Abstract
The neurotransmitter glutamate causes excitotoxicity in the human retina. In neonatal rats, the degree of glutamate-induced retinal damage depends on age at administration. To elucidate the sensitivity to glutamate on various developing stage of retina, we investigated glutamate-induced retinal damage and glutamate target cells on each postnatal day (PND). Newborn rats received a single subcutaneous administration of
Introduction
Glutamate, an excitatory neurotransmitter in the retina, is known to cause excitotoxicity, a phenomenon in which neuronal cell death occurs following excessive excitatory neurotransmitters. Excitotoxicity is linked to stroke, hypoglycemia, trauma, epilepsy, and chronic neurodegenerative diseases such as Huntington’s disease, acquired immunodeficiency syndrome dementia complex, amyotrophic lateral sclerosis, and Alzheimer’s disease (Lipton and Rosenberg 1994; Rothstein 1996). In the retina, excitotoxicity is believed to play an important role in retinal ischemia/reperfusion injury and more recently in neuronal loss in glaucoma (Donello et al. 2001; Martin et al. 2002; Sullivan et al. 2006).
The neurotoxic effects of glutamate are mainly mediated by stimulation of N-methyl-
The thinning of inner retina composed of internal limiting membrane to the inner nuclear layer has been recognized in glutamate-induced excitotoxic animal models (Kanno et al. 1991), indicating that glutamate injures not only RGC but also other inner retinal cells. However, the excitotoxic effects of glutamate against inner retinal cells other than RGC have not been clarified.
Glutamate-induced retinal damage in neonatal rats is a well-known animal model of glutamate-induced excitotoxicity in the retina. The retina is not fully developed in newborn rats, taking approximately 3 weeks to mature (Zucker et al. 1994), and the degree of retinal damage depends on age at glutamate administration (Kanno et al. 1991; Guerin et al. 2011). Here, we investigated retinal excitotoxicity in neonatal rats by administering
Material and Method
Animals
Pregnant female Sprague-Dawley (SD) rats purchased from Charles River Laboratories (Shiga, Japan) were maintained in specific pathogen-free conditions, with ad libitum access to a commercial diet (CRF-1 30 kGy; Oriental Yeast, Tokyo, Japan) and water. Pregnant animals were housed individually in plastic cages with paper-chip bedding (Shepherd Specialty Papers, Richland, MI) in an air-conditioned room at 23°C ± 3°C and 55% ± 15% relative humidity with a 12-hr light/dark cycle. Animals were maintained and treated in accordance with the Guide for the Care and Use of Laboratory Animals at our institution, which is certified by the Association for Assessment and Accreditation of Laboratory Animal Care. All experimental procedures were approved by the Institutional Animal Care and Use Committee of Astellas Pharma Inc.
Experimental Procedures
In experiment 1, neonatal SD rats were divided into 14 treatment groups of 2 animals each. The animals of each group were weighed and injected once subcutaneously with 10 µl of 2.4 mol/L monosodium glutamate per gram of body weight (Sigma-Aldrich, St. Louis, MO). The day of injection was varied from PNDs 1 to 14 (e.g., rats from group 1 were injected only on PND 1 while rats from group 14 were injected only on PND 14). Clinical signs were monitored daily in all treatment group animals starting from their respective injection days through PND 21. The rats of all 14 treatment groups were terminated on PND 21 by exsanguination under isofluorane anesthesia, and the eyes were removed for further analysis. Untreated normal animals were terminated on PNDs 1 to 14 or 21, and the eyes were removed. In experiment 2, neonatal SD rats were divided into 4 treatment groups of 6 animals each. The animals of each group were weighed and injected once subcutaneously with 10 µl of 2.4 mol/L monosodium glutamate per gram of body weight. The day of injection was varied on PNDs 4, 6, 8, and 10. The eyes were removed 6 hr after dosing for apoptotic cell analysis.
Tissue Fixation and Processing
In experiment 1, the right eyes were fixed in 4% phosphate-buffered glutaraldehyde and postfixed in 5% phosphate-buffered formalin, embedded in paraffin, sectioned at 3 µm, and stained with hematoxylin and eosin (HE). The posterior eyecups of the left eyes were fixed in 10% phosphate-buffered formalin for 1 hr, cryoprotected in 30% sucrose, embedded in optimal cutting temperature (OCT) compound (Sakura-Finetek, Tokyo, Japan), and stored at −80°C until used for immunohistochemistry. In experiment 2, eyes of 2 rats on each PND were fixed in 4% phosphate-buffered glutaraldehyde and postfixed in 5% phosphate-buffered formalin and stained with HE. Eyes of 2 rats on each PND were fixed in 10% phosphate-buffered formalin and embedded in paraffin for terminal deoxynucleotidyl transferase–mediated dUTP digoxigenin nick end labeling (TUNEL) method. The posterior eyecups of 2 rats were fixed in 10% phosphate-buffered formalin for 1 hr, cryoprotected in 30% sucrose, and embedded in OCT compound.
Immunohistochemistry
Cryopreserved tissues were sectioned for immunohistochemical labeling using the indirect fluorescence method. Antigen retrieval for Chx10 and Pax6 immunostaining was performed by incubation in Immunosaver (Nisshin EM, Tokyo, Japan) at 100°C for 15 min and citrate-buffered solution (Dako, Carpinteria, CA) at 105°C for 5 min, respectively. Retinal sections were blocked for 20 min in 1% bovine serum albumin (BSA) in 50 mM Tris-buffered saline (TBS) with pH 7.6. The primary antibodies listed in Table 1 were diluted in 1% BSA in TBS, and tissue sections were incubated with antibodies overnight at 4°C. After washing in TBS, secondary antibodies were applied at room temperature for 1 hr. These included rabbit anti-mouse immunoglobulin G (IgG) or goat anti-rabbit IgG conjugated with Alexa TM 488 (green fluorescence; Molecular Probes, Eugene, OR; diluted 1:100). For Chx10 immunostaining, rabbit anti-sheep IgG (Dako) diluted 1:200 was used as a secondary antibody and Alexa TM 488-conjugated goat anti-rabbit IgG (Molecular Probes) was used as the third antibody. All fluorescent specimens were viewed using a Leica TCS SP confocal microscope (Leica Microsystems GmbH, Wetzlar, Germany) equipped with a krypton–argon laser. Laser lines and emission filters were optimized with Leica TCS Power Scan software. Image brightness and contrast were adjusted using Adobe Photoshop Elements (version 11.0; Adobe Systems, San Jose, CA).
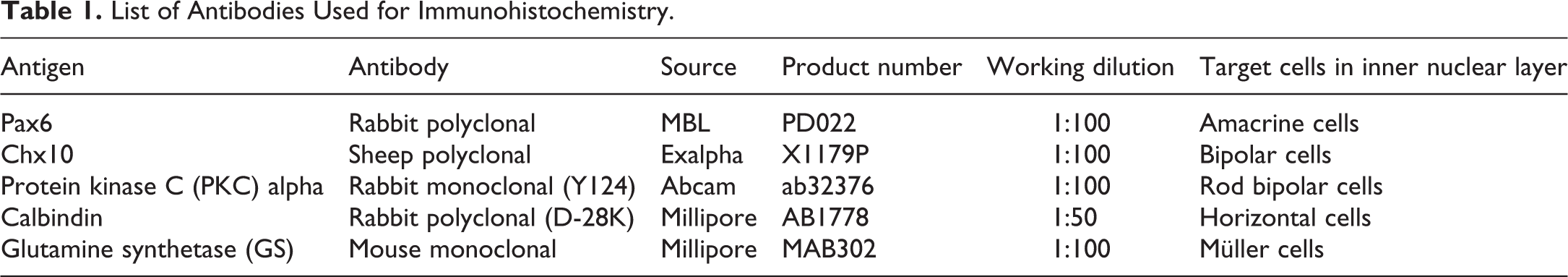
List of Antibodies Used for Immunohistochemistry.
TUNEL Method
Apoptotic cells were labeled with TUNEL method using an in situ apoptosis detection kit (Takarabio, Shiga, Japan) in accordance with the manufacturer’s instructions.
Morphometric Analysis of Inner Retinal Ratio
All light microscopic images were taken using ImageScope (Leica Biosystems, Wetzlar, Germany) from scanned images with ScanScope XT (Leica Biosystems). Total retinal thickness (internal limiting membrane to pigment epithelium) and inner retinal thickness (internal limiting membrane to the inner nuclear layer) were individually measured from HE slides using ImageScope (Leica Biosystems). Measurements were taken at the central retina (approximately 500 µm from the optic disc) and peripheral retina (approximately 500 µm from both sides of the ciliary bodies), and the inner retinal ratio [(inner retinal thickness/total retinal thickness) ×100] was calculated.
Results
Histopathology in the Central Retina
We evaluated age-dependent sensitivity of the retina to glutamate in neonatal rats treated with
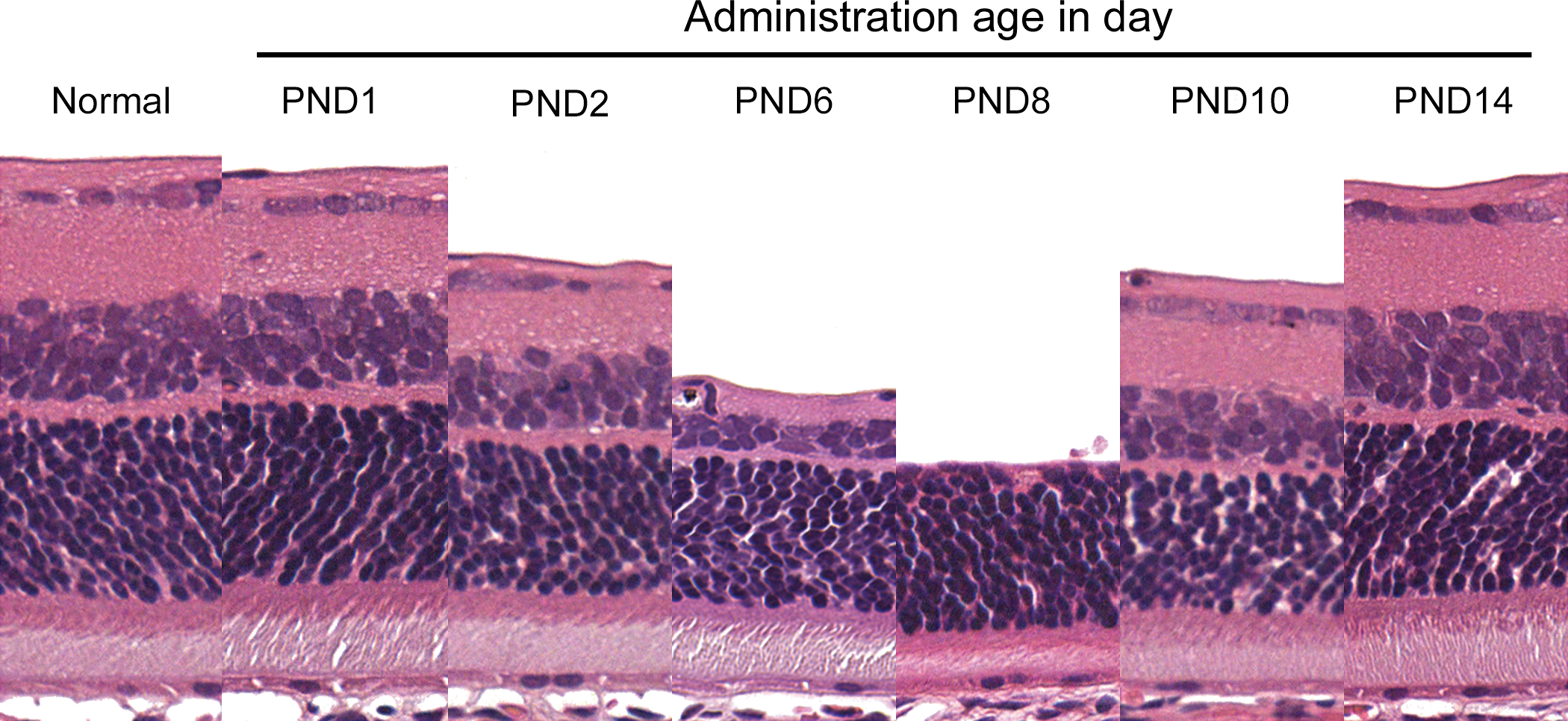
Sequential retinal changes at the central retina on postnatal day (PND) 21 after single administration of
Initial Change in the Inner Retina
To investigate the initial change of a single administration of
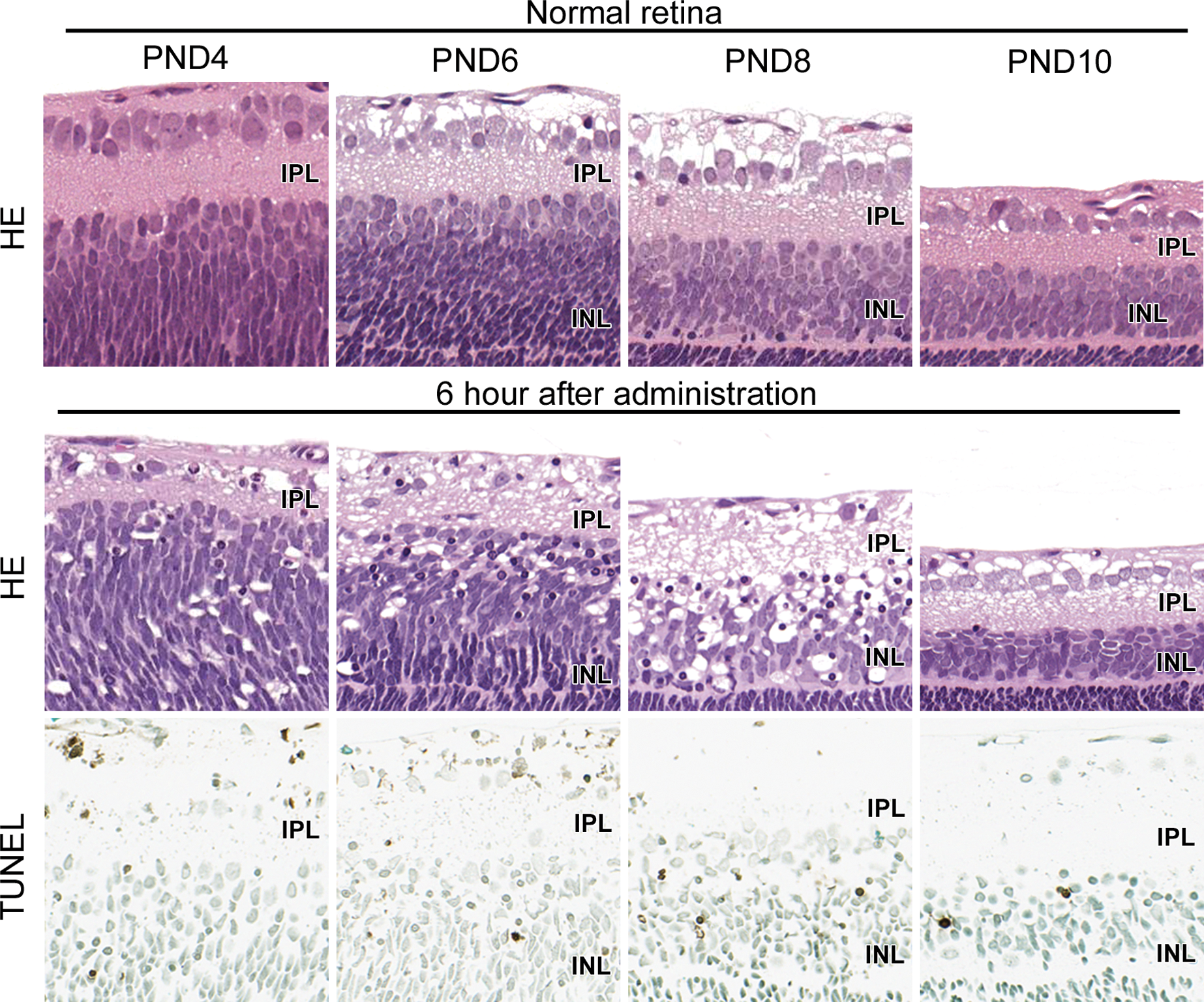
Initial change in inner retina on postnatal days (PNDs) 4, 6, 8, and 10 at 6 hr after dosing. Many pyknotic nuclei appeared selectively in inner retina, some of which were positive for terminal deoxynucleotidyl transferase–mediated dUTP digoxigenin nick end labeling method. The majority of pyknotic nuclei were located in the inner area of the inner nuclear layer. The number of pyknotic nuclei peaked in rats treated on PND 8 and was very few in rats treated on PND 10. IPL = inner plexiform layer; INL = inner nuclear layer.
Immunohistochemical Analysis of the Inner Nuclear Layer
Because inner nuclear layer consists of multiple types of cells, immunohistochemical analysis was conducted on PND 21 to identify glutamate target cells in the inner nuclear layer of rats treated on PNDs 6, 8, and 10. Pax6, Chx10, protein kinase C (PKC) alpha, calbindin, and glutamine synthetase (GS) were used as amacrine cell, pan-bipolar cell, rod bipolar cell, horizontal cell, and Müller cell markers, respectively. Pax6 and Chx10 were localized to nuclei, whereas PKC alpha, calbindin, and GS were in cytoplasm. In normal rats, Pax6-positive cells were detected in 3 to 4 layers in the inner area of the inner nuclear layer, while Chx10- and PKC alpha-positive cells were detected in 2 to 3 and 1 to 2 layers, respectively, in the outer area. Calbindin-positive cells were scattered in the outer edge of the inner nuclear layer, and GS-positive cells were recognized in 1 layer in the middle of the inner nuclear layer. In rats treated on PND 6, the number of Pax6-positive cells was decreased to 2 to 3 layers, whereas the number of positive cells for the other cell type markers was the same as in normal. In rats treated on PND 8, Pax6- and Chx10-positive cells were decreased to 1 layer, PKC alpha–positive cells were scattered, and calbindin-positive cells were not detected. GS-positive cells appeared the same in all samples. There was no change in rats treated on PND 10 for any markers (Figure 3). These results show that glutamate target cells in the inner nuclear layer were amacrine cells on PND 6 and amacrine cells, bipolar cells, and horizontal cells on PND 8. We also assessed the initial change in
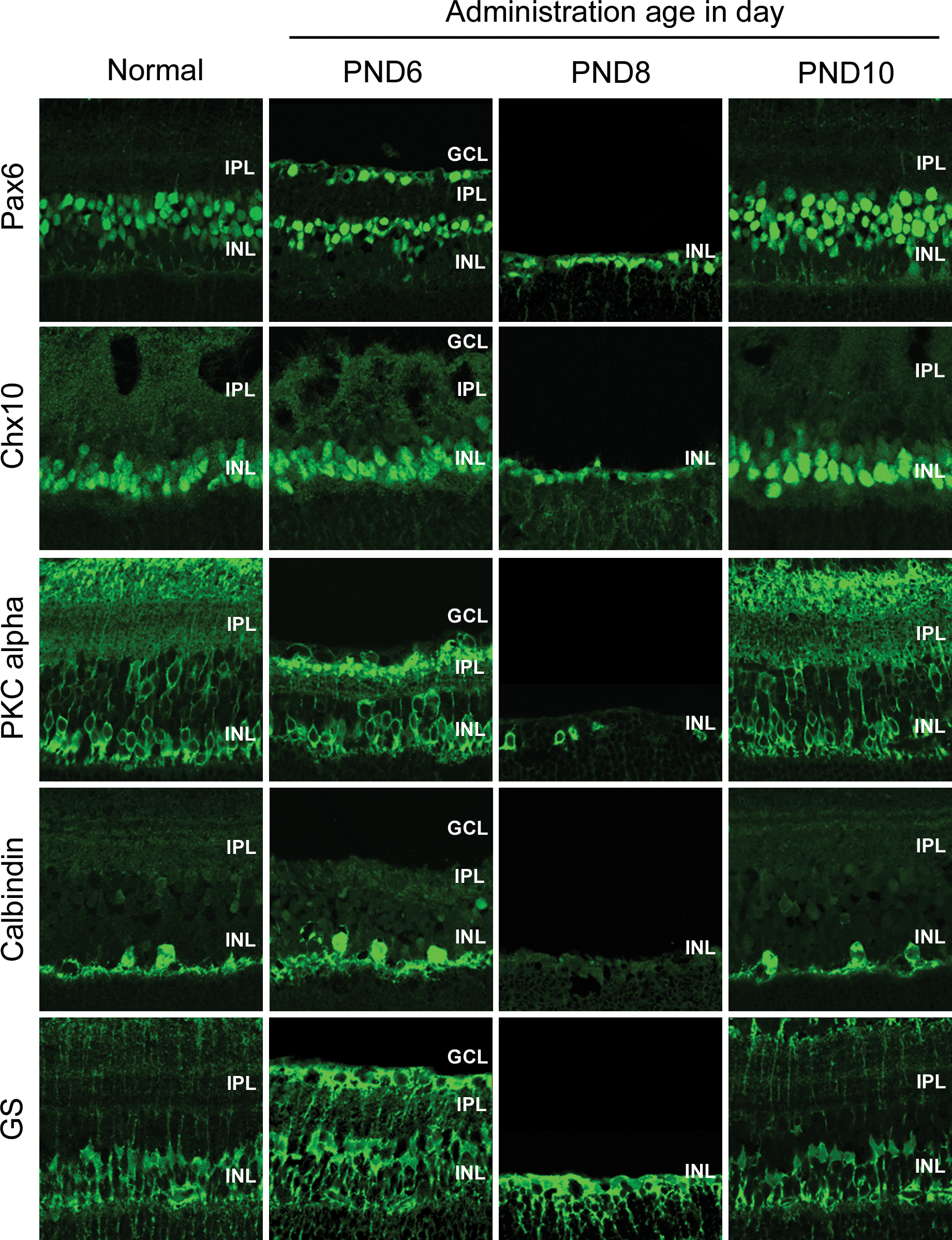
Distribution of each type of cells in the inner nuclear layer on postnatal day (PND) 21 and glutamate target cell on PNDs 6, 8, and 10. Immunohistochemical analysis was performed in the inner nuclear layer of the central retina on PND 21 after single administration of
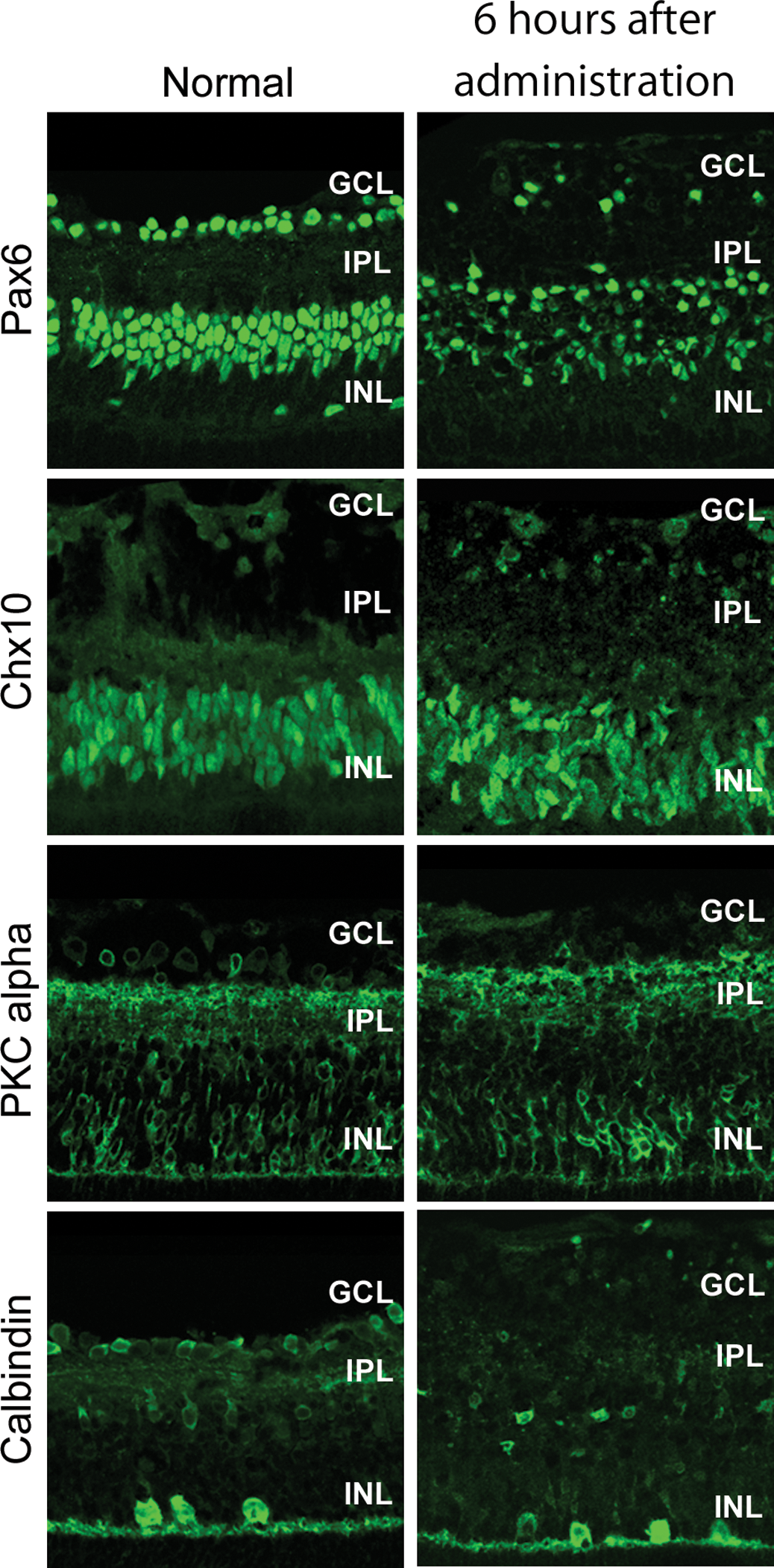
Initial change of retinal cells in the inner nuclear layer on postnatal day (PND) 8. Immunohistochemical analysis of retinal cells in the inner nuclear layer of central retina was performed in normal rats on PND 8 and in rats treated on PND 8 at 6 hr after administration of
Histopathology in the Peripheral Retina
The degree of retinal lesions induced by chemicals has been occasionally different between central and peripheral retina (Yoshizawa et al. 2011). We, therefore, compared histopathological changes on PND 21 in the peripheral retina with those in the central retina. Inner retina in the peripheral retina was also thinning in rats treated on PND 2 compared to normal retina. In the retina of rats treated on PND 8, inner retinal layers were very thin but still present, in contrast to the central retina, where the inner layer was virtually lost. The inner retinal thinning in rats treated on PND 10 was milder than those on PND 8. In rats treated on PND 14, the thickness of inner retinal layers was the same as in normal (Figures 1 and 5). On subsequent PND treatment, the pattern and degree of damage in the peripheral retina was similar to that in the central retina; however, inner retinal thinning in the peripheral retina was milder than those in the central retina on PND 8.
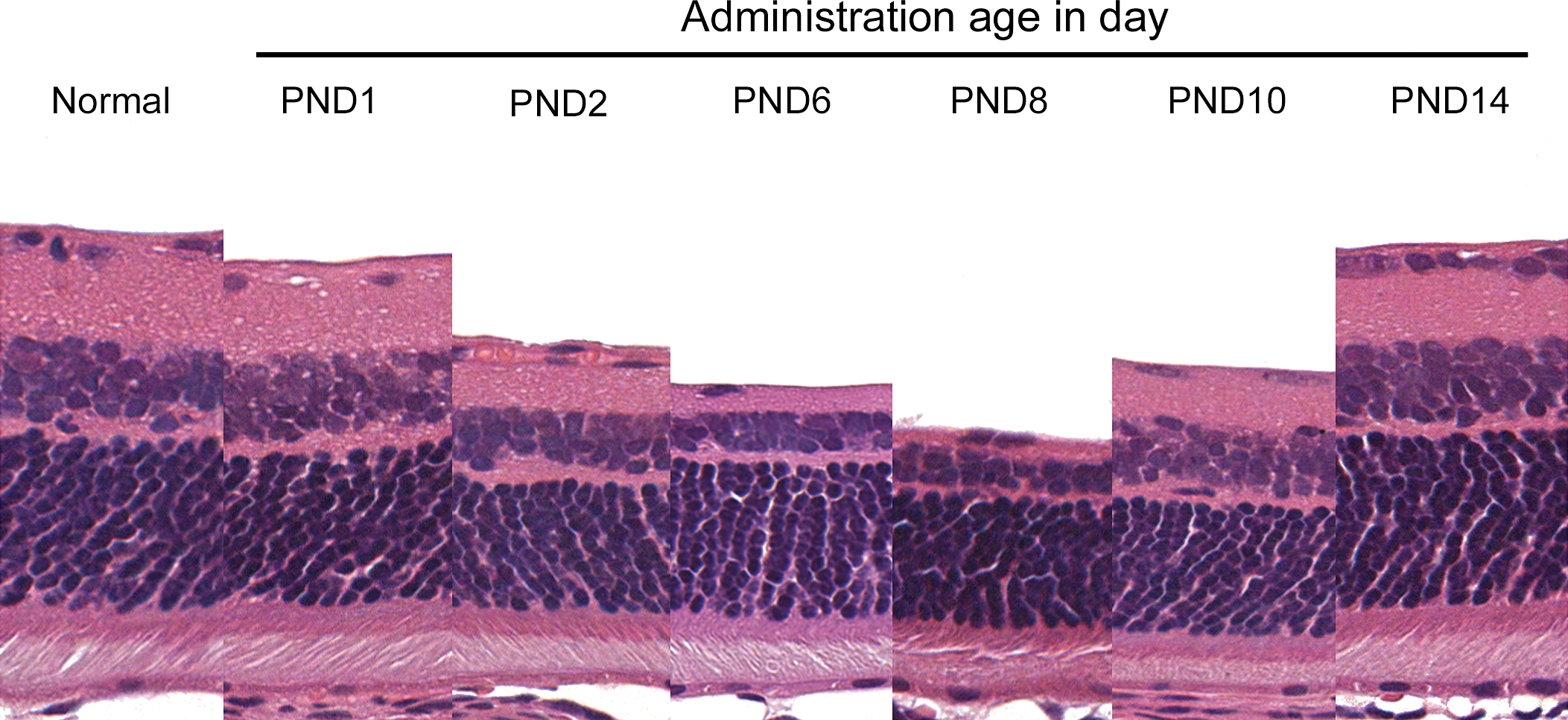
Sequential retinal changes at the peripheral retina on postnatal day (PND) 21 after single administration of
Morphometric Analysis of the Thickness of Inner Retina
To investigate sequential changes in inner retinal thickness, inner retinal ratio was calculated in the central and peripheral retina of rats treated on each PND. Inner retinal ratio on PND 21 was decreased in rats treated after PND 2 in both central and peripheral retina and was smallest in rats treated on PNDs 8 and 9, respectively. Inner retinal ratio in the central retina was smaller than that in the peripheral retina in rats treated on PND 8 but showed no marked difference between central and peripheral retina in rats treated on other PND (Figure 6). Changes in the ratio of nerve fiber layer to ganglion cell layer, inner plexiform layer, and inner nuclear layer, which comprise the inner retina, were similar to the inner retinal ratio (data not shown). Hence, the age at treatment when inner retinal thinning was most marked was younger in central retina than peripheral retina. In rats treated on PND 8, inner retinal thinning in the central retina was severer than that in the peripheral retina.
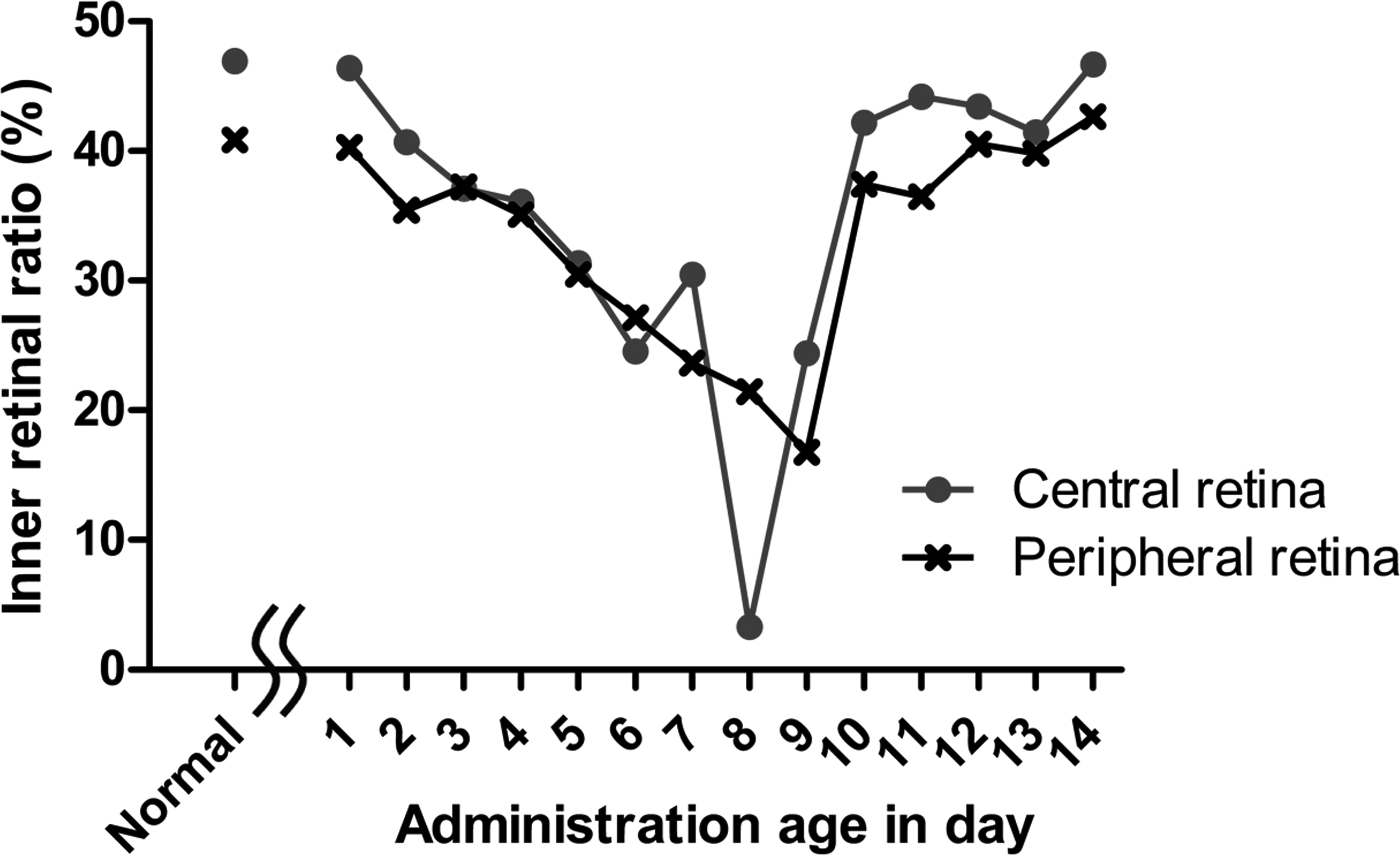
Inner retinal ratio on postnatal day (PND) 21 after single administration of
Discussion
The present study indicated the sequential changes in glutamate-induced retinal damage and glutamate target cells on each PND at administration in neonatal rats. Morphological and morphometric analyses revealed that inner retina showed thinning in rats treated after PND 2, and that within the central retina, the thinning was most marked in rats treated on PND 8. Given that retinal tissue is still undergoing development in neonatal rat, and that the expression of NMDA receptors and metabotropic glutamate receptors changes with age (Brandstatter, Koulen, and Wassle 1998; Grunder et al. 2000), it appeared likely that development of retinal cells makes inner retina increase sensitivity to glutamate until PND 8. The inner retinal thinning in the central retina of rats treated on PND 9 was far milder than that in rats treated on PND 8. Blood–retinal barrier segregates the circulating blood from retinal interstitial fluid and regulates the exchange of compounds between the circulating blood and the retina. In addition, vascular endothelial cells in inner retina and Müller cells express glutamate transporters and play a central role in retinal glutamate homeostasis and in preventing glutamate accumulation (Kanno et al. 1991; Pow and Barnett 1999; Sakurai et al. 2015). We proposed that the milder damage in rats treated on PND 9 than on PND 8 was caused by more effective glutamate homeostasis in older animals, which have developed mechanisms to better regulate glutamate.
We also investigated initial change of glutamate-induced retinal damage on each PND. Pyknotic nuclei were observed in inner retina, some of which were positive for TUNEL method. The number of cells with pyknotic nuclei increased in rats treated on PND 8 compared to PNDs 4 and 6, which correlated with the thinning of inner retina. It has previously been shown that glutamate-induced retinal damage correlates with apoptosis as inferred from caspase-3 and -9 activation and DNA fragmentation (Lam et al. 1999; Denes et al. 2011; Guerin et al. 2011). We concluded that the progressive thinning of the inner retina in treated rats is due to apoptosis, which is restricted to inner retina.
Glutamate target cells on PNDs 6, 8, and 10 were identified by immunohistochemical analysis. The number of Pax6-positive cells decreased in rats treated on both PNDs 6 and 8. The number of Chx10- and PKC alpha–positive cells decreased, and calbindin-positive cells were lost in rats treated on PND 8. GS-positive cells were observed in retina of all rats. Therefore, glutamate target cells in inner nuclear layer appeared to be amacrine cells on PND 6 and amacrine cells, bipolar cells, and horizontal cells on PND 8. The number of Müller cells did not decrease. Because horizontal cells were thought to have little impact on the thickness of inner nuclear layer, given that the retina in normal rats has only a few calbindin-positive cells, thinning of inner nuclear layer in rats treated on PND 6 was mainly caused by decrease in amacrine cell numbers, and thinning on PND 8 largely reflected the decrease in amacrine and bipolar cell numbers. Amacrine cells appeared to be more sensitive to glutamate than bipolar cells on PND 8 because the number of Pax6-positive cells decreased in rats treated on PND 8 at 6 hr after dosing and because apoptotic cells were mainly detected in inner area of inner nuclear layer which was the location of Pax6 expression. It had been indicated that amacrine cells were developed precedingly to bipolar cells by analysis of developing period of retinal cells with 3H-thymidine administration on each PND (Cepko et al. 1996; Marquardt and Gruss 2002). Therefore, the greater glutamate sensitivity of amacrine cells than bipolar cells in younger retina might be due to their earlier development. Each retinal cell expresses different types of glutamate receptors; NMDA receptors are expressed in RGC, amacrine cells, and cone bipolar cells; and AMPA and kainate receptors are expressed in RGC, amacrine cells, bipolar cells, and horizontal cells (Brandstatter, Koulen, and Wassle 1998; Dhingra and Vardi 2012). Therefore, the diversity of expressing glutamate receptors might also mediate sensitivity to glutamate in the retinal cells.
The inner retina, which consists of nerve fiber layer, ganglion cell layer, inner plexiform layer, and inner nuclear layer, also showed thinning in the peripheral retina of rats treated after PND 2. These inner retinal layers were virtually absent at the central retina in rats treated on PND 8 but persisted in the peripheral retina, although they were much thinner than in normal. Morphometric analysis revealed that the inner retinal ratio was smallest in rats treated on PND 8 in the central retina and on PND 9 in the peripheral retina. It is known that central retina develops precedingly compared to peripheral retina (Prada et al. 1991; Marquardt and Gruss 2002). Furthermore, we detected outer plexiform layer in normal rats from PND 6 in the central retina, but from PND 8 in the peripheral retina (data not shown). Therefore, glutamate-induced retinal thinning in the peripheral retina was considered to be milder than that in the central retina of rats treated on PND 8 because of the difference in developing stage. The treated age when inner retinal thinning was most marked was younger in central retina than in peripheral retina because of the difference in developing stage of homeostatic capability of glutamate level.
The inner retina is atrophic in patients with degenerative retinal diseases such as ischemia/reperfusion injury and glaucoma. It is suggested that glutamate accumulates in retinal tissue and excitotoxicity induces neural cell damage in those diseases (Donello et al. 2001; Martin et al. 2002; Sullivan et al. 2006). Therefore, mechanism identification of glutamate-induced excitotoxicity in animal models will provide a better understanding of excitotoxicity and degenerative retinal disease in human.
In summary, we clearly demonstrated that glutamate-induced retinal thinning was due to apoptosis of inner retinal cells and that this effect was most marked within the central retina in rats treated on PND 8 and in the peripheral retina in those treated on PND 9. Retinal thinning was more severe in the central than in the peripheral retina of rats treated on PND 8. The number of amacrine cells, bipolar cells, and horizontal cells was decreased by
Footnotes
Acknowledgments
The authors thank Ms. N. Saitoh and Ms. S. Yamamoto for their excellent technical assistance and Dr. S. Nakatsuji for his valuable advice and comments.
Authors Contribution
Authors contributed to conception or design (HM); data acquisition, analysis, or interpretation (HM, TI, MK, MM, JY); drafting the manuscript (HM); and critically revising the manuscript (HM, TI, MK, MM, JY). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
