Abstract
Microphysiological systems (MPS), commonly known as organs-on-chips, are a rapidly advancing technology that promises to impact many areas of medical and toxicological pathology. In this minireview, the history of MPS and its potential utility in safety assessment are described with the toxicologic pathologist in mind. Several MPS development focus areas are defined, and recent progress in the area is highlighted. MPS will likely become an important tool for the toxicologic pathologist as part of our role in the safety assessment process within the pharmaceutical, biotechnology, medical device, and cosmetic and agrichemical industries.
Successful agrichemical, medical device, pharmaceutical, cosmetic, and biotechnology development depends on well-designed and translatable nonclinical efficacy and safety assessment studies. The cost of development in these industries is high and continues to escalate, especially for the pharmaceutical and biotechnology industries as disease targets become more difficult and the regulatory burden to demonstrate efficacy and safety increases. Recent estimates for development of a single new molecular entity from bench to bedside is US$2.87 billion (DiMasi, Grabowski, and Hansen 2016). Part of this high cost is driven by drug attrition which, especially during the clinical testing phase, is an expensive consequence of the limits in the predictive quality of preclinical and early clinical science. While there are many reasons for the high attrition rate that are beyond the scope of discussion for this review, the suboptimal translation of both efficacy and safety between
Spawned by both of these considerations and its mandate to accelerate the discovery and development of safe new medicines, the Food and Drug Administration (FDA) joined forces with the National Institute of Health (NIH) and Defense Advanced Research Projects Agency (DARPA) in 2010 in an effort to modernize toxicology by developing better models of human adverse response (Sutherland 2013). They issued a request for applications for novel research- and science-based technologies and funded one of the first large efforts for MPS development by the Harvard–Wyss group, the heart–lung micromachine for safety and efficacy testing (Huh et al. 2010; Figure 1). In the following year, an entire MPS program was developed by DARPA-FDA-NIH. The program defined both broad and specific goals for MPS and provided important early support for MPS investigators (Sutherland 2013). The National Center for Advancing Translational Sciences (NCATS) presently directs the tissue chip program (https://ncats.nih.gov/files/Tissue-Chip-factsheet.pdf; Low and Tagle 2017a).
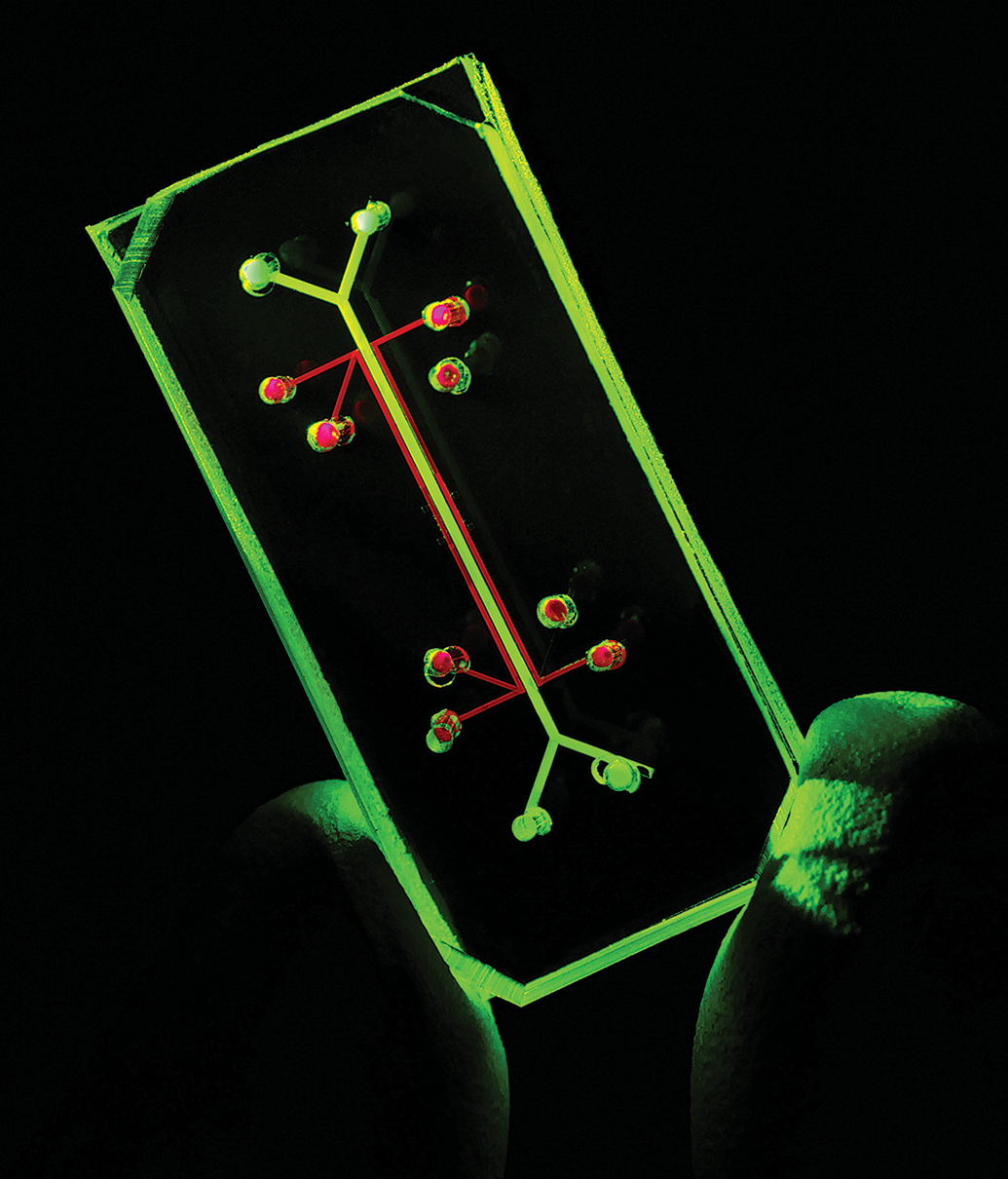
One of the pioneering microphysiological systems devices was the lung-on-a-chip. Using fluorescence light, the microfluidic input and output ports are highlighted here in red and green, respectively. These supply the functional portion of the chip which has two microchambers separated by a porous membrane. Human lung alveolar epithelial cells line one chamber and capillary endothelial cells line the other and mimic the air–fluid interface of the alveolus. Two air-filled chambers are next to the alveolar and endothelial cell chambers and provide the vacuum necessary to direct the biomechanics (expansion and relaxation of “breathing”) for the membrane between the two chambers. Courtesy of the National Center for Advancing Translational Sciences.
The momentum from these early efforts has led to investment and partnerships within the pharmaceutical industry which is driving additional advances in the MPS field as evidenced by efforts like the transatlantic think tank for toxicology (T4) workshop and the Innovation and Quality (IQ) Consortium’s Microphysiological Systems Working Group (Ewart et al. 2017; Marx et al. 2016). Briefly, T4 represents a consortium of pharmaceutical, academic, and government experts focused on driving a paradigm shift in drug development. In 2015, the group met in Berlin and critically examined the present state of MPS and approaches to advance the technology on a global scale. In their 2016 summary, the group envisioned a long-term impact of MPS that by 2030 could reduce current drug development cycle time from 13.5 to <6 years and total development costs from US$2.5 billion to <US$0.5 billion (Marx et al. 2016). T4 projected that the earliest impact would be observed in the drug discovery and lead optimization stages of drug development and would reduce or refine preclinical animal use and the design and conduct of phase I clinical testing. The IQ Microphysiological Systems Working Group is composed of pharmaceutical scientists from 13 different companies and recently partnered with the NCATS to create a road map for MPS use in drug discovery (reviewed in Ewart et al. 2017). In this commentary, the IQ group emphasized the importance of these partnerships and proposed early efforts focus on investigations evaluating mechanistic-based toxicity questions, advancing understanding of human drug disposition, and establishing early preclinical safety screens. Smirnova et al. (2018) envision MPS as an outstanding tool alongside computational modeling for enabling a systems approach to studying toxicologic (and therapeutic) effects of test agents. For the anatomic and clinical toxicologic pathologist, all of these approaches could result in our use of MPS data alongside traditional data sets in the support of our project development teams and collaborators.
While these are very exciting and provocative predictions, what exactly are MPS, what is the status of some leading research efforts, and how as toxicologic pathologists can we impact and synergize with this budding technology? MPS are engineered

A diagram of a 4-microphysiological systems (MPS) platform (left) and a MPS linking a 3 and 7 MPS to provide an interconnected human 10-MPS plate (right). For the 10-MPS plate, the 3- and 7-MPS plates are connected by microbore tubing which delivers a high flow rate and allows the plates to work as a one well-mixed system. Pneumatic connections deliver air and vacuum and drive pumps across the plates. The 10-MPS plate is approximately 17.5 cm × 25 cm. Reproduced from Eddington et al. (2018).
The microfluidics is designed to maintain a homeostatic and physiologic environment for the tissue or organ and works to control important factors such as oxygen tension, electrolyte balance, pH, and temperature. The tissue devices are formatted in such a way as to recapitulate the organ’s interaction with blood vessels, lymphatics, and muscle, bone, and connective tissue. Design elements are included that help imitate important functions of the tissue of interest such as the pumping action of the heart or the peristaltic movements of the intestine. Individual organs or tissues can be combined to form more complicated organ systems like the reproductive tract or cardiopulmonary system, and tiny sensors and controllers are used to orchestrate the system’s function or measure critical end points of interest. All of this is purposely designed to be scaled for enabling speed, reproducibility, and affordability (Figure 3).

A blood–brain barrier chip. The microphysiological system contains one channel with human endothelial cells and pericytes (visualized using red dye) and the other channel with human astrocytes (visualized using blue dye). Courtesy of the National Center for Advancing Translational Sciences.
The devices generally are formatted as plates or chips. Plates use liquid handling equipment like robotic pipettes and can be integrated with automated microscopes and plate readers (Figure 2). They have the advantage of scalability but lack the complexity that can be developed in chips. Chips use active microfluidic flow and are constructed for complex tissue or organ system MPS (Figure 4). Either approach may be applied in the design of single organ or multiple organ systems. The design can be two-dimensional (2-D) or three-dimensional (3-D) depending on the needs of the testing system. Examples of 2-D systems include monolayer cell cultures which may have a rather singular purpose in design and a specific testing output (e.g., organic ion transport across a hepatocellular membrane), while 3-D system systems like an organ spheroid incorporate more complicated design elements present in an organ system (e.g., drug transport and metabolism through the hepatic lobule). Clearly as the system goes from single organ and 2-D to multiple organ and 3-D, the complexity and variables requiring control and optimization become more onerous. Visionaries imagine the eventual development of a personalized human-on-a-chip system where a minimum number of organs are integrated to produce an amazing testing system of the future. Interconnected MPS that integrate up to 10 organ/tissue types are already a reality (Figure 2; Edington et al. 2018), and private companies with a sole focus on MPS design have been established globally and are collaborating broadly with government and industry (e.g., see TissUse at https://tissuse.com/enandEmulateBioathttps://emulatebio.com/).
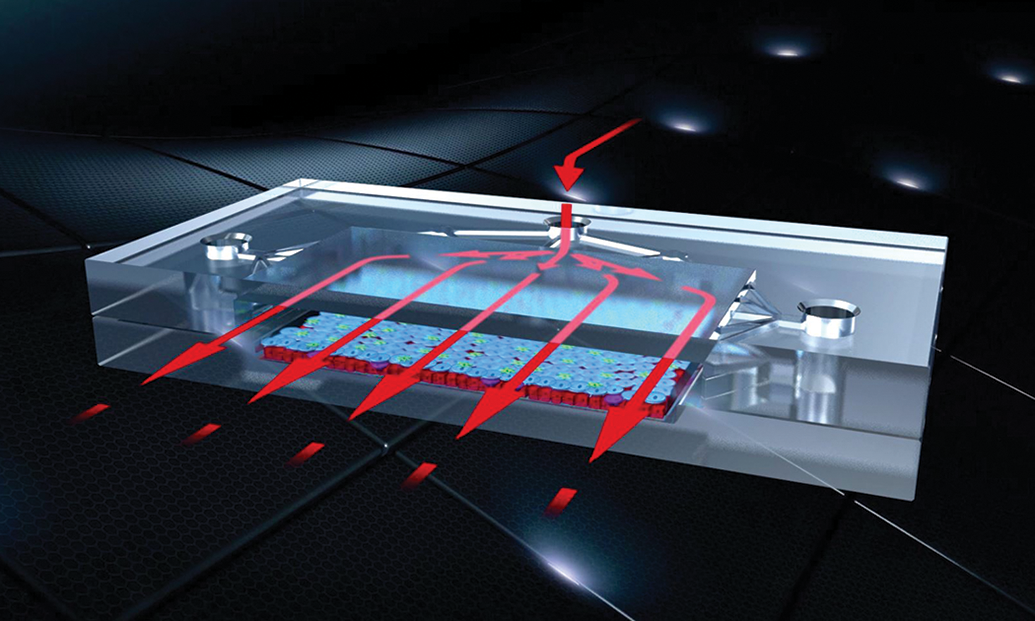
An example of a chip design for a microphysiological system which could be used for complex tissues or organs. Arrows show how fluids can move through the chip and mimic blood, lymph, or cerebrospinal fluid. Courtesy of the National Center for Advancing Translational Sciences.
In the design of these systems, the investigator’s objective is the accurate simulation of the entirety of human tissue or organ biology. This is a formidable task and only has been recently achievable because of great advances in biomaterial research using silicone-based devices in combination with polymers, ceramic or biological matrices, and textiles, all painstakingly designed to imitate the surface pattern, stiffness, and microarchitecture of the organ or tissue (Marx et al. 2016). Several cornerstones are discussed by the T4 group in the successful establishment of MPS technology. The first is the ability of the MPS to emulate the architecture of the human tissue or organ of interest. For example, for the liver, primary human hepatocyte cultures have been very useful in studying drug metabolism and modes of toxicity but are limited by the absence of normal architecture. A successful liver MPS would maintain the intimate relationship within the hepatic lobule, such as central vein, sinusoidal and portal blood flow, oxygen tension, metabolic differences between lobule zones, canalicular biliary flow, and so on.
The second cornerstone is more practical and related to longevity of the MPS itself. Using the primary human hepatocyte example again, testing using this approach requires acute collection of human cells and immediate plating and testing. Longer-term studies using primary human hepatocytes are challenged by the altered phenotype of the cells as they are maintained in a tissue culture system (Ewart et al. 2017). For MPS to have the impact that groups like T4 and IQ envision, as is the case for animal models, maintaining MPS performance over time will drive utility and reproducibility between laboratories located across the world and will be instrumental if MPS is to have maximum impact on drug development.
The third and fourth cornerstones are the most difficult and what truly establish the potential for MPS to replace and reduce the use of animal models. These milestones reflect the ability of MPS to mimic the actual pathophysiology that occurs in animal models and human disease. These include the normal replicative and repair processes specific to an organ or tissue as well as the response of the tissue or organ to a specific disease process (Marx et al. 2016). To monitor the normal function of the MPS and the effects of xenobiotics, the system must be constructed to reliably measure biomarkers using various sensors included in the system. Sensors may be engineered into the MPS or more traditional biomarkers may be measured by accessing the fluid as it leaves the MPS in a single or multiple organ system. In the latter case, standard analyzers could be modified to assess end points commonly examined in toxicology studies (i.e., clinical chemistry).
For MPS development, a dependable source of human cells is a requirement. There are many potential sources of human cells including immortalized cell lines, primary tissue explants or biopsies, embryonic stem cells (ESC), and inducible pluropotential cell (iPSC) lines. Immortalized cell lines have limitations in that with repeated passages, they often do not reflect the normal cell, usually because of loss or gain of various functions. Primary tissue explants or biopsies are directly applicable for MPS use but limited by supply, and ESC may provide excellent potential for MPS designs, but ethical considerations are problematic. The most exciting work in producing a reproducible and reliable source of human cells for MPS is in the use of iPSC lines. These cells are derived from adult donors and can be reprogrammed or differentiated into different primary human cell types. MPS may be the mechanism to take these cells and provide the appropriate conditions for them to form 2-D or 3-D testing systems (Raasch et al. 2018).
Virtually, every organ and tissue is presently being interrogated for use in MPS and specifically in the toxicology specialty (Marx et al. 2016; Truskey 2018). Recent research using multiorgan MPS has demonstrated the potential for these tools in safety assessment (Oleaga et al. 2016; Vernetti et al. 2017). Oleaga and colleagues designed a 4 organ pumpless, gravity-driven system that was animal serum free and composed of a human cardiac, muscle, neuronal, and liver module. Important considerations for the MPS design were the maintenance of physiological relevant oxygen and nutrient distribution and sheer stress within each module (Figure 5). The investigators’ objectives included creating a flexible system whereby the modular system could be adapted and added to using additional tissue/organ types or alternative experimental conditions. It was also important to incorporate sensors that directly measure specific functions of the cell types (e.g., electrophysiology for neurons or contractile activity of cardiomyocytes). The investigators demonstrated stability of the MPS over a 28-day period which is comparable in length to the 1-month investigative new drug enabling good laboratory practices (GLP) toxicology studies commonly done in animal models. Marketed drugs with established side effect profiles were tested in 7-day studies which approximates the frequently used non-GLP dose range finding studies completed during the lead optimization stage of a preclinical program supporting drug development. Doxorubicin, atorvastatin, valproic acid, acetaminophen, and
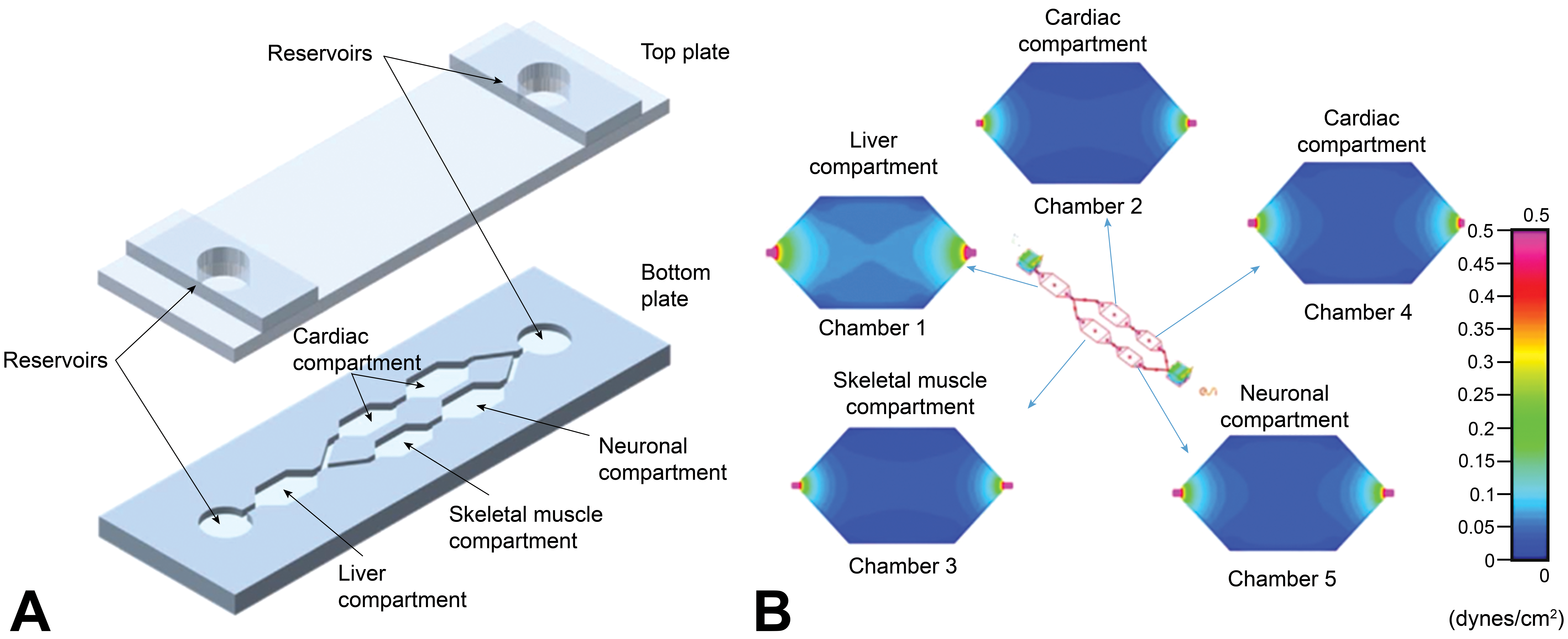
A diagram of the five human tissue microphysiological system platform used by Oleaga et al. (2016). A pumpless, gravity-based system (A) was used to simulate flow and shear stress (B) between the organs. The differential sheer stress across each organ in dynes/square centimeter is indicated by the different colors. Reproduced from Oleaga et al. (2016).
In an impressive effort, Vernetti et al. (2017) led six universities in a collaboration that coupled five organ systems, the intestine, liver, kidney proximal tubule, blood–brain barrier, and skeletal muscle, into a MPS (Vernetti et al. 2017). These investigators modeled the major absorption, metabolism, and clearance pathways for oral exposure of several test compounds by including sequentially the intestine, liver, and kidney within the MPS design. Instead of physically coupling these modules as was done by using a connecting channel, these investigators transferred media (so-called functional coupling) from one organ module, often between universities to the next based on the specific metabolic requirements of the specific organ (Figure 6).
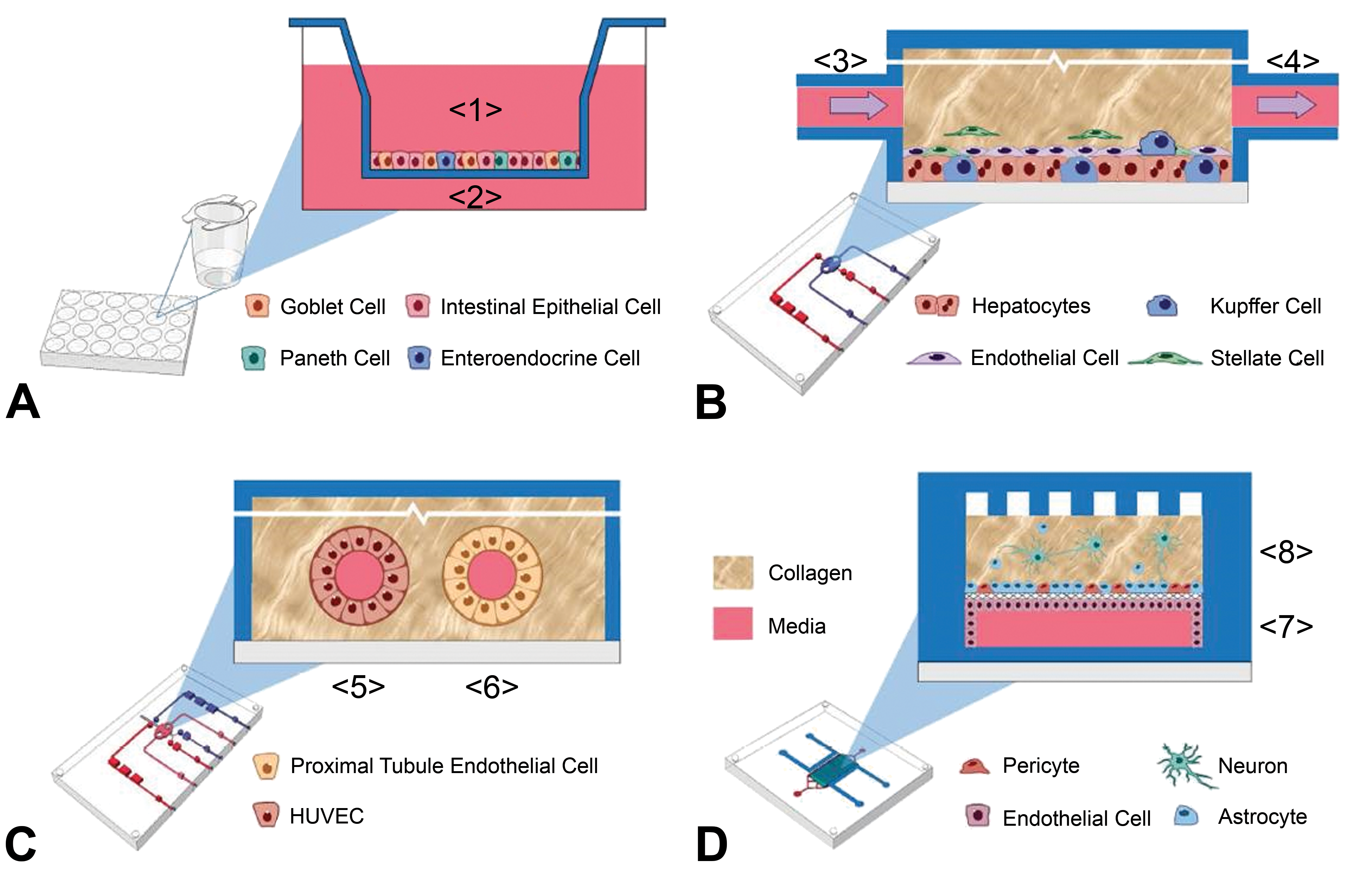
Illustration of the four human organ (intestine-A, liver-B, kidney-C, and brain-D) microphysiological systems (MPS) used by Vernetti et al. (2017) for functional MPS coupling and testing. Numbers 1 to 8 indicate the functional sequence of flow through the MPS. Reproduced from Vernetti et al. (2017).
Three test agents were evaluated in this system: terfenadine, trimethylamine (TMA), and vitamin D3. These compounds were selected to compare their absorption, metabolism, and excretion as well as toxicity within the MPS to clinical data. TMA is an industrial chemical that is also produced in tobacco smoke and present in the diet and is nephrotoxic after intestinal uptake and metabolism by the liver to trimethylamine N-oxide (TMAO). A four-module MPS (intestine, liver, kidney, and blood–brain barrier) successfully recapitulated the expected metabolic and excretory path for TMAO in humans. Terfenadine, an antihistamine removed from the market because of its propensity to prolong the QT interval and cause Torsade’s de Pointes, was evaluated using a four-module MPS (intestine, liver, kidney, and blood–brain barrier) and two-module MPS (liver and skeletal muscle). The four-module MPS predicted the metabolic and excretory path accurately and the two-module MPS ably demonstrated the muscle toxicity caused by inhibiting K+ current (Vernetti et al. 2017).
As many experts have noted, there are several challenges that will need to be overcome before MPS becomes an integrated part of safety assessment (Ewart et al. 2017; Hughes, Kostrzewski, and Sceats 2017; Low and Tagle 2017a, 2017b, 2018; Marx et al. 2016; Vernetti et al. 2017). These include the technical challenges associated with complex multiorgan MPS systems and the scaling of these systems to support automation, validation, and integration into a drug discovery and toxicology laboratory workflow. There are also limits with human cell sourcing which hopefully will be overcome with advances in iPSC technology (Raasch et al. 2018).
Toxicologic pathologists have an immense appreciation of the complexity of the structure and function of the tissue or organ and the interplay of organ systems within the whole animal. We focus on using our training in comparative medicine to assess a xenobiotic’s risk–benefit ratio for humans. MPS technology is developing rapidly but the complexity of the human organism is enormous. As Watson, Hunziker, and Wikswo discuss in a recent
Low and Tagle (2017b) specially bring this challenge forward in a review in the same issue and present both biological and technical challenges of integrating multiple organs-on-a-chip into an MPS homunculus. Some of these challenges were highlighted earlier including sourcing of human iPSC cells and scaling of the organs for use across laboratories. Other biological challenges include the development of a universal media, the appropriate vascularization of the tissue or organ, incorporation of tissue-specific immune components, and adapting the MPS for the influence of hormonal and circadian axes. Technical challenges include issues with xenobiotic adsorption to the chip polydimethylsiloxane, differences in flow rates between platforms, variations in required oxygenation and nutrient levels between different organs, difficulties with incorporation of biosensors for biomarker monitoring, and potential for sterility lapses. Despite these challenges, the pull from regulatory groups like the FDA for advances in MPS is impressive to date as evidenced by the NIH-funded Tissue Chip Testing Center. Data produced from MPS efforts will likely be well received by regulatory agencies currently requiring animal testing to predict human efficacy and safety. As toxicologic pathologists, we are in an excellent position to help be the bridge between the MPS developer, toxicologist, and regulatory scientist.
Biomedical engineers may not know or fully appreciate it yet, but toxicologic pathologists (anatomic and clinical) have the opportunity to collaborate in the development, validation, and application of MPS for human safety and risk assessment. In development, our comparative pathology training could help identify key design elements in MPS that solve specific issues in animal to human model translation and establish their biological and translational relevance (Ewart et al. 2017). Toxicologic pathologists appreciate the intricacy of the animal model and have experienced the pitfalls of translating animal model data to assess human risk. We can use our individual and institutional experiences to prioritize direct methods and biomarkers (e.g., biosensors) for monitoring MPS performance and response to toxic challenge. We understand the animal models and the value they provide in preventing serious adverse events after human exposure. As MPS are evaluated as tools for toxicology, there will be a need for high confidence that these models are equal or superior to present models. Toxicologic pathologists should be part of the team comparing MPS and animal model data. In fact, based on our training and experience, we should be engaged at the ground level in the systematic development and application of MPS within our industry. If MPS validation is done comprehensively and collaboratively, toxicologic pathologists will gain an arsenal of MPS tools that can help us understand the relevance of an animal toxicity to the human. We can partner in the development of novel study designs using these MPS to help interpret animal toxicology data sets. Finally, we can support integration of MPS results with other data sets produced during preclinical development and increase the overall impact and predictability of the data we produce, analyze, and interpret.
How do we best find these opportunities to partner? First, globally, organizations like the Society of Toxicologic Pathologists (STP), European Society of Toxicologic Pathologists (ESTP), and Japanese Society of Toxicologic Pathologists (JSTP), and others can help create training opportunities where newly boarded or board-eligible veterinary pathologists join laboratories developing these methods. The second is for toxicologic pathologists already in industry to seek out and collaborate with their toxicology partners in safety assessment organizations who are investigating these methods. Ideally, NCATS envisions that much of the commercial and industrial validation will be done at contract research organizations (CRO) which provide an additional avenue for involvement for many toxicologic pathologists who work in the CRO industry (Low and Tagle 2017b). Finally, those of us who are in midcareer may have the opportunity for sabbaticals or similar experiences and could consider working with an organization developing and validating MPS. A specific opportunity for both toxicologic clinical and anatomic pathologists is potentially available at two new independent Tissue Chip Testing Centers (Low and Tagle 2017b). These testing centers are working to develop the MPS platforms, so that they will perform reproducibly as a step toward validation. Additionally, a toxicologic pathologist could collaborate with one of the over 12 institutes and centers that are contributing their expertise to MPS development. It is likely within a decade that MPS will be a key tool for our employers, and by embracing it now, we will bring value to the technology and our own expertise as toxicologic pathologists.
Footnotes
Author Contributions
The author (DR) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. The author gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
