Abstract
The purpose of this symposium report is to summarize information from a session 3 oral presentation at the Society of Toxicologic Pathology Annual Symposium in Raleigh, North Carolina. Mice are genetically tractable and are likely to play an important role in elucidating environmental, genetic, and aging-related mechanisms of urinary dysfunction in men. We and others have made significant strides in developing quantitative methods for assessing mouse urinary function and our collaborators recently showed that aging male mice, like men, develop urinary dysfunction. Yet, it remains unclear how mouse prostate anatomy and histology relate to urinary function. The purpose of this report is to share foundational resources for evaluating mouse prostate histology and urinary physiology from our recent publication “Impact of Sex, Androgens, and Prostate Size on C57BL/6J Mouse Urinary Physiology: Functional Assessment.” We will begin with a review of prostatic embryology in men and mice, then move to comparative histology resources, and conclude with quantitative measures of rodent urinary physiology.
Prostate Embryology, Histology, and Urinary Physiology Testing in Mice
Prostate Embryology
Prostates were originally defined as structures that “lie on the urethra near the neck of the bladder” and their “ducts must open into the prostatic urethra at the level of the verumontanum.” 1 In most animals, several distinct lobes are grouped as single prostate. The human prostate was originally divided into 5 distinct lobes 1 corresponding to 5 discrete “prostate buds” that originate in the urogenital sinus during embryogenesis. 2 The lobes included a dorsal lobe (openings distal to the verumontanum), a middle lobe (openings proximal to the verumontanum), 2 lateral lobes (openings on each side of the verumontanum), and a ventral/anterior lobe (often absent or atrophic in adults with ventral openings into the urethra). 1 The mouse prostate is divided into 4 bilaterally symmetrical lobes: dorsal, anterior, lateral, and ventral. These lobes originate as series of buds in the urogenital sinus. 3 These buds have a similar localization to human prostate buds, 3 and based on the similar bud location, dorsal, anterior, and lateral lobes of mouse were hypothesized to correspond to the dorsal, middle, and lateral lobes of man. 1 Mouse and human prostate development are remarkably similar. They rely on similar molecular mechanisms for prostate specification (occurs during mouse embryonic day [E] 13-15 and human gestation week 8-9), ductal bud formation (mouse E16-18; human ∼10-11 weeks), bud elongation and branching morphogenesis (mouse P1-40; human ∼11 weeks and after), and ductal canalization (mouse P3-50; human ∼11 weeks and after). 4 Mouse and human prostate development also respond in comparable fashion to environmental hormone and toxicant exposures. 5 –8 Therefore, prostate development in mouse should be regarded as highly relevant to prostate development in man.
Prostate Histology
Contemporary definitions divide the human prostate into the peripheral, transition, and central zones based on the gross anatomic characteristics and propensity for disease. 9 The peripheral zone, defined in human based off its propensity for developing prostatic carcinoma, closely corresponds to the dorsal prostate in mouse based on anatomic location 1 and molecular signatures. 10 The central zone, defined by its high propensity for squamous metaplasia, closely corresponds to the anterior prostate in mouse. 11 The transition zone is the common site of benign prostatic hyperplasia, but as mice do not develop localized and nodular hyperplasia in their prostate, there isn’t a clear correlating lobe in the mouse. 11 A recent hypothesis suggests that mouse prostate ductal segments existing between the urethra and rhabdosphincter may be the most relevant correlate to the human prostate transition zone, but this region of prostate is largely uncharacterized in mice. For this reason, we have compiled serial sections starting at the bladder neck and extending to the pelvic urethra in order to fully characterize the prostatic urethra of the mouse (Figures 1 and 2).
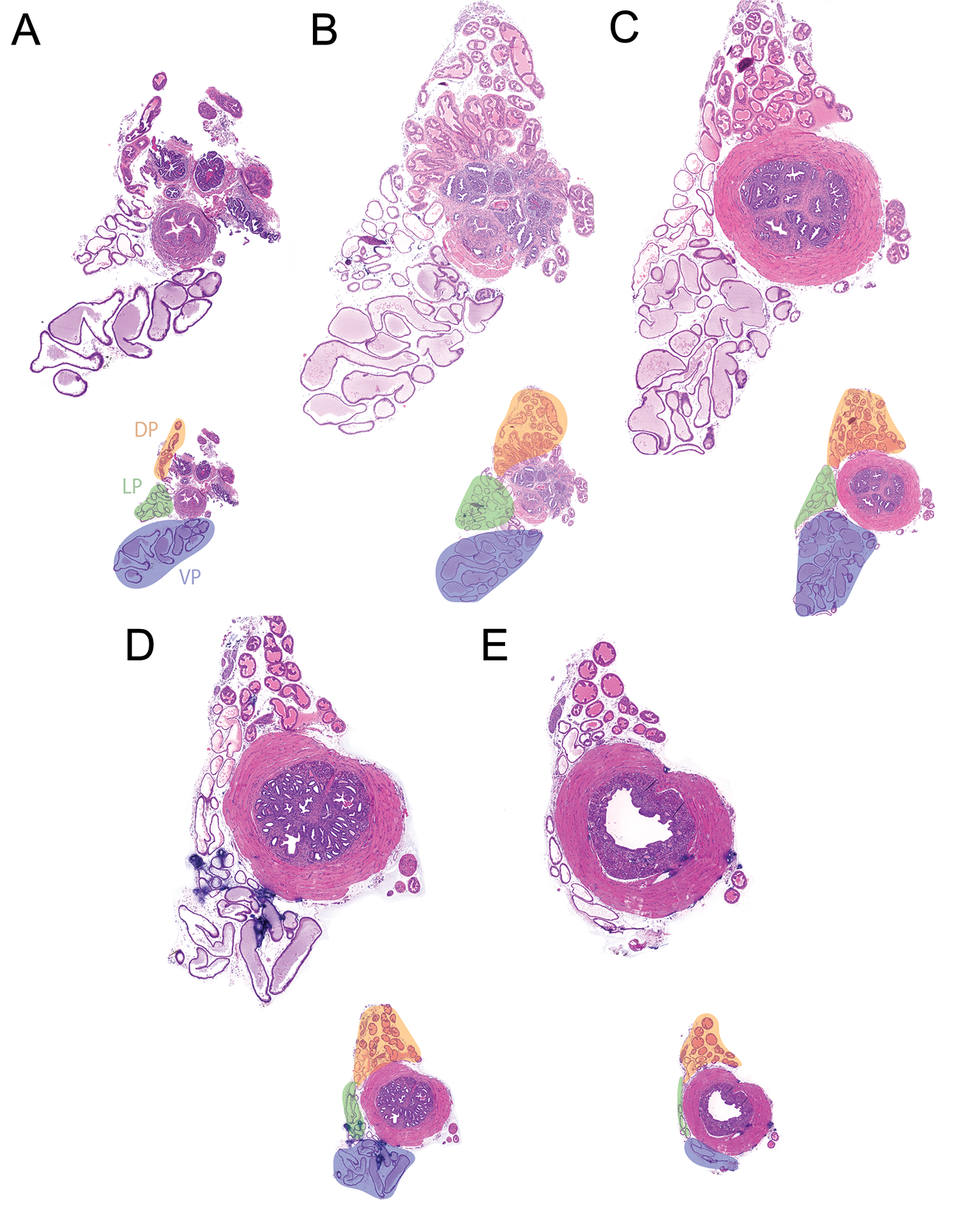
Serial images from the prostatic urethra of a 9-week-old intact male C57BL/6J mouse. The lower urinary tract was removed in whole through 5 cuts: bilateral ureters, bilateral vas deferens, and urethra at the level of the pelvic inlet. The bladder, seminal vesicles, anterior prostate, and hemi-dorsal, lateral, and ventral lobes were removed. The prostatic urethra and remaining prostate lobes were fixed in 10% formalin, oriented in the transverse plane, and embedded in paraffin. The block was faced and 45 serial 5-µm-thick sections were taken; the first, 11th, 22nd, 33rd, and 44th sections are depicted in (A) to (E). A, The first section near the bladder neck and each consecutive image (B-E) is 550 µm deeper. Below each tile scan is an image highlighting the dorsal prostate (DP, orange), lateral prostate (LP, green), and ventral prostate (VP, blue). We consider (C) a target region as it contains a large section of DP, LP, and VP, as well as a region of the rhabdosphincter containing ejaculatory duct, urethra, seminal vesicle (SV), and periurethral prostate duct segments (PUPDS). In adjacent nonideal sections (B and D), it can be difficult to distinguish urethral glands, SV, and PUPDS. A video tour and tile scans of all sections are available at https://doi.org/10.25548/16-QQG2.
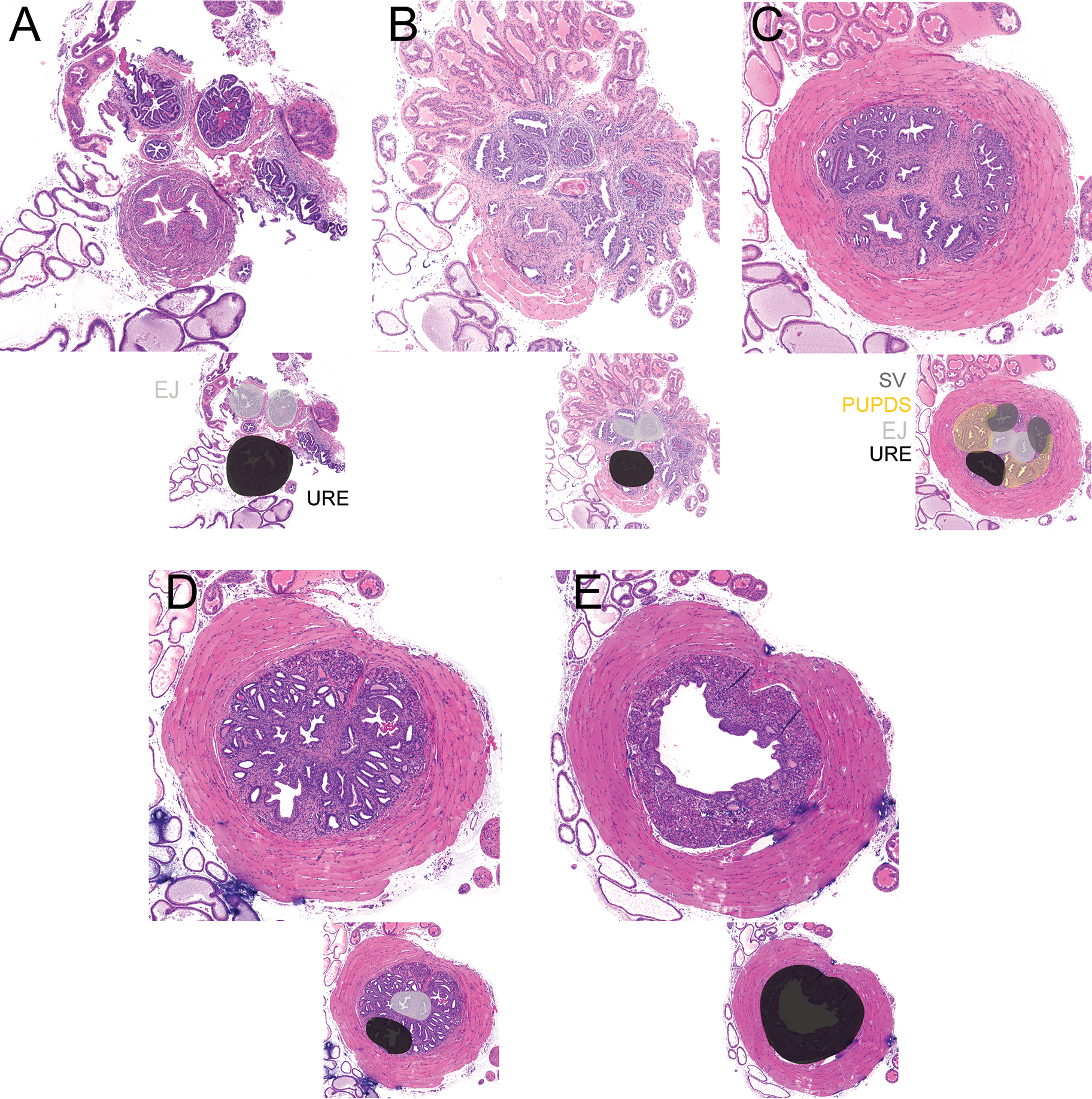
Urethral insets of the prostatic urethra of tile scans in Figure 1. The lower urinary tract was removed in whole through 5 cuts: bilateral ureters, bilateral vas deferens, and urethra at the level of the pelvic inlet. The bladder, seminal vesicles, anterior prostate, and hemi-dorsal, lateral, and ventral lobes were removed. The prostatic urethra and remaining prostate lobes were fixed in 10% formalin, oriented in the transverse plane, and embedded in paraffin. The block was faced and 45 serial 5-µm-thick sections were taken; the first, 11th, 22nd, 33rd, and 44th sections are depicted in (A) to (E). A, The first section near the bladder neck and each consecutive image (B-E) is 550 µm deeper. Below each urethral inset is an image highlighting ejaculatory ducts (EJ, light gray) and urethra (URE, black). We consider (C) a target region as it contains a region of the rhabdosphincter containing EJ, URE, seminal vesicle (SV, dark gray), and periurethral prostate duct segments (PUPDS, yellow). In adjacent nonideal sections (B and D), it can be difficult to distinguish urethral glands, SV, and PUPDS.
We have created a narrated prostate lobe dissection video using teaching mice (https://doi.org/10.25548/0KNR) in order to show prostate lobe identification and dissection. Mouse prostate lobes are difficult to distinguish, and the presence of pelvic fat and other glandular structures complicates prostate lobe identification and dissection. To assist in teaching this critical skill, we introduced tdTomato expression in Pbsn4+ luminal cells to pigment the prostate and enable rapid identification of anterior, dorsal, lateral, and ventral lobes. The mice were created by breeding Tg(Pbsn-cre)4Prb/J (Pbsn4cre, stock number 026662) 12 mice to B6.Cg-Gt(ROSA)26Sortm14(CAG-tdTomato)Hze/J (R26R-Tdtomato, Jax stock #007914) 13 mice. The prostate lobes of the resulting male offspring appear pink and are readily differentiated from white periprostatic fat, ampullary gland, and seminal vesicles. We have also developed a method for preparing histological sections in a reproducible transverse plane by orienting prostatic urethras in agar prior to dehydration, clearing, and paraffin infiltration (see https://doi.org/10.25548/0QFR).
Quantitative Urinary Physiology Testing in Mice
We routinely use 2 assays to measure urinary function in mice. The first is the void spot assay (VSA) and this assay is schematized here (https://doi.org/10.25548/0QF4). Void spot assay is performed in the vivarium where mice are housed and allows quantitative analysis of mouse void spots (spots of mouse urine). Whatman grade 540 (Fisher Scientific no. 057163-W) filter papers are cut to the size of the cage and placed on the bottom surface. Mice are placed in the cage (singly housed) with food ad libitum but no water for 4 hours starting at 8 to 11
The second assay for assessing mouse urinary function is cystometry and is used to evaluate mouse bladder pressures and bladder emptying in response to continuous filling at a defined rate. Bladder pressure and frequency of emptying can be altered in response to many disease states, including but not limited to diabetes, infection, and outlet obstruction. By measuring bladder pressure throughout the urination cycle, we can detect increases in baseline pressure and pressure at initiation of voiding (indicative of obstruction), reductions in time between voids (indicative of urinary frequency), and other changes from baseline indicative of urinary dysfunction in mice. We provide an overview of cystometry at https://doi.org/10.25548/0QFG. To perform cystometry, mice are anesthetized with urethane (1.43 g/kg subcutaneously, which in our experience provides a more stable plane of anesthesia than intraperitoneal urethane injection). We begin surgery 30 minutes after urethane dosing. An incision is made in the ventral abdomen to expose the bladder. Bladder length and diameter are measured for bladder volume calculation. A purse string suture is placed in the bladder dome. Then, polyethylene cystostomy tubing (PE50, outer diameter 0.58 mm, inner diameter 0.28 mm) is inserted into the bladder through the center of the suture and the purse string suture is secured to hold the tubing in place with 2 to 3 mm of tubing within the bladder. The abdominal wall and skin are then closed separately using a simple interrupted pattern. Finally, the exterior tubing is secured to the ventral abdominal skin with 2 simple interrupted sutures. Mice are placed on a heat pad for 1 hour after the procedure. To perform cystometry, the exposed tube is connected to a 3-way stopcock, and the other 2 arms of the stopcock are connected to an infusion pump (Harvard Apparatus, Holliston, Massachusetts) and pressure transducer (Memscap AS, Skoppum, Norway). Intravesical pressure is recorded continuously using a PowerLab data collection system (ADI Instruments, Colorado Springs, Colorado), while room temperature sterile saline (0.9%) is infused into the bladder at a rate of 0.8 mL per hour. Mice are placed in lateral recumbency above a force transducer (Model FT03, Grass Instruments), with a 3-dimensional printed urine collection funnel, Warwick, RI. The force transducer is calibrated with known volumes of saline to create a pressure-volume conversion. The mass of voided urine is recorded continuously using PowerLab. At least 1 hour of voiding activity is recorded and 3 to 5 consecutive voids, occurring after stabilization of micturition cycles, are used for analyses.
We recently used VSA to assess urinary function at 6, 7, 8, 9, 10, 12, and 14 weeks of age in intact male C57BL/6J mice. 15 All measured end points changed with age. 15 We believe these are background age-related changes in healthy adult male mice and are potentially behavioral but likely not pathological. Therefore, as with other assays, it is important to consider magnitude of change and concurrent findings when determining whether a statistical change in a VSA parameter is indicative of disease.
Also using VSA and cystometry, we have established baseline urinary characteristics of male and female C57BL/6J mice. 15 We found female mice start to urinate at a higher bladder pressure, reach a higher maximal bladder pressure during voiding, urinate quicker, and urinate a greater volume than male mice. 15 We also found that these baseline difference in males and females are likely due to testosterone and not anatomical differences, such as presence of prostate. 15
Concluding Remarks
We have documented clear similarities in human and mouse during prostate development, provided human prostate zone equivalents based on propensity of disease, and shared resources for evaluating mouse prostate anatomy and histology, and urinary physiology. Further studies are needed to continue to build comparisons between mouse and human so we can make well-founded conclusions about anatomic, histologic, and physiologic changes that translate to human disease. All animal experiments were conducted under an approved protocol from the University of Wisconsin Animal Care and Use Committee and conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All instruction videos and histology images were deposited into the GUDMAP consortium database and are fully accessible at: https://doi.org/10.25548/16-QQG2. 18
Footnotes
Acknowledgments
The authors would like to thank Hongsuda Tangmunarunkit and Laura Pearlman for their assistance and efforts in making inclusion of all of our instructional videos possible. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Funded by National Institutes of Health grants: U54 DK104310, Summer Program in Undergraduate Urologic Research (U54 DK104310S1), U01DK110807, R01ES001332, R01DK099328, F31ES028594, TL1TR002375, and University of Wisconsin–Madison, School of Veterinary Medicine.
