Abstract
Cytological bone marrow evaluation is utilized in nonclinical toxicology studies to characterize hematopoietic effects when the combined interpretation of histologic and complete blood count data does not yield sufficient information. Results from cytological bone marrow examination should be interpreted in the context of variability observed in concurrent control animals with consideration of cytologist experience and historical/published data. Cytological bone marrow differential counts and cellular morphologic findings from 130 (66 male, 64 female) healthy control cynomolgus monkeys from nonclinical toxicology studies were retrospectively analyzed. Myeloid to erythroid (M:E) ratios and the percentage of total cells for each cell type were determined from differential cell count data. M:E ratios ranged from 0.6:1 to 2.3:1. Percentages of total granulocytic cells, total erythroid cells, and lymphocytes ranged from 26.6% to 60.6%, 25.7% to 52.2%, and 5.5% to 40.4%, respectively. Monocytes, plasma cells, mast cells, and mitotic figures were typically <1% of total cells. Notable morphologic findings included occasional giant neutrophilic metamyelocytes and band neutrophils, ring-shaped band neutrophil nuclei, metarubricyte nuclear blebbing and binucleation, multiple or nonfused megakaryocyte nuclei, and emperipolesis. These results represent cytological bone marrow findings from healthy control cynomolgus monkeys utilized in nonclinical toxicology studies and provide insight into expected background variability.
Standard evaluation of the hematopoietic system in nonclinical toxicology studies includes histological evaluation of the bone marrow and review of complete blood count (CBC) data. If the combined evaluation of these standard assays does not yield sufficient information to fully characterize toxicologic effects, further analysis of the hematopoietic system by cytological bone marrow evaluation may be warranted as described by Reagan et al. (2011). Test article–related cytological bone marrow effects should be identified in the context of the relative degree of variability observed in concurrent control animals. Experience of the cytologist as well as internal and published historical control data may assist in the identification of test article–related effects, particularly if data from control animals are limited or do not appear to be representative (e.g., low sample size or control samples of poor quality).
The cynomolgus monkey (
Method and Material
Control monkeys of both sexes with complete quantitative femoral bone marrow cytological evaluations performed by one of the authors (C.M.C., L.C.C., or A.D.A.) were identified retrospectively. A total of 66 males and 64 females were included from studies conducted between 2008 and 2016.
Cynomolgus Monkeys and Husbandry
All study protocols and animal housing conditions were reviewed and approved by MPI Research Institutional Animal Care and Use Committee. Male and female cynomolgus monkeys originating from Mainland Asia (China or Vietnam), with ages ranging from 2 to 8.25 years, were housed at an Association for Assessment and Accreditation of Laboratory Animal Care–accredited facility. Species-specific housing was provided in climate-controlled rooms with a 12-hr light/dark cycle. Monkeys were fed a standard primate diet (Lab Diet® Certified Primate Diet #5048; PMI Nutrition International, Inc., Richmond, IN) with free access to water and additional food items occasionally provided as environmental enrichment. Animals were deemed clinically healthy prior to inclusion on study based on the evaluation of vendor health records/screens, physical exams, and pretest hematology, coagulation, and clinical chemistry profiles. At the termination of each study, animals were euthanized via intravenous barbiturate overdose (e.g., administration of pentobarbital or pentobarbital-based euthanasia solution).
Hematology Evaluation
CBCs were evaluated to assess animal health and determine if any animals had hematologic abnormalities typical of underlying bone marrow pathology that would warrant exclusion from the study. Prior to euthanasia, whole blood was collected from the femoral vein or other suitable blood vessel and anticoagulated with K3EDTA. CBCs were performed using an ADVIA® 120 or 2120 (Siemens Healthcare Diagnostics, Tarrytown, NY) within 24 hr of blood collection. End points evaluated included total leukocyte, neutrophil, lymphocyte, monocyte, eosinophil, basophil, large unclassified cell, erythrocyte, reticulocyte, and platelet counts, hemoglobin concentration, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration. Blood smears were prepared, stained with Wright–Giemsa using an automated stainer (Hematek® Slide Stainer; Bayer HealthCare LLC, Tarrytown, NY), and reviewed by light microscopy as warranted by institutional standard operating procedures.
Bone Marrow Smear Collection and Cytological Evaluation
Two femoral bone marrow smears were prepared for each animal using the paintbrush collection technique (Reagan et al. 2011; Provencher Bolliger 2004). Briefly, the femur was removed immediately following euthanasia and the bone marrow was exposed. A clean, natural-bristle paintbrush was moistened in 5% bovine serum albumin. A modest amount of bone marrow from the grossly red (active) area of the proximal femur was collected with the brush and deposited on labeled glass slides in three wavy streaks. Smears were air-dried and stained twice with Wright–Giemsa using an automated stainer (Hematek Slide Stainer). Cytological evaluations were performed by a diplomat of the American College of Veterinary Pathologists (C.M.C, L.C.C., or A.D.A.). Smears were evaluated subjectively by light microscopy at low power (10× and/or 20× objectives) for overall quality, cell preservation and distribution, adequacy of megakaryocyte numbers, and the presence of stromal elements. At high power (50× and/or 100× oil objectives), a minimum of a 400-cell differential count was performed (Reagan et al. 2011), with all cells in each included field counted to maintain representative ratios of cell types. Counts included differentiation of myeloblasts, promyelocytes, neutrophilic myelocytes, neutrophilic metamyelocytes, band neutrophils, segmented neutrophils, eosinophils (all stages), basophils (all stages), myeloid mitotic figures, rubriblasts, prorubricytes, basophilic rubricytes, polychromatophilic rubricytes, metarubricytes, erythroid mitotic figures, lymphocytes, monocytes, plasma cells, and mast cells. The presence and adequacy of polychromatophils were subjectively assessed during the differential cell count. Morphologic evaluations were performed at high power.
Calculations and Statistics
For each sex, myeloid to erythroid (M:E) ratios and the percentages of individual cell types, total granulocytic cells, and total erythroid cells relative to total bone marrow cells were calculated. Percentages were compiled, Dixon’s outlier range statistic was applied, and group medians were calculated. To compare sexes, data were evaluated for normal distribution using the D’Agostino and Pearson omnibus normality test, and Mann–Whitney
Results
Hematology Evaluations
No hematologic changes were observed that would warrant the exclusion of any animal for the bone marrow evaluations. All individual values for CBC endpoints were within the expected ranges for biological and/or procedure-related (i.e., decreases in red cell mass and/or increases in reticulocyte counts resulting from repeated blood collections) variation.
Cytological Bone Marrow Cell Differential Counts and Morphologic Evaluations
A total of 66 male and 64 female monkeys had complete cytological bone marrow evaluations performed by one of the authors (C.M.C., L.C.C., or A.D.A.). Bone marrow smears obtained immediately following euthanasia typically yielded high-quality smears with adequate cellularity for performing differential cell counts. There were predominantly hematopoietic cells, although variable numbers of adipocytes and low numbers of other nonhematopoietic components (e.g., adipocytes, hemosiderin-laden macrophages, osteoblasts, capillary structures, etc.) were also observed. There were generally small but moderate amounts of blood in the background. Hematopoietic cells at all stages of maturity were observed, and maturation of the myeloid and erythroid lineages appeared orderly and complete. There were no statistically significant differences in percentages of cell types and calculated end points between sexes, and data for both sexes were combined. The summarized bone marrow differential cell count data are presented in Table 1.
Summarized Femoral Bone Marrow Cell Differential Counts for Healthy Control Male and Female Cynomolgus Monkeys Originating from Mainland Asia (China or Vietnam).
a
b
c
Morphologic descriptions and progression through stages of maturation for granulocytic, erythroid, and megakaryocytic precursors were used for classification of cell types on this study and have been previously described (Provencher Bolliger and Fontaine 1998; Harvey 2012a; Olver 2010; Radin and Wellman 2010) but are not otherwise described here.
Granulocytic series
Median total granulocyte composition of the total cell count was 41.9% (central 95%, 25.6–60.6%) for both sexes combined, which included myeloblasts (median, 0.7%), promyelocytes (1.0%), neutrophilic myelocytes (4.3%), neutrophilic metamyelocytes (3.9%), band neutrophils (11.4%), segmented neutrophils (17.7%), eosinophilic cells of all stages (1.6%), and <1% basophilic cells of all stages and myeloid mitotic figures. Notable morphologic features of granulocytes included large to giant neutrophilic metamyelocytes and band neutrophils and band neutrophils with ring-shaped nuclei.
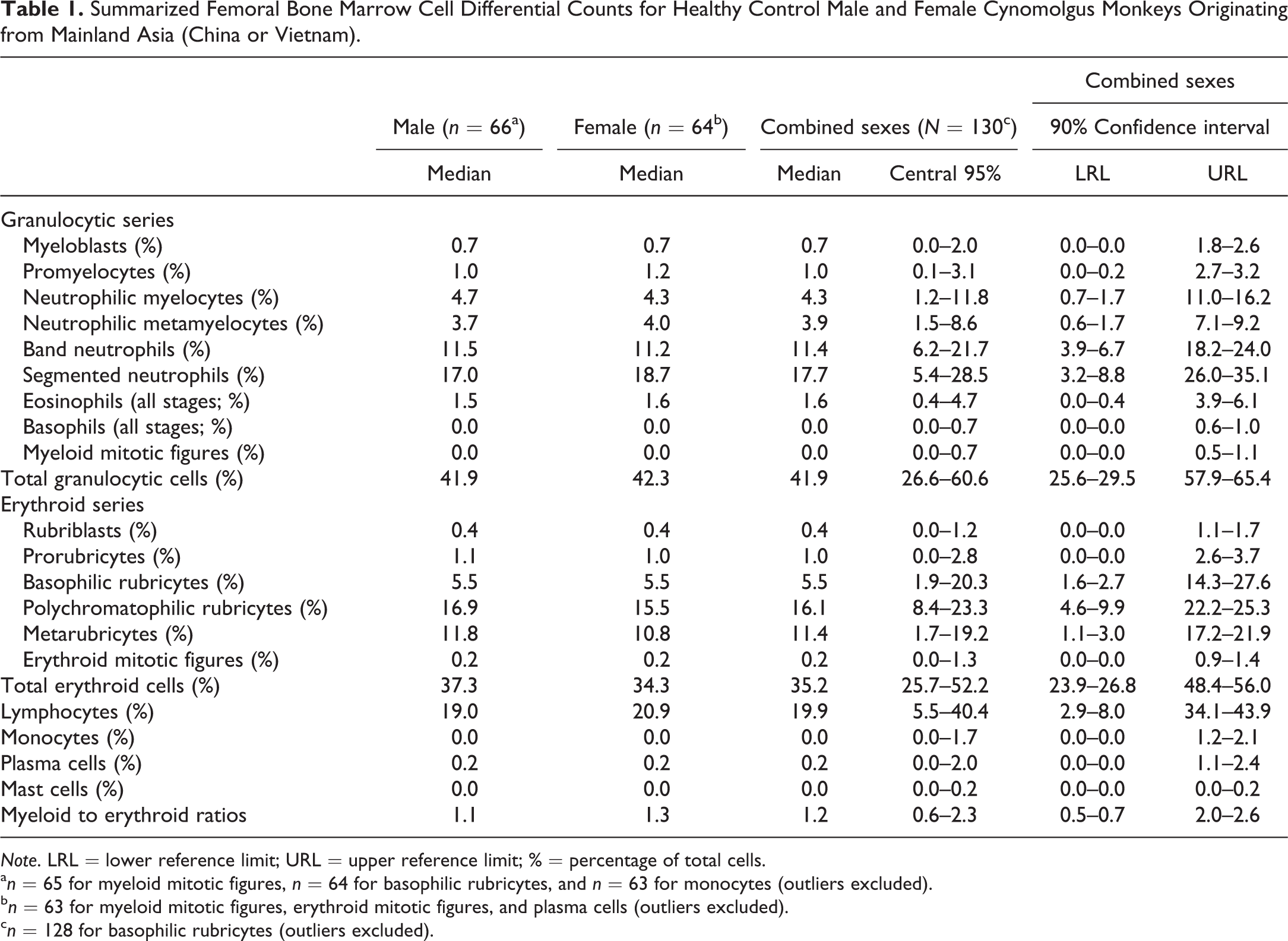
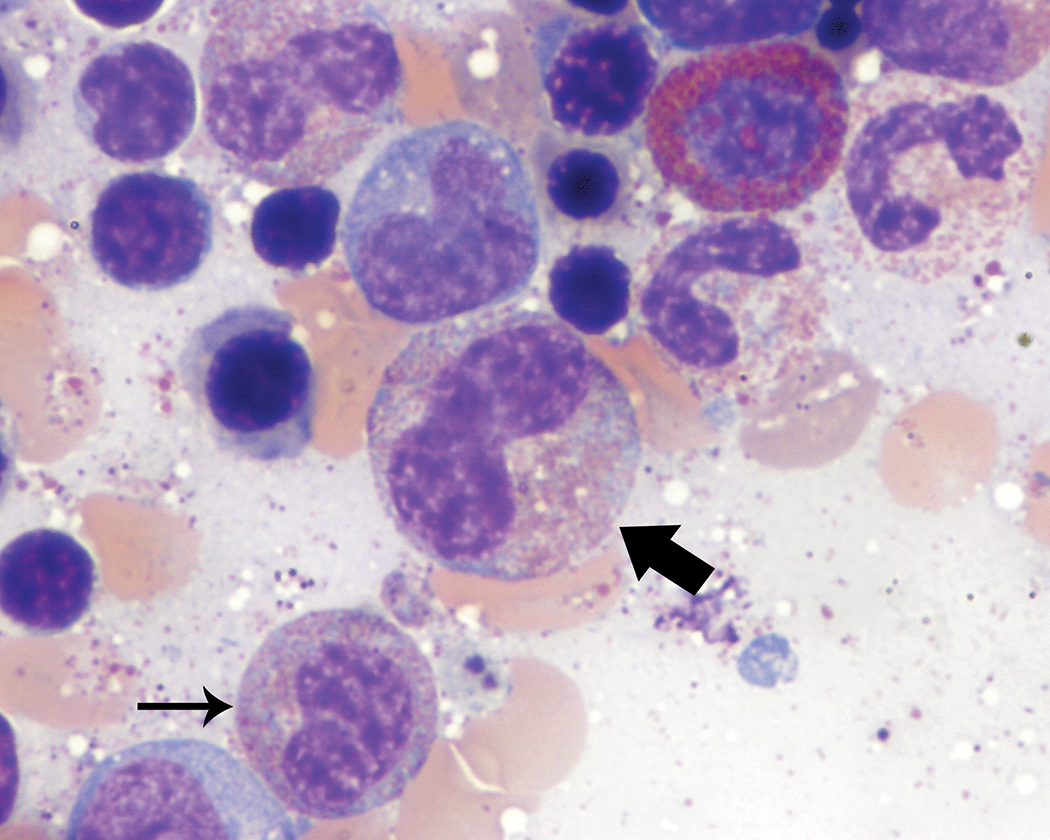
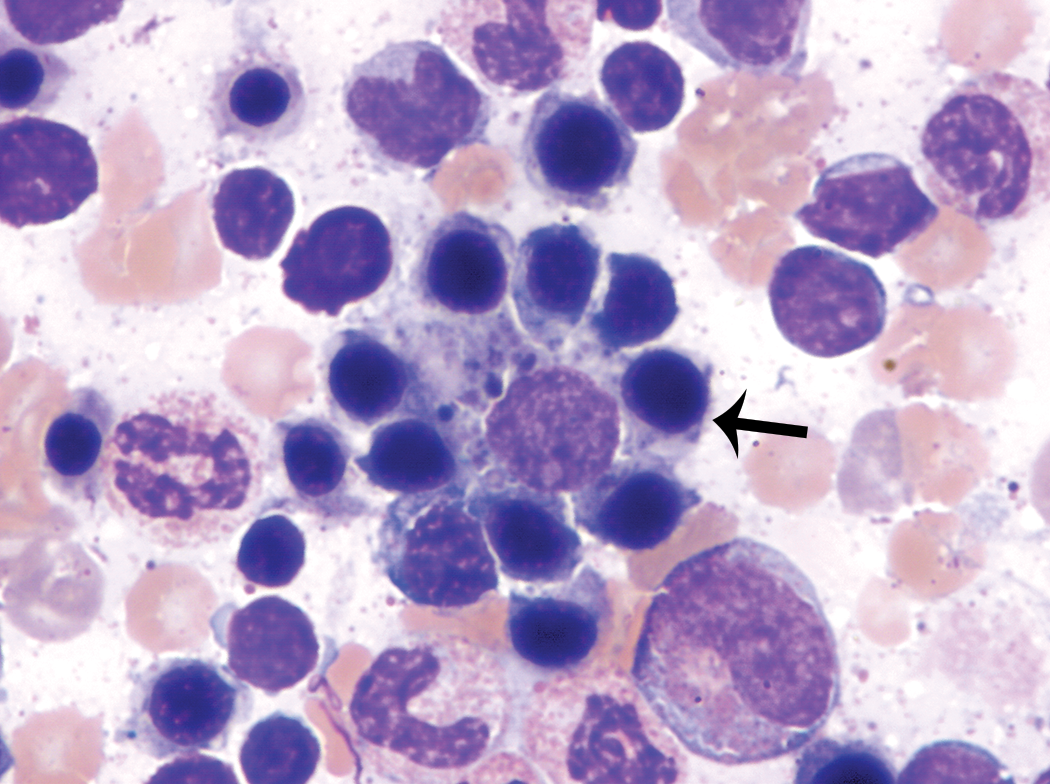
Large to giant neutrophilic metamyelocytes (Figure 1) and band neutrophils (Figure 2) were observed in low numbers in most animals, although rare individuals had slightly greater numbers of these forms. With the exception of their size, which was up to approximately twice the diameter of typical neutrophilic metamyelocytes and band neutrophils, the morphology of these cells was otherwise unremarkable.
Giant form of a neutrophilic metamyelocyte (thick arrow). Giant forms of neutrophilic metamyelocytes were up to twice the diameter of typical metamyelocytes but otherwise had unremarkable morphology. A more immature granulocyte (thin arrow) is present near the giant form for size comparison. Wright–Giemsa stain.
Giant form of a band neutrophil (thick arrow). Giant forms of band neutrophils were up to twice the diameter of typical band neutrophils but otherwise had unremarkable morphology. A typical band neutrophil (thin arrow) is present adjacent to the giant form for size comparison. Wright–Giemsa stain.
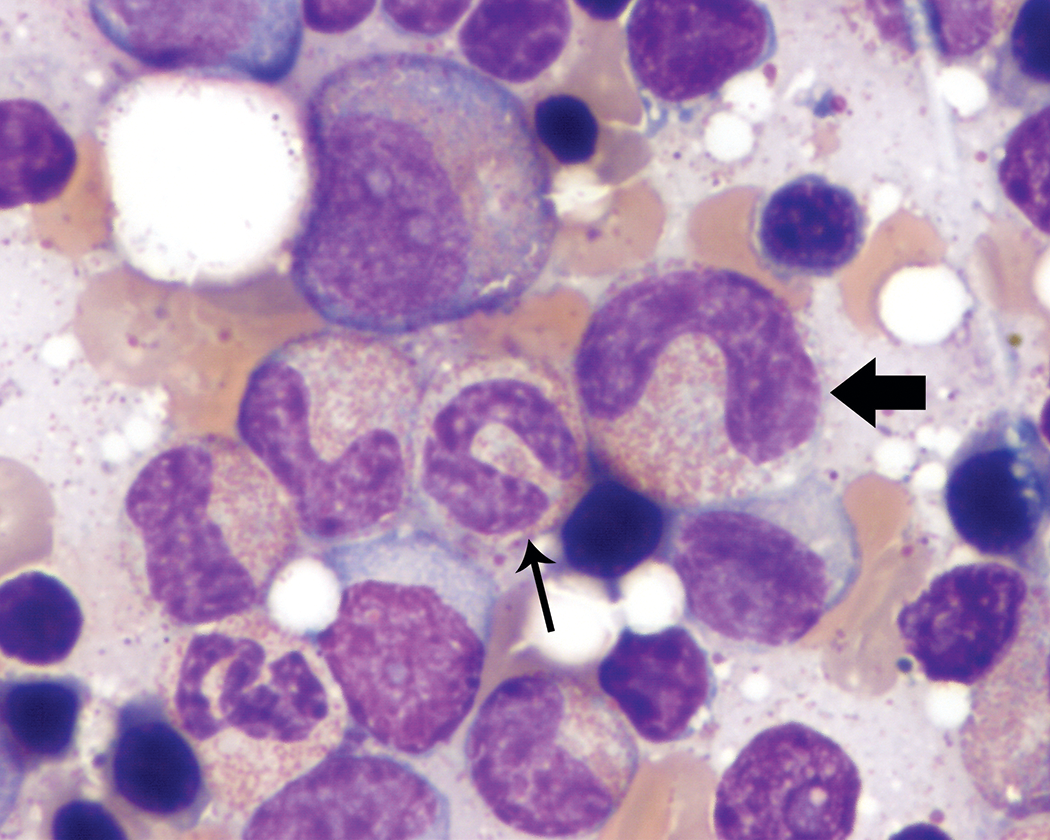
Band neutrophils typically had curved nuclei with characteristic “U,” “C,” or “S” shapes that lacked focal narrowing or constriction. Ring- or “O”-shaped nuclei (Figure 3) also were observed in low to occasionally moderate proportions of the band neutrophil population in a few animals. However, most animals evaluated did not have these band neutrophil forms observed during bone marrow smear review.
Band neutrophil with a ring-shaped nucleus (thick arrow). Wright–Giemsa stain.
Erythroid series
Median total erythroid composition of the total cell count was 35.2% (central 95%, 25.7–52.2%) for both sexes combined, which included rubriblasts (median, 0.4%), prorubricytes (1.0%), basophilic rubricytes (5.5%), polychromatophilic rubricytes (16.1%), metarubricytes (11.4%), and <1% erythroid mitotic figures. Polychromatophils were observed in low numbers amid the blood background on all smears but were not enumerated. Notable morphologic findings within the erythroid lineage included nuclear blebbing/lobulation and binucleation of metarubricytes.
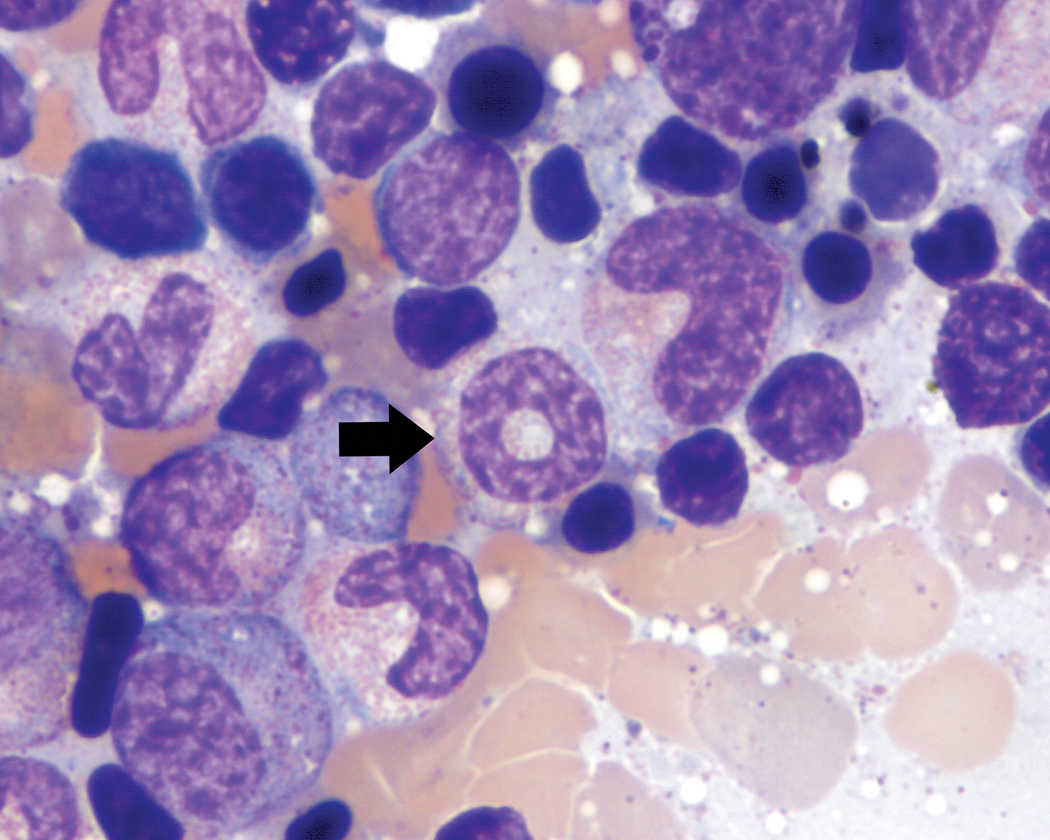
Metarubricyte nuclei were typically round and pyknotic with dense, homogeneous chromatin. However, metarubricyte nuclear blebbing/lobulation, which encompassed a wide range of shapes, including elongated, peanut-shaped, and trilobulated forms (Figure 4), was present at varying degrees in many of the animals. They tended to be observed in low proportions of metarubricytes in most animals, but several animals had frequent irregular nuclear forms. There were also occasional metarubricytes with nuclear fragmentation or binucleation (Figure 4) and rare cells with partially extruded nuclei. Only rare animals had no metarubricyte nuclear blebbing/lobulation, fragmentation, or binucleation observed.
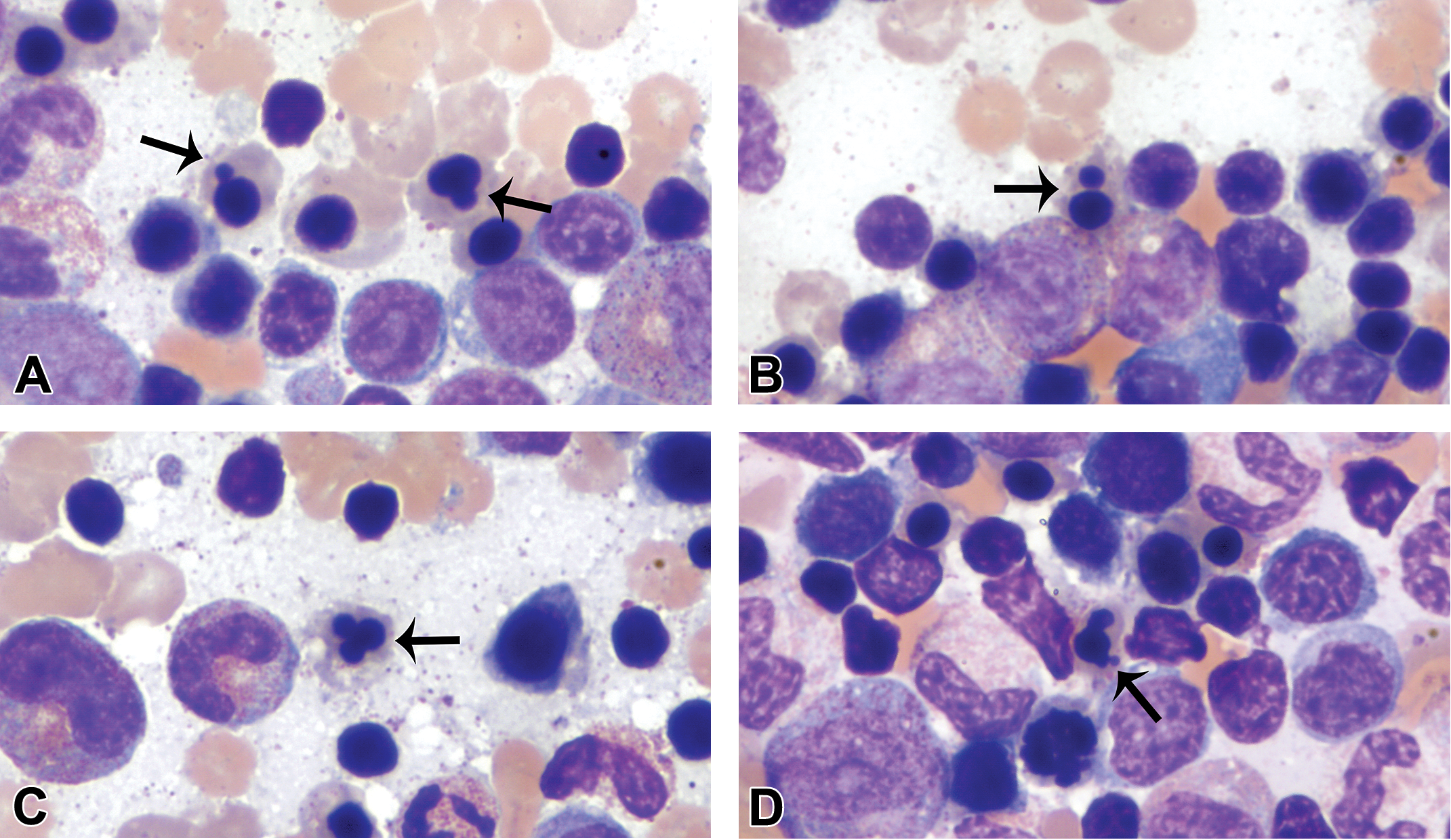
Metarubricyte nuclear blebbing/lobulation and binucleation (thin arrows). These metarubricyte nuclear changes encompassed a wide variety of shapes, including small single blebs (A), nuclear fragmentation or binucleation (B), trilobulation (C), and irregular blebbing/lobulation (D). Wright–Giemsa stain.
Megakaryocytic series
Megakaryocyte numbers were evaluated semiquantitatively (decreased, adequate, or increased) and were generally present in low numbers (<1/500 cells). Megakaryocyte numbers were considered adequate for all animals. Megakaryocytes were typically mature with rare megakaryoblasts and promegakaryocytes observed. In a few animals, there were rare megakaryocytes with multiple nuclei or nonfused nuclei and mature cytoplasm (Figure 5). Rare emperipolesis (Figure 6), typically involving segmented neutrophils, was also observed in a few animals.
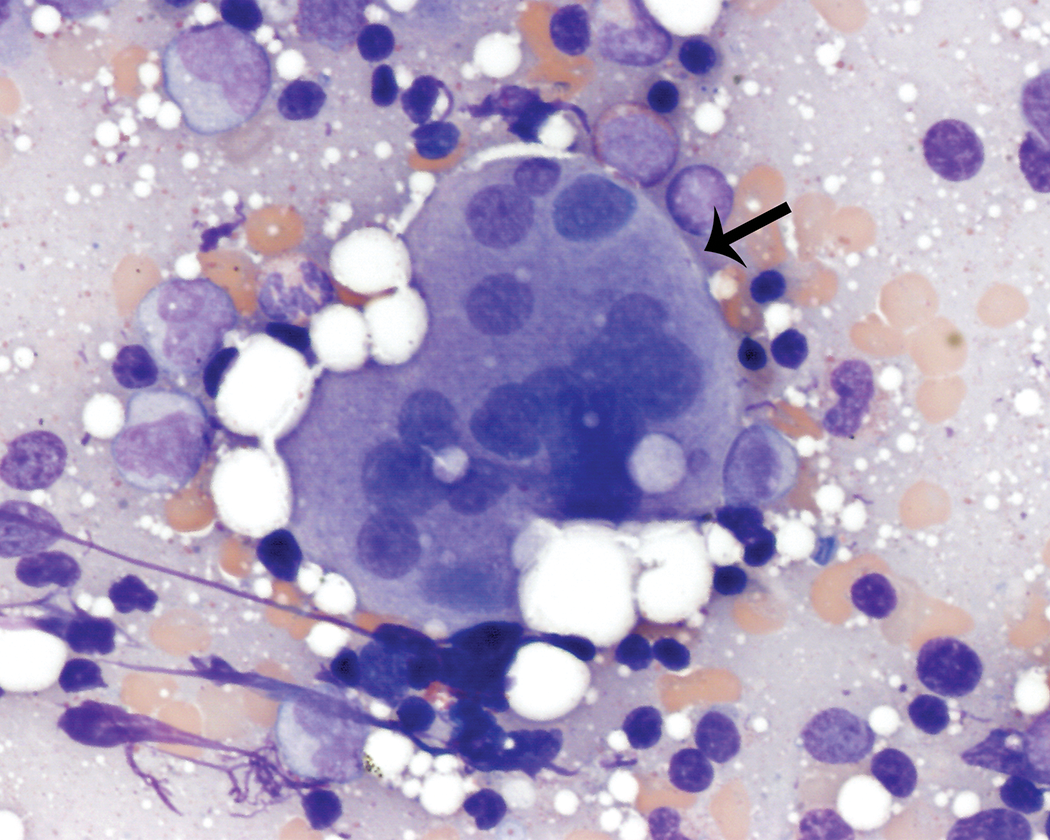
Multiple or nonfused megakaryocyte nuclei (thin arrow). Wright–Giemsa stain.
Megakaryocyte with emperipolesis (thin arrow). Wright–Giemsa stain.
Lymphocytic series
Lymphocytes represented a median of 19.9% (central 95%, 5.5–40.4%) of the total cell count for the combined sexes, although individual values tended to be highly variable. Lymphocytes were predominantly small and mature with high nuclear to cytoplasmic ratios, although a few large and less mature lymphocytes with mildly increased cytoplasmic basophilia were observed.
Monocytic series
Only rare mature monocytes were observed, with a median of 0.0% (central 95%, 0.0–1.7%) of the total cell count for both sexes combined. More immature monocytic cells could not be reliably distinguished from myeloblasts through myelocytes and were included in the granulocytic series counts.
Plasma cells
Median plasma cell composition of the total cell count was 0.2% (central 95%, 0.0–2.0%) for both sexes combined. Plasma cells had a characteristic morphologic appearance. Rare Mott cells with variable numbers of rounded, homogenous blue Russell bodies were observed. Plasma cells were distributed unevenly throughout the smears.
M:E ratios
The median M:E ratio for both sexes combined was 1.2:1. The central 95% interval was 0.6:1 to 2.3:1.
Nonhematopoietic elements
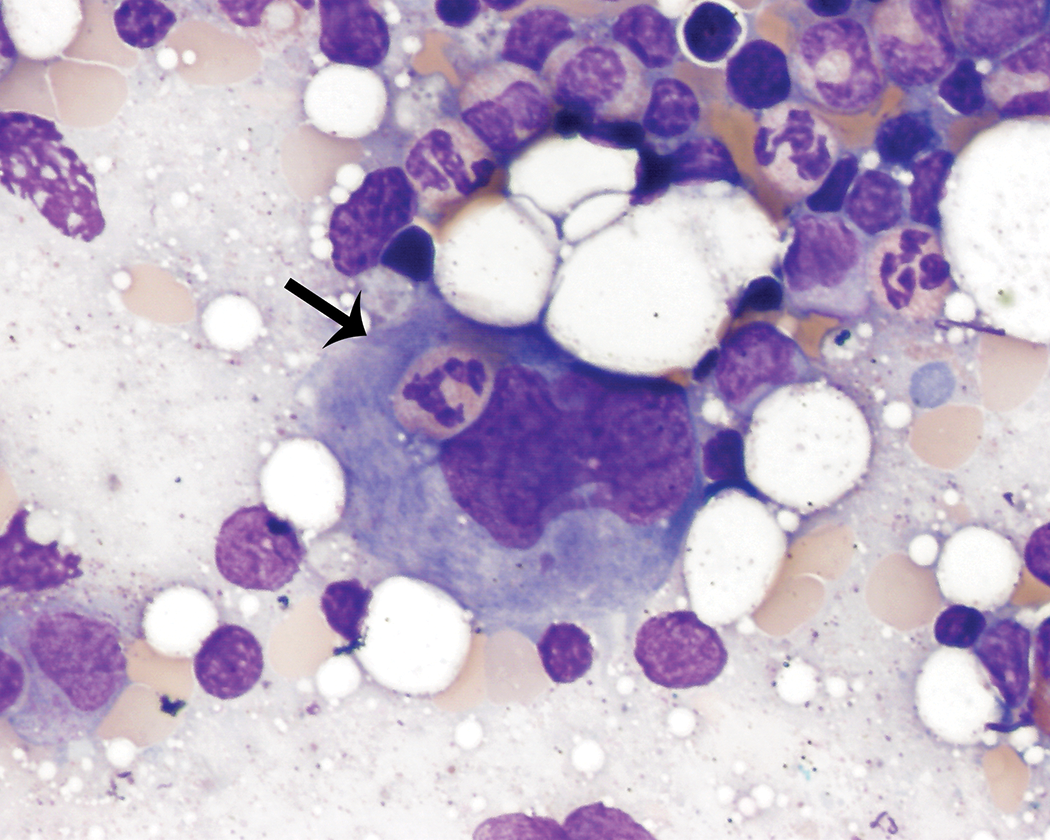
Other cellular elements were not included in the bone marrow cell differential counts and tended to be unevenly distributed across bone marrow smears from the same animal. These findings were typically infrequent, but the incidence was variable among animals. These elements included rare to occasionally moderate numbers of adipocytes, few hemosiderin-laden macrophages, including rare nurse cells (Figure 7), few osteoblasts present singly and in small aggregates (Figure 8), and rare osteoclasts, fibroblasts, endothelial cells, and capillary structures. All of these nonhematopoietic cell elements were cytologically unremarkable.
Macrophage surrounded by erythroid precursors in various stages of maturation (nurse cell; thin arrow). The macrophage contains a small amount of blue-gray pigment consistent with hemosiderin. Wright–Giemsa stain.
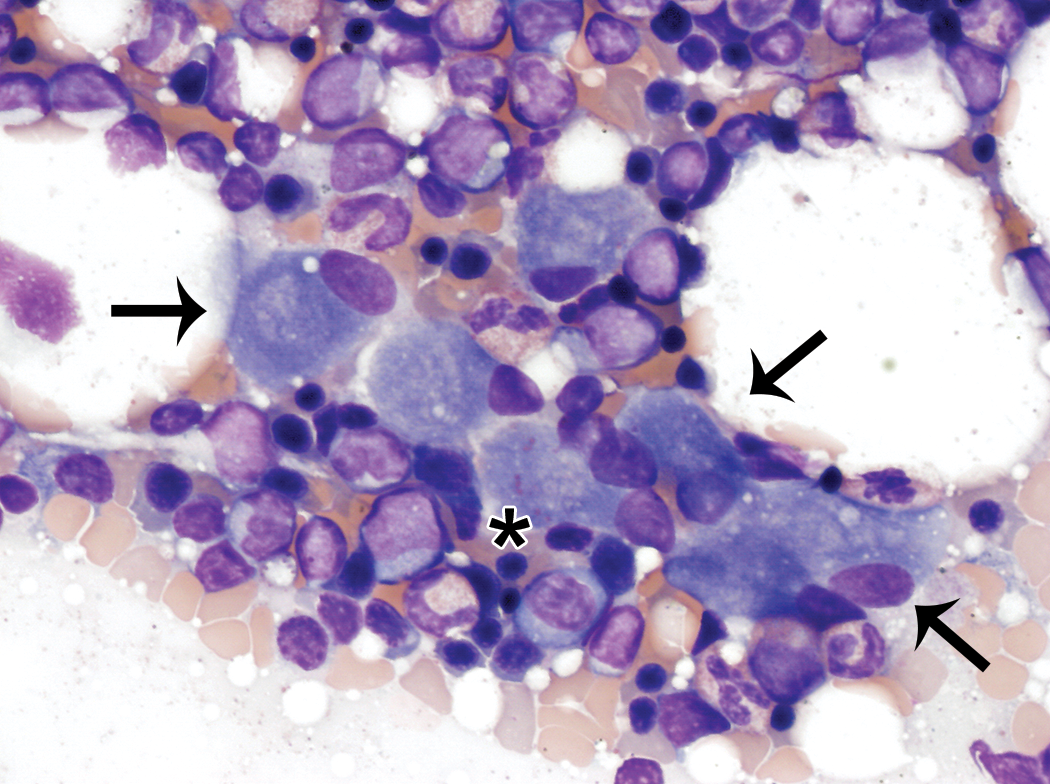
Small aggregate of osteoblasts (thin arrows). Osteoblasts were also observed singly and rarely contained small amounts of pink material (star) consistent with osteoid. They were typically observed in low numbers and were unevenly distributed throughout the smears. Wright–Giemsa stain.
Discussion
Complete cytological bone marrow cell differential counts and morphologic assessment of cell types were performed on 130 healthy control cynomolgus monkeys originating from Mainland Asia (China or Vietnam). Visual inspection of male and female data did not reveal any sex differences considered biologically or interpretively meaningful, and there were no statistically significant differences between the two sexes for any cell type or M:E ratios. Therefore, data for both sexes were combined for analysis.
Published cytological findings in healthy cynomolgus monkey bone marrow are limited. Provencher Bollinger and Fontaine evaluated the cellular composition of cynomolgus monkey bone marrow using smears prepared by extracting a small plug of marrow from sectioned femoral bone at necropsy. Monkeys included were young adults that were sexually mature and were purchased from a single vendor (Provencher Bolliger and Fontaine 1998). There were no statistically significant differences between sexes (11 male, 12 female), and both sexes combined had mean M:E ratios of 1.02 (range, 0.75–1.29), 47.44% (38.11–56.37%) total granulocytic cells, 46.17% (39.55–55.11%) total erythroid cells, and 5.27% (2.56–7.97%) total lymphoid cells, with ranges also reported for individual cell types (Provencher Bolliger and Fontaine 1998). Central 95% intervals from the present study were compared to ranges reported by Provincher Bolliger and Fontaine. The M:E ratios were more variable in this study (0.7–2.3), and many central 95% intervals were wider than previously published ranges, which may have been due the greater number and diversity of animals included in this study. Central 95% intervals for total granulocytic cells were generally comparable but wider (26.6–60.6%) than the published range, where the total erythroid cells were lower in this study (25.7–52.2%). The most notable difference was in the percentages of lymphocytes, which was greater in this study (5.5–40.4%) relative to the published range. Other differences included lower proportions of neutrophilic metamyelocytes (1.5–8.6% vs. 7.27–12.68%) and segmented neutrophils (5.4–28.5% vs. 17.10–30.56%), with increased proportions of band neutrophils (6.2–21.7% vs. 2.51–6.88%) observed in this study. Also, there tended to be decreased proportions of prorubricytes (0.0–2.8% vs. 1.60–5.07%) and metarubricytes (1.7–19.2% vs. 26.46–36.39%) in this study; rubricytes in the previous study were not categorized as basophilic or polychromatophilic, so a direct comparison could not be made.
Cytological classification of hematopoietic precursor cells based on morphology is somewhat subjective due to the continuum of microscopically visible features between consecutive stages of maturation and the lack of universal thresholds between these stages of maturation. Additionally, differential count data from three individual cytologists were included in this report. Variation in individual cytologist cell classification thresholds may have contributed to differences in cell-type percentages between studies. Although variable degrees of hemodilution may have affected the percentages of several cell types, most notably lymphocytes and segmented neutrophils (Provencher Bolliger and Fontaine 1998), hemodilution was generally minimal to mild, and large differences attributable to hemodilution were not expected.
Current methods used for cytologic evaluation of bone marrow by light microscopy involve a certain degree of inherent procedure-related imprecision as related to variations in smear quality, sampling/counting location, and nonhomogeneous distribution of cell types within smears. Differences in bone marrow collection procedures may also introduce smear variability, including the variability in cell proportions and distributions, and M:E ratios. Other sources of variation may include differences in monkey origin, age, husbandry, and vendor (Provencher Bolliger and Fontaine 1998; Provencher Bolliger et al. 2010). Data reported by Provencher Bolliger and Fontaine were obtained from extracted plugs of marrow from young adult, sexually mature monkeys from a single vendor, while data reported here were obtained from marrow collected using the paintbrush technique from 2- to 8.25-year-old that were purchased from multiple vendors. It was suspected that combinations of these factors and cytologist variation accounted for the majority of the differences in percentages of bone marrow cell types between this and the previously reported study. Within the context of nonclinical toxicology studies, the use of one cytologist for bone marrow evaluation and interpretation on a single study is recommended to help limit the impact of these factors in introducing variability into bone marrow data.
There were several notable morphologic observations among granulocytic, erythroid, and megakaryocytic lineages that deviated from the standard cytological descriptions of these cells types, although these findings tended to be present infrequently and did not influence interpretation or toxicologic relevance of bone marrow findings. These findings included large to giant neutrophilic metamyelocytes and band neutrophils, occasional ring-shaped band neutrophil nuclei, and nuclear blebbing/lobulation or binucleation of metarubricytes, multiple or nonfused megakaryocyte nuclei, and emperipolesis.
Large to giant forms of neutrophilic metamyelocytes and band neutrophils were observed in this study and have been previously reported as a small proportion of the metamyelocyte and band neutrophil populations in healthy cynomolgus and rhesus monkeys (Provencher Bolliger and Fontaine 1998; Switzer 1967). Rare giant neutrophilic myelocytes have also been reported in healthy cynomolgus monkeys (Provencher Bolliger and Fontaine 1998) but were not observed during this study.
Ring-shaped nuclei in promelocyte through band neutrophil stages may be seen in bone marrow from healthy rats and mice (Provencher Bolliger 2004; Provencher Bolliger et al. 2010). However, to the authors’ knowledge, ring-shaped nuclei in band neutrophils have not been previously reported in healthy monkeys. This finding was limited to a few animals that typically had low proportions of band neutrophils with ring-shaped nuclei, and its significance is uncertain.
Nuclear blebbing/lobulation and binucleation of metarubricytes were observed in these animals, although the incidence was variable among individuals, ranging from none to frequent. Similar metarubricyte nuclear blebbing/lobulation has been reported in the bone marrow of healthy rats (Provencher Bolliger 2004) and humans (Bain 1996), which suggested that these findings represented biological variation in these monkeys. However, nuclear blebbing/lobulation of late erythroid precursors has been observed in cynomolgus monkeys in conjunction with a strong regenerative erythroid response but also has been interpreted as dyserythropoiesis when a regenerative response to decreased red cell mass is lacking (Walters, Smith, and Walker 2013), emphasizing the importance of recognizing this feature as a potential background finding in control cynomolgus monkeys. Hematopoietic stress, resulting from multiple blood collection procedures, is common in cynomolgus monkeys during nonclinical toxicology studies. Although procedure-related decreases in red cell mass could stimulate a regenerative erythroid response, these monkeys generally lacked alterations in red cell mass or reticulocyte counts to suggest that the presence of metarubricyte nuclear blebbing/lobulation was secondary to a strong regenerative erythroid response or altered erythroid production.
Multiple or nonfused megakaryocyte nuclei were also rarely observed in a few individuals. Multiple separate nuclei in megakaryocytes have been described as a dysplastic abnormality in dogs and cats (Harvey 2012b). However, similar findings have been occasionally observed in the bone marrow of healthy rats and described as detached or nonfused nuclei (Provencher Bolliger 2004) and in apparently healthy humans and described as megakaryocyte multinucleation (Bain 1996). Multiple or nonfused megakaryocyte nuclei were rare and were not associated with other evidence of megakaryocyte dysplasia, suggesting that this may sporadically occur in healthy cynomolgus monkeys. However, separation of nuclei during bone marrow smear preparation could not be excluded and may be a consideration when evaluating bone marrow smears prepared with the paintbrush technique.
In this study, rare emperipolesis of hematopoietic cells within megakaryocytes was observed in a few animals. Emperipolesis of hematopoietic cells within megakaryocytes has been reported in various species, including dogs and cats (Harvey 2012b), rats (Lee 1989), and humans (Bain, Clark, and Wilkins 2010; Breton-Gorius 1981). Although emperipolesis has been associated with conditions that stimulate thrombopoiesis or alter leukocyte adhesion molecule expression (Harvey 2012b), it has also been observed in healthy rats (Lee 1989) and humans (Bain, Clark, and Wilkins 2010; Breton-Gorius 1981). To the authors’ knowledge, it has not previously been reported in healthy cynomolgus monkeys. The significance of this finding is unclear.
Conclusions
Bone marrow smear preparation using the paintbrush technique immediately following euthanasia yielded cellular specimens of good quality for cytological evaluation. Bone marrow cell differential counts and cytological findings from 130 healthy control cynomolgus monkeys were reported. The cellular composition of the bone marrow in this study was generally comparable to previously reported ranges, although the lymphocyte counts observed in this study were higher than those previously reported. Notable morphological features observed in control cynomolgus monkey bone marrow included large to giant forms of neutrophilic metamyelocytes and band neutrophils, ring-shaped band neutrophil nuclei, nuclear blebbing/lobulation and binucleation of metarubricytes, multiple or nonfused megakaryocyte nuclei, and emperipolesis; these findings were generally rare or observed in low numbers, with the exception of nuclear blebbing/lobulation of metarubricytes, which tended to have variable incidences among individual animals. The findings reported here demonstrate the variability among bone marrow cell differential counts and occasional hematopoietic cell morphologic findings that are expected in healthy control cynomolgus monkeys and should be considered during smear evaluation and data interpretation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
