Abstract
Arteritis/polyarteritis occurs spontaneously in many species used in preclinical toxicology studies. In Göttingen minipigs, arteritis/polyarteritis is an occasionally observed background change. In the minipig, this finding differs in frequency and nature from age-related polyarteritis nodosa in rats or monkeys, and Beagle pain syndrome in dogs. In minipigs, it can be present in a single small- or medium-sized artery of an organ or a few organs and is most commonly recorded in the cardiac and extracardiac blood vessels, vagina, oviduct, rectum, epididymis, spinal cord, pancreas, urinary bladder, kidneys, and stomach. The etiology is unknown although it has been considered in minipigs as well as in rats, dogs, and monkeys to be possibly immune mediated. This background change is important with respect to its nature and distribution in the minipig in order to distinguish it from drug-induced vascular changes, which might occur in similar locations and have similar morphologic features. This review summarizes the morphology, incidence, and predilection sites of arteritis as a spontaneously occurring background change and as a drug-induced vasculopathy in the minipig, and also describes the main aspects to consider when evaluating vascular changes in Göttingen minipig toxicity studies and their human relevance.
Arteritis or polyarteritis is defined as inflammation of the blood vessels that carry oxygenated blood away from the heart. It is characterized by the presence of mixed inflammatory cells within and around the blood vessel wall with concomitant vessel wall damage as indicated by fibrin deposition, collagen degeneration, and necrosis of endothelial and smooth muscle cells, with or without lumen occlusion (Robinson and Robinson 2016). The inflammation can extend into the periphery of the vessel and include the connective tissue surrounding the vessel (periarteritis). Clinical manifestations depend on the type of vessel involved, the localization, and the nature of the inflammatory process. Vascular inflammation can be the manifestation of a primary autoimmune disorder or secondary to other conditions such as infectious diseases, connective tissue disorders, or hypersensitivity disorders (Lintermans et al. 2014). The pathogenesis of primary systemic vasculitides is not completely understood and may be the result of an interplay between immunological, hormonal, and environmental factors in genetically predisposed individuals (Avcin et al. 2008; Lintermans et al. 2014). Viral, bacterial, or parasitic infectious agents may be involved in triggering an aberrant immune response. The pathophysiology of secondary vasculitides involves microbial antigens in conjunction with infectious diseases, autoantigens in connective tissue diseases and nonmicrobial antigens in hypersensitivity disorders (Avcin et al. 2008; Lintermans et al. 2014).
In preclinical safety studies in rodent and nonrodent species, arteritis or polyarteritis can occur spontaneously or can be induced by various classes of compounds. For standard laboratory species such as rat, dog, and monkey, spontaneous and drug-induced arteritis or polyarteritis are well described and are discussed in the literature and referenced in this review. However, arteritis or polyarteritis in the minipig, increasingly used as a nonrodent laboratory species, has not been reviewed in the literature to the same extent as for rat, dog, or monkey. Arteritis/polyarteritis in minipigs is an occasionally observed background change in all types of toxicology studies (Dincer and Skydsgaard 2012; Jeppesen and Skydsgaard 2015). This spontaneous condition in the minipig differs from rats, dogs, and monkeys in its frequency and features. It is therefore important to be aware of this background change in the minipig with respect to its nature and distribution in order to distinguish it from test article–related changes. This review summarizes the morphology, incidence, and predilection sites of arteritis as (A) a spontaneously occurring background change in Göttingen minipigs (a commonly used breed in toxicity testing) and (B) drug-induced vasculopathy focusing on compounds that have been studied in the minipig. This review also discusses how drug-induced vascular injury (DIVI) can be differentiated from spontaneous arteritis in Göttingen minipigs (C) and how its spontaneous or drug-induced occurrence compares with other species (D), as well as discussing its human relevance (E).
(A) Spontaneously Occurring Arteritis in Minipig
Arteritis/polyarteritis is an occasionally observed spontaneously occurring background change in Göttingen minipigs used in preclinical safety studies. It can be present in a small- or medium-sized single artery of a single organ or several organs in an animal or in several animals in a study. The severity is generally minimal or mild but can occasionally be observed at moderate levels with no age and sex predilection. The prevalence of the finding ranges from 0.06 to 0.29%; it is most commonly recorded with descending frequency in the cardiac and extracardiac blood vessels, vagina, oviduct, rectum, epididymis, spinal cord, pancreas, urinary bladder, kidneys, and stomach; less frequently in the larynx; and rarely in esophagus, sciatic peripheral nerve, eyeball, tongue, thyroid, lung, duodenum, thymus, spleen, trachea, adrenal, and liver (data derived from Helke et al. 2016; Table 1). In this table, the number of organs examined can vary between toxicology studies, especially in the reproductive system where there can be a small number of organs that can influence the incidence of the finding. Also note that Table 1 reports “inflammation, vessel” or “inflammation, vascular” in different organs. These are commonly used in International Harmonization of Nomenclature and Diagnostic (INHAND) criteria nomenclature as a synonym of arteritis or polyarteritis in laboratory species (Berridge et al. 2016). Other synonyms of arteritis used in the INHAND cardiovascular publication for the rat and mouse are degeneration/necrosis, necrosis/inflammation, and inflammation (Berridge et al. 2016). These are associated with location modifiers such as medial or mural (affected blood vessel distribution) and artery (affected blood vessel type; Berridge et al. 2016).
The etiology of spontaneous arteritis in minipigs is unknown. It is considered possibly to be immune mediated in common with polyarteritis in Beagle dogs (also known as Beagle pain syndrome) and polyarteritis nodosa in rats or monkeys (Percy and Barthold 2007; Porter et al. 2003; Son 2004). Since Göttingen minipigs are bred under “specific pathogen-free” (SPF) conditions and kept in laboratories under well-controlled hygienic conditions, vascular lesions secondary to infectious porcine diseases in toxicology studies have not been reported.
The Incidence of Spontaneously Occurring Arteritis Observed in Short and Long-term Preclinical Safety Studies in Göttingen Minipigs (North America and European Union).
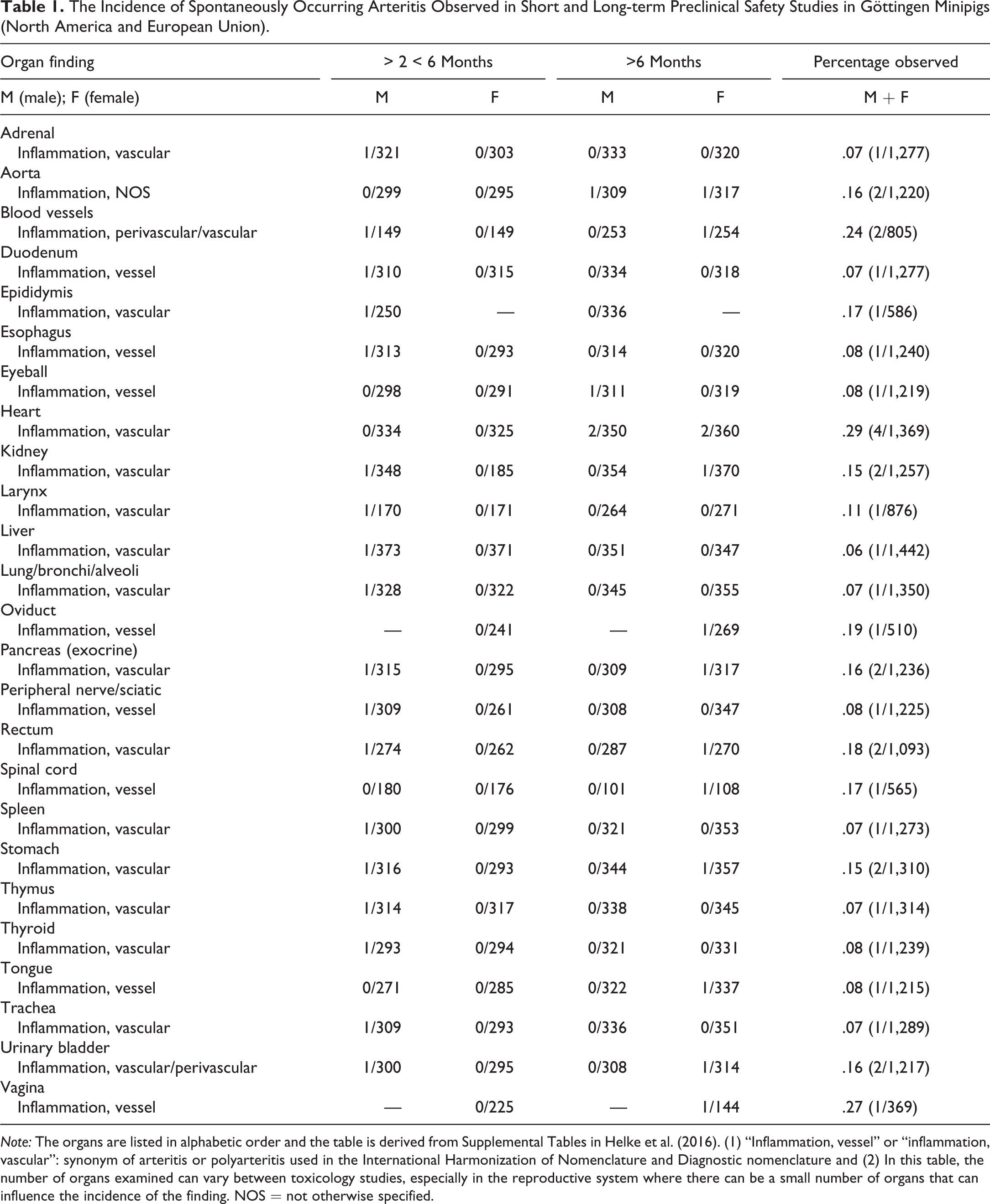
Note: The organs are listed in alphabetic order and the table is derived from Supplemental Tables in Helke et al. (2016). (1) “Inflammation, vessel” or “inflammation, vascular”: synonym of arteritis or polyarteritis used in the International Harmonization of Nomenclature and Diagnostic nomenclature and (2) In this table, the number of organs examined can vary between toxicology studies, especially in the reproductive system where there can be a small number of organs that can influence the incidence of the finding. NOS = not otherwise specified.
Currently, the diagnostic term “arteritis” or “arteritis/periarteritis” is used for describing this pathology in toxicology studies in Göttingen minipigs. It is generally seen in 2 forms including the fibrinoid necrotic form that is characterized by inflammatory cell infiltrates in all layers of the artery associated with necrosis and fibrin deposits in the vascular wall (Figure 1) and the chronic form that is characterized by thickening of the arterial wall due to fibrosis with a few mixed inflammatory cells generally extending to the periphery of the artery (periarteritis; Figure 2).
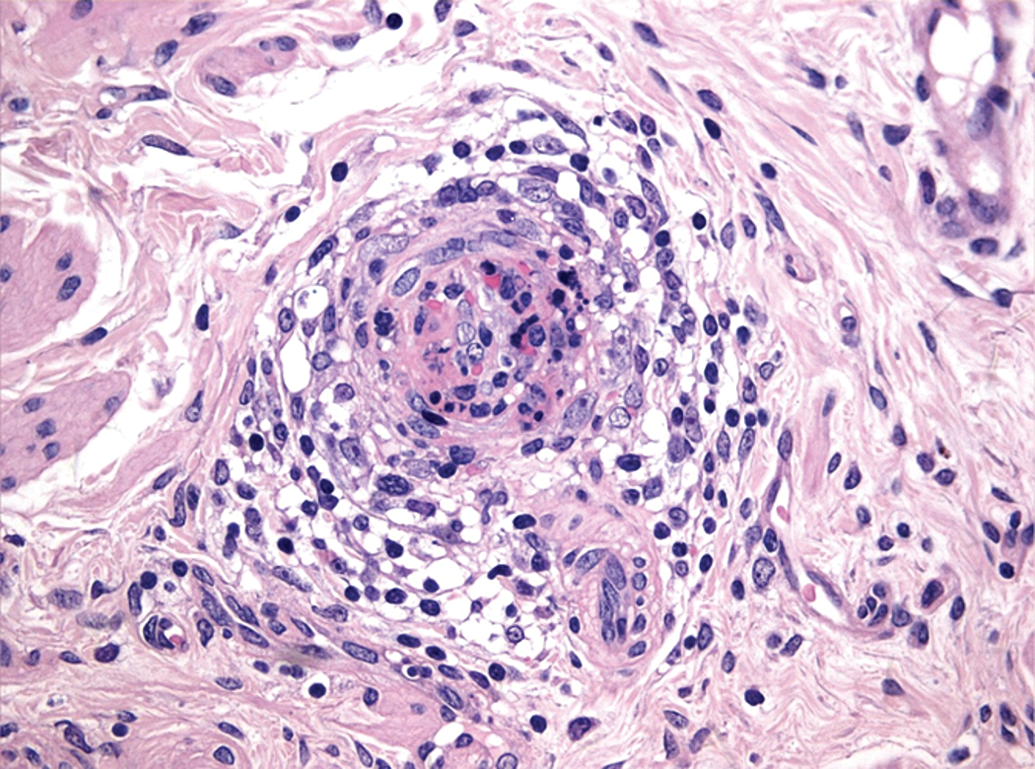
Spontaneous fibrinoid necrotic arteritis/periarteritis in the mesentery of a Göttingen minipig characterized by mixed inflammatory cell infiltrates in all layers and the periphery of the artery with necrosis and fibrin deposits in the vascular wall. Hematoxylin and eosin.
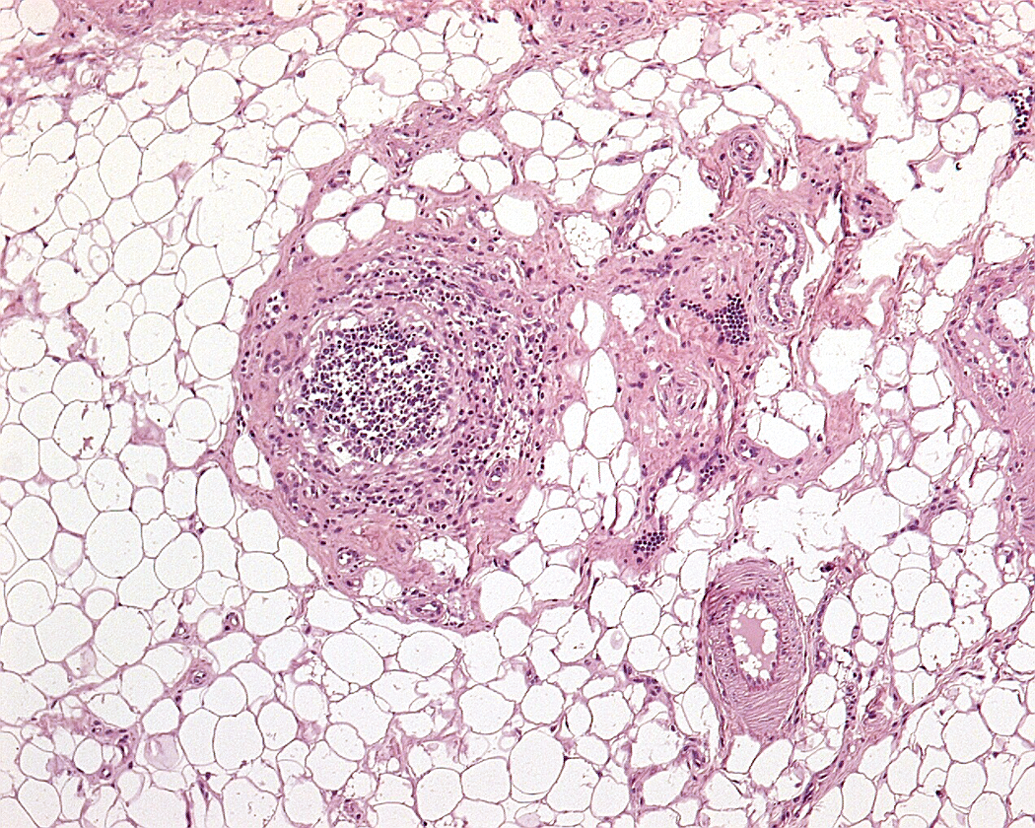
Spontaneous chronic arteritis/periarteritis in the intestinal serosa of a Göttingen minipig characterized by fibrotic thickening of the arterial wall with mixed inflammatory cells extending to the periphery of the artery. Hematoxylin and eosin.
Arteritis/polyarteritis has been described occasionally in conjunction with thrombocytopenic purpura syndrome (also known as hemorrhagic syndrome), an important spontaneously occurring disease in Göttingen minipigs (Carrasco et al. 2003; Maratea et al. 2006). The etiology of thrombocytopenic purpura syndrome is unknown but may be related to a type III hypersensitivity reaction based on the combined presence of thrombocytopenia and membranoproliferative lesions in the renal glomeruli (Carrasco et al. 2003; Dincer and Skydsgaard 2012). Carrasco et al. (2003) have noted necrotizing arteritis in 2 of the 11 minipigs with thrombocytopenic purpura syndrome in various tissues including kidney, stomach, skeletal muscle, and dermis. Maratea et al. (2006) described a spectrum of arterial lesions in 9 SPF Göttingen minipigs including morphologic features of arteriosclerosis (intimal proliferation, medial thickening, luminal stenosis, thrombosis, medial necrosis, and deposits of myxoid matrix material) affecting small- to medium-sized muscular arteries and arterioles in various organs and extraparenchymal tissues (vessels of renal pelvis and coronary arteries) in all animals and arteritis/periarteritis in 4 of the 9 minipigs. The etiopathogenesis of these lesions was not identified.
Arterial Changes Secondary to Catheterization and Intravenous Administration
Arteritis may also occur secondary to mechanical damage like catheterization. In humans, arteritis is described as a possible side effect during selective arterial catheterization to treat patients with primary and metastatic tumors of the liver (Belli et al. 1997). Such catheterization can lead to tortuosity of the arteries, stenosis, and occlusion of vessels (Belli et al. 1997). Similar arteritis can also be observed in Göttingen minipig after hepatic arterial catheterization for the administration of drugs. Alterations are seen at the administration site and also in the vicinity of occasional small- or medium-sized arteries (not previously reported).
Arteritis/periarteritis with thrombi is observed sporadically in the lungs of Göttingen minipigs after intravenous dosing. This may lead to thromboembolic pneumonia in some cases (Jeppesen and Skydsgaard 2015).
(B) Drug-induced Arteritis in the Minipig
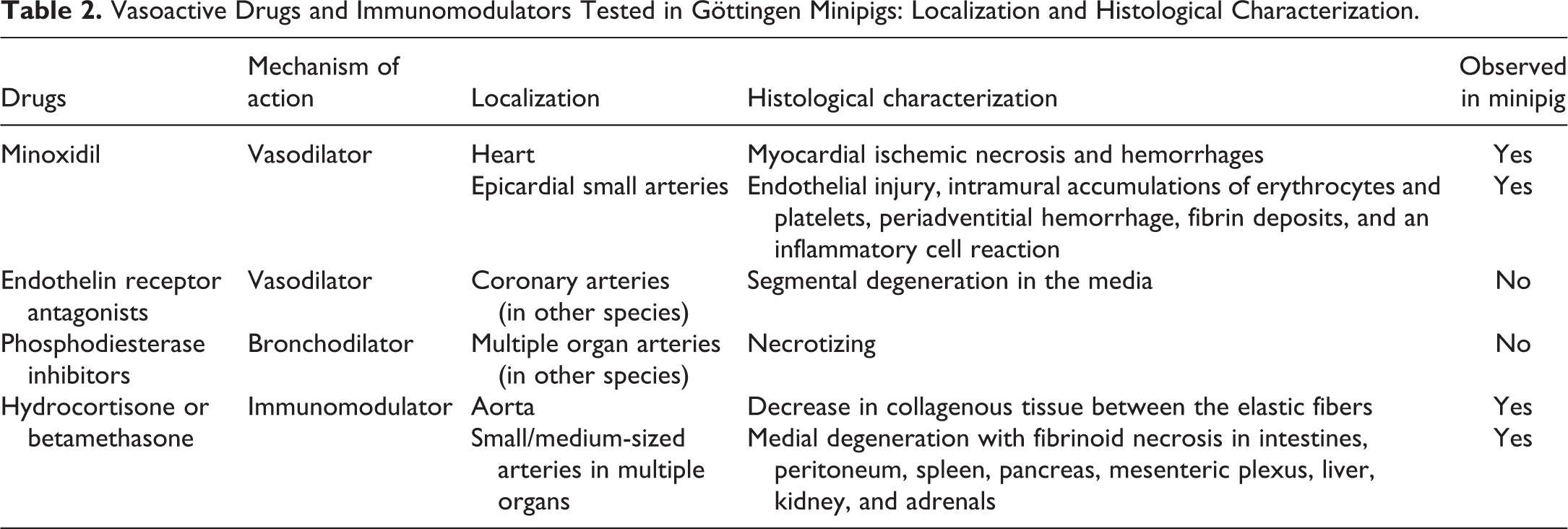
DIVI has not been as extensively studied in Göttingen minipigs as in dogs, monkeys, or rats. Despite the limited usage of minipigs as a nonrodent laboratory species, information is available on vasoactive drugs that affect vasculature directly, such as potassium channel openers and endothelin receptor antagonists, and other drugs affecting vasculature indirectly such as immunomodulators (e.g., hydrocortisone, betamethasone). Available literature is referenced below, and information is also summarized in Table 2.
Vasoactive Drugs and Immunomodulators Tested in Göttingen Minipigs: Localization and Histological Characterization.
Arterial Changes with Vasoactive Drugs
A potassium channel opener (minoxidil) is one of the vasoactive drugs studied in Göttingen minipigs. Minoxidil, a long-acting vasodilating antihypertensive agent, reduces arterial blood pressure by direct action on arteriolar smooth muscle (Herman et al. 1989). It causes myocardial ischemic necrosis and hemorrhage in minipigs. An ischemic origin for the necrosis has been suggested by the location of the lesions and by the pharmacologic effects of Minoxidil (Herman et al. 1989). In minipigs, the necrosis is most frequent in the left ventricular papillary muscles. The hemorrhagic lesions may be caused by overstretching of the vascular walls due to excessive, prolonged vasodilation. Gross epicardial or endocardial hemorrhages are seen in the atria and ventricles. The atrial lesions grossly show diffuse redness and microscopically comprise interstitial edema, extravasated erythrocytes, and infiltration of mononuclear inflammatory cells. Hemorrhages, occurring on the epicardial surfaces, are often associated with lesions that involve small arteries with 3 to 6 layers of medial smooth muscle cells and are characterized by endothelial injury, intramural accumulations of erythrocytes and platelets, periadventitial hemorrhage, fibrin deposits, and an inflammatory cell infiltration. These lesions preferentially involve the left atrium in pigs (Herman et al. 1989).
Endothelin A receptor antagonists (vasodilators), which inhibit the vasoconstrictor effect at the endothelin A receptor and have been investigated for treatment of various cardiovascular diseases, have not caused pathology in Göttingen minipigs, whereas cardiac arteriopathy has been seen in dogs and monkeys (Schneider et al. 2007).
Phosphodiesterase inhibitors (e.g., phosphodiesterase type III or IV inhibitor, bronchodilators) can result in necrotizing panarteritis in various organs but in Göttingen minipigs, vascular effects with phosphodiesterase inhibitors have not been reported in the literature.
Arterial Changes with Immunomodulatory Drugs
Immunomodulatory drugs such as glucocorticoids and cyclophosphamide are used to treat autoimmune diseases and associated vascular inflammation; however, they may be detrimental especially at high doses and prolonged application (Teng and Chatham 2015). They have been shown to induce vascular lesions in many species including humans. The nature and distribution of the vascular lesions induced by these drugs are usually different from those produced by vasoactive drugs. They are frequently associated with significant adverse effects in a number of organs including myocardium as well as suppression of bone marrow and immune function (Greaves 2012). Vascular inflammation due to infection as a consequence of immunosuppression is well recognized, either by direct infection of the vascular wall or via immunological effects such as type II, III, or IV hypersensitivity reactions, triggered by viral, bacterial, or fungal agents (Teng and Chatham 2015).
In view of the complexity of possible mechanisms involved with immunomodulation, the pathogenesis of arteritis in preclinical safety studies mostly remains uncertain. Considering the non-SPF status of species such as dog and monkey and the high degree of immunosuppression, often in conjunction with changes in the gut, occurrence of arteritis is likely to be a consequence of excessive systemic exposure to microbial antigens. In minipigs, there is only very limited experience regarding the safety evaluation of immunomodulatory drugs in comparison with other species. In Göttingen minipigs, 2-week oral studies with hydrocortisone and betamethasone showed arteritis (Figures 3–5) in various organs/tissues at daily doses of ≥30 mg/kg hydrocortisone and ≥30 mg/kg betamethasone, as well as additional changes in a number of other organs, which included the heart and the immune system. In the hydrocortisone study, affected arteries were aorta (decrease in collagenous tissue between the elastic fibers) and small- and medium-sized arteries of various organs (medial degeneration with fibrinoid necrosis in small and large intestines, peritoneum, spleen, pancreas, mesenteric plexus, liver, kidney, and adrenals). In the betamethasone study, similar degenerative arterial lesions were present as in the hydrocortisone study but with lower incidence and severity and mainly observed in the mesenteric plexus, renal pelvis, and spleen (unpublished data from Novartis archives).
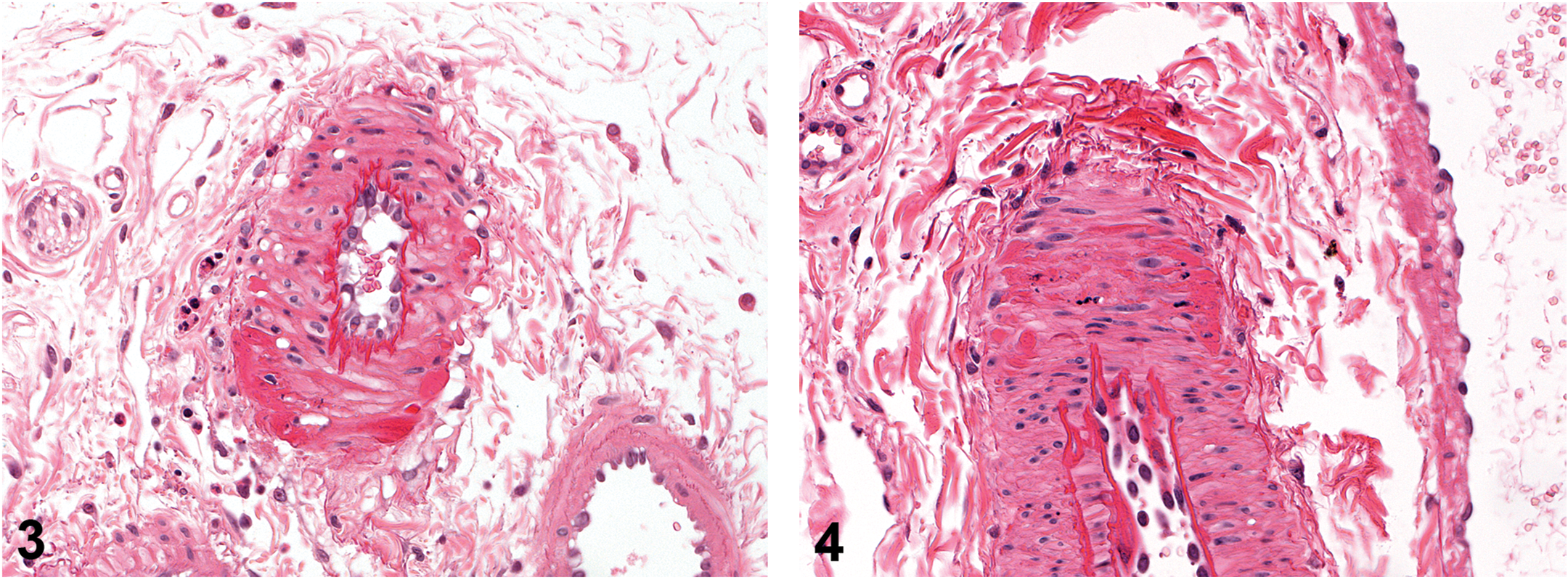
Drug-induced fibrinoid necrotic arteritis in a medium-sized mesenteric artery of a Göttingen minipig orally treated with hydrocortisone characterized by fibrinoid necrotic foci in the arterial wall with scattered mixed inflammatory cells in the adipose tissue of arterial periphery. Hematoxylin and eosin.
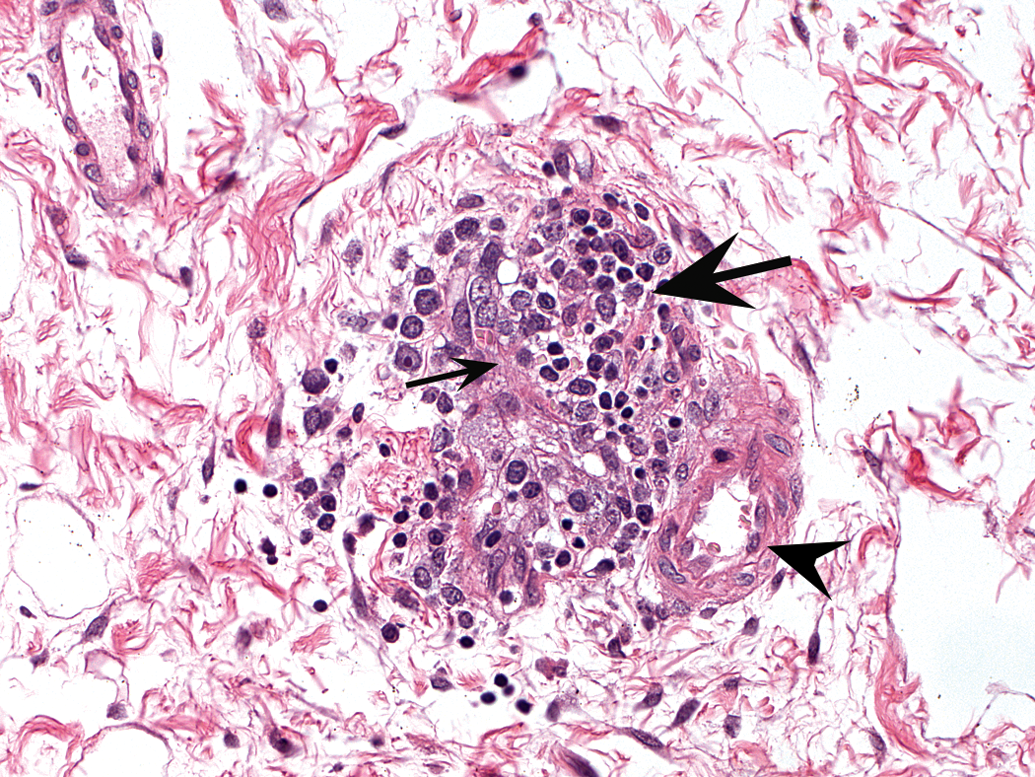
Drug-induced arteritis in a pelvic arteriole of the kidney of a Göttingen minipig orally treated with betamethasone characterized by prominent endothelial cells (arrowhead), thickened arterial wall with fibrin (thin arrow), and luminal and peripheral mixed inflammatory cells (thick arrow). Hematoxylin and eosin.
Arterial Changes with Radiation-based Treatment
Acute necrotizing vasculitis in swine can be seen after endovascular brachytherapy, a radiation-based treatment on reopened stenotic vessels, to prevent restenosis. This radiation-associated vasculitis resembles the localized lymphocytic vasculitis seen in human tissues exposed to external radiation. Vascular effects are seen in the target vessel and beyond its adventitia (Fajardo et al. 2002). Vascular damage induced by ionizing radiation is considered to be related to degenerative/apoptotic endothelial cells leading to increased vascular permeability and acute inflammatory response (Lombardini et al. 2013).
(C) Comparison between Spontaneous and Drug-induced Arteritis in Minipigs
The arterial changes in toxicity studies with minipigs should be investigated carefully regardless of whether it appears to be dose dependent or not. When vascular injury is observed in toxicity studies, the clinical signs, anatomic distribution, and microscopic appearance of the lesion can shed light on potential mechanisms. Detailed analysis of anatomical distribution and morphologic description of the arteritis are important to distinguish different mechanisms of drug-induced vascular changes from each other as well as from spontaneously occurring conditions. However, histopathology alone is unlikely to be definitive in differentiating drug-induced changes from changes of spontaneous origin. Other aspects such as general condition of the animal, other findings, reversibility, presence of the lesion in another species, dose–response, drug pharmacokinetics, pharmacodynamics, pharmacological activity, and therapeutic indication need to be considered when evaluating vascular injury in minipigs, as in other species.
Spontaneous arteritis occurs occasionally in Göttingen minipigs and the distribution of the change may easily mimic a drug-induced change. The historical background incidence is critical for the accurate interpretation of such lesions when multiple-treated animals are affected, and/or the lesions have an unusual distribution.
In exploratory studies with limited tissue evaluations, organs such as cardiac and extracardiac blood vessels, vagina, oviduct, rectum, epididymis, spinal cord, pancreas, urinary bladder, kidneys, and stomach, where arteritis is most commonly recorded, are recommended to be examined, as they are most likely to demonstrate drug-induced vascular lesions in Göttingen minipigs (Helke et al. 2016).
Arteritis can also be observed in some infectious and metabolic diseases (Grand 2012; Van Vleet and Ferrans 1986). The SPF status of Göttingen minipigs used in preclinical safety studies should grant protection from infectious agents. In the presence of infectious diseases after a possible loss of the SPF status (e.g., during transportation or after changes in husbandry), identification of other organ changes apart from vascular lesions, as well as identifying the causative agents, can help to differentiate spontaneously occurring or drug-induced vascular lesions from those occurring as a consequence of infectious disease.
Translation of DIVI occurring in preclinical species, including minipigs, to humans is unclear. With few exceptions, for example, dopamine, small molecule drugs that result in preclinical DIVI have not been shown to cause vascular injury in humans and have long records of safe clinical administration (Frazier et al. 2015). In humans, DIVI represents about 3% of all vasculitides. The skin and kidney are the most frequently affected organs. Spontaneously occurring vascular conditions in man are not well understood but are believed to be immune mediated as in animals. The presence of preclinical DIVI should not necessarily halt development or progression of a potential drug. However, further preclinical investigative work is required as well as consultation with regulatory authorities to help select the most appropriate study designs and end points for these studies. Cautious clinical dose selection/escalation with appropriate safety margins is necessary once clinical progression is initiated (Frazier et al. 2015).
(D) Minipig versus Other Species
Spontaneous Arteritis
In rats, spontaneously occurring arteritis is age related and described as a chronic progressive disease, also known as “polyarteritis nodosa.” Currently, the draft INHAND manuscript on rodent cardiovascular system proposes the following terminology for vascular lesions: (i) degeneration/necrosis, medial, or mural, artery and (ii) inflammation, medial, or mural, artery (Berridge et al. 2016). The pathogenesis has not been resolved but is thought to be possibly immune mediated. The incidence is higher in males, and arterial lesions are most frequently seen in medium-sized arteries of the mesentery, pancreas, testis, kidneys, and most other organs except lungs. Microscopically, affected arteries show fibrinoid degeneration, thickening of the media, and infiltrating leucocytes consisting of mononuclear cells and a few neutrophils with or without thrombosis and recanalization (Percy and Barthold 2007). In rats, vasoactive compounds such as phosphodiesterase type 4 inhibitors may induce necrotizing panarteritis in the mesentery, microscopically comparable to spontaneous polyarteritis nodosa (Mecklenburg et al. 2006).
In dogs, spontaneous polyarteritis is well known as “Beagle pain syndrome” or idiopathic canine polyarteritis, juvenile polyarteritis syndrome, necrotizing vasculitis, arteritis, periarteritis, or spontaneous disseminated panarteritis. Its occurrence in Beagle dogs is relatively common, with an incidence of 7.5% in 2004 (Son 2004). The etiology is unknown; however, immune-mediated injury has been suspected but not reliably confirmed by immunohistochemical or immune function studies. Small- to medium-sized muscular arteries are affected in multiple organs. The arteries most consistently affected are found in the heart (predominantly extramural branches of the right coronary artery), the cranial mediastinum, and the cervical spinal meninges. The lesion can be acute, subacute, or chronic. Microscopically, acute arterial changes range from histiocytic–lymphocytic periarterial infiltration to transmural neutrophil inflammation with medial fibrinoid necrosis with or without thrombosis. Subacute and chronic lesions are characterized by intimal hyperplasia and medial hypertrophy with varying degrees of ruptured internal elastic laminae and perivascular mononuclear inflammation (Clemo et al. 2003; Son 2004). In dogs, the spontaneous condition and its differentiation from drug-induced arterial injury are discussed by Clemo et al. (2003). Although idiopathic canine polyarteritis can generally be differentiated from DIVI in dogs, Clemo et al. (2003) recommend a critical examination of vascular pathology together with good understanding of clinical, pharmacological, and mechanistic data associated with the drug, since there may be an increased incidence of idiopathic canine polyarteritis in animals receiving various compounds, suggestive of an exacerbation of the spontaneous condition.
In monkeys, arteritis or polyarteritis is rarely observed as a background change, as it is seen in rats or dogs. Two isolated cases of polyarteritis nodosa, similar to the commonly described condition in rats, have been seen in cynomolgus macaques (Chamanza 2012). In 1 case, immunohistochemical staining was performed and showed that most infiltrating cells were T lymphocytes and histiocytes, suggesting a cell-mediated component to the pathogenesis. In both cases, affected vessels were located in a variety of organs: the kidney, heart, gall bladder, stomach, pancreas, small and large intestine, mesentery, spleen, urinary bladder, pancreas, and uterus, with sparing of the pulmonary vessels. These cases were very similar to those described in humans (Porter et al. 2003). Spontaneous vascular lesions in monkeys are also observed secondary to primary diseases such as atherosclerosis, infection, endocarditis, or aberrant parasite migration or to neoplasms invading soft tissue in or around major vessels (April and Keith 1998).
Drug-induced Arteritis
Available information in the literature comparing drug-induced arteritis in the minipig, rat, dog, and monkey is detailed below and also summarized in Table 3.
Vasoactive Drugs and Immunomodulators with Vascular Injury in the Minipig Compared with Rat, Dog, and Monkey.
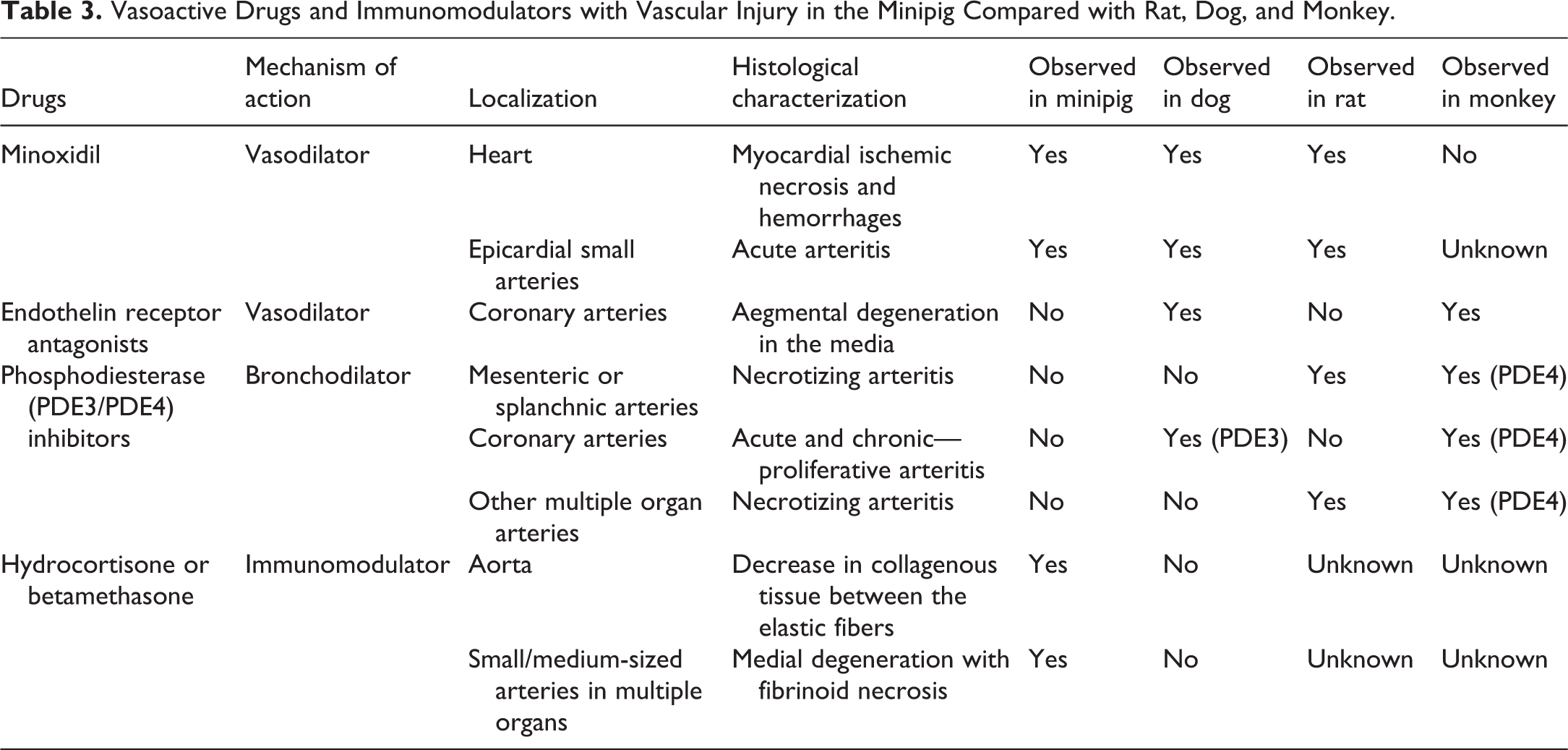
The potassium channel opener minoxidil causes arteritis, myocardial ischemic necrosis, and hemorrhages in minipigs as well as in dogs and rats. In minipigs, the necrosis in the left ventricular papillary muscles is less severe compared to dogs. Gross epicardial or endocardial hemorrhages involving the atria and ventricles are also more severe in dogs. Hemorrhages occurring on the epicardial surfaces involve the right atrium in dogs and the left atrium in pigs (Herman et al. 1989). In rats, cardiac lesions with minoxidil are similar to minipigs and dogs but are more frequent and severe in older rats than in younger rats (Herman et al. 1996). In monkeys, myocardial necrosis was not observed, but cardiac hypertrophy and dilation were recorded in oral studies according to the Food and Drug Administration (“Minoxidil” 2017).
Endothelin A receptor antagonists cause cardiac arteriopathy, characterized by segmental degeneration of the media of coronary arteries of dogs and monkeys, but not rats or Göttingen minipigs, even though plasma concentrations at steady state and overall exposure in terms of area under the curve(0–24h) were higher in minipigs and rats than in dogs (Stephan-Gueldner and Inomata 2000; Albassam et al. 1999). Therefore, the differences between the species could not be explained by differences in exposure in regard to developing arteritis with endothelin A receptor antagonists. In monkeys, coronary arteriopathy was seen following intravenous or oral administration of an endothelin antagonist (Albassam et al. 1999) or following oral administration of an adenosine agonist (agonist of a potent vasodilator; Albassam et al. 1998). In dogs, cardiac arteriopathy was seen after endothelin receptor antagonist administration by continuous intravenous infusion (Stephan-Gueldner and Inomata 2000).
Drug-induced arterial injury in rats is known for many vasoactive compounds, including phosphodiesterase type IV (PDE4) inhibitors, nonspecific phosphodiesterase inhibitors, selective phosphodiesterase type III (PDE3) inhibitors, or adenosine agonists. The pathogenic mechanism of the various phosphodiesterase inhibitors may be different from each other. Phosphodiesterase inhibitors result in necrotizing panarteritis in mesenteric vessels of rats. Apart from the mesentery, arteritis is also seen in vessels of pancreas, testis, spleen, and liver. This is a well-known species-specific side effect of phosphodiesterase inhibitors. It has been proven that PDE4 inhibition causes inflammation in the mesentery in the first instance, leading to vasculitis, and PDE3 inhibition results in inflammation in the vessels, followed by mesenteritis (Mecklenburg et al. 2006). In dogs, vascular changes are present only in coronary arteries and cardiac muscle with PDE3 inhibitors. The pattern of the changes is time dependent. Acute changes include segmental medial necrosis and hemorrhage in coronary arteries, which are followed by more chronic proliferative changes in the intima (smooth muscle hyperplasia with mucoid ground substance) and adventitia (fibroplasia). For many vasodilator-induced vascular injuries in dogs, arterial changes are often restricted to coronary arteries, and the nature of the changes in vessels yields important clues to their drug-induced nature such as the presence of medial and adventitial hemorrhage, which is absent in idiopathic polyarteritis (Losco et al. 2004; Clemo et al. 2003). Drug-induced arteriopathy is also reported in nonhuman primates following PDE4 inhibitor administration. It mainly affects the heart and splanchnic arteries (Losco et al. 2004). Vascular effects in the Gottingen minipig have not been observed after treatment with PDE inhibitors.
Following administration of immunomodulators (hydrocortisone or betamethasone) to the minipig, there is a thickening of the right ventricular wall and edema of the subepicardial tissues in the heart and fibrinoid necrotic foci in arterial walls with scattered mixed inflammatory cells in arteries in multiple organs. Immunomodulators also affect the immune system and cause other organ toxicities. Hydrocortisone and betamethasone were also given to rats at similar doses for similar durations. Although effects on the immune system were present in this species, no vascular alterations were observed (unpublished data from Novartis archives). To our knowledge, hydrocortisone or betamethasone effects on the cardiovascular system of monkeys and dogs have not been studied.
(E) Human Relevance
In minipigs, cardiovascular physiology and anatomy, ventricular performance, electrophysiology, and coronary artery distribution are similar to those in humans (McInnes 2012). As such, minipig models are relevant to study human diseases related to the vascular system. Until now, various minipig models have been successfully used to reproduce cardiovascular human diseases and have helped our understanding of the mechanisms and pathophysiology of the diseases. As examples, the coronary atherosclerosis model has been studied in the minipig (Swindle et al. 2012; Getz and Reardon 2012; Hamamdzic and Wilensky 2013), and in the cardiac allograft vasculopathy minipig model mimicking human heart transplantation, cyclosporine A was shown to induce proliferation and migration of smooth muscle cells from the media to the intima, leading to intimal thickening (Amano et al. 2015). In humans, arteritis or polyarteritis includes a range of different forms or entities, which has been classified with variable success according to vessel diameter, role of immune complexes, presence of specific autoantibodies, granuloma formation, organ specificity, and even population demographics (Mitchell 2015). Among them, polyarteritis nodosa is characterized by a segmental transmural necrotizing inflammation of small- or medium-sized muscular arteries, involving renal and visceral vessels but sparing the pulmonary circulation (Mitchell 2015). There are different forms of polyarteritis nodosa that may be etiologically and clinically distinct including classic idiopathic, cutaneous, and the form associated with chronic hepatitis (Mitchell 2015; Forbess and Bannykh 2015). To our knowledge, spontaneous cases of arteritis/polyarteritis in the minipig have not been considered as a model for the human disease(s). This could be explained by differences in the characteristics of minipig and human arteritis and by the limited knowledge of arteritis mechanisms in the minipig.
Differences between minipig and human arteritis have also been shown with vasoactive and immunomodulatory drugs. Following administration of minoxidil, patient autopsies have not revealed the characteristic hemorrhagic lesions seen in minipigs, dogs, and rats (“Minoxidil” 2017). Myocardial ischemic necrosis was occasionally seen but occurred in the presence of known preexisting coronary artery disease and was also seen in patients never exposed to minoxidil (“Minoxidil” 2017). Following administration of endothelin receptor antagonists, dogs are suspected to be more susceptible to the development of arteriopathy compared with monkeys or humans, while minipigs had no lesions. When administered bosentan, a nonselective endothelin A/B receptor antagonist, no adverse events related to drug-induced arteritis have been shown in patients although it induces canine coronary arteritis (Albassam et al. 1999; Jones et al. 2003). Following administration of phosphodiesterase inhibitors, especially PDE4 inhibitors, mesenteric arteritis has not been seen in man, unlike in rats. However, because of a report of arteriopathy in nonhuman primates (Losco et al. 2004), arteries require careful monitoring in man. PDE3 inhibitors have been shown to cause arteritis in rats and dogs. With long-term usage, they have also been shown to increase mortality in patients through an undetermined mechanism (Movsesian and Alharethi 2002; Banner and Press 2009). There were no data available regarding the cardiovascular effects of hydrocortisone or betamethasone in human, but in general, glucocorticoid treatment in humans is known to be associated with an increased risk of cardiovascular disease, which can be explained by hypertension and metabolic effects of glucocorticoid treatment on other pathways, especially impaired glucose tolerance, diabetes, and dyslipidemia (Strohmayer and Krakoff 2011). Arteritis as a direct side effect of glucocorticoids could, however, not be found in the literature.
Summary
Arteritis/polyarteritis in toxicology studies with Göttingen minipigs can occur spontaneously or can be induced by xenobiotics or as a consequence of mechanical vascular damage in specific experimental settings. Since swine are the preferred animal model for cardiovascular research, it is important to be aware of vascular changes in this species with respect to their nature and distribution in order to distinguish spontaneous from experimentally induced changes.
Spontaneous arteritis is occasionally seen in Göttingen minipigs, affecting a single small- or medium-sized artery or arteries of one or more organs in any type and duration of preclinical safety study. It is usually observed as a fibrinoid necrotic lesion or as chronic arteritis/periarteritis, mostly commonly in the cardiac and extracardiac blood vessels, vagina, oviduct, rectum, epididymis, spinal cord, pancreas, urinary bladder, kidneys, and stomach. This spontaneous condition in Göttingen minipigs differs from Beagle pain syndrome and polyarteritis nodosa of rats in its nature, predilection sites, and prevalence.
Despite the somewhat limited usage of Göttingen minipigs as a nonrodent laboratory species, information is not available in the literature on drug-induced arteritis/polyarteritis in this species. In light of the available data, it appears that the minipig is not the most sensitive species for drug-induced arterial changes, particularly with vasoactive drugs. The dog is the most sensitive species to vascular injury of coronary arteries, and the rat is the most sensitive species to vascular injury of mesenteric arteries. In the minipig, there are no vascular changes with endothelin receptor antagonists and phosphodiesterase inhibitors. In contrast, these classes of compounds show changes in rats, dogs, or monkeys. Drugs like minoxidil and immunomodulators that affect the vasculature of the minipig induce similar morphological changes as observed in dogs and rats with regard to nature and predilection sites, which allow easy differentiation of these induced changes from spontaneous changes.
In conclusion, vascular injury in the minipig, as in other species, has to be interpreted in the context of all clinical, pharmacological, and mechanistic data associated with the drug in order to differentiate drug-induced from spontaneous lesions and to clarify equivocal and exacerbated background changes to reach firm conclusions. A detailed morphologic description can also help to distinguish different mechanisms of drug-induced vascular changes from each other as well as from spontaneous changes.
Footnotes
Author Contributions
All authors (ZD, VP, AM, UJW, SM) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
