Abstract
Introduction:Among major trauma patients in the emergency department, the leading cause of morbidity and mortality is a hemorrhagic shock. The low oxygen flow with hypovolemia in trauma patients is believed to play a significant role. Hence, oxygen supply is essential in severe trauma patients with massive hemorrhage. This study aimed to investigate the effect of oxygen supply in hypoxic condition and variable treatments such as pentoxifylline (PTX), glycerol, hypertonic saline (HTS), protease inhibitor, and dexamethasone (DEXA) in macrophage and T cells. Method:Nitric oxide synthase (iNOS) and macrophage migration inhibitory factor (MIF) were measured for macrophage. MIF, interleukin (IL)-2, and IL-8 were measured for T cells. T cell viability was measued by MTT assay. Results: Pentoxifylline decreased iNOS expression mostly followed by glycerol under hypoxia. Under the hyperoxia, PTX and other treatments decreased iNOS expressions in macrophage. MIF expression was lowered with PTX under hypoxia. PTX, glycerol, HTS, and protease inhibitor were effective under hyperoxia in macrophage. PTX increased T cell survival under hypoxia. Under the hyperoxia, IL-2 expressions were upregulated with PTX, glycerol, and HTS. PTX and other treatments were effective for IL-8. Our results indicate that the PTX and the other agents tested reversed the effects of stimulation of lipopolysaccharide, PGE2 in hypoxia or hypoxia. Conclusion:Our study demonstrated potential usefulness in improving immune systems during severe inflammatory conditions similar to septic shock possibly caused by massive hemorrhage.
Introduction
Among major trauma patients admitted to the emergency department, hemorrhage is the leading cause of morbidity and mortality. Hemorrhagic shock is one of the most common types of shock. 1 Shock is a disequilibrium of the circulatory system. During a shock, a significant difference between vascular loading capacity and intravascular volume distribution leads to decreased tissue perfusion, cellular hypoxia and microvascular aberrations causing metabolic damage, such as anaerobic glycolysis and lactic acidemia. 2 Production of reactive oxygen species by neutrophils and tissue hypoxia is a significant phenomenon in hemorrhage shock-induced inflammatory response.3,4 In the event of various injuries, macrophages that defend early invaders and T lymphocytes that are stimulated by macrophages play an important role in immunity from a cellular immunological aspect.
However, despite proper resuscitation for severe trauma and massive hemorrhage, early systemic inflammatory response syndrome caused by excessive inflammation and later compensatory anti-inflammatory response syndrome caused by immunoparalysis have resulted in multiple organ dysfunction syndrome or sepsis. 5 The diminished flow of oxygen associated with hypovolemia in trauma patients is believed to play a significant role in shock and cell damage; however, the specific mechanism has yet to be identified. There is also controversy over the benefits of the supply of high concentration oxygen in addition to oxygen in the atmosphere to shock patients. We do not know the influence of hypoxia in a damage mechanism of cell unit with shock that can occur at an early stage of trauma, nor the influence of hypoxia on hyperinflammatory macrophages and T cells that are important to immunity.
This study aimed to investigate the effect of high concentration of oxygen supply in hypoxic condition and variable treatments such as pentoxifylline (PTX), glycerol, hypertonic saline (HTS), protease inhibitor, and dexamethasone (DEXA) in macrophage and T cells. In other words, we would like to investigate oxygen and various treatments for hyperinflammation and immunoparalysis that affect sepsis from severe trauma and massive hemorrhage.
Materials and methods
Cell culture and cell stimulation
Human acute monocytic leukemic cell line (THP-1 cells) (ATCC TIB-202, Manassas, VA, USA) and lymphocytic leukemic cell line (Jurkat cells Clone E6-1) (ATCC TIB-152, Manassas, VA, USA) each were maintained in RPMI-1640 (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum, 2 mM glutamine, 10 mM HEPES, 100 U/mL penicillin/streptomycin at 37oC in 5% CO2 incubator. Cells were cultured to a density of 5 × 105 cells/mL. Cell viability, as determined by tyropan blue dye exclusion, was >99%. For macrophage differentiation, the THP-1 cells were prepared in a 75T-flask and 1 µL/mL of PMA (sigma-Aldrich Co., St. Louis, MO, USA) was added for 3 days. The supernatant was discarded and washed with 10 mL of phosphate buffer saline (PBS), followed by the addition of 5 mL of trypLETM express (Gibco Co, Denmark) to take the attached cells off the floor. After the addition of 10 mL of fresh medium and the reaction mixture `was centrifuged for 5 min at 500 × g get the differentiated THP-1 cells. Therefore, THP-1-derived macrophages and Jurkat cells were used for the experiments. Prostaglandin E2 (PGE2) (1 μg/mL) (Sigma-Aldrich Co., St Louis, MO, USA) stimulation was used to demonstrate the effect of immunosuppression. Lipopolysaccharide (LPS) (1 μg/mL) (Sigma-Aldrich Co., St Louis, MO, USA) induction was used to simulate the effect of endotoxin.
First, the differentiated THP-1 cells were plated at a density of 1 × 104 cells/mL in 24 well plates and were placed under normoxia (20% O2) followed by hypoxia (1% O2) for 30min. After that, macrophages were stimulated with LPS (1 μg/mL) followed by variable treatments such as PTX, glycerol, HTS, protease inhibitor, and DEXA, followed by normoxia (20% O2) or hyperoxia (80% O2) for 2 h. After 12 h, the levels of macrophage migration inhibiting factor (MIF) and inducible nitric oxide synthase (iNOS) expression were measured by western blots and concentration of MIF in supernatant was measured by ELISA. Second, Jurkat cells (2 × 106 cells/mL) were subjected to normoxia followed by hypoxia for 30 min. After that, Jurkat cells induced with PGE2 (1 μg/mL) with various treatments such as PTX, glycerol, HTS, protease inhibitor, and DEXA, followed by either normoxia or hyperoxia for 2 h. After 12 h, their metabolic state, as indicated by cell survival, was measured using an MTT assay. The levels of MIF, interleukin 2 (IL-2), and interleukin 8 (IL-8) expression were measured by Western blots. Hypoxic insult is referred to as cells cultured under hypoxic conditions. Hyperoxic treatment is administered to cells with hypoxic insult followed by hyperoxia. This study was carried out with the approval of the Korea University Guro Hospital Institutional Review Board (approval number: 2019GR0188).
Enzyme-linked immunosorbent assay (ELISA) for MIF
The MIF concentration in the culture supernatants was measured by ELISA (RayBio® Cat# ELH-MIF-001). The optical density at 450 nm was measured on an automated microplate reader (Bio-Rad Laboratories Inc., Hercules, CA, USA). A standard curve was generated by plotting the optical density versus the log of the MIF concentration.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) viability assay
The tetrazolium dye, MTT, is widely used to assess the viability or the metabolic state of the cells. The MTT-colorimetric monocyte mediated cytotoxicity assay is based on the ability of living cells to reduce MTT into formazan by mitochondrial succinate dehydrogenase in viable cells. After treatment at the different culture conditions, Jurkat cells were plated in 96-well flat-bottom tissue culture plates to attain a final concentration of 2 × 106 cells/mL. After incubation for 12 h at 37oC, the resultant Jurkat cell viability was determined by the MTT viability assay (ATCC, Manassas, VA, USA).
Western blot analysis for nitric oxide synthase, MIF, IL-2 and IL-8 expression
The cells were washed 2 times in cold PBS and then centrifuged for 10 min. Cell pellets were suspended in 10 μL per 2 × 106 cell/mL pro prep protein extraction buffer. Incubated on ice for 10 min and then centrifuged at 3000 × g for 15 min at 4°C. The total protein concentration was determined by the Bradford method using a Bradford solution (Sigma Co. Louis, MO, USA). The prepared protein was used for Western blot analysis. Expression of iNOs, MIF, IL-2, and IL-8 proteins were quantified by Western blot analysis. The primary antibodies used anti-human iNOs, MIF, IL-2, and IL-8 (1:500; R&D systems) and the secondary antibody used 1:1000 horseradish peroxidase-labeled anti-rabbit antibody (R&D systems).
Data and statistical analysis
All results were expressed as the mean ± SD. Statistical significance was performed using a t-test, one-way ANOVA, and the Mann–Whitney U test using SPSS 18.0 (SPSS Inc, Chicago, IL). Each experiment was repeated twelve times at least.
Results
The effect of oxygen and medicines in the nitric oxide synthase expression in macrophages in hypoxic condition
The mean iNOs expression (iNOs/Actin) ratio was increased significantly when cells were exposed to hypoxia, compared with normoxic conditions ( iNOs expression in macrophages under hypoxia and oxygen supply. After hypoxic insult (1% O2), macrophage was stimulated with LPS. The iNOS expression was significantly increased after hypoxic insult and LPS stimulation (from 1.00 to 3.79, 3.16), and the iNOS expression was most significantly restored with PTX and followed by glycerol (from 3.16 to 0.35, 0.98). The oxygen supply under LPS and hypoxic insult, the iNOS expression was restored with PTX and followed by HTS, protease and glycerol, respectively, compared to oxygen and LPS stimulation (from 1.03 to 0.54, 0.68, 0,72 and 0.87). Hypoxia means 1% oxygen supply in 30 min. Hypoxia-oxygen supply means that 80% of oxygen is supplied for 2 h after 1% oxygen supply for 30 min. Data are presented as mean ± SD. *
The effect of oxygen and medicines in the MIF expression in macrophage in hypoxic condition
The mean MIF expression (MIF/Actin) ratio was significantly elevated after the hypoxic insult compared to normoxic conditions ( MIF expression in macrophage under hypoxia and oxygen supply. After hypoxic insult (1% O2), macrophage was stimulated with LPS. The MIF expression was significantly increased after hypoxic insult (from 1.00 to 1.49), and the MIF expression was restored with PTX, compared to hypoxic injury (from 1.49 to 1.06). The MIF expression was lowered with PTX, glycerol, HTS, and protease inhibitor when oxygen was administered to LPS-stimulated macrophage under hypoxic insult (from 0.90 to 0.65, 0.67, 0.75, 0.71). Hypoxia means 1% oxygen supply in 30 min. Hypoxia-oxygen supply means that 80% of oxygen is supplied for 2 h after 1% oxygen supply for 30 min. Data are presented as mean ± SD. *
The effect of oxygen and medicines in the cell viability of Jurkat cells (MTT)
Jurkat cells survival was decreased significantly by hypoxic condition ( Cell viability in Jurkat cell under hypoxia and oxygen supply. After hypoxic insult (1% O2), Jurkat cell was stimulated with PGE2. MTT value was significantly decreased after hypoxia and PGE2 stimulation (from 1.00 to −13.99, −21.29), and MTT value was restored with PTX and followed by glycerol (from −21.29 to −1.69, −6.91). The MTT value was increased significantly with PTX when oxygen was administered to PGE2-stimulated Jukat cell under hypoxic insult (from −1.86 to 10.63). Hypoxia means 1% oxygen supply in 30 min. Hypoxia-oxygen supply means that 80% of oxygen is supplied for 2 h after 1% oxygen supply for 30 min. Data are presented as mean ± SD. *
The effect of oxygen and medicines in the IL-2 expression in Jurkat cells
After hypoxic insult and PGE2 stimulation, the IL-2 expression was a little decreased without statistical significance. However, various agents did not restored IL-2 expression in hypoxic condition. In the oxygen supply under hypoxic conditions, the IL-2 expression was restored significantly by the addition of PTX, glycerol, and HTS in cells subjected to hypoxia and PGE2 stimulation ( IL-2 expression in Jurkat cell under hypoxia and oxygen supply. The IL-2 expression was a little decreased without statistical significance after hypoxic insult (1% O2) and PGE2 stimulation (from 1.00 to 0.91, 0.82); however, various medicines did not restored the IL-2 expression. In the oxygen supply in hypoxic Jurkat cells, the IL-2 expression was restored by pentoxifylline, glycerol, and hypertonic saline, compared to the oxygen supply in PGE2-stimulated cells under hypoxia (from 0.79 to 0.93, 0.90 and 1.09). Hypoxia means 1% oxygen supply in 30 min. Hypoxia-oxygen supply means that 80% of oxygen is supplied for 2 h after 1% oxygen supply for 30 min. Data are presented as mean ± SD. *
The effect of oxygen and medicines in the IL-8 expression in Jurkat cells
The hypoxia increased a little the IL-8 expression without statistical significance. PTX, glycerol, HTS and Dexa significantly downregulated IL-8 expression in PGE-stimulated Jurkat cells under hypoxia ( IL-8 expression in Jurkat cell under hypoxia and oxygen supply. The IL-8 expression was increased after hypoxia and PGE2 stimulation (from 1.00 to 1.26), and the IL-8 expression was significantly decreased with Dexa and followed by PTX, glycerol, and hypertonic saline, respectively (from 1.26 to 0.93, 0.99, 1.00 and 1.05). PTX and Dexa significantly further downregulated IL-8 expression when oxygen was administered to PGE2-stimulated Jukat cell under hypoxic insult (from 1.08 to 0.73, 0.83). Hypoxia means 1% oxygen supply in 30 min. Hypoxia-oxygen supply means that 80% of oxygen is supplied for 2 h after 1% oxygen supply for 30 min. Data are presented as mean ± SD. *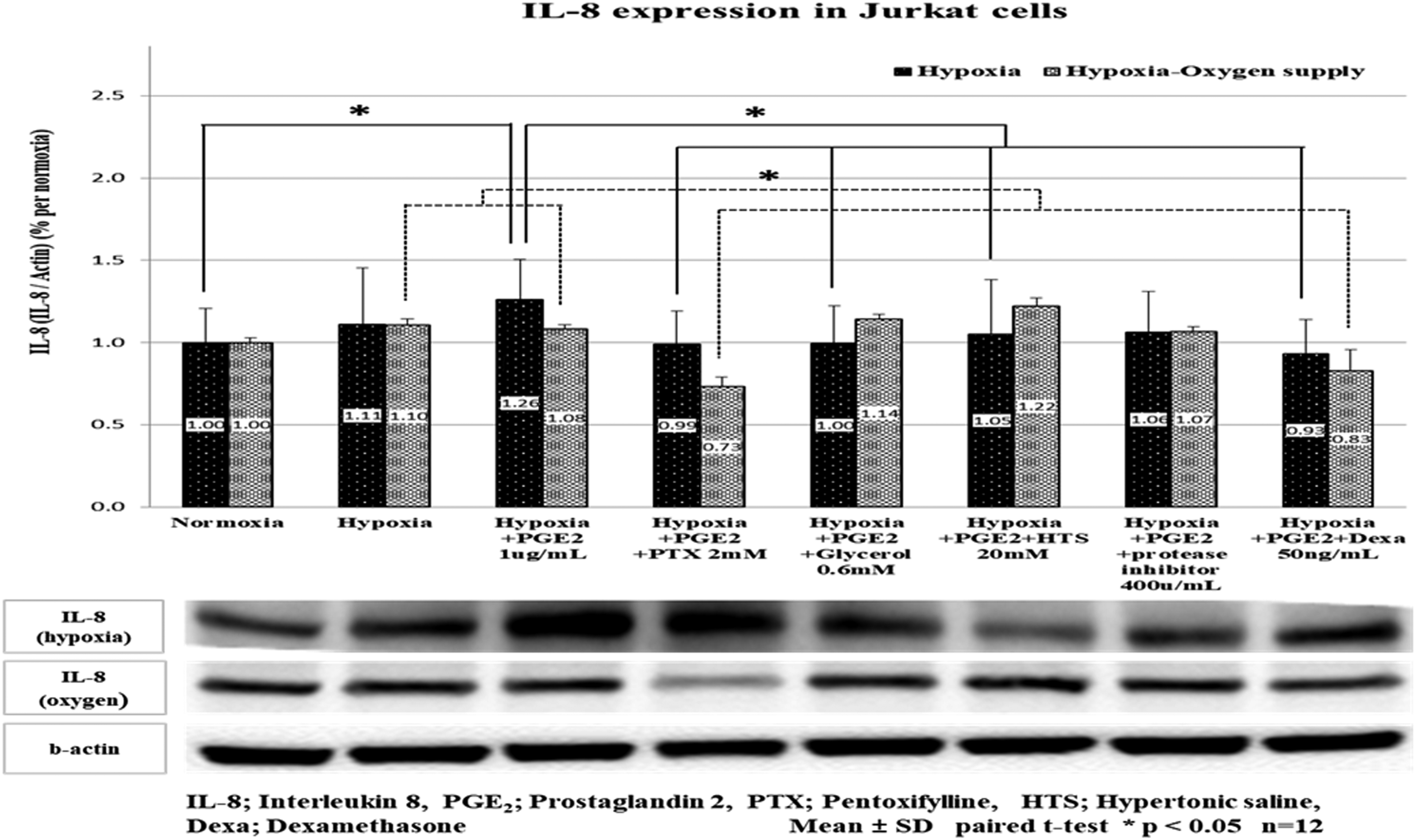
The effect of oxygen and medicines in the MIF expression in Jurkat cells
The hypoxia increased a little the MIF expression without statistical significance. PTX, protease inhibitor and Dexa significantly downregulated MIF expression in PGE2-stimulated Jurkat cells under hypoxia ( MIF expression in Jurkat cell under hypoxia and oxygen supply. The MIF expression was increased after hypoxia and PGE2 stimulation (from 1.00 to 1.25); however, Dexa, protease inhibitor, and pentoxifylline restored the MIF expression in PGE2-stimulated Jurkat cells under hypoxia (from 1.25 to 0.88, 0.88 and 0.93). Dexa downregulated MIF expression when oxygen was administered to PGE2-stimulated Jukat cell under hypoxic insult (from 1.24 to 0.93). Hypoxia means 1% oxygen supply in 30 min. Hypoxia-Oxygen supply means that 80% of oxygen is supplied for 2 h after 1% oxygen supply for 30 min. Data are presented as mean ± SD. *
Discussion
It is important to maintain the homeostasis of trauma patients until surgical treatment in order to alleviate hyper-inflammatory and immune-paralysis conditions to prevent the occurrence of post-traumatic secondary complications, such as sepsis, multiple organ failure. And also hypoxia in trauma patients should be play a important role in shock. 6 Data derived from recent studies showed that sepsis was associated with a higher risk of long-term mortality. 7 In the past, inflammation was believed to cause early mortality and the anti-inflammatory compensatory response caused organ failure, immune suppression, and mortality subsequently. 8 Nevertheless, analysis of septic tissue samples and severe trauma revealed sustained and concurrent inflammatory and anti-inflammatory state caused by dysfunctional innate and suppressed adaptive immunity, which together resulted in prolonged organ injury and mortality. 9 Our previous study showed the ameliorating effect of PTX in neuronal cells subjected to hypoxia. Further, the combined exposure to HTS and PTX resulted in a possible neuroprotective effect in neuronal cells after hypoxic injury. 10 Regulation and balancing have been known to play an important role in sepsis control. Massive blood loss triggers hemorrhagic and septic shock and eventually leads to death in the absence of appropriate intervention in severe trauma patients. Therefore, this study investigated the effects of oxygen supply and variable medicines on macrophages and T cells under hypoxia.
Macrophages, which are derived from bone marrow monocytes, play a vital role in the inflammatory response following hypoxia or infection. Macrophages induce the production of high levels of pro-inflammatory cytokines. Macrophages synthesize high levels of reactive nitrogen and oxygen intermediates. They are microbicidal and enhance the ability to resist pathogens. In vitro, macrophages are classically activated by the bacterial cell wall component LPS. Recognition of pathogen-related molecular patterns such as endotoxin (LPS) ignites the activation. 11 Also, the reduced capacity of blood monocytes after endotoxin stimulation in sepsis is associated with poor short- and long-term outcomes. 12 T cells play a major role in immunity, which has been reduced in sepsis and multiple organ failure.13,14 For the experiment, PGE2 was used as a representative of immunosuppressive substances. PGE2 is an inflammatory mediator derived from arachidonic acid following the catalytic action of PGE synthases and cyclooxygenase isoenzymes (COX-1 and -2). 15 The PGE2 suppression on T cells, natural killer cells and macrophages is mediated via specific receptors (EP2 and EP4). The interaction between PGE2 and specific receptors activates multiple pathways underlying the induction of anti-inflammatory and immunosuppressive genes. 16
It is well known that tissue injury and multiple organ dysfunction occur in sepsis when excessive iNOs production induces nitric oxide (NO) synthesis.17-19 In our research, the increased iNOs after hypoxia and LPS stimulation was restored to PTX and glycerol injection, and the production of iNO was restored to PTX, HTS, protease inhibitors, glycerol when oxygen was supplied. Therefore, hypoxia increased the iNOs that were important to the occurrence of shock, was able to see the efficacy of oxygen injection and PTX, and had similar effects on MIF, indicating that they had an effect on inflammatory reactions.
As in other studies, cell proliferation in hypoxia has decreased and further decreased by PGE2. 20 However, PTX and glycerol treatments restored Jurkat cell viability under PGE2 stimulation, which simulates immunosuppression. Interestingly, the results of IL-2 expression differed under hypoxia and high concentration of oxygen supply. In hypoxia, the PGE2 stimulation decreased the expression of IL-2, and the IL-2 expression was not restored by variable agents, whereas in the high concentration of oxygen supply in hypoxic condition group, the IL-2 expression was restored by PTX, glycerol, and HTS. IL-2 plays an important role in immune homeostasis, especially in determining the magnitude and duration of primary and memory immune responses and plays an essential role in downregulating immune responses. The loss of IL-2 leads to severe autoimmunity due to abnormal activation of T cell elimination.21,22 IL-2 is known for anti-apoptotic signaling, and effects on glycolysis and cellular metabolism, which are essential for the long-term survival of T cells.23,24 The contrasting results under hypoxia and high concentration of oxygen supply in hypoxic condition are attributed to the effect of oxygen. Therefore, it is estimated that there are various multiple factors other than IL-2 in hypoxia when it comes to the proliferation of T cells. In hypoxia-insulted Jurkat cells, stimulation with PGE2 increased the IL-8 expression, whereas treatment with various agents decreased the IL-8 expression. Following high concentration of oxygen supply in hypoxic condition, exposure to PTX and Dexa significantly decreased IL-8 expression. IL-8 is used as an evaluation factor of immunity that results in sepsis and multiple organ failure in traumatic patients and is an chemokine and angiogenic factor produced by alveolar macrophages, T cells, and epithelial cells in response to a variety of stimuli, including LPS, IL-1, and hypoxia. IL-8 increased in hypoxia, as in other studies, to show that it was related to the suppression of cell proliferation.25,26 PTX in our study shows meaningful results by restoring IL-8 during hypoxia and high concentration of oxygen supply in hypoxic condition. MIF antagonizes glucocorticoid inhibition of T cells proliferation in vitro by restoring IL-2 and IFN-γ production, as other studies, and plays a critical role in the host control of inflammation and immunity. 27 Although there was no change in hypoxia in our study, we could see that there was also a change in hypoxia condition as it was increased in injection of PGE2 and restored by PTX, protease inhibitor and Dexa. In high concentration of oxygen supply in hypoxic condition, the results are similar to hypoxia, which is different from IL-2, so research on hypoxia will be needed in the results are different from normal oxygen groups.
We investigated using clinically used treatments such as PTX, glycerol, HTS, protease inhibitor, and DEXA. It is known that there are no effective medicines other than adequate oxygen and fluid to maintain proper blood flow in hypoxia and shock. However, glycerol, which has the effect of reducing cell edema caused by shock and hypoxia, was significant in our study to restore the proliferation of T cells from the suply state of high concentration of oxygen as well as anti-inflammatory effects. The protease inhibitor used was a urinary trypsin inhibitor extracted and purified from human urine. It is an intrinsic serine-protease inhibitor that suppresses the infiltration of neutrophils and release of elastase and chemical mediators. Urinary trypsin inhibitors are believed to have an anti-inflammatory effect, although their role in the T cells was minimal, as is used in pancreatitis, septic shock, and hemorrhagic shock. Corticosteroids are the most investigated and commonly used drugs for a wide range of inflammatory conditions; however, the effects on macrophages and T cells in hypoxia are minimal and further research is required. PTX is effective in enhancing cell viability, iNOs production, and the expression of IL-2, -8, and MIF. In addition to altered red cell deformity and improved microcirculatory blood flow, the attenuation of inflammatory response after the hemorrhagic shock has been established by the activity of PTX as an adjuvant to conventional fluid resuscitation in hemorrhagic shock. 28 HTS demonstrated equivalent effectiveness in attenuating inflammatory conditions in this study. HTS has been used widely as a volume expander, and the number of studies showed enhanced microcirculatory blood flow and neutrophil adhesiveness after HTS resuscitation. 29 This will require the development of various medicines or the research of combinations of medicines.
This research has some limitations. Instead of using the monocytes and T cells, we conducted an experiment using THP-1 cells and Jurkat cells. The THP-1 cell and Jurkat cell lines, a human acute monocytic leukemic cell and lymphocytic leukemic cells, is believed to be partially different from the true monocyte and lymphocyte cells, but is widely used in various studies such as cell proliferation, cell therapy, which is thought to be suitable for use in various literature. Second, clinical trials have not been conducted, but it is expected if sufficient research is done; we will proceed later
Conclusions
Our results indicate the effect of PTX, HTS, and other agents administered with oxygen therapy on macrophage and Jurkat cells under various insults and stimulations, and the PTX and the other agents tested reversed the effects of of stimulation of LPS, PGE2 in hypoxia or hypoxia. Also, our study demonstrated the potential usefulness in improving both innate and adaptive immune systems in severe inflammatory conditions similar to septic shock possibly caused by massive hemorrhage. Clinical studies in the future are required to overcome the limitations associated with this in vitro study.
Footnotes
Acknowledgments
We are grateful to our colleague Qiuyu C, Bang IY, and Korean shock society member for participation in the study and helpful discussion and we appreciated Song DJ who conducted a statistical review.
Author contributions
YDC, SJP, and SHC conceived the idea. JYK, SHC, CSL, and TGS designed the experiments and interpreted the data. YDC, WSY, HJC, KHK, and SHC performed the experiments. YDC and SHC wrote the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2018R1A2B6004283) and partially supported by a Korea University Grant.
Ethics approval
Ethical approval for this study was obtained from „Korea University Guro Hospital Institutional Review Board (Approval Number/2019GR0188).
