Abstract
Detecting and monitoring exocrine pancreatic damage during nonclinical and clinical testing is challenging because classical biomarkers amylase and lipase have limited sensitivity and specificity. Novel biomarkers for drug-induced pancreatic injury are needed to improve safety assessment and reduce late-stage attrition rates. In a series of studies, miR-216a and miR-217 were evaluated as potential biomarkers of acute exocrine pancreatic toxicity in rats. Our results revealed that miR-216a and miR-217 were almost exclusively expressed in rat pancreas and that circulating miR-216a and miR-217 were significantly increased in rats following administration of established exocrine pancreatic toxicants caerulein (CL) and 1-cyano-2-hydroxy-3-butene (CHB) as well as in rats administered a proprietary molecule known to primarily affect the exocrine pancreas. Conversely, neither microRNA was increased in rats administered a proprietary molecule known to cause a lesion at the pancreatic endocrine–exocrine interface (EEI) or in rats administered an established renal toxicant. Compared with amylase and lipase, increases in miR-216a and miR-217 were of greater magnitude, persisted longer, and/or correlated better with microscopic findings within the exocrine pancreas. Our findings demonstrate that in rats, miR-216a and miR-217 are sensitive and specific biomarkers of acute exocrine pancreatic toxicity that may add value to the measurement of classical pancreatic biomarkers.
Introduction
Many commonly prescribed medications have been linked to drug-induced pancreatic damage (Trivedi and Pitchumodi 2005). Although the majority of these cases are mild to moderate in severity, serious cases can occur, with mortality reaching 30% in cases of severe pancreatitis (Hung and Lanfranco 2014; Jones et al. 2015). Monitoring pancreatic injury during nonclinical drug development and in the clinic can be challenging due to the absence of reliable biomarkers. Classical biomarkers amylase and lipase have limited sensitivity for the diagnosis of pancreatitis (Treacy et al. 2001), with large increases typically required for reliable identification of injury (Zhang and Rouse 2014). Detection of amylase and lipase is additionally hindered by their short half-lives, which are less than or equal to 13.7 hr and 17.7 hr, respectively, in humans and far shorter in rodents (less than 2 hr for each; Junge 1985). Amylase and lipase also have poor specificity for exocrine pancreatic injury. Serum amylase increases can occur with salivary gland disease, macroamylasemia, and several other sources of intra-abdominal inflammation/pathology; similarly, increases in serum lipase can be seen with lingual gland disease as well as other sources of intra-abdominal pathology (Banks 1997; Matull, Pereira, and Odonoghue 2006; Smotkin and Tenner 2002). Nonspecific elevations in amylase and lipase can also occur following administration of certain drugs, a phenomenon that may be due to altered secretion or exocytosis (Pezzilli et al. 2011). In addition, since both amylase and lipase are renally excreted, diminished glomerular filtration can slow their clearance from the blood, further complicating the diagnostic value of these enzymes (Junge 1985; Hudson and Strombeck 1978).
MicroRNAs (miRNAs) are a family of small, noncoding RNAs that control gene expression through translational inhibition or degradation of messenger RNAs (mRNAs; He and Hannon 2004). Individual miRNAs or miRNA clusters may be expressed in a tissue- or disease-specific pattern (Lagos-Quintana et al. 2002; Mikaelian et al. 2013), which could provide an inherently high degree of specificity. In addition, miRNAs released into biofluids can be easily detected using available, sensitive assay systems (Laterza et al. 2009). Recent studies have demonstrated their utility as biomarkers for a variety of diseases, including drug-induced liver injury, cardiovascular disease, and cancer (Volinia and Croce 2013; Wang et al. 2009; Creemers, Tijsen, and Pinto 2012; Peng et al. 2016). Furthermore, miRNAs show particular promise in a drug development paradigm, as they are extraordinarily well conserved across species and often demonstrate notable stability in plasma (Mitchell et al. 2008; Laterza et al. 2009).
Several miRNAs have been shown to have value as potential biomarkers of acute exocrine pancreatic injury. Statistically significant increases in miR-216a have been reported in Sprague-Dawley rat models of acute pancreatic injury induced by
We investigated the utility of miRNAs as biomarkers of exocrine pancreatic injury, first confirming the expression of miR-216a and miR-217 in pancreas relative to other tissues and then performing a series of rat studies using established pancreatic toxicants CL and CHB as well as two proprietary compounds: compound A is primarily associated with exocrine pancreatic lesions and compound B primarily exerts damage at the endocrine–exocrine interface (EEI; Brenneman et al. 2014). To examine specificity, we also evaluated these biomarkers in a para-aminophenol (PAP) model of nephrotoxicity (Davis et al. 1983). Our results revealed that dose-responsive increases in miR-216a and miR-217 were present in Wistar Han rat models of exocrine pancreatic toxicity induced by CL, CHB, and the proprietary compound primarily associated with exocrine pancreatic damage, but not in Sprague-Dawley rats administered the proprietary compound associated with EEI damage or Wistar Han rats administered PAP. Increases in miR-216a and miR-217 persisted longer, were of greater magnitude, and often correlated better with exocrine pancreatic histopathology than did amylase and lipase. Taken together, our findings indicate that in Wistar Han rats, miR-216a and miR-217 are sensitive and specific biomarkers of exocrine pancreatic injury with utility superior to that of amylase and lipase.
Material and Method
Study Design
All
CL
Wistar Han rats (
CHB
Wistar Han rats (
Compound A
Wistar Han rats (
Compound B
Sprague-Dawley rats (
PAP
Wistar Han rats (
Measurement of miRNAs
Total RNA was isolated from approximately 120 µl of rat serum using the miRNeasy Mini Kit (Cat No. 217004; Qiagen) at the time points previously described following manufacturer’s instructions. The total RNA was eluted in 30 µl of RNase-free water. miRNAs were quantified using TaqMan MicroRNA Assays (Applied Biosystems, Carlsbad, CA) according to manufacturer’s instructions. Briefly, 10 μl of RNA was used in a 15 μl of reverse transcription reaction using Taqman miRNA Reverse Transcription Kit (Cat No. 4366597; Applied Biosystems); real-time (RT) PCRs were performed in duplicate using 1.33 μl of RT product each. Copy numbers of miRNA were determined using a standard curve of synthetic miRNA (Integrated DNA Technologies). The data were expressed as copies per microliter serum.
For measurement of tissue miRNAs, human FirstChoice® Total RNA Survey Panel was purchased from Ambion (Cat No. AM6000). Approximately 50 to 100 mg of Wistar Han rat or C57Bl/6 mouse heart, pancreas, kidney, liver, lung, skeletal muscle, spleen, and thymus tissues were homogenized in Qiazol, and RNA was extracted using the miRNeasy Mini Kit according to manufacturer’s instructions (Cat No. 217004; Qiagen). Approximately 50 ng of total tissue RNA was used to quantify miRNA as described above. The data were expressed as copies per nanogram RNA.
Clinical Chemistry
Serum levels of amylase, lipase, and a full panel of clinical chemistry parameters were measured at the time points previously described using Advia 1200 Chemistry System from Siemens according to manufacturer’s instructions.
Histopathology
Pancreas sections were fixed in 10% neutral-buffered formalin and embedded in paraffin wax. Processed tissues were sectioned at a thickness of 4 to 5 µm, stained with hematoxylin and eosin for microscopic examination, and then examined histologically by a board-certified veterinary pathologist. Tissues were scored for atrophy (encompassing features such as vacuolation/degeneration and single-cell necrosis) and inflammatory cell infiltration (either mixed cell or mononuclear cell-predominant). Lesion severity was scored on a 5-point scale where 0 =
Electron Microscopy
Three millimeter biopsy punch samples were taken from formalin-fixed paraffin embedded (FFPE) tissue blocks and processed for electron microscopic examination. Briefly, the FFPE tissue pieces were rehydrated by processing through xylene (3×), graded ethanols (100%, 95%, 70%, and 50%), and rinsed in phosphate buffer. The tissue pieces were refixed in phosphate-buffered Karnovsky’s Fixative (2% methanol-free formaldehyde + 2.5% glutaraldehyde) followed by postfixation in phosphate buffered 1% osmium tetroxide. The samples were dehydrated through graded ethanols (50%, 70%, 95%, and 100%), rinsed in propylene oxide (PO), and transitioned through graded epoxy resin (with PO). The tissue samples were then embedded in pure epoxy resin and cured at 60°C. Sections (∼0.6 µM) were prepared from selected tissue blocks, adhered to glass slides, and stained with Toluidine Blue O for light microscopic examination. Thin sections (∼90 nm) were prepared of select areas of interest, counterstained with uranyl acetate and lead citrate, and examined via transmission electron microscopy (Hitachi H-7100). Representative digital images were recorded and analyzed by a board-certified veterinary pathologist.
Statistical Analysis
The values of experimental data were expressed as the mean ± standard error of the mean and analyzed with 2-way analysis of variance or correlation analysis using Pearson calculations (Graph Pad prism software, San Diego, CA). Differences with
Results
Selection of Candidate miRNAs through Tissue Profiling
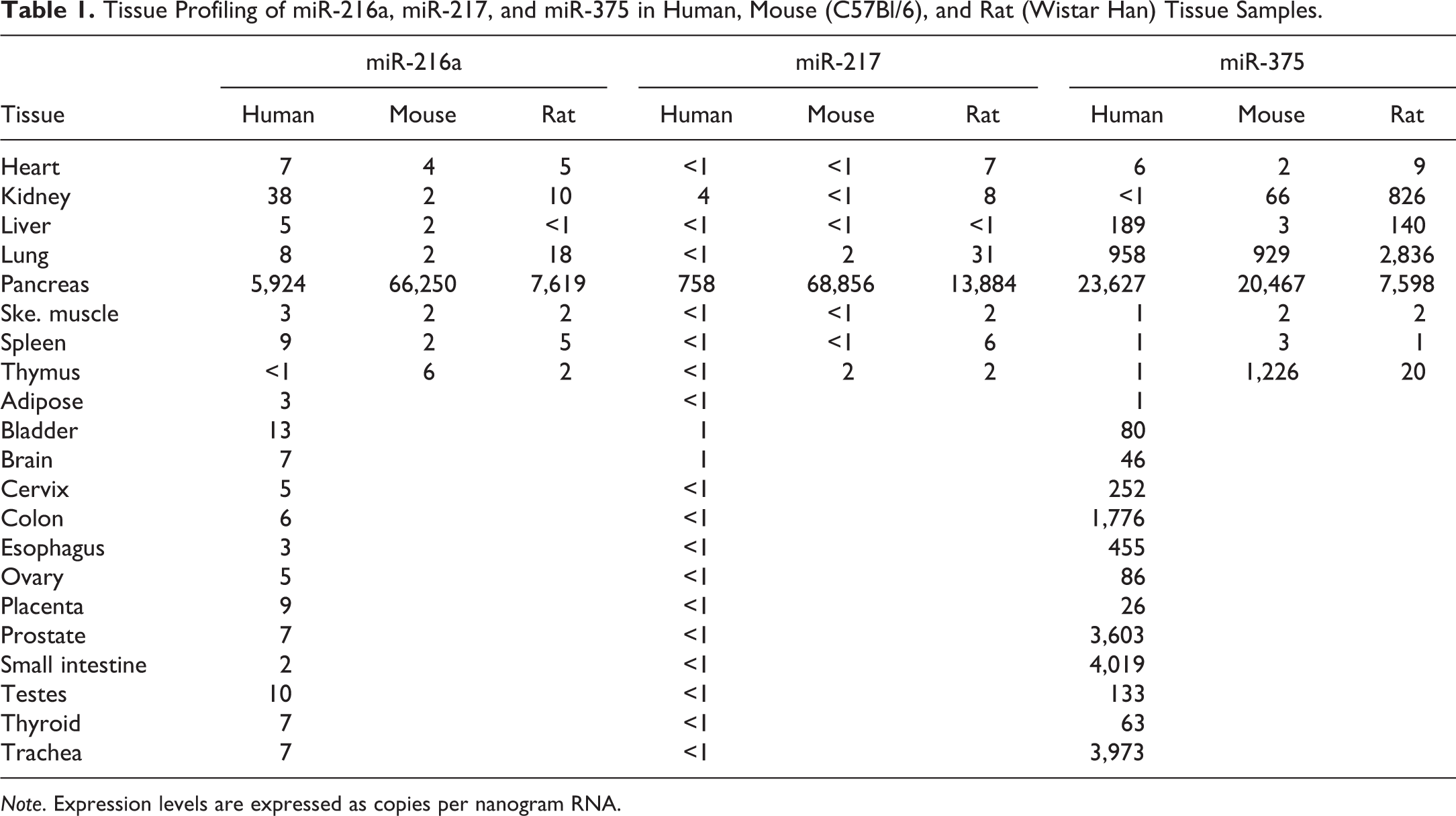
Previous miRNA array studies demonstrated that miR-216a and miR-217 are highly pancreas-specific in both humans and rats (Endo et al. 2013; Szafranska et al. 2007), and miR-375 has also been proposed as a biomarker of endocrine and exocrine pancreatic injury (Erener et al. 2013; Usborne et al. 2014). To verify that these 3 miRNAs are specifically and abundantly enriched in the pancreas across species, expression levels of miR-216a, miR-217, and miR-375 were measured in 21 human, 8 mouse, and 8 rat tissue samples (Table 1). Our results indicated that miR-216a and miR-217 were almost exclusively expressed in the pancreas across the human, mouse, and rat tissues examined. Although miR-375 was highly expressed in human, mouse, and rat pancreas, there were also low to medium expression levels present in several other tissues, including lung, small intestine, trachea, prostate, and colon in 1 or more species. As a result, miR-216a and miR-217 were selected to be evaluated as biomarkers in rat models of exocrine pancreatic toxicity.
Tissue Profiling of miR-216a, miR-217, and miR-375 in Human, Mouse (C57Bl/6), and Rat (Wistar Han) Tissue Samples.
In Vivo Qualification of miR-216a and miR-217 as Biomarkers of Exocrine Pancreatic Toxicity in Rats
CL study
CL, a cholecystokinin analogue, induces pancreatic lesions through excessive stimulation of protein secretion, resulting in autophagy/acinar degeneration and necrosis (Kim 2008; Zhang et al. 2014). Total doses of 45 and 150 µg/kg were selected based on previous reports (Usborne et al. 2014) to produce varying degrees of exocrine pancreatic damage.
Following dosing, minor clinical signs (rough hair coat and/or decreased activity) were observed in rats administered 45- or 150-µg/kg CL. Compared with rats administered vehicle, rats administered 45- or 150-µg/kg CL had significantly higher (though in a nondose-dependent fashion) serum amylase and lipase concentrations at the 1-, 3-, and 6-hr time points after dosing, with the highest values at the 3-hr time point. Amylase and lipase values were comparable to vehicle levels by 24-hr postdose (Figure 1A and B). In rats administered 45- or 150-µg/kg CL, serum miR-216a and miR-217 were higher in a time- and dose-dependent fashion, with the highest values at the 6- and 24-hr time points for miR-216a and miR-217, respectively. Significantly higher serum miR-216a concentrations were detected at the 3- and 6-hr time points and at the 1-, 3-, 6-, and 24-hr in the 45-µg/kg and 150-µg/kg dose groups, respectively, and significantly higher serum miR-217 were detected at all time points in both CL dose groups (Figure 1C and D). Although both miR-216a and miR-217 remained high at the 48-hr time points, the differences (compared with vehicle data) in miR-217 values were of greater magnitude than those of miR-216a.
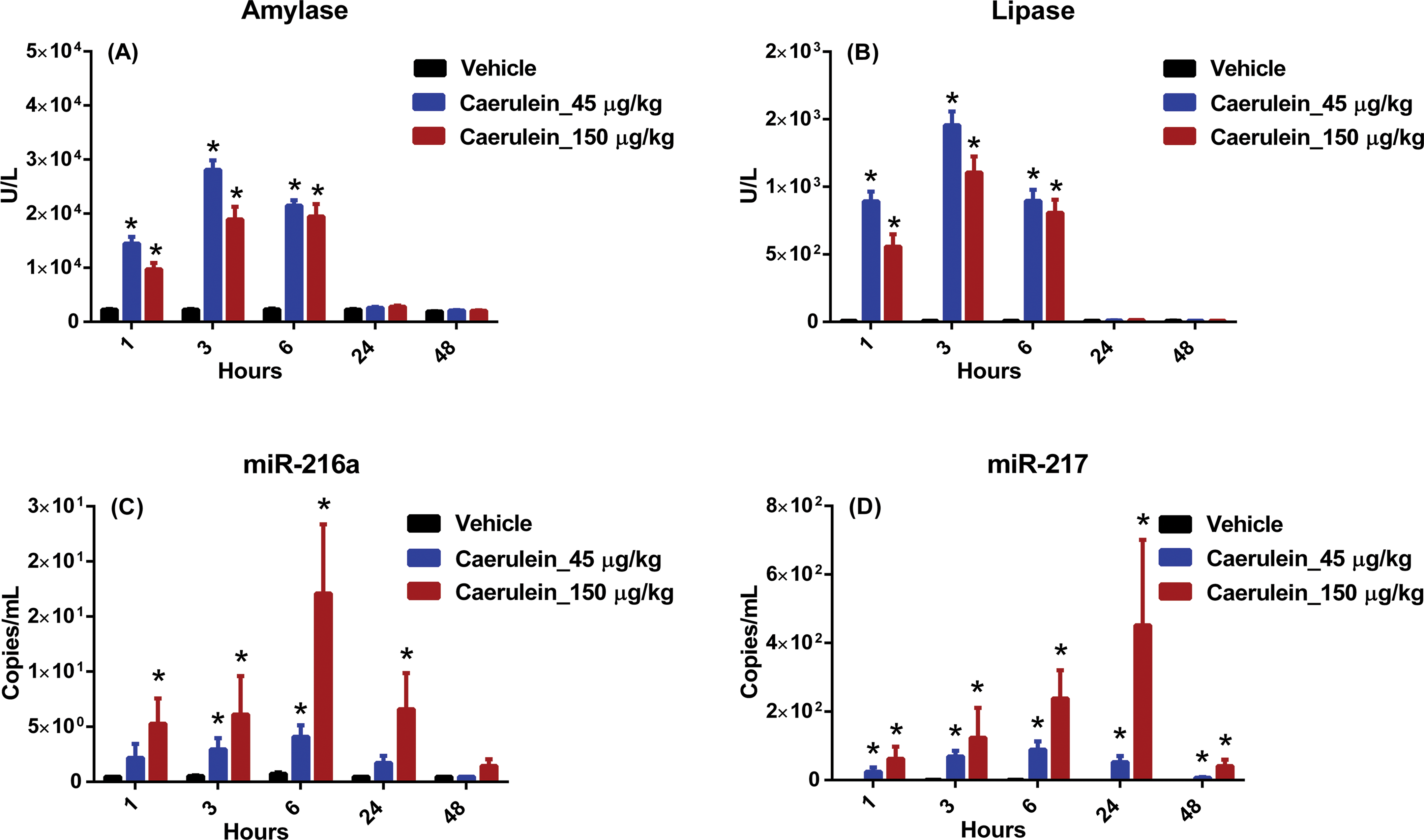
Caerulein (CL) study biomarker data. Wistar Han rats were administered 3 hourly doses intraperitoneally of either vehicle (black) or CL to achieve a total dose of 45 µg/kg (blue) or a total dose of 150 µg/kg (red). Following dosing, serum samples were collected at 1, 3, 6, 24, and 48 hr and analyzed for (A) amylase, (B) lipase, (C) miR-216a, and (D) miR-217. Statistically significant changes (2-way analysis of variance,
No macroscopic lesions were present in rats administered CL. Microscopic lesions were present in the exocrine pancreas of rats administered 45 or 150 µg/kg of CL and consisted of mild to marked atrophy of acinar cells characterized by loss of zymogen granules, reduced cell size, single-cell necrosis, and cytoplasmic vacuolation. Also evident was increased prominence of fibrous stroma and minimal to moderate inflammatory infiltrates composed of neutrophils, lymphocytes, plasma cells, and/or macrophages (Figure 2B and C). No effect on islets was noted. There were no CL-related lesions in the liver or kidney in any animals.
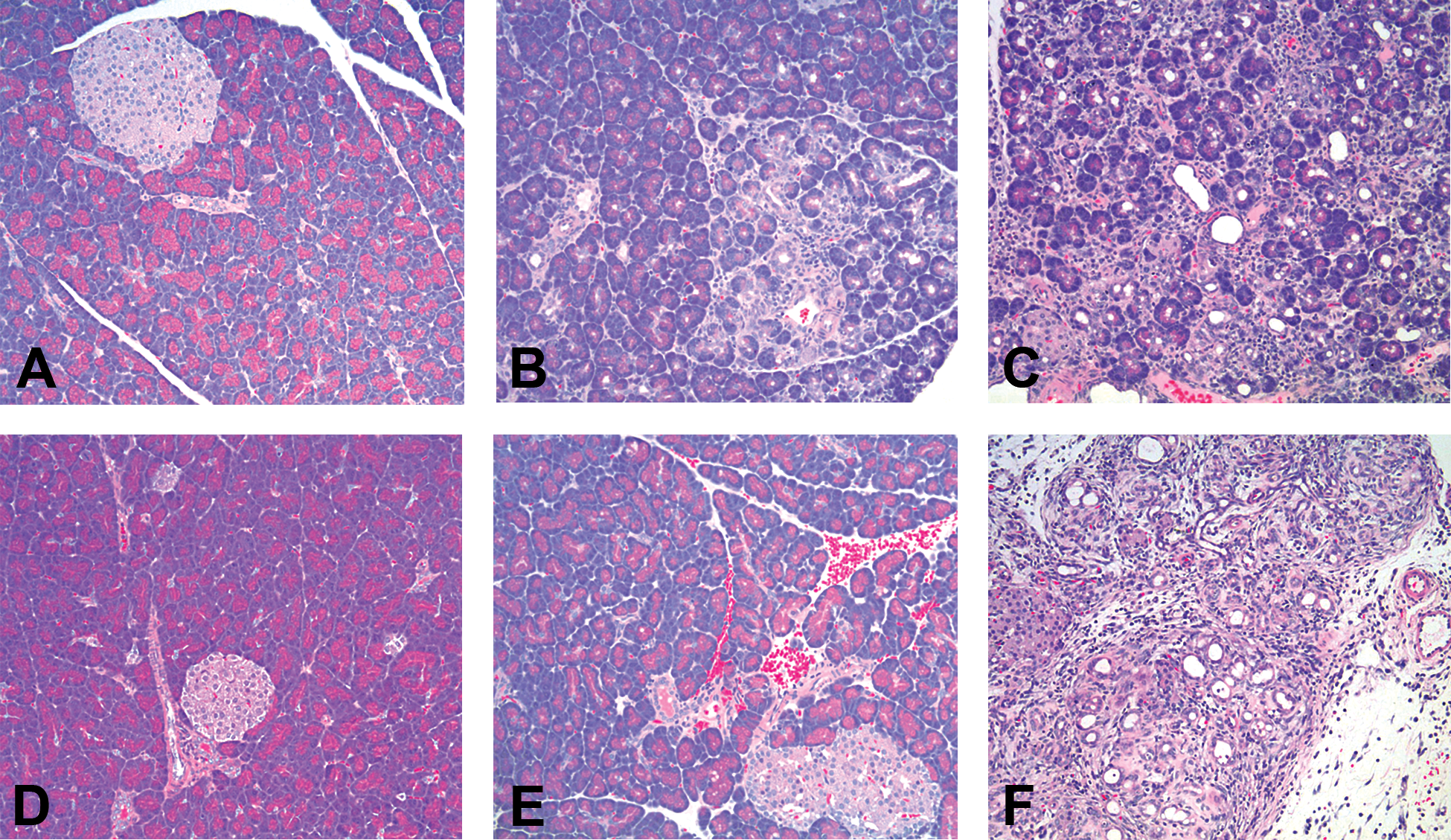
Light microscopy of pancreas from Wistar Han rats (100× original magnification, hematoxylin and eosin stain). (A) Pancreas from rat administered appropriate vehicle for caerulein (CL) was normal. Pancreas from rats administered a total dose of (B) 45 µg/kg CL or (C) 150 µg/kg CL showed dose-dependent acinar cell atrophy, single-cell necrosis, mixed cell inflammation, and increased prominence of fibrous stroma, as well as occasional regions of hemorrhage. (D) Pancreas from rat administered appropriate vehicle for 1-cyano-2-hydroxy-3-butene (CHB) was normal. Pancreas from rats administered (E) 75-mg/kg CHB was normal on light microscopy, while pancreas from rats administered (F) 150-mg/kg CHB showed widespread acinar cell atrophy, single-cell necrosis, mixed cell inflammation, and increased prominence of fibrous stroma.
CHB study
CHB, a naturally occurring plant nitrile originating from cruciferous vegetables, causes selective damage to the exocrine pancreas, possibly through alteration of glutathione metabolism, resulting in acinar cell apoptosis and acinar atrophy (Maher, Chernenko, and Barrowman 1991; Wallig, Gould, and Fettman 1988). Doses of 75 and 150 mg/kg were chosen based on previous reports (Usborne et al. 2014; Wang et al. 2014) to produce varying degrees of pancreatic damage. Following dosing, minor clinical signs (rough hair coat and/or decreased activity) were observed in rats administered 150-mg/kg CHB.
Compared with vehicle-dosed rats, those administered 150-mg/kg CHB had higher serum amylase and lipase values at the 6- and 24-hr time points after dosing, with the highest values and achieving statistical significance at the 24-hr time point, returning to vehicle levels by the 48-hr time point (Figure 3A and B). Compared with rats administered vehicle, rats administered 75-mg/kg CHB had slightly higher miR-217 values at the 24- and 48-hr time points after dosing. In rats administered 150-mg/kg CHB, higher serum miR-217 levels were observed at the 24-, 48-, 72-, and 144-hr time points after dosing, with statistical significance achieved at 24, 48, and 72 hr (Figure 3D). Rats administered 150 mg/kg also had significantly higher serum miR-216a at the 24- and 48-hr time points, with values returning to vehicle levels by 72 hr (Figure 3C).
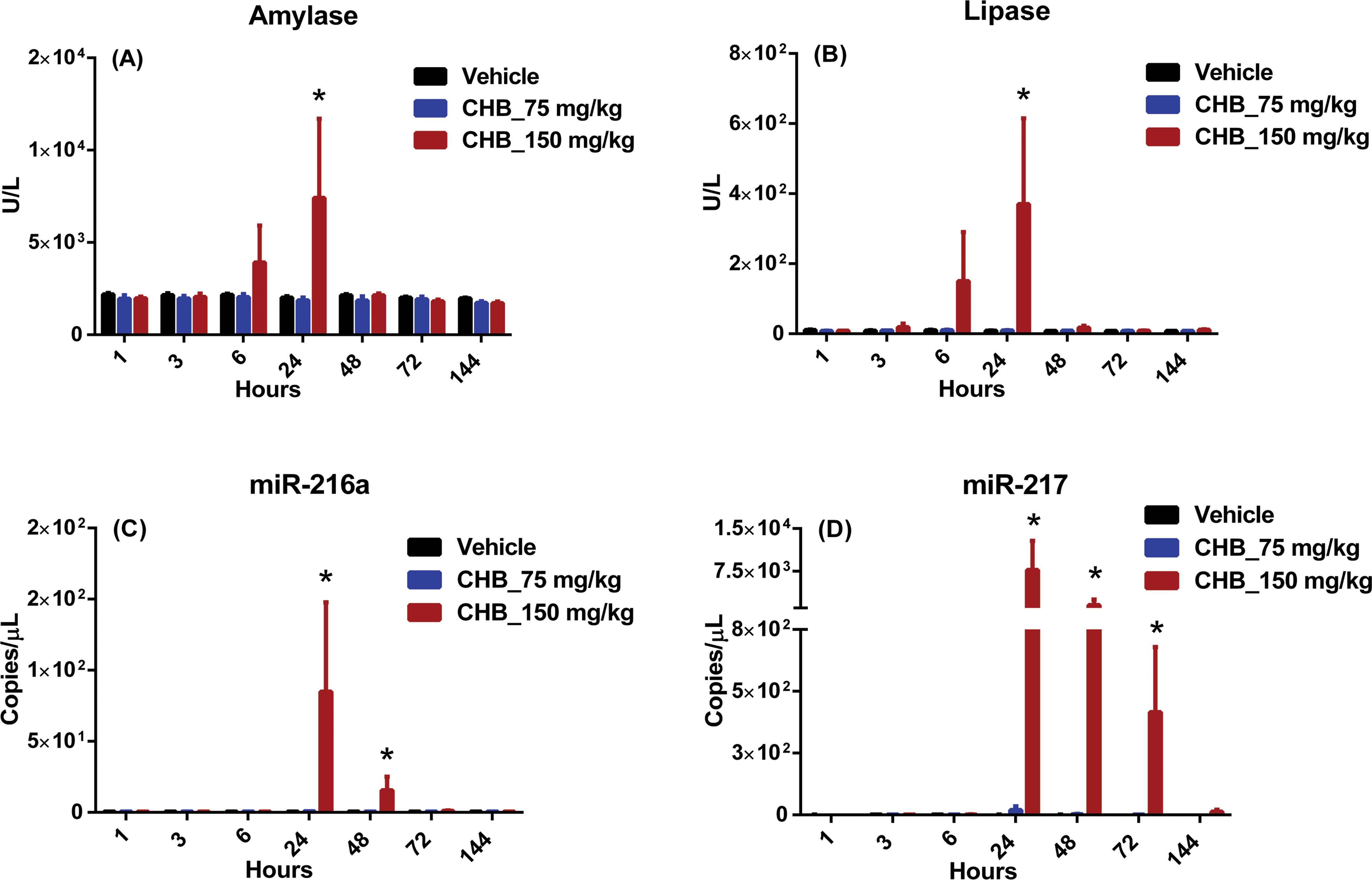
1-cyano-2-hydroxy-3-butene (CHB) biomarker data. Wistar Han rats were administered a single subcutaneous dose of vehicle (black) or CHB at 75 mg/kg (blue) or 150 mg/kg (red). Following dosing, serum samples were collected at 1, 3, 6, 24, 48, 72, and 144 hr and analyzed for (A) amylase, (B) lipase, (C) miR-216a, and (D) miR-217. Statistically significant changes (2-way analysis of variance,
Rats administered CHB had no macroscopic lesions. Microscopically, lesions were noted in the exocrine pancreas of rats administered 150 mg/kg/day of CHB, consisting of mild to severe atrophy of acinar cells characterized by edema, loss of zymogen granules, reduced cell size, cytoplasmic vacuolation, and single-cell necrosis. Multifocal regions of acinar loss with collapse of surrounding connective tissue were also present, along with minimal to moderate inflammatory cell infiltrates composed of neutrophils, lymphocytes, plasma cells, and/or macrophages (Figure 2F). No effect on islets was noted. There were no CHB-related lesions in liver or kidney in any animals.
To investigate causes of putative miRNA increases in animals lacking light microscopic pancreatic lesions, sections of acinar tissue from all dose groups were evaluated by electron microscopy. There were no findings in pancreatic tissue from rats administered vehicle. However, ultrastructural examination of acinar cells from rats administered 75 mg/kg/day revealed fragmentation and vacuolation of zymogen granules as well as dilation of the rough endoplasmic reticulum (Figure 4B). Acinar cells from rats administered 150 mg/kg/day had similar ultrastructural changes as well as membrane whorls and terminal bulbs affecting the rough endoplasmic reticulum. Animals in this dose group additionally showed release of zymogen granules into the interstitium as well as lysis and degeneration of acinar cells (Figure 4C).
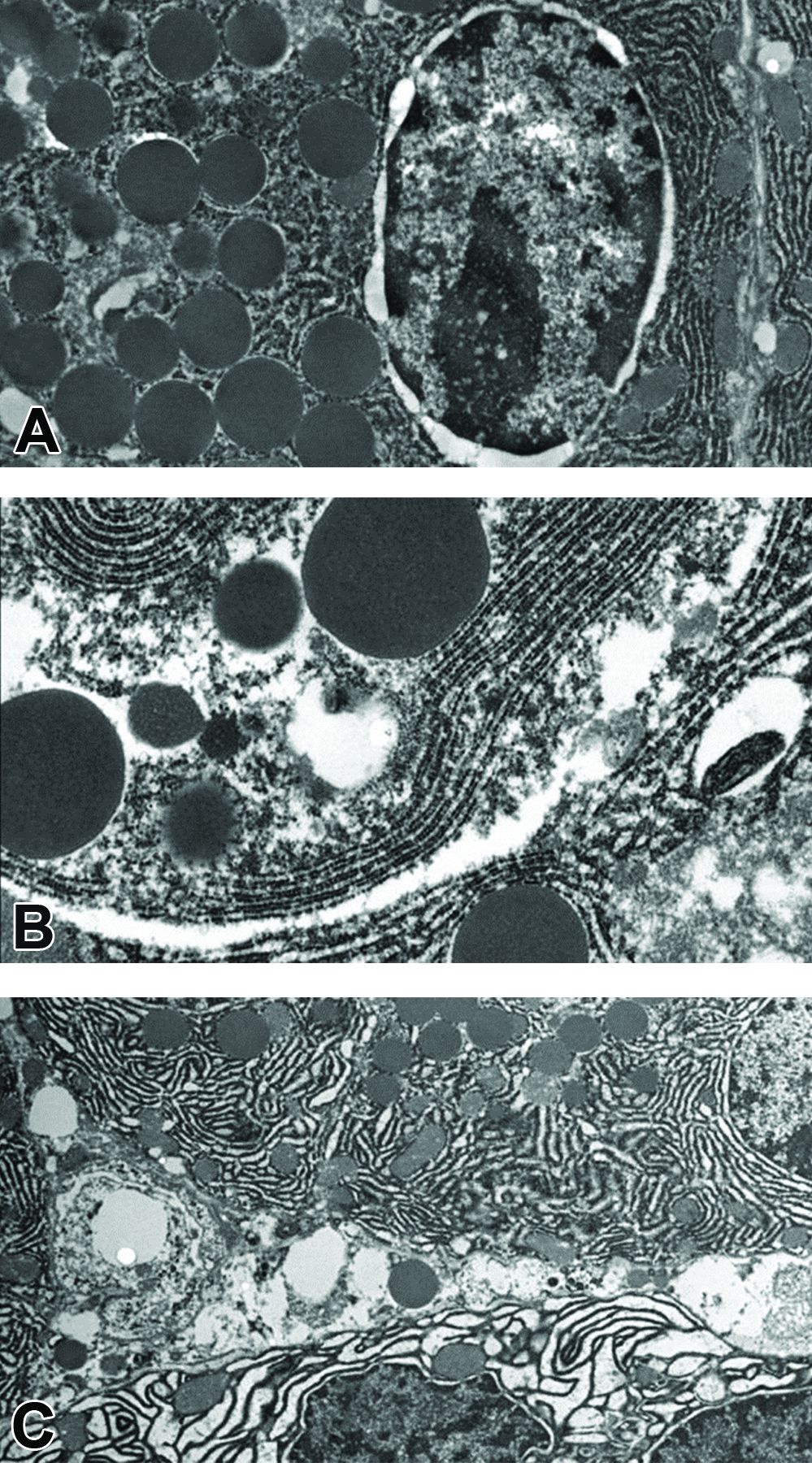
Ultrastructural micrographs of Wistar Han rat pancreatic acinar cells. (A) Normal acinar cell (5,000× magnification); (B) acinar cell from rat administered 75-mg/kg 1-cyano-2-hydroxy-3-butene (CHB) showing mild dilation of rough endoplasmic reticulum and vacuolation of zymogen granules (10,000× magnification); (C) acinar cell from rat administered 150-mg/kg CHB showing dilation of rough endoplasmic reticulum and release of zymogen granule into interstitium (5,000× magnification).
Compound A study
An undisclosed small molecule that has been associated with exocrine pancreatic toxicity in nonclinical rat studies (compound A) was selected as a tool compound, with doses of 100 or 300 mg/kg chosen to produce varying degrees of damage. Following dosing, clinical signs consisting of decreased activity and/or variably diminished skin turgor was observed in rats administered 100- or 300-mg/kg compound A. Two rats dosed at 300 mg were euthanized (1 each at 72 and 96 hr) prior to planned study conclusion for humane reasons, due to moribund condition.
There were no serum amylase changes in rats administered compound A (Figure 5A). Compared with rats administered vehicle, rats administered compound A had sporadically, neither significantly nor dose dependent, higher serum lipase values, with the highest values noted at 72 hr in 1 rat administered 300 mg/kg and at 120 hr in 1 rat administered 100 mg/kg (Figure 5B). In rats administered 100- or 300-mg/kg compound A, serum miR-216a values were higher in a time- and dose-dependent fashion at time points from 24 to 120 hr with the highest values noted at 120 hr; levels achieved statistical significance at 48 and 96 hr in rats administered 100 mg/kg and at all time points from 24 to 120 hr for rats administered 300 mg/kg. Serum miR-217 values were also higher compared with data from animals exhibited vehicle, in a time- and dose-dependent fashion at time points from 24 to 120 hr, with the highest values noted at 120 hr. Differences achieved statistical significance at the 24- and 48-hr time points in rats administered 100 mg/kg and at time points from 24 to 120 hr in rats administered 300 mg/kg. The fold changes in miR-217 were of greater magnitude than those seen with miR-216a (Figure 5C and D). Rats administered compound A also had minimal to marked increases in ALT, AST, ALP, GGT, GLDH, total bilirubin, and cholesterol values at the 120-hr time point (data not shown).
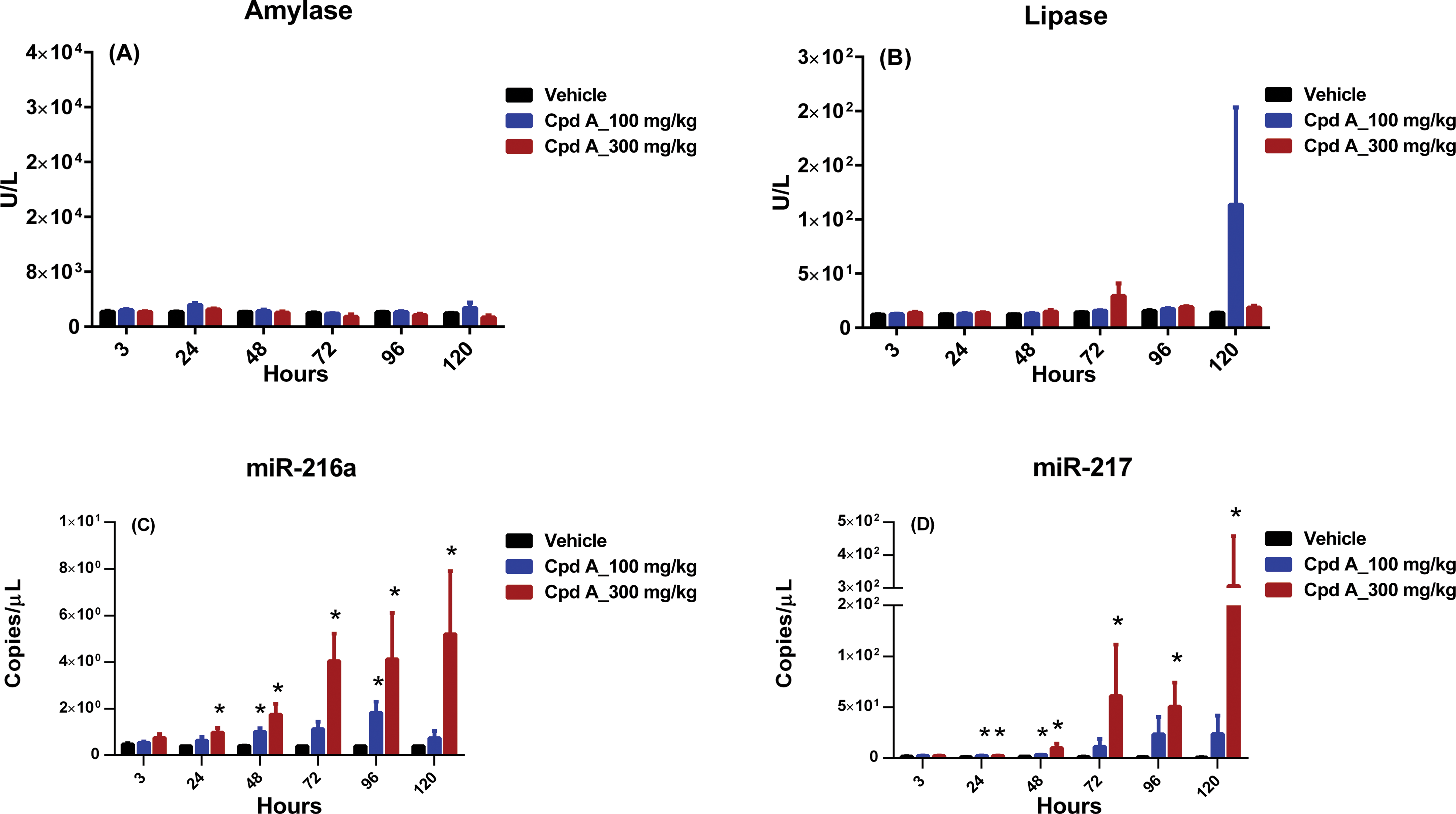
Compound A biomarker data. Wistar Han rats were dosed orally once daily for 3 days with vehicle (black) or compound A at 100 mg/kg (blue) or 300 mg/kg (red). Following dosing, serum samples were collected at 3, 24, 48, 72, 96, and 120 hr and analyzed for (A) amylase, (B) lipase, (C) miR-216a, and (D) miR-217. Statistically significant changes (2-way analysis of variance,
Macroscopically, smaller than expected pancreases and/or dark coloration of the pancreatic tissue were seen in rats in both dose groups. Microscopically, lesions were noted in the exocrine pancreas of rats administered compound A at 100 or 300 mg/kg, consisting of minimal to marked atrophy of acinar cells characterized by reduced cell size, cytoplasmic vacuolation, and single-cell necrosis as well as acinar loss with collapse of remaining connective tissue and minimal to mild inflammatory cell infiltrates composed of neutrophils, lymphocytes, plasma cells, and/or macrophages (Figure 6B and C). No effect on islets was noted. Rats in both dose groups also had microscopic lesions within the liver, consisting of minimal bile duct hyperplasia, minimal to mild single-cell necrosis of hepatocytes, and/or minimal to mild vacuolation of centrilobular hepatocytes. There were no compound-associated renal lesions in any animals administered compound A.
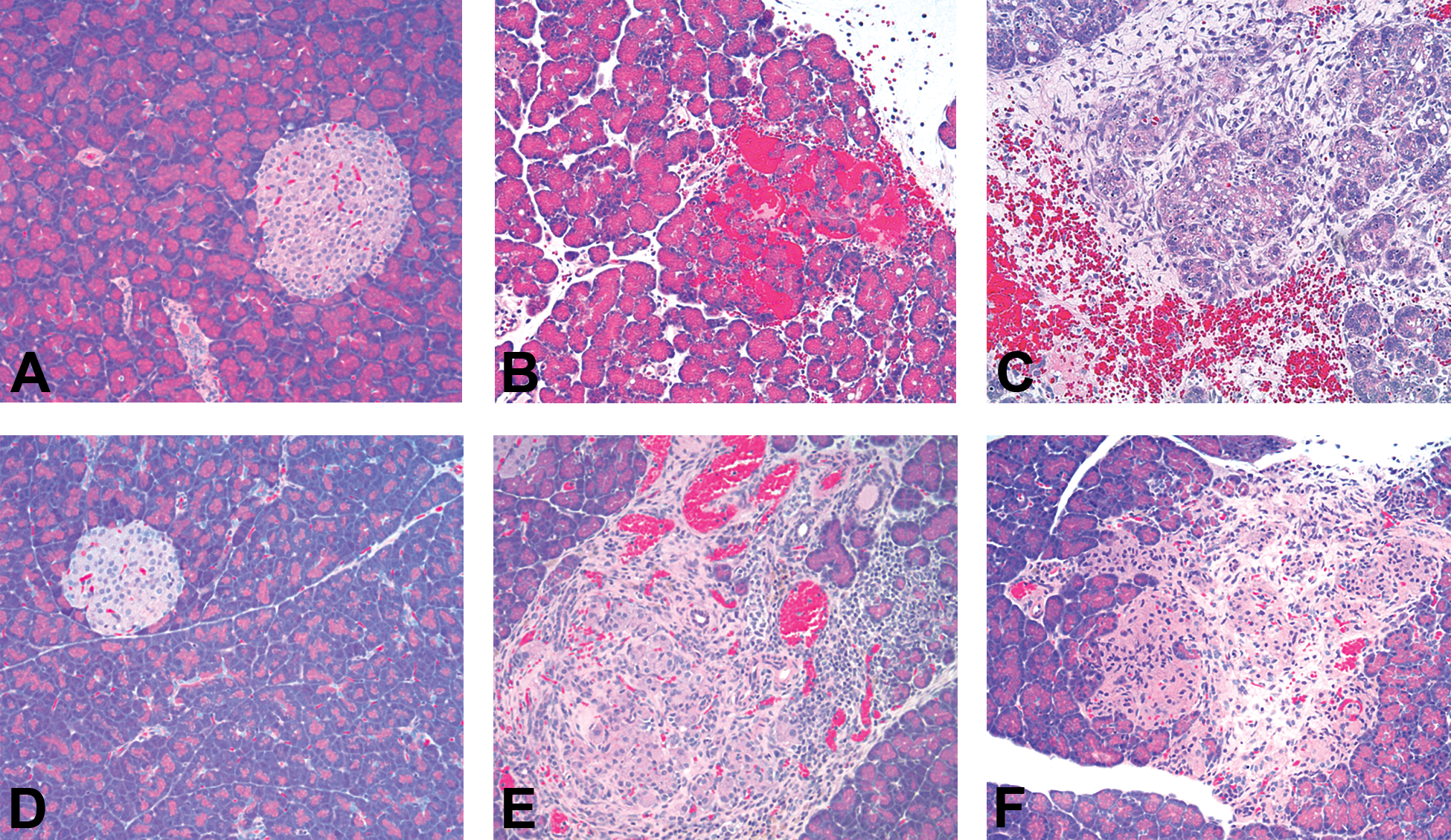
Light microscopy of pancreas from (A–C) Wistar Han and (D–F) Sprague-Dawley rats (100× original magnification, hematoxylin and eosin stain). (A) Pancreas from rat administered appropriate vehicle for compound A was normal. Exocrine pancreatic tissue from rats administered (B) 100-mg/kg or (C) 300-mg/kg compound A showed dose-dependent acinar cell atrophy, single-cell necrosis, mixed cell inflammation, and increased prominence of fibrous stroma, as well as occasional regions of hemorrhage. (D) Pancreas from rat administered appropriate vehicle for compound B was normal. Pancreatic tissue from rats administered (E) 10-mg/kg or (F) 100-mg/kg compound B showed lesions at the endocrine–exocrine interface, with multifocal regions of acinar cell atrophy and single-cell necrosis. Additional findings included peri-islet fibrin deposition and mixed cell inflammation.
Compound B study
An undisclosed test article known to cause pancreatic toxicity affecting the EEI (compound B) was selected as a tool compound, with doses of 10 or 100 mg/kg chosen based on previous reports (Brenneman et al. 2014) to produce varying degrees of damage. Following dosing, there were no changes in serum amylase, lipase, miR-216a, or miR-217 in rats administered 10- or 100-mg/kg compound B (data not shown).
Macroscopically, dark coloration affecting the pancreatic tissue was seen in rats of both dose groups. Microscopic findings were present in both acinar tissue and islets, localized adjacent to the junction of endocrine and exocrine pancreatic tissue. These findings consisted of atrophy characterized by reduced cell size and/or cellular loss, cytoplasmic vacuolation, and single-cell necrosis. Additional findings included hemorrhage, fibrin deposition, and minimal to mild inflammatory cell infiltrates composed of neutrophils, lymphocytes, plasma cells, and/or macrophages with occasional aggregates of hemosiderin-laden macrophages (Figure 6E and F). There were no compound-related liver or kidney lesions in any animal administered compound B.
PAP, a Nephroxicant, Did Not Cause Changes in miR-216a or miR-217
In order to evaluate specificity of miR-216a and miR-217 compared with amylase and lipase, cohorts of Wistar Han rats (
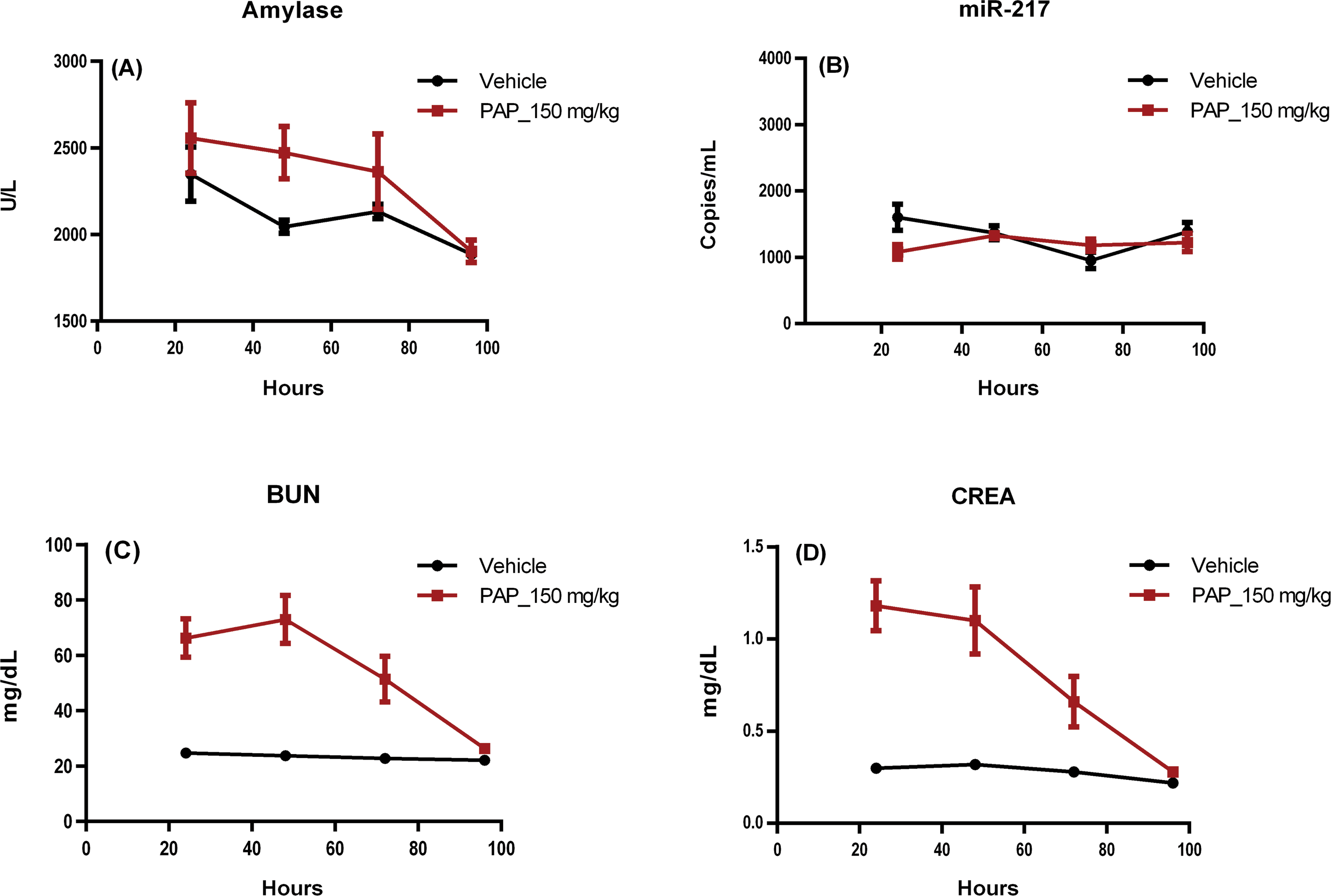
Para-aminophenol (PAP) biomarker data. Wistar Han rats were dosed orally once daily for 3 days with either vehicle (black) or PAP at 150 mg/kg (red). Following dosing, serum samples were collected at 24, 48, 72, and 96 hr and analyzed for (A) amylase, (B) miR-217, (C) blood urea nitrogen, and (D) creatinine. miR-216a levels were below the level of detection in all animals at all time points. Error bars represent standard error of the mean.
Microscopically, minimal to moderate renal tubular degeneration/necrosis/regeneration accompanied by the presence of minimal to mild cellular casts were seen in rats administered PAP. There were no compound-related lesions within the liver or pancreas of any animal administered PAP (Supplemental Figure).
Comparison of miRNA-216a and miRNA-217 with Amylase and Lipase
In order to evaluate the comparative utility of putative biomarkers miR-216a and miR-217 and conventional biomarkers amylase and lipase to detect microscopic exocrine pancreatic lesions, values from the peak (determined by the mean of all animals per dose group) day of each biomarker were plotted against the pancreatic histopathology sum score for the corresponding animal. In rats administered CL or compound A, better correlations were present between histopathology scores and miR-216a and/or miR-217 (Figure 8B and F) compared with those of amylase or lipase (Figure 8A and E), indicating that following exocrine pancreatic damage, miR-216a and miR-217 values may be better able to reflect the severity of microscopic lesions. Furthermore, following administration of CL, CHB, or compound A, a greater dynamic range was seen with miR-216a and miR-217, and these biomarkers were increased for a longer duration than were amylase and lipase (Figures 1, 3, and 5). Following the administration of compound A, miR-216a and miR-217 increased earlier than did amylase and lipase (Figure 5). Finally, in rats administered PAP to induce nephrotoxicity, notable (though statistically insignificant) increases in amylase were present, while miR-216a and miR-217 remained unchanged (Figure 7), indicating greater tissue specificity of these miRNA biomarkers for the exocrine pancreas compared with amylase. Taken together, these data provide strong evidence that miR-216a and miR-217 represent promising biomarkers of exocrine pancreatic toxicity with better sensitivity and specificity than amylase and better sensitivity than lipase.
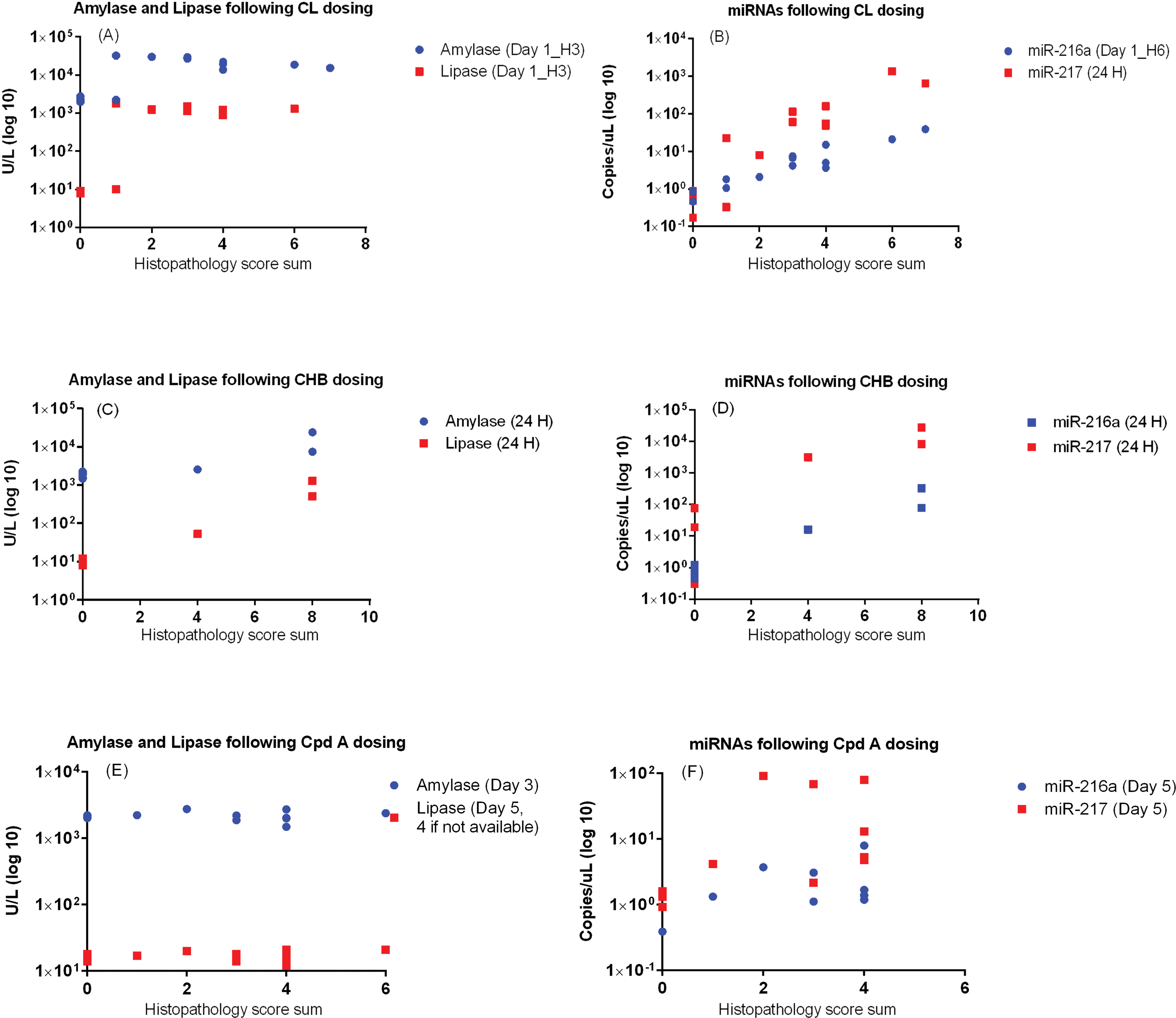
Comparison of miR-216a and miR-217 with amylase and lipase. Scatter plots showing individual values from the peak day (determined by the mean serum concentration of all animals per dose group) of amylase, lipase, miR-216a, and miR-217 measurements versus pancreatic histopathology score sum for each corresponding animal for (A–B) caerulein (CL), (C–D) 1-cyano-2-hydroxy-3-butene (CHB), and (E–F) compound A. There were no biomarker changes in animals administered compound B. Correlation coefficients are as follows: for CL, (A) amylase:
Discussion
The absence of reliable pancreatic biomarkers represents a major impediment to recognizing and monitoring exocrine pancreatic injury during all stages of drug development. The traditional biomarkers amylase and lipase are hindered by limited sensitivity and lack of specificity as well as short half-lives. Recently, several miRNAs (including miR-216a, miR-217, miR-375, and miR-216b) have been investigated as putative biomarkers of acute exocrine pancreatic injury in animal models of experimental pancreatitis induced by the administration of chemical secretagogues (CL), basic amino acids (
The abundance, ease of selection and amplification, and stability of serum/plasma miRNAs make them attractive biomarker candidates; in addition, tissue specificity is reported to be very high in many cases. However, gaps in understanding remain, including details about their half-lives, the exact mechanism(s) of their release, and variability of individual miRNAs during different disease states (Sasseville, Mansfield, and Brees 2014; Campion et al. 2013), though information about the latter is becoming increasingly available (Guo et al. 2014). Also until recently, high-throughput mechanisms for performing quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR), the method favored by many investigators for determining miRNA expression, had not been extensively evaluated and compared, though this is also being progressively remedied (Mestdagh et al. 2014; Farr et al. 2015; Tan, Khoo, and Tan 2015).
The tissue distributions of our candidate miRNAs were a critical consideration. Using tissue profiling, we were able to confirm that only miR-216a and miR-217, but not miR-375, were highly and nearly exclusively expressed in human, rat, and mouse pancreatic tissue. This substantiated previous reports of miR-216a and miR-217 enrichment within the human pancreas (Liang et al. 2007). Although our expression data failed to distinguish between exocrine and endocrine pancreas, the notable predominance of acinar cells in normal pancreas (approximately 90%) increases the likelihood that candidate miRNAs would be expressed in exocrine tissue (Szafranska et al. 2008). Additional confidence came from prior investigations utilizing laser capture microdissection, which demonstrated that miR-216a is enriched in acini but not islets of Langerhans; concurrently, miR-375 was shown to be highly expressed in the islets but not acinar cells (Endo et al. 2013). Although similarly detailed investigation of miR-217 in nonneoplastic pancreatic tissue does not currently exist in the literature, expression has been shown in a sample of normal pancreatic acinar cells by
Our investigations utilized two well-described tool compounds, CL and CHB, both of which cause injury to the exocrine pancreas. Following administration of CL or CHB, we observed brief increases in amylase and lipase that normalized by 24 and 48 hr postdose, respectively. In contrast, in rats administered CL or CHB, serum miR-216a and miR-217 were increased for a longer duration (possibly indicating longer half-lives for both putative biomarkers) and also had greater dynamic range. In addition, compared with amylase and lipase data, similar (CHB) or better (CL) positive correlation was noted between maximal candidate miRNA values and pancreatic histopathology scores (Figure 4A–D).
Occasional increases in miR-217 were observed in animals administered 75-mg/kg CHB in the absence of either traditional biomarker elevations or light microscopic changes, a finding that prompted ultrastructural evaluation of the exocrine pancreas. Acinar cell lesions detected by electron microscopy in animals from this dose group included dilation of rough endoplasmic reticulum accompanied by fragmentation of zymogen granules, findings that were also noted (accompanied by further lesions) in rats administered 150-mg/kg CHB. These findings are similar to those previously reported in rats dosed with CHB and are thought to represent cellular apoptosis and/or necrosis (Wallig, Gould, and Fettman 1988). Although further investigation is obviously needed, these findings may indicate that in some situations, miR-217 may be more sensitive than light microscopy at detecting exocrine pancreatic damage.
To further examine the utility of miR-216a and miR-217 as biomarkers of exocrine pancreatic injury, we selected 2 proprietary compounds that have incited distinctive pancreatic lesions in rats. While amylase and lipase showed minimal value as markers of pancreatic damage in rats administered compound A, miR-216a and miR-217 increased in a dose-dependent manner, with multiday duration and wide dynamic range. Although there were also hepatic lesions present in rats dosed with compound A, it was not thought that this was likely to be a source of miR-216a or miR-217, given negative tissue expression findings for these miRNAs in the liver.
Compound B primarily affects endothelial cells at the periphery of the islet; however, there is subsequent damage via compromise of the islet–acinar portal system (presumed to be ischemic in nature) to the adjacent endocrine and acinar tissue. Brenneman et al. reported a lack of test article–related increases in amylase or lipase in rats dosed with this compound; rather, they describe a dose- and duration-dependent decrease in amylase across studies. In our study, there were no test article–related effects on either amylase or lipase; neither were there test article–related increases in either putative miRNA, despite the common presence of exocrine pancreatic damage affecting tissue adjacent to the islets. The reason for the lack of effects on either traditional or miRNA biomarkers in rats dosed with compound B is unknown. Although it is possible that corresponding effects on biomarkers were not captured due to timing, this seems unlikely considering the number and frequency of sampling time points in our study and the extensive measurements pursued by Brenneman et al. as well as (in the case of our investigation) the prolonged duration of the higher values for both miRNAs compared with amylase and lipase data. It is perhaps more likely that the absence of biomarker signals in response to damage localized to this area is a result of inherent heterogeneity of acinar tissue, which exhibits distinct morphologic and physiologic features in peri-insular (immediately adjacent to the islets) and tele-insular (more distant from the islets) regions (Aughsteen and Kataoka 1993; Pandiri 2014), including differences in secretory granule content (Gingras and Bendayan 1992). In fact, prior work has demonstrated that in dogs, glucose and glucagon exert an inhibitory effect on exocrine pancreatic secretion on amylase, lipase, and protease (Nakajima and Magee 1970), and that in rats, the peri-insular acinar cells are more resistant to degranulation than are tele-insular cells in response to pilocarpine injection (Putzke and Said 1975). Other factors that may have contributed to a lack of biomarker signals include possible asynchronous development of lesions within the pancreas resulting in the reduction in peak biomarker concentrations in circulation and/or unintended test article–related inhibition of miRNAs themselves (Gumireddy et al. 2008; Zhang et al. 2010). Importantly, this was the only study we performed in which Sprague-Dawley (as opposed to Wistar Han) rats were utilized, since we wished to precisely recapitulate the previously described EEI lesion. It is unknown whether rat strain played a role in the development of the lesion, putative effects on miRNA release following compound B administration, or both, although increases in miR-216a and miR-217 have been previously demonstrated in several Sprague-Dawley rat models of exocrine pancreatic toxicity (Endo et al. 2013; Usborne et al. 2014; Goodwin et al. 2014). From these data, we concluded that miR-216a and miR-217 have poor utility as biomarkers for compound B–induced pancreatic lesions affecting the EEI in Sprague-Dawley rats.
One of the most notable flaws of both amylase and lipase is their lack of specificity. In order to demonstrate improved utility of our candidate miRNAs over traditional biomarkers, it was crucial to establish that increases in miR-216a and miR-217 are more specific to exocrine pancreatic damage than are increases in amylase and/or lipase. Using a rat model of PAP-induced nephrotoxicity, we observed trends toward increases in amylase, most likely due to diminished glomerular filtration of this enzyme. miR-216a and miR-217 were unaffected in these animals, indicating that neither renal tubular damage nor decreases in glomerular filtration rate impact the circulating levels of our putative miRNAs.
Our data demonstrate that miR-216a and miR-217 represent promising biomarkers of exocrine pancreatic toxicity in rats and may add value when assessed alongside amylase/lipase in nonclinical studies. Tissue expression profiling indicated that both are almost exclusively expressed in pancreas in rats as well as mice and humans. Compared with amylase and lipase, increases in miR-216a and miR-217 were of greater magnitude, persisted longer, and/or had better positive correlation with exocrine pancreatic damage arising from toxicants that cause primarily acinar lesions. Furthermore, in a rat model of nephrotoxicity, trends toward increases were observed in amylase but not in miR-216a and miR-217. Taken together, our results indicate that as exocrine pancreatic biomarkers in rats, miR-216a and miR-217 have better sensitivity and specificity than amylase, better sensitivity than lipase, and better dynamic range than both amylase and lipase. Although further qualification is necessary to fully characterize the ability of these biomarkers to detect and monitor exocrine pancreatic injury, our report provides evidence that miR-216a and miR-217 are likely to add value to nonclinical rat studies in which acinar damage could occur.
Footnotes
Acknowledgments
Special thanks to Karrie Brenneman, Ellen Evans, William Reagan, Frederick Sace, Dalia Kalabat, Rolla Yafawi, and the Pfizer La Jolla Histology group for their contributions to and/or critical review of this article.
Author Contribution
Authors contributed to conception or design (JW, WH, ST, WHu, AJ, AV); data acquisition, analysis, or interpretation (JW, WH, ST, TB, WB, HG, WE, WHu, AV); drafting the manuscript (JW, AV); and critically revising the manuscript (JW, WH, ST, TB, WB, HG, WE, WHu, AJ, AV). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
