Abstract
Atherosclerosis places a significant burden on humankind; it is the leading cause of mortality globally, and for those living with atherosclerosis, it can significantly impact quality of life. Fortunately, treatment advances have effectively reduced the morbidity and mortality related to atherosclerosis, with one such modality being percutaneous intervention (PCI) to open occluded arteries. Over the 40-year history of PCI, preclinical models have played a critical role in demonstrating proof of concept, characterizing the in vivo behavior (pharmacokinetics, degradation) and providing a reasonable assurance of biologic safety of interventional devices before entering into clinical trials. Further, preclinical models may provide insight into the potential efficacy of these devices with the appropriate study design and end points. While several species have been used in the evaluation of interventional devices, the porcine model has been the principal model used in the evaluation of safety of devices for both coronary and endovascular treatments. This article reviews the fundamentals of permanent stents, transient scaffolds, and drug-coated balloons and the models, objectives, and methods used in their preclinical evaluation.
Keywords
For its global burden of morbidity and mortality, atherosclerosis has been termed the “twenty-first century epidemic” (Estol 2011). Significant advancements have been made in the prevention and treatment of atherosclerosis through lifestyle modulation and medical therapy, though there remains the need for the acute treatment of arterial stenoses and occlusions through nonmedical means (Hansson 2005; Califf et al. 1989; Neamtu et al. 2014). As a median between medicine and surgery, percutaneous intervention (PCI) is a minimally invasive, catheter-based approach that has revolutionized the treatment of occlusive arterial disease. PCI and the field of interventional cardiology originated in 1977 with the first balloon angioplasty (BA), and over its forty-year history, PCI has matured and progressed with steady-stream development of bare metal stents (BMS) and drug-eluting stents (DES), fully bioresorbable scaffolds (BRS), and drug-coated balloons (DCB) for the treatment of both coronary and peripheral arterial diseases (Bertrand 2016). This modest list of interventional device families fails to take into account the plethora of accessory devices used in the assessment, access, and preparation of occlusive arterial lesions prior to treatment, as well as the more recently developed interventional devices for the treatment or repair of valvular and structural heart diseases which are beyond the scope of this review.
Since the inception of interventional cardiology, preclinical models have been essential in demonstrating proof of concept, confirming safety, and, with the more recent development of drug-eluting and/or bioresorbable devices, characterizing the in vivo behavior (e.g., pharmacokinetics, degradation) of these devices. Preclinical models have also played a cursory role in predicting clinical efficacy, as there are limitations in the extrapolation of preclinical outcomes to clinical efficacy. Entailed herein is an overview of the essential classes of interventional devices for the treatment of occlusive arterial disease and the methods by which these devices are assessed preclinically.
Balloons, Stents, and Scaffolds: A Brief History of Interventional Cardiology
Congruent with the proverb “necessity is the mother of invention,” the field of interventional cardiology has been built on a history of invention and reiteration based on the progression of need. Along the evolutionary time line of interventional cardiology, there are five main families of devices intended for the interventional treatment of occlusive arterial disease: balloons, DCB, BMS, DES, and bioresorbable vascular scaffolds. Additional devices evolved along with these, including specialized guidewires, catheters, and atherectomy devices, though these are beyond the scope of this review. Of the five main families, BA was the first developed in the 1970s, but its effectiveness was limited by abrupt vessel closure due to elastic recoil, vessel wall dissection, and resultant negative remodeling (Simsek and Serruys 2011). Still today, BA can serve as the first line of treatment in restoring patency to an occluded artery prior to stent/scaffold implantation, and BA has more recently developed into a second family of interventional devices, DCB, which are most commonly used in peripheral applications (Barkat, Torella, and Antoniou 2016).
BMS originated in the 1990s to overcome the shortcomings of BA, permanently supporting an occluded artery to prevent elastic recoil and negative remodeling and to “tack down” vessel dissection flaps. But BMS met with its own shortcomings, namely that of in-stent restenosis (ISR) that plagued nearly a third of patients treated with a BMS (Burt and Hunter 2006).
The turn of the 21st century marked the dawn of a new era for PCI with the development of DES, which have effectively reduced the frequency of ISR of BMS from 30% to single digits (Bangalore et al. 2012). This success of DES has been a key contributor in the rapid growth of interventional cardiology whereby coronary intervention is now the most frequently performed therapeutic procedure in medicine (Stefanini and Holmes 2013). Today, the fast-paced development of DES continues in order to enhance treatment of a broader range of patients (e.g., diabetics, high bleeding risk patients) and lesion subsets (e.g., chronic total occlusions [CTOs], left main, diffuse disease, challenging bifurcations).
Most recent in the clinical history of PCI is the BRS, though its actual concept and development historically matches that of DES (Zidar, Lincoff, and Stack 1994). Similar to permanent stents, BRS is designed to mechanically and (typically) pharmacologically treat the occlusive lesion in the short term. However, in contrast to permanent stents, BRS gradually resorbs to leave no functional permanent implant, thus allowing for restoration of more normal arterial function (Brie et al. 2016; Im, Jung, and Kim 2017). This bioresorption process necessitates additional considerations from the preclinical perspective, often requiring long term in vivo characterization.
Each of these families of interventional devices continues to mature with the intent of improving safety, deliverability, and efficacy with the development of new platform designs and metal alloys; the application of alternative polymer-free, bioresorbable and durable polymer coatings; and the investigation of new drugs and drug-coating formulations for enhancing healing while suppressing restenotic potential. And throughout this development process, preclinical models serve as the cornerstone for confirming safety and for investigating the potential efficacy of these devices.
Preclinical Models for the Evaluation of Vascular Interventional Devices
A variety of animal models have been used for the assessment of interventional devices. Rodents, rabbits, dogs, swine, sheep, goats, and nonhuman primates have been the prevalent ones utilized, and a full discourse detailing the advantages and disadvantages of each model are available in the references provided (Iqbal et al. 2016; Perkins 2010; Jeremy and Thomas 2010; Bayes-Genis et al. 2000). Of these species, swine are the most commonly used and recognized by regulatory agencies relative to their physiologic and anatomic similarities to humans for both coronary and peripheral applications (Schwartz et al. 2008; Schwartz, Edelman, Carter et al. 2002, 2004; EMEA 2007; Food and Drug Administration 2008). Secondarily, rabbits (and more specifically the iliofemoral arteries) serve as an acceptable model primarily for coronary stents. Sheep and goats are suitable alternatives to swine for both coronary and peripheral applications, though the vascular response in these species is not as well characterized. Because of their prevalent usage, swine will be the focus of this discussion.
Objectives of Preclinical Evaluation
Three overarching objectives govern the preclinical evaluation of interventional devices:
In vivo characterization: This objective is applicable to devices that deliver or elute pharmacologic(s) and/or have degradable components, both of which have become mainstream in the realm of endovascular devices. Before evaluation in vivo, the expectations for how an implant will perform, especially with regard to its pharmacokinetics and/or degradation, should be predictable based on in vitro evaluation. The objective of in vivo characterization then is to verify in vitro expectations, to ensure reproducible kinetics in a biological setting, and to formulate an appropriate program for determining safety preclinically based on this in vivo performance profile over time.
Safety (biocompatibility): It is imperative to confirm that the response of a living biological system to the device is congruent with the safety expected in the clinical setting for which the device is intended. Thus, safety studies should be conducted with procedures and outcomes which mimic those of the clinical setting. Further, for devices that deliver or elute pharmacologic(s), safety assessment includes dose range evaluation to establish subtherapeutic to toxic levels. In alignment with the replacement, reduction, refinement (3Rs) for the ethical treatment of animals, the safety evaluation, if intended for regulatory submission on the final product, should ideally be designed and executed with the intent of also satisfying biocompatibility requirements in accordance with International Organization for Standardization (ISO) 10993 (e.g., subparts 4 [hemocompatibility], 6 [local effects], and 11 [systemic toxicity]; ISO Technical Committee 2018).
Proof of concept or “efficacy”: This third objective is a catchall for evaluations outside of the realm of studies intended for regulatory submissions. Whereas the preceding objectives follow the path of “bench to bedside,” studies with this objective often assume a reverse route from “bedside to bench” to validate clinical observations, to offer insight into the pathobiology of tissue healing following injury and implant, and/or to determine the potential clinical efficacy of a device relative to another (Suzuki, Yeung, and Ikeno 2008b). For the latter regarding efficacy, the end points, such as the reduction of neointimal (NI) hyperplasia or rate of endothelialization and maturation, should be specific to hone the means of evaluation.
Study Design
Contrasting with pharmaceuticals, two aspects govern the effectiveness of a preclinical study for the evaluation of a medical device: time and technique. In contrast to a pharmaceutical agent in which the biological response is dose dependent, the response to a medical device is time dependent (Schuh 2008). Time points for the evaluation of safety should align to the critical phases of healing and thus are relatively standardized. However, many interventional devices for occlusive arterial disease also include biologically active and/or degradable components, rendering both time and dose as relevant factors to be considered (Table 1). The goal is to be able to predict accurately the response of the tissue around the implant as it reaches quiescence.
Recommended Sample Numbers and Time Points for In Vivo Characterization and Safety Evaluation of Stents, Scaffolds, and Balloons.

a Minimum recommendation to satisfy International Organization for Standardization (ISO) 10993-11 requirement for systemic biocompatibility (ISO Technical Committee, 2006).
As to technique, appropriate use and implantation govern the outcome of a study. Improper device sizing (length, diameter), inaccurate placement, and over or under expansion can negatively impact a study’s outcomes. Alignment with the intended clinical use is a critical component in the preclinical evaluation of these interventional devices. Dual antiplatelet medication and implant procedures, including the evaluation of both single and overlapping devices, should match those of the intended clinical use. Further, the design of the preclinical program for the evaluation of a stent, scaffold, or balloon should include not only evaluation of the device intended for clinical use but should also include the evaluation of devices using the highest drug dose/polymer load (e.g., 3–10× greater than the drug dose density of the final product) to establish a reasonable safety margin for clinical use (Schwartz et al. 2008).
The design of a preclinical program for a device is a holistic process that considers not only safety but also pharmacokinetics and degradation, as applicable. The input of the pathologist in the assessment of these devices is on safety; thus, the remainder of this review will focus on the methodology applied in the determination of safety.
Determining Safety of Endovascular Devices
Three main tiers contribute to the determination of safety (Table 2), and within these tiers are a multitude of parameters to be assessed. These three tiers include in vivo imaging, systemic safety, and the vessel response as assessed by histology and scanning electron microscopy (SEM).
Tiers of Safety Assessment of Endovascular Devices.

Note. CBC = complete blood count; CT = computed tomography; EEL = external elastic lamina; IEL = internal elastic lamina; IVUS = intravascular ultrasound; NI = neointima(l); OCT = optical coherence tomography; QCA = quantitative coronary angiography; SEM = scanning electron microscopy; TIMI = thrombolysis in myocardial infarction.
In Vivo Imaging
As angiography is used in the delivery, implantation, and follow-up assessment of endovascular devices in the clinical setting, angiographic end points in preclinical studies are easily obtained and provide the most clinically relevant measure of device safety (Figure 1). As listed in Table 2, angiography is used in qualitatively assessing device migration, aneurysm, vessel dissection, and blood flow (thrombolysis in myocardial infarction flow), and relevant angiographic parameters include lumen diameter, device: artery ratio, late loss, and percent diameter stenosis.
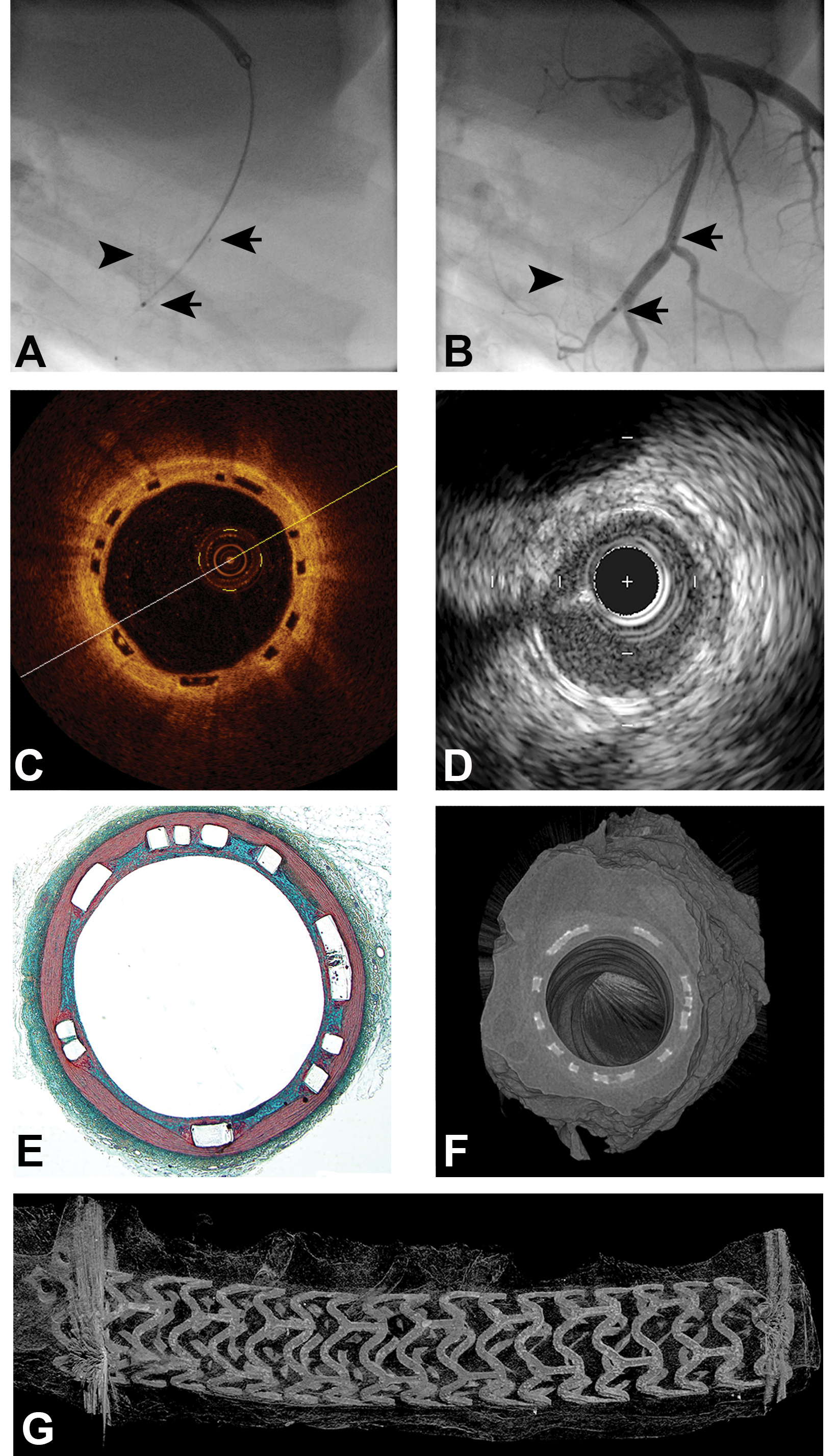
Representative images from porcine coronary artery implanted 28 days prior with bioresorbable scaffolds (BRS) to illustrate modalities used in the evaluation of safety of endovascular devices. (A) Angiography. Arrowhead depicts location of implanted metallic stent faintly visible in the right coronary artery. Arrows identify the location of the proximal and distal metallic markers of a BRS along the length of the guidewire in the left anterior descending artery. (B) Angiography. Arrow depicts location of implanted metallic stent faintly visible in the right coronary artery. Arrowheads identify the location of the proximal and distal metallic markers of a BRS. Contrast medium demonstrates patency of the BRS implanted artery. (C) Optical coherence tomography. Due to their polymeric nature, struts of BRS do not reflect light and therefore appear as clear (black) rhomboids. (D) Intravascular ultrasound. BRS struts are visualized as brighter foci distributed circumferentially around the arterial wall. (E) Histology, Movat’s pentachrome, original objective 2×. BRS-implanted artery has circumferential apposition of struts to the internal elastic lamina and complete neointimal coverage. Minor amount of fibrin around struts is a common observation in relation to drug-eluting devices at this 28-day time point. (F) Cross-sectional microCT. Arterial cross section appears analogous to that depicted by light microscopy with circumferential placement of struts and complete neointimal coverage. (G) Longitudinal microCT. Longitudinal uniformity of expansion and inferences regarding retained integrity of the polymeric BRS struts can be made through microCT. Blooming artifact created by proximal and distal metallic markers obscures visualization of scaffold ends.
Optical coherence tomography (OCT) and intravascular ultrasound (IVUS), two imaging modalities based on light and sound reflectance, respectively, are becoming increasingly used in the in vivo assessment of interventional devices. OCT, used most often in the coronary setting because of its limited depth of tissue penetration, has a high resolution (10 µm) that allows for a preliminary assessment of vessel metrics, tissue features, and vessel response (Figure 1; Giulio and Vasile 2008; Hamdan et al. 2012; Suzuki et al. 2008a; Won et al. 2014). In contrast, the sound-based imaging of IVUS offers lower resolution (>100 µm) but a greater depth of tissue penetration (Figure 1
In addition to in vivo metrics, research is ongoing to create algorithms for both OCT and IVUS to allow for three-dimensional rendering and for characterization of tissue features based on their light or sound reflectance, respectively (Malle et al. 2013; Garcia-Garcia, Costa, and Serruys 2010; Toutouzas et al. 2015). Together, with continued characterization and correlation to histological outcomes, OCT and IVUS may one day be able to replace, at least in part, the requirement for interim termination of animals, aligning with the 3Rs of animal research (Gonzalo et al. 2009; Murata et al. 2010; Nakatani et al. 2016). Furthermore, relying more heavily on in vivo imaging as opposed to histological end points may facilitate more rapid turnaround for translating preclinical results to clinical use and outcomes. Of note, however, with both OCT and IVUS, the passage of guidewires and imaging catheters can result in endothelial denudation within the region of interest and may therefore impact perceived endothelialization assessed by light microscopy or SEM at termination.
While quantitative coronary angiography, OCT, and IVUS are invasive means of in vivo imaging used at implant and at study termination, high-resolution radiography and microCT are two additional imaging modalities employed after explant of endovascular implants that allow for detailed assessment of the implant features relative to the surrounding tissue. High resolution radiography is mainly applicable only to metal stents, whereas microCT can be used for either metallic or polymeric implants and offers high resolution for the assessment of implant morphology and integrity (Figure 1). Relative to BRS, which is designed to acquire structural discontinuities over time, microCT can provide insight into this process and provide assurance that the acquired discontinuities occur at a time when there is adequate tissue coverage to avoid thrombosis and/or distal embolization.
Systemic Safety
The maintenance of health and well-being of animals through the duration of the testing period provides the first insight into a device’s safety. Morbidity and mortality in a preclinical study should be minimal and well-documented as to whether or not it is related to the device being tested (FDA 2010; Perkins 2010; Schwartz, Edelman, Carter et al. 2002, 2004). Evaluation of weight; body temperature; clinical signs; serum chemistry for liver, renal, or systemic imbalances; and hematology (complete blood count [CBC]) with comparison of each of these from baseline at implantation to termination ensures the maintained health of an animal over the course of a study. Complete necropsy with gross and histological assessments of the target site, downstream tissues (e.g., myocardium, skeletal muscle), and organs that may be impacted by the biological activity of the device (e.g., by degradants or eluted bioactive agents) is essential in this systemic evaluation. Additionally, organs related to clinical signs at the time of termination should be evaluated, such as the brain should neurological signs be observed. Organ weights may be required to help satisfy certain requirements for establishing systemic biocompatibility (ISO Technical Committee 2006).
As indicated, the effects related to the implantation procedure and/or device on downstream tissues should be assessed as part of a thorough evaluation of systemic safety (FDA 2010). For defined adjacent tissues and downstream organs, such as the heart, kidney, and brain, these should be evaluated grossly with bread loafing at routine (∼1 cm) intervals to ensure adequate gross evaluation; sampling for histological evaluation should include any tissues subjacent to the implanted region as well as representative samples throughout the distribution of the implanted artery (Schwartz, Edelman, Carter et al. 2002). Similarly, for devices evaluated in the lower limbs (e.g., iliac and femoral arteries), downstream skeletal muscle and the highly vascular coronary band should have representative samples collected and evaluated (Kolodgie et al. 2016). Pertinent observations in these tissues include infarction, replacement fibrosis, inflammatory infiltrates, vascular lesions (e.g., fibrinoid necrosis), and/or evidence of thrombo-embolization or embolization of material related to accessory devices, the test device, or both. Depending on the device and accessory devices used during implantation, these materials can vary from crystallized drug and nonbirefringent hydrophilic coating to birefringent polymeric fragments (Babcock et al. 2013). Polarized light microscopy thus is valuable in the evaluation of these downstream tissues as well as of other peripheral, highly vascular organs such as the lungs. Foreign materials as these, often bearing clinical significance, have been increasingly reported in the literature (Chopra et al. 2017; Grundeken et al. 2015; Mehta et al. 2010; Stanley et al. 2016; El-Najjar and Robinson 2012), and therefore, from a preclinical perspective, these observations should be well-documented. In preclinical studies, the clinical significance of observations made in downstream tissues cannot be concretely defined and depends on clinical symptoms and the frequency, distribution, and severity of associated lesions. Thus, a preclinical study should be designed to ensure that such lesions, when observed, can be attributed to either the control or test device.
Vascular Response: Histology and SEM
Proper handling and processing of treated arteries for histological evaluation is imperative as the quantitative analysis of the residual lumen, NI proliferation, and vessel remodeling are important aspects of the safety assessment of endovascular devices. At termination, arteries are ideally perfusion fixed under physiologic pressure to ensure lumen expansion. In addition to the treated segment proper, proximal and distal naive segments (within 5 mm immediately adjacent to the implanted segment) should be collected for assessment of regional effects of the treatment or device. As opposed to metal stents, in which arterial dimensions are largely constrained by the metallic implant, arteries treated with BA/DCB or in the advanced phases of degradation following implant of a BRS may be more challenging to fix in their expanded state as the contracture induced by fixation and dehydration during processing can be exacerbated in these arteries. With unconstrained arteries, perfusion fixation under physiologic pressure should be performed to assist in maintaining circular cross sections; additionally, the perimeter measurements may be applied in order to derive area measurements in these tissues (Yazdani et al. 2014). Hence, as indicated previously, in vivo imaging by angiography or more preferably OCT or IVUS can be a valuable alternative to standard histomorphometry.
As the preponderance of endovascular prosthetics are metal-based, conventionally arteries are embedded in a polymer resin and are either ground, as in the case of nitinol stents, or thin-sectioned on a rotary microtome using a tungsten carbide blade. Arteries treated by BA (whether with or without drug) and arteries implanted with softer polymeric devices may be suitable for paraffin embedding. A review of polymer resins commonly used and considerations for histological preparation of these and other devices is available (Rousselle and Wicks 2008). As for any implanted device, the integrity of the tissue–device interface impacts the value of the specimen. Routine stains used for histological evaluation include hematoxylin and eosin for morphology and stains for elucidation of the elastin-containing layers for histomorphometrics, such as Movat’s pentachrome, elastin Verhoeff-Van Gieson, or elastic Masson’s trichrome (Figure 2). Additional histochemical stains and immunohistochemistry may also serve useful for specific assessments.
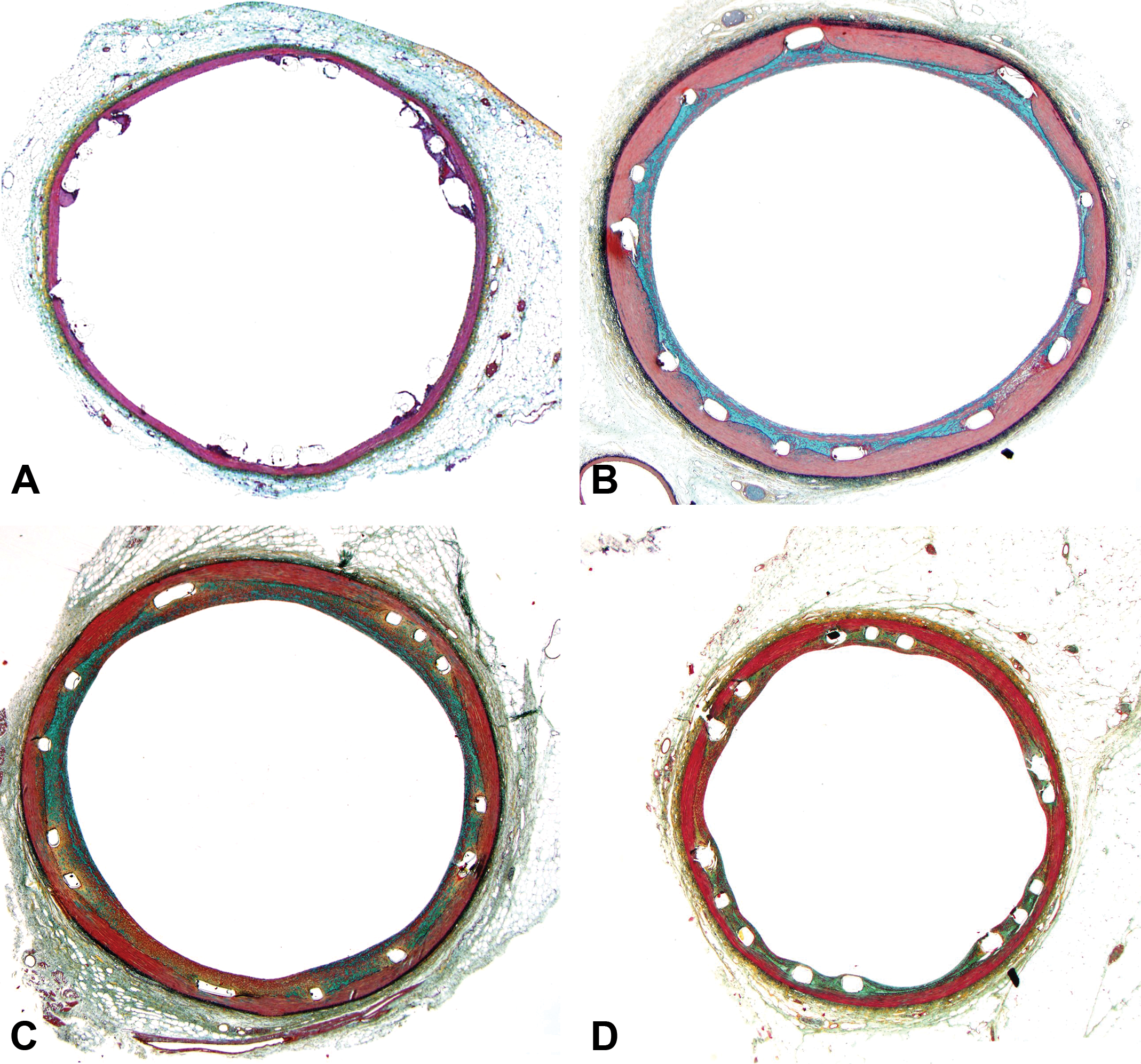
Representative photomicrographs of porcine coronary arteries after implantation of drug-eluting stent illustrating the progression of normal healing. (A)–(D) Movat’s pentachrome, original objective 2×. (E)–(H) Hematoxylin and eosin, original objective 20×. (A) and (E). Three days. Struts are covered by a thin layer of fibrin with intermixed leukocytes including neutrophils, macrophages, and multinucleated giant cells. (B) and (F) Twenty-eight days. Struts are sequestered by a smooth muscle cell rich neointima dispersed by proteoglycan-based extracellular matrix. Small amounts of fibrin are localized immediately around the struts, and few multinucleated giant cells are in juxtaposition to the strut. (C) and (G). Six months. The neointima sequestering struts has matured to become increasingly dense with smooth muscle cells. (D) and (H) Twelve months. Relative to proceeding time points, the neointima has continued to mature and contract to form a thin layer over struts.
Regardless of the anatomical location, there is a finite repertoire of vascular responses to an intervention (injury) or implanted device (Table 3). Thus, the histological evaluation can be relatively straightforward, though the full interpretation of relevant observations requires an understanding of the device materials, implantation procedures, kinetics of the bioactive agents and/or polymers, and performance characteristics of the endovascular device. Add to that the background responses that can occur in the porcine model, and the interpretation of device safety can become convoluted.
Essential Histomorphological Parameters in the Evaluation of Safety.

Note. EEL = external elastic lamina; IEL = internal elastic lamina; SMC = smooth muscle cell.
Histomorphometry
Histomorphometry by computerized planimetry is used for the measurement of specific anatomical features of the artery including the area within the EEL and internal elastic lamina (IEL) and the lumen area. Medial area, NI area, and percent area stenosis are then calculated from these parameters. Other relevant parameters include NI thickness (from adluminal aspect of strut/IEL to lumen surface) and radial gap width (separation between IEL and abluminal aspect of strut). By comparison of these outcomes between the control and the test article, an indication regarding potential efficacy can be obtained. Further, comparison of these metrics over time can provide insight into vessel remodeling that can occur with BRS and BA/DCB. Importantly, while comparison of vessel metrics between the reference vessel and the treated segment is a practical means for determining parameters such as percent stenosis and remodeling index (ratio of treated segment EEL: reference segment EEL) by in vivo imaging, caution should be taken in using reference vessel metrics obtained by histology due to the artifacts that can occur during excision and handling of the heart and implanted vessels and during processing due to higher tissue shrinkage in the reference (naive) vessel segment (Dobrin 1996).
Histomorphology
In the morphological evaluation of the vascular response, it is imperative to develop a rigorous, semiquantitative means for assessing the critical parameters that reflect on the safety of a device. Whether by a cross-sectional, quadrant-based, or strut-based approach, a multitude of scoring conventions have been used and published (de Prado et al. 2013; Otsuka et al. 2014; Wilson et al. 2007; Buszman et al. 2014). The methods for scoring should be clearly stated, consistent between control and test devices and throughout time points, and reproducible to ensure suitable interpretation for the determination of safety. Provided in Table 3 is an overview of these critical parameters and considerations in their assessment.
In the context of safety, histomorphometry and histomorphology, while seemingly distinct parameters, should be interpreted in coordination. For example, radioactive stents or stents eluting cytotoxic drugs (e.g., Actinomycin D) demonstrated morphometrical efficacy in reducing NI proliferation in the short term; however, morphologically, treated arteries had positive remodeling (radial gap), excess fibrin, medial SMC loss, and thrombus formation in association with these seemingly positive morphometric outcomes (Figure 3). Longer term follow-up in porcine arteries and clinical outcomes demonstrated that the vessel injury, mural necrosis, and adverse positive remodeling induced by these treatments were later compensated for by heightened NI proliferation and (re)stenosis (Schwartz, Chronos, and Virmani 2004; Kaluza et al. 2001; Virmani et al. 1998).
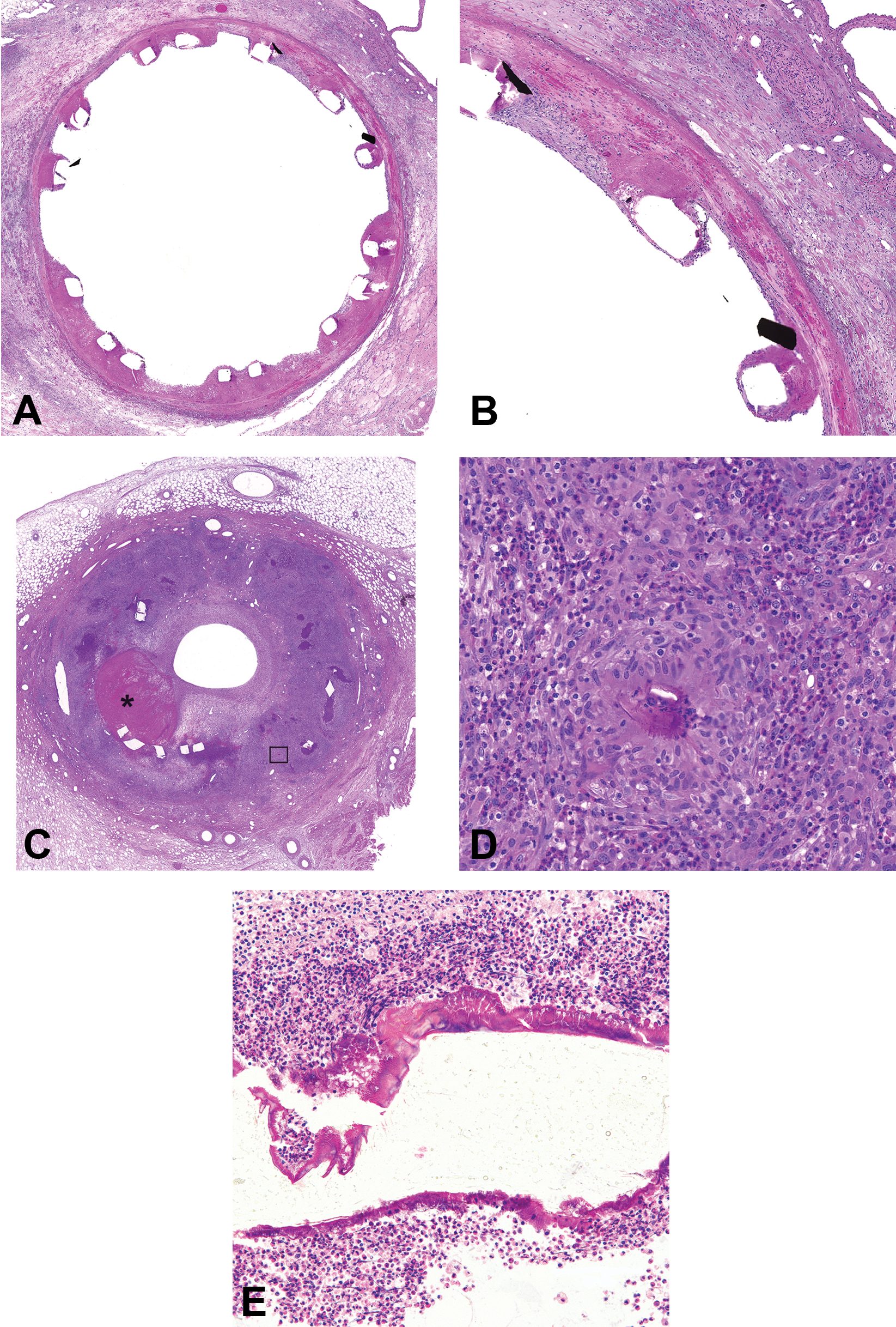
Representative images from porcine coronary arteries after implantation of endovascular devices. (A) Twenty-eight days after implant with a metallic stent eluting paclitaxel, a cytotoxic antiproliferative agent. Struts are surrounded by fibrin and are malapposed from the arterial wall due to positive remodeling and medial smooth muscle cell loss. Hematoxylin and eosin. (B) Higher magnification of (A) depicting loss of medial smooth muscle cells with replacement by fibrin and extravasated erythrocytes. Hematoxylin and eosin. (C) Six months after implant with bioresorbable scaffolds, there is marked lumen narrowing due to neointimal proliferation, residual thrombus (*), and effacement of the arterial wall due to marked eosinophilic granulomatous inflammation. Hematoxylin and eosin. (D) Higher magnification of the boxed region (C) in which Splendore-Hoeppli material is observed in association with migrated polymer coating. Hematoxylin and eosin. (E) Bioresorbable scaffolds strut encompassed by Splendore-Hoeppli material and degenerate granulocytes 188 days after implant. Hematoxylin and eosin.
In the porcine model, a close correlation has been demonstrated between vessel injury, inflammation, and (re)stenosis (Kornowski et al. 1998, 1999). However, this model is predisposed to develop granulomatous inflammation to vascular implants, which is often indiscriminate of the device and may have eosinophilic involvement (Perkins 2010; Schwartz et al. 2008; Wilson et al. 2009; Wilson 2011). With the presence of inert foreign materials, though rare, Splendore-Hoeppli can be observed in association with these eosinophilic infiltrates (Figure 3; Hussein 2008). Therefore, interpretations should take these host-related responses into account, understanding that these responses may be model-related. Hence, for studies conducted in swine, it is important to include both a control and test device in each animal (Schwartz et al. 2008; Perkins 2010).
SEM
Ultrastructural evaluation of arteries by SEM is a standard means for assessing endothelialization and the surface morphology to ensure these are consistent with a safe vascular response. Endothelial cell pavementing, alignment, and morphology; patency of side branches; and the presence and numbers of adherent platelets or leukocytes are important parameters that can be qualitatively and quantitatively assessed (Schwartz et al. 2008; de Prado et al. 2013).
Assessing the Potential for Clinical Efficacy
There is no perfect preclinical model that reflects clinical outcomes, though the commensurate nature of vessel responses to injury and implantation in the preclinical setting provides the pathologist reasonable assurance as to a device’s safety to enter into clinical use (Perkins 2010; FDA 2010; Virmani et al. 2003).
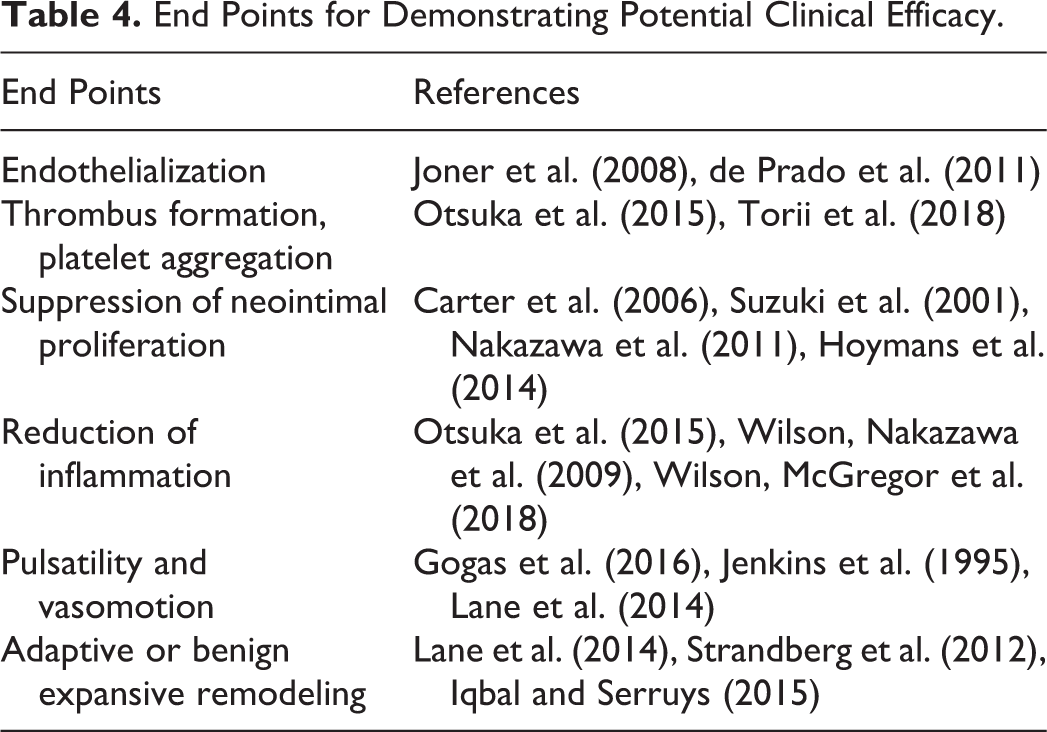
Taking this a step further, it may be possible, through a well-designed and targeted approach, to preclinically investigate end points that may roughly translate to clinical efficacy (FDA 2010). Provided in Table 4 is a sampling of some of these targeted end points and studies designed toward demonstrating potential clinical efficacy. Still, precisely which end points correlate best between preclinical outcomes with clinical efficacy is unclear. Take, for example, the seemingly straightforward outcome of the reduction of NI proliferation or (re)stenosis. In the porcine model, this potential efficacy end point is often absent or lost in studies greater than 30 days for DES and DCBs when compared to nondrug eluting controls, despite these devices clearly demonstrating clinical efficacy (Perkins 2010; Schwartz et al. 2008; Wilson et al. 2007; Yazdani et al. 2014; Tepe et al. 2014; Gray and Granada 2010).
End Points for Demonstrating Potential Clinical Efficacy.
There are several specialized preclinical models that exist with the intent of simulating clinical conditions in order to allow better prediction of clinical outcomes. The comparison of vascular healing between standard preclinical models and diseased human coronary arteries has been well described (Virmani et al. 2003; Nakazawa et al. 2007; Perkins 2010; Schwartz et al. 2004). However, human atherosclerosis is complex with many variables and related disease conditions. On a lesion basis, there are simple lesions to heavily calcified lesions, lesions located in the left main coronary artery, CTOs, and vulnerable plaque; and on a patient basis, there are comorbidities such as obesity, metabolic syndrome, hypertension, diabetes, and chronic renal failure. In relation to devices for treating atherosclerotic occlusions, the complexity of the clinical setting cannot be matched by any one preclinical model. Specialized preclinical models tend to be costly, of limited availability, not sufficiently characterized, and/or harbor too much biologic variability for routine use in safety evaluation (Perkins 2017; Iqbal et al. 2016). Despite these limitations though, these models have proven valuable in enhancing our understanding of vascular pathophysiology and disease conditions as they relate to atherosclerosis and occlusive arterial disease. Although specialized models are not yet for mainstream use in safety assessment (Schwartz et al. 2008), their continued development and characterization will benefit device development and testing.
A final consideration regarding the translation of preclinical observations to clinical outcomes relates specifically to BRS, whereby the degradation rate of a device is independent of the species or disease state of the tissue (Nakatani et al. 2016; Campos et al. 2015; Waksman et al. 2017). Due to the difference in healing rates between the normal, juvenile arteries of the standard (porcine) preclinical model versus the aged, diseased arteries of humans (Virmani et al. 2003), certain end points indicative of vascular restoration, such as adaptive (expansive) remodeling and vasomotility, are likely to be delayed in the clinical setting.
Conclusion
Preclinical models have contributed to our understanding of restenosis and enabled continuous improvement in and development of interventional treatments for coronary and peripheral occlusive arterial disease. Relative to its anatomical and physiologic similarities to humans, swine have served as the preferred species for use in the evaluation of interventional devices. While the porcine species has served well in the evaluation of proof of concept, confirmation of safety, and characterization of performance of these devices, there is still much to learn, with research ongoing in the assessment of potential efficacy.
This review has focused on the preclinical evaluation of interventional devices designed for the treatment of occlusive arterial disease, though the realm of interventional devices for the treatment of other cardiovascular maladies is ever expanding. From valves and valve repair devices, septal and atrial appendage occluders, pacemakers, pacing and defibrillator leads, vessel closure devices, to heart pumps, interventional cardiology is an ever-expanding field in need of preclinical assessment, and the trained pathologist, to ensure safety for clinical use.
Footnotes
Author Contribution
All authors (LP, MR) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
