Abstract
A3, generated as a monoclonal antibody against rat malignant fibrous histiocytoma cells, recognizes somatic stem cells in rats. We analyzed the distribution of A3-positive cells in dextran sulfate sodium (DSS)–induced colonic lesions consisting of regenerating mucosa and fibrosis. Male 6-week-old F344 rats were administered 5% DSS in drinking water for 5 to 7 days, and lesions at recovery stage were also examined. In untreated control adult colons, A3-positive cells are localized around the crypts where stem cell niche is formed. Histopathologically, in colons of DSS-administered rats, mucosal atrophy, inflammatory cell infiltration, and fibrosis were observed in the lamina propria; thereafter, mucosal epithelia were desquamated, and crypts were decreased gradually with decrease in surrounding A3-positive cells. At the early recovery stage, crypts showed regeneration with reappearance of A3-positive cells. Interestingly, A3-positive cells aggregated in desquamated mucosa surface of fibrosis. Aggregated A3-positive cells coexpressed with vimentin, Thy-1, and partly CK19 but did not react simultaneously with α-SMA. Likely, aggregated A3-positive cells may be rescue cells with nature of both mesenchymal and epithelial cells to maintain self-renewal after injury in the colon. A3 antibody would become a useful tool to investigate the participation of stem cells in rat colonic lesions.
Introduction
Newly developed monoclonal antibody may come to be an indispensable tool for understanding the kinetics of cells of specific population. We generated an antibody, named A3, by using a tumor cell line derived from rat malignant fibrous histiocytoma (MFH) as the antigen; MFH is considered to be derived from immature mesenchymal cells. 1,2 Interestingly, it was found that A3 labels stem cells in the bone marrow and hair follicle as well as immature mesenchymal cells (such as pericytes); these cells are regarded as somatic stem cells. 2,3 Recently, the epitope of A3 is regarded as the sugar chain (unpublished data); particularly, the N-linked sugar chain may be related to differentiation of embryonic stem cells and inducible pluripotential. 4 In adult rat colon, A3-positive cells participate in the intestinal stem cell niche as immature mesenchymal cells that are different from myofibroblasts around the crypts; more interestingly, some epithelial cells in the crypts expressed the A3 antigen. 4 Therefore, the further properties of A3-positive cells, which may take part in the colonic lesion development/regeneration, were investigated by using a rat model of inflammatory bowel diseases (IBDs), induced by dextran sulfate sodium (DSS). The DSS treatment can induce inflammation in rat colons, followed by ulceration and fibrosis. 5,6
Human IBDs, including ulcerative colitis and Crohn disease, are characterized by mucosal inflammation/ulcer and subsequent fibrosis. 7 The intestinal stem cell niche including myofibroblasts may play important roles in the development of these lesions. 8 The properties of intestinal stem cells and myofibroblasts have not yet been fully investigated. Myofibroblasts, appearing in rat cutaneous fibrosis, could be supplied from somatic stem cells such as pericytes reacting to A3. 9 In the present study, we investigated the distribution of A3-positive cells and the relationship between A3-positive cells and myofibroblasts in DSS-induced rat colonic lesions. In addition, it was shown that A3 labels certain epithelial cells of developing hair follicles, 3 and A3-positive cells participate in re-epithelialization of the epidermis during cutaneous wound healing (unpublished data). Therefore, this study also focused on the appearance of A3-positive cells in ulcer areas of DSS-induced colonic lesions. The present study showed that somatic stem cells, recognized by A3, can be closely related to the regeneration of colonic mucosa as rescue cells keeping the barrier from external environment under progress of re-epithelialization after injury. The A3 antibody would be useful to investigate the pathogenesis of chemically induced rat colonic lesions in the toxicologic study.
Materials and Methods
Dextran Sulfate Sodium–Induced Rat Colon Lesions
F344/DuCrj male rats, aged 6 weeks, were obtained from Charles River Laboratory (Yokohama, Japan). Rats were maintained in a room at 22°C ± 3°C with a 12-hour light–dark cycle, fed a standard diet (DC-8; CLEA Japan, Tokyo, Japan), and supplied with tap water ad libitum. After 1-week acclimatization, rats were allocated to 3 groups in order to obtain colon samples of early injury/inflammation, fibrosis, and regeneration (recovery process); they were divided into the control, DSS-treated, and recovery groups. To analyze the different colon lesions including the early injury, advanced injury, early recovery, and healing stages, rats in the DSS-treated and recovery groups were administered with 5% DSS (average molecular weight 36,000-50,000; MP Biomedicals, Irvine, California) in drinking water. The colon was sampled from each rat. Rats in the control group (n = 3) were given tap water and killed on day 10 after the starting of administration. Rats in the DSS-treated group were administered DSS until each examination point on days 3, 5, and 7 (n = 3/each point) after starting the DSS administration. Rats in the recovery group were killed on days 1, 3, and 5 (n = 3/each point) after the cessation of 5-day DSS administration, by replacing the DSS with tap water. The animal experiments were conducted following the institutional guidelines and were approved by the ethical committee of Osaka Prefecture University for the Care and Use of Experimental Animals (No.27-103).
Tissue Preparation and Histology
The “Swiss roll” samples 10 were applied to colon samples for the most part. Then, these samples were fixed in periodate-lysine-paraformaldehyde (PLP) solution for 6 hours at 4°C, and the rests were frozen immediately in the Tissu Mount (Chiba Medical, Saitama, Japan) and stored at −80°C. The PLP-fixed samples were then embedded in paraffin by acetone-methyl benzoate-xylene (AMeX) method 11 and were sectioned at 4-µm in thickness and stained with hematoxylin and eosin for morphological examination. In addition, the Azan-Mallory staining was performed for collagen deposition. The fresh frozen samples were sectioned at 10-µm thickness for immunohistochemistry and double immunofluorescence.
Immunohistochemistry and Double Immunofluorescence
Immunohistochemical conditions are detailed in Table 1. The following antibodies were used: vimentin and α-SMA for mesenchymal cells and myofibroblasts; Thy-1 for immature mesenchymal cells and pericytes; cytokeratin19 (CK19) for immature epithelial cells; cytokeratin AE1/AE3 (CK AE1/AE3) for re-epithelialization cells; and RECA-1 for endothelial cells. For negative control, primary antibodies were substituted with nonimmunized immunoglobulin G (IgG).
Primary Antibodies Used for Immunohistochemistry and Double Immunofluorescence.
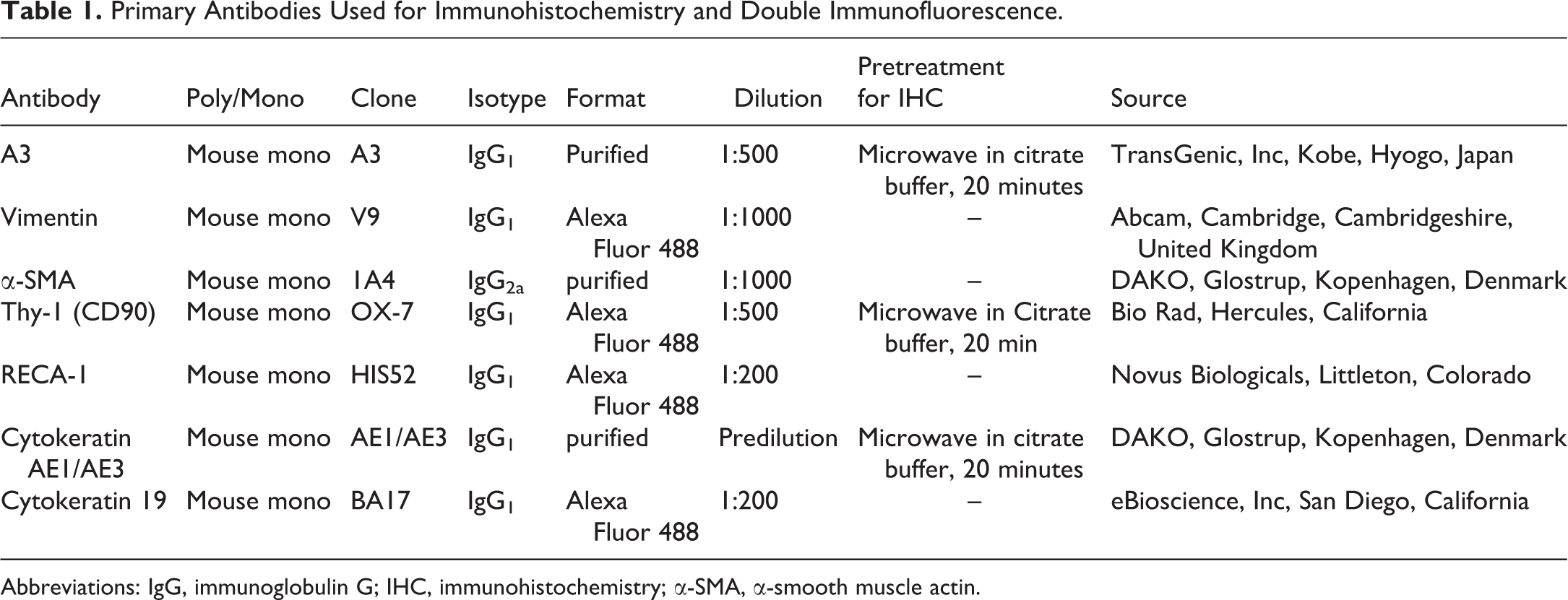
Abbreviations: IgG, immunoglobulin G; IHC, immunohistochemistry; α-SMA, α-smooth muscle actin.
The PLP–AMeX-processed tissue sections were used for vimentin, Thy-1, α-SMA, and CK AE1/AE3 immunohistochemistry. The sections were treated with 5% skimmed milk in phosphate-buffered saline (PBS) for 15 minutes and were incubated with each primary antibody overnight at 4°C. The sections were then incubated with 3% H2O2 in PBS for 15 minutes to quench endogenous peroxidase, followed by 1-hour incubation with horseradish peroxidase–conjugated secondary antibody (Histofine Simple Stain Rat MAX-PO; Nichirei, Tokyo, Japan). Positive reactions were detected with 3, 3′-diaminobenzidine (DAB; Nichirei). Sections were counterstained lightly with hematoxylin. The sections were scanned with VS120 Virtual Slide System (Olympus, Tokyo, Japan).
For double immunofluorescence, fresh frozen sections were used with combinations of A3/Thy-1, A3/vimentin, A3/CK19, A3/RECA-1, and A3/α-SMA. The sections were fixed in the PLP solution at room temperature for 15 minutes and were treated with 10% normal goat serum in PBS for 15 minutes to reduce nonspecific reactions. Then, the sections were incubated overnight with A3 at 4°C, followed by an incubation with goat antimouse IgG1 secondary antibody conjugated with Alexa Fluor 568 (Thermo Fisher Scientific, Waltham, Massachusetts). Subsequently, the sections were incubated with the counterpart primary antibody directly labeled with Alexa Fluor 488 for 1 hour. For the A3/α-SMA double immunofluorescence, the sections were incubated with both primary antibodies simultaneously, followed by an incubation with Alexa Fluor 568-conjugated goat antimouse IgG1 and Alexa Fluor 488-conjugated goat antimouse IgG2a secondary antibodies (Thermo Fisher Scientific). The sections were coverslipped with mounting medium including 4′,6-diamino-2-phenylindole (Fluoro-KEEPER Antifading Reagent; Nacalai Tesque, Inc, Kyoto, Japan) and were captured with Nikon C1Si confocal microscope (Nikon, Tokyo, Japan) and EZ-C1 Viewer (Nikon).
Results
Histopathological Findings in DSS-Induced Colonic Lesions
In control colons, no significant changes were seen (Figure 1). The lesion induced by DSS was limited to the distal colon. At the early injury stage of DSS-treated group on day 3, the colon had flattened mucosal epithelium with edematous interstitium and mild infiltrates of inflammatory cells such as neutrophils, lymphocytes, and macrophages, mainly seen in the lamina propria (Figure 2A). At the advanced injury stage of DSS-treated group on day 5, the distorted crypts, lined irregularly by flattened or cuboidal epithelial cells, were seen in addition to the mucosal injury (Figure 2B). Inflammation was extending from the lamina propria to the submucosa. Furthermore, mucosal ulcer occurred sporadically, and collagen deposition began to appear in varying degrees in the lamina propria. At the early recovery stage of DSS-treated group on day 7 or recovery group on day 1, the fibroplasia developed both in the lamina propria and in submucosa, accompanied by a loss of crypts, gradually progressing with desquamation of the mucosal epithelia (Figure 2C). Partly, mucosal regeneration by monolayered epithelial cells were observed in the ulcerative region, indicating re-epithelialization. At the healing stage of recovery on days 5 and 7, where re-epithelialization was developing after the cessation of DSS administration, regenerating crypts, rimmed by thin and irregularly arranged epithelial cells, were seen in the fibrotic lesion of the lamina propria; the crypts were dilated in varying degrees (Figure 2D). In addition, the ulcerative region with a decreased collagen deposition was covered by the regenerating epithelial cells. The fibrotic lesion seen in the DSS-administered rats was confirmed by the Azan–Mallory stain; the deposition of collagen was seen in both lamina propria and submucosa, resulting in the thickening of submucosa. Collagen deposition was gradually decreased with mucosal regeneration (re-epithelialization).
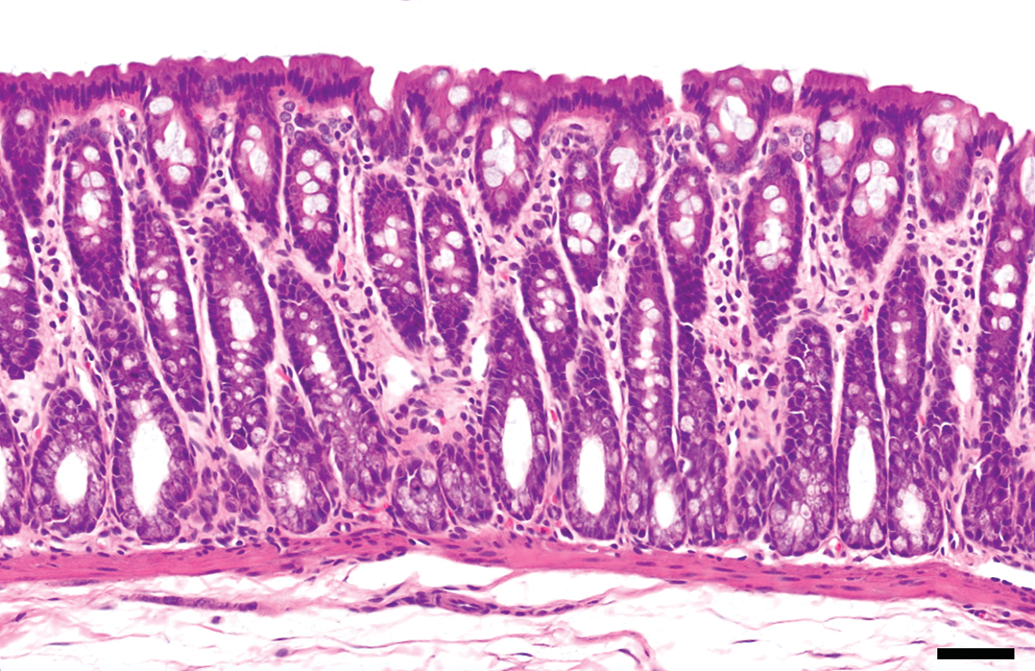
Histology of control colon. No significant changes are seen. Hematoxylin and eosin. Bar = 50 μm.
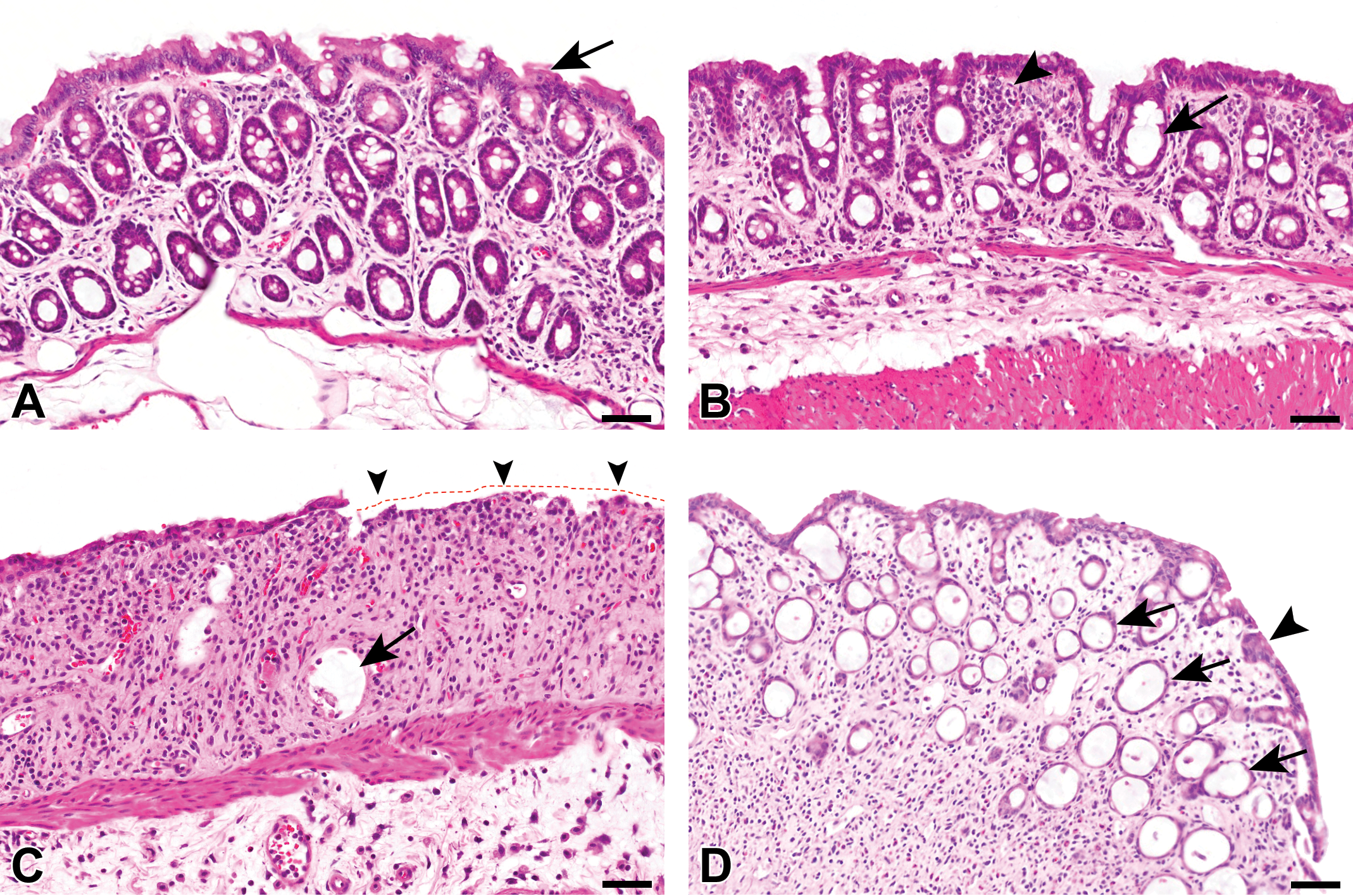
Histopathology of dextran sulfate sodium (DSS)–induced colonic lesions. A, At the early injury stage, the flattened mucosal epithelium (arrow) and edema in the lamina propria are seen. B, At the advanced injury stage, the crypts are distorted (arrow) with infiltrates of inflammatory cells (arrowhead) in the lamina propria. C, At the early recovery stage, fibroplasia in the lamina propria and loss of crypts are seen, accompanied with desquamation of the mucosal epithelium (red dotted line with arrowheads); the crypts are dilated without epithelial cell lining (arrow). D, At the healing stage, there are many regenerating crypts (arrows), rimmed by thin and irregularly arranged epithelial cells; the mucosal epithelium shows an early regeneration (reepithelialization) in the ulcerative region (arrowhead). Hematoxylin and eosin. Bar = 50 μm.
Distribution of A3-Positive Cells in DSS-Induced Colon Lesions
In colons of adult rats, A3-positive cells are localized exclusively around the crypts. 4 The A3 expression tends to surround the base to middle of crypt. In addition, A3 labels endothelial cells of capillaries and vessels in the lamina propria and the submucosa. By immunoelectron microscopy, A3-recognizing antigen was localized on cell membrane and in cytoplasm of the labeled cells as fine granular reactions. 4,12
At the early injury stage of the DSS-induced colonic lesions, A3-positive cells were localized exclusively around the crypts, as seen in control colons (Figure 3A, inset). At the advanced injury stage, A3-positive cells around the distorted crypts disappeared; endothelial cells of newly formed vessels in the lesion reacted faintly to A3 antibody (Figure 3B, inset). Interestingly, at the early recovery stage, the surface of the fibrotic lesion with desquamated mucosa was partly covered by A3-positive cells, showing a spindle or fusiform configuration (Figure 3C, inset). Furthermore, endothelial cells reacted more clearly to A3 in the fibrotic area. At the healing stage, a large number of A3-positive cells were aggregated in the surface of fibrotic lesion with desquamated epithelial cells, whereas A3-positive cells were few and sporadic in the fibrotic lesion beneath the monolayered regenerating epithelial cells (Figure 3D). Furthermore, in addition to the endothelial cells reacting to A3, A3-positive cells began to be seen around the regenerating crypts (Figure 3D, inset).
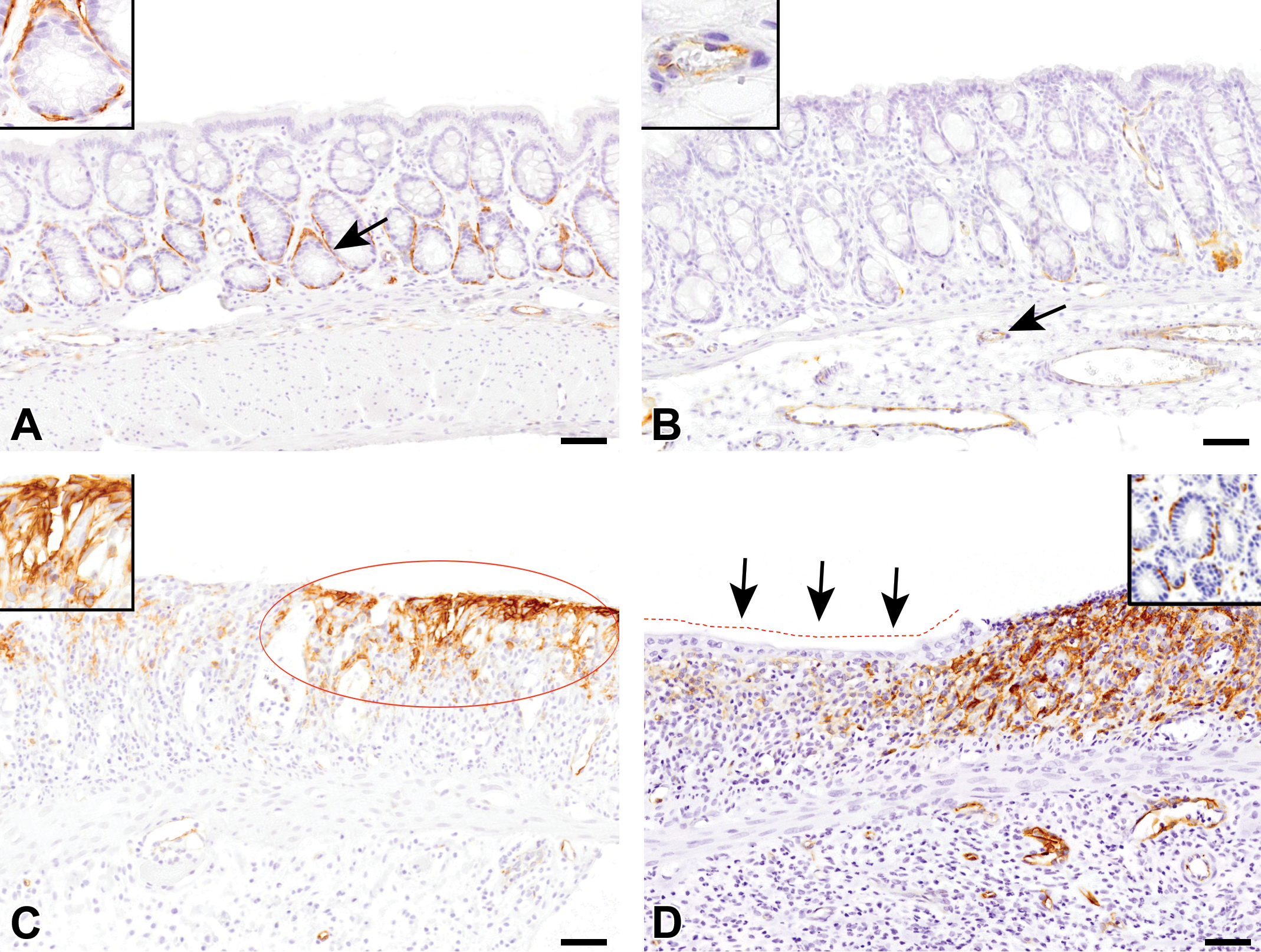
Immunohistochemistry for A3 in dextran sulfate sodium (DSS)–induced colonic lesions. A, At the early injury stage, A3-positive cells are seen around the crypts (inset: higher magnification of the arrowed area). B, At the advanced injury stage, cells reacting to A3 around the crypts disappear, while endothelial cells of neovascularization react to A3 (inset: higher magnification of the arrowed area). C, At the early recovery stage, A3-positive cells are aggregated in the surface of the ulcerative region where the epithelium is completely desquamated (circle); A3-positive cells show a spindle and fusiform configuration (inset). D, During the healing stage, many A3-positive cells are seen in the fibrotic lesion with desquamated mucosa, while there are few in the region of re-epithelialization (red dotted line and arrows). In addition, A3-reacting cells begin to be seen around the regenerating crypts (inset). Counterstained with hematoxylin. Bar = 50 μm.
Characterization of A3-Positive Cells Appearing in DSS-Induced Colon Lesions
To further characterize the A3-positive cells appearing in the DSS-induced colon lesions, single immunohistochemistry (with antibodies to vimentin, Thy-1, AE1/AE3, and α-SMA) or double immunofluorescence (with antibody combinations of A3/Thy-1, A3/vimentin, A3/CK19, A3/RECA-1, and A3/α-SMA) was performed. A large number of vimentin-positive mesenchymal cells were seen in the lamina propria and submucosa of the affected colon (Figure 4A). Thy-1-positive cells were present exclusively in the upper region (beneath the desquamated mucosa) of the affected lamina propria (Figure 4B). α-SMA antibody, a marker for myofibroblasts, 9,12 labeled spindle-shaped cells in lamina propria arranged vertically to the muscularis mucosae (Figure 4C) as well as smooth muscle cells in the preexisting vessels and muscularis mucosae (internal control).
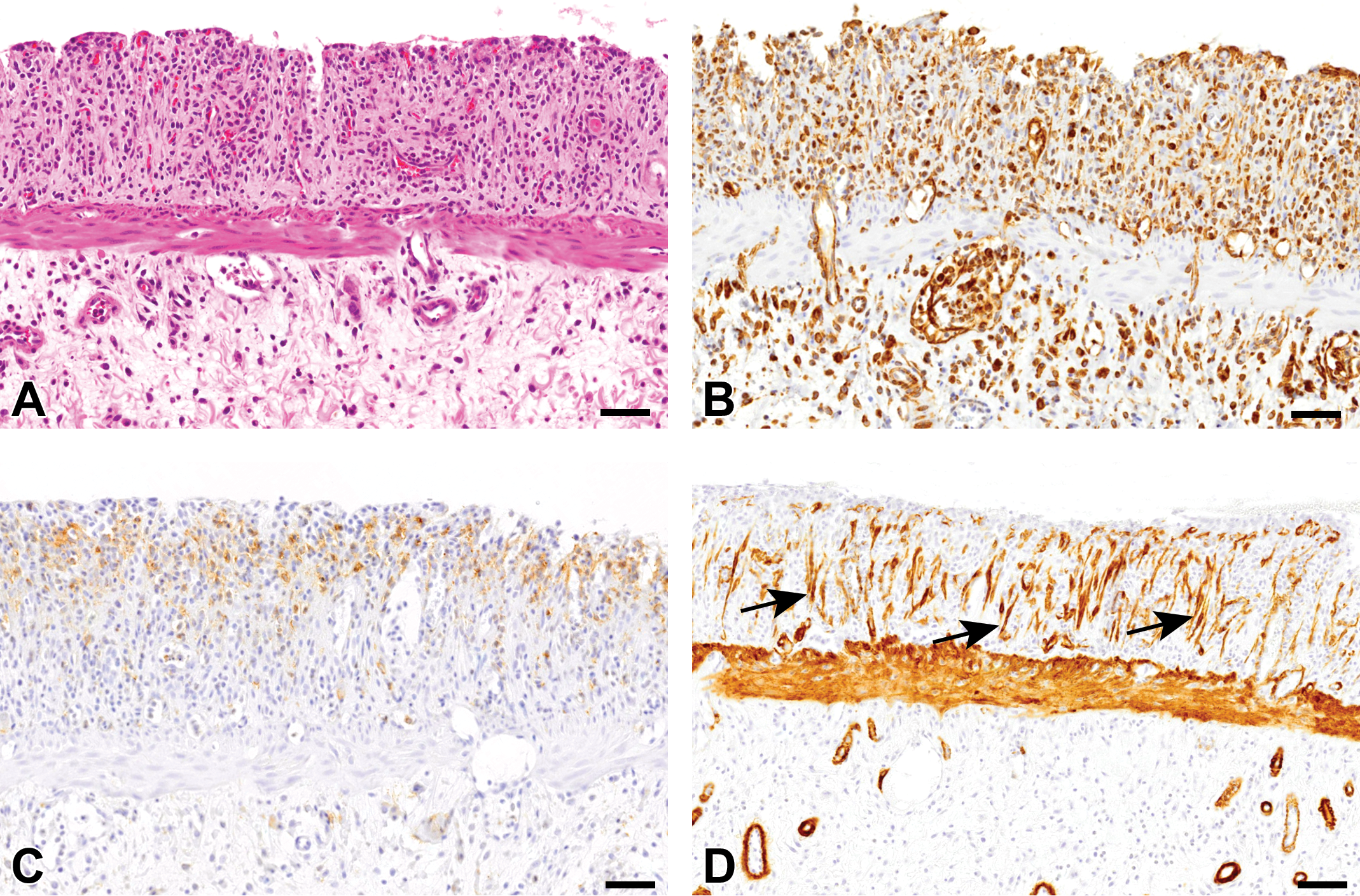
Immunohistochemistry for vimentin (mesenchymal cells), Thy-1 (immature mesenchymal cells), and α-smooth muscle actin (α-SMA; myofibroblasts) in dextran sulfate sodium (DSS)–induced colonic lesions at the early recovery stage. A, Histopathology of DSS-induced colonic lesions at the early recovery stage. Hematoxylin and eosin. B, Many vimentin-positive cells are seen diffusely in the fibrotic lesion. C, Thy-1-positive cells are seen mainly in the upper region of the fibrotic lesion. (D) α-SMA-expressing myofibroblasts in the fibrotic lesion are arranged vertically to the muscularis mucosae (arrows). Counterstained with hematoxylin. Bar = 50 μm.
At the early recovery stage with the greatest fibrosis and the healing stages with decreased fibrosis/re-epithelialization, the aggregated A3-positive cells, seen in the surface of fibrotic lesion beneath the desquamated mucosa, reacted simultaneously to Thy-1 (Figure 5A and B) and vimentin in varying degrees (Figure 5C and D); apparently, the double-positive cells with A3/Thy-1 and A3/vimentin tended to be greater at the early recovery stage than at the healing stage. More interestingly, in the surface of the fibrotic lesion, there were A3-positive cells coexpressing CK19 (Figure 5E and F). Regenerating epithelial cells, appearing in the surface of the fibrotic lesion, reacted to CK AE1/AE3, whereas the aggregated A3-positive cells did not react to AE1/AE3 (Figure 6). The A3 and CK AE1/AE3 double immunofluorescence was not performed, since the isotype of both antibodies is mouse IgG1. Endothelial cells of newly formed or preexisting blood vessels in the affected areas reacted both to A3 and to RECA-1 (Figure 7A and B). α-SMA-expressing myofibroblasts in the fibrotic lesion did not react to A3 (Figure 7C and D).
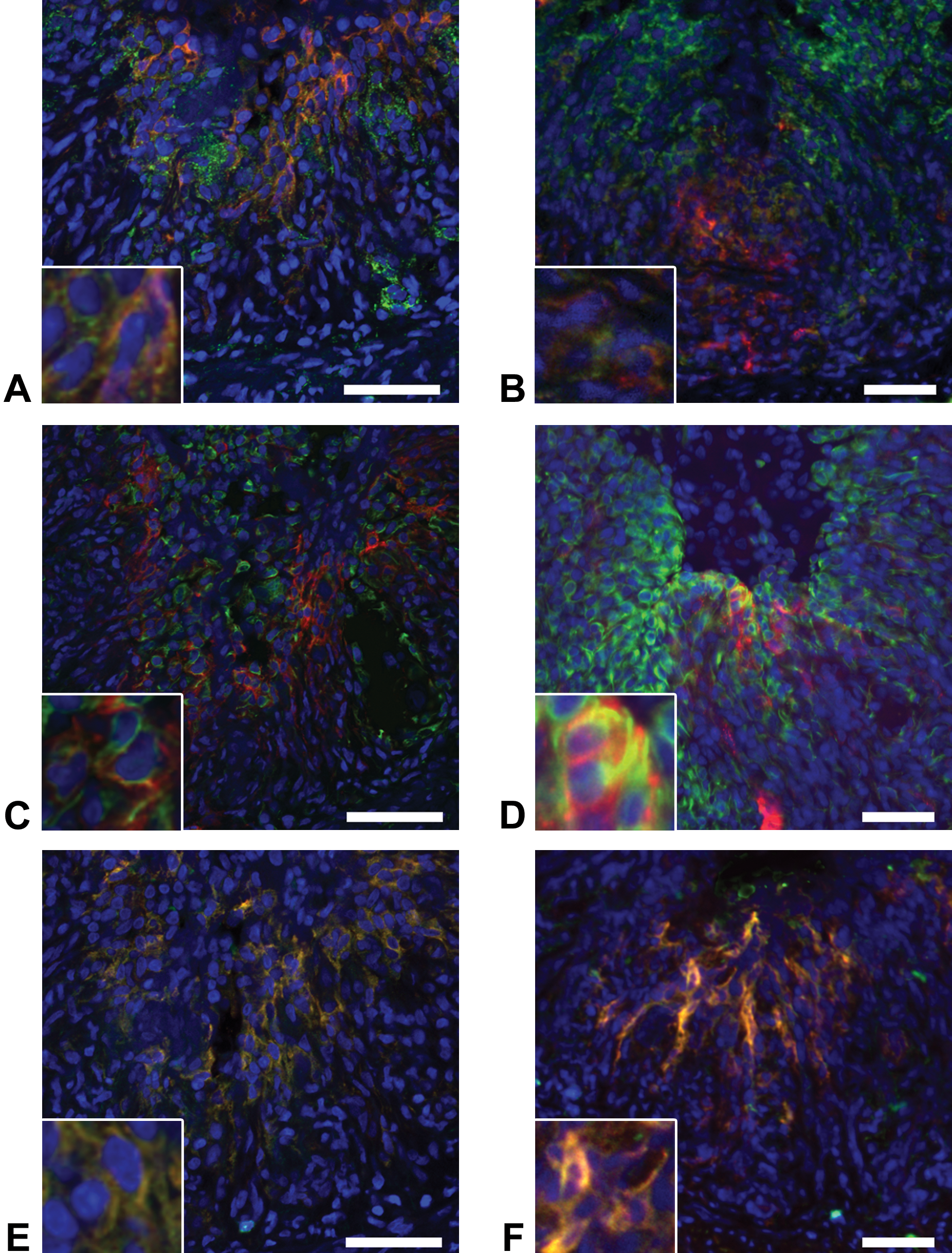
Double immunofluorescence for (A and B) A3 (red)/Thy-1 (green), (C and D) A3 (red)/vimentin (green), (E and F) A3 (red)/CK19 (green) in dextran sulfate sodium (DSS)–induced colonic lesions. Aggregated A3-positive cells at the (A, C, E) early recovery and (B, D, F) healing stages react simultaneously to Thy-1, vimentin, and CK19. Blue color indicates nuclei (with DAPI). Yellow color indicates a double positive reaction. Bar = 50 μm.
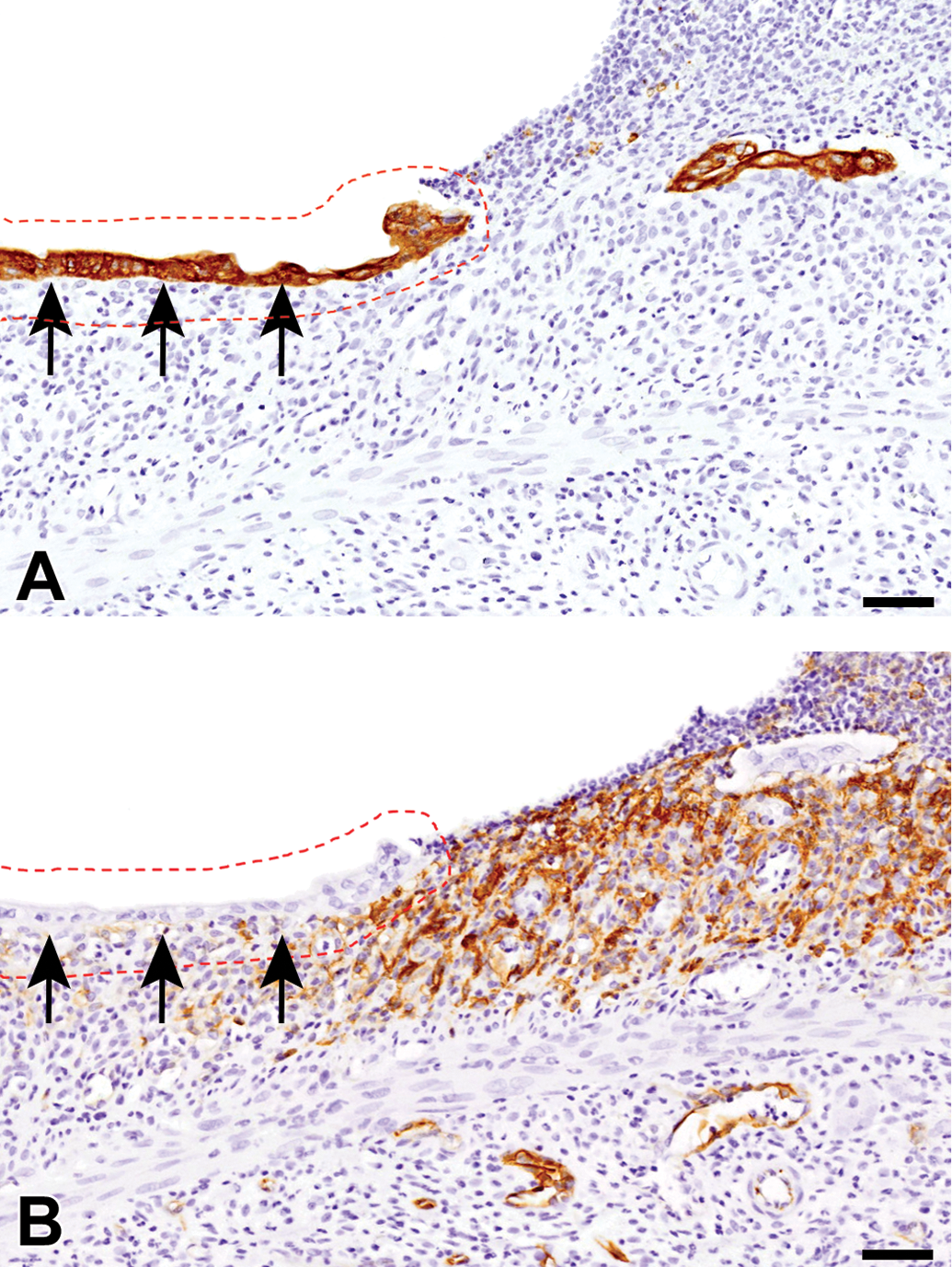
Immunohistochemistry for (A) CK AE1/AE3 and (B) A3 in dextran sulfate sodium (DSS)–induced colonic lesion at the healing stage. Mucosal epithelial cells in the region of re-epithelialization (red dotted lined area with arrows) do not react with A3. Counterstained with hematoxylin. Bar = 100 μm.
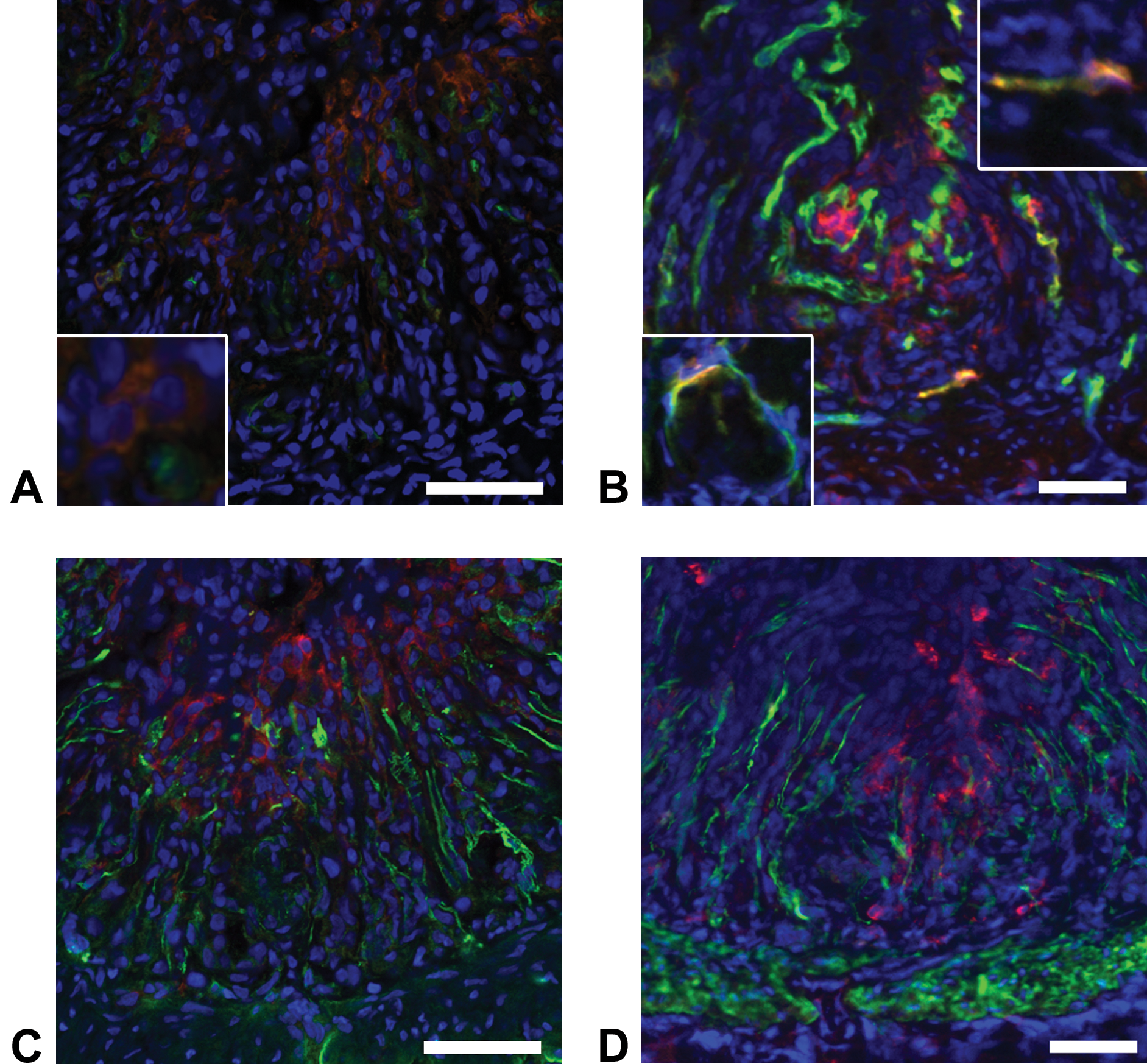
Double immunofluorescence for (A and B) A3 (red)/RECA-1 (green), (C and D) A3 (red)/α-smooth muscle actin (α-SMA; green) in dextran sulfate sodium (DSS)–induced colonic lesions at the early recovery stage. Endothelial cells of newly formed blood vessels (B; lower left inset) or preexisting blood vessels (B; upper right inset) in the fibrotic lesion react to A3 at the (A) early recovery and (B) healing stages. A3-positive cells do not react to α-SMA at the (C) early recovery and (D) healing stages. Blue color indicates nuclei (with DAPI). Yellow color indicates double a positive reaction. Bar = 50 μm.
Discussion
Dextran sulfate sodium has been used in rats and mice to induce colonic lesions, of which histopathological findings resemble those of human IBDs. 13,14 At the early stage after the administration in the DSS-treated group, the DSS-induced colonic lesions were characterized by interstitial edema, inflammatory cell reaction, and desquamating mucosal epithelial cells. At the advanced injury stage, epithelial elements of mucosa and crypts were irregularly arranged and disappeared with ulcer and fibrosis; in addition, neovascularization was seen. 5,15 To evaluate samples showing reparative fibrosis with decreased fibrosis and regeneration of mucosa and crypts, we set up the recovery groups after cessation of dosing. By examining samples, we could analyze different colonic lesions at the early injury, advanced injury, recovery, and healing stages as mentioned in “Results” section; the greatest fibrosis with mucosal desquamation was seen in the early recovery stage, and the fibrosis with regenerating epithelial cells in the mucosa and crypts as well as decreased collagen deposition was at the healing stage.
In human IBDs, the fibrotic lesion in the colon was the most characteristic lesion, indicating intractable chronic process. 8,16,17 α-SMA antibody has been used to detect myofibroblasts capable of producing a large amount of collagens, resulting in the development of incurable fibrosis in cases of liver cirrhosis and atrophic kidney 18 ; therefore, the derivation and characteristics of myofibroblasts have been focused on the development of fibrosis. In the present DSS-induced rat colonic lesions, α-SMA-expressing myofibroblasts were seen, being arranged vertically to the muscularis mucosae in the lamina propria; the positive cells were most abundant in the fibrotic area. In the double immunofluorescence, α-SMA-positive myofibroblastic cells were not corresponding to cells reacting to A3. In the pulmonary and cutaneous fibrosis rat models, although α-SMA-positive myofibroblasts were also seen, A3-positive cells did not react to α-SMA in double immunofluorescence 9,12 ; these findings were in agreement with those in the present colonic lesions with fibrosis. It has been considered that myofibroblasts seen in fibrotic lesion might be derived from immature mesenchymal cells. 19 -22 Thy-1 is a marker of immature mesenchymal cells. 23 It is interesting to note that there were cells reacting simultaneously to A3/Thy-1 or A3/vimentin in the present colonic fibrotic lesion. A3/Thy-1-positive immature mesenchymal cells in the colonic fibrotic lesion may be a progenitor of myofibroblasts. It is interesting to investigate the relationship between A3-positive immature mesenchymal cells and α-SMA-expressing myofibroblasts in the present colonic fibrotic lesion.
More interestingly, this study showed that there were cells reacting to A3 in the surface of the fibrotic lesion, where the mucosal epithelium was completely desquamated (ulcer lesion). The aggregated A3-positive cells simultaneously expressed vimentin or Thy-1. As mentioned above, vimentin is a mesenchymal marker and Thy-1 is known to be a marker for immature mesenchymal cells. The mesenchymal stem cells in pathological lesions have been speculated to be derived from bone marrow stem cells as a possible contributor of fibrotic lesion development 21,24 ; recently, such bone marrow–derived stem cells and connective tissue stem cells such as pericytes are called “Muse cells” (multilineage-differentiating stress-enduring cells). 25 The aggregated A3-positive immature mesenchymal cells might be derived from the bone marrow stem cells and pericytes, partly because it is reported that A3 labels bone marrow stem cells and pericytes in connective tissues. 2
Interestingly, some A3-positive cells appearing in the surface of the fibrotic lesion also reacted to CK19, which is used as a marker for immature epithelial cells. 26,27 Therefore, A3/CK19-double positive cells are regarded as immature cells with epithelial nature which is under regeneration. It has been considered that bone marrow–derived stem cells may participate in regeneration of damaged intestinal mucosal epithelial cells; the cell population of donor-derived epithelial cells increased in regenerative epithelial cells after bone marrow transplantation; these cells play an important role as rescue cells. 28 -30 The rescue cells could keep the barrier from external environment under progress of re-epithelialization. 31 The A3-positive cells seen in the surface of fibrotic lesion simultaneously reacted not only to CK19 but also to Thy-1 or vimentin. It has been reported that “Muse cells” can have multilineage differentiations such as epithelial and mesenchymal cells. 32 Although the significance of A3-postive cells should be investigated more, cells reacting simultaneously to A3/Thy-1 or A3/CK19, which were seen in the surface of colonic fibrotic lesion, are considered as “Muse cells”; these A3-positive cells are regarded as rescue cells in the colonic lesions.
In adult rat colon, the A3-postitive cells around the crypts are regarded as cells participating in the intestinal stem cells niche. 4 Basically, the intestinal stem cells in the crypts play important roles in regeneration of mucosal epithelial cells. 33,34 At the advanced injury stage, in addition to loss of crypts, A3-positive cells disappeared around the crypts, indicating the abnormal intestinal conditions due to inflammation and fibrosis. At the healing stage, A3-positive cells reappeared around the regenerating crypts. The A3-positive cells were different from α-SMA-expressing myofibroblasts (as a component of the intestinal stem cell niche). 4,35 -37 These findings indicate that A3-positive immature cells may be important for regeneration of mucosa as an element of intestinal stem cell niche.
In conclusion, the present study using DSS-induced colonic lesions showed that A3-positive cells might participate in re-epithelialization as rescue cells with nature of both mesenchymal and epithelial cells in the desquamated mucosa. In addition, the reappearance of A3-positive cells around the crypts in the fibrotic lesion indicated that A3-positive cells were the important cell element of intestinal stem cell niche for mucosal regeneration. Furthermore, endothelial cells reacting to A3 were related to newly formed blood vessels. A3 labels somatic stem cells in the rat bone marrow and pericytes with immature mesenchymal nature. 2 A3-positive cells seen in the present DSS-induced colonic lesions are regarded as “Muse cells” as a contributor for tissue repair. A3 antibody would become a useful tool to investigate the participation of somatic stem cells in chemical-induced pathological settings in rats.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article. This work was supported partly by JSPS KAKENHI Grant Numbers 26292152 and 19H03130 (to J.Y.) and by the Platform Project for Supporting Drug Discovery and Life Science Research [Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)] from AMED and Grand Numbers 3P18 am 0101123 (to J.Y.).
