Abstract
Histology of medical devices poses a variety of unique challenges. Comprehensive histologic assessment of medical devices often requires spatial context and high-quality retention of the device–tissue interface. However, the composition of many medical devices is often not amenable to traditional paraffin embedding and thus alternative specialized methodologies such as hard resin embedding must be used. Hard resin embedding requires specialized laboratory technical expertise and equipment, and the fixation techniques and resin composition used markedly impact the feasibility of immunohistochemistry. For the continuity of spatial context during histologic evaluation, additional imaging methods such as macrophotography, radiography, micro-Computerized Tomography (microCT), or magnetic resonance imaging (MRI) can be used to guide sectioning and to complement histologic findings. Although standardized approaches are scarce for medical devices, important considerations specific to medical device histology are discussed, including general specimen preparation, special considerations for devices by organ system, and the challenges of immunohistochemistry. Histologic preparation of medical devices must be thoughtful, thorough, and tailored to achieve optimal histologic outcomes for complex, valuable, and often limited implant specimens.
Keywords
In interventional and implant testing studies, the optimal histology protocol is contingent upon many variables and cannot be improvised. Medical devices are designed to overcome failing anatomical structures or to rectify impaired physiology. The variety of medical device designs created require a customized approach for proper evaluation. The pathologist must be involved early in protocol design and must have a good understanding of the surgical procedure, test article, and animal model and of study objectives in order to set the stage for a successful and meaningful ex vivo assessment (Nikula and Funk 2016). “First, do no harm” is a precept that histopathologist involved with medical device assessment must always consider and one that requires knowledge, experience, and thoughtfulness to honor study after study. This article aims to inventory and describe successful strategies and provides analytical tools and practical solutions which if followed can shepherd a preclinical study across the minefield of medical device histology and pathology. The chief operating principle of histology is to optimize the integrity of the specimens and when applicable of the implant–host tissue interface. Histopathologists are trained to most adequately handle and evaluate normal and naturally diseased bodies and organs and as a rule receive no training in handling surgical sites or organs implanted with biomedical implants. These skills are generally learned “on the job” or through the rare internship opportunities available throughout the industry or in specialized academic institutions. The pharmaceutical industry has long-established standards and time-tested practices that provide a clear framework for junior pathologists and histologists involved in drug development. In the medical device field, a few FDA guidance documents have been published but are still fragmentary, covering specific types of devices (i.e., stent or drug-eluting stents, valves, vascular sealers; FDA 1996; ISO 10993-6:2016 n.d.; Holmes et al. 1998; Schwartz et al. 2008; FDA 2008). These guidance documents primarily focus on the in vivo testing to include study designs, animal models, and clinical end points such as imaging modalities. The methodologies in these documents are generally sparse guides for addressing the practical minutia of tissue handling, collections, and histological preparations, which are critical to the success of post mortem evaluation. This article will provide practical details and methods for histologic preparations that medical testing standards and guidance documents do not address.
Sample Procurement and Macroscopic Trimming
The microscopic evaluation of an implantation or surgical site requires the utmost preservation of the implant–tissue interface and surgical planes. Any degree of fresh tissue dissection to directly expose the implant surface or surgical site invariably produces handling artifacts that are detrimental to microscopic evaluation and can sometimes completely preclude the evaluation of the tissue interface. Macroscopic evaluation can sometimes be successfully accomplished with minimal tissue disruption after the entire tissue has been thoroughly fixed. It is essential to establish a dialogue with study directors, surgeons, and interventionalist to educate all stakeholders to the risks of excessive handling and temper the temptation for aggressive dissections. This open channel of communication between the study team should be maintained along the evaluation of the samples since it allows to (a) discern between handling artifacts and test article–dependent findings and (b) refine correlation between clinical observation with pathological findings.
Precise and cautious tissue handling should be imperative in cases such as: Acute (<2 week) follow up in surgical sites where tissue cohesion is reliant on fibrin rather than fibrous connective tissue. Tissue that has been purposely denatured through heat or chemical treatment (decellularized scaffold) can also be particularly delicate in the early stages. Acute evaluation of energy-based therapies (i.e., cardiac ablation or pulmonary vein isolation sites, renal or other denervation therapies; Leshem et al. 2017).
The optimal way to harvest and preserve such sites is to use perfusion fixation and forego direct macroscopic observations and process the entire territory in paraffin followed by step sectioning into the area of interest. Direct observations of acutely treated tissues often result in tearing of fibrin, denatured collagen and muscle resulting in artifacts in tissue sections that can confound interpretation. Tissue trimming is a critical step and the choice of trimming planes imparts an experimental bias that must be carefully interpreted. Generally, hollow structures or tissues and devices that have an axis of symmetry are best trimmed in a plane that is perpendicular to such axis. This approach controls for the influence of angulation and tangentiality on microanatomy and quantitative area measurements. If volumetric data are sought, fundamental principles of stereology (Marcos, Monteiro, and Rocha 2012; Boyce, Boyce, and Gundersen 2010) must be applied to the sampling strategy. More generally, treatment and implantation sites should be sampled in a standard, planned, and deliberate fashion in representative areas. Medical devices can be highly engineered and may contain multiple materials that result in many possibly different interfaces between the biomaterial and host tissue that must all be assessed with dedicated or combined samples as necessary. Multiple and compound sectioning planes may be necessary to thoroughly assess tissue response. Tissue sectioning may also be guided by specific objectives such as measurement of the thermal spread of a lesion in a given plane or assessment of transmurality. When standard cassette and block size (∼25 × 40 mm) is limiting, oversized cassettes, embedding molds, and slides can be used (∼50 × 70 mm) to preserve the integrity across large areas or minimize the fragmentation of sites that are even larger than oversize slides can accommodate. The embedding medium must be appropriate for the device type (i.e., paraffin or plastic). An algorithm for sample procurement and tissue trimming is presented in Table 1.
Algorithm for Sample Procurement and Macroscopic Trimming.
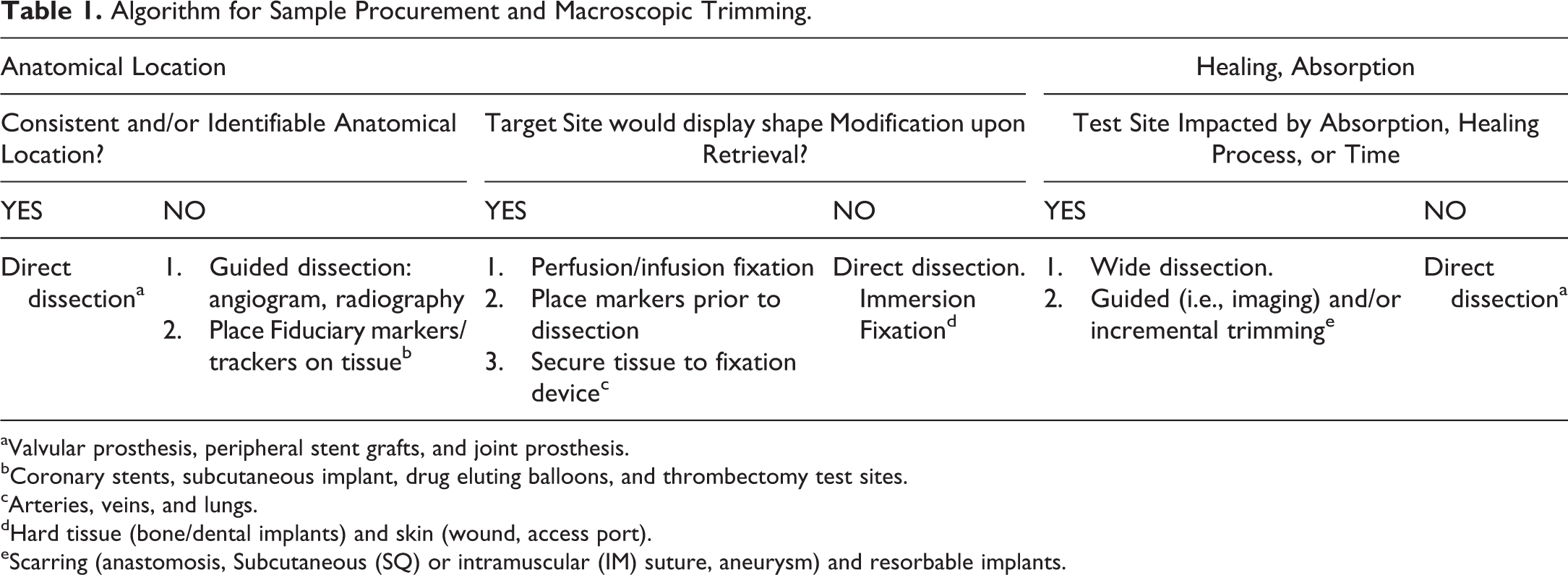
aValvular prosthesis, peripheral stent grafts, and joint prosthesis.
bCoronary stents, subcutaneous implant, drug eluting balloons, and thrombectomy test sites.
cArteries, veins, and lungs.
dHard tissue (bone/dental implants) and skin (wound, access port).
eScarring (anastomosis, Subcutaneous (SQ) or intramuscular (IM) suture, aneurysm) and resorbable implants.
Imaging techniques such as microCT or tissue MRI can be of assistance whenever the implant site has become embedded in healed tissue or the implant has been absorbed and the tissue is remodeled. These techniques can also assist in quantifying residual biomaterial and general healing characteristics (Tempel-Brami et al. 2015; Rousselle et al. 2019; Chagnon, Guy, and Jackson 2019).
Specimen Fixation and Preservation
Procedures developed for conventional safety assessment of pharmaceuticals and biologics take into account the slow rate of passive diffusion of tissue fixatives such as 10% neutral buffered formalin (NBF). It is advised that for optimal fixation by direct immersion, sample thickness should not exceed 3 to 5 mm (Ruehl-Fehlert et al. 2003). Implant sites and experimental surgical sites can rarely be trimmed fresh without compromising the integrity of the specimen or disrupt the implant interface. It is frequently advantageous or necessary to collect and fix larger explants (e.g., large excision wound), an entire organ (heart with stented vessels and bone with implant), or several organs together (joint, brain with skull, kidneys with associated supplying vasculature, and underlying psoas muscles in renal denervation protocols). Since implantable devices should ideally be tested to mimic the clinical setting and preferentially using the same surgical equipment and interventional techniques, most surgical and implant studies are conducted in large animal models. As a result, the experimental sites and/or downstream tissues are much larger than is advisable to collect and fix to obtain optimal results under standard processes. Good practices that can mitigate the limitations of fixative diffusion include perfusion or infusion fixation (brain, heart, and lungs), scoring of supporting tissues to expose inner portions (bones, brain, and skin), immersion in a large volume of fixative with agitation, and ultrasound- or microwave-assisted fixation if these modalities are compatible with the implanted device/biomaterial.
Perfusion fixation: This technique has become the standard for the heart (through aortic cannulation) and the head with brain (carotid cannulation) yielding excellent results. Premortem heparinization is necessary to prevent clotting and allow clearance of the vasculature by a isotonic physiological solution (i.e., phosphate-buffered saline) prior to perfusion with 10% NBF. When collected whole, the lungs should preferably be filled with fixative cannulating the trachea with a volume of fixative that is appropriate for the species and sufficient to expand the airways and alveoli. In large species, this can be impractical or burdensome, and immersion fixation generally yields adequate results for the objectives at hand. If infusion is performed, it is best to counterbalance the pressure applied by the column of fixative by the immersion of the lungs in a large volume of fixative while they are being infused through the airways.
Immersion fixation: The recommended volume of fixative is 15–20:1 fixative to tissue ratio and the absolute minimal recommended volume would be 10:1 fixative to tissue ratio. Practically this translates into ∼3 L of fixative or nearly 1 Gal for an average pig heart or 2 L for the brain (∼1/2 Gal).
Bone fixation: Small bones (i.e., rodents, rabbits) can be immersion fixed, provided soft tissue are sufficiently removed. Larger bones should always be pretrimmed for adequate fixation by immersion. If maintaining the integrity of the surgical or implant site precludes trimming and exposure of the marrow cavity, perfusion fixation of the relevant territory is warranted and must be considered. This principle applies to the skull with brain for species as small as rabbits if the skull cap cannot be open and brain must be evaluated histologically for collateral injury. There is a belief that large tissues such as bones (long bones and vertebrae) can be flash frozen for microCT imaging and subsequently thawed in fixative for histology. This practice is misguided because large tissues, especially bone cannot be frozen fast enough to avoid the formation of ice crystals and lysis of bone and/or marrow cells upon thawing. Bones subjected to this procedure and cycles of freezing and thawing before, during, and after microCT are marred with poor preservation of the soft tissue elements (marrow, osteoblasts/osteoclasts, and connective tissue cells) resulting in very poor histology results. This practice is strongly discouraged and should never be used. It is always best to fix tissues prior to CT or MRI imaging.
Fixation for immunohistology: Target epitopes can become excessively cross-linked and unreactive with prolonged formalin exposure. To optimize the chances of success, fixed tissue should be transferred to 70% ethanol solution as soon as fixation is achieved, which may vary from 12to 72 hr for soft tissues, based on tissue thickness and the use of perfusion techniques to 72 to 96 hr for larger nonperfused tissues such as bone (Hassan et al. 2017) or large muscles or entire heart. Seventy percent ethanol is a preservative but not a fixative and should not be used alone. In our experience, most histology grade antibodies known to work with formalin-fixed paraffin-embedded tissues can generally be compatible with resin embedding (i.e., Spurr’s resin embedding; Figure 1). Some epitopes can require frozen tissues, and appropriate contingencies should be considered for sampling and tissue preservation, as applicable (Baskin 2014).
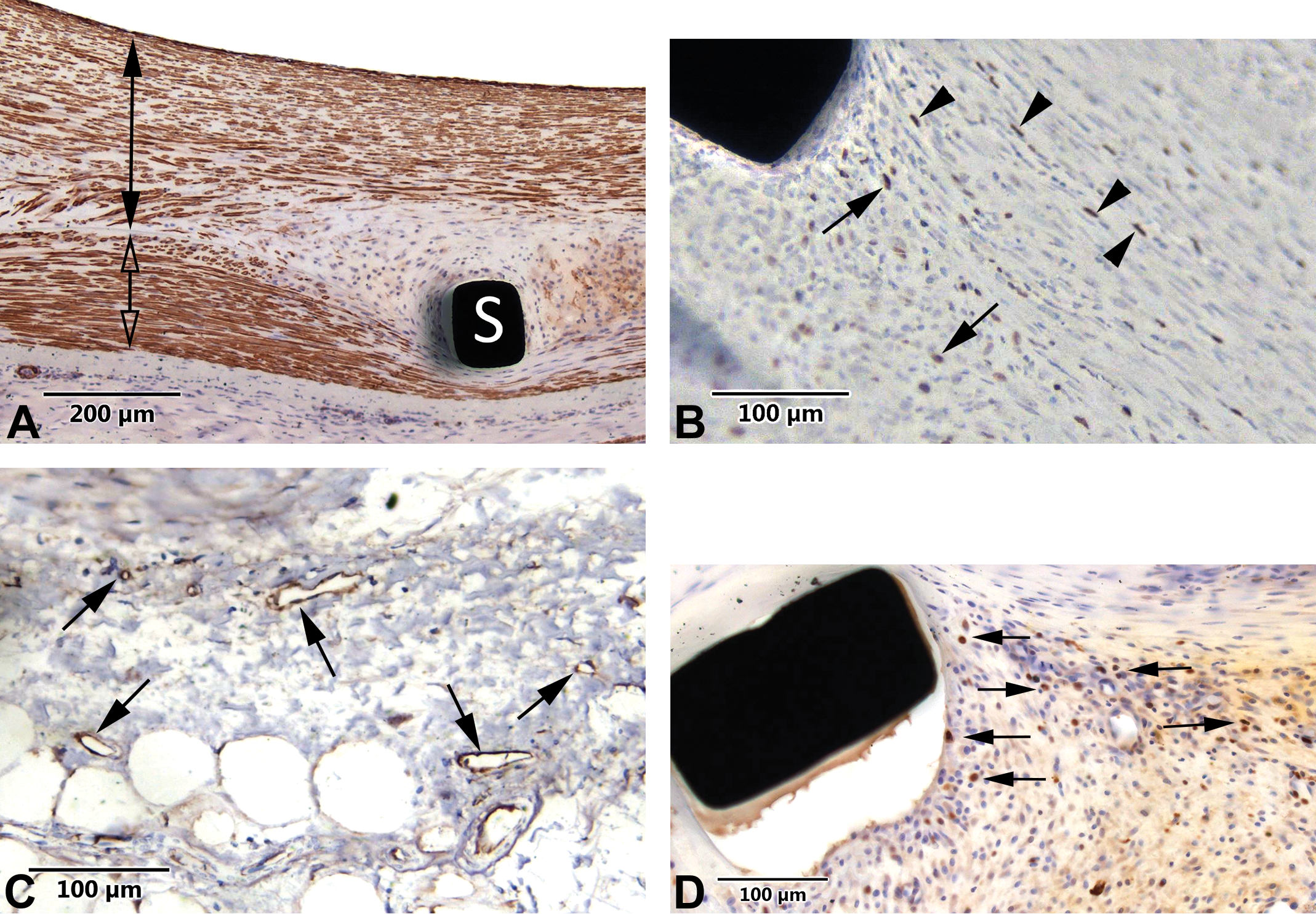
Representative images of the capability of Spurr’s method epon-embedded tissue to provide available epitopes displaying high compatibility for immunohistochemistry. This compatibility is present regardless if its utilizing a rotary microtome under 5 μ sections ([A] [smooth muscle cell actin in the neointima = solid black double arrow and in the media = clear double arrow] and [B] [Ki67 labeling for apoptotic cells; solid black arrows and arrow heads]) or thicker sections ([C] [CD31 labeling for endothelial cells; solid black arrows, field adjacent to the device] and [D] {ki67; solid black arrows]). The amount of metal content in any given device (black areas in sections) doesn’t seem to affect the capabilities of the resin to label for immunohistochemistry.
Processing
Histologic processing of tissue is aimed at imparting the specimen with sufficient stiffness to be sectioned and stained histochemically or by immunohistochemistry (Baskin 2014). The industry standard of gradual alcohol dehydration followed by xylene clearing and paraffin infiltration and embedding often constitutes the best practice except as follows: The biomaterial (or tissue, e.g., undecalcified bone) is harder than paraffin and insufficiently supported upon microtomy, producing tearing and various section artifacts. The biomaterial reacts with one or several reagents (e.g., excessive swelling, dissolution). The biomaterial is not maintained through microtomy and/or staining due to floating and/or detachment. If the tissue interface is well preserved, this is not necessarily a hindrance to evaluation and paraffin processing may still be a viable option. However, some reviewers can be keen on seeing the product in microscopic images which may warrant the selection of a different preparation technique such as plastic embedding and ground/micropolished or laser cut slides. The biomaterial is soluble in one or several reagents (e.g., hydrogels), which may require snap freezing and cryosectioning. The material may still be dissolved and disappear during the staining procedure. Some biomaterials may react with one or several of the processing reagents, including the embedding medium (paraffin, methyl methacrylate [MMA] monomer, epon resin, etc.). Reagent incompatibility may produce swelling, softening, fragmentation, unraveling, delamination, dissolution, and so on. When faced with untested material, it is best to investigate reactivity of the unimplanted biomaterial against a battery of possible reagents in order to identify any incompatibilities or problems in advance of preparing study materials to slide. A reagent test can be run with standardized samples of the unimplanted material, photographed with metric calibration before and after the exposure to the reagents at relevant temperatures and durations (Vertenten et al. 2008). This is an easy and very practical way to identify any problematic reagent and, if a substitution is available, prevent processing loss or alterations of biomaterials by using a substitute to the deleterious reagent. Additional considerations and examples are presented in Table 2.
Algorithm for Sample Handling and Tissue Preparation.
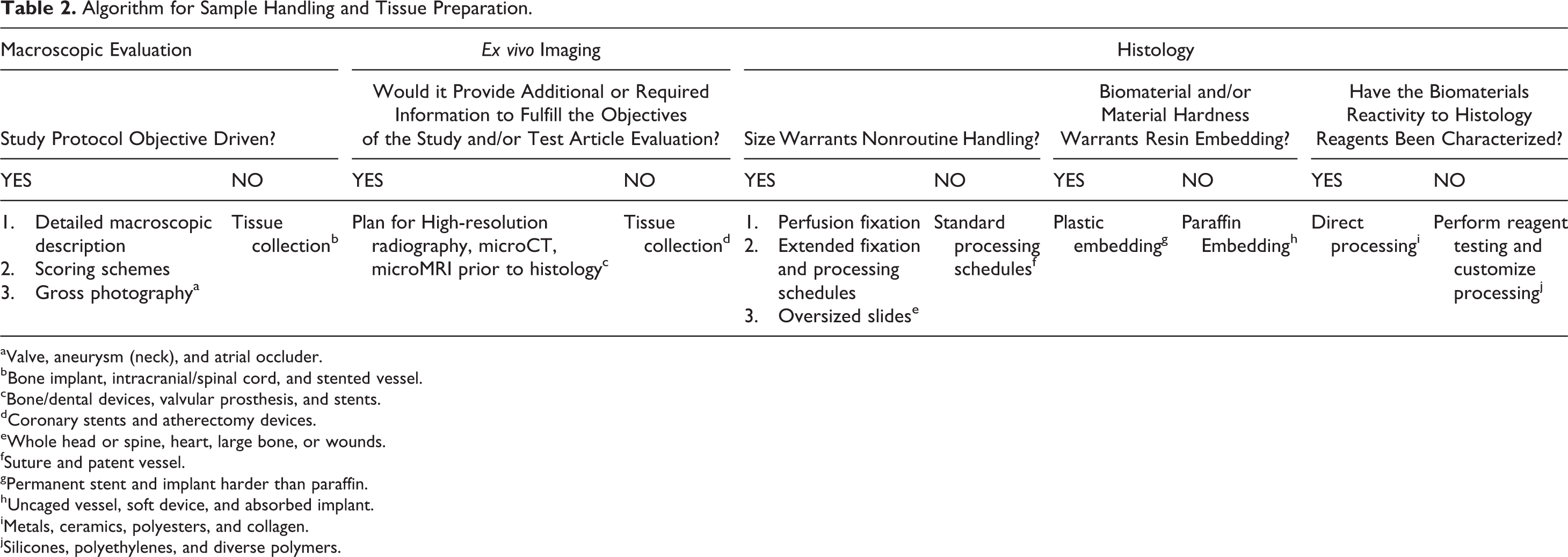
aValve, aneurysm (neck), and atrial occluder.
bBone implant, intracranial/spinal cord, and stented vessel.
cBone/dental devices, valvular prosthesis, and stents.
dCoronary stents and atherectomy devices.
eWhole head or spine, heart, large bone, or wounds.
fSuture and patent vessel.
gPermanent stent and implant harder than paraffin.
hUncaged vessel, soft device, and absorbed implant.
iMetals, ceramics, polyesters, and collagen.
jSilicones, polyethylenes, and diverse polymers.
Microtomy
Biomaterials embedded in paraffin are cut using standard microtomes and disposable blades at 5 μ. Some biomaterials, generally solid polymers, may curl up upon sectioning or float away in the water bath or during staining. If the surrounding tissue and interface are not otherwise affected, loss of the biomaterial does not hinder microscopic assessment, and evaluation of the tissue response or implant integration can still be accomplished. Loss of the biomaterial has the effect of replacing a clear piece of polymer by a negative clear space which is indifferent from a pathology standpoint. In order to avoid or minimize tissue disruption, it is important to carefully adjust water bath temperature at microtomy. When a polymer or biomaterial is porous and becomes invested by fibrovascular tissue (e.g., expanded Polytetrafluoroethylene (ePTFE) or polyester mesh or weaved fabric such as Dacron), it is generally possible to maintain the biomaterial and associated tissue within the section with careful and skilled microtomy handling. Use of charged or coated slides can often further improve the success rate. Biomaterials embedded in plastic medium can be cut using microtomy techniques similar to paraffin-embedded tissues, at 5 μ or thinner. The use of hard microtomy blades such as Tungsten-carbide is often warranted due to the presence of hard materials including metals or calcified bone. Care must be taken to use a blade profile appropriate for the microtome (sledge vs. rotary) and block type (bone, soft tissue, etc.). Resharpenable blades generally offer good hardness and optimal stability. Some biomaterials are too bulky or too hard to be sectioned using microtomy techniques and must be prepared using precise sawing followed by grinding and micropolishing to optical finish. There are several possible technical platforms available for each of the required steps, to include diamond wire, disc, or band sawing and various manufacturers of grinding/polishing equipment. These techniques have been described elsewhere (Rousselle and Wicks 2008; Erben 1997) and follow principles developed for petrographic studies. In short, thin wafers are cut with a diamond-coated blade. Wafer thickness may vary based on block size and saw precision and is typically in the range of 300 to 800 μ. Each cut further depletes the tissue block from the travel of the cutting apparatus through the tissue (kerf loss). Based on thickness of the cutting device, the total amount of tissue required to produce a slide can vary from 300 to 400 μ at a minimum to up to 1 mm or more. True serial sections cannot be produced using this technique.
A new technology based on femtosecond laser sectioning was developed that allows noncontact sectioning of plastic blocks in a true serial fashion, generating tissue sections that are only 10 to 15 μ thick (Richter et al. 2017). The main advantage of this technique is the ability to produce true serial sections in a very repeatable fashion and to conserve the specimen. One drawback of the technology is that the laser cannot penetrate and cut through opaque materials such as metals or ceramics. Thus, these materials are typically visible in conventional ground/micropolished histologic sections (Figure 1) and will appear in “negative” as an optically clear space on the slide. Specimen size is also routinely limited to a maximum of 25 × 35 mm.
Staining
Implants and surgical sites processed in paraffin can be suitably stained with all conventional techniques otherwise available in paraffin-embedded tissues. Plastic embedding can be more constraining, although these limitations primarily apply to tissues embedded in MMA, whereas sections obtained from tissues embedded in epon (Spurr) can typically be stained with most histochemical and immunohistology procedures working in paraffin, including in Spurr ground/micropolished preparations (Figure 1, panels c and d). In our experience, once tissue sections are fully deplasticized, the staining properties and antigen retrievability are very similar to that of deparaffinized tissue sections. MMA-embedded tissues are more challenging to stain, and the array of procedures available is limited (Torgersen, Takle, and Andersen 2009). Immunohistology procedures are notoriously challenging in MMA-embedded tissues, which is believed to be related to antigen denaturation during the exothermic process of MMA polymerization upon block curing (Baskin 2014). It has been theorized that curing under refrigerated condition (chilled water bath and/or in a freezer) improved antigen preservation and suitability of tissue sections for immunohistology. True comparative studies verifying this theory are lacking, and it is just as likely that difficulties in staining MMA-embedded tissues are related to variations in deplasticization quality and completeness and masking of antigens or structural components by residual MMA (Bako et al. 2015). We have found that more extensive deplasticization of MMA-embedded sections, for instance using immersion in MMA monomer, greatly improves staining suitability of thin MMA sections. Nonetheless, it remains important to control the speed of MMA polymerization and to monitor specimen temperature through the process as excessive heat can indeed be generated and spoil specimens if not closely monitored. Additional considerations and examples are presented in Table 3.
Algorithm for Tissue Sectioning and Staining.
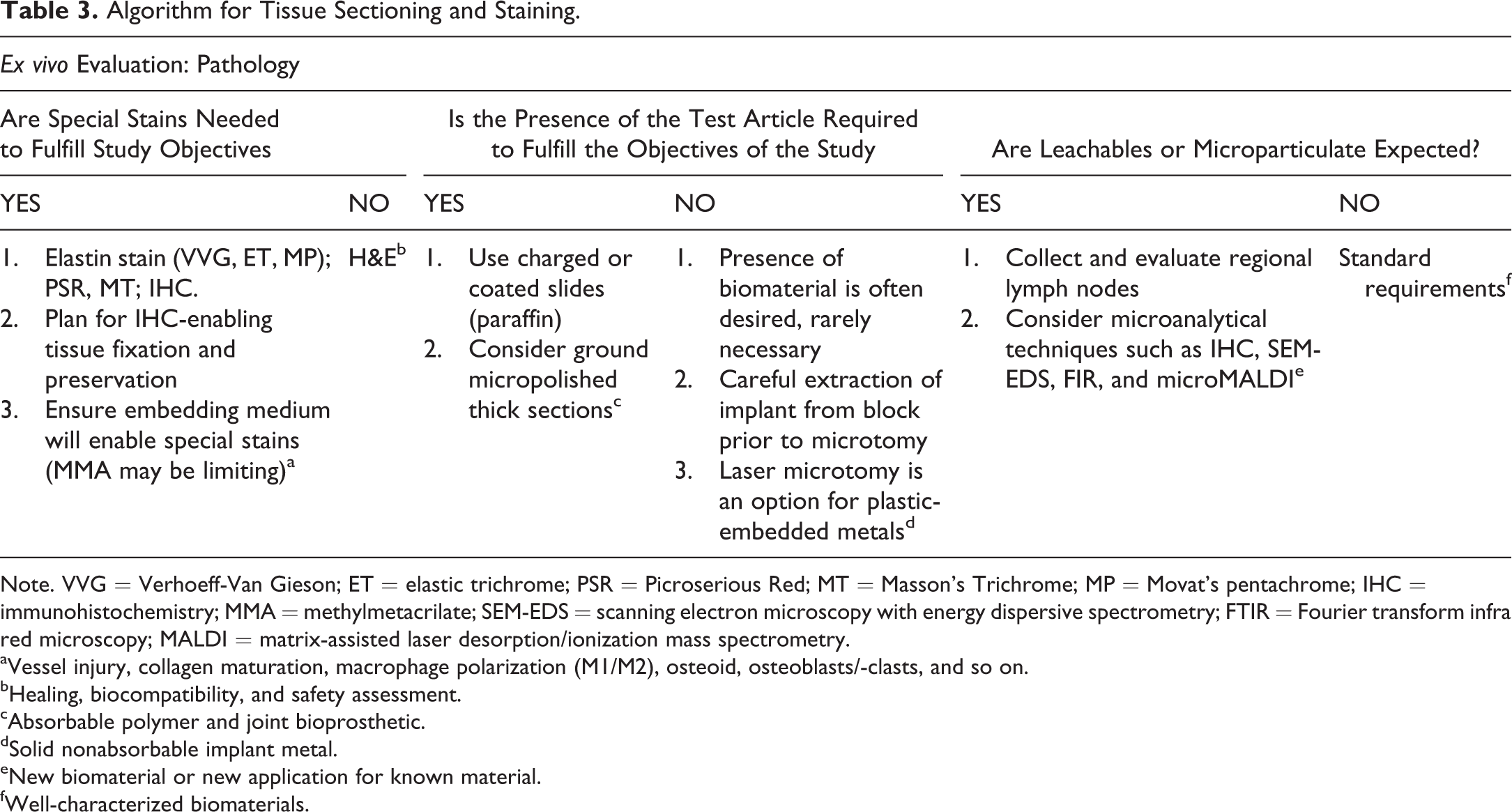
Note. VVG = Verhoeff-Van Gieson; ET = elastic trichrome; PSR = Picroserious Red; MT = Masson’s Trichrome; MP = Movat’s pentachrome; IHC = immunohistochemistry; MMA = methylmetacrilate; SEM-EDS = scanning electron microscopy with energy dispersive spectrometry; FTIR = Fourier transform infra red microscopy; MALDI = matrix-assisted laser desorption/ionization mass spectrometry.
aVessel injury, collagen maturation, macrophage polarization (M1/M2), osteoid, osteoblasts/-clasts, and so on.
bHealing, biocompatibility, and safety assessment.
cAbsorbable polymer and joint bioprosthetic.
dSolid nonabsorbable implant metal.
eNew biomaterial or new application for known material.
fWell-characterized biomaterials.
Application of Histology Strategies to Different Organ Systems
The following section presents an organ system–based review (as outlined in Figure 2) of recommended histological strategies.
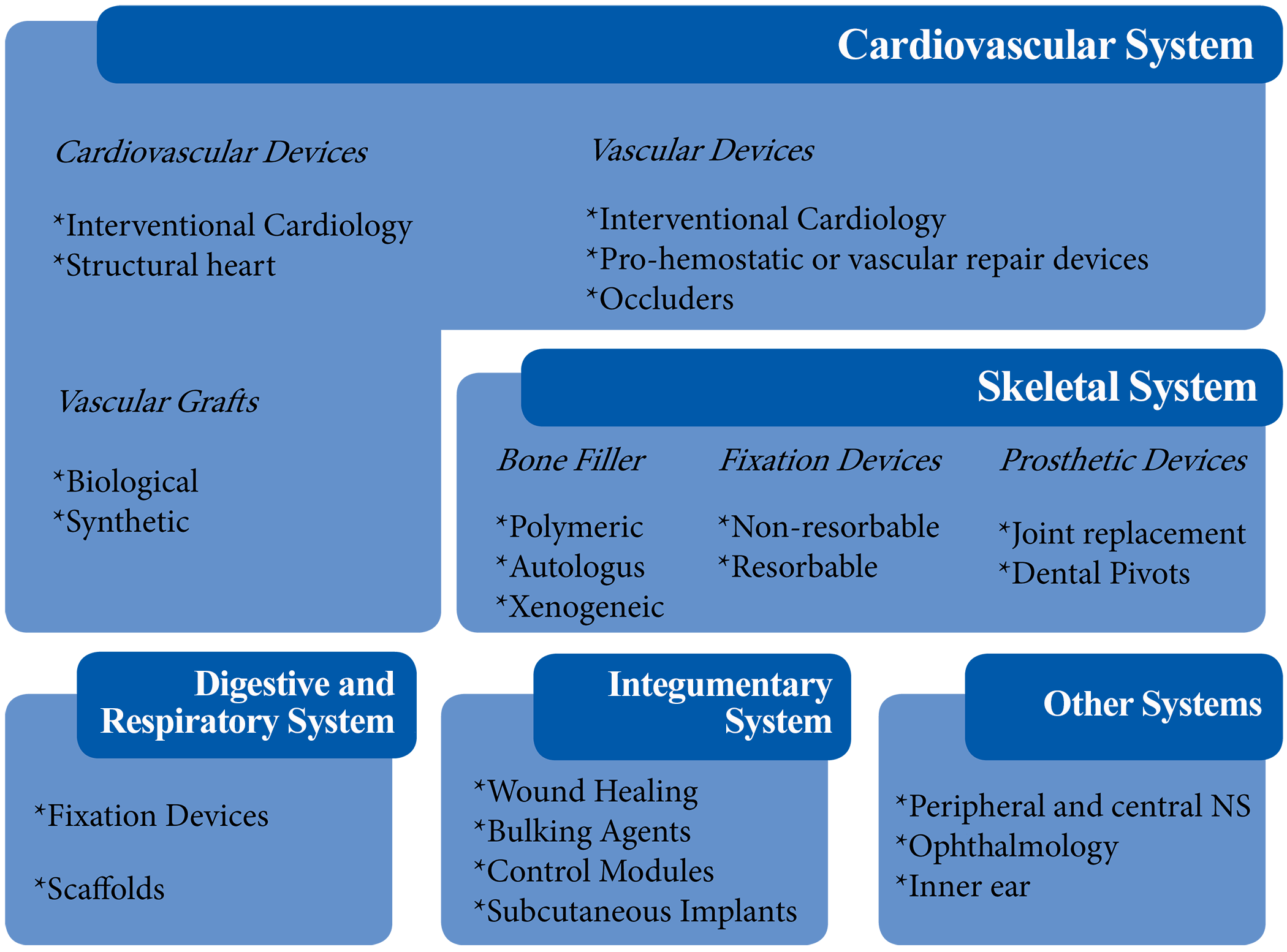
Histology strategies in different organ systems.
For devices that deliver a drug or release by-products or leachable, a tissue list for collection, weighing, and systemic evaluation is provided in ISO 10993-11:2017 Annex E (ISO 10993-6:2016 n.d.).
Cardiovascular System
Devices within and/or affecting the cardiovascular system require consideration given to the effects on local and distant tissues (Table 4), to include contact with blood within the circulatory system. For the preclinical safety assessment of cardiovascular devices intended for FDA approval, a guidance document is available regarding the general conduct of these studies. Safety assessment of these devices is often multifactorial and may require assessment of local and distant thrombosis/thromboembolism, thrombogenicity testing, and neointimal formation and other histologic features of device–tissue interface reaction to correlate with additional imaging modalities such as post mortem radiography, microcomputerized tomography, scanning electron microscopy, and histomorphology.
Algorithm for Downstream Organs and System Effect Evaluation.
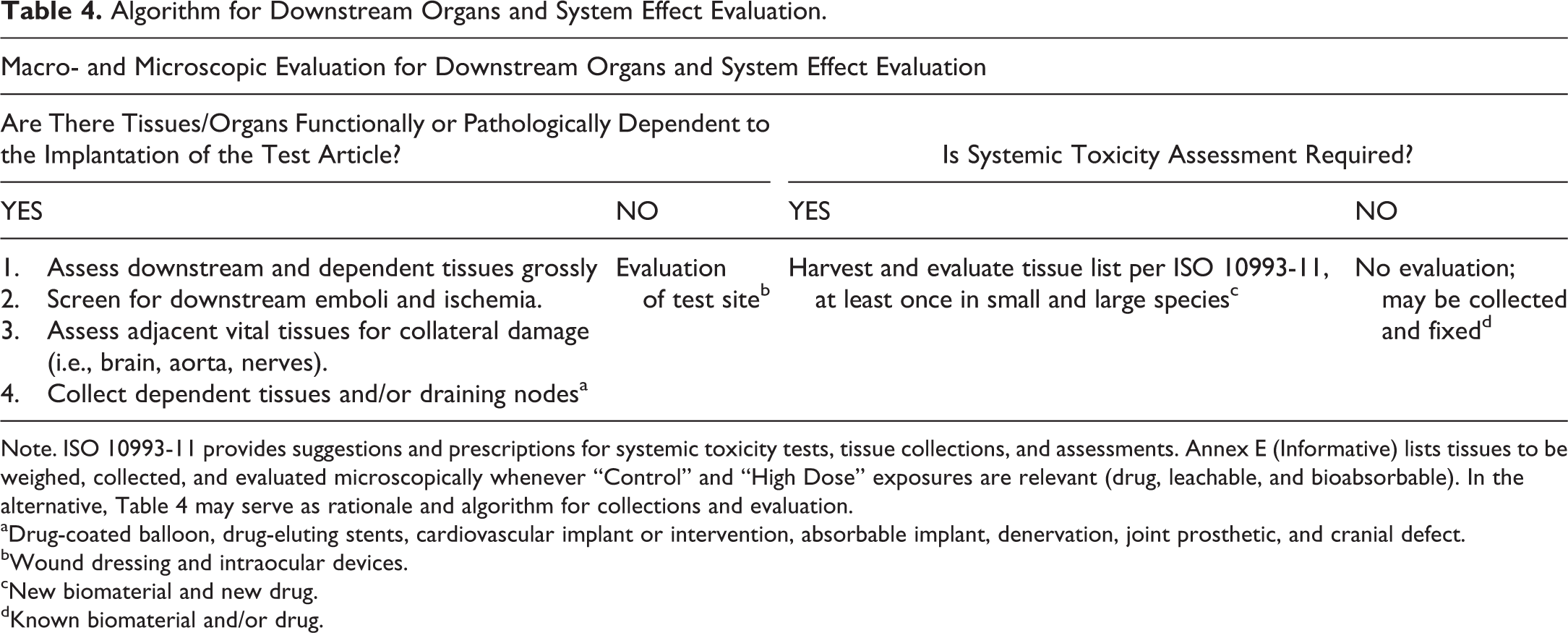
Note. ISO 10993-11 provides suggestions and prescriptions for systemic toxicity tests, tissue collections, and assessments. Annex E (Informative) lists tissues to be weighed, collected, and evaluated microscopically whenever “Control” and “High Dose” exposures are relevant (drug, leachable, and bioabsorbable). In the alternative, Table 4 may serve as rationale and algorithm for collections and evaluation.
aDrug-coated balloon, drug-eluting stents, cardiovascular implant or intervention, absorbable implant, denervation, joint prosthetic, and cranial defect.
bWound dressing and intraocular devices.
cNew biomaterial and new drug.
dKnown biomaterial and/or drug.
a) Cardiac Devices
Nonimplanted devices and energy delivery
Interventional catheters and guidewires are assessed for mechanical damage and healing characteristics within the exposed vessel tract. Critical end points must include an assessment of structural integrity, thrombogenicity, stenosis, and downstream embolization. General principles of tissue preparation discussed above can readily apply. Angiographic records are often necessary to optimize tissue collections and histological sampling. Ablation therapies (i.e., pulmonary vein and atrial chambers) generally rely on localized delivery of heat or cryo-energy along specific anatomical planes until isolation is achieved electrophysiologically (Leshem et al. 2017).
Structural devices
There is a wide and ever-growing array of devices intended to correct heart structure. Some implants are intended to replace a defective structure such as one of the four valves, others are correcting defective conformation (annuloplasty, ventriculoplasty), others occlude developmental or dilated or weakened cavities (septal and appendage occluders; Carpenter et al. 2013), and others still create new flow channels to divert blood in abnormally high-pressure situation (shunt devices). For all structural implants, a comprehensive and well-documented gross description is essential and should be accompanied by detailed gross photography. The evaluation should assess the position and conformation of the implanted device, the presence of thrombus, its tissue integration and coverage, and any abnormal deposits or vegetative deposits. Tissue dissection and trimming should be methodical and conservative to offer good visual exposure but also conserve anatomical relationships and interfaces. Radiographic and/or computerized tomographic imaging before and after dissection are useful to document position, structural integrity of frame members, and assess calcification. The excised implant site will typically require plastic embedding and ground/micropolished slide preparation due to frequent involvement of large metal frame members or dense compound constructs associating a densely weaved metal and other biomaterials such as polyester fabric, ties, and collagenous bioprosthetic layers. Heart valve implants are examples of constructs that involve an assembly of several biomaterials. Histopathological approaches have evolved overtime and state-of-the-art methods have been recently described (Tellez, Dillon, and Rousselle 2017). Methods for annuloplasty devices are also described elsewhere (Ramot et al. 2016). The histology strategy for safety assessment must ensure that all biomaterials exposed to body fluids and tissue are representatively sampled and sectioned in order to assess hemocompatibility and local tissue response. The evaluation must support conclusions on thrombogenicity, local irritation, integration, and healing at the implantation site. In addition, potential downstream effects (i.e., thromboembolic events and infarction) must be investigated (Table 4).
b) Vascular Devices
Vascular histology presents a number of challenges that must be well understood prior to designing a collection and sampling strategy and interpreting pathology and morphometry data. Post mortem elastic recoil is a confounding and frustrating issue that affects any uncaged vessel with a rich elastin framework. Arteries, namely, peripheral arteries and to a much lesser degrees veins are subject to passive recoil upon the loss of blood pressure. Current guidelines promote pressurized fixation in order to mitigate the effects of this natural property on important study end points such as lumen patency or wall thickness. It is important to note that recoil also occurs longitudinally and that measurements taken in vivo can generally not be transcribed into the fixed tissue, unless the length of the vessel is maintained through attachment to a rigid support. As reported, histology grade fixatives do not fix elastin (Fung and Sobin 1981). Whereas pressurized fixation indirectly reduces recoil through fixation of other tissue components (primarily collagen), it does not prevent elastic recoil entirely (Takemura et al. 1993). This effect is further aggravated by tissue shrinkage that occurs during processing. The magnitude of this postmortem artifact can be very significant, and it has been reported that for iliac and femoral arteries in swine, the decrease in lumen area attributable to elastic recoil and tissue shrinkage during processing can be as much as 65% (Tellez et al. 2015). Therefore, lumen area measurements and patency outcome derived from computerized morphometry must be interpreted with due caution in unsupported vessels (Figure 3, panels a and b).
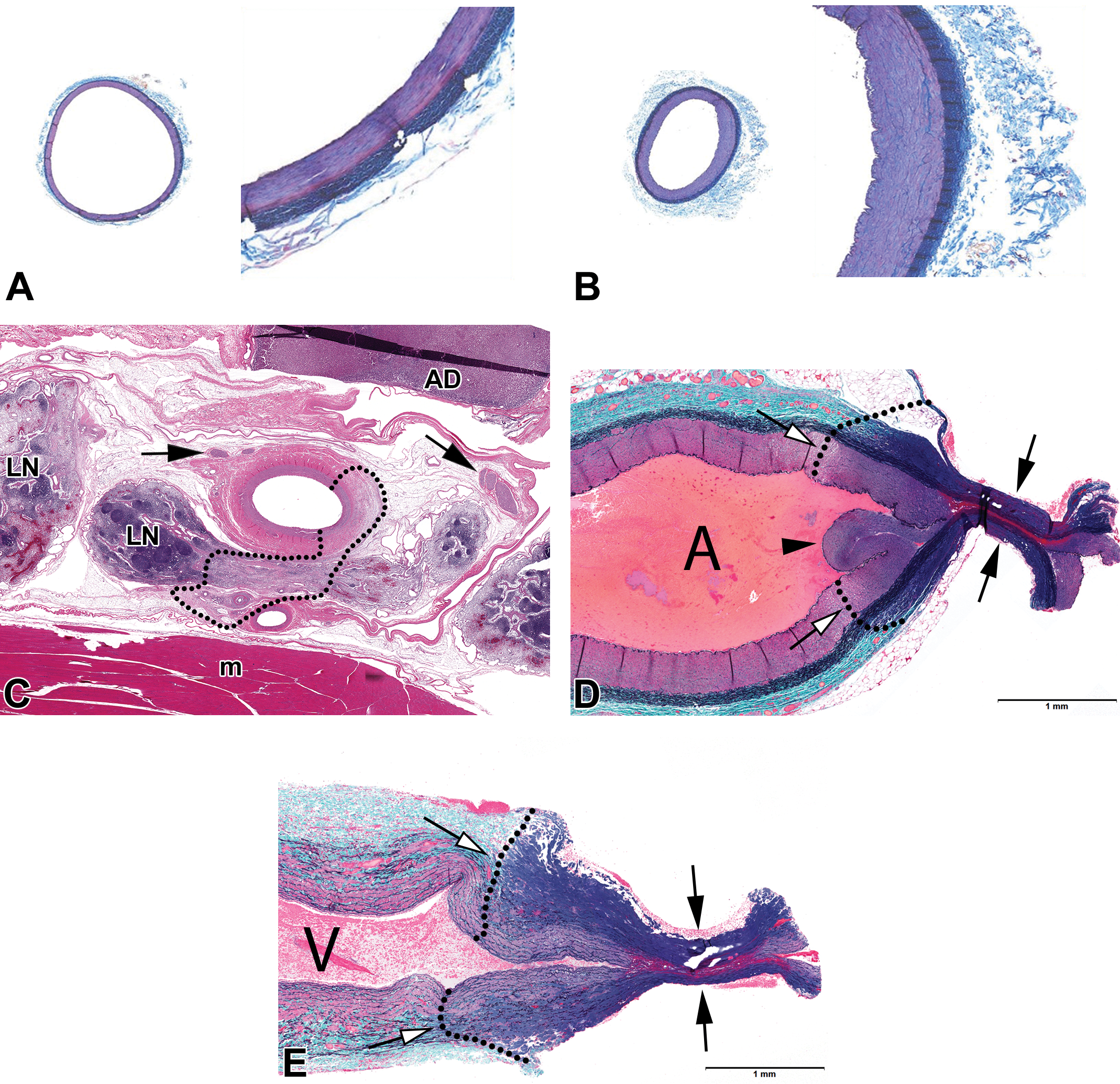
Representative histological images of low magnification (left) and high magnification (right) of a femoral swine arteries, where a intraluminal scaffold polymeric filler compatible with paraffin embedding was used (A) and a contralateral femoral artery without any intraluminal support (B). Important differences can be noted. Panel a displays a in vivo–like vascular diameters where the vascular wall is thinner, endothelium is flat and smooth muscle cells in the medial layer display a straight array. In contrast, (B) show a corrugated intimal layer, a thick neointimal wall, and an evident decrease in vascular area demonstrating the unaffected recoil properties of the elastic fibers despite pressure perfusion fixation and dehydration processing. (C) display a representative cross section image of the effects in a renal artery and surrounding tissue of a catheter-based renal denervation device. The dotted line display the thermal injury extent (AD = adrenal, m = muscle, LN = lymph node; clear arrows = nerves). (D) and (E) shows a Gomori’s trichrome stain demonstrating the effects of the thermal surgical vessel hemostatic sealing in an artery (D) and vein (E) (White arrow = edge of thermal injury in the media; Black arrow = seal crushed by clamp jaws; Arrowhead = necrotic média pressed in the vessel lumen; A = artery; V = vein).
It is best to sample and section vascular tissue across the axis of symmetry of the lumen to optimize stenosis/restenosis assessment. Oblique or longitudinal sectioning schemes are usually ill-advised because they can seldom be performed at a consistent and precise location due to vessel tortuosity and/or masking of the tissue by reactive fibrous tissue deposition. Longitudinal sections are unsuitable for stenosis measurement on the basis of pure geometry and common sense. The implant site should be sampled at representative levels proportionally to vessel length and to include proximal and distal reference. It is generally recommended to sample vascular devices every 5 to 10 mm, although exceptions can be made if there is an expectation of relative homogeneity through the length of the implanted vessel. Any abnormalities detected angiographically or macroscopically should also be sampled to ascertain that the worst outcome through the vessel length is being considered and reported upon.
In order to precisely evaluate mural integrity, vascular tissue histology should always include an elastin stain. Procedures that also support assessment of fibrosis and/or proteoglycan matrix deposition are most suitable (e.g., Elastin Trichrome, Gomori’s Elaastin Trichrome, Movat’s Pentachrome).
Interventional devices (thrombectomy, energy delivery, drug delivery)
Histology methods for thrombectomy devices are described elsewhere in this issue. Energy delivery systems have been studied extensively as a potential treatment for drug resistant hypertension (renal denervation) and other disease entities (pulmonary hypertension, Chronic Obstructive Pulmonary Disease (COPD), abdominal pain, etc.). These treatments involve a catheter that is removed upon the completion of the procedure. Therefore, targeted tissues can be routinely processed in paraffin. The preservation of local anatomical relationships at the treated site is of paramount importance to assess for collateral damage and also somewhat accurately measure treatment extension at the point of delivery (Figure 3, panel c). Suitable methods for renal denervation models have been described (Tellez et al. 2013; Rippy et al. 2011; Sakakura et al. 2015).
Patches, hemostatics, sealers
There is a wide variety of devices under study intended for vascular access sealing or vascular repair. Most surgical implants are collagen-derived and intended to be surgically applied, others are composed of bioabsorbable polymers and a subset of these can be delivered interventionally upon vascular egress. Collagenous implants can be processed and embedded in paraffin, whereas biopolymers may or may not require plastic embedding based on implant hardness and absorption status. In order to minimize trimming artifacts (artificial tissue disruption upon gross sectioning), it is sometimes advisable to process the implanted vessel whole and trim prior to embedding or embed the vessel in epon. These decisions are made on a case-by-case basis based on prior experience and intended histology outcome. Histologically we have found that acute thermal injury, namely, collagen denaturation can be very exquisitely outlined using Gomori’s Elastin Trichrome stain (Figure 3, panels d and e). Electrosurgical vessel sealers are evaluated following a dedicated FDA guidance document (FDA 1996). Histopathological end points must include quantitative analysis of acute thermal damage spread in the target tissue. This represents an exception to the recommendation of sampling the tissue via cross sections of the lumen. Vascular seals are best sampled and cut perpendicularly to the seal, hence in the axis of the sealed vessel, such that longitudinal thermal injury spread can be adequately measured. It is further stipulated in the guidance document that the report should include high-quality images of the seal depicting how measurements were taken. The Gomori’s Elastin stain greatly facilitates the evaluation of thermal spread (i.e., collagen denaturation). Chronic assessment is primarily focused on healing characteristics, evidence of leakage, and collateral damage. Tissue collection for chronic time points can be challenging due to the amount of scaring and fibrous tissue deposition. The use of fiduciary markers placed at the time of surgery, for instance, long nonabsorbable sutures (e.g., prolene) can be helpful in tracking the treatment sites at necropsy.
Occluders, leads, and sensors
Vascular occluders are used to isolate a territory (e.g., aneurysm, tumor, varicose vein) and are assessed for efficacy (occlusion) and safety (vascular leakage, vessel integrity, healing, collateral damage) using histological methods suitable for the implant (paraffin or hard plastic). The sectioning must be perpendicular to the vessel/occluder axis to support the measurement of percent occlusion in the targeted vessel. The evaluation should particularly focus on areas where the device may have its greatest diameter which is established through radiographic imaging or macroscopic appearance, as these areas generally represent the site of maximum occlusion. It is also often necessary to sample the occluded segment at several levels to increase the chances of capturing regions of maximum occlusion and of vascular injury potentially caused by overstretch. Regardless of histological appearance, patency assessment must be compared to terminal angiographic data for interpretation. It is not unusual to observe a network of vascular channels within reorganized occluded segments, particularly with porous implants (e.g., nitinol self-expanding wire mesh) which do not necessarily translate into functional patency angiographically. Sensors and electrostimulation leads can be placed intraluminally or along the adventitia of a vessel, along nerves or within a target tissue such as brain parenchyma. General sampling methods apply (cross sectioning). It is notably important to section any unique interface that may display a specific tissue response by virtue of the nature of the biomaterial or uniqueness of the microanatomical relationships with the surrounding tissue. These devices are highly engineered and can assemble many different components. A thorough understanding of the implant design and elements interfacing with the tissue as well as implantation track is necessary in order to sample the tissue at meaningful positions. It is also prudent to ascertain that none of the components will adversely react and be transformed by exposure to histology reagents. The presence of silicone insulation on electrical components can be particularly problematic because certain silicones can swell excessively during the exposure to organic solvents used in the histology process which may significantly impact histological presentation and ability to accurately assess vessel patency. Reagent testing prior to histology processing is highly recommended.
c) Vascular Grafts
Biological
Bioprosthetic vascular grafts such as autologous vein grafts are used in vascular bypass models. Tissue handling and processing generally follows guidelines applicable to blood vessels. Particular attention to anastomotic sites is critical as they may be a site predisposed to failure or regions of greater biological reactivity and neointimal response.
Synthetic
Synthetic grafts have evolved from simple polymer constructs tube (e.g., ePTFE) to highly engineered constructs often assembling several biomaterials that may include absorbable or nonabsorbable synthetic or biological polymers, stem cells, and/or drug or growth factors. The composition and histology and pathology end points must be carefully considered and taken into account in designing successful histology methods. Untested polymers must be tested against histology reagents and the choice of embedding method (i.e., paraffin vs. plastic) must be weighed against protocol objectives and material behavior through the process as tested and qualified by a method optimization phase. Sampling strategies generally follow that of biological grafts.
Skeletal System (Bones, Teeth, Ligaments)
Specificities, Limitations, and General Considerations
Bone fixation is especially challenging due to very slow penetration of fixative solutions through cortical bone and cartilage. Fixation will regularly be inadequate through immersion alone unless the marrow cavity and/or trabecular bone are exposed directly to the fixative using a bone saw, provided the distance between the exposed surface and the target region of interest is not too great (preferably less than 2 cm). Whenever large bones need to be left intact, the region should be perfusion-fixed prior to dissection and collection. High-resolution imaging such as contact radiography or microCT is frequently utilized to assess bone healing and regeneration, and these data sets are always very useful and sometimes indispensable to inform tissue sampling and sectioning.
a) Bone Fillers, Cartilage Replacement
Bone fillers and cartilage repair implants are generally tested in defects or critical-size defects, for instance, drill holes within metaphyseal or epiphyseal bone or within the articular surface. The rabbit and sheep are the most commonly used species. The rat and dog are often used for cranium defect models (Muschler et al. 2010). Typically, a standard size drilled defect or bone flap is created for the application of a putty or test article. Main challenges are associated with sampling and sectioning the defect sites accurately, particularly after healing and remodeling have occurred. While microCT imaging can be extremely helpful in gauging the location of experimental sites (Chagnon, Guy, and Jackson 2019), it is not fool proof and, under current techniques available, requires that imaging interpretation be transcribed into the physical specimen, with some level of human variability or imprecision. We have found that whenever possible, it is best to place fiduciary markers during surgery at the time of defect creation, for instance, small metallic pins placed into the bone on diametrically opposed sides of the defect and parallel to it. Such markers provide visual confirmation for the exact location and direction of the defect site, even when the sites are fully healed. The markers must be sized and positioned such that they do not interfere with experimental site healing or biomechanics. Based upon filler composition, bone defects can be decalcified and processed in paraffin or embedded in plastic (MMA) for thin or ground/micropolishing slide preparation (Figure 4). Additional methodological principles and specifics are discussed elsewhere in this issue.
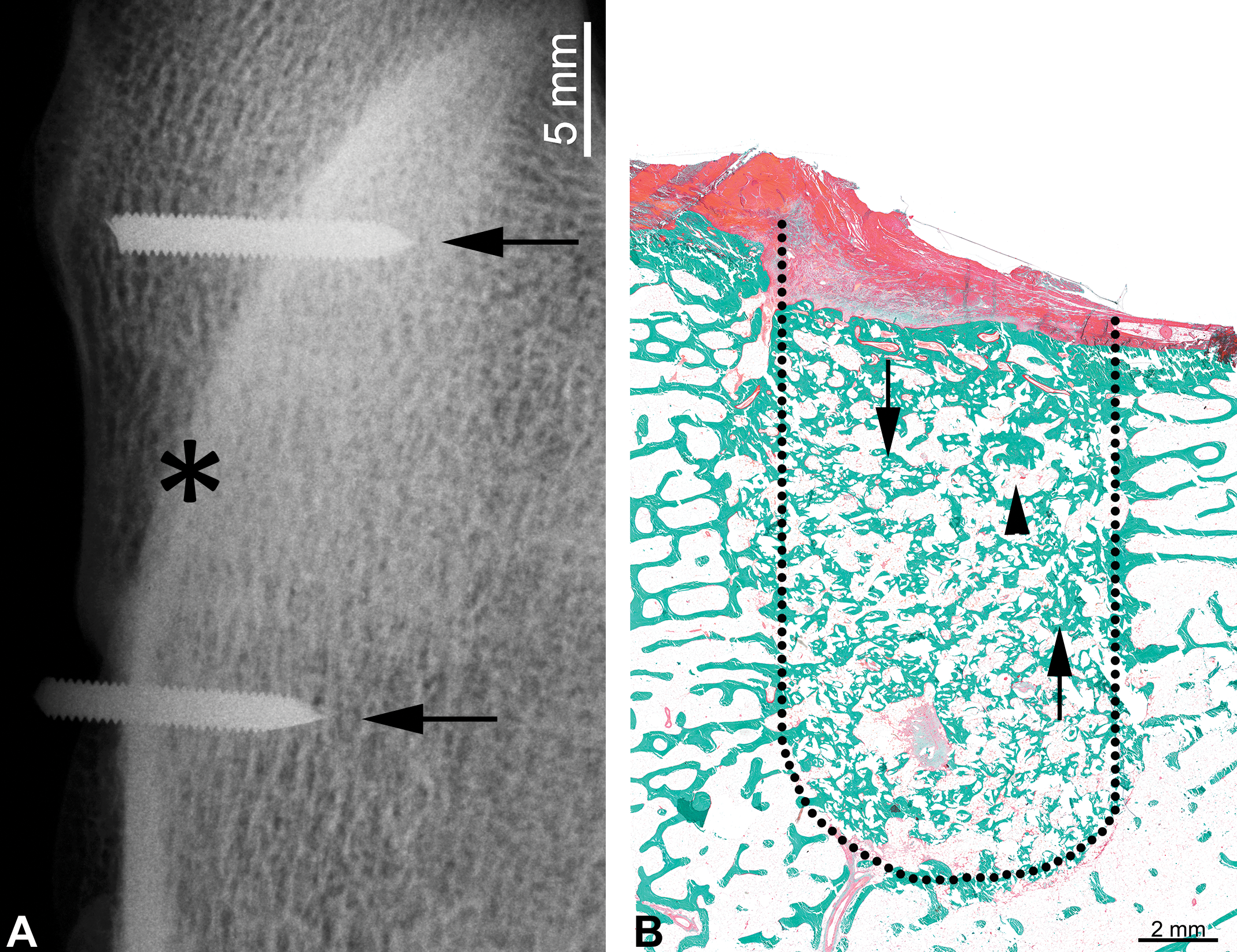
Representative image of a radiograph where orthopedic device can be observed with physical identifiable markers (A). Once the exact location and orientation of the experimental site is confirmed, a histological section can be successfully performed through the bisecting axis (asterisk). (B) displays in a Goldner trichrome stain, a bone defect area (dotted line) showing new bone formation (arrows) and residual bioglass (arrowheads).
b) Fixation Devices
Nonresorbable
Nonresorbable fixation devices such as screws, plates, wires, and meshes require plastic embedding and sectioning using grinding/micropolishing techniques or laser sectioning, recognizing that with the later the device itself will not be cut and will not be present in the tissue section. When the device, for instance, a pin or long plate prevents presectioning of the bone to expose the medullary cavity for fixation, perfusion fixation of the territory should be considered prior to harvesting dissection and subsequent immersion fixation. Cryopreservation is not an adequate mode of preservation for large bones as specimen size is incompatible with snap-freezing and freezing artifacts are unavoidable.
Absorbable and polymer fixation devices
Resorbable or polymer fixation devices can be more challenging histologically because they may be more difficult to visualize radiographically or macroscopically or due to gradual absorption and disappearance. In addition, some materials such as polymers may react with histology reagents and should be tested prior to preparing study tissues. Fixation devices that include bioactive components or stem cells remain challenging to study with current techniques, namely, concurrently with plastic embedding due to difficulties in using immunohistochemistry with MMA embedding. Spurr (epon) embedding is an attractive alternative, but its optimization remains elusive in large bones likely due to epon’s greater viscosity compared to MMA.
Prosthetic devices (joint replacements, dental pivots)
Joint replacement implants present histology challenges related primarily to the size of the surgical sites and amount as well as hardness of biomaterial involved (titanium alloys, bioceramics, or hard polymers). Fixation recommendations discussed above should be carefully followed. Optimal preservation of implant site integrity relies on embedding and sectioning of large intact joints, which requires very long processing and plastic infiltration schedules for quality results. These limitations should be considered at study design in order to manage expectations. Large MMA batches are also prone to exothermic auto-acceleration of the polymerization process and must be monitored very closely and temperature-controlled during curing and hardening. Dental implants can generally be handled and processed in a manner similar to fixation screws. Bioabsorbable dental implants present challenges for sampling and sectioning when the site becomes difficult to identify due to tissue healing and remodeling and availability of microCT imaging can be critical.
Digestive and Respiratory System
Specificities, Limitations, and General Considerations
Gastrointestinal and respiratory devices constitute a very heterogeneous family of implants that are designed to fulfill a wide array of missions from surgical apposition (staples), sealing, functional occlusion, stenotic opening and caging, drug delivery, and so on. They can be permanent, bioabsorbable, or retrievable. Endoscopic imaging and/or contact radiography can often be used to assist with sampling and sectioning.
a) Fixation Devices: Staples
The integrity of staple sites is best maintained using plastic embedding (Spurr epon preferably) followed by microtomy thin sectioning. Stainless steel or even harder staples can generally be satisfactorily cut using such technique, whereas removal of staples in fixed or paraffin-embedded tissue prior to sectioning generally creates unacceptable tearing artifacts that impede microscopic evaluation and may confound interpretation of tissue apposition and healing. These sites are cut across the anastomosis or staple line.
b) Scaffolds: Biliary, Esophageal
Metallic and bioabsorbable stents are developed for use in stenotic biliary and esophageal conditions. The canine and swine model is most commonly used (Shatzel et al. 2016). General sampling and preparation techniques follow that of scaffolds in other locations such as vasculature. Pathology changes and end points are site-specific and include epithelial/mucosal integration, ulceration, reactive hyperplasia, and/or polyp formation. Ascending bacterial infection (biliary) and bile salt precipitates can complicate healing outcome and interpretation.
Integumentary System
a) Bulking Agents
Bulking agents are most commonly comprised of natural extracellular matrices to include collagen, proteoglycans, autologous fat, or synthetic materials such as silicones or hydrogels. These implants can typically be prepared using conventional paraffin techniques and special stains appropriate for the test material (e.g., trichrome stains, picro-sirius red). Silicones can require reagent substitutions to prevent excessive swelling, and hydrogels are often dispersed or dissolved through histology processing, leaving a clear empty space in tissue sections.
b) Control Modules, Sensors
Modules that control internal leads or sensors, supply power, and relay data (implantable pulse generators, implantable cardioversion defibrillator, etc.) are generally implanted subcutaneously. Evaluation of local biocompatibility and healing at the site of implantation can be conducted after the extraction of the module followed by conventional paraffin histology of the tissue capsule or may be processed in situ through plastic embedded for optimal preservation of the implant–tissue interface. Certain batteries may not be safely cut into and should be removed prior to processing or cutting. Specimen size must be carefully considered, and processing and infiltration times tailored accordingly and supraproportionally for quality results.
c) Breast Implants, Supporting Mesh
Biomaterials used for breast replacement or support are generally soft and can be permanent or bioabsorbable. Histology approaches focus on the implant–connective tissue interface and can generally be performed with conventional paraffin techniques. These techniques generally offer optimal preservation of the tissue capsule and interface, although the implant material itself may become displaced and replaced by an optically clear space in the tissue sections. This does not generally impede the evaluation of biocompatibility, healing, and safety. If the presence of the implant material and integrity of the interface is mandated, preparation in plastic medium may be warranted.
Urinary and Reproductive Tract
Vaginal, uterine, prostate, and urethral anatomy is highly species-dependent, and it is challenging to work with a model that adequately supports the assessment of devices developed for human conditions such as prostatic hyperplasia or urinary incontinence. Some species have sigmoid inflexions that impede access, others have bony features that limit biomechanics. The canine prostate is relatively suitable and most commonly chosen for implantation and testing of intraprostatic devices or urethral drug delivery catheters and balloons. Provided that species anatomy is well understood, tissue collections and preparation present no major challenge, and device characteristics dictate histology methodologies.
Other Systems
a) Ocular Devices
Main challenges associated with ocular devices relate to the difficulties in producing quality histology preparations with this tissue, namely, for adequate fixation and careful handling and trimming and have been described elsewhere (Teixeira and Render n.d.) and within this issue (Drevon-Gaillot 2019).
b) Inner Ear
Inner ear cochlear electrodes and sensors interfacing with the acoustic nerve are developed and tested for local biocompatibility and safety. The inner ear and cochlea are naturally embedded in a very dense bone and notoriously difficult to assess histologically. Due to the tight bone structure, histology processing and infiltration times must be extended for quality results in plastic. Histological preparation techniques for human ear have been described (Rau et al. 2013). The different orientation of animal middle and inner ear structures compared to humans requires adaptation of some of the techniques discussed.
Recommendations and Summary
Implants and devices histology is nonroutine and requires qualified planning and discussion with a qualified histology laboratory and pathologist prior to study start. Maintaining the integrity of the site and interfaces is paramount. There are very few established standards, consensus documents (Holmes et al. 1998; Schwartz et al. 2008; FDA 2008), and guidelines available that contain practical technical details (i.e., stents). For everything else, sponsors and investigators have to rely on industry practices and expertise maintained in a few laboratories. New technologies (laser sectioning for MMA) and a shift from MMA to Epon plastic are opening up new options for special stains and immunohistochemistry, but these methods and protocols remain very specialized and expensive.
Footnotes
Author Contributions
All authors (SR, JW, BT, AT, MO) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
