Abstract

It is with great interest that I have read the publication on “spontaneous cholangiofibrosis” by Chen et al in a recent issue of Toxicologic Pathology. 1 I certainly agree that this finding is extremely rare and worth publishing. Liver lesions similar to those shown by Chen and colleagues 1 in figures 1 and 2 were induced in rats by various chemical carcinogens 2 but were only occasionally observed in untreated rodents as mentioned by the authors. I would like to draw your attention, however, to previous controversial debates on the nature of “cholangiofibrosis” (CF) in rodents and propose that the lesion found by Chen et al 1 should be classified as “cholangiofibroma” rather than “cholangiofibrosis.” While cholangiofibroma undergoes an expansive growth with compression of the adjacent liver parenchyma and protrusion of the liver surface in case of superficial localisation 2,3 like the spontaneous lesion demonstrated by Chen and colleagues, 1 CF is characterized by retraction of the surrounding parenchyma and depression of the liver surface if located superficially. 2,4
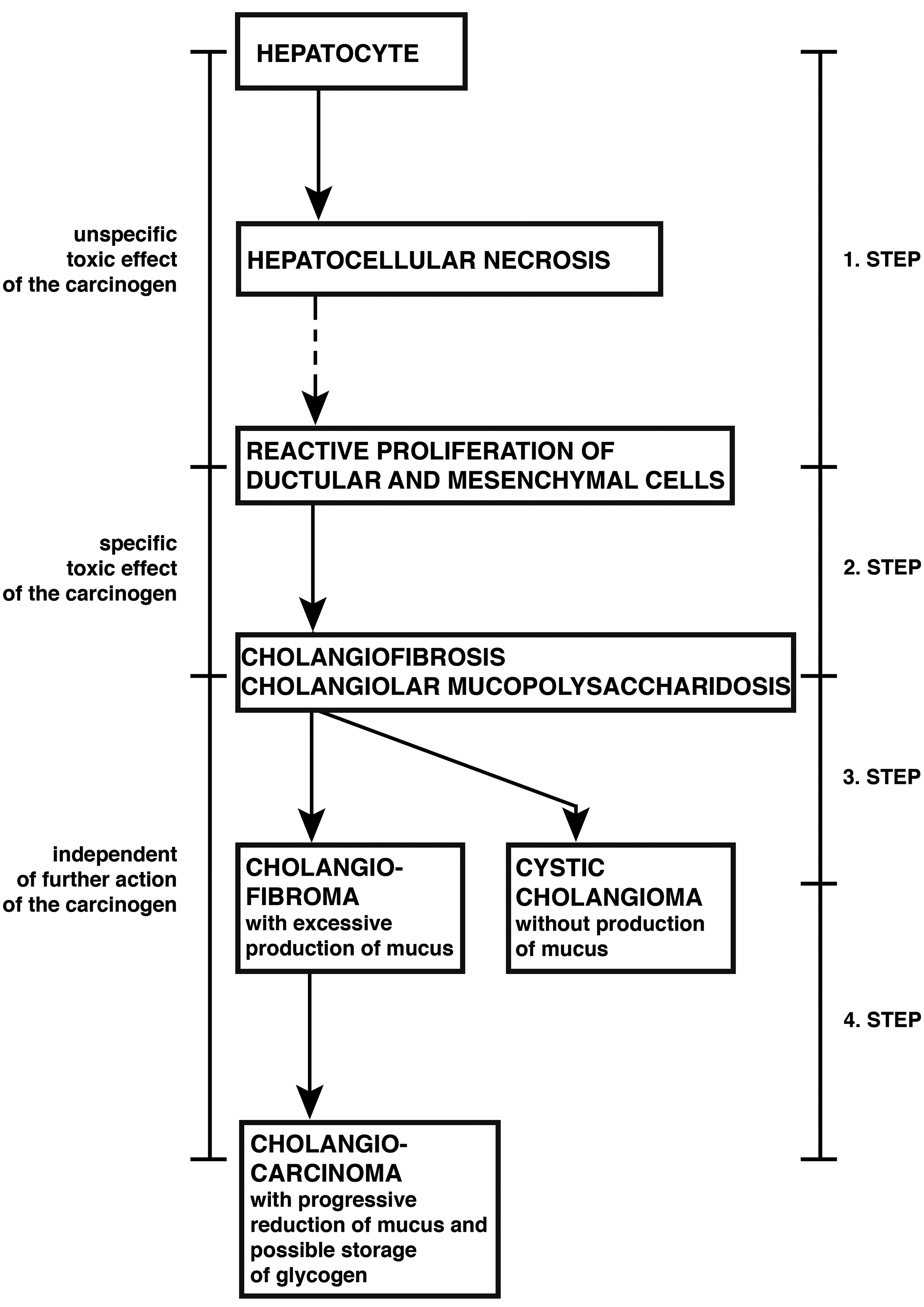
Diagram of the histogenesis and cytogenesis of cholangiocellular tumors induced in the rat by N-nitrosomorpholine. 3
The term “cholangiofibrosis” was coined by Opie 5 who considered the lesion a non-neoplastic precursor of cholangiocarcinoma (“malignant cholangioma”), deriving from a proliferation of ductular cells and fibroblasts in rats after administration of 4-dimethyl-aminoazobenzene (“butter yellow”) in the diet. In addition, Opie described that CF usually shows a strong production of mucus and frequently also an infiltration by inflammatory cells. 5 Some authors called the lesion “adenofibrosis,” 4 implicating that it originates from hepatocytes, but most authors agree that it results from a proliferation of cholangiolar cells. 2,6 –8 This cell type corresponds to the “oval cell” described by Farber 9 as a phenotype appearing early after exposure of rats to several chemical hepatocarcinogens, but it may also occur after severe parenchymal damage produced by various other noxae. 10 The proliferation of oval cells is usually associated with proliferation of mesenchymal and infiltration by inflammatory cells. Popper and colleagues 10 called this response to liver injury “ductular cell reaction.” The significance of oval cells for cellular differentiation and carcinogenesis in the liver has long been under discussion. While their postulated differentiation into hepatocytes and hepatocellular neoplasms has remained debatable, 11 –13 there is general agreement that they are precursors of cholangiocellular neoplasms in rodents. 2,7,14 With respect to cholangiocellular lesions induced in rats by N-nitrosomorpholine (NNM) our own studies indicated that the early proliferation of the cholangiolar (oval) cells is an unspecific reaction to dose-dependent severe toxic liver damage, particularly parenchymal necrosis rather than a neoplastic process. 15 A similar view has been taken by others who applied various chemical carcinogens to rats. 16 The pronounced production of mucus by goblet cells intercalated in the cuboidal or columnar epithelium of the abnormal cholangioles in CF and related cholangiocellular lesions has been interpreted as a specific carcinogen-induced metabolic aberration called “cholangiolar mucopylsaccharidosis” 11,15,17 in analogy to the hepatocellular glycogenosis preceding the majority of hepatocellular neoplasms. 18,19 Several authors favored the designation of the cellular changes characterizing the abnormal cholangioles as “intestinal metaplasia,” 7,20 –23 although the absence of staining for laminin β2 and β3 in the basement membrane associated with this lesion indicated that its differentiation is not equivalent to that seen in the gut. 7 In rats exposed to furan, a pathogenetic link between mucus CF and cholangiocellular neoplasms producing mucus has also been demonstrated at the molecular level by an overexpression of cell receptors Neu and Met in both types of lesion. 23 In the same animal model, persisting epigenetic as well as genetic changes were identified as potential causes of the carcinogenic effect. 24
From our experience in rats exposed to NNM at different dose levels for limited time periods (stop model), we concluded that mucus CF is actually a preneoplastic lesion which—once established—even in the absence of further carcinogen treatment may give rise to various types of cholangiocellular neoplasm (Figure 1), namely, cystic cholangiomas 15 and mucus cholangiofibromas which may progress to cholangiocarcinomas after long lag periods. 2,3 Both the transformation of CF into cystic cholangiomas and the progression from mucus cholangiofibromas to cholangiocarcinomas is regularly accompanied by characteristic changes in the cellular phenotype, particularly also a loss of the mucus-producing goblet cells. In subpopulations of the cholangiocellular carcinomas, the production of mucus may be replaced by a transient storage of glycogen, which in contrast to the typical glycogen rosettes in hepatocytes, is monoparticular. 25 It has remained obscure what factors determine the development of either benign cystic cholangiomas or cholangiofibromas and cholangiocarcinomas from CF, but it may be assumed that the dose-dependent degree of cellular injury by the carcinogen is different.
Studying cholangiocellular lesions in rats exposed to furan, Maronpot et al 26 questioned the preneoplastic nature of CF and considered it to be essentially malignant or premalignant because they did not observe qualitative differences between furan-induced mild CF and neoplasms diagnosed as cholangiocarcinomas, some of which were transplantable in syngeneic rats and metastasized after several serial passages. We have previously argued against this view for several reasons: (1) even extended CF may only replace lost liver parenchyma without any indication of expansion beyond the confines of the liver lobe; (2) the ductular component of CF may sometimes undergo involution, leaving nothing but a scar; (3) progression of CF frequently leads to cystic cholangiomas which represent a benign end-stage lesion, while only a second neoplastic derivative of CF the cholangiofibroma is potentially malignant and may eventually give rise to cholangiocarcinoma. 2
Differential diagnosis of CF and cystic cholangiomas is not a major problem, since the cysts of benign cholangiomas are usually free of mucus and are lined by a single layer of flat or cuboidal cells without any goblet cells. 12,15,27 Sometimes, we observed the development of morphologically similar cystic cholangiomas without discovering any preceding oval cell proliferation and CF. Evert et al 28 have demonstrated that this pathogenesis is the rule rather than the exception after transplantation of pancreatic islands into the livers of diabetic rats. The cholangiomas developing under these conditions emerged from cholangiocytes adjacent to the transplanted islands and turned out to be benign end-stage lesions like those originating from mucus CF in other models. The progression from peri-insular ductules to cystic cholangiomas after islet cell transplantation in diabetic rats was accompanied by a translocation of the insulin receptor from the cell membrane into the cytoplasm and an increase in expression of insulin-related signaling proteins (insulin-receptor-substrate-1, Raf-1, Mek-1) in the cholangiolar cells involved. 28
Distinguishing cholangiofibroma from CF is difficult, since gradual transitions between these lesions exist. In contrast to CF, cholangiofibroma is not only characterized by expansion rather than contraction but usually also shows a more pronounced production of mucus and particularly collagen fibers in central parts of these tumors. 2,3 All of these features apply to the lesion described by Chen and colleagues 1 as documented in figures 1 and 2 of their publication. Observing the biological behavior of cholangiofibromas in different stop protocols, we found a merely expansive growth for weeks and months after withdrawal of the carcinogenic agents, substantiating the classification of these neoplasms as benign. 2,3,11,12 It takes long lag periods until mucus cholangiofibromas may become malignant and progress to invasive cholangiocarcinomas. Similar results were obtained in rats exposed to other chemical carcinogens such as methapyrilene hydrochloride 29 and the mycotoxin phomopsin. 30 Several authors did not clearly separate cholangiofibromas from cholangiocarcinomas, since the epithelium of the glandular component of mucus cholangiofibromas, in addition to the goblet cells, frequently shows considerable cellular atypia with strong cytoplasmic basophilia, many mitotic figures, nuclear and nucleolar abnormalities, and variable numbers of necrotic cells. 7,8,22,23,26,31 In addition, large numbers of fibroblasts within peripheral tumor portions may give rise to the false impression of invasive growth. However, only unequivocal invasion and metastases prove malignancy. In more than 100 cholangiofibromas developing in situ after exposure of rats to various oral carcinogenic regimens (NNM, thioacetamide, 2-acetylaminofluorene, choline-deficiency/DL-ethionine-supplementation), we detected neither invasion nor metastases. This justifies the classification of the mucus cholangiofibroma—including the spontaneous lesion observed by Chen et al 1 —as a benign albeit potentially malignant liver tumor.
