Abstract
Regulatory guidances for nonclinical toxicity testing require brain evaluation but do not require a specific analytical strategy. The Society of Toxicologic Pathology (STP) has produced “best practice” recommendations for brain sampling and processing in general toxicity (GT) studies in adult rodents and nonrodents as well as developmental neurotoxicity (DNT) studies in rodents. This article explains acceptable brain trimming strategies as described in these 2 STP documents. Figures in the DNT and GT “best practices” illustrate coronal brain trimming at specific levels as defined by discrete external and internal anatomic landmarks. However, the text of both “best practice” papers states that institutions may choose different brain trimming levels or other planes (e.g., a longitudinal orientation) as long as key structures are sampled and trimming is consistent among individuals across the study. The STP-recommended number of brain levels to evaluate (7 or 8 coronal sections for GT and DNT studies, respectively) may need to be increased if neurotoxicity is considered possible or likely based on in-life clinical findings or other risk factors (chemical structure, known mode of action, etc.).
In the last 15 years, the Society of Toxicologic Pathology (STP) has produced 2 “best practices” documents that provide recommendations for nervous system sampling—including appropriate brain trimming levels—for use in regulatory-type nonclinical toxicity studies. One paper details techniques to be utilized for general toxicity (GT) studies (i.e., those in which no risk of neurotoxicity is anticipated) in adult rodents and nonrodents (Bolon et al. 2013), while the other describes methods to be employed for dedicated developmental neurotoxicity (DNT) studies in rodents (Bolon et al. 2006). In the author’s judgment, these 2 STP “best practice” papers have been viewed positively by both sponsors and regulators as a means of facilitating a more systematic histopathologic analysis of the brain because existing regulatory guidance documents require microscopic evaluation of multiple brain structures but do not dictate the trimming planes needed to reveal them (Organisation for Economic Co-operation and Development 1995a, 1995b, 1997, 2007, 2011; U.S. Environmental Protection Agency 1998a, 1998b, 1998c; U.S. Food and Drug Administration 2000). Nonetheless, the author’s recent interactions with client institutions, colleagues, and regulatory scientists have shown that the STP “best practice” recommendations have been misconstrued in some cases. The current article provides the author’s opinion regarding how the published STP “best practice” recommendations for DNT and GT studies should be interpreted with respect to implementing an appropriate brain trimming scheme for regulatory-type nonclinical studies.
The first consideration in formulating a brain trimming strategy is to define the brain regions that need to be assessed. Primary brain domains itemized for analysis in one or more regulatory guidelines (listed from rostral to caudal and dorsal to ventral) include olfactory bulbs, cerebrum (i.e., cerebral cortex [typically interpreted to imply those regions that support the associative, motor, sensory, and visual functions]), basal nuclei (i.e., caudate/putamen), hippocampus, thalamus, hypothalamus, midbrain, cerebellum, pons, and medulla oblongata (Bolon et al. 2011). Both the DNT and GT “best practice” documents were crafted by the STP so that their implementation will reliably sample these brain regions. Many other essential brain structures will be visible in brain sections prepared according to these 2 STP “best practices.” The major “ancillary” areas are enumerated in 1 STP “best practice” document (Bolon et al. 2013) and a related research paper (Rao, Little, and Sills 2014) and may be identified readily in brain anatomy atlases for common test species: mice (Franklin and Paxinos 2007; Paxinos et al. 2007), rats (Ashwell and Paxinos 2008; Paxinos, Ashwell, and Törk 1994; Paxinos and Watson 1997, 2004), dogs (Singer 1962; Palazzi 2011 [beagle]), pigs (Felix et al. 1999; Bjarkam et al. 2017), and nonhuman primates (Palazzi and Bordier 2008 [marmoset]; Paxinos et al. 2008 [rhesus macaque]; Szabo and Cowan 1984 [cynomolgus macaque]). Papers with annotated figures of brain sections that localize key structures to be considered for analysis in trimming planes similar to those shown in the STP “best practice” paper for nervous system sampling in GT studies have been published for adult rats (Rao, Little, and Sills 2014) and adult nonhuman primates (Pardo et al. 2012).
The second question that must be addressed in crafting an effective brain sampling strategy is selection of the trimming procedure. This point is multifaceted and necessitates independent decisions regarding (1) the angle and (2) the positions of the trimming cuts.
The preferred trimming angle in most nonclinical studies is the coronal orientation, which is the illustrated alignment in the STP “best practice” papers for both DNT and GT studies (Bolon et al. 2006, 2013). Rodent brains are small, and so generally they are viewed on standard-sized (75 × 25 mm) glass slides as full coronal sections showing both halves of the brain (Bolon et al. 2006, 2013). Nonrodent brains typically are trimmed to produce a coronal hemisection (i.e., one half of the brain) in either 1 or 2 parts (Garman 2003; Pardo et al. 2012; Bolon et al. 2013), although they also may be viewed as full coronal sections if oversized slides (e.g., 75 × 50 mm) are used. The rationale for preferring the coronal orientation for brain trimming is that this plane displays brain structures using the most common perspective illustrated in standard brain anatomy atlases for animals and humans. Such extensively annotated atlases are vital tools for pathologists seeking to learn regional macroscopic anatomy and to determine the identity of brain domains harboring neurotoxic lesions, which underscores the favored standing of the coronal orientation as an STP-recommended “best practice” for effective brain trimming.
For some purposes, however, the best angle for brain trimming actually may be in another orientation. For example, the longitudinal midsagittal plane is ideal for preparing sections of cerebellum from which morphometric measurements will be acquired in DNT studies (Garman et al. 2016). Similarly, a longitudinal parasagittal plane combined with several coronal hemisections may be a beneficial strategy for screening the brain in GT studies (Jordan et al. 2011), especially if the rostrocaudal continuity of white matter tracts is a key parameter of interest (Bolon et al. 2013). Such longitudinal orientations are documented as suitable alternatives in the STP “best practice” papers for GT and DNT studies even though sections taken at this angle are not included in the illustrations (Bolon et al. 2006, 2013).
The indispensable point with respect to selecting the angle and positions for brain trimming is reproducibility. The STP “best practice” documents specifically showcase coronal images representing 2 suitable trimming schemes (Bolon et al. 2006, 2013), but an attentive review of the text reveals in both documents that the figures are meant as examples and not to be construed as the only acceptable trimming planes. Practical experience reinforces this intent, as trimming planes in rodent brains (especially for juvenile and runted adult mice) typically cannot be positioned to reliably obtain the 3 recommended trimming planes in which cerebellum is visible (Bolon et al. 2013). Therefore, institutional brain trimming procedures should emphasize consistent procurement of highly homologous sections (i.e., where brain sections are essentially identical for all animals in a study; Garman et al. 2016) rather than trying to conform to the specific positioning of cuts as shown in the images in the 2 STP “best practice” papers for nervous system sampling.
The third matter to be resolved in devising an effective brain trimming strategy is to decide the scope of brain sampling that is necessary. The 2 STP “best practice” papers recommend analysis of 7 coronal sections for GT studies in adult rodents and nonrodents (Bolon et al. 2013) and 8 coronal sections for DNT studies for young rodents (Bolon et al. 2006). In the author’s view, evaluation of more than 7 to 8 sections will likely be required during dedicated neurotoxicity studies (i.e., where in-life clinical findings, chemical structure, known or suspected mode of action, or other factors suggest that brain toxicity is likely or certain). This increased (or “enhanced”) sampling scheme, which is performed to ensure that more brain regions are available for examination, is usually accomplished by increasing the number of trimming planes. Such enhanced sampling may be implemented in either an “automatic” or “targeted” fashion, with the choice of which strategy to use being left to the discretion of the institution.
The “automatic” approach to enhanced sampling is to increase the number of brain levels from which sections are prepared, without targeting any specific ancillary brain domains for histopathologic assessment. A simple means for accomplishing this feat in rodent brains that have been trimmed according to the STP recommended “best practices” for evaluation at 7 coronal planes (Bolon et al. 2013) is to procure a section soon after facing into the block and then a second section after stepping into the block for a particular distance (usually set between 0.5 to 2.0 mm for standard 4-mm-deep tissue cassettes). An alternative is to trim rodent brains to produce several more brain slabs than the usual 7 or 8 blocks; in the author’s experience, the integrity of the resulting thin brain blocks (approximately 2 to 2.5 mm) may be maintained more effectively by making trimming cuts using a brain matrix with prepositioned slots to guide cutting rather than by freehand positioning of the blade. In nonrodents, enhanced sampling requires that more brain blocks be taken than the 7 levels shown in the STP “best practices” for GT studies (Bolon et al. 2013). The easiest “automatic” option for completing this task is to double the number of trimming planes (e.g., as shown in Pardo et al. 2012; Figure 1) and then place 1 or 2 hemisection pieces from many or most planes into separate cassettes (Figure 2). The use of brain hemisections is feasible—as recognized in the STP “best practice” paper for GT studies—only if “bilateral or contralateral clinical signs are not observed ante mortem, bilateral gross lesions are not detected at necropsy, and a unilateral pattern of lesion distribution has not been demonstrated previously for the same or a structurally similar test article” (Bolon et al. 2013, p. 1035). If these preconditions are not met, bilateral sampling of the brain trimming planes is advisable in both rodents and nonrodents.
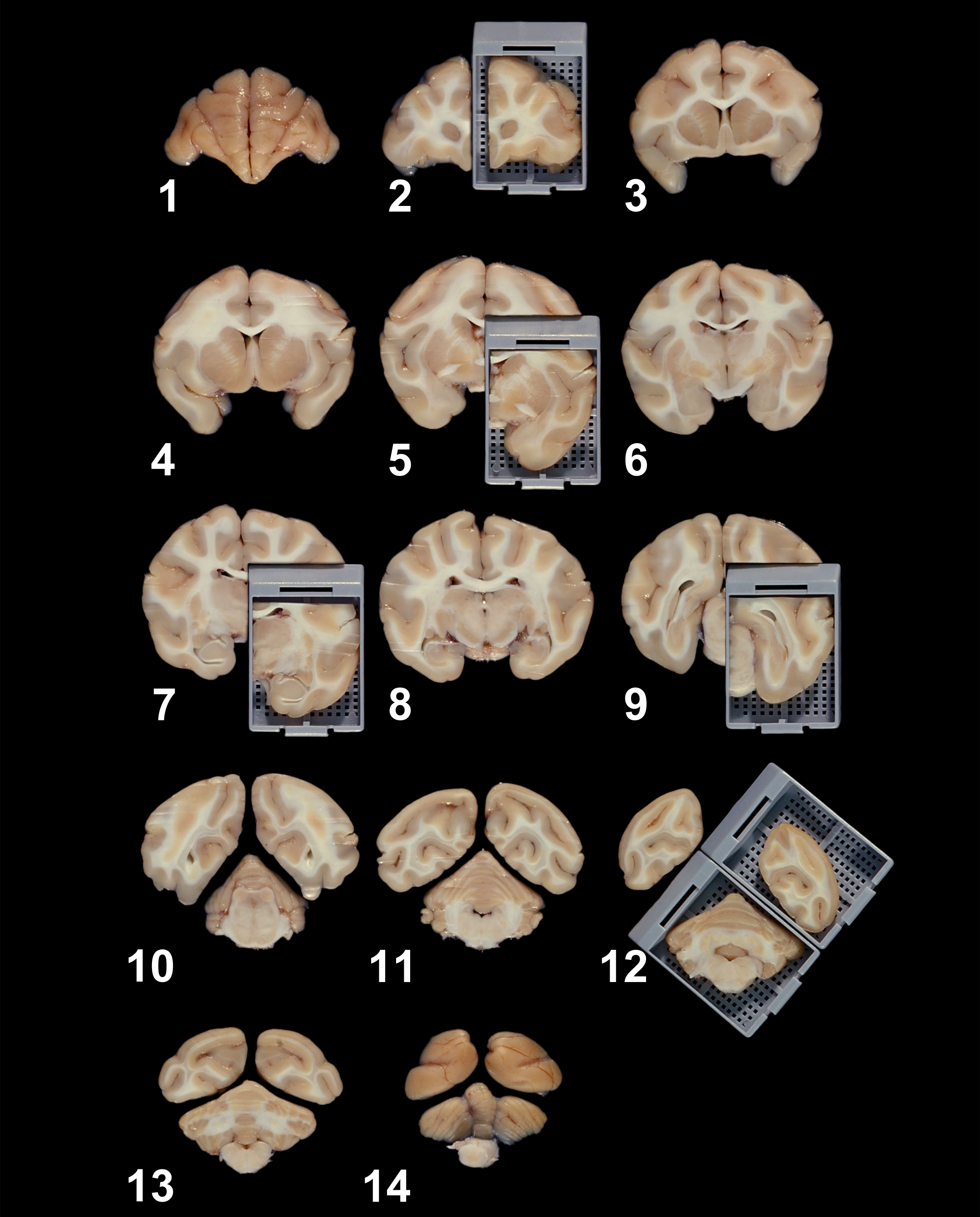
A suitable alternative baseline trimming scheme for nonrodent brains (shown here for a cynomolgus monkey [Macaca fascicularis]), demonstrating the rostral aspects of fourteen 4-mm-thick cross sections from which brain samples will be acquired. The tissue blocks in the gray cassettes of cross sections 2, 5, 7, 9, and 12 represent 6 of the 7 recommended sections for nonrodent brains illustrated in the Society of Toxicologic Pathology “best practice” document for nervous system sampling during general toxicity studies (Bolon et al. 2013). For embedding, the rostral (i.e., visible) surfaces of the pieces inside the cassettes will be placed “down” in the embedding mold so that it will be the first portion to be sectioned. Source: Image reproduced from Pardo et al. (2012) by permission of Dr. Ingrid D. Pardo and Sage Publications.
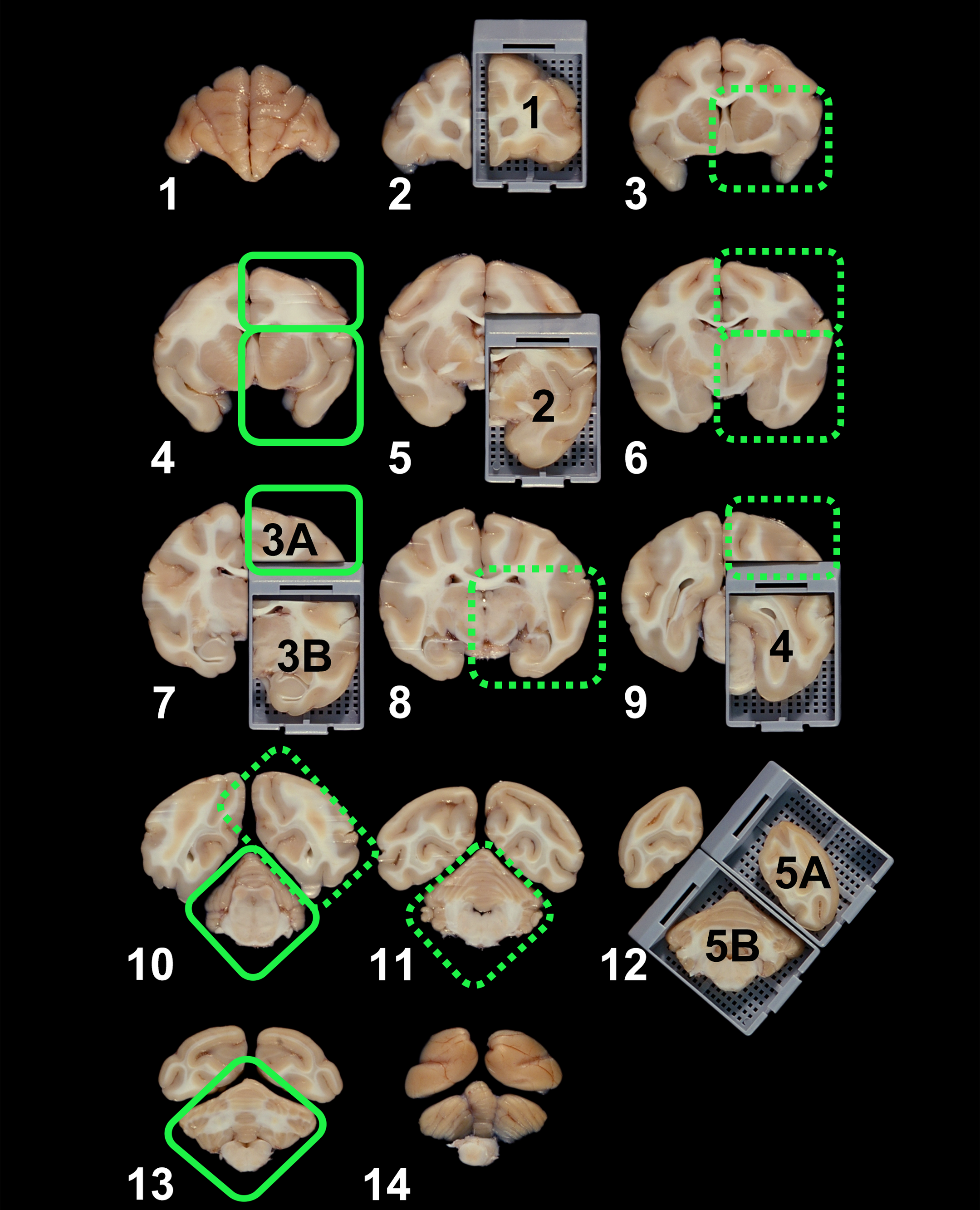
Additional trimming planes to consider for automated implementation of “enhanced” brain sampling (as preferred by the author) are depicted as green boxes overlying Figure 1. Areas in the solid boxes (N = 5) should be prioritized since they contain several previously unsampled major brain domains, while those in dotted boxes (N = 7) may also be warranted since they fall at a distance from samples acquired from histologically similar brain regions in nearby cross sections. The white numbers identify the 14 planes as enumerated in the original paper (Pardo et al. 2012), while the black numbers over tissue blocks label the 7 brain sections recommended for histopathologic evaluation of nonrodent brains in the published Society of Toxicologic Pathology “best practices” for nervous system sampling during general toxicity studies (Bolon et al. 2013).
The “targeted” approach to expanded brain sampling is founded on the trimming planes set in the STP “best practice” documents (Bolon et al. 2006, 2013), which then are supplemented by assessing other brain regions in which neurotoxicant-induced effects have been correlated with the induction of in-life neurological signs. This evidence-based approach requires considerable knowledge of structure/function correlations for major brain regions (e.g., Bolon et al. 2013). The variable nature of the possible effects to be investigated also means that it may be difficult to incorporate details for this adaptable “targeted” approach into standard operating procedures (SOPs). For this reason, the author suggests that institutions write their brain trimming SOPs and study protocols (or protocol amendments) using an “automatic” strategy for expanded brain sampling, with text that permits inclusion of any additional trimming planes that may be needed to include potential “target” sites that fall outside the regular complement of brain blocks.
The 2 published STP “best practice” recommendations for nervous system sampling and processing provide flexibility for institutions when selecting among several angles and many possible positions when deciding how to trim the brain (Bolon et al. 2006, 2013). A consistent orientation of brain trimming planes among all subjects in a study should be the driving focus rather than rigid adherence to the few visual examples of suitable orientations as illustrated in figures within the 2 STP “best practice” documents. Reproducible brain trimming at any angle which reliably unveils critical brain structures that are known targets for neurotoxicants (Bolon et al. 2013; Rao, Little, and Sills 2014) is the most effective strategy for following the STP “best practice” recommendations for brain sampling, and in the author’s opinion it should reliably meet regulatory agency expectations regarding neurotoxic hazard identification and risk assessment.
Footnotes
Acknowledgments
The author thanks Dr. James Morrison and Dr. Daniel Patrick for their critical review of the draft manuscript, Dr. Ingrid Pardo for providing the high-resolution version of Figure 1, and Ms. Beth Mahler for optimizing the resolution of annotations to ![]() .
.
Author Contributions
The author (BB) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. The author gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
