Abstract
Transcatheter prosthetic valves are heralding a new era in interventional cardiology and affording real therapeutic options to categories of patients currently medically disqualified, namely the elderly and higher risk individuals. An increasing variety of novel artificial valve designs and delivery systems are being tested preclinically. Cardiologists and surgeons are generally well-equipped to assess deliverability and function; however, methods for pathological evaluation of animals enrolled in transcatheter valve implant testing are scant, often vague, and far from consensual. Through this manuscript, we present and discuss a comprehensive evaluation platform that is proving reliable, reproducible, effective, and applicable to most, if not all, types and locations of valvular prostheses.
Valvular heart disease is estimated to affect approximately 2.5% of the population of the United States and sharply increases after the age of 65 years owing to the predominance of degenerative etiologies, causing significant morbidity and mortality to these patients (Iung and Vahanian 2011). Historically, the main etiology for valvular disorders was rheumatic disease; however, in recent years, we have seen a shift in demographics and primary etiology of alteration in the valvular systems. With prevalence of 2.5% in the overall population, degeneration of the mitral or aortic valves now leads to valvular heart disease etiology. With the advancement of therapies and increased human longevity, it is suggested that approximately 10% of the general population over 75 years of age, with all the additional comorbidities, are affected by this pathology (Iung and Vahanian 2011). In younger patients (low and intermediate risk for surgery) diagnosed with valvular disease, surgical replacements remain the treatment of choice (Nishimura et al. 2014). With the advancements in minimally invasive technologies, such as transcatheter delivery of replacement valves in the mitral, aortic, tricuspid, and pulmonary locations, the interventional cardiologist’s therapy arsenal has expanded to new horizons and is offering treatment opportunities to previously ineligible patients with valvular diseases.
Generally, and excluding a subset of novel approaches, the valves delivered by a minimally invasive approach consist of similar components: (1) a metallic frame manufactured from varying alloys allowing for either balloon expansion or self-expansion, (2) a sheet of pericardium from heterologous species (i.e., bovine or porcine) mounted on the metallic frame and geometrically engineered to create a functional valve, and (3) a polymer-based outer seal added to some designs as a result of clinical data. These basic components remain as innovative design advancements such as device miniaturization, profile optimization, and modifications of frame architecture work to improve the performance of the valvular prosthesis, decrease mechanical impact to adjacent native structures, and enhance deliverability (ensure proper topographical placement and long-term anchoring in the intended location).
Guidelines have been issued to establish a baseline according to which all transcatheter heart valves should be evaluated or, at a minimum, parameters to be considered. These guidelines are clear for in vivo end points pertaining to deliverability and function of the valve prosthesis and the imaging techniques that should be used. However, the recommendations for postmortem evaluation of preclinical valve specimens are not described in detail (ISO 5840-3). We present a comprehensive evaluation plan applicable to cardiac valve programs and large animal preclinical model applicable to either porcine or ovine model. This plan encompasses macroscopic examination, computed tomography (CT) imaging modalities, and their coregistration with histopathological evaluation and is supported by extensive practical laboratory experience.
Macroscopic Evaluation
Compared to other cardiovascular devices, valves are relatively large in size and tissue explants include a significant amount of surrounding tissues. For these reasons, it is appropriate to employ large animal models that have the proper anatomical dimensions closest to human anatomy. While regulatory agencies do not mandate particular species in the current guidelines, commercially available valvular prosthesis has been tested and approved on the basis of studies utilizing swine and sheep. Handling of the tissues prior to the pathologist’s evaluation must be minimized, and proper fixation (immediately following sample procurement at necropsy) is of paramount importance. Fixation of the heart with the valve in place is recommended by cannulating the aorta and flushing the blood remnants with isotonic solution (i.e., phosphate-buffered saline) until the fluid exiting the heart runs clear. This is immediately followed by flushing with fixative (i.e., 10% neutral-buffered formalin solution). Routinely, this is performed by holding roughly 1 L of fixative at approximately 100 cm above the heart such that the fluid runs through via vertical gravitational pull to ensure rapid, proper fixation of the innermost layer (i.e., myocardium and endothelium). The whole heart is then immersed in a minimum of 10:1 tissue to fixative volume for approximately 48–72 hr. If the sample is to be transferred for histopathological evaluation, it is imperative that the sample is packaged in a manner to alleviate risk of desiccation via exposure to air (i.e., covered with dampened gauze) during transit. Special care should be taken while handling the heart to ensure the integrity of the valve is preserved during packaging and transit. The described approach allows for proper valve procurement while permitting complete standard pathological evaluation of the myocardium.
Ideally, a high-resolution radiograph should be taken upon tissue harvest at the test facility and at the histopathology test site, at two orthogonal views (lateral and superior) to visualize the valve assembly in situ and assess placement within the native tissue architecture. The radiograph will display the location of the device, any potential displacement, and alterations such as fractures or conformation anomalies that may have occurred in vivo or postmortem (Figure 1). The initial macroscopic evaluation consists of examination of the myocardium for any macroscopic abnormalities. Thereafter, depending on the location of the device, surrounding tissue should be methodically dissected to expose both inflow (anterograde flow through the valve opening) and outflow (exit flow side) surfaces while examining all heart chambers and valves for any abnormalities. The adjacent surrounding tissue should be preserved intact in sufficient amount all around the device as to allow for evaluation of the interface between the implant and the native host tissue histopathologically. High-definition macroscopic images should be captured including the atrial and ventricular sides to illustrate the condition of the valve frame healing as well as the condition of the leaflets and other device components and any abnormality noted during the macroscopic evaluation (Figure 2). The tubular configuration of the valve implant sites presents particular optical challenges for macroscopic imaging and requires coaxial illumination conditions and high depth of field to ensure that images are sharp and legible through the entire depth of the conduit and valve surfaces.
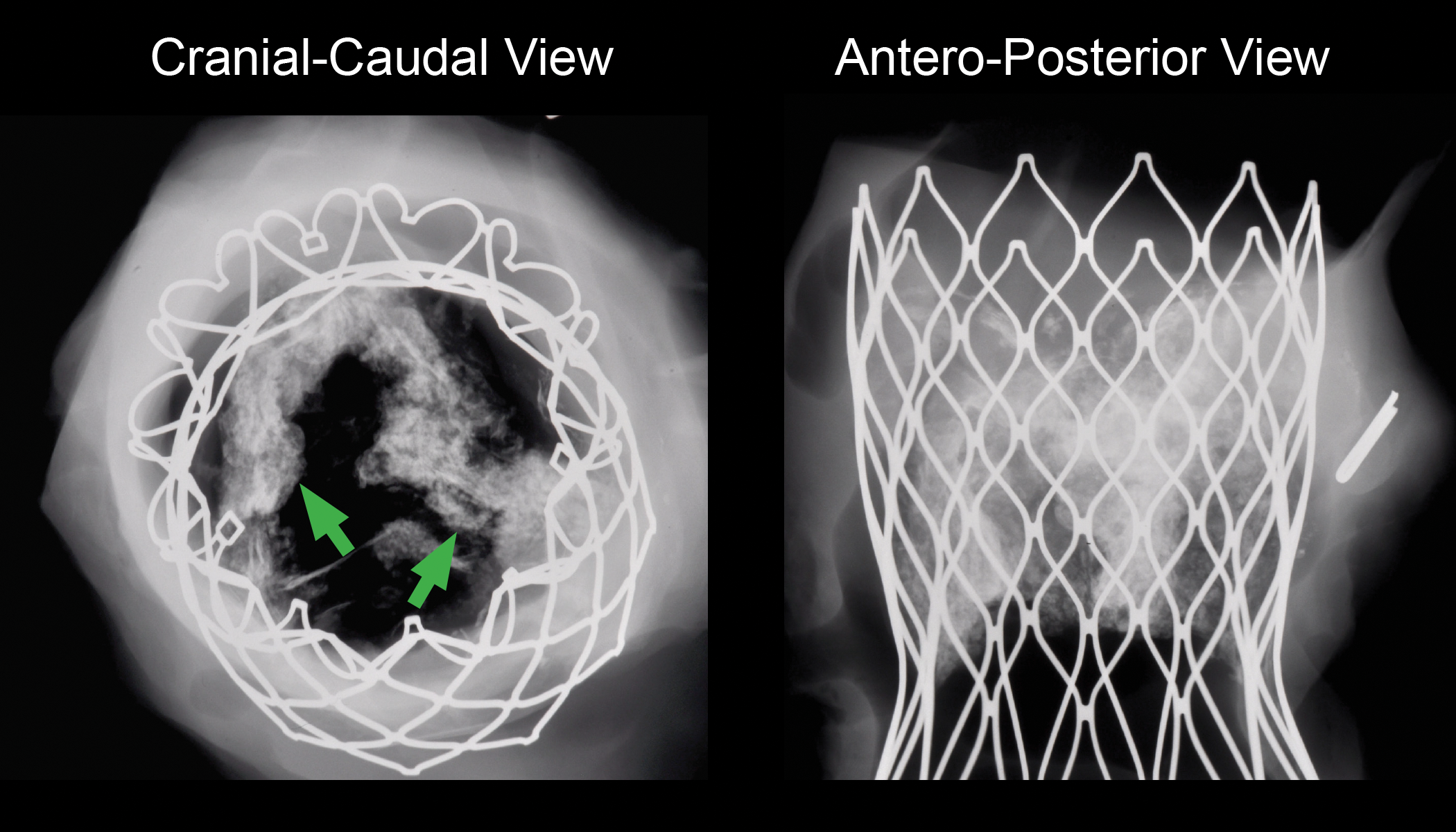
Representative images of digital high-definition X-ray radiography. The use of high-definition radiography in orthogonal positions (cranial–caudal and anteroposterior views) allows the evaluation of structural integrity and fracture analysis of the metallic frame, conformation anomalies, and the location of the leaflets, normally radiolucent and only visible due to the extent of heavy calcification (green arrows).
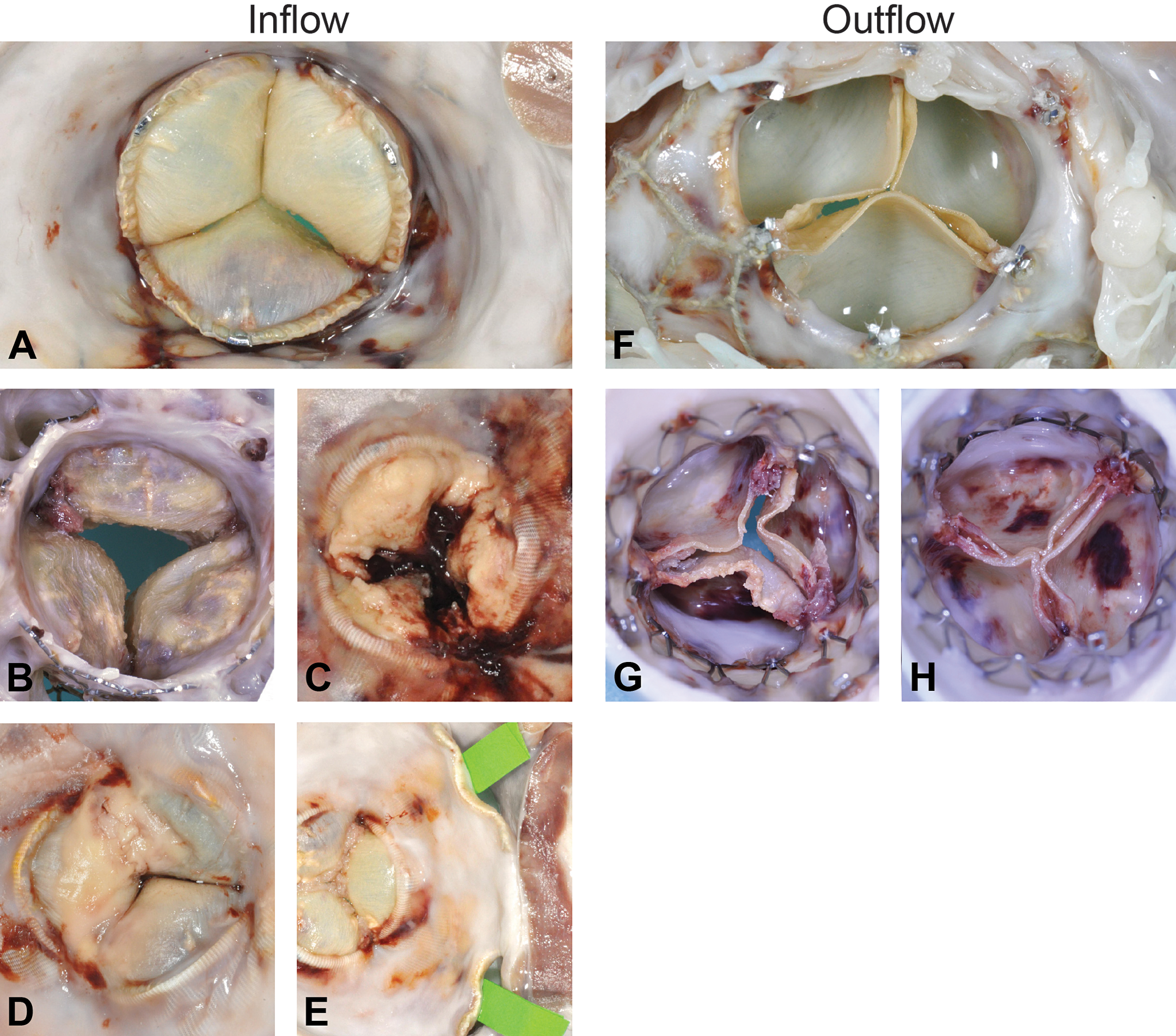
Representative images of valves during macroscopic evaluation. A comprehensive macroscopic evaluation approach consists of gross assessment of the inflow and the outflow of the valve. In the upper panel, images represent normal inflow (a) and outflow (f). Representative abnormal features are presented beneath. Deposits on the leaflets are quantified and characterized macroscopically, indicating the location of the calcification, lipidic, or pannus deposition (b) as well as thrombus and fibrin deposits (c). The presence of focalized/generalized septic endocarditis characterized by macroscopically thin, tan-yellow deposits on the cusps with mild to moderate calcification is an important part of the quantitative score (c). This feature eventually may progress, resulting in increased leaflet thickness or and leaflet fusion (d). The apposition of the valve to the native structure (i.e., annulus or conduit) is imperative and when there are paravalvular gaps (e, green tape) for correlation with in vivo imaging to determine clinical relevance. These same features should be searched and evaluated in the outflow of the valve (g, h).
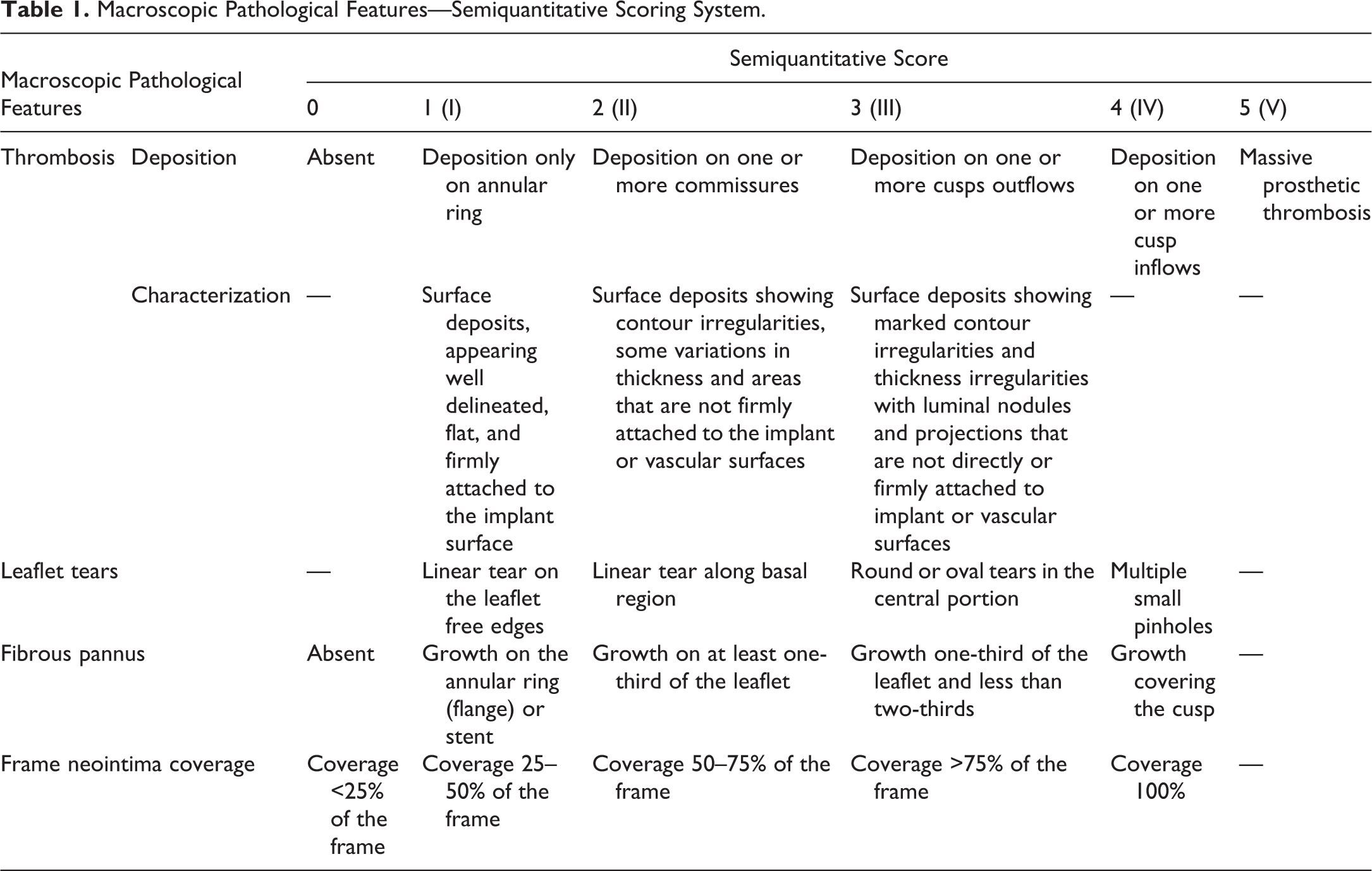
A semiquantitative scoring system was developed to properly document healing characteristics and macroscopic findings observed during the gross examination of the implant site to include at a minimum distribution and severity abnormal deposits such as thrombus or calcified deposits and healing response such as frame coverage a pannus formation on the cusps. Any implant alterations (tears) are scored for both the inflow and outflow aspects. Regurgitation channels and any other abnormalities are also noted. All these features are recorded on schematics of the cusps and frame. Figure 3 presents a representative example of the schematic employed during macroscopic examination of an aortic valve. This schematic provides a relational view among common pathological features and location thereof (i.e., valve cusp, valve base, and valve commissure) and constitutes a permanent visual record of the implant site characteristics and trackable abnormalities. Table 1 presents the semiquantitative scoring system with a description of each parameter. These tools are essential to issue a comprehensive gross evaluation of the valve in a transparent and reproducible manner.
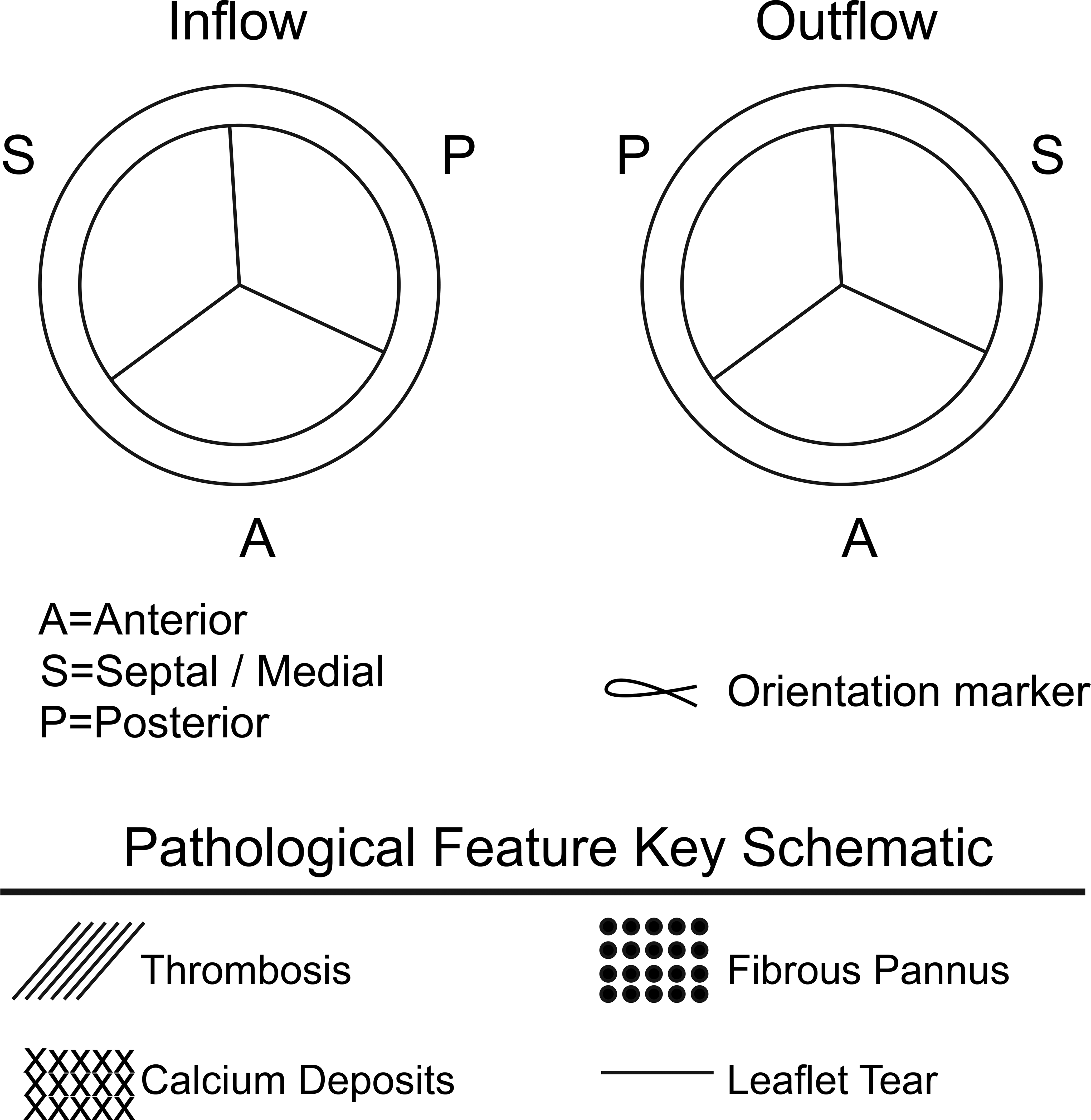
Schematic of macroscopic valve evaluation. Both aspects of the device, inflow and outflow, as well as all individual leaflets (posterior, anterior, and septal/medial) must be evaluated for the presence of thrombosis (///), fibrous pannus formation (:::), calcium (xxx), and leaflet tears (---). This suggested scoring system along with macroscopic images provides a clear, documented understanding of the gross evaluation.
Macroscopic Pathological Features—Semiquantitative Scoring System.
Nondestructive Imaging
Upon dissection, the excised implant site (atria and ventricles removed) is radiographed at high resolution under 3 orthogonal views (en face and two orthogonal profiles). These radiographs are used for calcification analysis and to assess the integrity of the scaffold using a semiquantitative grading system.
The following nondestructive imaging modalities have demonstrated great value in the comprehensive evaluation of valve technologies.
Micro-computed Tomography (micro-CT or µCT)
CT allows for the nondestructive and cost-effective, three-dimensional (3-D) visualization of tissues and devices based on volumetric reconstruction of radiopacity. Micro-CT or µCT takes advantage of ex vivo conditions (explanted specimens, unrestricted radiologic exposure, and limited field of view [FOV]) to great resolution benefits (e.g., 10–50 µm).
The value of 3-D digital rendering of valvular explants via µCT relies on an ability to separate the main components of the specimen, namely, soft tissue (leaflets and myocardium), abnormal deposits (calcification or thrombi), and frame based on radiopacity and material density differences. Once digitally reconstructed and visualized, these components can be segmented and measured.
There are numerous factors that contribute to a successful µCT evaluation of valvular prostheses. Sample stillness during scanning is critical, and the specimen must be prevented from drying during lengthy scanning sessions to avoid motion artifacts. Radiographic artifacts related to the presence of a metal frame (blooming or shadowing) present a significant challenge for this application and under certain conditions can result in pronounced shadowing or streaking artifacts precluding accurate extraction of radiographic signal associated with the soft tissue (frame noise, i.e., beam hardening artifacts based on mechanism that produce dark streaks between 2 high attenuation objects, like metal, with surrounding bright streaks). The extent of this effect must be minimized using appropriate acquisition parameters (i.e., scanning power, specimen orientation). Postprocessing methods can also mitigate shadowing metal artifacts.
Following acquisition, image analysis is typically conducted via a series of customized, automated software-based algorithms tailored to a study’s specimens, treatment type (e.g., drug or device), and investigative end points. For a standard valvular prosthesis, this first involves segmenting the volume into the valve leaflets, soft tissue, metallic cage, and calcification. Postprocessing may use low-pass filters to reduce metal streaking artifacts, flatten the image volume to remove blooming around metal edges, and threshold using area and intensity filters to segment the metal frame. A spectral filter to smooth tissue boundaries and remove noise, along with a shifted segmentation range, may then be used to isolate a specific parameter (i.e., calcification) in the preprocessed volume. In the example of calcification, this parameter is typically represented by voxels maintaining a Hounsfield Unit value above 1,000. Another low-pass filter and a wide intensity range may then be applied to extract the soft tissue component of the valves. In some cases, a connected components algorithm removes any segmented objects not connected to the larger tissue volume. Once a volume is segmented into its component parts, further quantitative evaluation is largely a mathematical endeavor, and algorithm outputs can be customized accordingly. In addition to quantitative information, digital analysis of µCT volumes provides a visual representation of segmented components, which may be pseudocolored for 3-D renderings (Figures 4 and 5).
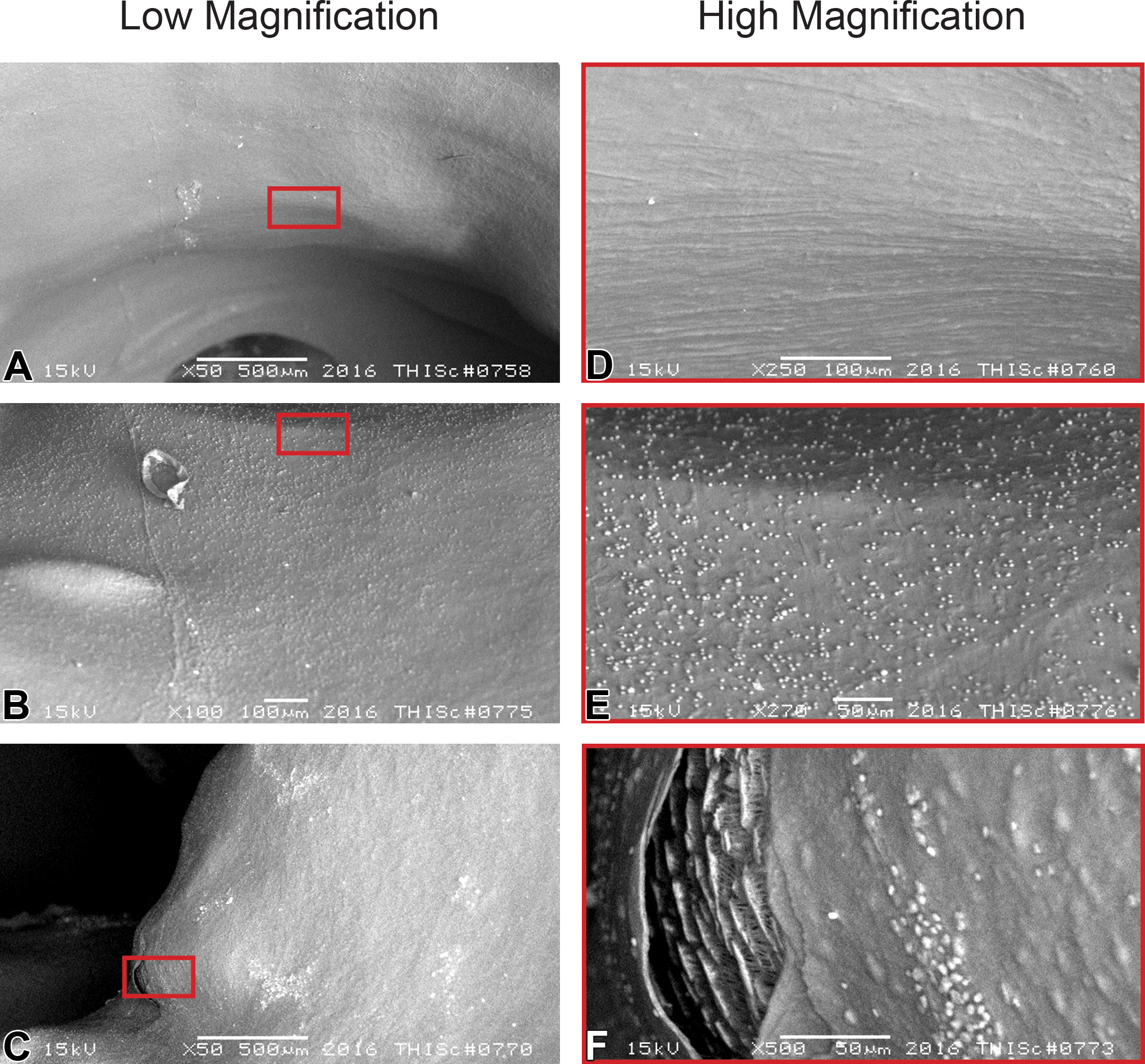
Representative images of scanning electron microscopy of leaflets. The complete, even, and smooth endothelial coverage, with no to minimal platelet aggregation, is the desirable biocompatibility response in an implanted device (images a and d). Nonflow obstructing platelet deposition (absence of thrombus) would be considered normal and expected since it is a normal foreign body response (images b and e). The integrity and potential interruptions of the device components (i.e., microscopic tears, c and f) need to be carefully evaluated and correlated with in vivo and histological features.
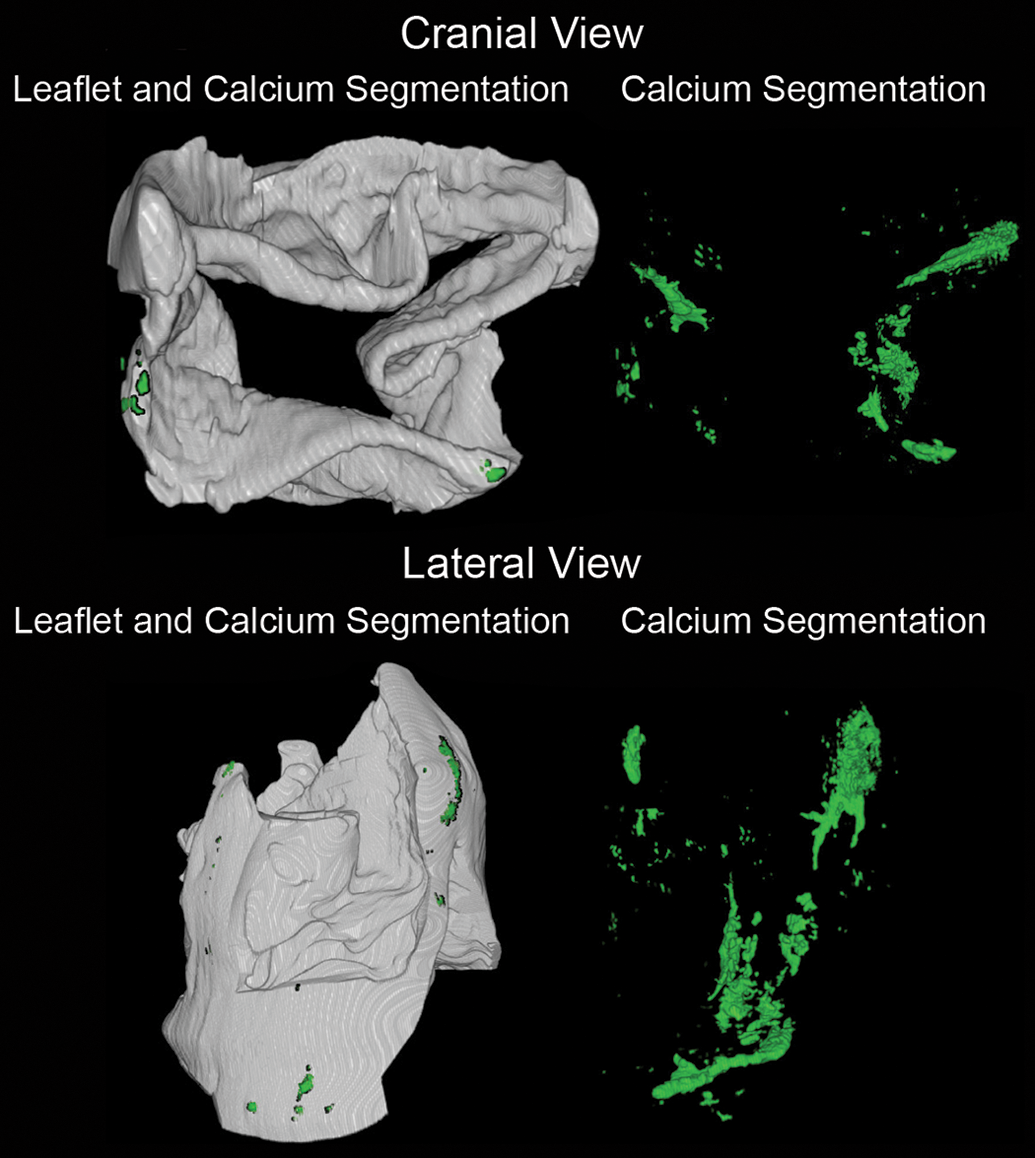
Representative images of microcomputed tomography (µCT). Displayed in the left panel is an example of a postprocessed scan utilizing soft tissue settings. The valve leaflets (gray) are overlapped with the calcium segmentation (green) in both cranial (upper images) and a lateral view (lower images). Under the proper settings and using metal deletion techniques, the segmentation allows for a representation of the soft tissue (myocardium, pericardium leaflets, and annulus) without the expected beam hardening and scatter artifact. The µCT image acquisition under allows the specific densities of each component to be isolated under specific segmentations which permit correlation of location and quantification of metrics (i.e., percentage, area, and extension).
Generally, the quality of a scan is highly dependent on the settings and may be limited by the specific µCT scanner available. Acquisition conditions (e.g., power, current, views, and frame averaging) should be optimized for any given µCT scanner, as they are often not consistent between manufacturers or models. For detailed analysis of valvular prostheses, high-resolution (e.g., 10–50 µm) scanning is required. Higher resolution scans offer additional measurement precision but may limit the maximum FOV captured in a single scan and may take additional time (and scans). For larger preclinical samples (e.g., swine mitral valves) acquired at high resolution (e.g., 20–50 µm), multiple scans may be required to visualize the entire region of interest (ROI). Sequential scans can be and stitched together to form a single contiguous ROI volume for analysis.
The chief advantage of µCT evaluation as applied to valve implants is the ability to assess cusp and conduit calcification in a comprehensive and unbiased manner. Current methods for calcification assessment rely on biochemical analysis of cusp samples collected in arbitrary if consistent locations (i.e., sliver of cusp dissected out at tissue trimming). Calcification is also evaluated histologically on standard sections of the cusps using routine (hematoxylin and eosin [H&E]) and special stains (e.g., Von Kossa [VK]). The standard and arbitrary location of these biochemistry and histology samples belies the generally random and varied distribution of calcium deposits in collagenous prosthetic valves. These techniques, while helpful in identifying extremes cases of calcification, can underestimate or overestimate calcification in situations of intermediate calcification due to sampling bias. µCT offers a critical advantage because it is comprehensive and informs on the severity and distribution of the calcium deposits and their relationship to other abnormalities such as thrombi, tears of specific structural or anatomical features. Using appropriate standards, calcium deposits segmented by µCT can be precisely quantified for volume distribution and calcium density.
In devices using bioresorbable components, the presence and rate of resorption can be visualized and quantified by radiographic segmentation and volumetric quantification of this component over time.
Availability of 3-D µCT representations can be very helpful in assessing material integrity such as frame fractures and help direct histology sectioning to specific planes or areas of interest where features of specific interest have been identified macroscopically and/or radiographically (CT-guided histology sectioning). This provides an unprecedented opportunity to prepare representative sections for more relevant analysis. Consequently, macroscopic, microscopic, and µCT findings (data and the actual images) can be coregistered and correlated to provide confidence that the implant is thoroughly assessed prior to clinical testing. These benefits have been recognized by the Food and Drug Administration in a recent update to the guidance for best practices calling for the use of µCT for postmortem explant imaging and assessment in preclinical studies (Iung and Vahanian 2011).
Surface Evaluation by Scanning Microscopy
The scanning electron microscope (SEM) is used to evaluate low- and high-magnification surface texture and topography. It is used to provide resolutions that cannot be seen by the human eye and can be used on almost any material that can be dehydrated not limited to biologicals. While the SEM will not provide any dynamic parameters, the extreme high-resolution images that SEM is capable of acquiring include a scanning area reaching up to 300,000×. Electrons behave like light only when they are manipulated in a vacuum environment. Vacuum pumps are used to obtain and maintain differing levels of vacuum between the electron gun and the fluorescent screen. The SEM can be used under 2 settings: low and high vacuum. Under both settings, the sample needs to be dehydrated by either graded alcohol (LV) or critical point drying (HV). The advantage of using low vacuum is the elimination of critical point drying and metallic coating, which allows the sample to be rehydrated and used for other purposes, such as other nondestructive imaging techniques and/or further histopathological assessment.
A heavy element like gold gives a good yield of secondary electrons (used for nonconductive samples that will be charged up or samples that contain volatile components, i.e., water) producing a higher resolution image. Additionally, gold is a preferred element since it is easily applied in a thin coating (10 nm) via sputter coater. The advantage of high vacuum is greater resolution due to the critical point drying and the above-mentioned metallic coating. However, important is to note that once a sample is critical point dried and metal coated, it is irreversible; therefore, it cannot be used for another purpose.
The objectives of SEM evaluation of a valvular prosthesis can be summarized in three main objectives: (a) the resulting high magnification produced by SEM allows a scrutinized survey of the surface of areas such as leaflets to assess proper endothelialization, rule out leaflet degeneration, or score subendothelial proliferation and migration (Mow and Pedersen 1999; Stein et al. 1989), (b) leaflet integrity assessment (superficial tear or fracture exposing underlying components), and (c) platelet adhesion and thrombosis assessment. Figure 6 shows a panel of images under low (a, b, and c) and high (d, e, and f) magnification in which representative images of endothelialization (a and d), platelet adhesion (b and e), and microscopic ruptures (c and f) are evaluated.
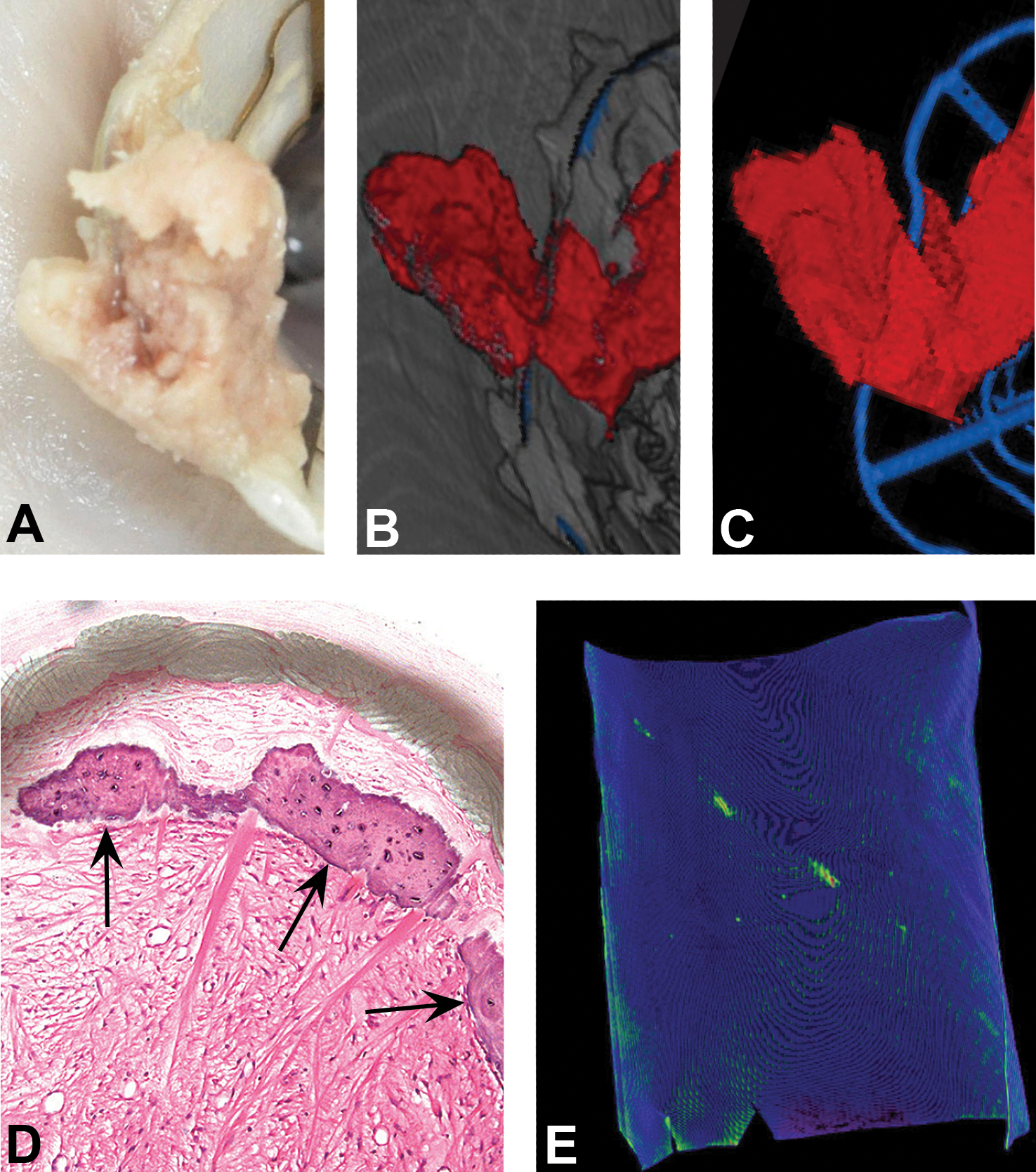
Representative images of microcomputed tomography (µCT) segmentation. In addition to metal and calcium, other different tissue components observed during macroscopic assessment (a, thrombus over the frame) can be segmented to provide a nondestructive visualization and potentially quantification (b, same thrombus under µCT segmenting for soft tissue and metallic frame). All segmentations can be combined or isolated (c, thrombus segmented in red and metal in blue) for optimal viewing and analysis. The 3-D rendering resulting from the µCT scanning allows a model in which the sample can be digitally rotated in all axis observing each segmentation in the best ideal perspective for better understanding and orientation. The level of detection of the µCT is in the scale of 5 µms which allow the highest level of detection available which can be inconspicuous at macroscopic assessment and at the sectioning level for histopathological evaluation from true osseous metaplasia (clear arrows indicate narrow rim of osseous metaplasia, d) to mineralization (swine pericardium sheet fixed in 0.6% glutaraldehyde and injected in diagonal pattern with a 2% calcium phosphate solution, e).
Histology and Microscopic Evaluation
Due to the technical challenges associated with preparing histologically complex and large bioprosthetic implants such as a valve explants, the conventional approach has been to dissect out slivers from each cusp for preparation in standard size paraffin slides. The frame and conduit have sometimes been overlooked or in best cases prepared in methylmetacrylate (MMA) plastic for histology. MMA embedding is widely used for hard tissues such as bone but presents limitations for slide staining (histochemical and immunohistochemical) that have impeded its use for cardiovascular and other soft tissue applications. Advances in plastic histology, namely, processing and infiltration in epon (Spurr), are now making it possible to embed large and complex tissue explants in plastic and retain nearly all staining options available in paraffin. Tissue processing is achieved by tissue dehydration (through gradual alcohols) followed by clearing with organic solvents (typically xylene or acetone). In valvular prosthesis, amount and type of implant and associated surrounding tissue will vary, and the processing technique and schedules have to be customized accordingly to achieve thorough processing, full clearing, and infiltration. Processing and infiltration times increase supraproportionally with specimen size in a near logarithmic fashion. Therefore, care must be taken to customize processing protocols very carefully to produce good results devoid of artifacts that can support the production of high-quality slides. For large valves, this can result in extended protocols of up to 4 days for processing followed by several weeks of tissue infiltration in epon.
Sectioning Planes and Stains
Routine paraffin sectioning of dissected cusp samples has typically been radial sections of the cusp alone, excluding its attachment to the frame and not including the annulus or the frame and conduit. Infiltration in Spurr epon supports a nondestructive approach axially through the cusp, down to the base of the leaflet with the frame and native tissue (i.e., native valve, annulus, myocardium, and native artery) represented in Figure 7, panel 1. Using these techniques, the cusps can be cut at 5 microns and stained with the conventional staining panels desirable for evaluation of healing, integration, inflammation, and calcification, to include H&E, Movat’s pentachrome, VK, and/or CD-31 immunostaining for endothelial cells or other immunostains (SMA, Ki67, etc.) or diagnostic stains (i.e., GRAM stain for bacteria) as necessary. Additional sectioning may be obtained to assess regions of the explant not typically accessible or investigated through conventional approaches in paraffin such as additional axial or cross sections through the cusps and/or frame to assess valve commissures and varying levels in the conduit, for instance, to track and assess specific macroscopic observations. Due to the presence of a large amount of metal, these sections are prepared using the grinding/micropolishing technique using thick, albeit optically clear wafers (∼100–200 µm thickness) polished to ∼60–100 µm to optical finish and surface stained with H&E for general assessment and/or a trichrome or elastin trichrome stain for fibrin and connective tissue (Figure 7, panel 2; Figure 8).
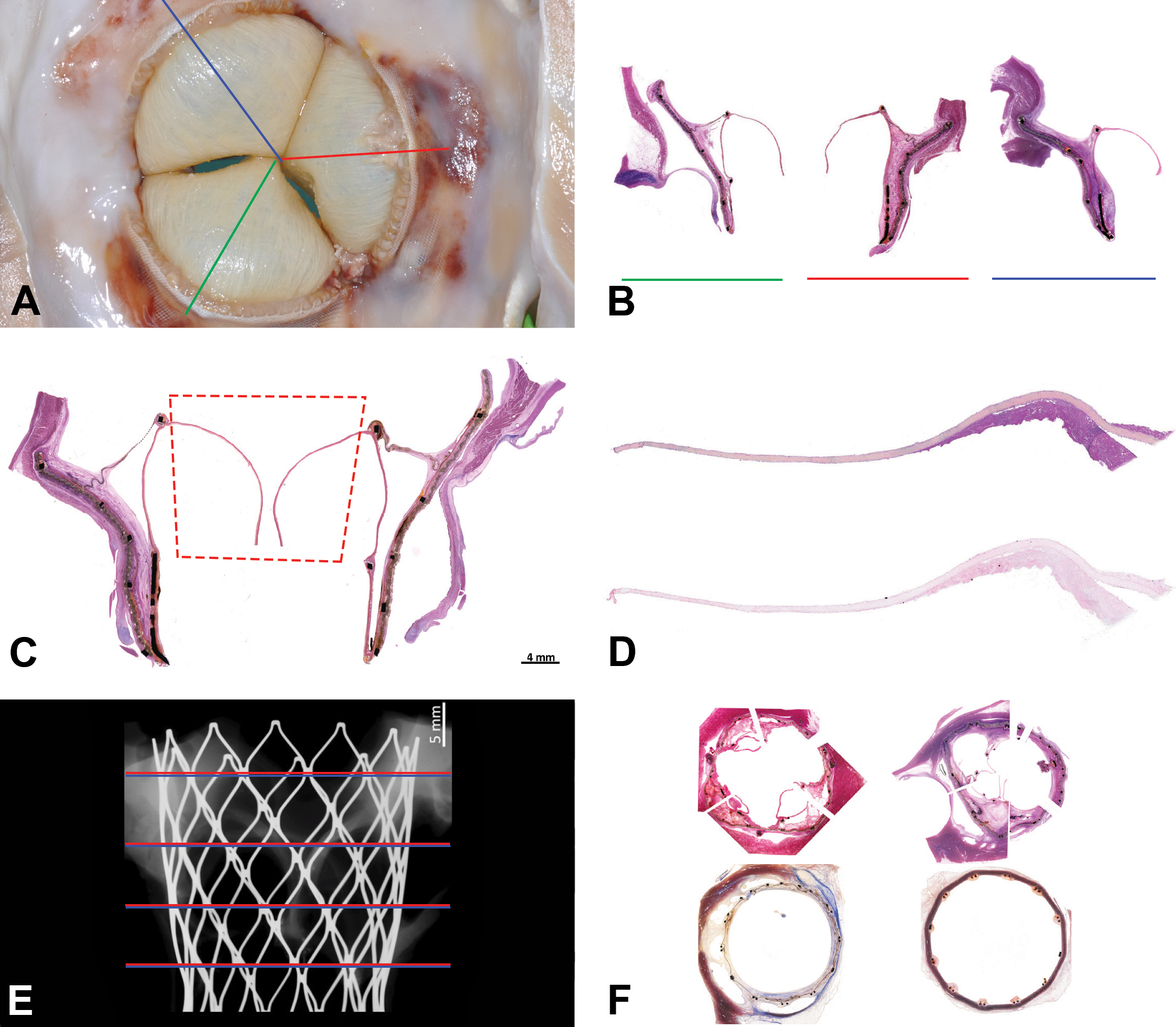
There are 3 main approaches that we suggest must be utilized to completely perform a proper preclinical evaluation of a valvular prosthesis by histopathology. Radial approach (a and b): The entire embedded valve is sectioned in the radial plane through the center of each leaflet to include the frame and surrounding tissue. This provides a clear view of neointima, pannus formation, and the coverage of the leaflets via a comprehensive perspective of the valvular apparatus with the native surrounding tissue. Coronal approach (c): A coronal wafer is taken from the plastic block that will display the frame and the leaflets from its base to its cusps and the relationship between the device and the native tissue. Leaflet approach (c and d): From the coronal wafer taken from the valve, the leaflet is separated for thin microtomy (c red square). This thin microtomy (d) would allow not only its histopathological evaluation but serial 5 µ sections in which special stains (i.e., Von Kossa, Movat’s pentachrome, Masson’s trichrome, and picrosirius red) and immunohistochemistry is possible. Cross-section approach (e and f): Once all the sections are taken, the block is reconstituted and sectioned transversely (e red and blue lines represent 2 different stains at each level) with a near complete circumference (f) the valve can be evaluated from along the length of the valve progressively, from proximal to distal, with different stains (f).
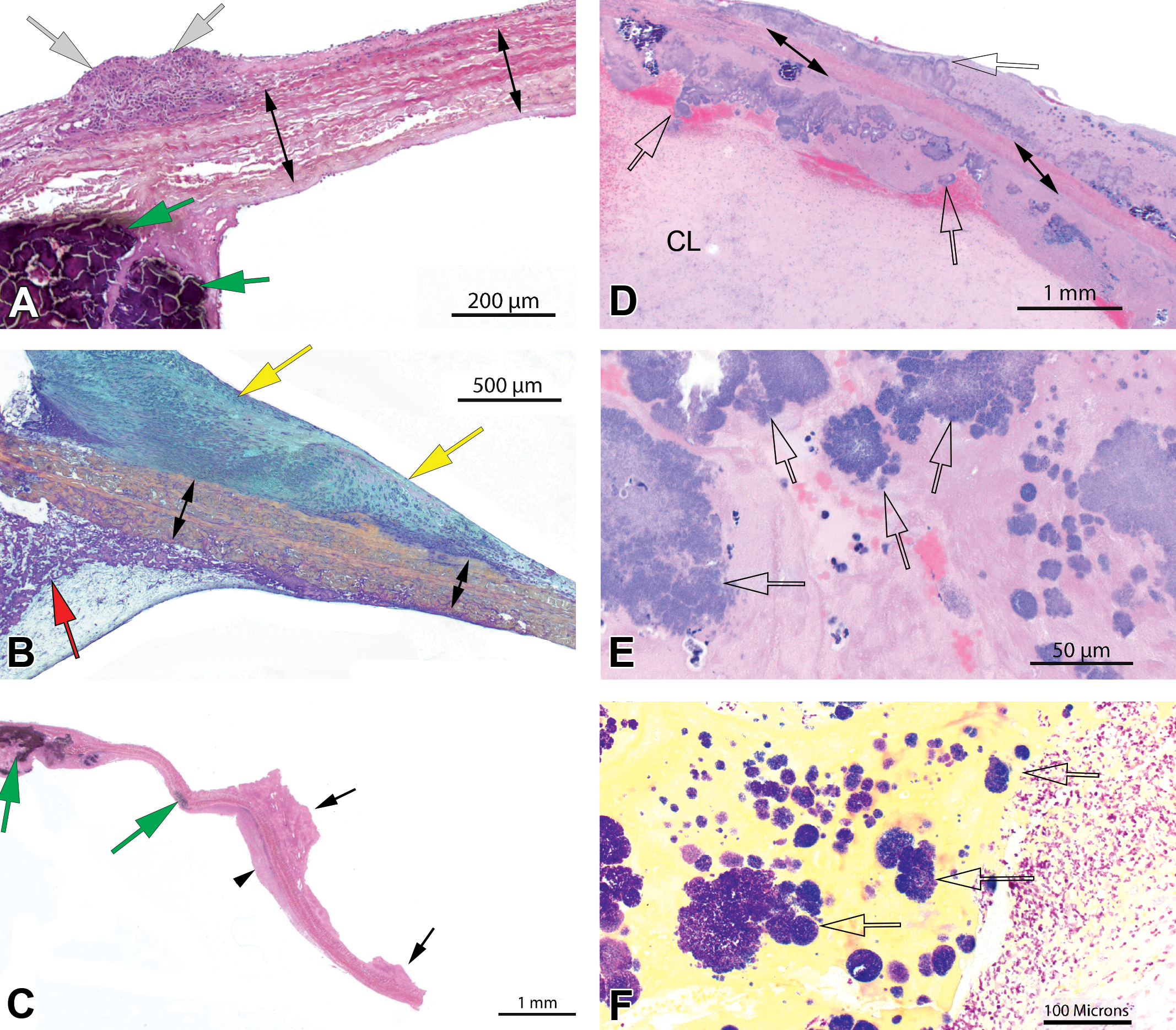
Representative microscopic pathological features. Localized inflammation (a, white arrows) consistent with low-grade foreign body response, a common feature found along the surfaces of collagenous bioprosthetic cusps (a, double arrows) and/or conduit. Fibromuscular pannus formation (b, yellow arrows) at the base of the collagen cusp (b, double arrows) is within normal limits as shown and may potentially impair the functionality of the valve when thicker and extensive. There is residual fibrin (b, red arrow) with associated calcium deposition at the base of and in the cusp (a, c, green arrows). A fibrin mat (c, arrowhead, outflow) and small fibrin thrombi (c, black arrows, inflow) are visible along the surfaces of the cusp. Bacterial vegetative endocarditis is a common complication in the swine model and is associated with abundant fibrin deposition or cusp thrombosis on either side of the cusp with inflammatory cells, namely, neutrophils and bacterial colonies (d, e, f, clear arrows). A postmortem clot can be attached to the cusps and vegetations (d, CL) and should not be confused with thrombosis. A Gram stain can be helpful in characterizing bacteria. Image f shows the tip of the collagenous cusp implant with Gram positive bacterial colonies (f, clear arrows) and purulent inflammation (f, right side of image).
Once the radial sections are obtained, the residual block fragments can be reconstructed and glued back anatomically together using appropriate clear spacers. The block is rotated 90° to procure sequential transverse (coronal) sections distal to proximal in the conduit. These sections include the frame and are prepared using grinding and micropolishing and stained with H&E and/or a special polychromic stain (i.e., elastin trichrome). The transverse sections are equivalent slides prepared from vascular stents and show the implant–tissue interface circumferentially with portions of residual cusps and commissures at applicable levels (Figure 7, panel 3).
Semiquantitative Microscopic Scoring of Pathological Features
Functional end points are evaluated in vivo using various imaging modalities (CT, sonograms, and echocardiogram). The objectives of the postmortem pathology evaluation of a valvular prosthesic implant include frame and bioprosthesis integration and local biocompatibility as well as general healing characteristics with safety evaluation as a prime objective.
Our evaluation approach is systematic, addressing healing and pathological features associated with the functional elements (cusps) and the supporting frame from the luminal side to the interface with the surrounding tissue (annulus, myocardium and/or native artery, and leaflets). The histology techniques that we have developed are designed to support all these end points in an optimal and integrated manner, optimizing the integrity of the tissue and implant.
Key histopathology end points should include endothelial coverage, thrombus or fibrin deposition, calcification and the formation of neointima (fibrocellular pannus), and its maturity. A nonexhaustive list of other changes evaluated includes inflammation, neovascularization, necrosis, fibrosis, hemorrhage, and the presence of bacteria. These parameters can be scored semiquantitatively on a 0–4 scale according to severity (Table 2). Specific implant features such as atrial cuffs, apical anchoring system, transseptal, or transatrial access points may require additional sections and evaluation.
Microscopic Pathological Features—Semiquantitative Scoring System.
Thrombosis and fibrin deposition (i.e., fibrin pannus) are paramount to safety assessment for any vascular implant, and valves are no exception. Technically, pannus is defined as a layer of vascularized fibrocellular tissue on the surface of a preexisting tissue element. It often derived from organization of a fibrinous deposit and by extension we refer to this early deposit as fibrinous pannus. Fibrinous pannus differs from thrombus deposition in that it is a generally thin (up to a few 100 microns) and homogeneous mat of fibrin that is firmly attached to the cusp material and appears self-limited and shows no delamination. By contract, thrombus deposition is generally irregular, heterogeneous, may be partially delaminated, and often appears progressive with leading edge(s) of newer fibrin deposition. It is important to distinguish fibrin pannus deposition from thrombosis as they have important and distinct safety implications. A degree of self-limited and stable (i.e., nondelaminated) fibrinous pannus is common and expected on the surface of bioprosthetic valves, and it can be considered as the incipient biological event leading to true fibrocellular pannus formation overtime. Excessive pannus formation can lead to an alteration in valve biomechanics and hence may impact function but does not typically represent an embolic risk. By contrast, thrombus deposition must be assessed for its inherent embolic potential. Evaluation of organization, delamination, and/or progression as well as distal organs is paramount to assessing the significance of valve thrombi.
Endothelialization of intravascular implant sites is always regarded as a favorable outcome. In valve implants, the conduit is typically covered by endothelialized and maturing neointima relatively quickly, in a manner and along timelines that are very similar to what is observed in large vessels implanted with stents or stentgrafts. A lack of endothelialization in the conduit may indicate instability, malapposition, or impaired healing. Endothelialization of collagenous bioprosthetic cusps is often restricted to the base due to the combined effects of limited tissue remodeling and high shear and biomechanical stress (friction against opposing cusp). Endothelial coverage is sometimes more extensive along the outflow side where shear and friction stress is less extreme.
Implant calcification is usually readily identifiable on H&E stains for its dark purple staining and the cracked/brittle appearance of the tissue area. It can be confirmed using special stains such as VK that stain calcium deposits dark brown to black. Microscopic evidence of calcification is useful and its extent is a factor in severity scoring. However, it should not be interpreted in isolation to avoid sampling bias. A thorough assessment of implant calcification should involve results from the macroscopic, radiographic, and microscopic assessment and preferably a tissue µCT visualization and quantification as well.
Valve local biocompatibility is evaluated via the inflammatory response along its planes and at the interface between the device and the annulus. A degree of foreign body response is expected and may vary based on the nature of the biomaterial(s) and study duration or species. It is generally useful to score inflammation along the various structural elements (cusps, graft, frame, and annular/myocardial interface). Inflammatory cell types should also be scored individually in order to provide insights on the nature of the response. A foreign body response is most commonly characterized by foreign body giant cells along the eliciting material surrounded by one or several layers of macrophages and lower proportions of lymphocytes and/or eosinophils. The presence of large proportions of neutrophils may indicate some degree of irritation, particularly if it persists over time. Solid collections of neutrophils may be a telltale sign of underlying bacterial infection, and in this instance, a Gram stain may be useful as an additional screening and diagnostic tool, although a negative result may not preclude a bacterial etiology. A tissue response showing large proportions of lymphocytes, namely, along the annular and myocardial interfaces, may suggest a cell-mediated hypersensitivity response to one of the implant components. Such reactions are rare since valve constructs generally use well-known and characterized biomaterials. Modifications imparted to them are typically aimed at reducing calcification and improving biomechanical properties and typically do not significantly alter antigenicity in animal models.
Evaluation of Downstream Tissues
Downstream embolization is evidently a major risk associated with any cardiac implant. While a comprehensive necropsy is advised, relevant downstream organs should be examined with particular attention (at a minimum lung for a right-side implant and heart, brain, and kidneys for a left-side implant). These organs are at a higher embolization risk due to disproportionate fraction of blood flow they receive and they should also be surveyed microscopically for emboli and evidence of embolization, infarction, or ischemia. Additional tissue may be examined (e.g., spleen or liver but they generally yield no implant-related findings). We recommend fixing these filtering organs in toto for subsequent serial slicing (“breadloafing”) and gross examination by the study pathologist for emboli and/or any changes possibly consistent with ischemia. Any gross lesions as well as systematic samples are trimmed and prepared to slide for microscopic evaluation. This plan constitutes a diligent approach to embolization risk assessment.
Discussion
Translational research has played a vital role in scientific and medical advances of the past century. Animal models have afforded the scientific community a better understanding of pathological processes and biological responses to therapies in humans. Progress supported by preclinical research has transformed the quality of life and medical prognosis for countless patients and has offered treatment options to patients previously deemed terminal (Festing and Wilkinson 2007). Transcatheter or surgically implanted valvular prostheses are emblematic of this paradigm. Under the proper ethical framework and the supervision of trained scientific personnel, with profound understanding of the responsibilities entailed in performing translational research (Matthiessen, Lucaroni, and Sachez 2003), evaluation of valvular prostheses can be conducted efficiently, respectfully, and humanely with scientific integrity. Among the ethical imperatives of preclinical animal studies is the responsibility to reduce animal use and hence generate quality and actionable data. Some devices can be deployed and tested in multiple locations (i.e., intravascular devices, stents, and drug-coated balloons), which is an approach consistent with the concept and mandate of the 3Rs (Würbel 2017). For other implants such as valves, it is difficult and sometimes impossible to find multiple sites that are sufficiently equivalent anatomically or biologically to justify such approach. By and large, valvular prostheses are limited to one device per animal in order to yield relevant translational data.
Preclinical studies are primarily directed at assessing safety. As for other devices, safety end points are often difficult or impossible to assess due to inherent anatomical or physiopathological differences (Gallegos et al. 2005). For instance, implantation in nondiseased naive valvular locations (in different species such as dogs, DeAnda et al. 1995; goats, Iwasaki et al. 2002; sheep, Gregoric et al. 2004; calves, Gregoric et al. 2004; and pigs, Gross et al. 1997; Smerup et al. 2004) contrasts with the likely heavily calcified, fibrotic, or deteriorated target location in the clinical setting. Some models (i.e., sheep) have been reported to have failed to be predictive of thrombotic risk with early generation valve implants (Li et al. 2007).
Valve prostheses are complex devices biomechanically, and their engineering requires the assembly of a variety of components and biomaterials. This often includes a metallic or polymeric supporting frame and a functional element (most typically collagenous leaflets) with various ties and supporting fabrics assembling these components.
Vast amounts of relevant data can be acquired in vivo (i.e., fluoroscopy, echocardiography, CT, hematology, etc.). A comprehensive ex vivo approach is equally important in order to assess biointegration, healing, local biocompatibility, and downstream impact in order to ultimately formulate an opinion on implant safety.
This review proposes a comprehensive imaging and pathological framework as a guide for an integrated valvular prosthesis evaluation. Based on our experience with a variety of valvular prosthesis designs, we have found that this platform is sufficiently thorough and lends itself well to correlations with in vivo parameter. Additionally, this approach overcomes limitations and artifacts frequently impeding valve evaluations. For instance, many laboratories excise small portions of the leaflets for biochemical analysis of calcium content and for paraffin embedding and light microscopy. This produces a sampling bias (Ca analysis) as well as a disruption of specimen integrity (histology) which in our opinion is unnecessary and suboptimal.
We included scanning electron microscopy for the evaluation of valve prosthesis. This technology is tailored to detailed evaluation of surfaces and has demonstrated its usefulness for other devices to help characterize reendothelialization, deposits, and integrity of vascular surfaces. This technology remains underused for the evaluation of valvular prosthesis and should be considered specially when characterizing surface healing and fibrin deposits or thrombosis.
The uCT approach described herein provides a detailed and topographical view of calcium deposits, that is, quantifiable and free of sampling bias. The histological methods that we advocate preserve explant integrity and support polychromic and immunohistochemical stains.
Some of the techniques described herein such as semiquantitative macroscopic scoring by qualified personnel and detailed and systematic photographic documentation of implant sites can be easily implemented. Other techniques such as wet tissue µCT or whole implant plastic histology require specialized and costly equipment and may be performed collaboratively. With respect to µCT, we have found that image acquisition is often well understood. However, postprocessing protocols tailored to soft tissue, calcium, and frame segmentation can be challenging, particularly with respect to managing shadowing and blooming artifacts in soft tissues to an acceptable level. Sample (i.e., frame) orientation during scanning, density of frame members, use of filters, and postprocessing protocols all play a role in imaging outcome, and the development of new scanning and postprocessing methods will surely improve in years to come.
Conclusion
We propose a new and comprehensive guide for preclinical postmortem evaluation of valve implants that is applicable to all or most valvular prostheses currently under evaluation. Based on study objectives, this approach provides an “à la carte” menu of analytical tools and may be customized to optimize the outcome of the evaluation and yield quality data that are objective, unbiased by sampling, and contribute to advancing the field of valve safety assessment.
Footnotes
Acknowledgments
The authors would like to present special thanks to the team of Image IQ (Cleveland, OH), specially to our colleagues Brett A. Hoover, Amit Vasanji, and Zach Kaufman who have provided outstanding scientific and technical expertise in the application of microcomputed tomography on valvular prosthesis. The authors would also like to thank the team of Texas Heart Institute (Houston, TX), specially to Pamela Potts and Sara Lee who have always been supporting our team with their incredible scanning electron microscope experience. Special thanks to Grace James from Alizee Pathology who participated in the creation of this article.
Authors’ Contribution
All authors (AT, KD, SR) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
