Abstract
Among many short-term, subchronic, and chronic toxicology studies with ammonium perfluorooctanoate (PFOA), the gastrointestinal tract has not been identified as a target organ for PFOA-related toxicity in laboratory animals where the corresponding serum PFOA concentrations typically approach several orders of magnitude higher than the general human population. These lack of gastrointestinal tract-related findings were in direct contrast to an epidemiological observation where a positive trend was observed for ulcerative colitis, an idiopathic chronic inflammatory condition of the gut, in a Mid-Ohio River community whose drinking water contained higher levels of PFOA. This study was conducted to perform a histological reevaluation of large intestine sections in laboratory animals from 2 long-term toxicological studies: one was with Sprague Dawley rats that received ammonium PFOA in their diet for 2 years and the other one was with cynomolgus macaques that received daily capsules of ammonium PFOA for 6 months. In both studies, there was a lack of histological evidence of treatment-related inflammatory lesions that was suggestive of the occurrence of ulcerative colitis in these laboratory animals even under the most rigorous treatment schedules. These findings do not offer support for the biological plausibility of the epidemiological associations reported.
Introduction
For over half a century, ammonium perfluorooctanoate (PFOA) was used in specialized industrial applications such as a processing aid in the manufacturing of fluoropolymers. 1 Perfluorooctanoate (C7F15CO2 −) is the dissociated form of ammonium PFOA and it can be detected in the environmental biota, including humans, at low part-per-billion levels, and the widespread presence of PFOA led to the US Environmental Protection Agency’s 2010/2015 PFOA Stewardship Program, which resulted in the voluntary reductions of PFOA emissions and phase out of its production by 8 manufacturers in the United States (see https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/fact-sheet-20102015-pfoa-stewardship-program). Due to the strength of C–F bonds, PFOA is extremely stable and it does not undergo further metabolism. It has long serum elimination half-lives in most species once absorbed, ranging from several weeks in laboratory animals to 2 to 3 years in humans. 2 –5 Both occupational (mainly airborne) and nonoccupational exposure sources (such as diet and drinking water) have been shown to contribute to the body burden of PFOA in humans. There have been several national government-sponsored evaluations related to health and exposure to PFOA. 6 –10
In toxicological studies, serum PFOA concentrations in laboratory animals typically approach several orders of magnitude higher than the general human population. Specifically, serum PFOA was usually in the range of 10,000 ng/mL or higher in the laboratory animals where biological responses were observed and these levels were higher than the serum PFOA concentrations reported for the US general population where the geometric mean is at 1.56 ng/mL per 2015 to 2016 Centers for Disease Control and Prevention National Health and Nutrition Examination Survey data. 11 Most toxicological effects reported can be attributed to adaptive changes in the liver due to nuclear receptor activation. 12 –17 Among the numerous short-term, subchronic, and chronic toxicology studies, the gastrointestinal tract was not identified as a target organ for PFOA-related toxicity in laboratory animals. However, in a Mid-Ohio Valley community cohort study of 32,254 residents and workers (median serum PFOA was 26 ng/mL with interquartile range 13-68 ng/mL), drinking water contained elevated levels of PFOA due to industrial release from a chemical plant. 18 The mean PFOA concentration in the water, sampled during 2002 to 2005 time period, was 3.55 ng/mL (range: 1.5-7.2 ng/mL), and the corresponding analytical lower detection limit was at 0.01 ng/mL. 19 A positive trend (P < .0001) was observed for an idiopathic chronic inflammatory condition of the gut, ulcerative colitis, by quartile of modeled cumulative PFOA exposure with the cut points of 158,586, and 3,500 ng/mL/yr and the respective odds ratios, compared to reference, of 1.71 (95% confidence interval [CI]: 0.89-3.27), 2.05 (95% CI: 1.07-3.91), and 3.05 (95% CI: 1.56-5.96). A similar positive trend was also reported for the subset of workers (n = 3,713) whose median serum PFOA was 112.7 ng/mL (interquartile range: 56-256 ng/mL). 20 While significant trends between PFOA exposure and ulcerative colitis were reported, 18,20 these epidemiological observations were restricted to a single population of higher exposed individuals (than the general population) and it contrasts with the toxicological data where the gastrointestinal tract was not identified as a target organ in laboratory animals under rigorous treatment schedules up to 300 ppm of ammonium PFOA administered in the diets to rats for 2 years. 13,17 There were no clinical or macroscopic observations or microscopic evidence of ulcerative colitis in either of these 2 chronic studies (serum PFOA concentrations were not measured in either study). Similarly, there were no “ulcerative colitis-like” symptoms or histological changes reported in cynomolgus macaques when they were given ammonium PFOA for 6 months, with the highest steady state serum PFOA concentration being approximately 158,000 ng/mL. 16 It should be noted that these animal studies were conducted 2 to 3 decades ago (the 2 chronic dietary studies with PFOA in rats were conducted in 1981 to 1983 and 1990 to 1992, and the 6-month cynomolgus macaque study with PFOA was done between 1998 and 1999). We undertook this study to reevaluate the original study materials for any potential microscopic evidence of ulcerative colitis in rats and cynomolgus macaques treated for extended periods with PFOA. The histological sections of selected tissues were reexamined by an independent pathologist (not involved in the original studies) focusing on any evidence of inflammatory changes. This article presents the results from this histological review.
Methods
Archived tissues in the large intestine from a 2-year dietary study in rats 13 and a 6-month study in cynomolgus macaques (Macaca fascicularis) 16 were evaluated by a board-certified veterinary pathologist. Both studies were conducted in accordance with Good Laboratory Practices in facilities accredited by the International Association for the Accreditation of Laboratory Animal Care. All procedures involving laboratory animals were reviewed and approved by the Institutional Animal Care and Use Committee associated with the facility in which the laboratory animals were housed. Animal care and procedures followed the US Department of Health and Human Services guidelines that were available at the time of the study.
Provided below is a brief description of the experimental design for each study: For the 2-year dietary study in Sprague Dawley rats,
13
the histopathologic examination was performed on section of colon obtained from rats at the end of 2-year treatment period, from the control group (0 ppm, n = 50/sex) and the highest dietary ammonium PFOA dose group (300 ppm, equivalent to approximately 15 mg/kg/d, n = 50/sex). Only colon sections were collected from the rats. Given the dietary nature of the study design and to ensure/increase the accuracy of food consumption calculations, the animal husbandry included was hanging stainless steel cages with wire mesh floors to allow for collection of food spillage. The male rats in this study were single-housed, while female rats were group-housed (n = 2/cage). In addition, to minimize cross-contamination, rats from the control group were allocated in separate rooms from rats receiving test material. Environmental monitoring for the laboratory animal rooms was temperature and humidity controlled with a 12-hour light/dark cycle. Individual rats were uniquely identified by an ear tag along with a cage card. Certified Purina Laboratory Chow, with or without ammonium PFOA fortification (Ralston-Purina Co, St Louis, Missouri) and tap water (St Paul, Minnesota), were provided ad libitum to all rats throughout the study except when fasting was required. During the study, there were no abnormal clinical observations such as vomiting, diarrhea, or bloody stool that would suggest an ulcerative colitis–like condition in these rats. For the 6-month study with ammonium PFOA in cynomolgus macaques,
16
the histopathologic examination was performed on sections of cecum, colon, and rectum obtained from all male animals. Because there was no distinct gender difference in serum elimination of PFOA in cynomolgus macaques (which is approximately in the range of 1 month),
21
only male cynomolgus macaques were used in this study (3-9 years old, mean age was approximately 6 years). Each animal was individually housed in suspended stainless steel cages inside an animal room which was temperature and humidity controlled and was on a 12-hour light/dark cycle. Certified primate chow (Harlan Teklad # 8726C, Madison Wisconsin) was provided daily to all the animals in addition to fruit and cereal supplementation. City tap water (Madison, Wisconsin) was provided ad libitum to all animals throughout the study except when fasting was required. Animals received daily administration of gelatin capsules that were either empty (control) or containing ammonium PFOA. The given were 0 mg/kg/d (control, n = 6), 3 mg/kg/d (n = 4), 10 mg/kg/d (n = 6), and 30/20 mg/kg/d (n = 2). The 30/20 mg/kg/d dose reflected the highest dose group in the cynomolgus macaques study that received 30 mg/kg/d in the beginning of the study for 12 days with n = 6 animals. The dosing was suspended due to systemic toxicity (eg, bodyweight loss, qualitatively low food consumption, and few or no feces production), and it was resumed on study day 22 with a lower dose at 20 mg/kg/d with n = 2 animals only. During the study, there were no abnormal clinical observations such as vomiting, diarrhea, or bloody stool that would suggest an ulcerative colitis–like condition in these animals.
In both studies, all tissues were stained with hematoxylin and eosin at the time when the studies were conducted. The histological sections were examined by the current study pathologist without knowledge of the original study pathologists’ findings for each of the individual animals.
Statistics
The null hypothesis was that the proportion of microscopic observations for either Sprague Dawley rats or cynomolgus macaques was equal across the control and PFOA-dosed groups. The alternative hypothesis was that the proportion of findings was not equal. To conduct these categorical analyses, Fisher exact tests (2 tailed) were performed using SAS statistical software (version 9.4; SAS Institute, Cary North Carolina). For the rat study, when a statistically significant difference was found for a microscopic observation in the categorical analyses (P < .05), then differential white blood cell counts by sex (for leukocyte, neutrophil, and lymphocyte as 103 cells/mm3) were examined and reported as mean ± standard deviation for these animals. Student t test were calculated using Microsoft Excel (Office 365) with a statistically significant difference defined as P < .05.
Results
Histological review of large intestine sections of Sprague Dawley rats that received ammonium PFOA in their diet for 2 years or cynomolgus macaques that received daily capsules of ammonium PFOA for 6 months did not reveal any evidence of treatment-related inflammatory lesions. In both studies, the animals had spontaneous background parasitic diseases that involved the large intestines, which is common in these animal models. For the 2-year dietary study with ammonium PFOA in rats, the histopathological findings in large intestinal specimens are summarized in Table 1. The colon had typical mild lymphocytic infiltration as part of the normal mucosal immune system. The mucosal inflammatory cell population also contained a minimal population of neutrophils. The subjective description term “minimal” applies to a grade 1 histological change (1 being the least pronounced and 5 being the most pronounced). This subjective grade is determined by visual examination of the histological sections per study pathologist. The various grades are intended to provide a comparison of the relative severity of histological alterations and do not constitute a morphometric analysis. The neutrophil population in the intestinal mucosa was increased in 8 males and 4 females from the 300 ppm dose group compared to 0 males and 2 females in the control group. Comparing the control and treatment group distributions across the 4 categories, and collapsing by sex, resulted in a Fisher exact test P value = .002. Restricting the analysis to a comparison of only the neutrophilic infiltration and the essentially normal categories resulted in a Fisher exact test P value = .02. While the incidence of minimal neutrophilic infiltrates was statistically significantly elevated in both analyses, there was no histological incidence of overt inflammation (ie, infiltration by inflammatory cells such as leukocytes with the additional feature of tissue destruction and/or tissue reaction) in the large intestines from these ammonium PFOA-treated rats (Table 1).
Microscopic Observations of Colon Tissues for Male and Female Sprague Dawley Rats that Received o ppm or 300 ppm Ammonium PFOA in Their Diet for 2 Years.
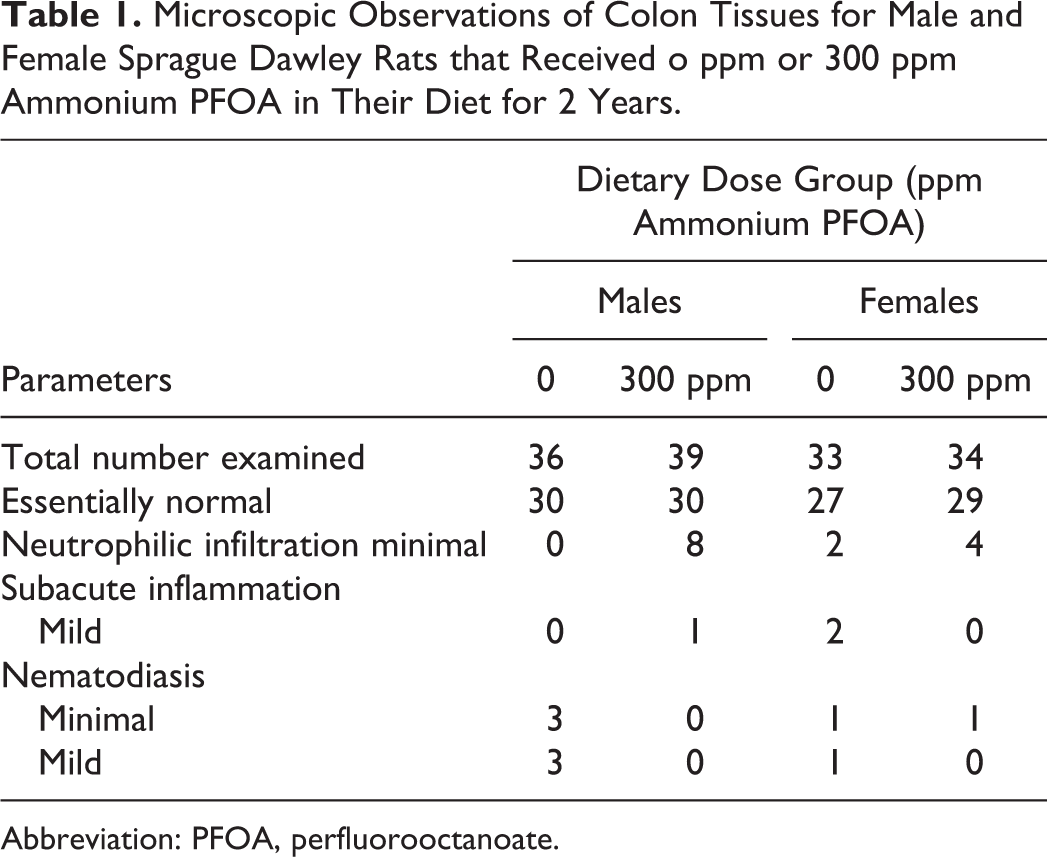
Abbreviation: PFOA, perfluorooctanoate.
Further support for an absence of treatment-related inflammation include (1) lack of histological evidence for chronic inflammation in the colonic sections of these rats (ie, absence of macrophage infiltration or fibrous lesions) and (2) normal hematological leukocyte counts for neutrophils and lymphocytes that were measured during the study (either at interim or at the end of the 2-year period). In male rats receiving control diet (0 ppm), the overall mean ± standard deviation for leukocyte, neutrophil, and lymphocyte counts were 10.8 ± 5.1, 2.6 ± 4.1, and 7.7 ± 2.6 × 103 cells/mm3, respectively. 13 In the eight 300 ppm dose group male rats that had mild neutrophilic infiltration, their mean ± standard deviation for leukocyte, neutrophil, and lymphocyte counts were 10.9 ± 2.0, 2.2 ± 1.0, and 8.0 ± 2.7 × 103 cells/mm3, respectively. These mean values were not statistically significantly different than the overall mean for the male control group. In female rats receiving control diet (0 ppm), the overall mean ± standard deviation for leukocyte, neutrophil, and lymphocyte counts were 9.0 ± 5.8, 1.9 ± 2.0, and 6.8 ± 4.9 × 103 cells/mm3, respectively. 13 In the four 300 ppm dose group female rats that had mild neutrophilic infiltration, their mean ± standard deviation for leukocyte, neutrophil, and lymphocyte counts were 5.6 ± 0.9, 1.7 ± 0.5, and 3.6 ± 0.7 × 103 cells/mm3, respectively. These mean values were not statistically significantly different than the overall mean for the female control group. For comparison purpose, the historic control normal ranges reported by the testing facility at the time when the study was conducted were 6 to 15 × 103 cells/mm3 for leukocytes, 0.5 to 4 × 103 cells/mm3 for neutrophils, and 5 to 14 × 103 cells/mm3 for lymphocytes.
In this study, there was Syphacia spp. (pinworm) infestation in the colon of a number of rats (6 control males, 2 control females, and 1 female from the 300 ppm dose group), but there were no definitive histological changes in the intestinal tissue associated with the presence of parasites. While the nematode parasite was prevalent in the laboratory rat colonies in the early 1970s and 1980s and parasitism was considered to be of no clinical significance, 22 it is unknown whether dietary ammonium PFOA treatment had an effect that could have resulted in the observation of parasitism in only 1 PFOA-treated rat, as compared to 8 control rats, in this particular study. It should be noted that the use of parasite-free laboratory animals was not a common practice during that time period. More recent information suggests pinworm infestation may be associated with increased cytokine production and other alterations that indicate some degree of immunomodulation and intestinal functional alteration associated with the parasitism. 23 –26
For the 6-month study with ammonium PFOA in cynomolgus macaques, the histopathological findings in large intestinal specimens are summarized in Table 2. The large intestinal mucosa commonly had a mild infiltration of lymphocytes and plasma cells with a few eosinophils and neutrophils. This inflammatory cell population was considered to be normal background histomorphology. Furthermore, there were no increases in the white blood cell populations (neutrophils, eosinophils, basophils, lymphocytes, and monocytes) in these animals. 16 Comparisons of the number of histologic evaluations that were essentially normal to the 2 combined “non-normal” categories for cecum and the 3 combined “non-normal” categories for colon across the 4 dose groups were not statistically significant (Fisher exact test, P = 1.0 for cecum, P = .67 for colon; Table 2). For the rectum, all animals were considered essentially normal for control and ammonium PFOA treatment groups. There were granulomatous lesions consistent with Oesophagostomum spp. infestation of the large intestine in 1 control animal and 1 ammonium PFOA-treated animal at 10 mg/kg/d. This nematode parasite was also common in the intestinal tract of nonhuman primates at the time the study was conducted. The large intestine specimens had no histological evidence of inflammation other than that directly associated with the parasitic granulomas.
Microscopic Observations of Cecum, Colon, and Rectum Tissues for Male Cynomolgus Monkeys that Received a Daily Oral Dose of Ammonium PFOA at Either 0, 3, 10, or 30/20 mg/kg/d for 6 Months.
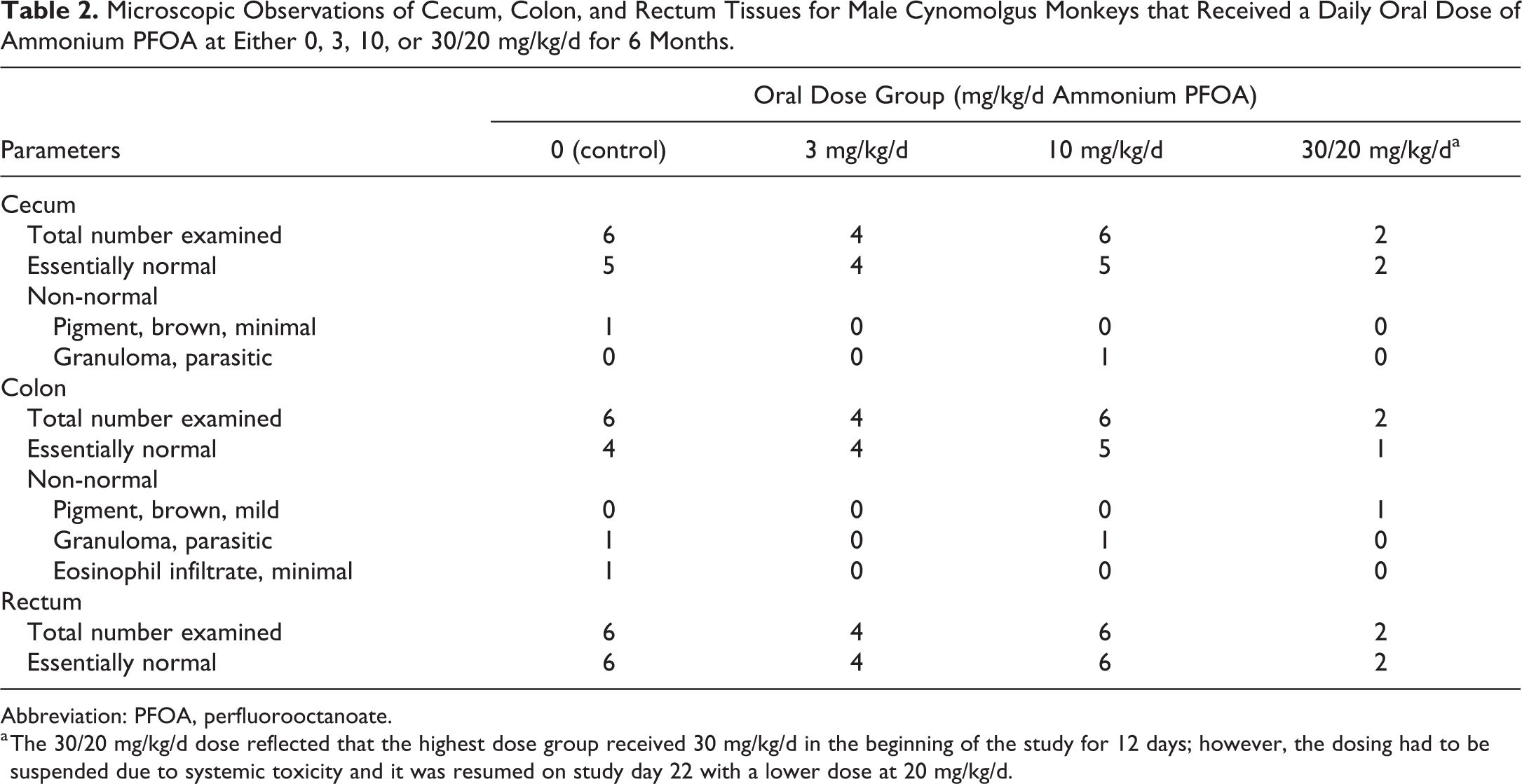
Abbreviation: PFOA, perfluorooctanoate.
a The 30/20 mg/kg/d dose reflected that the highest dose group received 30 mg/kg/d in the beginning of the study for 12 days; however, the dosing had to be suspended due to systemic toxicity and it was resumed on study day 22 with a lower dose at 20 mg/kg/d.
Discussion
This study used current pathology criteria to examine large intestine tissues from 2 published studies in rats and cynomolgus macaques for microscopic evidence of ulcerative colitis. This limited pathological review was conducted independently of the original study findings, and it was confirmed that there was no microscopic evidence of ulcerative colitis in either rats or cynomolgus macaques that received ammonium PFOA administration for an extended period of time for 2 years and 6 months, respectively. The conclusion from this histological review is consistent with the original study reports, which did not observe PFOA treatment-related abnormalities in the large intestines nor any PFOA treatment-related abnormalities in the other gastrointestinal tract tissues including esophagus, stomach, and small intestine (duodenum, jejunum, and ileum). Furthermore, it is worth noting that there were no abnormal histological findings in the key immune organs such as spleen, bone marrow, and lymph nodes in the original study reports. 13,16 Also, in the 2-year dietary bioassay study with ammonium PFOA in rats, there were no colorectal tumors nor intestinal tumors observed. These observations are important because research studies have suggested that the development of ulcerative colitis can be attributed to immune-related changes 27,28 as well as a possible association with colorectal tumors. 29 While the limited histological examination of immune organs does not fully constitute and represent the functional aspects of immune systems, the collective empirical evidence does not support a treatment-related effect of PFOA on the large bowel or the occurrence of ulcerative colitis in laboratory animals, which contrasts with some but not all epidemiological findings.
This retrospective review of large intestine specimens from rats and cynomolgus monkeys confirms the original study results that revealed no PFOA-associated inflammatory lesions that would suggest an association with ulcerative colitis in these species. However, these studies were general toxicology studies that were not designed to be an exhaustive investigation into the possible relationship between PFOA and ulcerative colitis. As is typical for general toxicology studies, only representative samples of the large intestine were subjected to histopathological analysis; thus, the specimen sampling may not have revealed multifocally distributed inflammatory lesions. In addition, the histopathological interpretation in the nonhuman primate study was complicated to some extent by the presence of inflammatory lesions associated with large intestinal parasites, which were commonly present in nonhuman primates at the time these studies were conducted. As a result, the absence of PFOA-associated intestinal inflammation in these 2 studies is not considered to be definitive but is one facet in the overall investigation of possible PFOA involvement in the pathogenesis of ulcerative colitis.
There are many small animal research models available for studying ulcerative colitis conditions and pathology, and the development of intestinal inflammation can be chemically induced, or via genetic modifications, to mimic the clinical appearance of ulcerative colitis observed in humans, under either acute or gradual regimes. 30 –34 Consequent to the frequent diarrheic episodes, the animals exhibit visible signs such as soft and/or bloody stools, rectal bleeding, and body weight loss. Macroscopically, intestinal tissue walls may present a loss of translucency (discoloration) and often colon segments are shortened with bleeding (Figure 1). In rodents, spleen weights may increase as a result of inflammation (Figure 1). Microscopically, loss of intestinal tissue integrity can be visualized and typically includes inflammatory infiltration into mucosa, submucosa, or muscularis externa and serosa. 30 –34 In laboratory rodents, the development of induction of ulcerative colitis–like conditions can be induced by chemical administration either acutely (within a few days) or chronically, depending on the dose or exposure duration. If PFOA can elicit ulcerative colitis, one would expect to observe this condition at the end of a 2-year exposure period, this was not the case (as discussed above).
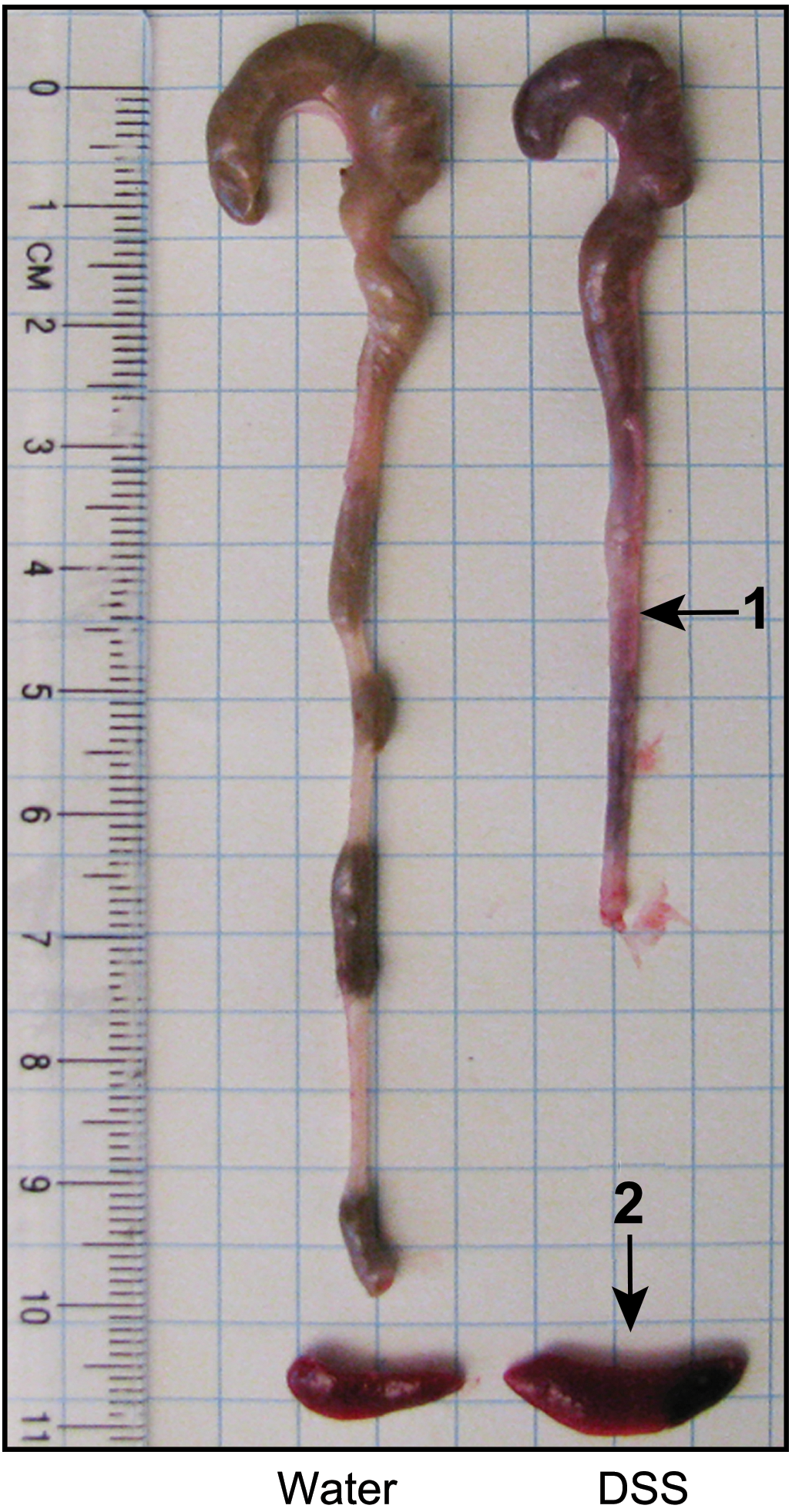
Macroscopic comparison of colon from 8-week C57BL/6 mice given either water or 2.5% dextran sulfate sodium (DSS, ulcerative colitis inducer) in drinking water for 7 days. Necropsy was performed on day 8. Arrows indicate: (1) shortened and bleeding colons and (2) enlarged spleen. Source: From Chassaing et al. 33 Image courtesy of Dr Benoit Chassaing, Georgia State University with reprint permission from John Wiley & Sons, Inc.
In humans, the pathological condition of ulcerative colitis is a form of inflammatory bowel disease (IBD) and together with Crohn disease, both diseases can result in inflammation and ulceration in the gastrointestinal tracts. 35 While Crohn disease most commonly appears as patches and affects the entire thickness of the intestinal wall in the upper bowel (small intestine), the pattern of inflammation in ulcerative colitis is more widespread and it affects the innermost lining (mucosa) of the lower bowel region (colon and rectum). In humans, clinical symptoms of ulcerative colitis do not occur suddenly; they tend to develop gradually over time and may include diarrhea, rectal bleeding, bloody stool, and anemia (as a result of blood loss). Even though the exact cause of ulcerative colitis in humans is unknown, infections, diet, environment, host microbiome populations, genetic polymorphisms, and compromised immune responses all have been thought to play a role in causing ulcerative colitis. 27,28,36
In epidemiological studies, the association between PFOA exposure and ulcerative colitis was studied in a community-based cohort of individuals that lived or worked in a region with elevated PFOA levels in drinking water. 18,20 Estimated PFOA exposure for each individual in the cohort was modeled based on the estimated intake of drinking water, and, when applicable, occupational PFOA exposure. Cases of self-reported autoimmune disease were validated through medical record review. In this regard, of the 596 self-reported ulcerative colitis cases, only 151 (25%) were medically validated. In the analysis of the combined cohort of community residents (n = 28,541) and workers (n = 3713), Steenland et al 18 reported an increased risk of medically validated ulcerative colitis by quartile of estimated PFOA exposure with adjusted rate ratios of 1.00 (referent), 1.76 (95% CI: 1.04-2.99), 2.63 (95% CI: 1.56-4.43), and 2.86 (95% CI: 1.65-4.96; P trend < .0001). In a subsequent analysis of only the workers in this combined cohort, Steenland et al 20 also reported significant positive trends (P = .05) between ulcerative colitis (28 cases) and quartile of estimated PFOA exposure in both unlagged and 10-year lagged analyses with the individual quartile estimates lacking precision. While positive trends were observed in both the combined cohort and the worker cohort, all 28 cases of ulcerative colitis in the worker cohort were included among the 151 cases in the combined cohort. As such, it is possible that the observed association in the community cohort was driven, at least in part, by workers with much higher exposure. An analysis of ulcerative colitis of the community alone in this cohort study was not reported as had been done for cancer. 37
Another important issue is the potential misclassification of ulcerative colitis cases. In the combined cohort, 151 cases of ulcerative colitis and 95 cases of Crohn disease were validated through medical record review. 18 Of these, 10 cases were validated for both ulcerative colitis and Crohn disease. There is some lack of clarity regarding whether the 10 cases were excluded or were included in the analyses for both diseases. Although not explicitly stated, these cases likely presented with indistinguishable features of both ulcerative colitis and Crohn disease and were diagnosed with indeterminate colitis. While most cases of indeterminate colitis are eventually classified definitively as ulcerative colitis or Crohn disease, 38 it is not known if the authors looked for any subsequent changes in diagnoses among the 10 cases.
Steenland et al 18 also suggested that the prevalence of ulcerative colitis in the study population (410 per 100,000 adults) may be in excess compared with a US general population prevalence of 263 per 100,000 adults, as reported by Kappelman 39 ; however, the prevalence of ulcerative colitis reported by Steenland et al may not reflect the true prevalence of ulcerative colitis in the community given that fewer than 50% of the eligible community participated in the combined study. It is possible that a higher proportion of individuals with known health conditions self-selected into Steenland et al’s study compared to those without any health conditions, thereby artificially inflating the true prevalence of ulcerative colitis. The prevalence rates of Crohn disease and other autoimmune disorders observed in this study by Steenland et al were also higher than prevalence rates reported in the literature, 40 which lends some support to this speculation. Further, ulcerative colitis is a well-established risk factor for the development of colorectal cancer, and in population-based cohorts, the risk of colorectal cancer in ulcerative colitis patients increases 2.4-fold. 41 However, it should be noted that in a cross-sectional study of this community, the likelihood of colorectal cancer among individuals in the highest PFOA quartile had a 40% reduction in colorectal cancer risk. 42
In a different community with general population level exposure to PFOA, Steenland et al 43 measured serum PFOA concentrations from archived serum samples obtained from primarily young white patients of ulcerative colitis and Crohn disease (serum samples collected spanning from 2004/2005 to 2012/2013 period with a mean collection year of 2008). These were patients from the Emory Health Care campus (Atlanta, Georgia). Interestingly, these data were compared to controls who were primarily non-Hispanic black, and their serum samples were collected from 2010 to 2013 with a mean collection year of 2011. Their respective arithmetic mean and median serum PFOA concentrations were 3.76 and 2.93 ng/mL for patients with ulcerative colitis, 3.63 and 1.78 ng/mL for patients with Crohn disease, and 2.46 and 1.33 ng/mL for controls. Because geometric means were not provided by the study authors, the median value is a good approximation to the geometric mean for the purpose of comparison: The geometric mean of serum PFOA for the general population in the United States was 4.12 ng/mL in 2007 to 2008 period (95% CI: 4.01-4.24), and in 2011 to 2012 period, the geometric mean was 2.08 ng/mL (95% CI: 1.95-2.22). 11 Logistic regression analyses by quintiles for ulcerative colitis versus controls/Crohn disease in relation to PFOA resulted in highly imprecise and nonmonotonic odds ratios, reference (1.00), 0.81 (95% CI: 0.22-2.93), 40.98 (95% CI: 11.67-150.34), 33.36 (95% CI: 11.32-119.36), 2.86 (95% CI: 0.94-8.75). Interpretation is difficult for several reasons: (1) measurement of the samples for PFOA was conducted well past the diagnosis of ulcerative colitis; (2) there was no information whether treatment, not just the disease, may have influenced the measurement of PFOA; (3) there was no mode of action suggested for such a small absolute difference in the PFOA measurements; and (4) statistical adjustment was not possible as the control samples were almost exclusively non-Hispanic blacks who, based on NHANES data, are known to consistently have had lower time-dependent geometric mean PFOA concentrations than whites. 11
In a different community in Sweden, approximately one-third of the population in Ronneby region was exposed to municipal drinking water that contained perfluroalkyls and polyperfluoroalkyls whose source was aqueous film-forming foam (AFFF) that had been used at a nearby military airbase between 1985 and 2013. 44 The cohort was identified as all Ronneby residents (n = 63,073) whose residence in Ronneby was for at least 1 year between 1980 and 2013, with the time period 1980 to 1984 considered the nonexposed period. During the study time period (1980-2013), 296 patients with ulcerative colitis were identified through the Swedish health registry system: 238 cases were considered never exposed, 20 cases were exposed in the early time period (1985-1994), 24 cases were exposed in the mid-exposure time period (1995-2004), and 14 cases in the late time period (2005-2013) whose exposure was considered the highest. The median serum PFOA level was 15 ng/mL, and the median serum perfluorohexanesufonate (PFHxS) and perfluorooctanesulfonate (PFOS) concentrations were 257 and 280 ng/mL, respectively. 45 The median PFOA concentration is less than the community residents from C8 Science Panel analyses but greater than the patients with ulcerative colitis from Emory University Healthcare study by Steenland et al. 43 There was no evidence of an increased trend of hazard ratios (HRs) for the diagnosis (morbidity or mortality) of ulcerative colitis during these time periods of exposure: not exposed (reference = 1.0), early exposure HR = 0.88 (95% CI: 0.55-1.4), mid exposure HR = 0.80 (95% CI: 0.56-1.32), and late exposure HR = 0.73 (95% CI: 0.42-1.28). Nor was there evidence for an increased risk for Crohn disease except in the early time period. To further investigate this exposed population, Xu et al 44 measured fecal calprotectin and fecal zonulin (biomarkers of intestinal inflammation and permeability, respectively) in a small subset of the Ronneby population as well as a nearby community. There was a negative trend of calprotectin with the combined increased exposure to PFOA, PFOS, and PFHxS, which indicated a lower degree of gut inflammation among the highly exposed. There was no association between fecal zonulin and exposure to the sum of these 3 perfluoroalkyls. Xu et al concluded that their findings provided no evidence of an association between exposure to AFFF and risk of IBD (Crohn disease or ulcerative colitis).
The association that has been reported in the 3 existing epidemiological studies as discussed previously by Steenland et al 18,20,43 might insinuate a need for direct studies of PFOA on ulcerative colitis in laboratory animals to better inform the biological plausibility. However, our reevaluation of 2 previously published laboratory studies did not show a treatment-related effect of PFOA suggestive of ulcerative colitis in laboratory Sprague Dawley rats or cynomolgus macaques and therefore does not support the biological plausibility of the epidemiological associations previously mentioned. 18,20,43 To date, the exact cause(s) of ulcerative colitis in humans remains recondite, and factors such as diet, genetics, gut microbiome, environment, and immune functions have been suggested to possibly influence the initiation and the development of this complicated pathological condition. 28 There are also studies suggesting associations between the prevalence of ulcerative colitis and colorectal tumors in humans. 29 The genesis of our study reported herein was retrospective in nature and applied only to selected tissue pathology rereview of a subchronic study in cynomolgus macaques and a chronic study in rats. A major limitation of the current study is related to the samples available for examination from the 2-year dietary study in rats. Specifically, only colon samples were collected from that study and the possibility of a lesion present in other parts of the large intestine (ie, cecum or rectum) cannot be completely ruled out. Histological sections of cecum, colon, and rectum samples were collected from the 6-month study with cynomolgus macaques. In addition, given that both studies revealed helminthic infection in the large intestine, which has been suggested to have beneficial effects on the development of ulcerative colitis, 34 this study was not able to determine if it played a protective role in these animals. Another limitation of this study encompasses the possible influencing factors mentioned above (eg, diet, genetics, gut microbiome, environment, and immune functions) that cannot be addressed by the current study design. The immune function is of particular research interest because there has been increased attention surrounding this topic.
Some studies have reported changes in immunoglobulin (Ig) concentrations in mice after (mostly) high-dose ammonium PFOA treatment; however, the observation was primary limited to decreased IgM with either no change in IgG or sometimes increased IgG or IgE. 46 –49 While the finding of PFOA at high doses suppresses the IgM response can be viewed as suggestive of immunosuppression, in reality, the absence of concomitant decreases in other Ig isotypes (eg, IgG and IgE) does not support such a conclusion. As a comparison, treatment of mice with a known immunosuppressant, cyclophosphamide, results in robust suppression of both IgM and IgG responses. 50 The observation with PFOA at high doses may also reflect immune system modulation as a result of significant weight loss observed at high PFOA concentrations and possibly accompanying increases in corticosteroids, which could lead to reduced B cell numbers in response to body weight loss. It is also possible that there were altered B cell class switch such that less IgM and more IgG or IgE are secreted. None of the published studies specifically examined these factors. In addition, recent advancement in the IBD research has suggested a more defined pattern of T helper type 2–mediated changes of the releases of several cytokines (eg, increased secretion of interleukin 5 [IL-5] and IL-13 and decreased secretion of IL-4) when colonic tissues were isolated from mice that had been induced to develop ulcerative colitis. 27,51 Even though no studies have evaluated the cytokine release profiles from the colonic tissues in PFOA-treated animals, a few studies have evaluated the effects of PFOA in vitro on intracellular cytokine pathways. Singh et al 52 suggested that PFOA induced p38, caspase activation, and nuclear factor kappa B (NF-κB) in a dose-dependent manner in human mast cells. However, these doses induced cell death (as indicated by caspase activation) and they were in contrast with the results reported by Corsini et al, 53 which showed that PFOA suppresses NF-κB activation in human promyelocytic cell line THP-1. Therefore, the effects of PFOA on cytokine production remain inconsistent and clear conclusions cannot be drawn.
Within the limit of the study design, our present data did not ratify a treatment-related effect of PFOA on the large bowel or the occurrence of ulcerative colitis in either cynomolgus macaques or rats. There might be some possible noncausal biological explanations for the positive association observed between PFOA and prevalence of ulcerative colitis reported in epidemiological studies, including: Displacement of PFOA by the therapeutic drugs prescribed for the treatment of ulcerative colitis: In clinical medicine, a standard therapy for ulcerative colitis is 5-aminosalicylate-based drugs such as sulfasalazine, balsalazine, olsalazine, or mesalazine.
54
–56
5-Aminosalicylates can attenuate the inflammation and cell proliferation of the affected tissues, in part, by activating peroxisome proliferator-activating receptor gamma (PPARγ) which initiates a cascade of negative feedback signals on the responsive cells to suppress the production of chemokines and cytokines.
55
Peroxisome proliferator-activating receptor gamma is a xenosensor nuclear receptor and it is highly expressed in the colonic epithelial cells and adipose tissues, as well as kidney, small intestine, liver, macrophages, and in T and B cells of humans and rodents.
57,58
There are many naturally occurring endogenous ligands for PPARγ such as fatty acids and cyclopentenone prostaglandins, and gut microbiome has also been suggested to enhance its activity.
31,36
Other PPARγ-activating agents include antidiabetic agents such as rosiglitazone, a drug commonly used to treat type II diabetes.
59
Mechanistic studies have shown that 5-aminosalicylates can compete with rosiglitazone for binding sites (on the PPARγ), and at therapeutic concentration, rosiglitazone can be displaced from their sites.
55,56
While the medication history in the cohort studied by Steenland et al
18,20
is unknown, it is possible that people diagnosed with ulcerative colitis were under standard medical intervention such as 5-aminosalicylate for therapeutic purpose. Given that PFOA is highly bound to serum proteins
60
and it also is a weak activator of PPARγ,
61
it is possible that ubiquitously bound PFOA can be displaced in the presence of 5-aminosalicylates. The displacement of PFOA by 5-aminosalicylates can then possibly result in a slightly higher serum PFOA level in patients with ulcerative colitis. Increased retention of PFOA with altered organic anion transporter functions: Efflux organic anion transporter proteins play a critical role in the intestinal barrier functions and it has been shown that 2 efflux transporters (ABCG2 and ABCB1) have decreased expressions from the colonic and rectal biopsy samples obtained from patients with ulcerative colitis.
62
It also has been shown the uptake of 5-aminosalicylate (drug administered to patients with ulcerative colitis) is mediated, in part, by human intestinal and hepatic OATP1B1, OATP1B3, and OATP2B1 in vitro.
54
Interestingly, given that urinary excretion is the primary pathway of eliminating PFOA from the body, an active area of research in the past few years has been on the evaluation of renal organic anion transporters and polypeptides and their roles in the elimination kinetics of PFOA once absorbed.
63
–66
Studies have demonstrated that PFOA is a substrate of these same transporters identified for 5-aminosalicylate described above
66
; therefore, it is possible that PFOA potentially can compete with similar substrates for trafficking across cells. In the case where the subjects having altered transporter expression due to ulcerative colitis condition, it may result in increased retention of PFOA in the blood due to competition. While it is possible that PFOA can be a substrate for other transporters, more studies are needed to verify these hypotheses.
In addition to these possible noncausal explanations, an issue of reverse causality where the disease or treatment might affect serum PFOA concentrations (as discussed above) would not be germane with the community study data reported by Steenland et al 18,20 where serum PFOA concentrations were based on exposure model and not from actual serum measurements. However, the possibility of reverse causation would be a potential limitation in the data reported by Steenland et al 43 where these investigators relied on serum measurement and not an exposure model for PFOA. On that note, even though the 2-year rat study data by Butenhoff et al 13 reported only nominal dietary doses (30 and 300 ppm) and serum PFOA was not measured, the internal body dosimetry in these rats should be quite appreciable based on a 13-week dietary study reported by Perkins et al. 67 In that study, dietary dose-dependent increases in serum PFOA concentrations were observed in male Sprague Dawley rats. At the end of the 13-week treatment period, the mean serum PFOA concentrations were 7,100 ± 1,200 ng/mL, 41,000 ± 13,000 ng/mL, 70,000 ± 16,000 ng/mL, and 138,000 ± 34,000 ng/mL for the 1, 10, 30, and 100 ppm dose groups, respectively. Therefore, the lack of serum PFOA information from the 2-year study in rats by Butenhoff et al 13 does not limit the pathological data interpretation reported herein.
In conclusion, review of histological sections of the large intestine from 2 chronic studies in rats and cynomolgus macaques exposed to PFOA did not show evidence of a treatment-related inflammatory response suggestive of ulcerative colitis. These findings do not support the observed epidemiological associations 18,20,43 that have been reported between PFOA and ulcerative colitis.
Footnotes
Acknowledgments
The authors would like to acknowledge Kara Andres (3M Company) for her expert support in the statistical analyses.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sue Chang, Sarah E. Kleinschmidt, Geary W. Olsen, Carol A. Ley, and Oyebode A. Taiwo are employees or former employees of the 3M Company. George Parker is an employee of Charles River Laboratories that was contracted by 3M Company to conduct the histopathology review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by 3M Company, former manufacturer of PFOA and related materials.
