Abstract
Several chemicals and pharmaceuticals increase the incidence of hemangiosarcomas (HSAs) in mice, but the relevance to humans is uncertain. Recently, canine HSAs were identified as a powerful tool for investigating the pathogenesis of human HSAs. To characterize the cellular phenotype of canine HSAs, we evaluated immunoreactivity and/or messenger RNA (mRNA) expression of markers for hematopoietic stem cells (HSCs), endothelial cells (ECs), a tumor suppressor protein, and a myeloid marker in canine HSAs. Neoplastic canine cells expressed EC markers and a myeloid marker, but expressed HSC markers less consistently. The canine tumor expression results were then compared to previously published immunoreactivity results for these markers in human and mouse HSAs. There are 2 noteworthy differences across species: (1) most human HSAs had HSC marker expression, indicating that they were comprised of tumor cells that were less differentiated than those in canine and mouse tumors; and (2) human and canine HSAs expressed a late-stage EC maturation marker, whereas mouse HSAs were negative, suggesting that human and canine tumors may retain greater differentiation potential than mouse tumors. These results indicate that HSA development is variable across species and that caution is necessary when discussing translation of carcinogenic risk from animal models to humans.
Keywords
Introduction
Hemangiosarcomas (HSAs) are aggressive, malignant tumors of endothelial cells (ECs) which are believed to arise from a variety of endothelial types, such as hepatic sinusoidal, venous, arterial, and capillary endothelium, as well as bone marrow-derived stem cells. 1 These tumors arise spontaneously at relatively high incidence in several mouse strains compared to other species. The overall tumor incidence is 5% in control male and female B6C3F1 mice. 1 In contrast, incidences are 10-fold lower in rats (∼0.5% in F344 rats 1 ) and rare in humans (<1% of all sarcomas, 2 which account for <1% of all adult solid malignancies 3 ). HSAs arise in 24 out of 100,000 canines (0.024%, >200-fold lower than mice), which is considered to be higher than humans, and is estimated to represent 7% of all canine malignant tumors. 4 Several breeds, including Golden Retrievers, Boxers, and German Shepherds, are known to have increased incidences of spontaneous HSAs. 1,4 Similar to humans, canine HSAs are highly malignant and metastasize frequently, and the affected dogs often die acutely from vascular rupture especially those in the spleen (American College of Veterinary International Medicine Fact Sheet: Hemangiosaroma, http://www.acvim.org/Portals/0/PDF/Animal%20Owner%20Fact%20Sheets/Oncology/Onco%20Hemangiosarcoma.pdf).
Various chemicals, including industrial, agricultural, and pharmaceutical agents, have been documented to increase the incidence of HSAs in 2-year carcinogenicity studies, primarily in mice. These agents include DNA reactive agents, such as vinyl chloride and thorotrast, 1 and non-DNA reactive agents, such as 2-butoxyethanol, 5 troglitazone (peroxisome proliferator-activated receptor γ activator), 6 and pregabalin (α2δ subunit of voltage gated calcium antagonist). 7 Despite an increased understanding of the mode of action, 1,7 –11 HSAs induced by a variety of chemicals in rodent bioassays, particularly in mice, have increasingly become a significant concern to regulatory authorities leading to a delay in regulatory approval for pharmaceuticals.
Characterizing the cellular phenotype of this tumor in preclinical species was undertaken to better understand and assess the risk to humans. We previously evaluated human and mouse HSAs by immunohistochemistry (IHC) for expression of hematopoietic stem cell (HSC) markers (CD45, CD34, CD117, and/or CD133), EC markers (vascular endothelial growth factor receptor 2 [VEGF-R2], CD31, and factor VIII–related antigen [FVIII-RA]), and a myeloid lineage marker (CD14). 12,13 The distribution of these markers in the course from bone marrow HSC to mature ECs has been summarized elsewhere. 12,13 Mouse HSAs had IHC staining patterns consistent with endothelial progenitor cells (EPCs) that expressed CD34, VEGF-R2, and CD31, but not CD45, FVIII-RA. 13 Human HSAs expressed CD45, CD117, and FVIII-RA in addition to CD34, VEGF-R2, and CD31. 12 The contribution of myeloid-lineage cells in human EC tumors is possible since some HSA cases were positive for CD14 immunoreactivity. 12 These results indicate that mouse HSAs possibly arise from different progenitor cells and/or develop by a different mechanism than humans. These data suggest that results of 2-year carcinogenicity studies in mice need to be interpreted cautiously to evaluate and predict sarcomagenic potential of chemicals toward human ECs.
The pathogenesis of EC tumors in canines is not well understood, but accumulated evidence has shown that, similar to mice and humans, canine HSAs may arise from EPCs rather than differentiated ECs. 14 –18 Canine HSA cell lines have been shown to express CD117, CD133, CD34, and in some cases, CD45 and CD14. 14 Cells from sphere-forming cultures of canine HSAs expressed HSC markers (CD133, CD117, and CD34), EC markers (CD105, CD146, and αvβ3 integrin), and myeloid lineage markers (CD115 and CD14). 15 Additionally, positive immunoreactivity for CD117 and FVIII-RA in canine HSAs has been reported in some studies. 16,17,19,20 These data indicate that the canine model may be potentially more relevant to understanding the pathogenesis of the human EC tumors compared to the mouse model. 15 Additionally, a recent genomic study in canine and human HSAs reported that canine HSAs closely model human HSAs on a genomic level, and suggested that canine HSAs are a powerful tool for investigating the pathogenesis of human HSAs. 18
In the present study, we evaluated the immunoreactivity and/or messenger RNA (mRNA) expression of HSC markers (CD133, CD117, CD45, and CD34), EC markers (VEGF-R2, CD31, and FVIII-RA), and a myeloid lineage cell marker (CD14) in 11 spontaneous canine HSAs. These tumors were also immunostained for the p53 tumor suppressor protein, which is frequently positive in human HSAs. Since the antibodies used to label the various tissue markers in the canine were generated to target either human and/or rodent proteins with minimal to no validation information for use in the canine species, mRNA expression was measured to corroborate the IHC results, as well as to provide expression data for those markers for which successful IHC assays could not be achieved. Immunoreactivity for each marker was compared to previous immunoreactivity results in humans and mice to determine how the cellular phenotype of canine HSAs compares to human and to mouse tumors.
Materials and Methods
Tumor Samples
Eleven cases of canine HSAs (from years 2011 to 2013) were identified from the tissue archives of Auburn University, and their formalin-fixed, paraffin-embedded (FFPE) tissue specimens retrieved. Diagnoses of HSAs were made and confirmed from hematoxylin and eosin-stained slides by board certified veterinary pathologists (R.C.C. at Auburn University and J.M.K. and L.A.O. at Pfizer). Animal data, including age, sex, and tissue site, were available for all cases (Table 1). All 11 canine cases were utilized for the IHC evaluations; however, only 5 of these cases had adequate mRNA integrity or amounts required for reverse transcription-polymerase chain reaction (RT-PCR) analysis. Normal spleen tissue was obtained from 2 nontreated control beagle dogs (Marshall Laboratory, North Rose, NY) from nonclinical toxicity studies to use as normal tissue comparators for the IHC and RT-PCR analysis.
Summary of Canine Samples.
Abbreviations: F-S, female spayed; HSA, hemangiosarcomas; M-C, male castrated.
IHC
Information of primary antibodies and reagents used for immunoreactivity are summarized in Table 2. Formalin-fixed, paraffin-embedded tissues were cut at 5 µm and placed on glass slides, deparaffinized, and rehydrated through graded alcohols. Slides were rinsed in distilled water, followed by antigen retrieval using proteinase K (S3020; Dako, Carpinteria, CA) for 5 minutes at room temperature for FVIII-RA or heat-induced antigen retrieval using a Decloaker pressure chamber (BioCare Medical, Pacheco, CA) for 5 minutes at 120°C in EDTA, 1× (005500, diluted to 1× using distilled water; ThermoFisher Scientific, Waltham, MA) for CD45, CD31, and p53, Borg solution (BD1000G1; BioCare Medical, Pacheco, CA) for CD34, or Citra solution (HK086-0 K; Biogenix, Houston, TX) for CD117. Slides with heat-mediated antigen retrieval were cooled for approximately 5 minutes at room temperature. After rinsing slides in distilled water, drawing a barrier around a tissue specimen using a water repellent pap pen, and rinsing slides in Tris-buffered NaCl solution with Tween 20, pH 7.6 (TBST, S3306; Dako, Carpinteria, CA), the slides were loaded onto an automated slide stainer (intelliPATH, BioCare Medical, Pacheco, CA) for automated processing. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide (BP2633500; ThermoFisher Scientific, Waltham, MA) for 10 minutes, followed by rinsing slides in TBST and blocking nonspecific binding sites in Protein Block, Serum-Free (X0909; Dako, Carpinteria, CA). Sections were then incubated with each primary antibody at desired concentration for desired time (Table 2). Serum collected from host species for each primary antibody was used as a negative control. Sections were incubated with Labelled Polymer—Dako (Carpinteria, CA) REAL EnVision-HRP, Mouse (K4001) or Rabbit (K4003) for 30 minutes. After rinsing slides in TBST, immunoreactivity was visualized by incubation in DAB+, Liquid (K3468; Dako, Carpinteria, CA) for 5 minutes and resulted in brown staining. Slides were then counterstained with Mayer hematoxylin, dehydrated, and cover-slipped. CD133, VEGF-R2, and CD14 could not be analyzed since optimization of the IHC method for these antigens was not successful. Microscope slides were scanned on the whole digital slide scanner (Aperio AT2; Leica, Buffalo Grove, IL) at the ×40 objective setting, and images were saved in .svs format. Representative images of the immunoreactivity results (Figures 1A-F and 2A-F) were digitally captured using the Aperio ImageScope software v11.2.0.780 at a virtual magnification of ×20.
Antibodies and Negative Controls Used for Immunohistochemistry.
Abbreviations: FVIII-RA, factor VIII–related antigen; RT, room temperature; VEGF-R2, vascular endothelial growth factor receptor 2.
a The dilution range recommended by each manufacturer in a product sheet was used in initial experiments for the method optimization of each antibody. If these initial attempts were successful, further experiments were performed to optimize the antibody dilution for each staining upon the discussion with the pathologist (L.A.O.).
b No antigen retrieval, as well as, antigen retrieval methods including Proteinase K incubation for 5 minutes at room temperature, heat-mediated antigen retrieval in Citra solution, EDTA (×1), or Borg solution with an array of dilutions of the antibody were performed during the method optimization, but none of these were successful in the current study.
c CD83 is known as a myeloid cell marker and expressed in dendritic cells. It was reported to be expressed in human infantile hemangioma endothelium. 21
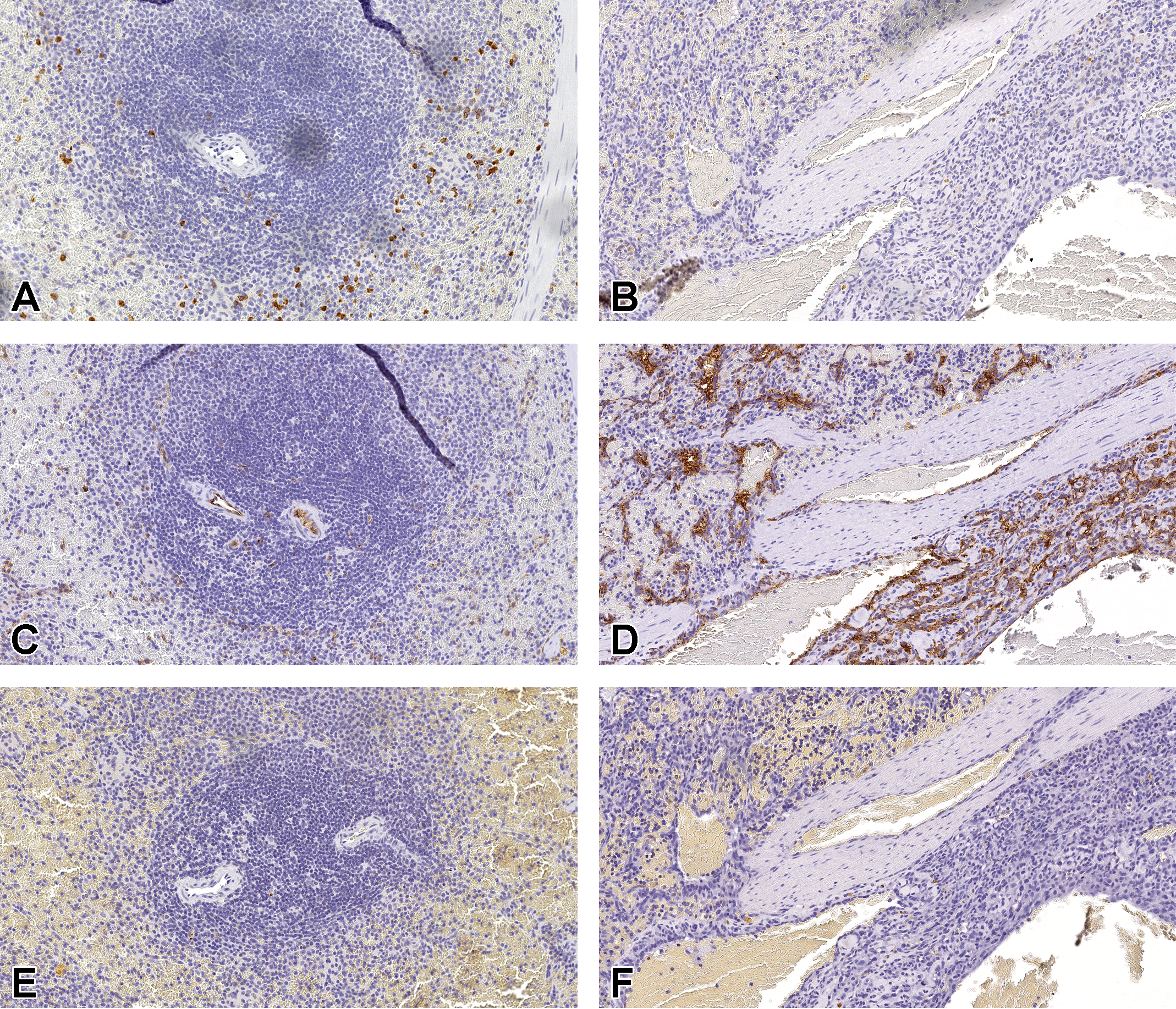
Tissue expression of CD117 (A and B), CD34 (C and D), and CD45 (E and F) were evaluated in spleens from a normal dog (A, C, and E) and a dog (HSA-13, see Table 1) with a splenic HSA (B, D, and F). The CD117 expression was present in granulocytes (A) but lacking in normal endothelium (A) and HSA tissue (B). The CD34 expression was strong in normal vascular endothelium (C), as well as, in HSA tissue (D). The CD45 expression was negative in normal endothelium (E) and HSA tissue (F). Original scans ×20. HSA indicates hemangiosarcoma(s).
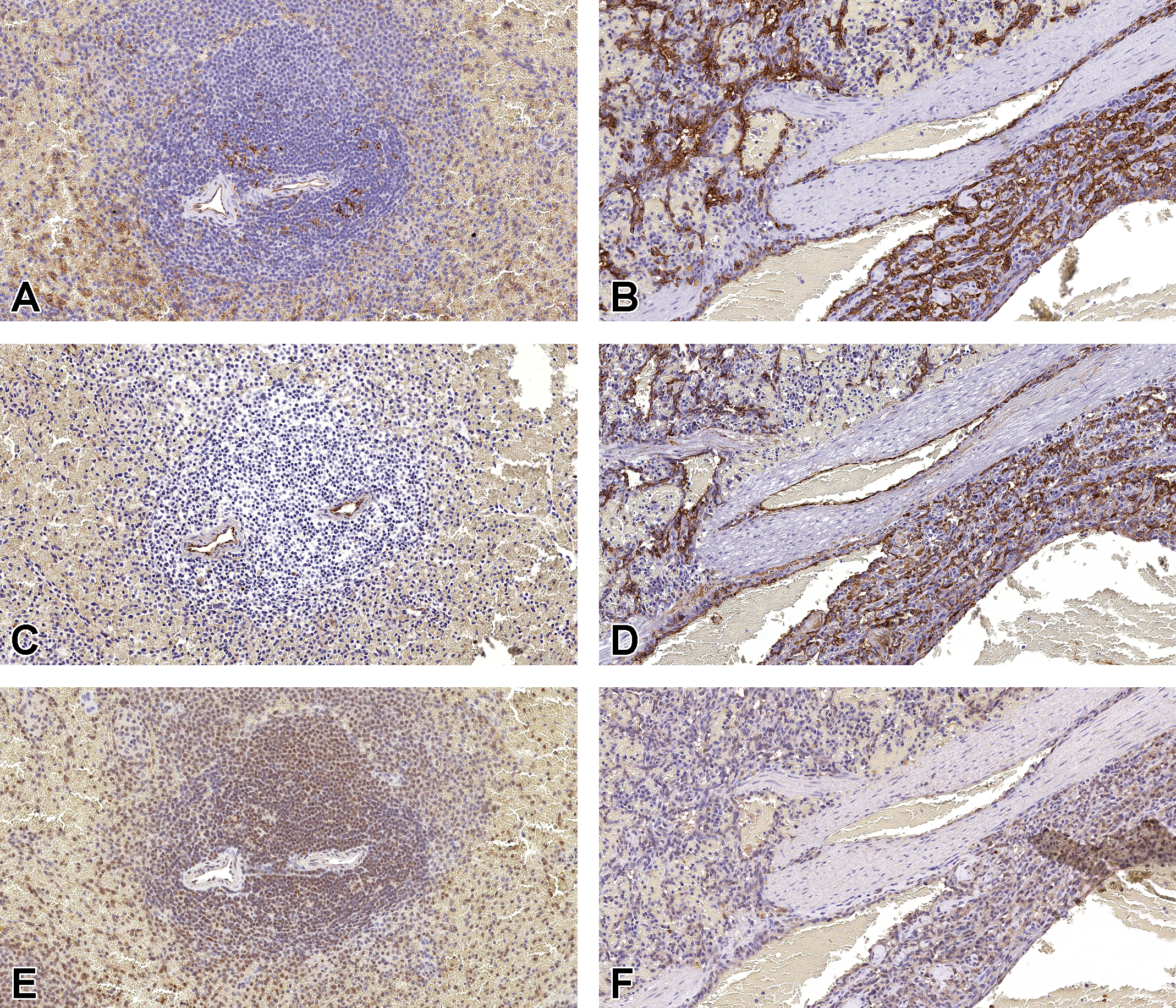
Tissue expression of CD31 (A and B), FVIII-RA (C and D), and p53 (E and F) were evaluated in spleens from a normal dog (A, C, and E) and a dog (HSA-13, see Table 1) with a splenic HSA (B, D, and F). The CD31 expression was moderate in normal splenic vascular and sinusoidal endothelium (A) and strongly expressed in HSA tissue (B). The FVIII-RA expression was strong in normal vascular endothelium (C) and was moderately expressed in HSA tissue (D). Nuclear p53 expression was moderate in sinusoidal endothelium and lymphocytes in the normal splenic tissue (E), as well as, in HSA tissue (F). Original scans ×20. FVIII-RA indicates factor VIII–related antigen; HSA, hemangiosarcoma(s).
IHC Evaluation
IHC-stained whole slide sections were evaluated by the veterinary pathologist (L.A.O.) for overall immunostaining intensity of the positive cells and were classified into one of the following grades as per previous studies 12,13 : negative, weak, moderate, or strong. These scoring criteria were applied to the HSA neoplastic cells and normal ECs present within nondiseased canine spleens.
RNA Isolation
Total RNA was isolated from one 5-µm section of FFPE tissue using a Qiagen RNeasy FFPE Kit (Cat #73504; Qiagen, Germantown, MD). Briefly, samples were deparaffinized using 1 mL of xylene, vortexed vigorously for 10 seconds and centrifuged for 2 minutes. After precipitation of the sample, the supernatant was removed by vacuum suction without disturbing the pellet. Residual xylene was removed by washing the pellet with 1 mL of 100% ethanol, vortexed and centrifuged again for 2 minutes. The supernatant was removed for a second time through vacuum suction without disturbing the pellet. With the lids open, the pellets were further dried in a vacufuge for 15 minutes at 37°C. To reverse formaldehyde modification of nucleic acids, 150 μL of Buffer PKD was added and the sample was mixed by vortexing followed by addition of 10 μL of proteinase K which was mixed by gentle pipetting. The samples were then incubated at 56°C for 15 minutes followed by 80°C for 15 minutes and then on ice for 3 minutes. Samples were then centrifuged for 15 minutes at 20,000 × g after which the supernatant was transferred to a new microcentrifuge tube. To remove DNase, 16 μL of DNase Booster Buffer and 10 μL DNase I (Cat#79254; Qiagen, Germantown, MD) stock solution were added and each sample was mixed through inverting followed by a quick centrifuge of the tube and incubation at room temperature for 15 minutes. A total of 320 μL of Buffer RBC was added to the lysate to adjust binding conditions followed by 720 μL of 100% ethanol. Up to 700 µL of the sample was then transferred to an RNeasy spin column placed in a 2 mL collection tube and centrifuged for 15 seconds at 20,000 × g before discarding the flow through. This was repeated with the remainder of the sample until no aqueous ethanol mixture remained. The column was then washed with 500 μL Buffer RPE for 15 seconds at 20,000 × g followed by an additional 500 μL buffer RPE wash and centrifugation for 2 minutes at 20,000 × g. Following these washes, the RNeasy MinElute spin columns were placed in a new 2 mL collection tube. With the lids open, the tubes were centrifuged at full speed for an additional 5 minutes. Total RNA was eluted from the solid support matrix by applying 30 µL of RNase free water (Cat #129112; Qiagen, Germantown, MD) and collected in a sample tube through centrifugation at 20,000 × g for 1 minutes. After elution, 1 µL of each sample was evaluated for total RNA concentration using standard 260 nm absorbance on a NanoDrop 1000 (Thermo Scientific) UV-Vis spectrophotometer. The isolated total RNA sample was stored at −80°C until needed.
Complementary DNA Library Generation
Isolated RNA samples were reverse transcribed to complementary DNA (cDNA) using the Life Technologies, Carlsbad, CA, High Capacity RNA-to-cDNA Kit (Cat# 4387406) following a protocol outlined in the manufacturer’s directions. Briefly, up to 2 µg of total RNA was used per 20 µL 1× reaction. For each reaction, 10 µL of the 2× RT Buffer, 1 µL of the 20× enzyme mix, and up to 9 µL of the sample (volume require to deliver appropriate amount of total RNA) were mixed and volume adjusted to a final 20 µL reaction volume using RNase-free water. Reverse transcription was performed for all samples at 37°C for 60 minutes followed by 95°C for 5 minutes. Sample cDNA libraries were stored at −20°C until expression analysis was performed.
Preamplification Reactions for a TaqMan Array Micro Fluidic Card
Complementary DNA samples were preamplified using the Preamplification Pool that was purchased with the TaqMan Array Micro Fluidic Cards following a protocol outlined in the manufacturer’s directions. Briefly, as the TaqMan Array Micro Fluidic Card format was 64, four fill reservoirs were loaded per sample. For each reaction, 4 μL of the custom TaqMan PreAmp Pool, 8 µL of the 2× TaqMan PreAmp Master Mix (Cat# 4384267; ThermoFisher Scientific, Waltham, MA), and up to 4 µL of the cDNA (up to 100 ng) were mixed and volume adjusted to a final 16 µL reaction volume using RNase-free water. Preamplification was performed for all samples at 95°C for 10 minutes followed by 14 cycles of denaturing at 95°C for 15 seconds and annealing at 60°C for 4 minutes. Samples were then held at 99.9°C for 10 minutes before cooling to 4°C. Preamplified samples were stored at −20°C until expression analysis was performed.
TaqMan Array Micro Fluidic Card Protocol
The quantitative RT-PCR (qRT-PCR) reaction was performed using the TaqMan Probe-Based Gene Expression Analysis and QuantStudio 12K Flex Real-Time PCR Systems (ThermoFisher Scientific, Waltham, MA). Target gene and endogenous controls were run in triplicate for each probe set on predesigned TaqMan Low Density Arrays. For each sample, 14 µL of preamplification product was added to 206 µL of nuclease free water and 220 µL of 2X Master Mix (Cat # 4352042; ThermoFisher Scientific, Waltham, MA) for a total volume of 440 µL. A total of 100 µL of sample was added to each port and 4 fill reservoirs were loaded per sample after which the plate was centrifuged, sealed and loaded into the QuantStudio 12K Flex Real-Time PCR System, and run. Default thermal cycling conditions were as follows: 2 minutes at 50°C, 10 minutes at 90°C and 40 cycles of 15 seconds at 90°C followed by 1 minute at 60°C.
Quantitative RT-PCR and Data Analysis
Quantitative RT-PCR was performed on a QuantStudio 12K FlexTM 7 qRT-PCR system (Life Technologies, Carlsbad, CA). ExpressionSuite Software version 1.0.3 (Life Technologies, Carlsbad, CA) was used to generate automated threshold values for signal amplification for a majority of samples. Automated thresholds were rarely adjusted manually. Amplification plots resulting in Ct values >35 were discarded and considered below the limit of detection, as were those samples with plots that generated a Ct value but did not display a trend of logarithmic amplification. All Ct values were exported from the ExpressionSuite software and relative quantification calculations were performed in Microsoft Excel 2010 (Microsoft Corporation, Inc, Redmond, WA).
Statistical analysis using multiple T-tests was performed using GraphPad Prism version 6.03 for windows (GraphPad Software, San Diego, California). Discovery was determined using the 2-stage linear step-up procedure of Benjamini, Krieger, and Yekutieli, with Q = 1%. Each row was analyzed individually, without assuming a consistent standard deviation. A P value greater than or equal to .05 was considered to be biologically significant.
Results
Representative IHC results (normal spleen and HSAs) for each marker are presented in Figures 1 and 2, and subjective IHC scoring for each marker is summarized in Table 3. Since tumor cells disrupted the normal vascular architecture in many of the HSA samples, tumor cell immunoreactivity could not be compared to the adjacent normal vascular endothelium. Instead, immunoreactivity intensity of tumor cells was compared to normal ECs from non-neoplastic canine spleens. Similarly, mRNA expression for all markers, including those for which IHC methods could not be optimized (VEGF-R2, CD14, and CD133), were evaluated in selected HSA cases and normal spleens. The HSA mRNA RT-PCR log2 fold change in delta cycle threshold (ΔCT) value compared to average ΔCT value of control spleens for each marker were reported and increased or decreased mRNA expression compared to normal spleen was indicated for each tumor evaluated; the results were summarized and compared to IHC results in Table 4. For most IHC markers (CD117, CD45, CD31, FVIII-RA, and p53) mRNA results paralleled immunoreactivity trends; the somewhat disparate CD34 results are discussed. For the case ID of HSA-05, tumor sample was taken from the liver even though the patient had tumor present in the spleen (Table 1). Due to sample size constraints, the liver tumor was used for IHC and the spleen tumor was used for mRNA. The liver derived tumor sample was comprised only of tumor without the presence of normal liver tissue. Therefore, IHC staining in tumor cells in liver origin sample was compared to unaffected normal spleen sinusoids present in the spleen section. Also, for consistency across patient samples, the spleen from case ID of HSA-05 was used for the evaluation of mRNA expression levels instead of the liver.
Staining for HSC and EC Markers and p53 in Canine HSAs.
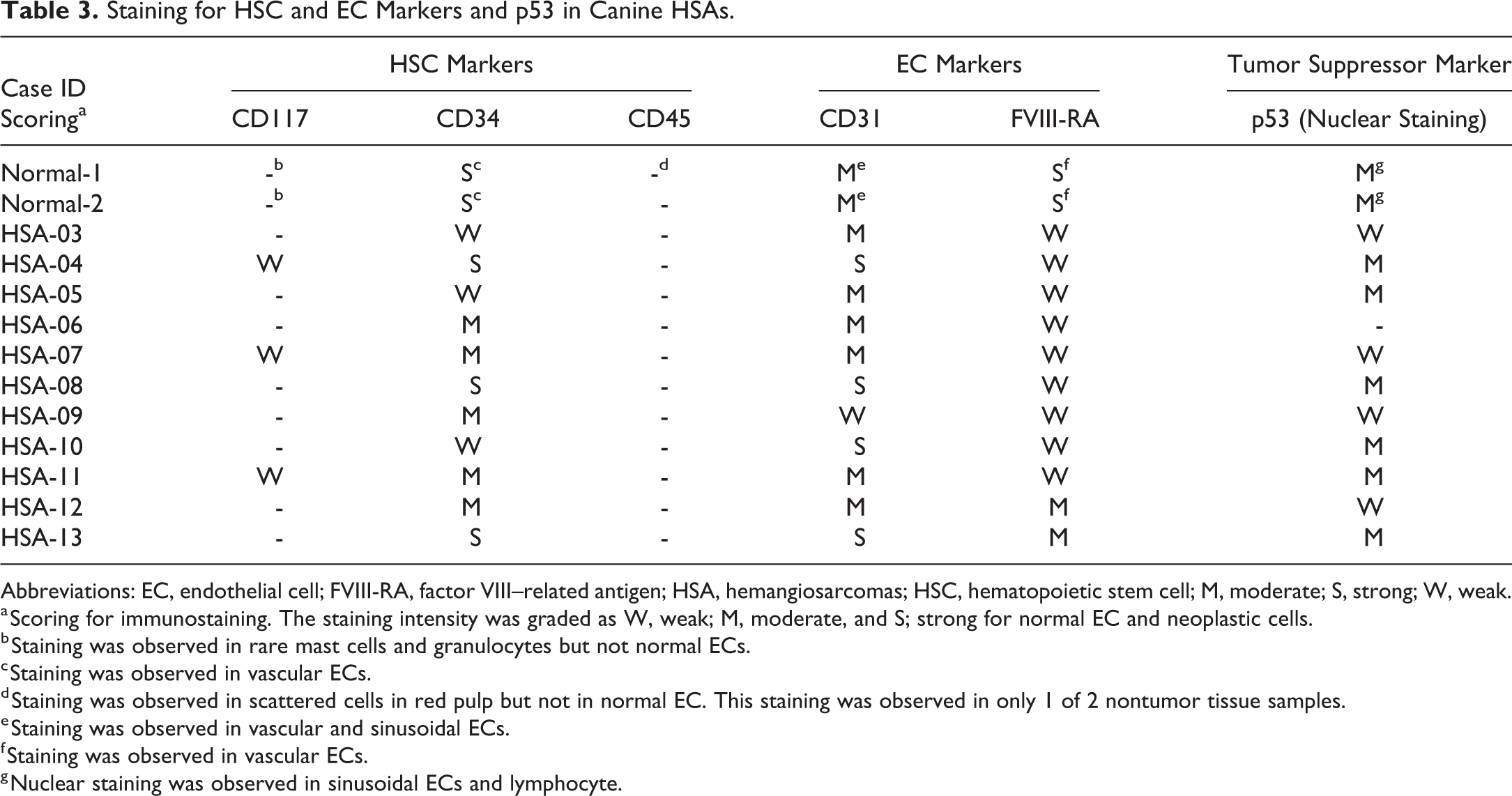
Abbreviations: EC, endothelial cell; FVIII-RA, factor VIII–related antigen; HSA, hemangiosarcomas; HSC, hematopoietic stem cell; M, moderate; S, strong; W, weak.
a Scoring for immunostaining. The staining intensity was graded as W, weak; M, moderate, and S; strong for normal EC and neoplastic cells.
b Staining was observed in rare mast cells and granulocytes but not normal ECs.
c Staining was observed in vascular ECs.
d Staining was observed in scattered cells in red pulp but not in normal EC. This staining was observed in only 1 of 2 nontumor tissue samples.
e Staining was observed in vascular and sinusoidal ECs.
f Staining was observed in vascular ECs.
g Nuclear staining was observed in sinusoidal ECs and lymphocyte.
The mRNA Expression for HSC, EC, and Myeloid Markers and p53 in Canine HSAs.
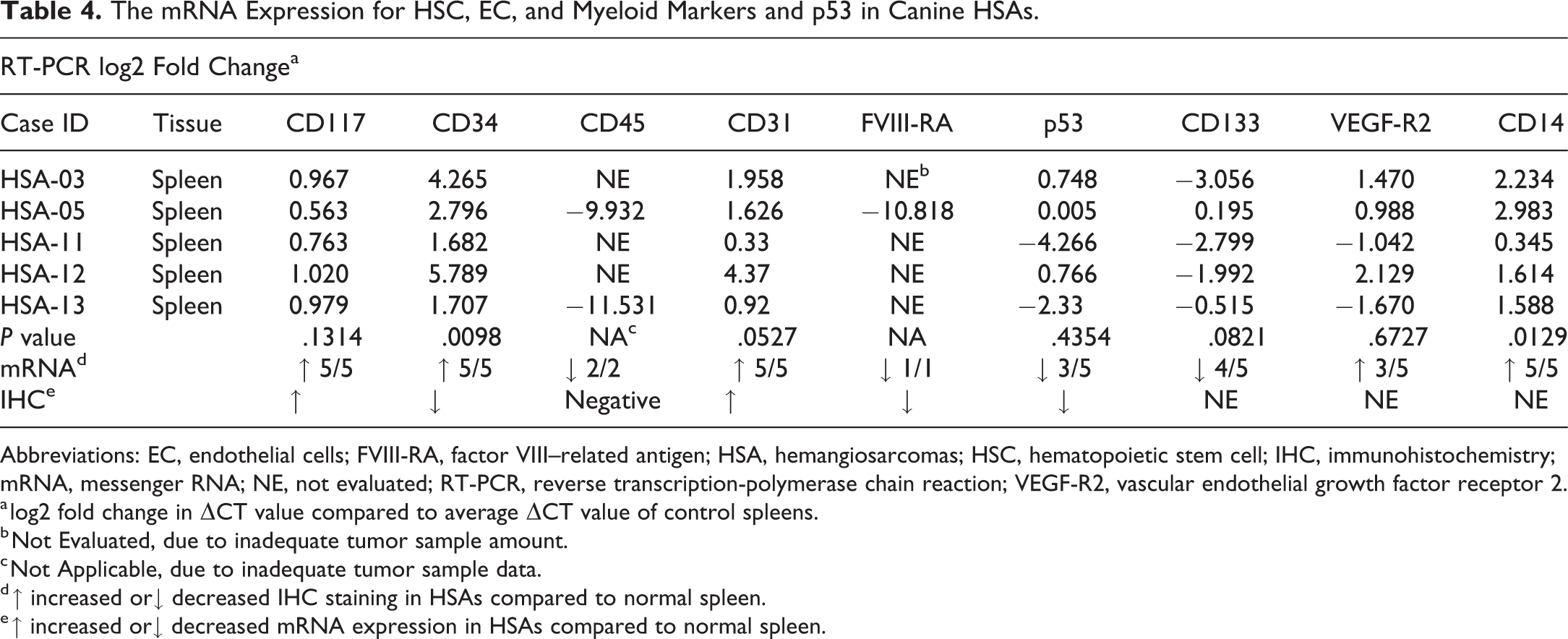
Abbreviations: EC, endothelial cells; FVIII-RA, factor VIII–related antigen; HSA, hemangiosarcomas; HSC, hematopoietic stem cell; IHC, immunohistochemistry; mRNA, messenger RNA; NE, not evaluated; RT-PCR, reverse transcription-polymerase chain reaction; VEGF-R2, vascular endothelial growth factor receptor 2.
a log2 fold change in ΔCT value compared to average ΔCT value of control spleens.
b Not Evaluated, due to inadequate tumor sample amount.
c Not Applicable, due to inadequate tumor sample data.
d ↑ increased or↓ decreased IHC staining in HSAs compared to normal spleen.
e ↑ increased or↓ decreased mRNA expression in HSAs compared to normal spleen.
Expression of HSC Markers
In control tissues, CD117 membrane and cytoplasmic immunoreactivity was observed in granulocytes and/or mast cells, but not in normal endothelium of spleen (Figure 1A). Three of 11 HSA cases were weakly positive for CD117 cytoplasmic immunoreactivity in some tumor cells, but most HSA cases were negative (Figure 1B). All (5 of 5) tumors had slightly higher CD117 mRNA expression (<2-fold) compared to normal spleen, but the average fold change was not statistically significant (Table 4). Even though mRNA and protein expression for CD117 were higher in canine HSAs compared to normal spleen, it was only minimally increased and approaching the lower limits of detection for these assays.
CD34 staining was observed in normal vascular endothelium (Figure 1C), as well as tumor cells (Figure 1D), in all HSAs cases. Although CD34 membrane and cytoplasmic immunoreactivity was strong in normal ECs (Table 3), the tumor cells immunostained more variable intensity (weak to moderate) in over half of HSA cases (8 of 11; Table 3). However, all (5 of 5) tumors had statistically significant (P value = .00098, Table 4) higher mRNA expression compared to control spleen. Even though staining intensity was more variable within the tumor cells compared to normal ECs, the higher mRNA expression in the HSAs could reflect an overall higher number of CD34-positive tumor cells within the HSAs compared to the number of ECs in normal spleen or it could be the result of posttranscriptional regulation.
CD45 cytoplasmic immunoreactivity was observed rarely in red pulp sinusoidal cells consistent with leukocytes in the normal canine spleen, but CD45 staining was not observed in normal ECs (Figure 1E) nor in the tumor cells of all HSA cases (Figure 1F). However, CD45 mRNA expression was lower in the tumors compared to normal spleen suggesting that the tumor cells outnumbered and/or displaced the leukocytes lowering the CD45 mRNA levels even further in the tumors (Table 4).
The immunoreactivity method for CD133 could not be successfully optimized in the canine. Since no alternative antibodies suitable for IHC were identified, we were not able to analyze CD133 immunoreactivity. The mRNA expression of CD133 was lower in the tumors (4 of 5) compared to normal spleen, but average fold change was not statistically significant (Table 4).
Expression of EC Markers and Tumor Suppressor Protein p53
Staining of CD31 and FVIII-RA was observed both in normal vascular endothelium (CD31: Figure 2A, FVIII-RA: Figure 2C) and tumor cells (CD31: Figure 2B, FVIII-RA: Figure 2D). Although CD31 membrane and cytoplasmic immunoreactivity was moderate in normal splenic vascular and sinusoidal endothelium (Figure 2A), tumor cells showed increased immunoreactivity as well as an overall increased number of positive cells (Figure 2B, Table 3). FVIII-RA membrane immunoreactivity was strong in normal ECs. In most HSA cases (Table 3), FVIII-RA immunoreactivity in tumors was weak to moderate and highly variable within and between tumors. The mRNA expression for both CD31 and FVIII-RA paralleled the IHC results (Table 4).
Nuclear p53 immunoreactivity was observed in normal vascular endothelium (Figure 2E) and in the tumor cells (Figure 2F) of most HSA cases (10 of 11; Table 3). The immunoreactivity intensity in tumor cells (negative to moderate) was generally less than normal endothelium (moderate). The mRNA expression for p53 was also lower in most tumors (3 of 5) compared to normal spleen, but the difference was not statistically significant (Table 4).
The immunoreactivity method for VEGF-R2 and myeloid lineage marker (CD14) could not be successfully optimized in canines, and since no alternative antibodies suitable for IHC were identified, we were not able to analyze for these markers. Both VEGF-R2 and CD14 mRNA expression was higher in tumors compared to normal spleens, and this difference was statistically significant for CD14 (P = .0129, Table 4). Neither the type of HSAs (cavernous vs solid) nor breed of canine tracked with any IHC marker or their respective mRNA expression results.
Comparison of Canine IHC Results With Mice and Humans
IHC staining results for HSC markers, EC markers, a myeloid lineage marker, and a tumor suppressor protein in human and mouse HSA specimens were previously reported. 12,13 To identify and highlight differences and similarities between the immunoreactivity results seen in canine HSA cases with those seen in human and mouse tumors, the immunoreactivity results for all 3 species are summarized in Table 5. Immunoreactivity results for CD133 13 and CD117 for mouse specimens were not available due to technical difficulties in optimizing the immunoreactivity technique (personal communication).
Comparison of Staining for HSC, EC, and Myeloid Markers and p53 in HSAs across Species.

Abbreviations: EC, endothelial cells; FVIII-RA, factor VIII–related antigen; HSA, hemangiosarcomas; HSC, hematopoietic stem cell; VEGF-R2, vascular endothelial growth factor receptor 2.
a Scoring for immunostaining. The staining intensity was graded as W, weak; M, moderate, and S; strong for normal EC and neoplastic cells. Since the intensity was not reported in detail for mouse tissues, 13 it was determined for each staining upon the discussion with the pathologist (T.A.C.).
b Results of spontaneous tumors, nongenotoxic chemically induced tumors, and genotoxic chemically induced tumors were combined as there was little difference between them.
c NA, not available.
d Of 12 human hemangiosarcoma samples, only one specimen showed scattered CD133-positive cells with strong intensity. 12
e Staining in the normal ECs ranged from weak to strong, depending on the tissue. In general, the staining was quite weak in the normal ECs of the liver though quite strong for the tumors within the same tissue (ie, the tumors in liver stained more intensely than the adjacent normal endothelium).
f Of 49 mouse hemangiosarcoma samples, only one sample was positive for FVIII-RA. 13
Presence of tumor cell immunoreactivity was not consistent for CD117 (HSC marker), CD45 (HSC marker), FVIII-RA (late-stage EC marker), and p53 across the species (Table 5). For example, in humans, CD117 immunoreactivity with moderate-to-strong intensity was observed in a majority of HSA cases (8 of 12). 12 In contrast, only weak CD117-positive immunoreactivity was observed in a few canine HSA samples (3 of 11; Table 3). CD45-positive immunoreactivity with strong intensity was observed in a majority of human HSA cases (7 of 9). 12 In contrast, CD45 staining was negative in all canine (Table 3 and Figure 1F) and mouse HSAs. 13 Factor VIII-RA was positive in all human 12 and canine HSA cases (Table 3 and Figure 2D), while it was negative in most mouse HSA samples. 13 p53 was positive for all cases of human HSA 12 and most canine HSA specimens (10 of 11; Table 3); however, p53 positivity was only observed in some samples of mouse HSAs (7 of 45). 13
Immunoreactivity for CD34 (HSC marker) and CD31 (EC marker) was observed in most HSA samples across all 3 species, however, immunoreactivity intensity in tumor cells compared to the internal vascular controls or normal ECs differed across the species (Table 5). In mice and humans, intensity of tumor cell immunoreactivity for CD34 and CD31 was compared to the adjacent normal vascular endothelium within the same specimen 12,13 ; whereas splenic tissue obtained from healthy canines was utilized as a source for basal level staining. In most mouse HSAs, tumor cells labeled for CD34 with increased intensity compared to the adjacent normal vascular endothelium. 13 The immunoreactivity intensity of CD34 in human HSA cases was similar to the internal vascular controls. 12 In the present study, in over half of the canine HSAs, tumor cells immunostained for CD34 with less intensity compared to normal ECs (Table 3). The immunoreactivity qualities of CD31-positive tumor cells in mouse and human HSAs were similar to that of the adjacent normal vascular endothelium. 12,13 However, in canines, tumor cells generally showed more intense immunoreactivity (ranging from weak to strong) compared to normal ECs (moderate; Figure 2A and B and Table 3).
Discussion
Based on the 11 HSA cases examined, the current study results suggest that the majority of canine HSAs are composed of cells expressing CD34, CD31, VEGF-R2, FVIII-RA, CD14 and p53, with lesser number of cells expressing CD117 and CD133. Considering that (1) some tumor ECs showed CD117 immunoreactivity and (2) immunoreactivity of FVIII-RA was relatively focal and weak compared to normal ECs, canine HSAs likely arise from EPCs with hematopoietic origin rather than differentiated ECs. This is the first study to comprehensively characterize markers of canine HSAs, but there are 2 major limitations to this study that should be addressed in future studies; namely small sample size and the use of archival tissue that has been stored in fixative for variable times. To overcome these limitations, future studies should be prospective and control time in fixative and collecting sufficient surrounding normal tissue to provide an internal control for each animal.
Markers utilized in the current study have been used in canine HSA studies elsewhere. For instance, CD31 and FVIII-RA have been used diagnostically and are in many publications. 22 –25 The VEGF-R2 IHC assay in canine tissue was not successful in our laboratory, but a recent paper 16 (which was published after our work was done) was able to get VEGF-R2 to work in canine tissue. The other markers to our knowledge have not been published in canine HSAs, but they have been used in other tumors and/or organ pathology, including CD117 (c-kit) in mast cell tumors and gastrointestinal-stromal tumors, 21 CD45 in lymphomas and myxomatous mitral valve disease, 26,27 and p53 and CD34 in many tumor types. 28,29
The IHC results for HSC markers (CD133, CD117, CD34, and CD45), EC markers (VEGF-R2, CD31, and FVIII-RA), a myeloid cell marker (CD14), and a tumor suppressor protein (p53) were compared across 3 species (mouse, 13 human, 12 and canine; Table 5). Although CD34 staining was observed in HSAs across all species, human HSAs derived from surgical specimens had more robust staining of remaining HSC markers (CD117 and CD45) than canine or mouse tumors. Most of the human HSA cases were positive for CD117 with moderate-to-strong immunoreactivity intensity. 12 CD117 was only weakly positive in some canine HSA specimens. Only human HSA cases were strongly positive for CD45, 12 with none of the mouse 13 or canine tumor cells having immunoreactivity. These results indicate human HSAs are likely to be derived from and composed of cells that have less differentiation compared to canine and mouse tumors. It is not clear if the lack of CD45 staining in canine and mouse tumor cells is caused by expression below the level of IHC detection, malignant tumor heterogeneity, or if the tumor cell of origin did not express CD45.
Results of immunoreactivity for CD117 in the present study were somewhat different from previous literature reporting CD117 staining in canine HSA samples. 17,19,20 Although scoring of immunoreactivity was not shown, Fosmire et al 19 reported that CD117 staining was detected in all HSA samples tested. Similarly, Sabattini et al 17 or Chen et al 20 reported CD117 immunoreactivity in a majority of canine HSA cases. However, Adachi et al 16 reported that only 30% of canine HSAs had weak CD117 staining, which more closely parallels the current observations. Although the Fosmire et al 8 and Adachi et al 16 studies utilized different CD117 antibodies, the Sabattini et al 17 and Chen et al 20 studies used the same antibody as the present study (A4502; Dako, Carpinteria, CA). Differences in instrumentation and detection systems utilized between the laboratories may have accounted for the disparate IHC results for this marker. We were able to detect low CD117 mRNA expression levels in the canine tumors, which suggests that the current CD117 IHC assay maybe less sensitive compared to the other investigators’ methods. Alternative methodology, such as in situ hybridization (ISH) for CD117 mRNA expression in the tumor samples, may assist with further tissue characterization of this marker in canine HSAs.
In the current study, CD45 cytoplasmic immunoreactivity was observed rarely in red pulp sinusoidal cells in normal canine spleen. Canine CD45 has been shown to have 2 isoforms which are variably expressed in leukocytes and that only upon culture stimulation did CD45 expression increase in lymphocytes. 30 A more recent paper showed increased CD45 positive cells in the skin of dogs with symptomatic Leishmaniasis which is an immune stimulated state. 31 Thus, the rare to absent CD45 immunostaining in the control spleens may reflect a nonimmune stimulated state.
The staining pattern of HSC markers (CD133, CD117, and CD34) in the present study differed from in vitro canine HSA cell lines since the expression of CD133 and CD117 was consistently detected across monolayer and sphere-culture canine HSA cell lines. 14,15 While CD133, CD117, and CD34 demonstrated a tendency to downregulate expression in monolayer culture for extended periods of time, 14 increased expression levels of these markers were observed within 2 weeks and sustained up to 11 to 12 months in sphere-cultured cell lines. 15 Thus, in vitro microenvironment factors may contribute to different expression levels and patterns of HSC markers between canine HSA cell lines in vitro compared to canine HSA tumors in vivo. Additionally, while CD45 staining was observed in some cases of in vitro canine HSA cell lines, 14 CD45 mRNA expression was very low in canine HSA samples in the current study. There are several possible explanations, one being that the tumor ECs at least in canines might have lost and/or downregulated CD45 protein expression during the pathogenesis of the disease. Alternatively, the complex interplay of inflammatory cell signaling with ECs in vivo 15,32 is not present in vitro which could affect marker expression.
Some similarities and differences were also identified in the immunoreactivity of EC markers across species. Although the immunoreactivity method for VEGF-R2 could not be successfully optimized in canines, VEGF-R2 mRNA expression was higher in tumors compared to normal spleens. Our results correlated with previously reported data. Tamburini et al 33 reported VEGF-R2 expression in canine HSA cell lines by immunoblotting, and Adachi et al 16 reported positive VEGF-R2 IHC staining in all canine HSAs evaluated and that 90% were high expressers. Additionally, a recent genomic study of canine HSAs reported that similar to human HSAs, VEGF-R2 and KDR genes were recurrently gained. 18
Immunoreactivity of FVIII-RA, which is considered to be the last marker expressed among the 3 EC markers, 34,35 was negative in most mouse HSAs 13 but positive in all human 12 and canine HSAs. These results indicate that, although mouse tumor cells are arrested at a stage prior to complete differentiation, tumor cells in humans and canines are likely to retain the ability to differentiate further.
FVIII-RA staining was predominantly weak in canine HSAs compared to the strong staining present in the normal spleen ECs. A recent investigation compared the staining of differentiated EC markers, CD31, and von Willebrand Factor (vWF, also known as FVIII-RA), to the degree of cellular atypia in canine HSA subtypes. 24 Cellular atypia negatively correlated with the immunostaining of both markers without relation to canine HSA subtype. Interestingly, CD31 but not vWF/FVIII-RA was detected in the tumor cells with the most severe atypia suggesting that cellular atypia along with EC differentiation markers can be used to identify differentiation levels in canine HSAs. 24
The mRNA expression of a myeloid lineage marker, CD14, was higher in all (5 of 5) canine tumors compared to normal spleen. Although CD14 immunoreactivity was negative in most human HSAs, a few cases showed patchy positivity for CD14 in tumor ECs. 12 The CD14 immunoreactivity was completely negative in mouse HSAs. Multiplex ISH or ISH in combination with IHC will be needed to confirm the cell of origin (neoplastic EC and/or immune cell).
In the current study, most canine HSAs showed positive immunoreactivity for p53, which is consistent with previous reports 36 –38 and similar to human HSAs. 12 In contrast, the presence of p53 immunoreactivity was less frequently observed in mouse HSAs (positive in only 7 of 45 samples). 13 Interestingly, p53 protein expression in normal tissues may also differ across species; in humans, p53 nuclear immunoreactivity was negative in many normal tissues, including liver, 39,40 and positivity was detected in some peripheral blood and lymphoid tissues including spleen. 40,41 In mice, p53 was not observed in normal tissues, including liver, spleen, and thymus. 42 In the current study, p53 immunoreactivity was observed in the normal canine spleen. Additionally, different tissues may have different expression regulation for p53 in each species. Transgenic mice carrying mutated p53 genomic fragments showed high levels of p53 protein expression in spleen, thymus, lymph nodes, and ovaries, lesser levels in lung and testis, and very low levels in liver. 42 In a recent whole-exome sequencing study in canine HSAs, TP53 was found to be one of the most frequent mutated genes. 43
In summary, the majority of canine HSAs that we examined were composed of cells expressing CD34, CD31, FVIII-RA, VEGF-R2, CD14, and p53, with a small number of cells expressing CD117 and CD133. Similar to mouse and human, canine HSAs are likely derived from HSCs or EPCs. However, the cascade of HSA development from EPCs appears to be variable across species; the pathogenesis of canine HSAs, like mouse, may not be relevant to humans. Human HSAs had more robust staining of HSC markers, indicating that they are comprised of tumor cells that are less differentiated than those in canine and mouse tumors. Human and canine HSAs expressed a late-stage EC maturation marker (FVIII-RA), whereas mouse HSAs were negative, suggesting that human and canine tumors may retain greater differentiation potential than mouse tumors. Together, with previous studies evaluating human and mouse HSAs, 12,13 the current study suggests that caution is needed when discussing the risk of sarcomagenic potential of chemicals to humans when testing is done using animal models.
Footnotes
Authors’ Note
Satoko Kakiuchi-Kiyota and Leslie A. Obert contributed equally to this work.
Acknowledgments
The authors gratefully acknowledge Alan Opsahl (Pfizer) for his assistance with the preparation of the microscopic figures for this manuscript. The authors also express their gratitude to Cynthia Hutchinson (Auburn University) and Renee Huynh (Pfizer) for retrieving the paraffin blocks and cutting sections of canine tumor and normal tissues. Although the immunoreactivity method for CD117 in mouse tissues could not be successfully optimized, the authors sincerely thank the IHC Lab of the Pathology Support Group of the Cellular and Molecular Pathology Branch of the National Toxicology Program, NIEHS, for their extensive efforts and time attempting to optimize the immunoreactivity method.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.K.-K., L.A.O., and M.D.R. are employees of Genentech/Roche, GlaxoSmithKline, and Sarepta Therapeutics, respectively. They were previously employed by Pfizer. S.X., T.M.C., J.M.K., D.M.C., and J.C.C. are employees of Pfizer. T.A.C. is an employee of EPL. S.M.C. is an employee of University of Nebraska Medical Center, and is a consultant for Pfizer but on issues not related to the present study. He did not receive payment for activities related to the present project. R.C.C. is an employee of Auburn University, and has not served as a consultant for activities related to the present project. Pregabalin for the treatment of neuropathic pain was developed by Pfizer, and it was reported that an increased incidence of a single tumor type, HSAs, was seen in male and female mice, but not in rat, in 2-year carcinogenicity studies. EPL has been involved with numerous toxicity studies that have involved the evaluation of vascular tumors in various species, including mice and canines.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from Pfizer Worldwide Research & Development.
