Abstract
Permanent vascular catheterization for intravascular access is one of the most commonly applied techniques used on rodents in pharmacology studies. However, use of the intravascular catheters is complicated by nontolerance due to thromboembolic disease and sepsis. We have undertaken an extensive pathologic and clinical analysis of an intravascular catheterization model in Wistar Han and Sprague-Dawley rats, with a particular focus on carotid artery catheterization with or without jugular vein catheterization, in order to define the pathologic mechanisms behind nontolerance and define clinical end points to ensure maximal animal welfare. Further, we have explored various potential solutions to increase the tolerance of the procedure. In these studies, indwelling catheters were found to cause a high degree of thromboembolic disease with infarction in the brain, cecal tip, and kidneys being the primary causes of nontolerance. Loss of greater than 10% body weight was determined to be the most sensitive indicator of nontolerance and was closely correlated with degree of renal parenchymal loss. Sepsis was noted as a very rare complication, indicating that routine aseptic surgical techniques are adequate for preventing this complication.
Keywords
Automatic blood sampling technology via intravascular catheterization (IVC), such as the DiLab® AccuSampler®, is widely used in drug discovery to facilitate exploratory rodent-based pharmacokinetic (PK) and PK/pharmacodynamic (PD) studies with minimal animal handling. Typically automated blood sampling uses a carotid artery catheterization (CAC) rat model with or without jugular vein catheterization (JVC). In this CAC with or without JVC model (CAC ± JVC), male rats underwent a surgical procedure that involved permanent ligation and insertion of an indwelling catheter into the left carotid artery with or without a second catheter in the right jugular vein. In our study, catheter design and the project licence allowed the reuse of rats in 2 different studies, which significantly reduced the total number of animals required. Typically, rats were kept for up to 3 weeks after surgical placement of the catheters, with timing of termination of animals clinically tolerating the procedure being based on operational requirements. Over a period of 8 months preceding this analysis, 228 rats were surgically implanted with CAC ± JVC. The nonclinical tolerance death or euthanasia rate of this model averaged 35.1%; with 4.4% of the animals dying unexpectedly without any clinical signs, while a further 30.7% were symptomatic necessitating euthanasia (data not shown). Animals displayed a range of presenting clinical signs including neurological signs (lameness, twitching, head tilt, circling, leaning, hypersensitivity, hind limb paralysis, vocalizing, and depression), body weight (BW) loss, dehydration, and general malaise (hunched posture, piloerection/rough hair coat, and chromodacryorrhea). An additional 12% of rats had to be euthanized due to catheter patency issues. Overall, 73% of CAC ± JVC rats were used in at least 1 study, and only 52.9% were reused in 2 studies. Because of this significant morbidity and animal attrition, an extensive pathologic and clinical analysis of catheterization models was undertaken to define the pathologic mechanisms behind nontolerance, define clinical end points to ensure maximal animal welfare, and explore various potential solutions to increase the tolerance of the procedure including catheter material, gauge, and heparin concentration.
Methods and Materials
Animals
All experimental procedures complied with and were licensed under the British Animals (Scientific Procedures) Act 1986. Male Wistar Han–Crl:WI(WH) or Sprague-Dawley–Crl:CD(SD) rats weighing between 250 and 370 g at the time of the surgery were obtained from Charles River, Manston. They were provided with ad libitum RM1 (E) SQC, Special Diet Services (Essex, United Kingdom) food pellets, and bottled drinking water filtered by reverse osmosis. The room temperature was set at 21°C ± 2°C with a relative humidity of 55% ± 10%, 15 to 20 air changes/hr. Light cycles are automatically controlled on a 12:12-hr light/dark cycle set at 100 Lux. When the animals were not on study, they were singly housed in conventional open top cages (1354G, Tecniplast, Italy) under a conventional barrier with paper wool and chewing wood stick medium blocks (Lillico, Surrey, United Kingdom) as enrichments and on Litaspen Premium woods chips bedding (Lillico, Surrey, United Kingdom). During study periods, they were singly housed in microdialysis cages from Dilab/CMA (Lund, Sweden) or in the Tecniplast Metabolic Cages for rats and were connected to a DiLab AccuSampler, with a typical PK/PD study duration of 24 hr. Catheters were flushed prior to connection to the DiLab AccuSampler with 10-IU/ml heparin/0.9% saline or at least once a week. The catheter design and project license allowed animals to be reused on a maximum of 2 studies over a 2- to 4-week period post-CAC/JVC surgery.
Study Design
In the initial 3 studies, data were collected from a total of 41 WH rats subject to IVC of the left carotid artery with or without catheterization of the right jugular vein. All animals were acclimatized in the facility for a minimum of 5 days before undergoing surgery. All the surgeries were performed under isoflurane general anesthesia using aseptic techniques. A heated mat set at 38°C was placed under the animals. All animals received Carprofen (Rimadyl®) 5mg/kg sc at the induction and once on the morning one day after the surgery. During the anesthesia recovery, animals were placed in a warmed incubator box at approximately 31°C until mobile and then returned to their cage. Moistened food pellets were available in the cage during the postsurgery recovery period. Following the surgery, animals were monitored and weighed daily until their euthanasia. After a recovery period of at least 2 days, asymptomatic animals were used on study.
In experiment 1 assessing routine procedures, 26 WH rats were surgically prepared as described below with a 3 French silicone catheter. During the surgery, catheters were flushed with a 10-IU/ml heparin/0.9% saline solution for a total of 33 UI/kg per catheter. Experiment 2 assessed the effect of catheter materials on clinical outcomes in 9 WH rats, substituting 2 French polyurethane catheters for the 3 French silicone catheter used in experiment 1. In experiment 3, a higher heparin concentration for flushing the 3 French silicone catheters directly after their implantation during the surgery was assessed in 6 WH rats implanted with CAC and JVC using 66-IU/ml heparin/0.9% saline solution for a total of 250 IU/kg per catheter. A fourth experiment in which the carotid artery was ligatured without any catheter placement was carried out to assess the impact of the dissection, manipulation, and ligation of the artery on the lesions observed in the model. Experiment 4 used 20 rats (10 WH and 10 SD), 8 ligated, and 2 sham surgical controls per strain. The surgical procedures in these models are detailed below.
Catheterization of the Carotid Artery
An incision was performed about 1.5 cm left of midline on the neck from the manubrium toward the head. A blunt dissection was performed down through the fat and connective tissue to expose the fascia between the sternomastoid and the sternohyoid muscles. This fascial plane was gently dissected through to expose the carotid artery and the vagus nerve. The area was irrigated with 2% lidocaine. An approximately 5- to 10-mm section of vessel was isolated and exposed without damaging the vagus nerve. A silk ligature was positioned at either end of the isolated portion of the artery. The caudal ligature was loosely tied, and the cranial ligature was tightly tied without twisting the carotid. An incision was made between the two ligatures, close to the tied caudal ligature. The catheter was introduced into the vessel and advanced toward the heart until the retention bead of the catheter was over the vessel incision site. Both ligatures were tied to secure the catheter; the caudal one was tied behind the retention bead to keep the catheter in place. Catheter was flushed with 10- or 66-IU/ml heparin/0.9% saline solution and locked with 500-IU/ml heparin/glycerol lock solution. A Vicryl subcuticular stitch closed the neck incision.
Catheterization of the Jugular Vein
A small incision was performed over the right jugular vein, directly over the visible pulse. The subcutaneous fat and connective tissue were dissected to expose the vessel. An approximately 5-mm section of vessel was isolated and exposed. A silk ligature was positioned at either end of the isolated portion of the vein. The caudal ligature was loosely tied, and the cranial ligature was tightly tied. Using fine Vanna scissors, a small incision was performed between these ligatures. The catheter was inserted into the vein toward the heart until the retention bead of the catheter passed the vessel incision site. Both ligatures were tied to secure the catheter, and the cranial one was tied behind the retention bead to keep the catheter in place. Catheter was flushed with 10- or 66-IU/ml heparin/0.9% saline solution and locked with 500-IU/ml heparin/glycerol lock solution. A Vicryl subcuticular stitch closed the neck incision.
Carotid Ligation Study
A small incision was performed about 1.5 cm just left of midline on the neck from the manubrium toward the head. A blunt dissection was performed down through the fat and connective tissue to expose the fascia between the sternomastoid and the sternohyoid muscles. This fascial plane was gently dissected through to expose the carotid artery and the vagus nerve. Area was irrigated with 2% lidocaine. An approximately 5- to 10-mm section of vessel was isolated and exposed without damaging the vagus nerve. A tight silk ligature was positioned around the artery. A Vicryl subcuticular stitch closed the neck incision.
Clinical Observations
Animals underwent a daily clinical observation throughout the duration of the study. Baseline BW was determined prior to the surgery and recorded daily following the surgery. Any animals displaying signs of pain or distress, including significant BW loss, considered more than 20% of their presurgery BW, were euthanized. In the initial study cohort, blood samples were collected prior to the necropsy from 7 terminal animals to measure the blood urea nitrogen (BUN) and creatinine to assess the renal function.
Euthanasia and Necropsy
Animals were euthanized by an overdose of pentobarbitone IV when a jugular vein catheter was present or by inhalation of a raised concentration of CO2 using an increasing displacement of 20% CO2 per minute. A complete necropsy was performed in all animals. Tissues collected at necropsy were fixed in 10% neutral-buffered formalin for approximately 48 hr followed by routine histologic processing. Briefly, dehydration and clearing steps were performed on a Tissue Tek VIP Tissue Processor (GMI, Inc, Ramsey, MN). The tissues were then embedded in paraffin and cut into 4-µm sections. Haematoxylin and eosin (H&E) staining was performed on the Leica ST5020 Multistainer (Leica Microsystems, Milton Keynes, United Kingdom) according to the manufacturer’s instructions. Histologic sections were examined on an Olympus BX-51 optical microscope, and photomicrographs captured with an Olympus ColorView IIIu digital camera with Cell D software (Soft Imaging Systems GmbH, Munster, Germany). One board-certified pathologist (R.E.A.) performed all histopathologic assessments in the study. Histopathologic examination was conducted on core tissues from all animals including brain, kidneys, heart, lung, liver, and spleen and any additional tissues with macroscopic abnormality, most frequently the cecum. Percentage of damaged renal tissue (infarction, tubular degeneration/regeneration, and tubular necrosis) was assessed separately for each kidney to the nearest 5% (or 1–5% if less than 5%) using the percentage affected of a standard field of view of the microscope on a standard midline longitudinal section. The percentage of total damaged renal parenchyma calculated as an average of both kidneys, for each animal individually prior to cohort calculations. Histopathological aging of thrombi and thromboemboli was performed on H&E sections using a scheme adapted from mouse models of deep vein thrombosis by Diaz et al. (2012) and human deep vein thrombosis by Fineschi et al. (2009; Figure 1). Briefly, features of phase I acute vascular thrombosis of approximately 2 days duration (Figure 1A) included a thin vessel wall, no reactions between the vessel wall and thrombus, and fibrin and platelet deposition with obvious Zahn’s lines (Diaz et al. 2012; Fineschi et al. 2009). Histologic features of subacute phase II vascular thrombosis of 3 to 8 days duration (Figure 1B) included some thickening of the vessel wall, early penetration by fibroblasts and endothelial budding, increased numbers of neutrophils and monocytes, and some cavity and fissure formation that contains loosely aggregated erythrocytes (Diaz et al. 2012; Fineschi et al. 2009). Phase III vascular thrombosis of 4 to 20 days duration (Figure 1C) included infiltration by capillaries, fibroblasts, mesenchymal cells, and haemosiderin-accumulating histiocytes, leukocytic infiltration that was coalescing and contained nuclear debris, and a free surface that may be covered by endothelium (Diaz et al. 2012; Fineschi et al. 2009).
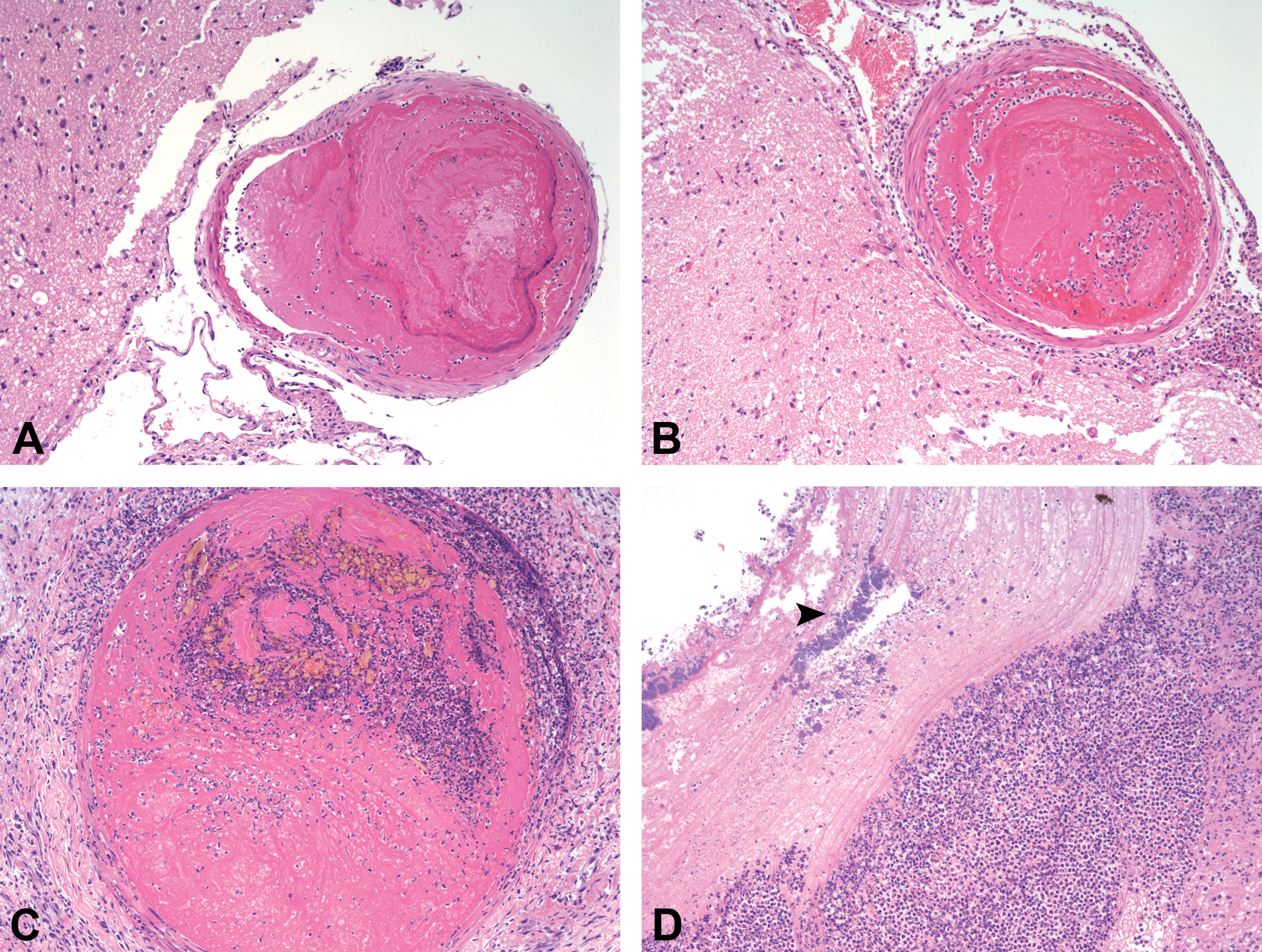
Kinetics of the microscopic appearance of intravascular thomboemboli in intravascular catheterization rats. Phase I acute thromboembolus in the meninges in a nontolerance rat euthanized 2 days after catheterization (A). Phase II thromboembolus in the meninges in a nontolerance rat euthanized 9 days after catheterization (B). Phase III subacute thromboembolus in a kidney in a nontolerance rat euthanized 12 days after catheterization (C). A jugular catheter–associated septic thrombus with bacterial colonies (arrow head) in the heart of a terminal rat in the heparin trial (D).
Results
Experiment 1: Pathological and Clinical Effects of Catheterization Models in Rats
Experiment 1 included a clinical and pathologic analysis of 26 CAC ± JVC rats (21 CAC only and 5 CAC + JVC) using a 3 French silicone carotid catheter with a 10-IU/ml heparin/0.9% saline solution for a total of 33-UI/kg per catheter. Of this cohort, 12 were terminally euthanized, 2 were found dead, and 12 were defined as nonclinical tolerance due to clinical signs. Terminal animals did not display any clinical signs, with the exception of a slight BW loss, on average of 2.32% (
Nonclinical tolerance animals displayed a variety of clinical signs including neurological signs (flaccid paralysis, twitching, head tilt, circling, leaning, and hind limb paralysis), dehydration, general malaise/pain signs (hunched posture, piloerection/ rough hair coat, hypersensitivity, chromodacryorrhea, vocalizing, and depression), and a more severe BW loss, averaging 11.31% (
Experiment 1: Summary of Individual Animal Data from Animals Receiving the Standard Protocol with the 3 French Silicone Catheter.
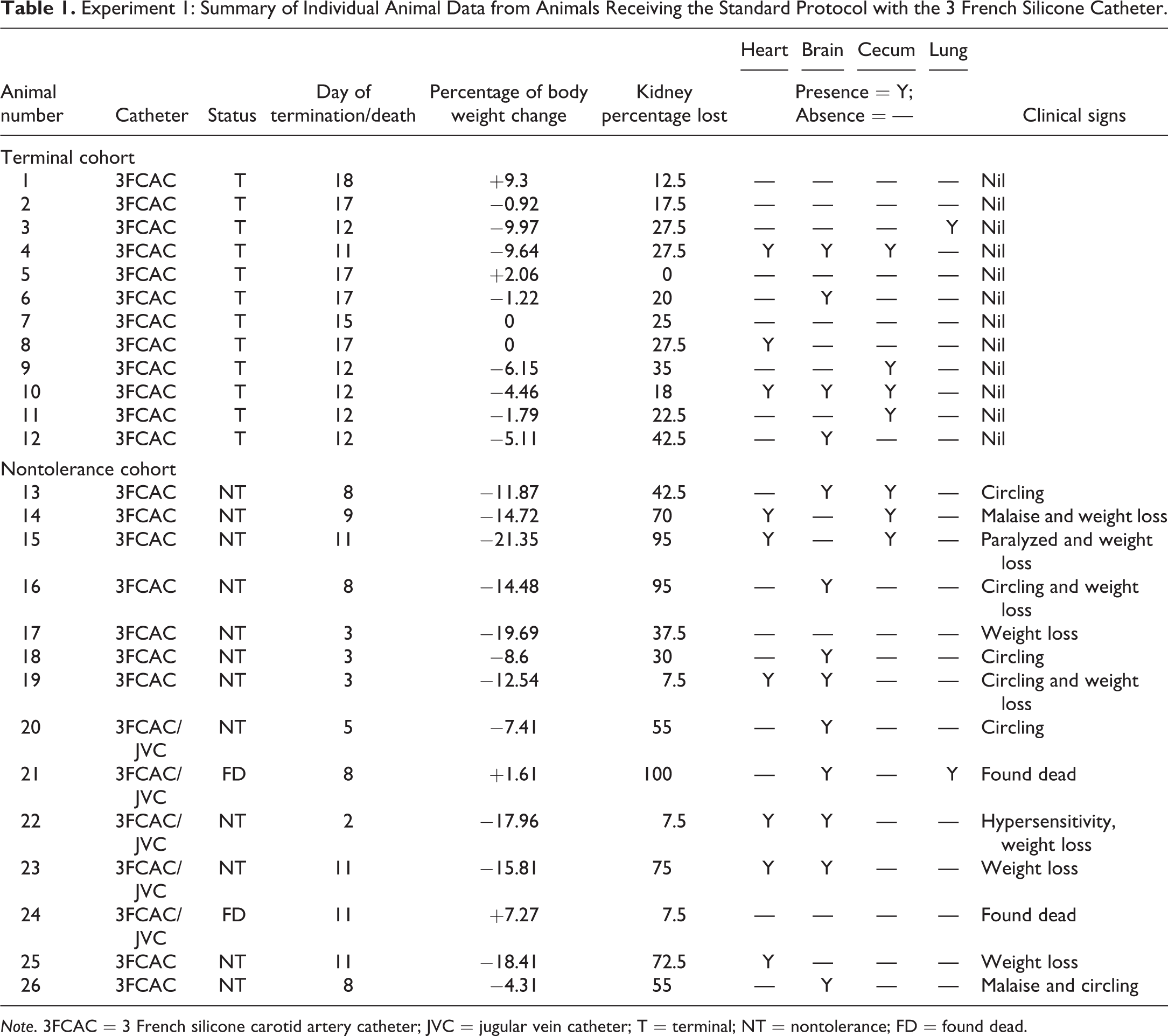
Incidence Table of Pathological Findings from Animals Receiving the Standard Protocol with the 3 French Silicone Catheter.
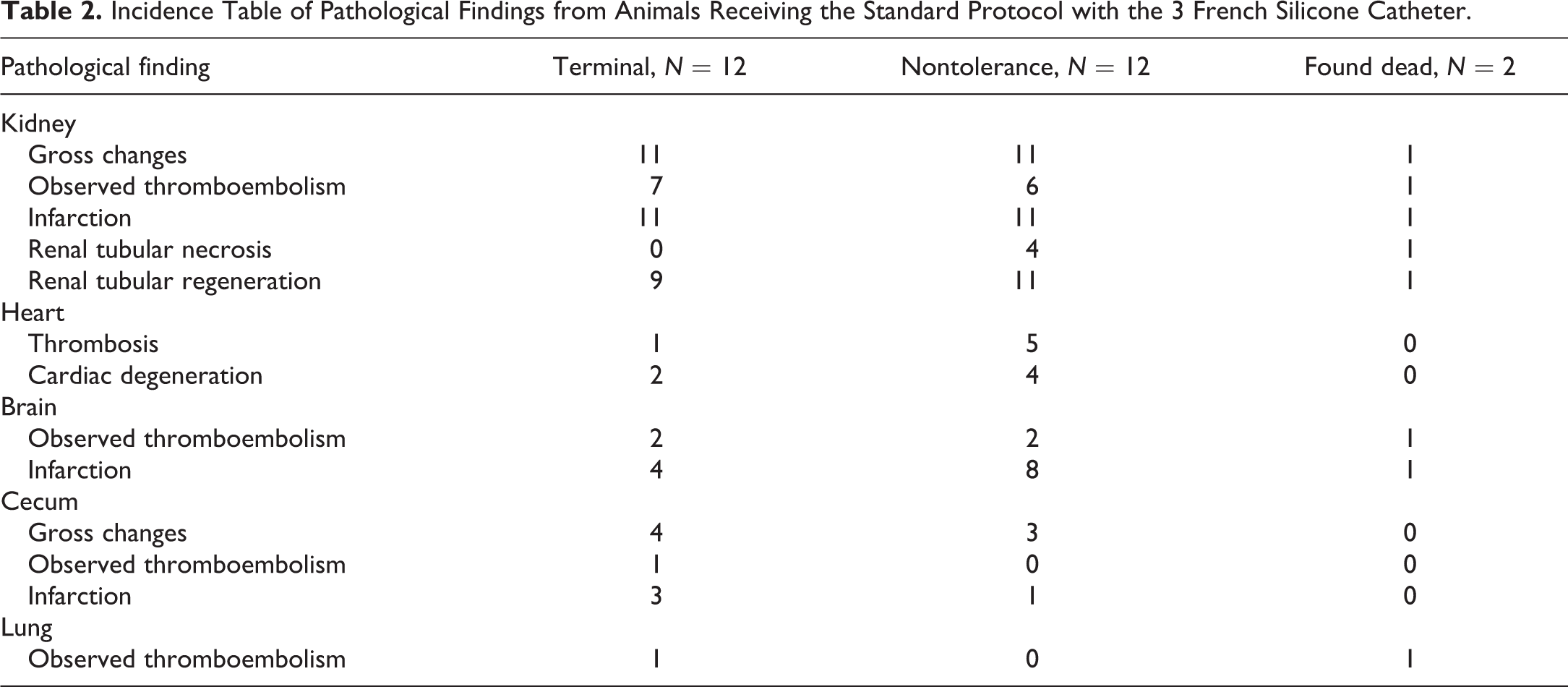
Summary of Thromboembolic Disease Incidence across Protocols in Intravascular Catheterization Models.
The types of lesions recorded in IVC rats with the 3 French silicone catheter were highly consistent, irrespective of whether the procedure was tolerated clinically, with a very high incidence of thromboembolic in both clinical tolerance, euthanasia and found dead cohorts. These included infarction of the cerebrum, kidneys, and cecal tip. Less frequently, other organs including the spleen, testis, heart, lung, and salivary glands were noted to contain thromboemboli, often associated with infarction of the tissue.
Macroscopically, renal infarcts were visible as variably sized areas of pallor on the renal cortices, which extended to the medulla as wedge-shaped regions on cut section (Figure 2A). Wedge-shaped areas of pallor in the kidneys are classical for infarcts, with the size and distribution of the infarct reflecting the region of tissue supplied by the occluded vessel (Mosier 2012). Macroscopically, cecal infarcts appeared as sharply demarcated dark red areas affecting 10% to 30% of the tip of the cecum (Figure 2B). Sometimes fibrinous adhesions were formed between the area of the infarct and the mesentery or other abdominal organs. Cerebral infarcts were rarely observed macroscopically, although occasional asymmetry of the cerebral hemispheres was noted in extensive cases.
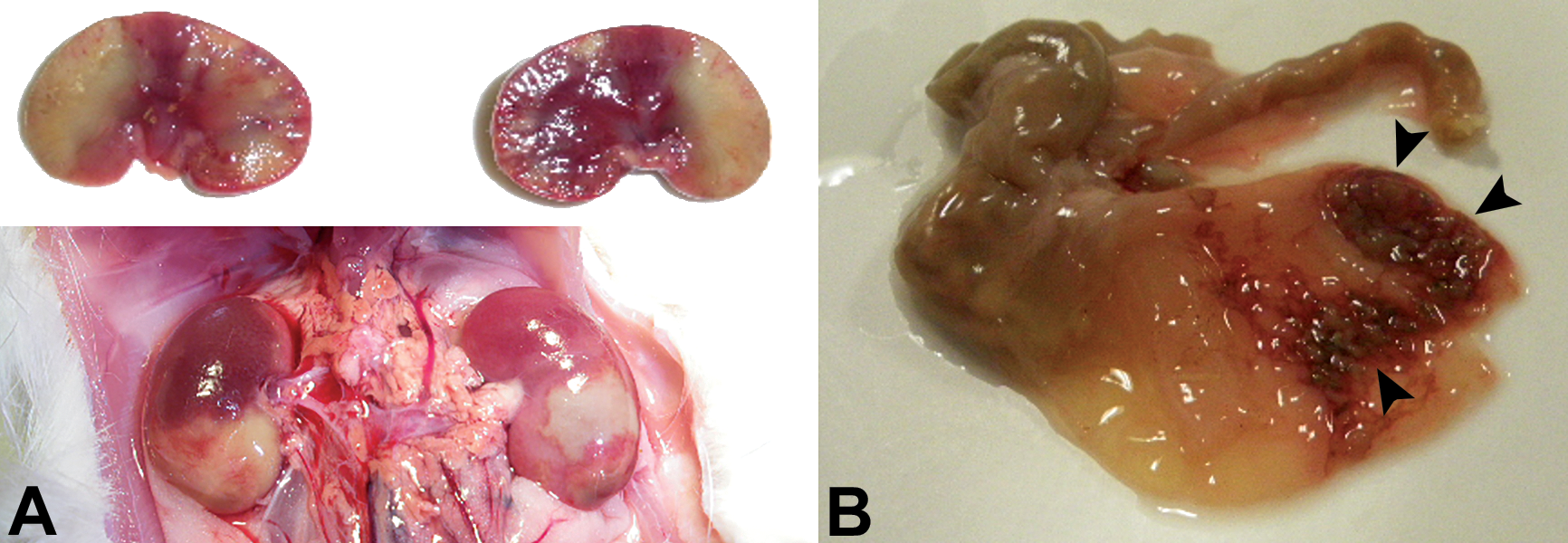
Macroscopic lesions associated with thromboembolic disease secondary to IVC were noted predominantly in the kidneys and cecum. In the kidneys, infarcts were visible as areas of pallor on the cortices (A, bottom) that extended into the medulla as wedge-shaped areas on cut section (A, top). Figure B shows cecal infarcts appeared as dark red sharply demarcated areas affecting 10% to 30% of the cecal tip (arrow heads).
On histopathologic analysis, lesions in IVC rats were classical for ischemic infarction secondary to occlusion of arterial blood supply by thromboemboli, predominantly affecting the kidneys (Figure 3A), cecum (Figure 3B), and cerebrum (Figure 3C). Thromboemboli were noted predominantly within the arterial blood supply of affected organs, and in rare cases, thrombi were noted to be propagating off the carotid catheter tip (Figure 3D) and observed within the aorta or proximal carotid artery in histologic sections of the heart and great vessels. Showering of thrombi into the aorta from the carotid artery catheter would account for the distribution of thromboemboli with secondary infarction into the arterial vasculature of many organs. Two animals, 1 found dead (CAC + JVC) and 1 terminal (CAC), were noted to have pulmonary thromboemboli, and in the found dead animal, the pulmonary thromboembolism was very large and considered the cause of death. The typical histologic appearance of infarcts consisted of regions of tissue which had undergone coagulative necrosis surrounded by a border of tissue reaction, including inflammation and fibrosis, which varied with the age of the lesions. Thromboemboli within vessels adjacent to or within the infarcts were noted in some but not all cases, probably due to the thrombus being outside of the plane of the examined section, though fibrinolysis of some thrombi may also have occurred. No histologic evidence of bacterial infection or septicemia was noted in this cohort of animals, indicating that the aseptic techniques used during catheter placement, maintenance, and use were adequate and that sepsis was not the primary cause of the thrombi.
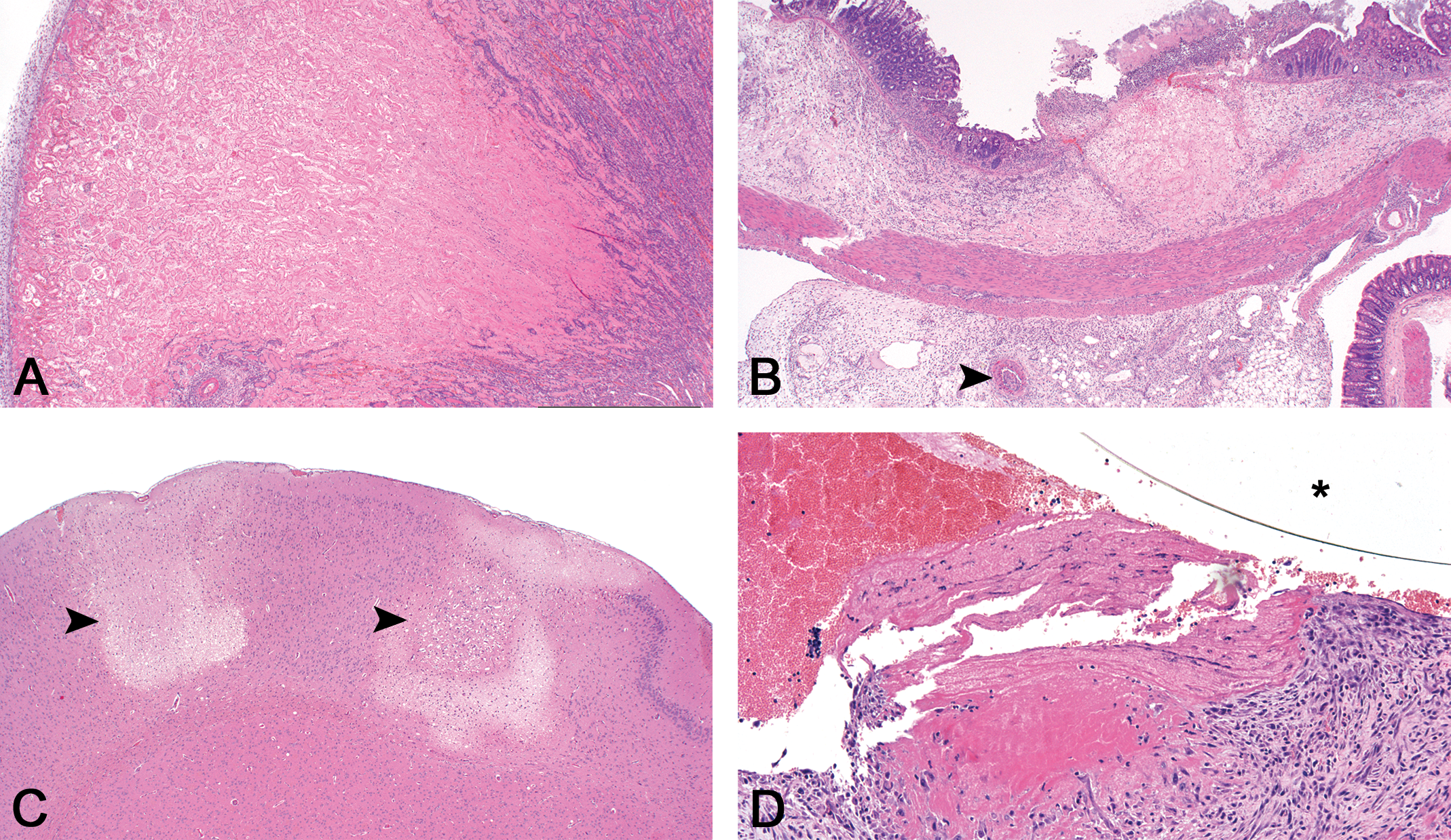
Microscopic examination of IVC infarcts in the kidneys, cecum, and cerebrum revealed that many were associated with thromboemboli in adjacent arteries. A wedge-shaped area of coagulative necrosis surrounded by peripheral inflammation and fibroplasia is classic for renal infarction (A). An infarct in the tip of the cecum (B) is associated with an arterial thrombus (arrow head). Two distinct regions (arrow heads) of infarction are present in the parietal cortex (C). A thrombus is propagating off the carotid artery catheter (*) and is partially organized at the site of attachment to the vessel wall (D). Haematoxylin and eosin.
IVC rats showed a very high incidence of renal infarction irrespective of clinical tolerance, with infarcts observed in 96% of animals (25/26), of which 56% (14/25) were associated with observed arterial thromboemboli (data not shown). Occlusive arterial thromboembolism was the cause of the infarcts, with the size of the infarct being attributable to the branch and size of the renal artery occluded. The degree of renal parenchyma lost to infarction was estimated from standard midline longitudinal histologic sections of the kidney and compared to the clinical status of the animals. Notably, terminal animals had less severe renal lesions, with an average loss of 23% (range 0–42.5%) versus animals which were euthanized for nontolerance which lost an average of 49% (range 7.5–100%) of renal parenchyma. Thus, the degree of renal parenchymal loss was correlated with clinical status of the animals. To further explore the effect of IVC on renal function, BUN and creatinine levels were examined at termination from a cohort of animals (
The total tissue area affected by cerebral infarction varied between individual rats; however, the infarction was typically unilateral and predominantly involved regions perfused by the middle cerebral artery including the lateral caudate putamen, parietal cortex, piriform cortex, hippocampus, and lateral thalamus (Nagasawa & Kogure, 1989). However, in 1 animal, infarcts were noted in the insular cortex at the level of the caudate putamen, diagonal band area, thalamus, and the contralateral cingulate cortex, which was a distribution more suggestive of a thromboembolic shower. Infarction of the cecum typically involved 10% to 30% of the cecal tip, with varying degrees of fibrosis and peritonitis relating to whether the infarction was transmural or involved only part of the wall. Thromboemboli were noted in some cases of cecal infarction.
Experiment 2: Catheter Material and Gauge
Experiment 2 was conducted to examine if catheter material and gauge could affect the incidence of thromboembolic disease in a cohort of 9 WH rats which underwent CAC surgery implanting a 2 French polyurethane catheter, in comparison to the standard 3 French silicone catheter. The histopathological incidence of thromboembolic disease in the experiment 2 cohort was compared to the experiment 1 cohort from the initial CAC ± JVC model analysis. Of the 9 rats, 3 were preterminally euthanized between days 5 to 10 postsurgery due to significant BW lost, on average 10.41% (
Experiment 3: Heparin Dosage Trial
As most thromboembolic lesions appeared to occur in the immediate postsurgical period, the effect of additional anticoagulative measures at the time of the surgery was assessed. To examine if increasing the heparin levels administered at the time of catheter placement affected the levels of thromboembolic disease, a cohort of 6 WH rats with 3 French silicone CAC + JVC underwent a trial using a higher concentration of heparin to flush the catheters directly after their implantation. In these rats, the catheter was flushed with 66-IU/ml heparin/0.9% saline solution and locked with 500-IU/ml heparin/glycerol lock solution, and this was compared to the experiment 1 cohort where catheters were flushed with a 10-IU/ml heparin/0.9% saline solution for a total of 33 UI/kg per catheter during surgery with the same lock solution. Of the 6 rats, 1 was preterminally euthanized on day 6 postsurgery due to significant BW lost (18%) in association with general malaise/pain signs. The 5 terminal animals, euthanized from 6 to 18 days following the surgery, did not display any clinical signs, with the exception of a slight BW loss, on average of 1.03% (
Experiment 4: Effect of Carotid Ligation without Catheterization on Thromboembolic Disease
To determine if surgical manipulation and ligation of the major vessels would induce thromboembolic disease or nonclinical tolerance, the carotid artery was surgically manipulated as for catheterization and ligated without catheter placement. None of the 10 HW and 10 SD rats, either undergoing carotid ligation (8/10) or sham surgery (2/10) had to be euthanized or died prior the end of the study. Animals in this analysis remained asymptomatic and maintained their BW for the duration of the experiment. Rats were observed clinically for 3 weeks, before euthanasia and examination of core tissues including brain, kidneys, heart, lung, liver, and spleen. There was no macroscopic or microscopic evidence of thromboembolic disease in any organ of any animal. Thus, carotid artery ligation and the associated surgery are insufficient to produce thromboembolic disease in either rat strain, and no difference in susceptibility was noted between WH or SD rats. Thus, the catheter placement, not the associated surgery and arterial ligation, is strongly implicated as the cause of the thromboembolic disease in IVC rats, with an incidence of 92% to 100% in animals receiving catheters, versus 0% in animals undergoing arterial ligation alone (Table 3). Again no evidence of infection or septicemia was observed in any of the rats, consistent with adequate aseptic technique.
Discussion
Thromboembolic disease with infarction was a significant problem in all of the IVC analyses conducted in this study (data summarized in Table 3), with an incidence of 83% to 100% in terminal animals receiving catheters and 100% in nontoleration animals. Infarction is the process of ischemic necrosis of tissue caused by occlusion of the arterial supply or venous drainage (Mosier 2012). In IVC rat models, the thromboemboli were present predominantly within the arterial system of many organs, consistent with a shower off the carotid artery catheter into the aorta, and thrombi on the carotid artery catheters were occasionally observed. Pulmonary thromboemboli could potentially propagate off the jugular vein catheter and enter the lung via the pulmonary artery, although thrombi from the carotid artery catheter could also potentially enter the lung via the bronchial arteries which carry the lungs oxygenated blood supply. Any thromboemboli from the jugular vein catheter entering the lung via the pulmonary arteries would be arrested in the capillary bed of the alveolar septa and would not enter the systemic circulation. Thus, the majority of thromboembolic lesions could be attributed to the carotid artery catheter.
Organs with poor collateral circulation or an end-arterial blood supply are more prone to infarction, as oxygenated blood is supplied to those tissues predominantly by a single artery. This explains the predominance of lesions in the kidneys, cecal tip, and brain. Thromboembolic disease is caused by 3 major factors known as Virchow’s triad, including endothelial injury, alterations in normal blood flow or turbulence, and hypercoagulability (Mosier 2012). For arterial thrombi, endothelial cell injury and blood flow turbulence are the predominant inciting factors ((Mosier 2012). In the CAC ± JVC model, endothelial injury from surgery and placement of the catheters as well as turbulent blood flow around the catheters are likely to be the predominant contributing factors. However, an inflammatory response to surgery and tissue handling may have promoted a procoagulant state (Shebuski & Kilgore, 2002). Supporting this hypothesis, it has been previously noted that rats undergoing permanent CAC have serum haptoglobin, and body temperature rises suggestive of a systemic inflammatory response (Fonseca et al. 2010). These authors also reported a high percentage of bacterial infection, which may account for this increase in inflammatory markers (Fonseca et al. 2010). In experiment 1, the majority of lesions were attributable to embolic showering off the CAC due to their anatomic site. However, the 5 rats that underwent dual CAC/JVC showed 100% nontolerance and included the 2 found dead rats. This suggests that the additional JVC catheterization may contribute to increased morbidity by promoting coagulopathy through inflammation associated with additional surgery and tissue handling or providing an additional nidus for clot formation with endothelial damage and blood turbulence.
In these studies on IVC rat models, a strong correlation between clinical tolerance and amount of renal parenchymal loss due to infarction was noted. Animals that tolerated the procedure lost on average between 14% and 23% of renal parenchymal versus nontolerance animals which lost 40% to 49%. Renal infarction is very rare as a background finding in this age and strains of rat, and all infarcts had a duration within the postsurgical interval, thus the renal infarction was directly attributable to the IVC. In animals that tolerated the IVC surgery, measurement of renal function using BUN and creatinine showed no evidence of azotemia. The kidney has enormous functional reserve, with an acute loss of greater than 75% of nephrons required before the development of azotemia, thus the normal clinical chemistry observed in these animals was consistent with the amount of renal damage observed and their clinical status (Tripathi et al. 2011). However, BUN and creatinine are relatively insensitive indicators of renal function, and as many IVC animals are used for PK/PD modeling of investigational drugs with potential renal excretion, the degree of renal damage observed in IVC rats may impact the validity of data taken from these animals. Thus, a recommendation of this study would be that postmortem investigations or the use of more sensitive biomarkers of renal function or acute kidney injury be used to assess renal parenchymal loss in critical PK/PD modeling studies, to ensure results were not affected by significant but potentially nonclinical kidney damage.
Loss of over 10% of initial BW and neurological symptoms were defined as clinical end points warranting euthanasia. While percentage of BW loss is a predictive measure of clinical tolerance, further clinical signs or humane end points that would help prevent spontaneous death or reduce the percentage of found dead animals were not identified, and additional work to identify more sensitive indicators of animal welfare is warranted.
Neurological clinical signs were a frequent occurrence following IVC surgery and a common cause of clinical nontolerance. In these studies, neurologic clinical signs were attributed to cerebral infarction. However, clinical neurological assessment showed a poor correlation with the presence of cerebral lesions. In the initial pathological assessment of 26 animals in experiment 1, of the 13 animals with neuropathology, only 61.5% (8/13) showed neurological clinical signs. Surprisingly, 33.3% (4/12) of asymptomatic terminal animals also had brain lesions. The poor correlation between clinical and pathologic assessment with regard to cerebral infarction in IVC rats likely reflects the insensitivity of standard clinical assessments to some of the more subtle neurologic impairments which would occur depending on the brain regions affected. Thus, to ensure optimal welfare, careful neurologic examination, perhaps with additional neurofunctional tests and observations, may be indicated in IVC rats.
Previously, Fonseca et al. (2010) had reported that renal infection and septicemia were a significant problem with permanent catheterization of the carotid artery in male Crl:SD rats. These authors reported an incidence of 80% kidney infection and 10% to 30% septicemia in their analyses as determined by kidney swab and blood cultures. However, no histologic analysis was conducted in this study, and the authors describe the macroscopic renal lesions noted in the rats as “white spots in the cortex similar to infarcts” (Fonseca et al. 2010). Histopathologic analysis is a more sensitive assessment of bacterial infection and septicemia, as bacteria are definitively identified within the tissue accompanied by an inflammatory response versus bacterial culture that has a high false positive rate due to contamination or terminal bacteremia. In this study, only 1 animal in experiment 3 on heparin analysis was noted to have bacterial infection. Thus, with adequate aseptic procedures, bacterial infection and septicemia are an infrequent complication of permanent vascular catheterization.
In this analysis, comparison of different catheters and increased heparin dose did not reduce the incidence of thromboembolic disease in IVC rats. It was noted on the histologic analysis that a majority of the infarcts were of an age similar to the postsurgical interval, suggesting a majority of thromboembolic lesions develop in the immediate postsurgical period. This suggests that additional anticoagulation measures prior to and in the 48- to 72-hr period following the surgery may significantly reduce the occurrence of thromboembolic disease. Likewise, minimizing handling of the catheters or omitting flushing during this period may reduce the showering of thromboemboli and allow thrombi associated with the catheters to either lyse or organize. Exploration of various anticoagulation regimes and flushing protocols warrants further study to optimize animal welfare.
Conclusion
In conclusion, a very high incidence of thromboembolic disease occurred secondary to IVC surgery in rats. Thrombi propagating off the catheters and showering into the aorta led to infarction in the kidneys, brain, and cecal tip, with the predominance of these organs explained by their vascular anatomy. Thromboembolic disease was the direct cause of clinical nontolerance in many animals, with cerebral infarction causing neurologic signs, and renal and cecal infarction resulting in BW loss and general malaise severe enough to warrant euthanasia. In this study, the amount of renal parenchymal loss secondary to infarction was highly correlated with nontolerance, and BW measurement was the most sensitive indicator of animal well-being and procedure tolerance. A percentage of BW lost over 10% of initial BW and neurological symptoms were the clinical end points warranting euthanasia identified in this analysis. There was a poor correlation between the occurrence of cerebral infarction and neurological clinical signs in the rats, suggesting a more detailed neurologic assessment may be indicated with IVC rats. The high incidence of thromboembolic disease in IVC rats irrespective of whether they clinically tolerate the procedure is of concern for accuracy of pharmacologic modeling and other studies these animals are used in, and postmortem or biomarker monitoring of renal function in PK/PD studies is highly recommended to ensure valid results are obtained.
Footnotes
Acknowledgments
The authors wish to thank Hannah Gunton, Wayne Attwell, Jeremy Wickens, Jayne Holby, Lisa-Marie Burrows, Lorrayne Stretton, Chris Wilson, Ben Stone, and Rosie Ochs for care they provided to those animals, the technical assistance with the surgery, in live data collection, necropsies, and histologic processing; and Dr. André Allavena for assistance with the statistical analysis.
Authors’ Contribution
Authors contributed to conception or design (RA, RW, JG, and MD); data acquisition, analysis, or interpretation (RA, RW, and MD); drafting the manuscript (RA and MD); and critically revising the manuscript (RA, RW, JG, and MD). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
