Abstract
It is unclear whether the process of spontaneous and chemically induced hemangiosarcoma and hemangioma formation in mice involves the transformation of differentiated endothelial cells (ECs) or recruitment of multipotential bone marrow–derived hematopoietic stem cells or endothelial progenitor cells (EPCs), which show some degree of endothelial differentiation. In the present study, immunohistochemical staining for hematopoietic stem cell markers (CD45 and CD34), EC markers (vascular endothelial growth factor receptor 2 [VEGFR2], CD31, and factor VIII–related antigen), and a myeloid lineage marker (CD14) was employed to better define the origin of hemangiosarcomas and hemangiomas in mice. Staining was negative for CD45, factor VIII–related antigen, and CD14 and positive for CD34, VEGFR2, and CD31, indicating that mouse hemangiosarcomas and hemangiomas are composed of cells derived from EPCs expressing CD34, VEGFR2, and CD31 but not factor VIII–related antigen. The lack of CD45 expression suggests that mouse vascular tumors may arise from EPCs that are at a stage later than hematopoietic stem cells. Since factor VIII–related antigen expression is known to occur later than CD31 expression in EPCs, our observations may indicate that these tumor cells are arrested at a stage prior to complete differentiation. In addition, myeloid lineage cells do not appear to contribute to hemangiosarcoma and hemangioma formation in mice.
Keywords
Introduction
Hemangiosarcomas are aggressive malignant tumors characterized by the presence of irregular vasoformative structures consisting of multilayered or solid clusters of endothelial cells (ECs; Hardisty et al. 2007). The occurrence of hemangiosarcomas is rare in humans, usually with no known cause. The only known causes of hemangiosarcomas in humans are a few rare genetic disorders and exposure to certain genotoxic agents, such as vinyl chloride and thorotrast (Cohen et al. 2009). Hemangiosarcomas have recently become the focus of considerable attention because a wide range of nongenotoxic chemical agents and pharmaceutical agents, such as peroxisome proliferator–activated receptor (PPAR) agonists, are reported to increase the incidence of hemangiosarcomas, primarily in mice as demonstrated in 2-year carcinogenicity studies (Cohen et al. 2009). We have proposed that the sarcomagenic PPARγ agonist, troglitazone, increased EC proliferation, and decreased apoptosis in mice led to an elevation in spontaneous mutations, resulting in hemangiosarcoma formation (Kakiuchi-Kiyota et al. 2009; Kakiuchi-Kiyota et al. 2011a, 2011b).
It is not well understood whether hemangiosarcomas originate from differentiated ECs that undergo mutations which endow them with malignant potential or from transformed endothelial progenitor cells (EPCs; Lamerato-Kozicki et al. 2006). Although the origin of EPCs and the course of differentiation to mature ECs are not well understood, there are numerous studies showing that adult bone marrow and peripheral blood contain EPCs that are capable of differentiating into mature ECs (Hristov and Weber 2004; Schatteman and Awad 2004; Urbich and Dimmeler 2004). It has been proposed that EPCs are derived from bone marrow hematopoietic stem cells. These hematopoietic stem cells have been demonstrated to express hematopoietic stem cell markers, including CD133, CD45, and CD34 (Hristov and Weber 2004; Urbich and Dimmeler 2004). Among these markers, CD133 is expressed in more immature stem cells, such as hematopoietic stem cells and/or early EPCs located in the bone marrow or in blood just after migration into the circulation (Hristov and Weber 2004). Purified CD133-positive cells can differentiate to ECs in vitro (Gehling et al. 2000; Kanayasu-Toyoda et al. 2003). Expression of CD133 is decreased in late progenitor cells and absent in mature ECs (Handgretinger et al. 2003; Hristov and Weber 2004). In contrast, expression of CD34 is not restricted to hematopoietic stem cells or early EPCs but is also present in late EPCs and mature ECs at lower levels (Hristov and Weber 2004; Urbich and Dimmeler 2004). CD45 is expressed in hematopoietic stem cells and in both lymphoid and myeloid cell lineages from progenitor to mature cells (Hermiston, Xu, and Weiss 2003). The course of CD45 expression from hematopoietic stem cells to EPCs is unclear.
Early EPCs located in the bone marrow or in blood just after migration into the circulation begin to express the EC marker, vascular endothelial growth factor receptor 2 (VEGFR2), in addition to CD133 and CD34 (Hristov and Weber 2004; Schatteman and Awad 2004; Urbich and Dimmeler 2004). Recently, it was reported that the frequency of circulating cells expressing CD133, CD34, and VEGFR2 collected from human peripheral blood was extremely low (approximately 84 per 106 mononuclear cells) and that >99% of these cells also expressed CD45 (Case et al. 2007). During differentiation from EPCs to mature ECs, the circulating EPCs lose CD133 expression and begin to express mature EC markers, such as CD31 and factor VIII–related antigen (or von Willebrand factor [vWF]), which is one of three components of factor VIII (Harach, Jasani, and Williams 1983; Hristov and Weber 2004; Schatteman and Awad 2004; Urbich and Dimmeler 2004). It has been proposed that VEGFR2 is the earliest EC marker that is expressed, followed closely by CD31 (Kanayasu-Toyoda et al. 2003; Hristov and Weber 2004).
Recently, the contribution of myeloid lineage cells in angiogenesis has been reported. Yoder et al. (2007) showed that cells expressing CD45 and CD14 (monocyte/macrophage cell surface antigen) also expressed EC markers, such as CD31, VEGFR2, and factor VIII–related antigen, and Ulex europaeus agglutinin-1 (UEA-1). Kim et al. (2009) demonstrated that macrophages (differentiated monocytes) that expressed both CD31 and F4/80 (macrophage cell surface antigen) directly contributed to angiogenesis.
In humans, Yonemori et al. (2011) reported that another hematopoietic stem cell marker c-kit expression was observed in hemangiosarcomas (15%). Although the sample number is small due to only case reports being available, CD45 expression has been shown to be negative in hemangiosarcomas (Lin et al. 1997; Forton, Van Parys, and Hertveldt 2005; Kuroda et al. 2009; Terada 2011). CD34, VEGFR2, CD31, and factor VIII–related antigen expression has been demonstrated in hemangiosarcomas and hemangiomas (Poblet, Gonzalez-Palacios, and Jimenez 1996; Breiteneder-Geleff et al. 1999; Itakura et al. 2008; Yonemori et al. 2011). Although the contribution of myeloid lineage cells to the formation of hemangiosarcomas in humans is unknown, coexpression of CD83 (dendritic cell marker) and EC markers (factor VIII–related antigen or UEA-1) was observed in infantile hemangiomas (Ritter et al. 2006). This suggests the participation of myeloid-derived cells in the formation of these benign tumors arising from ECs in humans. Canine hemangiosarcoma cell lines expressed c-kit, CD133, CD34, and in some cases, CD45 and CD14 (Lamerato-Kozicki et al. 2006). However, to the best of our knowledge, there is no literature regarding the expression of hematopoietic stem cell, EC, and myeloid lineage cell antigens in canine hemangiosarcomas/hemangiomas in vivo. In spontaneous hemangiosarcomas in Ink4c/p53-null mice, expression of CD34, VEGFR2, CD31, and factor VIII–related antigen was observed (Zindy et al. 2003); however, expression of CD133, CD45, and myeloid lineage cell antigens, such as CD14, in mouse hemangiomas or hemangiosarcomas has not been extensively investigated.
In the present study, we evaluated the staining of hematopoietic stem cell markers (CD133, CD45, and CD34), EC markers (VEGFR2, CD31, and factor VIII–related antigen), and a myeloid lineage cell marker (CD14) in spontaneous hemangiosarcomas and hemangiomas and in nongenotoxic and genotoxic chemically induced hemangiosarcomas and hemangiomas in mice. These tumors were also stained with the p53 tumor suppressor protein. These results contribute to the understanding of whether the recruitment of hematopoietic stem cells/EPCs and/or myeloid lineage cells are involved in hemangiosarcomas or hemangiomas in mice similar to that previously demonstrated in human hemangiosarcomas (Yonemori et al. 2011), canine hemangiosarcoma cell lines (Lamerato-Kozicki et al. 2006), and infantile hemangiomas in humans (Ritter et al. 2006) or whether differentiated ECs undergo mutations that result in the formation of tumors in mice.
Materials and Methods
Study Design
Paraffin blocks from B6C3F1 mice containing hemangiomas or hemangiosarcomas from various tissues were provided from the archives of the National Institute of Environmental Health Sciences (NIEHS) and Pfizer, Inc., and sectioned for immunohistochemical analysis of CD133, CD45, CD34, VEGFR2, CD31, factor VIII–related antigen, CD14, and p53. Chemical names, exposure routes, genotoxicity of chemicals, dose and duration of treatment, histopathological diagnosis, references, and archive information are described in Table 1. Sex and the number of hemangiosarcomas or hemangiomas for each tissue are described in Table 2.
Summary of long-term carcinogenicity and genetic toxicity studies.
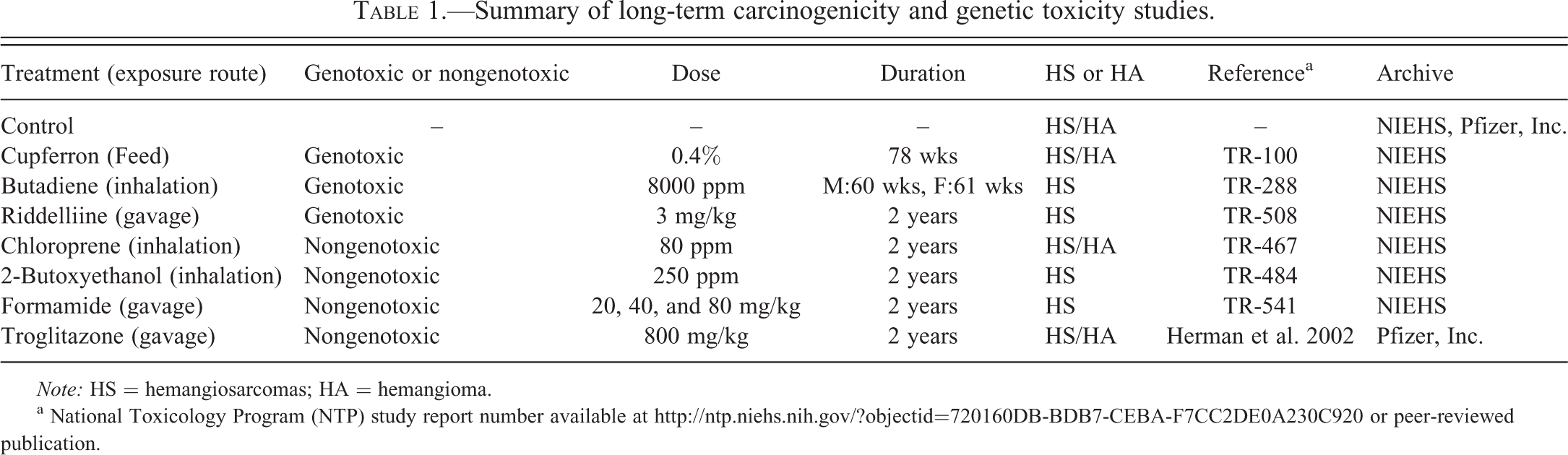
Note: HS = hemangiosarcomas; HA = hemangioma.
a National Toxicology Program (NTP) study report number available at http://ntp.niehs.nih.gov/?objectid=720160DB-BDB7-CEBA-F7CC2DE0A230C920 or peer-reviewed publication.
Summary of the tissues examined in the present study.
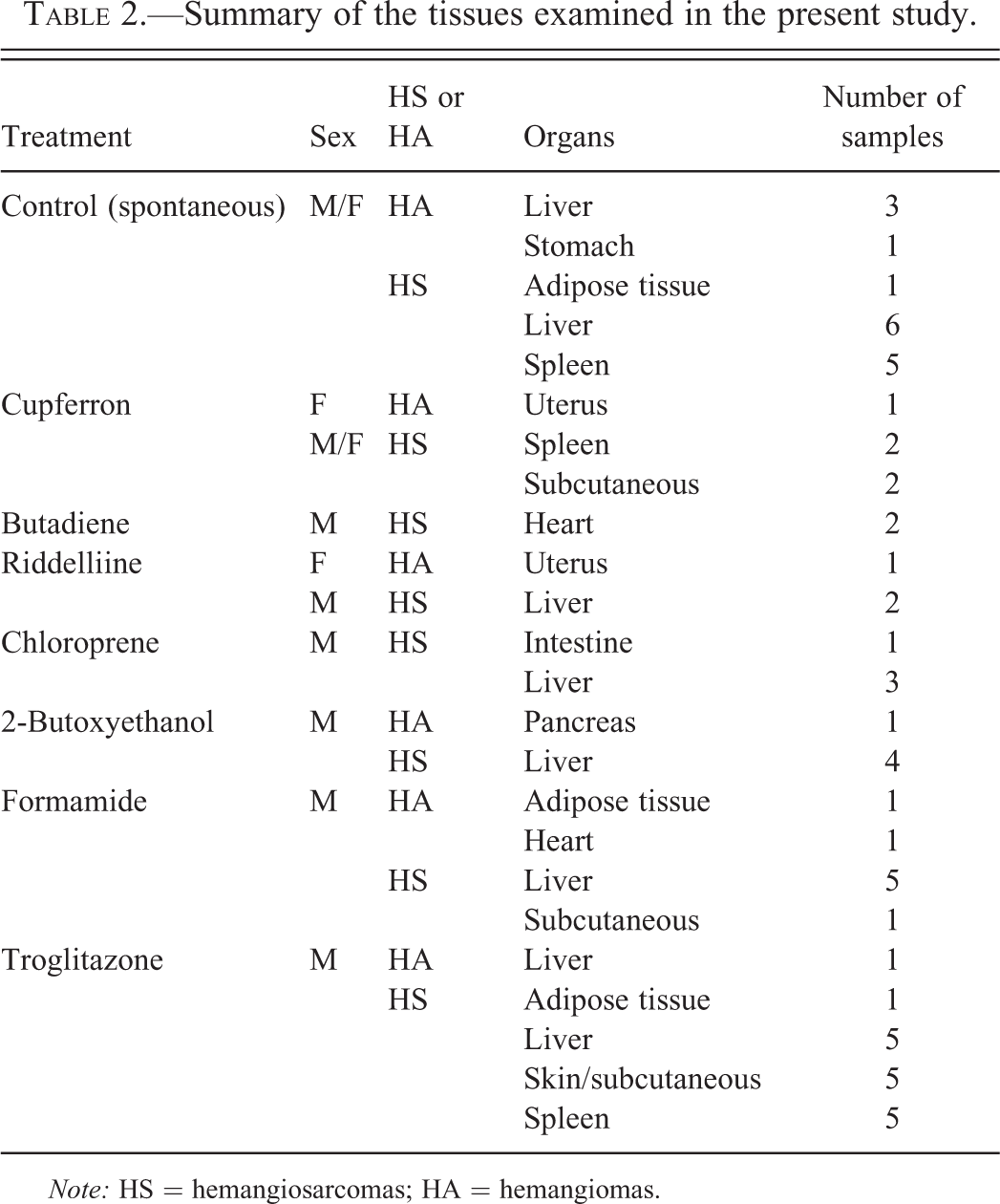
Note: HS = hemangiosarcomas; HA = hemangiomas.
Immunohistochemistry
Immunohistochemical procedures, including antibodies and negative controls for primary antibodies used in our study, are summarized in Table 3. Formalin-fixed and paraffin-embedded mouse tissues were deparaffinized and rehydrated through graded alcohols. Endogenous peroxidase activity was quenched with 3% H2O2, followed by heat-induced antigen retrieval using a Decloaker pressure chamber (Biocare Medical, Concord, CA) for 5 min at 120
Antibodies and negative controls used for immunohistochemistry.
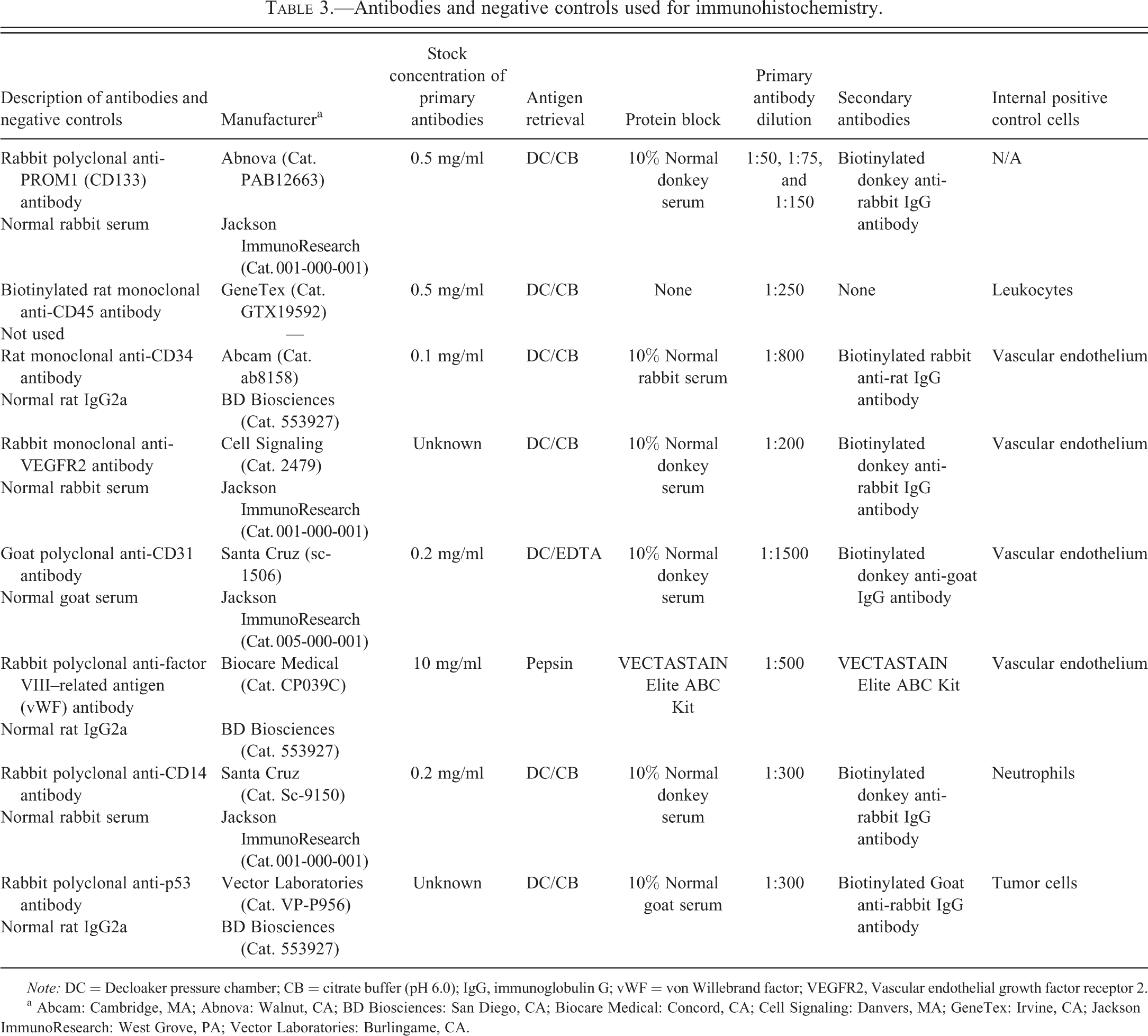
Note: DC = Decloaker pressure chamber; CB = citrate buffer (pH 6.0); IgG, immunoglobulin G; vWF = von Willebrand factor; VEGFR2, Vascular endothelial growth factor receptor 2.
a Abcam: Cambridge, MA; Abnova: Walnut, CA; BD Biosciences: San Diego, CA; Biocare Medical: Concord, CA; Cell Signaling: Danvers, MA; GeneTex: Irvine, CA; Jackson ImmunoResearch: West Grove, PA; Vector Laboratories: Burlingame, CA.
Immunohistochemical Evaluation
Immunohistochemically stained sections were semiquantitatively evaluated for the presence/absence of staining, the percentage of tumor cells expressing positive staining, and the overall staining intensity of positively stained tumor cells, and were subsequently classified into one of the following three grades: (−) = no positive staining in any of the tumor cells; (+) = positive staining in ≤25% of the tumor cells and/or minimal to mild staining intensity; and (++) = positive staining in >25% of the tumor cells and/or moderate to marked staining intensity.
Statistical Analysis
All statistical analyses were performed using Prism 5.01 (GraphPad software, La Jolla, CA). Fisher’s exact test was used to examine the correlation of staining between spontaneous and nongenotoxic chemically induced hemangiosarcomas, between spontaneous and genotoxic chemically induced hemangiosarcomas, between nongenotoxic and genotoxic chemically induced hemangiosarcomas, between spontaneous hemangiomas and chemically induced hemangiomas, and between hemangiomas and hemangiosarcomas. A probability of chance less than 5% was considered as significant.
Results
Hemangiosarcomas and hemangiomas collected from mice treated with or without nongenotoxic or genotoxic chemicals were immunostained with hematopoietic stem cell markers (CD133, CD45, and CD34), EC markers (VEGFR2, CD31, and factor VIII–related antigen), and a myeloid lineage cell marker (CD14) to investigate whether these tumors originate from differentiated ECs or stem cells/EPCs in mice. We also investigated the expression of the p53 tumor suppressor protein in these tumors. For each immunostain, no major differences were observed between males and females or between different tissues containing the vascular tumors, in terms of staining pattern and/or intensity (data not shown).
Hematopoietic Stem Markers
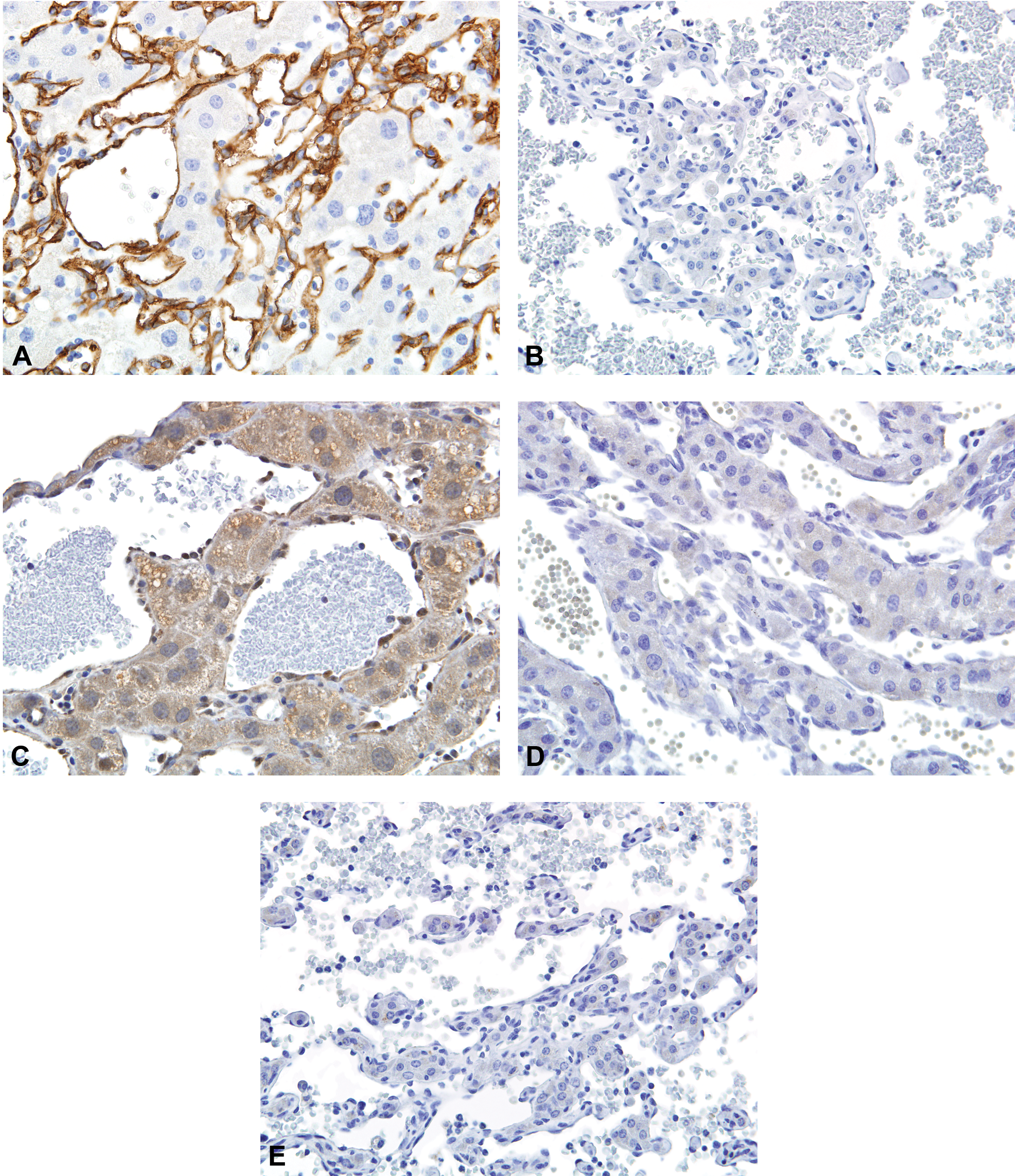
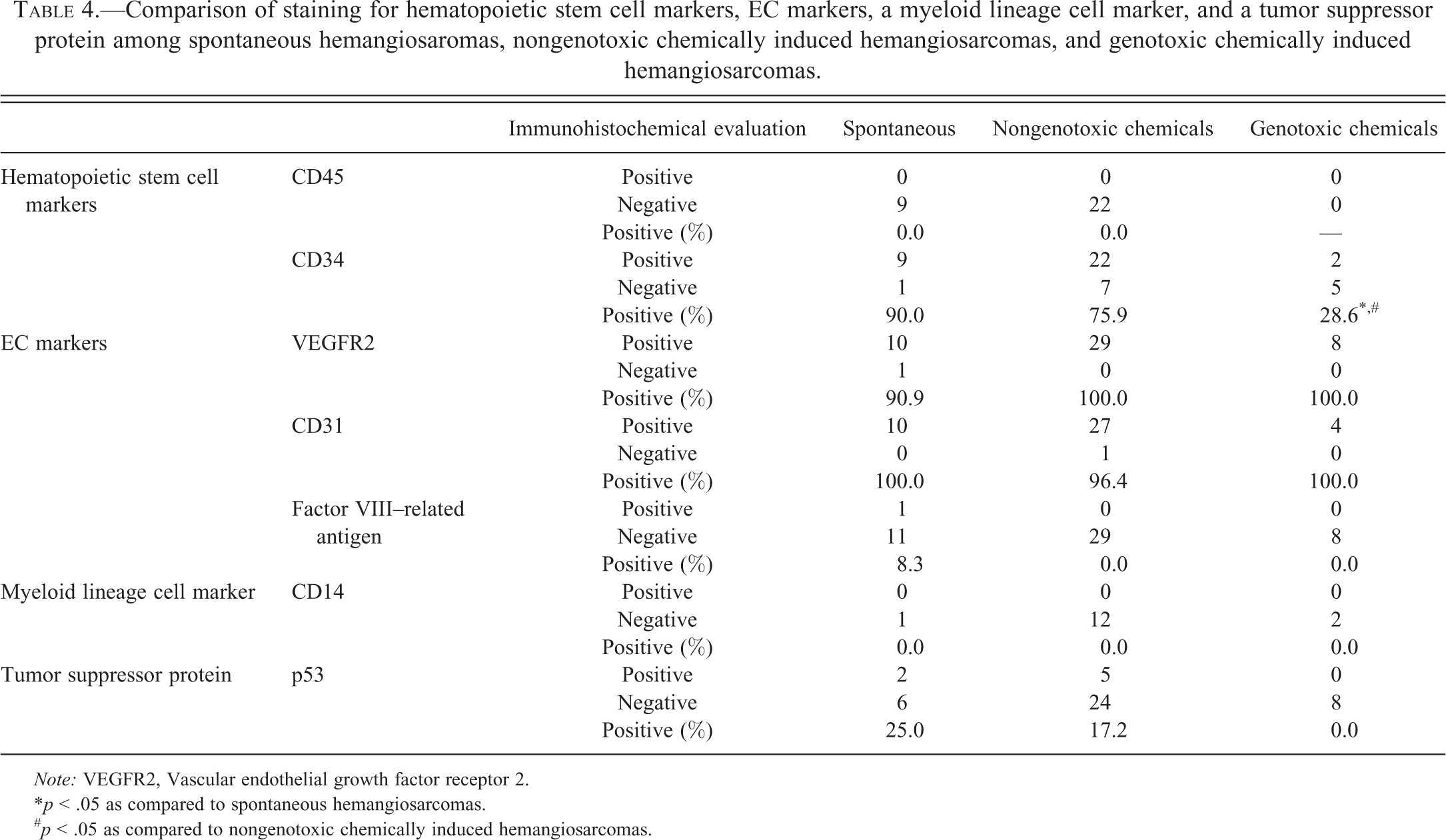
All hemangiosarcomas and hemangiomas were negative for CD45 staining (Figures 1A and 4A, Tables 4 and 5), despite positive membrane staining of internal control cells (leukocytes) within these sections. In contrast, both spontaneous and chemically induced hemangiosarcomas and hemangiomas were positive for CD34 staining, exhibiting intense membrane staining of a majority of the tumor cells (Figures 2A and 3A, Tables 4 and 5). Expression of CD34 is considered to diminish along the cascade of development from early EPCs through differentiated ECs (Hristov and Weber 2004; Urbich and Dimmeler 2004). In most instances in our study, tumor cells stained with increased intensity compared to the adjacent normal vascular endothelium (data not shown). While many spontaneous and nongenotoxic chemically induced hemangiosarcomas were positive for CD34 staining (90% and 76%, respectively), there was a significant decrease in the percentage of CD34 positively stained tumors (29%) among the genotoxic chemically induced hemangiosarcomas (Figure 2B, Table 4). Unfortunately, the staining method for CD133 could not be successfully optimized, and since no alternative antibodies suitable for immunohistochemistry were available, we were not able to analyze CD133 staining.
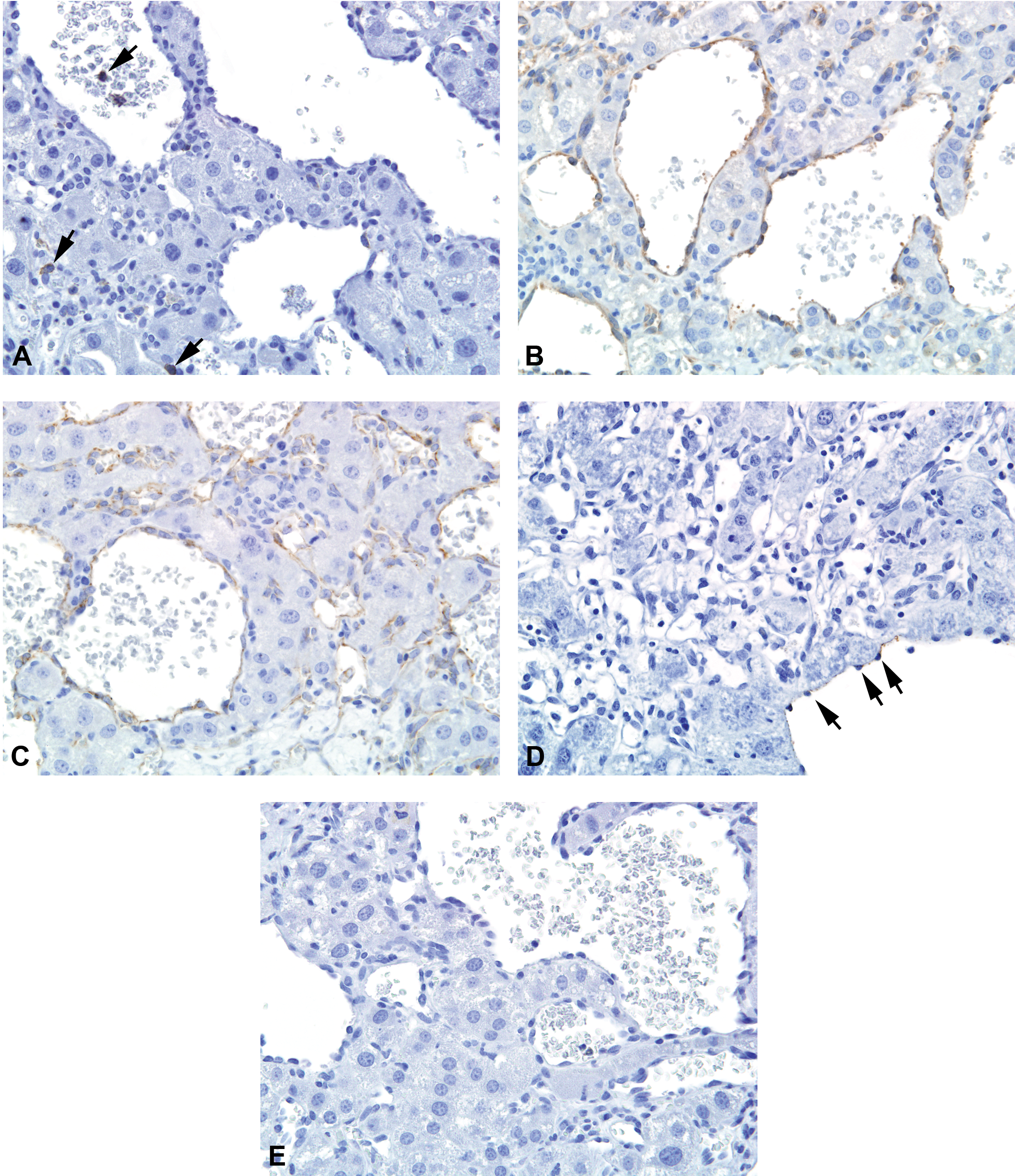
Immunostains for CD45 (A), vascular endothelial growth factor receptor 2 (VEGFR2) (B), CD31 (C), factor VIII–related antigen (D), and CD14 (E) in spontaneous and chemically induced hemangiosarcomas in mice. Tumor cells were positive for VEGFR2 (B) and CD31 (C). In contrast, staining for CD45 (A), factor VIII–related antigen (D), and CD14 (E) was negative in these tumors despite the positive staining of internal positive control cells (CD45: leukocytes, factor VIII–related antigen: endothelium in normal vasculature, and CD14: neutrophils) indicated by arrows (data not shown for CD14). Original magnification: 40×.
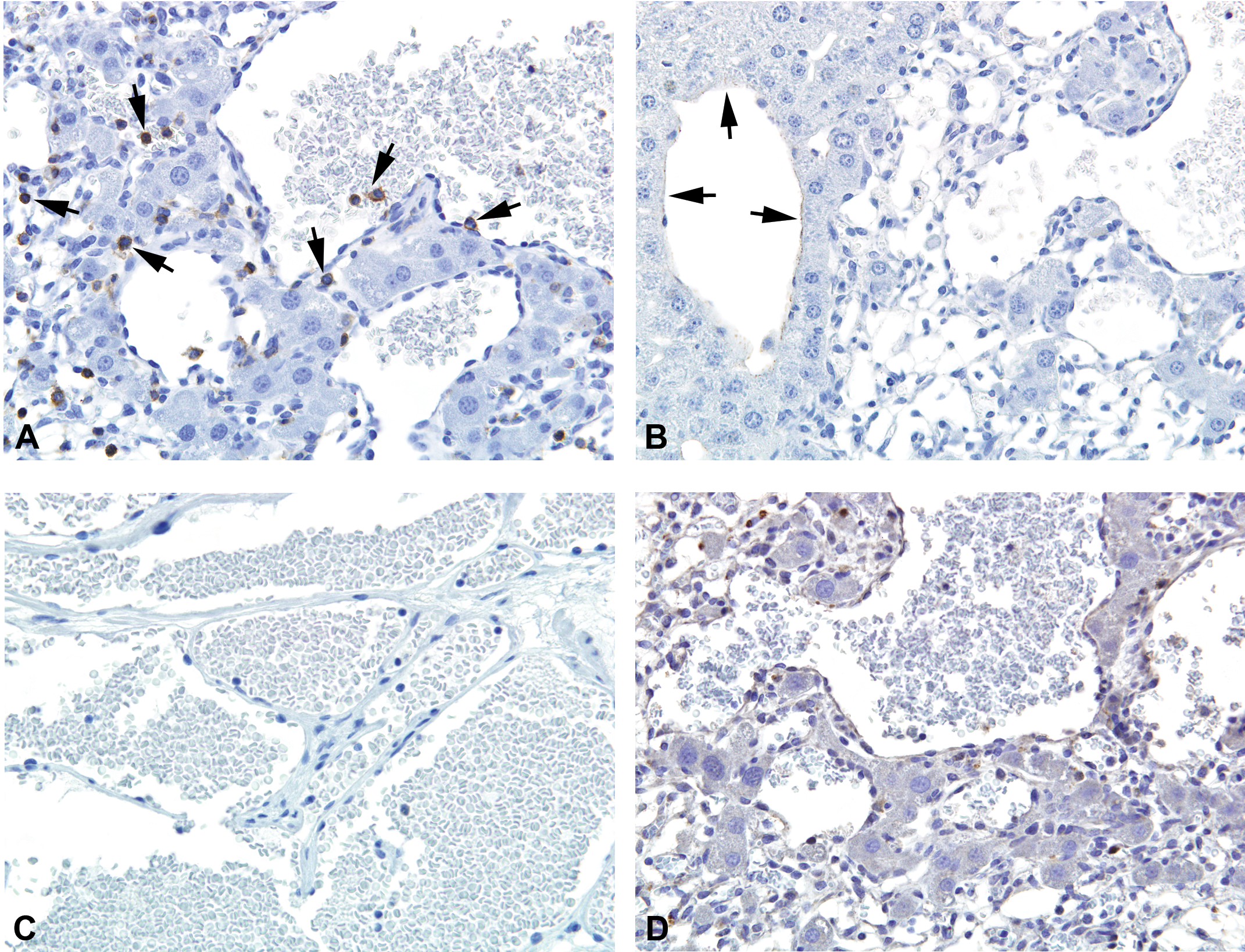
Immunostains for CD34 (A and B) and p53 (C, D, and E) in mouse hemangiosarcomas induced by formamide (A, C), 2-butoxyethanol (D), or cupferron (B, E). Many spontaneous and nongenotoxic chemically induced hemangiosarcomas were positive for CD34 staining (A). CD34 staining was negative in 5 of 7 genotoxic chemically induced hemangiosarcomas (B) although the positive staining was observed in the adjacent normal vascular endothelium (data not shown). Sections stained with p53 exhibited a nonspecific hepatocellular cytoplasm; however, specific intense nuclear positive staining was observed in some spontaneous and chloroprene- and formamide-induced hemangiosarcomas (C). p53 staining was negative in troglitazone- and 2-butoxyethanol-induced hemangiosarcomas (D). p53 staining was also negative in genotoxic chemically induced hemangiosarcomas (E). Original magnification: 40×.
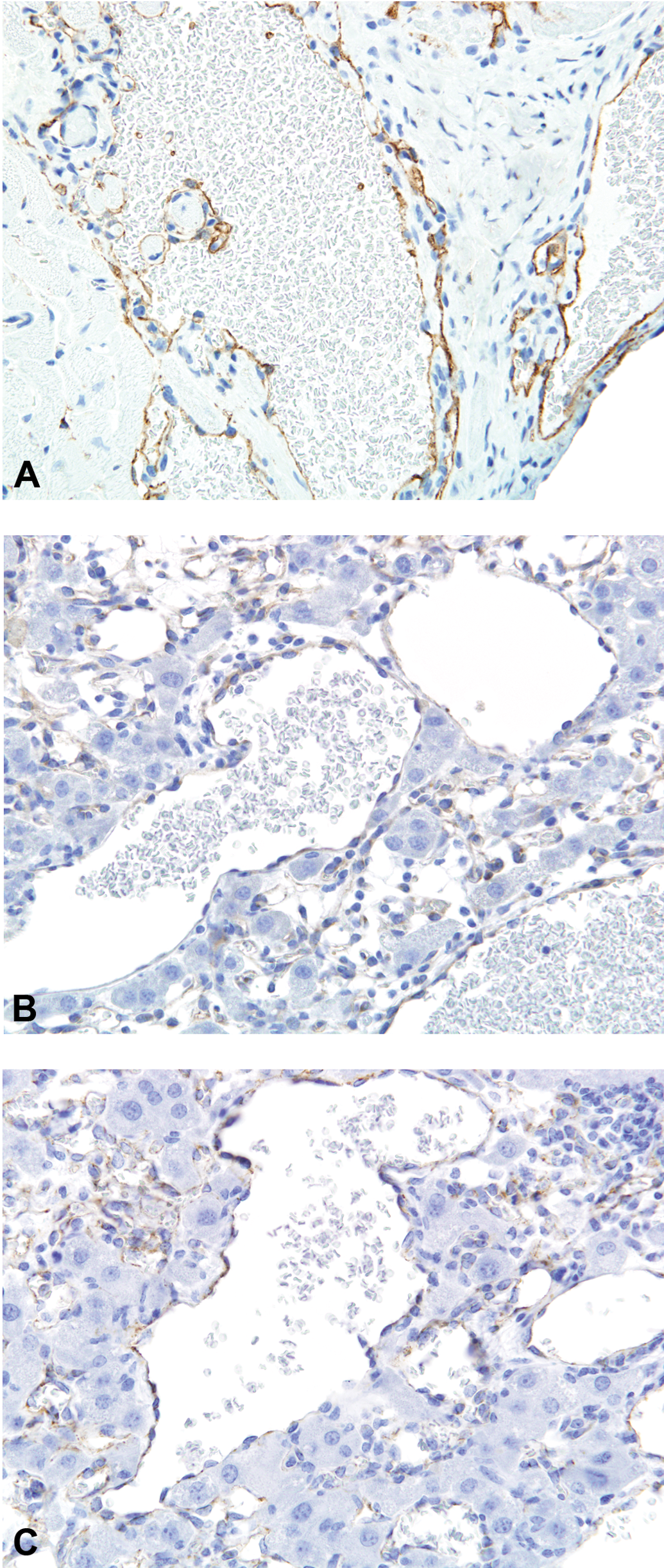
Immunostains for CD34 (A), vascular endothelial growth factor receptor 2 (VEGFR2) (B), and CD31 (C) in spontaneous and chemically induced hemangiomas in mice. Tumor cells were positive for CD34 (A), VEGFR2 (B), and CD31 (C). Original magnification: 40×.
Immunostains for CD45 (A), factor VIII–related antigen (B), CD14 (C), and p53 (D) in spontaneous and chemically induced hemangiomas in mice. Staining for CD45, factor VIII–related antigen, and CD14 was negative in these tumors despite the positive staining of internal positive control cells (CD45: leukocytes, factor VIII–related antigen, endothelium in normal vasculature, and CD14: neutrophils) indicated by arrows for CD45 (A) and factor VIII–related antigen (B) (data not shown for CD14). p53 staining was also negative in spontaneous and chemically induced hemangiomas (D). Original magnification: 40×.
Comparison of staining for hematopoietic stem cell markers, EC markers, a myeloid lineage cell marker, and a tumor suppressor protein among spontaneous hemangiosaromas, nongenotoxic chemically induced hemangiosarcomas, and genotoxic chemically induced hemangiosarcomas.
Note: VEGFR2, Vascular endothelial growth factor receptor 2.
*p < .05 as compared to spontaneous hemangiosarcomas.
# p < .05 as compared to nongenotoxic chemically induced hemangiosarcomas.
Staining for hematopoietic stem cell markers, EC markers, a myeloid lineage cell marker, and a tumor suppressor protein in spontaneous hemangiomas and chemically induced hemangiomas.
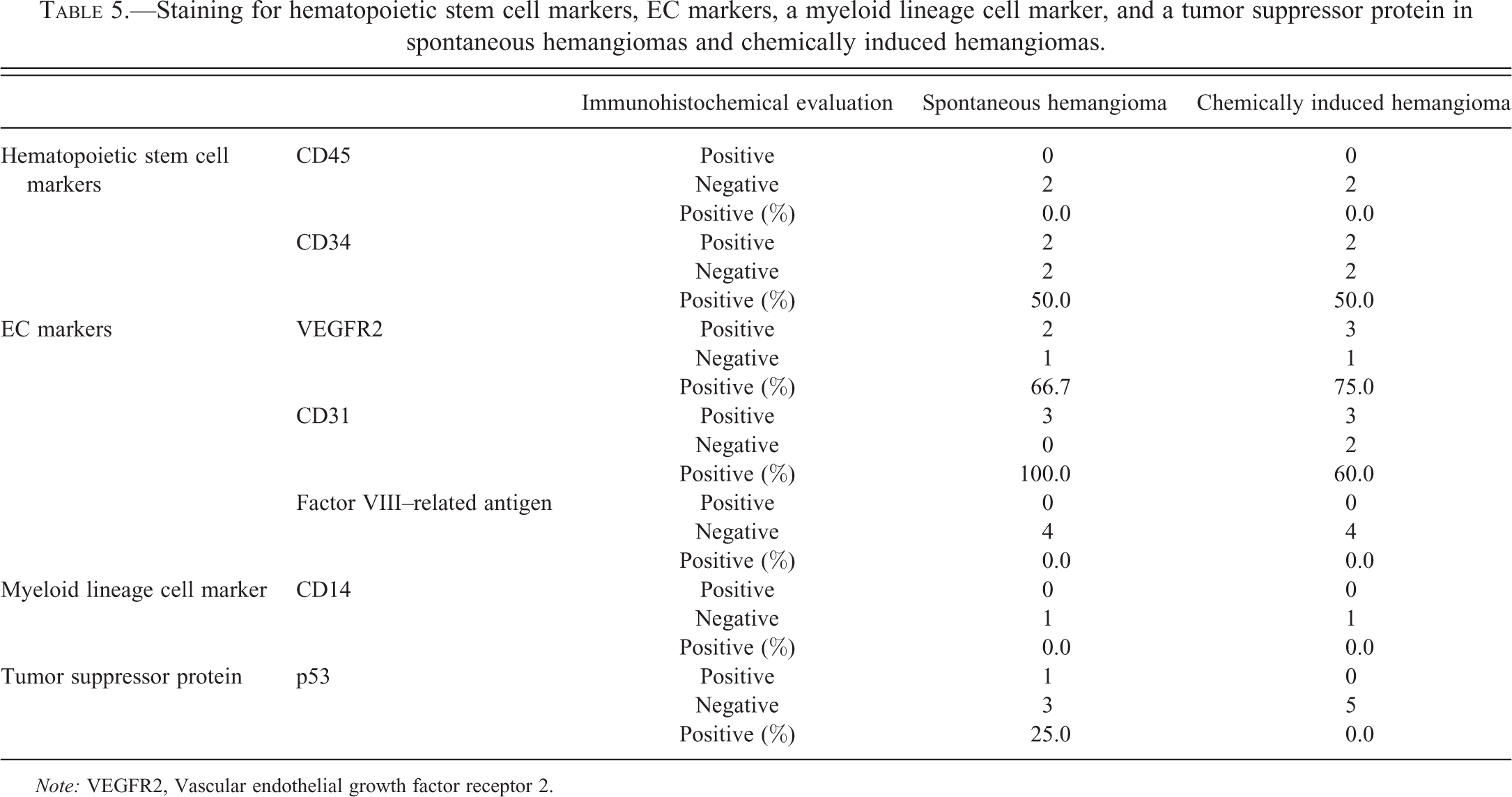
Note: VEGFR2, Vascular endothelial growth factor receptor 2.
EC Markers
Almost all spontaneous, nongenotoxic and genotoxic chemically induced hemangiosarcomas were positive for both VEGFR2 and CD31 staining (Figure 1B and C, Table 4). Similarly, staining for VEGFR2 and CD31 was also present in many spontaneous and chemically induced hemangiomas (Figure 3B and C, Table 5). For both immunostains, the reactive tumor cells exhibited intense diffuse staining, and the staining qualities were similar to that of the adjacent normal vascular endothelium. In contrast, factor VIII–related antigen was negative in most hemangiosarcomas and hemangiomas (Figures 1D and 4B, Tables 4 and 5) although endothelium in the adjacent normal vasculature was positive (internal positive control).
Myeloid Lineage Cell Marker and Tumor Suppressor Protein
Membrane/cytoplasmic staining was observed in neutrophils (a positive internal control); however, all hemangiosarcomas and hemangiomas were negative for CD14 staining (Figures 1E and 4C, Tables 4 and 5). p53 staining was observed in some spontaneous and some nongenotoxic chemically induced hemangiosarcomas (tumors from 1 of 4 chloroprene- and 4 of 6 formamide-treated mice, data not shown; Figure 2C) but in none of the 2-butoxyethanol and troglitazone-induced hemangiosarcomas (Figure 2D). Staining for p53 was also negative in the genotoxic chemically induced hemangiosarcomas (Figure 2E and Table 4) and in the spontaneous and chemically induced hemangiomas (Figure 4D and Table 5). Sections stained with p53 exhibited a moderate to marked amount of background and nonspecific cytoplasmic staining, especially in hepatocellular cytoplasm. Thus, staining was considered positive only when specific intense nuclear positive cells were observed.
Comparison of Staining in Hemangiosarcomas and Hemangiomas
Immunohistochemical staining of hematopoietic stem cell markers (CD45 and CD34), EC markers (VEGFR2, CD31, and factor VIII–related antigen), a myeloid lineage cell marker (CD14), and a tumor suppressor protein (p53) in spontaneous and chemically induced hemangiosarcomas in mice was compared with the staining of these markers in spontaneous and chemically induced hemangiomas in mice. No statistical differences were observed with the exception of an increased percentage of VEGFR2-positive hemangiosarcomas (Table 6). However, this statistical difference might be attributed to the small sample size of hemangiomas present in this study.
Comparison of staining for hematopoietic stem cell markers, EC markers, a myeloid lineage cell marker, and a tumor suppressor protein in hemangiomas and hemangiosarcomas.
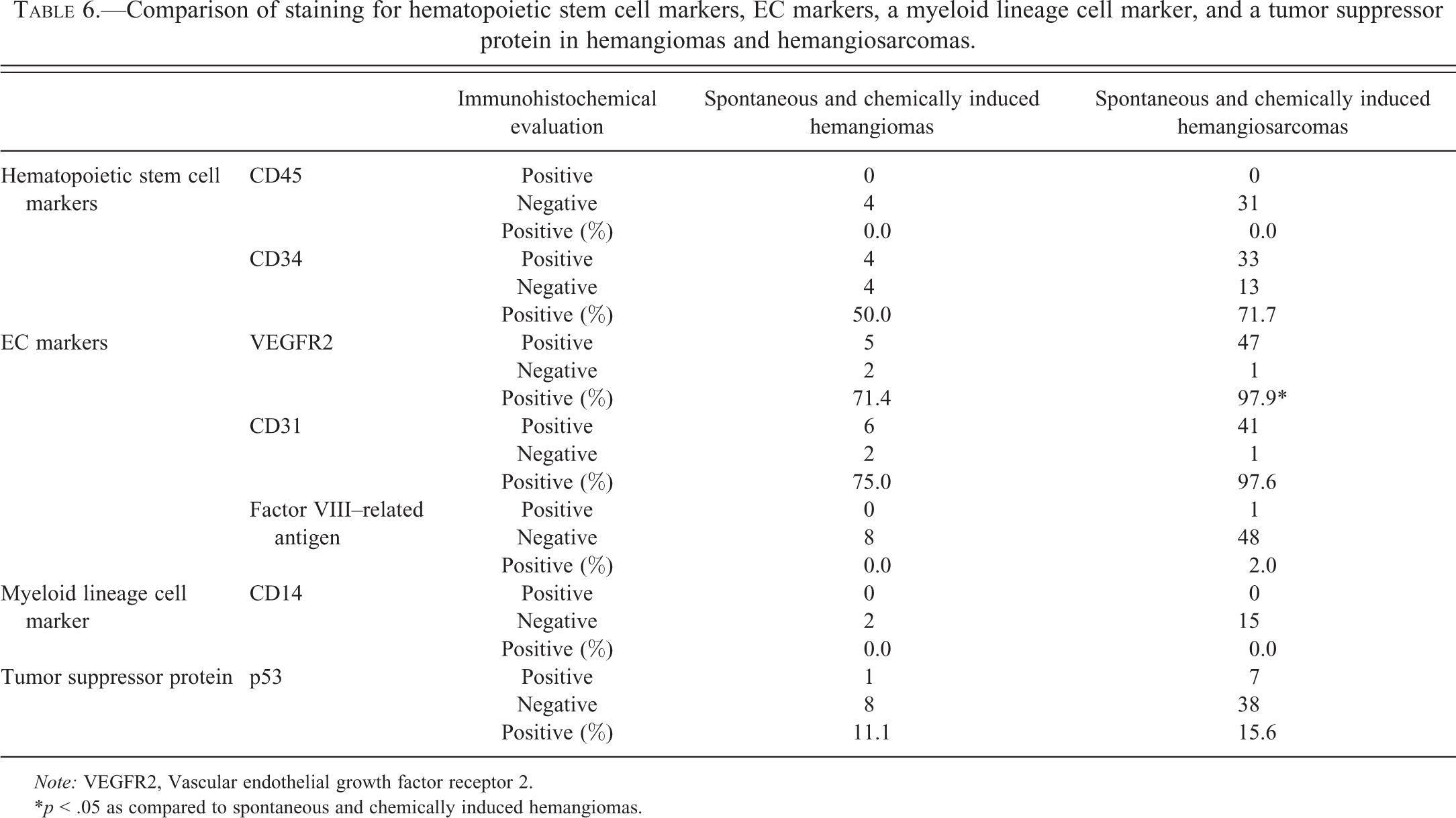
Note: VEGFR2, Vascular endothelial growth factor receptor 2.
*p < .05 as compared to spontaneous and chemically induced hemangiomas.
Discussion
Vascular tumors occur spontaneously at relatively high incidences in several mouse strains compared with the incidences in rats (less than 1%; National Toxicology Program [NTP] 2000) and humans (<0.001%; Weiss and Goldblum 2008), including B6C3F1 mice used in the present studies. According to NTP historical control information for B6C3F1 mice, the mean incidences of spontaneous hemangiosarcoma formation are 9.6% in the male and 3.0% in the female (ranges of 6–14% and 0–14%, respectively) in all organs combined (National Toxicology Program 2000). These vascular tumors occur in a variety of tissues, especially the liver, spleen, and bone marrow, frequently with multiple tissues being involved in an individual mouse. The incidence of these vascular tumors can be increased by a variety of chemicals, genotoxic or nongenotoxic (Cohen et al. 2009). The relevance of these mouse vascular tumors induced by nongenotoxic chemicals to humans has been questioned (Cohen et al. 2009; Kakiuchi-Kiyota et al. 2009; Criswell et al. 2012).
In the present study, we investigated whether the process of spontaneous and chemically induced hemangiosarcoma and hemangioma formation in mice involves the transformation of differentiated ECs with the ability for proliferation or multipotential bone marrow–derived hematopoietic stem cells or EPCs showing some differentiation using immunostaining with three hematopoietic stem cell markers, three EC markers, and a myeloid lineage cell marker.
CD133, CD45, and CD34 are hematopoietic stem cell surface antigens expressed on hematopoietic stem cells and bone marrow–derived EPCs. It is considered that CD133 is expressed on more immature cells, such as hematopoietic stem cells and early EPCs located in the bone marrow or in blood just after migration into the circulation (Hristov and Weber 2004; Urbich and Dimmeler 2004). Expression of CD133 is decreased in late progenitor cells and absent in mature ECs (Handgretinger et al. 2003). In contrast, expression of CD34 is not restricted to hematopoietic stem cells or early EPCs but is also present on late EPCs and mature ECs at lower levels compared with EPCs (Hristov and Weber 2004; Urbich and Dimmeler 2004). CD45 is expressed not only on hematopoietic stem cells but also on lymphoid and myeloid cell lineages from progenitor to mature cells as a pan-hematopoietic marker (Hermiston, Xu, and Weiss 2003; Urbich and Dimmeler 2004). The cascade of CD45 expression during the differentiation process from hematopoietic stem cells to EPCs is unclear. Among the three EC markers used in our study, VEGFR2 is considered to be expressed the earliest and can be seen in more immature cells that also express CD133 (Peichev et al. 2000). During the differentiation process, these cells lose CD133 expression and begin to express the EC markers, first CD31 and then factor VIII–related antigen (Kanayasu-Toyoda et al. 2003; Hristov and Weber 2004).
In the current study, staining for CD45 was negative in spontaneous and chemically induced hemangiosarcomas and hemangiomas in mice. In contrast, CD34-positive cells were observed in these tumors in mice. Due to technical difficulties, we were unable to analyze staining for CD133. These tumors were positive for VEGFR2 and CD31, however, factor VIII–related antigen staining was negative in both hemangiosarcomas and hemangiomas. The qualitative immunohistochemical staining pattern was similar between male and female mice, in tumors from all tissues examined, in hemangiomas and hemangiosarcomas, and in tumors arising spontaneously (in control mice) or in nongenotoxic or genotoxic chemically induced tumors. The staining for most antigens was also quantitatively similar for these comparisons, with the exception of decreased staining for CD34 in tumors from mice treated with genotoxic chemicals compared to controls and those from mice treated with nongenotoxic chemicals. This might be due to damage to CD34 induced by genotoxic chemicals.
It was reported that >99% of mononuclear cells expressing CD133, CD34, and VEGFR2 in peripheral blood also expressed CD45 (Case et al. 2007). Without knowing whether tumor cells in our study expressed CD133 in addition to CD34 and VEGFR2, we are unable to speculate further regarding CD45-positive cells. Expression of CD34 is considered to diminish along the progression of development from early EPCs through differentiated ECs (Hristov and Weber 2004; Urbich and Dimmeler 2004). In most cases in our study, tumor cells stained with increased intensity compared to the adjacent normal vascular endothelium. Based on the increased intensity of CD34 staining and the lack of factor VIII–related antigen staining in these tumors, these cells may be early EPCs that are not completely differentiated. It is still unclear whether circulating early EPCs directly contributed to the hemangiosarcoma formation or more immature stem cells contributed to the tumor formation and differentiated to EPCs, but then differentiation was arrested at the early EPC stage.
Canine hemangiosarcoma cell lines expressed c-kit and CD133 in addition to CD34 and, in some cases, CD45 (Lamerato-Kozicki et al. 2006). In humans, some studies have reported that immunostaining in hemangiosarcomas for CD45 was negative and c-kit staining was variable (15%; 5 of 34 samples; Lin, Weiss, and Battifora 1997; Forton, Van Parys, and Hertveldt 2005; Terada 2011; Yonemori et al. 2011). No reports are available regarding the expression of CD133 in these tumors in humans. These results indicated that more immature stem cells expressing c-kit, CD133, and/or CD45 contributed to hemangiosarcomas in dogs and humans. Expression of CD34, VEGFR2, CD31, factor VIII–related antigen, and other EC markers has been demonstrated in human and canine cases of hemangiosarcomas and/or hemangiomas (von Beust, Suter, and Summers 1988; Poblet, Gonzalez-Palacios, and Jimenez 1996; Lin, Weiss, and Battifora 1997; Breiteneder-Geleff et al. 1999; Forton, Van Parys, and Hertveldt 2005; Lamerato-Kozicki et al. 2006; Itakura et al. 2008; Terada 2011; Yonemori et al. 2011). Considering that factor VIII–related antigen expression, which has been shown to occur later than CD31 expression in EPCs, was also positive in these tumors, it is likely that the tumors arise from more immature cells but retain the ability to more fully differentiate in humans and canines.
CD14 is a monocyte/macrophage cell surface antigen, and the contribution of myeloid lineage cells to angiogenesis has recently been demonstrated (Yoder et al. 2007; Kim et al. 2009). In addition, it was reported that the coexpression of a dendritic cell marker and EC markers was observed in human infantile hemangioma (Ritter et al. 2006). It is possible that myeloid-derived cells are involved in hemangiosarcoma formation in dogs, at least in some cases (Lamerato-Kozicki et al. 2006). However, CD14 staining was absent in spontaneous and chemically induced hemangiosarcomas and hemangiomas in mice, indicating that there is no contribution of monocyte-/macrophage-derived cells to tumor formation.
Since the expression pattern of hematopoietic stem cell and EC markers in mouse hemangiosarcomas was similar to mouse hemangiomas in the current study, this may indicate that hemangiosarcomas could arise from benign EC tumors (hemangiomas) in mice. However, it is still questionable since in humans there is no evidence that hemangiosarcomas (rare tumors) arise from hemangiomas (common tumors) or any other benign precursor (Cohen et al. 2009).
We also investigated p53 expression in hemangiosarcomas and hemangiomas in mice. Wild-type p53 protein has a short half-life; however, the mutated form of p53 protein is stabilized and can be detected in the nucleus of neoplastic cells (Hong et al. 2003). It is well known in human tumors that inactivation of the tumor suppressor gene, p53, is involved in hemangiosarcomas but not hemangiomas. For example, in humans, immunostains for p53 were almost entirely negative in hemangiomas (Iwata et al. 1996), whereas nuclei of malignant cells in hemangiosarcomas stained positive for p53 protein (Meis-Kindblom and Kindblom 1998). Furthermore, p53 protein mutations in exons 5–8 were observed in spontaneous and vinyl chloride–induced hemangiosarcomas in humans (Hollstein et al. 1994; Naka et al. 1997). Multiple publications indicate the increased frequency of hemangiosarcoma formation in p53-mutant mice (Donehower et al. 1992; Zindy et al. 2003). In our study, similar to humans, p53 staining was negative in most hemangioma samples. Staining of p53 in nongenotoxic chemically induced hemangiosarcomas in mice was variable. It was absent in hemangiosarcomas collected from troglitazone-treated mice similar to previous reports (Duddy et al. 1999), in 2-butoxyethanol-treated mice and in most tumors collected from chloroprene-treated mice. However, immunostaining was observed in 4 of 6 formamide-induced hemangiosarcomas. Wild-type p53 protein can be stabilized and functionally inactivated by binding to the cellular murine double minus-2 (MDM2) protein so that overexpression of MDM2 may inactivate wild-type p53 protein in hemangiosarcomas (Zietz et al. 1998). Therefore, it is possible that formamide, but not other nongenotoxic chemicals, affected pathways that increase MDM2 protein, resulting in the stabilized p53 without inducing genetic mutation in p53.
In summary, this study indicates that the hemangiosarcomas and hemangiomas in mice are composed of EPCs expressing CD34, VEGFR2, and CD31 but not factor VIII–related antigen. Hematopoietic stem cell antigen, CD45, was negative, and myeloid-lineage cells do not appear to contribute to hemangiosarcoma and hemangioma formation in mice. The lack of factor VIII–related antigen expression may indicate that the mouse tumor cells are arrested at a stage prior to complete differentiation, although it is likely that tumor cells retain the ability to more fully differentiate in humans and canines. Our results indicate that mouse hemangiomas and hemangiosarcomas may arise by a different mechanism than canine EC tumors and possibly humans, and use of the rodent model for understanding the mechanism and relevance of EC tumors to humans might not be appropriate. Additional studies better defining the immunophenotype of vascular tumors are required.
Footnotes
Acknowledgments
The authors gratefully acknowledge Dr. Norimitsu Shirai at Pfizer, Inc., for his pathological expertise and the individuals at EPL, Inc.: the Histology Lab for retrieving the paraffin blocks and cutting sections, the Photo Lab for taking photos of immunostaining, and Beth Mahler for editing images. We also gratefully acknowledge the technical expertise of Heather Jensen, Otis Lyght, Geoffrey Hurlburt, Yvette Rebolloso, David Olson, Quashana Brown, and Natasha Clayton in the Immunohistochemistry Lab of the Pathology Support Group of the Cellular and Molecular Pathology Branch of the National Toxicology Program, NIEHS for development of the methodology and performance of the immunohistochemistry. Heather Jensen was particularly helpful technically and in assisting in preparation of the manuscript. This research was supported (in part) by the Division of the National Toxicology Program of the NIH, National Institute of Environmental Health Sciences. This article may be the work product of an employee or group of employees of the National Institute of Environmental Health Sciences (NIEHS), National Institutes of Health (NIH); however, the statements, opinions, or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the U.S. government.
Drs. Kakiuchi-Kiyota and Cook work for Pfizer, Inc.; Dr. Crabbs works for EPL, Inc., with a previous appointment at the National Toxicology Program; and Dr. Cohen has consulted for Pfizer.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by the Division of the National Toxicology Program of the NIH, National Institute of Environmental Health Sciences; however, the statements, opinions or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the U.S. government.
