Abstract
Crystalluria can involve the kidney and lower urinary tract, can produce acute and chronic effects, and occurs in all mammalian species. Most commonly urinary crystals contain calcium. Numerous other endogenous and exogenous substances can produce crystalluria. Crystals are identified in kidneys of many species, up to 100% in certain rat strains. More severe renal disease (acute tubular necrosis and chronic renal disease) can be secondary to crystal accumulation, such as observed with melamine–cyanuric acid in cats and dogs. Aggregation of crystals leads to calculi that act as urothelial abrasives with consequent regenerative proliferation. Accumulation in the kidney pelvis or bladder can lead to partial or complete obstruction and hydronephrosis. Long-term presence of urinary tract calculi in rodents leads to increased risk of urothelial tumors, but not in humans. Crystals in the lower urinary tract can act as irritants in rodents, but not in humans. It is critical that specific procedures are followed to optimize the presence of crystals in urine for diagnosis, including not fasting the animals. Numerous factors have been identified which can enhance or inhibit crystal formation. Extrapolation from animals for the threshold toxicity of crystals/calculi is appropriate but is not relevant for cancer risk assessment.
Keywords
Crystals are a normal component of the urine in the lower urinary tract and can also occur in the renal tubules (Karcher, McPherson, and Pincus 2017). In toxicologic pathology, the most common change observed in an assessment of the rodent urinary system is the frequent presence of calcium-containing crystals in the tubules of rats at the corticomedullary junction (Frazier et al. 2012; Lord and Newburne, 1990). With special stains, these can be demonstrated to be present in nearly all rats by 3 months of age. They tend to increase in number with the aging of the animals but do not appear to have toxicologic or functional significance.
Similar calcium crystals can occur in rodents and in humans in other parts of the tubule and in greater numbers, most notably after the ingestion of large amounts of ethylene glycol administered to rodents, other species or ingested as antifreeze by individuals mistaking it for alcohol (Guo et al. 2007). Under these circumstances, there are large amounts of calcium oxalate crystals deposited in the tubules which lead to acute renal failure. These can be readily visualized on a standard hematoxylin and eosin (H&E)-stained section but can be highlighted by evaluating the kidney tissue with polarized lenses as the crystals are birefringent.
Crystals can form in the urinary tract following ingestion of a wide variety of substances (Table 1). It is critically important to identify whether the crystals are composed of the administered material (or metabolites) or whether they are composed of endogenous substances such as calcium, urate, or cysteine. The approach for these in defining toxicologic effects and modes of action is different (Cohen et al. 2002).
Substances Producing Urinary Calculi When Administered to Rodents and/or Humans.
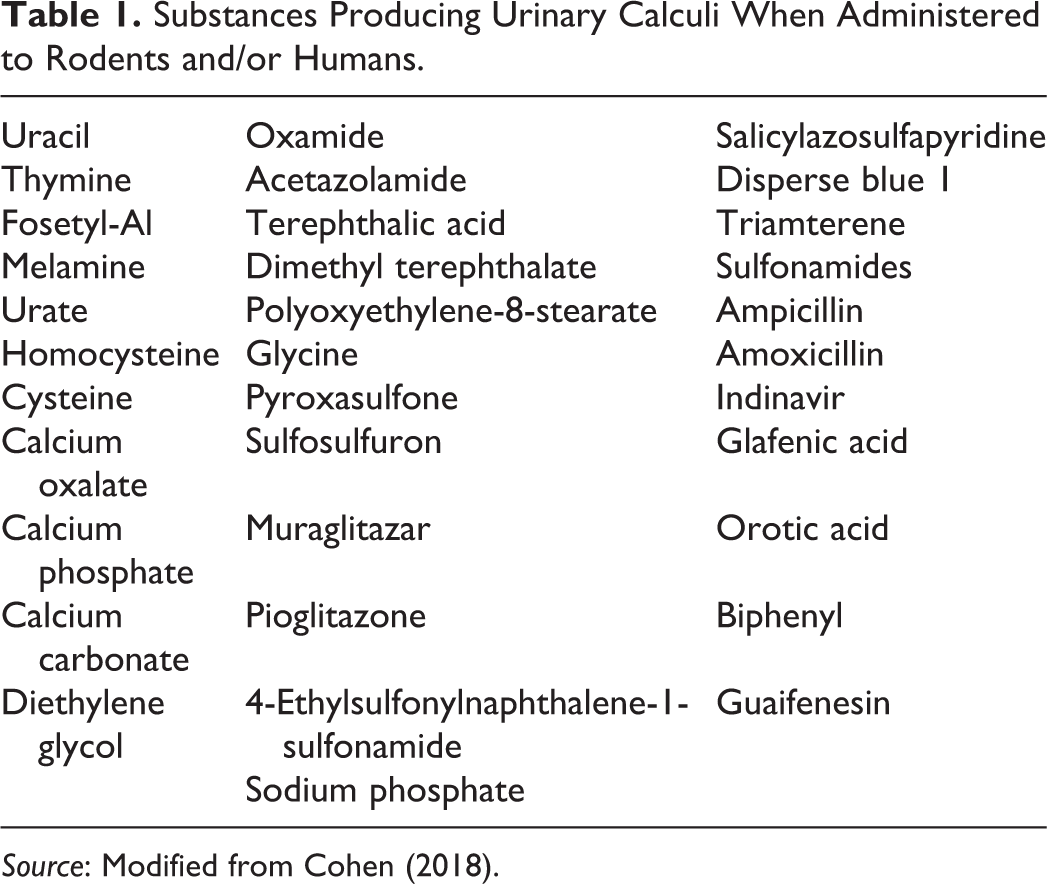
Source: Modified from Cohen (2018).
Melamine
More than 3 decades ago, melamine was demonstrated to form crystals in the lower urinary tract when administered to rats and mice (Dapson and Rinde 1993). In rats, there was an increased incidence of urothelial tumors, primarily in the urinary bladder. Melamine is a substance that is soluble when exposures are small, but when large amounts (3% of the diet to the rats) are administered, the solubility is saturated and crystals form. This is expected to occur in all species, not only in rodents but also in cats, dogs, and humans, if sufficiently large quantities are ingested. This was exemplified in an episode in which pet foods were adulterated with high levels of inexpensive melamine to artifactually produce a high nitrogen reading indicating high protein content in the pet food products because of the large percentage of nitrogen present in melamine (Dobson et al. 2008; Chen et al. 2009). This resulted in acute renal failure in a large number of cats and dogs seen by veterinarians. The source of the renal effects was quickly delineated by Daston and his colleagues (2008) at Proctor and Gamble as being due to the formation of melamine crystals. In addition, these pet food products had been adulterated not with pure melamine but with a mixture of melamine and cyanuric acid. A complex of the nonring nitrogen of melamine with the nonring oxygen of cyanuric acid lead to strong hydrogen bonding and a distinct lattice crystalline structure which is much less soluble than either melamine or cyanuric acid alone (Figure 1). This led to the precipitation of crystals composed of these two substances at lower concentrations than would have been anticipated for either of these substances alone.
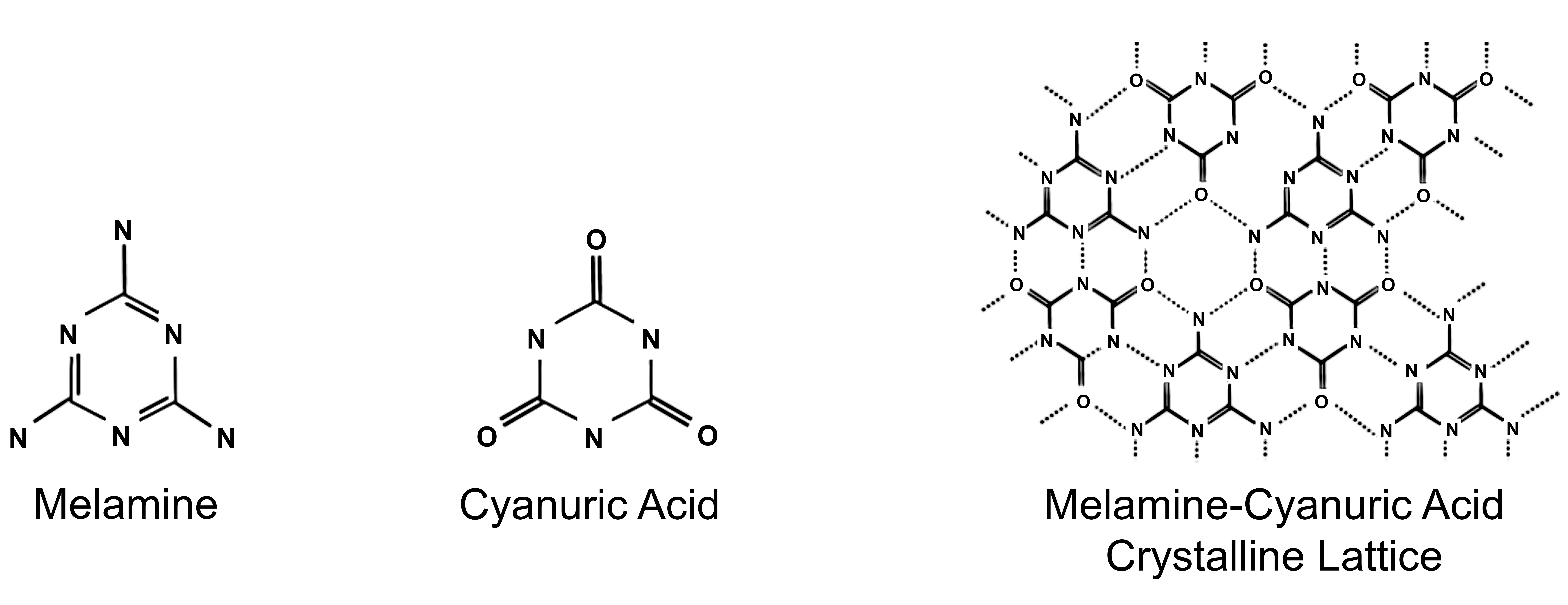
Chemical structures of melamine, cyanuric acid, and the crystalline lattice formed by hydrogen bonding of them. Source: From Dobson et al. (2008), with permission.
Tragically, a similar event occurred in human infants fed infant formula that had been adulterated with melamine (Guan et al. 2009; Gossner et al. 2009). The formula was their only source of nourishment. A large number of these infants developed acute renal disease and a few died.
These events highlight the overall mode of action of these substances based on well-defined physical chemical principles, that is, solubility. As long as the dose of the substance is below the concentration which produces crystals in the urine, there is essentially no toxicologic effect. Once the solution is saturated, crystals form and toxicologic responses occur in the urinary tract, particularly if the substance is excreted and concentrated in the urine. Urinary concentration is the critical parameter for comparison between species, not the blood level. The toxicologic effects related to these crystals is a well-defined threshold event (Cohen et al. 2002).
Retrograde Nephropathy
One of the long-term consequences of crystals in the urine is development of an entity referred to as retrograde nephropathy. The histopathology of this entity has been well described by Hard, Flake, and Sills (2009) in a study involving melamine administration to F344 rats. Tubule dilatation and tubule basophilia can be traced in the tubules from the papilla all the way through the medulla into the cortex (Figure 2). By the end of a 2-year experiment in rats, there is tubular loss and collagen deposition resulting in scar formation. It is particularly critical to distinguish this lesion from chronic progressive nephropathy and from α2u-globulin nephropathy in male rats. Similar changes with melamine have been reported in pets that have ingested the adulterated pet food and in infants fed the adulterated formula.
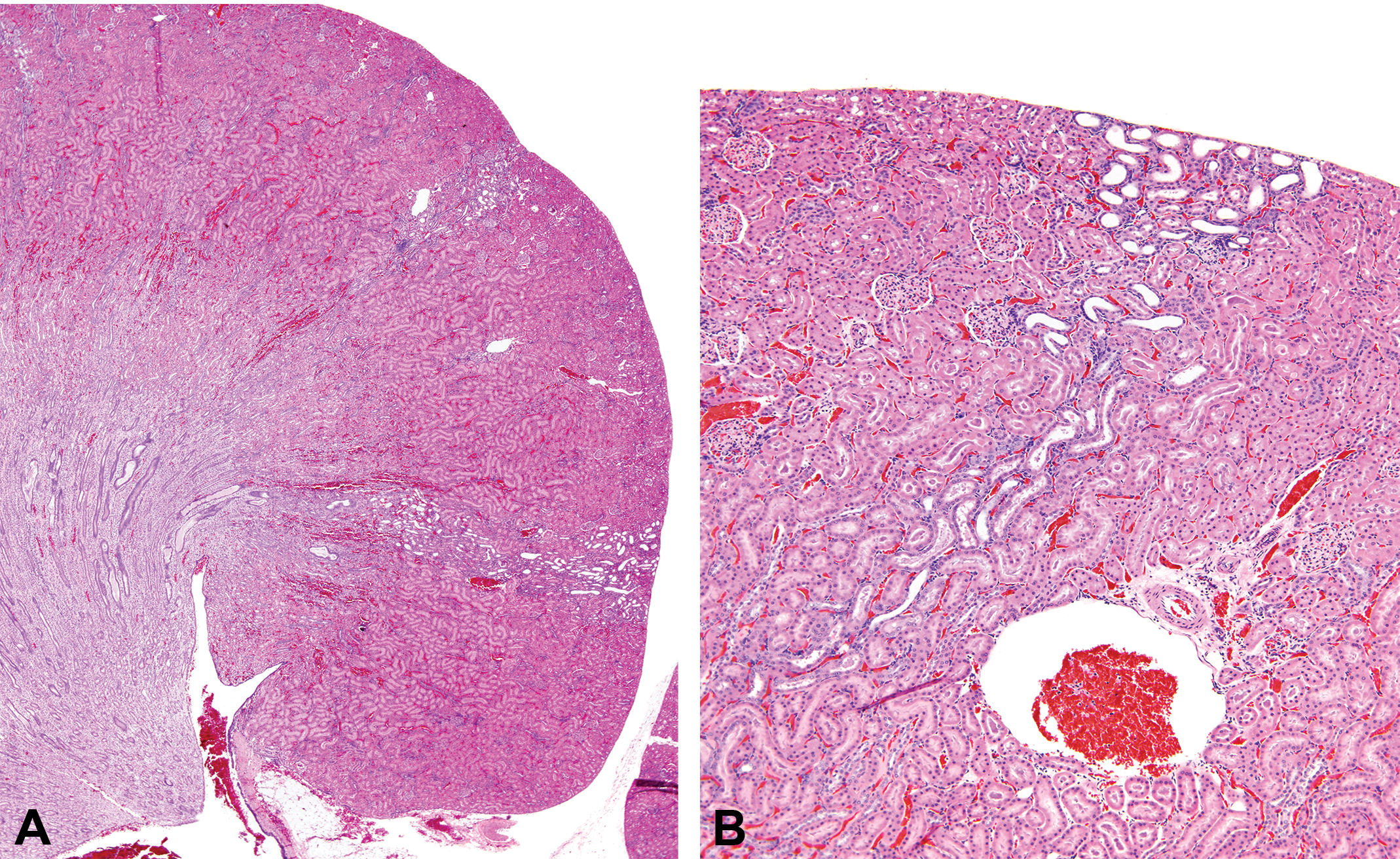
Retrograde nephropathy showing basophilic tubules tracking axially from the medulla through the cortex (low and high magnification). Source: From Hard, Flake, and Sills (2009), with permission.
Urothelial Effects
In rodents, the presence of crystals and calculi in the lower urinary tract can lead to chronic irritation of the urolthelium and regeneration with the ultimate formation of urothelial tumors (Clayson, Fishbein, and Cohen 1995; Cohen et al. 2002). This has been demonstrated not only with melamine but with a large number of other substances (Table 1), with the rat usually more susceptible than the mouse, and the male rat usually more susceptible than the female rat. The urinary crystals and calculi act as an irritant on the urothelial surface and lead to an increased incidence of urothelial tumors in rodents.
The mode of action analysis shows that the key events are ingestion of high doses of the test material leading to formation of the urinary tract crystals or calculi producing urothelial toxicity and consequent urothelial proliferation. Tumors occur in rodents if this persists for a long period of time.
The question is whether these tumorigenic effects occur in all species, whether rodents, pets, or humans. It turns out that the urothelial tumors secondary to urinary crystals or calculi do not predict to tumors in other species such as cats, dogs, nonhuman primates, and humans (Cohen et al., 2018). Interestingly, cats frequently develop urinary tract calculi, and yet lower urinary tract tumors in cats are exceedingly rare (Robinson et al. 2008). There are a variety of reasons for the lack of tumorigenesis in humans secondary to calculi but most notably is the difference in anatomy between the rodent and the human (DeSesso 1995; Liapis et al. 2015). Calculi certainly can occur in humans. The rodent is horizontal, so calculi can accumulate in the dome of the bladder without completely obstructing the urinary flow and thus can be present for the lifetime of the animal. In contrast, in humans the lower urinary tract has several points of narrowing where calculi lead to complete obstruction: where the kidney pelvis narrows to become the ureter, where the ureter goes over the pelvic brim, and where the ureter enters the wall of the urinary bladder. Of course, there is the outflow obstruction through the urethra. In humans, when calculi produce obstruction, an individual experiences excruciating pain with a demand for immediate removal of the calculus. Consequently, in humans, calculi are present for short periods of time and tumors do not develop. There are a few unusual circumstances where calculi can be present in the urinary bladder of humans for long periods of time, such as bladder diverticuli or neurogenic bladder secondary to paraplegia. Under these circumstances, the calculi nearly always become infected with bacteria, and there is a slight increased risk of developing urothelial tumors. However, this increased risk appears to be due to the bacterial cystitis, as chronic bacterial cystitis without calculi increases the risk of urothelial tumors at this same level (Cohen et al. 2002, 2007). Consequently, most regulatory authorities consider urinary tract crystals and calculi not to be relevant to human carcinogenesis. However, it is important to keep in mind that formation of urinary calculi is a toxic endpoint in humans, just like it is in other species, so determining this in toxicology studies is important for the risk assessment. The risk in humans for these noncancer toxicities secondary to calculi is similar as in rodent and nonrodent test species. However, the tumors that appear in rodents secondary to calculi do not appear to be relevant to humans (Cohen et al. 2002).
Table 1 lists substances that have been reported to produce calculi in rodents and/or humans. Several of these are actually essential ingredients in our diet, such as calcium and phosphate, others form endogenously, such as urate, cysteine, and uracil, but there are also others that are synthetic agrichemicals, pharmaceuticals, consumer products, or industrial chemicals. The important consideration regarding human risk assessment is that the toxicity is relevant to humans, but not the carcinogenicity. The quantitative risk assessment is based on the amount that is actually excreted in the urine and the solubility of the chemical.
In rodents, it appears that urinary crystals and calculi can produce cytotoxicity of the urothelium (Cohen et al. 2002, 2007). In contrast, crystals by themselves do not appear to be toxic to the human urothelium (Dominick et al. 2006). If crystals are present in the urine in humans, it is an indication either of a systemic effect, such as urate crystals in gout, or as an indication that calculi could form. Calculi, in contrast, produce the same types of toxicity, with urothelial irritation and regenerative proliferation in rodents, larger animals, or humans. If present for short periods of time, these heal rather quickly.
Consequences of obstruction acutely include acute pyelonephritis and hydronephrosis. Chronically, the kidneys become completely scarred with chronic pyelonephritis and develop end-stage kidney disease.
Calcification
The most common toxicologically significant kidney tubule crystals and urinary crystals and calculi in humans are calcium-containing (Liapis et al. 2015). In humans, 70% to 80% of urinary tract calculi are calcium-containing. Calcium calculi can form in individuals most commonly secondary to hypercalcemia, but can also occur in individuals who are normocalcemic or even hypocalcemic. There are numerous causes of hypercalcemia associated with urinary calculi formation including hyperparathyroidism, paraneoplastic syndromes, skeletal metastasis, various genetic diseases, vitamin A or D excess, milk alkali syndrome, and other causes including some medications (Liapis et al. 2015).
Calcification can occur in the renal parenchyma and in the lower urinary tract, and this is usually calcium phosphate or calcium oxalate. Dystrophic calcification can also occur with long-standing kidney disease, such as occurs in papillary necrosis with scarring (Liapis et al. 2015). Calcium-containing crystals can be readily identified utilizing only H&E stains due to their basophilic or purple appearance. In addition, calcium oxalate crystals can be readily identified using polarized light because of their birefringence, whereas calcium phosphate crystals are not birefringent. Stains for calcium are also available including the Von Kossa stain, which stains the phosphate, not the calcium, and therefore will not stain calcium oxalate crystals unless they are cross contaminated with phosphate. A more specific stain for calcium is the Alizarin Red stain (Liapis et al. 2015).
α2u-Globulin Nephropathy
Another cause of mineralization in the rodent kidney is α2u-globulin nephropathy (Cesta et al. 2013). This protein has much greater concentration in males than in females and is rat specific. Thus, the nephropathy is a phenomenon seen only in male rats, and the effects, both acute and chronic, due to α2u-globulin nephropathy are not relevant to humans. Nevertheless, in rodent studies, it is important to identify the presence of α2u-globulin nephropathy for an overall risk assessment of renal changes that are observed. Acutely, there are a number of features including cast formation and the appearance of cytoplasmic eosinophilic droplets in the proximal tubule cells. These can be seen on H&E stain but can be highlighted utilizing special stains such as Mallory-Heidenhain stain or a specific immunohistochemical stain for α2u-globulin. It is important to keep in mind that the Mallory-Heidenhain and immunohistochemical stains will stain the α2u-globulin in the normal rat kidney as well as in the kidneys in which it becomes bound to a specific chemical leading to toxicity. The difference is in the shape of the droplets. In the normal male rat kidney, the droplets are small, generally round, and do not accumulate to a great extent. In contrast, with α2u-globulin nephropathy, there is large droplet (crystal) formation along with angulation of the crystals and exfoliation of tubular cells into the lumen. In the long term, there is a linear mineralization that occurs in the renal papilla (Figure 3), which is quite distinctive and has been described by numerous authors (Cesta et al. 2013). The calcification is present as calcium hydroxyapatite (Trump et al. 1984).
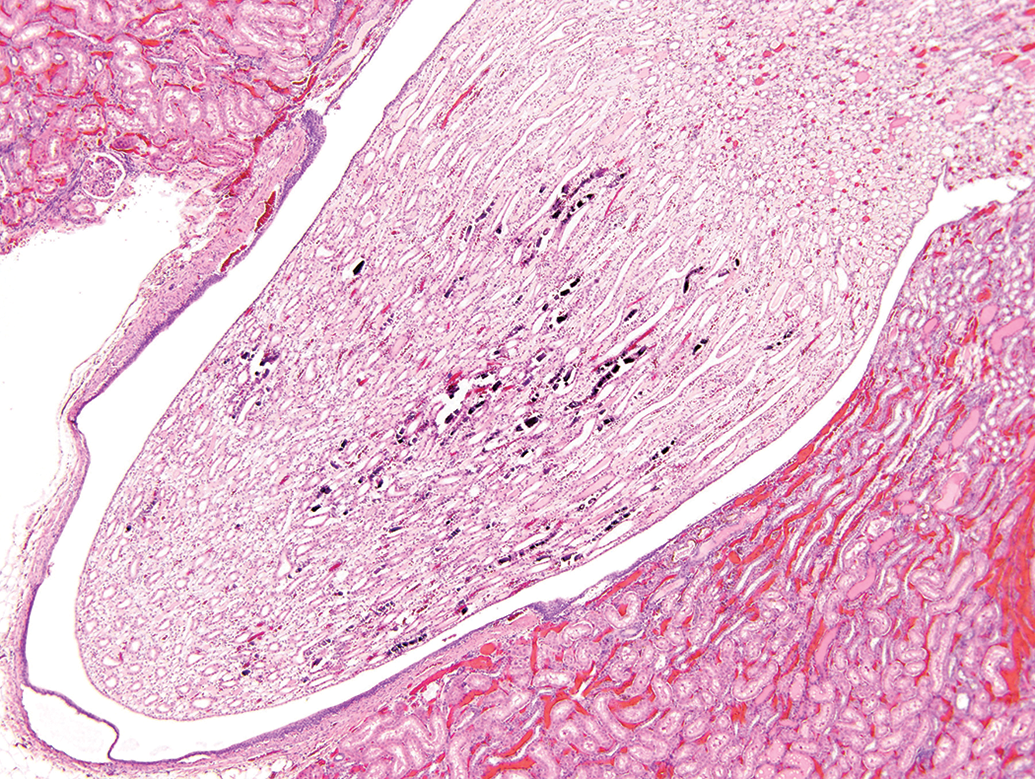
Linear papillary mineralization in chronic α2u-globulin nephropathy. Source: From Cesta et al. (2013), with permission.
Evaluation of Urine
For toxicologic evaluation for the presence of crystals and calculi, an important part of the assessment is to evaluate the urine (Cohen et al. 2007). Crystals can occur in the kidney tubules and be visualized by a variety of methods including stains, polarized light, or other more sophisticated methods if necessary. However, once the crystals are present in the lower urinary tract in the urine, they frequently are not present in the tissues, as they are washed away with the urine. Although they are sometimes adherent to the surface and can be visualized by scanning electron microscopy, it is unusual for them to actually become embedded into the tissue. Incorporation into the tissue can occur with continuous ulceration and inflammation. Tissue evaluation indicating the possible presence of calculi is the concurrent presence of inflammation.
Most importantly, however, the presence of urinary crystals or calculi is determined by urinary examination.Specific methods need to be applied for a detailed examination, particularly if it is crystals and not calculi that are causing the toxicity (Cohen et al. 2007). Most critically, the animals should not be fasted before urine is collected. Urine composition is critical to the formation of these crystals and calculi, and this most often is related to the time that the animal is actually consuming the chemical and food, since food will greatly alter the composition of the urine. Depending on the kinetics of the administered substance, if the crystals are composed of that chemical (or metabolite), toxicokinetics will play a major role in determining maximum concentration of the substance in the urine. It is at these times that urine needs to be collected since at lower concentrations the crystals may not be present. Since the crystals do not actually get into the tissue, they need to be constantly formed for them to be detected as they are being excreted in the urine.
Urine composition plays a critical role in the formation of crystals and calculi regardless of their composition. Urine undergoes diurnal variations in virtually all components, including pH, protein, electrolytes, and urea, primarily related to the time of food consumption (Cohen et al. 2007). All of these factors can influence the formation or dissolution of the crystals or calculi. A particularly important factor is urinary pH, since certain substances, such as calcium-containing crystals, will only form at neutral or higher urinary pH whereas others will form only at lower pH, such as urate crystals. When the animal stops eating, the urine rapidly becomes acidic, and if the crystals are calcium-containing, these will disappear. Keep in mind that rodents are nocturnal creatures, so it is critical to collect urine early in the morning.
It is also strongly recommended that fresh void urines be collected rather than overnight or 24-hr urines (Cohen et al. 2007). Fresh void urine avoids the confounding role of the crystals sticking to the metabolism cage but more importantly provides for immediate examination without time for the urine composition to change (such as the evaporation of the water or CO2) which can change the urinary pH and overall urine composition. It also avoids the possible dissolution of the crystals over time, or more importantly, the artifactual formation of certain crystals by standing. pH can also alter the ionization of chemicals, whether acidic or basic, greatly affecting the potential for the toxicity of the substance if toxicity is dependent on the ionized form or the unionized form.
A number of other factors have been identified in urine which contribute to the formation of crystals (Cohen 1995). Protein is one of these factors, acting most frequently as a nidus for the formation of the crystals, but occasionally calculi and crystals can be composed entirely of protein and other organic matter in the urine such as mucopolysaccharide. Rodents have much higher concentrations of protein in the urine than humans (Cohen 1995), even in patients with nephrotic syndrome. In addition, in male rodents, there is higher protein concentration than in the females, such as α2u-globulin in the male rat and mouse urinary protein in male mice. Another major factor is that the concentration of solutes that can form crystals such as calcium, magnesium, and phosphate can vary considerably across species. For unknown reasons, mice have much lower concentrations of these ions than rats or humans (Cohen 1995). Mice are much less susceptible to the development of calcium-containing crystals than the rat. Another major factor is the overall density of the urine. Rat and mouse urine has an osmolality of 1,500 to 3,000 mOsmol, whereas humans usually have an osmolality of 50 to 400. All of these factors play a role in the formation of the crystals, so it is necessary to measure all of them rather than relying on a single factor.
Examination for crystals in the urine can be frequently accomplished by collecting the sediment from the urine collection and examining it by light microscopy (Karcher, McPherson, and Pincus 2017; Cohen et al. 2007). A more sensitive method is to collect the crystals on a filter and examine them by scanning electron microscopy. This can be helpful in identifying not only the presence of crystals but their elemental composition if the microscope has a built-in reflective X-ray spectroscopy unit. This aids in identifying crystals that are of endogenous origin versus those that are composed of the exogenously administered chemicals or their metabolites. Identification of the material in a crystal is essential in the overall interpretation of the mode of action of the toxicity (Cohen et al. 2002). This can be accomplished by collecting the calculi and examining them be infrared spectroscopy. Even in the kidney tissue, ultrasensitive methods are available for identifying the composition of crystals even in the tubules, such as Fourier transform infrared microspectroscopy (Dobson et al. 2008). Examination by scanning electron microscopy is also helpful for identifying extremely small crystals that are not easily visible by light microscopy, such as a variant of calcium oxalate (Dominick et al. 2006).
In addition to measuring substances in the urine that can crystallize, it is also important to evaluate the urine for substances that act to inhibit the formation of the crystals, such as citrate, a chelating substance for calcium. For example, PPAR-γ agonists, such as muraglitazar (Dominick et al. 2006), produced urinary tract calcium-containing crystals secondary to an effect on the production of citrate in the liver. Citrate levels were greatly reduced, leading to hypocitratemia that, of course, leads to hypocitraturia. Calcium crystals were then able to form.
Conclusions
In summary, crystals are a normal component of the urine, with most mammalian species containing magnesium ammonium phosphate crystals (Cohen 1995; Cohen et al. 2007). In normal quantities, these are not toxic. However, there are numerous other types of crystals that can form in the kidney and/or in the urine itself which can produce toxicity. This is a high-dose phenomenon and is secondary to the presence of a quantity of material that can become insoluble and form crystals at a given concentration. If the exposure produces a urine concentration below which crystallization can occur, there is no toxicity. It is a threshold phenomenon. Acute effects of crystal formation include acute renal failure, such as secondary to ethylene glycol ingestion and calcium oxalate crystals in the kidney or can produce obstruction with consequent hydronephrosis and frequently associated acute pyelonephritis. In the long term, there are chronic changes including retrograde nephropathy, chronic hydronephrosis, and pyelonephritis and eventually end-stage kidney. Urothelial tumor formation is a toxic end point in rodents but does not appear to occur in humans and other species, such as cats (Cohen, 2018). For risk assessment purposes, the essential steps are identifying the formation of crystals and/or calculi as the inciting event, determining the composition of the crystals/calculi, and determining the dose response for the threshold amount necessary for formation of these crystals.
Footnotes
Acknowledgment
The author is grateful to Lora Arnold and Jeanne Bradford for their invaluable assistance in preparing this article.
Author Contributions
The author (SC) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. The author gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
