Abstract
The chemically induced accumulation of α2u-globulin protein in male rats causes specific renal lesions and subsequent nephropathy. Herein, we report additional parallel findings in the kidney of male rats consistent with obstructive and retrograde nephropathy. Kidney and urinary bladder samples were evaluated from Wistar rats treated with RG7129 for 2 week and 8 week and from an 8-week mechanistic study using females, intact and castrated males. Histopathological findings were present in intact males in all studies, including hyaline droplet accumulation and granular casts consistent with α2u-globulin nephropathy. In addition, tubular degeneration and regeneration, tubular changes extending from papilla to cortex, tubular dilation, and interstitial and luminal inflammation were observed consistent with retrograde and obstructive nephropathy. Renal and urinary lesions and their severity increased in a time- and dose-dependent manner. Urinalysis findings, including increases in leukocytes, protein, and in kidney biomarkers, kidney injury molecule 1 and clusterin, were present only in intact males. No treatment-related changes were observed in female rats or in castrated males. These results indicate that RG7129 induces α2u-globulin nephropathy, associated with retrograde and obstructive nephropathy secondary to precipitation in intact male rats only, constituting a species- and sex-specific syndrome that is not expected to occur in humans or other species.
Animal toxicology studies are necessary to assess the potential risk of chemicals to humans. However, in some instances, inferences from animal data to human may not be possible due to a species-specific response. In the rat, a number of chemicals have been shown to produce species-specific renal lesions characterized by intracytoplasmic hyaline droplet formation predominantly associated with accumulation of the male rat-specific α2u-globulin (Baetcke et al. 1991; Hard et al. 1993; Hard 2008; Swenberg 1993; Swenberg and Lehman-McKeeman 1999; Doi et al. 2007; Cesta et al. 2013). Such accumulation has not been observed in female rats or male rats lacking α2u-globulin and also poses no risk to humans, primarily due to the absence of a similar protein.
α2u-globulin, a low-molecular-weight protein member of the lipocalin family, is synthesized in the liver of male rats under multihormonal control, particularly androgen (Baetcke et al. 1991; Hard et al. 1993; Swenberg and Lehman-McKeeman 1999). This protein, present in the mature male rat, is not synthesized until puberty, and therefore, levels are diminished in both immature and castrated male rats (Hard et al. 1993; Swenberg and Lehman-McKeeman 1999). In female rats, estrogens inhibit hepatic synthesis of α2u-globulin (Hard et al. 1993; Swenberg 1993). Previous studies have demonstrated that compounds can bind to α2u-globulin and by this induce accumulation of α2u-globulin in male rats leading to nephropathy; this is not observed in female rats, castrated male rats, or rats that do not synthesize α2u-globulin (e.g., NCI-Black Reiter rats). Such accumulation has also not been observed in mice; although mice produce a protein similar to α2u-globulin (mouse major urinary protein [MUP]), this does not cause nephropathy because MUP does not bind with chemicals that induce accumulation of protein droplets (Baetcke et al. 1991; Hard et al. 1993).
α2u-globulin nephropathy is characterized by the rapid accumulation of hyaline droplets in the P2 segment of the renal proximal tubule cells following chemical treatment (Swenberg and Lehman-McKeeman 1999; Swenberg 1993; Hard et al. 1993; Baetcke et al. 1991). The accumulating protein in these droplets is α2u-globulin. The main mechanism of α2u-globulin nephropathy is the reversible and noncovalent binding of a chemical or metabolite to α2u-globulin. This reduces lysosomal degradation of α2u-globulin, leading to its accumulation in the phagolysosomes (Swenberg and Lehman-McKeeman 1999; Swenberg 1993). Subsequent additional histological changes also occur following continued treatment with chemicals that induce α2u-globulin accumulation. These include single-cell degeneration and necrosis in the P2 segment of the proximal tubule and the formation of granular casts at the cortico–medullary junction (Hard et al. 1993; Swenberg and Lehman-McKeeman 1999). Cellular degeneration and necrosis in the kidneys trigger a compensatory increase in renal cell proliferation in the P2 segment of the renal proximal tubule, which is dose related and time related (Swenberg and Lehman-McKeeman 1999; Swenberg 1993; Hard et al. 1993). It has also been reported that further continued exposure may cause the formation of linear papillary mineralization, exacerbation of chronic progressive nephropathy (CPN), and the development of renal tumors as a consequence of the sustained cell proliferation (Swenberg and Lehman-McKeeman 1999; Swenberg 1993; Hard et al. 1993).
Exposure to chemicals can also induce obstructive and retrograde nephropathy in the rat (Hard, Flake, and Sills 2009). Typical histopathological features of obstructive nephropathy include tubular dilation and inflammation due to intraluminal crystal deposits causing urinary tract obstruction (Frazier et al. 2012). In contrast, retrograde nephropathy is characterized by tubule basophilia and tubule dilation involving all zones from cortex down to papilla preferentially at the poles of the kidney without marked inflammation due to an ascending effect originating from the lower urinary tract (Hard, Flake, and Sills 2009; Frazier et al. 2012). While obstructive and retrograde nephropathy have been previously described, they have not been observed in combination with α2u-globulin nephropathy.
The studies we describe aimed to examine the toxicology of a chemical, RG7129. RG7129 is a central nervous system –penetrating compound designed for inhibiting the β-site amyloid precursor protein cleaving enzyme 1 for the treatment of Alzheimer’s disease. A fluorine containing oxazines is the basis of its chemical structure. We report drug-induced renal and urinary bladder lesions in intact male rats via mechanisms involving α2u-globulin accumulation, combining three pathological processes: α2u-globulin nephropathy, retrograde nephropathy, and obstructive nephropathy that has not been previously described in the literature.
Materials and Methods
Oral gavage studies were performed in Wistar rats to assess the toxicology of a small molecule RG7129 (developed by F. Hoffmann-La Roche Ltd, Basel, Switzerland). RG7129-induced toxicity was compared with vehicle control animals. RG7129 was prepared as a stock suspension with a nominal concentration of 80 mg/g free base and was diluted to relevant doses/concentration with the vehicle control.
Animals
Male and female Wistar rats (HanRCCWIST [SPF], Harlan Laboratories Ltd, Horst, the Netherlands) aged approximately 8 weeks were habituated for 1 or 2 weeks. Animals were kept in an air-conditioned room under periodic bacteriologic control at a temperature of 22°C ± 2°C, with 40% to 80% humidity and a 12-hr light/dark cycle and background music coordinated with light hours. Animals were housed in pairs in environmentally enriched Makrolon boxes (type III [2-week pilot toxicity study, 2-week toxicity study, and 8-week mechanistic study]/type IV [8-week Good Laboratory Practice (GLP) toxicity study], with autoclaved sawdust bedding). A pelleted standard rodent diet and tap water were supplied ad libitum. Animals were randomly assigned to dose groups based on body weight using the data collection software Provantis V8.4 (Instem Life Sciences, Stone, UK). In all studies, the animals were kept in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International and treated in accordance with the guidelines of the Swiss Animal Welfare Act. All procedures were in accordance with the respective Swiss regulations and approved by the Cantonal Ethical Committee for Animal Research.
Study Design, Clinical Observations, and Mortality
2-week pilot toxicity study
A 2-week non-GLP pilot toxicity study was conducted in 36 Wistar rats. Daily doses of vehicle (0 mg/kg/day; solution containing methylparaben [0.18%], propylparaben [0.02%], hydroxypropyl methylcellulose [1.25%], docusate sodium [0.1%], purified water [98.45%]) or RG7129 (240 and 300 mg/kg/day) were administered by oral gavage at a volume of 4 ml/kg. Each dose group contained four male and four female rats. Dose levels were selected based on results from a previous pharmacokinetic study (data not shown). The study was prematurely terminated on day 3 after two days of treatment due to severe clinical signs (e.g., hypoactivity, piloerection, decreased muscle tone, loss of righting reflex) at both 240 and 300 mg/kg/day dose levels.
2-week toxicity study
A 2-week non-GLP toxicity study was conducted in 60 Wistar rats. Daily doses of vehicle (0 mg/kg/day) or RG7129 (20, 60, 120, or 180/240 mg/kg/day) were administered by oral gavage for 14 days at a volume of 5 ml/kg body weight (6.67 ml/kg for rats treated at 240 mg/kg/day). Each dose group contained six male and six female rats. Dose levels were selected based on results from a previous pharmacokinetic study (data not shown).
8-week GLP toxicity study
An 8-week GLP toxicity study was conducted in 112 Wistar rats. Animals were dosed daily by oral gavage at 0 (vehicle; solution containing citrate buffer pH 3, sorbic acid, avicel CL 611, xanthan gum, Tween 80) or RG7129 at 20, 100, and 200/160 mg/kg/day for 57/58 days at a volume of 5 ml/kg body weight. Each dose group contained 14 male and 14 female rats. Due to the occurrence of severe clinical signs (hypoactivity and piloerection) on day 2 at 200 mg/kg/day, treatment was ceased temporarily from day 3 to 7, and the high-dose level was reduced to 160 mg/kg/day from day 8 onward by reduction in dose volume to 4 ml/kg/day.
8-week mechanistic study
An 8-week mechanistic study was conducted in 96 Wistar rats. Daily doses were administered by oral gavage at 0 mg/kg/day (vehicle; solution containing 3% microcrystalline cellulose derivative, 0.3% xanthane gum, 0.2% polysorbate 80, 0.15% sorbic acid, 0.84% citric acid monohydrate, and 100% demineralized water ad libitum) or RG7129 at 20 and 160 mg/kg/day for 2, 15, or 58 days at a volume of 5 ml/kg body weight. Each dose group contained 12 intact males and 12 castrated males; animals were subdivided into three subgroups with three staggered necropsy dates on days 3, 16, and 59/60. Additionally, female animals were dosed daily by oral gavage (0 and 160 mg/kg/day) for 58 days at a volume of 5 ml/kg body weight, with six females per dose group. Dose levels were selected based on results from the previous toxicity studies and toxicokinetic data in rats.
Routine parameters
For all studies, clinical observations, mortality, body weight, food consumption, and body temperature (except for the 8-week mechanistic study) were recorded, and a detailed physical examination was performed (see Table 1 for details). Blood for laboratory investigations was taken in all studies as outlined in Table 1. Animals were sacrificed by CO2 inhalation and exsanguinated; all organs and tissues were collected, weighted (adrenal glands, brain, epididymides, heart, kidneys, liver, prostate gland, spleen, testes, and thymus), and macroscopically examined.
Summary of Measurements for All Studies.
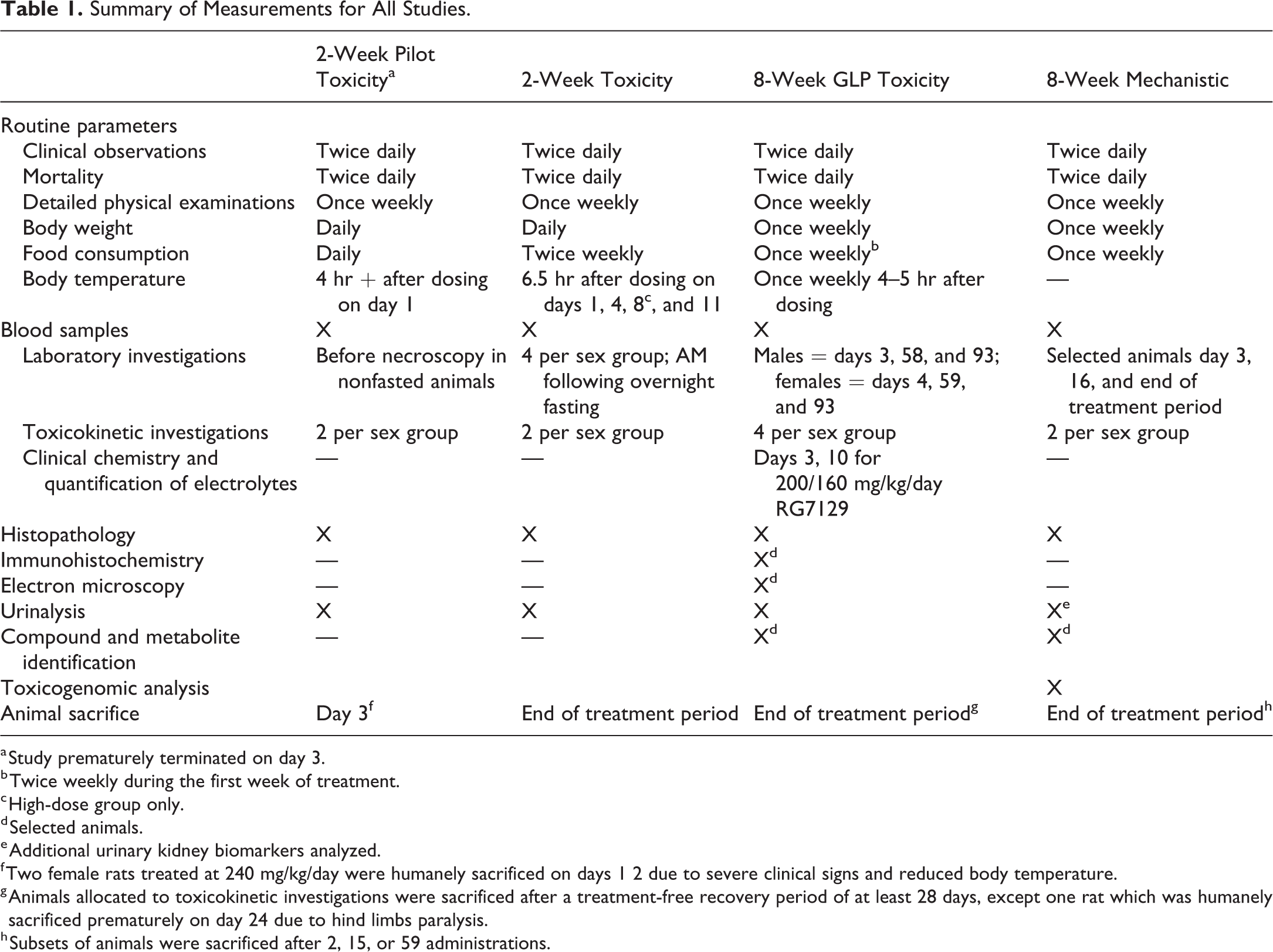
a Study prematurely terminated on day 3.
b Twice weekly during the first week of treatment.
c High-dose group only.
d Selected animals.
e Additional urinary kidney biomarkers analyzed.
f Two female rats treated at 240 mg/kg/day were humanely sacrificed on days 1 2 due to severe clinical signs and reduced body temperature.
g Animals allocated to toxicokinetic investigations were sacrificed after a treatment-free recovery period of at least 28 days, except one rat which was humanely sacrificed prematurely on day 24 due to hind limbs paralysis.
h Subsets of animals were sacrificed after 2, 15, or 59 administrations.
Histopathology
A complete tissue set was collected for histopathologic examination. Tissue samples were fixed in 10% buffered formalin for at least two days. Organs and tissues were embedded in paraffin, and sections were stained with hematoxylin–eosin. Additionally, kidney sections were stained with Heidenhain stain (Heidenhain’s iron hematoxylin) for detection of hyaline droplets within tubular epithelium or with periodic acid–Schiff reaction.
Immunohistochemistry
Deparaffinized kidney sections of selected males treated for 8 weeks with RG7129 were stained for α2u-globulin. The primary antibody was a prediluted (1:50) polyclonal goat anti-α2u-globulin antibody (R&D Systems Inc., Minneapolis, MN; catalog number AF586). The detection kit was the Ventana Omnimap DAB Detection Kit. This used the avidin–biotin–peroxidase complex method and a biotinylated secondary antibody of rabbit anti-goat on a Ventana Discovery XT immunostainer (Ventana Medical System, Tucson, AZ).
Electron microscopy
Tissue samples from kidneys of selected male rats treated for 8 weeks with 200/160 mg/kg/day RG7129 were examined by transmission electron microscopy. Formalin-fixed material was postfixed in glutaraldehyde, resin-embedded, trimmed to a size fitting copper grids of 3 mm in diameter, and ultrathin sections (100 nm) were prepared. Sections were contrasted using uranyl acetate and lead citrate. Ultrathin sections were evaluated using a Philips CM 10 electron microscope with focus on lysosomal structures in intact proximal epithelial cells.
Urinalysis
Urine was collected for approximately 6 hr from animals toward the end of dosing in metabolic cages (Tecniplast) and analyzed with Aution Max AX-4280 automated urine chemistry analyzer for the following parameters: specific gravity, pH, white blood cells, nitrite, protein, glucose, ketones, urobilinogen, bilirubin, erythrocytes/hemoglobin, color, and transparency. Additionally, collected urine from the 8-week mechanistic study was analyzed for urinary kidney biomarkers using the Myriad RBM assay (Austin, TX). The kidney biomarker Rat Kidney MAP assay was used to analyze the following biomarkers: β2-microglobulin, calbindin, clusterin, cystatin C, epidermal growth factor, glutathione S transferase α, glutathione S transferase μ, kidney injury molecule 1 (KIM-1), neutrophil gelatinase–associated lipocalin, osteopontin, tissue inhibitor of metalloproteinases 1, and vascular endothelial growth factor.
Compound and metabolite identification
Urine as well as uroliths detected macroscopically in intact male rats treated for 8 weeks were investigated for the presence of parent compound and metabolites from selected males by high-performance liquid chromatography (Atlantis T3 [Waters], 2.1 × 100 mm, 3 μm) and time-of-flight mass spectrometry (Xevo QTof, Waters, Milford, MA, USA).
Toxicogenomic analysis
Kidney samples from intact and castrated males treated at 0 or 160 mg/kg/day were collected at necropsy on days 3, 16, and 60 and analyzed. In addition, liver samples from intact males treated at 0 or 160 mg/kg/day were collected at necropsy on day 60 and analyzed. RNA isolation from rat kidney and liver, amplification, labeling, and hybridization were carried out as recommended by Affymetrix with minor modifications, and quality controls for RNA and for hybridizations were performed. Affymetrix Rat Genome U34A microarrays were used. Statistical analysis of the signals to detect expressed probe sets was performed using the Roche Affymetrix Chip Expression Analysis program.
Statistical Analysis
Group means and standard deviations for N ≥ 3 animals per group were calculated for clinical pathology parameters using ClinAxys software (Clinical Systems Ltd, Stokenchurch, UK), for organ weights using PathData software V6.2c2 (PDS, Birsfelden, Switzerland) and for in-life parameters using Provantis software (Instem Life Sciences).
Body weight data were assessed by a one-way analysis of variance, which includes the Bartlett test for variance, the Wilks–Shapiro test for normality (the Kolmogorov–Smirnov test for large numbers of animals), and the Dunnett test for pair-wise comparison. Food consumption was assessed by the Dunnett test for pair-wise comparison. For clinical pathology parameters (ClinAxys), group variances were assessed for homogeneity using Bartlett’s test. Group differences for homogeneous data were assessed using a parametric ANOVA followed by a pair-wise Student’s t-test. Group differences for nonhomogeneous data were assessed using a nonparametric ANOVA (Kruskal–Wallis test). A statistically significant finding across groups was followed by a Mann–Whitney U-test comparing individual treated groups with controls. Other appropriate tests were used as necessary. Dunn’s test or the Dunnett test were used for statistical analysis of organ weights in PathData.
Results
Drug-induced Obstructive and Retrograde Nephropathy Associated with α2u-globulin Nephropathy: Histopathological Observations
In all four studies, dose- and time-dependent histopathological changes were observed in the kidneys of intact male rats following RG7129 administration (Tables 2 and 3). These included increased accumulation of Heidenhain stain–positive hyaline droplets in the proximal tubular epithelium, tubular epithelial cells filled with intraepithelial hyaline droplets partly bulging into the lumen of some or all affected tubules, tubular degeneration and regeneration, tubular dilation of distal cortical and medullary tubules, and inflammatory processes. In addition, urinary bladder lesions were observed in intact male rats (Figures 1 –3).
Incidence of Renal and Bladder Lesions in Intact Male Rats in the 2-week Studies.
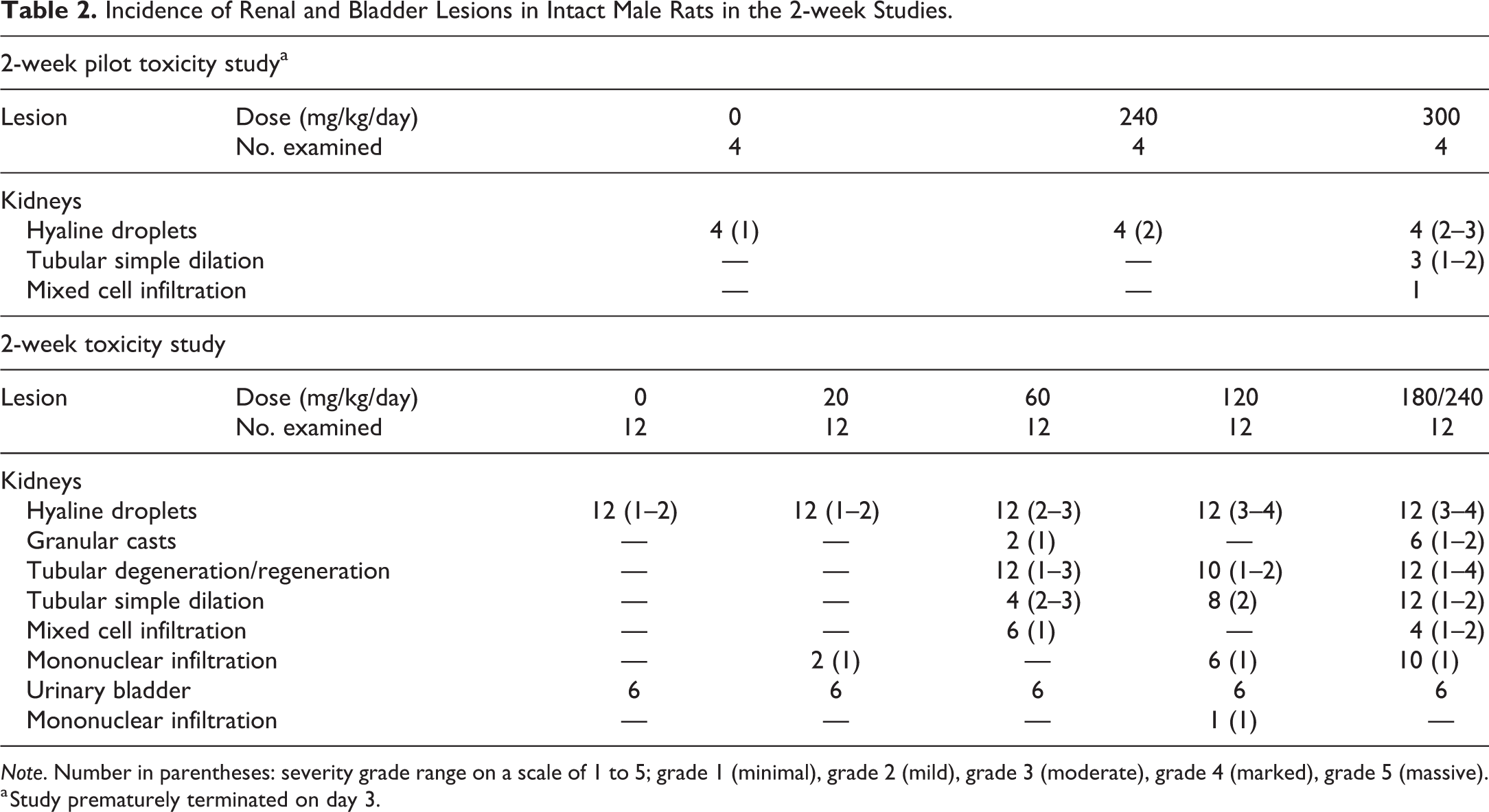
Note. Number in parentheses: severity grade range on a scale of 1 to 5; grade 1 (minimal), grade 2 (mild), grade 3 (moderate), grade 4 (marked), grade 5 (massive).
a Study prematurely terminated on day 3.
Incidence of Renal and Bladder Lesions in Intact Male Rats in the 8-week Studies.
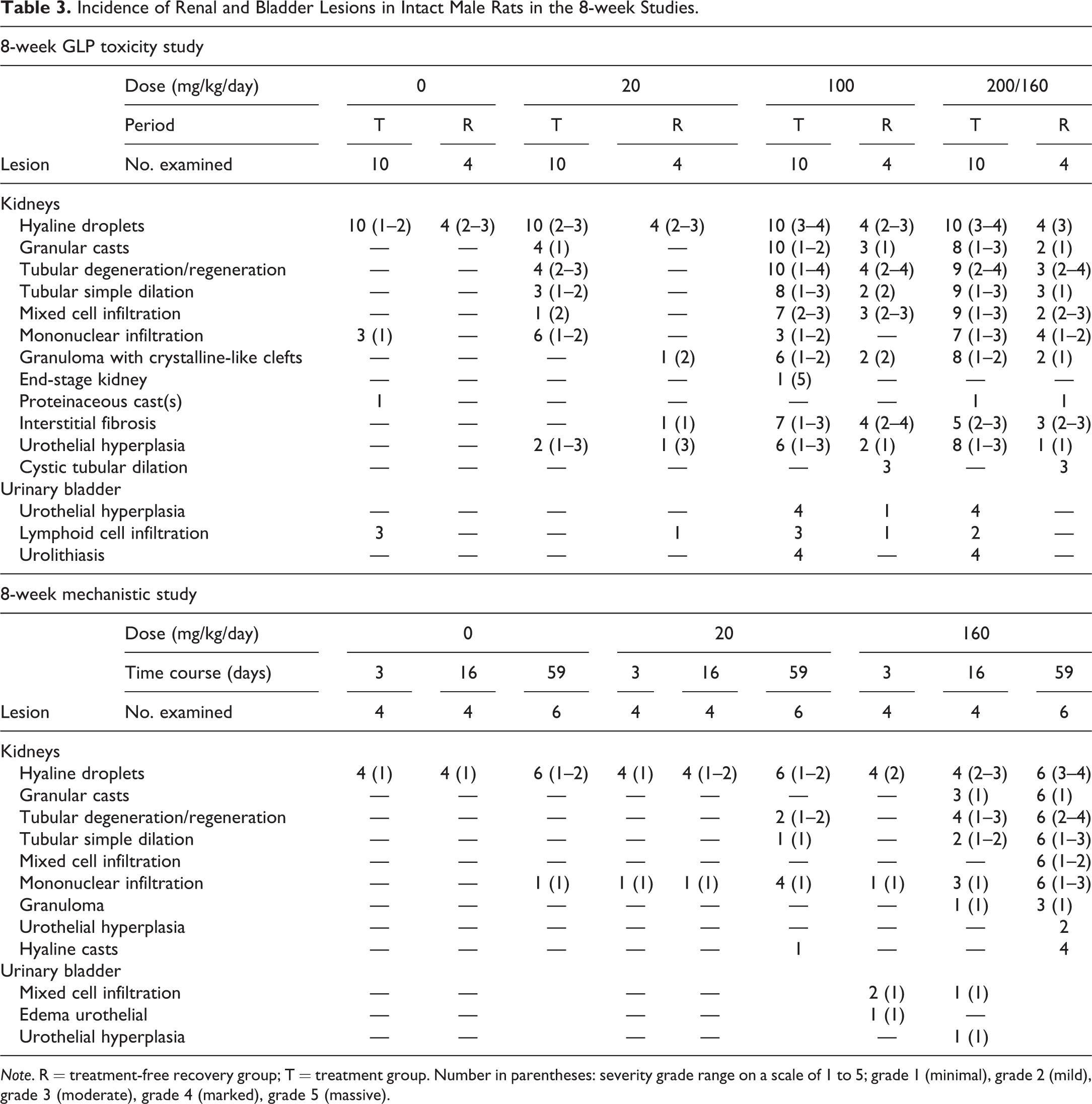
Note. R = treatment-free recovery group; T = treatment group. Number in parentheses: severity grade range on a scale of 1 to 5; grade 1 (minimal), grade 2 (mild), grade 3 (moderate), grade 4 (marked), grade 5 (massive).
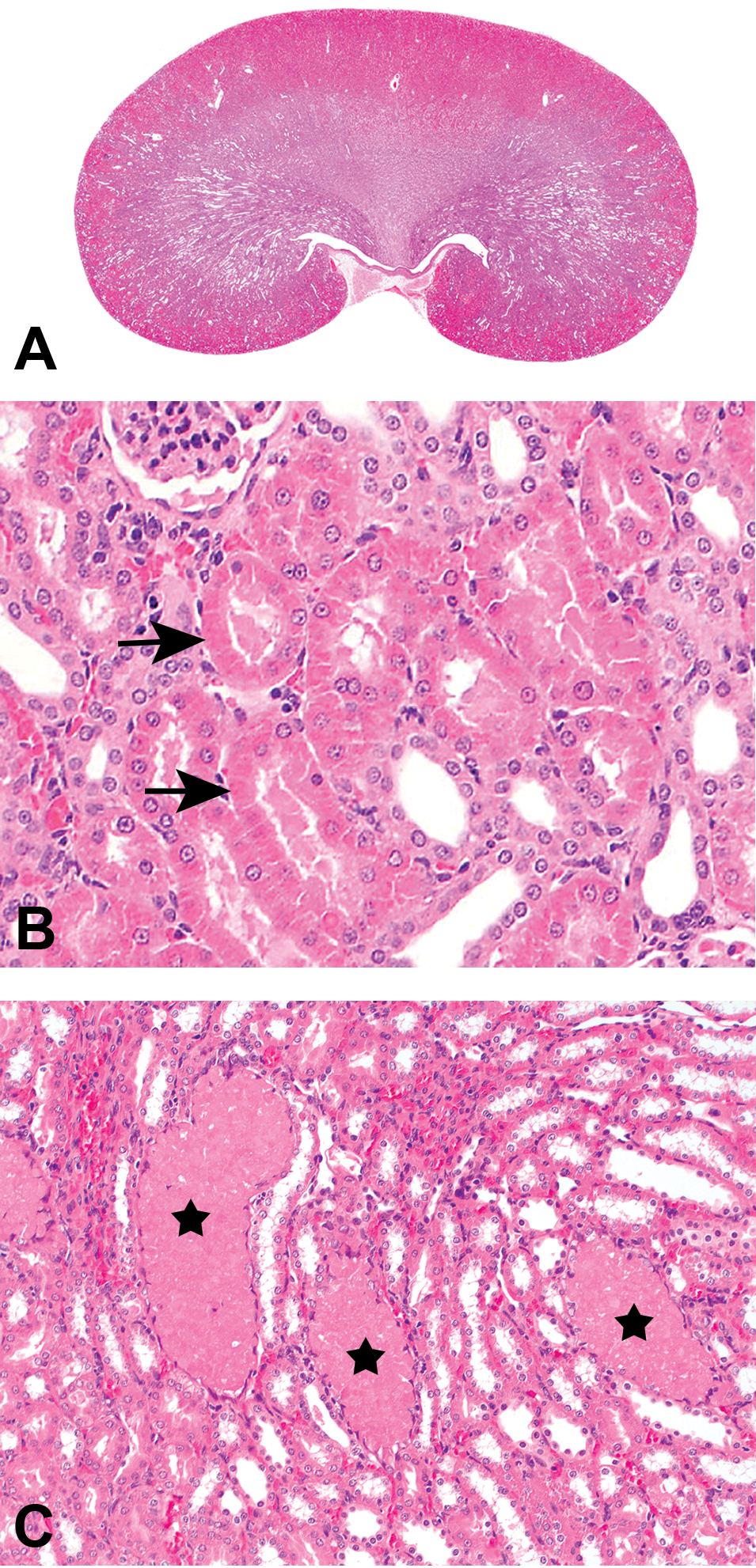
Kidney histopathological changes in treated intact male rats. (A) Multifocal nephropathy with predominance for cranial and caudal poles extending from cortex to the papilla in intact male rats exposed to 180/240 mg/kg/day of RG7129 for 14 days (2-week toxicity study); hematoxylin–eosin (HE). (B) Hyaline droplets (arrows) in proximal tubular epithelium in intact male rats exposed to 180/240 mg/kg/day of RG7129 for 14 days (2-week toxicity study); HE. (C) Intratubular granular casts (stars) in intact male rats exposed to 160 mg/kg/day of RG7129 for 59 days (8-week mechanistic study); HE.
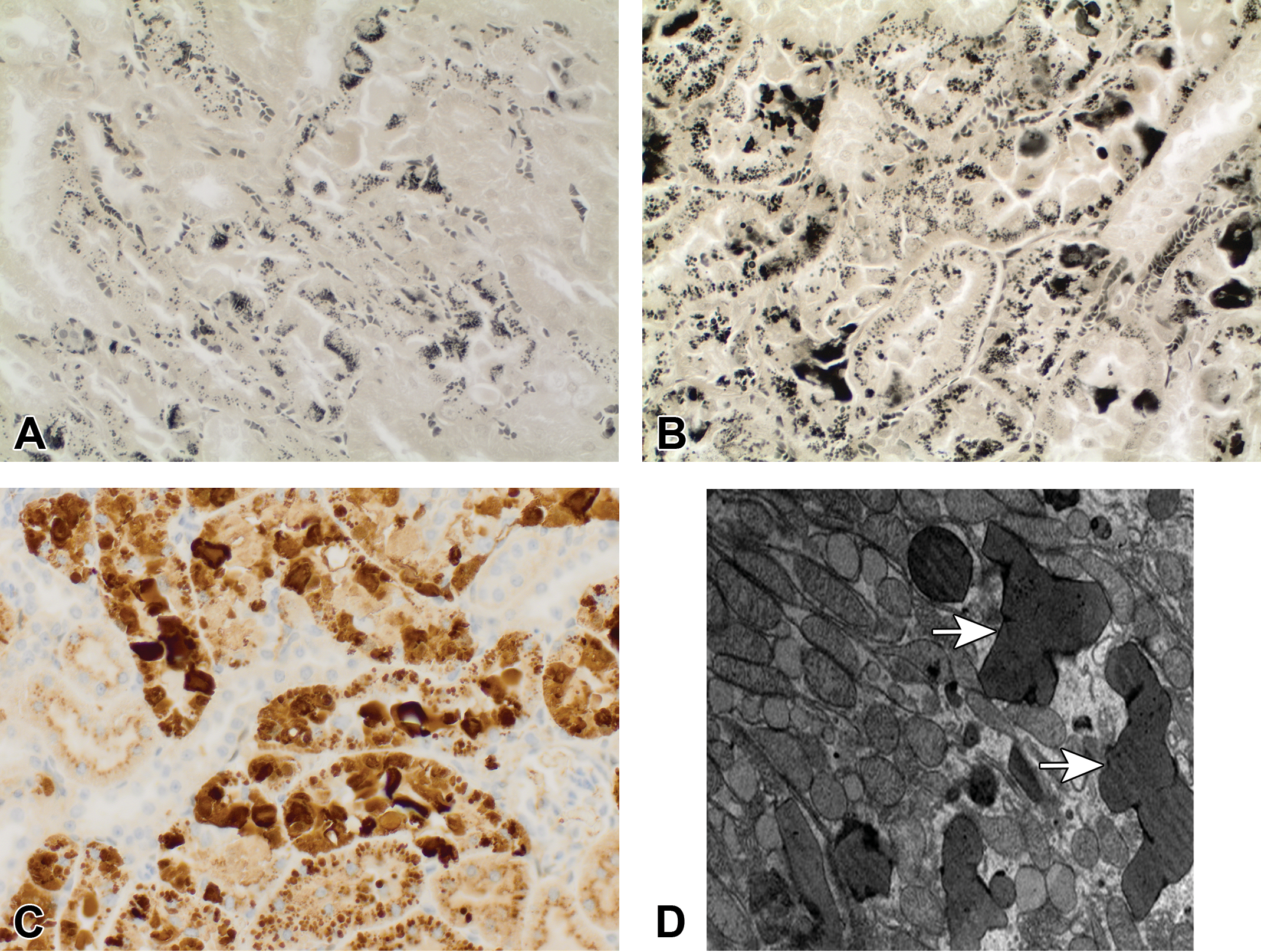
8-week GLP toxicity study. (A) Normal distribution of Heidenhain-positive (black stained) droplets in proximal tubular epithelium in intact male rats exposed to vehicle for 58 days; Heidenhain’s iron hematoxylin. (B) Increased accumulation of Heidenhain-positive (black stained) droplets in proximal tubular epithelium in intact male rats exposed to 200/160 mg/kg/day of RG7129 for 58 days; Heidenhain’s iron hematoxylin. (C) Increased accumulation of α2u-globulin in proximal tubular epithelium partly extending into the lumen in intact male rats exposed to 200/160 mg/kg/day of RG7129 for 58 days; anti-α2u globulin immunohistochemistry. (D) Cytoplasmic polyangular phagolysosomes (arrows) in proximal tubular epithelium in intact male rats exposed to 200/160 mg/kg/day of RG7129 for 58 days; transmission electron microscopy.
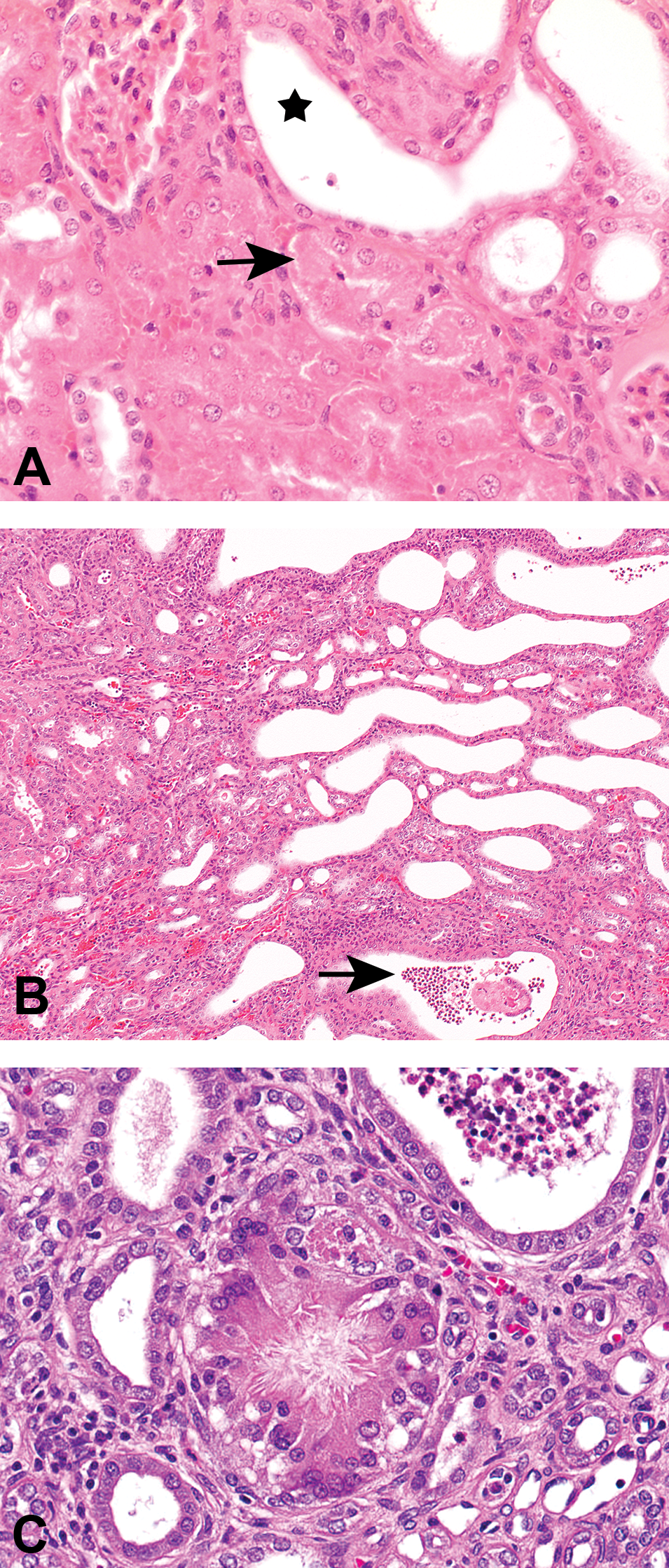
Kidney histopathological changes in treated intact male rats. (A) Tubular degeneration (black arrow), and tubular dilation (star) in intact male rats exposed to 160 mg/kg/day of RG7129 for 59 days (8-week mechanistic study); hematoxylin–eosin (HE). (B) Tubular dilation with intraluminal granulocytic infiltration and eosinophilic material (arrow) and tubular basophilia (regeneration) in intact male rats exposed to 160 mg/kg/day of RG7129 for 59 days (8-week mechanistic study); HE. (C) Granuloma with central crystalline-like clefts in intact male rats exposed to 200/160 mg/kg/day of RG7129 for 58 days (8-week GLP toxicity study); HE.
Nephropathy was present in intact males (Figure 1A). Kidney lesions consistent with α2u-globulin nephropathy were characterized by accumulation of hyaline droplets in the proximal tubules present already after 3 days of treatment (Figure 1B). The accumulation of hyaline droplets showed a dose- and time-dependent increase in severity, and the hyaline droplets were partly extended into the lumen of some or all affected tubules. The hyaline droplets were confirmed by Heidenhain stain and by immunohistochemistry with an anti-α2u-globulin antibody in selected males to be α2u-globulin (Figure 2A–C). Electron microscopic evaluation of the proximal tubular cells of the kidneys of high dose male rats showed increased incidence and severity of cytoplasmic large polyangular phagolysosomes in the renal tubular epithelial cells of the proximal convoluted tubules, compared with control males (Figure 2D). These phagolysosomes partially had a condensed crystalline core characteristic for α2u-globulin accumulation. Starting after 2 weeks of treatment, intact males showed intratubular granular casts at the junction of the cortex and outer medulla in addition (Figure 1C). The intratubular granular casts showed a dose- and time-dependent increase in incidence and severity and were accompanied by tubular degeneration and regeneration.
In addition to the typical lesions consistent with α2u-globulin nephropathy, lesions consistent with precipitation of RG7129 with secondary retrograde nephropathy and obstructive nephropathy were observed. These included tubular degeneration and regeneration characterized by single-cell necrosis (Figure 3A), tubular basophilia and simple tubular dilation with intraluminal granulocytic infiltration and eosinophilic material (Figure 3B), mixed cell infiltration, and interstitial fibrosis (Tables 2 and 3). Starting already at 3 days, widespread tubule dilation involving all zones, from cortex down to the papilla, was observed consistent with retrograde nephropathy (Table 2). Inflammatory infiltrates partly with granuloma formation indicative of early obstructive nephropathy were also present in some rats starting after 2 weeks of treatment. At 8 weeks, minimal to slight interstitial/intratubular granuloma(s)/granuloma-like lesions with intracellular eosinophilic material and crystalline-like clefts (i.e., suggestive of foreign body) were present in some animals (Figure 3C). The lesions mainly involved the cranial and/or caudal poles of the kidneys.
Urinary bladder lesions in the form of focal to diffuse and minimal to moderate urothelial hyperplasia were observed in the 8-week studies in intact males at ≥100 mg/kg/day (Table 3). The urothelial hyperplasia was associated with uroliths in one male at 100 mg/kg/day and one male at 200/160 mg/kg/day (Figure 4A and B). Moderate and diffuse urothelial hyperplasia (without uroliths) was present in one male at 100 mg/kg/day after the 4-week treatment-free period. One intact male at 160 mg/kg/day showed minimal multifocal inflammatory infiltrates besides urothelial hyperplasia (Figure 4C and D).
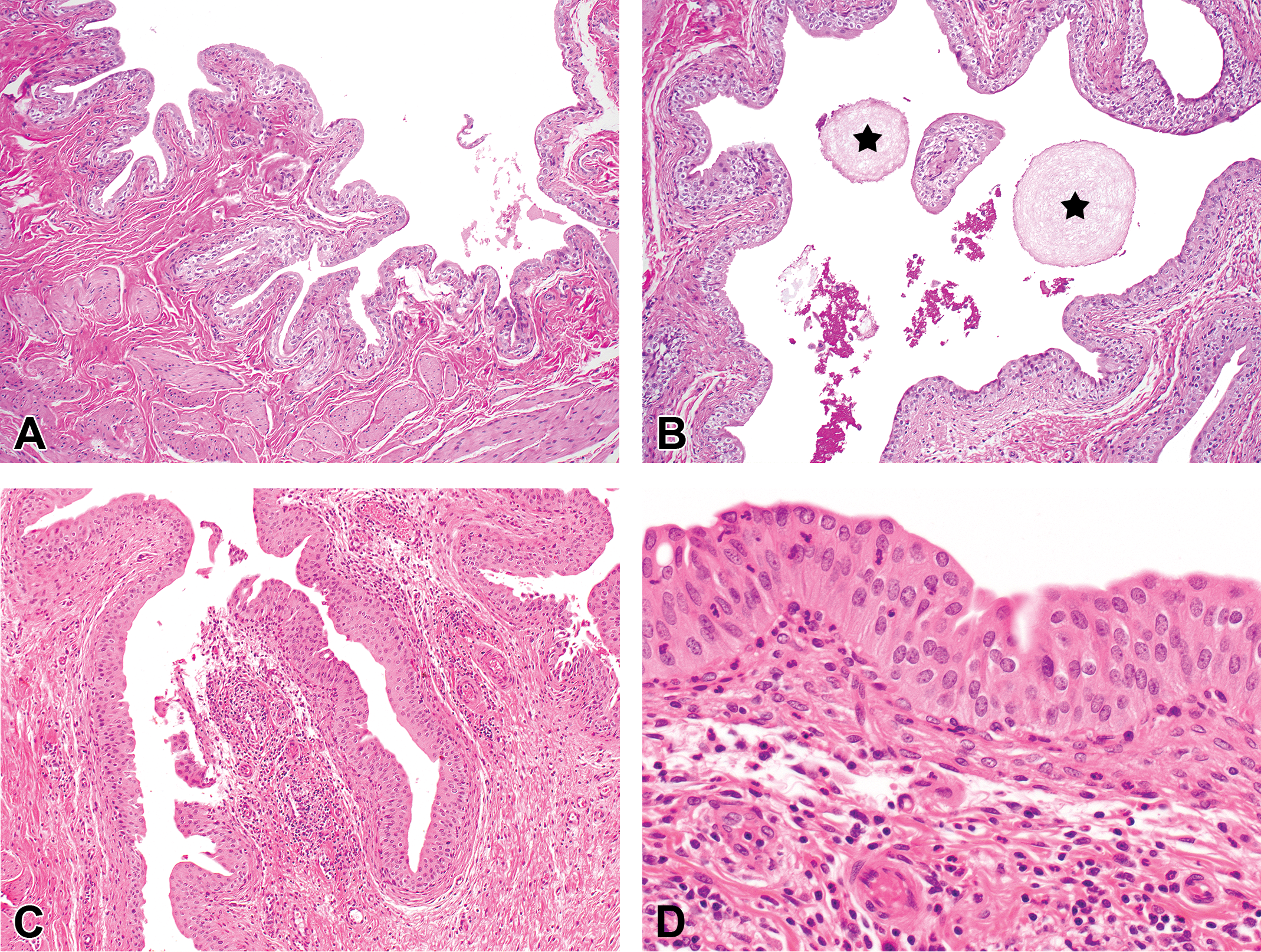
Urinary bladder histopathological changes. (A) Intact male rats exposed to vehicle for 58 days (8-week GLP toxicity study). (B) Uroliths (stars) in intact male rats exposed to 100 mg/kg/day of RG7129 for 58 days (8-week GLP toxicity study). (C and D) Mixed cell infiltration and urothelial hyperplasia in intact male rats exposed to 160 mg/kg/day of RG7129 for 16 days (8-week mechanistic study); hematoxylin–eosin.
Renal and urinary lesions were specific to intact male rats only, and no pathological changes were observed in female or castrated male rats. After 4-week of drug-free period, minimal to marked nephropathy and/or granuloma with crystalline-like clefts were present in intact males at ≥20 mg/kg/day indicating no reversibility.
Clinical Pathology
Hematology and coagulation parameters were unaffected by the administration of RG7129 in both male and female rats at all time points and dose levels. At 2 weeks, no associated serum chemistry findings were observed. Due to poor clinical condition and dehydration, blood urea nitrogen (BUN) and creatinine concentrations were elevated in males at 300 mg/kg/day in the 2-week pilot study. After 8 weeks, serum chemistry revealed RG7129-related increases in BUN in intact males compared with control males (Table 4).
Summary of Serum Chemistry and Urinalysis Findings for the 8-week GLP Toxicity Study and 8-week Mechanistic Study.
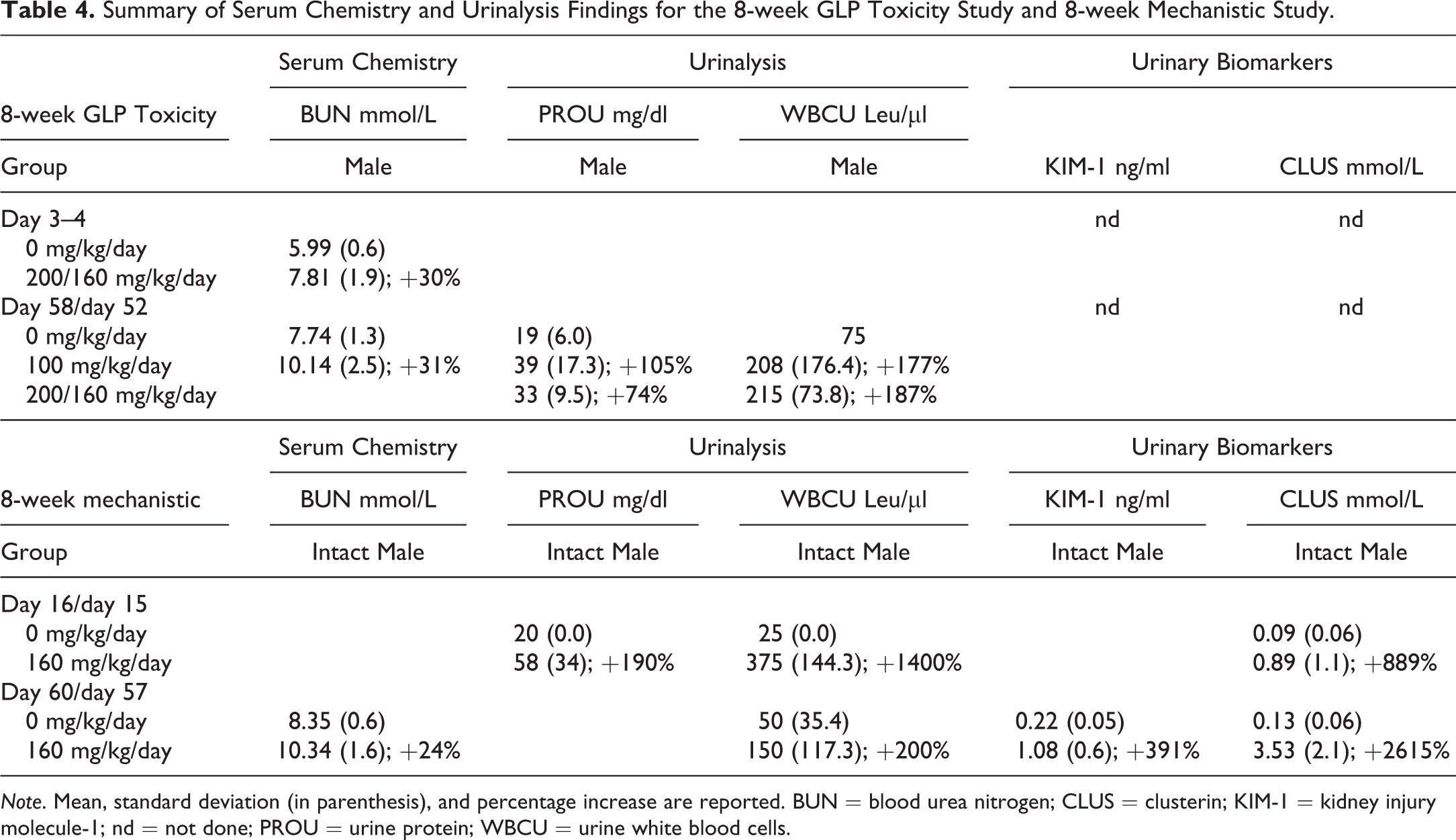
Note. Mean, standard deviation (in parenthesis), and percentage increase are reported. BUN = blood urea nitrogen; CLUS = clusterin; KIM-1 = kidney injury molecule-1; nd = not done; PROU = urine protein; WBCU = urine white blood cells.
Urinalysis findings related to RG7129 were present in intact males, including a moderate increase in urinary leukocytes (urinary leukocyte/granulocyte esterase activity) present after 2 weeks in intact males at 120 and 180/240 mg/kg/day and an increase in urinary protein (Table 4). This was also present in individual males previously treated at 200/160 mg/kg/day for 8 weeks after the 4-week recovery period on day 88. No RG7129-related urinalysis findings were observed in castrated males or females.
Urinary and serum kidney biomarkers were measured in the 8-week mechanistic study, and RG7129-related findings consisting of urinary KIM-1 and clusterin increases were also present in intact males only at 160 mg/kg/day from day 15 onward (Table 4). Both biomarkers were increased in a dose- and time-dependent manner and correlated with slight to marked nephropathy only. Animals with minimal nephropathy displayed no increase in urinary biomarkers. Additionally, one intact male at 160 mg/kg/day on day 60 with a marked nephropathy showed increases in serum KIM-1 and osteopontin. Additional findings in serum chemistry were considered unrelated to the nephropathy and consisted of increases in cholesterol in intact males, castrated male rats, and female rats compared with controls.
Toxicogenomic Analysis
At 8 weeks, toxicogenomic analysis showed that the dose level of 160 mg/kg/day of RG7129 had an impact on the kidney transcriptome in intact male rats only. These effects occurred on day 60, with marginal signs on day 16 and virtually no precursor sign on day 3. They consisted of upregulation of inflammatory markers, elements of tissue turnover and fibrosis, and known serum and urinary biomarkers of kidney insult, indicating tissue injury with inflammation and fibrotic processes on day 60 in intact males only, with early signs appearing on day 16 (data not shown). In the liver, α2u-globulin precursor transcripts PGCL3 and PGCL4 were detected but were comparable in intact males at 0 and 160 mg/kg/day on day 60.
Compound and Metabolite Identification
At 8 weeks, metabolite profiles in urine of intact and castrated male rats were qualitatively similar, and RG7129 and its oxidative metabolites M1, M2, and M5a/b were excreted in urine in both intact and castrated male rats (Figure 5). Uroliths found in the urinary bladder of selected intact male rats consisted primarily of unchanged parent compound (RG7129). Smaller amount of the major metabolites was also present.
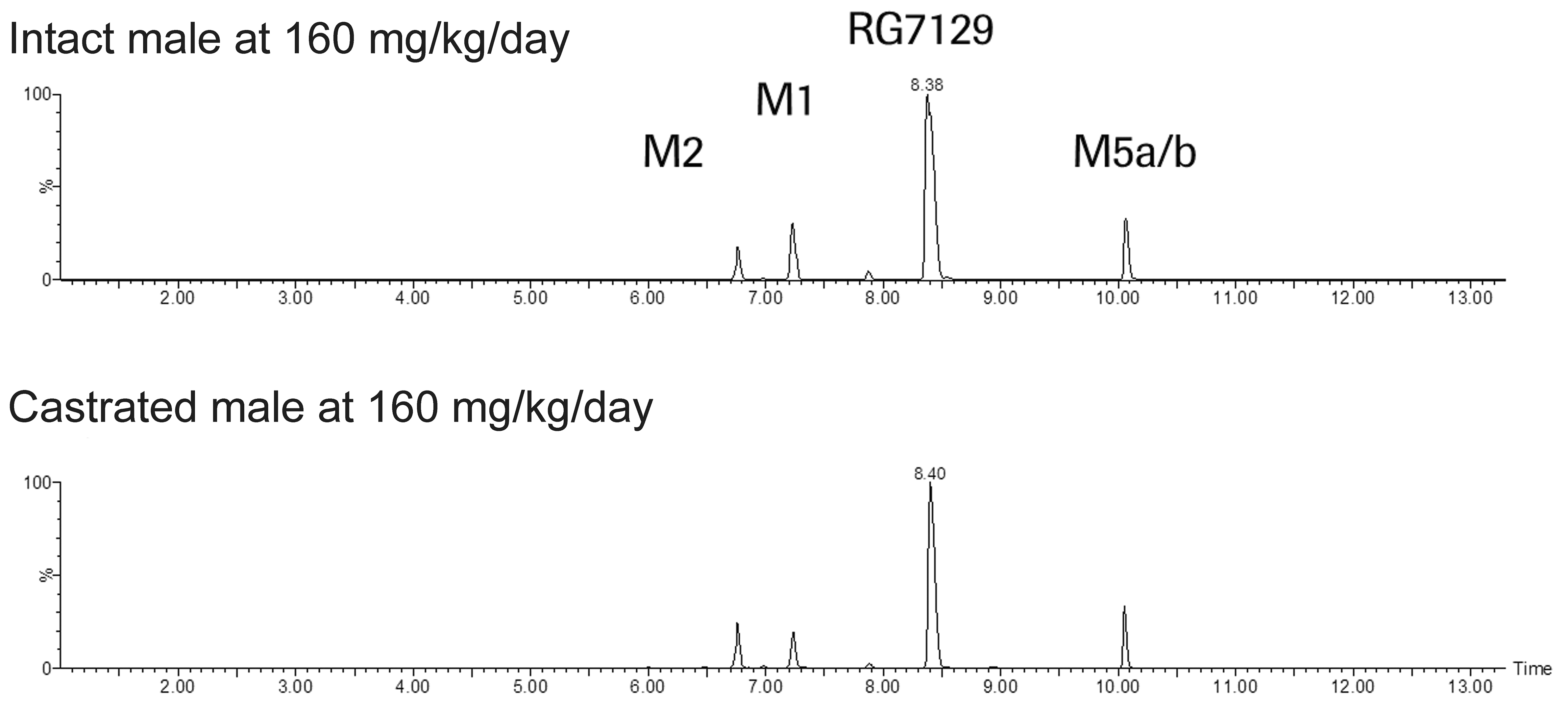
Metabolite profile in urine of male rats in the 8-week mechanistic study. Mass chromatograms obtained from urine of intact male and castrated male rats exposed at 160 mg/kg/day of RG7129.
Discussion
The studies reported herein demonstrated that daily administration of RG7129 resulted in three pathological processes in intact male rats kidneys. In addition to the classical α2u-globulin nephropathy, characterized by dose-dependent increases in accumulation of α2u-globulin droplets in the proximal tubular epithelium (confirmed by Heidenhain stain, immunochemistry and electron microscopy) and granular casts, lesions consistent with retrograde and obstructive nephropathy were observed. These additional pathological processes consisted of tubular degeneration and regeneration, tubular dilation with intratubular cellular casts, intraluminal eosinophilic material, interstitial inflammatory infiltrates, and interstitial fibrosis. Tubular changes extended from papilla to cortex, and the lesions mainly involved the cranial and/or caudal poles of the kidneys. Moreover, mixed cell infiltration was present, and the incidence of mononuclear cell infiltration was increased. Additionally, granuloma(s)/granuloma-like lesions with crystalline-like clefts were present in intact males receiving RG7129 at ≥20 mg/kg/day. Therefore, the histopathological findings observed in the kidney of intact male rats were considered to be a combination of three pathological processes following exposure to RG7129: α2u-globulin nephropathy, retrograde nephropathy, and obstructive nephropathy. This presents the first evidence of retrograde and obstructive nephropathy associated with α2u-globulin nephropathy.
α2u-globulin nephropathy is typically characterized by the accumulation of hyaline droplets in the P2 segment of proximal tubules and the formation of granular casts at the junction of the outer and inner stripes of the outer medulla. This represents the accumulation of cell debris where the P3 segment of the proximal tubule narrows into the descending limb of Henle (Hard 2008; Frazier et al. 2012). We observed the rapid accumulation of hyaline droplets and the formation of granular casts following exposure to RG7129 in intact male rats. Indeed, protein stain (i.e., Heidenhain stain; Hard 2008) and immunochemical staining confirmed that the accumulating protein observed within proximal tubules of intact male rats was α2u-globulin. Furthermore, the angular structure of the accumulated α2u-globulin within phagolysosomes of proximal tubular epithelium (Hard 2008) was also demonstrated by electron microscopy. The mechanism leading to the accumulation of α2u-globulin droplets in the proximal tubular epithelium in intact male rats was most likely the impaired lysosomal degradation of α2u-globulin in proximal tubular cells following reversible noncovalent binding of RG7129 to α2u-globulin, which leads to the accumulation of parent compound or metabolites (Hard et al. 1993; Swenberg and Lehman-McKeeman 1999). The absence of lesions in the kidney of castrated males and female rats clearly indicated that the lesions seen in intact male rats were dependent on the presence of α2u-globulin. However, RG7129 did not induce an increased synthesis of α2u-globulin shown by the toxicogenomic analysis of the liver where α2u-globulin precursor transcripts PGCL3 and PGCL4 were comparable in intact males at 0 and 160 mg/kg/day.
In addition to classical α2u-globulin nephropathy, we observed retrograde and obstructive nephropathy that was associated with α2u-globulin. Retrograde nephropathy was first defined by Hard and colleagues in 2009 after the reevaluation of several studies reporting histopathological changes induced by the oral administration of melamine in the rat kidney. Retrograde nephropathy was defined as a reflux effect stemming from the lower urinary tract. Examples include transient drug precipitation, infection, irritation, or partial obstruction that causes an ascending process up the papillary tubules into the cortex, where the lesions are ill-defined foci or areas of tubule basophilia coupled with dilation of the distal nephrons; the lesions are preferentially at the poles of the kidney (Hard, Flake, and Sills 2009; Frazier et al. 2012). The rat is likely to be predisposed to this condition as this species is spontaneously prone to vesicoureteral reflux (Sommer 1961; Corriere and Murphy 1967). In the present studies, this nephropathy was most likely caused by transient precipitation of RG7129 or its metabolites in the lower urinary tract due to a local high concentration. Obstructive nephropathy was considered due to precipitation of RG7129 or its metabolites within the tubules of the kidney. Obstructive nephropathy is characterized by tubular dilation and inflammation involving both mononuclear and neutrophil infiltration into the interstitium or tubule lumen due to intraluminal deposits causing tubule blockage (Hard et al. 1999; Frazier et al. 2012). The observation of outlines of intraluminal crystals, associated in some cases with multinucleated giant cells, confirmed the cause to be the precipitation of RG7129 and—to a lesser extend—its major metabolites in the collecting ducts due to a local high concentration by accumulation with α2u-globulin. The mechanistic study showed that retrograde and obstructive nephropathy only occurred in intact male rats, and pathological lesions were not observed in female or castrated male rats. This demonstrates a clear association between retrograde and obstructive nephropathy and the accumulation of α2u-globulin. The binding of α2u-globulin and subsequent accumulation in the kidney of intact male rats resulted in a local high compound load above the solubility limit, which lead to the combined nephropathy observed in these animals.
Changes in urine biomarkers KIM-1 and clusterin from day 15 onward in intact males only correlated with the histopathology findings of slight to marked nephropathy. Animals with minimal nephropathy displayed no increase in urinary biomarkers. Being increased in the mechanistic toxicity study already after 2 weeks, the two biomarkers were more sensitive than the traditional biomarker of renal injury BUN only being elevated after 8 weeks. However, animals with minimal nephropathy had no increase in any of the measured biomarkers of renal injury, limiting the use for clinically detecting early renal lesions in the current studies.
The toxicogenomic analysis of the kidney showed upregulation of inflammatory markers, elements of tissue turnover and fibrosis, and known serum and urinary biomarkers of kidney insult occurring on day 60 in intact males only, with marginal signs on day 16 and virtually no precursor sign on day 3. The findings correlated with the nephropathy and their different components of inflammation, tubular degeneration and regeneration, and interstitial fibrosis, respectively, from day 16 onward. However, there was no corresponding toxicogenomic signal to the histopathological finding of accumulation of hyaline droplets on day 3, limiting additional insights into the development of the nephropathy presented.
The absence of similar renal and urinary bladder lesions in female rats and male castrated rats indicated that the lesions reported above in intact males were dependent on the presence of α2u-globulin and further confirmed that the process of compound accumulation and sequential precipitation was α2u-globulin dependent. Besides the well-known α2u-globulin nephropathy, the accumulation of α2u-globulin and associated compound and/or metabolite was considered the prerequisite for the retrograde and obstructive nephropathies observed in intact male rats treated with RG7129.
In addition, the lesions in the urinary bladder consisting of inflammatory infiltrates and urothelial hyperplasia in intact males at high dose were considered to be RG7129 related and likely secondary to the chronic irritation by uroliths (Hard et al. 1999). Within these solid residues in the urinary bladder, we identified unchanged parent compound mixed with major metabolites, and this was most likely the cause of histopathological lesions in the kidney secondary to precipitation and vesicoureteral reflux. However, the sediment of the urine was unfortunately not checked for crystalluria.
Rats have a higher predisposition to developing drug precipitation in kidney tubules than humans. This is primarily due to the high urine-concentrating capacity occurring in the rat (Cohen, Wanibuchi, and Fukushima 2002; Khan and Alden 2002). An osmotic gradient develops in the loops of Henle to form the basis for a countercurrent system in which urine osmolality rises sharply from the glomerular filtrate to that in the papilla, where the terminal collecting ducts play a key role in the urine-concentrating effect (Oliver, Roy, and Jamison 1982; Khan and Alden 2002). Compared with humans, rats have extremely high levels of urinary osmolality, ranging approximately from 300 mOsmol/kg in the cortex to as high as 3000 mOsmol/kg in the papilla (Oliver, Roy, and Jamison 1982; Cohen 1995). The equivalent range in humans is approximately 100 to 1,000 mOsmol/kg (Cohen 1995). Furthermore, high urine pH, such as that of rats compared with humans (range of 5.5–7.5 vs. 5.0– 6.0, respectively), favors the formation of urinary crystals or precipitate in the rat (Cohen 1995). Moreover, α2u-globulin is a protein unique to adult male rats and absent in humans. Nephropathy developed as a consequence of α2u-globulin accumulation in male rats has been established to be a species- and sex-specific phenomenon (Baetcke et al. 1991; Hard et al. 1993); it does not occur in immature or female rats. No renal changes related to RG7129 were present in the female or castrated rat, including neither retrograde nephropathy nor obstructive nephropathy coupled with crystal precipitation. As mentioned before, it is well-known that female rats and castrated male rats do not develop α2u-globulin nephropathy. In female rats and castrated male rats, there was no α2u-globulin compound accumulation, and consequently, the high compound solubility observed in intact male rats was not reached and female and/or castrated rats did not develop retrograde and obstructive nephropathies. In addition, the nephropathies and histological manifestations reported herein are not relevant for humans or for species extrapolation in human risk assessment (Baetcke et al. 1991; Swenberg and Lehman-McKeeman 1999). This is because chemicals that bind to α2u-globulin do not bind to human members of the lipocalin superfamily of proteins (Baetcke et al. 1991; Swenberg 1993).
The studies presented here were limited in that binding of RG7129 to α2u-globulin was not demonstrated in vitro. In addition, renal tumors and exacerbation of CPN with prolonged treatment were not demonstrated due to limitations in the length of the current studies. It has been reported that renal tumors associated with α2u-globulin accumulation require at least 18 months of continuous exposure to the nephropathy-inducing chemical (Swenberg and Lehman-McKeeman 1999; Doi et al. 2007).
In conclusion, treatment with RG7129 resulted in significant changes only in the kidneys of intact male rats. Beside the lesions typical of α2u-globulin nephropathy, lesions consistent with retrograde and obstructive nephropathies were present in parallel. These were most likely secondary to precipitation due to a local high concentration of parent compound by accumulation with α2u-globulin. Importantly, retrograde and obstructive nephropathy have not been previously described in association to α2u-globulin. The absence of histopathological findings in female rats and castrated male rats with similar urine and kidney composition as intact male rats confirmed that the observed nephropathy was specific to the intact male rat. The current studies showed that all renal and urinary bladder findings secondary to the administration of RG7129 in intact male rats were dependent on the presence of α2u-globulin. However, as a species- and sex-specific syndrome, it is not expected to occur in humans or other species.
Footnotes
Acknowledgments
The authors would like to thank the numerous associates and scientists in the different functions for their excellent support. The authors also thank Anna Mestres-Missé of Meridian HealthComms Ltd (Plumley, UK) for medical writing assistance.
Author Contributions
Authors contributed to conception or design (BJ, CF, AE, JH, MA, BL); data acquisition, analysis, or interpretation (BJ, CF, AE, JH, MA, BL); drafting the manuscript (BJ); and critically revising the manuscript (BJ, CF, AE, JH, MA, BL). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
