Abstract
Mastic gum extracts are widely used as herbal remedies and are being tested for several clinical indications. Nevertheless, information on their safety is limited. RPh201 is an extract of the mastic gum, formulated and stabilized in a proprietary method, which is being developed as a novel drug candidate for neurological indications. The aim of this study was to assess the systemic toxic potential of RPh201, administered twice weekly by subcutaneous injections to minipigs, after 39 weeks of administration followed by a recovery period of 6 weeks. No clinical or dose-related signs were observed, but treatment-related findings were seen at the injection sites of the high-dose animals, composed of abscesses, chronic inflammation, and subcutaneous fibrosis. Abscesses >30 mm in size, graded as marked severity, were confined to the high-dose group and were considered as adverse. Minimal-slight subcutaneous and lymph nodes abscesses seen in control, low, and intermediate doses, related to the vehicle (cottonseed oil), were not considered as adverse. Additionally, minimal-to-slight cystic spaces or vacuolation related to the vehicle were observed in the skin, lymph nodes, kidney, and lungs. These findings were considered not to be adverse. The no-observed-adverse-effect level was considered to be 12.5 mg/kg/occasion.
Mastic gum is extracted from the Pistacia lentiscus plant, which grows in the Greek island of Chios (Suzuki et al. 2017). It has been traditionally used as a dietary additive and as a flavoring agent (Spyridopoulou et al. 2017), but it has also been widely used as a herbal remedy, mostly in the Mediterranean area (Jin et al. 2017). In recent years, there is a growing interest in the potential beneficial properties of mastic gum extracts, and a large number of scientific studies reported on their biological activities (Jin et al. 2017; Spyridopoulou et al. 2017; Suzuki et al. 2017). These range from antibacterial activities (Karygianni et al. 2014; Koychev et al. 2017; Miyamoto, Okimoto, and Kuwano 2014) via antiviral activities (Ezz Eldin and Badawy 2015; Loizzo et al. 2008; Ozcelik et al. 2005; Sakagami et al. 2009) to antioxidant activities (Ahmad et al. 2010; Gholami et al. 2016; Hatamnia et al. 2016; Tolooei and Mirzaei 2015; Toul et al. 2017). In addition, many studies have reported on the anticancer properties of the mastic gum extracts, both for solid and hematologic cancers (Balan et al. 2005; Balan et al. 2007; Dimas et al. 2009; Giaginis and Theocharis 2011; He et al. 2006; Loutrari et al. 2006; Magkouta et al. 2009; Moulos et al. 2009; Sakagami et al. 2009).
Taking into consideration this long list of potential positive properties, mastic gum extracts are being incorporated into health and care products and are a major component in the food industry (Vlastos et al. 2015). They are also being tested for many clinical indications such as Crohn’s disease (Kaliora et al. 2007a, 2007b), healing of peptic ulcers (Al-Habbal, Al-Habbal, and Huwez 1984; Huwez and Al-Habbal 1986; Huwez et al. 1998), and dental ulcers (Saez, Lopez, and Romero 2005; Takahashi et al. 2003). In spite of their increasing popularity, data on their safety are very limited.
RPh201 is an extract of the mastic gum, formulated and stabilized in a proprietary method, which is being developed as a novel drug candidate for neurological indications. It has already been shown to be effective for patients with previous nonarteritic ischemic optic neuropathy in a phase 2 clinical trial (Hazan et al. 2016). However, information on its long-term safety profile is limited. We have recently reported that RPh201 was found to have no adverse effects in Sprague-Dawley rats when administered subcutaneously (Ramot et al. 2017). In this study, injection site reactions were observed to be of greater severity in the high-dose group, but they were of mild severity and reversible.
In order to gain more information on the safety and toxicokinetic profile of RPh201, we have conducted a study accomplished in compliance with the Good Laboratory Practice (GLP) standards. The study included 39-week subcutaneous (twice weekly) administration in minipigs, with a 26-week interim phase and a 6-week recovery period.
Materials and Method
Animal Husbandry and Maintenance
A total of 32 male and 32 female Göttingen minipigs, approximately 3 to 4 months old, weighing 8.3 to 12.3 kg, were supplied by Ellegaard Göttingen Minipigs (Dalmose, Denmark). The animals were housed in a limited access animal facility. Room controls were set to maintain temperature and relative humidity at the range of 15 to 24°C and 40% to 70%, respectively. The rooms were artificially lit for 12 hr each day. The study animals were acclimatized for at least 4 weeks before treatment initiation.
Potable drinking water from the public supply was available without restrictions via automatic valves that were checked daily. All animals were supplied Special Diets Services SMP2 811571 Minipig Diet on arrival, and after a short period, animals were transitioned onto Teklad Minipig Diet 8753C; 8753C diet was supplied from no later than the start of pretreatment recordings until study completion.
The Göttingen minipig was chosen for this study based on physiologically similar cardiovascular, gastrointestinal, and, notably, integumentary systems to humans (Manno et al. 2016; Ramot et al. 2015; Vezzali et al. 2011).
The study was conducted in accordance with the requirements of the European Commission Directive 2004/10/EC, and in compliance with 21 CFR part 58, Good Laboratory Practice for Nonclinical Laboratory Studies, the UK Good Laboratory Practice Regulations (Statutory Instrument 1999 No. 3106, as amended by Statutory Instrument 2004 No. 994), and the Organization for Economic Cooperation and Development Principles of Good Laboratory Practice (as revised in 1997), ENV/MC/CHEM(98)17. The experiment was conducted at Envigo, Cambridgeshire, UK.
Test Material and Treatment Protocol
The test material, RPh201, was supplied in a BHT-stabilized cottonseed oil by Regenera Pharma, Nes-Ziona, Israel. BHT-stabilized cottonseed oil was also used as vehicle. An oil formulation has been used because the botanical drug substance is essentially insoluble in aqueous carriers. Cottonseed oil has been specifically used because it has relatively low irritability compared to other oils (2001). The formulation used in the safety study is the same as the one that has been used in the clinical studies in humans (phase 1–3). The study design is summarized in Table 1 and consisted of a control group and three RPh201 treatment groups. Main study animals were treated for 39 weeks with an interim sacrifice point at 26 weeks. Recovery animals were treated for 39 weeks followed by a 6-week off-dose period. Treatment was administered twice weekly as subcutaneous injections on the left and right sides of the flanks. Two sites on each side were marked and used in rotation.
Experimental Design, Group Allocation, and Number of Animals.

a Expressed in terms of the free base.
Clinical Observations and Mortality and Physical Examination
Animals were inspected visually at least twice daily, and pens were inspected daily for evidence of ill-health. A detailed weekly physical examination was performed on each animal. Detailed clinical observations were performed on both dosing days during weeks 1 to 4 and thereafter on a dosing day once weekly during weeks 5 to 13 and once every two weeks thereafter. These observations were done before injection, 1- to 2-hr postdosing and at the end of the same working day. The injection sites were observed 1- and 3-hr postdosing on dosing days. During the recovery phase, observations of the animals and their pens were recorded at least once per day.
The weight of each animal was recorded (prior to feeding) weekly during the acclimatization period, on the day that treatment commenced (week 0), weekly throughout the study, and before necropsy. Food consumption was calculated by weighing the amount of food supplied to each animal and subtracting any remaining food together with an additional estimate of any that had been spilt. This was recorded daily during the acclimatization period and throughout the study. The eyes of the animals were examined by means of a binocular indirect ophthalmoscope pretreatment, on weeks 25 to 27 and 37 to 39 on all main and recovery animals, and at recovery week 6 for all recovery animals.
Electroretinogram (ERG) and Tonometry
Under general anesthesia, an ERG was conducted on both eyes of main and recovery animals. Both scotopic and photopic ERG evaluations were carried out on the same day. The animals were habituated to darkness for a minimum of 2 hr prior to the scotopic evaluation. Under the same general anesthetic as for ERG, measurement of intraocular pressure was carried out via tonometry on both eyes of main and recovery animals. These investigations were performed pretreatment, on weeks 25 to 27 and 37 to 39 on all main and recovery animals, and at recovery week 6 for all recovery animals.
Electrocardiography
Electrocardiograph tracings were recorded predose and 0.5, 1, 2, 4, 8, and 24 hr after completion of dosing from all animals pretreatment and on weeks 13, 26, 37, and 39. Recovery animals were tested on recovery week 6.
Clinical Pathology and Toxicokinetics and Urinalysis
Blood samples were obtained via the jugular vein/cranial vena cava from all animals pretreatment, on weeks 13 and 26, at week 39 from both the main and recovery-phase animals, and at recovery week 6 from the recovery-phase animals. The blood was analyzed for hematological, coagulation, and biochemical parameters. Urine samples were collected at the same time points.
Blood samples for toxicokinetics and immunogenicity were obtained from the main-phase animals on day 1 and week 13, 26, and 39 at 0.5, 1, 2, 4, 8, and 24 hr after dosing. Samples from all treated animals and two samples from each control animal per occasion were analyzed. One sample around T max (2-hr time point) and one sample in the elimination phase (8-hr time point) were selected for analysis.
Euthanasia and Necropsy
Animals of the main study were sacrificed following 39 weeks of treatment with an interim sacrifice at 26 weeks. Recovery animals were sacrificed following 39 weeks of treatment and 6 weeks of recovery. Animals were euthanized with an overdose of sodium pentobarbitone solution (200 mg/ml) by intravenous injection followed by exsanguination. All animals were subject to a detailed necropsy.
Organ Weights
Organ weights of the following organs were recorded: adrenal glands, brain, epididymides, heart, kidneys, liver, lungs, ovaries, pituitary gland, prostate gland, spleen, testes, thymus, thyroid gland, and uterus with cervix.
Microscopic Observations
Tissues were preserved in 10% neutral-buffered formalin except for the testes that were preserved in modified Davidson’s fluid and the eyes that were preserved initially in Davidson’s fluid and subsequently transferred to 70% industrial methylated spirit (IMS).
Histopathological evaluation of hematoxylin and eosin–stained tissues was performed on all animals. The following tissues were evaluated: adrenals, thoracic aorta, brain, cecum, colon, dosing sites, duodenum, epididymides, esophagus, eyes, femur, gallbladder, Harderian gland, heart, ileum, jejunum, kidneys, lacrimal gland, liver, lungs, lymph nodes (mandibular, mesenteric, and left inguinal), optic nerves, ovaries, pancreas, parathyroids, Peyer’s patches, pituitary, prostate, salivary glands (submandibular, parotid, and sublingual), sciatic nerves, skeletal muscle, skin with mammary glands, spinal cord, spleen, sternum, stomach, testes, thymus, thyroid, trachea, urinary bladder, uterus with cervix, and vagina. In addition, Oil Red O staining was performed on the lungs from a few animals to confirm presence of oil, and Gram’s stain was performed on the injection sites from a few animals with abscesses to check for the presence of bacteria. Findings were either reported as “present” or assigned a severity grade. In the latter case, one of the following five grades was used—minimal, slight, moderate, marked, or severe.
Abscesses seen in the injection sites and lymph nodes were graded according to the following criteria: Minimal: between one and five abscesses up to 2 mm in size; slight: more than five abscesses up to 2 mm and/or abscesses up to 10 mm in size. Usually, only one abscess larger than 2 mm was present at a single-injection site; moderate: abscesses 10 to 30 mm in size; marked: abscesses larger than 30 mm in size.
Abscesses graded as moderate or marked were usually a single abscess that was detected macroscopically, although smaller abscesses (generally smaller than 2 mm) were sometimes present at the same injection site. The distinction between moderate and marked was made from the size of the macroscopic description, as abscesses larger than 30 mm were too large to fit on the slide and may also have ruptured during harvesting and preparation of the slide. Recovering abscesses usually occurred singularly at an injection site and were graded in the same way based on their size. The same grading system was used for abscesses in lymph nodes that were all graded as minimal or slight.
Statistical Analysis
All statistical analyses were carried out separately for males and females (except for electrocardiography, for which sexes were combined) using the individual animal as the basic experimental unit. A parametric analysis was performed for body weight, food consumption, electrocardiography, organ weight, and clinical pathology data if Bartlett’s test for variance homogeneity was not significant at the 1% level. The F 1 approximate test was applied. If the F 1 approximate test for monotonicity of dose-response was not significant at the 1% level, Williams’ test for a monotonic trend was applied. If the F 1 approximate test was significant, suggesting that the dose-response was not monotone, Dunnett’s test was performed instead. A nonparametric analysis was performed if Bartlett’s test was still significant at the 1% level following both logarithmic and square-root transformations. The H 1 approximate test, the nonparametric equivalent of the F 1 test described above, was applied. If the H 1 approximate test for monotonicity of dose-response was not significant at the 1% level, Shirley’s test for a monotonic trend was applied. If the H 1 approximate test was significant, suggesting that the dose-response was not monotone, Steel’s test was performed instead.
For clinical pathology data, if 75% of the data (across all groups) were the same value, Fisher’s exact tests were performed. Treatment groups were compared using pairwise comparisons of each dose group against the control. For organ weight data, analysis of covariance was performed using terminal body weight as covariate, unless nonparametric methods were applied. The treatment comparisons were made on adjusted group means in order to allow for differences in body weight, which might influence the organ weights. Significant differences between the groups compared were expressed at the 5% (p < .05) or 1% (p < .01) level.
Results
Clinical Observations (Including Ocular ERG and Tonometry, and Cardiac Electrocardiography) and Organ Weights
The behavior of the animals has been unaffected following 39 weeks of treatment, and there were no test article–related clinical signs. One female animal from group 1 had to be euthanized early on day 88 for welfare reasons after sustaining an injury during routine blood sampling.
There were no treatment-related effects on body weight or food consumption during the 39 weeks of treatment or 6-week recovery phase. The terminal investigations conducted following 39 weeks of treatment revealed lower absolute and body weight–adjusted liver weights in all treated groups when compared to controls (Table 2). This observation did not show a dose relationship, and the liver weights of individual animals showed considerable overlap with the concurrent control range of values. Considering also that there were no significant histopathological findings in the liver and the absence of any differences in liver weights after a 6-week off-dose period compared to controls, it was considered that any variation in liver weights was fortuitous and unrelated to treatment.
Liver Weights at Week 39.
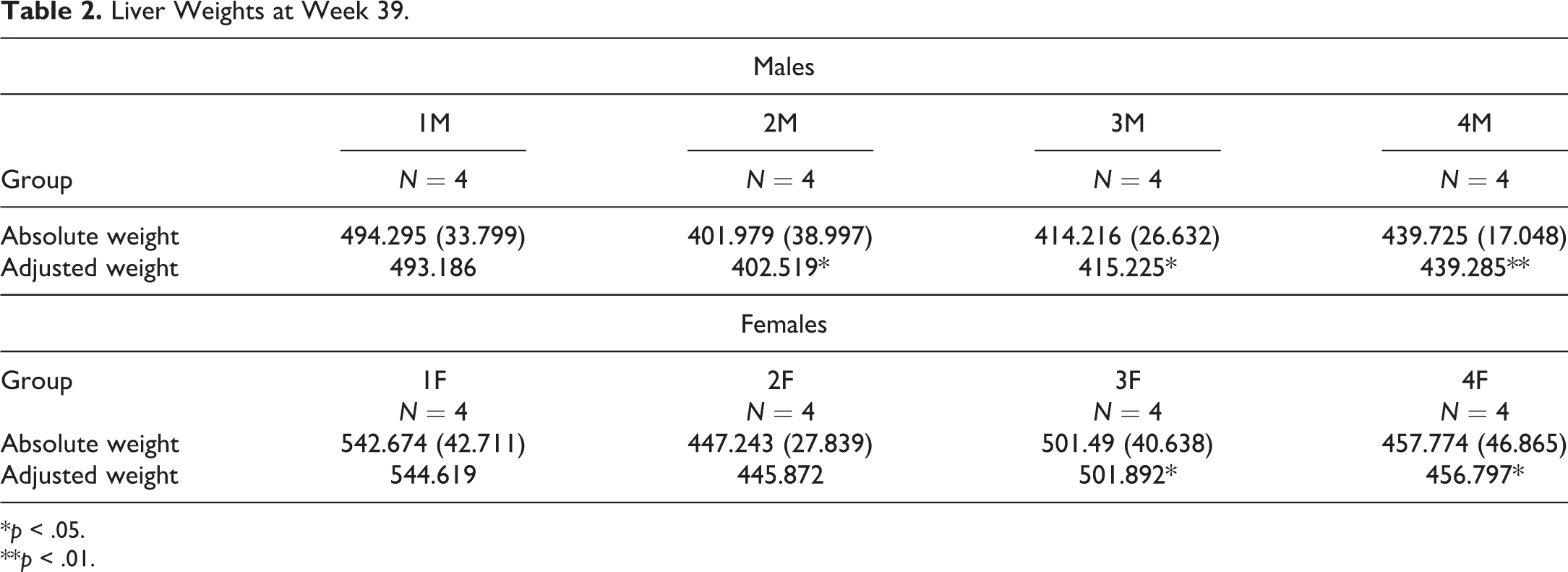
*p < .05.
**p < .01.
The terminal investigations, conducted following 26 weeks of treatment, did not reveal any organ weight differences from the control group, which were related to treatment.
All other differences from controls were minor, showed considerable overlap with the concurrent control range of values or lacked dose dependency, are not considered to be test item related, and are therefore attributed to normal biological variation.
Posterior suture line opacity was observed in the lens of the eye of four animals: one male from group 2 at week 39; one male from group 4 at week 39; one female from group 4 at week 26, 39, and recovery week 6; and one female from group 4 at recovery week 6. There was no effect on the retina at ERG examination, and no ocular hypo or hypertension was evident. There were no effects on electrocardiography parameters.
Clinical Pathology
Hematology investigations conducted in week 26 of treatment revealed slightly higher mean neutrophil counts observed in both sexes receiving 12.5 or 50 mg/kg/occasion when compared to concurrent controls (Table 3). This slightly higher neutrophil count was still evident in the week 39 hematology investigations but only in males given 12.5 mg/kg/day when compared to concurrent controls. There was full recovery during the 6-week off-dose period, although this was assessed in females only due to clotted samples in the males.
Hematology Results.
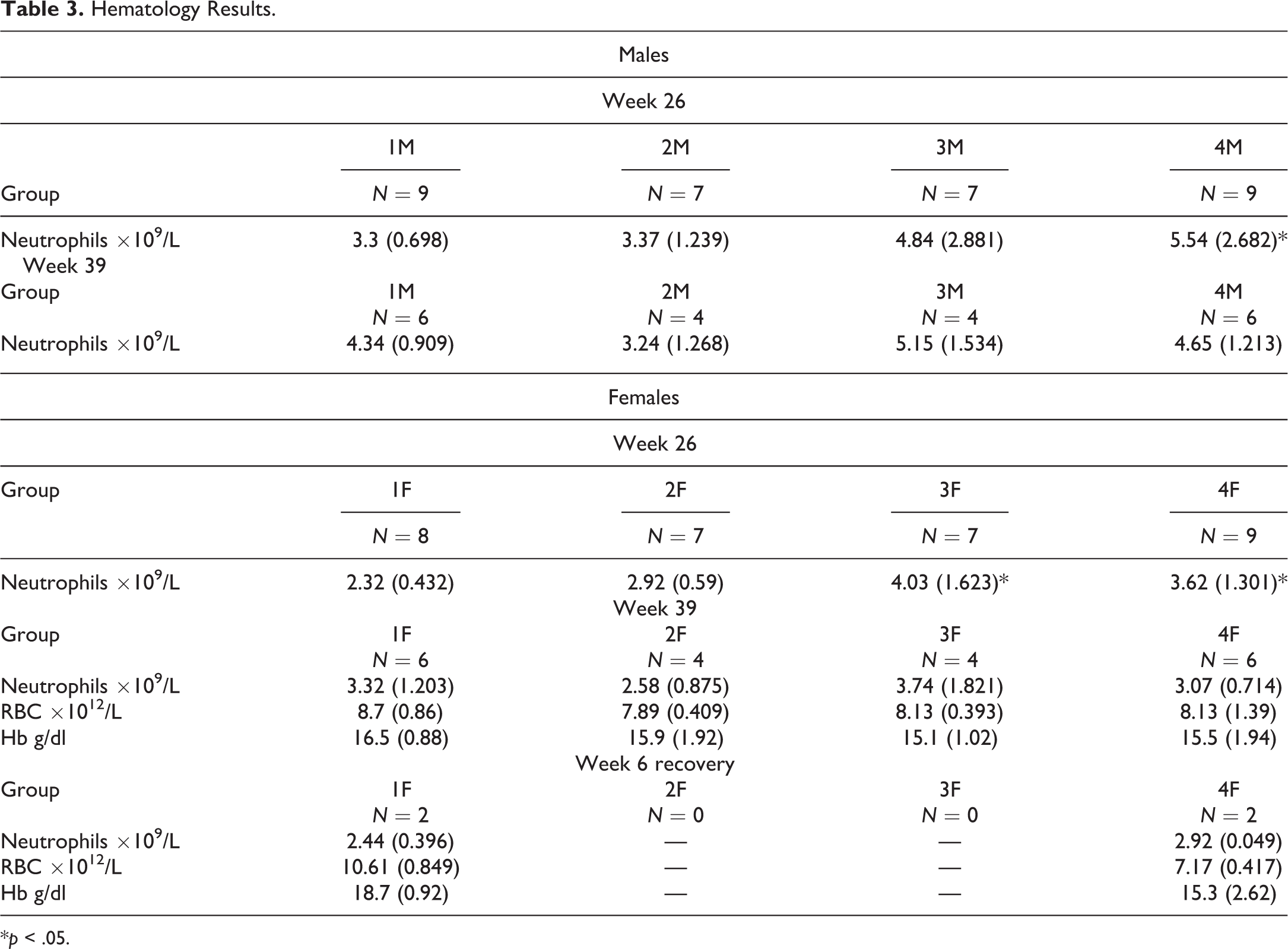
*p < .05.
In week 39, a lower mean erythrocyte count and hemoglobin concentration were observed in females receiving 50 mg/kg/occasion in comparison with the controls (Table 3). The lower mean values were mainly due to particularly low values observed for one animal. There was partial recovery in both parameters following 6 weeks off-dose, as one female still showed lower erythrocyte count and hemoglobin concentration. There were no other differences from the controls in clinical pathology or urinalysis parameters observed at the week 13, 26, or 39 investigations that were considered to be related to treatment.
Toxicokinetics
The rate of systemic exposure of minipigs to RPh201, measured as the two components masticadienonic acid (MDA) and isomasticadienonic acid (IMDA), appeared to be characterized by nonlinear, subproportional (dose dependent) kinetics over the dose range from 3.1 to 50.0 mg/kg/day on day 1 and during weeks 13, 26, and 39 of the 39-week subcutaneous toxicity study (data not shown). However, the extent of systemic exposure of minipigs to MDA and IMDA generally appeared to be characterized by dose-independent (linear) kinetics over the dose range 3.1 to 50.0 mg/kg/day on day 1 and during weeks 13, 26, and 39 of the 39-week subcutaneous toxicity study. Systemic exposure of minipigs to MDA was generally comparable to that of IMDA.
The data also provided evidence that the systemic exposure of female minipigs to MDA and IMDA was generally slightly lower than that of males after repeated administration and that there was some accumulation of MDA and IMDA at the 3.1 and 12.5 mg/kg/day dose levels in both sexes after repeated subcutaneous administration of RPh201.
Macroscopic Observations
Injection sites
Clear oily liquid was found at the injection sites of all groups at the 26- and 39-week sacrifice points. In the 6-week recovery group, clear oily liquid was also observed in controls and animals previously treated with 50.0 mg/kg/occasion with a similar incidence. Thickening was seen at the 39-week sacrifice point in both sexes treated with 50 mg/kg/occasion, control animals of both sexes, and in a single female treated with 12.5 mg/kg/occasion. Pale white areas were seen in all treated groups at the 26- and 39-week sacrifice points and in a male and female control animal. At the 6-week recovery group, pale areas were seen in controls and animals previously treated with 50.0 mg/kg/occasion with a higher combined incidence in previously treated animals. Raised areas were seen at the week 26 time point in females treated with 50 mg/kg/occasion and at the 39-week sacrifice necropsy in both sexes treated with 50 mg/kg/occasion, in female animals treated with 12.5 mg/kg/occasion, and in one female control animal. At the 6-week recovery necropsy, raised areas were seen in females treated with 50.0 mg/kg/occasion.
Lymph nodes
At the 26-week sacrifice necropsy, enlargement of the right and left inguinal lymph nodes was found in one female receiving 50.0 mg/kg/occasion, with pale areas also noted in the left inguinal node of a different female from the same dose group. At the 39-week and 6-week recovery sacrifice points, clear oily liquid, enlargement, and pale areas were seen in various lymph nodes (including axillary, inguinal, mandibular, and mediastinal lymph nodes) in animals treated with 50 mg/kg/occasion and in the control group. Enlargement was also seen in one female animal treated with 12.5 mg/kg/occasion, and pale areas were also seen in a male and female animal in this group.
Microscopic Observations
Treatment-related findings
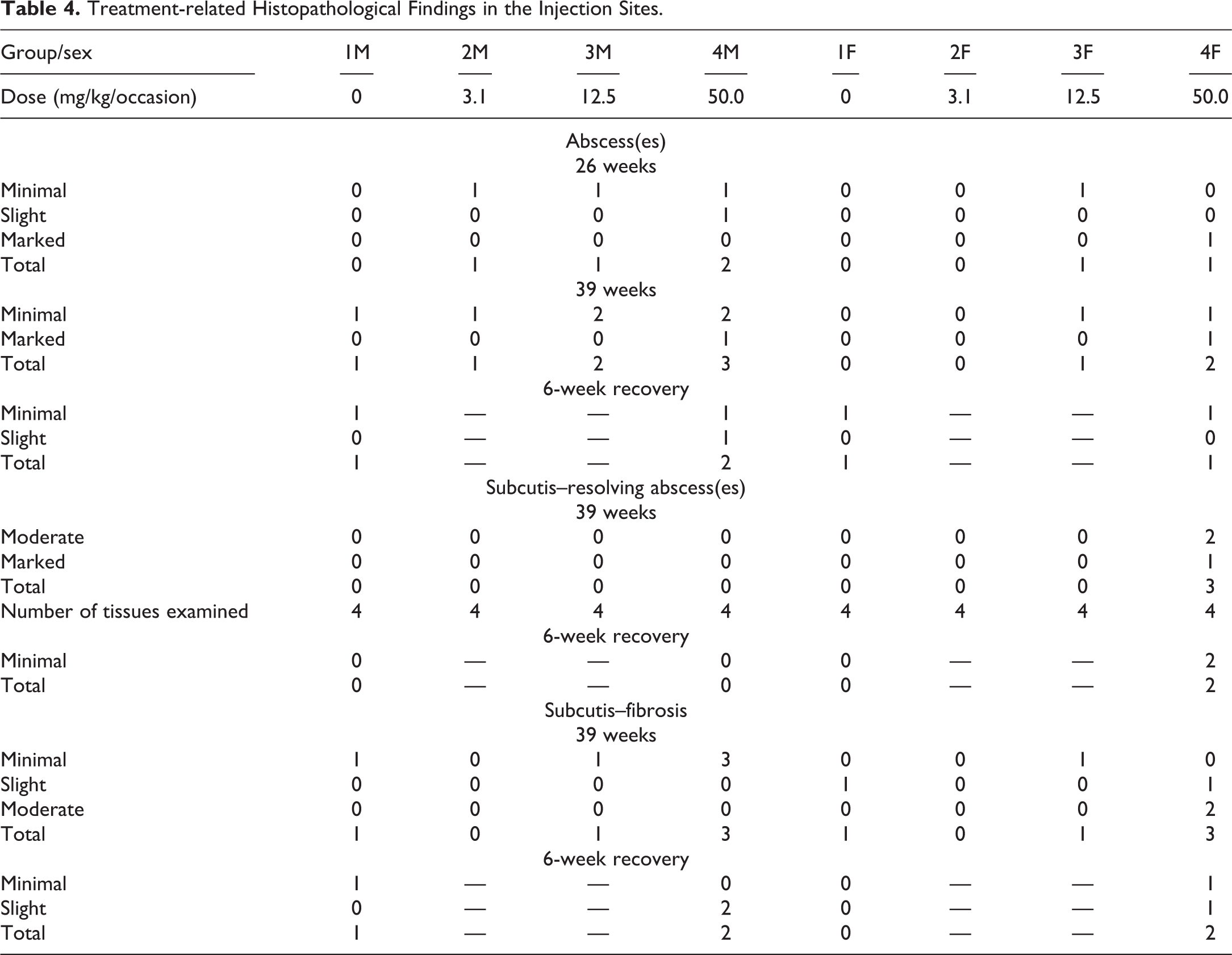
Treatment-related findings were observed at injection sites of animals from all sacrifice points and are summarized in Table 4. Since the findings were comparable for all four injection sites evaluated, only injection site 3 is presented in the table. The histopathological findings comprised of abscesses at the 26-week and 39-week sacrifice and recovery phase and recovering abscesses and an increase in fibrous tissue in the 39-week sacrifice and recovery phases. Abscesses were characterized by a circular space containing aggregates of neutrophils with a variable chronic inflammatory component and a fibrous capsule of varying thickness. Minimal-graded abscesses were also seen in the control group, and this grade is considered to be related to the vehicle. Abscesses graded as moderate or marked were confined to animals receiving 50 mg/kg/occasion. Gram stain was performed on the injection sites from a few animals with abscesses to check for the presence of bacteria, and no bacterial colonies were present. Resolving abscesses were seen at the 39-week sacrifice point in females receiving 50.0 mg/kg/occasion. These were characterized by fibrous tissue, mineralization, and with few neutrophils present. At the 6-week recovery sacrifice, the severity of the abscesses, resolving abscesses, and fibrosis was confined to minimal or slight, which indicated partial recovery from the findings seen at 26 and 39 weeks. Representative photos of the abscesses from the main phase are shown in Figure 1A–E. Representative photos of the resolving abscesses from the recovery phase are shown in Figure 1H–J.
Treatment-related Histopathological Findings in the Injection Sites.
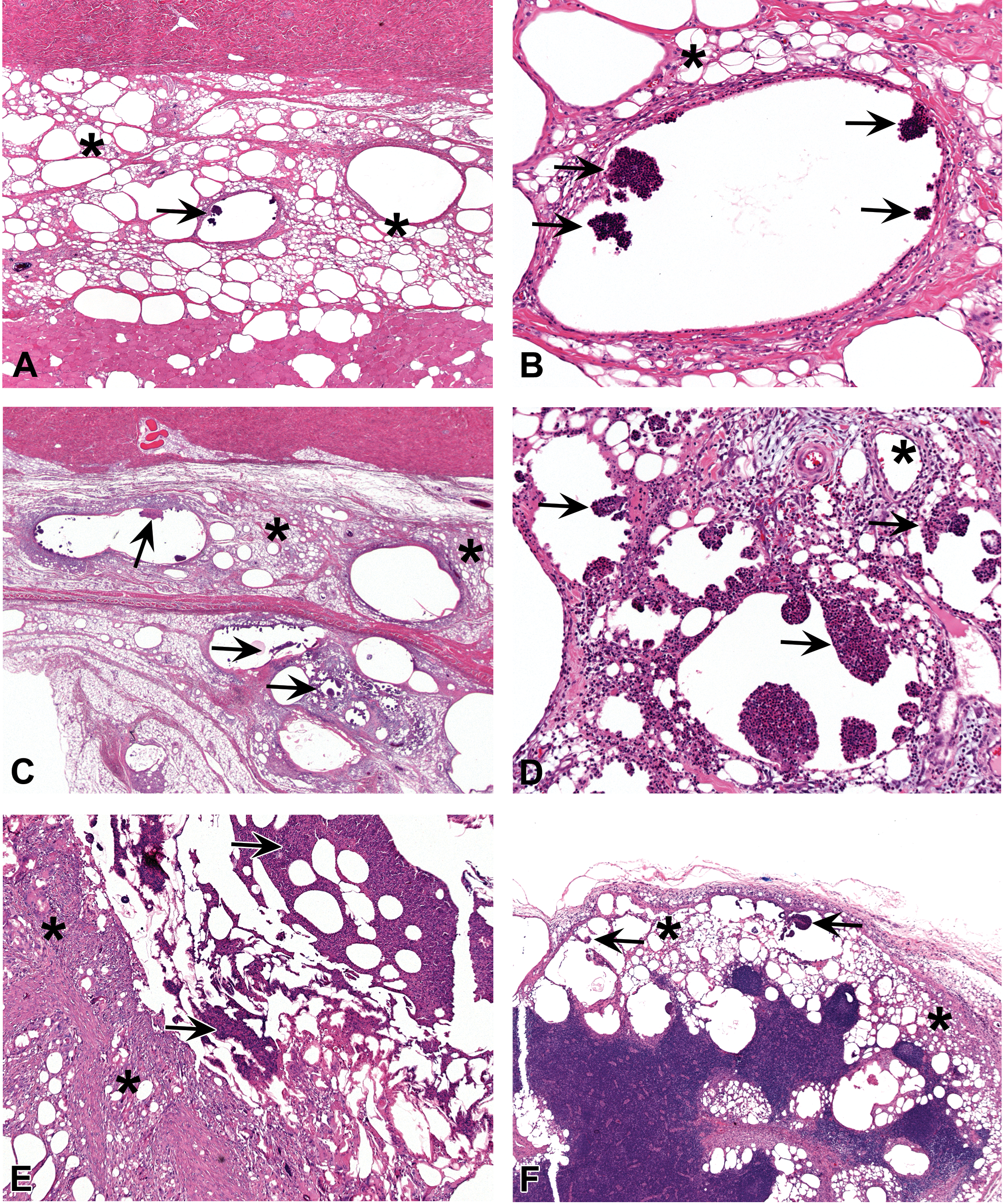
(A) Histopathological section of the subcutaneous injection site in minipig from the high-dose group (50 mg/kg), main phase of the study (i.e., sacrifice at the end of 6 months of treatment). The tissue reaction consists of chronic inflammation (asterisks). These findings were comparable to the control group and were considered as related to the vehicle. In addition, abscesses (arrow) in the subcutis (minimal grade) were seen, characterized by a cystic space containing aggregates of neutrophils with a variable chronic inflammatory component and a fibrous capsule of varying thickness. H&E. (B) Histopathological section of the subcutaneous injection site in minipig from the high-dose group (50 mg/kg), main phase of the study. Higher magnification of the previous photo. Note the abscess (arrows) in the subcutis (minimal grade), characterized by a cystic space containing aggregates of neutrophils with a variable chronic inflammatory component and a fibrous capsule of varying thickness. The surrounding tissue reaction consists of chronic inflammation (asterisk). H&E. (C) Histopathological section of the subcutaneous injection site in minipig from the high-dose group (50 mg/kg), main phase of the study (i.e., sacrifice at the end of 6 months of treatment). The tissue reaction consists of chronic inflammation (asterisks), and intermixed with empty spaces reflecting the washed-out cotton-seed oil vehicle. These findings were comparable to the control group and were considered as related to the vehicle. In addition, abscesses (arrows) in the subcutis (slight grade) were seen. H&E. (D) Histopathological section of the subcutaneous injection site in minipig from the high-dose group (50 mg/kg), main phase of the study. Higher magnification of the previous photo. Note the abscess (arrows) in the subcutis (slight grade). The surrounding tissue reaction consists of chronic inflammation (asterisks). H&E. (E) Histopathological section of the subcutaneous injection site in minipig from the high-dose group (50 mg/kg), main phase of the study. The tissue reaction consists of chronic inflammation (asterisk), and intermixed with empty spaces reflecting the washed-out cotton-seed oil vehicle. Note the abscess (arrows) in the subcutis (marked grade). This grade of lesion is considered treatment related. H&E. (F) Histopathological section of the inguinal lymph node (regional to the injection site) in minipigs from the intermediate-dose group (12.5 mg/kg), main phase of the study (i.e., sacrifice at the end of 6 months of treatment). The tissue reaction consists of chronic inflammation (asterisks), and intermixed with empty spaces reflecting the washed-out cotton-seed oil vehicle. These findings were comparable to the control group and were considered as related to the vehicle. In addition, minimal-grade abscesses (arrows) were seen. H&E. (G) Histopathological section of the inguinal lymph node (regional to the injection site) in a minipig from the intermediate-dose group (12.5 mg/kg), main phase of the study. Higher magnification of the previous photo. Note the minimal-grade abscess (arrows). The surrounding tissue reaction consists of chronic inflammation (asterisk). H&E. (H) Histopathological section of the subcutaneous injection site in a minipig from the high-dose group (50 mg/kg), recovery phase of the study. A resolving abscess. The tissue reaction is characterized by fibrous tissue, mineralization (arrows), and with only few neutrophils present (asterisk). H&E. (I) Histopathological section of the subcutaneous injection site in minipig from the high-dose group (50 mg/kg), recovery phase of the study. Another example of a resolving abscess. The tissue reaction is characterized by fibrous tissue (arrow) and with only few neutrophils present (asterisk). H&E. (J) Histopathological section of the subcutaneous injection site in minipig from the high-dose group (50 mg/kg), recovery phase of the study. Area of fibrosis (asterisks). The increase in subcutaneous fibrosis was also seen in the 39-week and 6-week recovery sacrifice, mainly in the high-dose animals. H&E.
Vehicle-related findings
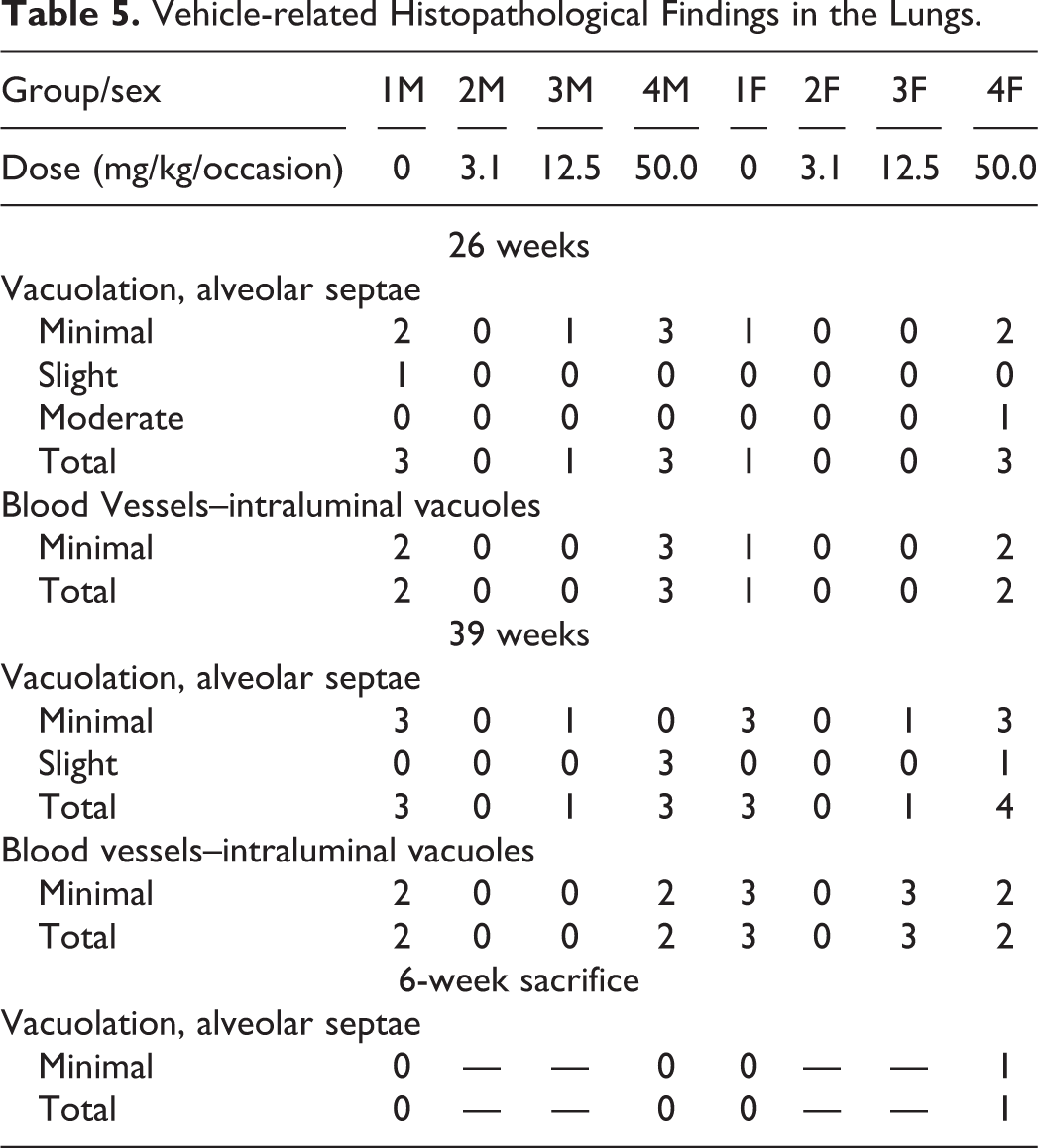
Lungs: Vacuolation in the alveolar septae and intraluminal vacuoles in blood vessels were seen in control animals and those receiving 12.5 and 50 mg/kg/occasion (Table 5). Oil Red O staining was performed on the lungs with vacuolation from a few animals to confirm the presence of oil, and all the tissues stained were positive confirming the presence of oil vacuolation in the alveolar septae and intraluminal vacuoles in blood vessels. Partial recovery was evident for all findings at the 6-week recovery point.
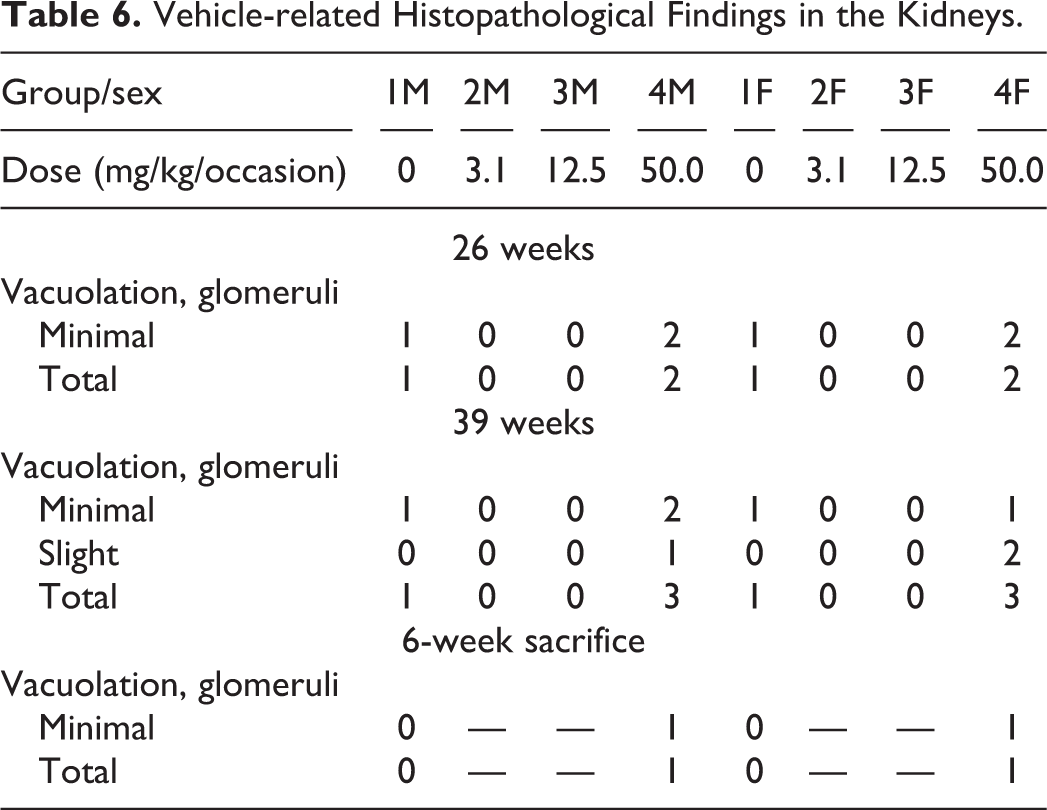
Kidneys: Vacuolation in the glomeruli was seen in control animals and those receiving 50.0 mg/kg/occasion (Table 6). Partial recovery was evident for all findings at the 6-week recovery point.
Injection sites: Cystic spaces and chronic inflammation were seen in all treated groups and controls with the severity generally correlating with the volume of vehicle administered (Table 7). The chronic inflammation was characterized by mononuclear cells (lymphocytes and macrophages), foreign body giant cells, fibrosis, and occasional neutrophils. Incidence and severity of the findings did not show signs of recovery after 6 weeks.
Vehicle-related Histopathological Findings in the Lungs.
Vehicle-related Histopathological Findings in the Kidneys.
Vehicle-related Histopathology Findings at Injection Sites.
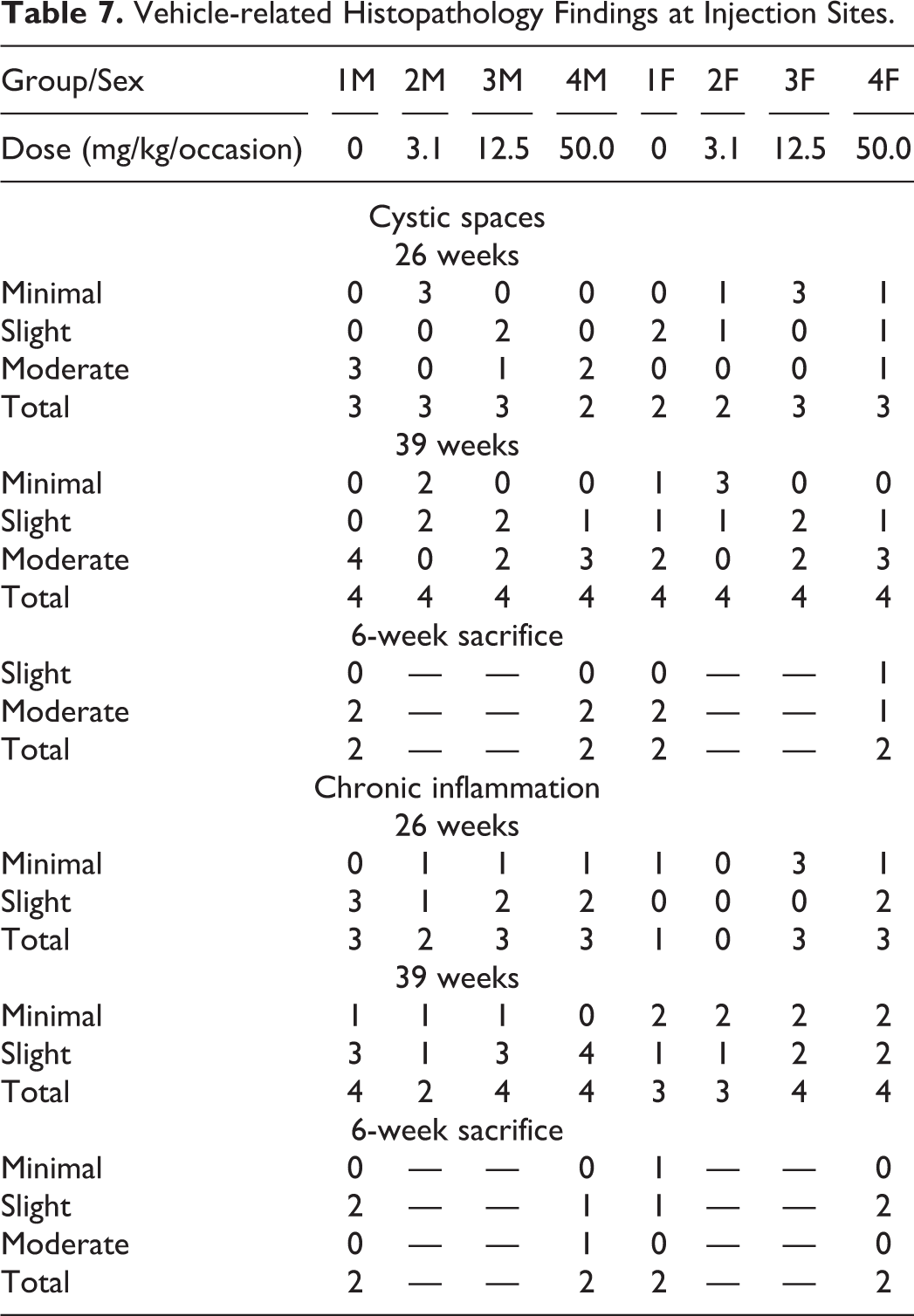
Findings similar to those at the subcutaneous injection sites were seen in the routine skin section of one control male and one male receiving 50.0 mg/kg/occasion at the 26- and 39-week sacrifice point. Such findings were not observed at the 6-week recovery group. Lymph nodes: Cystic spaces and chronic inflammation were seen in the left inguinal lymph node of all treated groups and controls at 26 week, 39 week, and recovery phase with the severity generally correlating with the volume of vehicle administered (Table 8). Chronic inflammation was characterized by mononuclear cells (lymphocytes and macrophages), foreign body giant cells, fibrosis, and occasional neutrophils. Abscesses, graded as minimal, were seen in the left inguinal lymph node of a few animals killed at 26 weeks in all treated groups but also in a female control killed after 39 weeks of treatment and in the recovery group. They were also seen in other lymph nodes in all three phases of this study, which were examined histopathologically due to being macroscopically abnormal. These consisted of aggregates of neutrophils contained within a small circular space surrounded by a minimal fibrous capsule. Representative photos of the abscesses in the inguinal lymph nodes from the main phase are shown in Figure 1F–G. Considering the overall incidence of abscesses in the left inguinal lymph node and other lymph nodes in the study, this finding was considered to be related to the vehicle. The incidence and severity of the findings at the 6-week sacrifice point were similar to the 39-week sacrifice point, therefore not showing signs of recovery.
Findings Related to the Vehicle in the Left Inguinal Lymph Node.
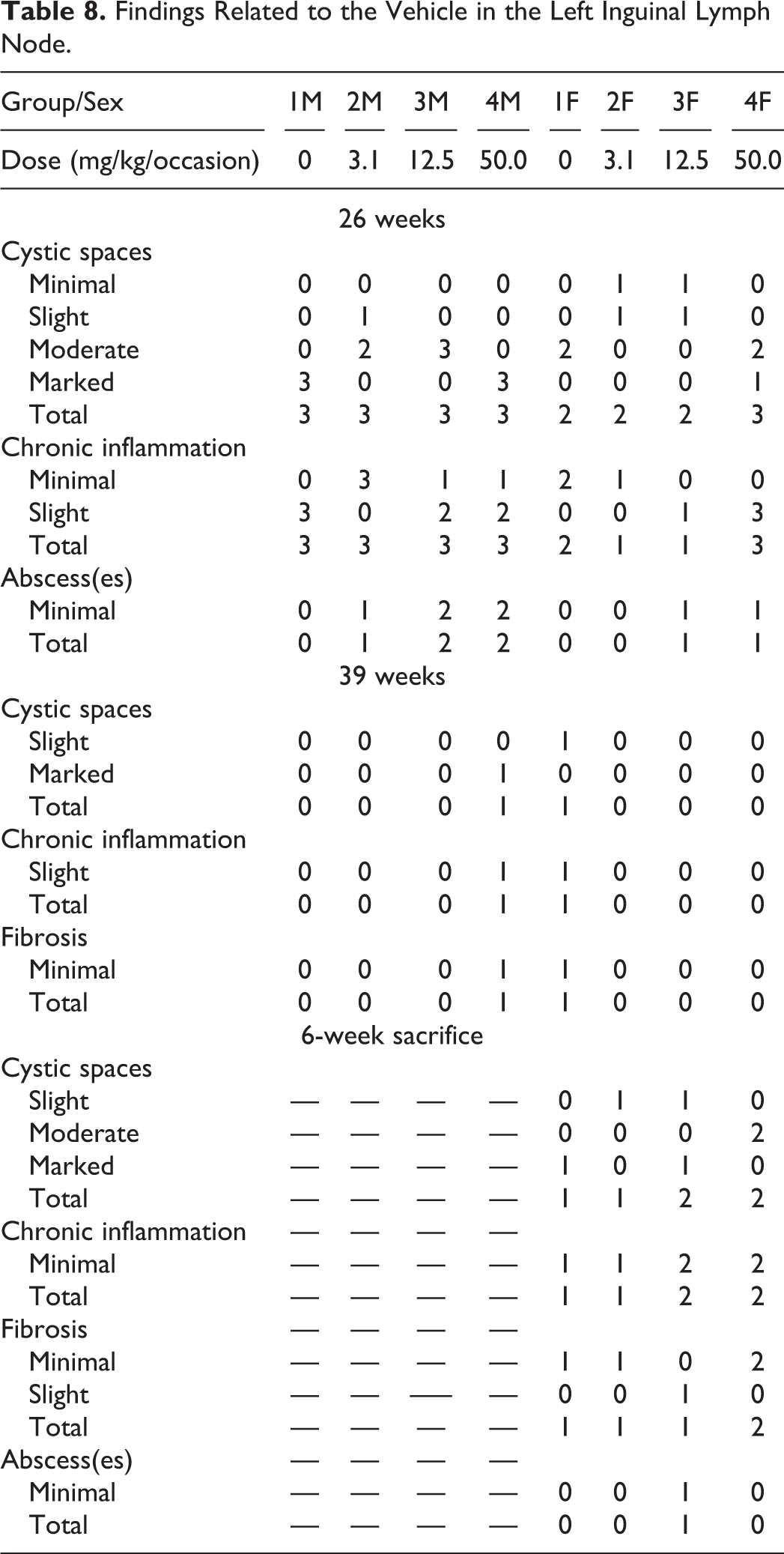
Similar findings were also seen in the right inguinal lymph node and in the axillary, mediastinal, medial iliac, aortic, and superficial inguinal lymph nodes when a macroscopic abnormality was seen. Such abnormalities were present most commonly in controls and animals receiving 50.0 mg/kg/occasion.
Discussion
The aim of this study was to assess the systemic toxic potential of RPh201, administered twice weekly by subcutaneous injections to minipigs, after 39 weeks of administration followed by a recovery period of 6 weeks. This study shows that RPh201 was well tolerated for 39 weeks with no clinical or dose-related signs observed and with no changes in body weight, food consumption, ERG, intraocular pressure, electrocardiography or blood chemistry, and urinalysis parameters.
Treatment-related findings were seen at the injection sites. Abscesses, which were scored as minimal or slight, and subcutaneous fibrosis that was associated with pale areas were observed at the injection sites at the 12.5 or 50 mg/kg/occasion dose levels. These findings correlated with a small increase in peripheral neutrophil count. At the 12.5 mg/kg/occasion dose level, these abscesses were scored as minimal to slight and therefore were considered to be nonadverse and at this dose level or below there were no findings considered to be of toxicological importance. However, in animals given 50 mg/kg/occasion, abscesses graded above slight corresponded with macroscopically thickened areas (with cystic pockets of dark fluid) or raised areas (which contained either dark fluid or green purulent material). The subcutaneous fibrosis seen at the injection sites was considered secondary to chronic inflammation and was again more pronounced in animals given 50 mg/kg/occasion and to a lesser extent in animals given 12.5 mg/kg/occasion. There was partial recovery from both the abscess formation and fibrosis.
Necrosis was not seen in any of the injection sites, and bacterial colonies were not seen in the abscesses (i.e., confirmed negative by Gram staining). Therefore, these abscesses were concluded to be sterile. Formation of sterile abscesses in the skin and muscle was observed previously in humans, following repeated injections of oil-based formulations (Cordes and Arnold 1949; Jones 1996; Starmark, Forsman, and Wahlstrom 1980). Chronic inflammation can result due to irritation from the presence of the drug in a location with a relatively slow rate of absorption. The acute inflammation seen in a few injection sites may represent a milder reaction or an early stage of the process, which might progress to abscess formation. Even though necrosis was not seen at any injection site and no treated animals showed any clinical signs relating to the injection sites or any effect on food consumption or bodyweight, the abscesses seen at the injection sites of some animals receiving 50 mg/kg/occasion were considered adverse due to their relatively large size, that is the size of the abscess was the “critical effect size,” which defines the breaking point between adverse and nonadverse changes (Kerlin et al. 2016).
Cystic spaces were recorded in the subcutaneous injection sites, and these correlated with an abnormal oily clear liquid material recorded macroscopically, which is considered to be due to the presence of the vehicle (BHT-stabilized cottonseed oil). Thickened areas recorded macroscopically in the subcutaneous injection sites generally correlated with chronic inflammation in the subcutis seen microscopically. Findings related to treatment with the vehicle were seen in the lungs, kidneys, lymph nodes (inguinal, axillary, mediastinal, medial, iliac, aortic, and mandibular lymph nodes), injection sites, and routine skin and subcutis sections. Generally, the incidence and severity reflected the volume of material injected, with the controls showing a similar incidence of findings to the animals receiving 50 mg/kg/occasion. The chronic inflammation in the subcutis was considered to be related to the cystic spaces caused by the vehicle. The findings in the routine sections of the skin and subcutis were considered to be due to ventral movement of injected material from the dorsally located injection sites. After a 6-week recovery period, the lungs and kidney showed partial recovery, while the findings in the subcutaneous injection sites and lymph nodes were similar to that seen at the end of the treatment period. These findings were considered as not adverse since they were of minimal-to-slight severity.
The trafficking of foreign material, such as oil-based formulations, via lymphatic vessels has been known for many years (Clark and Clark 1917). Such trafficking can be mediated by antigen presenting cells or as passive absorption (Ramot et al. 2009). Alternatively, movement of oil-based compounds can occur via the circulatory system (Peyre et al. 2004). This can result in the formation of lipogranuloma at distant organs such as the peritoneum (Ramot et al. 2009).
Vegetable oils, and among them cottonseed oil, are capable of solubilizing highly lipophilic materials and are in commercial use for many parenteral formulations (Strickley 2004). Nevertheless, information on the potential adverse effects related to the use of these vehicles, and specifically during preclinical studies, is limited. Therefore, the information on the lack of adverse effects associated with the use of the cottonseed oil vehicle will be of importance for future drug safety assessments utilizing vegetable oils as a vehicle.
Lens opacities, including suture line abnormalities, are described as a normal background lesion in the Göttingen minipig (Loget 1995; Loget and Saint-Macary 2000). According to Loget and Saint-Macary (2000), the background incidence of lens abnormalities in the Göttingen minipig is reported to be 2.8% for suture line abnormality, 8.3% for focal nuclear opacity, 19.4% for posterior cortical pinpoint opacity, and 8.3% for posterior capsular opacity (Loget and Saint-Macary 2000).
Lens focal nuclear opacities were reported in other animal species (e.g., dogs, rats, and Yucatan minipigs) and are nonprogressive and do not interfere with vision. It is probable that this abnormality appears later in life and should not be considered adverse (Loget 1995).
The nature and the appearance of the findings, the age of occurrence, and the pattern of progression are in accordance with the background incidences of posterior suture line opacities, which are seen in this species, strain, and age. Furthermore, no effects were observed following the ERG or tonicity assessments of the eyes or clinical signs that would suggest impaired eye function. It is therefore highly unlikely to be associated with the administration of the test compound, especially if similar findings were not seen in any other species that received the compound. In conclusion, under the conditions of this study, the no-observed-adverse-effect level was considered to be 12.5 mg/kg/occasion, after 39 weeks of administration followed by a 6-week recovery period.
Footnotes
Authors’ Note
Drs. Zadik Hazan, Andre Lucassen, and Konstantin Adamsky are employees of Regenera Pharma; Abraham Nyska is a consultant to Regenera Pharma.
Authors Contribution
Authors contributed to conception or design (ZH, AL, KA, VR, NY, MS, HE, AN); data acquisition, analysis, or interpretation (YR, ZH, AL, KA, VR, NY, MS, HE, AN); drafting the manuscript (YR, ZH, AL, KA, VR, NY, MS, HE, AN); and critically revising the manuscript (YR, ZH, AL, KA, VR, NY, MS, HE, AN). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Regenera Pharma, Nes-Ziona, Israel.
