Abstract
Although necrosis and apoptosis are uncommon, autophagy of sensory neurons (ASN) in trigeminal and dorsal root ganglia is a very common, spontaneous finding in cynomolgus monkeys (Macaca fascicularis). Data from one author’s (Butt) laboratory showed 12 of 22 studies (year range 2017 to 2019) that included the evaluation of sensory ganglia from cynomolgus monkeys had at least one control animal with ASN. Autophagy of sensory neurons is characterized by a distinct cell membrane, cytoplasm filled with autolysosomes, disintegrated nuclear membrane, and/or globules of degraded chromatin. Since these changes are consistent with autophagy and indicate an irreversible state, a diagnosis of autophagy is preferred instead of necrosis or degeneration. Sensory ganglia are not commonly evaluated in nonclinical toxicology investigations so many pathologists may be unaware of this common change. Especially due to the typically small group size of monkey studies, the observation of this change in sensory ganglia may lead to a faulty interpretation that this change is due to the test article. This article describes the light microscopic and ultrastructural characteristics of neuronal autophagy in trigeminal and dorsal root ganglia and provides historical control data of the incidence of this change in cynomolgus monkeys.
In nonclinical studies, toxicologic pathologists must identify and characterize test article-related morphologic changes and determine the dose levels associated with such changes, a task that may be greatly complicated by the presence of spontaneous (background) and artifactual changes. Interpreting the relationship of a spontaneous change to a particular test article may be especially complicated by one or more of the following: relatively small group sizes, inclusion of an unfamiliar tissue and/or morphologic change, and/or a lack of published information and/or institutional experience with a particular tissue/morphologic change. Studies in monkeys that include evaluation of sensory ganglia may include all of these complicating factors, plus typical study design (1 control group, 3 test article dose levels) means there is a 75% change of a single incidence of a spontaneous change occurring in a test article-treated animal. Sensory ganglia are seldom listed as a required tissue for morphologic evaluation in regulatory guidelines 1 and thus are often absent from the list of tissues to be examined microscopically. This may be an important oversight, since the vascular barrier in sensory ganglia is generally more fenestrated/permeable than comparable barriers in the brain and spinal cord, 2 meaning neurons in sensory ganglia may be more exposed to various test articles than neurons in the brain or spinal cord. Recent recommendations by a committee of the Society of Toxicologic Pathology 3 include the microscopic evaluation of selected sensory ganglia in investigations where toxicity/effects in the peripheral nervous system are suspected based on clinical signs. Given the potential for exposure of sensory neurons to nearly any administered small molecule, biologic, or biosimilar, evaluation of sensory (typically one or more dorsal root) ganglia may be warranted in most studies.
A spontaneous change in cynomolgus monkeys likely to cause confusion is autophagy of sensory neurons (ASN) in dorsal root ganglia (DRG), trigeminal ganglia, and likely other sensory ganglia. To the best of the authors’ knowledge, ASN was first reported in a textbook on toxicologic pathology, 4 with later clarification 5 that autophagy was the suspected mechanism of the morphologic appearance at microscopic evaluation.
This communication describes the light microscopic and ultrastructural features of trigeminal and DRG ASN in control cynomolgus monkeys and provides incidence data for this change from studies evaluated at Tox Path Specialists, LLC.
Table 1 describes the morphologic characteristics of ASN as observed in sections of sensory ganglia prepared using common fixation (aldehydes ± osmium tetroxide), embedding (paraffin or plastic), and staining methods for evaluation using light or electron microscopy. Figures 1 and 2 provide examples of these morphologic characteristics.
Morphologic Characteristics of Autophagy of Sensory Neurons.

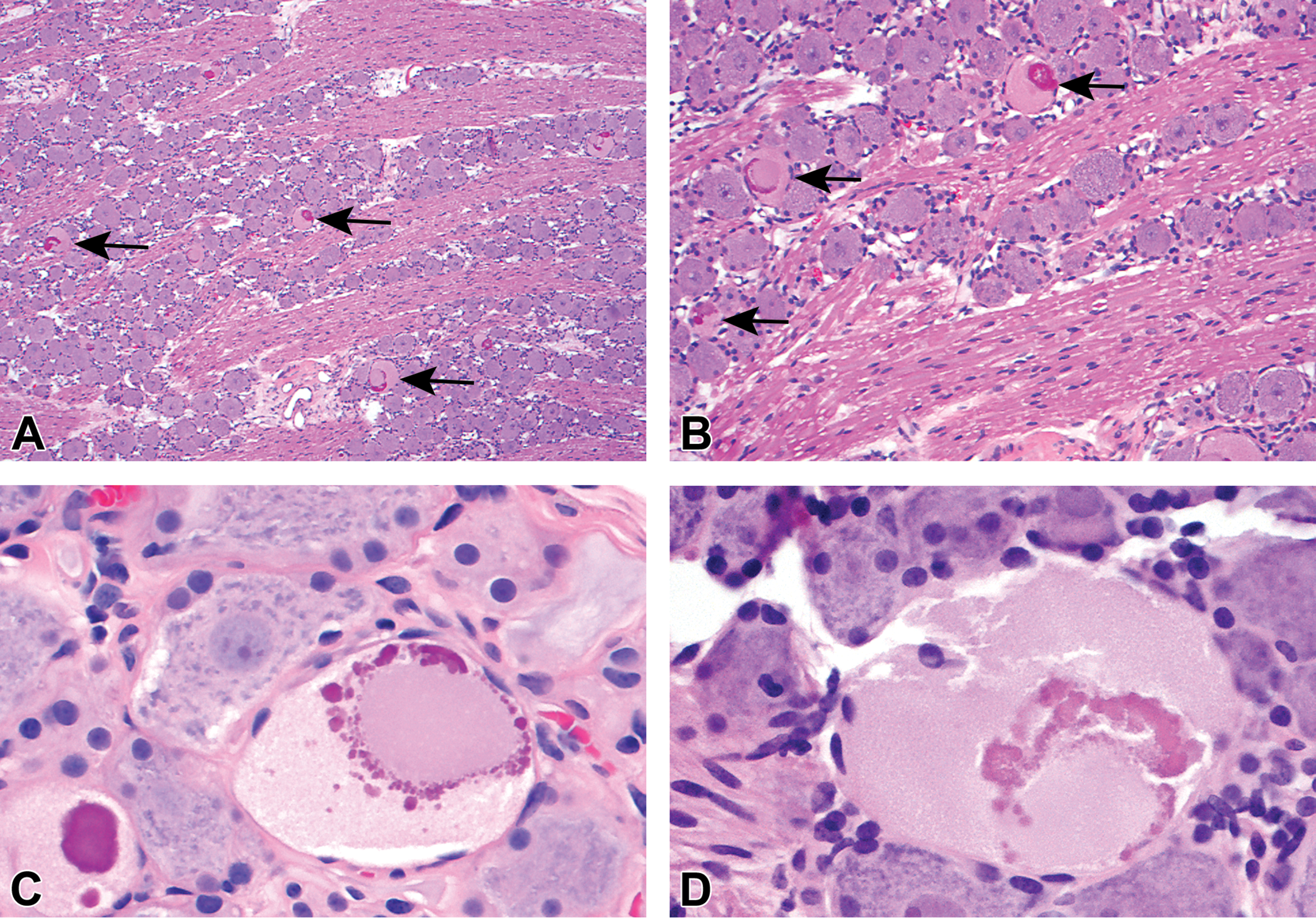
A, Intravascular perfusion fixation with 10% neutral buffered formalin, paraffin embedding, hematoxylin and eosin (H&E) stain, 5 µ section. Lumbar dorsal root ganglion from a control cynomolgus monkey. Numerous autophagic neurons (some indicated by arrows) are scattered throughout the ganglion. B, Intravascular perfusion fixation with 10% neutral buffered formalin, paraffin embedding, H&E stain, 5 µ section. Trigeminal ganglion from a control cynomolgus monkey. Multiple autophagic neurons (arrows) are distributed among morphologically normal neurons. Inflammation, architectural disruption or other reactive or degenerative changes are not evident. In the affected neurons, degraded chromatin (dark eosinophilic material) fills or largely surrounds the prior nucleus. C, Intravascular perfusion fixation with 10% neutral-buffered formalin, paraffin embedding, H&E stain, 5 µ section. Cervical dorsal root ganglion from a control cynomolgus monkey. Two neurons are affected. Both contain globules of darkly eosinophilic, degraded chromatin and have cytoplasm compartments filled with pale, eosinophilic granules but no discernible organelles. In the larger neuron on the right, globules of degraded chromatin surround a structure suspected to be the prior nucleus. D, Intravascular perfusion fixation with 10% neutral buffered formalin, Paraffin embedding, H&E stain, 5 µ section. Trigeminal ganglion from a control cynomolgus monkey. The affected neuron is larger than adjacent neurons. Nissl substance, clearly visible in surrounding neurons, is not discernible in the affected neuron. The morphologic features are similar to those described in C.
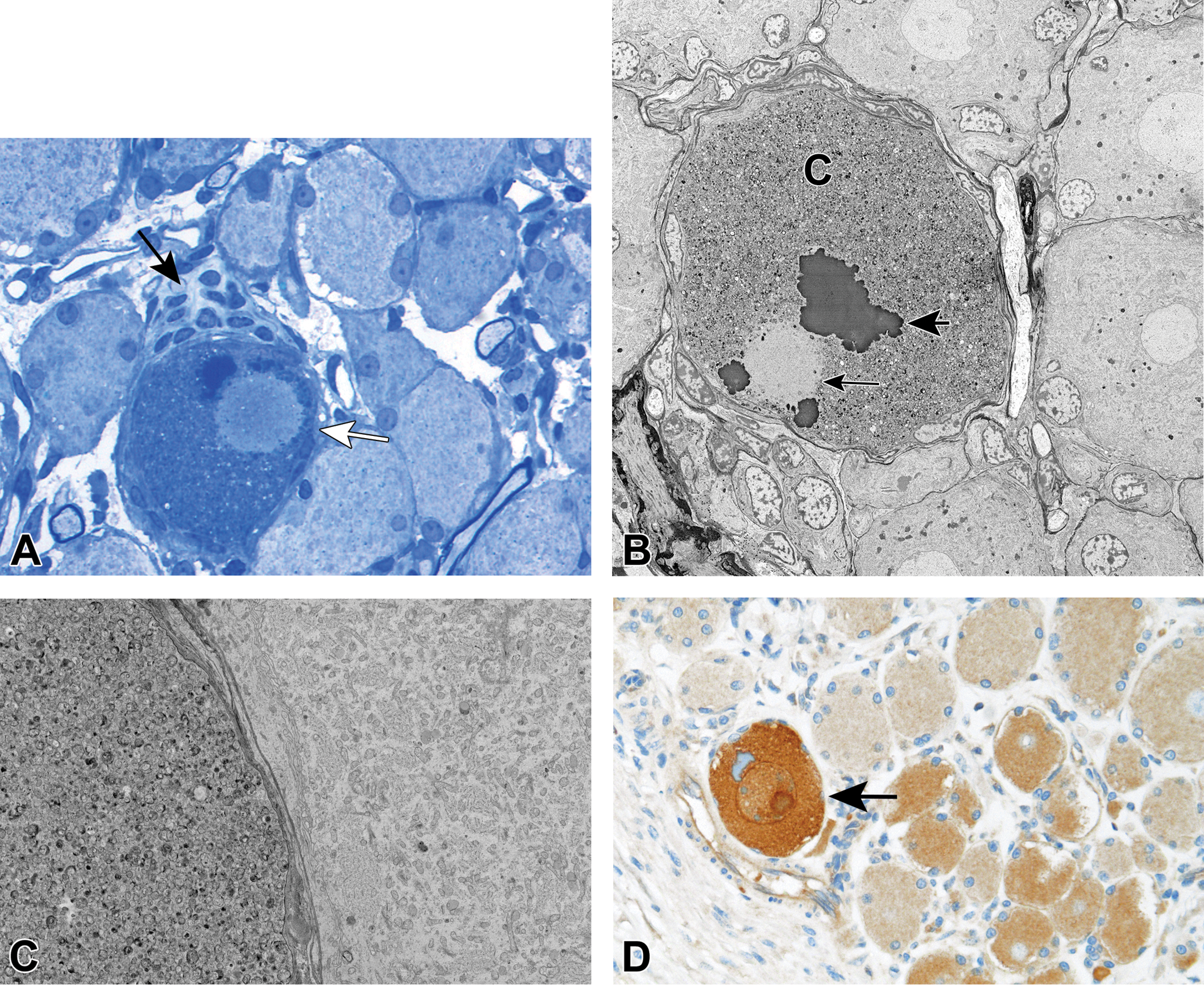
A, Intravascular perfusion fixation with 10% neutral-buffered formalin followed by immersion for 2 hours in 2% osmium tetroxide, Resin embedding, Toluidine blue stain, 1 µ section Lumbar dorsal root ganglion from a control cynomolgus monkey. A single affected neuron (white arrow) is adjacent to multiple unaffected neurons. The cytoplasm is filled with numerous small granules. Nissl substance, clearly visible in surrounding neurons, is not discernible in the affected neuron. The satellite glial cells surrounding the affected cell are flattened and appear less numerous except at one portion of the cell border (black arrow). The prior nucleus is distinct but lacks a membrane. Condensed chromatin is adjacent to the disintegrating prior nucleus. B, Immersion fixation in 2% methanol free formaldehyde/2.5% glutaraldehyde for 24 hours, followed by immersion for 1 hour in 1% osmium tetroxide, then immersion in 0.5% uranyl acetate overnight. Resin embedding, 0.2% lead citrate stain (1 minute), <100 nm section lumbar dorsal root ganglion from a control cynomolgus monkey. An autophagic neuron is surrounded by unaffected neurons. In the affected cell, the nuclear membrane has disintegrated (small arrow), degraded chromatin is present (large arrow), and the cytoplasmic compartment (C) is filled with autolysosomes. The satellite glial cells surrounding the affected cell are absent or flattened. C, Immersion fixation in 2% methanol free formaldehyde/2.5% glutaraldehyde for 24 hours, followed by immersion for 1 hour in 1% osmium tetroxide, then immersion in 0.5% uranyl acetate overnight. Resin embedding, 0.2% lead citrate stain (1 minute), <100 nm section lumbar dorsal root ganglion from a control cynomolgus monkey. An affected neuron (left) and a normal neuron (right). Organelles (mitochondria, profiles of endoplasmic reticulum) are clearly visible in the normal cell. In the affected cell, the cytoplasm compartment is filled with round, membrane bond, variably osmiophilic, autolysosomes. Note the outer cell membranes of both neurons are distinct and intact. D, Immersion fixation in 10% neutral-buffered formalin 24 hours, Paraffin embedding, 5 µ section; immunohistochemistry staining with mouse anti-ATG16L1 (MBL International Corp., clone 1F12) followed by unconjugated rabbit anti-mouse (Abcam, AB133469), and detected by Ventana Medical Systems, Inc. OmniMap anti-Rb HRP and ChromoMap DAB kit Lumbar dorsal root ganglion from a control cynomolgus monkey. The section is labeled with a primary antibody directed against ATG16L1 IHC, a marker for autophagy. The presence of ATG16L1 is indicated by the intense chromogen reaction (brown color) of a neuron to the left (arrow); this neuron also has features of ASN. The notably less pronounced chromagen reaction in the other neurons was judged to be nonspecific/background staining of no diagnostic relevance.
The lack of any association with myelin/Schwann cell processes, the appearance (in many affected cells) of what appears to be a prior nucleus, and the presence of degraded/clumped chromatin suggest these structures were neuronal cell bodies and not the proximal axon. Multiple morphologic characteristics (cytoplasm compartment filled with autolysosomes; disintegrated nuclear membrane; globules of degraded chromatin; general lack of a response to the autophagic cells) indicate the changes are due to autophagy. Preliminary immunohistochemical staining by one of the authors (Fuji) indicates neurons with features of ASN may stain for the presence of ATG16L1 (autophagy-related protein 16-1). ATG16L1 is part of a protein complex necessary for autophagy). 7 Figure 2D (section from a lumbar dorsal root ganglion) shows an autophagic sensory neuron staining positively for the presence of ATG16L1.
Because the morphologic features of ASN indicate cell death via a process of autophagy, a diagnosis of “Autophagy” is warranted and accurate, much like a diagnosis of “Necrosis” is warranted and accurate when cells display the appropriate characteristics. “Degeneration” has been suggested as a term to diagnose/record the presence of ASN, but with the additional knowledge presented in this communication regarding the ultrastructural changes, “Autophagy” is the preferred terminology. The term “Degeneration” implies a reversible change and/or a change in a cell or tissue to a less functional form, but neither of these conditions exist with ASN. These neurons undergoing autophagy are dead. In situations where the user’s glossary may prevent use of the term “Autophagy” (and it is acknowledged that the diagnostic glossary for the Standard for Exchange of Nonclinical Data does not currently contain the term “Autophagy,”), then a sufficient description in the report correlating the chosen term to the actual nature of ASN is suggested.
At Tox Path Specialists, LLC, ASN was observed in one or more control cynomolgus monkeys from 12 out of 22 studies (evaluated between 2017 and 2019) in which trigeminal and/or DRG were reviewed. In those 12 studies (Table 2), the overall incidence of ASN (trigeminal and DRG combined) in control animals was approximately 32% (80 out of 247 ganglia), but at any anatomic specific level of sensory ganglia, the incidence was often greater than 50%. It is the experience of the lead author that if ASN is present in an animal, multiple ganglia are often affected. To the knowledge of the authors, ASN has no known clinical implications.
Incidences of Autophagy of Sensory Neurons in Paraffin Embedded, H&E Stained Sections From Selected Ganglia From Cynomolgus Monkeys (Controls) From Various Studies (2017 to 2019) Evaluated at Tox Path Specialists, LLC.
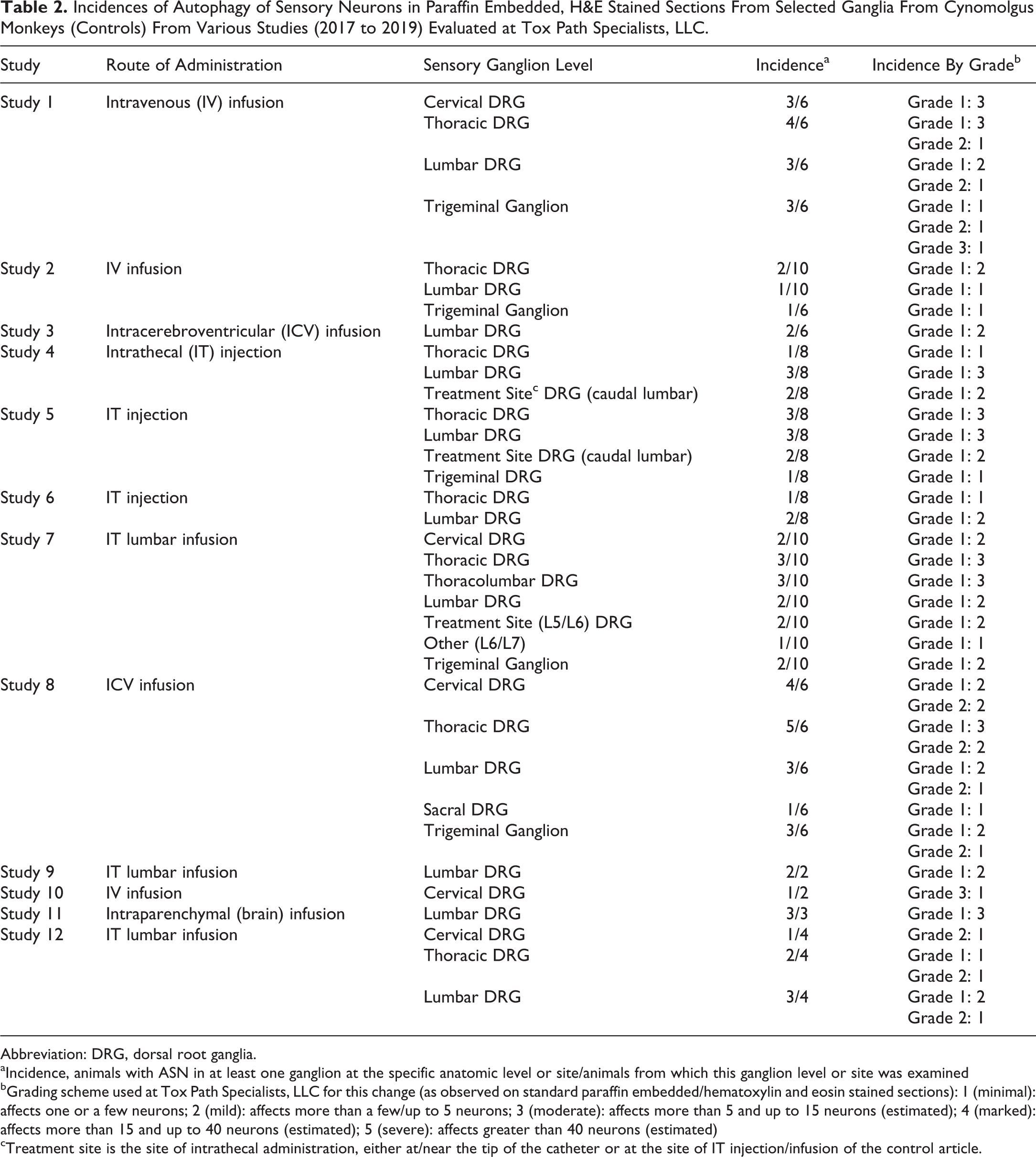
Abbreviation: DRG, dorsal root ganglia.
aIncidence, animals with ASN in at least one ganglion at the specific anatomic level or site/animals from which this ganglion level or site was examined
bGrading scheme used at Tox Path Specialists, LLC for this change (as observed on standard paraffin embedded/hematoxylin and eosin stained sections): 1 (minimal): affects one or a few neurons; 2 (mild): affects more than a few/up to 5 neurons; 3 (moderate): affects more than 5 and up to 15 neurons (estimated); 4 (marked): affects more than 15 and up to 40 neurons (estimated); 5 (severe): affects greater than 40 neurons (estimated)
cTreatment site is the site of intrathecal administration, either at/near the tip of the catheter or at the site of IT injection/infusion of the control article.
Recognition of the morphologic characteristics of ASN in cynomolgus monkeys combined with knowledge concerning the possible incidence of this change should assist the toxicologic pathologist in accurately recording and interpreting the significance of ASN in any particular study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
