacaine mixture at the ear sites. A field block was also done at the interscapulum site a few minutes prior to incision. The subcutaneous tissues were approximated to obliterate dead space and the skin closure completed with microdroplets of surgical glue.
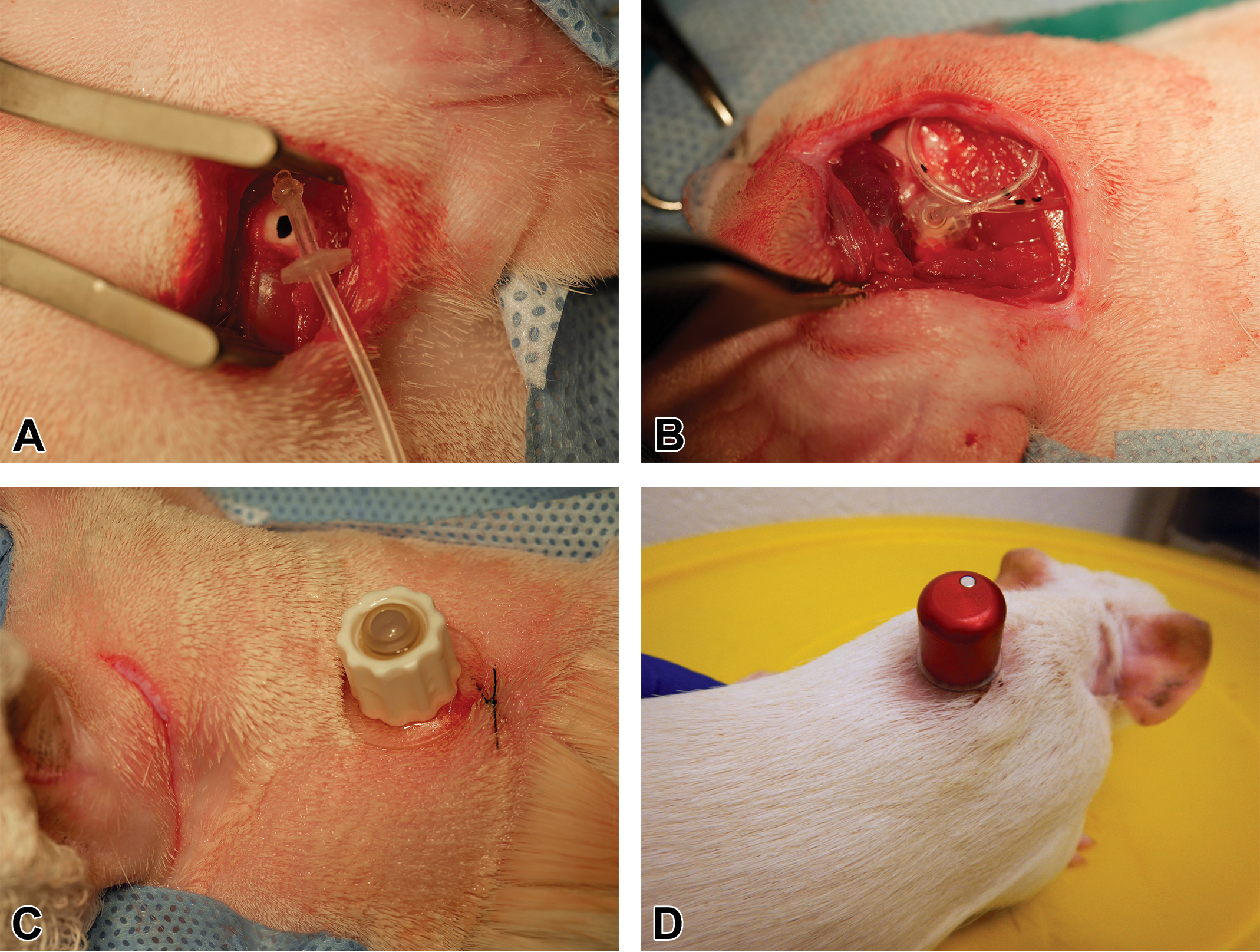
Surgical procedures. Hole drilled in temporal bone (A), catheter with disk (B), and catheter exit site in scapular region with protective cap (C and D).
Postoperative Procedures
Following surgery, the animals received intramuscular atipamezole (1 mg/kg) to help reverse xylazine effects and speed up recovery from anesthesia. The animals were kept on nasal oxygen and on a warm water heating pad throughout the anesthesia until sufficiently recovered to permit return to the housing pool. Also, a single application of topical antibiotic ointment (Polytopic) on the surgical sites was performed along with a second subcutaneous injection of lactated Ringer’s. A protective cap was placed when the VAB was not in use, permitting immediate group housing of the animals following surgery without the worry of device damage and more importantly facilitating recovery from surgery and general well-being of the animals (Figure 1C and D). Prophylactic eye lubricant was administered daily for a total of 3 days inclusively to prevent postanesthetic corneal drying and damage. In addition, subcutaneous injections of lactated Ringer’s were administered daily for 3 days.
Study Design
Animals were assigned to 2 treatment groups as indicated in Table 1.
Study Design.
Starting 2 weeks after surgery, test formulations (sterile saline or gentamicin; 70 μL/dose) were administered bilaterally to the middle ear by slow injection followed by a flush of sterile saline (20 µL). Animals were given a total of 4 doses, twice weekly, and were monitored for up to 9 weeks postsurgery. Throughout the study, animals were monitored for general health at least weekly (detailed clinical examinations and measurement of body weight).
Auditory Brain Stem Response
Animals underwent bilateral ABR assessment at 4, 8, and 16 kHz at 3 occasions: 2 (prior to dosing), 6, and 9 weeks following surgery. Animals were anesthetized by intramuscular injection of a cocktail (ketamine 64 mg/kg and xylazine 9.6 mg/kg). Recordings were performed using a Tucker Davis Systems BioSig (Tucker-Davis Technologies, Alachua, FL). Each test was performed beginning at 90 dB sound pressure levels (SPL) and decreasing in intensity using 5 dB steps down to 0 dB SPL. The signal pulse was by free-field presentation, 5 milliseconds duration and 0.5 milliseconds gate time, averaged 512 times per intensity, and the threshold at the intensity level 5 dB above the no electroencephalogram activity level determined. The contralateral ear was occluded using a foam earplug during ipsilateral assessment.
Necropsy
Prior to necropsy, all animals were anesthetized with isoflurane and humanely euthanized by exsanguination. The external ears, skin, and excess tissue were removed, and the skull was disarticulated. The front part of the skull was removed by cutting it rostrally to the tympanic bulla. The remainder of the skull was cut in half through the midline, and PFF solution (85% aqueous saturated picric acid, 10% formaldehyde, and 5% formic acid) was gently injected into the right tympanic bulla. Right temporal bones were placed in precooled PFF solution and then refrigerated at 4°C. Left cochleae were stained with succinate dehydrogenase, fixed with 10% buffered formalin, and dissected to remove organ of Corti in order to perform the cytocochleograms.
Histopathology
Decalcification of the right temporal bones was assessed periodically by X-rays. Following complete decalcification, the tissues were embedded in paraffin, step-sectioned (5-μm-thick sections at 75-µm intervals), mounted on glass slides, stained with hematoxylin and eosin, and evaluated by a board-certified pathologist.
Cytocochleograms
Whole mounts of left cochleae were examined at a magnification of ×20 with an Olympus microscope equipped with a scale (reticle) built into 1 eyepiece. In random order sequence, each organ of Corti was divided in 250-µm segments along its entire length, and missing hair cells were counted for each of these segments. The numbers of missing hair cells for each group of 4 consecutive segments were added to provide a number of missing hair cells representing approximately 5% of the total length of the organ of Corti. In order to calculate the percentage of missing hair cells, the sum of missing hair cells within each of these 5% sections was divided by the expected total number of hair cells in the corresponding 5% section (according to an in-house database built from examination of naive guinea pig cochleae).
All animal procedures were conducted in accordance with the policies and recommendations of the Canadian Council on Animal Care and under approval from the Institutional Animal Care and Use Committee of Senneville Charles River Laboratories.
Results
Clinical Condition and Surgical Recovery
Transient body weight loss and clinical signs related to the surgical procedures (scabs/skin or fur discoloration) were observed during the first postoperative week. However, overall, the recovery was very good, with return to normal food intake and grooming behavior within days of surgery. Body weight gain was noted by the second week of recovery, and animals administered saline remained in good condition for the remainder of the study.
Catheters remained implanted throughout the study period; the cap protected the ports from potential damage (eg, due to scratching or cage mates) and no noteworthy findings at the catheter exteriorization site were noted.
Middle ear administration of gentamicin resulted in vestibular effects, including head tilt, uncoordination, and abnormal gait, and was associated with reduced food and water intake, with a general decline in overall condition.
Auditory Brain Stem Response
Auditory brain stem responses were within expected ranges for guinea pigs 2 weeks after surgical implantation of the catheter. Following twice-weekly saline administration at weeks 3 and 4, a slight increase in ABR was observed at 6 or 9 weeks postsurgery. The increase in threshold was up to +15 to +30 dB at 9 weeks after surgery.
In animals that received gentamicin, the ABR response was absent or measurable only with very loud stimuli at 6 weeks postimplantation (4 weeks after the start of dosing) and correlated with severe hair cell loss observed microscopically.
Histopathology
In all the temporal bones examined, there was either no or minimal inflammation in the middle ear (Figure 2A). All temporal bone catheterization sites showed normal healing, without excessive inflammation or granulation tissue (Figure 2B). A few macrophage aggregates occasionally with rare neutrophils were observed in the tympanic cavity, near the ossicles and oval window, of a single middle ear (Figure 2C and D). Hemorrhage, fibrosis, or otosclerosis were not seen in any of the ears examined. In all saline control animals, cochleae showed normal morphology, and the integrity of the organ of Corti, including hair cells, was demonstrated (Figure 2E). In contrast, in animals administered gentamicin, the organ of Corti showed severe microscopic changes characterized by hair cell and support cell loss and replacement by scar-like tissue, interpreted to be secondary to gentamicin-induced necrosis (Figure 2F).
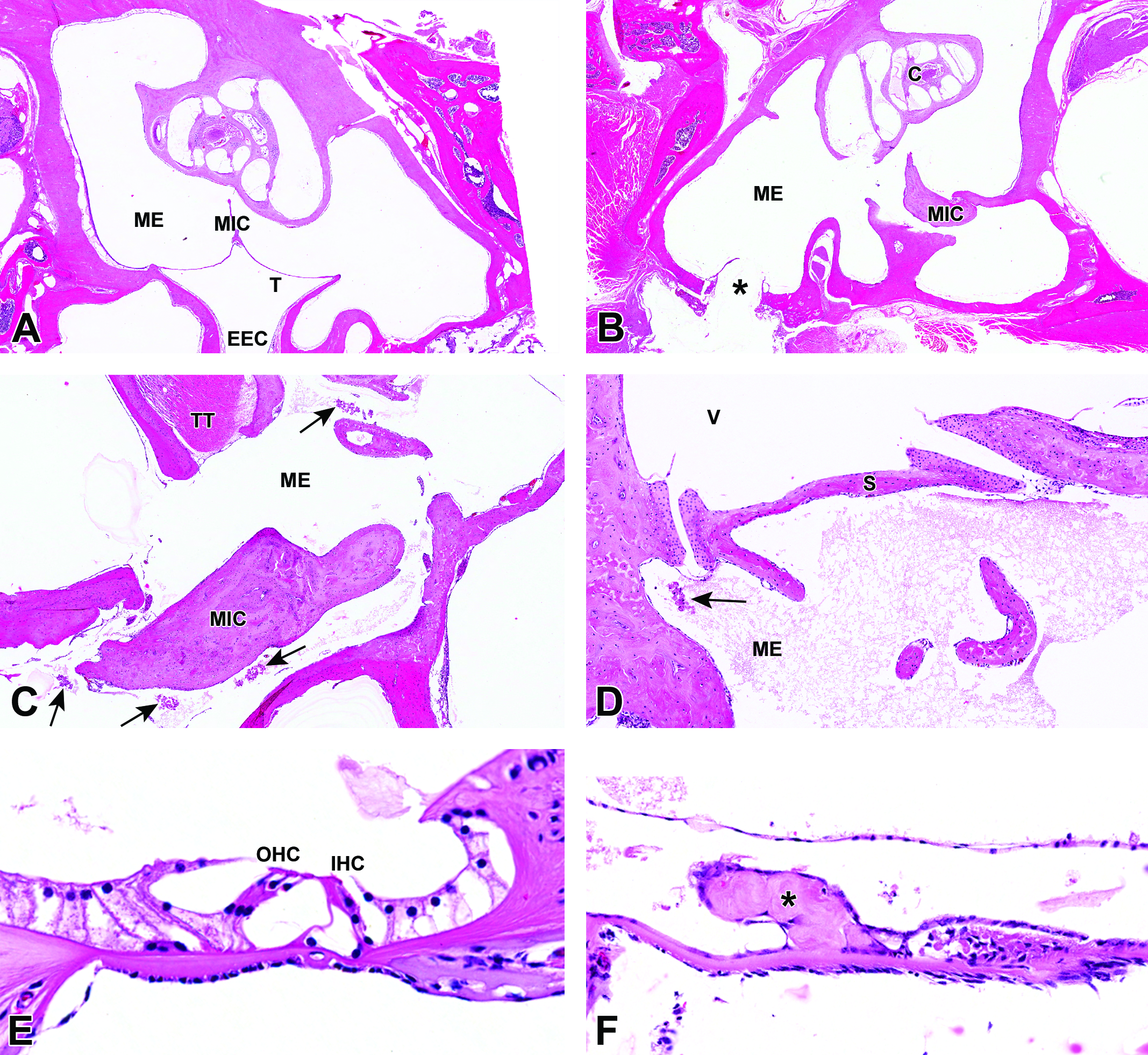
Histopathological evaluation of catheterized middle ears shows no significant inflammation in middle ear (A) and normal healing of temporal bone catheterization site (B). Minimal macrophage aggregates with few neutrophils were noted in a single middle ear (C and D). Normal organ of Corti in a saline-administered ear (E) compared to severe hair cell and support cell loss secondary to necrosis in a gentamicin-administered ear (F). ME = middle ear; MIC = malleoincudal complex; T = tympanic membrane; EEC = external ear canal; C = cochlea; TT = tensor tympani muscle; V = vestibule; S = stapes; OHC = outer hair cells; IHC = inner hair cells; arrows = macrophage aggregates; * = hair cell and support cell necrotic debris.
Cytocochleograms
All cytocochleograms from saline control animals yielded normal inner and outer hair cell counts, with no more than the expected loss of a few individual hair cells multifocally (Figure 3A). Cytocochleograms from the animals administered gentamicin revealed a profound loss of both inner and outer hair cells (Figure 3B).
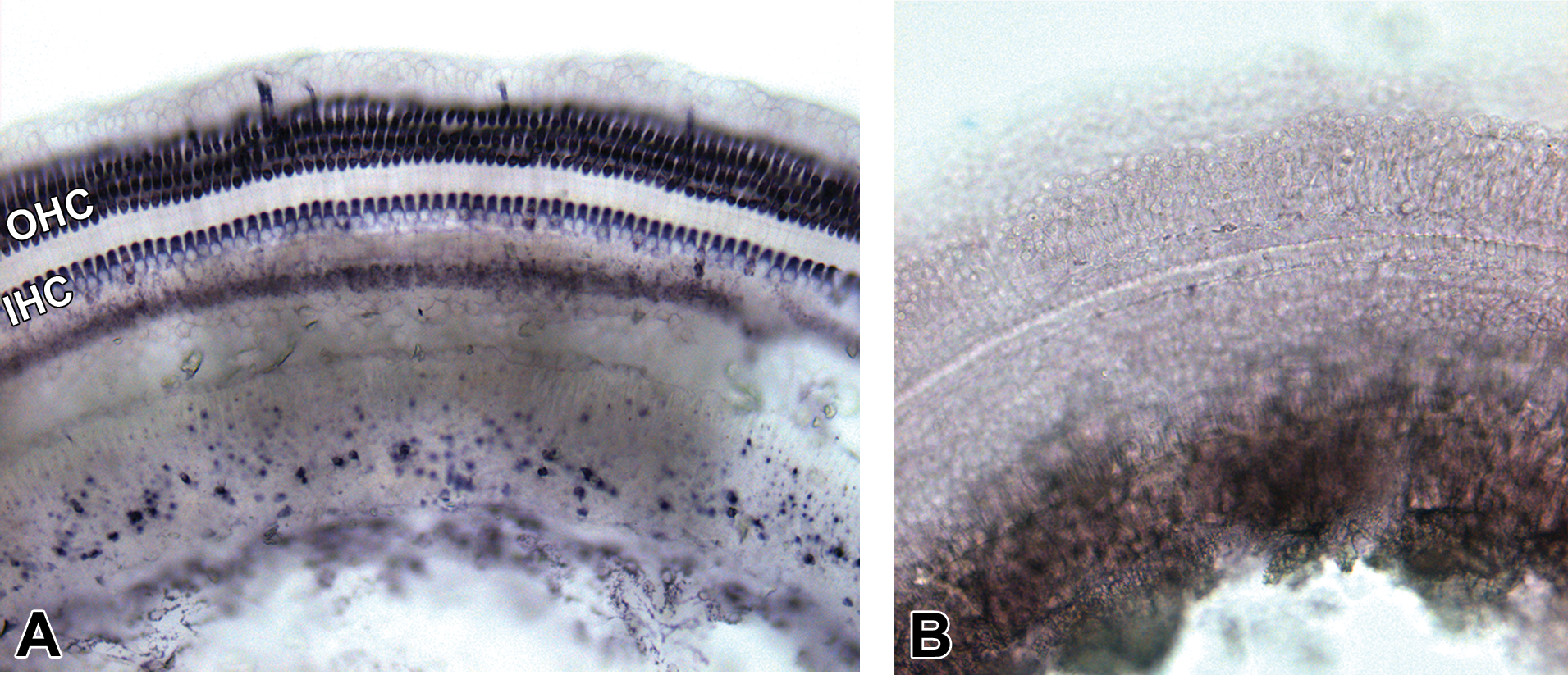
Cytocochleograms. Normal cytocochleogram from saline administered ear (A) and cytocochleogram from a gentamicin-administered ear showing diffuse, severe outer and inner hair cell loss (B). OHC = outer hair cells; IHC = inner hair cells.
Discussion
In the authors’ experience, long-term middle ear catheterization is often associated with significant morbidity and preterminal mortality which is mostly secondary to the surgical procedures and/or to the presence of the catheter in the middle ear. Microscopic changes such as inflammation, fibrosis, and otosclerosis in the middle ear are frequent complications which can be associated with hearing loss and can thus become significant confounding factors in the ABR evaluation as well as in the histopathological examination. However, successful middle ear catheterization is preferable to repeated intratympanic injections that may represent an animal welfare issue because of the repeated general anesthesia episodes required and which are associated with increased risks of middle ear infection and persistent tympanic perforation. 17,18 A similar technique would almost certainly have the same benefits in other species such as mice, rats, chinchillas, cats, dogs, sheep, or monkeys, which are all susceptible to the same complications. We chose guinea pigs in the present context because they are widely used in hearing loss and otoprotection studies and because their size (larger than mice or rats which represent a bigger surgical challenge but small enough to be easily housed, handled, and decalcified) and docile temperament make them easy to work with. 19
In the current experiment, the model was considered suitable for use in studies requiring repeated administration to the middle ear for at least 9 weeks in duration. The model ensured high animal welfare standards by permitting group housing while associated with a quick postoperative recovery and minimal changes in ABR threshold. Histopathological evaluation and cytocochleograms demonstrated that the surgical procedure and the presence of the catheter did not cause any significant deleterious changes to the middle and inner ear of saline control animals. The absence of inflammatory changes, fibrosis, and otosclerosis in the middle ear as well as the preserved integrity of the organ of Corti confirmed the long-term sustainability of this model for middle ear infusion administration. The expected hair cell loss observed in the cochleae of animals administered gentamicin confirmed successful drug administration to the middle ear with subsequent diffusion to the inner ear.
Footnotes
Acknowledgments
The authors would like to thank Elridge Edwards, Carmen Côté, Isabelle Jeanson, and Michèle Lauzon for their outstanding technical work.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
