Abstract
Pregnant Sprague-Dawley rats were orally administered di(n-butyl)phthalate (DBP; 100 mg/kg/day) on gestation days (GD) 12 to 21. We investigated the male offspring and probed morphological alterations in Sertoli cells at 7, 9, 14, and 17 weeks of age. Parameters assessed in this study included offspring number, sex ratios, body weights, testis weights, seminiferous tubule (ST) profile numbers and diameters, number of vimentin-labeled Sertoli cells, and both testosterone and follicle-stimulating hormone (FSH) levels. Testicular weight/body weight ratios and the numbers and diameters of ST in maximum transverse testicular sections were statistically similar at weeks 7 and 9; however, at weeks 14 and 17, they were statistically different and displayed higher BrdU-positive Sertoli cells/Sertoli cell ratios in the DBP treatment group. Noteworthily, the serum FSH levels were higher and testicular testosterone levels were lower in the DBP treatment group. To our knowledge, the present study is the first to report that in utero DBP exposure significantly increased Sertoli cell numbers and their cellular proliferation from postpuberty to adulthood, with a significant decrease in testicular testosterone and an increase in FSH.
Phthalates are present in a wide variety of products, from personal care and cosmetic products to printing inks, pharmaceutical coatings, and medical devices. Phthalates provide flexibility and other desirable characteristics and thus are used in a variety of products. Di(n-butyl)phthalate (DBP) is a widely used plasticizer (Rodriguez-Sosa et al. 2014). The pharmacokinetics of DBP in pregnant Sprague-Dawley rats has been performed and reported at 50, 100, and 250 mg/kg. Major findings from these studies were that the fetus displayed about 50% the levels of DBP observed in the mother. In addition, the data illustrate that DBP is rapidly absorbed, distributed, and metabolized to mono-butyl-phthalate (MBP), which can then undergo biotransformation to give the phase II metabolite MBP-glucuronide (MBPG). MBPG was the major metabolite excreted in urine and about 77% of the dose was eliminated via urine within 24 hr. In an additional study, DBP was dosed at 100, 250, and 500 mg/kg/day (GD 12–21) with the lowest dose (100 mg/kg/day) delaying preputial separation (Mylchreest et al. 1999). Based upon these reports, and doses we have previously used in other studies from our laboratories to study DBP effects on Leydig cells (Wakui, Takahashi, et al. 2012; Wakui, Muto, et al. 2012), we selected 100 mg/kg/day (GD 12–21) dosing for the current study. However, human DBP exposures are reported to be less than 10 mg/kg/day (Kavlock et al. 2002).
In mammals, Sertoli cells display proliferative activity only during embryogenesis and perinatal development (Kluin, Kramer, and de Rooij 1984). In rats, fetal and postnatal periods up to about 2 weeks of age constitute to be Sertoli cell mitosis stage. At around 15–18 days of age, the blood–testis barrier forms, but Sertoli cells continue differentiating in order to sustain full spermatogenesis. While around puberty, Sertoli cell number apparently decreases (Steinberger and Steinberger 1971; Orth 1982; Orth, Gunsalus, and Lamperti 1988; Vergouwn, Jacobs, and Huiskamp 1991; Riera et al. 2012; Hu et al. 2014). Thereafter, Sertoli cell numbers stabilize throughout adult life in nonseasonal species (Richburg and Boekelheide 1996). Sertoli cell differentiation is indispensable for testis formation, because these cells form critical spermatogenic cell attachment sites. The interactions between Sertoli cells and spermatogenic cells are critical for successful spermatogenesis (Hess, Cooke, and Bunick 1993; Alam et al. 2010). Thus, Sertoli cell number is a sperm-producing capacity determinate in animals, including man into adulthood (Hess, Cooke, and Bunick 1993). Researchers have reported that in utero DBP exposure induces adverse Sertoli cell effects during perinatal periods; the exposure results in a significant Sertoli cell number reduction, but their numbers do recover up until puberty (Kleymenova et al. 2005; Hutchison et al. 2008; Auharek et al. 2010; Rodriguez-Sosa et al. 2014; van den Driesche et al. 2015). However, Sertoli cell alteration data after in utero DBP exposure in rats from postpuberty to adulthood have appeared to be deficient. Consequently, we sought to investigate morphological alterations in Sertoli cells at 7, 9, 14, and 17 weeks in male offspring whose mothers had been orally administered DBP (100 mg/kg/day) on GD 12 to 21.
Material and Method
Animals, Chemicals, and Experiment Design
DBP (99.8% pure) was obtained from Tokyo Kasei Kogyo Co. (Tokyo, Japan). Eight-week-old time-mated female Sprague-Dawley rats (F0 = 40) were procured from Japan SRL Co. (Shizuoka, Japan); gestation day 0, the day copulation was confirmed.
Animals were individually housed in a HEPA-filtered, mass-air-displacement room maintained on a 12-hr light–dark cycle at 22 ± 2°C and 55 ± 5% relative humidity and fed a conventional diet (MF, Oriental Yeast, Tokyo, Japan). All experimental procedures were conducted under Animal Care and Used Committee approval at Azabu University School of Veterinary Medicine. We followed the guidelines set by the National Institutes of Health and Public Health Service Policy on the Humane Use and Care of Laboratory Animals. Pregnant rats (20 per group) were treated intragastrically (i.g.) with 100 mg/kg/day DBP (Tokyo Kasei Kogyo Co.) in corn oil (Nacalai Tesque Inc., Tokyo, Japan), and the remaining 20 rats were dosed with an equivalent volume of corn oil (control group: ∼0.5 ml/animal, i.g.) on days 12 through 21 postconception. The phthalate dose was chosen based on several previous studies describing adverse effects on fetal rats at the level of 100 mg/kg/day (Mylchreest et al. 1999; Barlow and Foster 2003; Motohashi, Wempe, Mutou, Okayama, et al. 2016; Motohashi, Wempe, Mutou, Takahashi, et al. 2016). The offspring were sexed at birth, and litters were reduced on day 1 so that each dam was left with 10 offspring (5 males and females per dam). Therefore, each treatment group comprised 100 male offspring. Weaning was performed on day 21 postpartum and then males were placed 3 per cage; the female offspring were anesthetized with isoflurane and killed by CO2 overdose. For each treatment group, the male offspring were randomized mathematically. At 7, 9, 14, and 17 weeks of age, animals (n = 20 per group per time point) were anesthetized with isoflurane and killed by CO2 overdose. The remaining rats were applied for bromodeoxyuridine (BrdU) incorporation experiments (n = 5 per group per time point). Testes were removed, fixed in the modified Davidson’s fluid (MDF) for 2 days, embedded into paraffin, and then cut into serial 4-μm transverse sections.
Vimentin Staining and Analysis
Vimentin labeling was analyzed using the avidin–biotin complex (ABC) method. After deparaffinization and hydration, 4-µm thick sections, so 4 sections × 20 rats × 2 testes = 160 microscopy slides, were treated with 0.3% H2O2 (10 min) and then blocked with 10% goat or horse serum in phosphate-buffered saline (PBS, 10 mM KPO4, 150 mM NaCl, pH 7.4; 20 min). Next, sections were rinsed in PBS and treated with a vimentin antibody (Sigma-Aldrich, St. Louis, MO; diluted 20-fold, 12 hr), which was detected with biotinylated IgG (Vector Laboratories, Burlingame, CA; diluted 100-fold, 3.0 hr), followed by ABC-peroxidase staining (Vector Laboratories) and diaminobenzidine (DAB; Sigma-Aldrich) for 1.0 hr. Then sections were immersed in Tris-HCl-buffered 0.02% 3,30-DAB tetrahydrochloride (DAB) containing 0.1 M sodium azide and 0.005% hydrogen peroxidase for 6.0 min. Sections were counterstained with Mayer’s hematoxylin (1.0 min). As a negative control, primary antibody was substituted with nonimmunized serum. The cross-sectional diameter of seminiferous tubules (STs) with a length:width ratio of less than 2 was examined, and the number of Sertoli cells in all vimentin antibody-stained sections was counted using image analysis (Nikon Coolscope, Tokyo, Japan). Total numbers of ST and vimentin-positive Sertoli cells in the testicular transverse sections of each testicular section were quantified.
BrdU Incorporation
For immunohistochemical detection of proliferating Sertoli cells on days 7, 9, 14, and 17, 5 control rats and 5 DBP dose group rats were randomly picked and administered an intraperitoneal injection of 5-BrdU (Zymed Laboratories, San Francisco, CA) at a dose of 100 mg/kg body weight. Rats were anesthetized and killed using pentobarbital overdose at 3.0-hr post-BrdU injection. The testes were immediately removed, cut into 4 serial sections, and fixed in MDF for 2.0 days and then embedded into paraffin for sectioning at a thickness of 4.0 µm, 5 sections × 5 rats × 2 testes = 50 microscopy slides. The slide sections were dewaxed and then microwaved at 900 W (15 min; 100 nM citrate buffer). Endogenous peroxidase activity was quenched by incubating methanol containing 0.3% H2O2 (30 min). The slides were incubated in blocking serum (10% normal rabbit serum; 20 min at room temperature) and then incubated with a rat anti-BrdU monoclonal antibody (ab623; Abcam, Cambridge, United Kingdom) using the BrdU cell proliferation kit (Abcam). Periodic acid–Schiff stain was used to counterstain the sections.
Then, in each testis, we counted the number of BrdU-positive Sertoli cells per tests per ST via Coolscope (Tokyo, Japan) and calculated the BrdU-positive Sertoli cells/(BrdU-positive Sertoli cells + BrdU-negative Sertoli cells). BrdU staining was localized at the nucleus, and Sertoli cell counting was validated using the exclusive diagnosis method. BrdU-positive cells capture Sertoli cells in S-face division (DNA is replicated); the morphology of BrdU-positive Sertoli cells localized to the nucleus was different from mature Sertoli cells. The nucleus of BrdU-positive cells was relatively large and quite different as compared to other spermatogenic cells; 4 different pathologists (S.W., E.K., H.T., and H.I.) counted the BrdU-positive cells and BrdU-negative Sertoli cells.
Hormone Assays
Testicular testosterone (T) and follicle-stimulating hormone (FSH) levels were measured using radioimmunoassay methods as described previously (Mylchreest et al. 1999; Fisher 2004; Wakui, Muto, et al. 2012; Wakui, Shirai, et al. 2013) from individual testes from 5 male rats per group at each endpoint. After dissection, testes were snapped into small tissues and frozen on liquid nitrogen and stored at −80 ± 10°C until analysis; rat T enzyme-linked immunosorbent assay (ELISA) kit (ab108666) and rat FSH ELISA kit (kt-15332) were used. Testes were defrosted and homogenized individually (500 µl PBS, pH 7.4); this solution was extracted with diethyl ether (2.0 ml), shaken (5.0 min), and placed in methanol cooled with dry ice. The nonaqueous portion of the extract was removed, allowed to air-dry overnight in a fume hood, and reconstituted in assay buffer. The assay limit of detection was 40 pg/testis.
Statistical Analysis
For each set of mean values, standard deviation and standard error of means (SEMs) were calculated and compared by Scheffé’s F test (p < .05) using the statistical computer analysis system StatView-J version 5.0 (Abacus Concepts Co. Ltd., California).
Results
Pregnant Sprague-Dawley dams were i.g. administered DBP (100 mg/kg/day) on GD 12 to 21. Compared to control group (corn oil), the treatment group (i.e., DBP 100 mg/kg/day) did not alter body weight. The average body weights (Figure 1A) were statistically similar to control group (p > .05) at weeks 7, 9, 14, and 17. Additionally, the DBP treatment group showed no decrease in litter size, pup survival rates, alteration of sex ratio, or body weight of male pups compared to controls (data not shown). The relative testicular weights (testicular weight/body weight; Figure 1B) were statistically similar to control treatment group at weeks 7 and 9, whereas at weeks 14 and 17, the DBP treatment group displayed a statistically significant (p < .05) lower testicular weight/body weight ratio; the testes were smaller in the DBP dose group at weeks 14 and 17. We used light microscopy methods to evaluate the diameters and numbers of STs per testis. The ST numbers were statistically similar at weeks 7 and 9 but were statistically greater (p < .05) than control at weeks 14 and 17 (Figure 1C). In contrast, the average ST diameter was statistically lower (p < .05) than control at weeks 14 and 17 (Figure 1D). Vimentin staining revealed that the numbers of vimentin-positive Sertoli cells per ST were similar at weeks 7 and 9 but were statistically higher (p < .05) via the DBP group at weeks 14 and 17 (Figure 1E, 2). Furthermore, the total number of Sertoli cells was statistically similar at weeks 7 and 9 but became statistically higher in the DBP treatment group at weeks 14 and 17 (Figure 1F, 2). BrdU immunohistochemical staining revealed that Sertoli cell numbers (BrdU-positive cells) per ST were similar at 7 and 9 weeks but became statistically higher (p < .05) in the DBP dose group at weeks 14 and 17 (Figure 1G, 3). When the BrdU-positive Sertoli cells/Sertoli cell ratios were plotted (Figure 1H, 3), the ratios were statistically similar at weeks 7 and 9 but statistically higher (p < .05) at weeks 14 and 17. The testicular T levels via the DBP treatment group were significantly lower (p < .05) than control group at all 4 time points (i.e., weeks 7, 9, 14, and 17; Figure 1I), whereas the serum FSH levels were statistically higher (p < .05) in the DBP treatment group at all 4 time points (i.e., 7, 9, 14, and 17 weeks; Figure 1J).
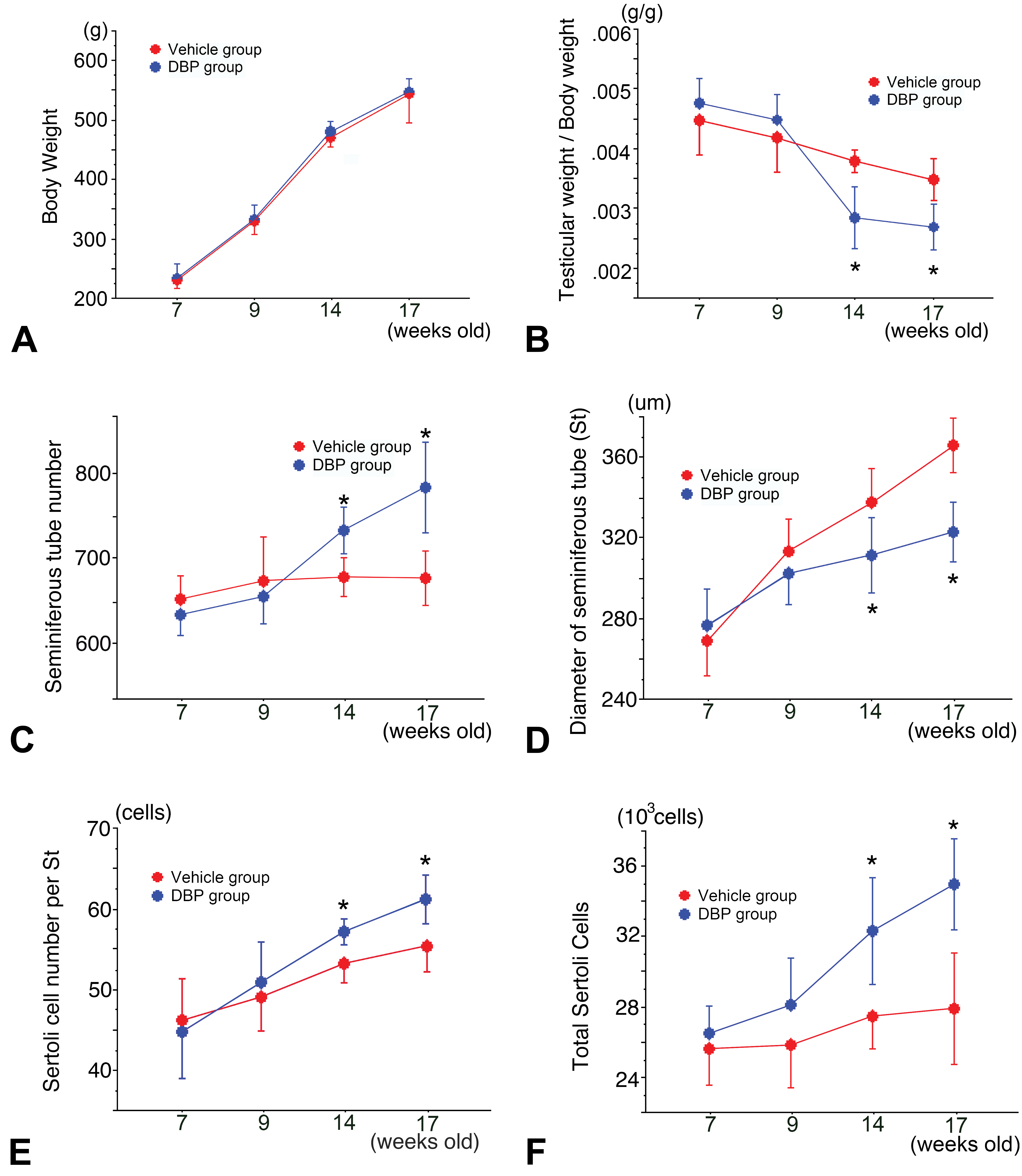
(A) Male pup body weights via dams exposed to 100 mg/kg/day (di(n-butyl)phthalate [DBP] group; rats = 100) or corn oil (vehicle group; rats = 100) on gestation days 12 to 21. Mean ± SD, Scheffé’s F test, *p < .05. (B) Relative testicular weights (testicular weight/body weight), vehicle versus DBP dose group; 20 rats were applied at each point. Mean ± SD, Scheffé’s F test, *p < .05. (C) Number of seminiferous tubules in the maximum testicular transverse sections, vehicle versus DBP dose group; 20 rats were applied at each point. Mean ± SD, Scheffé’s F test, *p < .05. (D) Diameter of seminiferous tubules in the maximum testicular transverse sections, vehicle versus DBP dose group; 20 rats were applied at each point. Mean ± SD, Scheffé’s F test, *p < .05. (E) Number of Sertoli cells per seminiferous tubule, vehicle versus DBP dose group; 20 rats were applied at each point. Mean ± SD, Scheffé’s F test, *p < .05. (F) Total number of Sertoli cells, vehicle versus DBP dose group; 20 rats were applied at each point. Mean ± SD, Scheffé’s F test *p < .05. (G) Number of bromodeoxyuridine (BrdU)-positive Sertoli cells per seminiferous tubule, vehicle versus DBP dose group; 5 rats were applied at each point. Mean ± SD, Scheffé’s F test, *p < .05. (H) Number of BrdU-positive Sertoli cells/(number of BrdU-positive Sertoli cells + number of BrdU-negative Sertoli cells), vehicle versus DBP dose group; 5 rats were applied at each point. Mean ± SD, Scheffé’s F test, *p < .05. (I) Testicular testosterone levels, vehicle versus DBP dose group; 5 rats were applied at each point. Mean ± SD, Scheffé’s F test, *p < .05. (J) Serum follicle-stimulating hormone levels, vehicle versus DBP dose group; 5 rats were applied at each point. Mean ± SD, *p < .05.
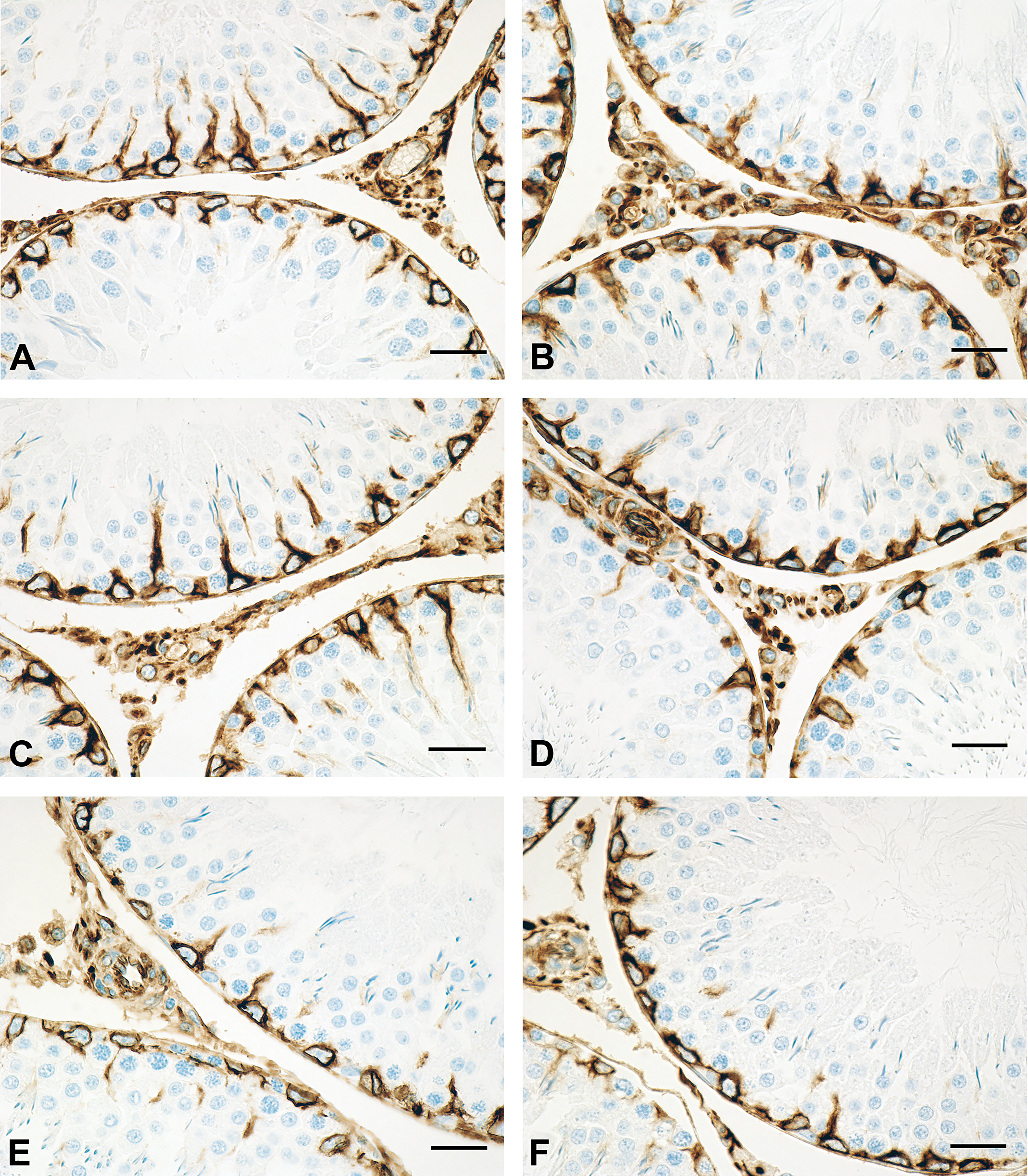
(A) Representative vimentin staining in rat testes at 7 weeks following exposure to corn oil (vehicle group) on gestation days 12 to 21. Vimentin-positive Sertoli cells are observed. Bar = 50 µm. (B) Representative vimentin staining in rat testes at 7 weeks following exposure to 100 mg di(n-butyl)phthalate (DBP)/kg/day (DBP group) on gestation days 12 to 21. Vimentin-positive Sertoli cells are observed; compared to vehicle (A), the differences were not statistically different. Bar = 50 µm. (C) Representative vimentin staining in rat testes at 9 weeks following exposure to corn oil (vehicle group) on gestation days 12 to 21. Vimentin-positive Sertoli cells are observed. Bar = 50 µm. (D) Representative vimentin staining in rat testes at 9 weeks following exposure to 100 mg DBP/kg/day (DBP group) on gestation days 12 to 21. Compared to control (C), the differences were not statistically different. Bar = 50 µm. (E) Representative vimentin staining in rat testes at 14 weeks following exposure to corn oil (vehicle group). Vimentin-positive Sertoli cells are observed. Bar = 50 µm. (F) Representative vimentin staining in rat testes at 14 weeks following exposure to 100 mg DBP/kg/day (DBP group) on gestation days 12 to 21. Compared to control (E), the vimentin-positive Sertoli cell numbers were statistically higher. Bar = 50 µm. (G) Representative vimentin staining in rat testes at 17 weeks following exposure to corn oil (vehicle group) on gestation days 12 to 21. Vimentin-positive Sertoli cells are observed. Bar = 50 µm. (H) Representative vimentin staining in rat testes at 17 weeks following exposure to 100 mg DBP/kg/day (DBP group) on gestation days 12 to 21. Compared to control (G), the vimentin-positive Sertoli cell numbers were statistically higher. Bar = 50 µm.
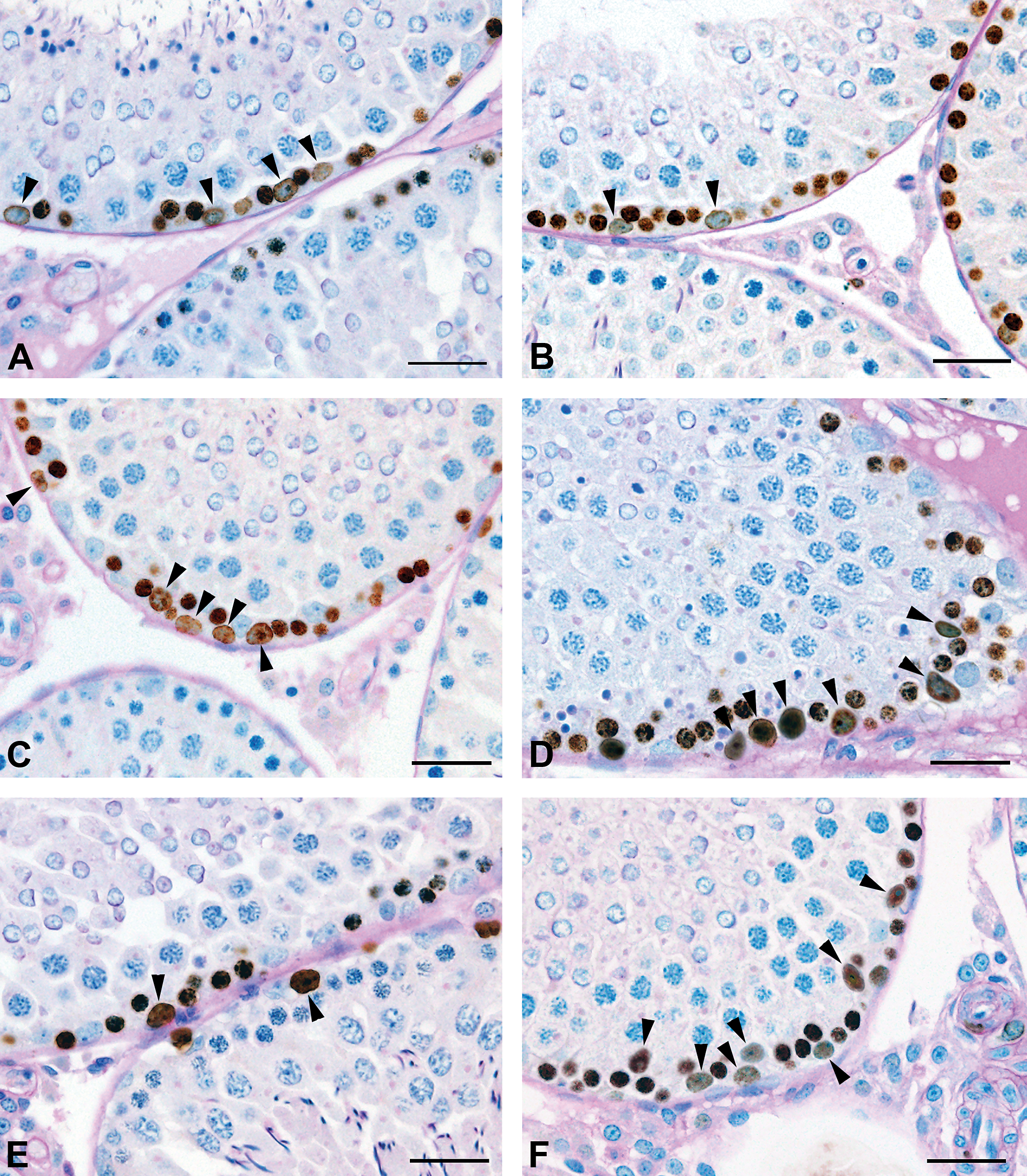
(A) Representative immunohistochemical assay by bromodeoxyuridine (BrdU) from rat testes at 7 weeks old following exposure to corn oil (vehicle group) on gestation days 12 to 21. The BrdU-positive Sertoli cells (black arrowheads) are denoted. Bar = 20 µm. (B) Representative immunohistochemical assay by BrdU from rat testes at 7 weeks old following exposure to 100 mg of di(n-butyl)phthalate (DBP)/kg/day (DBP group) on gestation days 12 to 21. The BrdU-positive Sertoli cells (black arrowheads) are denoted. Bar = 20 µm. (C) Representative immunohistochemical assay by BrdU from rat testes at 9 weeks old following exposure to corn oil (vehicle group) on gestation days 12 to 21. The number of BrdU-positive Sertoli cells (black arrowheads) are denoted. Bar = 20 µm. (D) Representative immunohistochemical assay by BrdU from rat testes at 9 weeks old following exposure to 100 mg DBP/kg/day (DBP group) on gestation days 12 to 21. BrdU-positive Sertoli cells (black arrowheads) are denoted. Bar = 20 µm. (E) Representative immunohistochemical assay by BrdU from rat testes at 14 weeks old following exposure to corn oil (vehicle group) on gestation days 12 to 21. BrdU-positive Sertoli cells (black arrowheads) are denoted. Bar = 20 µm. (F) Representative immunohistochemical assay by BrdU from rat testes at 14 weeks old following exposure to 100 mg DBP/kg/day (DBP group) on gestation days 12 to 21. BrdU-positive Sertoli cells (black arrowheads) are observed. Bar = 20 µm. (G) Representative immunohistochemical assays by BrdU from rat testes at 17 weeks old following exposure to corn oil (vehicle group) on gestation days 12–21. BrdU-positive Sertoli cells (black arrowheads) are observed. Bar = 20 um. (H) Representative immunohistochemical assay by BrdU from rat testes at 17 weeks old following exposure to 100 mg DBP/kg/day (DBP group) on gestation days 12 to 21. BrdU-positive Sertoli cells (black arrowheads) are observed. Bar = 20 µm.
Discussion
Phthalates are widely distributed in the environment and designated as endocrine active substances (Fisher 2004; Wakui, Muto, et al. 2012; Wakui, Shirai, et al. 2013; Motohashi, Wempe, Mutou, Okayama, et al. 2016; Motohashi, Wempe, Mutou, Takahashi, et al. 2016), which means that they can interact or interfere with normal hormonal action. Previous epidemiological, animal, and molecular studies have shown that exposure of 3-week-old rats to DBP (500 mg/kg) disrupts spermatogenesis with testicular atrophy (Alam et al. 2010). The present study demonstrates that rats exposed in utero to DBP (100 mg/kg/day) showed (1) significant testicular atrophy with changes in testicular weight/body weight ratio and (2) a significant ST number and diameter decrease with age (i.e., weeks 14 and 17). However, the mechanisms causing spermatogenesis disruption induced by in utero DBP exposure have not been fully elucidated (Alam et al. 2010).
Based upon specific biochemical and morphological alterations after phthalate exposure, many researchers have deduced that phthalate esters target Sertoli cells (Richburg and Boekelheide 1996; Boekelheide et al. 2009). Previous studies have illustrated that male rats exposed in utero to DBP (500 mg/kg/day; GD 12–21 or 13.5–21.5) displayed severely reduced Sertoli cells. During the perinatal period, spermatogonia were decreased by ∼50% but recovered to normal levels by weeks 3 to 4 (Kleymenova et al. 2005; Hutchison et al. 2008; Scott et al. 2008; Auharek et al. 2010; Rodriguez-Sosa et al. 2014; van den Driesche et al. 2015).
There were 2 treatment groups in the present study; both groups used the male offspring, treatment group 1 being control (corn oil only; F1 n = 100 from 20 F0) and treatment group 2 (F1 n = 100 from 20 F0) were exposed in utero to DBP (100 mg/kg/day; i.g., GD 12–21). In this study, the average body weights were not statistically different (Figure 1A); the pup survival rates, sex ratio, and litter sizes were similar in the 2 treatment groups. However, denoting testicular atrophy, the DBP treatment group testes were smaller (i.e., 14 and 17 weeks of age) than the control (Figure 1B). ST numbers of the DBP dose group were statistically greater (p < .05) than the control at weeks 14 and 17 (Figure 1C). In contrast, average ST diameters were statistically lower (p < .05) than the control at weeks 14 and 17 (Figure 1D). In the maximum testicular transverse sections, the increased quantity of ST is associated with a lower ST diameter, results that further confirm the testicular atrophy. Furthermore, the total number of Sertoli cells was statistically higher in the DBP treatment group at weeks 14 and 17 (Figure 1F). BrdU immunohistochemical staining revealed that Sertoli cell numbers (BrdU-positive cells) per ST were statistically higher (p < .05) in the DBP dose group at weeks 14 and 17 (Figure 1G). We surmise that the increased number of Sertoli cells per ST is a consequence of testicular atrophy. In an attempt to further indicate cell proliferation, BrdU-positive Sertoli cells/total (BrdU positive + BrdU negative) Sertoli cell ratios were plotted (Figure 1H); the ratios were statistically higher (p < .05) in the DBP treatment group at weeks 14 and 17.
The testicular T levels via the DBP treatment group were significantly lower (p < .05) than the control group at all 4 time points (i.e., weeks 7, 9, 14, and 17; Figure 1I). We previously demonstrated that in utero DBP exposure decreased T levels by causing dysfunction of Leydig cells (Wakui, Muto, et al. 2012; Wakui, Shirai, et al. 2013; Shirai et al. 2013; Motohashi, Wempe, Mutou, Okayama, et al. 2016; Motohashi, Wempe, Mutou, Takahashi, et al. 2016). Testicular T is a determinant of spermatogenesis, which binds with the androgen-binding protein of Sertoli cells to produce glycoprotein (β globulin). Reports indicate that lower levels of T and FSH result in low spermatogenesis (Orth 1982; Sharpe et al. 2003; Chaudhary et al. 2005). On the other hand, we have observed that Sertoli cells proliferate in adult rats with in utero exposure to 3,3′,4,4′,5-pentachlorobiphenyl (PCB126) but T and FSH concentrations were similar to vehicle-treated group (Wakui, Takahashi, et al. 2012). In the present study, serum FSH levels were statistically higher (p < .05) in the DBP treatment group at all 4 time points (i.e., 7, 9, 14, and 17 weeks; Figure 1J). To our knowledge, the present study is the first to report that in utero DBP exposure significantly increased Sertoli cell numbers and their cellular proliferations from postpuberty to adulthood, with a significant decrease in testicular T and an increase in serum FSH.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported via the Asabi University Research Services Division of the Promotion and Mutual Aid Corporation for Private Schools of Japan and Grants-in-Aid (C) #25450473 received from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
