Abstract
The design and execution of toxicology studies supporting vaccine development have some unique considerations relative to those supporting traditional small molecules and biologics. A working group of the Society of Toxicologic Pathology Scientific and Regulatory Policy Committee conducted a review of the scientific, technical, and regulatory considerations for veterinary pathologists and toxicologists related to the design and evaluation of regulatory toxicology studies supporting vaccine clinical trials. Much of the information in this document focuses on the development of prophylactic vaccines for infectious agents. Many of these considerations also apply to therapeutic vaccine development (such as vaccines directed against cancer epitopes); important differences will be identified in various sections as appropriate. The topics addressed in this
*This Points to Consider article is a product of a Society of Toxicologic Pathology (STP) Working Group commissioned by the Scientific and Regulatory Policy Committee (SRPC) of the STP. It has been reviewed and approved by the SRPC and Executive Committee of the STP but does not represent a formal best practice recommendation. The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of their respective agencies and organizations. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to the editor.
Introduction
The development and use of vaccines have evolved substantially since Edward Jenner first used exudate from cowpox lesions to inoculate people against smallpox. The underlying concept for vaccine development, however, is essentially the same today as it was then: expose people to an antigen to prime development of protective immunity against disease. Vaccines may be prophylactic or therapeutic. Prophylactic vaccine development is different from traditional drug development, as the target is a foreign protein that should have no or very little homology to human- or animal-expressed proteins. Therapeutic vaccines, in contrast, typically target endogenous proteins or neoantigens to cure existing disease, and as such have some different nonclinical considerations. Several therapeutic vaccines are currently being or have been investigated for potential treatment of a diverse number of diseases such as Alzheimer disease, Parkinson disease, and multiple types of cancer. 1 –3 Vaccines are being developed using a wide variety of modalities, and formulations/therapies may include adjuvants or other immunological modulators to promote the desired immune response.
Regulatory toxicology studies in support of vaccine clinical trials incorporate similar design elements to standard small molecule and biotherapeutic toxicity studies, with inclusion of additional points intended to evaluate acute phase and immunogenic responses to the vaccine antigens. With respect to pathology, regulatory toxicology studies for vaccines share many of the same fundamental points as routine toxicology studies, such as macroscopic and microscopic observations, organ weights, and clinical pathology parameters. However, there are considerations that the pathologist involved in vaccine development programs should be aware of to help guide protocol development, tissue collection, and study interpretation. Specifically, these include species selection, route of administration, nonantigen vaccine components, coadministered compounds such as adjuvants, the use of devices or techniques for accurate dose administration and draining lymph node and injection site tissue collection. In addition to the standard toxicology studies conducted for vaccines, other studies such as neurovirulence, biodistribution, or environmental risk (eg, livestock transmission) studies may be required depending on the vaccine modality and indication, and veterinary pathologists may be involved in study design and interpretation of study data as well. A host of other studies may be conducted for vaccines, and pathologists may be enlisted to provide support.
The purpose of this article is to serve as a “Points to Consider” document for toxicologic pathologists in vaccine development and includes practical information to help guide pathologists in the conduct of these studies. The focus of the discussions here is related to the development of vaccines for human use. Although regulatory aspects differ slightly, the same principles and practices described here could be applied to vaccines being developed for veterinary use.
Regulatory Guidelines for the Nonclinical Safety Assessment of Vaccines and Adjuvants
Until the 1990s, there was little regulatory guidance on vaccine development. The first comprehensive nonclinical pharmacology and toxicology prophylactic vaccine regulatory guideline was issued in 1997 by the European Medicines Agency (EMA), 4 which has subsequently been replaced by World Health Organization (WHO) guidance documents on assessment of vaccine safety. 5 Guidelines specifically addressing the use of adjuvants in vaccines include a WHO guidance 6 (2014) and an EMA guidance (2005). 7 The Food and Drug Administration (FDA) defers to the WHO guidelines for the development of prophylactic vaccines intended to protect against microbial disease. The WHO guidance documents guide the nonclinical development of vaccines for many countries, although additional studies on vaccine safety may be expected in some countries. There are several disease-specific vaccine guidances that should be referred to for certain indications (eg, dengue, Ebola, HIV). 8– 10 There are no International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use guidances specifically for vaccine development and nonclinical safety assessment. It is important to note that regulatory expectations for vaccine development are evolving, and for these reasons, vaccine developers and producers need to keep apprised of current global regulatory requirements to meet regulatory expectations for marketing approval worldwide. Regulatory guidance in the development of therapeutic vaccines is currently limited and includes US FDA’s “Clinical Considerations for Therapeutic Cancer Vaccines” 11 and EMA’s “Guideline on the evaluation of anticancer medicinal products in man.” 12 Although these guidelines focus primarily on clinical development, they briefly include considerations for nonclinical studies.
Regulatory requirements may vary depending on the nature of the vaccine (eg, peptide, conjugate, nucleic acid, viral vector, live virus); its components including the presence of adjuvants, adsorbants, or other; and the intended target population. Specific guidelines (general list can be found in Table 1) should be referred to when designing nonclinical studies for new vaccine candidates. Most prophylactic vaccines require only 1 repeat-dose toxicology study in a single species, which is generally expected to include a recovery arm to assess the reversibility and to detect any delayed toxicity, such as a delayed immune response. Regulatory requirements to support administration in humans are similar to those for other biopharmaceuticals, with some exceptions (Table 2). Generally, stand-alone single-dose toxicity and safety pharmacology studies prior to clinical studies are not required for vaccines (with the exception Japanese regulatory authorities, which presently do ask for safety pharmacology assessments for vaccines). If it is determined that such studies are necessary, it is generally sufficient to assess these effects within the repeat-dose toxicology study supporting the first-in-human clinical trial. Additionally, nucleic acid–based vaccines using DNA or RNA vectors will likely require biodistribution and possibly persistence studies and may require other safety studies, for example, neurovirulence in the case of potentially neurotropic live virus constructs. For additional information, the reader is referred to the many excellent reviews published on vaccine study design and regulatory considerations. 13 –17
Selected Regulatory Guidelines for Vaccines.
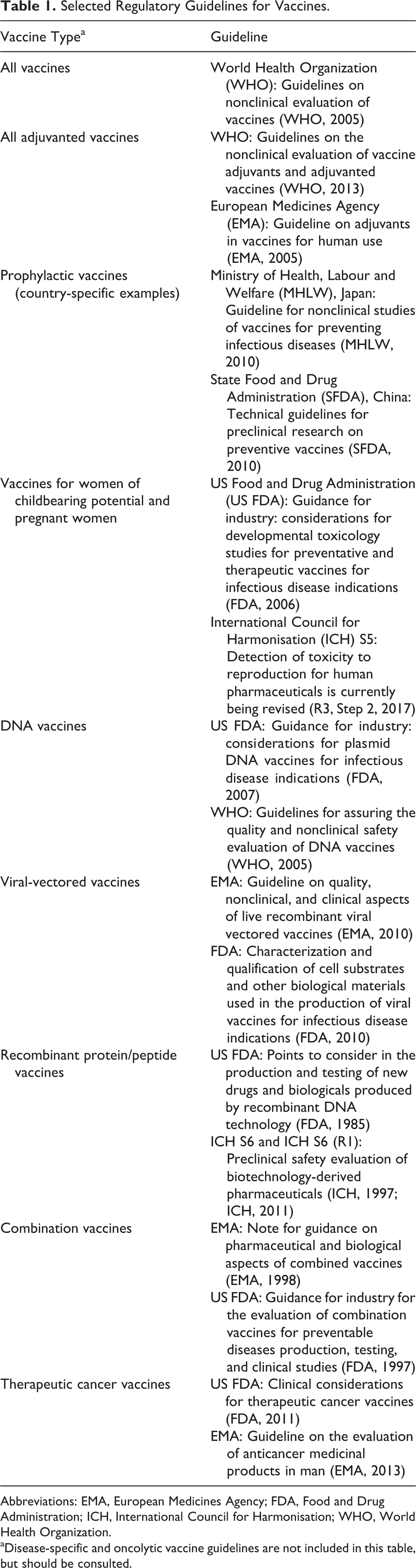
Abbreviations: EMA, European Medicines Agency; FDA, Food and Drug Administration; ICH, International Council for Harmonisation; WHO, World Health Organization.
aDisease-specific and oncolytic vaccine guidelines are not included in this table, but should be consulted.
Types of Studies Performed for Different Modalities.
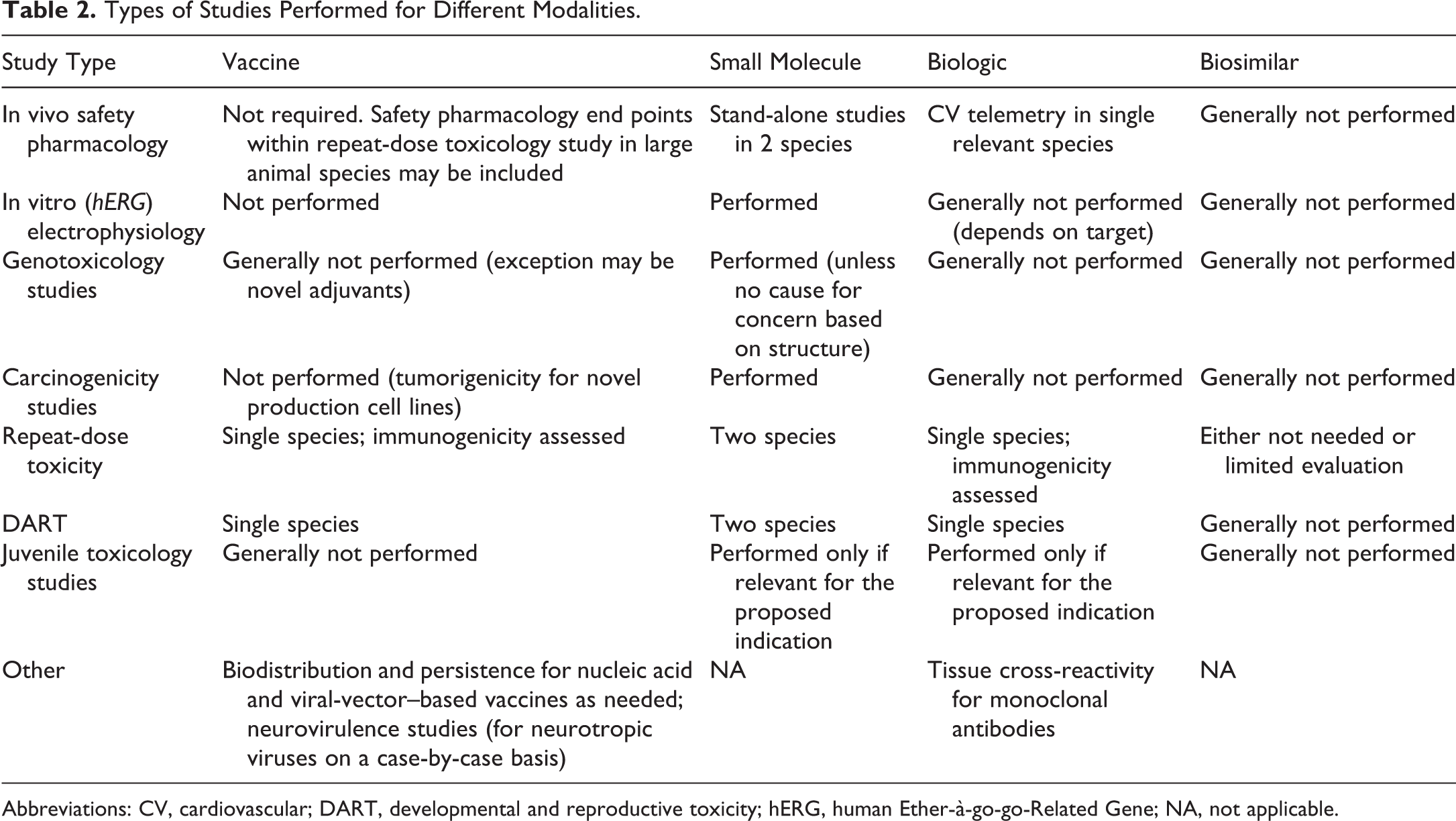
Abbreviations: CV, cardiovascular; DART, developmental and reproductive toxicity; hERG, human Ether-à-go-go-Related Gene; NA, not applicable.
Therapeutic vaccines are a heterogeneous class of vaccines that contain immunogenic substances capable of inducing antigen-specific, active (humoral or cell-mediated) immunity for use in treating a number of diverse diseases (eg, Alzheimer disease, Parkinson disease, multiple types of cancer). 1 Therapeutic vaccine regimens may include administration of active components that modulate the immune system, such as programmed cell death protein-1 and programmed death ligand-1 (PD-1/PDL-1) inhibitors, cytotoxic T-lymphocyte associated protein 4 (CTLA-4) inhibitors, and tyrosine kinase inhibitors. Inclusion of these components is essential for overcoming tolerance to self-antigens, which would prevent elicitation of the immune response needed for a therapeutic effect. Any active components should have a complete safety package to support the route of administration, dose, and so on. Additionally, devices intended for clinical administration of vaccines must have an adequate safety package for submission to the FDA (and subject to device-specific guidelines), and the pathologist may or may not be involved in the evaluation of these devices.
Adjuvants
Many vaccine formulations contain adjuvants. Adjuvants used in conjunction with a vaccine antigen may increase or prolong an immune response, retain an antigen at the site of injection, and/or modulate the type of immune response to the antigen (Th1 vs Th2) to enhance the success of the vaccine. 6,18 –20 Although adjuvants have an immune-stimulatory effect, regulatory authorities in the United States and European Union do not consider them to be active components of the formulation. Instead, they are considered vaccine constituents as described in the Code of Federal Regulations (US 21CFR 610.15 (USCFR [2015]). Established adjuvants include those that are used in marketed vaccines, of which the most common are the aluminum salts (eg, aluminum hydroxide, aluminum phosphate, potassium aluminum sulfate) that have been in many products and have decades of safety information. In contrast, novel adjuvants are those that have a limited safety database and/or are not yet used in marketed products. It should be noted that there is not complete uniformity in the regulatory expectations for novel adjuvants between the EMA and WHO guidelines. The EMA Guideline on Adjuvants in Vaccines for Human Use recommends that adjuvants be tested alone in 2 species (rodent and nonrodent) unless otherwise justified, whereas the WHO guidelines indicate that including an adjuvant alone arm in the repeat-dose toxicology study in single species is generally acceptable. Therefore, decisions around species selection for adjuvanted vaccines may require scientific discussion with regulatory authorities. There are several adjuvants that were initially developed as novel adjuvants (eg, MF59 [Fluad], AS01B [Shingrix], CpG [22-mer ODN 1018 ISS] [Heplisav-B]), but which are now in approved products and have postapproval safety data.
Vaccine Study Design
Considerations for the design of nonclinical studies for vaccines include species selection, dose selection, and dosing regimen and may be different between therapeutic and prophylactic vaccines. In addition, the components of the vaccine should reflect the anticipated clinical doses and administration routes. Any specialized equipment for vaccine delivery (eg, electroporation units, microneedles) in nonclinical studies should, as closely as possible, match the device intended for clinical use. However, there may be equipment limitations due to the small body size of some nonclinical species.
Species Selection
The key criterion for species selection in vaccine toxicity testing is a demonstrable immune response to the administered antigen(s). The WHO guidelines 5 specify that the animal species should develop “an immune response similar to the expected human response after vaccination.” However, this may not be feasible as even in nonhuman primates (NHP) the immune response does not necessarily translate to that of humans 21 and the immune response need not necessarily reflect the type of immune response anticipated in humans (eg, a humoral response may be assessed even if the vaccine elicits a cellular response). The WHO guidelines 5 also suggest that the chosen animal species ideally “be sensitive to the pathogenic organism or toxin under consideration.” Presumably, this is a consideration for modified live viruses, which may revert to a more virulent form or inactivated viruses that may be incompletely inactivated. Selecting a species that is susceptible to the pathogen or reflects the course of infection in humans may be difficult. Therefore, in practical terms, demonstrable immunogenicity to the vaccine (antigen) is accepted for most vaccine toxicology studies.
The rat and rabbit are the most commonly used species for vaccine studies, as their small size is cost-effective and they both tend to generate immune responses to administered vaccine antigens. Other laboratory animal species such as mice, guinea pigs, ferrets, 22 NHP, dogs, and minipigs 23 are also used both in vaccine toxicology studies and as nonclinical models for vaccine development. For therapeutic cancer vaccines, the choice of the appropriate species for evaluation is often on a case-by-case basis with consideration of antigen sequence homology versus endogenous human proteins and pharmacologic activity of coadministered materials (eg, CTLA-4 and/or PD-1/PDL1 antagonists). 24 Specific considerations for the various test species for vaccine toxicology studies are included below.
Rabbits
The rabbit has been the traditional species for vaccine toxicology studies, as they tend to generate a robust immune response to administered antigens. Their larger size also allows for administration of the full human dose/dose volume (typically 0.5 mL) into a single intramuscular (IM) site, and their blood volume permits a greater number of repeated blood samplings than from rodents. 25 The disadvantages of using the rabbit can include less familiarity of necropsy staff and pathologists with rabbit anatomy, a paucity of publications on background pathology findings in rabbits, a limited historical control database (HCD), housing costs, and the current lack of a standardized system of nomenclature, tissue sampling, and trimming guidelines for rabbit. The forthcoming International Harmonization of Nomenclature and Diagnostic Criteria publication on neoplastic and non-neoplastic findings in rabbits will substantially improve consistency in nomenclature and pathology findings in this species. Rabbits have also been reported to have a limited immune response to adjuvants that trigger a toll-like receptor-9 (TLR9) response in mice and humans, such as CpG oligodinucleotides (ODN). 26 Therefore, the rabbit may not be an ideal toxicology species for vaccine formulations that contain CpG ODN. A recent publication also suggested that stress in rabbits may lead to inflammation in the heart, which may confound study interpretations. 27 For these reasons, some companies have transitioned to using the rat as their primary toxicology species for vaccine studies.
Rats
Rats are small and easy to handle, have relatively low housing costs, and do not require specialized training for necropsy. In addition, the rat anatomy is well understood by most technicians, and tissue trimming guides are readily available. For the pathologist, the advantages of the rat include a well-harmonized and standardized nomenclature 28 and a more ubiquitous understanding of background or spontaneous histopathology and variability of clinical pathology parameters. The primary disadvantages of using the rat are limited blood volume and small muscle mass for vaccines administered by IM injection. The small muscle mass constraints may be overcome by administering in multiple sites or using a lower dose volume that is allowed per the WHO guideline (dosing on a milligram per kilogram [mg/kg] basis) IM. 25 The small muscle mass makes IM administration challenging, and errors in precision of the injection are common with individuals inexperienced in this technique. Brief anesthesia with isoflurane may be used to improve the placement accuracy of IM injection without apparent impact in other study parameters. Many companies have transitioned from using the Sprague-Dawley (SD) rat to using the Wistar Han (WH) rat as they generally have fewer age-related background findings. 29 Because the WH rat strain is smaller than other rat strains commonly used for toxicology (eg, SD), it may be advisable to initiate vaccine studies in 10- to 12-week-old WH rats rather than the more typical age of 8 to 10 weeks so that they are slightly larger in size at study start.
Mice
Mice have been less commonly used than rabbits or rats as the toxicology species for vaccine development, primarily because of their small size. However, if the mouse has been used as the model in nonclinical pharmacology or efficacy studies, there is no reason they cannot be used as the toxicology species as long as a reliable and consistent immune response is generated to the vaccine antigens. Since administration of a full human dose is not generally possible, vaccine studies in mice are conducted using a dose that achieves some multiple(s) of the anticipated human clinical dose on a mg/kg basis. Historical control database may be limited within an institution on the specific strain utilized; however, for the standard mouse strains, there is published literature and databases (eg, Registry of Industrial Toxicology-Animal Data; https://reni.item.fraunhofer.de/reni/public/rita/) on background findings to serve as a good resource to the pathologist. 30 In addition, as in the rat, the mouse anatomy is well understood by most technicians and tissue trimming guides, 31 –33 and a harmonized and standardized system of nomenclature is readily available. 34
Other Small Nonclinical Species
Other small animals, such as the ferret, guinea pig, cotton rat, and hamster, have also been used for vaccine safety studies. 25,35 The guinea pig is occasionally used for local tolerance studies and routinely for the abnormal toxicity/general safety test 36 (a quality control test conducted to ensure no contaminants have been introduced during product manufacturing), which is required by some countries (eg, Russia, China). Guinea pigs are relatively unique in that mast cell degranulation may be initiated with immunoglobulin G (IgG) as well as IgE, so high IgG titers may result in a species-specific anaphylactic-like response. 37 Ferrets have been used primarily for vaccine safety studies for influenza vaccines, as they are susceptible to the disease and often serve as both the nonclinical efficacy and toxicology species. 38,39 These species have the disadvantages of small size, generally limited HCD, and limited published strain-specific background findings with incidences. 25 There may also be limitations on the availability of assays and reagents (eg, for measuring immune responses, acute-phase reactants) for some of these species.
Nonhuman Primates
Nonhuman primates have a number of advantages for vaccine studies, such as having relatively large muscles to enable IM administration of a full human dose and a high blood volume for sampling. The muscle size also enables use of special delivery techniques, such as electroporation devices. Nonhuman primates are not commonly used for toxicology studies of vaccines against infectious disease, although they are used often as nonclinical models to evaluate immune induction to vaccine candidates and sometimes to support juvenile studies. Nonhuman primates should be used for toxicology studies only if there are no other relevant animal species, which is may be the case with therapeutic vaccines, with or without coadministeration with biotherapeutic molecules. When using NHP, the source of the animals should be considered. For example, cynomolgus monkeys (
Minipigs
As with NHP, minipigs have the advantage of a large muscle mass to facilitate IM administration of a full human dose and have ample blood volume for repeated blood collections. They are not commonly used as a toxicology species for vaccines, but have been developed as a model for studying intradermal or topical vaccines. 43 Minipigs have the advantage of having similar physiological, skin microanatomical, and immunological characteristics as humans. 44,45 A recent increase in minipig publications 46 may make this a more commonly used species going forward. There may be limitations on anatomic knowledge for technical staff, tissue trimming guidelines, availability of assays and reagents (eg, for measuring immune responses, acute-phase reactants), varying degrees of pathologist familiarity with evaluating the species, and HCD.
Dogs
From a practical perspective, the dog could be viewed as a good candidate species for vaccine toxicology studies due to their large size (amenable to full human dose), substantial blood volume enabling serial sampling, and an abundance of publicly available and/or proprietary HCD. However, dogs have seldom been used as models for studying vaccine pharmacology, efficacy, or safety. An important consideration in the use of dogs is their high sensitivity to polysorbate 80 (Tween80), a common component of vaccines, which causes an anaphylactic-like reaction. 47,48 There is also little historical immunology-specific data, limited availability of assays and reagents for Beagle dogs as compared to rodents, 49 and studies of comparative immunology between humans and dogs are limited.
Study Groups
Study groups should include, at a minimum, a saline control group (formulation buffer may also be used but is less common) and vaccine group(s). If an adjuvant is a component of the vaccine formulation, inclusion of an adjuvant control group should be considered to comfortably interpret the clinical pathology and histological findings in those vaccine groups. If the adjuvant has an extensive safety database and is in numerous marketed products, an adjuvant control group may not be necessary (eg, aluminum salt adjuvants such as AlPO4, Al[OH]3). Dose groups for therapeutic vaccines may vary depending on the components of the vaccine. Animal numbers for vaccine studies are determined similarly as in other routine toxicology studies and specific recommendations are included in the WHO guidance documents. 5,6
Dose Volume and Administration Location
Most vaccines are administered IM, although intradermal, subcutaneous, oral, and intranasal (IN) administrations are also sometimes used. Although uncommon, intravenous administration of vaccines may be effective. 50 Because the WHO guidance documents indicate that the full human dose should be administered whenever feasible, IM administration of the full human dose (typically 0.5 mL) to the rabbit or other large animals is straightforward, and the hindlimb or the dorsolumbar musculature is most commonly used. When the dosing regimen requires multiple administrations, IM dosing may alternate between the left and right sides. Administration of the full human dose volume into a single muscle of rodents or other small animals, however, is generally not feasible. A maximum IM dose volume in the rat at one site is recommended to be ≤250 µL and must be in a muscle large enough to support the dose volume. 25,51 In general, this limits the injection sites to the hindlimb and the volume is typically split between both hindlimbs, although lower dose volumes may be administered in the lumbar muscles. In the rat, the quadriceps muscles are most commonly used; however, other muscles of the hindlimb, including the biceps femoris or gastrocnemius muscles, may be utilized.
Mice or other small species will require much lower dose volumes (≤100 µL) per IM site. When administering lower doses than those anticipated for use in humans, there should be immunological data to support an immune response to the dose administered. A mg/kg exposure multiple for the formulation components may be a scientifically valid alternative for discussion with regulatory agencies. 5,6 Alternatively, it may be possible in some cases to increase the concentration of the vaccine antigens and adjuvants such that the full human dose of these components may be administered. However, this is not a commonly used approach and there are possible concentration-based immunological or physical confounding effects that could be introduced. If such an approach is taken, however, risk may be mitigated through evaluation in non-Good Laboratory Practices studies prior to initiating the pivotal toxicology studies.
Subcutaneous vaccine administration sites may differ by species, although most species may be readily injected in the interscapular region, dorsal neck, shoulder, or flank. Subcutaneous locations in most species except for mouse may accommodate up to 5 mL/kg/site or the full human dose. 52
Intranasal administration delivers the vaccine to the nasal mucosa and IN vaccines have been developed mainly for influenza but have been tested for a number of respiratory viruses.
39,53
The IN route offers the induction of both a systemic and local/mucosal immunity and the production of higher local IgA antibody and cell-mediated immunity compared to parental routes, as its main advantages.
54
It also provides a noninvasive or needle-free delivery system, which contributes to patient comfort/compliance, and requires a relatively smaller antigenic dose.
55
Clinically, the main challenges for IN delivery of vaccines include the potential passage of the vaccine to the olfactory bulb of the brain and other potentially adverse effects in patients with allergies or asthma.
55
To adequately assess the potential effects of vaccine delivery to the brain, species with nasal anatomy closer to that of humans, such as the cynomolgus monkey, should be considered.
55,56
Species such as the rat and dog, in which the olfactory bulb is protected by the presence of a transverse lamina, are generally not considered suitable, as the vaccine may not reach the olfactory bulb, limiting reliable prediction of brain delivery-associated effects.
56
A further disadvantage of using rodents and other smaller animals for safety testing of IN vaccines includes the potential for the vaccine to reach the lungs. Evaluation of the administration site in IN vaccines may be challenging, particularly in species with a large nasal mucosal surface area. Mucosal delivery of vaccines also includes oral vaccines as have been developed for
Intradermal or transcutaneous vaccine administration has appeal, as this method of delivery tends to use less material (dose-sparing), generally does not require adjuvant, and serves as a “needle-free” alternative to vaccination. 58 Microneedle techniques are also utilized for intradermal vaccine administration. 59 To date, there has been mixed success with intradermal delivery of live-attenuated vaccines. 58,60 –63 These methods of delivery require use of adequate delivery devices, of which there are several in development. 64 –66 Because of the similarity with human skin, the pig is often used as a nonclinical model for safety testing. 67 The pathologist should be aware of “dippity pig syndrome” that is characterized by reddened skin and open sores along the dorsum in healthy young pigs, which may overlap the region of dose administration and confound the toxicology interpretation (Figure 1). The lesion usually begins resolving within a few days of onset. A cause for this syndrome is uncertain. 68
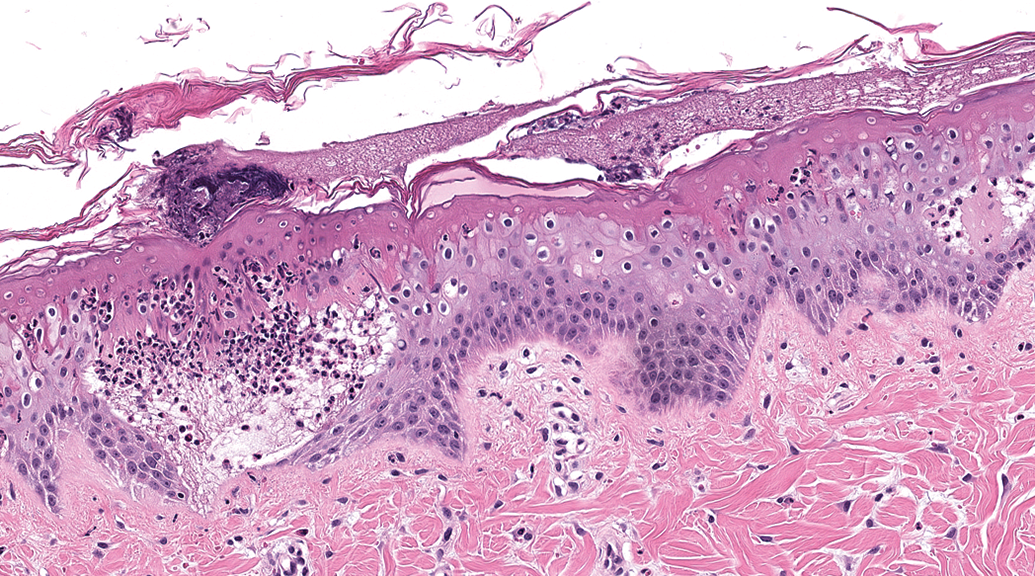
Photomicrograph of dorsal skin from a minipig with “dippity pig” syndrome stained with hematoxylin and eosin. Note the presence of epithelial edema, infiltrates of neutrophils, and superficial crust.
Dose Number and Dosing Interval
The dose number and dosing interval should reflect the strategy planned for clinical use. The number of doses should, at a minimum, represent the anticipated number of clinical doses given in a regimen over a year; however, it is suggested in repeat-dose toxicology studies to add an additional dose (N + 1, where N = the number of doses to be administered clinically). Prophylactic vaccines against infectious agents are often clinically administered periodically, with months or even years between administrations. In toxicology studies, the time between dose administrations may be shortened. Doses are usually administered 2 to 3 weeks apart, based on the kinetics of the primary and secondary antibody response in the animal species. Time points separated by less than 2 weeks are generally not used. For therapeutic vaccines, the timing of dose administration and number of doses generally reflect the clinical dosing regimen for at least 1 cycle (for cancer treatment expectations, refer to ICHS9; Non-Clinical Evaluation of Anticancer Pharmaceuticals), and dose administration methods should be as close to that used in the clinic as possible (eg, electroporation devices).
Technical Considerations of Dose Administration
Since the primary target tissue in most vaccine studies is the injection site, accurate administration of the test material is essential to facilitate identification and precise collection of the injection site(s) at necropsy. Some test facilities have found it advantageous to briefly anesthetize small species with isoflurane for the IM injections to ensure accuracy of administration and reduce local trauma due to animal movement during the injection. In the authors’ experience, use of anesthesia has not been associated with any confounding effects in study parameters. In-life techniques for facilitating the accurate administration of the test material include (1) anatomic localization of the injection site; (2) application of specific dosing parameters, including needle length/gauge, injection approach, and directionality; (3) consideration of species variation in anatomy; (4) manual or chemical restraint as necessary; and (5) utilization of appropriate marking of the area of the injection. At necropsy, these techniques may be coupled with collection of wide margins to ensure the accurate collection of the injection site(s).
Injection Sites
Injection site delineation is important to ensure accurate collection at necropsy. Injection site collection from animals with larger muscle area, such as the rabbit and NHP, may be more challenging to sample as compared to those with much smaller muscles. The injection site may be shaved and marked to delineate the location of needle entry and vaccine deposition. This is often done with a superficial pen or ink marking at the time of injection, which sometimes requires repeated application at regular intervals due to fading. An alternative method of marking has been to delineate an injection area by permanent tattoos to surround or otherwise highlight a central area for injection. This requires tattoo administration at least a week prior to dosing but can reduce staff effort and time in remarking the skin over the course of the study. Histologically, tattoo ink pigment may be associated with minimal lymphohistiocytic infiltrates within the dermis (Figure 2).
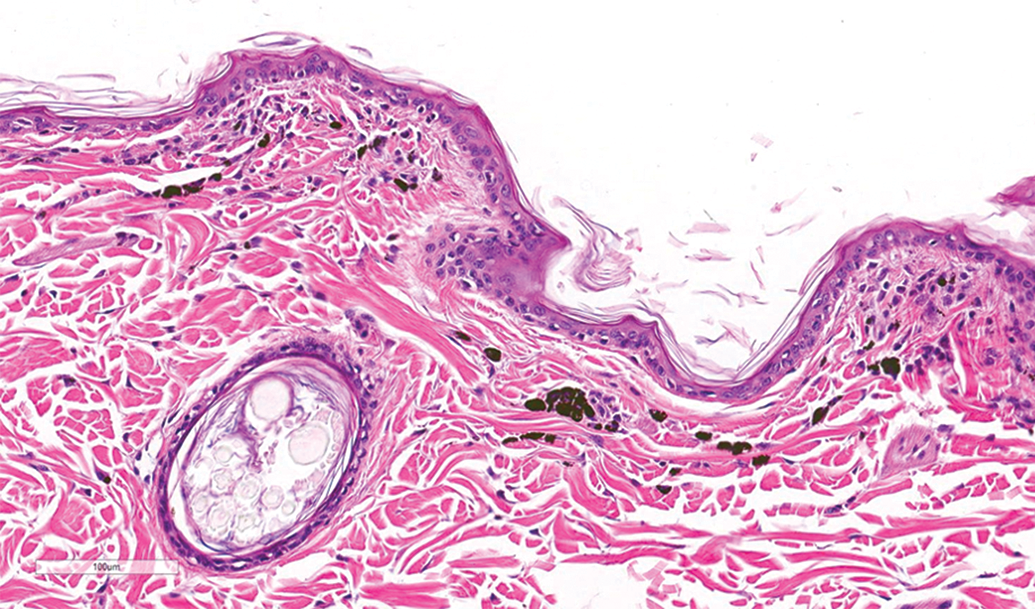
Photomicrograph of skin from tattoo site near intramuscular injection site in a ferret stained with hematoxylin and eosin. Pigment-laden macrophages and small numbers of mixed lymphocytes and plasma cells infiltrate the superficial dermis in a focal area.
Exterior marking has the benefit of easy identification of the injection site for the technical staff and may have benefit for the necropsy staff. However, the highly pliant nature of rabbit skin may make skin marking an inconsistent way to delineate the desired IM target. Alternative methods of identifying the injection site other than by marking may allow for more consistent and better dosing and collection. The use of anatomic landmarks may provide consistent localization of the site of administration, assisting staff during in-life and postmortem procedures (Figure 3A-C). The most reliable anatomic landmarks tend to be bones in the region of the injection site(s). For example, the femur is a commonly used landmark for injections into the quadriceps or biceps femoris muscles, with the articulation of the hip and the knee forming 2 points of reference and the length of the femur forming another. By palpating hip and knee joints, the midfemur may be located, and the vaccine consistently administered into the adjacent defined muscle belly such as quadriceps or biceps femoris muscles. This method also improves collection of sites, as necropsy and tissue trimming staff can use the same landmarks to locate the injection site region.
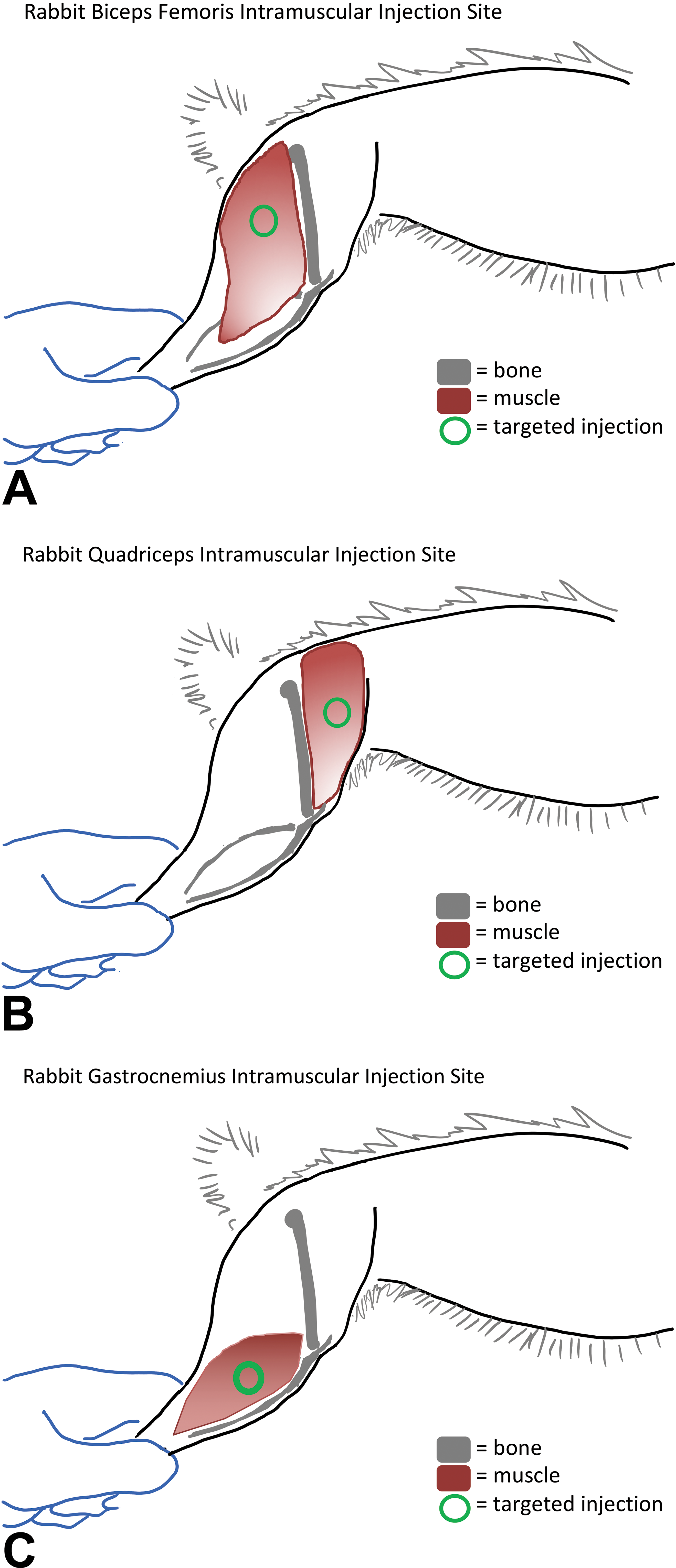
Diagram of injection site localization in rabbits. A, Diagram of injection site location in biceps femoris. Injection should be perpendicular to skin/muscle belly, at approximate midpoint of femur and 1 to 2 cm caudal to femur. B, Diagram of injection site location in quadriceps. Injection should be perpendicular to skin/muscle belly, at approximate midpoint of femur and 1 to 2 cm cranial to femur. C, Diagram of injection site location in gastrocnemius. Injection should be perpendicular to skin/muscle belly, just proximal to the midpoint of tibia/fibula and ∼1 cm caudal to tibia.
Directionality and Depth of Injections
The direction and depth of injection should be considered. The technical staff should be trained to have consistent orientation, injection site direction, and injection depth. For example, if the area of injection is marked on the skin, the technical staff should pay attention to where the material is ultimately injected—the needle might enter the skin in the delineated region, but be injected outside the demarcated area (Figure 4). This is particularly a concern for subcutaneous injections, but is also important for IM injections. Additionally, needles for IM injections should be oriented consistently (preferably perpendicular to the muscle) to provide greater accuracy and consistency, and reduced effects on regional structures such as the sciatic nerve and knee joint.
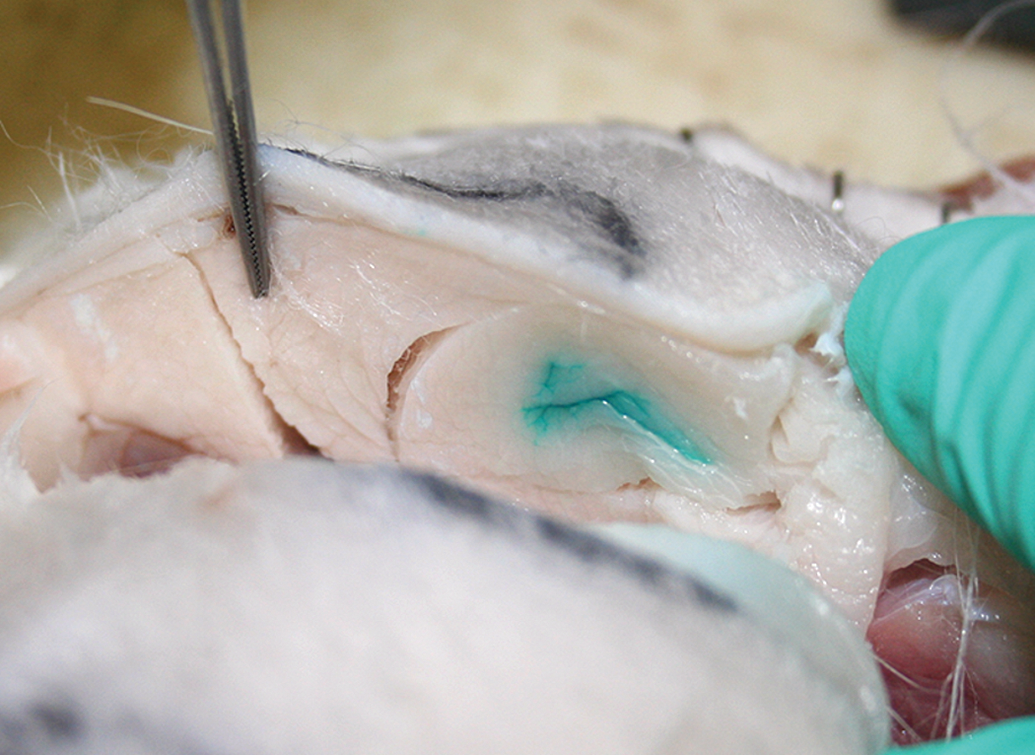
Macroscopic photo after intramuscular injection of green marking dye in a rabbit. Note that the injected material is in line with or external to the circle inked on the skin to mark the outer rim of the injection site. Careful attention to needle direction during dose administration is essential for accurate injection site collection at necropsy.
Needle Length
Needle length is important to consider both within a study and between studies to reduce nonspecific variation in the injection site histology. For IM injections, longer needles in rodents may run the risk of causing deeper than desired injection of material, such as into the stifle joint region, muscle bundles deep to the targeted muscle, and in or around the sciatic nerve. A needle length of 3/8″ is appropriate for IM delivery in many animals, including rabbits, monkeys, dogs, and ferrets. Longer needles such as 5/8″ may be associated with administration of material into underlying muscle beds, even in NHP. Shorter needles may be appropriate in smaller species, such as mice, rats, and hamsters (eg, 3/16″) and should be considered on a case-by-case basis. In general, the proximal portion of the muscle is thicker than the distal portions; thus, there is a lower risk of passing through the intended muscle. Variations in needle length may impact the consistency of the location of where material is injected, even if directionality is consistent.
In-Life Assessments
The in-life assessments in vaccine studies are the same as for other repeat-dose toxicology studies with inclusion of postvaccine administration-specific end points, such as body temperature and injection site evaluations. Specific recommendations are detailed in the WHO 5,6 and other regulatory guidance documents.
Local Toxicity/Reactogenicity
Evaluation of the vaccine administration site is an important component of the in-life assessment in vaccine studies. Assessment of the injection site often utilizes a prospectively defined scoring system for erythema (redness) and edema (swelling) for which many use a modification of scoring of skin irritation described by Draize et al, 69 as well as characterization of other injection site changes such as vesiculation, ulceration, eschar formation, and any potential evidence of significant toxicity, including limb impairment. However, use of a specific scoring system is not a requirement if injection site observations over the course of the study are accurately captured and described. Additionally, body temperature before and after vaccine administration is evaluated and may be measured at the same time as the injection site observations. 6
Clinical Pathology
The clinical pathology panels in vaccine studies are similar to those for small molecules and include blood (hematology, clinical chemistry, and coagulation) and urine (urinalysis). 70,71 Additionally, 1 or more serum acute-phase proteins (APPs) are typically measured as indicators of an immune response. 72,73 Cytokines may also be used as a measure of the immune response, but are less common because of their short half-life, wide physiological range, and high inter and intra-animal variation.
Timing of blood collection for clinical pathology is usually 1 to 3 days after the first and last dose administrations and at the recovery-phase necropsy, although other time points may be included. Blood collection 1 to 3 days after dosing is timed to capture any acute-phase response, which is usually short-lived, depending on the parameter being evaluated. For rodent studies, blood volume and collection limitations are often an issue. For studies in which multiple blood collections for clinical pathology and serology are required, rats may be divided into subgroups to allow collection for various assays (such as half for hematology and other half for chemistry; other biomarkers or serology may be accommodated with either subset of animals depending on the volume requirements or, if needed, an additional cohort of animals). Serum collection for assessing the antigen-induced immune response usually occurs prior to dose initiation and at the end of the dosing and recovery phases; however, time points could vary depending on study needs (eg, when comparing different prime-boost regimens).
Clinical Chemistry and APPs
The clinical chemistry panel includes various end points for assessment of general health as well as markers for inflammation and APP. Creatine kinase (CK) is occasionally included as a marker of muscle damage at the injection site and may help differentiate acute-phase responses associated with the vaccine components from those occurring secondary to physical trauma. However, often there is poor concordance between local tissue injury (measured by CK) and the systemic inflammatory response (measured by APPs). 72
Due to the lack of sensitivity in the routine clinical chemistry parameters to monitor an acute-phase response, fibrinogen and species-specific APPs are routinely measured to assess potential vaccine reactogenicity. Hepatic synthesis of APPs (positive APPs) may downregulate the synthesis of other proteins, including albumin, which are identified as negative APPs. However, there are no defined cutoffs for changes in these APPs to help with decision-making. The presence of an acute-phase response in vaccine studies is of little significance to the overall study conclusions if the immune response to the antigen is as expected and no unexpected or adverse systemic findings are observed.
Species differences in APPs must be considered when designing a vaccine toxicology study. For example, C-reactive protein (CRP) is a sensitive indicator of the acute-phase response and the most commonly utilized major APP for humans, rabbits, dogs, and NHP. 72 However, CRP is a poor marker of an acute-phase response in rodents. Instead, the major APPs for rodents are α1-acid glycoprotein and α2-macroglobulin. 73 When measuring APPs, it is very important to understand how an assay is being performed; there may be notable differences in results due to differences in assay sensitivity between different instruments and reagent systems, and thus data should not be compared between different methods and laboratories. Some of these APPs (such as CRP, haptoglobin, and serum amyloid A) may utilize human assays or species-specific assays, so species cross-reactivity should be critically evaluated and assay validation should be performed in relevant species to assure satisfactory performance.
Evidence of an acute-phase response indicative of reactogenicity to the vaccine formulation may be evident in the routine chemistry panel as increases in globulins and decreases in albumin. Globulins increase slowly after vaccine administration and are long-lived, sometimes remaining elevated until the end of the recovery period. These elevations are often very slight (<1.2× relevant control) and not always statistically significant. Decreases in albumin are often evident within days of vaccine administration and as with globulins; the change may be slight and variably statistically significant. Therefore, reductions in the albumin:globulin (A:G) ratio after the first vaccine administration may reflect reduced albumin, whereas later in the study, the decreased A:G ratio may reflect increased globulins.
Hematology
Changes in hematology (complete blood count) parameters are useful in assessing the inflammatory response caused by the administration of vaccines and their components. Findings should be compared to concurrent controls, even when baseline data (prior to dose administration) are available, as hematology data can be influenced by study procedures. Most commonly observed changes in hematology parameters include increased white blood cells with increased neutrophils, monocytes, and/or lymphocytes. Eosinophils and basophils are not reliable as indicators for inflammation, as they are generally present in very small numbers. The inflammatory response is often more prominent with vaccines formulated with adjuvants. The inflammatory leukogram tends to be variable depending on the antigen and adjuvant and may range from negligible to remarkable. Different adjuvants can impact the severity and character of hematology changes. Stress/excitement may also result in increased neutrophils, monocytes, and/or lymphocyte counts, and study procedures should be taken into consideration for identifying vaccine/adjuvant-related hematology changes.
Coagulation
The coagulation panel typically includes coagulation times (prothrombin time and activated partial thromboplastin time [aPTT]) and fibrinogen. Fibrinogen is a clotting factor and an APP that is present at a higher basal concentration (generally 200-300 mg/dL range) in the blood to maintain normal clotting. Due to its high basal concentration and the upper limit of the assay, the maximal increase in fibrinogen is typically not more than 4- to 5-fold, making it a minor to moderate reactant for most species. However, fibrinogen is still an effective marker for inflammation/acute-phase reaction in most species, and small increases are usually meaningful. Clinically increased CRP levels are known to cause false increases in aPTT, as CRP has binding affinity for phospholipids, interfering with the aPTT assay. 74
Other Considerations in Clinical Pathology Findings
Intramuscular injection, a common route of administration for vaccines, causes local tissue injury and incites an inflammatory response, creating more variability in data. Sometimes, it can be difficult to parse out changes attributed to the injection procedure from those caused by the immune response to vaccine/adjuvants. In such cases, the saline control group is often helpful in identifying changes attributed to the injection procedure, as well as the evaluation of CK, a marker of muscle damage that could indicate injection site muscle trauma.
Therapeutic vaccines, which contain other active components, may have changes related to those components or to the vaccine target. Immune oncology checkpoint inhibitors, for example, can elevate the white blood cell count, particularly lymphocytes. 75 Therapeutic vaccines may also have intended alterations in clinical pathology parameters. For instance, vaccines targeting cholesterol biogenesis would be anticipated to have reductions in serum cholesterol concentrations as a therapeutic end point.
Anatomic Pathology
In toxicology studies for prophylactic vaccines, the pathology evaluation focuses primarily on the local response to the vaccine at the injection site and draining lymph node. Different concentrations of antigen infrequently alter the findings at the injection site and draining lymph node, so in this way, antigen dose often has little impact on the pathology findings. The dose and type of adjuvant in the vaccine formulation may have a greater impact on the pathology data than the antigens, and thus different formulations under consideration for human testing should be evaluated in the toxicology study. Because the antigen is a foreign protein, there is rarely a concern for immune cross-reactivity with an endogenous protein. This is in contrast to therapeutic vaccines, for which an endogenous antigen is used, and therefore tissues in which cross-reactivity may be of concern should be evaluated histologically.
Necropsy
Macroscopic Findings
Vaccines formulated with an adjuvant often have gross enlargement of the draining lymph nodes, in contrast to vaccines formulated without adjuvant in which lymph node enlargement is less frequent. The spleen may also be enlarged. Macroscopic injection site findings may range from unremarkable to ulceration or scabbing. Changes at the injection site should be differentiated from damage associated with injection site preparation (shaving and disinfection). However, most often, there are either no findings or slight discoloration of the skin or underlying muscle in the injection site region. Therapeutic vaccines may contain other active components, which may be associated with gross treatment-related findings (although in the experience of the working group, this is uncommon).
Tissue Lists and Organ Weights
Organ weights are routinely collected in vaccine toxicology studies consistent with the Society of Toxicologic Pathology (STP) best-practice guidance on organ weight collection. 76,77 In vaccines against infectious diseases, there is often no change in organ weights, although the spleen may on occasion have slight increases in absolute and/or relative weight. Although not recommended in the STP best-practice guideline, draining lymph nodes may be weighed to help in the assessment of immunogenicity and lymphoid reactivity. However, the value should be considered on a study- and species-specific basis. 78 The value of weighing draining lymph nodes in rodents and other small laboratory animals may be limited by interanimal variation in immune reactivity, sampling error, and small size, all of which may contribute to wide variation in lymph node weight.
The WHO guidelines include an extensive tissue list. 5 The majority of tissues suggested for collection are consistent with those collected for routine toxicology studies (including the injection site[s] and draining lymph nodes), but with the addition of larynx and lacrimal gland. Presumably, microscopic evaluation of the larynx is intended for inhaled or ingested vaccines. The inclusion of lacrimal gland was a result of a squalene-based adjuvant-related finding in rabbit lacrimal glands. 79 Because of the nature of vaccine study design (ie, generally no dose ranges), all tissues from all dose groups are processed to slide and evaluated at the end of the dosing period. In the case that multiple dose levels of the same formulation are evaluated, limiting evaluation to targets only at lower dose groups may be acceptable. Adequate collection of the injection site(s) and their corresponding draining lymph node(s) (Table 3) is essential, so care must be taken in ensuring that these are properly and consistently collected. Reliable collection of the injection site depends on consistency in the location of dose administration (see section “Dose Administration”). The draining lymph nodes can easily be identified or verified prestudy by methylene blue injection at the site of intended administration in the toxicology species of choice. Tissue collection at the recovery phase is intended to evaluate resolution of findings observed at the end of the dosing phase and potential delayed toxicities. The same tissues should be collected at the recovery-phase necropsy as at the dosing-phase necropsy. Microscopic evaluation of tissues from recovery phase animals may include all tissues collected or could be limited to the injection site, draining lymph nodes, spleen, and any organs with changes at the end of the dosing phase, with or without a subset of additional tissues as deemed appropriate.
Draining Lymph Nodes.
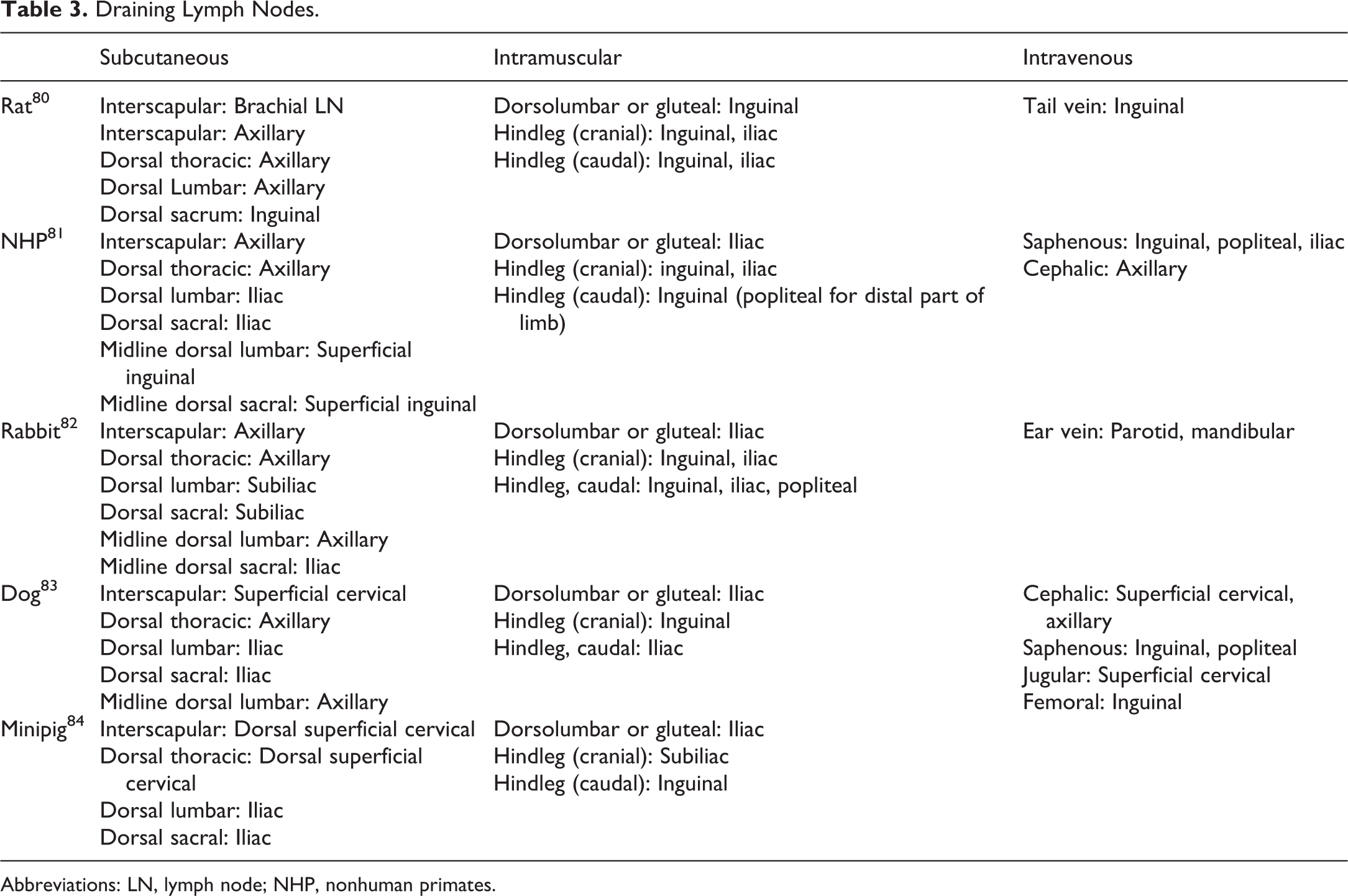
Abbreviations: LN, lymph node; NHP, nonhuman primates.
Collection and Trimming of the Injection Site
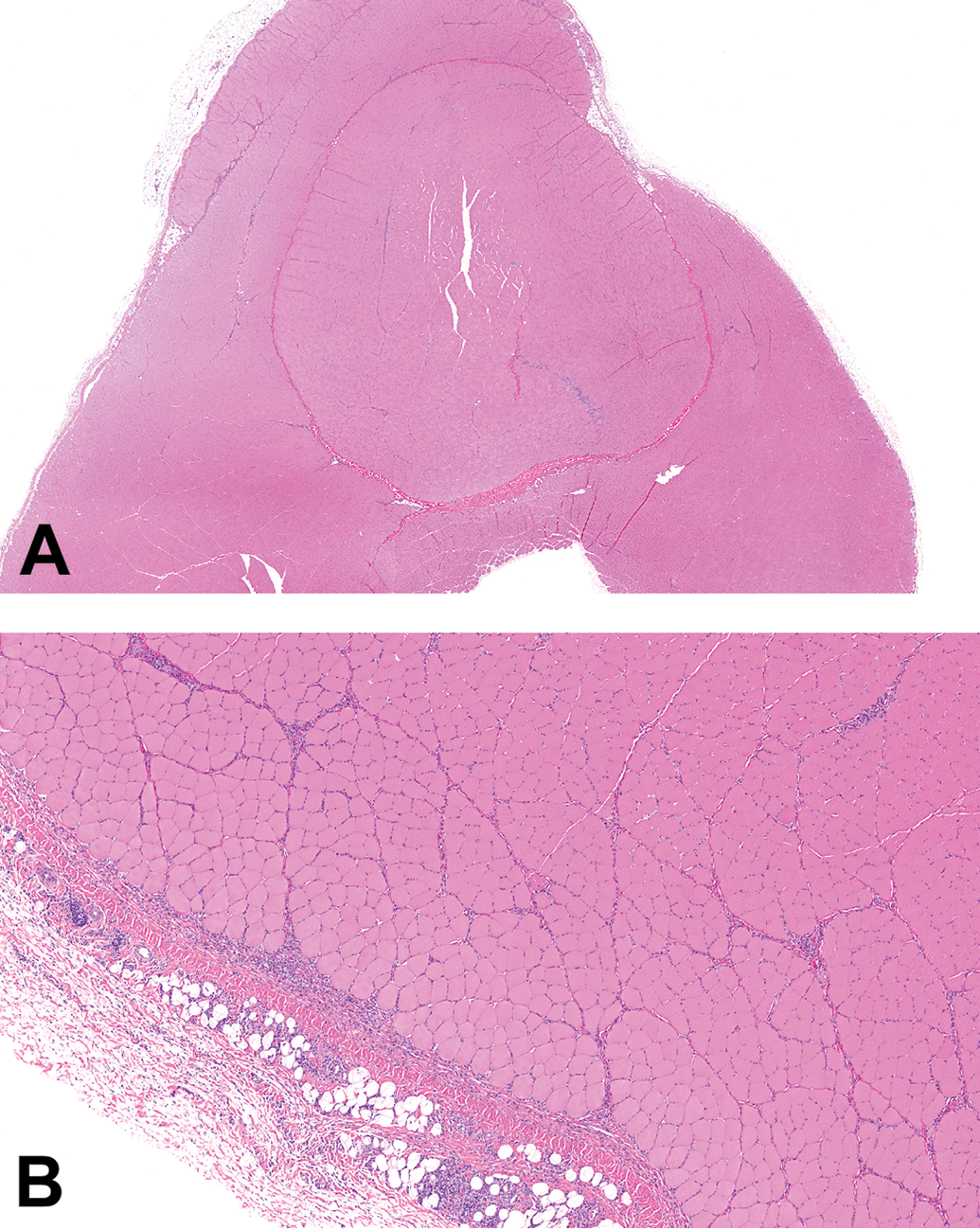
For IM injections, the injection site muscle and overlying skin should be collected and evaluated. These do not need to be kept together and may be evaluated separately, although retaining the overlying skin with the muscle may reveal the needle tract. In rabbits, where the skin is only loosely connected to the underlying muscle, maintenance of the muscle with overlying skin may not add value for identifying injection tracts. At the time of collection, staples may be used at the edge of the excision site in rabbits to securely attach the skin to the underlying musculature and avoid the slipping of the skin over the muscle and the eventual differential shrinkage of the skin compared to the muscle upon fixation (Figure 5). After fixation, the staples may be removed, and the tissues will remain opposed for trimming. To preserve anatomical correlation after necropsy, the hindlimb of animals up to rabbit size may be successfully removed and fixed in totality prior to trimming and embedding. For evaluation of the muscle of the injection site, it is helpful to have 3 to 5 cross sections of the region for histological evaluation (depending on the animal size), as there will be variation in the extent of the change depending on whether the evaluation is at the center of the injection site or along the margins (Figure 6). However, other strategies may also be successful. Inclusion of the deep margins of the muscle at the injection site is often helpful in evaluating the full extent of the changes.
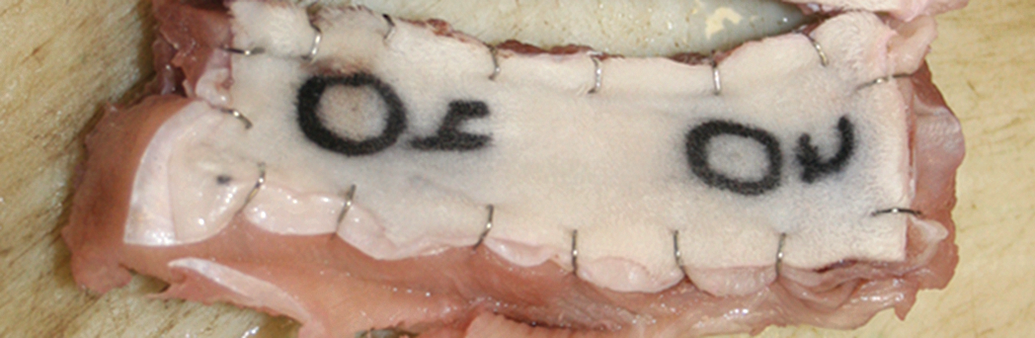
Macroscopic photo of an excised intramuscular injection site in the rabbit epaxial musculature and skin (circles indicate injection sites 1 and 2). The use of surgical staples along the cut edge of skin and muscle helps to maintain in vivo orientation. Every cut edge of skin
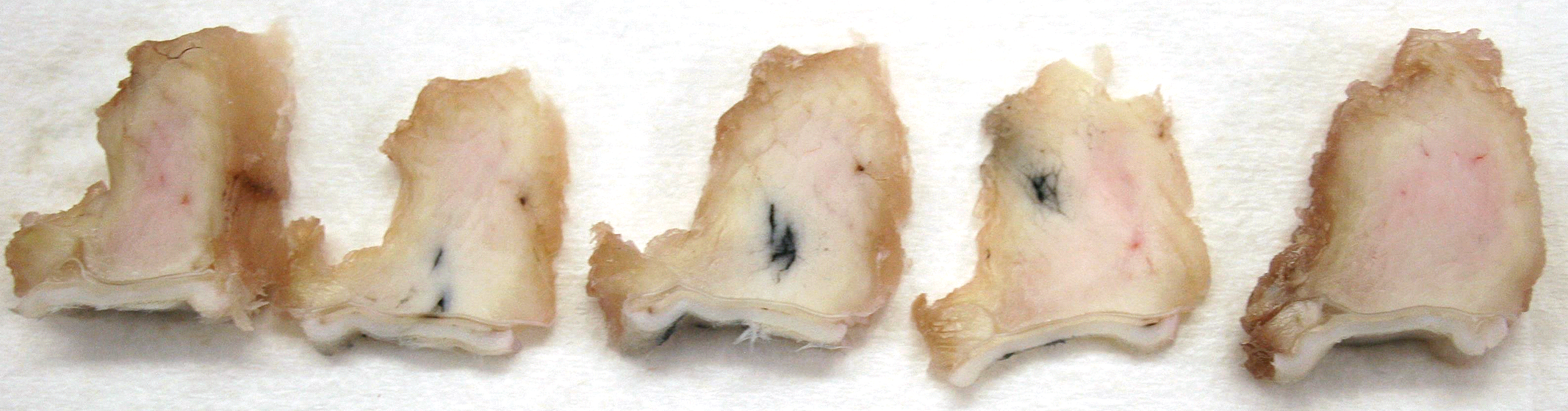
Macroscopic photo of 5 cross sections through an intramuscular (IM) injection site in rabbit after IM injection of green marking dye. Note that the injection site is seen only in the middle 3 sections. Injected dye is seen at varying depths in the muscle as well.
For subcutaneous injections, the underlying muscle, overlying skin, and the subcutaneous compartment are typically collected and evaluated. These are often kept together and evaluated as a whole. Similar to IM injection sites, evaluation of multiple spaced cross sections provides a method of ensuring accurate assessment of the injection site despite possible dosing variability.
For IN vaccines, multiple levels of the nasal cavity should be collected for histopathology 85 –87 along with the nasal-associated lymphoid tissue (NALT), tonsillar tissue (if applicable), the nasopharynx/pharynx, larynx, trachea, draining lymph node for the respiratory tract such as (tracheo)bronchial lymph nodes, lungs, and the brain. In rodents (which lack tonsils), NALT located in the ventral aspects of the caudal nasal cavity at the entrance of the nasopharynx is considered to be the equivalent of the Waldeyer ring in humans and can be assessed on nasal cavity sections; in monkeys, NALT is present throughout the nasal cavity and tonsillar tissue is present within the nasopharyngeal septum. 56 Dogs have abundant tonsillar tissue that can be sampled separately, but as discussed before, dogs are rarely used in IN vaccine studies since the presence of a transverse lamina and olfactory recess in the nasal cavity in this species limits their use to predict brain delivery-associated effects.
Other Considerations for Tissue Collection
Although damage to the skeletal muscle and trauma or vaccine-induced inflammation in the region around the sciatic nerve or joint may be common issues in IM vaccine studies, they may become particularly problematic if they are more frequently identified in the vaccine groups (Figure 7). To rule out any potential questions regarding systemic effects on the skeletal muscle, nerves, or joints, it may be of value to collect these tissues from sites distant to the site of vaccine administration. For example, if both the hindlimbs were used for injection, collection of a skeletal muscle, joint, and nerve from the forelimb or, for rodents, the entire forelimb, may be beneficial. These additional tissues may be held in reserve for evaluation if needed or may be processed to slide and evaluated as part of the study.
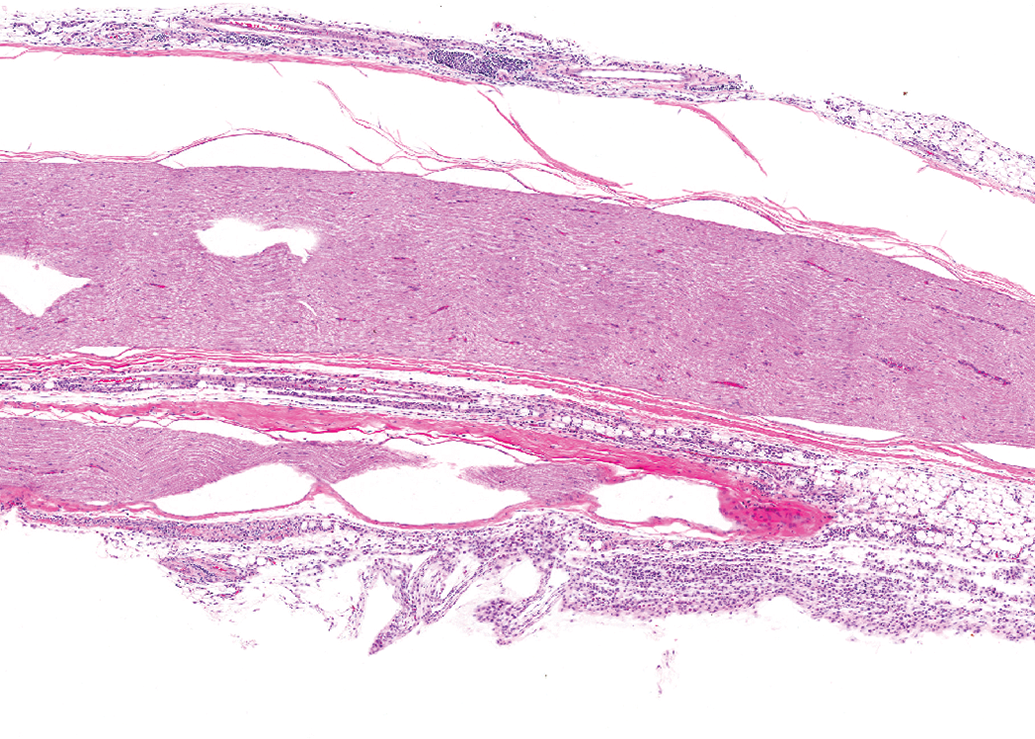
Photomicrograph of sciatic nerve stained by hematoxylin and eosin from a Wistar (Han) rat administered ISCOMATRIX. Perineural inflammatory cell infiltrates are present around the nerve due to vaccine administration at or near the sciatic nerve.
Microscopic Findings
Microscopic findings in vaccine studies are usually at the injection site and surrounding tissues and in the draining lymph nodes. There may also be findings in other lymphoid organs, such as the spleen. Comparing the microscopic findings of the vaccine to those of the saline controls is important to understand the effect of the vaccine formulation in its entirety. However, within the microscopic component of the pathology report, it is important to differentiate, where possible, the effects of adjuvant from the effects of antigen. For studies that lack an adjuvant control, this may require reference to historical data. Differences between microscopic findings with the adjuvant alone and the vaccine formulated with the adjuvant are relatively infrequent. Since other components such as immune checkpoint inhibitors are included in therapeutic vaccines, changes specific to those components may be evident histologically (such as increased immune cell infiltrates into tissues). 75,88 However, details on the microscopic findings outside the injection site(s) and draining lymph nodes with various therapeutic vaccine regimens are beyond the scope of this article.
Injection Sites
Most vaccine studies have multiple doses administered over the period of 2 weeks or more. Therefore, microscopic findings may be complex, as they range from acute to chronic with responses not only to the injected material but also to physical trauma from the needle or injected material (eg, tissue compression). Determining how many injection sites should be evaluated microscopically should be based on the study design. If a full dose was administered in alternating sites, then evaluation of all sites can give a more temporal understanding of the injection site findings. In this case, the injection sites should be independently identified and evaluated. If the dose was administered at multiple sites at each dosing day, then it would be adequate to evaluate a single site (with collection of all sites). In this case, if multiple sites are evaluated, findings may be recorded individually or as a composite diagnosis(es). For therapeutic vaccines, different components of the regimen may be administered at different sites; injection sites for each unique component are usually evaluated independently. Regardless of how many injection sites are evaluated, it is helpful to include multiple levels of each injection site, if possible, to accurately capture the microscopic characteristics and severity (Figures 6 and 8). If microscopic findings are not identified, particularly with formulations containing adjuvants, it may be of value to either section deeper into the block (rats and mice) or retrim wet tissue to ensure accurate evaluation of injection site findings.
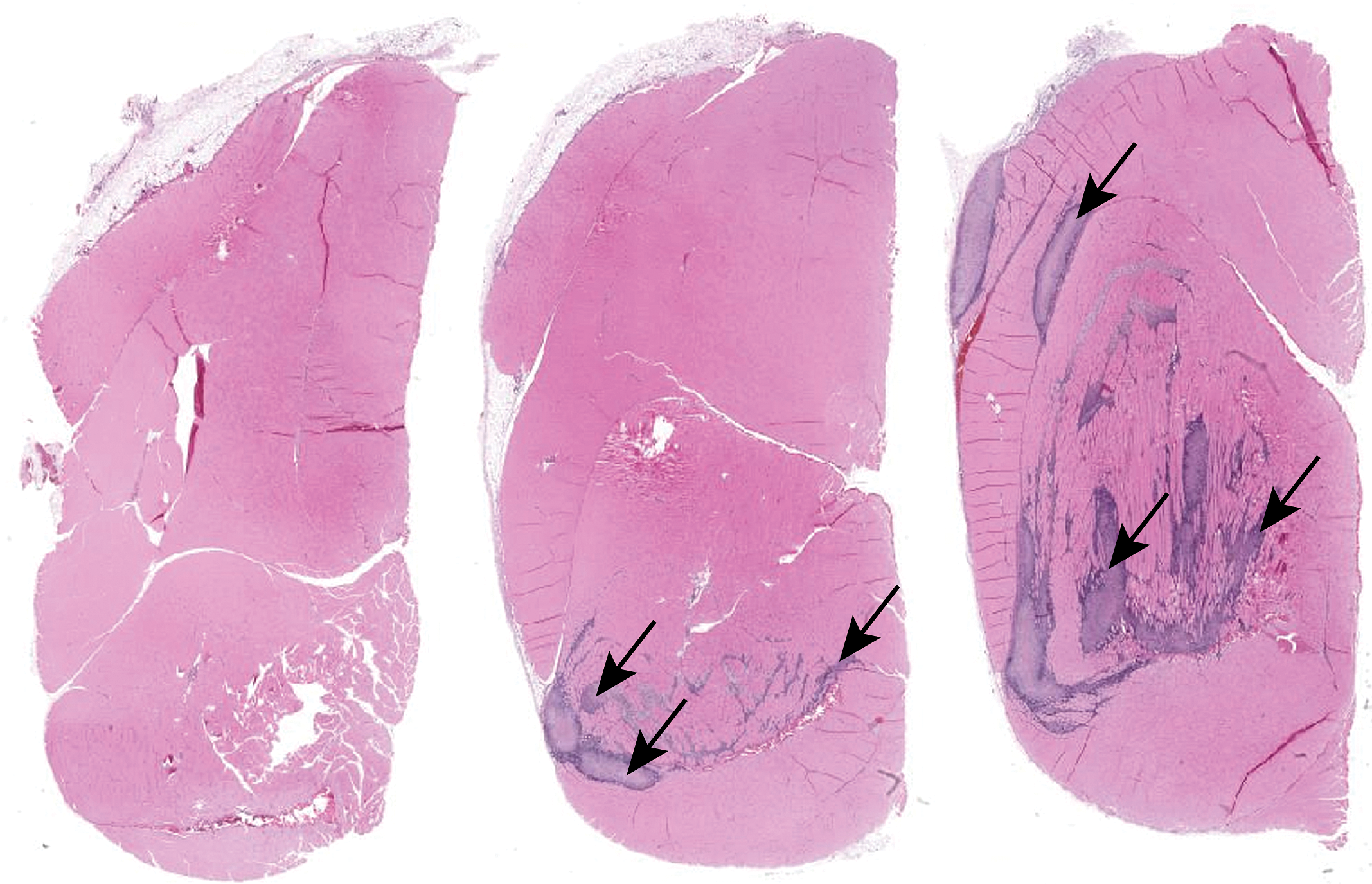
Photomicrograph of the quadriceps muscle injection site stained with hematoxylin and eosin from a Wistar Han rat administered aluminum phosphate adjuvant. Note that inflammatory cell infiltrates are negligible in the section to the left, slight in the middle section, and prominent in the section to the right (arrows). It is important that more than 1 section be evaluated to accurately identify and diagnose injection site findings.
The severity of lesions and type of inflammatory cell infiltrates at the injection site will vary depending on the vaccine components. Most injection sites include some degree of inflammation or inflammatory cell infiltrate, muscle degeneration or necrosis, edema, hemorrhage, and/or fibrosis, depending on the timing of necropsy after administration. Intramuscular administration of saline causes scant inflammation, often comprised of scattered small aggregates of macrophages or other mononuclear inflammatory cells with or without individual myocyte degeneration or necrosis, edema, hemorrhage, and/or fibrosis (Figure 9). Some antigens alone are highly immunogenic and may cause notable inflammation at the injection site, even when formulated without an adjuvant, although many have changes similar to those identified with saline administration.
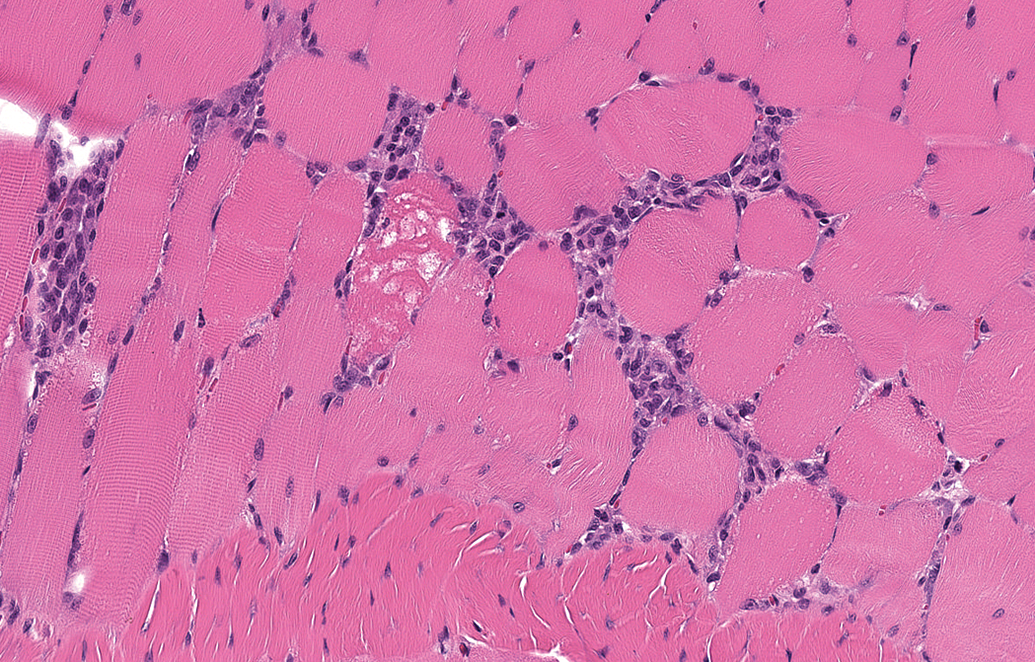
Photomicrograph of an intramuscular injection site stained with hematoxylin and eosin from a rat after administration of saline. Note the myofiber degeneration/necrosis and minimal inflammatory cell infiltrate.
Aluminum-containing formulations consistently result in inflammation at the injection site. Acute changes are characterized by infiltrates of macrophages (multinucleated cells are uncommon), neutrophils (heterophils in rabbits), and eosinophils, with variable degrees of hemorrhage, inflammation, and individual muscle cell degeneration/necrosis (Figure 10). Aggregates of neutrophils may be present within the lesion and generally resolve; these should not be confused with bacterial contamination of the injection site. Basophilic to gray granular material (interpreted to be aluminum) is typically present both within macrophages and extracellularly at the injection site. Adjuvant material may collect along and distend fascial planes at the injection site. Macrophages may surround moderate amounts of extracellular adjuvant material displacing tissue. In the chronic stage, the injection site will have fewer granulocytic cells and the adjuvant-containing macrophages will often be surrounded by lymphocytes and fewer plasma cells (Figure 11A–B). This change is considered an indicator of the recovery process. Adjuvant material and the associated macrophages are usually present at recovery, although often slightly less prominent.
Photomicrograph of the quadriceps muscle injection site stained with hematoxylin and eosin from a Wistar Han rat administered aluminum phosphate adjuvant intramuscular. Note the presence of macrophages and granulocytes (including eosinophils) admixed with granular material and necrotic skeletal myofibers. Original objective 20×.
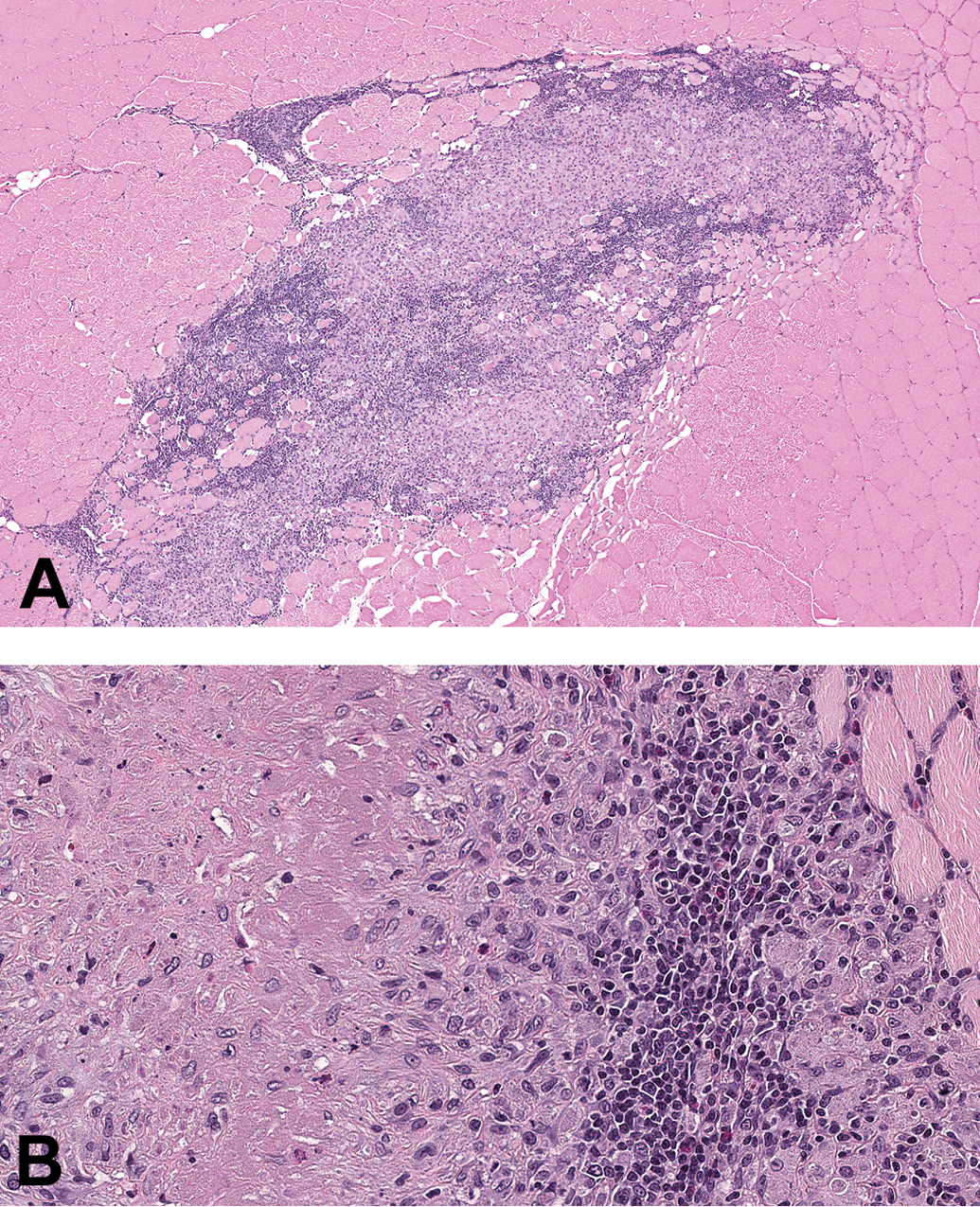
Photomicrographs of the quadriceps muscle injection site stained with hematoxylin and eosin from a Sprague-Dawley rat administered aluminum-containing adjuvant. Chronic inflammation characterized by central adjuvant, cellular debris, and accumulations of macrophages surrounded by lymphocytic infiltrates. (A) Original objective 20× and (B) Original objective 20×.
Non aluminum–containing adjuvants variably incite inflammation and/or necrosis at the injection site. Inflammation, hemorrhage, and edema tracking along the muscle fascial planes may also be evident with these adjuvants (Figure 12A–B). Administration of ODN adjuvants (eg, CpG-ODN), which drive TLR-9 activation, has histological changes similar to saline. However, when they are coadministered with aluminum, changes are generally more severe than aluminum alone. Squalene-based oil emulsion adjuvants may elicit inflammation with vacuolated macrophages.
Photomicrograph of the quadriceps muscle injection site stained with hematoxylin and eosin from a Wistar Han rat administered ISCOMATRIX adjuvant intramuscular. Inflammatory cell infiltrates dissect between muscles and muscle fascicles, rather than displacing tissue. (A) Original objective 2× and (B) Original objective 10×.
Consistent diagnostic terminology is important for comparisons of findings between studies, programs, and organizations. For injection sites that have both the muscle and the skin, it may be helpful to subcategorize the injection site into injection site: muscle and injection site: skin. Because injection sites may be quite complex, splitting diagnoses in the data capture system may reduce, rather than improve, clarity of the injection site findings. Instead, the use of simple diagnostic terms with microscopic details (eg, specific inflammatory cell types, muscle degeneration/necrosis, edema, hemorrhage) specified and/or elaborated upon in the study report narrative may be beneficial. The use of other diagnoses should be included as needed to differentiate test groups (eg, fibrosis). Most injection site inflammatory findings can be diagnosed as infiltrate, cell type or as inflammation, cell type (eg, neutrophilic, mixed, lymphocytic, plasmacytic, mononuclear). The addition of chronicity modifiers may be included (acute, subacute, chronic, or chronic-active). 89 When inflammation includes many macrophages, the authors prefer the use of macrophage or histiocyte as a modifier rather than granulomatous. The use of the term “granulomatous inflammation” may be misleading, as it is evocative of etiologies such as fungal or mycobacterial infection. However, some formulations that contain oil or crystalline material may induce a foreign-body response, and in such cases, the pathologist may opt to use the modifier “granulomatous.” Severity grading should take into consideration the amount of tissue affected, the intensity of the inflammatory infiltrate, and the degree of associated tissue damage. Thus, if multiple sections of the injection site were evaluated microscopically, the severity modifier would be based on the average severity across all 3 sections. In rats, the severity of findings can be greater at the injection site in females than in males, attributable to their smaller muscle size. The criteria for the severity modifiers are typically described within the pathology text.
With IM administration, it is common to see injection material outside the region of administration, whether by extension along fascial planes, movement out through the injection tract, or from inaccurate administration (Figures 7, 12, and 13). Particularly with rats and mice, inadvertent administration of material into an incorrect location is occasionally encountered, including unintentional administration of the vaccine into the wrong muscle bed (eg, administration into the gastrocnemius rather than the caudal thigh muscles such as the biceps femoris muscle). Because of this, regional structures may be at risk for needle-related damage or vaccine-associated inflammation. The extent of this effect will vary depending on the site of administration, the size of the test species, and the experience of the technicians administering the material. In the hindlimb, inflammation around the sciatic nerve due to local extension of the vaccine is sometimes observed (regardless of the species; Figure 7). In smaller species, such as the rat and mouse, periosteal or periarticular extracapsular inflammatory infiltrates may occasionally be observed around the knee joint in the hindlimb (Figure 13), and rarely, inflammatory infiltrates, with or without injection material, may be seen within the joint capsule. In the pathology report, these findings may be described as an extension of the injection site, to differentiate it from a systemic (immune-mediated) effect. When designing a vaccine study in rodents, consider collection of joint(s) distant from the injection site for potential microscopic evaluation in case there is any ambiguity as to the cause of any joint findings encountered near the local injection site.
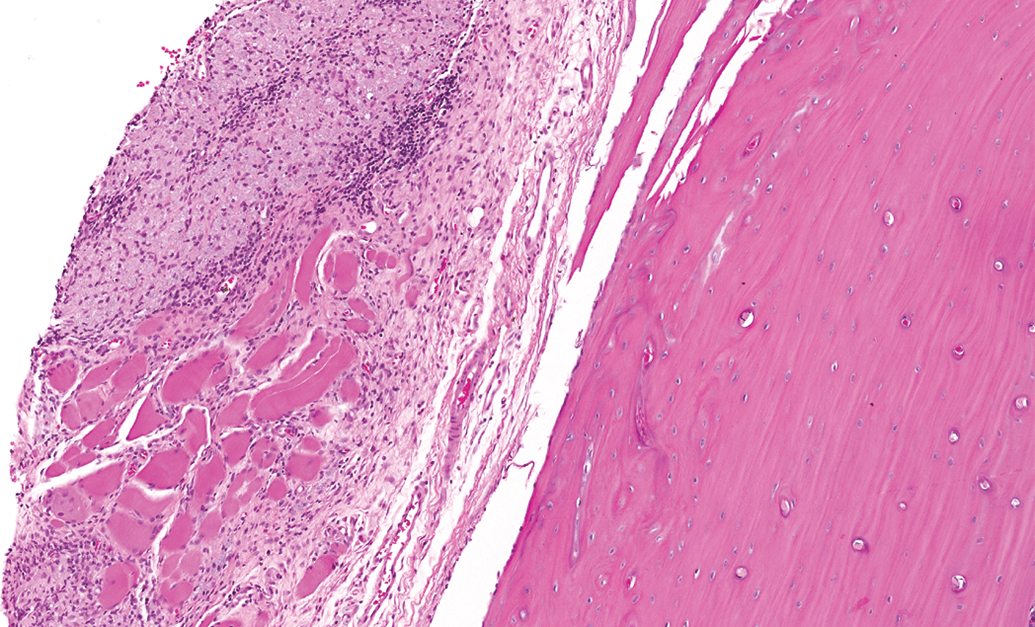
Photomicrograph of connective tissue adjacent to the femur stained with hematoxylin and eosin from a Wistar Han rat administered an aluminum-containing adjuvant intramuscular. Note the accumulation of macrophages containing lightly basophilic granular material (adjuvant).
Draining Lymph Nodes and Spleen
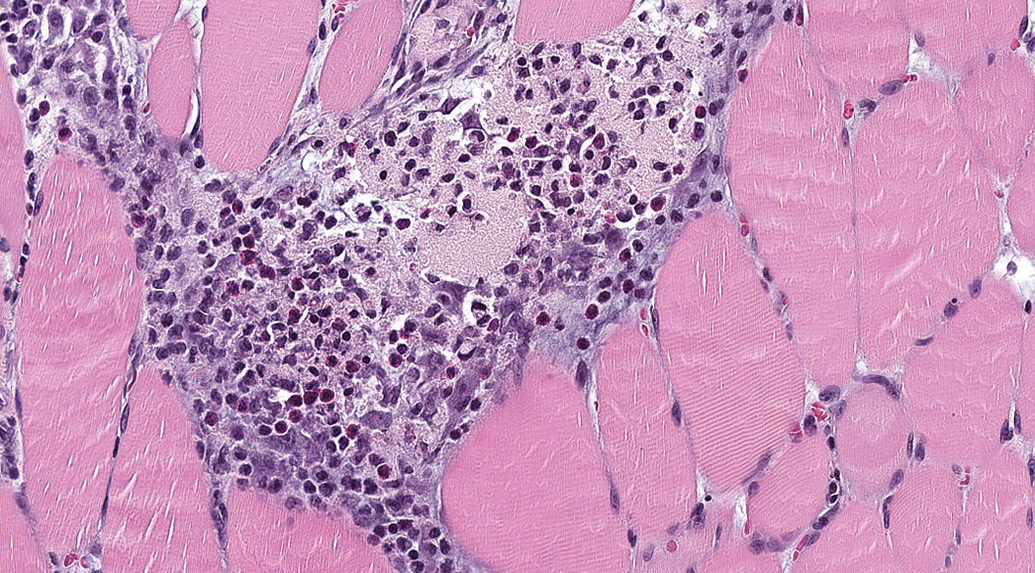
Microscopic evaluation of the draining lymph nodes may be challenging simply because of inconsistencies in orientation at tissue embedding and sectioning, particularly for rodents. This variation should be taken into consideration when interpreting the findings. The most common finding is increased cellularity and/or number of the germinal centers in association with antigens with or without adjuvants, although adjuvants may increase the incidence/severity of this finding. This finding may also be present in the spleen, although this is not as common as in the draining lymph nodes and may vary with the formulation. In animals administered aluminum-containing formulations, the draining lymph nodes will have aggregates of macrophages containing basophilic to gray granular material, reflecting accumulation of aluminum draining from the injection site (Figure 14). The region of the lymph node within the histological section can dramatically impact the severity of macrophage aggregates. Aggregates may be evident both within the cotex and sinusoids of the lymph nodes. Additionally, there may be significant plasmacytosis within the draining lymph node(s). While germinal center changes may be recovered 4 weeks from the last dose, the accumulation of aluminum-containing macrophages is often unchanged 4 weeks from the end of the dosing phase.
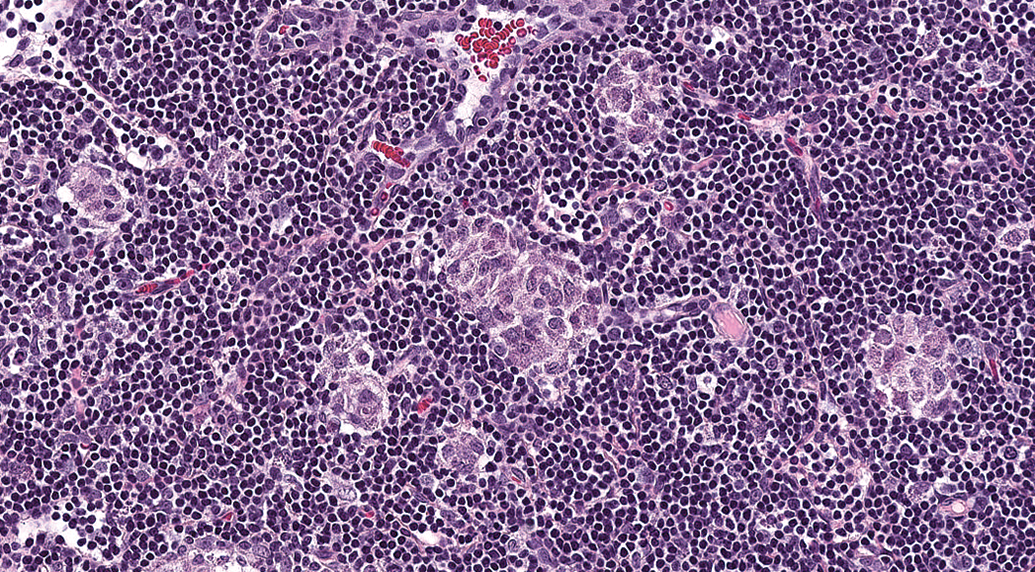
Photomicrograph of the draining lymph node stained with hematoxylin and eosin from a Wistar Han rat administered an aluminum-containing adjuvant formulation. Note the aggregates of macrophages containing granular material (adjuvant).
Other Tissues
Treatment-related microscopic findings in other tissues beyond injection site, draining lymph node, and the spleen are uncommon in prophylactic vaccines, although there may be exacerbations of species-specific background findings (eg, cardiac changes in rabbits) as a result of study activity-related stress. 27 Therapeutic vaccines may have histological findings related to coadministration of other components. 88,90,91 Many therapeutic vaccines are evaluated in NHP. Studies using NHP have fewer animals per group and NHP have notable interanimal variation in background inflammatory infiltrates (particularly in Macaques of Chinese origin), 40 which may make interpretation of treatment-related immune cell infiltrates into tissues challenging. In these cases, the HCD may be of notable value in interpreting the study findings.
Data Integration
Vaccine-associated inflammatory reactions at the injection sites or systemically may be correlated with elevations in body temperature, elevations in white blood cells, and alterations in clinical chemistry and coagulation (ie, fibrinogen) findings. Animals administered saline often have no or only limited clinical signs, clinical pathology findings, or microscopic observations, and depending on the immunogenicity of the antigen, there may also be limited findings in vaccines administered without adjuvants. When adjuvants are included in the formulation, most anatomic and clinical pathology findings will be related to the adjuvant with some contribution from the antigens. It may be of value to parse out the impact of the adjuvant component of the formulation, when possible, as this may help identify antigen-specific effects of the vaccine. Elevations in sensitive biomarkers (eg, CRP, α2-macroglobulin, α1-acid glycoprotein, fibrinogen) may correlate with findings at the injection site (including inflammation, tissue trauma, myofiber degeneration/necrosis, and/or hemorrhage). However, good concordance between clinical pathology findings and microscopic findings at the injection site and draining lymph nodes may not be observed. In these cases, the clinical pathology findings may be more sensitive than the injection site and lymphoid organs in reflecting acute-phase response or immune stimulation by the antigen(s).
The injection site inflammation may be robust, particularly when formulated with an immune activator (ie, adjuvant). When unaccompanied by clinical signs (other than transient elevations in body temperature and redness/swelling at the injection site) and with evidence of reversibility (ie, no evidence of long-term impairment, excessive scarring/fibrosis, progression, etc), injection site findings, even marked to severe, are generally considered nonadverse. Microscopic inflammatory changes at the injection site and draining lymph nodes/spleen are typically ascribed as consistent with those observed with administration of other vaccines or aluminum-containing formulations. Occasionally, however, a vaccine or an adjuvant development candidate can be associated with atypical findings, which may be considered adverse (ie, any effect that impairs the animals’ physical or physiological functions 92 ). Ulceration of the skin overlying the IM injection site, severe edema or abscessation, extensive necrosis, progression rather than reversibility, and so on may require additional efforts and discussions to determine adversity. Therapeutic vaccines may have similar findings at the injection sites and in lymphoid tissues, although target- or modality-related findings may also be present in other tissues. Ultimately, the decision to proceed into the clinic with any vaccine, prophylactic, or therapeutic demonstrating more severe or unusual local inflammatory changes will have to include a risk–benefit analysis. Since a precise threshold for local tolerability may be difficult to define, it is often helpful to place nonclinical study findings in context by comparison to other marketed compounds.
Conclusions
Regulatory toxicology studies supporting human vaccine clinical trials have many similarities to studies supporting other biopharmaceutical products. With respect to pathology, toxicology studies for vaccines share the same fundamental end points as routine toxicology studies (ie, macroscopic and microscopic evaluation, organ weights, and clinical pathology parameters). Clinical pathology typically includes additional biomarkers to measure the acute-phase response and the selection of the APP(s) must be appropriate to the species being evaluated. Key histological end points in vaccine studies are the findings in the injection sites and draining lymph nodes. Therefore, accurate and consistent vaccine administration is essential for optimized injection site collection at necropsy and trimming postnecropsy. The information presented here is intended to help pathologists and other team members involved in vaccine development programs. This “Points to Consider” paper reviewed some basic vaccine regulatory and study design considerations, addressed important technical concerns for vaccine studies, and discussed the microscopic and clinical pathology findings that are commonly identified in vaccine studies.
Footnotes
Acknowledgments
The authors thank Amber Doan and Beth Mahler for their artistic contributions to this article (Figure 3), and Xavier Palazzi for contributing figure 1.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
