Abstract
The INHAND (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions) Project (www.toxpath.org/inhand.asp) is a joint initiative of the societies of toxicologic Pathology from Europe (ESTP), Great Britain (BSTP), Japan (JSTP), and North America (STP) to develop an internationally accepted nomenclature for proliferative and nonproliferative lesions in laboratory animals. The purpose of this publication is to provide a standardized nomenclature for classifying lesions observed in most tissues and organs from the dog used in nonclinical safety studies. Some of the lesions are illustrated by color photomicrographs. The standardized nomenclature presented in this document is also available electronically on the internet (http://www.goreni.org/). Sources of material included histopathology databases from government, academia, and industrial laboratories throughout the world. Content includes spontaneous lesions, lesions induced by exposure to test materials, and relevant infectious and parasitic lesions. A widely accepted and utilized international harmonization of nomenclature for lesions in laboratory animals will provide a common language among regulatory and scientific research organizations in different countries and increase and enrich international exchanges of information among toxicologists and pathologists.
Table of Contents
Introduction
General (Multisystemic) Pathology
Cardiovascular System
Digestive System
Endocrine System
Hematolymphoid System
Hepatobiliary System
Integumentary System
Mammary Gland
Nervous System
Female Reproductive Tract
Male Reproductive System
Respiratory System
Skeletal System and Tooth
Soft Tissue and Skeletal Muscle
Special Senses
Urinary System
1. Introduction
The INHAND Project (International Harmonization of Nomenclature and Diagnostic Criteria) is a joint initiative of the societies of toxicologic pathology from Europe (European Society of Toxicologic Pathology—ESTP), United Kingdom (British Society of Toxicological Pathologists—BSTP), Japan (Japanese Society of Toxicologic Pathology—JSTP), and North America (Society of Toxicologic Pathology—STP) to update the existing World Health Organization (WHO)/International Agency for Research on Cancer and Society of Toxicologic Pathology (STP)/Standardized System of Nomenclature and Diagnostic Criteria nomenclature systems. The INHAND nomenclature and the related diagnostic criteria represent a consensus of experienced toxicologic pathologists and were reviewed by the INHAND-GESC (INHAND-Global Editorial and Steering Committee) for compliance with INHAND principles. Members of the societies of toxicologic pathology had the opportunity to comment to the draft version during a 60-day period. The initial series of nomenclature publications were focused on lesions in rats and mice. With interest of the US Food and Drug Administration in the use of published terminology standards and the decision of the Clinical Data Interchange Standards Consortium initiative on Standard for the Exchange of Nonclinical Data (SEND) to model the controlled terminology (CT) based on the INHAND nomenclature, the INHAND project was extended to other laboratory animal species including the monkey, rabbit, mini-pig, fish, and dog.
Although the INHAND nomenclature and diagnostic criteria represent a preferred international standard nomenclature for lesions identified in nonclinical studies, recommendations for diagnostic criteria and preferred terminology may not be applicable in all situations. The purpose of specific experiments or the specific context of a given study may require modifications to this standardized nomenclature and diagnostic criteria. The appropriate diagnoses are ultimately based upon the scientific judgment of the study pathologist.
The present publication provides standardized terms and diagnostic criteria to be used in nonclinical toxicology studies conducted in laboratory beagle dogs. Throughout this publication, lesions applicable for use in toxicology studies in dogs are tabulated by organ system. The terms and tabulations are built on the existing INHAND rodent nomenclature. In most instances, the description and definition of the rodent lesion also applies to the dog and therefore is not further described. This publication focuses on lesions that are unique to the dog and are not observed in rodents and lesions in dogs that share the same terminology with a rodent lesion but display different morphologic features. Lesions that are unique to rats or mice and are not to be used in dogs are denoted accordingly in the tabulation. The tabulated lesions are categorized according to the following characteristics: “common,” “uncommon,” “not observed but potentially relevant,” and “not applicable.”
The distinction between common and uncommon lesions is based on the occurrence in untreated beagle dogs in the authors’ experience and is not based on published references. The uncommon category is reserved for changes that are observed only sporadically as spontaneous findings in most dog studies or those that are induced almost exclusively by xenobiotics. “Not observed but potentially relevant” are changes that have not been described or observed in dogs; however, the use of these terms has been considered permissible, should a lesion meet the diagnostic criteria. The category “not applicable” refers to lesions and terms specific to rodents, such as chronic progressive nephropathy in the kidney; the use of these terms in dogs is not considered appropriate. It should be kept in mind that the dogs used in toxicology studies are usually of young age and are only on study for a relatively short time (usually not more than 52 weeks), a fraction of the normal life span of dogs. The health status of individual dogs is usually checked carefully, and the individual dogs selected for a study are in excellent condition. For these reasons, spectrum and frequency of changes observed in this population are different from those in diagnostic laboratories, and common age-related lesions are rarely seen. Because neoplasms represent a very rare event in toxicity studies in dogs, neoplasms are generally excluded and only included if they are of relevance, that is, if the finding has been recorded in a toxicity study. For a description and the diagnostic criteria for tumors in dogs, the reader is referred to the fascicles of the International Histological Classification of Tumors of Domestic Animals published by the Armed Forces Institute of Pathology in conjunction with the American Registry of Pathology and the World Health Organization, the textbook Tumors of Domestic Animals, 1 and/or current literature. Whenever possible, the equivalent rodent term/SEND terminology should be used for any tumors not specifically addressed in this manuscript, as appropriate.
In addition to the journal publication, the nomenclature and diagnostic criteria for the dog are also available online. 2 The online version contains additional images and useful links to differential diagnoses characterizing it as a practical tool for diagnostic work. In addition, all INHAND publications are available at the website of the Society of Toxicologic Pathology. 3 Several manuscripts and texts have been published on background lesions in the laboratory beagle, and the reader is directed to the general bibliography at the end for further reading.
The recommended nomenclature is generally descriptive rather than diagnostic based on standard hematoxylin and eosin–stained paraffin-embedded sections only. Histochemical or immunohistochemical staining characteristics may be addressed in the comments section of the respective lesion. Such special techniques may be required in some situations, but a comprehensive discussion of these methods is outside the scope of this publication. Systemic nonproliferative lesions that occur across organs systems and are not specific to an organ are reviewed in the section on systemic pathology. Instead of “synonyms” for each term, as was used in some earlier rodent publications, the nonrodent publications have used the notation “other term(s).” While these synonyms or other terms have been used historically, the primary listed term is the preferred term and will link to the CT in SEND.
Findings included in this nomenclature system may be further specified by modifiers. Criteria are given for modifiers that are of particular relevance. These modifiers should be consistently applied. Additional modifiers not provided in this nomenclature system may describe the location, tissue type, or duration, among others. General principles of the INHAND nomenclature have been published separately. 4 As an excellent resource for the pathology in dogs, see the textbook on Pathology of Domestic Animals, 5 which also provides extensive reference lists. As new information becomes available, new terms will be needed from time to time and a request for this new term will be applied by “change control.” 2,3
2. General (Multisystemic) Pathology
Findings that may be observed in multiple tissues are listed in Table 2.1 with associated diseases and conditions as well as tissues, in which they are usually observed. An associated comment or description is available in this chapter for terminology marked with an asterisk. For description of other terminology, the reader is referred to the chapter of the corresponding organs.
Microscopic Findings of Systemic Pathology (Generally Used Preferred Terms): Dog.
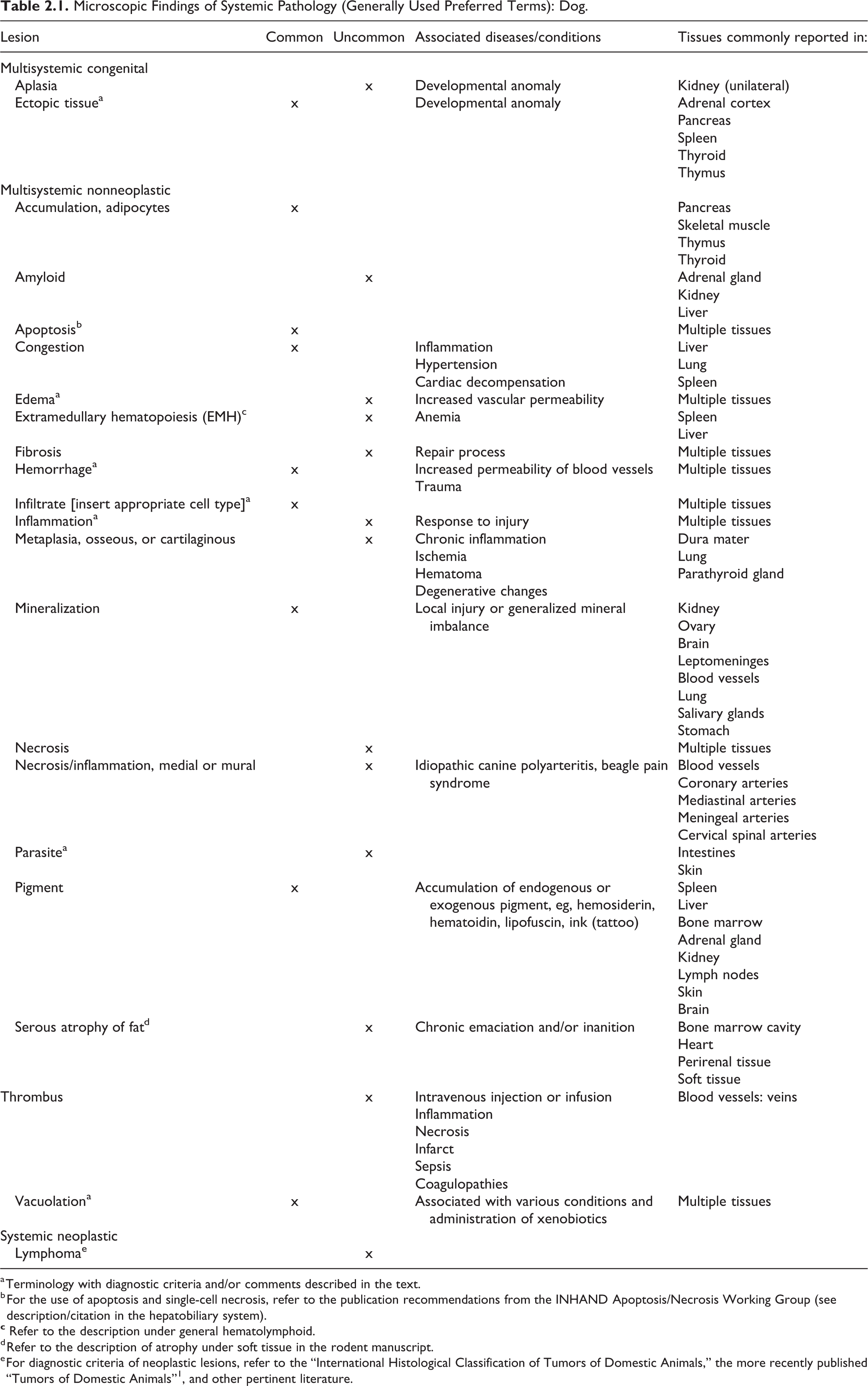
a Terminology with diagnostic criteria and/or comments described in the text.
b For the use of apoptosis and single-cell necrosis, refer to the publication recommendations from the INHAND Apoptosis/Necrosis Working Group (see description/citation in the hepatobiliary system).
d Refer to the description of atrophy under soft tissue in the rodent manuscript.
e For diagnostic criteria of neoplastic lesions, refer to the “International Histological Classification of Tumors of Domestic Animals,” the more recently published “Tumors of Domestic Animals”1, and other pertinent literature.
Ectopic Tissue—Multiple Tissues
Other Term(s)
Accessory (tissue).
Pathogenesis/Cell of Origin
Pathogenesis varies, depending on the originating tissue organ, for example: Shedding off of fragmented adrenal tissue during development Failure of appropriate migration or involution during development (thyroid gland, thymus)
Acquired autoimplantation after trauma or surgery (spleen)
Diagnostic Features
Predominantly normal tissue in an abnormal location.
Comment
In most instances, ectopic tissues are asymptomatic and represent an incidental finding during necropsy or microscopic examination.
Edema—Multiple tissues
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Increased permeability of the vascular system leading to excessive accumulation of vascular fluid in tissues due to increased hydrostatic pressure in venous capillaries while an unchanged colloid–osmotic pressure is maintained (stasis) decreased colloid–osmotic pressure while blood pressure remains normal (hypoalbuminemia) damage to the capillary endothelium (inflammatory, toxic, allergic, hormonal dysregulation) obstruction of lymph drainage a combination of the above (a coincidence of more than one underlying reasons is frequent)
Diagnostic Features
Swelling of the affected tissue; microscopically, the structures appear less dense.
Differential Diagnoses
Inflammation: Presence of inflammatory infiltrates.
Fibrosis: Increase in collagenous or elastic fibers.
Lipoproteinous (alveolar): Lipoproteinous is, unlike edema, characterized by eosinophilic, Periodic acid–Schiff (PAS)-positive exudate.
Comment
Edema is frequently noted in the subcutaneous tissue and the lungs but can occur in any organ and tissue.
Hemorrhage—Multiple Tissues
Other Term(s)
Bleeding
Extravasation
Pathogenesis/Cell of Origin
Extravasation of red blood cells.
Diagnostic Features
Red blood cells in the parenchyma, interstitial space, or body cavity, outside the vascular system.
Differential Diagnoses
Congestion. Accumulation of blood within the vascular system.
Comment
Hemorrhage, bleeding, extravasation, or the escape of blood from the blood vessels may be observed in virtually every organ and tissue and is microscopically characterized by the extravasal presence of erythrocytes in tissues. Usually, the following etiologies are discerned: Trauma Erosion Rupture Diapedesis Asphyxia
For detailed descriptions of the various types, the reader is referred to textbooks of general pathology. Most relevant in toxicologic pathology is hemorrhage by diapedesis, caused by, for example, allergic reactions, infections, toxic agents, thrombocytopenia, disorders of blood coagulation, and so on. Extravasation of red blood cells in areas of exsanguination as part of the euthanasia process is generally not recorded as part of a general toxicology study as this is considered an agonal and procedural finding, unrelated to the objective of the study.
Infiltrate [insert appropriate cell type]—Multiple Tissues
Other Term(s)
Aggregates, inflammatory cell.
Modifier
Eosinophil, (histiocyte), lymphocyte, macrophage (preferred terminology over “histiocyte”), mixed cell, mononuclear cell, neutrophil, plasma cell, polymorphonuclear cell.
Diagnostic Features
Foci of cell infiltrates
Absence of tissue damage
Differential Diagnosis
Inflammation: In addition to the inflammatory cell infiltrates, additional features are edema, tissue damage, hemorrhage, and/or fibrosis.
Comment
Frequently used term to describe the presence of inflammatory cells without significant accompanying inflammation, tissue damage, or morphologic evidence of cellular injury, noted in a variety of organs and tissues. May be within limits of normal. The term should be used with a descriptive modifier of the cell type, for example, infiltrate, lymphohistiocytic.
Inflammation—Multiple Tissues
Other Terms
“-itis,” specific to the organ affected, for example, encephalitis, gastritis, pneumonia, and so on.
Comment
For organ-specific, detailed characteristics, refer to the description in the respective organ section.
Various approaches to further characterize inflammation are used by pathologists, including chronicity (peracute, acute, subacute, etc), location (perivascular, peribiliary, etc), and others. To implement a descriptive terminology, the use of the term “inflammation” is preferred over the “-itis” terminology, and the indication of the predominant cell type(s) in the diagnosis is recommended over the conventional use of chronicity, for example: Inflammation, neutrophilic Inflammation, lymphocytic Inflammation, plasmocytic Inflammation, histiocytic
When applicable the terms are combined, for example: Inflammation, lymphoplasmocytic Inflammation, lymphohistiocytic
Further description by location and distribution is recommended.
Parasite—Multiple Tissues
Comment
For details on the diagnostic criteria of parasites, the reader is referred to textbooks on veterinary parasitology. While parasitic structures can be observed in multiple organs and tissues of domestic dogs, their occurrence in laboratory dogs used in toxicity studies is very limited due to the stringent hygiene conditions in breeding facilities and laboratories. Parasites that may be noted occasionally include the nematode Toxocara canis and the arthropods Demodex canis and Sarcoptes scabiei. When parasites are observed, they should be recorded as “parasite” and, if possible, specified further in a comment to the finding. Parasite may be recorded as “present” or graded depending on the data collection system used in the toxicology study.
Vacuolation—Multiple Tissues
Other Term(s)
Fatty change, lipidosis, lipid accumulation, phospholipidosis, and so on.
Comment
The descriptive term vacuolation is the preferred term over the etiologic terminology listed above. It is considered good practice to add modifiers such as lipid, fatty change, or phospholipidosis if the nature of the vacuoles has been confirmed by special methodology, for example, lipid stain or electron microscopy. In the case of lipid vacuolation, micro- and macrovesicular vacuolation may be also indicated by modifier or comment.
3. Cardiovascular System—Heart, Heart Valves, and Blood Vessels
Introduction
Review of the incidence of spontaneous cardiovascular lesion in dogs has been published, 6,7 and protocol for the collection and dissection of the heart in dogs is available. 8 For detailed general considerations of the cardiovascular system, refer to the INHAND publication on the rodent cardiovascular system. 9
In nonclinical studies, both morphological and functional end points are essential in the identification of any potential interaction between drugs and cardiovascular structures. As a minimum, the standard morphological evaluation of the cardiovascular system includes heart weight measurement and conventional light microscopic histopathology of the myocardium and valvular leaflets in the heart, aorta, and blood vessels within tissues. Evaluation of serum levels of heart-specific biomarkers, electron microscopy, and immunohistochemistry may represent essential investigative tools to better identify the pathological process caused by xenobiotics. Functional assessments, such as blood pressure and echocardiography, can provide valuable in-life information correlating with morphological alterations observed at the postmortem examination.
Additional structures of the conduction system may also be evaluated following careful sampling. 10 The microscopic evaluation of the vascular structure in the other organs is performed as part of evaluation of each specific tissue. This may reveal the presence of either organ-specific vascular changes (possibly part of an organ-specific toxicity) or widespread vascular changes visible in several organs pointing toward a systemic vascular injury.
Heart
Introduction
At necropsy, similar to other lab animal species, the heart is sampled with the root of the large vessels and fixed in buffered formalin solution. The heart is generally opened prior to immersion in the fixative in order to ensure adequate fixation and eliminate large blood clots occupying the cardiac chambers. Conventional light microscopic histopathology of the heart is based on a thorough and consistent microscopic evaluation of hematoxylin and eosin–stained section of all relevant compartments and structures of the heart, including the ventricular, atrial, and septum wall, the valves, and the coronary vessels. This allows an accurate identification of changes in the muscular cells, extracellular matrix, conduction system within the myocardium, and the vascular structures within the myocardium and the adjacent epicardial tissue. Attention should also be given to regional distribution of lesions within the heart. For example, subendocardial zones in the left ventricle and left papillary muscles, especially near insertion site of chordae tendineae, are especially vulnerable to ischemic-based myocardial lesions. 11,12
The recommended terminology of microscopic lesions observed in the heart of dogs is presented in Table 3.1.
Microscopic Findings of the Cardiovascular System: Heart; Dog.

a Finding more frequent as an induced change.
b Terminology addressed in the General Pathology Section.
c Terminology with diagnostic criteria and/or comments described in the text.
d For a comprehensive list of neoplasms and tumor-like lesions, refer to the “International Histological Classification of Tumors of Domestic Animals,” the more recently published “Tumors of Domestic Animals”1, and other pertinent literature.
Hemorrhage—Heart, Heart Valve
Comment
General aspects and a description of hemorrhages are included in the General Pathology section. In the heart, subendocardial, epicardial, and valvular hemorrhages (“heart valve hematomas”) are often observed and readily noted at necropsy. In addition, myocardial hemorrhages may be noted microscopically. It is recommended to indicate the location as descriptor or modifier to the term.
Vacuolation, Conduction System—Heart
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Purkinje fiber.
Diagnostic Features
Round (spherical) empty spaces (vacuoles) usually centrally located in Purkinje fibers.
Differential Diagnoses
Not applicable.
Comment
Vacuolation of the conduction system is occasionally observed in beagle dogs. In a publication on morphologic evaluation of the heart in dogs and monkeys, Keenan and Vidal 7 reported prominent vacuolation of Purkinje fibers in 15% of the males and 12% of the females, while Bodié and Decker 6 reported this change in 34% of the male and 28% of the female control dogs. Authors also noted that vacuolation varied in prominence but was more evident in fibers following vascular adventitia into the ventricles.
Rhabdomyoma, Cardiac—Heart
Other Term(s)
Rhabdomyomatosis; congenital glycogenic tumor.
Pathogenesis/Cell of Origin
Exact histogenesis is uncertain, although conducting fiber, myocardial fiber, and pluripotent embryonic cells are considered to be origin.
Diagnostic Features
Tumor is well circumscribed, expansile, and nonencapsulated.
Solitary or multiply occurrence in myocardium.
Tumor consisted of tightly arranged, ovoid to irregular swollen cells that have distinct cell borders with a deeply eosinophilic cytoplasm and varying degrees of cytoplasmic vacuolation and single, oval to elongate, peripherally located nucleus with 1 or 2 prominent nucleoli.
Tumor cells contain abundant cytoplasmic glycogen and the so-called spider cells occasionally appear.
No mitotic activity.
Differential Diagnoses
Rhabdomyosarcoma: Highly pleomorphic and high mitotic activity with abnormal mitotic figures. Locally infiltrative with frequent distant metastases. Frequently showing necrosis and hemorrhage.
Lipoma: Well-demarcated, lobulated mass. Consist of mature fat cells containing a single fat vacuole with eccentrically located nucleus. Tumors are usually separated into lobules by fibrous septa.
Glycogen storage disease (so-called glycogenosis): No well-circumscribed nodules. This disease is characterized by glycogen accumulation in the heart, skeletal muscles, liver, kidneys, or muscular layer of esophagus.
Special Techniques for Diagnostics
The PAS reaction with or without diastase digestion is applied for the detection of glycogen in neoplastic cells. Immunohistochemically, neoplastic cells are demonstrated positively for desmin and myoglobin but negatively for smooth muscle actin or vimentin.
Comment
Cardiac rhabdomyoma has been reported in various animals including humans and guinea pigs. It is a controversial issue whether cardiac rhabdomyoma is a true neoplasm or hamartoma. Human cardiac rhabdomyomas are caused by mutation in the TSC1 and TSC2 gene. A mouse model of cardiac rhabdomyoma is associated with loss of the TSC-1 gene in ventricular myocytes. 13 One of the most interesting aspects of these tumors is their tendency to undergo spontaneous regression. 14 Rhabdomyoma can occur in the myocardium, skeletal muscles of the larynx, and in the head region in both human and animals.
Heart Valves
Observation of drug-induced valvulopathy in postmarketing surveillance in man has led to withdrawal of some commercialized compounds, such as appetite-suppressant drugs. This has increased the interest in the evaluation of heart valves during preclinical studies.
Histopathological examination of the cardiac valves in all laboratory animal species, including dogs, may suffer from inconsistencies and artifacts related to both the technical approach and the inherent small size of the valves. The trimming/sectioning of the heart may greatly influence the type, number, and correct orientation of valves within the histological sections, which finally affects the accuracy of the microscopic examination. Therefore, in those studies where drug-related effects are suspected to occur in the cardiac valves, the pathologist may need to adapt the preparation of the heart and trimming of valves to ensure the best representation of most or all 4 cardiac valves. Although congenital valvular anomalies (pulmonary stenosis, aortic stenosis) can be observed in dogs, 15 these are very uncommon in beagle dogs purpose-bread for biomedical research and used in toxicity studies.
The recommended terminology of microscopic lesions observed in the heart valves of dogs is presented in Table 3.2.
Microscopic Findings of the Cardiovascular System: Heart Valve; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
Degeneration, Myxomatous, Valve—Heart—Heart Valve
Comment
Myxomatous valvular degeneration is a common valvular lesion in older dogs, affecting chiefly the left atrioventricular valve. 15 Macroscopically, the valve may appear irregular, shortened, thickened, opaque, with discrete to prominent nodules that may extend to chordae tendineae. Microscopically, thickening of the spongiosa by loose fibroblastic glycosaminoglycan-rich tissue and degeneration of the fibrosa with often hyalinized and fragmented collagen fibers are prominent features. The presence of myxomatous tissue at the base of the heart, near the aortic valve and aorta, is a common normal component of the heart of dogs 7 and should not be confused with myxomatous degeneration.
Hemorrhage—Heart—Heart Valve
Comment
General aspects and a description of hemorrhages are included in the General Pathology section. In the heart, subendocardial, epicardial, and valvular hemorrhages (“heart valve hematomas”) are often observed and readily noted at necropsy. In addition, myocardial hemorrhages may be noted microscopically. It is recommended to indicate the location as descriptor or modifier to the term.
Myxoma, Valve—Heart Valve
Species
Dog (term does not exist in rodent goRENI).
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Multipotent vasoformative cells originating in subendocardial layer.
Diagnostic Features
Grossly, multilobular, soft, gelatinous mass; may have hemorrhagic areas; can obstruct the blood flow.
Myxomas are covered by endothelial cells and are composed of an abundant hypocellular myxoid matrix with stellate or globular cells, blood vessels, and smooth muscle cells.
May have capillary-like channels lined by endothelial cells.
Differential Diagnoses
Myxosarcoma: high cellular pleomorphism, presence of numerous mitotic figures, metastasis
Special Techniques for Diagnostics
Histochemical stain such as PAS and Alcian blue stain may be used to confirm the acid mucopolysaccharide nature of the myxoid matrix, and IHC may be used to confirm the presence of endothelial cells and mesenchymal nature (vimentin-positive) of myxoma cells.
Comments
Rare in dogs (common primary cardiac tumor in humans).
Will often disseminate by embolization.
Associated with hemodynamic consequences according to size and anatomical location.
Myxosarcoma, Valve—Heart Valve
Species
Dog (term does not exist in rodent goRENI).
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Multipotent vasoformative cells originating in subendocardial layer.
Diagnostic Features
Grossly, multilobular, soft, gelatinous mass may have hemorrhagic areas; can obstruct the blood flow.
As for the benign form, myxosarcomas are covered by endothelial cells and are composed of an abundant hypocellular myxoid matrix with stellate or globular cells, blood vessels, and smooth muscle cells, with high cellular pleomorphism, numerous mitotic figures, and areas of necrosis. Areas of cartilaginous or osteoid-like differentiation can also be present.
Presence of metastasis in lymph nodes and viscera.
Differential Diagnoses
Myxoma—No cellular pleomorphism, mitotic figures or metastasis.
Special Techniques for Diagnostics
Histochemical stain such as PAS and Alcian blue stain may be used to confirm the acid mucopolysaccharide nature of the myxoid matrix, and IHC may be used to confirm the presence of endothelial cells and mesenchymal nature (vimentin-positive) of myxosarcoma cells.
Comments
This tumor is extremely rare in dogs. As for the benign form, cardiac myxosarcoma may be associated with hemodynamic consequences according to the size of the mass and anatomical location.
Blood Vessels
The microscopic evaluation of the vasculature in organs is generally performed as part of the evaluation of each specific organ. This may reveal the presence of either organ-specific vascular changes (possibly as part of an organ-specific toxicity) or widespread vascular changes visible in several organs pointing toward a systemic vascular injury. Specific collection of larger vessels may be required in particular when intravascular administration of xenobiotics is performed (eg, through bolus injection or slow infusion), with potential for local and/or systemic vascular injury. Vascular injury in dogs, as well as in other laboratory animal species, can occur as a spontaneous pathological change or be drug induced. In preclinical drug development, vascular injury can be caused by a wide array of compounds encompassing small and large molecules (such as monoclonal antibodies) and antisense oligonucleotides. Several papers have been published to better characterize drug-induced vascular injury and associated biomarkers in preclinical studies. 16 –19 However, the distinction between the spontaneous and drug-induced vascular injury may still be challenging and a correct identification of the histological pattern of vascular changes is crucial.
The observation of a vascular injury may reflect an organ-specific toxicity or represent a widespread change visible in several organs and, therefore, suggest the occurrence of a systemic vascular disorder.
Histopathological evaluation of the heart and the other organs according to standard guidelines for organ trimming is considered sufficient to ensure the thorough evaluation of the vascular tree.
The recommended terminology of microscopic lesions observed in the blood vessels of dogs is presented in Table 3.3.
Microscopic Findings of the Cardiovascular System: Blood Vessels; Dog.

a Terminology addressed in the General Pathology section.
b Terminology with diagnostic criteria and/or comments described in the text.
Intramural Plaque, Fatty—Blood Vessel
Other Term(s)
Atheroma, fibrofatty plaque.
Pathogenesis/Cell of Origin
Intimal/medial accumulation of lipids intermixed with inflammatory cells and extracellular matrix. Lesion typically associated with atherosclerosis.
Diagnostic Features
Grossly, affected vessels are enlarged, with thickened, cord-like walls; plaque may protrude into the lumen.
Microscopically, consists of an accumulation of free lipids (may have cholesterol clefts) or intracellular lipids (usually within foamy macrophages) expanding the media and extending into the intima, with disruption of the internal elastic lamina.
Associated with variable amount of inflammatory cells, smooth muscle cells, and extracellular matrix.
Plaque may be mineralized and have a necrotic core.
Infrequently, plaque may be ulcerated and associated with vascular thrombosis and hemorrhage.
Differential Diagnoses
Arteriosclerosis—Thickening of the intima/media by hyaline material (mucopolysaccharides, fibrin) or by laminar proliferation of smooth muscle cells and extracellular matrix, without accumulation of lipids.
Special Techniques for Diagnostics
Intramural fatty plaque may be highlighted with histochemical stains for elastic fibers, fat, and mineralized material and immunohistochemical stain for macrophages and smooth muscle cells.
Comment
Intramural fatty plaques in arteries/arterioles, characteristically associated with atherosclerosis, are rare spontaneous finding in dogs as dogs are resistant to the development of atherosclerosis. However, this lesion is seen infrequently in dogs following developing hypercholesterolemia associated with endocrinopathies.
The intramural fatty plaque in dogs will typically affect the vasculature of the heart, brain, and kidney and less often other vasculatures; deposition of lipids typically begins in the middle and outer layer of the media, eventually extends to the intima and affects more extensively the small muscular arteries. In contrast, intramural fatty plaques in humans are present primarily in the intima and affect primarily the major elastic arteries (aorta, carotid) and the large and medium caliber muscular arteries (coronary, popliteal).
Necrosis/Inflammation, Media or Wall, Artery—Blood Vessel (Figures 3.1 and 3.2)
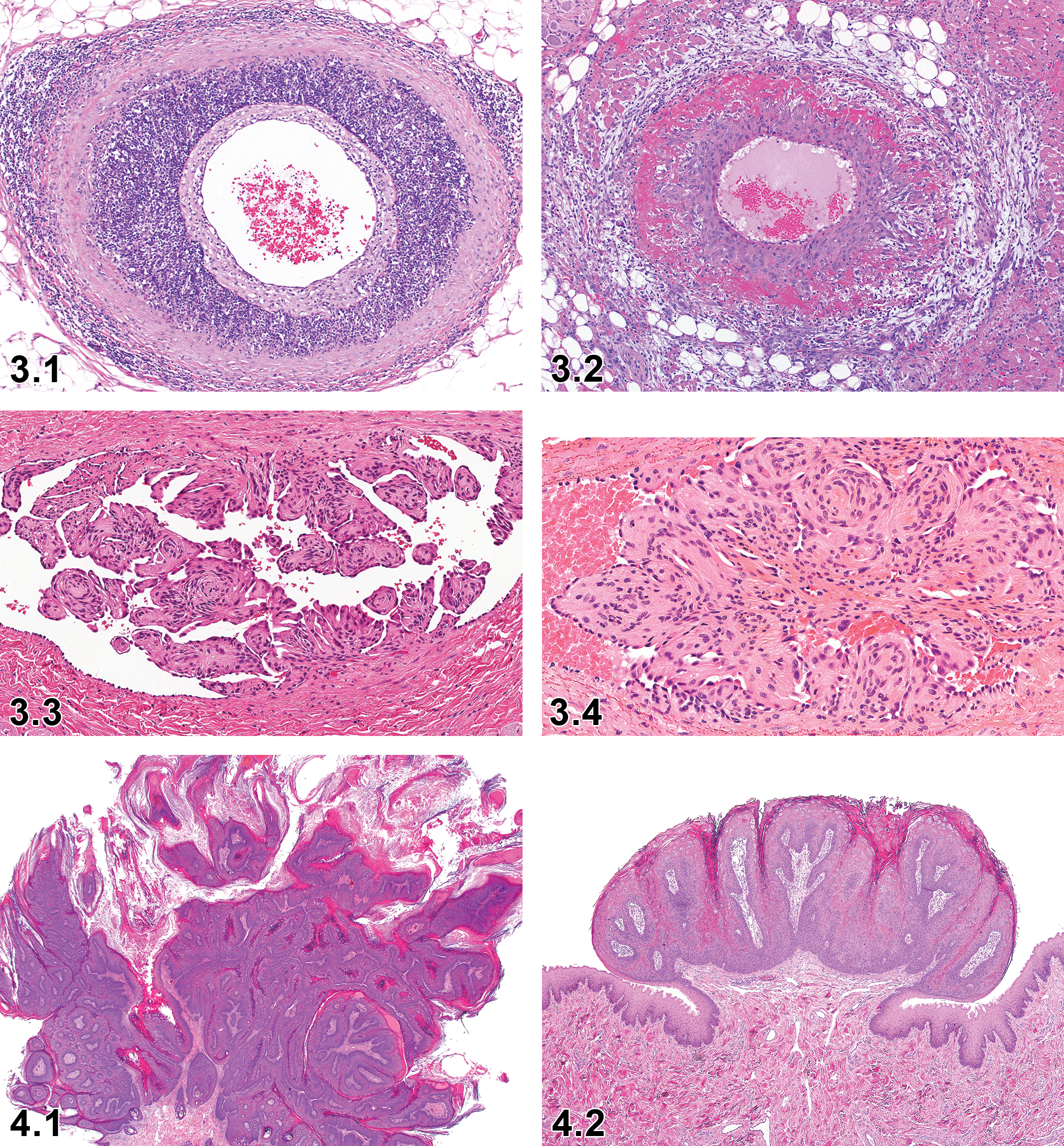
Dog, artery, necrosis/inflammation medial or mural, artery, H&E. Courtesy of Eric van Esch.
Other Term(s)
Beagle pain syndrome, idiopathic canine polyarteritis, canine juvenile polyarteritis syndrome, steroid-responsive meningitis-arteritis.
Pathogenesis/Cell of Origin
Necrotizing polyarteritis; exact etiopathogenesis unknown, a Th2-mediated immune response, and upregulation of CD11a integrin and activation of metalloproteases are suspected.
Diagnostic Features
Acute to chronic necrotizing fibrinoid arteritis affecting the media to the totality of the vascular wall of small to medium size muscular arteries of young beagles and medium to large breed dogs; typically affects the coronary, mediastinal, meningeal, and cervical spinal arteries but can also be observed in arteries of other organs.
Morphological appearance is variable depending on the number of febrile episodes and stage of the disease. Characterized by intimal proliferation, homogenous eosinophilic material and karyorrhectic debris in media, transmural to periarterial infiltrates of predominantly neutrophils with scattered lymphocytes, plasma cells and macrophages, variable degree of hemorrhage, rupture of internal elastic laminae, medial hypertrophy, and fibrosis.
Followed potentially by thrombosis and infarction within affected tissues.
Differential Diagnoses
Hemorrhage, vascular: Presence of extravasated erythrocytes in vascular wall without wall injury.
Inflammation, vascular: Infiltrates of inflammatory cells in vascular wall without evidence of cell injury.
Comment
It is suggested to use necrosis/inflammation, medial or mural, artery to describe necrotizing inflammation in arteries. It can be difficult to differentiate idiopathic canine polyarteritis from drug-induced vascular injury, especially when an increased incidence of chronic lesion consistent with this disease is present following treatment with a vasoactive drug. Vasodilator-induced lesions are generally characterized by medial/adventitial hemorrhage and necrosis with minimal inflammation (acute) to intimal and adventitial proliferative changes (chronic); are typically limited to the coronary arteries (extramural and intramural), often associated with hemodynamic and myocardial changes; and may be associated with atrial hemorrhage. Vasoconstrictor-induced lesions usually affect small size arteries in a variety of tissues and include medial thickening and necrosis, with hyalinization. Hypersensitivity vasculitis (type IV) affects small vessels (not limited to arteries) of skin or of other tissues and is characterized by transmural nonnecrotizing vasculitis with infiltrates of mononuclear and variable number of eosinophils, generally with no fibrinoid necrosis or thrombosis.
The site predilection, dose–response, and clinical and laboratory changes can be differentiating. In drug-induced vascular injury, vascular lesions are generally restricted to coronary arteries, and clinical signs are variable, likely unrelated to vascular effects. Idiopathic canine polyarteritis will usually show prominent transmural to periarterial inflammation with little or no hemorrhage, will typically affect arteries of the heart and of many other tissues as well, myocardial changes will only rarely be present, and idiopathic canine polyarteritis is not associated with hemodynamic changes or atrial hemorrhage. Clinically, acute idiopathic canine polyarteritis is typically associated with pyrexia, anorexia, reluctance to move, cervical rigidity and pain, with elevated polymorphonuclear cell count and concentration of IgA and acute phase proteins in serum and cerebrospinal fluid. In the chronic form of the disease, progressive atrophy of temporal and cervical muscles and neurological deficits may occur.
Hyperplasia, Intravascular Papillary Endothelial—Blood Vessel (Figures 3.3 and 3.4)
Other Term(s)
Reactive vascular proliferation; proliferation, intravascular endothelial; angiomatosis, intravascular.
Pathogenesis/Cell of Origin
Endothelial proliferation in response to traumatic injury followed by thrombosis, inflammation, and stasis within vascular bed.
Diagnostic Features
Papillary formations are totally confined to cystic dilated vascular lumina.
Lesion is covered by a single layer of endothelium.
Fibrous pseudocapsule containing residual smooth muscle of the preexisting vessel wall may be present.
Myriad of small delicate papillae projections into the lumen may be seen.
Inflammatory infiltrates and thrombus may be present.
Proliferating endothelial cells appear prominent or plump but lack significant pleomorphism and mitotic figures.
Differential Diagnoses
Hemangiosarcoma: Proliferation of atypical endothelial cells form vascular channels (capillary to cavernous) and solid cellular masses. Mitotic figures can be common and bizarre. Local invasion and metastases are often present.
Hemangioma: Proliferating endothelial cells show slight cytological abnormalities and form variably sized vascular spaces (capillary to cavernous) and compression of surrounding tissues.
Endothelial hyperplasia: Proliferation of normally present endothelial cells without papillary proliferation.
Special Techniques for Diagnostics
Immunostaining for vimentin, von Willebrand factor, and CD31 are applied for identification of endothelial cells.
Comment
Canine intravascular papillary endothelial hyperplasia has been surveyed by Gamlem and Nordstoga. 20,21 This lesion is thought to represent an abnormal morphologic type of organizing thrombus. Therefore, it should not be confused with preneoplastic or neoplastic lesions. 22 Scrotal vascular hamartoma in dogs may be the same entity of this lesion.
4. Digestive System—Oral Cavity, Salivary Glands, Esophagus, Stomach, Intestines, and Exocrine Pancreas
Introduction
For detailed general considerations on the digestive system, refer to the INHAND rodent publication. 23
This chapter provides a set of standardized terms, diagnostic criteria, and examples for the upper and lower digestive tract, as well as for the salivary glands and the exocrine pancreas of beagle dogs used in toxicologic pathology studies.
The digestive tract is the entry site into the body for orally administered test articles. An irritant test article may lead to local acute lesions at this first site of contact to the body, and the digestive tract can be affected by adverse drug reactions. On the other hand, orally administered test articles usually reach systemic exposure, sufficient to be toxic to other organs, without noticeable effect on the digestive tract.
A distinctive feature of the digestive tract is the high proliferative rate of the epithelium, making it particularly sensitive to agents interfering with cell division but resulting also in a high regenerative capacity. Because of the large surface area of these tissues, accurate assessment of potential treatment effects is almost entirely dependent on a thorough gross examination and sampling of focal lesions.
For the classification of neoplastic lesions, refer to the “International Histological Classification of Tumors of Domestic Animals: Tumors of the Alimentary System of Domestic Animals,” published by the Armed Forces Institute of Pathology in conjunction with the American Registry of Pathology and the World Health Organization, 24 the more recently published “Tumors of Domestic Animals,” 25 and other pertinent literature.
Upper Alimentary Tract (Oral Cavity, Tongue, Pharynx, and Esophagus)
Most commonly, microscopic lesions in the upper alimentary tract are observed in tissues that are required to be examined microscopically, based on regulatory guidelines and the preferences of the laboratories, for example, esophagus, pharynx, tongue. In most instances, other lesions in the oral cavity and pharynx are examined only when gross lesion were recorded. Table 4.1, based on the rodent manuscript, lists the nonproliferative and nonneoplastic proliferative changes of the upper alimentary tract.
Microscopic Findings in the Digestive System: Oral Cavity and Esophagus; Dog.

a This and other malformations are rarely observed in toxicity studies and generally represent gross observations; a microscopic correlate may be required, however.
b Terminology addressed in the General/Systemic Pathology section.
c Finding more frequent as an induced change.
d Terminology with diagnostic criteria and/or comments described in the text.
Mineralization—Oral Cavity
Other Term(s)
Calcinosis circumscripta
Tumoral calcinosis
Ectopic mineralization
Pathogenesis/Cell of Origin
The pathogenesis of calcinosis circumscripta is not known, and a variety of causes have been discussed.
Diagnostic Features
Sharply defined, irregular areas of amorphous, calcified (Kossa positive) material.
Surrounded by epithelioid and multinucleated macrophages.
Peripheral fibrosis.
Differential Diagnoses
Not applicable.
Comment
In the dog, calcinosis circumscripta represents an entity described in text books as a primarily subcutaneous change; it is also observed periarticular and has been described in the tongue of dogs. The lesion is characterized by sharply defined, irregular areas of amorphous, calcified (Kossa positive) material, surrounded by epithelioid and multinucleated macrophages, and peripheral fibrosis. Predisposed are young dogs and large breeds, in particular German shepherd. 24,26,27 The use of the term is not recommended, rather the term “mineralization” should be used instead, and, if considered appropriate, a comment could be added indicating that the finding is consistent with calcinosis circumscripta.
Eosinophilic Granuloma—Oral Cavity
Pathogenesis/Cell of Origin
Eosinophilic granuloma (EG) is suspected to be due to an inherited defect in eosinophil regulation.
Diagnostic Features
Diffuse dermal eosinophilic inflammation.
Foci of degranulating eosinophils.
Mast cells and epithelioid macrophages may be present.
Eosinophilic cell debris surrounding collagen fibers (flame figures).
Acanthosis or ulceration of the overlying epithelium.
Differential Diagnoses
Oral mast cell tumor: Anaplastic mast cells and eosinophils are diffusely present, whereas in the EG, there is a predominance of eosinophils with a few normal mast cells.
Comment
Eosinophilic granulomas are rare in dogs, the features are similar to those observed in cats. Eosinophilic granuloma may be observed at all ages, is more common in dogs younger than 3 years old and in males, and has been noted in various breeds; there is a predilection in Siberian Huskies and cavalier King Charles spaniels. 24,25
Papilloma, Squamous Cell—Oral Cavity (Figures 4.1 and 4.2)
Other Term(s)
Oral papillomatosis.
Pathogenesis/Cell of Origin
Hyperplastic response to viral infection; canine papillomavirus 1.
Diagnostic Features
Verrucous lesion with a thick squamous epithelium and a fibrous stalk.
Marked acanthosis.
Degeneration of epithelial cells in the stratum granulosum and spinosum with clear cytoplasm and condensed nucleus.
Basophilic intranuclear inclusions in the outer spinosum layers.
Regressing lesions show marked lymphoid infiltrates (T cells).
Differential Diagnoses
Not applicable.
Comment
Typically, a lesion of young dogs. Outbreaks of papillomatosis can occur in experimental dog colonies. The papillomas regress spontaneously after 1 to 2 months, and the dog is protected from reinfection through antibody-mediated immunity. 28,29
Stomach
Unlike the rodent, the stomach of the dog consists of glandular mucosa only and is devoid of a nonglandular mucosa; therefore, the terminology for the nonglandular stomach of rodents is not applicable for the dog and was omitted. As in other species, the glandular mucosa of the dog is composed of various glands that are present in specific regions of the organ. To assure a thorough examination of the stomach, examination of representative sections of all regions is mandatory. In the dog, these include the cardia, fundus, and pylorus regions with correspondingly named glands. The recommended nomenclature for nonproliferative and nonneoplastic proliferative lesions in the stomach of beagle dogs used in toxicity studies are listed Table 4.2.
Microscopic Findings in the Digestive System: Stomach; Dog.

a Terminology addressed in the General/Systemic Pathology section.
b Finding more frequent as an induced change.
c Terminology with diagnostic criteria and/or comments described in the text.
Cellularity Increased, Lymphocyte—Stomach (Figure 4.3)
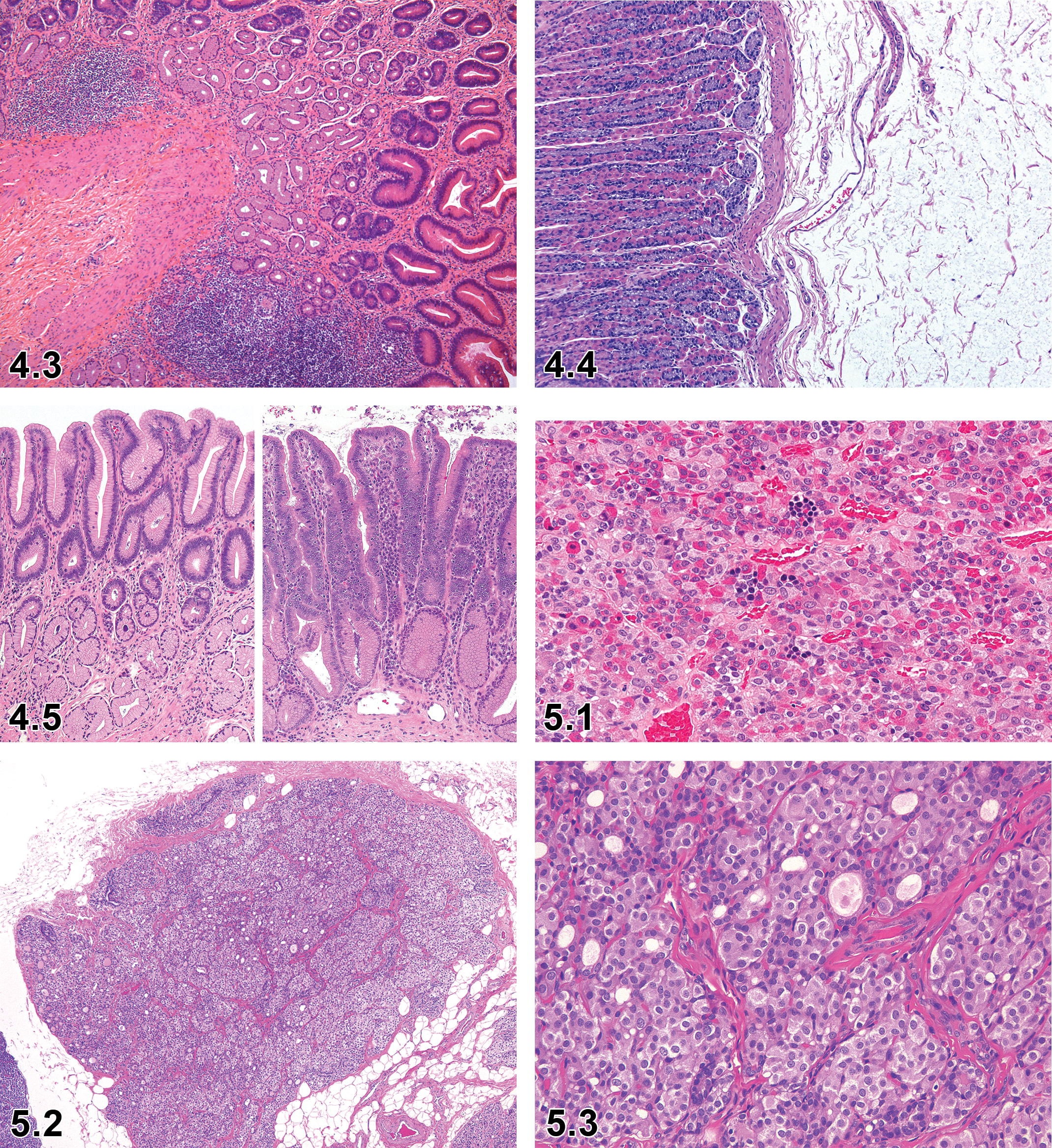
Dog, stomach, pylorus, cellularity increased, lymphocytes, H&E. Courtesy of Dr Klaus Weber, AnaPath GmbH.
Other Term(s)
Hyperplasia, lymphoid follicles
Hyperplasia, lymphocyte
Pathogenesis/Cell of Origin
Increased number of lymphocytes and plasma cells.
Diagnostic Features
Increased size of lymphoid follicles, due to increased number of B- and/or T-lymphocytes, plasma cells, and macrophages.
Increased number of lymphoid follicles.
Increase in the number of lymphocytes.
Differential Diagnoses
Not applicable.
Comment
Increased lymphoid cellularity, increased size of lymphoid nodules, or increased number of lymphoid nodules are rare findings in untreated healthy dogs but occur more frequently in diseased animals and also may be seen frequently as an induced change. It is considered a benign, nonneoplastic, reactive response to an immune stimulus. If the change pertains to lymph follicles, the sublocation may be added.
Edema—Stomach (Figure 4.4)
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Increased permeability of the vascular system leading to excessive accumulation of vascular fluid in tissues (see also General Pathology section).
Diagnostic Features
Swelling of the stomach wall, without increase in inflammatory cell infiltrates or fibrosis.
Differential Diagnoses
Inflammation. Presence of inflammatory infiltrates.
Fibrosis. Increase in collagenous or elastic fibres.
Comment
In the dog, edema of the stomach wall as the only lesion is rare and, if noticed, may be due to incidental intoxication, 30 which is unlikely to occur in a laboratory setting.
Inflammation—Stomach
Other Term(s)
Lymphoplasmacytic gastritis
Scirrhous eosinophilic gastritis
Comment
Variants of inflammation include lymphoplasmacytic gastritis and scirrhous eosinophilic gastritis.
Lymphoplasmacytic gastritis is characterized by a thickening of the stomach wall by lymphocytes and plasma cells infiltrating area of fibrosis. In their investigation of accompanying gastric lesions in dogs with intestinal abnormalities, Lidbury et al 31 found that lymphoplasmacytic gastritis was the most prevalent entity. Scirrhous eosinophilic gastritis is a rare condition in the dog with a focal or multifocal distribution. A concomitant arteritis has been described; an allergic reaction or hypersensitivity was suspected. 32
Hyperplasia, Focal, Mucosa—Stomach (Figure 4.5)
Other Term(s)
Stenosis, pyloric.
Comment
In old dogs, an acquired hyperplastic lesion has been described as pyloric stenosis, characterized by annular thickening of the pyloric region, due to hyperplasia of the glands, granulation tissue in the submucosa, and hypertrophy of the lamina muscularis. 24
In toxicity studies in dogs, hyperplasia of the pylorus region may be observed as a treatment-related finding. 33
Hypertrophy, Chronic, Pylorus—Stomach
Other Term(s)
Giant rugal hypertrophy, giant hypertrophic gastritis, chronic hypertrophic gastritis.
Pathogenesis/Cell of Origin
Gastric glands.
Diagnostic Features
Thickening of large areas of the gastric mucosa by hypertrophic/hyperplastic glands; grossly resembling cerebral gyri.
Lesion may be focal or diffuse.
Secondary folds of muscularis mucosae and submucosa may be present.
Foveolar and glandular hyperplasia.
Progressive loss of parietal cells, replaced by mucous cells.
Cystic dilatation of mucous glands.
Differential Diagnoses
Parietal cell hyperplasia, in association with Zollinger-Ellison syndrome.
Comment
Generally, the change is rare and breed dispositions have been reported, for example, Basenji and Drentse Patrijshond dog. 24,34 There are anecdotal case reports for other breeds and species. The diffuse variant is considered to be analogous to Menetrier disease in humans.
Polyp, Hyperplastic (Regenerative)—Stomach
Pathogenesis/Cell of Origin
Gastric epithelium.
Diagnostic Features
Epithelial lesion that protrudes above the mucosal surface.
Thickened cystic epithelium with a fibrous stalk.
Variable histiocytic or lymphoplasmacytic inflammation.
Epithelial cells are similar to those of the adjacent normal tissue, no dysplasia.
Nuclei are of normal size and staining intensity.
Mitotic rate may be slightly increased; mitoses in the base of crypt.
Differential Diagnoses
Inflammatory polyp: Normal epithelium, covering granulation tissue with various inflammatory infiltrates or lymphocytic foci.
Adenomatous polyp: Tubular or papillary growth, basophilia, loss of nuclear polarity, increased mitotic rate along the whole gland.
Comment
Most commonly, canine stomach polyps are observed in the pylorus region.
Polyp, Inflammatory—Stomach
Other Term(s)
Benign lymphoid polyp.
Diagnostic Features
Normal gastric epithelium
Epithelium covers granulation tissue
Variable inflammatory infiltrates or lymphoid foci with well-differentiated germinal centers.
Differential Diagnoses
Hyperplastic polyp: Thickened cystic epithelium with a fibrous stalk.
Comment
As per Head et al, 24 examples of inflammatory polyps have been described in the dog; in a retrospective analysis, Taulescu et al 35 report 3 cases of inflammatory polyps.
Small and Large Intestine (Duodenum, Jejunum, Ileum, Cecum, Colon, Rectum)
The morphology of the canine intestine is basically similar to that of rodents and humans, but relative size and length of the cecum of dogs are smaller and shorter than the rodent cecum. The ileum communicates only with the colon, and the ileocolic valve is an apparent structure when this region is opened at the gross examination.
Table 4.3 summarizes the suggested nomenclature of nonproliferative and nonneoplastic proliferative lesions in the small and large intestine of the dog.
Microscopic Findings in the Digestive System: Small and Large Intestines; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General/Systemic Pathology section.
c Finding more frequent as an induced change.
Ectopic Tissue—Small Intestines
Modifier
Fundic gland, pancreas.
Pathogenesis
Presence of pancreatic cells or fundic glands in the mucosa or submucosa.
Diagnostic Features
Pancreatic acini with or without islets of Langerhans, in the mucosa or submucosa of intestine, adjacent to the mesenteric attachment.
Comment
Presence of ectopic gastric mucosa with feature of the gastric fundic gland in the small intestine, in addition to ectopic pancreas as described in the rodent manuscript, is reported in Beagle dogs. 36
Dilatation, Crypt—Small and Large Intestine
Other Term(s)
Cyst, mucosa
Dilation, gland
Pathogenesis/Cell of Origin
Unknown.
Diagnostic Features
Marked dilatation of crypt showing oval, round, pear-shaped, or elongated shape.
Lined by intestinal epithelium including goblet cells.
Lumen may contain mucus and cellular and nuclear debris, but inflammatory cells are few.
Differential Diagnoses
Diverticulum: Extension of glands through muscularis mucosae into submucosa.
Comment
Mucosal cysts appear more common in duodenum and jejunum than large intestine and are supposed to be acquired as part of degenerative process. 37
Salivary Glands
Table 4.4 summarizes the suggested nomenclature for nonproliferative and nonneoplastic proliferative changes of the salivary glands.
Microscopic Findings in the Digestive System: Salivary Glands; Dog.

a Terminology addressed in the General/Systemic Pathology section.
b Finding more frequent as an induced change.
c Terminology with diagnostic criteria and/or comments described in the text.
Metaplasia, Necrotizing—Salivary Glands
Other Term(s)
Necrotizing sialometaplasia, salivary gland infarction. 38,39
Pathogenesis/Cell of Origin
Trauma-induced compromise of blood vessels; immune-mediated vascular damage has been discussed.
Diagnostic Features
Ischemic necrosis
Inflammation
Squamous metaplasia
Differential Diagnoses
Not applicable.
Comment
The condition occurs in small breeds (terriers), preferably in the submandibular gland.
Metaplasia, Squamous cell—Salivary Glands
Other Term(s)
Squamous metaplasia; ductal squamous metaplasia.
Pathogenesis/Cell of Origin
Squamous transformation of ductal or acinar epithelium due to vitamin A deficiency.
Diagnostic Features
Squamous epithelium replaces the cubic/cylindrical epithelium of the salivary gland acini and ducts.
Differential Diagnoses
Not applicable.
Comment
The condition occurs in small breeds.
Exocrine Pancreas
Table 4.5 summarizes the suggested nomenclature for nonproliferative and nonneoplastic proliferative lesions in the exocrine pancreas of dogs.
Microscopic Findings in the Digestive System: Pancreas; Dog.

a Terminology addressed in the General/Systemic Pathology section.
b Finding more frequent as an induced change.
c Terminology with diagnostic criteria and/or comments described in the text.
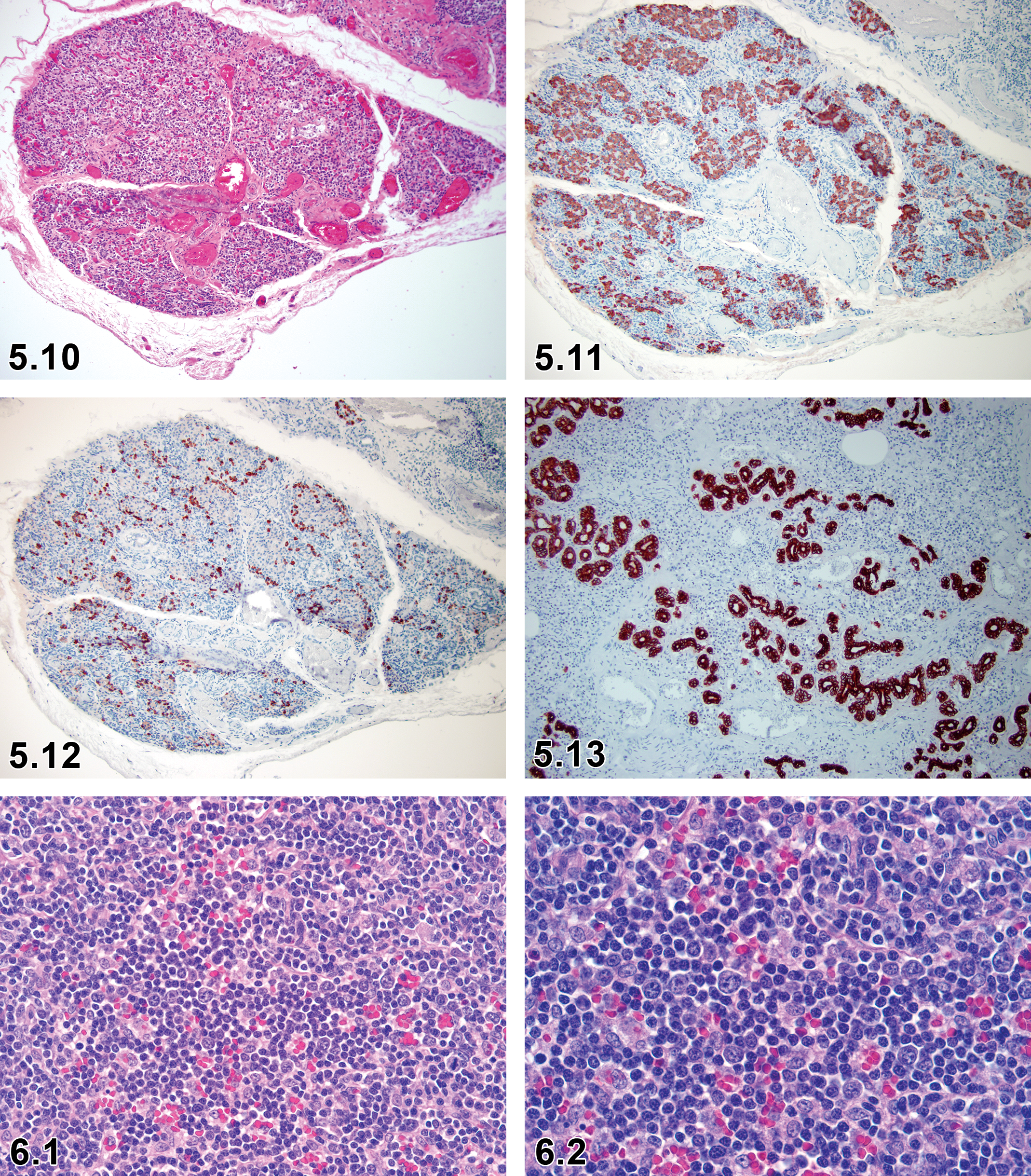
Proliferation, Extra-Islet Endocrine Cell (Nesidioblastosis)—Pancreas (Figures 5.10–5.13)
Comment
Nonneoplastic proliferation of islet and ductular tissue. 40 For a more detailed description, refer to the section on endocrine pancreas in endocrine system.
5. Endocrine System—Pituitary, Pineal Gland, Thyroid Gland, Parathyroid Gland, Adrenal Cortex and Medulla, Endocrine Pancreas
Introduction
This chapter provides a recommended nomenclature for classifying microscopic lesions observed in the endocrine system of the laboratory beagle dog in toxicity studies. The endocrine system is divided into the pituitary, pineal, thyroid, parathyroid, adrenal cortex and medulla and the endocrine pancreas (islets of Langerhans). For detailed general considerations of the endocrine system, refer to the INHAND publication on the rodent.
Pituitary Gland
The pituitary gland (hypophysis) occupies a bony recess (sella turcica) in the basisphenoid bone and is attached to the hypothalamus by the infundibular stalk. The pituitary gland has 2 major compartments including the (1) adenohypophysis (anterior lobe) composed of the pars distalis, pars tuberalis, and pars intermedia and (2) neurohypophysis (posterior lobe) composed of the pars nervosa infundibulum and lobus nervosus. Macroscopically, the adenohypophysis is vascular and soft, whereas the neurohypophysis is pale with the texture of brain tissue.
The adenohypophysis consists of 3 types of endocrine cells identified by routine H&E staining: acidophils, basophils, and chromophobic cells. The frequency of each cell type varies according to the age, sex, and physiological and pathological states. The acidophils have a generally uniform pattern of distribution with increased numbers near the central region and produce growth hormone and/or prolactin. The basophils occur in greater numbers at the periphery of the adenohypophysis and produce adrenocorticotropic hormone (ACTH), thyroid-stimulating hormone, luteinizing hormone, or follicle-stimulating hormone. The chromophobic cells appear in moderate numbers near the central region and produce ACTH. The pituitary also contains supporting cells (folliculostellate cells) and undifferentiated stem cells. The pars tuberalis is composed of epithelioid cells, sinusoids, and occasionally small follicles lined by folliculostellate or endocrine cells. The pars intermedia is located adjacent to the neurohypophysis and is separated from it by a fine layer of vascularized pial connective tissue. The majority of the cells in the pars intermedia are chromophobic cells which produce both ACTH and melanocyte-stimulating hormone. Immunohistochemistry techniques are required to accurately identify the specific cell populations.
The neurohypophysis is joined to the hypothalamus by the infundibular stalk and is composed of nonmyelinated axons and capillaries supported by modified glial cells (pituicytes). The capillaries in the pars nervosa are termination sites for unmyelinated axons, which originate from the hypothalamic neurosecretory neurons. Axons arising from supraoptic and paraventricular nuclei terminate in the pars nervosa. Both oxytocin and vasopressin (antidiuretic hormone) are synthesized in supraoptic and paraventricular nuclei as large precursor molecules, which contain both active hormones and their associated neurophysins. As the biosynthetic precursor molecules travel along the axons in secretion granules from the neurosecretory neurons, the precursors are cleaved into the active hormones.
The recommended terminology of microscopic lesions observed in the pineal gland of dogs is presented in Table 5.1.
Microscopic Findings in the Endocrine System: Pituitary Gland; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
Extramedullary Hematopoiesis: Pituitary Gland (Figure 5.1)
Other Term(s)
Hematopoietic cell proliferation; erythropoiesis; extramedullary erythropoiesis.
Pathogenesis/Cell of Origin
Extramedullary hematopoietic cells.
Diagnostic features
Small clusters of hematopoietic cells are randomly distributed in the interstitium of the pars distalis.
Usually consist of erythroid cells, but granulocytic precursors and/or megakaryocytes are occasionally present.
Not associated with the parenchymal necrosis or degeneration.
Differential Diagnoses
Mononuclear cell infiltrates; lymphocytes and histiocytic cells present alone or in addition to mature myeloid cells.
Focal inflammation: infiltration of leukocytes or lymphocytes associated with cellular necrosis or degeneration.
Pineal Gland
The pineal gland is located on the dorsal midline of the diencephalon and is part of the epithalamus. The gland forms the caudal boundary of the roof of the third ventricle. The pineal gland is mainly composed of epithelioid pinealocytes, neurons, and supporting glial cells. Pinealocytes produce and secrete melatonin and also contain serotonin. The neurons connect to the central nervous system, and the gland is innervated by the sympathetic system. The glial cells are characterized by numerous filaments, and their processes form a glial layer bordering connective tissue spaces. Myelinated and unmyelinated nerve fibers are also present.
The recommended terminology of microscopic lesions observed in the pineal gland of dogs is presented in Table 5.2.
Microscopic Findings in the Endocrine System: Pineal Gland; Dog.

a Terminology addressed in the General Pathology section.
Thyroid Gland
The thyroid gland originates as a thickened plate of epithelium in the ventral oropharynx to form the thyroglossal duct, which extends along the midline to the region of the larynx in the fetus. The paired lobes of the thyroid gland develop from the thyroglossal duct on either side of the larynx and proximal trachea. The ultimobranchial bodies fuse with the thyroid and deliver the C cells (neural crest origin) to each thyroid lobe. Accessory thyroid tissue, which lacks C-cells, is common in the dog and may be located anywhere from the larynx to the diaphragm. Thyroglossal duct cysts may be present in the ventral anterior cervical region and represent a postnatal, persistent portion of the thyroglossal duct.
The major components of the thyroid gland are follicular cells and C-cells (parafollicular cells). The follicular cells form follicles, which contain liquid colloid, the stored form of thyroglobulin. These follicular cells are flattened, cuboidal, or columnar depending on the biosynthetic activity of the gland. C-cells are present in the wall of the follicle or between follicles and form large clusters in the central region of the thyroid lobes. The C-cells predominantly synthesize calcitonin and also smaller quantities of calcitonin gene-related peptide, somatostatin, and bombesin, among other minor hormones.
The recommended terminology of Microscopic lesions observed in the thyroid gland of dogs is presented in Table 5.3.
Microscopic Findings in the Endocrine System: Thyroid Gland; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
C-Cell Complex—Thyroid Gland (Figures 5.2 and 5.3)
Other Term(s)
Special parafollicular cell complex.
Pathogenesis/Cell of Origin
Remnant of cells of ultimobranchial body origin.
Diagnostic Features
Clusters of C-cells in various developmental stages.
Small numbers of undifferentiated epithelial cells, ducts, and cysts are present.
Follicular cells in various stages of differentiation form small follicles with or without colloid.
C-cell complexes can be seen in intra- and extrathyroidal tissue with no fibrous capsule.
Contains follicles in various stages of differentiation with undifferentiated cells, small follicles, with small lacunae and some larger follicles.
Differential Diagnoses
C-cell hyperplasia, focal: Focal proliferation of C-cells and free of other cells including follicular cells and remnants of ultimobranchial bodies.
Special Techniques for Diagnostics
Immunohistochemistry: Positive for vimentin, anti-19S-thyroglobulin, and calcitonin. The immature follicular cells and C cells are positive for vimentin and 19S-thyroglobulin. 41 –43
Comment
Vimentin is expressed by the immature follicular cells derived from the ultimobranchial anlage. The vimentin filaments may participate in thyroglobulin synthesis and folliculogenesis. When the follicular cells accumulate colloid in the follicular lumens, the vimentin immunoreactivity disappears. Typical thyroid follicles have no immunoreactivity for vimentin.
In the dog, the calcitonin-producing C cells are very common and particularly prominent; these cells are regarded as normal and do not have to be recorded in toxicity studies. The C cells are frequently noted in the perithyroidal tissue near the hilus of the thyroid gland and along the main branches of the thyroid artery. C-cell complexes need to be distinguished from C-cell hyperplasia, which should be diagnosed only if there is a significant increase in C-cell numbers throughout each thyroid lobe compared to age-matched controls. To achieve this, both thyroid lobes should be sectioned longitudinally in a consistent manner. 44,45
Accumulation, Adipocyte—Thyroid Gland (Figures 5.4 and 5.5)
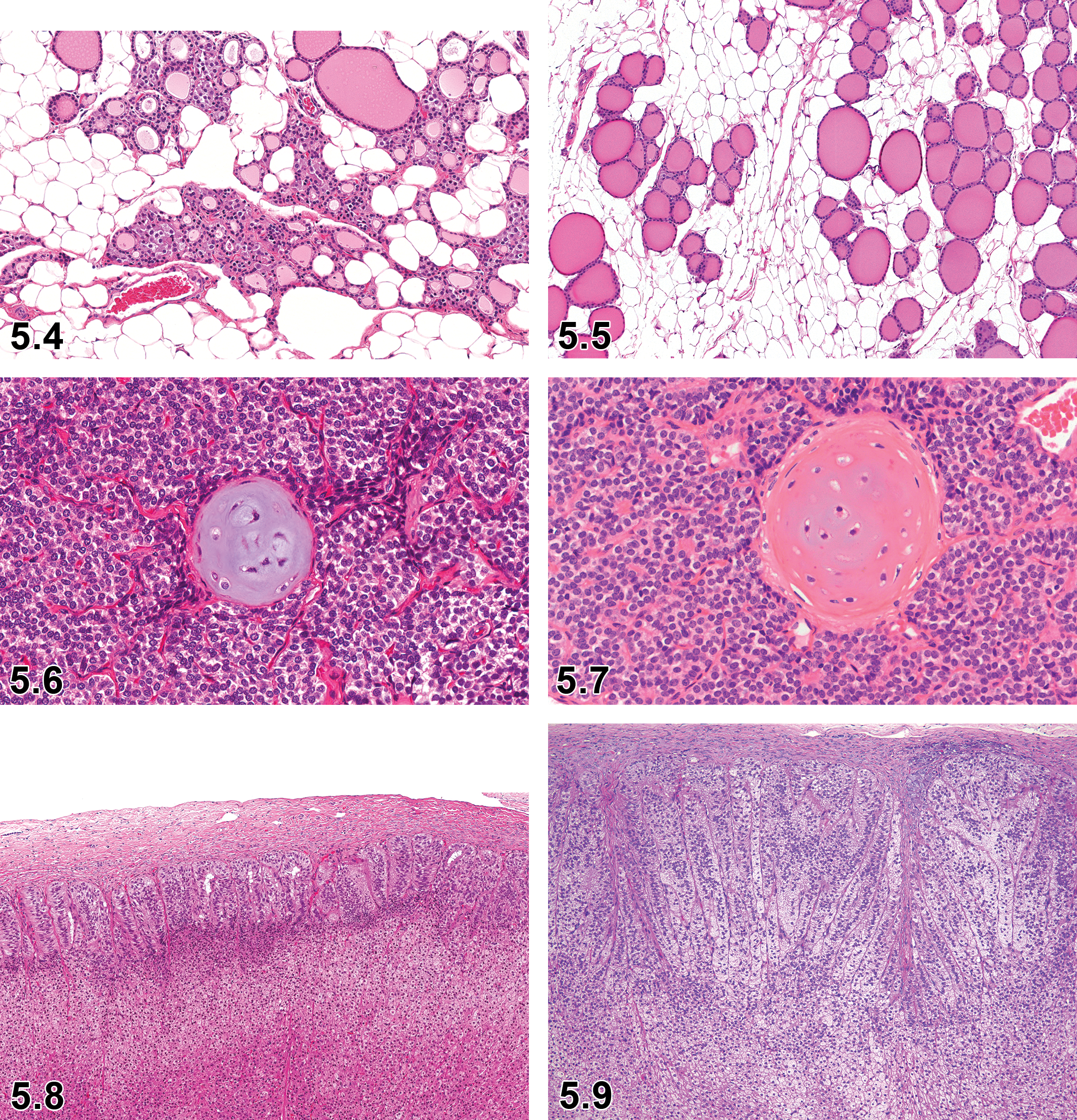
Dog, thyroid gland, adipocyte accumulation, H&E.
Dog, pancreas, extra-islet endocrine cell proliferation, H&E.
Other Term(s)
Fat infiltration, fat replacement, infiltration, adipocyte, lipomatosis.
Pathogenesis/Cell of Origin
Mature adipocytes.
Diagnostic Features
Mature adipose cells present focally or diffusely in the stroma.
No degenerative change is observed.
Number of follicles may be reduced.
Comment
Slight accumulation of adipocytes is recognized as an incidental or background lesion in healthy dogs frequently observed in preclinical studies, but usually not recorded. When the parenchymal tissue is severely or almost completely replaced by adipocytes, the observation should be recorded including indicating the severity.
C-Cell Hyperplasia—Thyroid Gland
Comment
In the dog, the calcitonin-producing C cells are very common and particularly prominent. They are regarded as normal and, therefore, do not necessarily have to be recorded in toxicity studies. They are frequently noted in the perithyroidal tissue near the hilus of the thyroid gland and along the main branches of the thyroid artery. C-cell complexes need to be distinguished from C-cell hyperplasia, which should be diagnosed only if there is a significant increase in C-cell numbers throughout each thyroid lobe compared to age-matched controls. To achieve this, both thyroid lobes should be sectioned longitudinally in a consistent manner. 44,45
The difference between focal C-cell hyperplasia and C-cell adenoma is subjective and arbitrary. As a general guideline, the size of an adenoma should exceed several thyroid follicles; these smaller adenomas will not compress thyroid follicles and not exhibit a capsule. 45
Parathyroid Gland
The 4 parathyroid glands are of endodermal origin derived from the pharyngeal pouches in close association with the primordia of the thymus. The external gland is on the proximal surface of the thyroid lobe, and the internal gland is embedded within the medial lateral thyroid lobe.
The parathyroid glands contain a single type of endocrine cell, namely, the chief cells. The parenchyma of the parathyroid glands consists of densely packed, folded, branching cords of polygonal chief cells separated by a delicate stroma of reticular and collagen fibers with occasional fibrocytes. The cords are usually 1 to 2 cells thick arranged in a trabecular or rarely an acinar arrangement.
The chief cells synthesize and secrete parathyroid hormone (PTH) that regulates systemic calcium homeostasis. The amount of cytoplasm reflects the level of activity of the chief cells. Chief cells with increased synthesis and secretion of PTH are hypertrophied with increased cytoplasm and reduced eosinophilia or vacuolation of the cytoplasm.
The recommended nomenclature for microscopic lesions observed in the parathyroid gland of dogs is presented in Table 5.4.
Microscopic Findings in the Endocrine System: Parathyroid Gland; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
Ectopic Tissue, Cartilage—Parathyroid Gland (Figures 5.6 and 5.7)
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Developmental anomaly.
Diagnostic Features
Mature cartilaginous tissue appears in the interstitium.
No evidence of tissue damage or inflammation.
Differential Diagnoses
Metaplasia: Metaplasia can occur when there is abnormal stimulation of tissue growth, generally due to aberrant wound healing from persistent toxic insult or constant mechanical disruption of the injured area.
Adrenal Gland (Cortex and Medulla)
The adrenal glands are located bilaterally craniomedial to the kidneys. The outline of the left gland is flattened dorsoventrally and oval in the cranial portion and cylindrical in the caudal projection, and that of the right gland has an acute angular bend with its cortex projecting cranially. The adrenal gland is composed of 2 different tissues forming the adrenal cortex and medulla. The cortex originates from mesenchymal cells of the coelomic mesoderm. These cells initially proliferate near the genital ridge and form the fetal cortex and differentiate to form the adult cortex while the fetal cortex regresses. The medulla arises from neural crest ectoderm cells, which migrate from their point of origin into the developing mesodermal mass. The medulla is separated from the cortex by a delicate network of reticular and loose connective tissues.
Cortical zones consist of the zona glomerulosa, zona fasciculata, and zona reticularis. The outermost cortical zone is the zona glomerulosa, and the cells are in arches and clusters that constitute approximately 25% of the adrenal cortex. This zone produces aldosterone which regulates blood pressure and extracellular fluid volume by acting at distal and collecting tubules of the kidney to promote sodium retention and potassium excretion. The zona fasciculata occupies approximately 50% of the cortex, and cells in this zone are arranged in long anastomosing cords or columns, separated by small capillaries. They are responsible for the secretion of glucocorticoid hormones (eg, cortisol), which have diverse homeostatic actions on many organ systems including an increase in glucose production, decreased lipogenesis, and immunosuppression in high concentrations. The innermost cortical zone is the zona reticularis, which has a relatively random and loose network of cells and composes 25% of the cortex and produces sex hormones (androgens and estrogens). The adrenal cortex is dependent on trophic hormones secreted from the pituitary gland.
The adrenal medulla is composed of chromaffin cells, which produce catecholamine hormones, norepinephrine (noradrenaline), and epinephrine (adrenaline). Epinephrine is involved in stress and induces the “fight or flight” response, whereas norepinephrine functions as a major sympathetic neurotransmitter.
The recommended nomenclature for microscopic lesions observed in the adrenal gland of dogs is presented in Table 5.5.
Microscopic Findings in the Endocrine System: Adrenal Gland; Dog.

a Terminology addressed in the General Pathology section.
b Terminology with diagnostic criteria and/or comments described in the text.
Hyperplasia/Hypertrophy, Zona Glomerulosa—Adrenal Cortex (Figures 5.8 and 5.9)
Other Term(s)
Hyperplasia, zona glomerulosa; hypertrophy, zona glomerulosa; thickening, zona glomerulosa.
Pathogenesis/Cell of Origin
Response to endocrine stimulation of the zona glomerulosa.
Diagnostic Features
Diffuse increased thickness of the zona glomerulosa due to increased cell number or hypertrophy of zona glomerulosa cells.
The cells of the zona glomerulosa may contain eosinophilic cytoplasm or may be vacuolated (normal or increased numbers and size of vacuoles).
Cytoplasm of zona glomerulosa cells will be eosinophilic when there are reduced cytoplasmic lipid droplets.
Differential Diagnoses
Not applicable.
Special Techniques for Diagnostics
Morphometrical analysis to determine the thickness of zona glomerulosa.
Ultrastructural examination may be useful.
Comment
Drug-induced thickening of the zona glomerulosa can occur with increased aldosterone secretion and increased number of zona glomerulosa cells. Calcium channel blockers have been reported to induce hypertrophy/hyperplasia of the zona glomerulosa. Increased thickness of the zona glomerulosa can occur in dogs with pulmonary hypertension due to heartworm (Dirofilaria immitis) infestation. 46
Endocrine Pancreas: Islets of Langerhans
The endocrine pancreas originates from the foregut endoderm, and the endocrine cells are arranged in small foci of periductular cells or as aggregates called the islets of Langerhans. The islets distribute throughout the pancreas including the right lobe, body, and left lobe. The body and left lobe have various sized islets, while the right lobe has small islets.
There are 3 major endocrine cell types that produce glucagon (α cells), insulin (β cells), and somatostatin (δ cells). Small number of endocrine cells produce pancreatic polypeptide (PP of F cells), ghrelin, gastrin, or substance P (enterochromaffin cells). These endocrine cells are located in specific regions of the islets. In the body and left lobe, β-cells are distributed in all parts of the islet, and α-cells are scattered in the center or periphery of the islet regardless of the size of the islet. Fewer δ-cells are present in the center or periphery of the islets. In the right lobe, β-cells occupy the majority of the islet with few scattered δ-cells. 47
Table 5.6 contains the recommended nomenclature of microscopic lesions observed in the endocrine pancreas of dogs.
Microscopic Findings in the Endocrine System: Endocrine Pancreas (Islets of Langerhans); Dog.

a Terminology addressed in the General Pathology section.
b Terminology with diagnostic criteria and/or comments described in the text.
Proliferation, Extra-Islet Endocrine Cell—Endocrine Pancreas: Islets of Langerhans (Figures 5.10–5.13)
Other Term(s)
Nesidioblastosis.
Pathogenesis/Cell of Origin
Congenital anomaly or neogenesis of islet cells arising from ductal epithelium.
Diagnostic Features
Nonneoplastic diffuse or disseminated proliferation of pancreatic islets.
Irregular shaped islets, ductuloinsular complexes, and budding of endocrine cells.
Associated adenomatosis may be seen.
Various-sized and irregularly outlined islet cell aggregations that are multifocal or diffuse.
Small clusters of endocrine cells are often distributed around proliferative pancreatic ducts or ductules.
Acinar cells are atrophied, degenerate with vacuolated cytoplasm, irregular-shaped, and have apoptosis adjacent to this lesion.
Severe cases have small pancreatic organ volume.
Differential Diagnoses
Islet cell hyperplasia—Proliferation of islet cells but not exocrine and ductular cells.
Islet cell adenoma—Focally proliferation of islet cells with compression of peripheral tissue and/or encapsulation.
Islet cell carcinoma—Local invasion of capsule and acinar tissue and with proliferation of fibrovascular stroma. Cellular anaplasia and cellular and nuclear pleomorphism are common.
Special Techniques for Diagnostics
Immunohistochemical staining for endocrine cells and duct cells is useful to identify proliferating cells.
Comment
Nesidioblastosis in dogs is a nonneoplastic diffuse or disseminated hyperplasia of pancreatic islets and ductules. In the lesions, the relative numbers of alpha cells is decreased, and there is no significant change in the number of beta and delta cells. Canine nesidioblastosis is not usually demonstrated as a functional disorder of the endocrine pancreas, whereas in humans, it is characterized by persistent hyperinsulinemic hypoglycemia due to defective nonneoplastic proliferation of β-cells and usually occurs in newborns and rarely adults. 48 –50
6. Hematolymphoid System—General Terminology, Bone Marrow, Thymus, Spleen, Lymph Node, and Other Lymphoid Tissues
Introduction
For general considerations on the hematolymphoid system, refer to the rodent manuscript. In addition, guidance on the microscopic pathology of the hematolymphoid system is the subject of an STP Position Paper 51 and is also included in a special issue of Toxicologic Pathology: A Monograph on Histomorphologic Evaluation of Lymphoid Organs, Volume 34 (5); 2006.
This chapter includes microscopic findings of the hematolymphoid system, namely bone marrow, thymus, lymph node, spleen, mucosal associated lymphoid tissue (MALT), other lymphoid structures, that is, tertiary lymphoid structures and serosa-associated lymphoid structures (TLSs and SALSs), and general hematolymphoid changes. The tabulated list is based on the nonneoplastic changes described in the INHAND manuscript for rodents. Detailed descriptions will focus on findings that have unique features in the dog compared with the rodent, while the reader is referred to the rodent document for all other findings. Findings are tabulated with an indication of their prevalence or applicability to vehicle control beagle dogs used in nonclinical toxicology studies. While the rodent manuscript details descriptive, conventional (traditional), and enhanced nomenclature, it is the opinion of nonrodent working group for the dog that more descriptive microscopic findings are generally more appropriate in the dog in general toxicology studies.
General Terminology
General terms that can be used across organs and tissues of the hematolymphoid system are presented Table 6.1.
Microscopic Findings in the Hematolymphoid System: General Terms; Dog.

a Finding more frequent as an induced change.
b Finding addressed in the General Pathology section.
c Terminology with diagnostic criteria and/or comments described in the text.
Erythrophagocytosis—Bone Marrow, Spleen, Lymph Nodes (Figures 6.1–6.3)
Other Term(s)
Erythrophagia.
Pathogenesis/Cell of Origin
Erythrophagocytosis is the main route of normal and pathophysiological clearance of damaged erythrocytes. Resident bone marrow macrophages and red pulp macrophages are essential in this process. 52
Diagnostic Features
Ingestion of red blood cells especially by macrophages.
Differential Diagnoses
Artifact.
Emperipolesis: Active penetration or engulfment of one cell by another which remains intact, 53 whereas phagocytosis includes the degradation of ingested cells or materials.
Comment
Erythrophagocytosis is an important physiologic process to recycle iron from senescent or damaged erythrocytes to extract heme. 54 In diseased animals, the severity of the process may be increased.
Vacuolation, Macrophage—Bone Marrow, Spleen, Lymph Nodes, Thymus, Other Lymphoid Tissues (Figures 6.4–6.5)
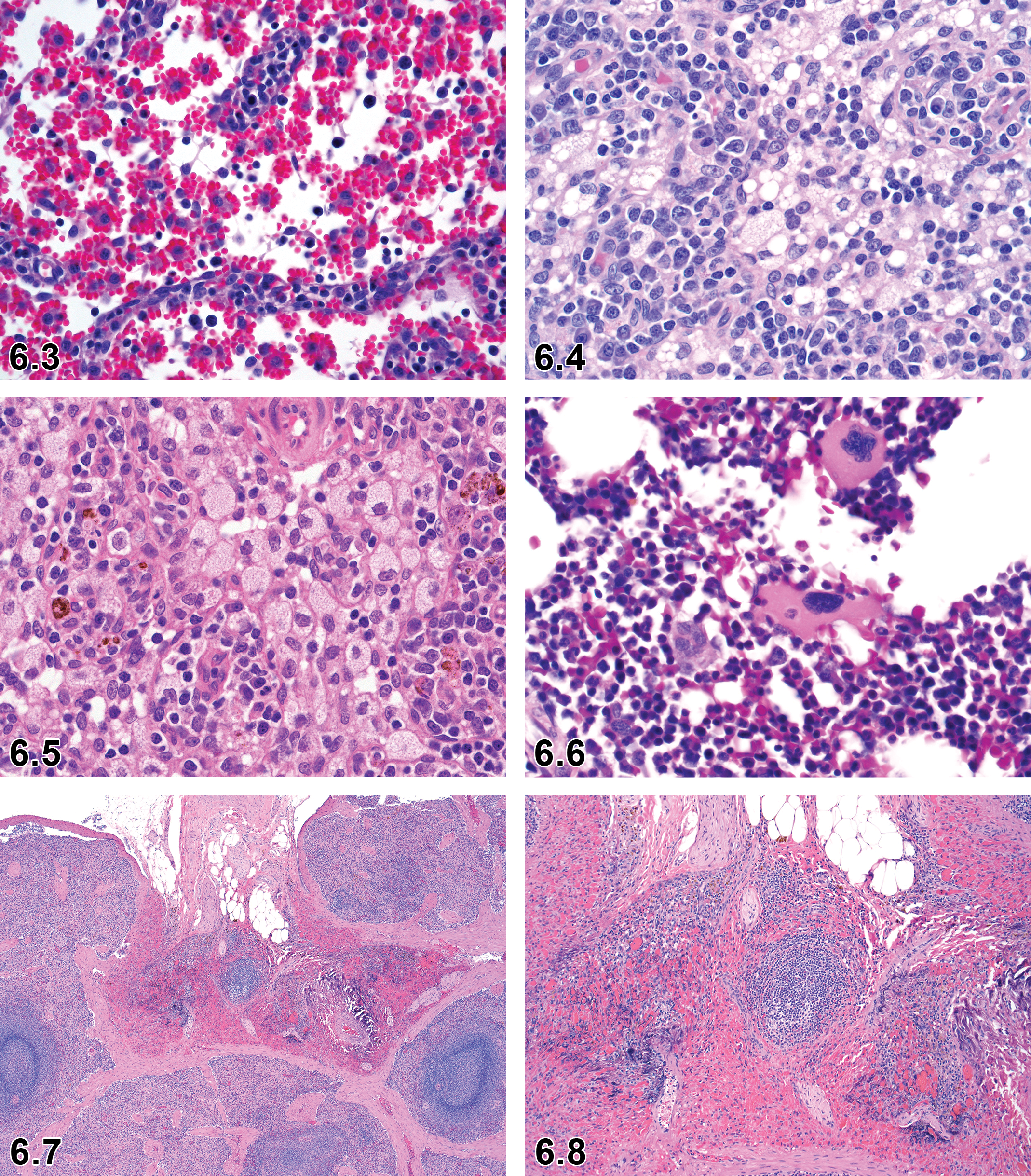
Dog, lymph node, erythrocytosis and erythrophagocytosis, H&E. Courtesy of Eric van Esch.
Other Term(s)
Phospholipidosis.
Comment
The descriptive (general) term “Vacuolation, macrophages” is the preferred term over phospholipidosis. The term phospholipidosis may be used as a comment or modifier but should only be used if verified by other methods, for example, electron microscopy. 55
Bone Marrow
In nonclinical toxicity studies, hematology data in conjunction with microscopic examination of the bone marrow provide an assessment of overall hematopoietic cellularity. If the combined data are inadequate, then cytologic evaluation of the bone marrow smears (from rib or sternum) may be considered for assessing the effect of test articles on the hematopoietic system. 51,56 Other techniques, such as flow cytometric evaluation, clonigenic assays, and electron microscopy, may also be helpful in certain situations. 56
In dogs, bone that contains red marrow, preferably rib, sternum, or vertebrae, is sampled for the microscopic evaluation of the bone marrow. Long bones such as proximal humerus or femur should be avoided because the diaphyseal marrow cavities of these bones may be almost completely replaced by fat. 56
The recommended microscopic terminology for the bone marrow of dogs is presented in Table 6.2.
Microscopic Findings in the Hematolymphoid System: Bone Marrow; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Finding addressed in the General Pathology section.
c Finding more frequent as an induced change.
Emperipolesis—Bone Marrow (Figure 6.6)
Other Term(s)
Engulfment of another cell.
Pathogenesis/Cell of Origin
Megakaryocytes.
Diagnostic Features
Active penetration or engulfment of one cell by another which remains intact. 53
Differential Diagnoses
Phagocytosis: The process of phagocytosis includes the degradation of ingested cells or materials.
Comment
Emperipolesis is often noted in megakaryocytes containing intact neutrophils or other hematopoietic cells. The significance of the finding is unclear as it can be observed in healthy animals but also associated with disease.
Thymus
The thymus should be trimmed transversely through the middle of the organ after fixation to include both lobes. The cortex and medulla should be evaluated separately considering the cortex: medulla ratio average across multiple lobules.
The thymus involutes (shrinks) with age in dogs. The thymus of beagle dogs undergoes progressive involution during the period from 6 to 23 months of age. 57 Therefore, the thymus weights are of low if any significance. Keratinization of Hassall’s corpuscles is prominent in young dogs as in minipigs and nonhuman primates.
The recommended microscopic terminology for the thymus of dogs is presented in Table 6.3.
Microscopic Findings in the Hematolymphoid System: Thymus; Dog.

a Finding addressed in the General Pathology section.
b Finding more frequent as an induced change.
Spleen
Routinely, histopathological evaluation of the spleen involves examination of a single cross-section, which is sufficient to evaluate the red pulp and includes the serosal surface, but white pulp may be not adequately present. A longitudinal section will increase the amount of lymphoid tissue (white pulp) available for assessment. 58 The role of the spleen in dogs is primarily a storage organ, whereas in rodents, it is primarily defensive. 59 Due to the at times massive barbiturate-related congestion in euthanatized dogs, spleen weight is of little value in nonclinical toxicologic pathology studies.
The recommended microscopic terminology for the spleen of dogs is presented in Table 6.4.
Microscopic Findings in the Hematolymphoid System: Spleen; Dog.
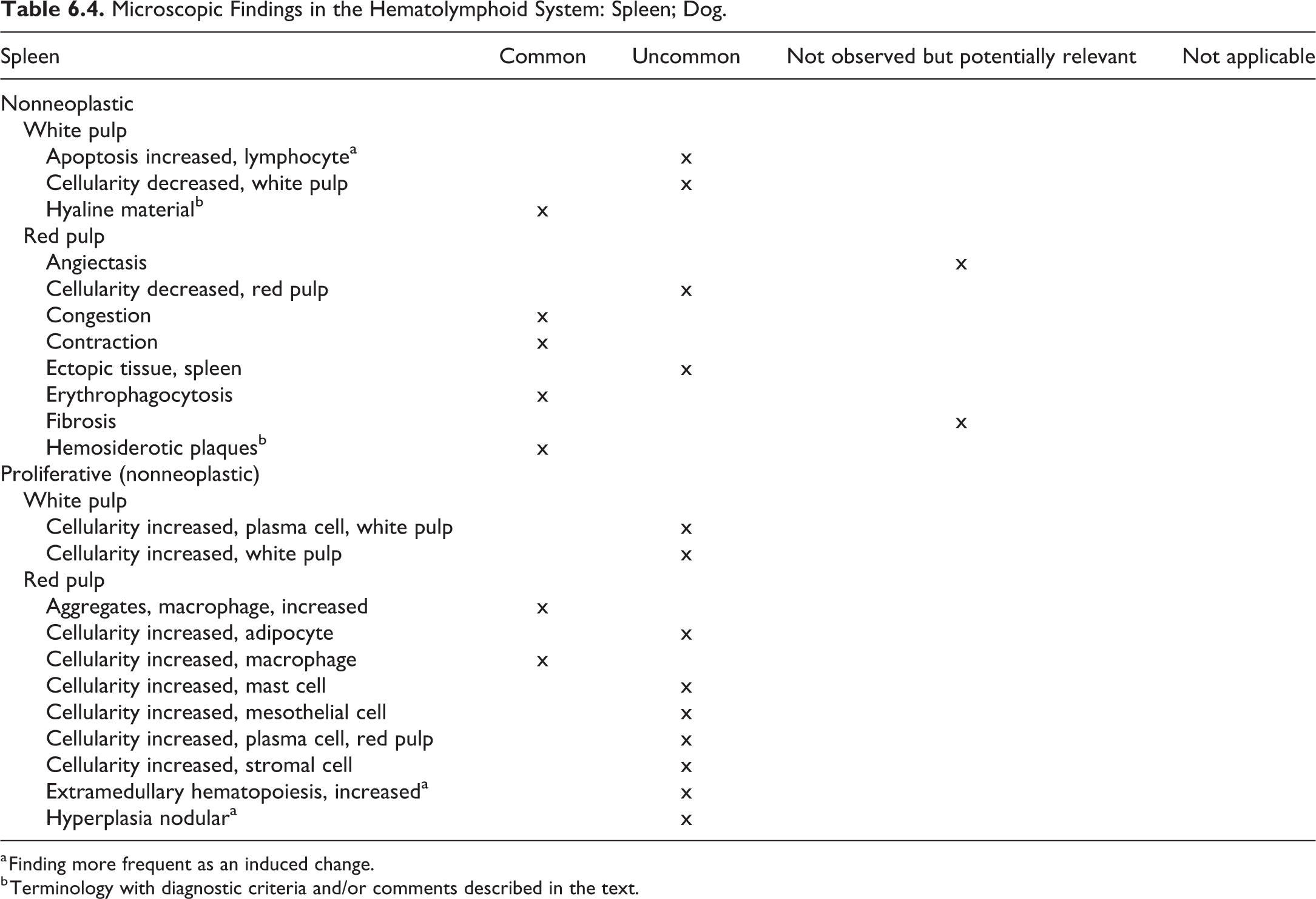
a Finding more frequent as an induced change.
b Terminology with diagnostic criteria and/or comments described in the text.
Hyaline Material—Spleen, Lymph Nodes (Figure 6.9)
Dog, lymph node, hyaline material, H&E.
Other Term(s)
Hyaline deposits.
Diagnostic Features
Hyalinized material is occasionally seen in the follicles of lymph nodes and spleen. 60
Differential Diagnoses
Amyloid deposition.
Immunoglobulin accumulation, probably mediated by immune complexes.
Hemosiderotic Plaque—Spleen (Figures 6.7 and 6.8)
Other Term(s)
Siderotic plaque, siderofibrotic plaque, sidero-calcific plaque, Gamna-Gandy bodies.
Pathogenesis
Likely sequela of prior hemorrhage
Dystrophic change secondary to trauma
Diagnostic Features
Present in the fibrous connective tissue of the capsule, trabeculae, and/or around the hilar vessels and consists chiefly of bilirubin, hemosiderin, and/or calcium deposits. May not be seen macroscopically in young dogs but are recognized macroscopically in older dogs as yellow or grey encrustations or nodules.
Comment
Senile change, no toxicologic significance.
Lymph Node
Typically examined lymph nodes in toxicologic pathology studies include a section of mandibular and mesenteric lymph node, but other nodes may be sampled as indicated by the study design or target biology. Ideally, a standardized midsagittal section of lymph node should be obtained to include all the lymph node compartments, which constitute the follicles and interfollicular cortex, the paracortex (or deep cortex), and the medulla, including medullary cords and medullary sinuses (Table 6.5). 61
Microscopic Findings in the Hematolymphoid System: Lymph Node; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
Hyaline Material—Spleen, Lymph Nodes (Figure 6.9)
Other Term(s)
Hyaline deposits.
Diagnostic Features
Hyalinized material is occasionally seen in the follicles of lymph nodes and spleen. 60
Differential Diagnoses
Amyloid deposition.
Immunoglobulin accumulation, probably mediated by immune complexes.
Mucosa-Associated Lymphoid Tissue
The MALT is usually organized into nonencapsulated accumulations or diffuse collections of lymphocytes scattered along most mucosal surfaces. The MALT, a normal anatomical structure, is considered a secondary immune tissue, which initiates immune responses to specific antigens encountered at or via the mucosal barrier. Following antigen stimulation, lymphocytes forming MALT may proliferate, as is commonly noted with inflammation, but test articles or xenobiotics may also directly stimulate MALT. It would be inappropriate to record normal or stimulated MALT as mononuclear or lymphocytic infiltrates or inflammation. The most common examples of MALT are gastrointestinal-associated lymphoid tissue in the stomach, intestines, and gallbladder; bronchial-associated lymphoid tissue in the lung; nasal-associated lymphoid tissue in the nasal cavity; conjunctiva-associated lymphoid tissue in the eyelids. Less frequently, MALT may be encountered in sections of trachea, larynx, urinary bladder, reproductive tract, and the ducts of various exocrine glands.
Tonsils represent another secondary lymphoid tissue that may be examined in toxicity studies in dogs. In the dog, 3 tonsils are present, the lingual tonsil, the paired palatine tonsil, and the pharyngeal tonsil. 62 For further anatomical and histological details, please refer to anatomical textbooks or the excellent review article by Casteleyn et al. 62
The recommended microscopic terminology for the mucosa-associated lymphoid tissue of dogs is presented in Table 6.6.
Microscopic Findings in the Hematolymphoid System: Mucosa-Associated Lymphoid Tissue (MALT); Dog.
a Finding more frequent as an induced change.
Other Lymphoid Tissues (TLSs and SALSs)
For the definition and function of TLSs and SALSs, the reader is referred to the rodent manuscript. Usually, these structures are examined microscopically in toxicologic pathology studies when they are included in the section of the associated tissue (like MALTs).
The recommended microscopic terminology for other lymphoid tissues of dogs is presented in Table 6.7.
Microscopic Findings in the Hematolymphoid System: Other Lymphoid Tissues (Tertiary Lymphoid Structures and Serosa-Associated Lymphoid Structures); Dog.

7. Hepatobiliary System—Liver and Gall Bladder
Introduction
The liver is a common target organ for chemical-induced toxicity in all species, including dogs, because most drug exposure occurs via the oral route and the liver receives the absorbed chemical directly via the portal vein. The liver metabolizes (biotransforms) many of these substances, termed xenobiotics, for excretion as water-soluble compounds. During biotransformation, intermediate compounds can pose a risk of cellular injury to hepatocytes and biliary epithelium.
Liver
Liver pathology has been covered in detail in the rodent INHAND document 63 as well as in other publications. 64,65 This section provides recommended terminology for the hepatobiliary system in the laboratory beagle dog during preclinical toxicity studies with diagnostic criteria and histology images included only for nomenclature where there are distinguishing features of the process that differ between the dog and the rodent.
In all organs and tissues, including those of the hepatobiliary system, tumors are very rarely observed in toxicologic pathology studies in beagle dogs and, therefore, are generally not included in this manuscript. For the nomenclature and diagnostic criteria of the neoplastic changes, refer to the chapter on hepatobiliary neoplasms in the textbook of Tumors of Domestic Animals. 66,67
Anatomy
A dog liver consists of 4 lobes; the left lobe divided into the left lateral lobe and left medial lobe; the right lobe divided into the right lateral lobe and right medial lobe; the quadrate lobe; and the caudate lobe divided into the papillary process and caudate process. The liver is supplied with blood from the portal vein and the hepatic artery.
The extrahepatic bile duct system includes the hepatic, cystic, and common bile ducts and the gallbladder, which is situated between the left medial and the quadrate liver lobes.
Histology
Histology of dog hepatobiliary system is generally similar to rodents and has been described. 63 A distinct histologic feature of the dog liver is a spiral smooth muscle that surrounds the hepatic veins starting at the larger central veins and persisting in sublobular and larger veins until the hepatic vein is formed. This muscle can constrict in response to various endogenous and xenobiotic mediators and alter blood flow from the liver. 68,69
Physiology
Physiology of dog hepatobiliary system is generally similar to rodents and has been described. This functional heterogeneity across the lobule is characterized by greater drug metabolism via the cytochrome P450 enzymes in the centrilobular regions of the liver. Periportal regions, supplied with the most oxygenated blood, are generally more active in synthetic activities. The zonation develops as a result of the oxygen gradient across the acinus or the gradient of hormones and nutrients.
The recommended microscopic terminology for the liver of dogs is presented in Table 7.1.
Microscopic Findings in the Hepatobiliary System: Liver; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
c Finding more frequent as an induced change.
d For criteria and description of erythrophagocytosis, refer to the Hematolymphoid System/General Terms.
e For the recommended use of infiltrate, refer to Infiltrate [insert appropriate cell type] in the section General/Multisystemic Pathology.
Congenital Vascular Anomaly—Liver (Figure 7.1)
Other Term(s)
Microvascular dysplasia.
Pathogenesis/Cell of Origin
Abnormal development of portal vasculature.
Diagnostic Features
Congenital portosystemic shunt: Anomalous vascular connection between portal vein and systemic venous system, abnormal portal tracts with small or absent portal vein, proliferated arterioles, and hepatocellular atrophy.
Portal vein hypoplasia: Similar changes in the portal tract, but no shunt vessel is present. Portal vein hypertension with ascites is possible.
Differential Diagnoses
Portal vein thrombosis.
Comment
Congenital vascular anomalies are occasionally observed and are generally macroscopic findings made at necropsy, except for primary hypoplasia of the portal vein, which does not involve a shunt vessel and requires histologic confirmation. 70 Affected animals have small livers, and histologic features in these dogs include portal vein hypoperfusion, small or absent portal veins, proliferated hepatic arterioles, and hepatocyte atrophy. Primary hypoplasia of the portal vein resembles portosystemic shunts histologically, but affected animals often have portal hypertension and resultant ascites. In the absence of a diagnosed shunt and particularly in the presence of portal hypertension, the most likely diagnosis is portal vein hypoplasia.
Ductal plate anomaly—Liver (Figures 7.2 and 7.3)
Other Term(s)
Ductal plate malformation, juvenile polycystic disease/congenital hepatic fibrosis, congenital dilatation of the large and segmental bile ducts (macroscopic observation).
Pathogenesis/Cell of Origin
Persistence or aberrant remodeling of the embryonic ductal plate.
Diagnostic Features
Extensive bands of portal bridging fibrosis containing numerous small to dilated irregular tortuous bile ducts.
Hypoplastic or absent portal veins, compensatory arteriolar proliferation.
No nodular regeneration, only minimal inflammation.
Differential Diagnoses
Congenital vascular disorders.
Chronic biliary obstruction or inflammation.
Special Techniques for Diagnostics
Cytokeratin 7 or 19 to identify biliary epithelium.
Comment
Apoptosis—Liver
Other Term(s)
Programmed cell death.
Pathogenesis/Cell of Origin
Mitochondrial or intrinsic pathway.
Death receptor or extrinsic pathway.
Perforin/granzyme pathway.
Diagnostic Features
Cytoplasmic and nuclear condensation.
Nuclear fragmentation.
Intact plasma membrane until late in the process.
Lack of inflammation.
Tingible body macrophages may be present.
Differential Diagnoses
Single-cell necrosis: Single-cell necrosis results from acute cellular injury and is characterized by cell and nuclear swelling, karyolysis, karyorrhexis, karyopyknosis, pale eosinophilic cytoplasm, cytoplasmic vacuoles, loss of cellular detail, cellular debris, and inflammation.
Comment
A comprehensive description of the processes involved and a guidance on which terminology to use is provided by the publication of the INHAND Apoptosis/Necrosis Working Group. 72 In brief:
Use necrosis and apoptosis as separate diagnostic terms.
Use modifiers to denote the distribution of necrosis (eg, necrosis, single cell; necrosis, focal; necrosis, diffuse; etc).
Use the combined term apoptosis/single-cell necrosis when
there is no requirement or need to split the processes, or
when the nature of cell death cannot be determined with certainty, or
when both processes are present together.
The diagnosis should be based primarily on the morphological features in H&E-stained sections. When needed, additional, special techniques to identify and characterize apoptosis can also be used.
Bile Plug—Liver (Figure 7.4)
Other Term(s)
Cholestasis, icterus, pigment, pigment deposition.
Pathogenesis/Cell of Origin
Obstruction of bile flow, hepatocellular injury.
Diagnostic Features
Green-brown bile pigment, primarily bilirubin, in hepatic bile canaliculi.
Pigment may appear in Kupffer cells (after rupture of bile canaliculi).
Differential Diagnoses
Other pigments, including hemosiderin, lipofuscin, porphyrin, hematoxylin, and formaldehyde: A comprehensive discussion of differentiation is included in the rodent manuscript under pigmentation. 63
Proof of bile pigment can be achieved by special stains (eg, iodine test or Hall test).
Intranuclear Brick Inclusions—Liver (Figure 7.5)
Other Term(s)
Acidophilic crystalline intranuclear inclusions, paracrystalline intranuclear inclusions.
Pathogenesis/Cell of Origin
The genesis is unknown.
Diagnostic Features
Crystalline, rectangular to rhomboid brightly eosinophilic homogenous bodies.
Size is up to 15 µm long.
Nuclear membrane occasionally distorted by the inclusions.
No morphological evidence suggesting cytoplasmic degeneration.
Ultrastructurally, a basic periodic structure is observed in the crystal that appears to be composed of helical filaments. No structural abnormality is noted in the cytoplasm, nucleus, or nuclear membrane in the cells containing these crystals.
Differential Diagnoses
Viral inclusion bodies.
Heavy metal inclusions.
Special Techniques for Diagnostics
Brick inclusions are positive with acid-fast staining 73,74 ; it can be confirmed by electron microscopy.
Comments
Brick inclusions are occasionally observed in young beagle dogs using toxicological studies, although it commonly appears in older dogs. There are no evidences of biological or pathologic significance. Similar inclusions occur in the proximal tubular epithelial cells in kidneys in dogs. There are no evidences relating to heavy metal intoxication nor viral infection.
Inflammation—Liver
Other Term(s)
Hepatitis.
Comment
Diagnostic term “inflammation” should be used carefully. The term inflammation is the recommended, preferred term over hepatitis or any etiologic terminology; the etiology, when determined, may be added as a comment or descriptor to the finding. The type of inflammation is usually added as a modifier and preferably should include the predominant cell type (eg, neutrophilic, lymphoplasmocytic, histiocytic, etc). Indicating the chronicity as a modifier (peracute, acute, subacute, chronic) is acceptable. Traditionally, hepatic inflammatory responses have been classified as acute, subacute, chronic, granulomatous, and so on. These terms are somewhat interpretative, lack precise definitions, vary depending upon study duration, usually do not consist of a singular cell type, and do not have exclusive pathognomonic features. A more descriptive approach is recommended and can be qualified by lesion distribution or use of subclassification and discretionary qualifiers.
Rarefaction—Liver (Figure 7.6 and 7.7)
Dog, liver, cytoplasmic rarefaction, H&E.
Other Term(s)
Cytoplasmic rarefaction, excess glycogen accumulation, ballooning degeneration, corticosteroid hepatopathy.
Pathogenesis/Cell of Origin
Excessive endogenous or exogenous corticosteroid exposure leads to significant accumulation of cytoplasmic glycogen beyond that seen in fed dogs, most likely due to a disturbed balance between glycogenesis and glycogenolysis. 75 –77
Diagnostic Features
Enlarged hepatocytes (ballooned) with a clear cytoplasm and well-defined eosinophilic cytoplasmic membrane, appearing less densely stained, with fine strands of cytoplasm (accumulation of glycogen).
Increased sinusoidal neutrophil accumulation.
The change appears panlobular with prominence of midlobular and centrilobular zones.
Differential Diagnoses
Storage disease.
Vacuolation: Well-defined, single or multiple rounded vacuole(s).
Special Techniques for Diagnostics
To detect glycogen, PAS staining with diastase digestion is routinely applied using tissues which are fixed with alcoholic fixative or in frozen sections.
Comment
Steroid hepatopathy is limited to dogs primarily, although rabbits and cats are reported to have a similar, but less pronounced response to corticosteroids. Postprandial and toxicity-associated glycogen accumulation have also been described in rodents and marmosets. 75 Endogenous steroid excess is the result of Cushing disease or syndrome, due to neoplasia of the pars distalis of the pituitary gland or functional neoplasms of the adrenal cortex. This finding is uncommon as background in laboratory dogs used in preclinical studies. Administration of compounds with direct or indirect corticosteroid activity can also produce this lesion. If deemed appropriate or necessary, qualifiers such as increased or decreased may be added or the grade of rarefaction can be recorded. Because glycogen is frequently evident in hepatocytes, it is typically only recorded if a drug-related effect is suspected.
Vacuolation, Hepatocytes—Liver (Figures 7.8–7.10)
Other Term(s)
Fatty change, steatosis, lipidosis, lipid accumulation; phospholipidosis.
Pathogenesis/Cell of Origin
Excessive accumulation of vacuoles (generally lipids) in the cytoplasm
Diagnostic Features
Macrovesicular: Hepatocytes contain a large well-defined single rounded vacuole within each cell. Nucleus and cytoplasm displaced to the periphery. A few hepatocytes may contain one or more smaller vacuoles.
Microvesicular: Hepatocytes partially or completely filled with numerous small lipid vacuoles. Affected hepatocytes may have a “foamy” appearance. Small vacuoles do not normally displace the nucleus to the periphery.
Differential Diagnoses
Cytoplasmic rarefaction.
Special Techniques for Diagnostics
Frozen sections of unfixed or fixed tissue can be stained for fat (oil red O, Sudan IV) and are helpful in recognizing microvesicular lipidosis and in differentiating fat from other substances that may accumulate in vacuoles.
Comment
The descriptive term vacuolation is the preferred term over the etiologic terminology listed above (Other Terms).
It is considered good practice to add modifiers such as lipid or fatty change, if the nature of the vacuoles has been confirmed by special methodology, for example, lipid stain. In the case of lipid vacuolation, micro- and macrovesicular vacuolation may be also indicated by modifier or comment; other qualifiers and comments may be added (eg, location).
It is recommended to diagnose phospholipidosis, characterized by multiple irregular to round clear membrane-bound cytoplasmic vacuolation, as vacuolation and indicate phospholipidosis as modifier, descriptor, or comment. Verification by electron microscopy or immunohistochemical (IHC) staining (LAMP-2) 78 is required (Figure 7.6C).
Adenoma, Hepatocellular—Liver (Figure 7.11)
Comment
A rare benign tumor in the liver of beagle dogs, which has been observed in toxicologic pathology studies. The neoplasm is included in the INHAND rodent manuscript of the hepatobiliary system. 63 For the diagnostic criteria in dogs of this and other neoplastic findings, readers are encouraged to refer the chapter on hepatobiliary neoplasms in the textbook Tumors of Domestic Animals 66 and/or current literature.
Gallbladder and Biliary Tree
Table 7.2 summarizes the recommended nomenclature for gall bladder changes that can be observed in dogs used in preclinical toxicological studies.
Microscopic Findings in the Hepatobiliary System: Gallbladder and Biliary Tree; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
Ectopic Tissue, Pancreas—Gallbladder (Figure 7.12)
Dog, gallbladder, ectopic tissue, pancreas, H&E. Courtesy of Charlotte Maria Dalsgaard.
Comment
For a detailed description of the ectopic pancreas tissue in the gallbladder, refer to the rodent manuscript (Ectopic Tissue, Pancreas). To the authors’ knowledge, there is only one reference available that describes this lesion in the dog, specifically in a Siberian Husky. 79 A case of ectopic pancreatic tissue in a beagle dog has been brought to the authors’ attention; refer to the image (Figure 7.12).
Hemorrhage—Gallbladder
Other Term(s)
Bleeding
Extravasation
Pathogenesis/Cell of Origin
Extravasation of red blood cells.
Diagnostic Features
Red blood cells outside of the vascular system in the parenchyma, interstitial space, or cavity of the hollow organ.
Differential Diagnoses
Congestion. Accumulation of blood within the vascular system.
Comment
Hemorrhage, bleeding, extravasation, or the escape of blood from the blood vessels may be observed in virtually every organ and tissue and is microscopically characterized by the extravasal presence of erythrocytes in tissues. For the types of hemorrhages observed, refer to the comment under Hemorrhage in the General Pathology section. The exact location of the hemorrhage (eg, “submucosa”) may be added as a modifier, locator, or comment, depending on the data acquisition system and the pathologist’s preference.
Infarct—Gallbladder (Figure 7.13)
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Occlusion of the cystic artery.
Diagnostic Features
Transmural coagulative necrosis Fibrin thrombi
Differential Diagnoses
Gall bladder mucocele may occur concurrently or infarction may occur as a single entity.
Special Techniques for Diagnostics
Phosphotungstic acid hematoxylin staining may help identify arterial thrombi.
Comment
The pathogenesis of arterial thrombosis is not known. 80
Mucocele—Gallbladder (Figure 7.14)
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Etiology is unknown, may be related to decreased gallbladder motility, bile stasis, altered bile composition, and viscosity.
Diagnostic Features
Dilatation with accumulation of mucoid secretion.
Mucosal proliferation with projecting epithelial fronds.
Mucus-filled cysts.
Necrosis and rupture of the wall may ensue.
Differential Diagnoses
Cystic mucinous hyperplasia of the gall bladder mucosa.
Extrahepatic biliary obstruction.
Special Techniques for Diagnostics
The PAS may help highlight the mucus in the lumen of the gall bladder.
Comment
This condition may cause extrahepatic biliary obstruction. 81,82
Hyperplasia, Cystic Mucinous—Gall Bladder (Figure 7.15)
Other Term(s)
Cystic hyperplasia, cystic mucinous hypertrophy, papillary adenomatous hypertrophy.
Pathogenesis/Cell of Origin
Unknown.
Diagnostic Features
Macroscopically, gallbladder is enlarged and consisted of thickened wall with numerous multilocular, gelatinous translucent cysts.
Thickened mucosa by numerous fronds and cysts.
Mucosal membrane consists of high columnar epithelial cells containing and secreting abundant mucinous materials.
Little or no inflammatory infiltrate.
Differential Diagnoses
Gallbladder mucocele.
Special Techniques for Diagnostics
Not applicable.
Comments
Diagnostic overlap with gallbladder mucocele as some cases may have elements of cystic mucinous hyperplasia as well as excessive production of abnormal mucus. Regarded as an age-related disorder, so very uncommon in the laboratory beagles generally used in preclinical toxicology studies. This finding may be associated with exposure to progestational agents. 83,84
8. Integumentary System (Skin)
Introduction
This section provides a proposed standardized nomenclature for classifying lesions observed in the skin and appendages of dogs, namely epidermis, dermis, hair follicles, sebaceous glands, apocrine (epitrichial) glands, eccrine (atrichial) glands, and nailbed using standard H&E-stained paraffin sections only. Lesions in the mammary gland are covered separately. Dermal lesions, which were covered in the soft tissue, nervous system, or cardiovascular system INHAND rodent manuscripts, are also covered separately, under those respective systems. The nomenclature of skin lesions in the dog differs from the rodent most strikingly in the classification of neoplasms. The authors recommend the use of the WHO nomenclature for domestic animals or the more recently published text titled “Tumors of Domestic Animals”1 for reference if a skin neoplasm is identified in chemical or pharmacological toxicity studies.
In this section, only notable differences between dogs and rodents will be addressed, as the anatomy of dog skin is comparable to that of other mammals. For detailed general considerations on the integument, please refer to the INHAND publication on rodent integument, 85 and, for a detailed description of the anatomy and physiology of canine skin, refer to Muller and Kirk’s Small Animal Dermatology. 86 Surveys of skin lesions in beagle dogs are included in the publications of Sato et al 49 and Scudamore. 60 Parasitic diseases in the dog include flea infestations (Ctenocephalides canis and others), sarcoptic mange (Sarcoptes scabiei), and demodectic mange (Demodex canis). For details on the diagnostic criteria of parasites, the reader is referred to textbooks on veterinary parasitology. In laboratory settings of toxicologic pathology studies, parasitic diseases of the skin are very rare. When parasites are observed, they should be recorded as “parasite” and, if possible, specified further in a comment to the finding. Also refer to the General/Multisystemic Pathology section.
In general, the thickness of dog skin ranges from 0.5 to 5.0 mm, with the thickest areas on the head, dorsum of the neck, back, and sacrum. 86 In a recent study, the thickness of beagle dog skin was referred with 1.3 to 1.4 mm at different measurement sites. 87 The epidermis of normal haired skin varies from 0.1 to 0.5 mm with 2 to 3 nucleated keratinocyte layers. 88 Rete ridges are not found in the normal haired dog skin but may be observed at the nasal planum, footpads, and scrotum. The epidermal surface varies from undulating at the densely haired skin of the back to heavily folded at the skin of the abdomen. 88 The surface of footpads is papillated. Beside keratinocytes, the epidermis comprises several other cell types, for example, melanocytes, Langerhans cells and Merkel cells. 89 The melanocytes are located in the basal layer of the epidermis, in the outer root sheath of hair follicles, in the hair matrix, and in ducts of sebaceous and sweat glands. Single melanocytes can also be found in the superficial dermis. Long dendritic processes of melanocytes extend between keratinocytes and transfer pigment-containing melanosomes to keratinocytes. 86
The canine dermis does not usually have dermal papillae. The dermis is sparsely populated with cells, but there can be several mast cells around superficial dermal blood vessels. 86
The beagle dog, most commonly used in preclinical safety studies, has a short coat composed of primary (outercoat) and secondary (undercoat) hair. Canine hair follicles are arranged in clusters of several primary hairs surrounded by groups of smaller secondary hairs. Each primary hair is accompanied by sebaceous glands, apocrine sweat glands, and arrector pili muscles, whereas secondary hairs may have sebaceous glands. 90 Sebaceous glands and apocrine sweat glands are distributed throughout the haired skin, whereas eccrine sweat glands are limited to the footpads. Besides these adnexal glands, dogs have specialized glands including circumanal glands, glands of external ear canal, the anal sac gland, and the tail gland. Circumanal glands and tail gland (supracaudal gland) are composed of hepatoid cells, which can also be found in the prepuce. Anal sac glands are composed of abundant large sebaceous glands and apocrine sweat glands, whereas the glands of the external ear canal (ceruminous glands) are apocrine glands. Meibomian glands are modified sebaceous glands. 86
Table 8.1 summarizes the microscopic lesions that can be observed in the skin of beagle dogs in toxicological safety studies.
Microscopic Findings in the Integument: Skin; Dog.
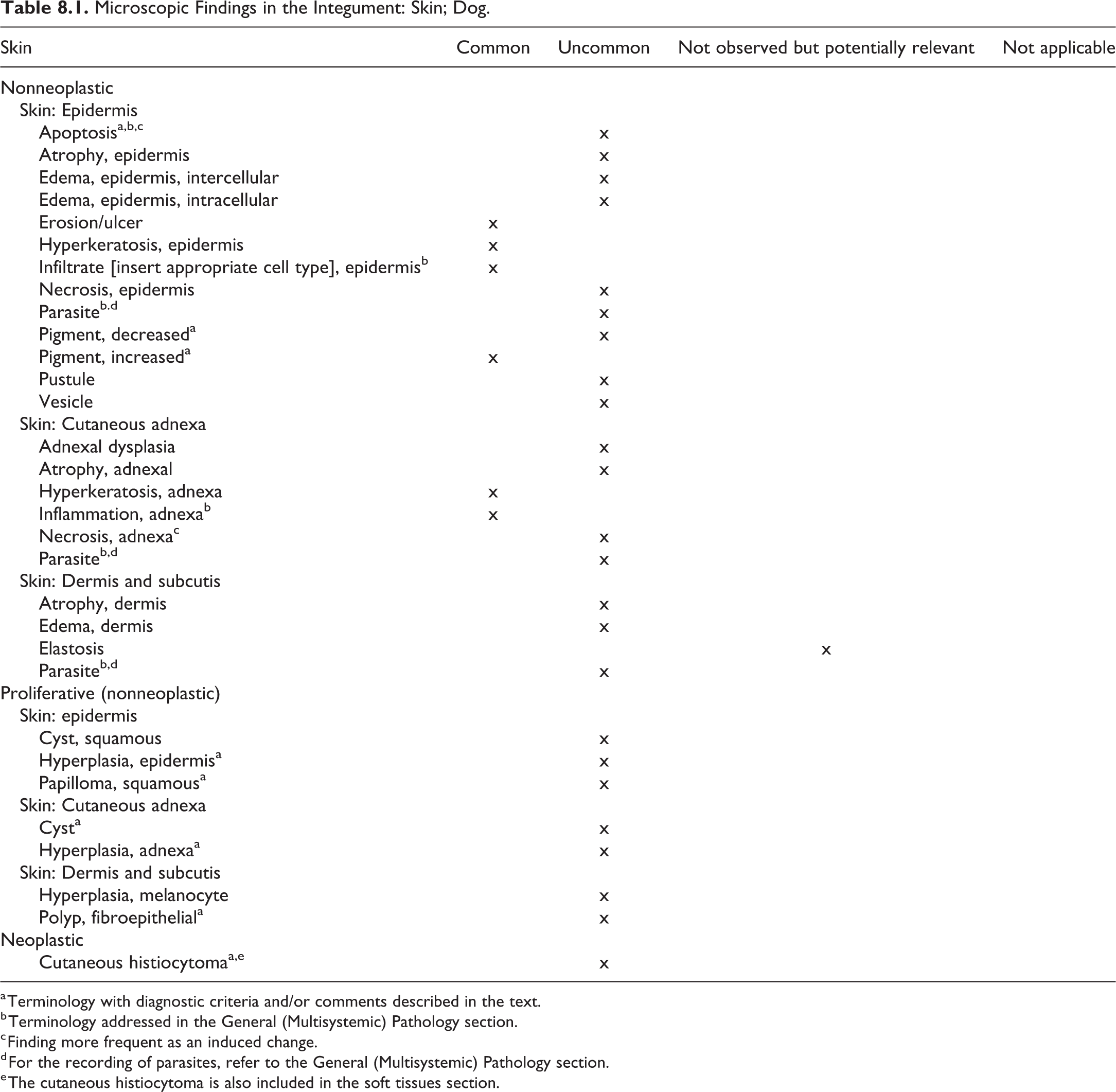
a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General (Multisystemic) Pathology section.
c Finding more frequent as an induced change.
d For the recording of parasites, refer to the General (Multisystemic) Pathology section.
e The cutaneous histiocytoma is also included in the soft tissues section.
Apoptosis—Skin
Other Term(s)
Programmed cell death.
Pathogenesis/Cell of Origin
Mitochondrial or intrinsic pathway
Death receptor or extrinsic pathway
Perforin/granzyme pathway
Diagnostic Features
Cytoplasmic and nuclear condensation
Nuclear fragmentation
Intact plasma membrane until late in the process
Lack of inflammation
Tingible body macrophages may be present
Differential Diagnoses
Single-cell necrosis: Single-cell necrosis results from acute cellular injury and is characterized by cell and nuclear swelling, karyolysis, karyorrhexis, karyopyknosis, pale eosinophilic cytoplasm, cytoplasmic vacuoles, loss of cellular detail, cellular debris, and inflammation.
Comment
A comprehensive description of the processes involved and a guidance, which terminology to use is provided by the publication of the INHAND Apoptosis/Necrosis Working Group 72 . In brief:
Use necrosis and apoptosis as separate diagnostic terms.
Use modifiers to denote the distribution of necrosis (eg, necrosis, single cell; necrosis, focal; necrosis, diffuse; etc.).
Use the combined term apoptosis/single-cell necrosis when
there is no requirement or need to split the processes, or
when the nature of cell death cannot be determined with certainty, or
when both processes are present together.
The diagnosis should be based primarily on the morphological features in H&E-stained sections. When needed, additional, special techniques to identify and characterize apoptosis can also be used.
Pigment Decreased—Skin
Other Term(s)
Hypopigmentation, hypomelanosis.
Pathogenesis/Cell of Origin
Decreased pigmentation reflects lower amounts of melanin pigment in the epidermis either microscopically or macroscopically (loss of skin or hair pigmentation). While it may be associated with congenital or acquired idiopathic defects on melanization in some dog breeds, in toxicologic pathology, most often it is the result of the effects of certain chemicals (eg, monobenzyl ether of dihydroquinone) on melanocytes, inflammatory disorders that affect melanization or destroy melanocytes, hormonal disorders, or dermatoses featuring hydropic degeneration of basal keratinocytes. In dermatoses associated with hydropic degeneration of basal cells and subsequent hypopigmentation, the underlying superficial dermis usually reveals pigmentary incontinence characterized by increased size and/or number of melanosomes. The mechanism of pigmentary incontinence is unclear but could include epidermal injury and melanin release to the superficial dermis through a damaged basement membrane.
Diagnostic Features
Decreased or absent pigment in a normally pigmented epidermis.
Melanin clumps and/or increased size and/or number of melanophages in the underlying dermis (pigmentary incontinence).
Comments
Decreased pigmentation may be associated with pigmentary incontinence. In general, pigmentary incontinence is not recorded separately in toxicity studies, but may be described as a component of a distinct lesion with melanin release to the superficial dermis.
Pigment Increased—Skin
Other Term(s)
Hyperpigmentation, melanosis.
Pathogenesis/Cell of Origin
Increased deposition of melanin within the epidermis. Skin pigmentation is affected by local inflammation because melanocytes respond to inflammatory mediators by increasing or decreasing melanogenesis and by altering melanin transfer to keratinocytes.
Diagnostic Features
Deposition of excessive amounts of melanin within the epidermis.
It may be confined to the stratum basale or present throughout all epidermal layers.
Increased melanin may be present within keratinocytes and/or dermis (within melanophages).
Comments
Hyperpigmentation is a common, nonspecific finding that can be observed in many skin diseases associated with chronic inflammation, numerous hormonal dermatoses, as well as some developmental and neoplastic disorders. In general, this finding is not described or recorded separately in toxicity studies but may be described as a component of a complex inflammatory finding or may be recorded when describing a chronic or pronounced change. Increased pigment may be observed in the hormone responsive skin of dogs that have been administered the dopaminergic and prolactin-inhibiting agent, bromocriptine. 91,92
Hyperplasia, Epidermis—Skin
Comment
Epithelial hamartoma (pigmented epidermal nevus, linear epidermal hamartoma, verrucous epidermal nevus), Cutaneous horn (papillomatous hyperplasia), and Seborrheic keratosis are considered entities to be summarized under epidermal hyperplasia. A comment may be added to the diagnosis to further specify the lesion.
Epidermal hamartomas are congenital or acquired malformations that are commonly recognized only later in life. In the dog, association with papilloma virus has been described in cases. Inflamed linear epidermal nevus is a very rare lesion in dogs. The lesion is characterized by focal irregular epidermal papillary hyperplasia, laminated hyperkeratosis, and keratohyalin granules and may be present in the stratum granulosum and may be hyperpigmented. The inflamed linear epidermal nevus is characterized by prominent parakeratosis, neutrophilic pustules, and variable dermal inflammation.
The spontaneous cutaneous horn is a very rare lesion in beagle dogs. It may occur at any age and has been observed in association with other hyperplastic or neoplastic skin entities. Microscopically, the change is characterized by an exophytic cylindrical formation of compact keratin, focally hyperplastic epidermis, extensive, compact, laminated, ortho- or parakeratotic hyperkeratosis. On the surface, the stratum corneum forms columns.
Seborrheic keratosis is a benign, acanthotic, and often pigmented epidermal proliferation; other features include an exophytic or slightly endophytic growth pattern, monomorphic and basaloid appearance of proliferating keratinocytes, and multifocal keratin accumulations that may form small horn cysts, and hyperpigmentation due to numerous melanin granules distributed throughout proliferating keratinocytes. The lesion is rarely diagnosed in the dog. So far, no breed, sex, or site predilection was recognized. The lesion seems to occur more frequently in older dogs
Squamous Papilloma—Skin
Other Term(s)
Idiopathic squamous papilloma, warts.
Pathogenesis/Cell of Origin
Originates from squamous epithelium of the epidermis.
Diagnostic Features
Papillary mass
Hyperplastic stratified squamous epithelium
Supporting dermal stroma
Differential Diagnoses
Cutaneous horn
Cutaneous exophytic papilloma: canine papillomavirus (CPV) induced neoplastic lesion and pedunculated or cauliflower-like circumscribed alopecic hyperkeratotic mass, observed mainly on the head, eyelids, or paws. Often regresses spontaneously. Histopathologically, proliferating keratinocytes show a variable viral cytopathic effect and intranuclear inclusion bodies.
Cutaneous inverted papilloma: Canine papillomavirus induced, single or multiple, raised, firm neoplasm with a central keratin-filled pore; cup-shaped appearance. Occurs most commonly on the abdomen or groin, also on limbs and digits. Does not regress spontaneously. Histopathologically, it may be cup shaped with a central core of keratin. Keratinocytes may show a viral cytopathic effect.
Cyst, Adnexa—Skin
Comment
A number of cystic lesions with hyperplastic properties have been described in the skin of dogs. Usually, their terminology reflects the origin and/or their features. Examples are:
Infundibular cyst (epidermoid cyst, epidermal cyst, epidermal inclusion cyst)
Isthmus cyst
Panfollicular cyst (Trichoepitheliomatous cyst)
Dermoid cysts
Sebaceous duct cyst
Subungual epithelial inclusion cyst
Apocrine cyst (apocrine cystomatosis)
Ceruminous cyst
Ciliated cyst
Comedone, pressure point
These conditions are generally rarely observed in toxicologic pathology studies. Should these lesions occur, it is recommended to diagnose them as cyst with a descriptive modifier. For the diagnostic criteria, refer to the section Epithelial and Melanocytic Tumors of the Skin by Goldschmidt and Goldschmidt 93 in the textbook “Tumors of Domestic Animals”, to “Integumentary System” by Mauldin and Kennedy 27 in Jubb, Kennedy, and Palmer’s Veterinary Pathology, Volume I, the WHO fascicle Histological Classification of Epithelial and Melanocytic Tumors of the Skin of Domestic Animals, 94 and/or pertinent literature.
Hyperplasia, Adnexa—Skin
Comment
Apocrine (sweat gland) hamartomas, fibroadnexal hamartomas, follicular hamartomas, and sebaceous hamartomas are considered entities to be summarized under adnexal hyperplasia. The lesions originate from apocrine, follicular, and/or sebaceous epithelial cells. Microscopically, they are characterized by an accumulation of disorganized but mature specialized cells or tissues indigenous to the particular site. The lesions represent proliferative lesions of hair follicles, sebaceous glands and ducts, apocrine glands, or pilosebaceous units. A comment may be added to the diagnosis to further specify the lesion.
Apocrine (sweat gland) hamartomas are rare but have been described in dogs; they are proliferating sweat glands in the superficial dermis and/or panniculus; apocrine glands are larger than normal, ectatic and lined by attenuated epithelium; the overlaying epidermis may be hyperplastic.
Fibroadnexal hamartomas (adnexal nevus, focal adnexal dysplasia, folliculosebaceous hamartoma) occur most commonly on the distal limbs, pressure points, and in interdigital areas. The lesions may be congenital or acquired following trauma to the area. Microscopic features include hyperplastic, distorted, and dilated hair follicles; no connection to the skin surface and no hair bulbs; surrounded by dense, mature collagen; and variable inflammation.
Follicular hamartomas (follicular nevus) are rare lesions that occur more frequently in younger dogs. Predisposed sites include the limbs and neck. Microscopically, the lesions consist of clusters of very large anagen hair follicles that extend more deeply than adjacent normal follicles. In the majority of cases, hair follicles within a cluster are uniform and have the appearance of primary follicles; associated glands may be enlarged. The lesions are surrounded by hyalinized collagen, and secondary folliculitis may occur.
Sebaceous hamartomas are regarded as an uncommon variant of the fibroadnexal hamartoma characterized by an intradermal proliferation of predominantly large sebaceous glands and ducts. They are located in the mid to deep dermis without involvement of superficial dermis; hair follicles are small and malformed; the lesions are surrounded by mature collagen.
Polyp, Fibroepithelial—Skin
Other Term(s)
Cutaneous tag, skin tag, acrochordon, fibrovascular papilloma.
Pathogenesis/Cell of Origin
Proliferative fibrovascular lesion
Diagnostic Features
Mature dermal collagenous tissue that can be highly vascularized.
No adnexal structures within the lesion but may be hyperplastic at the base of the polyp.
Covered by an irregular, hyperplastic, hyperkeratotic, and hyperpigmented epidermis.
Few amounts of inflammatory cells.
Secondary ulceration and neutrophilic inflammation due to trauma may occur.
Comment
Fibroepithelial polyps are fibrovascular lesions that rather represent a proliferative response to trauma or inflammation than actual neoplasms. They occur mostly on the trunk, sternum, and pressure points of the limbs and may be solitary or multiple.
Cutaneous histiocytoma—Skin (Figures 8.1–8.3)
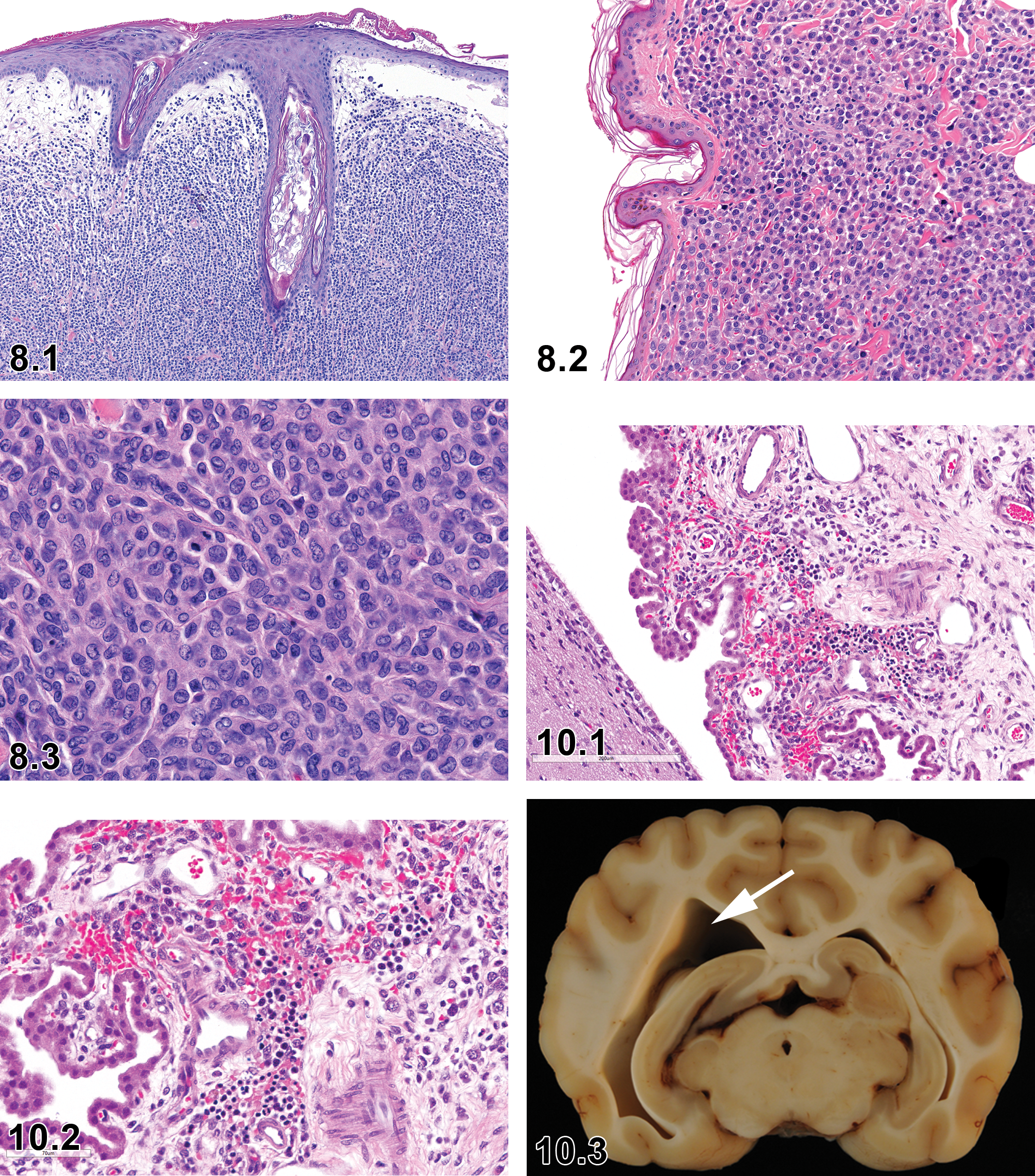
Dog, skin, histiocytoma, H&E. Courtesy of Eric van Esch.
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Langerhans cells.
Diagnostic Features
Grossly, a dome-shaped protrusion of the skin, hairless, often abraded, and ulcerated.
Dermal (and epidermal) infiltrate of densely packed, mildly pleomorphic round cells, arranged in cords and nests.
Histiocytic-like neoplastic cells, ovoid, bean-shaped, or convoluted nuclei, lightly eosinophilic cytoplasm.
Often numerous mitotic figures.
Typically, wedge shaped, that is, narrow basis and wider near the epidermis, 95 “top heavy” epidermal focus. 27,96
Little or no stroma.
Lymphocytic infiltrates at the base of the neoplasm may be present during regression.
Immunohistochemistry may be used to provide definitive proof 97 ; canine cutaneous histiocytomas are considered a Langerhans cell tumor and express CD1a, MHC II, CD11c, CD18, and often E-cadherin. 96,98
Differential Diagnoses
Epitheliotropic T-cell lymphoma: When considered because of epithelial involvement, differentiation is best done by IHC.
Plasmacytoma: Sheets of round cells, in nests or poorly defined cords, pleomorphic nuclei, mono, bi-, or multinucleated, no or few mitoses, scant to moderate eosinophilic or basophilic cytoplasm, amyloid may be present. The IHC may be required for definite differential diagnosis; MUM1 is considered the most reliable marker. 99
Cutaneous histiocytosis: Coalescence of perivascular infiltrates in the subcutis and deep dermis without involvement of epidermis.
Comment
Usually a solitary lesion of young dogs, less than 3 years old, but has been observed in dogs of all ages. The lesion occurs in all breeds, predisposed are brachycephalic breeds, including boxers, but also dachshund, Doberman pinschers, and cocker spaniels. Spontaneous regression is a feature. 96,100
9. Mammary Gland
Introduction
For detailed general considerations, refer to the INHAND rodent publication. 101 A detailed description of estrous cycle-related changes in the canine female genital tract, including the mammary gland, is provided in a review by Rehm et al 102 ; cycle-related changes of the mammary gland are also presented in Table 11.1 of the female reproductive system. The following findings may be observed as part of the normal estrus cycle in dogs: fibrosis in early diestrus, apoptosis (single-cell necrosis), and pigment accumulation in proestrus. 103
In routine toxicologic pathology studies, 1 section of the mammary gland, usually one of the inguinal glands, is examined per female and male. The section should include the nipple, the duct, and a representative amount of glandular tissue. In male dogs, glandular tissue may be difficult to obtain; the presence of ductular structures is considered sufficient in these cases.
Substantial differences between dogs and rodents are observed in the morphology and terminology of mammary gland neoplasms; for a detailed description of the neoplastic and tumor-like lesions, refer to the “International Histological Classification of Tumors of Domestic Animals: Mammary Tumors of the Dog and Cat,” published by the Armed Forces Institute of Pathology in conjunction with the American Registry of Pathology and the World Health Organization, 104 the more recently published “Tumors of Domestic Animals,” 105 and other pertinent literature.
The recommended terminology of microscopic lesions in the mammary gland of dogs is presented in Table 9.1.
Microscopic Findings in the Mammary Gland; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
Apoptosis—Mammary Gland
Other Term(s)
Programmed cell death.
Pathogenesis/Cell of Origin
Mitochondrial or intrinsic pathway
Death receptor or extrinsic pathway
Perforin/granzyme pathway
Diagnostic Features
Cytoplasmic and nuclear condensation
Nuclear fragmentation
Intact plasma membrane until late in the process
Lack of inflammation
Tingible body macrophages may be present
Differential Diagnoses
Single-cell necrosis: Single-cell necrosis results from acute cellular injury and is characterized by cell and nuclear swelling, karyolysis or karyorrhexis, nuclear pyknosis, pale eosinophilic cytoplasm, cytoplasmic vacuoles, loss of cellular detail, cellular debris, and inflammation.
Comment
A comprehensive description of the processes involved and guidance on which terminology to use is provided by the publication of the INHAND Apoptosis/Necrosis Working Group.
72
In brief: Use necrosis and apoptosis as separate diagnostic terms. Use modifiers to denote the distribution of necrosis (eg, necrosis, focal; necrosis, diffuse, etc). Use the combined term apoptosis/single-cell necrosis, when there is no requirement or need to split the processes, or when the nature of cell death cannot be determined with certainty, or when both processes are present together. The diagnosis should be based primarily on the morphological features in H&E-stained sections. When needed, additional, special techniques to identify and characterize apoptosis can also be used.
10. Nervous System—Brain, Spinal Cord, and Peripheral Nerve
Introduction
Species used for neurotoxicological research exhibit 3 different general models of brain architecture based on the prominence of cerebrocortical subdivisions (archicortex, paleocortex, neocortex). The rodent model, which also applies for the rabbit, is characterized by a lissencephalic cerebral surface and large archicortex. The carnivore (dog, cat) model shows prominent gyri and sulci in the cerebrum, which abuts the cerebellum but does not cover it in contrast to the primate model. The carnivore model shows far less complex cerebrocortical subdivisions than the primate. The most striking difference between carnivore and rodent brain is the presence of a well-developed neocortex in carnivores. In the spinal cord, a notable difference between rodent and carnivore is the much larger size of the dorsal funiculus in carnivores. The location and size of spinal motor tracts show notable species differences: the corticospinal tract of rats is found in the dorsal funiculus, while in carnivores and primates, it is located in the lateral funiculus; compared to carnivores, the rodent shows smaller sensory tracts in the dorsal funiculus. In the brain stem, the olivary nucleus is enlarged in carnivores and primates compared to rodents, reflecting also the greater lateral expansion of the cerebellar hemispheres. The size of the medial and lateral lemniscus is increased in carnivores and primates compared to rodents. The profile of the vermis on the caudal surface of the cerebellum is generally straight in rodents and primates but is twisted into a serpentine loop in carnivores because of its elongation in these species. The corpus striatum is unified in rodents but divided in carnivores and primates into caudate and lentiform nuclei. The information summarized here came from a review article, 106 and the reader is referred to that publication for additional details. Other pertinent references include:
A comparative brain atlas of rat, dog, and monkey in Fundamental Neuropathology for Pathologists and Toxicologists, Chapter 3. 107
The atlas “The Beagle brain in stereotactic coordinates” 108 is a useful reference for neuroanatomy in the dog.
The large brain size of the carnivore compared to rodents necessitates a different approach to trimming and embedding the brain; a recommended trimming scheme is presented in an STP position paper by Bolon et al 109 resulting in 7 sections/blocks.
The nomenclature of lesions of the nervous system in the dog differs from the rodent most strikingly in the classification of neoplasms, which is outside the scope of this document, given the very low incidence of nervous system neoplasms in the young age and well-controlled breeding programs of purpose-bred dogs commonly used in general toxicity studies. For a detailed description of the neoplastic and tumor-like lesions, refer to “Tumors of Domestic Animals” 110 and other pertinent literature.
Table 10.1 summarizes the recommended terminology of microscopic lesions observed in dogs used in toxicity studies.
Microscopic Findings in the Nervous System: Brain, Spinal Cord, and Peripheral Nerve; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
c Refer to description in the section “Cardiovascular System.”
Extramedullary Hematopoiesis—Choroid plexus (Figures 10.1 and 10.2)
Other Term(s)
Hematopoietic hyperplasia; myeloid hyperplasia; erythroid hyperplasia; megakaryocytic hyperplasia; hematopoietic cell proliferation; myeloid metaplasia.
Pathogenesis/Cell of Origin
Hematopoietic progenitor cells.
Diagnostic Features
Hematopoietic cells of 3 lineages (myeloid, erythroid and megakaryocytic) may be present.
Proportions of cell types varies.
Any of the lineages may be predominant.
Each lineage is often represented by cells in various stages of differentiation.
Differential Diagnoses
Neoplasms of hematopoietic origin:
Leukemia, myeloid; Leukemia, erythroid; or Leukemia, megakaryocytic:
Tumor cells are often all at one stage of differentiation especially in less well-differentiated tumors.
Other organs are often involved with the same neoplastic cells.
Some leukemia present with minimal tissue involvement but with a high white blood cell count and numerous immature forms/blasts in the blood.
Mitosis, apoptosis, and necrosis may be present.
Lymphoma:
Cell morphology is different from that of myeloid, erythroid or megakaryocytic cells.
Infiltrate, mononuclear cell.
Mononuclear cells without any tissue reaction.
Inflammation:
Mononuclear cells with reaction of surrounding tissue.
Comment
Extramedullary hematopoiesis (EMH) in the choroid plexus represents a very subtle finding in beagles; it has been noted in almost 35% of 19- to 56-week-old males and 20% of 24- to 85-week-old females. 111 In dogs from certain breeders, increased incidences were noted, and the observation was not necessarily associated with a pathologic condition. In another publication, EMH was described in dogs euthanatized because of seizures; no other remarkable hematopoietic findings were noted and no other site of extramedullary hematopoiesis was observed. 112 However, no coherent evidence for a connection between EMH and seizures was provided.
Dilatation, Ventricle—Brain (Figure 10.3)
Other Term(s)
Ventriculomegaly.
Pathogenesis/Cell of Origin
Unknown.
Diagnostic Features
Widening of the ventricular spaces (bilateral or unilateral).
Without changes in ependymal cells, white matter, glial/microglial cells.
Normal cerebrospinal fluid flow.
Differential Diagnoses
Hydrocephalus: Internal hydrocephalus is also characterized by a widening of the ventricular space; it is caused by disturbance or blockage of ventricular drainage and accompanied by degenerative changes, including atrophy and cavitation of the white matter.
Comment
Dilatation of the ventricle(s) or ventricular system is usually a diagnosis readily made at trimming. The need of histomorphologic correlation may require the repetition of the term. Vullo et al 113 reported 17 asymptomatic beagle dogs with ventricular dilation by magnetic resonance imaging and ultrastructure evaluations (normal ependymal cells, white matter, and glia/microglial cells with normal cerebrospinal fluid flow).
Meningioangiomatosis—Meninges (Figure 10.4)
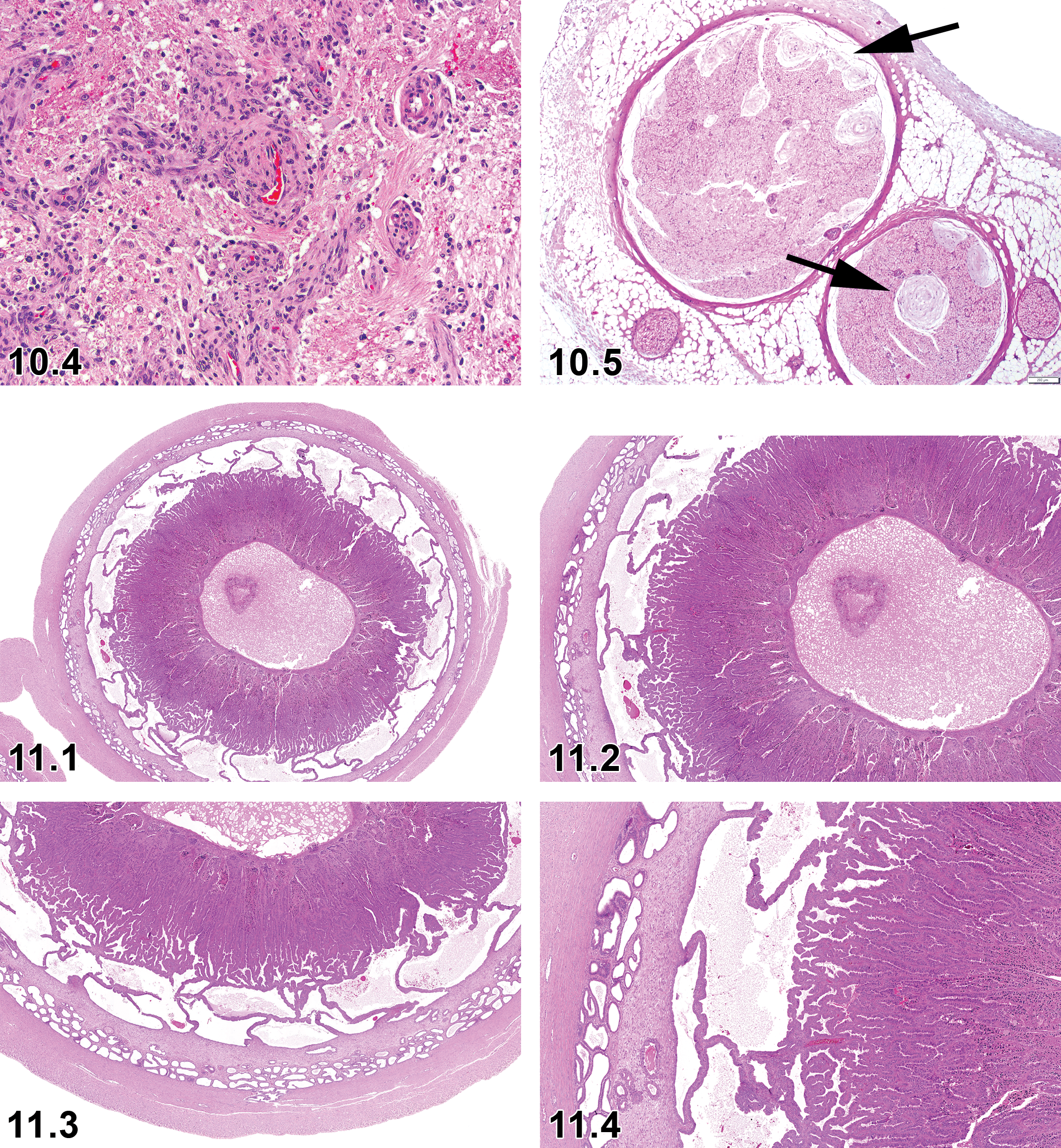
Dog, brain, meningioangiomatosis, H&E.
Biological Behavior
Meningioangiomatosis is a benign proliferative condition of the central nervous system, which is considered to be a vascular malformation or hamartoma (ie, a disorganized, benign mass composed of cells indigenous to the site) and not a neoplasm.
Other Term(s)
Not applicable.
Pathogenesis
This finding likely develops from mesenchymal, pluripotent cell with fibroblastic or meningothelial differentiation.
Diagnostic Features
Plaque-like thickening of the meninges of brain stem or cervical spinal cord of young dogs, which can be discerned grossly.
Characterized by a focal proliferation of blood vessels and spindle-shaped meningothelial cells extending from the meninges along Virchow-Robin’s spaces into the adjacent brain parenchyma.
Cells do not display atypia, pleomorphism, or mitotic activity.
Meningothelial cells especially those adjacent to the blood vessels are immunoreactive for vimentin.
Dense gliosis, rarefied neuropil, scattered Rosenthal fibers (thick, elongated, brightly eosinophilic, irregular structures in astrocyte processes), and slight white matter vacuolation are the accompanying features.
Comment
Meningioangiomatosis is a rare benign proliferative condition of central nervous system reported in humans and animals including young dogs. It is generally considered to be a hamartoma; however, whether meningioangiomatosis is hamartomatous, neoplastic, or a developmental defect remains controversial. 114 Affected dogs exhibit ataxia, circling, rotatory nystagmus, mild tetraparesis, proprioceptive deficits, or muscle atrophy. 115,116
Renaut Bodies—Peripheral Nerve (Figure 10.5)
Biological Behavior
Benign proliferation of endoneural collagen forming well-demarcated elliptical, lamellar structures containing a core of intensely eosinophilic fibers.
Other Term(s)
Not applicable.
Histogenesis
Mesenchymal origin endoneural collagen subtypes I, II and VI) and mucopolysaccharides
Pathogenesis
Physiologic adaptation of nerves (usually distal portion of nerves within fore and hind legs) hypothesized to be a response to mechanical stress imposed on them.
Diagnostic Features
Subperineurial accumulations of elongate, loosely whorled, mucin-rich, cell-poor connective tissue that occur in various peripheral nerves, especially in the distal branches of the sciatic nerve branches tibial and fibular nerve, and less commonly in autonomic nerves near viscera. 117
Renaut bodies (RBs) occur within nerve fibers but do not cause axonal degeneration or reactive gliosis/inflammation or encapsulation.
They range from 20 to 500 µm in diameter and hundreds of microns in length in longitudinal section
They are elliptical in shape when sectioned transversally and cylindrical in shape longitudinal and have onion-skin arrangement containing loosely textured, concentric lamellae of filamentous strands intermixed with sparse numbers of dark spindle shaped nuclei.
Special diagnostic techniques:
Histochemical results included positive acidic Alcian blue, Gomori’s trichrome, and Verhoeff Van Gieson’s and negative Periodic acid–Schiff, Congo red, and Luxol fast blue/cresyl violet.
Immunohistochemical results included positive vimentin and collagen (subtypes I, II, and VI) and negative neuron-specific enolase, S-100, glial fibrillary acidic protein, amyloid A component, desmin, alpha-sarcomeric actin, pancytokeratin, epithelial membrane antigen, and von Willebrand factor.
Transmission electron microscopy revealed loosely arrayed, circumferentially oriented collagen fibers intermixed with varying amounts of amorphous substance and finely fibrillar material. Most of the cells comprising the RBs are fibroblasts.
Differential Diagnoses
Schwannoma, mesenchymal neoplasia.
Comment
Renaut bodies have been described in humans at sites of chronic nerve entrapment 118 as well as in control beagle dogs 117,119,120 and horses. 119,121 The incidence of RB can increase in subchronic and chronic studies 120 and have been reported in hypertrophic neuropathy. 122 These lesions may serve to cushion adjacent nerve fibers from mechanical trauma. 119 This premise is supported by the experimental demonstration that RB may be induced by nerve compression, 120,123,124 suggesting that mechanical trauma is the primary pathogenesis for the change, although other mechanisms may be involved since affected autonomic nerves near viscera are not subjected to great mechanical stress. 117 The number of RB increases with age 125 and may vary by the vendor. 126
11. Female Reproductive Tract—Ovaries, Oviduct, Uterus, Cervix, and Vagina
Introduction
For general aspects on the female reproductive system, refer to the rodent publication. This chapter provides the recommended nomenclature for the canine female reproductive tract. Generally used preferred terms of systemic nonproliferative lesions across organ systems, such as hemorrhage, inflammation, or thrombus, are included in separate INHAND manuscripts and are therefore not included in this section, except where there are distinguishing features of the process relevant to the female reproductive system. In all organs and tissues, including those of the female reproductive system, tumors are very rarely observed in toxicologic pathology studies in beagle dogs and therefore are generally not included in this manuscript. For the nomenclature and diagnostic criteria of neoplastic changes, refer to the World Health Organization’s Histologic Classification Tumors of the Genital System of Domestic Animals, 127 the more recent publication on Tumors of Domestic Animals by Meuten1, and pertinent literature.
Anatomy and Physiology
The anatomy and histology of the canine female reproductive tract has been described extensively, and it is beyond the scope of this chapter to detail the morphology of this system and its cycle-dependent changes. The reviews by Rehm et al 102 and Chandra and Adler 128 are excellent references for this topic.
Briefly, the cycle is composed of proestrus (lasting 1-2 weeks), estrus (lasting 1-2 weeks), and diestrus (lasting between 2 and 3 months). Metestrus in the dog is a very short phase usually not distinguished from estrus. 102 The anestrus phase of the cycle is unique to the dog. It represents a quiescent phase following diestrus and lasting between 3 and 5 months. 102 Dog-specific peculiarities in the reproductive physiology include the presence of polyovular follicles, ovulation of an immature oocyte, and a periovulatory period with particularly high concentrations of circulating progesterone. 129 –131
In the female dog, the ovary, uterus, cervix, and vagina show marked cyclical changes, which are displayed in Table 11.1. For easy reference, the cycle changes in the mammary gland are included as well.
Normal Cyclic Changes in the Female Reproductive System.

Nomenclature
The recommended nomenclature for nonproliferative and nonneoplastic proliferative changes in beagle dogs used in general toxicology studies listed in Tables 11.2 to 11.6. The nomenclature is adapted from the rodent manuscript as listed on the go-RENI.org website. Diagnostic criteria for nonneoplastic changes are only described if they differ from those used in mice and rats.
Microscopic Findings in the Female Reproductive System: Ovary, Dog.

a Terminology addressed in the General Pathology section.
b Preovulatory follicles are luteinized in the dog and should not be diagnosed as a finding if the dog is in late estrus.
c Terminology with diagnostic criteria and/or comments described in the text.
Microscopic Findings in the Female Reproductive System: Oviduct, Dog.

a Terminology addressed in the General Pathology section.
Microscopic Findings in the Female Reproductive System: Uterus, Dog.

a Terminology addressed in the General Pathology section.
b Terminology with diagnostic criteria and/or comments described in the text.
Microscopic Findings in the Female Reproductive System: Cervix, Dog.

a Terminology addressed in the General Pathology section.
b For a description and diagnostic criteria, refer to Uterus (rodent).
Microscopic Findings in the Female Reproductive System: Vagina, Dog.

a Terminology addressed in the General Pathology section.
Ovaries
Adenoma—Ovary
Comment
A rare benign tumor in the ovary of dogs, which has been observed in toxicologic pathology studies in beagle dogs. In the INHAND rodent manuscript of the female reproductive system, tubulostromal adenoma and cystadenoma of the ovary are included. 132 For the diagnostic criteria in dogs of this and other neoplastic findings, refer to the fascicle on the genital system of the International Histological Classification of Tumors of Domestic Animals published by the Armed Forces Institute of Pathology in conjunction with the American Registry of Pathology and the World Health Organization, 127 the chapter on neoplasms of the genital systems in the textbook Tumors of Domestic Animals 133 , and/or current literature.
Oviduct, Uterus, Cervix, and Vagina
Hyperplasia, Segmental Cystic—Uterus (Figures 11.1–11.4)
Other term(s)
Pseudoplacentational endometrial hyperplasia (PEH).
Pathogenesis/cell of origin
Endometrial glands and stroma under the influence of progestogen stimulation.
Diagnostic features
Localized proliferation of the endometrium
Broad-based polypoid mass extending into the lumen or continuous band of protruding tissue.
Cystic epithelial proliferation of the endometrium separated into 2 layers.
The uterine surface endometrium is replaced by cavernous spaces lined by hyperplastic columnar epithelium with pale pink secretory cytoplasm.
The lower endometrium contains dilated basal glands.
Necrotic luminal surface and necrotic debris often observed in the lumen.
Differential diagnoses
Endometrial stroma polyp. The polyp contains substantial connective tissue stroma in addition to dilated glands and is pedunculated.
Comment
Segmental cystic endometrial hyperplasia or PEH of pseudopregnancy occurs occasionally; it has been observed in association with pyometra. It may be induced by a variety of sterile substances in the uterine lumen during the luteal phase 134,135 and intrauterine Escherichia coli suspension. 136 The ovary contains prominent and persistent corpora lutea. Use of the term deciduoma, while sometimes present in the literature, 137 is not recommended as it is not descriptive and does not reflect the pathogenesis of the finding. 138 Likewise, the use of the term pseudopregnancy is not recommended and should be restricted to the clinic.
Polyp, Endometrial Stroma—Uterus
Other term(s)
Endometrial polyp.
Pathogenesis/cell of origin
Proliferation of glandular and stromal elements.
Diagnostic features
Single or multiple projections of sessile or pedunculated masses into the uterine lumen.
Dilated uterine glands within a proliferating stroma.
Pedunculated polyps may protrude into the cervix and vagina.
Differential diagnoses
Segmental cystic hyperplasia. The polyp contains substantial connective tissue stroma in addition to dilated glands and is pedunculated.
Comment
In the rodent (see the rodent publication or the goReni website), endometrial stroma polyps are considered benign neoplasms. In the dog, as in other domestic animals, they can be observed as a spontaneous change that is considered hyperplastic in nature and not a neoplastic entity.
12. Male Reproductive System—Testis, Efferent Ducts, Epididymis, and Prostate
Introduction
Standardized nomenclature recommended for the reporting of microscopic lesions in the canine male reproductive tract is based on the INHAND project nomenclature previously published for the rat and mouse male reproductive system. 139
The male dog reproductive anatomy and histologic architecture differ from the rodent in several ways. Rodents can voluntarily retract the testes into the inguinal canal; dogs cannot. Canine testes that have not descended from the inguinal canal or abdominal cavity by about 6 weeks of age indicate cryptorchidism. 140 Unlike the funnel-like pattern in rodents where efferent ducts join together as a single common duct emptying into the head of the epididymis, dogs have multiple, short parallel efferent ducts emptying separately into the head. 141 Congenital blind-ending efferent ducts in the dog may predispose to sperm granulomas (via sperm stasis) near the epididymal head. 142 The prostate is the only accessory sex gland in the male dog. Anatomy of the canine penis includes an os penis (located dorsal to the urethra) and bulbus glandis (erectile tissue) which during mating contribute to copulation and lock (“tie”) the male and female, respectively.
Sexual Maturation in the Male Dog
The use of animals with sexually immature or peripubertal reproductive organs is not a sensitive means to detect potential treatment-related effects to adult reproductive tissues. Dogs may be 5 to 6 months of age at the start of routine toxicity studies, but sexual maturity occurs from 7 to 12 months of age, and this age can vary depending on the source of the animals. 140,143 Therefore, in male dogs at the lower end of this age spectrum, the male reproductive tract may often be undergoing sexual maturation, particularly during nonclinical studies of short duration (less than 13 weeks). Therefore, the reproductive tract of dogs may not be mature in some nonclinical studies. Unlike rats where spermatogenesis begins shortly after birth, in dogs, there is a postnatal dormant period of about 5 months before spermatogenesis starts. 140 At 5 to 6 months of age, the canine Sertoli cells begin to produce fluid which opens tubular lumina and may form vacuoles within the seminiferous epithelium. 140 Spermatocytes are evident at 6 to 7 months of age, followed by spermatids at 6 to 8 months of age, and then shedding spermatozoa at 7 to 10 months of age. 140 Early waves of spermatogenesis are not as efficient so there can be frequent germ cell sloughing, degeneration, and multinucleated cells with cellular debris present within the epididymis. 144 This is often not uniform across the canine testes since the individual seminiferous tubules vary in their rate of spermatogenesis. 144 This is in contrast to rodents where the rate of spermatogenesis is fairly uniform across the testes. 140
In contrast to rodents, dogs on a nonclinical study do not mature at the same rate. 140 Despite a similar age range, some dogs on study may have mature testes and epididymal ducts filled with sperm, whereas other dogs may have immature reproductive tracts or peripubertal seminiferous tubules with germ cell depletion and/or degeneration and epididymides devoid of spermatozoa. 140 Therefore, dogs on study may have varying microscopic presentation attributable to differences in their individual rate of sexual maturation and not due to test article administration. It is important to distinguish microscopic differences due to immaturity or peripuberty from those related to experimental treatment. The pathology report should address when it is challenging or not possible to differentiate germ cell degeneration attributed to sexual maturation from that which could be a treatment-related effect.
Documentation of sexual maturation can aid the interpretation of study findings. A survey by a Working Group of the Scientific and Regulatory Policy Committee of the STP showed that pathologists prefer the terms “immature” and “mature” and also use the term “peripubertal.” 145 However, some institutions may only record sexual immaturity since the default is reproductive maturity. The Canine Nonrodent Working Group of the INHAND project recommends that for nonclinical studies the state of sexual maturity should be recorded as “mature” when sexually mature and “immature” or “peripubertal” when sexually maturity has not been reached. These terms more accurately reflect the nature of changes related to an underdeveloped male reproductive tract and will help to better distinguish findings related to experimental treatment from those related to sexual maturation. Evaluation of the peripubertal testis is challenging, if not impossible, to assess from treatment-related findings to spermatogenesis. When recording maturation status, the pathologist should clearly refer to sexual maturation to avoid confusion with skeletal maturation. Where in the study data the state of sexual maturation is recorded which should be at the discretion of the institution. The INHAND project recommends recording sexual maturity as “immature,” “peripubertal,” “mature,” or, when sexual maturity cannot be determined, “indeterminate” under a “summary tissue,” as proposed by a working group of the STP or under the testis if a summary tissue is not available.
When assessing the maturation status, it is important to be aware that the canine male reproductive tract is not synchronized with respect to the rate of maturation of the testes, epididymides, and prostate. Maturation of the prostate may be delayed in relationship to the state of sexual maturation in the testes or vice versa. The difference in the state of maturation between the testes and prostate has been attributed to differences in the expression of the androgen receptor. 140 Pathologists usually identify testicular maturation by spermatogenesis (ie, presence of elongating spermatids) and epididymal tail containing spermatozoa. 144,145 Immature prostatic acini have narrow, undeveloped lumina lined by basophilic epithelium that is cuboidal to flattened with a high nuclear to cytoplasmic ratio. 49,143 The amount of connective tissue is relatively higher in the immature prostate than the mature prostate. 143 Maturing prostate can have a patchy distribution of immature small acini with wide interstitium and mature glands with scant stroma. Mature prostatic glands are dilated with epithelial infoldings lined by columnar cells with abundant eosinophilic cytoplasm and supported by scant stroma. 143 Luminal eosinophilic secretory material is often not observed.
Brief definitions of the three separate stages of sexual maturation for the male dog are: Immature: Immature animals have undeveloped testes, epididymides, and prostate. The immature testes lack spermatozoa within seminiferous tubules. From birth to about 5 months of age, the immature seminiferous tubules are contracted and populated only by Sertoli cells and centrally located gonocytes.
144,146
Subsequently, the underdeveloped seminiferous tubules are lined only by Sertoli cells and basally located spermatogonia.
144
There can be low numbers of spermatocytes. Spermatids and spermatozoa are not present. There are no or rare exfoliated germ cell debris and multinucleated cells. Immature epididymides have underdeveloped contracted epididymal ducts lined by a low epithelium.
49,144,146
Mature sperm are not present, but there may be rare germ cell debris in the lumina. The immature prostate is small and glands are not developed. Peripubertal: Peripubertal animals have seminiferous tubules in varying stages of development or may have developed testes but underdeveloped prostate. Seminiferous tubular lumina are partially dilated and there can be vacuoles within the seminiferous epithelium due to fluid production by Sertoli cells.
140,144
Tubular epithelium may vary from having a limited number of germ cells to being fully populated with germ cells. Some seminiferous tubules have germ cell development to mature spermatids. There is frequent degeneration of germ cells or multinucleated giant cells during early waves of spermatogenesis.
144
At or near the rete testis, there may be sperm stasis. Contracted or partially expanded epididymal ducts may contain increased numbers of sloughed degenerate germ cells evident as luminal cell debris.
144
Few spermatozoa may be present in the epididymal head or body but tend to be particularly absent from the epididymal tail. Maturation of the prostate may vary from the maturation state of the testes since prostatic maturation is not synchronized with testicular development.
144
If the testes are mature but the prostate still underdeveloped, the dog should be considered as peripubertal. Mature: Mature animals have developed testes and prostate. Mature testes have a full complement of developed seminiferous tubules with all 4 generations of germ cells (spermatogonia, spermatocytes, round spermatids, and elongating spermatids) present and mature spermatids present throughout the section. Residual bodies are present and signify recent release of mature spermatids.
140
Germ cell degeneration and multinucleated giant cells are less than observed for peripubertal testes.
144
Abundant spermatozoa generally expand the lumina of epididymal ducts in the corpus and tail.
144
There can be rare sloughed germ cells. Maturation of the prostate may vary, but in sexually mature animals, the prostate should be fully developed. In contrast, animals with developed testes but underdeveloped prostate are considered peripubertal.
Morphologic Evaluation of Canine Male Reproductive System
External genitalia (penis, prepuce, and scrotum), the spermatic cord (vas deferens, pampiniform plexus, cremaster muscle), and efferent ducts are generally not examined microscopically in routine nonclinical toxicity studies. The male reproductive tissues routinely evaluated in nonclinical toxicity studies are the testes, epididymides, and prostate. Histologic sectioning of the testis should include the tunica albuginea, parenchyma (seminiferous tubules and interstitium), and the rete testis. Microscopic examination of the epididymides should include the head (caput), body (corpus), and tail (cauda). Microscopic findings in the epididymis should include location modifiers such as head or tail. Longitudinal sampling of the epididymides is preferred, as it increases the opportunity to include the efferent ducts for evaluation, since they are located within the epididymal head of the dog. 140 Pathologists should be aware that the efferent ducts are embedded within the initial segment/head of the epididymis in dogs and not erroneously diagnose these normal structures as epididymal degeneration or epithelial hyperplasia. Additional reproductive tissues may be examined for specific studies but are not generally necessary to adequately assess the male reproductive tract in routine toxicity studies.
Testis
Canine seminiferous tubules are grouped into lobules separated by connective tissue and with ends emptying into a central rete testis within the fibrous mediastinum. 140 This histologic architecture influences how testicular injury morphologically presents in the dog. For example, injury to an affected seminiferous tubule can present as a change isolated to a lobule. In contrast, rats do not have lobular arrangement so morphologic changes to a seminiferous tubule may be dispersed throughout the testis. 140 Seminiferous tubule and/or stage-specific microscopic findings should include modifiers such as cell type (eg, Sertoli cells, germ cells, spermatogonia, spermatocytes, round spermatids, or elongating spermatids). Microscopic evaluation of the canine testis should be conducted with an attention to spermatogenic stages. 147,148 However, detailed stage aware evaluations are challenging for the dog 140 due to the following reasons: (1) germ cell layer associations are less synchronized than in the rat, (2) spermiation (release of mature spermatids) may occur over 2 stages, (3) the profile of elongating spermatid heads may present differently depending on the plane of section, (4) periodic acid—Schiff does not stain the spermatid acrosome until spermiation, 149 and (5) dogs have a higher incidence of spontaneous findings (eg, hypospermatogenesis, swollen spermatocytes, multinucleated giant cells, or changes associated with peripubertal development) compared to the rat.
The recommended terminology of microscopic changes observed in the testes of the dog is presented in Table 12.1.
Microscopic Findings of the Male Reproductive System: Testis; Dog.
aTerminology addressed in Systemic Pathology section.
bTerminology with diagnostic criteria or comments described in the text.
cFinding more frequent as an induced change.
dTerminology addressed in the Cardiovascular section.
Hypoplasia, Tubule—Testis (Figures 12.1 and 12.2)
Dog, testis, tubule hypoplasia, H&E, low magnification.
Other Term(s)
Atrophy/hypoplasia, focal hypoplasia, Focal subcapsular tubular atrophy or hypoplasia, segmental hypoplasia, Sertoli cell-only tubules.
Modifiers
Segmental.
Pathogenesis
The lobular distribution suggests that the group of tubular profiles belong to a single, coiled, seminiferous tubules never having been populated by germ cells, hence hypoplasia.
Diagnostic Features
Lobular distribution that is often wedge-shaped and situated in a subcapsular location.
Generally affects triangular clusters (segments) of varying numbers of seminiferous tubular profiles.
Affected tubules are completely devoid of germ cells and are only lined by Sertoli cells.
Tubule diameter smaller than normal tubules.
Tubular lumina are empty.
Affected tubules reside immediately adjacent to normal tubules.
Unilateral, but may be bilateral.
Differential Diagnoses
Atrophy, tubule: Alternatively, seminiferous tubules that have sustained permanent loss of germ cells (atrophy) may result in Sertoli cell-only tubules. Basement membrane thickening may be a feature.
Transitional areas (tubuli recti): Germ cells are reduced or absent at transitional areas between seminiferous tubules and the rete testis. 150 These areas are normal anatomy and not a finding to be diagnosed.
Hypospermatogenesis: A complete or partial loss of one or more germ cell layers that tend to have a patchy distribution throughout the testes.
Comments
Tubules lined by only Sertoli cells in dogs are attributed to hypoplasia of entire tubules or tubular segments presumably never populated by germ cells during testicular development. The lobular distribution is considered evidence that the affected tubular profiles belong to a single, convoluted, seminiferous tubule. 144 Descriptive terminology such as “Sertoli cell-only tubules” have been used or suggested by some authors, 49,151 and the finding can be confused with focal tubular atrophy. However, the high prevalence of this finding in dogs less than 1 year of age suggests “tubule hypoplasia” is an appropriate term. Tubule hypoplasia may be seen in dogs of any age. The etiology is presumably due to a reduced migration of germ cells to the affected segment.
Hypospermatogenesis—Testis (Figures 12.3 and 12.4)
Other Term(s)
Segmental hypospermatogenesis.
Modifiers
Segmental.
Pathogenesis
Hypospermatogenesis is attributed to transient failure/inefficiency of spermatogenesis affecting a segment of the seminiferous tubule resulting in an absence of one more generation of germ cells.
Diagnostic Features
Affects individual tubular profiles (segments) randomly distributed throughout the testes (may affect few, many, or the majority of tubules).
Tubules have a complete or partial absence of spermatocytes, round spermatids, and/or elongate spermatids.
When affecting only some of the germ cell generations, the affected tubular segments may be missing round spermatids and/or pachytene spermatocytes from the layers below the overlying elongate spermatids.
There may be reduction of tubular diameter.
Affected tubules may have prominent Sertoli cells or contain small amounts of luminal debris, multinucleated giant cells, and/or swollen spermatocytes.
Unilateral or bilateral.
Differential Diagnoses
Immature: The presence of elongate spermatids in hypospermatogenesis differentiates this finding from immaturity.
Peripubertal: Seminiferous tubules vary from having a limited number of germ cells to being fully populated with germ cells including development to mature spermatids. There is frequent degeneration of germ cells or multinucleated giant cells.
Transitional areas (tubuli recti): Germ cells are reduced or absent at transitional areas between seminiferous tubules and the rete testis. 150 These areas are normal anatomy and not a finding to be recorded in a nonclinical toxicology study
Hypoplasia, tubule: Triangular clusters (segments) of varying numbers of seminiferous tubular profiles completely devoid of germ cells and are lined by Sertoli cells only.
Atrophy, tubule: Permanent loss of germ cells (atrophy) may result in Sertoli cell-only tubules. Basement membrane thickening may be a feature.
Degeneration/atrophy, tubule: Some seminiferous tubules have degenerating germ cells and other tubules have partial or complete loss of germ cells that may result in Sertoli cell-only lined tubules.
Degeneration, tubule: Seminiferous tubules have degenerating germ cells not restricted to a specific germ cell type or stage.
Degeneration, germ cell: Cell type-specific or stage-restricted germ cell death demonstrating cytoplasmic eosinophilia, chromatin condensation/margination, and/or nuclear fragmentation. 139
Depletion, germ cell: Partial or complete loss of one or more germ cells layers in a more specific pattern and/or diffuse pattern. 139 Depletion related to treatment is typically a diffuse change to a uniform population of germ cells.
Comments
It is common for most testes to have a few tubules affected by segmental hypospermatogenesis. The finding varies widely in severity, with the lesion being marked or severe in some animals. 144,150,151 It has been reported to range from affecting <10% tubules per testis to greater than 75% of tubules per testis. 144 At higher severity levels, the size and/or weight of the testis may be reduced and the number of sperm in the epididymides may be reduced. 150 Distinguishing hypospermatogenesis from peripubertal testes or tubule degeneration/atrophy or germ cell depletion can be challenging. There is often overlap in the appearance of germ cell depletion and hypospermatogenesis. Germ cell depletion that is induced by xenobiotics or radiation tends to have a more widespread, diffuse distribution and often impacts a more uniform population of cells. In addition, the extent of the finding tends to correlate with the length of the treatment period. In contrast, hypospermatogenesis has a more random distribution throughout the testis, often affects different germ cell types, and the severity and/or cell types affected may not correspond to the treatment period. Hypospermatogenesis has been reported to occur at a higher incidence in younger control beagle dogs 151 ; however, the absence of various germ cell types is a normal feature during puberty, and when diagnosing hypospermatogenesis, there should be clear evidence of maturity (eg, mature spermatids, residual bodies, expansion of the epididymal tail). Since hypospermatogenesis is a transient inefficiency in spermatogenic activity, degenerate/multinucleated germ cells are infrequent. 140 The appearance of an increased incidence and/or severity of hypospermatogenesis with treatment may occur due to test article-related germ cell depletion. Therefore, testicular evaluations must consider dose-related exacerbation of this change and it is important to be aware of historic control data for the test site.
Swollen spermatocytes—Testis (Figure 12.5 and 12.6)
Other Term(s)
Enlarged spermatocytes.
Pathogenesis
Swollen spermatocytes are considered to be individual germ cells arrested during meiosis. These are likely arrested secondary spermatocytes that failed to undergo their second meiotic division. These swollen spermatocytes are often sequestered within a normally developing germinal epithelium.
Diagnostic Features
Abundant pale cytoplasm with eccentrically located nuclei.
Hyperchromatic nuclei (pyknotic) due to chromatin condensation.
Often mononuclear though authors have also shown binucleate.
Affects spermatocytes as single cells or as loose aggregates within a portion of the seminiferous tubule and are surrounded by germ cells undergoing normal maturation to round spermatids.
Affects a few tubules per testis.
Often bilateral.
Differential Diagnoses
Multinucleated giant cells: Multinucleated giant cells composed of spermatogenic cells (often round spermatids) reside within the seminiferous epithelium or lumen. 139
Degeneration, germ cell: Cell type-specific or stage-restricted germ cell death demonstrating cytoplasmic eosinophilia, chromatin condensation/margination, or nuclear fragmentation. 139
Comments
Swollen spermatocytes are a common incidental finding that can have a high prevalence reported to affect up to 93% of control beagle dogs that are 6 to 36 months of age. 151 The lesion is generally present at a low severity level affecting a few tubules per testis. Swollen spermatocytes are postulated to be secondary spermatocytes that did not undergo the second meiotic division and survived to later stages. 140 A low severity of swollen spermatocytes is generally not diagnosed. However, when the change is a prominent feature, diagnosis of swollen spermatocytes may be warranted.
Excurrent Ducts (Efferent Ducts and Epididymis)
In the dog, sperm are not typically observed in the head and proximal body of the epididymis and only become evident as they become more concentrated in the body and tail. 144 This is a normal feature in the dog and should not be recorded as a finding. One or more intranuclear eosinophilic inclusion bodies may be present in any region of the epididymis, but the head is considered the most common location. 152,153 Ultrastructurally, these inclusions have been reported to resemble secondary lysosomes and their significance is unknown. 152,153
Table 12.2 summarizes the histopathological lesions that can be observed in the efferent ducts and epididymis of beagle dogs in nonclinical toxicological studies.
Microscopic Findings of the Male Reproductive System: Efferent Ducts and Epididymis; Dog.

a Terminology addressed in Systemic Pathology section.
b Finding more frequent as an induced change.
c Terminology with diagnostic criteria or comments described in the text.
d Terminology addressed in the Cardiovascular section.
Adenomyosis—Efferent Ducts and Epididymis
Other Term(s)
Diverticulum, epithelial.
Pathogenesis
Origin unknown.
Diagnostic Features
Periductular connective tissue contains epithelial diverticuli of ducts that have penetrated through the smooth muscle layer.
Entrapped sperm may be within the lumen of epithelial diverticuli.
Sperm granuloma may occur secondary to entrapped sperm.
Differential Diagnoses
Not applicable.
Comments
Adenomyosis in the efferent ductules, epididymis, or vas deferens (deferent duct) is due to diverticula of epithelium that has extended through the smooth muscular wall supporting the ducts. 154,155 Adenomyosis should not be confused with intra-epithelial lumina and is distinct from adenosis of rodents, which are groups of epithelial cells with microlumina between ducts. The epithelial diverticuli in dogs may contain entrapped spermatozoa and are thought to contribute to the formation of sperm granuloma at locations considered unrelated to blind-ended efferent ducts near the epididymal head. 154 Estrogen has been suggested to play a role in adenomyosis. 155
Hyperplasia, Epithelium—Efferent Ducts and Epididymis
Other Term(s)
Not applicable.
Pathogenesis
Origin unknown.
Diagnostic Features
Proliferation of the epithelial cells lining the epididymis.
Differential Diagnoses
Cribriform change: The epididymis more commonly responds with cribriform change rather than epithelial proliferation. Cribriform change is attributed to collapse of epididymal ducts 140 associated with infoldings that can form 50-200 µm intraepithelial spaces (pseudoglandular cysts) lined by partially compressed epithelium. 151 Pseudoglandular spaces may contain eosinophilic content. Cribriform change has been described as a background finding in the dog at the junction of the epididymal body and tail. 144 Cribriform infoldings may also occur at the epididymal head in association with obstruction of the efferent ducts. 140 Sometimes cribriform change is considered a hyperplastic lesion. 140
Comments
Hyperplasia of the epididymal epithelium has been reported in beagle dogs older than 2 years of age and was most common in dogs older than 7 years of age. 156 Therefore, spontaneous epithelial hyperplasia is unlikely to be encountered in the age of dogs generally used in toxicology studies. Cribriform change is a change that is more likely to be observed than a true hyperplasia of the epididymal epithelium.
Accessory Sex Gland (Prostate)
Table 12.3 summarizes the histopathological lesions that can be observed in the prostate of beagle dogs in nonclinical toxicological studies.
Microscopic Findings of the Male Reproductive System: Prostate; Dog.

a Terminology addressed in Systemic Pathology section.
b Terminology with diagnostic criteria or comments described in the text.
c Terminology addressed in the Cardiovascular section.
d Not a preferred term; refer to the comments for hyperplasia, epithelium.
Dilatation, Cystic—Prostate (Figure 12.7)
Dog, prostate, cystic dilatation, H&E.
Other Term(s)
Dilatation, cystic acinar; dilatation, acinar/vesicle (not recommended, see comment).
Pathogenesis
Origin unknown.
Diagnostic Features
Focal or lobular “cystic” acini.
Acini may be lined by low cuboidal “atrophic” appearing epithelium or by columnar secretory epithelium.
Differential diagnoses
NA.
Comments
“Dilatation, cystic” is the preferred term over “dilatation, acinar/vesicle,” which is the term recommended for rodents. 139 Dogs have only a prostate and do not have seminal vesicles as opposed to rodents. Dilated acini are common, generally considered normal, and do not warrant diagnosis. Focal, cystic dilatation of acini in the canine prostate can be common. 144 When observed, the modifier term “cystic” should be considered.
Metaplasia, Squamous—Prostate (Figures 12.8–12.13)
Other Term(s)
Not applicable.
Pathogenesis
Replacement of the normal columnar epithelium by stratified squamous epithelium.
Diagnostic Features
Replacement of the normal prostatic glandular columnar epithelium by multiple layers of flattened epithelial cells.
Squamous metaplasia may preferentially affect the periurethral glands of the prostate rather than subcapsular glands. 157
Differential Diagnoses
Not applicable.
Comments
Replacement of the prostatic glandular epithelium by stratified squamous epithelium had been reported following treatment of dogs with estrogen. 157 –159 Squamous metaplasia of the prostatic epithelium may also be associated with inflammation.
Hyperplasia, Epithelium—Prostate
Other Term(s)
Diffuse hyperplasia.
Modifiers
Diffuse.
Pathogenesis
Proliferation of the epithelial cells lining the prostatic acini.
Diagnostic Features
Proliferation of acinar epithelium in a single layer that can be a diffuse change.
There may be papillary folding of the lining epithelium into the acinar lumina.
There may be cystic acini lined by flattened epithelium.
Differential Diagnoses
Dilatation, cystic: Focal cystic dilation of acini in the absence of epithelial hyperplasia elsewhere in the prostate.
Hyperplasia, atypical: A focal hyperplastic lesion with multilayered epithelium and cellular atypia.
Hyperplasia, reactive: Epithelial hyperplasia accompanied by inflammation.
Comments
“Hyperplasia, epithelium” is the preferred term over “hyperplasia, functional,” which was the term proposed for rodents, 139 but is no longer a preferred term. Prostatic hyperplasia has been reported as a common finding in mature beagles older than the young dogs generally included in routine toxicity studies. 160
13. Respiratory System
Introduction
Standardized INHAND nomenclature of proliferative and nonproliferative respiratory tract lesions in rats and mice was previously published. 161 This document follows a similar anatomical approach but will focus on lesions that are unique to the dog, particularly purpose bred beagle dogs commonly used in research laboratories. Consequently, the nasal cavity, larynx, trachea, bronchi, bronchioles, lung parenchyma, and pleura have been separated in this nomenclature scheme, even though there may be considerable redundancy regarding responses at various levels. For general considerations of the respiratory system, refer to the published rodent publication. 161 The standardized nomenclature for the dog, including respiratory tract lesions, presented in this document is also available electronically at the STP website 3 and in more detail at the goRENI web site. 2 The respiratory system is susceptible to injury caused by blood borne or inhaled xenobiotics and thus should be routinely examined in all toxicity studies. While the primary purpose of this publication is to provide a standardized nomenclature for classifying microscopic lesions, standardization of tissues sampled and examined is also important, particularly in inhalation studies. The respiratory tract is composed of multiple organs and ancillary tissues including the nasal cavity, nasopharynx, larynx, trachea, tracheal bifurcation, lung (7 lobes), and draining lymph nodes. The nasal cavity, larynx, and lung are complex organs which have multiple types of epithelia and specific anatomic structures that vary with location. Each of these organs, cells types, and structures must be individually examined for accurate assessment of the respiratory tract. For inhalation studies, it is recommended that tissues include 4 transverse levels of nasal cavity, 2 transverse levels of larynx, 1 or 2 transverse levels of trachea, 1 longitudinal (horizontal) section of carina, samples of all lungs lobes to include proximal and peripheral sections, and the tracheobronchial lymph nodes. Tissue sampling for noninhalation studies may be less extensive but should include at least 2 sections of lung (a proximal area near the main stem bronchi and an area at the periphery) and 1 section of trachea. One transvers section of the epiglottis and one transverse section of the oropharynx bordered by the tonsils may be (optionally) sampled as needed for specific studies. This sampling is illustrated in the Respiratory Collection Protocol of the Laboratory Dog, published by GlaxoSmithKline, Charles River Laboratories, and EPL (2006).
Nomenclature
Tables 13.1 to 13.7 summarize the microscopic findings that can be observed in the respiratory tract of dogs in preclinical toxicological studies. Basic terminology is based on the INHAND Rodent publications, with appropriate modifications and additions for the dog.
Nasal Cavity, Nasopharynx, and Paranasal Sinuses
The dog nasal cavity is anatomically more complex than humans, has a greater relative surface area (surface/volume), and has different amounts and distributions of epithelial cell types. 162 –164 The greatest difference is in the percentage of the nasal cavity lined by olfactory neuroepithelium. Overall, the dog nasal cavity is less similar to humans than nonhuman primates. 165 The degree of bend in the nasopharyngeal airways impacts local dosimetry of some inhaled agents. Rodents have little dorsoventral bend in the nasopharynx (15°), dogs have slightly more (45°), and primates have a marked bend (80° and 90° in macaques and humans, respectively). 164 In contrast to mice and rats, which are obligate nose breathers, the anatomy of the nasal and oral cavities of dogs, nonhuman primates, and humans allows for both nasal and oral breathing.
Microscopic Findings in the Respiratory System: Nasal Cavity, Nasopharynx, and Paranasal Sinuses; Dog.
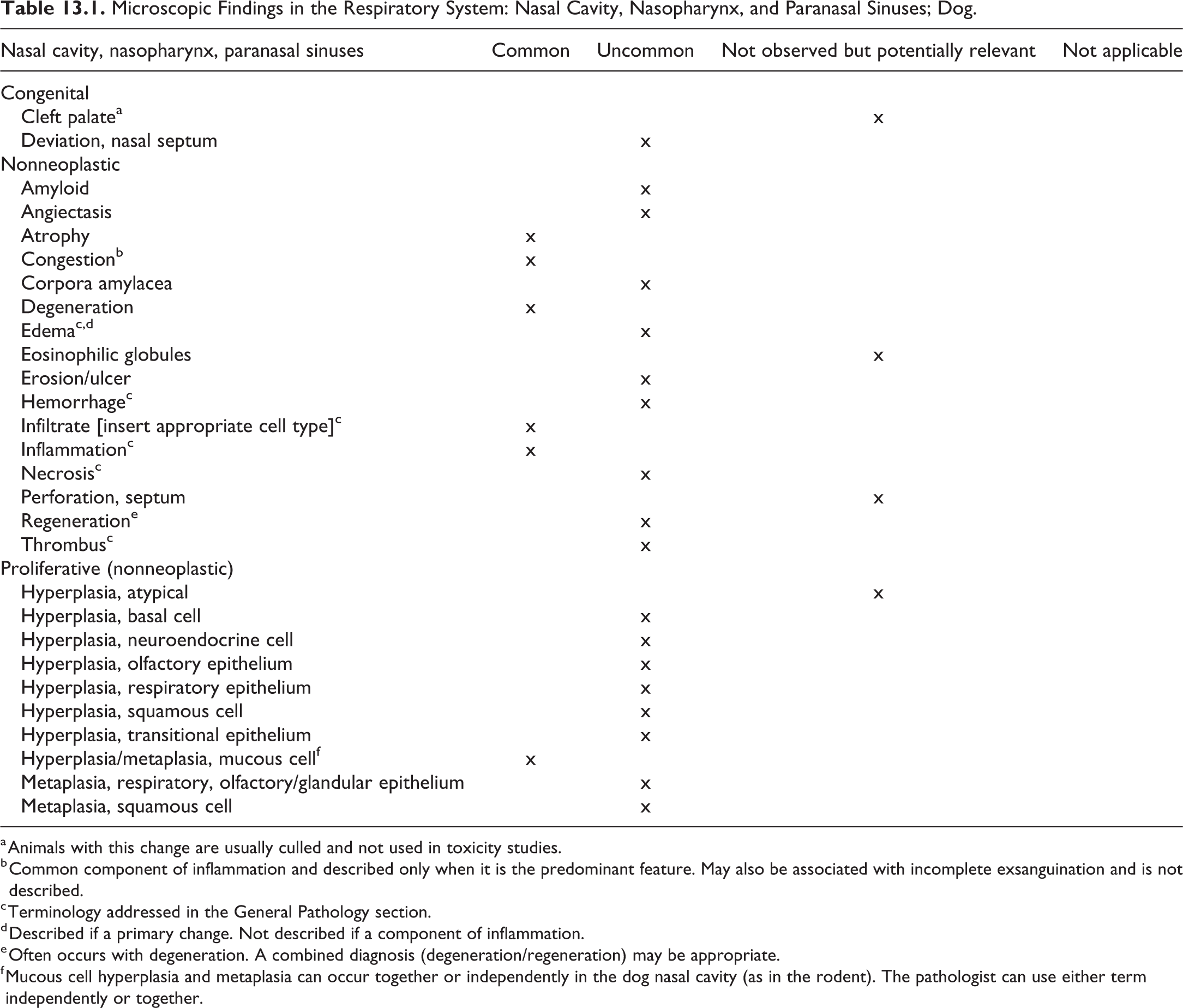
a Animals with this change are usually culled and not used in toxicity studies.
b Common component of inflammation and described only when it is the predominant feature. May also be associated with incomplete exsanguination and is not described.
c Terminology addressed in the General Pathology section.
d Described if a primary change. Not described if a component of inflammation.
e Often occurs with degeneration. A combined diagnosis (degeneration/regeneration) may be appropriate.
f Mucous cell hyperplasia and metaplasia can occur together or independently in the dog nasal cavity (as in the rodent). The pathologist can use either term independently or together.
Larynx and Carina
The larynx and carina in dogs are anatomically and histologically more similar to humans and respond to xenobiotics in a more similar fashion to humans than rats. 166,167
Microscopic Findings in the Respiratory System: Larynx; Dog.

a Common background finding. Describe only if greater severity/extent than commonly seen as an incidental background finding.
b Terminology addressed in the General Pathology section.
c Described if a primary change. Not described if a component of inflammation.
d Often occurs with degeneration. A combined diagnosis (degeneration/regeneration) may be appropriate.
e Often accompanied by hyperkeratosis. Hyperkeratosis may be the predominant feature and should be described in the narrative.
Microscopic Findings in the Respiratory System: Trachea; Dog.

a Common background finding. Describe only if greater severity/extent than commonly seen as an incidental background finding.
b Terminology addressed in the General Pathology section.
c Described if a primary change. Not described if a component of inflammation.
d Often occurs with degeneration. A combined diagnosis (degeneration/regeneration) may be appropriate.
Microscopic Findings in the Respiratory System: Bronchi; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Finding more frequent as an induced change in inhalation studies.
c Common background finding. Describe only if greater severity/extent than commonly seen as an incidental background finding.
d Terminology addressed in the General Pathology section.
e Described if a primary change. Not described if a component of inflammation.
f Often occurs with degeneration. A combined diagnosis (degeneration/regeneration) may be appropriate.
Microscopic Findings in the Respiratory System: Bronchioles; Dog.

a Finding more frequent as an induced change in inhalation studies.
b Terminology addressed in the General Pathology section.
c Described if a primary change. Not described if a component of inflammation.
d For a description, refer to Pigment/foreign material under Terminal Bronchioles or Alveoli of the rodent manuscript.
e Often occurs with degeneration. A combined diagnosis (degeneration/regeneration) may be appropriate.
f Terminology with diagnostic criteria and/or comments described in the text.
Microscopic Findings in the Respiratory System: Respiratory Bronchioles, Alveolar Ducts, and Alveoli; Dog.

a Diagnosis based on electron microscopy or immunohistochemistry.
b Terminology addressed in the General Pathology section.
c Common component of inflammation and described only when it is the predominant feature. May also be associated with incomplete exsanguination and is not described.
d Described if a primary change. Not described if a component of inflammation.
e Terminology with diagnostic criteria and/or comments described in the text.
f Common background finding as a minimal, focal, or multifocal change. Describe only if greater severity/extent than commonly seen as an incidental background finding.
g Suggested preferred terms: Fibrosis followed by locator or hemorrhage, followed by locator term.
h Alveolar hemorrhage is frequently accompanied by hemoglobin crystals and low numbers of macrophages and/or leukocytes (especially neutrophils). These are a component of hemorrhage and should not be recorded as separate findings.
i Use of the term “macrophage aggregation” is not recommended and was replaced by “macrophages increased”.
j Pigments such as anthracosis and hemosiderin are common background finding and should be described only if extensive.
k May be seen in large or small vessels including capillaries. Thromboemboli may be common in chronic infusion studies.
Microscopic Findings in the Respiratory System: Pleura; Dog.

a Terminology addressed in the General Pathology section.
Bronchi and Bronchioles
Pulmonary conducting airways of dogs are anatomically more similar to humans than rats and mice based on the presence (in dogs but not rats or mice) of cartilage and submucosal glands in intrapulmonary bronchi, of mucus cells as the primary secretory cell type in conducting airways, and the presence of several generations of respiratory bronchioles. 164 However, tracheobronchial branching patterns of laboratory animals differ from those of humans. Human tracheobronchial airways are dichotomous, that is, they are relatively symmetrical and daughter branches are of relatively equal diameter. In contrast, tracheobronchial airways of monkeys, rodents, and dogs are monopodial, that is, daughter branches are of unequal diameter and the smaller airway branches at a different angle from the larger airway. Humans and rodents have more generations of nonrespiratory bronchioles than dogs and monkeys. Club cells are present mainly in terminal and respiratory bronchioles of humans, monkeys, and dogs, but dog club cells are distinctive due to the large amount of glycogen in their cytoplasm. Overall, the epithelial types and distribution of secretory cells (mucus, serous, and club cells) lining airways are more similar in dogs to humans than those of rats or mice. 168
Aplasia/Hypoplasia Bronchial Cartilage—Lung–Bronchi
Pathogenesis/Cell of Origin
Congenital change. Aplasia, hypoplasia or dysplasia of bronchial cartilage.
Diagnostic Features
Lack of or incomplete development of the cartilage of the bronchial tree.
Absent, incomplete, rudimentary cartilaginous ring around bronchi.
Cartilage may be disorganized.
Bronchial glands and muscle fiber are present.
Bronchi are lined by a cuboidal to columnar epithelium which may be immature.
May be associated with alveolar emphysema, dilated bronchioles, and terminal airways.
Comment
This is a rare change that has been reported in nonlaboratory dogs. 169 –171 The change is often associated with dyspnea. Laboratory dogs with this change would likely be culled prior to being used in toxicity studies.
Hypertrophy/Hyperplasia, Smooth Muscle—Lung: Bronchi/Bronchioles
Pathogenesis/Cell of Origin
Smooth muscle cells in the submucosa of bronchi and bronchioles.
Diagnostic Features
Increased prominence of smooth muscle mass characterized by enlarged smooth muscle cells with or without increased nuclear density.
Smooth muscle cells are well differentiated and aligned appropriately.
Usually the thickened muscle layer is evenly distributed around the airways (depends on section).
May be associated with an adjacent inflammatory cell infiltrate.
No disruption, compression, or disturbance of adjacent tissues.
Differential Diagnoses
Tangential section through vessel: Other structures in section are cut tangentially, missing, or otherwise abnormal in shape.
Comment
Smooth muscle cell hypertrophy/hyperplasia is uncommon as a spontaneous finding but can be seen in cases of lung inflammation, allergic disease, and/or immune stimulation, especially following inhalation administration of certain xenobiotics. 172 Often it is observed as a focal/multifocal change rather than diffusely present throughout all areas of the parenchyma.
Fibrosis/Epithelial Hyperplasia—Lung: Alveoli (Figure 13.1 and 13.2)
Other Term(s)
Fibrosing alveolitis, segmental fibrosis, segmental subpleural septal fibrosis; these terms are no longer preferred.
Pathogenesis/Cell of Origin
Fibroblasts, bone marrow-derived inflammatory cells. Usually accompanied by epithelial hyperplasia.
Diagnostic Features
Areas of alveolar septal thickening due to fibrosis and interstitial inflammation. Thickened septa are typically lined by hyperplastic epithelium.
Subpleural location, extending from pleura into pulmonary parenchyma. May be wedge-shaped with pleural fibrosis.
Increased alveolar macrophages and squamous metaplasia may be present.
The predominance of inflammation and epithelial hyperplasia may vary with lesion age such that fibrosis and inflammation are the main features in some lesions whereas fibrosis and epithelial hyperplasia predominate in others.
Comment
Common background finding in beagle dogs. Unique to dogs. Differs from pulmonary fibrosis in its consistent subpleural location and characteristic alveolar septal fibrosis with epithelial hyperplasia. A high incidence of this finding, unrelated to test article administration, may be misinterpreted by pathologists unfamiliar with background pathology in laboratory beagles when it occurs in treated animals. 173 Possibly related to old infections with Filaroides or Toxicara species. 60
Macrophages, Increased—Lung: Alveoli (Figure 13.3 and 13.4)
Other Term(s)
Alveolar macrophage aggregation, alveolar macrophage accumulation, alveolar histiocytosis.
Pathogenesis/Cell of Origin
Bone marrow-derived monocyte precursors; resident interstitial and alveolar macrophages proliferate locally in adults.
Diagnostic Features
Increased numbers of alveolar macrophages over and above the expected number of randomly scattered individual macrophages within the alveolar spaces.
Often aggregated in multiple foci. However, some of the apparent aggregation and location may be an artifact of intratracheal instillation of fixative.
Comment
The term increased alveolar macrophages is intended to replace alveolar macrophage aggregation in the rat INHAND document, particularly for inhalation studies. 174 It is preferable to use increased alveolar macrophages as a stand-alone diagnosis and not as an umbrella term that includes other inflammatory, degenerative, or proliferative responses. The term also pertains to lesser numbers of interstitial macrophages that might accompany increased alveolar macrophages. The affected compartment should be addressed in the report narrative. Increased interstitial macrophages should only be diagnosed separately if the pulmonary interstitium represents the predominant location. If the macrophages from treated animals have cytoplasmic changes (pigment, vacuolation, foreign material, etc) that are distinct from features in controls (historical or current), these changes should be part of the diagnostic term. The term alveolar phospholipidosis is not a synonym for increased alveolar macrophages and should be reserved for accumulations of phospholipid-containing macrophages observed after treatment with cationic amphiphilic drugs. 175,176
Metaplasia, Osseous—Lung: Alveoli
Other Term(s)
Ectopic bone, ectopic ossification, pulmonary ossification, pulmonary osteophytes.
Pathogenesis/Cell of Origin
Transformation of differentiated mesenchymal cells into bone cells.
Diagnostic Features
Nodule(s) of bone tissue within the parenchymal interstitium.
Bone marrow may be present.
Differential Diagnoses
Mineralization: No differentiated bone tissue, that is, no cellular component.
Comment
Osseous metaplasia of the lung is an uncommon incidental finding in dogs. 49,60 The change is similar to that seen in rats but occurs at a lower incidence in dogs than in rats.
14. Skeletal System and Tooth
Introduction
For detailed general considerations of the skeletal system, please refer to the rodent publication. While the type and classification of bone and joints lesions is identical or very similar across species, their frequency in untreated individuals may differ substantially in the various species.
The standard bone samples examined during a toxicity study in dogs varies among institutions; they usually include a section of sternum or rib and a section of a long appendicular bone with the articular surface. The selected long bone should be trimmed longitudinally to include a section of the articular cartilage, epiphysis, growth plate, metaphysis, and cortical bone in the diaphysis. The distal femur with or without the proximal tibia, the femorotibial joint, or the proximal tibia are good options for evaluation of long bone in dogs. In standard toxicity studies, bone tissue is decalcified before it is subjected to the usual processing in the histology laboratory. Undecalcified sections may be prepared if the cortical remodeling, bone formation rate, and/or status of mineralization needs to be addressed.
The discussion here is focused on spontaneous lesions observed in young dogs (up to 2 years of age) used in preclinical studies. The assignment to the categories “common” and “uncommon” is subjective to a certain extent and refers to the incidence of lesions in untreated beagle dogs used in toxicity studies. Generally, nonproliferative skeletal abnormalities are uncommon spontaneous findings in young dogs and proliferative neoplastic lesions are rarely observed as background findings in routine toxicity studies due to the young age of animals and the short duration of studies compared to the dog’s natural life span.
Fractures in the bones are very rarely observed and joint lesions, more frequently seen in older dogs with a predilection in certain breeds, are also uncommon spontaneous findings in untreated young beagle dogs.
Bone
The recommended nomenclature of nonneoplastic and neoplastic bone changes in the dog is based on the INHAND manuscript for rodents and the online publication of the goReni website. Comments specific to the dog were added to the terms increased bone, trabeculae, and/or cortex and osteosarcoma. Terms added to the dog nomenclature that were not considered pertinent to the rodent included bone increased, trabeculae and/or cortex, fibrous dysplasia, and growth plate closed/partially closed/open. The recommended nomenclature is presented in Table 14.1. For a description of the neoplastic changes not pertinent to rodents but potentially to dogs, refer to the Histological Classification of Bone and Joint Tumors of Domestic Animals published by the Armed Forces Institute of Pathology in conjunction with the American Registry of Pathology and the World Health Organization, 177 the textbook Tumors of Domestic Animals, 178 and /or current literature. Genetic and congenital diseases were considered out of scope for this tabulation.
Microscopic Findings in the Skeletal System: Bone; Dog.
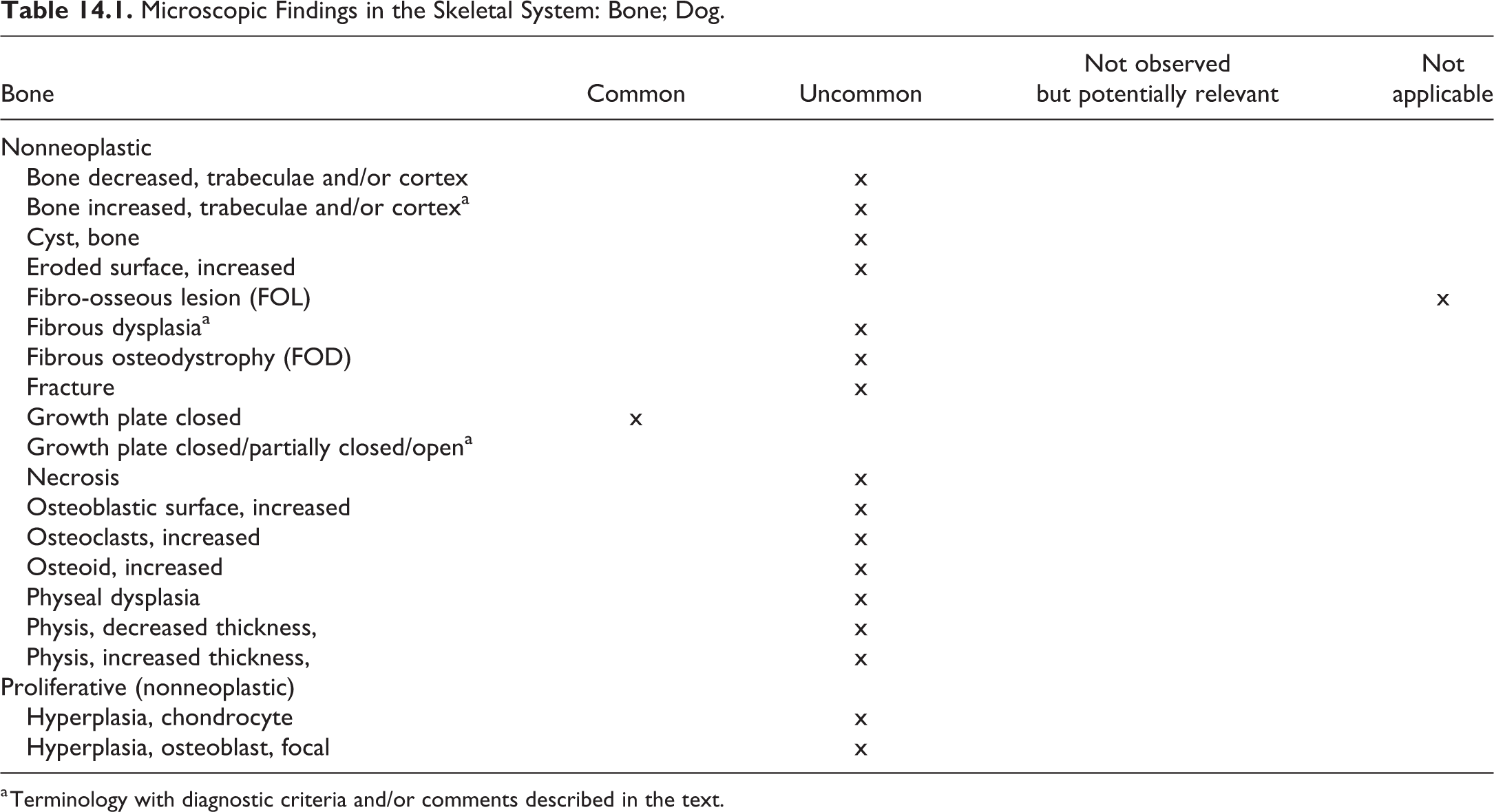
a Terminology with diagnostic criteria and/or comments described in the text.
Bone, Increased, Trabeculae, and/or Cortex—Bone
Comment
In the dog, there are a number of conditions that are characterized by excessive bone formation, “hyperostosis,” including craniomandibular osteopathy, hypertrophic osteopathy (HO), panosteitis, and canine hepatozoonosis. While craniomandibular osteopathy and panosteitis (also referred to as enostosis, eosinophilic panostitis, juvenile osteomyelitis, or fibrous osteodystrophy) have been described in other breeds than the beagle and canine hepatozoonosis infection should not occur in in-door kept animal colonies, HO could theoretically affect a beagle dog. Typically, however, it is observed in large breeds and boxers; to the author’s knowledge it has not been observed in toxicity studies with beagle dogs. Salyusarenko et al 179 characterized HO as pathologic periosteal bone formation, which is observed predominantly in the metacarpi or metatarsi (76%), distal and proximal long bones (66% and 38% respectively), carpi and tarsi (31%), and phalanges (21%). Hypertrophic osteopathy is described to occur secondary to other conditions, mostly pulmonary or intrathoracic neoplasms. 179,180 Dogs with primary lung neoplasia or osteosarcoma metastases in the lungs appear to be most often affected. The pathogenesis of HO is not known, a number of theories exist, increased limb blood flow is a consistent early change, and induction of vascular endothelial growth factor via cyclo-oxygenase-2 derived increase in prostaglandin E2 appears to be involved. 181 –183 Changes accompanying increased bone, for example, inflammation, should be either recorded separately or their occurrence and nature could be mentioned in the narrative of the report.
Fibrous Dysplasia—Bone
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Disorder of development of bone, associated with mutations of the GNAS1 gene 184 ; all the components of normal bone are present but they do not differentiate into their mature structures (ie, no rimming of trabeculae by osteoblasts and no evidence of lamellar replacement of woven bone).
Diagnostic Features
Well-circumscribed intramedullary lesions that can expand and distort the bone.
Fibrous dysplasia can involve a single bone (monostotic) or several bones (polystotic).
Characterized by disorganized trabeculae of woven bone that lacks conspicuous osteoblastic rimming, surrounded by a moderately cellular fibroblastic proliferation.
Nodules of hyaline cartilage may be present.
Cystic degeneration, hemorrhage, and foamy macrophages can occur.
Differential Diagnoses
Fibrous osteodystrophy: generalized condition; osteoblastic rimming present.
Ossifying fibroma: localization in the jaws; presence of osteoblastic rimming around trabeculae.
Comments
Fibrous dysplasia has been reported in a number of species, including the dog, and is usually observed as a solitary lesion but also has been reported in animals and man in the polyostotic form. It is a rare, nonaggressive, radiolucent, nonneoplastic lesion considered developmental in origin. 185 –187
Growth Plate Closed, Partially Closed, Open—Bone
Other Term(s)
Physis closed, partially closed, open.
Pathogenesis/Cell of Origin
Ossification of the growth plate
Diagnostic Features
Thickness of the physis depends on the rate of growth.
When growth is slowing, the layers of the physis narrow.
At the end of the process, the cartilage of the growth plate is replaced by a bony scar, which is remodeled into trabecular bone.
Differential Diagnoses
Not applicable.
Comment
Unlike the rodent, the physis of dogs closes when the animal reaches maturity and growth ceases. Usually, it is not considered necessary to record the status of physis closure in a standard toxicity study. However, the recording of physis closure may become necessary if a treatment causes or is considered to possibly cause a delay in physis closure, a premature physis closure, or maturity of other organs (eg, male and female genital tract organs) have to be compared against skeletal maturity.
In the untreated, healthy dog, growth plate closure of tibia and femur occurs at the age of 6 to 11 months 26 ; the closure of the distal femur physis in the beagle occurs between 250 and 325 days of age (8-11 months) in females slightly earlier than in males. 188 Similar data of epiphyseal closure of the distal femur are given by Zoetis et al 189 ; their publication provides a detailed survey of skeletal maturation and postnatal bone growth of femur and humerus across species including humans and dogs.
Joint
The recommended descriptive nomenclature for lesions in the joint of dogs is summarized in Table 14.2, which is based on the INHAND rodent nomenclature. It should be noted, however, that in beagle dogs used in toxicity studies, spontaneous lesions of the joints are rare, and neoplastic joint lesions are extremely rare. For a histomorphologic description of neoplasms not pertinent to rodents but potentially to dogs, refer to the WHO fascicle on Histological Classification of Bone and Joint Tumors of Domestic Animals, 177 the chapter on joint tumors 190 in the textbook Tumors of Domestic Animals, and/or current literature.
Microscopic Findings in the Skeletal System: Joint; Dog.

aTerminology with diagnostic criteria and/or comments described in the text.
b Finding more frequent as an induced change.
c Terminology addressed in Systemic Pathology section.
Degeneration, Articular Cartilage—Joint
Other Term(s)
Arthropathy
Pathogenesis/Cell of Origin
Articular cartilage; degenerative changes due to administration of xenobiotics.
Diagnostic Features
Vesicle and/or fissure formation in the articular cartilage.
Accumulation of fluid in vesicles, accumulation of debris within fissures (eg, collagen fibrils, eosinophilic material, granular extracellular matrix material).
Spindle-shaped cells adjacent to the fissures.
Degenerative changes in chondrocytes, for example, shrunken cytoplasm; shrunken, pyknotic nuclei; staining abnormalities such as poor staining of chondrocytes; dark purple matrix or lighter purple matrix (toluidine blue stain).
Flap formation or loss of superficial cartilage.
Clusters of chondrocytes, indicating regeneration.
Differential Diagnoses
Spontaneous degeneration; occurs in older dogs.
Comment
Quinolone arthropathy represents a typical example of drug-induced joint degeneration affecting major synovial joints; it has been observed in many species including the dog. 191 –193
Tooth
Teeth are not a part of the routine histopathological evaluation in dog toxicity studies but may be added to the spectrum of organs to be examined if considered necessary, for instance, if they are the subject of investigation. Dogs, in contrast to rodents, are diphyodont with deciduous and permanent dentition. Shabestari et al 194 describe the dental eruption time specifically in in beagle dogs; the deciduous dental formula is I 3/3, C1/1, M 3/3; the permanent dental formula is I3/3, C1/1, PM 4/4, M 2/3. Tooth lesions are uncommon in beagle dogs, 195 in part because the teeth are rarely examined and, in toxicity studies, the dogs are of young age. The incisor, canine, or mandibular tooth can be decalcified and evaluated if needed.
The recommended descriptive nomenclature for tooth lesions is presented in Table 14.3, which is based on the INHAND rodent nomenclature. Added terminology for the dog is limited to inflammation, periodontal (periodontal disease), which represents the most common dental disease in dogs, although it is rarely observed in young beagles used in toxicity studies. For histomorphologic criteria of dental neoplasms, refer to the WHO fascicle on Histological Classification of Tumors of the Alimentary System of Domestic Animals, 24 the chapter on tumors of the alimentary tract 25 in the textbook Tumors of Domestic Animals, and/or current literature.
Microscopic Findings in the Skeletal System: Tooth; Dog.
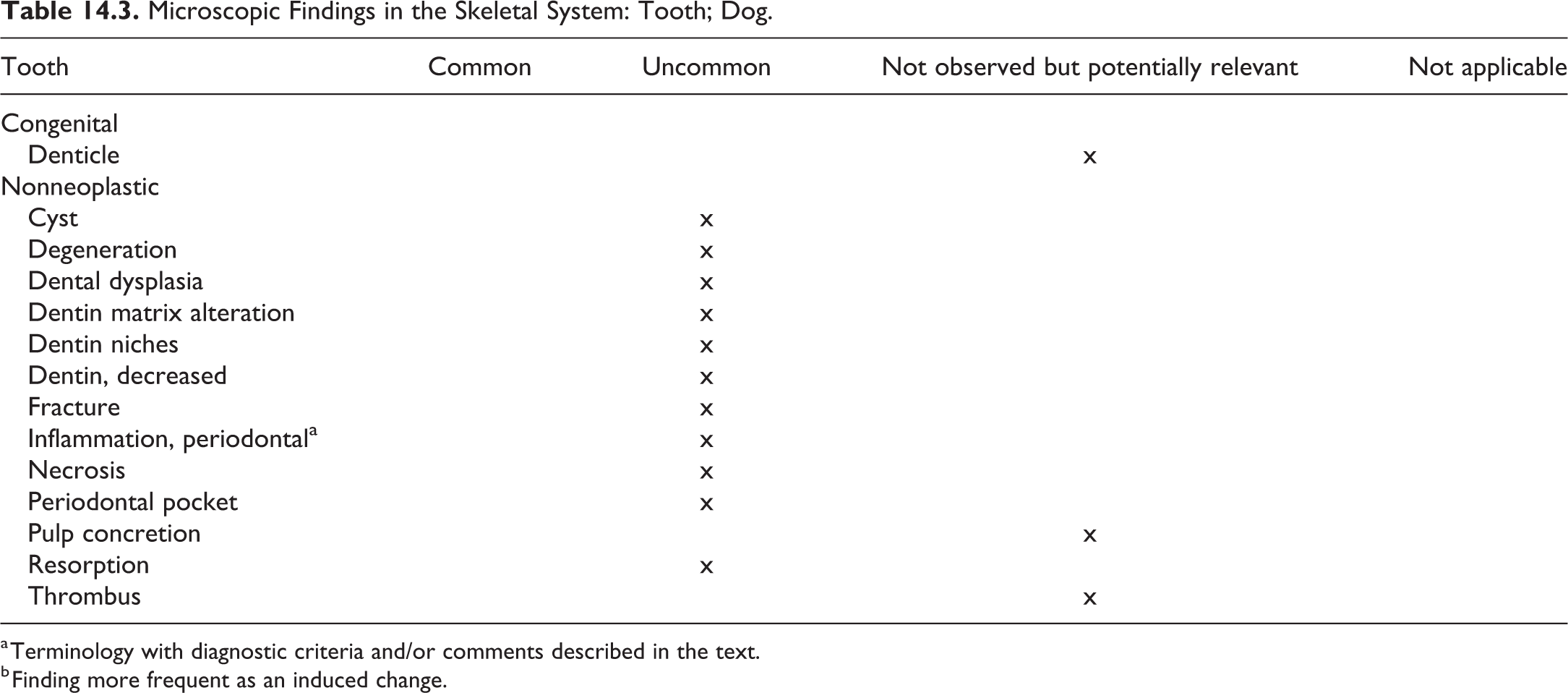
a Terminology with diagnostic criteria and/or comments described in the text.
b Finding more frequent as an induced change.
Inflammation, Periodontal—Tooth
Other Term(s)
Gingivitis
Periodontal disease
Pathogenesis
The process is usually initiated by plaque (tartar) accumulation and starts as acute inflammation (increased number of neutrophils and increased fluid); later on, plasma cell and macrophage accumulation follow.
Loss of gingival collagen fibers, caused by cytokines and enzymes of the plaque bacteria.
Apical recession of the tooth–gingiva junction and resorption of alveolar bone.
Destruction of the periodontium and periodontal ligament.
Resorption of alveolar bone, cementum, and root dentin lead to exfoliation of teeth.
Diagnostic Features
The diagnosis is in most instances readily made grossly (plaque formation, reddening, and swelling of the gingiva). Microscopically, it is characterized by the inflammatory process and the associated inflammatory cell infiltrates.
15. Soft Tissue and Skeletal Muscle
For general aspects of soft tissue and muscle lesions, please refer to the rodent manuscript. 196 Soft tissue and skeletal muscle lesions observed in the dog are generally identical to those observed in rodents and other domestic animals as well as those in humans. 196 Therefore, the nomenclature of nonneoplastic and neoplastic soft tissue and skeletal–muscle changes described in the INHAND manuscript for rodents and the structure of the rodent classification were adopted for the dog. Included are lesions of the soft and adipose tissue, smooth and skeletal muscle, and the mesothelium. Lesions of the synovium are described in the chapter on the skeletal system. For detailed general considerations on the soft tissue and skeletal muscle, please refer to the rodent publication.
While the type and classification of soft tissue lesions is identical or very similar across species, their frequency in untreated individuals may differ substantially in the various species. The assignments to the categories “common” and “uncommon” are subjective to a certain extent and refer to the incidence of lesions in untreated beagle dogs used in toxicity studies. The incidence may increase significantly under certain treatment regimens, such as granuloma formation observed in the subcutaneous connective tissue of dogs after the implantation of microchips 197 or skeletal muscle hypertrophy following inhibition or blockade of myostatin gene. 198
Soft Tissue and Adipose Tissue
The following tabulations (Tables 15.1 and 15.2) summarize the nonneoplastic lesions observed in the soft and adipose tissue and the smooth muscle of dogs. The terminology is based on the INHAND rodent terminology, and modifications or additions for the laboratory beagle dog were not considered necessary or adequate. As noted in the rodent manuscript, the careful recording of the nature, intensity, and duration of the inflammatory response in soft tissues to implants or injected substances is important for the assessment of their local tolerability. 196
Microscopic Findings in the Soft Tissue: General Terminology; Dog.
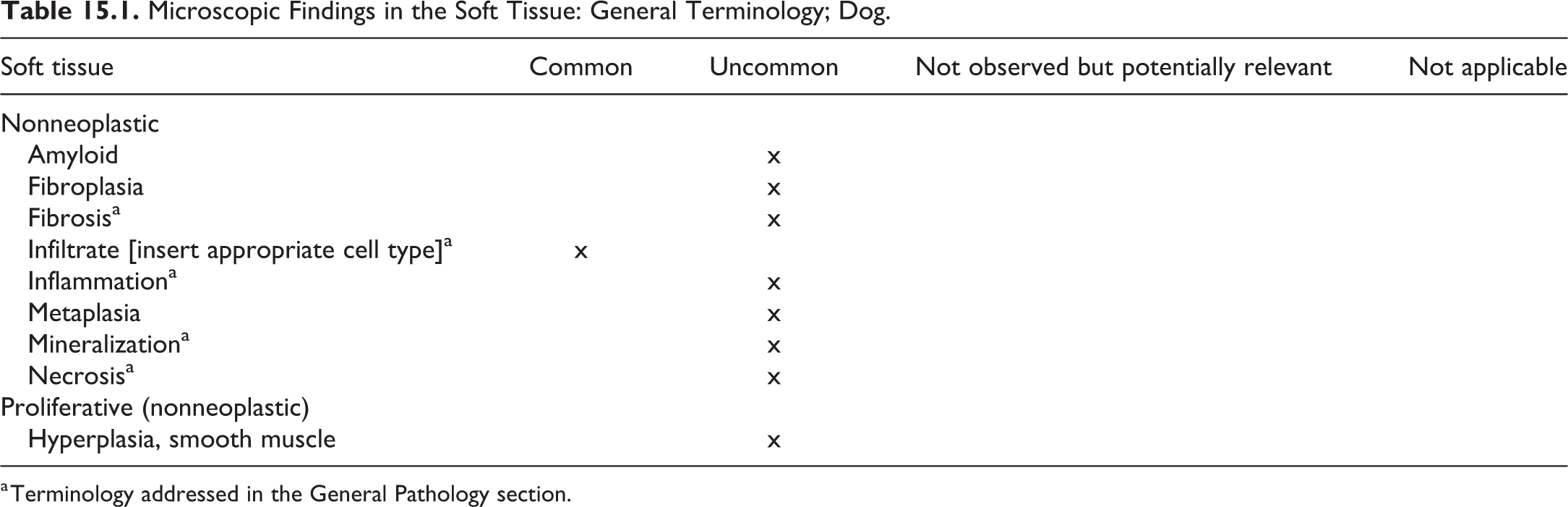
a Terminology addressed in the General Pathology section.
Microscopic Findings in the Soft Tissue: Adipose Tissue; Dog.

a Terminology addressed in the General Pathology section.
Skeletal Muscle
The special cellular structure of skeletal muscle, the different fiber types, and their varying sensitivity in the response to xenobiotics require a separate consideration of the pathological changes and also the extent of sampling and careful selection of sampling sites. 196 Skeletal muscle fiber types are characterized by the histochemical reaction for adenosine triphosphate and the pH lability of this reaction. 199,200 Species-specific type II subtypes have been identified for the dog: II A, II dog (unique to the dog), and II C. 201 An investigation based on reverse transcription polymerase chain reaction and confirmatory histochemistry, immune histochemistry, and sodium dodecyl sulfate polyacrylamide gel electrophoresis showed that limb and trunk muscles of dogs express myosin heavy chain (MHC) types 1, 2A, 2X, and that the so-called type 2 dog fibers express MHC-2X. MHC-2B type fibers were found in extraocular and laryngeal muscles. 202
Table 15.3 summarizes the lesions observed in the skeletal muscle followed by changes in the mesothelium (Table 15.4). The nomenclature applies to rodents and other laboratory animal species including the dog.
Microscopic Findings in the Soft Tissue: Skeletal Muscle; Dog.

a Terminology addressed in the General Pathology section.
b Terminology with diagnostic criteria and/or comments described in the text.
Microscopic Findings in the Soft Tissue: Mesothelium; Dog.

Regeneration—Skeletal Muscle
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Repair process following muscle injury
Diagnostic Features
Increased basophilia
Centrally located nuclei
Chains of nuclei
Disorganized appearance of muscle fibers in the case of tissue disruption
Muscle giant cells in the case of tissue disruption, indicative of abortive regeneration
Differential Diagnoses
Not applicable.
Comment
Skeletal muscle has a high regenerative potential. The process of skeletal muscle regeneration can be divided into 3 phases: inflammatory, satellite cell activation/differentiation, and maturation. 203 The inflammatory phase follows muscle injury and is characterized by initial infiltrates of neutrophils, followed by pro-inflammatory macrophages and anti-inflammatory macrophages, which are essential for the activation of satellite cells. Satellite cells are considered the myogenic stem cells; upon activation, they divide asymmetrically into new stem cells and myogenic progenitor cells (2nd phase). Eventually, the progenitor cells fuse into multinucleated myofibers with centrally located nuclei. During maturation (3rd phase), the nuclei move toward the periphery. 203,204
Mesothelium
In addition to the general terminology of soft-tissue lesions, findings unique to the mesothelium are limited to hyperplasia (Table 15.4).
Neoplastic Proliferative Lesions
In the experience of the authors, the cutaneous canine histiocytoma and the lipoma of the serosa represent relevant neoplastic findings, that is, these have been noted in the soft tissues in toxicologic pathology study in dogs; they are presented in Table 15.5. For a histomorphologic description of neoplasms, refer to the WHO fascicle on mesenchymal tumors of the skin and soft tissues, 95 the chapter on soft tissue neoplasms 99 in the textbook Tumors of Domestic Animals, and/or current literature.
Microscopic Findings in the Soft Tissue: Relevant Neoplastic Proliferative Lesions; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b For a histomorphologic description of lipoma, refer to the rodent manuscript, the WHO fascicle on mesenchymal tumors of the skin and soft tissues, the textbook Tumors of Domestic Animals 1, and/or current literature.
Histiocytoma, Canine Cutaneous—Skin (Figures 8.1–8.3; Section on Integument)
Other Term(s)
Not applicable.
Pathogenesis/Cell of Origin
Langerhans cells.
Diagnostic Features
Grossly, a dome-shaped protrusion of the skin, hairless, often abraded, and ulcerated.
Dermal (and epidermal) infiltrate of densely packed, mildly pleomorphic round cells, arranged in cords and nests.
Histiocytic-like neoplastic cells, ovoid, bean-shaped, or convoluted nuclei, lightly eosinophilic cytoplasm.
Often numerous mitotic figures.
Typically, wedge shaped, that is, narrow basis and wider near the epidermis, 95 “top heavy” epidermal focus. 27,96
Little or no stroma.
Immunohistochemistry provides the definitive proof of the diagnosis 97 ; canine cutaneous histocytomas are considered a Langerhans cell tumor and express CD1a, MHC II, CD11c, CD18, and often E-cadherin. 96,98
Differential Diagnoses
Epitheliotropic T-cell lymphoma: When considered because of epithelial involvement, differentiation is best done by IHC.
Plasmacytoma: Sheets of round cells, in nests or poorly defined cords, pleomorphic nuclei, mono-, bi-, or multinucleated, no or few mitoses, scant to moderate eosinophilic or basophilic cytoplasm, amyloid may be present. The IHC may be required for definite differential diagnosis; MUM1 is considered the most reliable marker. 99
Cutaneous histiocytosis: Coalescence of perivascular infiltrates in the subcutis and deep dermis without involvement of epidermis.
Comment
Usually a solitary lesion of young dogs, less than 3 years old, but has been observed in dogs of all ages. The lesion occurs in all breeds, predisposed are brachycephalic breeds, including boxers, but also dachshund, Doberman pinschers, and cocker spaniels. Spontaneous regression is a feature. 96,100
16. Special Senses—Eye and Glands
Introduction
For general aspects of special senses organs, refer to the rodent manuscript. The suggested standardized nomenclature for the ocular system is subdivided into the eye and the glands of the eye. The ear is not examined routinely in toxicologic pathology studies and is not included. Recommended nomenclature for microscopic lesions in the nose is addressed in the respiratory chapter.
Eye
The eye is a complex organ, and comprehensive review of the toxicological response of each component is beyond the scope of this manuscript. In brief, however, the eye is composed of tissues of ectodermal, neuroectodermal, and mesenchymal origin; therefore, abnormalities in development of any of them may contribute to the abnormal development of adjacent, sequentially developed tissue. Thus, this organ is particularly sensitive to in utero, as well as postnatal, toxic and teratogenic insults. In addition, the avascular nature of the lens and cornea and their resultant dependence on the aqueous humour for nutrition, as well as the immunoprivileged nature of the eye itself, are all considerations in the analysis of xenobiotic-related toxicity of the eye. Extraorbital glandular tissue, eye lids, muscles, and other adnexa develop independently of the globe, although toxic damage to these tissues may affect the health of the eye, making evaluation of the adnexa an important component of toxicity studies. (For example, cytotoxicity in the lacrimal gland may result in decreased tear production with subsequent damage to the cornea.) Systemic or topical administration of a drug or compound may result in toxicologically relevant ocular exposure, potentially compromising not just the structural but also the functional competency of this organ, so microscopic evaluation of the eye should generally be supplemented with clinical and ophthalmologic examination for complete analysis. In addition, the intimate association between the retina and the central nervous system may result in changes within the globe following exposure to certain neurotoxicants.
At necropsy, the eye should be enucleated and placed in an appropriate fixative immediately after euthanasia. Davidson’s fluid is most commonly recommended for fixation of the eye, as this fixative is particularly good for preservation of retinal architecture. Formalin and Bouin’s fixative have also been used in the past, but formalin fixation is not optimum for preservation of retinal detail, and Bouin’s fixative contains picric acid and provides a health and safety hazard, if not handled correctly. Some pathologists prefer injection of a small amount of fixative into the globe in addition to immersion fixation; however, for routine toxicology studies, this method is usually not necessary. Paraffin-embedded, H&E-stained sections of eye, trimmed in a sagittal orientation at the midline of the eye to capture the optic nerve/optic cup, are usually adequate for routine histological examination.
Adnexal lacrimal glands are present in the dog in both the orbit and within the third eyelid, referred to in the latter location as the gland of the third eyelid or the nictitans gland. Please note that Harderian glands, as recognized in the rodent, are not present in dogs. Also note that the tapetum lucidum, a reflective layer of the choroid, is well developed in the canine eye and is generally viewed in routine vertically oriented histologic sections.
The recommended nomenclature of lesions in the eye of dogs is presented in Table 16.1.
Microscopic Findings in Special Sense Organs: Eye; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
c Finding more frequent as an induced change in inhalation studies.
d Common artifact.
Adhesion, Iris—Eye: Uvea, Iris, and Ciliary Body
Comment
While recognized uncommonly as a congenital finding in young beagle dogs, adhesion between the anterior edge of the iris and the endothelium of the cornea (anterior synechia) or the posterior edge of the iris and the anterior surface of the lens (posterior synechia) may also be acquired, most commonly as a sequela to anterior uveitis, trauma, or other inflammatory processes.
Infiltrate, Mixed Cell, Conjunctiva—Eye: Conjunctiva (Figure 16.1)
Dog, prostate, squamous metaplasia, H&E, high magnification. Courtesy of Eric van Esch.
Comment
Within the conjunctiva, mononuclear or lymphoid cell infiltrates may not be indicative of an inflammatory process but may instead represent CALT, a normal structure of the conjunctival mucosa, particularly prevalent on the bulbar surface of the third eyelid. 205,206 With chronic antigenic stimulation, lymphoid hyperplasia may occur in CALT structures.
Cyst—Eye: Uvea, Iris, and Ciliary Body
Other Term(s)
Not applicable.
Diagnostic Features
Fluid-filled structure lined by 1 to 2 layers of variably pigmented cuboidal uveal epithelial cells supported by a thin fibrous capsule.
Cysts may be ruptured during processing.
Differential Diagnosis
Uveal melanoma: not usually cystic. Commonly comprised of plump pigmented cells often with lesser numbers of spindloid cells.
Comment
Although not commonly reported as a congenital finding in laboratory beagles, uveal cysts may be noted in any breed of dog as a congenital or acquired finding. 207 Cysts arise from the posterior iris or ciliary body epithelium and may remain attached to the point of origin, or they may detach and be observed in the anterior chamber or adhered to the cornea, iris, or lens. 208
Degeneration, Microcystoid—Eye: Retina
Other Term(s)
Not applicable.
Diagnostic Features
One to multiple variably-sized spaces within the inner nuclear and plexiform layer that disrupt the retinal architecture.
Rupture can result in retinal atrophy.
Differential Diagnosis
Not applicable.
Comment
Most commonly observed in middle-aged or older beagle dogs as an incidental finding. 209 If encountered in a younger population, careful consideration should be made as to whether the observation is test article-related or spontaneous.
Degeneration, Tapetum Lucidum—Eye: Retina (Choroid)
Other Term(s)
Atrophy, tapetum lucidum.
Diagnostic Features
Changes in tapetal cells may include swelling, vacuolation, necrosis, or a complete loss of cells.
The alterations in tapetal cells may be associated with inflammatory changes.
Differential Diagnosis
Not applicable.
Comment
Ophthalmoscopic examination of eyes with tapetal degeneration typically shows loss of tapetal reflectivity with the development of focal, mottled, or diffuse zones of pigmentation. 210 Spontaneous focal tapetal degeneration in untreated dogs may rarely be seen, 211 but it is more likely to occur as a treatment-related change. It has been reported following administration of a number of different classes of therapeutic agents. 210,212,213 Due to the absence of a tapetum lucidum in humans, tapetal changes in dogs without alterations in other structures of the eye may not be relevant for humans.
Hyperplasia, Meibomian Gland—Eye: Eyelid
Other Term(s)
Hypertrophy/hyperplasia, Meibomian gland; hypertrophy, Meibomian gland.
Diagnostic Features
Enlargement of glands with increased number of sebaceous cells in acini, surrounded by basal (reserve) cells.
Differential Diagnosis
Adenoma, Meibomian gland: Regular architecture is distorted; basal cells may be prominent and growth pattern exophytic.
Eye Glands
The recommended terminology for microscopic lesions observed in the eye glands of dogs is presented in Table 16.2.
Microscopic Findings in Special Sense Organs: Eye Glands; Dog.
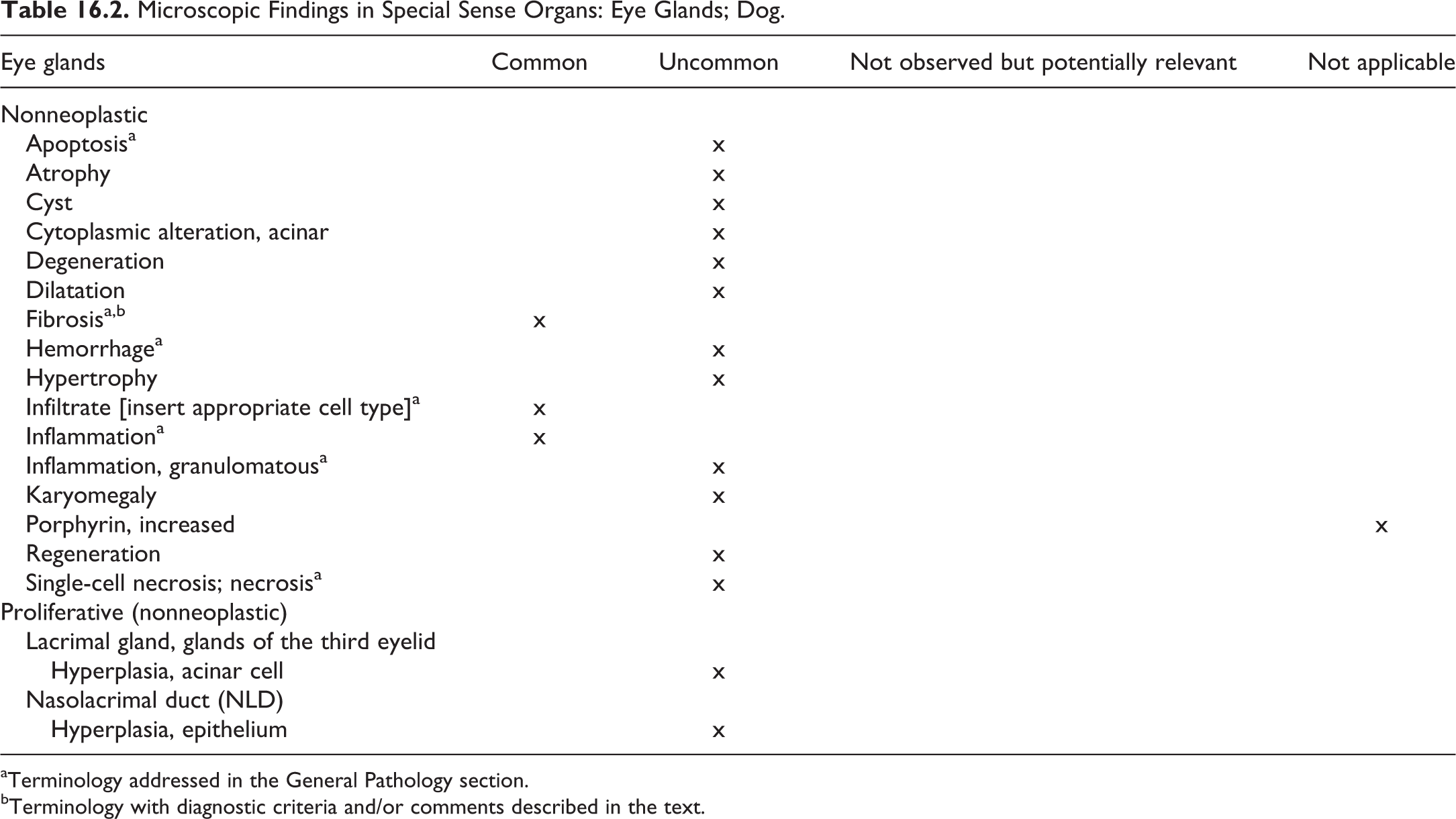
aTerminology addressed in the General Pathology section.
bTerminology with diagnostic criteria and/or comments described in the text.
Fibrosis: Lacrimal Gland
Comment
Focal fibrosis of the lacrimal gland, associated with mononuclear or lymphoid infiltrate and/or acinar atrophy, was reported as an occasional incidental finding in the beagle by Sato et al (Table 16.2). 49
17. Urinary System
Introduction
The urinary system pathology has been covered in detail in rodent INHAND document as well as in other publications. This chapter will provide a brief overview of the anatomical differences in the dog and recommended terminology and diagnostic criteria, along with selected representative images for the urinary system in the laboratory beagle dogs in preclinical toxicity studies. Readers are directed to the rodent INHAND for more in-depth review of histology and physiology of the urinary tract.
Understanding the mechanism, pathogenesis and importance of xenobiotic-induced changes in the urinary system requires accurate morphologic descriptions of those changes; therefore, a working understanding of the anatomy and physiology of the kidney is important. As in the rodent, the urinary system of the dog is comprised of paired kidneys and ureters, the urinary bladder, and urethra. The general anatomic locations of these organs in the retroperitoneum of the lumbar region of abdomen and pelvic cavity are similar to the rodent and other mammalian species. There are, however, 2 major anatomical features unique to the canine, compared to the rodent. These are comprised of the complete fusion of medullary pyramids resulting in the formation of a renal crest and the expansion of the proximal end of the ureters to form the renal pelvis. The functional consequence of these subtle anatomic differences is limited and there are no other significant anatomic differences of the canine urinary system (kidneys, ureters, and urinary bladder) compared with the rodent. 214,215 Compared with mice, where embryogenesis of the kidney is complete at birth, the production and maturation of new nephrons continues to postnatal day (PND) 10 in rats and PND 21 in dogs. 216
Kidney
The nephron, the functional unit of the kidney, consists of the renal corpuscle with the glomerulus, Bowman’s capsule, and the renal tubule, which encompasses the proximal convoluted tubule, the loop of Henle, the distal convoluted tubule, and the collecting tubule or duct which terminates at the renal calyx in the renal papilla. The nephron is supported by the interstitium. The counter current exchanger that establishes increasing osmotic gradient in the medulla interstitium is critical to urine concentration. The filtration unit of the nephron, the glomerulus, is a complex structure composed of a cluster of fenestrated capillaries closely adhered with visceral epithelia (podocytes) within an expanded sphere (Bowman’s capsule) with adjacent mesangial cells; the 2 cell types contribute to a negatively charged basement membrane which repels positively charged proteins. 215
Ultrafiltrate, composed of water, nitrogenous waste products, and small proteins (<60 kDa), exits Bowman’s capsule and enters the proximal convoluted tubule. “Fetal glomeruli” (ie, immature glomeruli) characterized by small diameter, decreased matrix, and dense, peripherally placed nuclei with scant Bowman’s space are frequently seen in dogs. 49,215 Glomerular filtration rate does not completely reach its maximum until 8 to 10 weeks of age in dogs, although it is considered functionally mature by week 6 compared with PND 28 to 32 in mice and PND 42 in rats. 217 –219 Proximal convoluted tubular epithelium is simple, columnar, and has a prominent microvillus border, which increases surface area for absorption of water, proteins, and other substances and secretion of uric acid, urea, creatinine, and other substances (certain xenobiotics). The inner and outer stripe of the outer medulla is much more differentiated in rodents than in humans, dogs, or monkeys. 216 Urine concentrating ability develops prenatally in dogs, but postnatally in mice, rats, and humans. 220 The epithelium of the loops of Henle and the distal convoluted tubules are also metabolically active and involved in finely balanced excretion/absorption of water, depending on current hydration status and electrolyte concentrations.
The kidneys are primarily exposed to drugs and chemicals through the blood via the paired renal arteries arising from the abdominal aorta. As such, the kidneys are a site of xenobiotic biotransformation, which may result in formation of toxic metabolites, which may be taken up into the cytoplasm of tubular epithelial and/or excreted in the urine. Renal proximal tubules are common targets for toxicity due in part to the expression of transporters that mediate the secretion and reabsorption of xenobiotics. 221 Excretion of chemicals and/or metabolites, which may be concentrated in the urine, may result in increased exposure of the lower urinary system. Finally, the deposition of biologic molecules (eg, therapeutic antibodies, immune complexes, or proteins) may occur in the glomerulus. The juxtaglomerular apparatus is a specialized aspect of the afferent arteriole and the distal convoluted tubule with a key role in the renin–angiotensin–aldosterone axis to control systemic blood pressure and is composed of the macula densa, juxtaglomerular cells, and extraglomerular mesangial cells. 215 Erythropoietin is produced by cortical interstitial fibroblasts associated with the distal convoluted tubules (extraglomerular mesangial cells) in the kidney.
The recommended terminology of microscopic findings in the kidney of dogs is presented in Table 17.1 (kidney) and 17.2 (renal pelvis).
Microscopic Findings in the Urinary System: Kidney; Dog.

a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology section.
c Finding more frequent as an induced change.
Microscopic Findings in the Urinary System: Renal Pelvis; Dog.

a Finding more frequent as an induced change.
b Terminology addressed in the General Pathology section.
Immature Glomerulus—Kidney (Figure 17.1 and 17.2)
Dog, kidney, immature glomeruli, H&E.
Other Term(s)
Fetal glomerulus.
Pathogenesis/Cell of Origin
Incomplete development of glomerulus (congenital).
Diagnostic Features
Depending on the stage of development, the diagnostic features may vary:
Immature glomeruli are of smaller size than fully developed glomeruli.
There may be a significantly broader space between the developing capillary tuft and Bowman’s capsule.
Developing glomerular tuft may appear as vesicle, comma or s-shaped, or capillary loop.
Developing glomerular tuft appears as dense, basophilic accumulation of cells.
Developing glomerular tuft is lined by dark stained epithelium cells (podocytes).
Differential Diagnoses
Not applicable.
Comments
In the postnatal period, immature glomeruli are normally present as kidney development is not completed at birth. 222 In the kidney of the peripubertal and adult dog, a few immature glomeruli are frequently observed, considered a background finding, and are only reported in toxicologic pathology studies if their number is significantly increased. They have been described in conjunction with other congenital renal lesions, such as renal dysplasia and juvenile nephropathy. 222 –224
Hypertrophy, Juxtaglomerular Cell—Kidney
Biological Behavior
Adaptive change
Sublocation
Cortex
Pathogenesis/Cell of Origin
Stimulation of the renin–angiotensin system; increased demand for renin.
Diagnostic Features
Increased size of the afferent arterioles of glomeruli, in severe cases also efferent arterioles.
Invariably associated with hyperplasia of the juxtaglomerular cells.
Increased granularity of the afferent arterioles, that is, renin-containing cells.
Special Procedures
Differential Diagnoses
Not applicable.
Comments
Hypertrophy of the juxtaglomerular cells is in most if not all instances associated with hyperplasia of juxtaglomerular cells; in these cases, the combination term hyperplasia/hypertrophy, juxtaglomerular is considered appropriate. The lesions have been observed after administration of angiotensin-converting enzyme inhibitors 229 and angiotensin II antagonists. 230 Experimentally, the change has also been described following adrenalectomy, sodium depletion, hypertension, and renal ischemia.
Lipidosis, Glomerular—Kidney (Figure 17.3 and 17.4)
Other Term(s)
Glomerular vacuolation, renal glomerular lipidosis.
Pathogenesis/Cell of Origin
Deposition of lipid-containing foam cells in the glomerulus.
Diagnostic Features
Lipid accumulation characterized by deposition of fine vacuoles in mesangial cells of glomeruli, giving rise to foam cells.
Focal or segmental lipid accumulation is often observed and hyaline droplets deposition frequently accompanies this change.
Differential Diagnoses
Not applicable.
Comments
Glomerular lipidosis is a segmental change in mesangial cells of the glomerular tuft with aggregation of lipid-laden foam cells; this lesion did not appear to affect overlying podocytes. 231 The foam cells were observed in capillary lumina or within the mesangium and distribute focally or segmentally within affected glomeruli. 49,60,232 The pathogenesis of glomerular foam cell accumulation remains unknown and is likely multifactorial. Glomerular lipidosis was identified in renal biopsies from 46 dogs evaluated by the International Veterinary Renal Pathology Service. Glomerular lipidosis was the sole diagnosis in 5 of 46 cases (11%), all of which were proteinuric. All 5 dogs had at least 1 additional clinicopathologic abnormality consistent with renal disease, including hypertension (4), azotemia (3), and/or hypoalbuminemia (2). 233
Mineralization—Kidney
Comments
Features of renal corticomedullary tubular mineralization in dogs are generally identical to those observed in rodents and are captured in the INHAND publication on the rat and mouse urinary system. 234 Mineralization of medullary tubules, similar to that in the corticomedullary region and similar to that noted in rats, has also been described in dogs. 49 Shiga et al 235 describe vascular mineralization in the renal papilla of dogs; the lesion reportedly occurs in males and females, it is not age related, and the severity does not increase with age.
Vacuolation, Tubule—Kidney (Figure 17.5)
Comments
Vacuoles may be seen in proximal tubule epithelium under circumstances which indicate either physiological or toxicological change. Among species used in toxicity studies, vacuolation is most commonly observed in beagle dogs as a normal variation, where both proximal and distal tubules may contain fine lipid vacuoles, 232 and is seen more commonly in females. 49,60 The severity of spontaneous vacuolation of tubules may be increased by various drugs and chemicals, including vehicles, and may become degenerative in nature. Vacuolation of the proximal tubule epithelium due to increased lipid is a rare phenomenon and is an indication of toxic change. 236 Cytoplasmic lipid droplets can be confirmed by Oil Red-O stain. 237 A reversible vacuolation of proximal tubule epithelium (osmotic nephrosis) is seen in laboratory animals following the administration of hypertonic sugar solutions, dextran, and polyethylene-conjugated proteins. Tritium-labeled dextran accumulates in lysosomes, and vacuoles induced by sugar solutions and dextran at lower severities may represent reversible lesions. Ionic and nonionic X-ray opaque contrast media can also induce lysosomal vacuoles in the proximal tubule epithelium, but it usually does not accompany significant functional or permanent structure damage. 232 Administration of cationic amphophilic drugs has been associated with phospholipidosis, an accumulation of exogenous materials within lysosomes. 232 Lysosomes may be considered organelles that enable detoxification and cell survival by storage and degradation of a foreign material. From this standpoint, lysosome pathology may indicate cytotoxicity possibly leading to cell death and therefore may be considered adverse, although minimal to mild phospholipidosis is generally considered nonadverse. 238
Mesoblastic Nephroma—Kidney (Figures 17.6–17.8)
Other Term(s)
Mesenchymal derivative of nephroblastoma.
Pathogenesis/Cell of Origin
Tumor cells of myofibroblastic lineage.
Diagnostic Features
Nonencapsulated and predominantly monomorphic neoplasm.
Interlacing bundles of homogenous spindle cells with small basophilic nuclei and scant eosinophilic cytoplasm.
Widespread myxoid pattern.
Fascicular and wavy patterns are also present.
Matrix full of collagen fibrils.
Differential diagnoses
Not applicable.
Comment
A rare benign tumor in dogs; the collagen fibrils in the matrix can be demonstrated by a positive Masson trichrome stain; immunohistochemically, the tumor cells are positive for vimentin and show myofibroblastic lineage with positive reaction for smooth muscle actin or fibronectin.
Nephroblastoma (Embryonal Nephroma, Nephroblastomatosis, and Wilms’ Tumor [Human Nomenclature])—Kidney
Comment
A rare malignant tumor in dogs, which has been observed in toxicologic pathology studies in beagle dogs. The neoplasm is included in the INHAND rodent manuscript of the urinary tract. 234 For the diagnostic criteria in dogs of this and other neoplastic findings, refer to the fascicle on the urinary tract of the International Histological Classification of Tumors of Domestic Animals published by the Armed Forces Institute of Pathology in conjunction with the American Registry of Pathology and the World Health Organization, 239 the chapter on neoplasms of the urinary system 240 in the textbook Tumors of Domestic Animals, and/or current literature.
Ureter, Urethra, and Urinary Bladder (Tables 17.3 –17.5)
Microscopic Findings in the Urinary System: Ureter; Dog.
a Terminology addressed in the General Pathology section.
Microscopic Findings in the Urinary System: Urethra; Dog.

a Terminology addressed in the General Pathology section.
Microscopic Findings in the Urinary System: Urinary Bladder; Dog.
a Terminology with diagnostic criteria and/or comments described in the text.
b Terminology addressed in the General Pathology Section.
Degeneration/Regeneration; Smooth Muscle—Urinary Bladder (Figure 17.9 and 17.10)
Dog, kidney, mesoblastic nephroma, H&E.
Other Term(s)
Regeneration, degeneration, detrusor myopathy.
Pathogenesis/Cell of Origin
Degeneration and regeneration of smooth muscle fibers/bundles in the wall of the urinary bladder.
Diagnostic Features
Myopathic lesions may be complex; consisting of focal to multifocal areas of smooth muscle fiber degeneration, atrophy, and/or regeneration.
Regeneration is often characterized by increased cytoplasmic basophilia and nuclear changes (mitoses).
Other features may include individual cell necrosis, enlarged smooth muscle nuclei and nucleoli, and/or occasional mitotic figures.
Differential Diagnoses
Not applicable.
Comments
Degenerative lesions of the urinary bladder muscular tunic, consisting of perinuclear vacuolation, have been observed in various dog breeds; additional degenerative features such as atrophy and foci of necrosis as well as fiber regeneration (cytoplasmic basophilia and mitotic figures) are the hallmarks of the detrusor myopathy, for which the beagle dog appears to be particularly susceptible. 241 In addition, the finding seems to be more prevalent in dogs with inflammatory changes in the mucosa, arteritis, and generally after catheterization. 241
Rhabdomyosarcoma, Botryoid Type—Urinary Bladder (Figures 17.11–17.13)
Dog, urinary bladder, rhabdomyosarcoma, botryoid type, H&E, macro.
Other Term(s)
Rhabdomyosarcoma, botryoid embryonal.
Pathogenesis/Cell of Origin
Tumor cells of embryonic myoblasts.
Diagnostic Features
Nonencapsulated and multilobulated neoplasm arising in neck region of urinary bladder.
Interlacing bundles of elongated mesenchymal cells with small basophilic nuclei and scant to moderate filamentous cytoplasm.
Occasional “strap-like” and/or multinucleated cells usually present.
Cell bundles are usually surrounded by a myxomatous stroma.
Distinct cross-striations may be present in some cells with phosphotungstic acid hematoxylin stain.
Differential Diagnoses
Not applicable.
Comment
A rare malignant tumor in juvenile dogs arising in the neck region of the urinary bladder comprised of embryonic myoblasts. 242 –245 With embryonal tumor types, including the botryoid type arising in the urinary bladder, neoplastic cells may exhibit differentiation ranging from myoblastic to myotubular (ie, multinucleated cells). Neoplastic cells are often desmin, myoglobin, and vimentin positive. 243 Ultrastructurally, myofilaments arranged in parallel arrays or z-band structures are usually present. 243 Distant metastasis has been reported. 246,247
Footnotes
Authors’ Note
All procedures used to prepare macroscopic and microscopic images of animal specimens for this article were performed in accordance with regulations and established guidelines for humane treatment of research animals and were reviewed and approved in advance by an Institutional Animal Care and Use Committee.
Acknowledgments
The authors wish to express their thanks to the memberships of the BSTP, ESTP, JSTP, and STP for comprehensive reviews, excellent comments, and helpful edits. And we wish to express our sincere appreciation to Charlotte Maria Dalsgaard, Eric van Esch, Beth Mahler, Charlotte Keenan, Rupert Kellner, Catherine Schafer, Kenneth Schafer, and Klaus Weber for their help and contributions in the preparation of this publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
