Abstract
Thermoresponsive materials have the ability to respond to a small change in temperature—a property that makes them useful in a wide range of applications and medical devices. Although very promising, there is only little conclusive data about the cytotoxicity and tissue toxicity of these materials. This work studied the biocompatibility of three Food and Drug Administration approved thermoresponsive polymers: poly(N-isopropyl acrylamide), poly(ethylene glycol)-poly(propylene glycol)-poly(ethylene glycol) tri-block copolymer, and poly(lactic acid-co-glycolic acid) and poly(ethylene glycol) tri-block copolymer. Fibroblast NIH 3T3 and HaCaT keratinocyte cells were used for the cytotoxicity testing and a mouse model for the in vivo evaluation. In vivo results generally showed similar trends as the results seen in vitro, with all tested materials presenting a satisfactory biocompatibility in vivo. pNIPAM, however, showed the highest toxicity both in vitro and in vivo, which was explained by the release of harmful monomers and impurities. More data focusing on the biocompatibility of novel thermoresponsive biomaterials will facilitate the use of existing and future medical devices.
Keywords
Biomaterials have been used in medical devices since antiquity (Huebsch and Mooney 2009). These materials interface with biological systems with the purpose of evaluating, treating, augmenting, or replacing organs or functions of the body (Williams 1987). The degree of sophistication of currently used biomaterials has increased significantly over the years (Huebsch and Mooney 2009), in particular after the immergence of stimuli-responsive materials.
Thermoresponsive materials exhibit a thermoresponsive phase transition often termed a lower critical solution temperature (LCST; Chung et al. 1999). Below their LCST, these polymers present water-soluble and hydrophilic characteristics, but above it they become hydrophobic, aggregate and harden. Consequently, thermoresponsive materials have been used in applications where solidification following an increase in temperature is beneficial, for example, in drug delivery, coatings, and sealants (Bawa et al. 2009). The inherent advantages of thermoresponsive hydrogels in delivering pain relief medications and other drugs, as exudate absorption materials, as barriers to bacteria and viruses, and being permeable to oxygen, have also contributed to their use in dermal care management (Rogero et al. 2003).
The International Organization for Standardization (ISO 10993-5) requires cytotoxicity assessment of all medical devices (2009). Despite the great potential of existing thermoresponsive materials, however, there has been little conclusive research regarding their local toxicity (Cooperstein and Canavan 2013). For instance, the toxicity of poly(N-isopropyl acrylamide) (pNIPAM), the most investigated thermoresponsive polymer, is still being debated (Hoffman 2014). In fact, there are very few publications regarding the toxicity of thermoresponsive polymers compared to numerous publications focusing on their applications.
Here, we investigate the biocompatibility of three of the most studied and FDA-approved (Diniz et al. 2015; Makadia and Siegel 2011; Shagan et al. 2018; Kim et al. 2017) thermoresponsive materials: poly(lactic acid-co-glycolic acid) (PLGA) and poly(ethylene glycol) tri-block copolymer (PLGA-PEG-PLGA), poly(ethylene glycol)-poly(propylene glycol)-poly(ethylene glycol) tri-block copolymer (Pluronic F-127), and pNIPAM (Figure 1). These polymers possess LCST values around body temperature (Plunkett et al. 2006; Wang et al. 2011; Yu, Zhang, and Ding 2011), which makes them ideal for dermal therapy. The investigated polymers are administered topically in a variety of applications. For example, Pluronic F-127 was found to enhance cutaneous wound healing in rats (Kant et al. 2014) and was used for topical nitric oxide delivery (Champeau, Seabra, and de Oliveira 2017). PLGA-PEG-PLGA and pNIPAM have been investigated for improving ocular delivery of various drugs (Gao et al. 2010; Hsiue et al. 2002). Since an important prerequisite for a successful dermal system is being nontoxic and nonirritant, both in vitro and in vivo toxicity evaluation were conducted (Schmolka 1972).
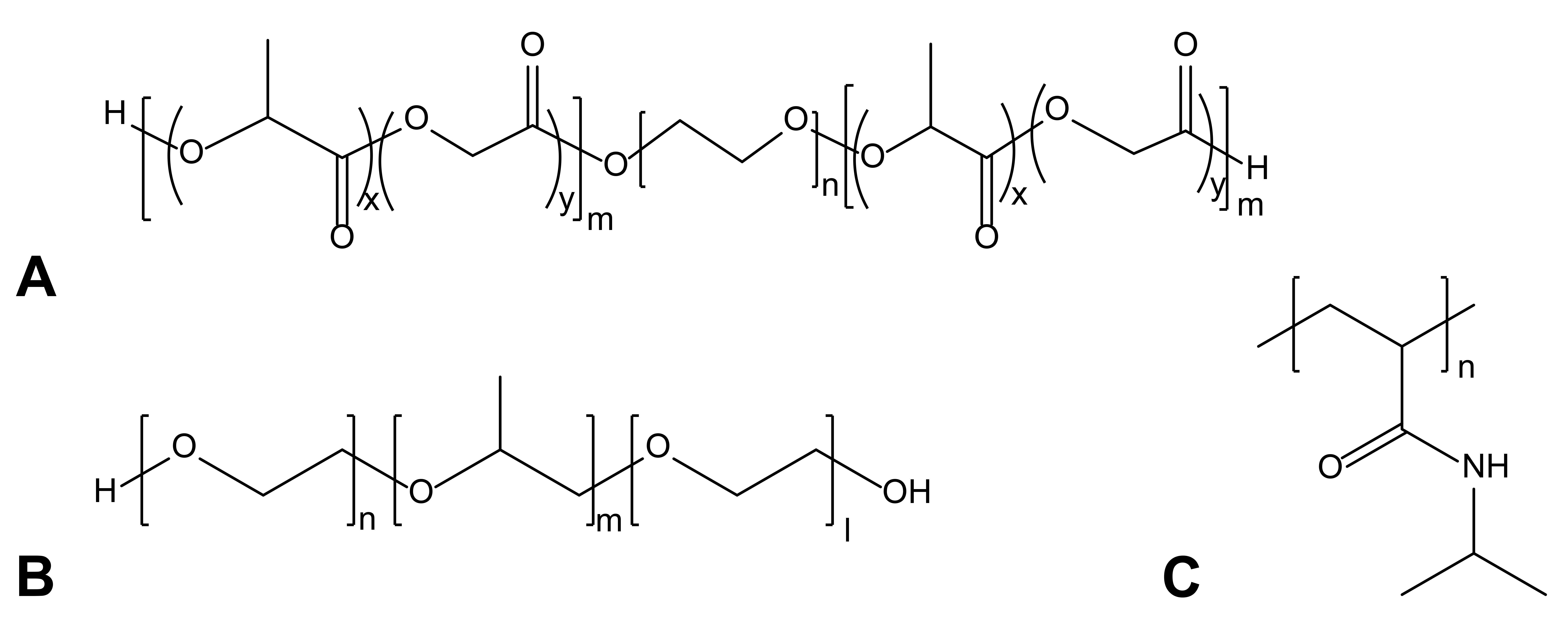
Chemical structures of (A) poly(lactic acid-co-glycolic acid) and poly(ethylene glycol) tri-block copolymer (PLGA-PEG-PLGA), (B) poly(ethylene glycol)-poly(propylene glycol)-poly(ethylene glycol) tri-block copolymer (Pluronic F-127), and (C) poly(N-isopropyl acrylamide) (pNIPAM).
Materials and Methods
Chemicals
PLGA-PEG-PLGA and Pluronic F-127 were purchased from Sigma-Aldrich, Inc. (St. Louis, MO). pNIPAM was purchased from Polysciences, Inc. (Warrington, PA). Dulbecco’s modified Eagle medium (DMEM), minimal essential medium (MEM-Eagle), fetal calf serum (FCS),
A stock solution of each of the tested polymers was prepared in double distilled water (DDW). Various hydrogels with concentrations ranging from 7% to 23% were obtained by further dilution with DDW. Solutions were carefully mixed and stored at 4°C overnight to get a homogenous solution. Hydrogels too soft and weak to be applied on the skin or too viscous to process at body temperature were excluded from the study. The gelation times of each concentration solution was assessed by the tilting method (Vadnere et al. 1984; Deshmukh et al. 2010). In brief, 1 ml aliquot of each hydrogel was placed on a tilted glass slide (45°C) at 37°C. Gelation was said to have occurred when the meniscus stopped moving. The time until gelation was monitored visually using a timer. Three replicates were measured each tested group (n = 3). Results are given as average ± standard deviation.
The toxicity of the different hydrogels was evaluated on fibroblast NIH 3T3 and HaCaT keratinocyte cells in comparison to control cells (without exposure to the hydrogels). Cells were grown in DMEM supplemented with 10% FCS, penicillin-streptomycin-nystatin and
All animals were cared for in compliance with protocols approved by the Council for Animal Experiments, State of Israel Ministry of Health, in conformity with the Animal Welfare Law guidelines (published in 1994). C57 Black (Bl) female mice were purchased from Envigo (Jerusalem, Israel); mice were subjected to 7 days quarantine and acclimation period before the beginning of the experiment. During the experiment, mice were housed together, in groups of five, in a temperature- and humidity-controlled environment at a 12:12-light/dark cycle. Each examined group included five mice. Experimental hydrogels (200 µl) were topically administered once daily on a shaved region (1 × 1 cm) on the flanks of mice under anesthesia with 1% isoflurane (Piramal Critical Care, Inc., Bethlehem, PA) in oxygen. Mice were immediately recovered after hydrogels administration. DDW was used as a control. We noted that the tested hydrogels can penetrate and absorbed through the skin (Lufton et al. 2018; Yurdasiper, Ertan, and Heard 2018; Rancan 2016). Therefore, each hydrogel was administrated daily and the treated area was remained uncovered for the entire period of the experiment. After five days, mice were sacrificed using isoflurane anesthesia followed by carbon dioxide.
Skin samples were harvested, and tissues were fixed and preserved in 10% neutral-buffered formalin. Histopathological evaluation was performed on all animals in all (control and treated) groups. The histopathological sections of the skin samples (i.e., trimming of the tissue) were taken from the middle of the sample, to include entire thickness, from the epidermis to subcutis. After dehydration and embedding in paraffin wax, sections of the tissues were cut at 5-µm thickness and stained with hematoxylin and eosin. One section was tested from each mice. The histological evaluation of the skin pertaining to treatment sites was made by a subjective description of the observed tissue reaction, based on parameters describing grade of tissue tolerance (i.e., inflammation, necrosis, and fibrosis). The scoring of the lesions was done semiquantitatively, using a 5-point grading scale (0–4), taking into consideration the severity of the changes (0 = no change, 1 = minimal change, 2 = mild change, 3 = moderate change, 4 = marked change), based on the criteria explained by Schafer et al. (2018).
Histological images were acquired using an Eclipse 50i microscope (Nikon Instruments Inc., Melville, NY) and a SPOT Insight 4 Meg FW Color Mosaic camera with SPOT4.5.9.1 software (Diagnostic Instruments Inc., Sterling Heights, MI).
Statistical comparisons were performed with a Prism 6 GraphPad (La Jolla, CA). A Student’s t test was used. p values <.05 were considered to reflect statistical significance.
Results and Discussion
We investigated the toxicity of three hydrogels from thermoresponsive polymers. In order for these hydrogels to be suitable for administration on the skin, they should have low viscosity at refrigerator temperature but rapidly solidify after administration. Therefore, we started this study by correlating the concentration with the times required for gelation, thus finding the optimal polymer concentrations for each system (Figure 2A).
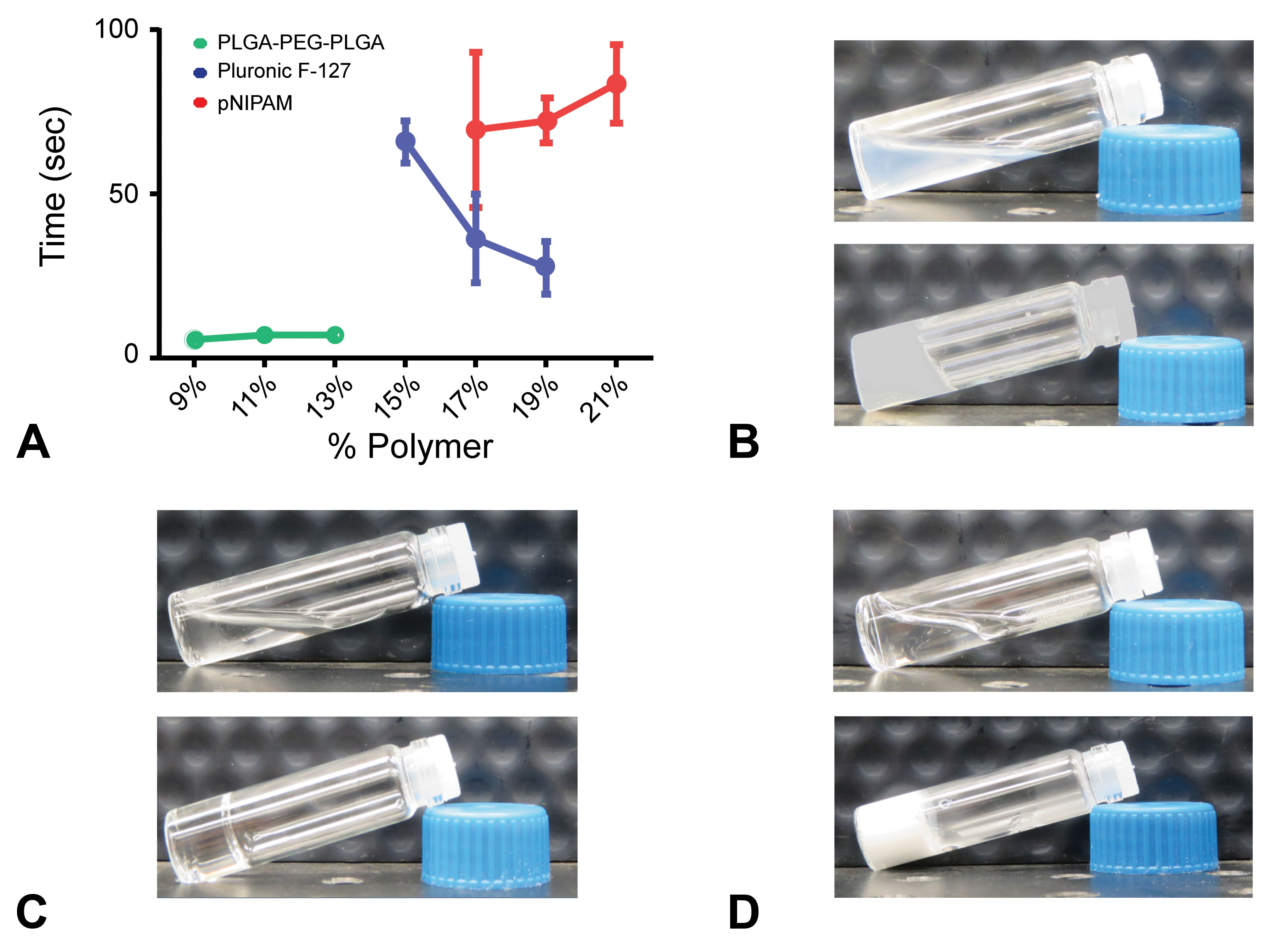
(A) The effect of concentration on gelation time of thermoresponsive hydrogels (data are means ± SD). (B) Poly(lactic acid-co-glycolic acid) and poly(ethylene glycol) tri-block copolymer (PLGA-PEG-PLGA), (C) poly(ethylene glycol)-poly(propylene glycol)-poly(ethylene glycol) tri-block copolymer (Pluronic F-127), and (D) poly(N-isopropyl acrylamide) (pNIPAM) before and after temperature changes.
Pluronic F-127 solution showed a concentration-dependent behavior: increasing the concentration decreased gelation time. For example, a 15% (w/v) concentration solution gelled within 70 sec compared to less than 30 sec for the 19% soution (Figure 2A and C). Increasing the concentrations of PLGA-PEG-PLGA and pNIPAM did not appear to have a significant effect on the gelation time. Based on a trade-off between concentration (i.e., the desire to work with minimal concentrations) and gelation times, we chose to work with 9% w/v PLGA-PEG-PLGA (Figure 2A and B) and 19% w/v pNIPAM and Pluronic F-127 (Figure 2A, C, and D).
Thermoresponsive systems with similar concentrations as found here have been suggested for the delivery of various drugs. For example, 20% aqueous Pluronic F-127 hydrogels have been suggested for dermal and rectal administration of indomethacin (Miyazaki et al. 1984, 1987, 1995). In vivo studies have suggested that Pluronic F-127 is of practical use as a cell and drug carrier (Khattak, Bhatia, and Roberts 2005) at concentrations of 15% to 20% w/w. Similarly, a 20% pNIPAM-gelatin solution has been successfully implemented for postsurgery adhesion control (Ohya et al. 2005).
The cytotoxicity of the various hydrogels was evaluated on NIH 3T3 (Figure 3A) and HaCaT cell lines (Figure 3B). Hydrogels were either in direct contact with cells (direct, Figure 3C) or in transwell inserts where they would be isolated from cells (indirect contact with soluble elements). In both the direct and indirect assays, viability (as a percentage relative to the control—without hydrogels) was assessed after 24 hr by the MTS assay. Trends between the cytotoxicity levels produced by each tested hydrogel were similar between the two cell cultures; the HaCaT cell line, however, was generally less sensitive. PLGA-PEG-PLGA demonstrated the best results in terms of cell toxicity (Figure 3A, B), with no toxicity to both cell lines. The cytotoxicity induced by Pluronic F-127 was slightly higher, with 75% and 100% viability for NIH 3T3 and HaCaT, respectively, in the indirect assay. Direct exposure caused a drastic decrease in cell viability, to around 25% and 65% for the NIH 3T3 and HaCaT cells, respectively.
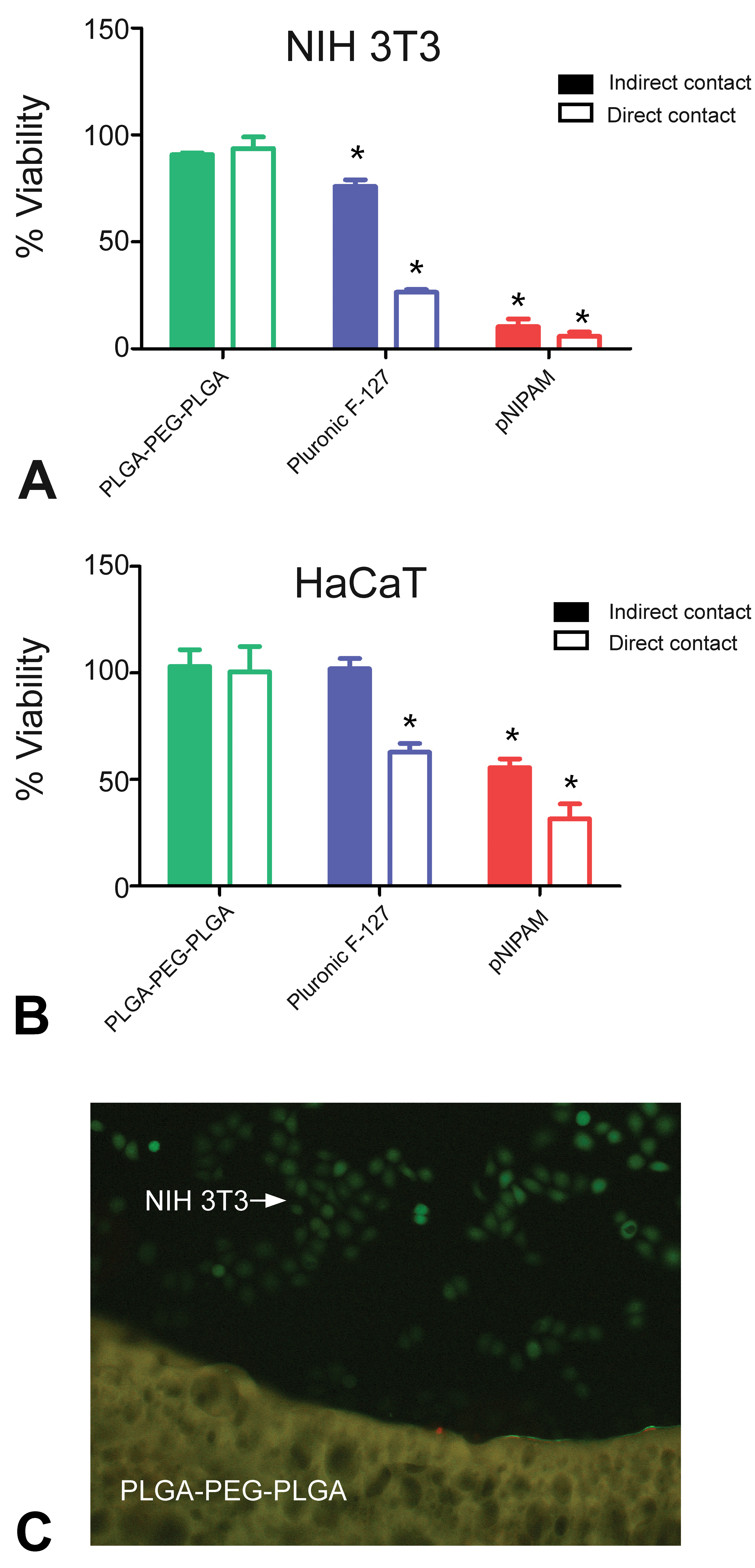
Viability 24 h after exposure to thermoresponsive hydrogels. (A) NIH 3T3 fibroblast and (B) HaCaT keratinocyte. Data are means ± SD of the percentage viability derived from the MTS assay of cells with or without indirect contact with the polymers (full column—indirect, hollow column—direct), n = 4. The results were compared using a Student’s t test (*p < .05). (C) Fluorescence microscopy of the margins of the poly(lactic acid-co-glycolic acid) and poly(ethylene glycol) tri-block copolymer (PLGA-PEG-PLGA) hydrogel and surrounding NIH 3T3 cells (for better clarity, cells were stained using a live/dead kit).
The cytotoxicity induced by pNIPAM was significantly higher compared to the other hydrogels. Fibroblast cells showed 10% and 5% viability for the direct and indirect, respectively. For the HaCaT cells, cytotoxicity was less pronounced with 30% and 55% viability for the direct and indirect assays, respectively. The relatively high cytotoxicity produced by pNIPAM has been attributed to impurities in the commercially available pNIPAM, to the presence of leftover monomers and to toxic degradation products (Cooperstein and Canavan 2013; Patenaude and Hoare 2012).
The negative effect of Pluronic F-127 on cell viability has been attributed to its amphiphilicity (Khattak, Bhatia, and Roberts 2005) and its ability to bind cell membrane and alter its microviscosity (Melik-Nubarov et al. 1999). This notion was supported by our experiments where Pluronic F-127 presented toxicity also when placed in the inserts, without direct contact with the cells (indirect in Figure 3A and B).
Overall, PLGA-PEG-PLGA presented no cytotoxicity, followed by Pluronic F-127 with a very mild toxicity, in particular demonstrated in the direct assay. pNIPAM exhibited the highest cytotoxicity levels in both the direct and indirect assays. These results are in agreement with previous studies that suggested that most of the toxicity of hydrogels is associated with unreacted monomers, oligomers, and initiators that leach out during application (Peppas et al. 2000).
The ability of the tested hydrogels to induce skin inflammation was evaluated in vivo (Figure 4). C57BL/6 mice were shaved in the dorsal region and randomly assigned to one of the each group: Control, 19% Pluronic F-127, 9% PLGA-PEG-PLGA, and 19% pNIPAM (n = 5).
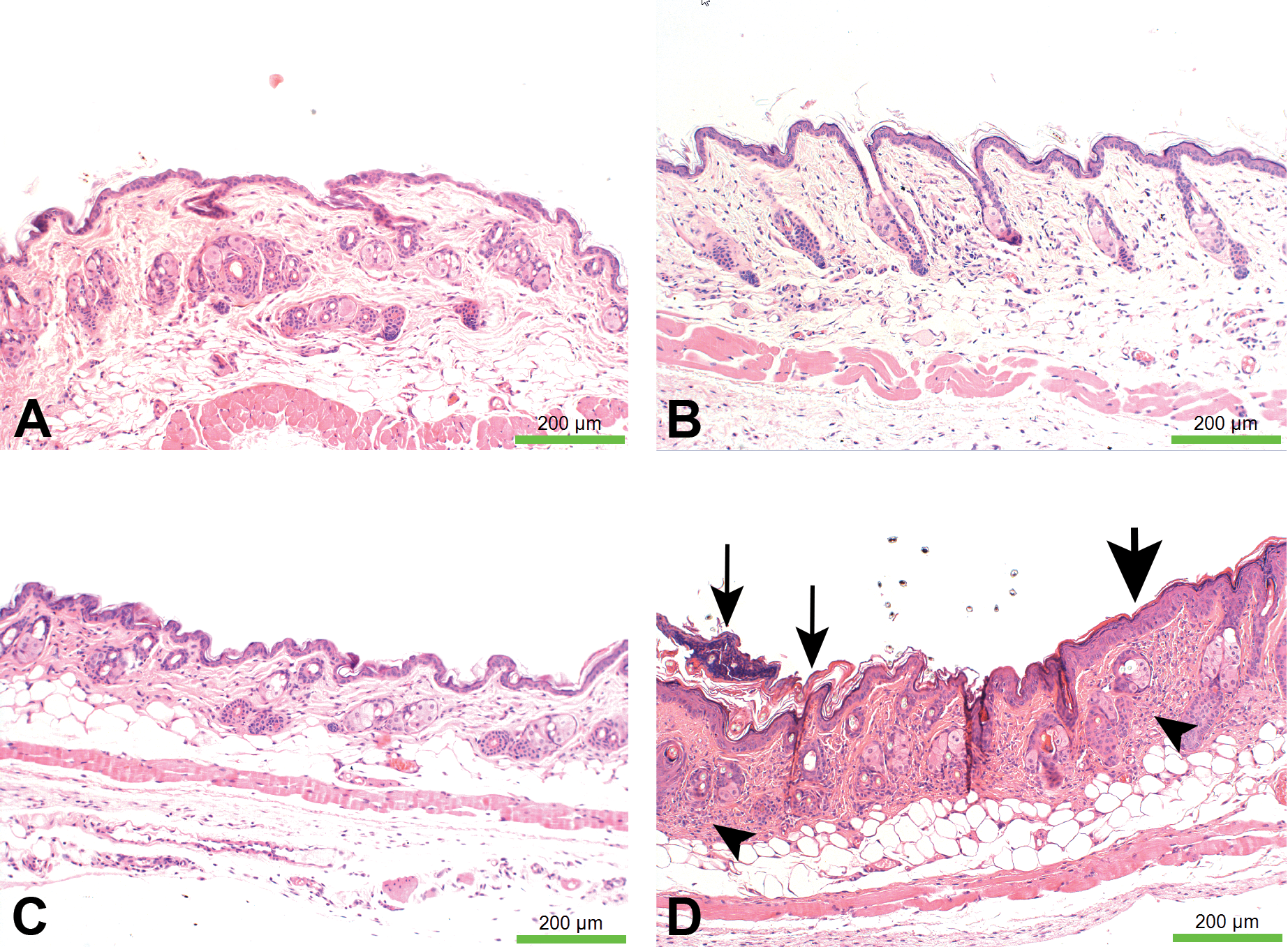
Tissue reaction to 200 µl of tested thermoresponsive hydrogels applied daily on the skin, shown on hematoxylin–eosin staining of skin (A), control (B), poly(lactic acid-co-glycolic acid) and poly(ethylene glycol) tri-block copolymer (PLGA-PEG-PLGA) (C), Pluronic F-127 (D), poly(N-isopropyl acrylamide) (pNIPAM). Histological scores for specific morphological changes (E) and average (F) inflammation were compared using a Student’s t test (data expressed as means ± SD, *p < .05, n = 5 per group). Arrows indicate areas of inflammation. Photographs are representative views. In the micrograph from a mouse administered with pNIPAM (D), there is parakeratosis and crust formation (thin arrows), hyperkeratosis, epidermal hyperplasia (thick arrow), and dermal mononuclear cells infiltration associated with fibrosis (arrowheads).
Each treatment was administered daily for five days. Mice administrated with PLGA-PEG-PLGA or Pluronic F-127 did not show any sign of adverse effects or clinical signs of toxicity, mice administered with pNIPAM revealed a mild erythema limited to the site of administration area. After five days, mice were euthanized and a histological evaluation, based on the inflammation degree scoring system, was made of the skin and underlying subcutaneous tissue. All mice in the control and the Pluronic F-127 groups did not show any signs of inflammation (an average inflammation score of 0; Figure 4A, C, F). 4 or 5 mice in the PLGA-PEG-PLGA group had no signs of inflammation, with one mice showing minimal hyperkeratosis and mild hyperplasia with minimal rete pegs (an average inflammation score of 0.2; Figure 4B, E, F). In contrast, mice administered with pNIPAM showed crust formation, minimal parakeratosis, minimal hyperkeratosis, minimal to mild hyperplasia, and minimal mononuclear cells infiltration (average inflammation score of 1.2; Figure 4D–F).
In vivo results generally mirrored the results seen in vitro (Figure 3): PLGA-PEG-PLGA and Pluronic F-127 showed minimal or no toxicity while pNIPAM induced a mild toxicity (all toxicity levels were lower than 1.5 on a scale of 0–4). Polymeric hydrogels usually show good biocompatibility when in contact with body fluids and tissue, mainly because they are made of water-soluble molecules and water (Mizrahi et al. 2013). The formation of the 3-D structure did not prevent the release of leftover monomers and impurities, as evident from both in vitro and in vivo results. We note that the agreement between the in vitro and in vivo results is an important factor in the assessment of any medical device, in particular when the proposition is to reduce or replace the use of animals due to ethical concerns (Rogero et al. 2003).
Conclusions
With the emergence of sophisticated medical devices, next-generation biomaterials will require enhanced biocompatibility. The thermoresponsive hydrogels studied in this work did not present a meaningful toxic effect both in the in vitro and in vivo evaluations, suggesting that these biomaterials may be acceptable for use in a dermal medical device. Nevertheless, we note that PLGA-PEG-PLGA and Pluronic F-127 showed the best results in terms of biocompatibility, which probably makes these hydrogels more suitable for skin application than pNIPAM. Similar studies to the one described here, focusing on the biocompatibility of next-generation biomaterials, will facilitate a better understanding of current systems and improve the functionality and safety of future medical devices.
Footnotes
Acknowledgments
The authors would like to thank the Russell Berrie Nanotechnology Institute, Technion, for its scholarship for excellent students in nanoscience and nanotechnology research.
Author Contributions
All authors (SY, AS, AN, BM) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results received funding from the Israeli Science Foundation (No. 505/17) and the Polk Foundation.
