Abstract
Surgical and laparoscopic implantation of mesh devices is on the rise for a variety of applications. The complexity and range of evolving mesh designs calls for consistent and detailed pathologic evaluation in determining host responses and assessing overall safety. This review addresses the components of evaluation of mesh implants in animal models, with emphasis on histologic parameters, semiquantitative scoring matrices, and morphometric analyses that have been specifically adapted to this class of implants. Necropsy assessment should include implant persistence, architecture, and associated host responses such as exudation and adhesions. Microscopic evaluation should focus on primary relevant responses such as bioresorption, integration/tissue ingrowth, neovascularization, and inflammation. Selection of the best means of processing and evaluation can be complicated, as meshes may include one or more biologic components (e.g., collagen), synthetic polymer fibers, coatings, and other molecules. The architecture of some meshes can influence tissue responses and complicate sampling, sectioning, and evaluation. Recognition of specific study objectives and knowledge of anticipated responses helps to determine the appropriate histologic or immunochemical stains, while understanding of mesh composition and anticipated persistence in tissue determines the suitability of paraffin or resin embedding, and both guide the evaluation of mesh devices in the preclinical setting.
Use of surgically implanted meshes dates to the late 19th century, but it was following the mid-20th-century boom in manufacture of synthetic polymers that meshes underwent increased clinical usage to the point that today procedures involving mesh implantation are some of the most common surgeries performed worldwide (Baylón et al. 2017; Köckerling et al. 2018). Clinical applications for mesh implants include surgical or laparoscopic approaches in a variety of locations, including breast reconstruction, abdominal/pelvic hernia repair, and hiatal hernia repair (Baylón et al. 2017; Todros, Pavan, and Natali 2016). The evolving designs within this complex group of implants require a consistent and detailed pathologic evaluation which is critical in determining host responses and to assess overall safety. Yet, regardless of the particular device and circumstances of implantation, the goals for determining overall safety are fundamentally similar.
The pathologist plays a key role in evaluating biological responses to implants, by characterizing and quantifying such responses grossly and microscopically. It is also critical, though, for the pathologist to place such responses in context with regard to their pathophysiologic significance. This is where an experienced investigative team is critical. Fundamental knowledge of the expected biologic inflammatory, repair, and remodeling response in a given model is necessary, as well as anticipation of typical intrinsic foreign body responses, understanding of the gross and microscopic appearance of various implant components at different times postimplantation, and the type and degree of changes that are anticipated and acceptable at a given time point.
Regardless of mesh implant technique (e.g., open/surgical or laparoscopic) or location (e.g., abdominal, preperitoneal, subcutaneous, and hiatal), there are particular features that should be evaluated (Deeken, Faucher, and Matthews 2011; Müller-Stich et al. 2014). Depending on the feature, both semiquantitative and morphometric approaches may be possible. Specific parameters important for pathologic evaluation include the presence, degree, and characterization of:
fibrosis, which may include interstitial/dissecting fibrosis and presence of fibrous capsule;
inflammation, including amount, cellular composition and distribution, and distance from material/tissue interface;
degree and composition of tissue ingrowth, particularly into porous, woven, and absorbable implant materials. This may include fibroblasts, collagenous tissue, blood vessels, adipocytes, and inflammatory cells (Trandafir, Popa, and Vasile 2017; Brown and Badylak 2013);
degenerative or other changes in peri-implant tissue including necrosis, vascularization, fatty infiltration, mineralization, and osseous metaplasia; and
implant material-specific parameters such as integrity, fragmentation, debris presence, and location of remnants of degraded material.
The overarching goal of this analytic approach is to determine the presence of favorable responses (e.g., appropriate implant bioresorption or integration into host tissue) compared to complications and adverse effects (i.e., postsurgical adhesions, hernia recurrence, and mesh rejection) to arrive at a conclusion regarding biocompatibility or safety of an implant in a specific preclinical setting.
Animal models for mesh implant research include both large (swine, sheep) and small (rat, rabbit) animal species (Zhu, Schuster, and Klinge 2015; Müller-Stich et al. 2014; Deeken, Faucher, and Matthews 2011). With regard to general study design, it is ideal to compare similar rather than disparate mesh designs/compositions, both from a pathophysiology perspective, and because it may adequately blind pathologists to treatment groups in instances of highly distinct implants. Study durations are usually determined in part by the anticipated persistence of meshes following implantation and are influenced by experience with predicate devices.
Mesh Implant Biomaterials and Architecture
The intent of this article is not to serve as an overview of biomaterials science, to review specific commercially available mesh products, or to address relative advantages or disadvantages of mesh types in various applications. Nevertheless, understanding of the scope of available biomaterials is important in giving the pathologist perspective on how to implement gross and histologic matrices for preclinical evaluation of implants. There is an ever-evolving series of synthetic and biologic materials from which mesh implants are manufactured (Baylón et al. 2017; Zhu, Schuster, and Klinge 2015), and each material may provide its own set of challenges. Broadly speaking, several classes of mesh implant are routinely used for a range of clinical applications, and constituents include synthetic nonabsorbable or synthetic absorbable materials, and biologics such as modified collagen (of porcine or human dermal or visceral origin; Baylón et al. 2017; Köckerling et al. 2018). Permanent or absorbable barrier constituents may also be present.
The most common primary implant components to date include synthetic nonabsorbable polypropylene (PP), polyester, and polytetrafluorethylene; synthetic absorbable polylactic-co-glycolic acid; polyhydroxybutyrate or related polymers; biologic origin material such as dermal and visceral origin collagen; and sometimes additional components such as silk, hydrogel, oxidized cellulose, and omega-3 fatty acids (Trandafir, Popa, and Vasile 2017; Zhu, Schuster, and Klinge 2015; Müller-Stich et al. 2014; Yang et al. 2017; Faucher et al. 2015). Component fibers may be monofilament or polyfilament, and some devices are composite and include more than one type of material.
Above and beyond primary material composition, higher level architectural features including layering, weave/knit, porosity, thickness, and fenestration may introduce even more complexity to implants (Zhu, Schuster, and Klinge 2015). Fibers can be united to form fabrics in a number of ways. They may be knitted (with fibers stitched/interlocked together), warp-knitted (a specific knit allowing cutting/trimming without losing integrity), woven (two sets of fibers interlaced at right angles), or nonwoven (an irregular, amorphous tangle), with knitted variants allowing greater inherent porosity and elasticity (Zhu, Schuster, and Klinge 2015). Porosity, or the interstitial space between mesh components, can affect tissue infiltration and inflammatory response, with pores over 75 µm or larger causing less inflammation when compared to smaller pores, lower connective tissue response with less scar tissue formation (Zhu, Schuster, and Klinge 2015). Larger pores are also reported to be associated with lower less risk of fistula formation, less mineralization, and less bridging of individual granulomas to form confluent, encapsulating layers (Zhu, Schuster, and Klinge 2015). In addition, implants may include two or more materials, for example, a primary fiber mesh with an anti-adhesive coating, and a third component to hold the other two together. Anti-adhesive/anti-inflammatory coatings, generally designed to bioresorb over a short time frame, may be part of intraabdominal implants (Zhu, Schuster, and Klinge 2015; Trandafir, Popa, and Vasile 2017).
Macroscopic Evaluation
Thorough macroscopic evaluation should be performed at necropsy, with appropriate in situ and ex vivo photographic documentation. Persistence of implant should be noted when possible, and semiquantitative scoring matrices may be used to estimate the percentage of bioresorbable implant present. Folding/wrinkling may be noted, especially for implants on the peritoneal wall. Assessment of implant-related adhesions is particularly important with intraabdominal implants. Any adhesions of omentum or viscera to implants should be noted in situ. A variety of adhesion scoring paradigms exist, with the overarching goals being to record the extent of adhesions (along the implant surface or edges), adhesion tenacity, and the involvement of viscera. Reflecting its normal function in the abdominal cavity, the greater omentum is frequently adhered to the visceral aspect of peritoneal implants. Depending on implant location, among other variables, there can also be involvement of the spleen, liver, stomach, small intestine, and cecum-colon (Figure 1). Tenacity of adhesions can range from fibrinous to fibrous. Adhesions may be extremely loose, to the point of incidentally breaking down during even careful necropsy manipulation, or more durable but still overtly fibrinous and easily separated. Given enough chronicity, adhesions may become fibrous and resistant to separation, either as bands or plaques, and can only be separated via incision. Migration of implants from initial implantation sites (e.g., following disruption of anchor/suture sites) should also be noted, as should any separation or delamination of implant components.
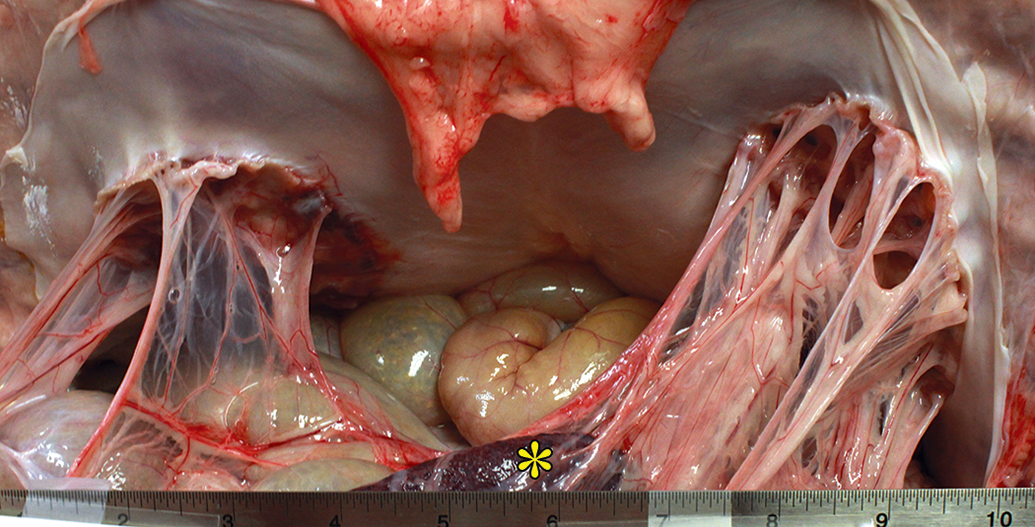
Necropsy view of two mesh implants in situ, along the ventral abdominal parietal peritoneal surface. There are omental adhesions to both, at both the free edges and central surface of the implants. The spleen (asterisk) is indirectly involved in the adhesions at right.
Following documentation of adhesions and appropriate exposure of implants, such as by reflection of the ventral abdominal wall to expose peritoneal implants, and careful preparation of implant surfaces, digital photographs of the implant surfaces may be obtained. The percentage of surface involvement in adhesions may then be evaluated semiquantitatively at necropsy or subsequently using morphometric software on digital images containing an appropriate calibrated ruler (Figure 2). Similar morphometric analysis is commonly performed for quantification of implant contraction compared to nonimplanted devices. Excessive fibrosis should be noted, as well as implant folding, wrinkling, or migration. For esophageal hiatal implants, assessment of localized stricture/stenosis may be performed (Muller-Stich et al. 2014). Other potential findings at necropsy include implant-associated erythema, fibrin, hemorrhage, or hematoma. Any seroma should be noted, especially as lesions can be difficult to thoroughly assess microscopically and may be lost during sectioning/histological processing. Any indication of inflammation such as exudation, abscessation, or granuloma formation should be noted.
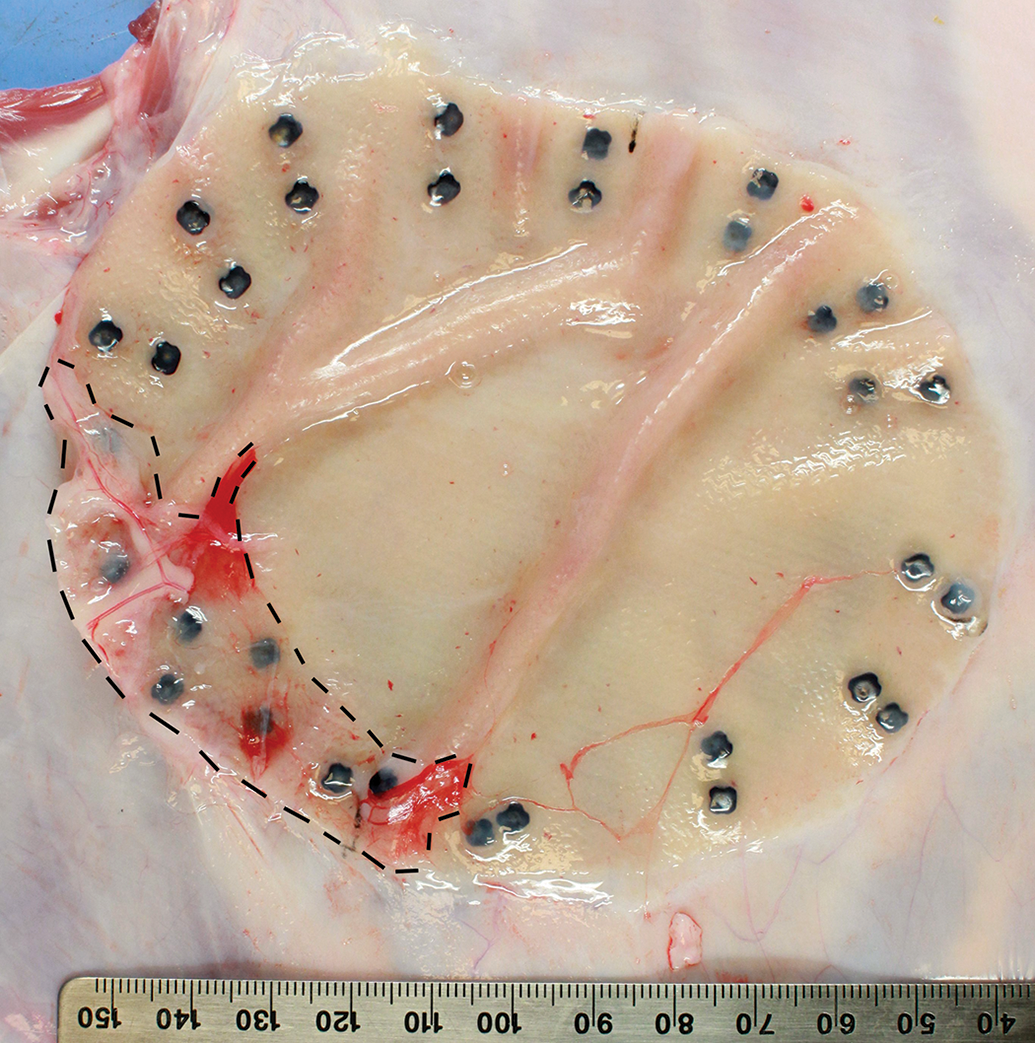
Necropsy view of mesh implant in situ on abdominal wall. Excess/adherent tissue has been dissected away to allow morphometric measurement of area involved in omental adhesions (interrupted line). Overall tissue reaction on the visceral peritoneal implant surface is minimal. Note double row of fixation devices along the implant periphery.
As is the case with histopathologic evaluation, there is no single set of parameters that applies to all preclinical situations, and no single scoring system is uniformly applicable. However, examples of basic scoring matrices for several gross pathology parameters are shown in Table 1.
Macroscopic Scoring Matrices.
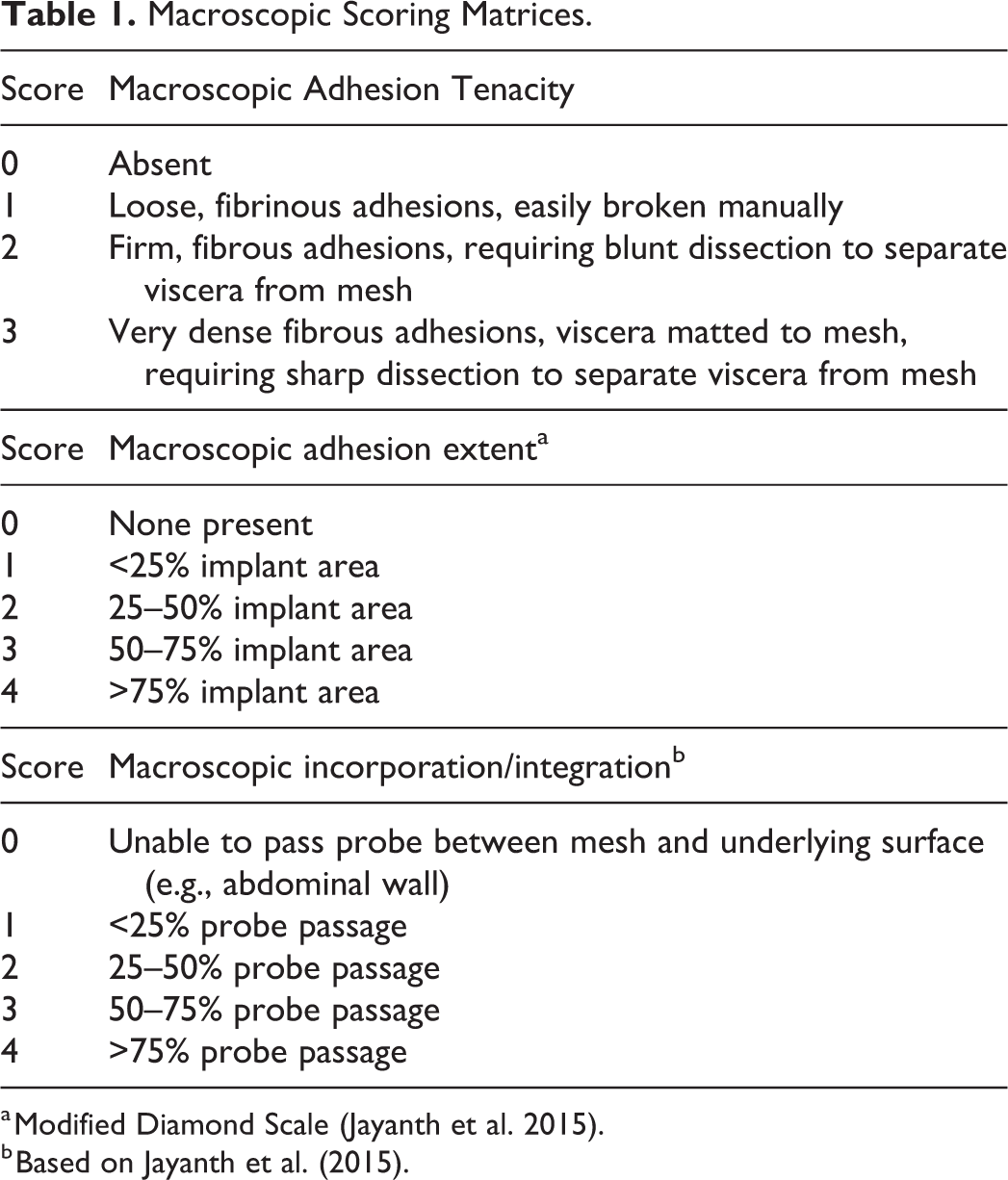
a Modified Diamond Scale (Jayanth et al. 2015).
b Based on Jayanth et al. (2015).
At necropsy or thereafter, implants may be subdivided to generate separate samples for subsequent evaluation including histology, mechanical testing, molecular analysis, and SEM. Trimming notes are helpful in documenting mesh presence (see Figure 3) as well as any other findings that might benefit from histologic evaluation.
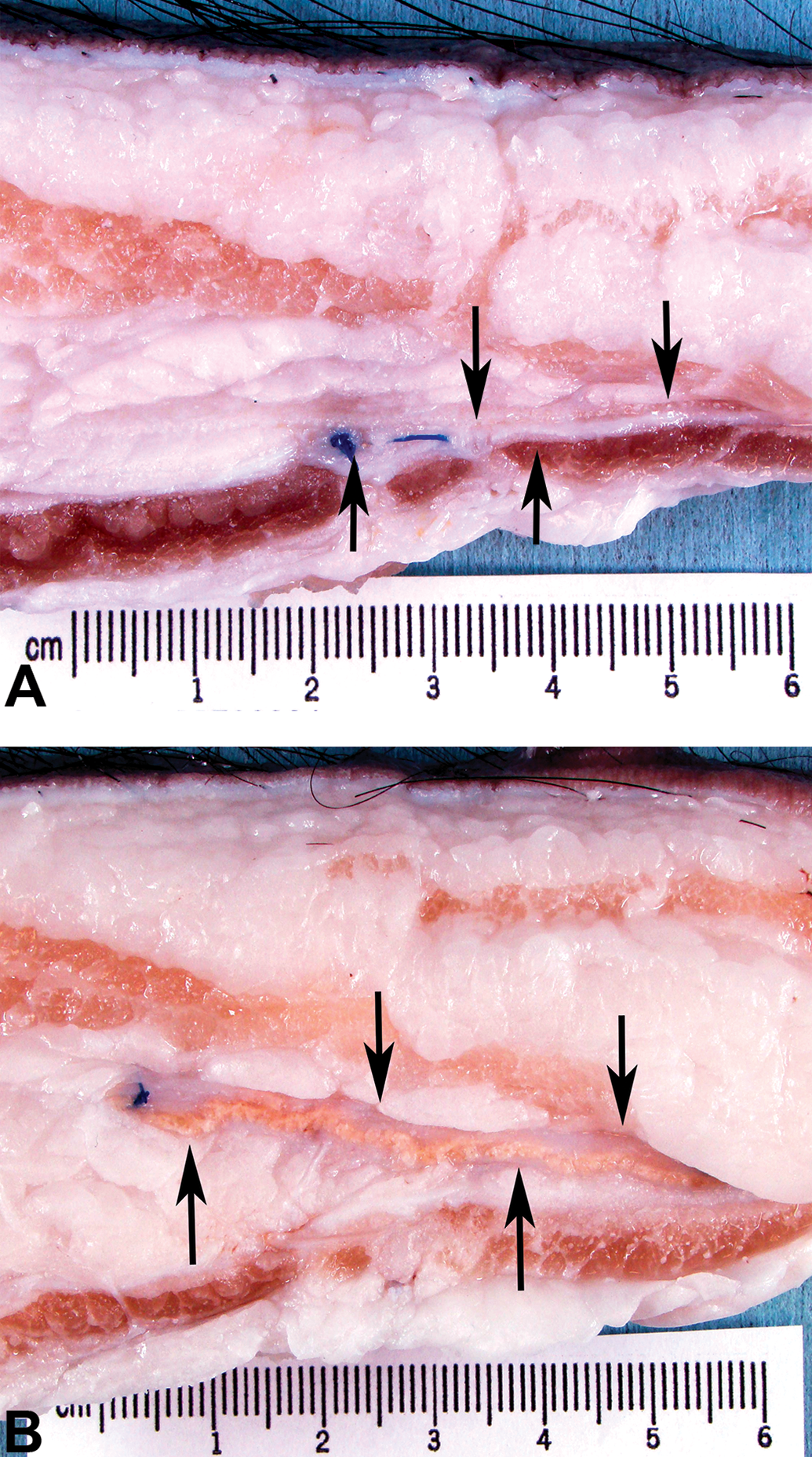
Transverse sections through subcutaneous implants and surrounding tissue at necropsy. Arrows indicate a thin implant deep within the preperitoneal adipose tissue, with the implant in panel A being thin and white and that in panel B being thicker and pink tan.
Thought should be put into the sampling process, and determining the number, size, and relative location of tissue blocks generated for histopathologic analysis. It may be important to be able to visualize implant areas distant from anchor or suture sites, in order to obtain a specific view of tissue–mesh interface away from possibly confounding tissue responses. Conversely, and particularly at chronic time points involving bioresorbable implants, it may be useful to have anchor or suture sites visible as orienting landmarks. In general, there is great value in preserving tissue surrounding, abutting, or adhered to implants, thereby allowing most complete analysis of tissue responses at and transitioning away from implants.
Histopathologic Evaluation—General Considerations
Generation of histologic sections of mesh implants is itself a specialized procedure, the details of which are determined by the physical and chemical characteristics of implant components. Selection of embedding medium may depend on implant durability/pliability, processing options may depend on potential for introducing artifactual changes such as swelling or shrinkage, and responsiveness to sectioning is influenced by factors such as overall implant presence, which may decrease with bioresorption. When in doubt, method development should be performed on unimplanted, postmortem implanted, or explanted devices. Depending on the particular material constituents, resin or paraffin embedding may be appropriate choices, and microtome sectioning is usually sufficient. Transverse sections suffice for most morphologic and morphometric evaluations but generation of en face sections that contain more linear mesh fiber profiles may be called for in some instances, such as evaluation of mesh weave architecture.
Histopathologic analysis relies on H&E staining for overall evaluation of cellular and tissue responses, and collagen-specific stains such as Masson’s trichrome (MT) are not necessary but can be useful and visually striking adjuncts in highlighting fibroproliferative responses at chronic time points, particularly for nonpathologist reviewers. Indirect indication of collagen fiber size and arrangement can be obtained by examination of picrosirius red (PSR)-stained sections, and this may assist with interpretations of connective tissue chronicity and maturity. Thick fibers consistent with more mature collagen, typically type I collagen stain orange-red under polarized light, while thin fibers consistent with less mature collagen, typically type III collagen, stain green (Rich and Whittaker 2005). Immunohistochemical stains may provide information about the type of collagenous response (e.g., collagen types I and III) or cellular response (e.g., CD68 for macrophages, CD31 for endothelium, and smooth muscle actin for myofibroblasts). Assessment of both cell proliferation and cellular apoptosis may also be made (e.g., Ki67 and TUNEL, respectively).
There is no single scoring system that is uniformly applicable across all implant types that will answer all the questions that arise during device development and preclinical evaluation. The fundamental goal of scoring tissue responses, however, remains the comprehensive evaluation of tissue responses with contextual discussion and comparison to predicate devices whenever possible. Specific histopathologic evaluation includes scoring parameters typical of implant safety studies, as well as some that are specific to meshes. Histology should be performed using semiquantitative analysis when possible, consistent with regulatory guidelines (i.e., ISO-10993-6 Annex E). Examples of scoring matrices for selected parameters are presented in Table 2 and shown graphically in Figure 4. Figure 5 illustrates the range of acute to chronic tissue responses for several different implant classes.
Histopathologic Scoring Matrices.
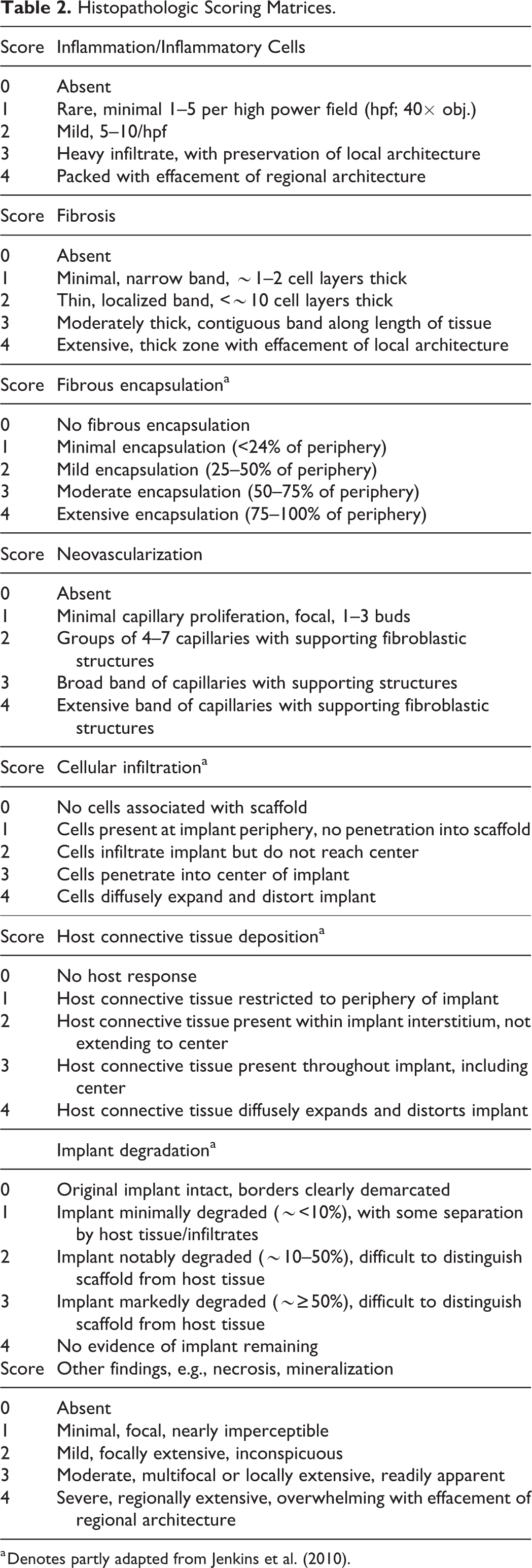
a Denotes partly adapted from Jenkins et al. (2010).
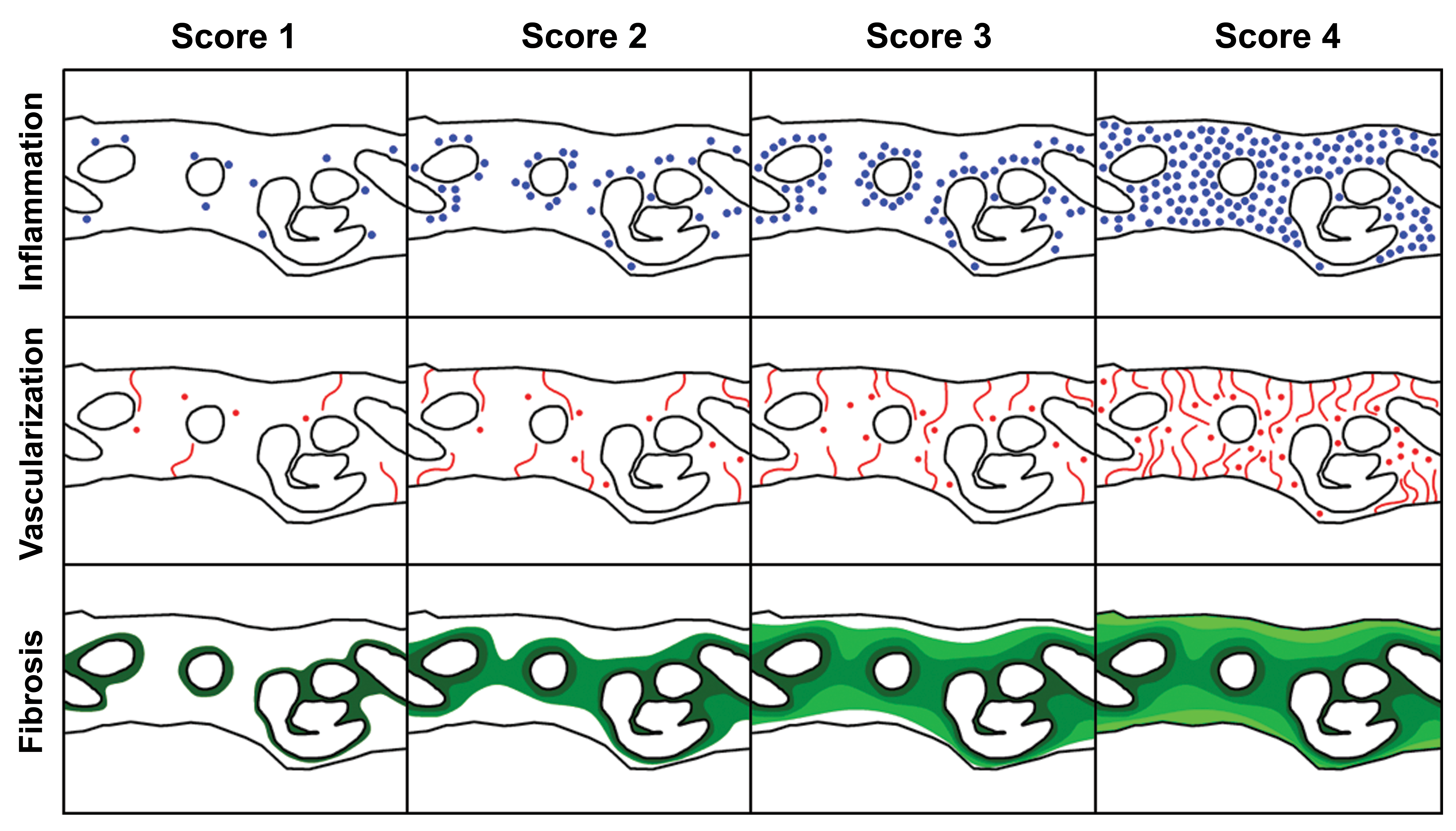
Schematic representations of relationship between degree and distribution of mesh associated tissue responses and corresponding histopathologic scores for inflammation, vascularization, and fibrosis.
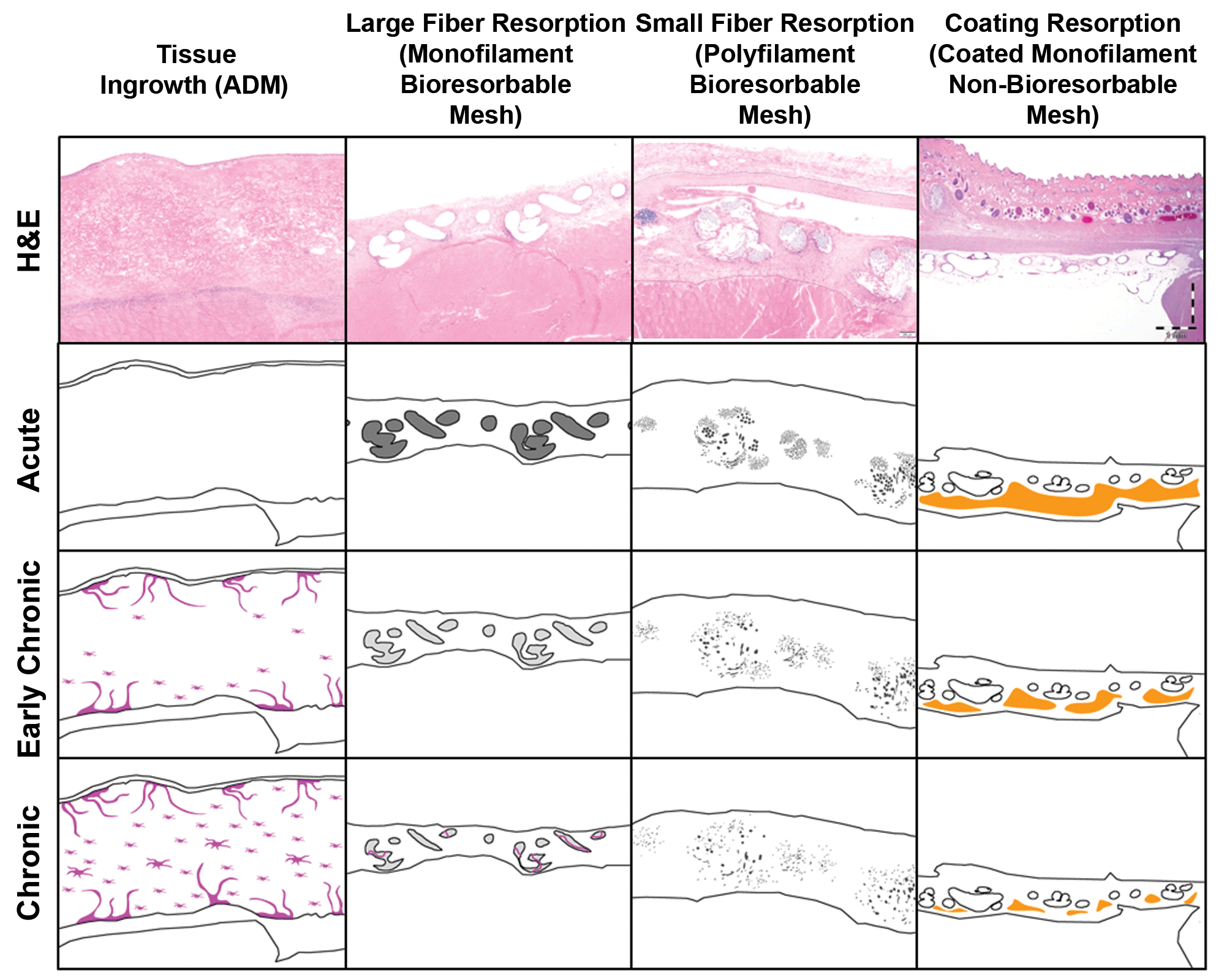
Schematic representations of acute, early chronic and late chronic responses of various types. ADM: acellular dermal matrix.
Regarding specific cellular responses, inflammation is an important parameter, and evaluation should include both the overall degree of infiltrates and the amounts of cellular subtypes (e.g., neutrophils, macrophages, and multinucleated giant cells). The relative locations of inflammatory infiltrates may also provide important information regarding differential responses to various implant components (Brown et al. 2012) and the possibility of incidental, model-related inflammation (such as occurs following incision/disruption of skeletal muscle). Whether inflammatory infiltrates are restricted to the periphery of an implant or extend within an implant may reflect biologic processes of interest. Inflammation spatially unrelated to the implant, and possible model related should be accounted for but in most cases should be kept separate from scores of implant-associated infiltrates. Inflammatory responses to various implant components are shown in Figures 6 and 7.
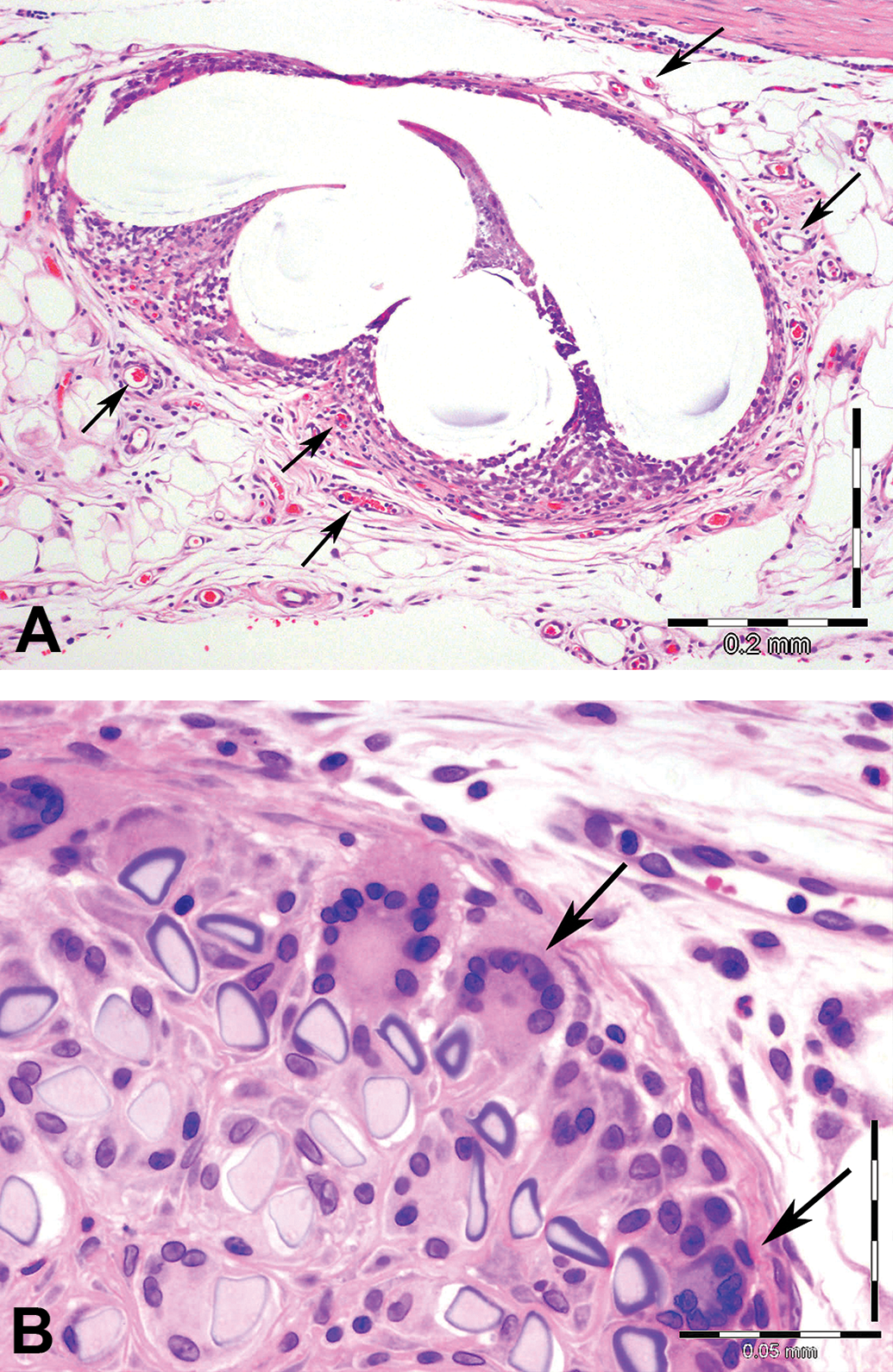
(A) High magnification view of nonbioresorbable monofilament implant showing a localized response around a mesh knot (large white/empty lobulated shape). Capillary profiles (arrows) are present at the periphery, while the fibers are more closely surrounded by few fibroblasts, very sparse collagen, and a mild, mixed inflammatory cell population. The cellular-proteinaceous exudate extends into the space between the fibers, which themselves are completely intact. (B) High magnification view demonstrating typical inflammatory response to a polyfilament bioresorbable implant. Fibers are separated by macrophages and peripherally surrounded by a discrete row of multinucleated giant cells (arrows).
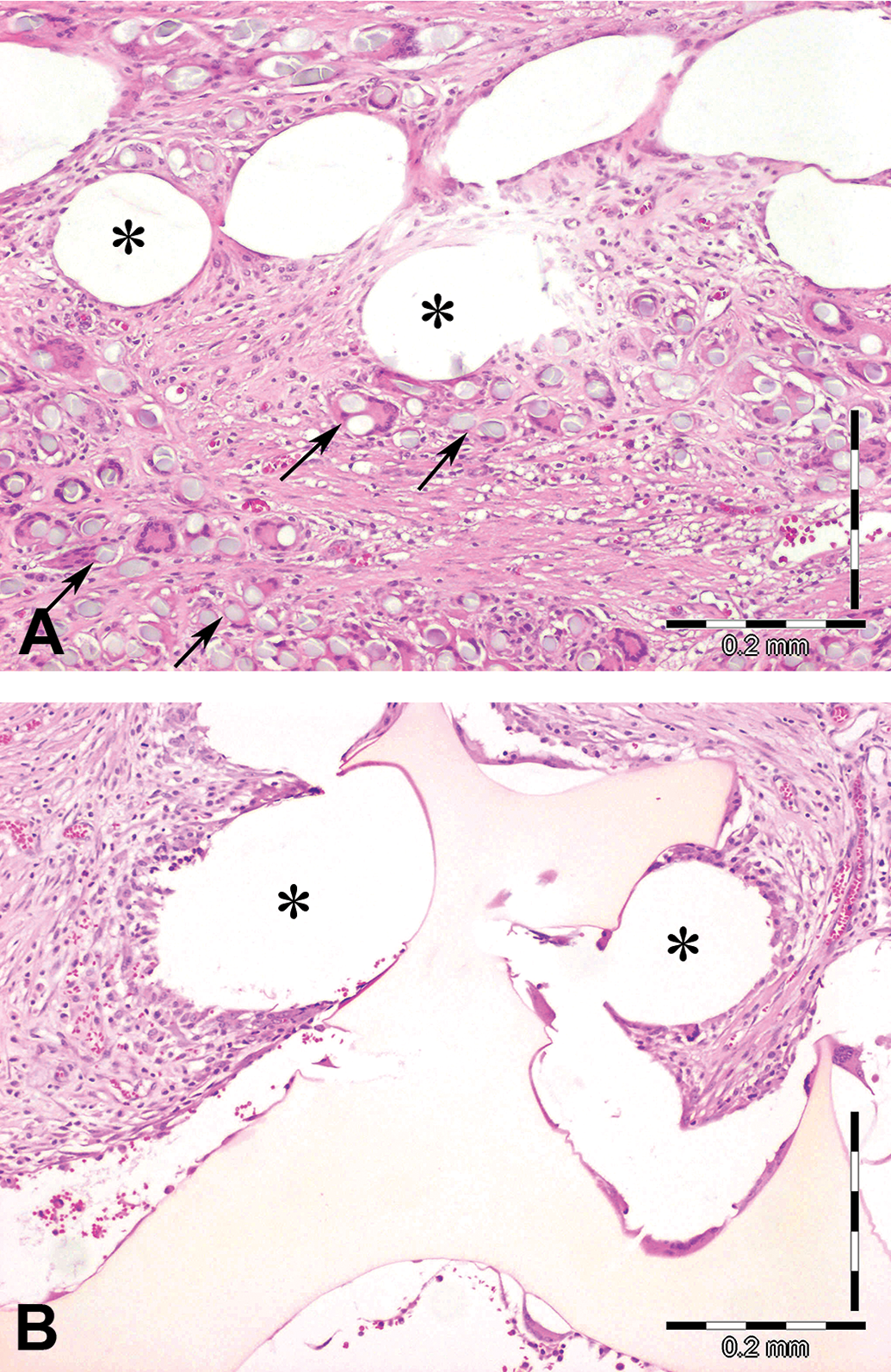
(A) Composite implant includes both large, nonbioresorbable monofilament zones (spaces indicated by asterisks) and more numerous, smaller, polyfilaments (arrows) which are undergoing phagocytic bioresorption. The mesh interstitium is moderately expanded by collagen, fibroblasts, mononuclear inflammatory cells, and fiber-associated multinucleate giant cells. (B) High magnification of mesh implant with coating (yellow at center and bottom). Both the large nonbioresorbable monofilaments (asterisks) and the pale yellow coating are peripherally accompanied by macrophages and multinucleated giant cells. The interstitium is expanded by fibroblasts, inflammatory cells, scant collagen, and few capillaries.
Other implant-associated parameters are particularly critical for determination of implant integration. These include neovascularization, both overall degree and location within and around an implant, and the presence of fibroblasts and deposition of extracellular matrix (particularly collagen). It may be useful to separate the cellular/fibroblastic and matrical/collagenous components of the fibrotic response, and the absolute and relative amounts of these may change as responses mature. Encapsulation of fibrovascular tissue around implants can be scored, as can ingrowth into implants. Regarding tissue ingrowth, scoring paradigms or descriptive assessments may be significantly dependent upon implant architecture, as discussed in a subsequent section.
In conjunction with evaluation of fibroplasia/fibrosis, pathologists may also be asked to determine whether scar tissue has formed. Given the often loose and colloquial definition of “scar,” such evaluation should occur with caution and should include consideration of both macroscopic features (e.g., irregular appearance and stricture) and microscopic ones (abundant, dense, mature, and disorderly collagenous tissue).
Pathologic evaluation should include presence of implant-associated necrosis, protein exudation, mineralization, and osseous metaplasia. Fatty infiltration of peritoneal implants must be differentiated from deposits of omental fibro-adipose tissue that may implant on the visceral surface subsequent to in vivo adhesion formation. Fat necrosis and mineralization may follow incidental tissue trauma during implantation, and this should be differentiated from implant mineralization or implant-induced necrosis.
Histologic evaluation of implant bioresorption/degradation reinforces the macroscopic assessment at necropsy and also provides information about specific implant components. Large PP or similar nonbioresorbable fibers remain prominent across time points. Resorbable implant components are usually typically associated with some degree of inflammatory cell accumulation, neovascularization, or fibrosis. Following resorption and late stage remodeling, however, implants may be difficult to identify definitively and may be indistinguishable from adjacent, nonimplanted sites. Figures 8 –10 show progression of chronic responses in several different implant classes. Evidence of coating resorption in coated implants can be derived from changes in coating integrity, staining, and cross-sectional area, as well as the presence of macrophages, foam cells, and giant cells along appropriate implant surfaces.
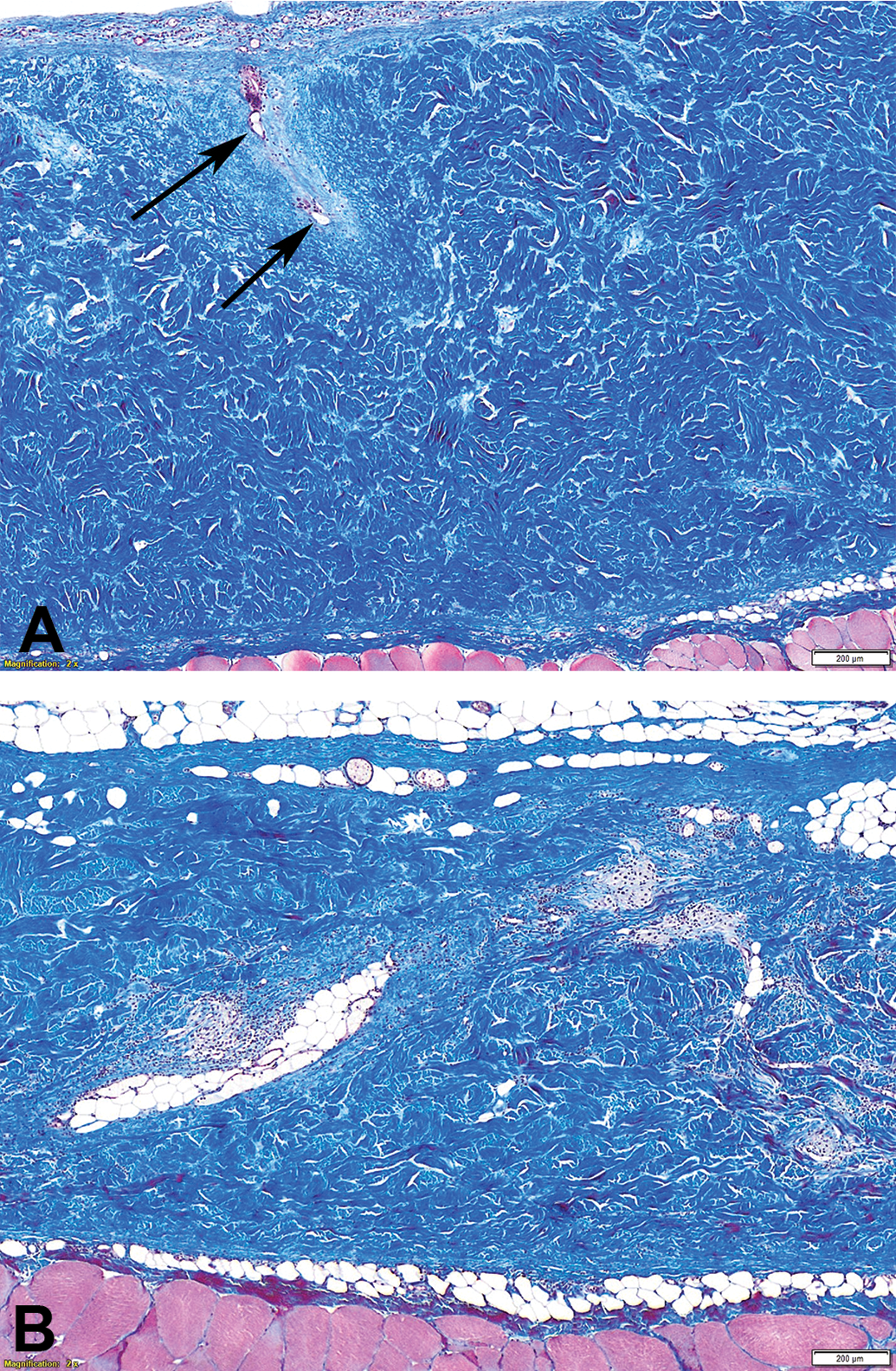
Bioresorbable polyfilament, Masson’s trichrome stain. Early chronic appearance of the implant at day 14 (panel A) shows a thin layer of collagenous tissue along the mesh, and inflammatory cells gathered around the fiber periphery. Panel B shows complete bioresorption and remodeling at the late chronic time point (day 180) with only a thin band of collagenous tissue remaining.
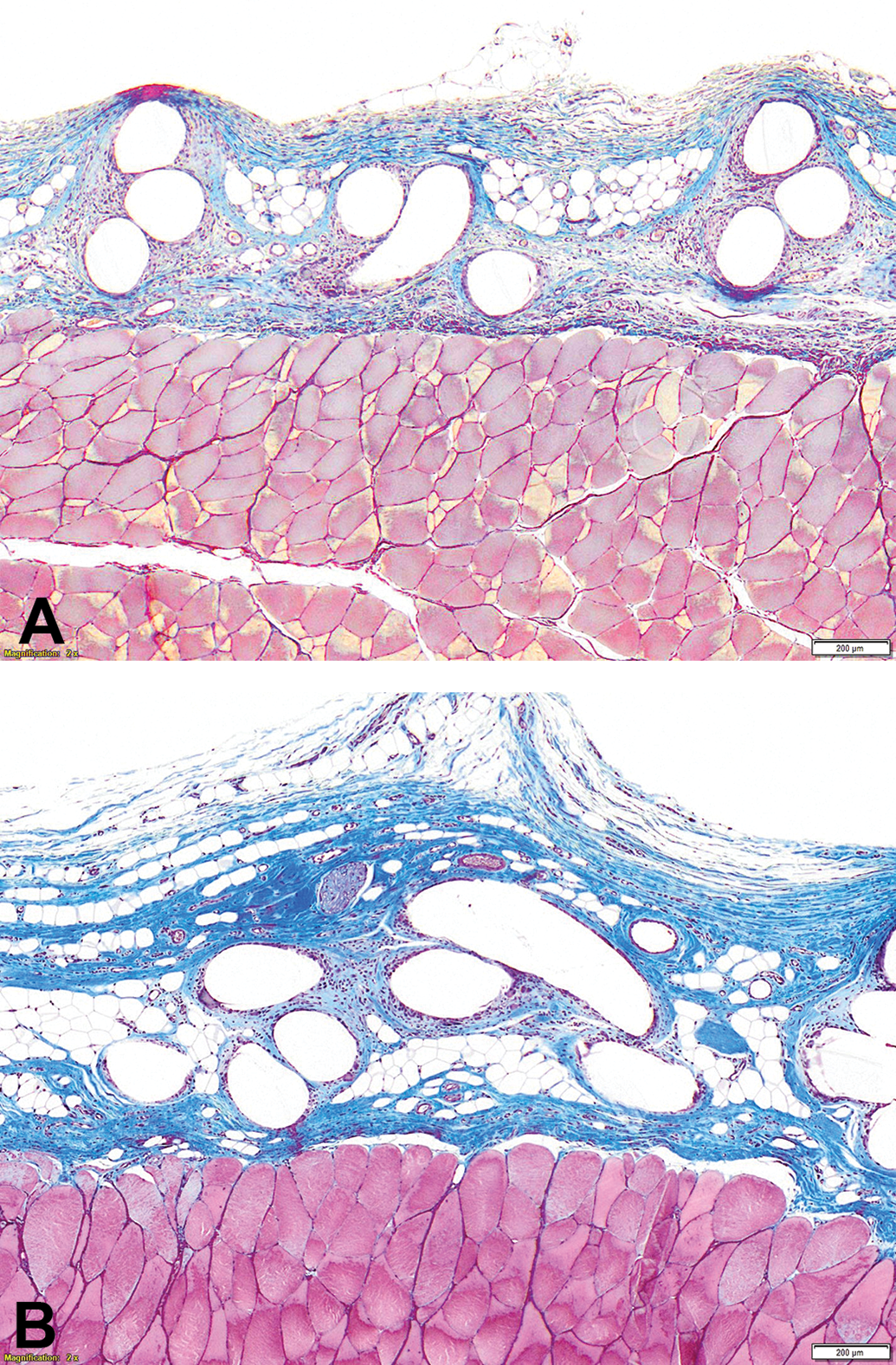
Acellular dermal matrix, Masson’s trichrome stain. At the early chronic time point (day 28; panel A), there is thin peripheral fibroproliferative response, extending into the follicular remnant at top left (arrows). At the late chronic time point (day 180; panel B), there is more notable interstitial infiltration by adipose and fibrovascular tissue, and slight implant consolidation.
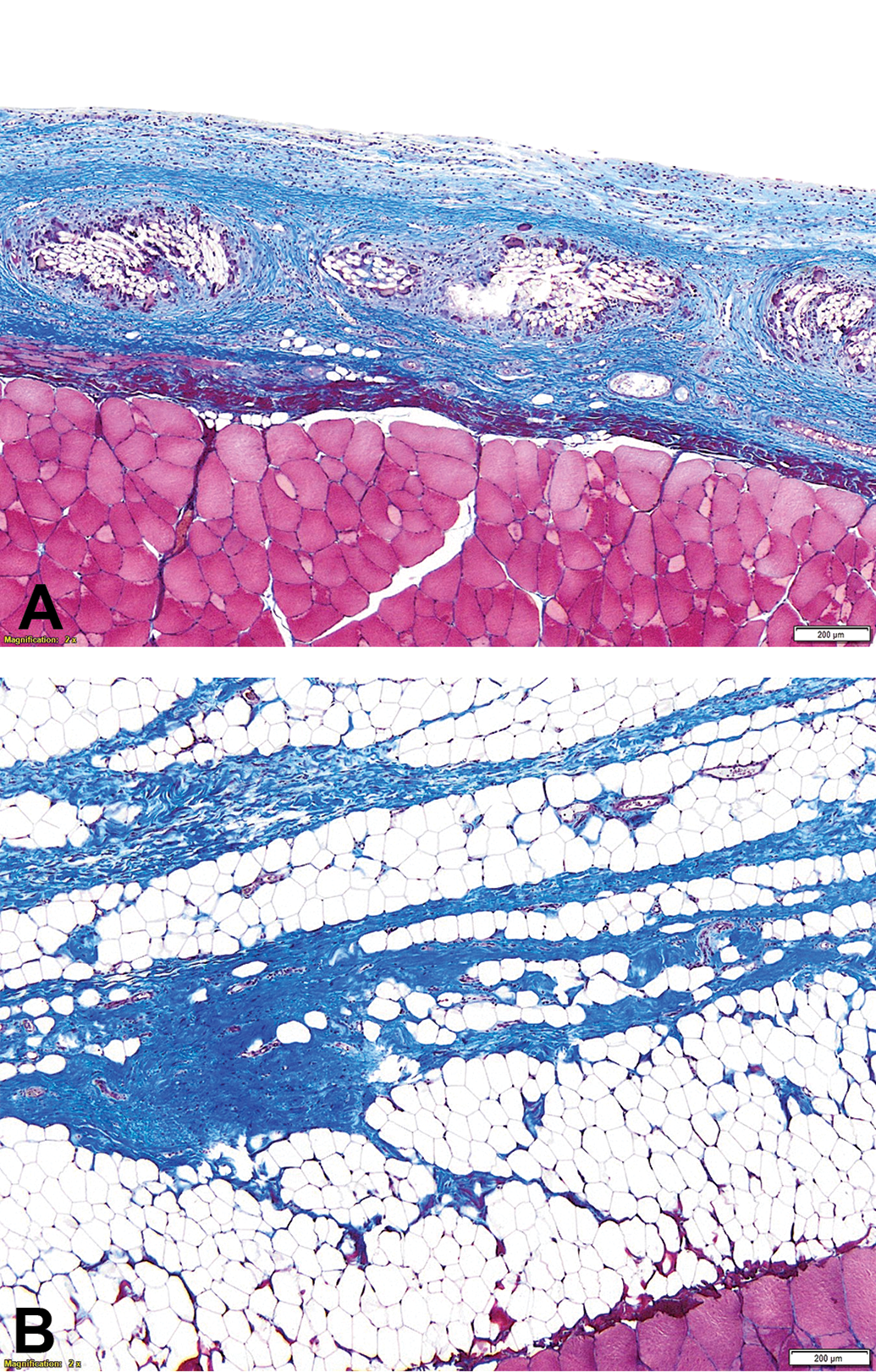
Synthetic nonabsorbable monofilament, Masson’s trichrome stain. The mesh is unchanged from the early chronic time point (day 28; panel A) through the late chronic time point (day 180; panel B), while the tissue response exhibits maturation and consolidation.
Histopathologic Evaluation—Special Considerations
Assessment of response to implants may be complicated by factors above and beyond inherent tissue–material interaction. Changes present in tissue section may be influenced by the size of samples, as well as the specific site within the implant (e.g., distance from edge center or fenestrations). Particular anatomic location (e.g., subcutis, preperitoneal, or intraperitoneal space) may also impact the ability of cellular constituents to access an implant (via direct migration or following vascular ingrowth). Iatrogenic tissue trauma occurring during implantation can introduce spurious changes, for example, within skeletal muscle or adipose tissue, where cellular injury and necrosis are likely to incite fibrosis and granulomatous inflammation, respectively. Similarly, attempts should be made to determine whether fibrin exudation or hemorrhages, when present, are related to the interventional model or the implant itself. When possible, complications such as seroma (Figure 11), infection, or idiosyncratic hypersensitivity responses should be ascribed to implant material or to model-related causes. The same should be done with findings related to accessory, nonimplant material such as cyanoacrylate tissue glue, anchor, or tissue closure sutures (Dumanian, Tulaimat, and Dumanian 2015; commonly polydioxanone or PP), or permanent or absorbable fixation devices such as tacks or other fasteners.
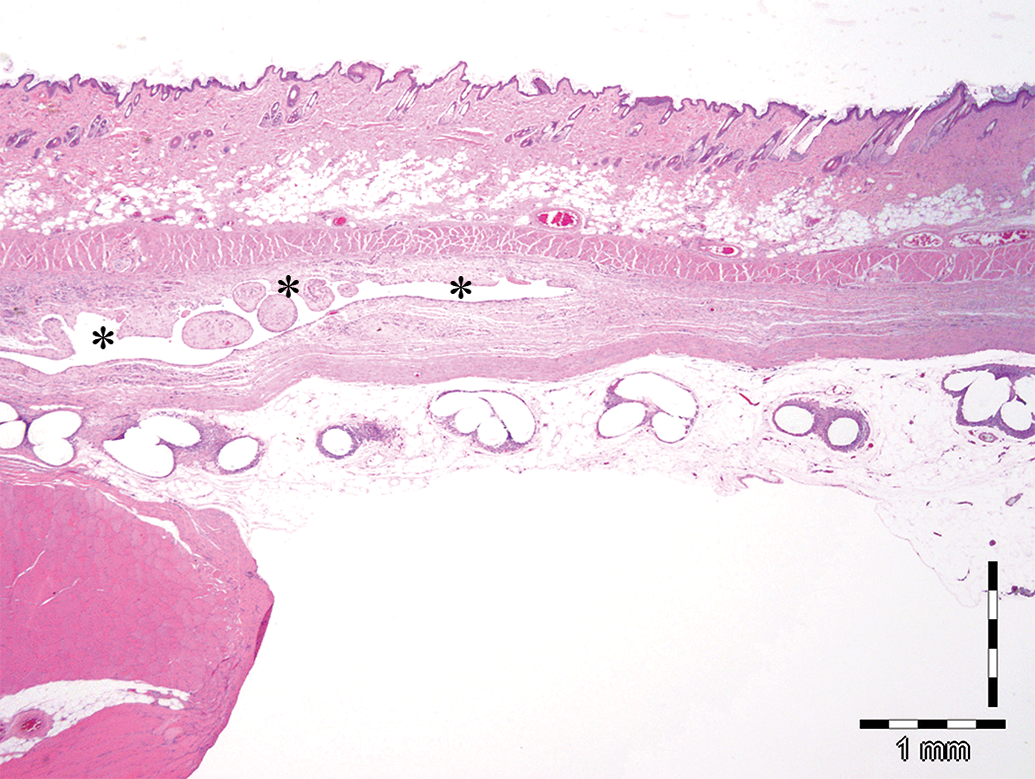
Low magnification view of peritoneal implant in abdominal wall defect model. Asterisks indicate seroma pocket (nonrelated to the implanted mesh and likely consequent to the implantation procedure) within abdominal wall underlying the implant. Note that such fluid-filled pockets collapse during sectioning, resulting in a flattened, empty, or nearly empty profile.
Regardless of the particular histopathologic scoring matrices that are utilized, the specific architecture of some classes of scaffolds may impact both the approach to histopathologic evaluation and the underlying tissue responses as well (e.g., by influencing exposure to inflammatory cells and hastening or retarding cellular ingrowth).
Because of their derivation from normal tissue, acellular dermal matrices can pose a specific challenge by mimicking host tissue. Because the collagen of acellularized matrices is indistinguishable from mature host collagen, differentiation of novel versus preexistent collagen can be difficult but can be accomplished via the slight tinctorial differences (in H&E, trichrome, and PSR-stained sections) between thick, mature, chronic collagen and thin, immature, novel collagen (Rich and Whittaker 2005). Implant collagen bundles may be thick, bright eosinophilic, and acellular in comparison to the narrow or amorphous, pale pink, paucicellular deposits of interstitial collagen that typical of newly formed host tissue. Collagenous matrices may be differentiated by architectural features indicative of their derivation site, such as the crosshatched arrangement of thick collagen bundles typical of dermis, empty linear to rounded spaces representing former sites of follicular and adnexal elements, or presence of elastic fibers (prominent with MT staining). The scaffolds are initially acellular, and acellular foci may remain through early time points. Tissue ingrowth into acellular dermal matrices may be first noted along the angled, linear gaps that represent prior dermal sites of hair follicles, with further tissue infiltration along the implant surface and also extending into the mesh interstitium. Containing abundant if densely packed interstitial space, collagenous scaffolds can eventually become obscured by a robust fibroproliferative response (Trandafir, Popa, and Vasile 2017).
Tissue infiltrative and inflammatory responses also can be influenced by monofilament or polyfilament fiber composition. Fundamentally, even terms such as “interstitial” must be applied with care. PP monofilaments are common and owing to the size and rigidity of the material, fibers tend to be large and, because they are nonbioresorbable, well defined. As a consequence, “interstitium” may refer to the space outside the fibers but within the weave of such monofilament areas. In contrast, polyfilament meshes contain both, this extra-fiber/intraweave space as well as additional potential space within the polyfilaments themselves, and device integration/incorporation.
While histopathology is necessary for evaluation of tissue responses, SEM can be very useful in evaluating changes in scaffolds and coating, whether postimplantation or following benchtop modification. This generally consists of bioresorptive responses such as fiber shrinkage or pitting and diminishment or fragmentation of coating layers (Figure 12).

Scanning electron micrographs of portions of explanted meshes showing polyfilament mesh with sparse associated adherent material (panel A) and monofilament mesh with localized resorptive change (panel B; arrow).
With their clinical prevalence and increasing frequency, mesh devices represent a common subject of preclinical evaluation, which is important for medical device pathologists to understand. Selection of tissue processing and embedding may vary with mesh composition, such as presence of biologic components (e.g., collagen), synthetic polymer fibers, coatings, and other components. The complexity and range of evolving mesh designs calls for consistent and detailed pathologic evaluation to determine host responses and assess safety. Thorough macroscopic and microscopic evaluation in animal models depends on histologic parameters, semiquantitative scoring matrices, and morphometric analyses that are appropriate to this class of implants. Implant persistence, architecture, and associated host responses should be noted at necropsy. Microscopic evaluation focuses on evaluation of bioresorption, integration/tissue ingrowth, neovascularization, and inflammation. Recognition of specific study objectives, mesh characteristics, and knowledge of anticipated responses at given time points helps to determine the appropriate tissue processing, staining, evaluation, and interpretation of safety profile of mesh devices in the preclinical setting.
Footnotes
Author Contributions
Authors (JK, RM, FG) contributed to conception or design, data acquisition, analysis, or interpretation; drafting the manuscript (JK, RM); and critically revising the manuscript (FG). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
